Water Cleaning Adsorptive Membranes for Efficient Removal of Heavy Metals and Metalloids
Abstract
:1. Introduction: A Broader Context
1.1. The Problem of Heavy Metals and Metalloids
1.2. General Removal Methods of Heavy Metals and Metalloids
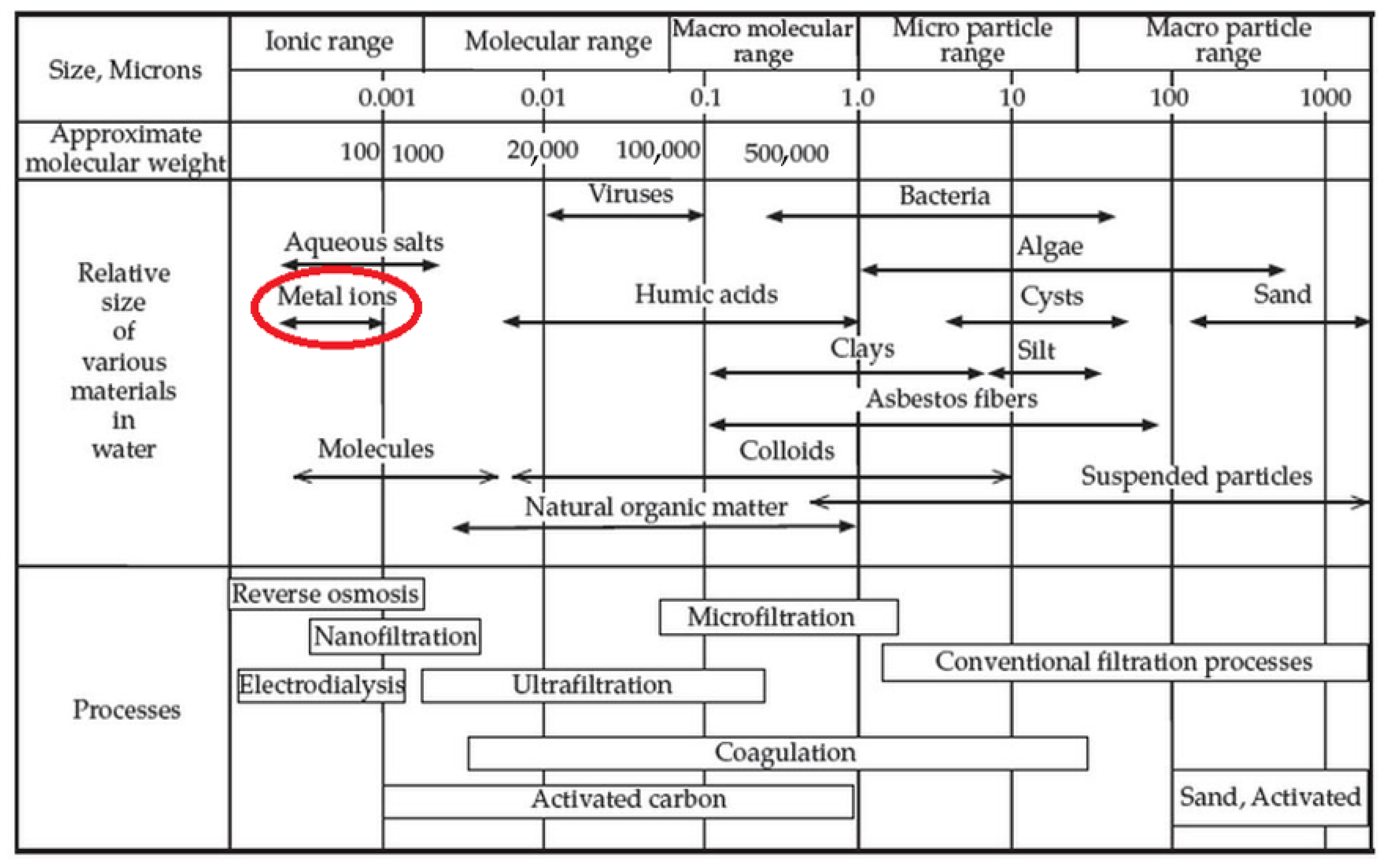
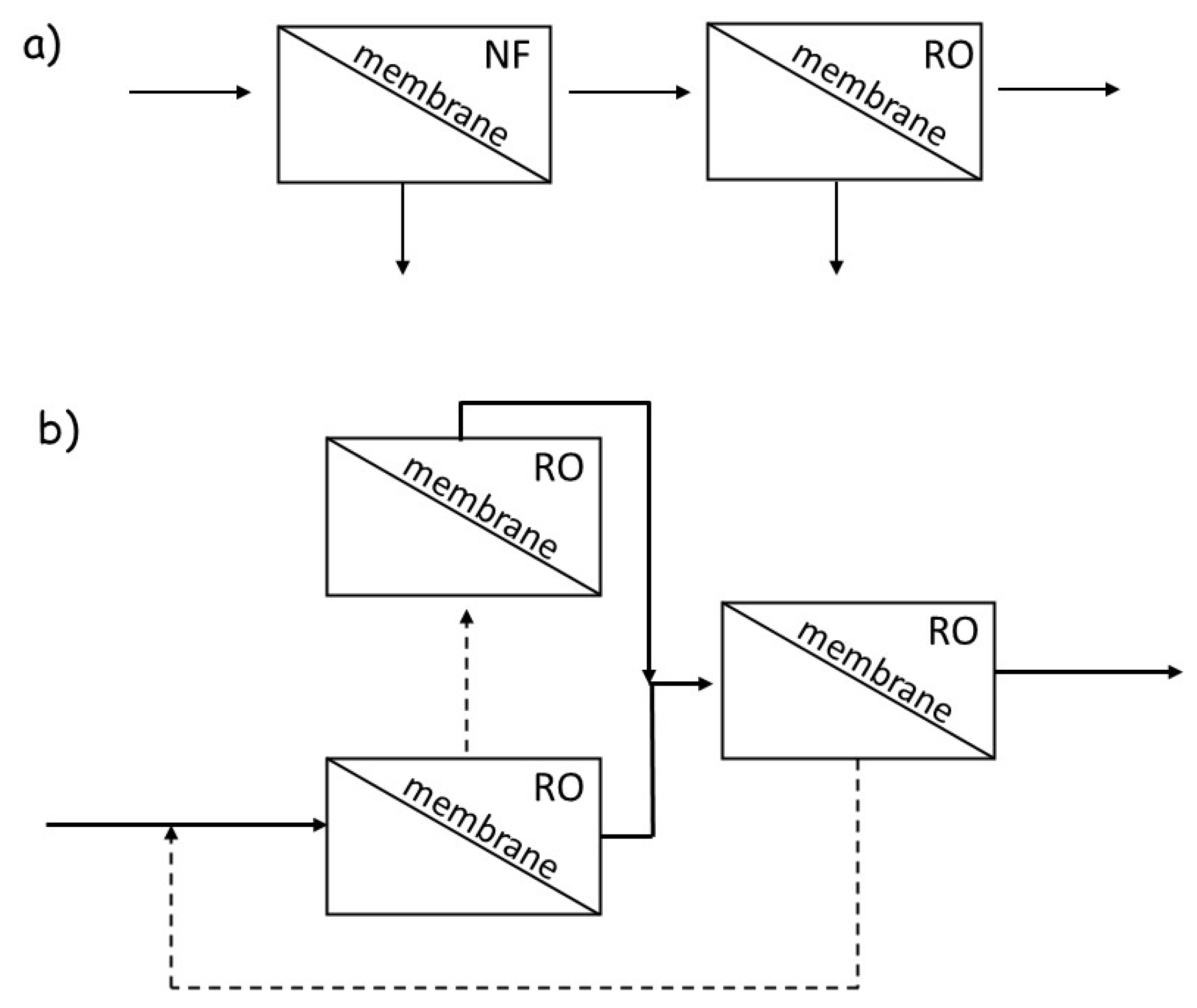
1.3. A Glimpse at Membrane Processes for Heavy Metals and Metalloids Removal
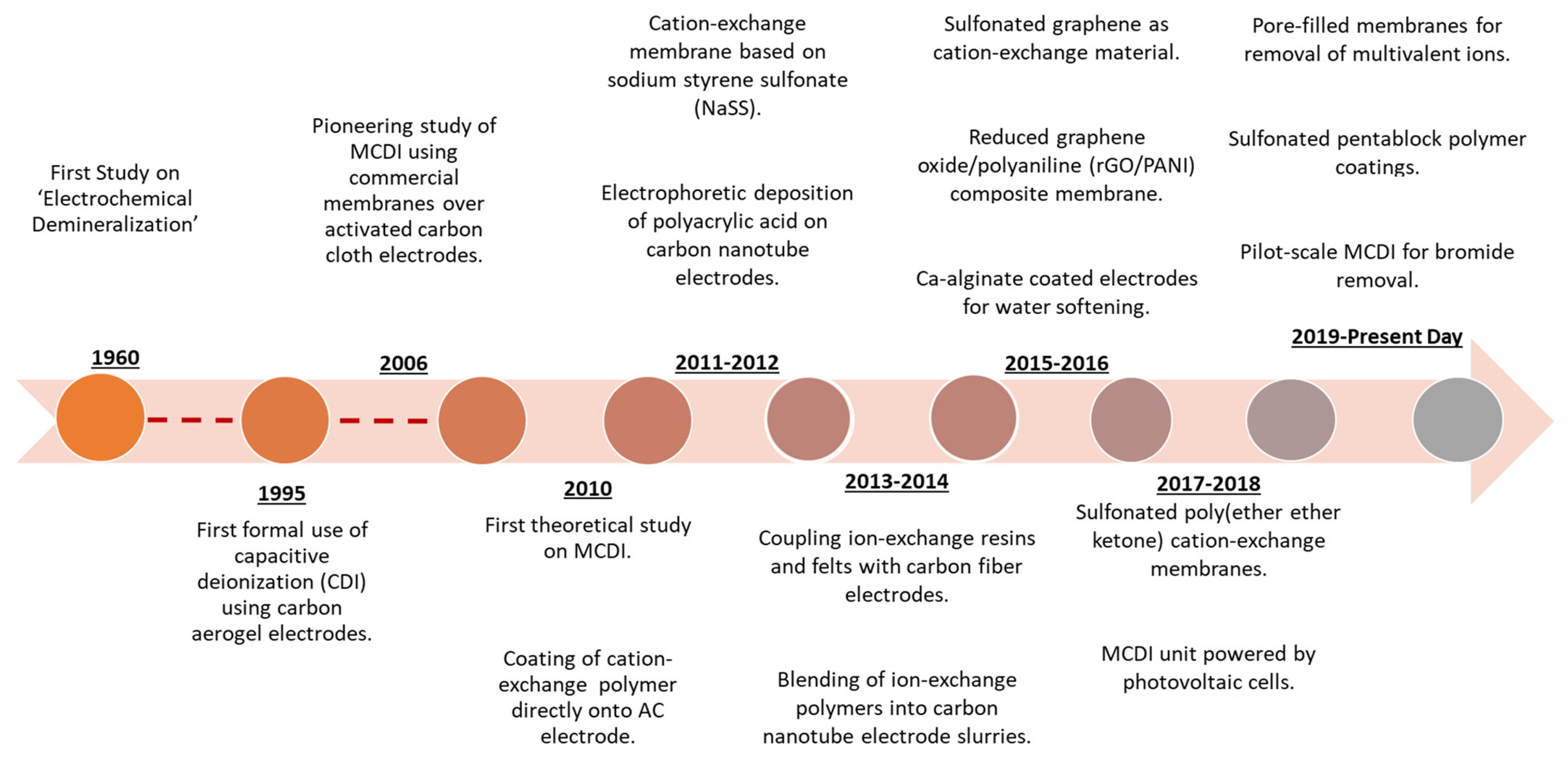
2. Adsorptive Membrane Technology
2.1. Adsorptive Membranes
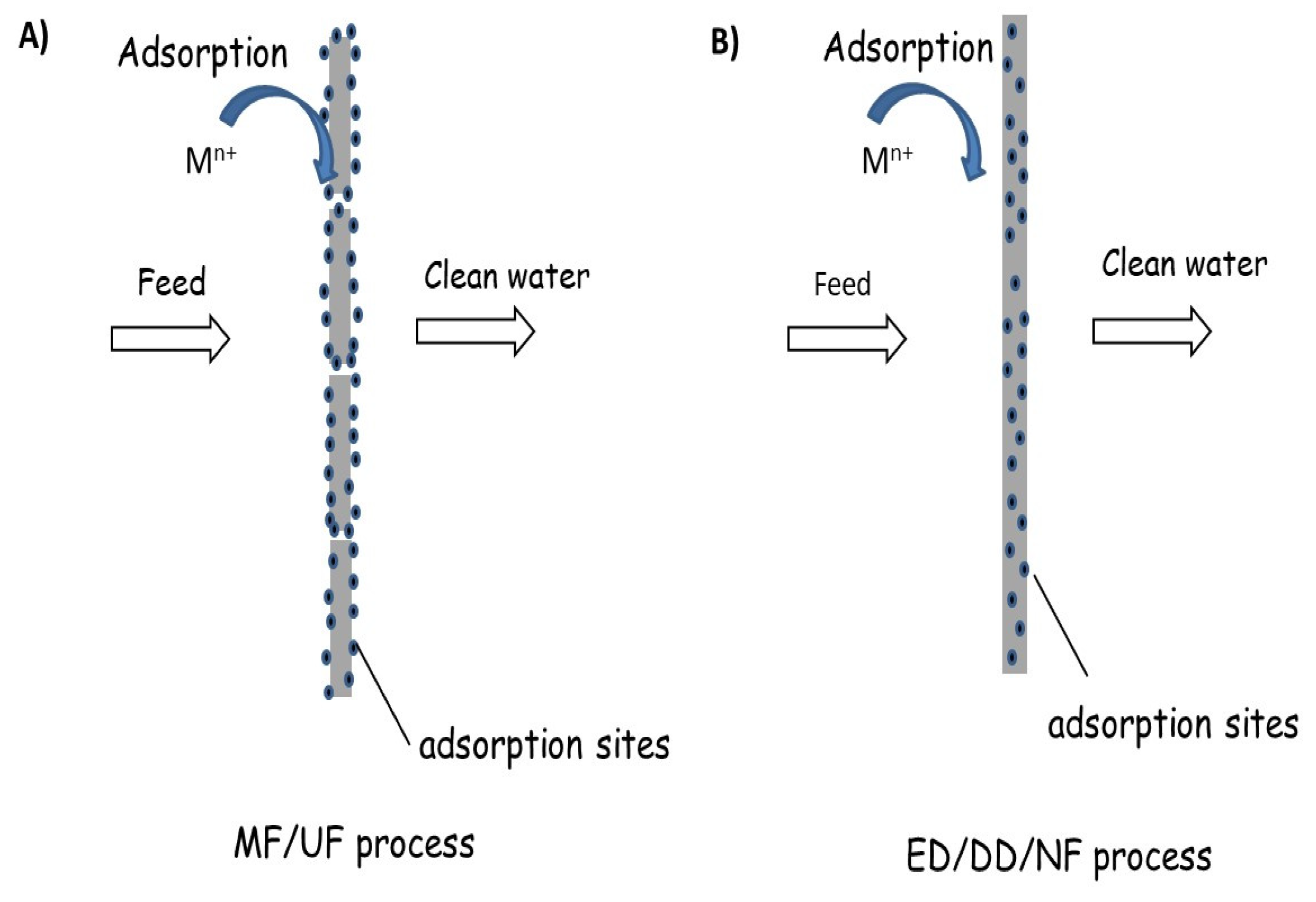
2.1.1. Ways to Introduce Adsorbents into/onto Separation Membranes
2.1.2. Types of Adsorptive Membranes
Bio-Based Adsorptive Membranes
Bio-Inspired Adsorptive Membranes
Inorganic Adsorptive Membranes
Functionalized Polymers-Based Adsorptive Membranes
Mixed Matrix Adsorptive Membranes
3. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kinuthia, G.K.; Ngure, V.; Beti, D.; Lugalia, R.; Wangila, A.; Kamau, L. Levels of heavy metals in wastewater and soil samples from open drainage channels in Nairobi, Kenya: Community health implication. Sci. Rep. 2020, 10, 8434. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Khan, E.; Ilahi, I. Environmental Chemistry and Ecotoxicology of Hazardous Heavy Metals: Environmental Persistence, Toxicity, and Bioaccumulation. J. Chem. 2019, 2019, 6730305. [Google Scholar] [CrossRef]
- Vaseashta, A.; Maftei, C. (Eds.) Water Safety, Security and Sustainability; Springer International Publishing: Cham, Switzerland, 2021. [Google Scholar]
- Zhao, M.; Xu, Y.; Zhang, C.; Rong, H.; Zeng, G. New trends in removing heavy metals from wastewater. Appl. Microb. Biotechnol. 2016, 100, 6509–6518. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Monier, M.; Abdel-Latif, D. Modification and characterization of PET fibers for fast removal of Hg(II), Cu(II) and Co(II) metal ions from aqueous solutions. J. Hazard. Mater. 2013, 250, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Takano, J.; Miwa, K.; Fujiwara, T. Boron transport mechanisms: Collaboration of channels and transporters. Trends Plant Sci. 2008, 13, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Badruk, M.; Kabay, N.; Demircioglu, M.; Mordogan, H.; Ipekoglu, U. Removal of boron from wastewater of geothermal power plant by selective ion-exchange resins. I. Batch sorption–elution studies. Sep. Sci. Technol. 1999, 34, 2553–2569. [Google Scholar] [CrossRef]
- Oki, T.; Kanae, S. Global hydrological cycles and world water resources. Science 2006, 313, 1068–1072. [Google Scholar] [CrossRef]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef]
- Kabay, N.; Güler, E.; Bryjak, M. Boron in seawater and methods for its separation—A review. Desalination 2010, 261, 212–217. [Google Scholar] [CrossRef]
- Güler, E.; Kaya, C.; Kabay, N.; Arda, M. Boron removal from seawater: State-of-the-art review. Desalination 2015, 356, 85–93. [Google Scholar] [CrossRef]
- Gemici, Ü.; Tarcan, G.; Helvacı, C.; Somay, A.M. High arsenic and boron concentrations in groundwaters related to mining activity in the Bigadiç borate deposits (Western Turkey). Appl. Geochem. 2008, 23, 2462–2476. [Google Scholar] [CrossRef]
- Geneva, S. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Hilal, N.; Kim, G.; Somerfield, C. Boron removal from saline water: A comprehensive review. Desalination 2011, 273, 23–35. [Google Scholar] [CrossRef]
- Eaton, S.V. Effects of boron deficiency and excess on plants. Plant Physiol. 1940, 15, 95. [Google Scholar] [CrossRef]
- Xu, F.; Goldbach, H.E.; Brown, P.H.; Bell, R.W.; Fujiwara, T.; Hunt, C.D.; Goldberg, S.; Shi, L. Advances in Plant and Animal Boron Nutrition; Springer: Heidelberg, The Netherlands, 2007. [Google Scholar]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mazumder, M.J.; Al-Attas, O.; Husain, T. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci. Total Environ. 2016, 569, 476–488. [Google Scholar] [CrossRef]
- Tseng, W.-P. Effects and dose-response relationships of skin cancer and blackfoot disease with arsenic. Environ. Health Perspect. 1977, 19, 109–119. [Google Scholar] [CrossRef]
- Nable, R.O.; Bañuelos, G.S.; Paull, J.G. Boron toxicity. Plant Soil 1997, 193, 181–198. [Google Scholar] [CrossRef]
- Guo, Q.; Zhang, Y.; Cao, Y.; Wang, Y.; Yan, W. Boron sorption from aqueous solution by hydrotalcite and its preliminary application in geothermal water deboronation. Environ. Sci. Pollut. Res. 2013, 20, 8210–8219. [Google Scholar] [CrossRef]
- Fu, F.; Zeng, H.; Cai, Q.; Qiu, R.; Yu, J.; Xiong, Y. Effective removal of coordinated copper from wastewater using a new dithiocarbamate-type supramolecular heavy metal precipitant. Chemosphere 2007, 69, 1783–1789. [Google Scholar] [CrossRef]
- Ghosh, P.; Samanta, A.N.; Ray, S. Reduction of COD and removal of Zn2+ from rayon industry wastewater by combined electro-Fenton treatment and chemical precipitation. Desalination 2011, 266, 213–217. [Google Scholar] [CrossRef]
- Kurniawan, T.A.; Chan, G.Y.; Lo, W.-H.; Babel, S. Physico–chemical treatment techniques for wastewater laden with heavy metals. Chem. Eng. J. 2006, 118, 83–98. [Google Scholar] [CrossRef]
- Barakat, M. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Usmana, M.; Zarebanadkouki, M.; Waseem, M.; Katsoyiannis, I.A.; Ernst, M. Mathematical modeling of arsenic(V) adsorption onto iron oxyhydroxides in an adsorption-submerged membrane hybrid system. J. Hazard. Mater. 2020, 400, 123221. [Google Scholar] [CrossRef] [PubMed]
- Inglezakis, V.J.; Stylianou, M.A.; Gkantzou, D.; Loizidou, M.D. Removal of Pb(II) from aqueous solutions by using clinoptilolite and bentonite as adsorbents. Desalination 2007, 210, 248–256. [Google Scholar] [CrossRef]
- Argun, M.E. Use of clinoptilolite for the removal of nickel ions from water: Kinetics and thermodynamics. J. Hazard. Mater. 2008, 150, 587–595. [Google Scholar] [CrossRef]
- Khelifa, A.; Moulay, S.; Naceur, A. Treatment of metal finishing effluents by the electroflotation technique. Desalination 2005, 181, 27–33. [Google Scholar] [CrossRef]
