The Prawn Palaemon adspersus in the Hypersaline Lake Moynaki (Crimea): Ecology, Long-Term Changes, and Prospects for Aquaculture
Abstract
:1. Introduction
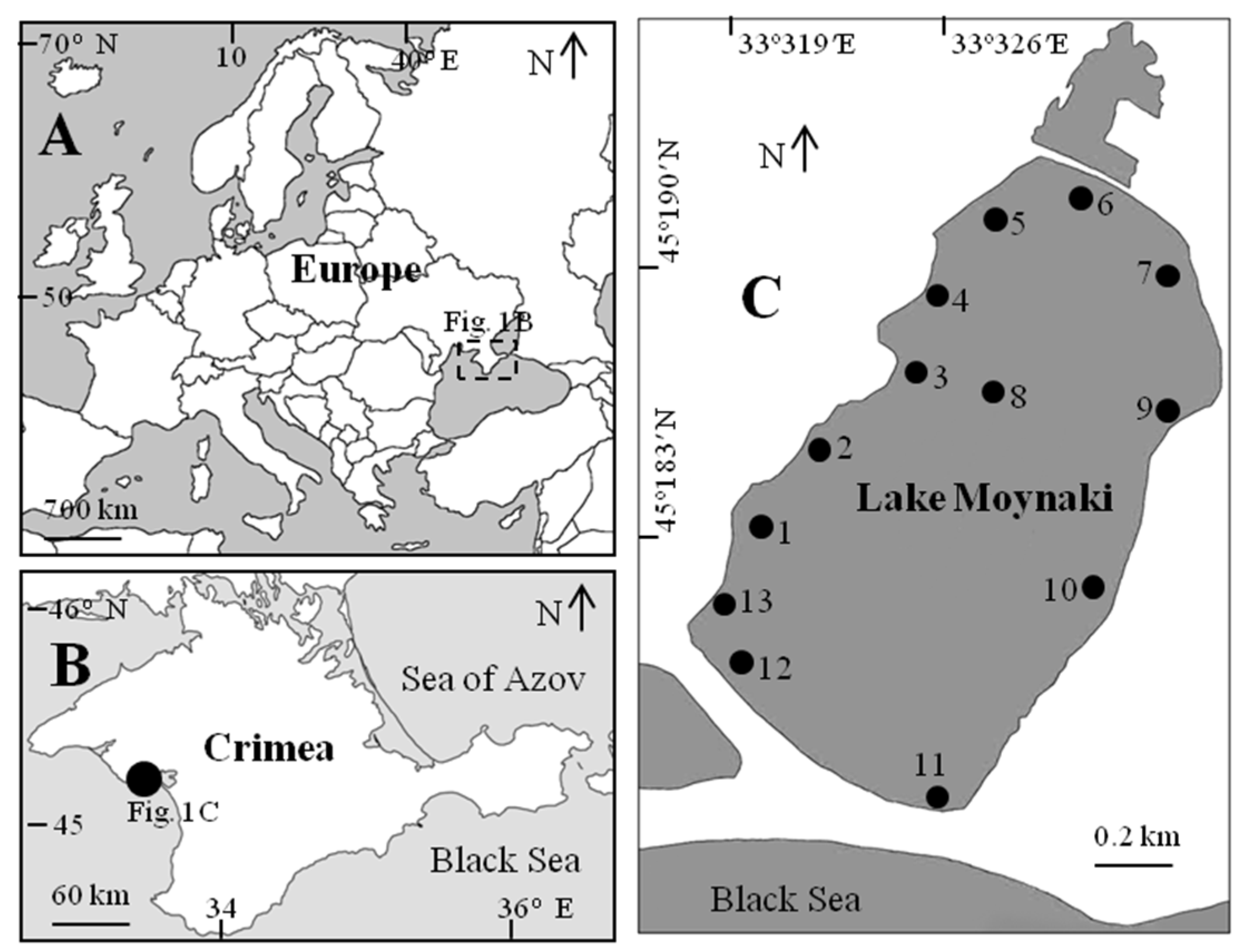
2. Materials and Methods
3. Results
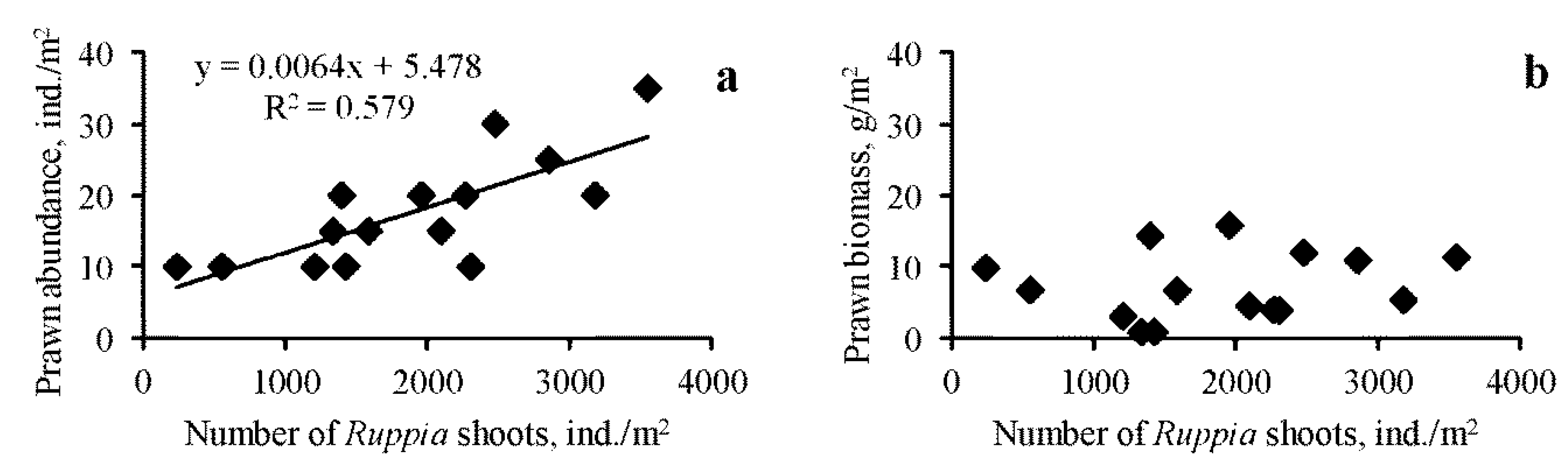
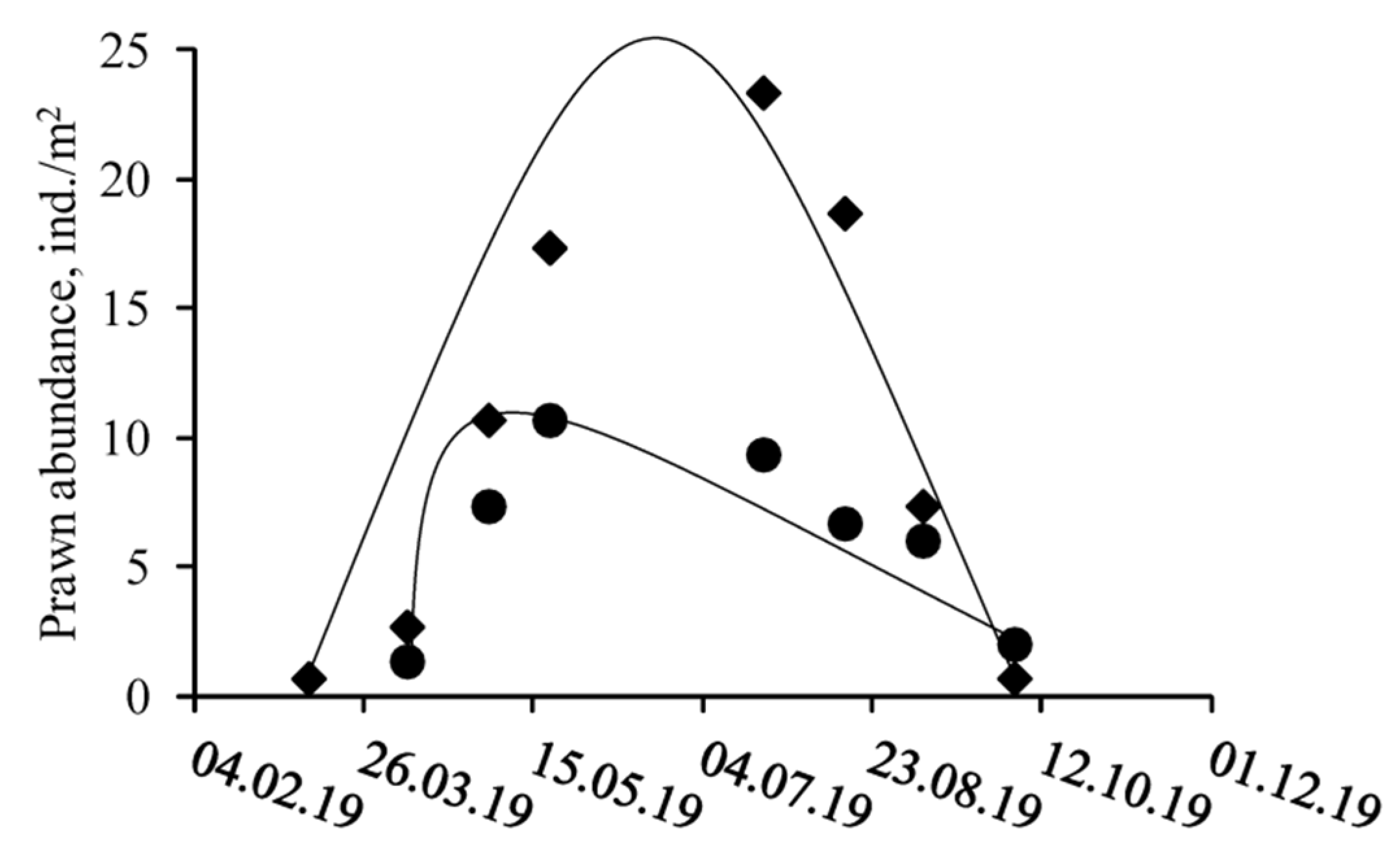
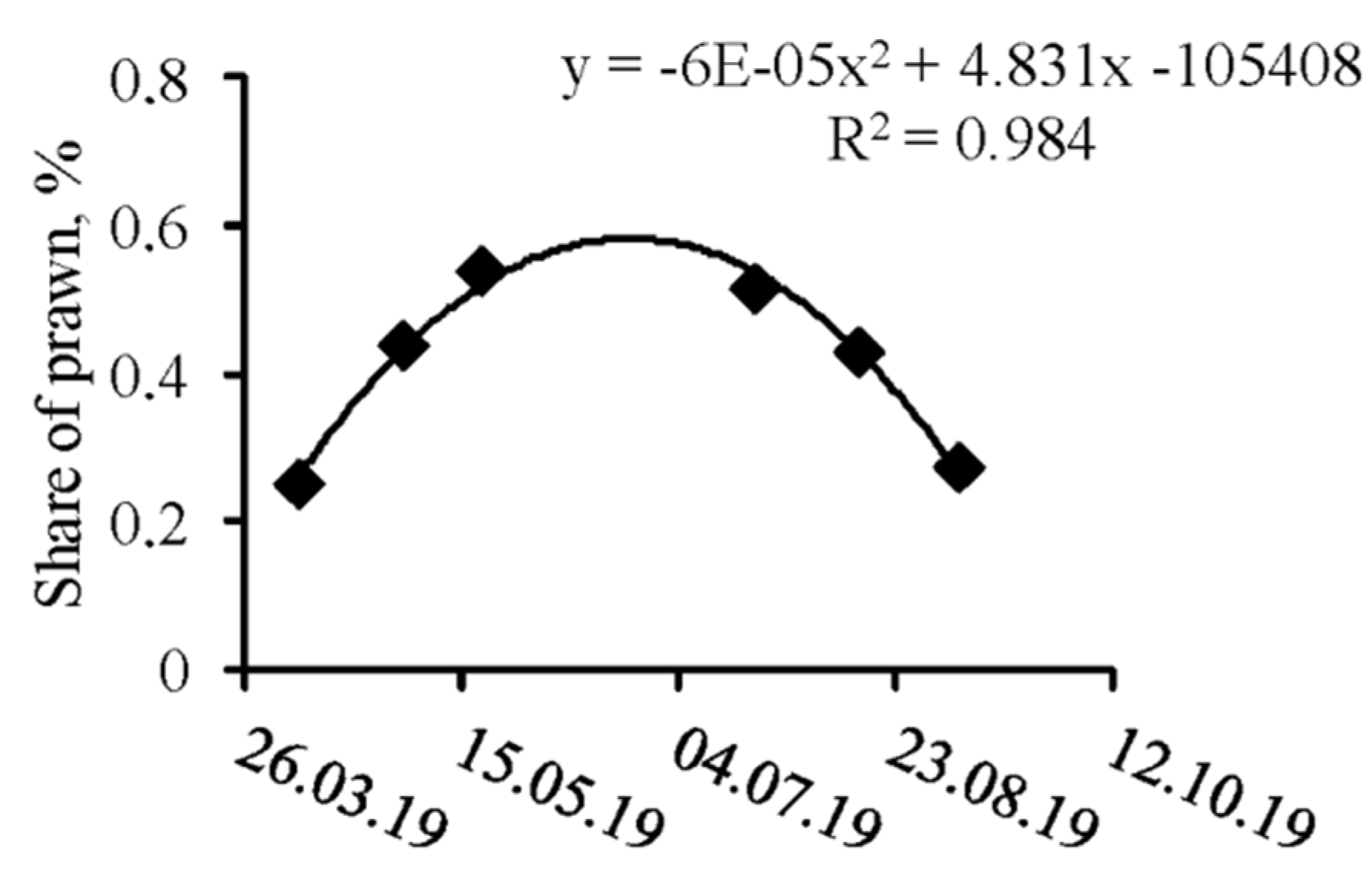
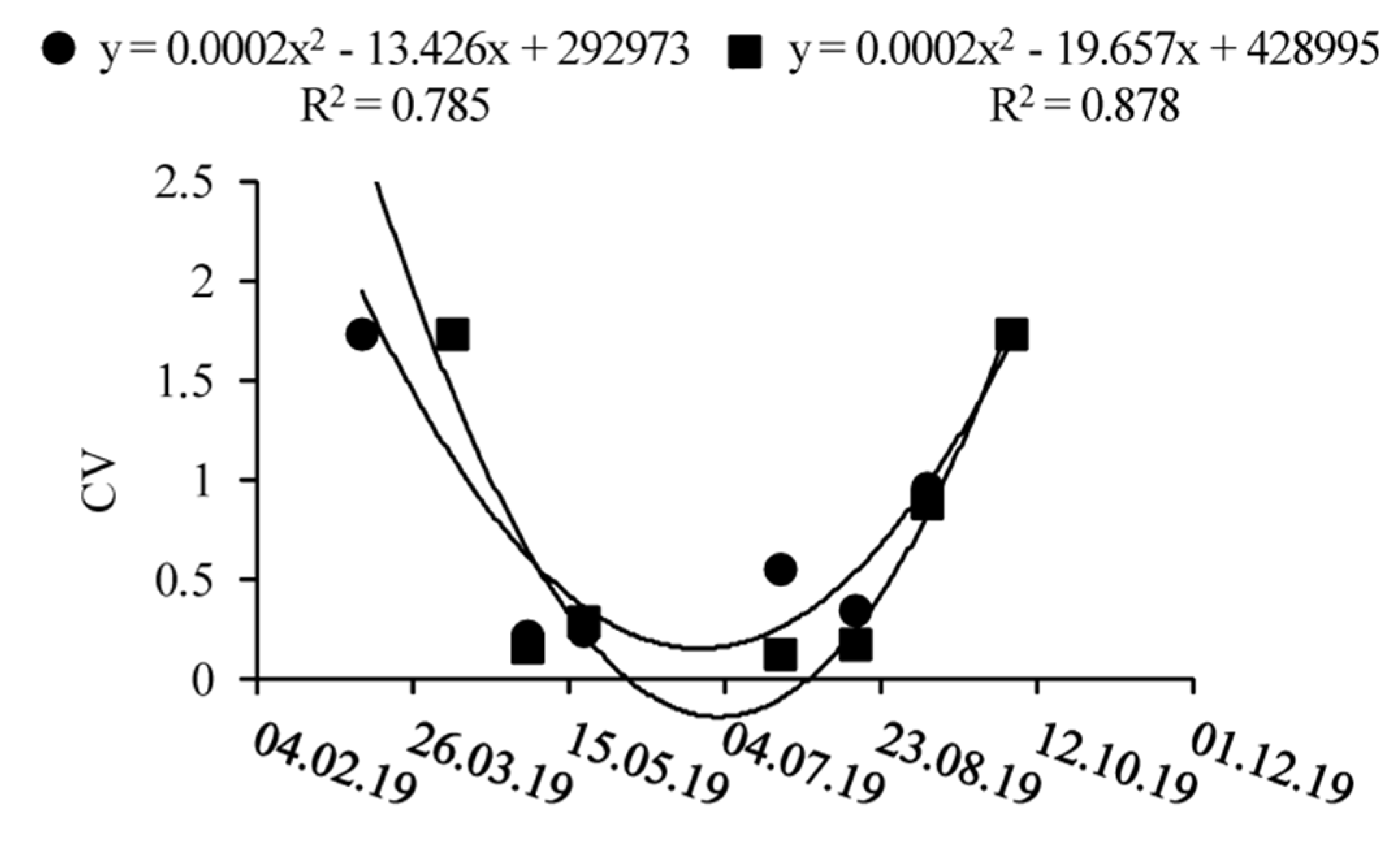
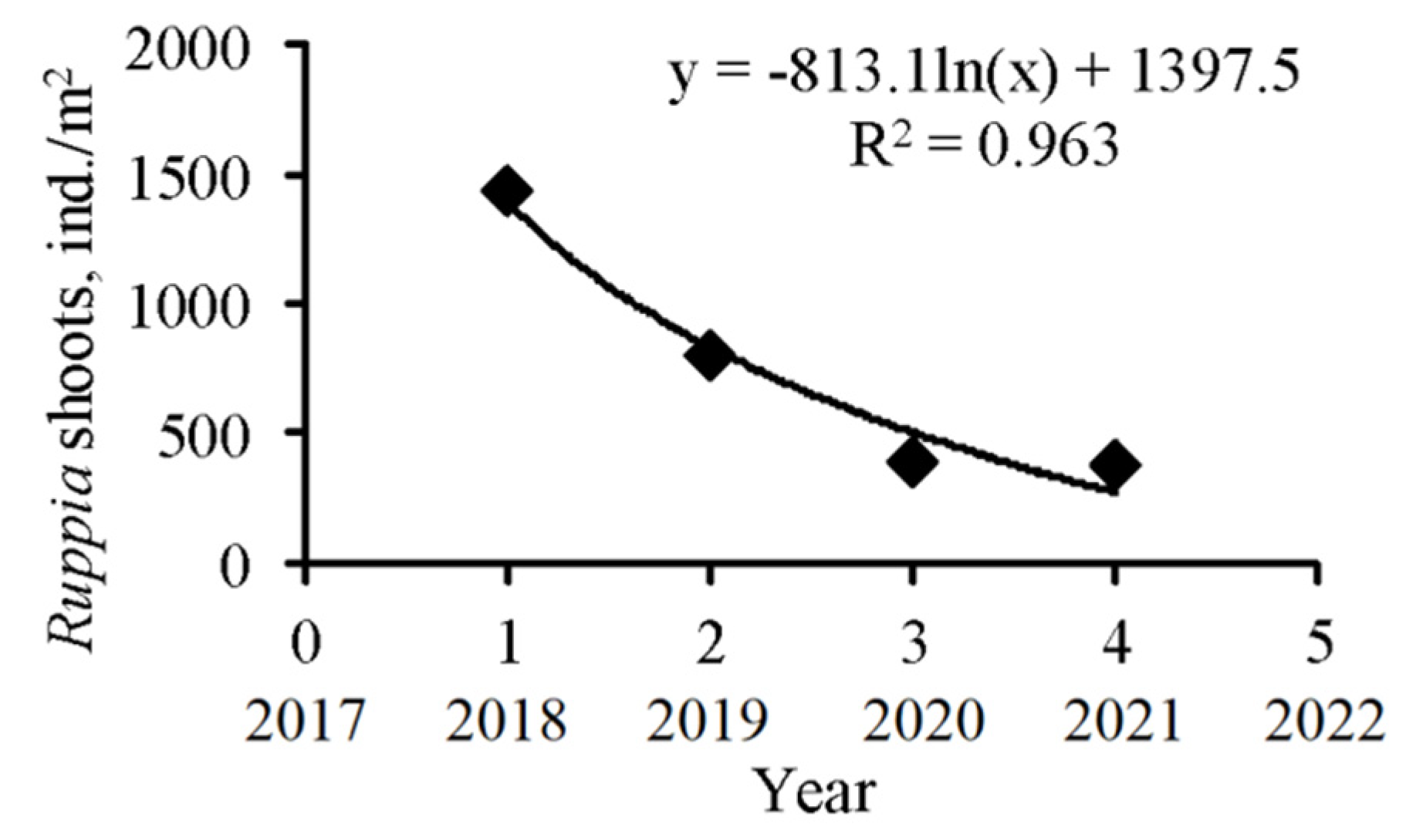
3.1. Spatio-Temporal Dynamics
3.2. Length, Mass, Fecundity
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Williams, W.D. Salinity as a determinant of the structure of biological communities in salt lakes. Hydrobiologia 1998, 381, 191–201. [Google Scholar] [CrossRef]
- Saccò, M.; White, N.E.; Harrod, C.; Salazar, G.; Aguilar, P.; Cubillos, C.F.; Meredith, K.; Baxter, B.K.; Oren, A.; Anufriieva, E.; et al. Salt to conserve: A review on the ecology and preservation of hypersaline ecosystems. Biol. Rev. 2021, 96, 2828–2850. [Google Scholar] [CrossRef]
- Pickens, A.H.; Hansen, M.C.; Hancher, M.; Stehman, S.V.; Tyukavina, A.; Potapov, P.; Marroquin, B.; Sherani, Z. Mapping and sampling to characterize global inland water dynamics from 1999 to 2018 with full Landsat time-series. Remote Sens. Environ. 2020, 243, 111792. [Google Scholar] [CrossRef]
