In Situ Pumping–Injection Remediation of Strong Acid–High Salt Groundwater: Displacement–Neutralization Mechanism and Influence of Pore Blocking
Abstract
:1. Introduction
2. Materials and Methods
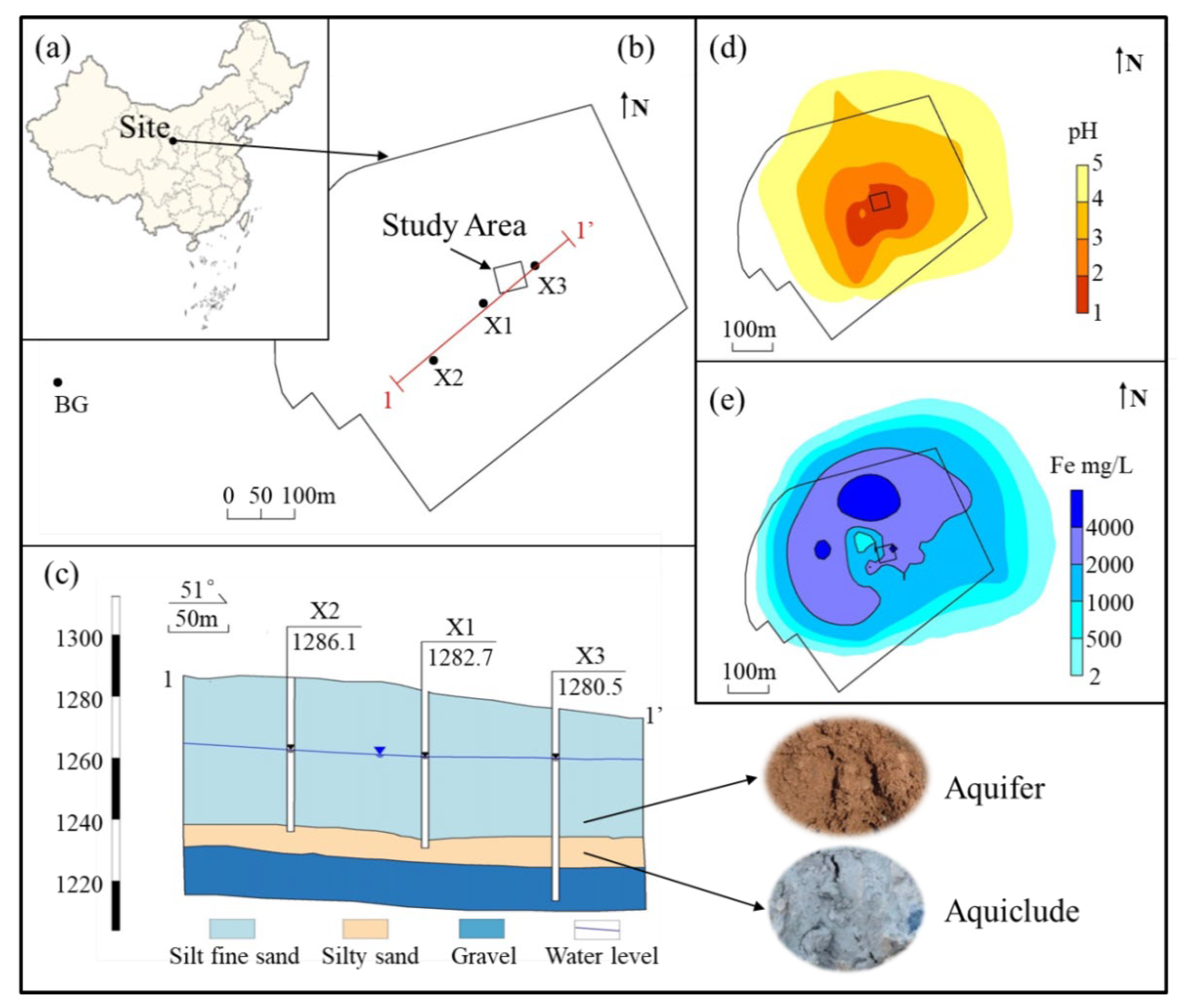
2.1. Study Site
2.1.1. Hydrogeology Conditions
2.1.2. Characteristics of Groundwater Pollution
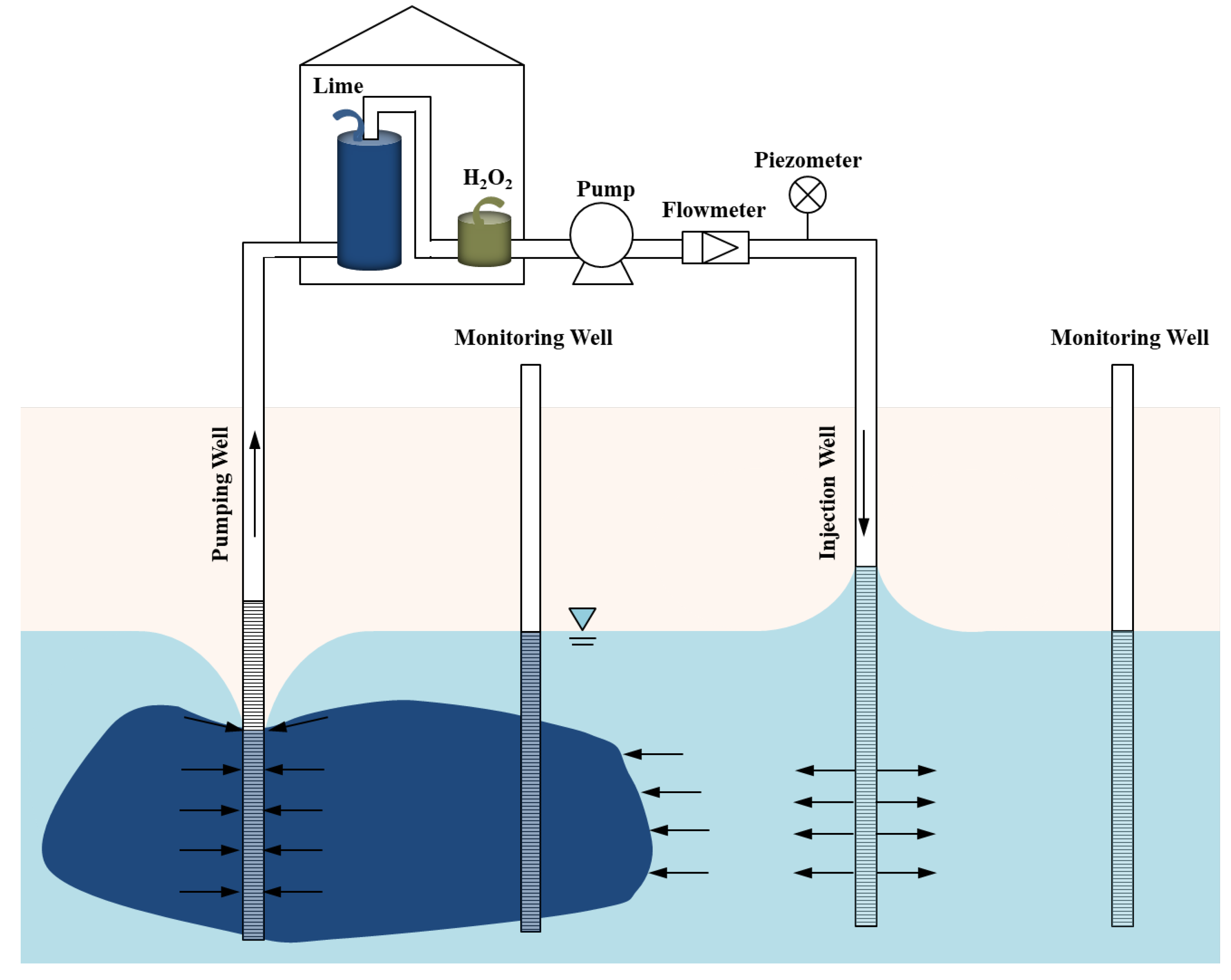
2.2. Operational Details
2.2.1. Pump–Treat–Inject Process
2.2.2. Experimental Process
2.3. Sample Collection and Analysis
2.3.1. Water Sample Analysis
2.3.2. Core Analysis
2.4. Analysis of the Data
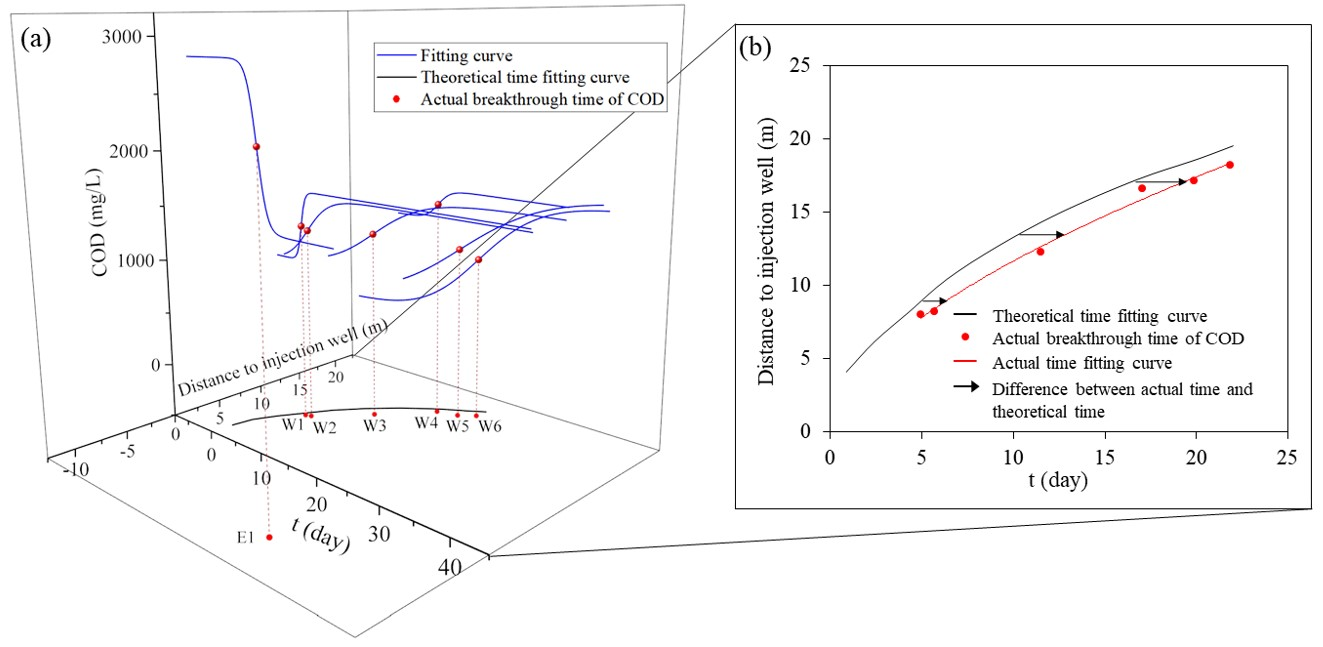
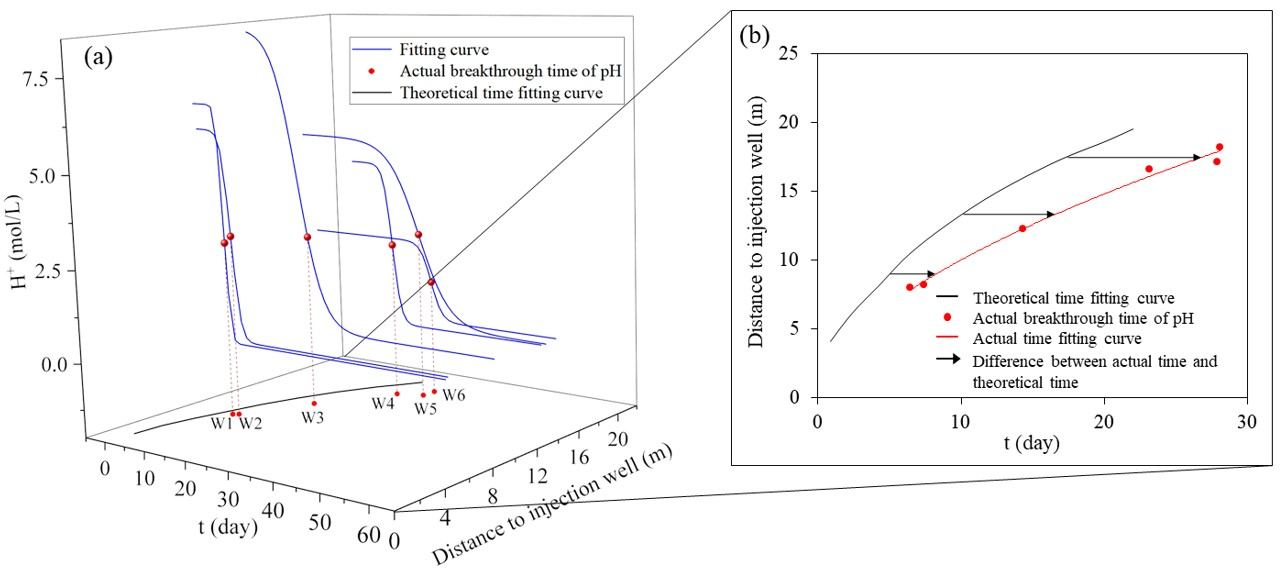
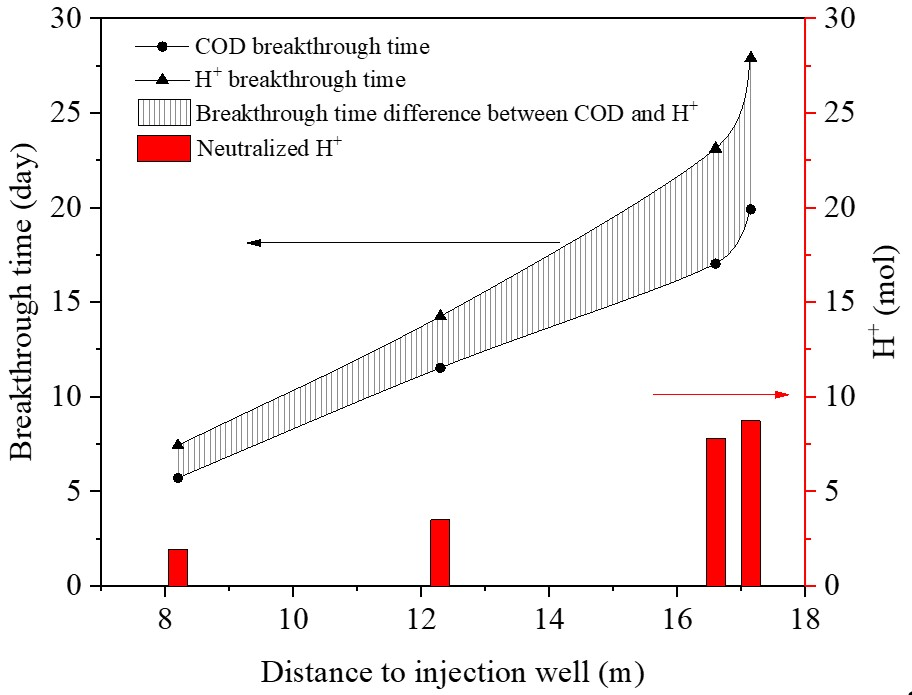
2.4.1. Breakthrough Time
2.4.2. Neutralized H+ Content
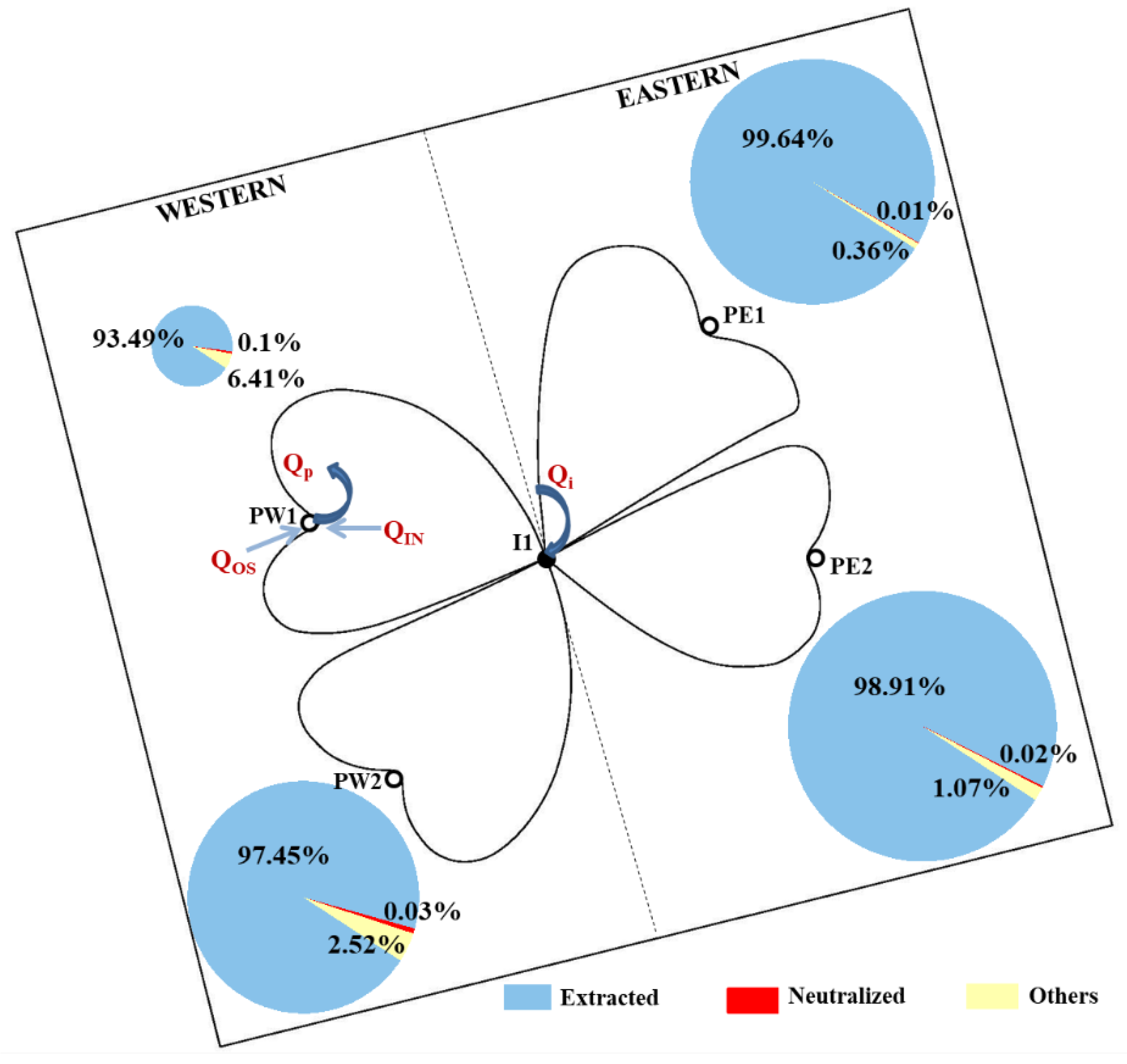
2.4.3. Extracted H+ Content
3. Results and Discussion
3.1. Displacement of Acidic Groundwater by Injected Water
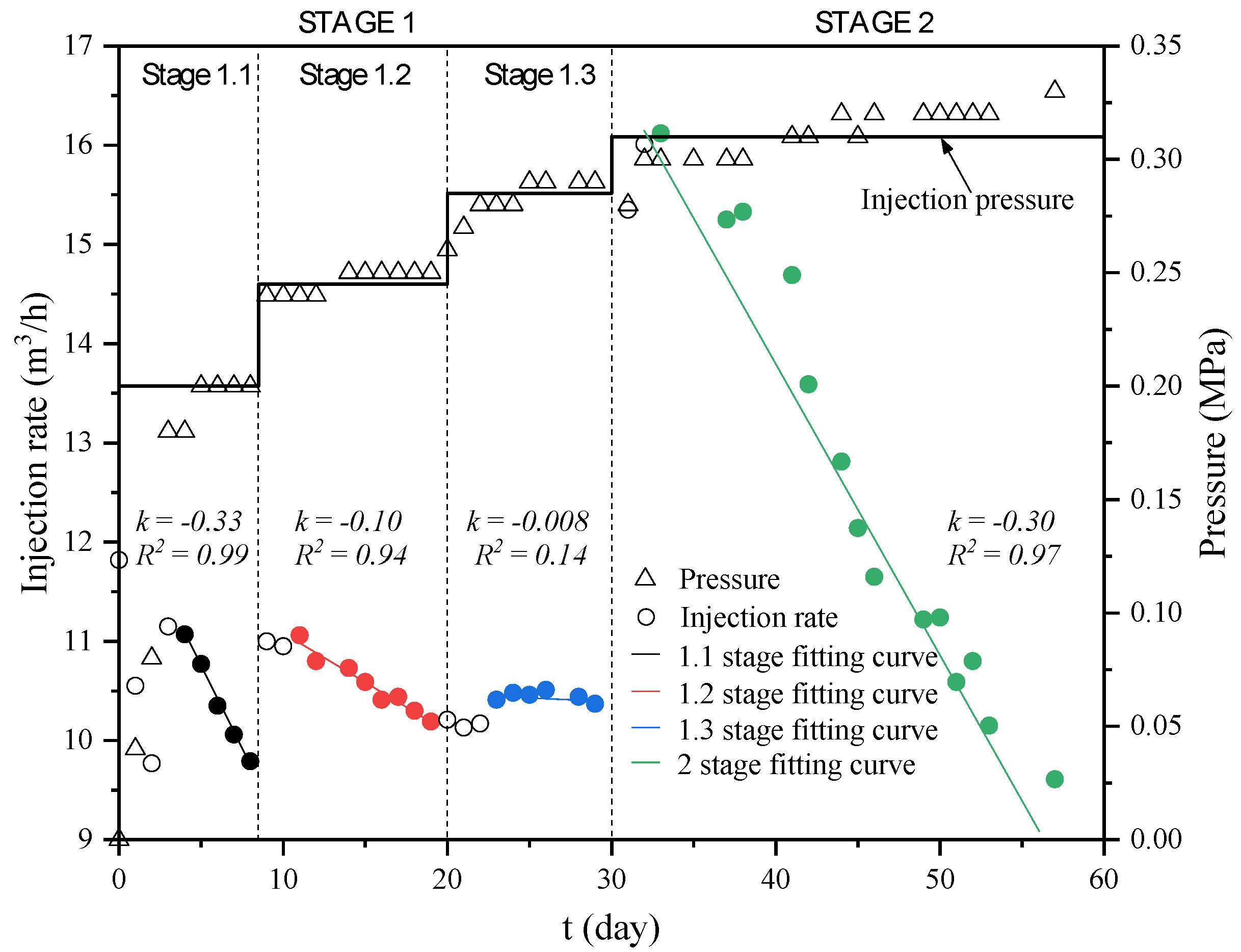
3.1.1. Water Injection Process
3.1.2. Flow and Dispersion Process of Acidic Groundwater
3.1.3. Attenuation of Permeability of Aqueous Medium
3.2. Displacement–Neutralization Process of Acidic Groundwater
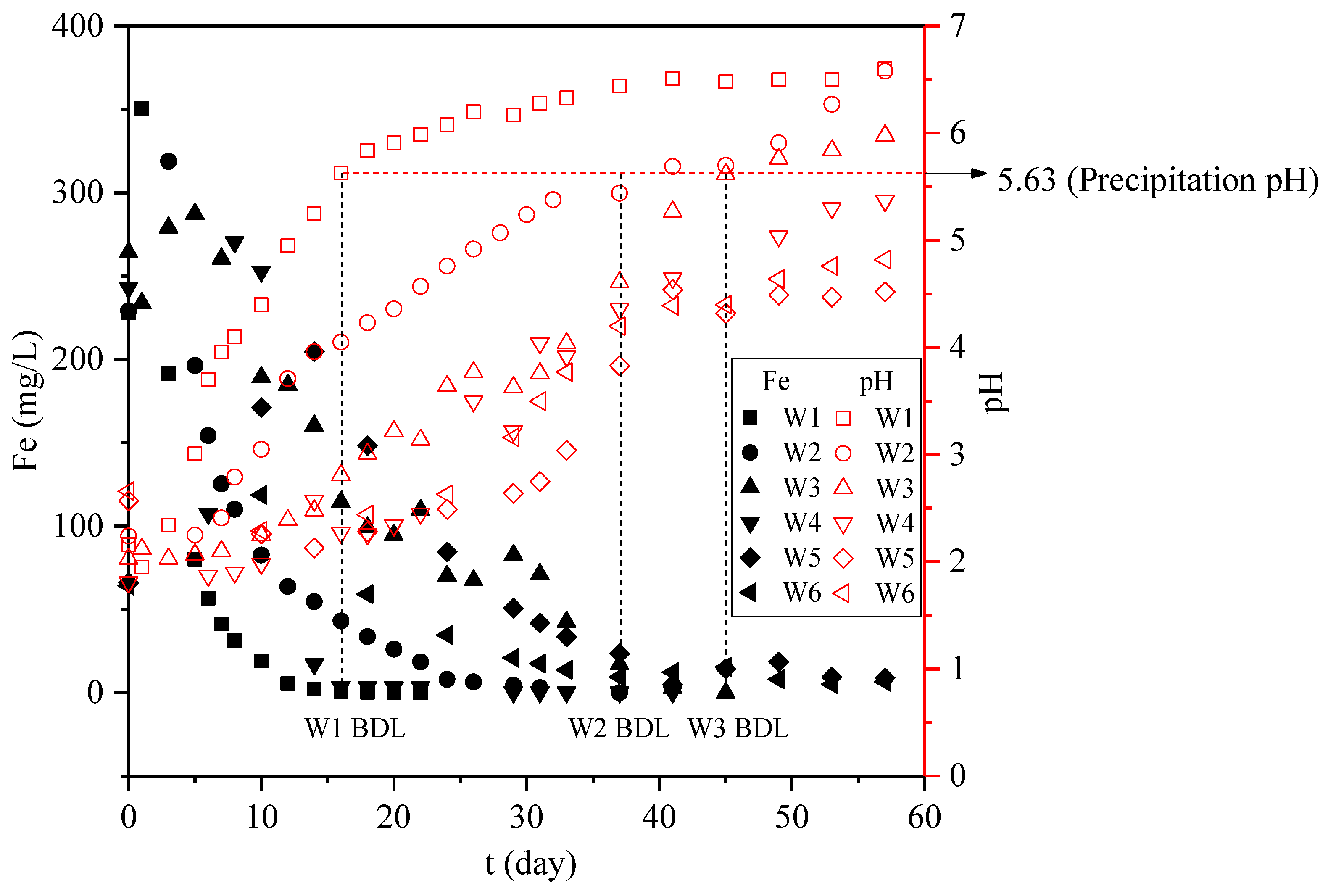
3.2.1. Dynamic Variation of Groundwater pH
3.2.2. Neutralization Amount of H+ in Groundwater
3.2.3. Equilibrium Analysis of H+ in Groundwater
3.3. Blocking Mechanism
3.3.1. Iron Concentration
3.3.2. Mineralogical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Güngör-Demirci, G.; Aksoy, A. Variation in time-to-compliance for pump-and-treat remediation of mass transfer-limited aquifers with hydraulic conductivity heterogeneity. Environ. Earth Sci. 2010, 63, 1277–1288. [Google Scholar] [CrossRef]