- Peter-Varbanets, M.; Zurbrügg, C.; Swartz, C.; Pronk, W. Decentralized systems for potable water and the potential of membrane technology. Water Res. 2009, 43, 245–265. [Google Scholar] [CrossRef]
- Mohammad, A.W.; Othaman, R.; Hilal, N. Potential use of nanofiltration membranes in treatment of industrial wastewater from Ni-P electroless plating. Desalination 2004, 168, 241–252. [Google Scholar] [CrossRef]
- Al-Rashdi, B.; Somerfield, C.; Hilal, N. Heavy metals removal using adsorption and nanofiltration techniques. Sep. Purif. Rev. 2011, 40, 209–259. [Google Scholar] [CrossRef]
- Al-Rashdi, B.; Johnson, D.; Hilal, N. Removal of heavy metal ions by nanofiltration. Desalination 2013, 315, 2–17. [Google Scholar] [CrossRef]
- Kotrappanavar, N.S.; Hussain, A.; Abashar, M.; Al-Mutaz, I.S.; Aminabhavi, T.M.; Nadagouda, M.N. Prediction of physical properties of nanofiltration membranes for neutral and charged solutes. Desalination 2011, 280, 174–182. [Google Scholar] [CrossRef]
- Alzahrani, S.; Mohammad, A.W.; Hilal, N.; Abdullah, P.; Jaafar, O. Comparative study of NF and RO membranes in the treatment of produced water—Part I: Assessing water quality. Desalination 2013, 315, 18–26. [Google Scholar] [CrossRef]
- Cingolani, D.; Fatone, F.; Frison, N.; Spinelli, M.; Eusebi, A.L. Pilot-scale multi-stage reverse osmosis (DT-RO) for water recovery from landfill leachate. Waste Manag. 2018, 76, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Urase, T.; Kitawaki, H.; Rahman, M.; Rhahman, M.; Yamamoto, K. Modeling of arsenic rejection considering affinity and steric hindrance effect in nanofiltration membranes. Water Sci. Technol. 2000, 42, 173–180. [Google Scholar] [CrossRef]
- Teychene, B.; Collet, G.; Gallard, H.; Croue, J.-P. A comparative study of boron and arsenic (III) rejection from brackish water by reverse osmosis membranes. Desalination 2013, 310, 109–114. [Google Scholar] [CrossRef]
- Woods, W.G. An introduction to boron: History, sources, uses, and chemistry. Environ. Health Perspect. 1994, 102, 5–11. [Google Scholar]
- Bernstein, R.; Belfer, S.; Freger, V. Toward improved boron removal in RO by membrane modification: Feasibility and challenges. Environ. Sci. Technol. 2011, 45, 3613–3620. [Google Scholar] [CrossRef]
- Sagiv, A.; Semiat, R. Analysis of parameters affecting boron permeation through reverse osmosis membranes. J. Membr. Sci. 2004, 243, 79–87. [Google Scholar] [CrossRef]
- Greenlee, L.F.; Lawler, D.F.; Freeman, B.D.; Marrot, B.; Moulin, P. Reverse osmosis desalination: Water sources, technology, and today’s challenges. Water Res. 2009, 43, 2317–2348. [Google Scholar] [CrossRef]
- Garb, Y. Desalination in Israel: Status, Prospects, and Contexts, Water Wisdom; Rutgers University Press: New Brunswick, NJ, USA, 2010; pp. 238–245. [Google Scholar]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination: Energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef]
- Soo, K.W.; Wong, K.C.; Goh, P.S.; Ismail, A.F.; Othman, N. Efficient heavy metal removal by thin film nanocomposite forward osmosis membrane modified with geometrically different bimetallic oxide. J. Water Process Eng. 2020, 38, 101591. [Google Scholar] [CrossRef]
- He, M.; Wang, L.; Lv, Y.; Wang, X.; Zhu, J.; Zhang, Y.; Liu, T. Novel polydopamine/metal organic framework thin film nanocomposite forward osmosis membrane for salt rejection and heavy metal removal. Chem. Eng. J. 2020, 389, 124452. [Google Scholar] [CrossRef]
- Landsman, M.R.; Sujanani, R.; Brodfuehrer, S.H.; Cooper, C.M.; Darr, A.G.; Davis, R.J.; Kim, K.; Kum, S.; Nalley, L.K.; Nomaan, S.M. Water treatment: Are membranes the panacea? Annu. Rev. Chem. Biomol. Eng. 2020, 11, 559–585. [Google Scholar] [CrossRef]
- Bolisetty, S.; Peydayesh, M.; Mezzenga, R. Sustainable technologies for water purification from heavy metals: Review and analysis. Chem. Soc. Rev. 2019, 48, 463–487. [Google Scholar] [CrossRef]
- McNair, R.; Szekely, G.; Dryfe, R.A.W. Ion-Exchange Materials for Membrane Capacitive Deionization. ACS EST Water 2021, 1, 217–239. [Google Scholar] [CrossRef]
- Zhao, R.; Porada, S.; Biesheuvel, P.; Van der Wal, A. Energy consumption in membrane capacitive deionization for different water recoveries and flow rates, and comparison with reverse osmosis. Desalination 2013, 330, 35–41. [Google Scholar] [CrossRef]
- Mack, C.; Burgess, J.; Duncan, J. Membrane bioreactors for metal recovery from wastewater: A review. Water SA 2004, 30, 521–532. [Google Scholar] [CrossRef]
- Arican, B.; Gokcay, C.F.; Yetis, U. Mechanistics of nickel sorption by activated sludge. Process Biochem. 2002, 37, 1307–1315. [Google Scholar] [CrossRef]
- Kelly, C.J.; Tumsaroj, N.; Lajoie, C.A. Assessing wastewater metal toxicity with bacterial bioluminescence in a bench-scale wastewater treatment system. Water Res. 2004, 38, 423–431. [Google Scholar] [CrossRef]
- Holloway, R.W.; Achilli, A.; Cath, T.Y. The osmotic membrane bioreactor: A critical review. Environ. Sci. Water Res. Technol. 2015, 1, 581–605. [Google Scholar] [CrossRef]
- Thaçi, B.S.; Gashi, S.T. Reverse Osmosis Removal of Heavy Metals from Wastewater Effluents Using Biowaste Materials Pretreatment. Pol. J. Environ. Stud. 2019, 28, 337–341. [Google Scholar] [CrossRef]
- Chan, B.; Dudeney, A. Reverse osmosis removal of arsenic residues from bioleaching of refractory gold concentrates. Miner. Eng. 2008, 21, 272–278. [Google Scholar] [CrossRef]
- Ipek, U. Removal of Ni (II) and Zn (II) from an aqueous solutionby reverse osmosis. Desalination 2005, 174, 161–169. [Google Scholar] [CrossRef]
- Rodrigues Pires da Silva, J.; Merçon, F.; Guimarães Costa, C.M.; Radoman Benjo, D. Application of reverse osmosis process associated with EDTA complexation for nickel and copper removal from wastewater. Desalin. Water Treat. 2016, 57, 19466–19474. [Google Scholar] [CrossRef]