- He, C.; Liu, Z.; Wu, J.; Pan, X.; Fang, Z.; Li, J.; Bryan, B.A. Future global urban water scarcity and potential solutions. Nat. Commun. 2021, 12, 4667. [Google Scholar] [CrossRef]
- Anufriieva, E.V. How can saline and hypersaline lakes contribute to aquaculture development? A review. J. Oceanol. Limnol. 2018, 36, 2002–2009. [Google Scholar] [CrossRef]
- Van Stappen, G.; Sui, L.; Hoa, V.N.; Tamtin, M.; Nyonje, B.; de Medeiros Rocha, R.; Sorgeloos, P.; Gajardo, G. Review on integrated production of the brine shrimp Artemia in solar salt ponds. Rev. Aquac. 2020, 12, 1054–1071. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Suppression of Artemia spp. (Crustacea, Anostraca) populations by predators in the Crimean hypersaline lakes: A review of the evidence. Int. Rev. Hydrobiol. 2019, 104, 5–13. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Behavior of Gammarus aequicauda (Crustacea, Amphipoda) during predation on Artemia (Crustacea, Anostraca): New experimental results. Int. Rev. Hydrobiol. 2020, 105, 143–150. [Google Scholar] [CrossRef]
- Anufriieva, E.; Shadrin, N. The long-term changes in plankton composition: Is Bay Sivash transforming back into one of the world’s largest habitats of Artemia sp. (Crustacea, Anostraca)? Aquac. Res. 2020, 51, 341–350. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Gammarus aequicauda and Moina salina in the Crimean saline waters: New experimental and field data on their trophic relation. Aquac. Res. 2020, 51, 3091–3099. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Can Gammarus aequicauda (Amphipoda) suppress a population of Baeotendipes noctivagus (Chironomidae) in a hypersaline lake? A case of Lake Moynaki (Crimea). Aquac. Res. 2021, 52, 1705–1714. [Google Scholar] [CrossRef]
- Alexander, K.A.; Potts, T.P.; Freeman, S.; Israel, D.; Johansen, J.; Kletou, D.; Meland, M.; Pecorino, D.; Rebours, C.; Shorten, M.; et al. The implications of aquaculture policy and regulation for the development of integrated multi-trophic aquaculture in Europe. Aquaculture 2015, 443, 16–23. [Google Scholar] [CrossRef]
- Guerra-García, J.M.; Baeza-Rojano, E.; Jiménez-Prada, P.; Calero-Cano, S.; Cervera, J.L. Trends in aquaculture today. Marine amphipods as alternative resource. Biodivers. J. 2017, 8, 395–398. [Google Scholar]
- Jazdzewski, K.; Konopacka, A. Crustacea, excluding land isopods (Malacostraca excl. Oniscoidea). Cat. Faunae Pol. 1995, 1, 1–165. [Google Scholar]