- Ciampi, P.; Esposito, C.; Cassiani, G.; Deidda, G.P.; Rizzetto, P.; Papini, M.P. A field-scale remediation of residual light non-aqueous phase liquid (LNAPL): Chemical enhancers for pump and treat. Environ. Sci. Pollut. Res. 2021, 28, 35286–35296. [Google Scholar] [CrossRef] [PubMed]
- Antelmi, M.; Renoldi, F.; Alberti, L. Analytical and Numerical Methods for a Preliminary Assessment of the Remediation Time of Pump and Treat Systems. Water 2020, 12, 2850. [Google Scholar] [CrossRef]
- Truex, M.; Johnson, C.; Macbeth, T.; Becker, D.; Lynch, K.; Giaudrone, D.; Frantz, A.; Lee, H. Performance Assessment of Pump-and-Treat Systems. Groundw. Monit. Rem. 2017, 37, 28–44. [Google Scholar] [CrossRef]
- Park, Y.-C. Cost-effective optimal design of a pump-and-treat system for remediating groundwater contaminant at an industrial complex. Geosci. J. 2016, 20, 891–901. [Google Scholar] [CrossRef]
- Matott, L.S.; Rabideau, A.J.; Craig, J.R. Pump-and-treat optimization using analytic element method flow models. Adv. Water Resour. 2006, 29, 760–775. [Google Scholar] [CrossRef]
- Thornton, S.F.; Baker, K.M.; Bottrell, S.H.; Rolfe, S.A.; McNamee, P.; Forrest, F.; Duffield, P.; Wilson, R.D.; Fairburn, A.W.; Cieslak, L.A. Enhancement of in situ biodegradation of organic compounds in groundwater by targeted pump and treat intervention. Appl. Geochem. 2014, 48, 28–40. [Google Scholar] [CrossRef]
- Bortonea, I.; Ertob, A.; Nardoc, A.D.; Santonastasoc, G.F.; Chianesec, S.; Musmarrac, D. Pump-and-treat configurations with vertical and horizontal wells to remediate an aquifer contaminated by hexavalent chromium. J. Contam. Hydrol. 2020, 235, 103725. [Google Scholar] [CrossRef]