- Xu, P.; Capito, M.; Cath, T.Y. Selective removal of arsenic and monovalent ions from brackish water reverse osmosis concentrate. J. Hazard. Mater. 2013, 260, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Petrinic, I.; Korenak, J.; Povodnik, D.; Hélix-Nielsen, C. A feasibility study of ultrafiltration/reverse osmosis (UF/RO)-based wastewater treatment and reuse in the metal finishing industry. J. Clean. Prod. 2015, 101, 292–300. [Google Scholar] [CrossRef]
- Cséfalvay, E.; Pauer, V.; Mizsey, P. Recovery of copper from process waters by nanofiltration and reverse osmosis. Desalination 2009, 240, 132–142. [Google Scholar] [CrossRef]
- Chung, S.; Kim, S.; Kim, J.-O.; Chung, J. Feasibility of combining reverse osmosis–ferrite process for reclamation of metal plating wastewater and recovery of heavy metals. Ind. Eng. Chem. Res. 2014, 53, 15192–15199. [Google Scholar] [CrossRef]
- Mohsen-Nia, M.; Montazeri, P.; Modarress, H. Removal of Cu2+ and Ni2+ from wastewater with a chelating agent and reverse osmosis processes. Desalination 2007, 217, 276–281. [Google Scholar] [CrossRef]
- Qdais, H.A.; Moussa, H. Removal of heavy metals from wastewater by membrane processes: A comparative study. Desalination 2004, 164, 105–110. [Google Scholar] [CrossRef]
- Malamis, S.; Katsou, E.; Takopoulos, K.; Demetriou, P.; Loizidou, M. Assessment of metal removal, biomass activity and RO concentrate treatment in an MBR–RO system. J. Hazard. Mater. 2012, 209, 1–8. [Google Scholar] [CrossRef]
- Nanofiltration Membranes to Treat Industrial Wastewater from Heavy Metals. Available online: https://phys.org/news/2021-04-nanofiltration-membranes-industrial-wastewater-heavy.html (accessed on 18 April 2022).
- Gao, J.; Wang, K.Y.; Chung, T.-S. Design of nanofiltration (NF) hollow fiber membranes made from functionalized bore fluids containing polyethyleneimine (PEI) for heavy metal removal. J. Membr. Sci. 2020, 603, 118022. [Google Scholar] [CrossRef]
- Gherasim, C.-V.; Mikulášek, P. Influence of operating variables on the removal of heavy metal ions from aqueous solutions by nanofiltration. Desalination 2014, 343, 67–74. [Google Scholar] [CrossRef]
- Cui, Y.; Ge, Q.; Liu, X.-Y.; Chung, T.-S. Novel forward osmosis process to effectively remove heavy metal ions. J. Membr. Sci. 2014, 467, 188–194. [Google Scholar] [CrossRef]
- Zhao, P.; Gao, B.; Yue, Q.; Liu, S.; Shon, H.K. The performance of forward osmosis in treating high-salinity wastewater containing heavy metal Ni2+. Chem. Eng. J. 2016, 288, 569–576. [Google Scholar] [CrossRef]
- Mahmoud, A.; Hoadley, A.F. An evaluation of a hybrid ion exchange electrodialysis process in the recovery of heavy metals from simulated dilute industrial wastewater. Water Res. 2012, 46, 3364–3376. [Google Scholar] [CrossRef]
- Hamane, D.; Arous, O.; Kaouah, F.; Trari, M.; Kerdjoudj, H.; Bendjama, Z. Adsorption/photo-electrodialysis combination system for Pb2+ removal using bentonite/membrane/semiconductor. J. Environ. Chem. Eng. 2015, 3, 60–69. [Google Scholar] [CrossRef]
- Deghles, A.; Kurt, U. Treatment of tannery wastewater by a hybrid electrocoagulation/electrodialysis process. Chem. Eng. Process 2016, 104, 43–50. [Google Scholar] [CrossRef]
- Sadyrbaeva, T.Z. Removal of chromium (VI) from aqueous solutions using a novel hybrid liquid membrane—Electrodialysis process. Chem. Eng. Process 2016, 99, 183–191. [Google Scholar] [CrossRef]
- Dong, Y.; Liu, J.; Sui, M.; Qu, Y.; Ambuchi, J.J.; Wang, H.; Feng, Y. A combined microbial desalination cell and electrodialysis system for copper-containing wastewater treatment and high-salinity-water desalination. J. Hazard. Mater. 2017, 321, 307–315. [Google Scholar] [CrossRef]
- Su, Y.-N.; Lin, W.-S.; Hou, C.-H.; Den, W. Performance of integrated membrane filtration and electrodialysis processes for copper recovery from wafer polishing wastewater. J. Water Process Eng. 2014, 4, 149–158. [Google Scholar] [CrossRef]
- Nataraj, S.; Hosamani, K.; Aminabhavi, T. Potential application of an electrodialysis pilot plant containing ion-exchange membranes in chromium removal. Desalination 2007, 217, 181–190. [Google Scholar] [CrossRef]
- Lambert, J.; Rakib, M.; Durand, G.; Avila-Rodríguez, M. Treatment of solutions containing trivalent chromium by electrodialysis. Desalination 2006, 191, 100–110. [Google Scholar] [CrossRef]
- Moura, R.C.; Bertuol, D.A.; Ferreira, C.A.; Amado, F.D. Study of chromium removal by the electrodialysis of tannery and metal-finishing effluents. Int. J. Chem. Eng. 2012, 2012, 179312. [Google Scholar] [CrossRef]
- Kim, D.-H.; Choi, Y.-E.; Park, J.-S.; Kang, M.-S. Capacitive deionization employing pore-filled cation-exchange membranes for energy-efficient removal of multivalent cations. Electrochim. Acta 2019, 295, 164–172. [Google Scholar] [CrossRef]
- Ali, A.; Quist-Jensen, C.A.; Jørgensen, M.K.; Siekierka, A.; Christensen, M.L.; Bryjak, M.; Hélix-Nielsen, C.; Drioli, E. A review of membrane crystallization, forward osmosis and membrane capacitive deionization for liquid mining. Resour. Conserv. Recycl. 2021, 168, 105273. [Google Scholar] [CrossRef]
- Huang, Y.; Feng, X. Polymer-enhanced ultrafiltration: Fundamentals, applications and recent developments. J. Membr. Sci. 2019, 586, 53–83. [Google Scholar] [CrossRef]
- Chakraborty, S.; Dasgupta, J.; Farooq, U.; Sikder, J.; Drioli, E.; Curcio, S. Experimental analysis, modeling and optimization of chromium (VI) removal from aqueous solutions by polymer-enhanced ultrafiltration. J. Membr. Sci. 2014, 456, 139–154. [Google Scholar] [CrossRef]
- Huang, Y.; Wu, D.; Wang, X.; Huang, W.; Lawless, D.; Feng, X. Removal of heavy metals from water using polyvinylamine by polymer-enhanced ultrafiltration and flocculation. Sep. Purif. Technol. 2016, 158, 124–136. [Google Scholar] [CrossRef]
- Huang, Y.; Du, J.; Zhang, Y.; Lawless, D.; Feng, X. Batch process of polymer-enhanced ultrafiltration to recover mercury (II) from wastewater. J. Membr. Sci. 2016, 514, 229–240. [Google Scholar] [CrossRef]
- Ennigrou, D.J.; Gzara, L.; Romdhane, M.R.B.; Dhahbi, M. Cadmium removal from aqueous solutions by polyelectrolyte enhanced ultrafiltration. Desalination 2009, 246, 363–369. [Google Scholar] [CrossRef]