- Cartaxana, A. Growth of the prawn Palaemon longirostris (Decapoda, Palaemonidae) in Mira river and estuary, SW Portugal. J. Crustac. Biol. 2003, 23, 251–257. [Google Scholar] [CrossRef]
- González-Ortegón, E.; Cuesta, A.J. An illustrated key to species of Palaemon and Palaemonetes (Crustacea: Decapoda: Caridea) from European waters, including the alien species Palaemon macrodactylus. J. Mar. Biolog. Assoc. UK 2006, 86, 93–102. [Google Scholar] [CrossRef]
- Boltachev, A.R.; Statkevich, S.V.; Karpova, E.P.; Khutorenko, I.V. Black Sea grass shrimp Palaemon adspersus (Decapoda, Palaemonidae): Biology, fisheries, problems. Vopr. Rybolov. 2017, 18, 313–327. (In Russian) [Google Scholar]
- Berglund, A. Coexistence, size overlap and population regulation in tidal vs. non-tidal Palaemon prawns. Oecologia 1982, 54, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zenkevich, L. Biology of the Seas of the USSR; Interscience Publishers: New York, NY, USA, 1963; 955p. [Google Scholar]
- Pihl, L.; Isaksson, I.; Wennhage, H.; Moksnes, P.O. Recent increase of filamentous algae in shallow Swedish bays: Effects on the community structure of epibenthic fauna and fish. Neth. J. Aquat. Ecol. 1995, 29, 349–358. [Google Scholar] [CrossRef]