- Kahler, D.M.; Kabala, Z.J. Acceleration of groundwater remediation by deep sweeps and vortex ejections induced by rapidly pulsed pumping. Water Resour. Res. 2016, 52, 3930–3940. [Google Scholar] [CrossRef]
- Teramoto, E.H.; Pede, M.A.Z.; Chang, H.K. Impact of water table fluctuations on the seasonal effectiveness of the pump-and-treat remediation in wet–dry tropical regions. Environ. Earth Sci. 2020, 79, 435. [Google Scholar] [CrossRef]
- Sadeghfam, S.; Hassanzadeh, Y.; Khatibi, R.; Nadiri, A.A.; Moazamnia, M. Groundwater Remediation through Pump-Treat-Inject Technology Using Optimum Control by Artificial Intelligence (OCAI). Water Resour. Manag. 2019, 33, 1123–1145. [Google Scholar] [CrossRef]
- Takem, G.E.; Kuitcha, D.; Ako, A.A.; Mafany, G.T.; Takounjou-Fouepe, A.; Ndjama, J.; Ntchancho, R.; Ateba, B.H.; Chandrasekharam, D.; Ayonghe, S.N. Acidification of shallow groundwater in the unconfined sandy aquifer of the city of Douala, Cameroon, Western Africa: Implications for groundwater quality and use. Environ. Earth Sci. 2015, 74, 6831–6846. [Google Scholar] [CrossRef]
- Vahedian, A.; Aghdaei, S.A.; Mahini, S. Acid Sulphate Soil Interaction with Groundwater: A Remediation Case Study in East Trinity. APCBEE Proc. 2014, 9, 274–279. [Google Scholar] [CrossRef]
- Caraballo, M.A.; Macias, F.; Nieto, J.M.; Ayora, C. Long term fluctuations of groundwater mine pollution in a sulfide mining district with dry Mediterranean climate: Implications for water resources management and remediation. Sci. Total Environ. 2016, 539, 427–435. [Google Scholar] [CrossRef]
- Shin, D.; Kim, Y.; Moon, H.S. Fate and toxicity of spilled chemicals in groundwater and soil environment I: Strong acids. Environ. Health Toxicol. 2018, 33, e2018019. [Google Scholar] [CrossRef]