- Yin, N.; Wang, K.; Wang, L.; Li, Z. Amino-functionalized MOFs combining ceramic membrane ultrafiltration for Pb (II) removal. Chem. Eng. J. 2016, 306, 619–628. [Google Scholar] [CrossRef]
- Canizares, P.; Pérez, A.; Camarillo, R.; Linares, J. A semi-continuous laboratory-scale polymer enhanced ultrafiltration process for the recovery of cadmium and lead from aqueous effluents. J. Membr. Sci. 2004, 240, 197–209. [Google Scholar] [CrossRef]
- Oehmen, A.; Vergel, D.; Fradinho, J.; Reis, M.A.; Crespo, J.G.; Velizarov, S. Mercury removal from water streams through the ion exchange membrane bioreactor concept. J. Hazard. Mater. 2014, 264, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Katsou, E.; Malamis, S.; Loizidou, M. Performance of a membrane bioreactor used for the treatment of wastewater contaminated with heavy metals. Bioresour. Technol. 2011, 102, 4325–4332. [Google Scholar] [CrossRef]
- Mahmoudkhani, R.; Torabian, A.; Hassani, A.H.; Mahmoudkhani, R. Copper, cadmium and ferrous removal by membrane bioreactor. APCBEE Procedia 2014, 10, 79–83. [Google Scholar] [CrossRef]
- Aftab, B.; Khan, S.J.; Maqbool, T.; Hankins, N.P. Heavy metals removal by osmotic membrane bioreactor (OMBR) and their effect on sludge properties. Desalination 2017, 403, 117–127. [Google Scholar] [CrossRef]
- Marino, T.; Figoli, A. Arsenic removal by liquid membranes. Membranes 2015, 5, 150–167. [Google Scholar] [CrossRef]
- Ahmad, A.; Kusumastuti, A.; Derek, C.; Ooi, B. Emulsion liquid membrane for heavy metal removal: An overview on emulsion stabilization and destabilization. Chem. Eng. J. 2011, 171, 870–882. [Google Scholar] [CrossRef]
- Chang, S.H. Types of bulk liquid membrane and its membrane resistance in heavy metal removal and recovery from wastewater. Desalin. Water Treat. 2016, 57, 19785–19793. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Dutta, S.K.; Sikder, J.; Mandal, M.K. Computational and experimental study of chromium (VI) removal in direct contact membrane distillation. J. Membr. Sci. 2014, 450, 447–456. [Google Scholar] [CrossRef]
- Manna, A.K.; Sen, M.; Martin, A.R.; Pal, P. Removal of arsenic from contaminated groundwater by solar-driven membrane distillation. Environ. Pollut. 2010, 158, 805–811. [Google Scholar] [CrossRef]
- Murthy, Z.; Chaudhari, L.B. Application of nanofiltration for the rejection of nickel ions from aqueous solutions and estimation of membrane transport parameters. J. Hazard. Mater. 2008, 160, 70–77. [Google Scholar] [CrossRef]
- Murthy, Z.; Chaudhari, L.B. Separation of binary heavy metals from aqueous solutions by nanofiltration and characterization of the membrane using Spiegler–Kedem model. Chem. Eng. J. 2009, 150, 181–187. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. Npj Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Algieri, C.; Chakraborty, S.; Candamano, S. A Way to Membrane-Based Environmental Remediation for Heavy Metal Removal. Environments 2021, 8, 52. [Google Scholar] [CrossRef]
- Aijaz, M.O.; Karim, M.R.; Omar, N.M.A.; Othman, M.H.D.; Wahab, M.A.; Akhtar, M.; Uzzaman, M.A.; Alharbi, H.M.; Wazeer, I. Recent Progress, Challenges, and Opportunities of Membrane Distillation for Heavy Metals Removal. Chem. Rec. 2022, 22, e202100323. [Google Scholar] [CrossRef]
- Khulbe, K.C.; Matsuura, T. Removal of heavy metals and pollutants by membrane adsorption techniques. Appl. Water Sci. 2018, 8, 19. [Google Scholar] [CrossRef]
- Vo, T.S.; Hossain, M.M.; Jeong, H.M.; Kim, K. Heavy metal removal applications using adsorptive membranes. Nano Converg. 2020, 7, 36. [Google Scholar] [CrossRef]
- Adam, M.R.; Othman, M.H.D.; Kurniawan, T.A.; Puteh, M.H.; Ismail, A.F.; Khongnakorn, W.; Rahman, M.A.; Jaafar, J. Advances in adsorptive membrane technology for water treatment and resource recovery applications: A critical review. J. Environ. Chem. Eng. 2022, 10, 107633. [Google Scholar] [CrossRef]
- Kim, S.; Nam, S.-N.; Jang, A.; Jang, M.; Min, C.; Ahjeong, P.; Namguk, S.; Jiyong, H.; Heo, J.; Yoon, Y. Review of adsorption–membrane hybrid systems for water and wastewater treatment. Chemosphere 2022, 286, 131916. [Google Scholar] [CrossRef] [PubMed]
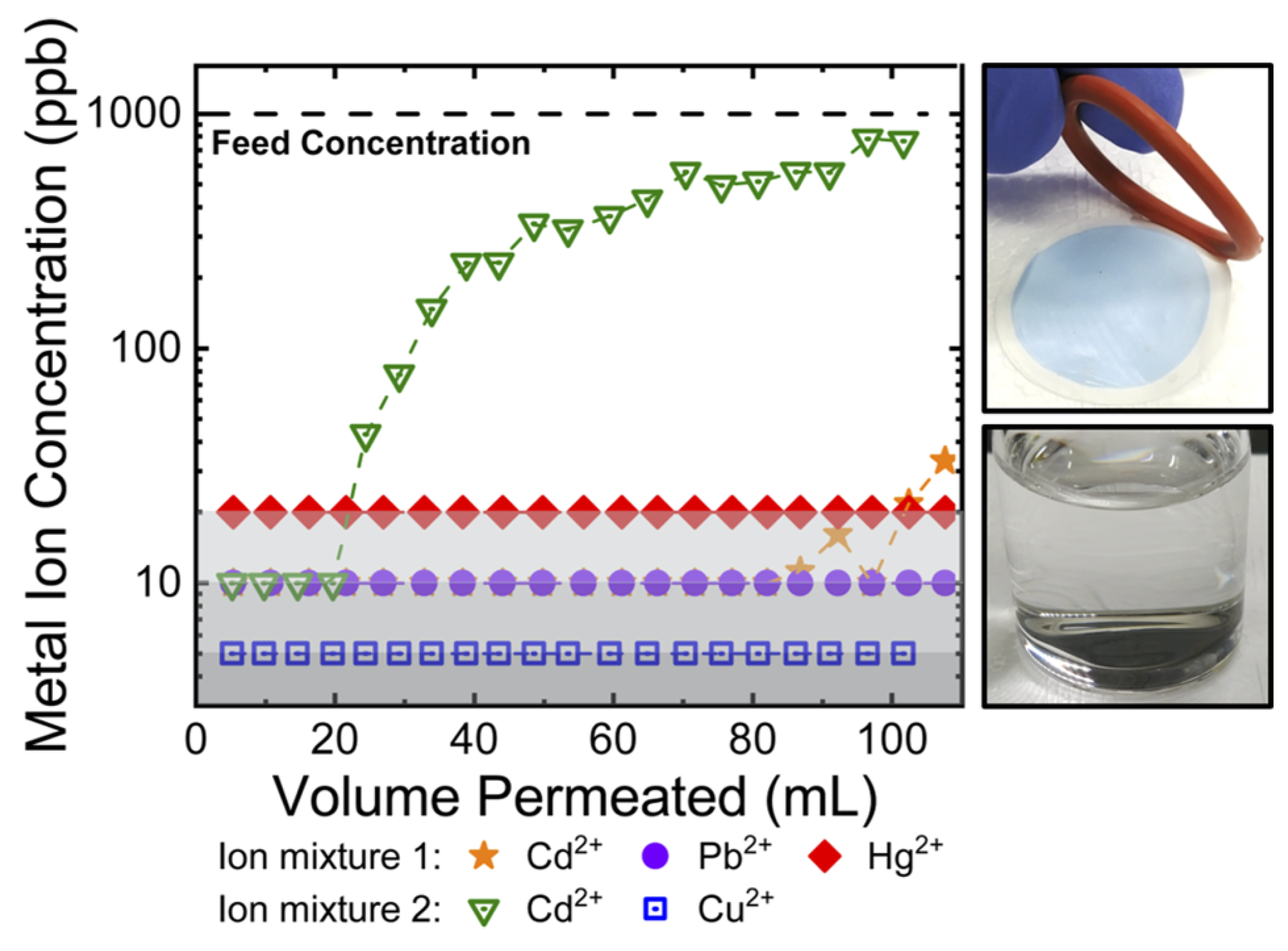
- Glass, S.; Mantel, T.; Appold, M.; Sitashree, S.; Usman, M.; Ernst, M.; Filiz, V. Amine-Terminated PAN Membranes as Anion-Adsorber Materials. Chem. Ing. Tech. 2021, 93, 1396–1400. [Google Scholar] [CrossRef]
- Chand, A.A.K.; Bajer, B.; Schneider, E.S.; Mantel, T.; Ernst, M.; Filiz, V.; Glass, S. Modification of Polyacrylonitrile Ultrafiltration Membranes to Enhance the Adsorption of Cations and Anions. Membranes 2022, 12, 580. [Google Scholar] [CrossRef]
- Usman, M.; Katsoyiannis, I.; Rodrigues, J.H.; Ernst, M. Arsenate removal from drinking water using by-products from conventional iron oxyhydroxides production as adsorbents coupled with submerged microfiltration unit. Environ. Sci. Pollut. Res. 2021, 28, 59063–59075. [Google Scholar] [CrossRef]
- Bolisetty, S.; Mezzenga, R. Amyloid–carbon hybrid membranes for universal water purification. Nat. Nanotechnol. 2016, 11, 365–371. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Li, H.; Yang, C. Chitosan membrane adsorber for low concentration copper ion removal. Carbohydr. Polym. 2016, 146, 274–281. [Google Scholar] [CrossRef]
- Liang, H.W.; Cao, X.; Zhang, W.J.; Lin, H.T.; Zhou, F.; Chen, L.F.; Yu, S.H. Robust and highly efficient free-standing carbonaceous nanofiber membranes for water purification. Adv. Funct. Mater. 2011, 21, 3851–3858. [Google Scholar] [CrossRef]
- Chitpong, N.; Husson, S.M. High-capacity, nanofiber-based ion-exchange membranes for the selective recovery of heavy metals from impaired waters. Sep. Purif. Technol. 2017, 179, 94–103. [Google Scholar] [CrossRef]
- Chitpong, N.; Husson, S.M. Polyacid functionalized cellulose nanofiber membranes for removal of heavy metals from impaired waters. J. Membr. Sci. 2017, 523, 418–429. [Google Scholar] [CrossRef]