- De La Rosa, I.L.; Rodríguez, A.; Raso, J.E. Seasonal variation and structure of a decapod (Crustacea) assemblage living in a Caulerpa prolifera meadow in Cádiz Bay (SW Spain). Estuar. Coast. Shelf Sci. 2006, 66, 624–633. [Google Scholar] [CrossRef]
- Bilgin, S.; Samsun, O.; Ozen, O. Seasonal growth and reproduction biology of the Baltic prawn, Palaemon adspersus (Decapoda: Palaemonidae), in the southern Black Sea. J. Mar. Biolog. Assoc. UK 2009, 89, 509–519. [Google Scholar] [CrossRef]
- Berglund, A. Niche differentiation between two littoral prawns in Gullmar Fjord, Sweden: Palaemon adspersus and P. squilla. Ecography 1980, 3, 111–115. [Google Scholar] [CrossRef]
- Lapinska, E.; Szaniawska, A. Environmental preferences of Crangon crangon (Linnaeus 1758), Palaemon adspersus Rathke 1837, and Palaemon elegans Rathke 1837, in the littoral zone of the gulf of Gdansk. Crustaceana 2006, 79, 649–662. [Google Scholar]
- Prazukin, A.V.; Anufriieva, E.V.; Shadrin, N.V. Cladophora mats in a Crimean hypersaline lake: Structure, dynamics, and inhabiting animals. J. Oceanol. Limnol. 2018, 36, 1930–1940. [Google Scholar] [CrossRef]