- Ha, Q.K.; Choi, S.; Phan, N.L.; Kim, K.; Phan, C.N.; Nguyen, V.K.; Ko, K.S. Occurrence of metal-rich acidic groundwaters around the Mekong Delta (Vietnam): A phenomenon linked to well installation. Sci. Total Environ. 2019, 654, 1100–1109. [Google Scholar] [CrossRef]
- Appleyard, S.; Wong, S.; Willis-Jones, B.; Angeloni, J.; Watkins, R. Groundwater acidification caused by urban development in Perth, Western Australia: Source, distribution, and implications for management. Aust. J. Soil Res. 2005, 42, 579–585. [Google Scholar] [CrossRef]
- Paulson, A.J.; Balistrieri, L. Modeling Removal of Cd, Cu, Pb, and Zn in Acidic Groundwater during Neutralization by Ambient Surface Waters and Groundwaters. Environ. Sci. Technol. 1999, 33, 3850–3856. [Google Scholar] [CrossRef]
- Indraratna, B.; Pathirage, P.U.; Banasiak, L.J. Remediation of acidic groundwater by way of permeable reactive barrier. Environ. Geotech. 2017, 4, 284–298. [Google Scholar] [CrossRef]
- Shin, D.; Lee, Y.; Park, J.; Moon, H.S.; Hyun, S.P. Soil microbial community responses to acid exposure and neutralization treatment. J. Environ. Manag. 2017, 204, 383–393. [Google Scholar] [CrossRef]
- Sung, P.H.; Doyun, S.; Hee, S.M.; Young-Soo, H.; Seonjin, H.; Yoonho, L.; Eunhee, L.; Hyun, J.; Yu, S.H. A Multidisciplinary Assessment of the Impact of Spilled Acids on Geoecosystems: An Overview. Environ. Sci. Pollut. Res. 2020, 27, 9803–9817. [Google Scholar]