- Weidman, J.L.; Mulvenna, R.A.; Boudouris, B.W.; Phillip, W.A. Nanoporous block polymer thin films functionalized with bio-inspired ligands for the efficient capture of heavy metal ions from water. ACS Appl. Mater. Interfaces 2017, 9, 19152–19160. [Google Scholar] [CrossRef]
- Weidman, J.L.; Mulvenna, R.A.; Boudouris, B.W.; Phillip, W.A. Nanostructured membranes from triblock polymer precursors as highcapacity copper adsorbents. Langmuir 2015, 31, 11113–11123. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Jia, Z.; Wen, J.; Li, S.; Peng, W.; Huang, R.; Xu, X. Progress in adsorptive membranes for separation–A review. Sep. Purif. Technol. 2021, 255, 117772. [Google Scholar] [CrossRef]
- Roper, D.K.; Lightfoot, E.N. Separation of biomolecules using adsorptive membranes. J. Chromatogr. A 1995, 702, 3–26. [Google Scholar] [CrossRef]
- Kubota, N.; Konno, Y.; Miura, S.; Saito, K.; Sugita, K.; Watanabe, K.; Sugo, T. Comparison of two convection-aided protein adsorption methods using porous membranes and perfusion beads. Biotechnol. Prog. 1996, 12, 869–872. [Google Scholar] [CrossRef]
- Sujanani, R.; Landsman, M.R.; Jiao, S.; Moon, J.D.; Shell, M.S.; Lawler, D.F.; Katz, L.E.; Freeman, B.D. Designing solute-tailored selectivity in membranes: Perspectives for water reuse and resource recovery. ACS Macro Lett. 2020, 9, 1709–1717. [Google Scholar] [CrossRef]
- Warnock, S.J.; Sujanani, R.; Zofchak, E.S.; Zhao, S.; Dilenschneider, T.J.; Hanson, K.G.; Mukherjee, S.; Ganesan, V.; Freeman, B.D.; Abu-Omar, M.M.; et al. Engineering Li/Na selectivity in 12-Crown-4-functionalized polymer membranes. Proc. Natl. Acad. Sci. USA 2021, 118, e2022197118. [Google Scholar] [CrossRef]
- Uliana, A.A.; Bui, N.T.; Kamcev, J.; Taylor, M.K.; Urban, J.J.; Long, J.R. Ion-capture electrodialysis using multifunctional adsorptive membranes. Science 2021, 372, 296–299. [Google Scholar] [CrossRef]
- Adil, H.I.; Thalji, M.R.; Yasin, S.A.; Saeed, I.A.; Assiri, M.A.; Chong, K.F.; Ali, G.A.M. Metal–organic frameworks (MOFs) based nanofiber architectures for the removal of heavy metal ions. RSC Adv. 2022, 12, 1433. [Google Scholar] [CrossRef] [PubMed]
- Anantharaman, A.; Chun, Y.; Hua, T.; Chew, J.W.; Wang, R. Pre-deposited dynamic membrane filtration—A review. Water Res. 2020, 173, 115558. [Google Scholar] [CrossRef]
- Ersahin, M.E.; Ozgun, H.; Dereli, R.K.; Ozturk, I.; Roest, K.; Van Lier, J.B. A review on dynamic membrane filtration: Materials, application and future perspectives. Bioresour. Technol. 2012, 122, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.S. Reverse Osmosis Membrane Research: Based on the Symposium on “Polymers for Desalination” Held at the 162nd National Meeting of the American Chemical Society in Washington, D.C., September 1971; Lonsdale, H.K., Podall, H.E., Eds.; Springer: Boston, MA, USA, 1972; pp. 379–403. [Google Scholar]
- Li, L.; Xu, G.; Yu, H. Dynamic Membrane Filtration: Formation, Filtration, Cleaning, and Applications. Chem. Eng. Technol. 2018, 41, 7–18. [Google Scholar] [CrossRef]
- Tanny, G.B. Dynamic membranes in ultrafiltration and reverse osmosis. Separ. Purif. Methods 1978, 7, 183–220. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, X.C.; Ngo, H.H.; Sun, Q.; Yang, Y. Anaerobic dynamic membrane bioreactor (AnDMBR) for wastewater treatment: A review. Bioresour. Technol. 2018, 247, 1107–1118. [Google Scholar] [CrossRef]
- Marcinkowsky, A.E.; Kraus, K.A.; Phillips, H.O.; Johnson, J.S.; Shor, A.J. Hyperfiltration studies. IV. Salt rejection by dynamically formed hydrous oxide Membranes. J. Am. Chem. Soc. 1966, 88, 5744–5746. [Google Scholar] [CrossRef]
- Usman, M.; Belkasmi, A.I.; Kastoyiannis, I.A.; Ernst, M. Pre-deposited dynamic membrane adsorber formed of microscale conventional iron oxidebased adsorbents to remove arsenic from water: Application study and mathematical modeling. J. Chem. Technol. Biotechnol. 2021, 96, 1504–1514. [Google Scholar] [CrossRef]
- Fang, X.; Li, J.; Li, X.; Pan, S.; Zhang, X.; Sun, X.; Shen, J.; Han, W.; Wang, L. Internal pore decoration with polydopamine nanoparticle on polymeric ultrafiltration membrane for enhanced heavy metal removal. Chem. Eng. J. 2017, 314, 38–49. [Google Scholar] [CrossRef]
- Pan, S.; Jiansheng Li, J.; Noonan, O.; Fang, X.; Wan, G.; Yu, C.; Wang, L. Dual-Functional Ultrafiltration Membrane for Simultaneous Removal of Multiple Pollutants with High Performance. Environ. Sci. Technol. 2017, 51, 5098–5107. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, Y.; Xiang, Q.; Liu, F.; Ling, C.; Wang, F.; Li, Y.; Li., A. A high-flux and anti-interference dual-functional membrane for effective removal of Pb(II) from natural water. J. Hazard. Mater. 2020, 384, 121492. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, Z.; Yuea, C.; Song, L.; Lva, Y.; Liu, F.; Li, A. Insight into the efficient co-removal of Cr(VI) and Cr(III) by positively charged UiO-66-NH2 decorated ultrafiltration membrane. Chem. Eng. J. 2021, 404, 126546. [Google Scholar] [CrossRef]
- Liu, H.; Zuo, K.; Vecitis, C.D. Titanium Dioxide-Coated Carbon Nanotube Network Filter for Rapid and Effective Arsenic Sorption. Environ. Sci. Technol. 2014, 48, 13871–13879. [Google Scholar] [CrossRef]
- Salehi, E.; Daraei, P.; Shamsabadi, A.A. A review on chitosan-based adsorptive membranes. Carbohydr. Polym. 2016, 152, 419–432. [Google Scholar] [CrossRef]
- Miretzky, P.; Cirelli, A.F. Cr(VI) and Cr(III) removal from aqueous solution by raw and modified lignocellulosic materials: A review. J. Hazard. Mater. 2010, 180, 1–19. [Google Scholar] [CrossRef]
- O’Connell, D.W.; Birkinshaw, C.; O’Dwyer, T.F. Heavy metal adsorbents prepared from the modification of cellulose: A review. Bioresour. Technol. 2008, 99, 6709–6724. [Google Scholar] [CrossRef]
- Wang, R.; Guan, S.; Sato, A.; Wang, X.; Wang, Z.; Yang, R.; Hsiao, B.S.; Chu, B. Nanofibrous microfiltration membranes capable of removing bacteria, viruses and heavy metal ions. J. Membr. Sci. 2013, 446, 376–382. [Google Scholar] [CrossRef]
- Karim, Z.; Mathew, A.P.; Kokol, V.; Wei, J.; Grahn, M. High-flux affinity membranes based on cellulose nanocomposites for removal of heavy metal ions from industrial effluents. RSC Adv. 2016, 6, 20644–20653. [Google Scholar] [CrossRef]