- Janas, U.; Piłka, M.; Lipińska, D. Temperature and salinity requirements of Palaemon adspersus Rathke, 1837 and Palaemon elegans Rathke, 1837. Do they explain the occurrence and expansion of prawns in the Baltic Sea? Mar. Biol. Res. 2013, 9, 293–300. [Google Scholar] [CrossRef]
- Burukovsky, R.N. Food composition of the shrimp Palaemon adspersus Rathke, 1837 (Crustacea Decapoda, Palaemonidae) in Karkinitsky Bay of the Black Sea in September 2016. Mar. Biol. J. 2019, 4, 12–23. (In Russian) [Google Scholar]
- Berglund, A. Different reproductive success at low salinity determines the estuarine distribution of two Palaemon prawn species. Ecography 1985, 8, 49–52. [Google Scholar] [CrossRef]
- Marin, I.N. Small Atlas of Decapoda in Russia; Association of Scientific Publications: Moscow, Russia, 2013; 145p. (In Russian) [Google Scholar]
- Anufriieva, E.V.; Shadrin, N.V.; Shadrina, S.N. History of research on biodiversity in Crimean hypersaline waters. Arid Ecosyst. 2017, 7, 52–58. [Google Scholar] [CrossRef]
- FAO. Fisheries and Aquaculture Information and Statistics Service. Aquaculture Production: Quantities 1950–2005. FISHSTAT Plus—Universal Software for Fishery Statistical Time Series [online or CD-ROM]. Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. Available online: http://www.fao.org/fi/statist/FISOFT/FISHPLUS.asp (accessed on 4 September 2022).