- Jeon, I.; Nam, K. Change in the site density and surface acidity of clay minerals by acid or alkali spills and its effect on pH buffering capacity. Sci. Rep. 2019, 9, 9878. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dai, Y.; Wang, Y.; Huang, X.; Pei, Q. Groundwater chemistry, quality and potential health risk appraisal of nitrate enriched groundwater in the Nanchong area, southwestern China. Sci. Total Environ. 2021, 784, 147186. [Google Scholar] [CrossRef]
- Olías, M.; Cánovas, C.R.; Basallote, M.D.; Macías, F.; Pérez-López, R.; González, R.M.; Millán-Becerro, R.; Nieto, J.M. Causes and impacts of a mine water spill from an acidic pit lake (Iberian Pyrite Belt). Environ. Pollut. 2019, 250, 127–136. [Google Scholar] [CrossRef]
- Zhang, Y.; He, Z.; Tian, H.; Huang, X.; Zhang, Z.; Liu, Y.; Xiao, Y.; Li, R. Hydrochemistry appraisal, quality assessment and health risk evaluation of shallow groundwater in the Mianyang area of Sichuan Basin, southwestern China. Environ. Earth Sci. 2021, 80, 576. [Google Scholar] [CrossRef]
- Gazea, B.; Adam, K.; Kontopoulos, A. A review of passive systems for the treatment of acid mine drainage. Miner. Eng. 1996, 9, 23–42. [Google Scholar] [CrossRef]
- Jiankang, Z.; Yong, Z.; Jin, C. Application Research on Pressure Recharge in Underground-water Source Heat Pump System. Explor. Eng. (Rock Soil Drill. Tunn.) 2010, 37, 55–58. [Google Scholar]
- Wu, J.Q.; Zhang, J.Z.; Li, X.H.; Shi, F.F.; Zhang, P.; Bai, M. Experiment on artificial pressure re-injection of geothermal water in Xi’an suburb. J. Water Resour. Water Eng. 2014, 25, 215–218. [Google Scholar]
- Zhao, B.l.; Mu, Z.Y.; Wu, J.X. Articifial Recharge of Groundwater (Translated Version); Water Conservancy Press: Beijing, China, 1980; pp. 32–33. [Google Scholar]
- Du, X.Q.; Lu, Y.; Li, S.T.; Ye, X.Y.; Wang, Z.J. Mechanism, Prediction and Control of Suspended Matter Blocking in Artificial Recharge of Groundwater. In Proceedings of the 8th China Water Forum, Harbin City, China, 2–3 August 2010; p. 146. [Google Scholar]