- Karim, Z.; Claudpierre, S.; Grahn, M.; Oksman, K.; Mathew, A.P. Nanocellulose based functional membranes for water cleaning: Tailoring of mechanical properties, porosity and metal ion capture. J. Membr. Sci. 2016, 514, 418–428. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, J.; Li, L.; Pan, K.; Cao, B. Poly(N,N-dimethylaminoethyl methacrylate) modification of a regenerated cellulose membrane using ATRP method for copper (II) ion removal. RSC Adv. 2013, 3, 20625–20632. [Google Scholar] [CrossRef]
- Isogai, A.; Fukuzumi, H. TEMPO-oxidized cellulose nanofibers. Nanoscale 2011, 3, 71–85. [Google Scholar] [CrossRef]
- Karim, Z.; Mathew, A.P.; Grahn, M.; Mouzon, J.; Oksman, K. Nanoporous membranes with cellulose nanocrystals as functional entity in chitosan: Removal of dyes from water. Carbohydr. Polym. 2014, 112, 668–676. [Google Scholar] [CrossRef]
- Mathew, A.P.; Oksman, K.; Karim, Z.; Liu, P.; Khan, S.A.; Naseri, N. Process scale-up and characterization of wood cellulose nanocrystals hydrolysed using bioethanol pilot plant. Ind. Crops Prod. 2014, 58, 212–219. [Google Scholar] [CrossRef] [Green Version]
- Božič, M.; Liu, P.; Mathew, A.P.; Kokol, V. Enzymatic phosphorylation of cellulose nanofibers to new highly-ions adsorbing, flame-retardant and hydroxyapatite-growth induced natural nanoparticles. Cellulose 2014, 21, 2713–2726. [Google Scholar] [CrossRef]
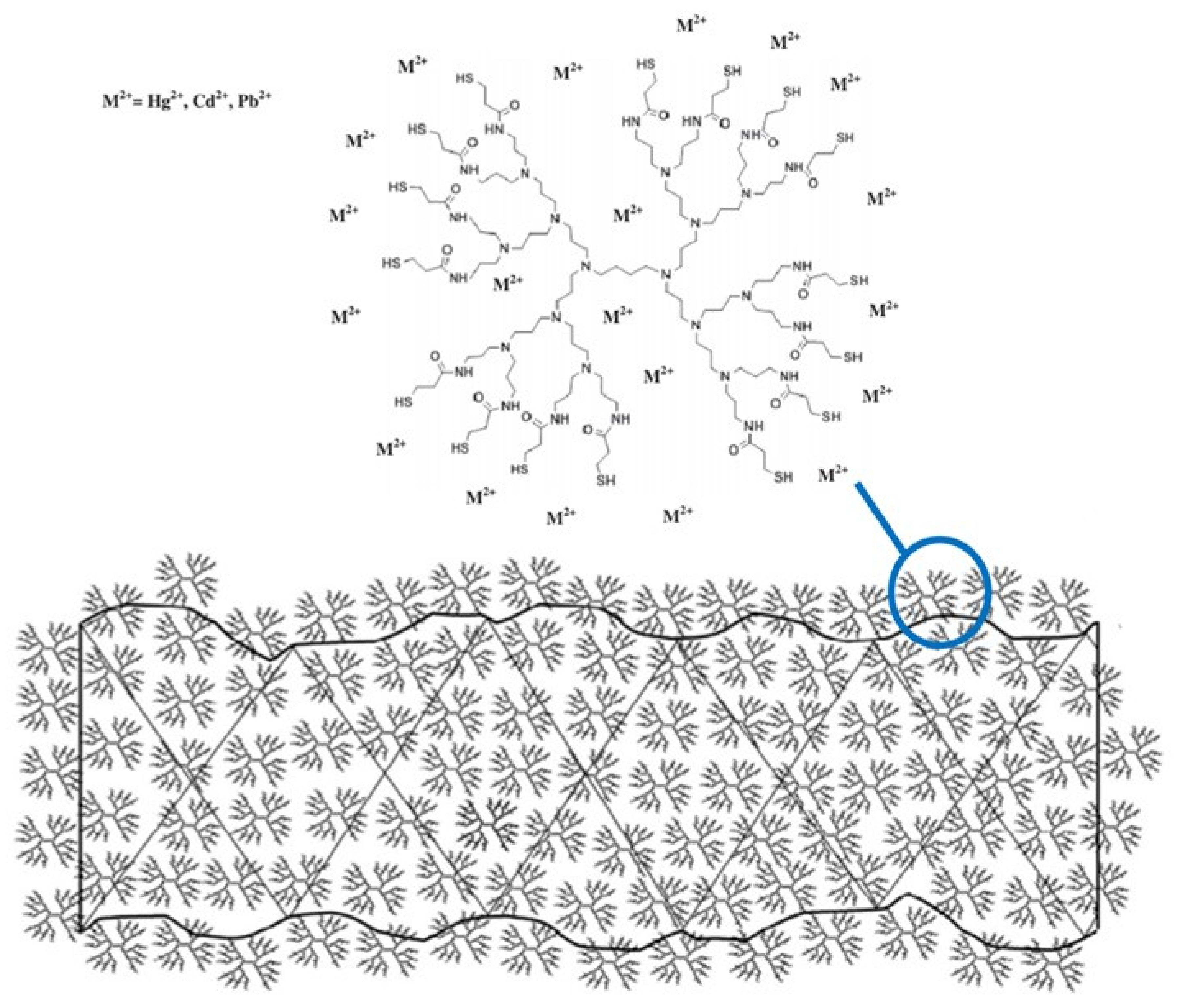
- Algarra, M.; Vázquez, M.I.; Alonso, B.; Casado, C.M.; Casado, J.; Benavente, J. Characterization of an engineered cellulose based membrane by thiol dendrimer for heavy metals removal. Chem. Eng. J. 2014, 253, 472–477. [Google Scholar] [CrossRef]
- Giwa, A.; Hasan, S.; Yousuf, A.; Chakraborty, S.; Johnson, D.; Hilal, N. Biomimetic membranes: A critical review of recent progress. Desalination 2017, 420, 403–424. [Google Scholar] [CrossRef]
- Gouaux, E.; MacKinnon, R. Principles of selective ion transport in channels and pumps. Science 2005, 310, 1461–1465. [Google Scholar] [CrossRef]
- Kopfer, D.A.; Song, C.; Gruene, T.; Sheldrick, G.M.; Zachariae, U.; De Groot, B.L. Ion permeation in K+ channels occurs by direct Coulomb knock-on. Science 2014, 346, 352–355. [Google Scholar] [CrossRef] [PubMed]
- Mitaa, K.; Sumikama, T.; Iwamoto, M.; Matsuki, Y.; Shigemi, K.; Oiki, S. Conductance selectivity of Na+ across the K+ channel via Na+ trapped in a tortuous trajectory. Proc. Natl. Acad. Sci. USA 2021, 118, e2017168118. [Google Scholar] [CrossRef]
- Wen, Q.; Yan, D.; Liu, F.; Wang, M.; Ling, Y.; Wang, P.; Kluth, P.; Schauries, D.; Trautmann, C.; Apel, P.; et al. Highly Selective Ionic Transport through Subnanometer Pores in Polymer Films. Adv. Funct. Mater. 2016, 26, 5796–5803. [Google Scholar] [CrossRef]
- Epsztein, R.; DuChanois, R.M.; Ritt, C.L.; Noy, A.; Elimelech, M. Towards single-species selectivity of membranes with subnanometre pores. Nat. Nanotechnol. 2020, 15, 426–436. [Google Scholar] [CrossRef]
- Zhang, H.; Hou, J.; Hu, Y.; Wang, P.; Ou, R.; Jiang, L.; Liu, J.Z.; Freeman, B.D.; Hill, A.J.; Wang, H. Ultrafast selective transport of alkali metal ions in metal organic frameworks with subnanometer pores. Sci. Adv. 2018, 4, eaaq0066. [Google Scholar] [CrossRef]
- Pedersen, C.J. Cyclic polyethers and their complexes with metals salts. J. Am. Chem. Soc. 1967, 89, 7017–7036. [Google Scholar] [CrossRef]
- DuChanois, R.M.; Heiranian, M.; Yang, J.; Porter, C.J.; Li, Q.; Zhang, X.; Verduzco, R.; Elimelech., M. Designing polymeric membranes with coordination chemistry for high-precision ion separations. Sci. Adv. 2022, 8, eabm9436. [Google Scholar] [CrossRef]
- Nunes, S.P. Block Copolymer Membranes for Aqueous Solution Applications. Macromolecules 2016, 49, 2905–2916. [Google Scholar] [CrossRef]
- Jha, A.K.; Tsang, S.L.; Ozcam, A.E.; Offeman, R.D.; Balsara, N.P. Master curve captures the effect of domain morphology on ethanol pervaporation through block copolymer membranes. J. Membr. Sci. 2012, 401, 125–131. [Google Scholar] [CrossRef]
- Buonomenna, M.G.; Tone, C.M.; De Santo, M.P.; Ciuchi, F.; Perrotta, E.; Zappone, B.; Galiano, F.; Figoli, A. Ordering phenomena in nanostructured poly(styrene-b-butadiene-b-styrene) (SBS) membranes for selective ethanol transport. J. Membr. Sci. 2011, 385–386, 162–170. [Google Scholar] [CrossRef]
- Jung, A.; Filiz, V.; Rangou, S.; Buhr, K.; Merten, P.; Hahn, J.; Clodt, J.; Abetz, C.; Abetz, V. Formation of integral asymmetric membranes of AB diblock and ABC triblock copolymers by phase inversion. Macromol. Rapid Commun. 2013, 34, 610–615. [Google Scholar] [CrossRef]
- Pendergast, M.M.; Dorin, R.M.; Phillip, W.A.; Wiesner, U.; Hoek, E.M. Understanding the structure and performance of self-assembled triblock terpolymer membranes. J. Membr. Sci. 2013, 444, 461–468. [Google Scholar] [CrossRef]
- Peinemann, K.-V.; Abetz, V.; Simon, P.F. Asymmetric superstructure formed in a block copolymer via phase separation. Nat. Mater. 2007, 6, 992–996. [Google Scholar] [CrossRef]
- Rzayev, J.; Hillmyer, M.A. Nanochannel array plastics with tailored surface chemistry. J. Am Chem. Soc. 2005, 127, 13373–13379. [Google Scholar] [CrossRef]
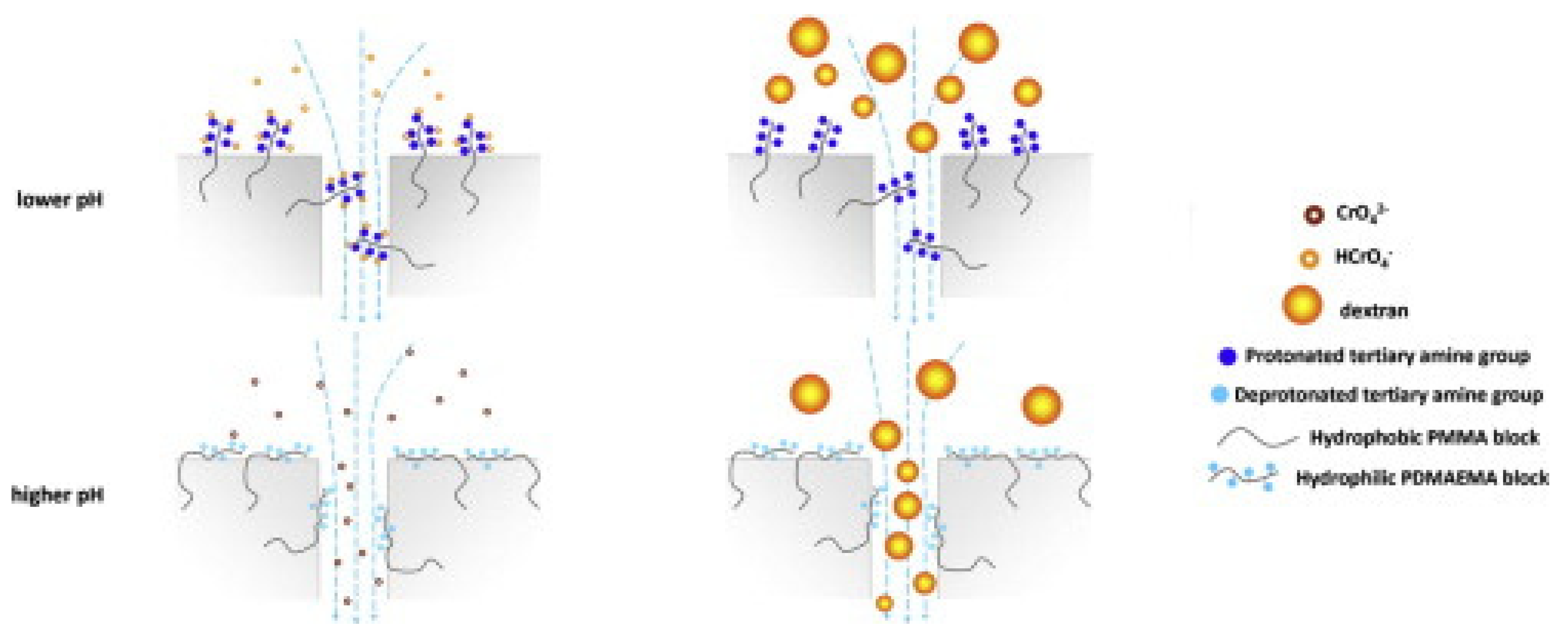
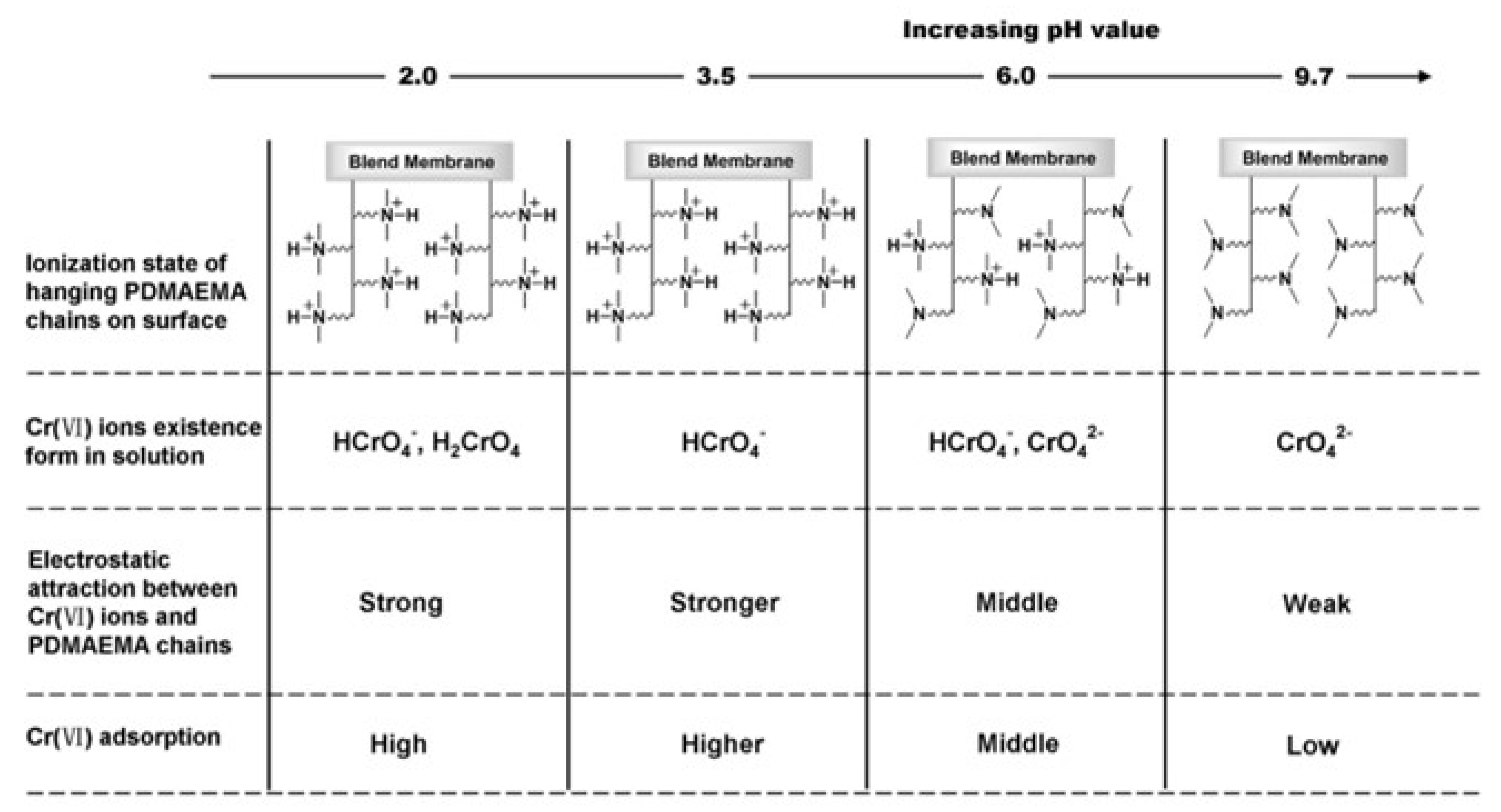
- Yao, Z.; Li, Y.; Cui, Y.; Zheng, K.; Zhu, B.; Xu, H.; Zhu, L. Tertiary amine block copolymer containing ultrafiltration membrane with pH-dependent macromolecule sieving and Cr (VI) removal properties. Desalination 2015, 355, 91–98. [Google Scholar] [CrossRef]
- Kausar, A. Investigation on self-assembled blend membranes of polyethylene-block-poly (ethylene glycol)-block-polcaprolactone and poly (styrene-block-methyl methacrylate) with polymer/gold nanocomposite particles. Polym.-Plast. Technol. Eng. 2015, 54, 1794–1802. [Google Scholar] [CrossRef]
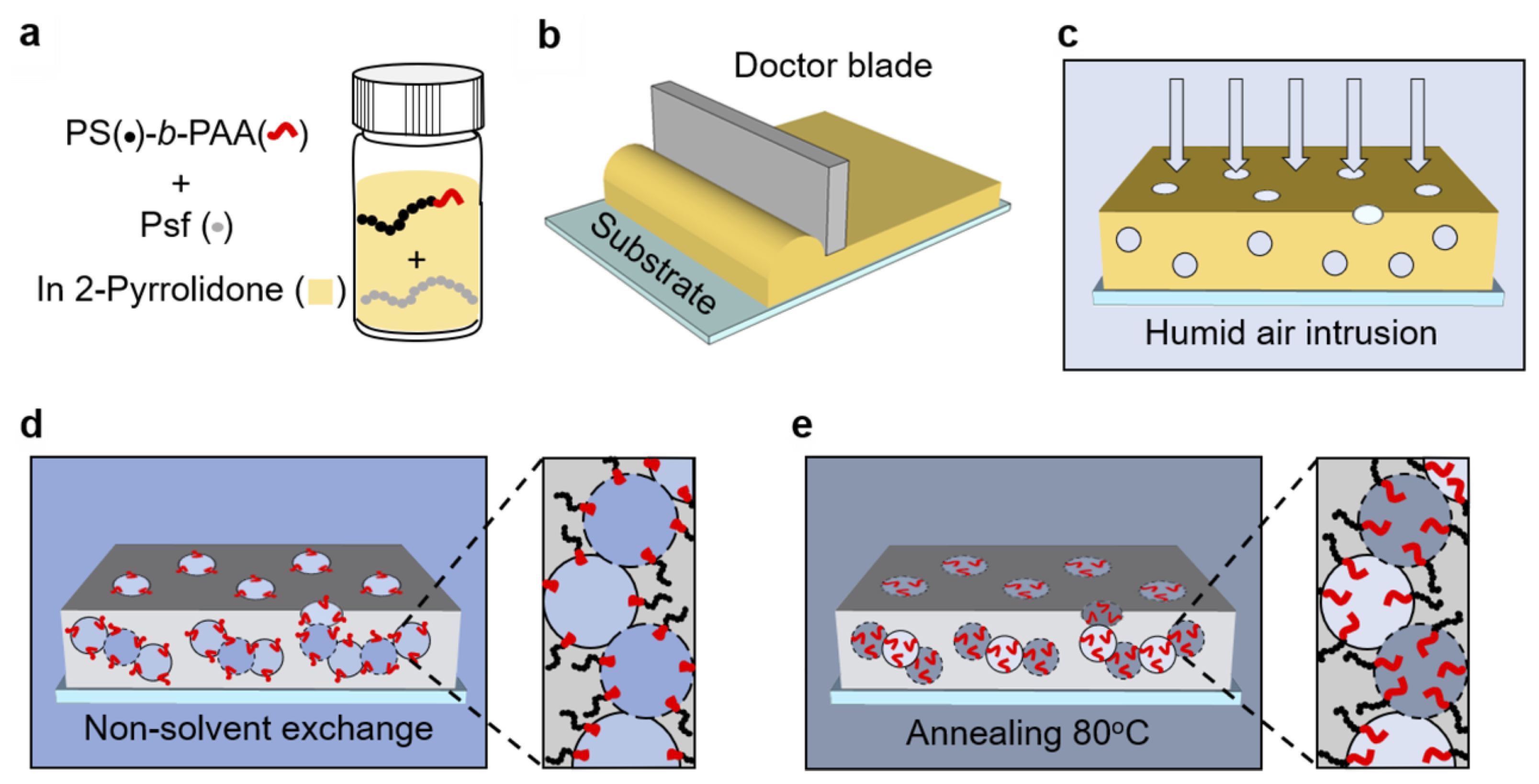
- Zhang, Y.; Vallin, J.R.; Sahoo, J.K.; Gao, F.; Boudouris, B.W.; Webber, M.J.; Phillip, W.A. High-affinity detection and capture of heavy metal contaminants using block polymer composite membranes. ACS Cent. Sci. 2018, 4, 1697–1707. [Google Scholar] [CrossRef]