- Bondarenko, L.V.; Yakovenko, V.A. Transformation of the species structure of crustaceans in Lake Moynaki to connection with its desalination. Bull. Dnipropetr. Univ. Ser. Biol. Ecol. 2000, 8, 100–105. (In Russian) [Google Scholar]
- Shadrin, N.V.; Yakovenko, V.A.; Anufriieva, E.V. Appearance of a new species of Cladocera (Anomopoda, Chydoridae, Bosminidae) in the hypersaline Moynaki Lake, Crimea. Biol. Bull. 2021, 48, 934–937. [Google Scholar] [CrossRef]
- Muller, P.H.; Neuman, P.; Storm, R. Tafeln der Mathematischen Statistic; VEB Fachbuchverlag: Leipzig, Germany, 1979; 280p. [Google Scholar]
- Thode, H.C. Testing for Normality; Marcel Dekker Inc.: New York, NY, USA, 2002. [Google Scholar]
- Plotnikov, I.S. Long-Term Changes in the Fauna of Free-Living Aquatic Invertebrates of the Aral Sea; ZIN RAN: Saint Petersburg, Russia, 2016; 168p. (In Russian) [Google Scholar]
- Baden, S.P.; Pihl, L. Abundance, biomass and production of mobile epibenthic fauna in Zostera marina (L.) meadows, western Sweden. Ophelia 1984, 23, 65–90. [Google Scholar] [CrossRef]
- Köhn, J.; Gosselck, F. Identification key for the Malacostraca of the Baltic Sea. Mitt. Aus Dem Zool. Mus. Berl. 1989, 65, 3–114. [Google Scholar] [CrossRef]
- Manent, P.; Abella-Gutiérrez, J. Population biology of Palaemon adspersus Rathke, 1837 (Decapoda, Caridae) in Fornells Bay, Balearic Islands, Western Mediterranean. Crustaceana 2006, 79, 1297–1308. [Google Scholar] [CrossRef]
- Schaffmeister, B.E.; Hiddink, J.G.; Wolff, W.J. Habitat use of shrimps in the intertidal and shallow subtidal seagrass beds of the tropical Banc d’Arguin, Mauritania. J. Sea Res. 2006, 55, 230–243. [Google Scholar] [CrossRef]
- Bilgin, S.; Ozen, O.; Ates, A.S. Spatial and temporal variation of Palaemon adspersus, Palaemon elegans, and Crangon crangon (Decapoda: Caridea) in the southern Black Sea. Estuar. Coast. Shelf Sci. 2008, 79, 671–678. [Google Scholar] [CrossRef]
- Fusté, X. Crustáceos decápodos de la Bahía de Els Alfacs (Delta del Ebro). Investig. Pesq. 1988, 50, 617–623. [Google Scholar]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef]
- Shadrin, N.V. The alternative saline lake ecosystem states and adaptive environmental management. J. Oceanol. Limnol. 2018, 36, 2010–2017. [Google Scholar] [CrossRef]
- Nielsen, A.; Hagerman, L. Effects of short-term hypoxia on metabolism and haemocyanin oxygen transport in the prawns Palaemon adspersus and Palaemonetes varians. Mar. Ecol. Prog. Ser. 1998, 167, 177–183. [Google Scholar] [CrossRef]
- Berglund, A.; Bengtsson, J. Biotic and abiotic factors determining the distribution of two prawn species: Palaemon adspersus and P. squilla. Oecologia 1981, 49, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Shadrin, N.; Anufriieva, E. Ecosystems of hypersaline waters: Structure and trophic relations. Zh. Obshch. Biol. 2018, 79, 418–427. (in Russian). [Google Scholar]
- Glamuzina, L.; Conides, A.; Prusina, I.; Ćukteraš, M.; Klaoudatos, D.; Zacharaki, P.; Glamuzina, B. Population structure, growth, mortality and fecundity of Palaemon adspersus (Rathke 1837; Decapoda: Palaemonidae) in the Parila Lagoon (Croatia, SE Adriatic Sea) with Notes on the Population Management. Turkish J. Fish. Aquat. Sci. 2014, 14, 677–687. [Google Scholar]
- Conides, A.; Tsevis, N.; Yannopoulos, C. Studies on sex ratios observed on the prawn Palaemon adspersus (Rathke, 1837) population at Messolonghi lagoon, Greece. Fresenius Environ. Bull. 1992, 1, 665–669. [Google Scholar]
- Figueras, A.J. Crecimiento de Palaemon adspersus (Rathke 1837) y P. serratus (Pennant 1777) (Decapoda: Natantia) en la ria de Vigo (S) de Espana. Rev. Investig. Pesqueira 1986, 50, 117–125. [Google Scholar]
- Guerao, G.; Ribera, C. Growth and reproductive ecology of Palaemon adspersus (Decapoda, Palaemonidae) in the western Mediterranean. Ophelia 2013, 43, 205–213. [Google Scholar] [CrossRef]