- Akhtar, M.S.; Nakashima, Y.; Nishigaki, M. Clogging mechanisms and preventive measures in artificial recharge systems. J. Groundw. Sci. Eng. 2021, 9, 21. [Google Scholar]
- Pavelic, P.; Vanderzalm, J.; Dillon, P.; Herczeg, A.; Magareyd, P. Assessment of the Potential for Well Clogging Associated with Salt Water Interception and Deep Injection at Chowilla, SA; CSIRO: Canberra, Australia, 2007. [Google Scholar]
- Dou, Z.; Zhang, X.; Zhuang, C.; Yang, Y.; Wang, J.; Zhou, Z. Saturation dependence of mass transfer for solute transport through residual unsaturated porous media. Int. J. Heat Mass Transfer 2022, 188, 122595. [Google Scholar] [CrossRef]
- Dou, Z.; Tang, S.; Zhang, X.; Liu, R.; Zhuang, C.; Wang, J.; Zhou, Z. Influence of Shear Displacement on Fluid Flow and Solute Transport in a 3D Rough Fracture. Lithoshere 2021, 2021, 1569736. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, Z.; Huang, G.; Zhi, D. Variations of Groundwater Quality in the Multi-Layered Aquifer System near the Luanhe River, China. Sustainability 2019, 11, 994. [Google Scholar] [CrossRef]
- Mengying, L. Investigation on Environmental Impact of Subway Construction in Jinan; Shanghai Jiao Tong University: Shanghai, China, 2018. [Google Scholar]
- Olsthoorn, T.N. The clogging of recharge wells, main subjects. In KIWA-Communications; KIWA: Rijswijk, The Netherlands, 1982. [Google Scholar]
- Yan, Y.; Yang, R.; Wang, X.; Yan, F.G.; Zhang, D. Analysis on Influencing Factors of Groundwater Recharge in Hohhot Area. West. Resour. 2014, 6, 193–195. [Google Scholar]