- Liu, H.; Peng, S.; Shu, L.; Chen, T.; Bao, T.; Frost, R.L. Magnetic zeolite NaA: Synthesis, characterization based on metakaolin and its application for the removal of Cu2+, Pb2+. Chemosphere 2013, 91, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wang, J.; Wu, D.; Li, X.; Luo, Y.; Han, C.; Ma, W.; He, S. Investigating the heavy metal adsorption of mesoporous silica materials prepared by microwave synthesis. Nanoscale Res. Lett. 2017, 12, 323. [Google Scholar] [CrossRef]
- Faghihian, H.; Naghavi, M. Synthesis of Amine-Functionalized MCM-41 and MCM-48 for removal of heavy metal ions from aqueous solutions. Sep. Sci. Technol. 2014, 49, 214–220. [Google Scholar] [CrossRef]
- Taba, P.; Budi, P.; Puspitasari, A. Adsorption of Heavy Metals on Amine-Functionalized MCM-48, IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; p. 012015. [Google Scholar]
- Joseph, I.V.; Tosheva, L.; Miller, G.; Doyle, A.M. FAU-Type Zeolite Synthesis from Clays and Its Use for the Simultaneous Adsorption of Five Divalent Metals from Aqueous Solutions. Materials 2021, 14, 3738. [Google Scholar] [CrossRef]
- Zhao, G.; Ren, X.; Gao, X.; Tan, X.; Li, J.; Chen, C.; Huang, Y.; Wang, X. Removal of Pb(II) ions from aqueous solutions on few-layered graphene oxide nanosheets. Dalton Trans. 2011, 40, 10945–10952. [Google Scholar] [CrossRef]
- Sitko, R.; Turek, E.; Zawisza, B.; Malicka, E.; Talik, E.; Heimann, J.; Gagor, A.; Feist, B.; Wrzalik, R. Adsorption of divalent metal ions from aqueous solutions using graphene oxide. Dalton Trans. 2013, 42, 5682–5689. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Li, J.; Ren, X.; Chen, C.; Wang, X. Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ. Sci. Technol. 2011, 45, 10454–10462. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Yang, Y.; Zhou, H.; Ye, T.; Huang, Z.; Liu, R.; Kuang, Y. Highly efficient removal of Cu (II) from aqueous solution by using graphene oxide. Water Air Soil Pollut. 2013, 224, 1372. [Google Scholar] [CrossRef]
- Hu, M.; Mi, B. Enabling graphene oxide nanosheets as water separation membranes. Environ. Sci. Technol. 2013, 47, 3715–3723. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Graham, N. Development of a stable cation modified graphene oxide membrane for water treatment. 2D Mater. 2017, 4, 045006. [Google Scholar] [CrossRef]
- Yeh, C.-N.; Raidongia, K.; Shao, J.; Yang, Q.-H.; Huang, J. On the origin of the stability of graphene oxide membranes in water. Nat. Chem. 2015, 7, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, T.; Zhang, L.; Yu, Q.J.; Hou, L.A. Dopamine crosslinked graphene oxide membrane for simultaneous removal of organic pollutants and trace heavy metals from aqueous solution. Environ. Technol. 2018, 39, 3055–3065. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.; Chung, T.-S. Nanometric graphene oxide framework membranes with enhanced heavy metal removal via nanofiltration. Environ. Sci. Technol. 2015, 49, 10235–10242. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Wang, Y. Covalently crosslinked graphene oxide membranes by esterification reactions for ions separation. J. Mater. Chem. A 2015, 3, 4405–4412. [Google Scholar] [CrossRef]
- Li, S.; Yang, P.; Liu, X.; Zhang, J.; Xie, W.; Wang, C.; Liu, C.; Guo, Z. Graphene oxidebased dopamine mussel-like cross-linked polyethylene imine nanocomposite coating with enhanced hexavalent uranium adsorption. J. Mater. Chem. A 2019, 7, 16902–16911. [Google Scholar] [CrossRef]
- Tan, P.; Sun, J.; Hu, Y.; Fang, Z.; Bi, Q.; Chen, Y.; Cheng, J. Adsorption of Cu2+, Cd2+ and Ni2+ from aqueous single metal solutions on graphene oxide membranes. J. Hazard. Mater. 2015, 297, 251–260. [Google Scholar] [CrossRef]
- Sitko, R.; Musielak, M.; Zawisza, B.; Talik, E.; Gagor, A. Graphene oxide/cellulose membranes in adsorption of divalent metal ions. RSC Adv. 2016, 6, 96595–96605. [Google Scholar] [CrossRef]
- Musielak, M.; Gagor, A.; Zawisza, B.; Talik, E.; Sitko, R. Graphene oxide/carbon nanotube membranes for highly efficient removal of metal ions from water. ACS Appl. Mater. Interfaces 2019, 11, 28582–28590. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, Y.; Liu, Y.-L.; Li, M.; Zhao, H.-Y.; Hou, L.-A. High flux MWCNTs-interlinked GO hybrid membranes survived in cross-flow filtration for the treatment of strontium-containing wastewater. J. Hazard. Mater. 2016, 320, 187–193. [Google Scholar] [CrossRef]
- Viraka Nellore, B.P.; Kanchanapally, R.; Pedraza, F.; Sinha, S.S.; Pramanik, A.; Hamme, A.T.; Arslan, Z.; Sardar, D.; Ray, P.C. Bio-conjugated CNT-bridged 3D porous graphene oxide membrane for highly efficient disinfection of pathogenic bacteria and removal of toxic metals from water. ACS Appl. Mater. Interfaces 2015, 7, 19210–19218. [Google Scholar] [CrossRef]
- Kang, H.; Shi, J.; Liu, L.; Shan, M.; Xu, Z.; Li, N.; Li, J.; Lv, H.; Qian, X.; Zhao, L. Sandwich morphology and superior dye-removal performances for nanofiltration membranes self-assemblied via graphene oxide and carbon nanotubes. Appl. Surf. Sci. 2018, 428, 990–999. [Google Scholar] [CrossRef]
- Krogman, K.; Lowery, J.L.; Zacharia, N.S.; Rutledge, G.C.; Hammond, P.T. Spraying asymmetry into functional membranes layer-by-layer. Nat. Mater. 2009, 8, 512–518. [Google Scholar] [CrossRef]
- Teeters, M.; Conrardy, S.; Thomas, B.; Root, T.; Lightfoot, E. Adsorptive membrane chromatography for purification of plasmid DNA. J. Chromatogr. A 2003, 989, 165–173. [Google Scholar] [CrossRef]
- Yang, H.; Viera, C.; Fischer, J.; Etzel, M.R. Purification of a large protein using ion-exchange membranes. Ind. Eng. Chem. Res. 2002, 41, 1597–1602. [Google Scholar] [CrossRef]
- Menkhaus, T.J.; Varadaraju, H.; Zhang, L.; Schneiderman, S.; Bjustrom, S.; Liu, L.; Fong, H. Electrospun nanofiber membranes surface functionalized with 3-dimensional nanolayers as an innovative adsorption medium with ultra-high capacity and throughput. Chem. Commun. 2010, 46, 3720–3722. [Google Scholar] [CrossRef]