- Stepanova, O.D.; Kibenko, V.A. The Black Sea grass shrimp (Palaemon adspersus Rathke, 1837) is a promising object of fishing in the Azov-Black Sea basin. Vestn. Kerchenskogo Gos. Morskogo Tekhnologicheskogo Univ. 2021, 3, 84–94. (In Russian) [Google Scholar] [CrossRef]
- Bilgin, S.; Samsun, O. Fecundity and egg size of three shrimp species, Crangon crangon, Palaemon adspersus, and Palaemon elegans (Crustacea: Decapoda: Caridea), off Sinop Peninsula (Turkey) in the Black Sea. Turk. J. Zool. 2006, 30, 413–421. [Google Scholar]
- Shadrin, N.V.; Anufriieva, E.V.; Belyakov, V.P.; Bazhora, A.I. Chironomidae larvae in hypersaline waters of the Crimea: Diversity, distribution, abundance and production. Eur. Zool. J. 2017, 84, 61–72. [Google Scholar] [CrossRef]
- Anufriieva, E.; Kolesnikova, E.; Revkova, T.; Latushkin, A.; Shadrin, N. Human-induced sharp salinity changes in the world’s largest hypersaline lagoon Bay Sivash (Crimea) and their effects on the ecosystem. Water 2022, 14, 403. [Google Scholar] [CrossRef]
- Jones, E.; Qadir, M.; van Vliet, M.T.; Smakhtin, V.; Kang, S.M. The state of desalination and brine production: A global outlook. Sci. Total Environ. 2019, 657, 1343–1356. [Google Scholar] [CrossRef] [PubMed]
- Anger, K.; Moreira, G.S.; Ismael, D. Comparative size, biomass, elemental composition (C, N, H), and energy concentration of caridean shrimp eggs. Invertebr. Reprod. Dev. 2002, 42, 83–93. [Google Scholar] [CrossRef]
- Janas, U.; Bruska, O. Energy values and energy resources of two prawns in Baltic coastal waters: The indigenous Palaemon adspersus and the non-indigenous Palaemon elegans. Oceanologia 2010, 52, 281–297. [Google Scholar] [CrossRef] [Green Version]
- Bal-Prylypko, L.V.; Lebskiy, S.O.; Lebskaya, T.K.; Menchinskaya, A.A. A research on biologically active compounds from black sea grass crab Palaemon adspersus. Chem. Technol. Control. Manag. 2019, 2019, 4. (In Russian) [Google Scholar]
- Bykova, V.M. Antarctic Krill. Manual; VNIRO: Moscow, Russia, 2001; 207p. (In Russian) [Google Scholar]
- Abete, P.; Testa, G.; Galizia, G.; Della-Morte, D.; Cacciatore, F.; Rengo, F. PUFA for human health: Diet or supplementation? Curr. Pharm. Des. 2009, 15, 4186–4190. [Google Scholar] [CrossRef] [PubMed]

| Area | Salinity g/L | Maximum Length, mm | Parameters of Equation W = aLb | Number of Eggs Per 1 g | References | ||||
|---|---|---|---|---|---|---|---|---|---|
| Females | Males | ||||||||
| Female | Male | a, 10−5 | b | a, 10−5 | b | ||||
| Mediterranean Sea, Parila Lagoon | <30 | 72.5 | 59.0 | 0.0003 | 2.13 | 0.0009 | 1.81 | 929 | [48] |
| Black Sea, Sinop Bay | 17÷18 | 82 | 70.0 | 0.7 | 3.25 | 0.2 | 2.93 | 725 | [22] |
| Mediterranean Sea, Mesolongi Lagoon | 33÷35 | 78.7 | 64.7 | 0.5 | 3.11 | 0.4 | 3.20 | - | [49] |
| Atlantic Ocean, the Vigo estuary | 33÷35 | 68.2 | 44.9 | - | - | - | - | - | [50] |
| Baltic Sea, Gdansk Bay | 6.4÷6.7 | 59.0 | 38.0 | 0.4 * | 3.17 * | - | - | - | [24] |
| Mediterranean Sea, western part | 33÷35 | 71.0 | 52.0 | 0.3 | 3.32 | 0.5 | 3.20 | - | [51] |
| Black Sea, Karkinitsky Bay | 17÷18 | 78.2 | 57.3 | - | - | - | - | 792 | [17] |
| Black Sea, Kerch strait | 11÷15 | 95.0 | - | - | - | - | - | 357 | [52] |
| Black Sea, Karkinitsky Bay | 17÷18 | 58.1 | 44.1 | - | - | - | - | - | [28] |
| Lake Moynaki | 49÷63 | 58 | 46 | 0.4 | 3.348 | 0.6 | 3.074 | 653 | New data |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakovenko, V.; Shadrin, N.; Anufriieva, E. The Prawn Palaemon adspersus in the Hypersaline Lake Moynaki (Crimea): Ecology, Long-Term Changes, and Prospects for Aquaculture. Water 2022, 14, 2786. https://doi.org/10.3390/w14182786
Yakovenko V, Shadrin N, Anufriieva E. The Prawn Palaemon adspersus in the Hypersaline Lake Moynaki (Crimea): Ecology, Long-Term Changes, and Prospects for Aquaculture. Water. 2022; 14(18):2786. https://doi.org/10.3390/w14182786
Chicago/Turabian StyleYakovenko, Vladimir, Nickolai Shadrin, and Elena Anufriieva. 2022. "The Prawn Palaemon adspersus in the Hypersaline Lake Moynaki (Crimea): Ecology, Long-Term Changes, and Prospects for Aquaculture" Water 14, no. 18: 2786. https://doi.org/10.3390/w14182786