- Huang, X.D.; Shu, L.C.; Liu, P.G.; Wang, E. An experimental study on the problem of blockage during recharging of water injection wells. J. Hydraul. Eng. 2009, 40, 430–434. [Google Scholar]
- Dandan, L.; Fei, L.; Deren, M. Optimization of Soil Heavy Metal Sequential Extraction Procedures. Geoscience 2015, 29, 7. [Google Scholar]
- Fetter, C.W. Contaminant Hydrogeology; Waveland Press, Inc.: Long Grove, IL, USA, 2008; p. 500. [Google Scholar]
- Zhao, H.; Zhang, J.; Chen, H.; Wang, L.; Yang, Z. Localization of Groundwater Contaminant Sources Using Artificially Enhanced Catchment. Water 2020, 12, 1949. [Google Scholar] [CrossRef]
- Siyu, Y.; Zhiping, L.; Shuli, W.; Qiaoling, Y.; Lili, L. Multi-model comparative analysis of nitrobenzene penetration curve in the sandy strata. Geotech. Investig. Surv. 2020, 48, 40–46. [Google Scholar]
- Bu, X.F.; Wan, W.F. Experimental Research on the Hydrodynamic Dispersion Characteristics of Loess Medium in Hilly Area of Western Henan Province. China Rural Water Hydropower 2021, 12, 27–31. [Google Scholar]

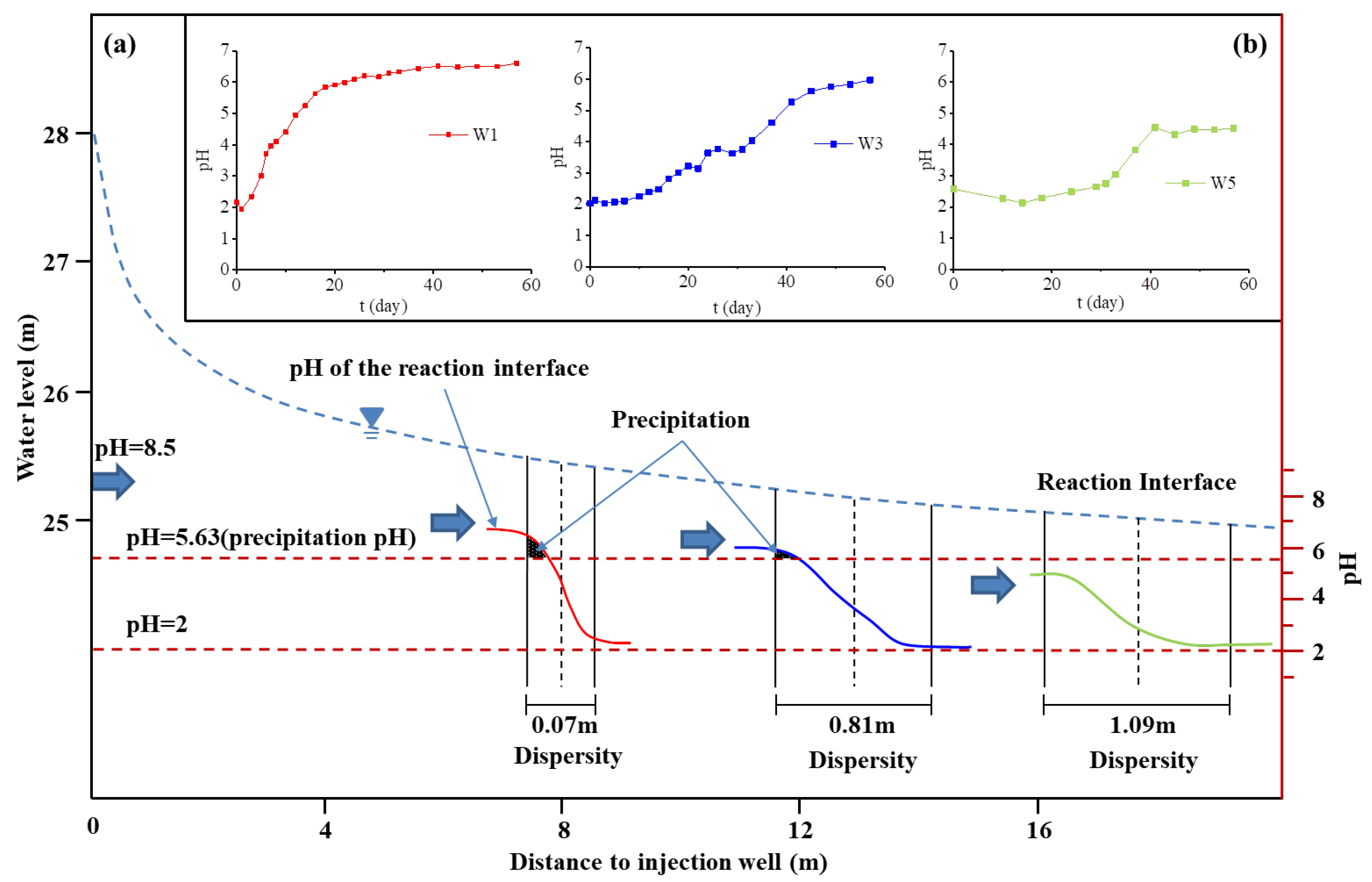
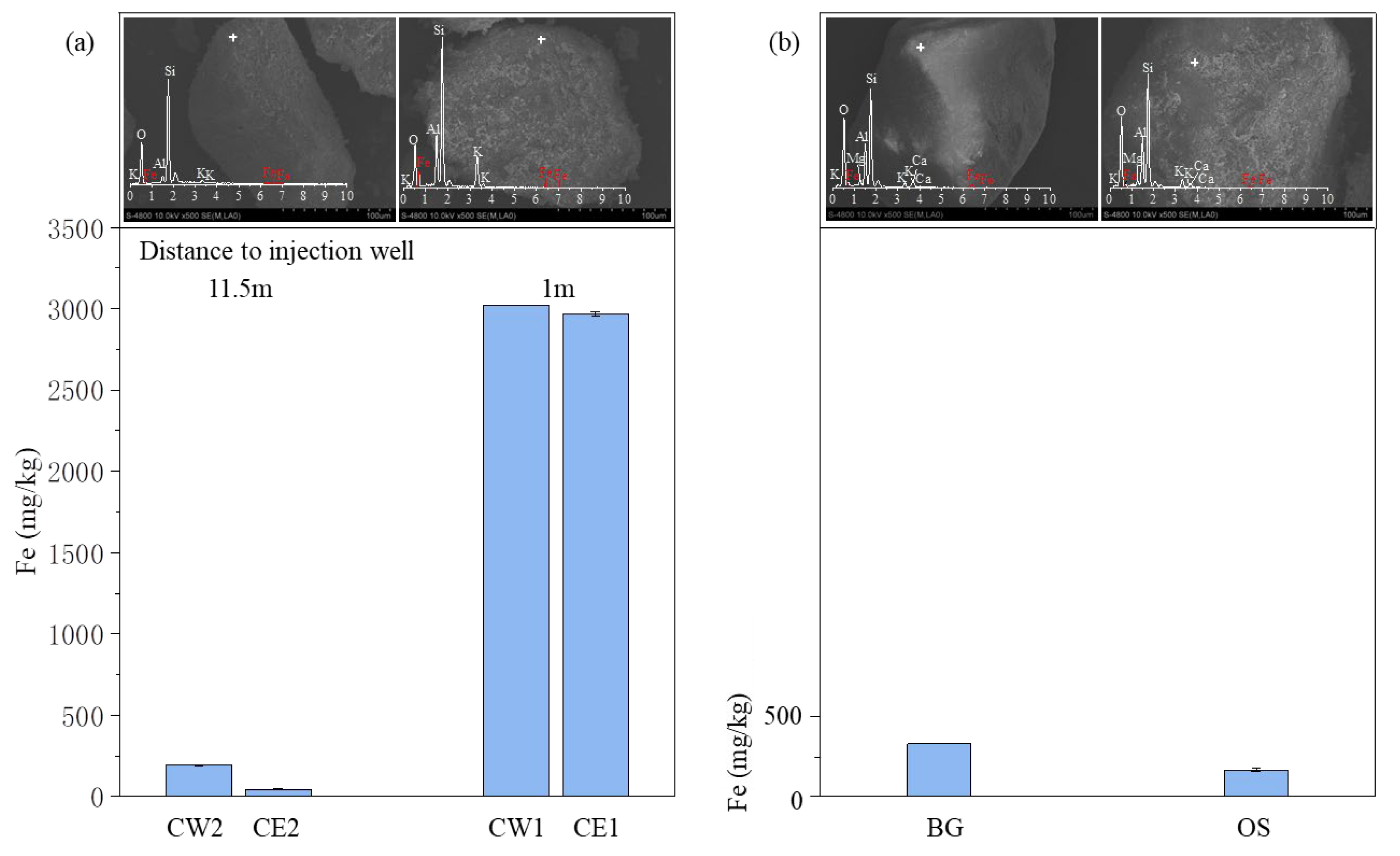
| Index | Pumped Groundwater | Injection Water (Average Value) | Monitor Well | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Western Zone | Eastern Zone | ||||||||
| W1 | W2 | W3 | W4 | W5 | W6 | E1 | |||
| pH | 1.29~1.8 | 8.5 | 2.15 | 2.24 | 2.03 | 1.81 | 2.57 | 2.66 | 1.2 |
| Fe (mg/L) | 912~2180 | — | 213.6 | 229.1 | 264.2 | 243.1 | 66.02 | 64.3 | 8880 |
| COD (mg/L) | 1249~4650 | 1610 | 820 | 94.5 | 1226.5 | 2287 | 320.5 | 311 | 9240 |
| HAc (mg/L) | 1050~3900 | 1560 | 728 | 520 | 1086 | 2130 | 290 | 280 | 8224 |
| Distance to the injection well (m) | 8 | 8.2 | 12.3 | 16.6 | 17.15 | 18.2 | 8.45 | ||
| Stage | Stage 1 | Stage 2 | Total | ||
|---|---|---|---|---|---|
| Stage 1.1 | Stage 1.2 | Stage 1.3 | |||
| Time | 0~8th | 9~19th | 20th~30th | 31st~57th | 57 d |
| Injection pressure (MPa) | 0~0.2 | 0.24~0.25 | 0.28~0.29 | 0.3~0.32 | — |
| Injection volume (m3) | 2022 | 2783 | 2671 | 8186 | 15,662 |
| Pumping wells | PW1, PW2 | PW1, PW2, PE1, PE2 | — | ||
| Equilibrium Area | Total (mol) | Neutralized | Extracted | Others | |||
|---|---|---|---|---|---|---|---|
| Amount | Proportion (%) | Amount | Proportion (%) | Amount | Proportion (%) | ||
| PW1 | 8648.69 | 8.72 | 0.10 | 8085.62 | 93.49 | 554.34 | 6.41 |
| PW2 | 25,705.49 | 7.81 | 0.03 | 25,049.46 | 97.45 | 648.22 | 2.52 |
| PE1 | 27,146.96 | 1.67 | 0.01 | 27,048.87 | 99.64 | 96.42 | 0.36 |
| PE2 | 29,756.84 | 5.23 | 0.02 | 29,433.67 | 98.91 | 317.95 | 1.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, F.; Zhang, J.; Chen, J.; Chen, H.; Barnie, S. In Situ Pumping–Injection Remediation of Strong Acid–High Salt Groundwater: Displacement–Neutralization Mechanism and Influence of Pore Blocking. Water 2022, 14, 2720. https://doi.org/10.3390/w14172720
Yuan F, Zhang J, Chen J, Chen H, Barnie S. In Situ Pumping–Injection Remediation of Strong Acid–High Salt Groundwater: Displacement–Neutralization Mechanism and Influence of Pore Blocking. Water. 2022; 14(17):2720. https://doi.org/10.3390/w14172720
Chicago/Turabian StyleYuan, Fang, Jia Zhang, Jian Chen, Honghan Chen, and Samuel Barnie. 2022. "In Situ Pumping–Injection Remediation of Strong Acid–High Salt Groundwater: Displacement–Neutralization Mechanism and Influence of Pore Blocking" Water 14, no. 17: 2720. https://doi.org/10.3390/w14172720