- Salehi, E.; Madaeni, S.; Rajabi, L.; Vatanpour, V.; Derakhshan, A.; Zinadini, S.; Ghorabi, S.; Monfared, H.A. Novel chitosan/poly (vinyl) alcohol thin adsorptive membranes modified with amino functionalized multi-walled carbon nanotubes for Cu(II) removal from water: Preparation, characterization, adsorption kinetics and thermodynamics. Sep. Purif. Technol. 2012, 89, 309–319. [Google Scholar] [CrossRef]
- Farjadian, F.; Schwark, S.; Ulbricht, M. Novel functionalization of porous polypropylene microfiltration membranes: Via grafted poly (aminoethyl methacrylate) anchored Schiff bases toward membrane adsorbers for metal ions. Polym. Chem. 2015, 6, 1584–1593. [Google Scholar] [CrossRef]
- Scharnagl, N.; Buschatz, H. Polyacrylonitrile (PAN) membranes for ultra-and microfiltration. Desalination 2011, 139, 191–198. [Google Scholar] [CrossRef]
- Chen, Y.; He, M.; Wang, C.; Wei, Y. A novel polyvinyltetrazole-grafted resin with high capacity for adsorption of Pb(II), Cu(II) and Cr(III) ions from aqueous solutions. J. Mater. Chem. A 2014, 2, 10444–10453. [Google Scholar] [CrossRef]
- Huang, M.-R.; Li, X.-G.; Li, S.-X.; Zhang, W. Resultful synthesis of polyvinyltetrazole from polyacrylonitrile. React. Funct. Polym. 2004, 59, 53–61. [Google Scholar] [CrossRef]
- Gölander, C.-G.; Kiss, E. Protein adsorption on functionalized and ESCA-characterized polymer films studied by ellipsometry. J. Colloid Interface Sci. 1988, 21, 240–253. [Google Scholar] [CrossRef]
- Lundström, I. Surface physics and biological phenomena. Phys. Scr. 1983, 1983, 5. [Google Scholar] [CrossRef]
- Ulbricht, M. Advanced functional polymer membranes. Polymer 2006, 47, 2217–2262. [Google Scholar] [CrossRef]
- Minko, S. Grafting on Solid Surfaces: “Grafting to” and “Grafting from” Methods. In Polymer Surfaces and Interfaces; Springer: Heidelberg, The Netherlands, 2008; pp. 215–234. [Google Scholar]
- Schroen, C.; Stuart, M.C.; Van der Voort Maarschalk, K.; Van der Padt, A.; Van’t Riet, K. Influence of preadsorbed block copolymers on protein adsorption: Surface properties, layer thickness, and surface coverage. Langmuir 1995, 11, 3068–3074. [Google Scholar] [CrossRef]
- Gopal, R.; Zuwei, M.; Kaur, S.; Ramakrishna, S. Surface Modification and Application of Functionalized Polymer Nanofibers. In Molecular Building Blocks for Nanotechnology; Springer: Heidelberg, The Netherlands, 2007; pp. 72–91. [Google Scholar]
- Kato, K.; Uchida, E.; Kang, E.-T.; Uyama, Y.; Ikada, Y. Polymer surface with graft chains. Progr. Polym. Sci. 2003, 28, 209–259. [Google Scholar] [CrossRef]
- Kumar, M.; Shevate, R.; Hilke, R.; Peinemann, K.-V. Novel adsorptive ultrafiltration membranes derived from polyvinyltetrazole-co-polyacrylonitrile for Cu(II) ions removal. Chem. Eng. J. 2016, 301, 306–314. [Google Scholar] [CrossRef]
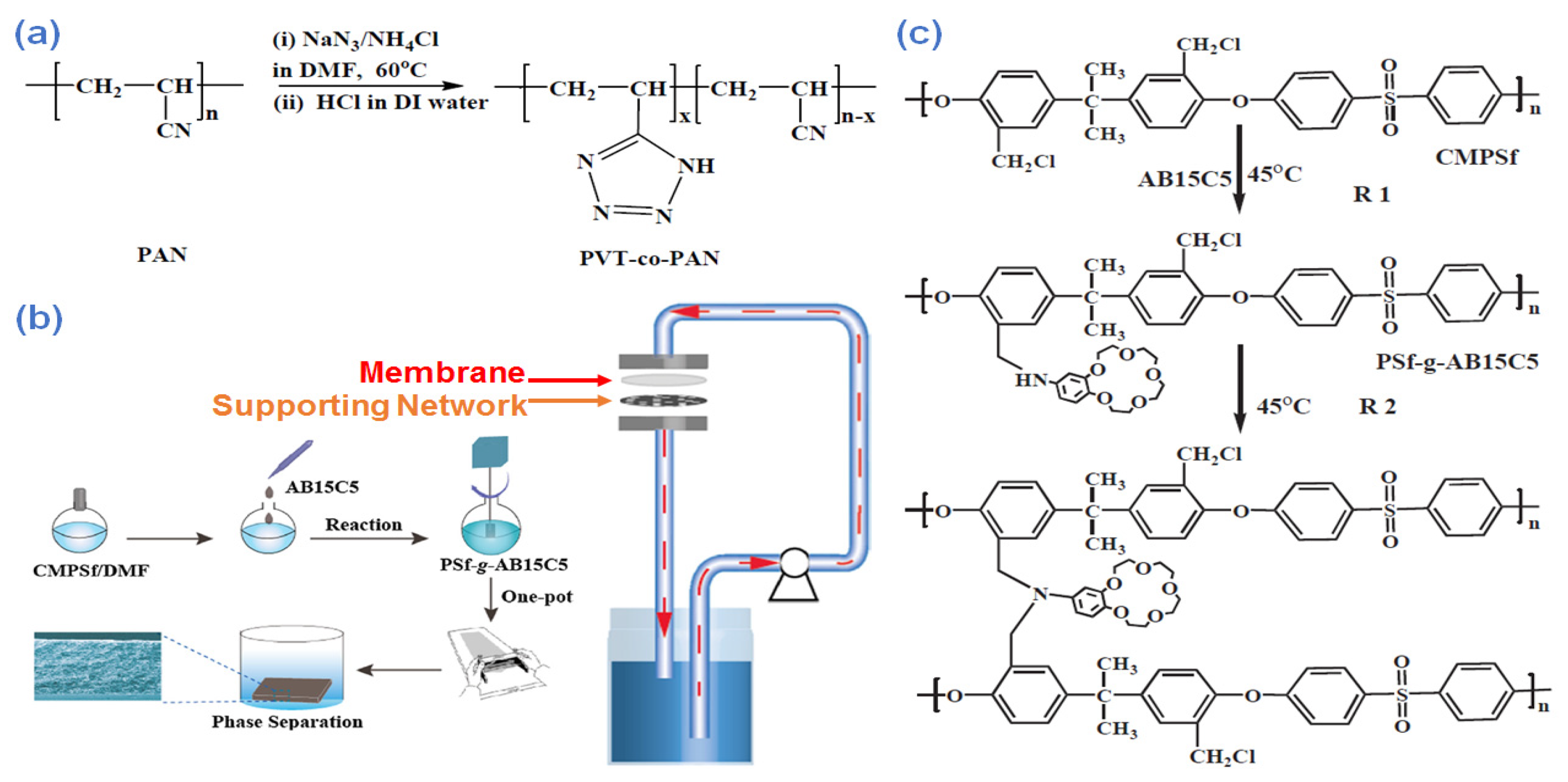
- Pei, H.; Yan, F.; Ma, X.; Li, X.; Liu, C.; Li, J.; Cui, Z.; He, B. In situ one-pot formation of crown ether functionalized polysulfone membranes for highly efficient lithium isotope adsorptive separation. Eur. Polym. J. 2018, 109, 288–296. [Google Scholar] [CrossRef]
- Wu, S.; Li, F.; Wang, H.; Fu, L.; Zhang, B.; Li, G. Effects of poly (vinyl alcohol) (PVA) content on preparation of novel thiol-functionalized mesoporous PVA/SiO2 composite nanofiber membranes and their application for adsorption of heavy metal ions from aqueous solution. Polymer 2010, 51, 6203–6211. [Google Scholar] [CrossRef]
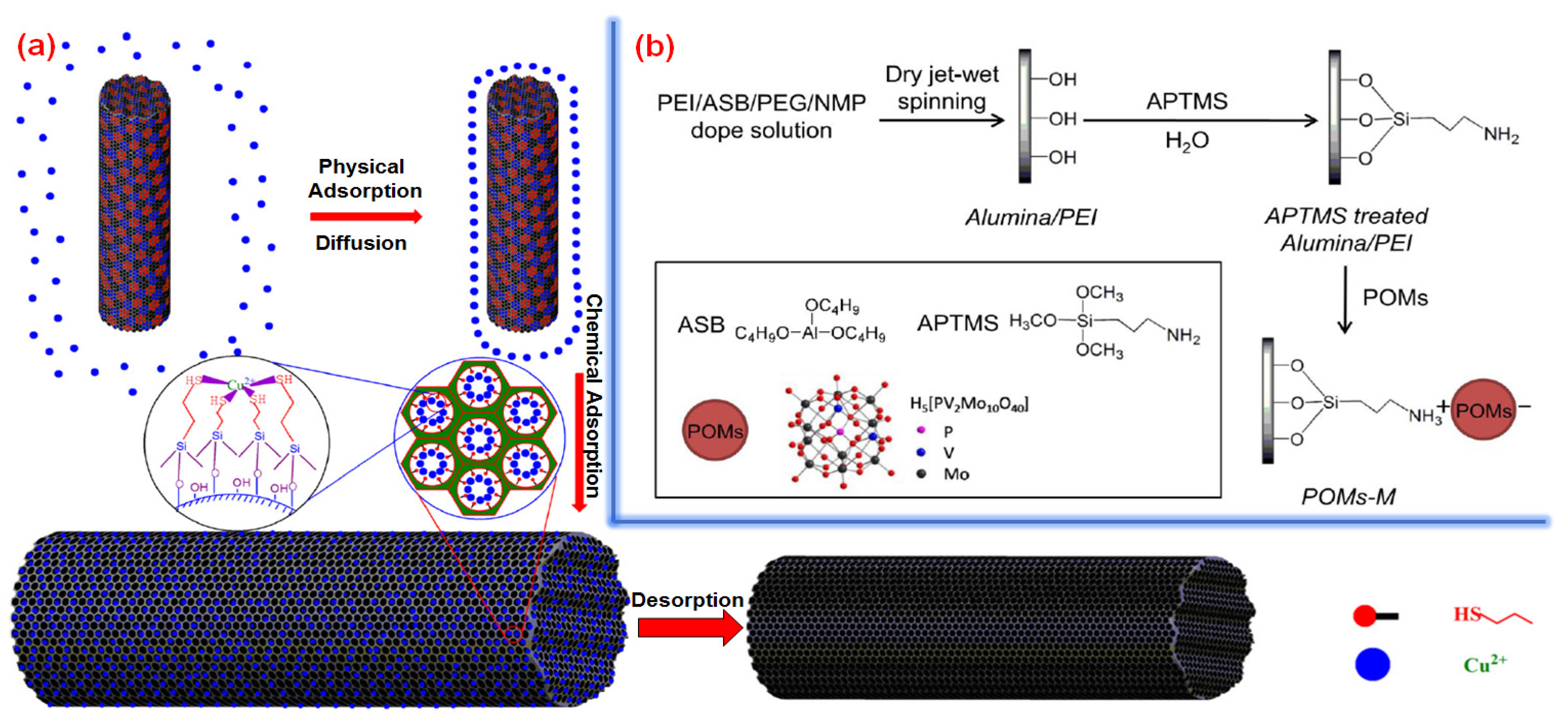
- Yao, L.; Zhang, L.; Wang, R.; Chou, S.; Dong, Z. A new integrated approach for dye removal from wastewater by polyoxometalates functionalized membranes. J. Hazard. Mater. 2016, 301, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Menkhaus, T.J.; Fong, H. Fabrication and bioseparation studies of adsorptive membranes/felts made from electrospun cellulose acetate nanofibers. J. Membr. Sci. 2008, 319, 176–184. [Google Scholar] [CrossRef]
- Yang, X.; Zhou, Y.; Sun, Z.; Yang, C.; Tang, D. Synthesis and Cr adsorption of a super-hydrophilic polydopamine-functionalized electrospun polyacrylonitrile. Environ. Chem. Lett. 2021, 19, 743–749. [Google Scholar] [CrossRef]
- Zhang, H.; Ruan, X.; Hu, Y.; Cheng, J.; Tan, P. Adsorptive characteristics of Cu2+, Ni2+ and Cd2+ on chitosan/poly (vinyl alcohol) nanofiber. Acta Sci. Circumstantiae 2015, 35, 184–193. [Google Scholar]
- Phan, D.-N.; Lee, H.; Huang, B.; Mukai, Y.; Kim, I.-S. Fabrication of electrospun chitosan/cellulose nanofibers having adsorption property with enhanced mechanical property. Cellulose 2019, 26, 1781–1793. [Google Scholar] [CrossRef]
- Aliabadi, M.; Irani, M.; Ismaeili, J.; Piri, H.; Parnian, M.J. Electrospun nanofiber membrane of PEO/Chitosan for the adsorption of nickel, cadmium, lead and copper ions from aqueous solution. Chem. Eng. J. 2013, 20, 237–243. [Google Scholar] [CrossRef]
- Zia, Q.; Tabassum, M.; Lu, Z.; Khawar, M.T.; Song, J.; Gong, H.; Meng, J.; Li, Z.; Li, J. Porous poly (L–lactic acid)/chitosan nanofibres for copper ion adsorption. Carbohydr. Polym. 2020, 227, 115343. [Google Scholar] [CrossRef]
- Shariful, M.I.; Sharif, S.B.; Lee, J.J.L.; Habiba, U.; Ang, B.C.; Amalina, M.A. Adsorption of divalent heavy metal ion by mesoporous-high surface area chitosan/poly (ethylene oxide) nanofibrous membrane. Carbohydr. Polym. 2017, 157, 57–64. [Google Scholar] [CrossRef]
- Wu, R.-X.; Zheng, G.-F.; Li, W.-W.; Zhong, L.-B.; Zheng, Y.-M. Electrospun chitosan nanofiber membrane for adsorption of Cu (II) from aqueous solution: Fabrication, characterization and performance. J. Nanosci. Nanotechnol. 2018, 18, 5624–5635. [Google Scholar] [CrossRef]
- Lakhdhar, I.; Belosinschi, D.; Mangin, P.; Chabot, B. Development of a bio-based sorbent media for the removal of nickel ions from aqueous solutions. J. Environ. Chem. Eng. 2016, 4, 3159–3169. [Google Scholar] [CrossRef]
- Wang, P.; Wang, L.; Dong, S.; Zhang, G.; Shi, X.; Xiang, C.; Li, L. Adsorption of hexavalent chromium by novel chitosan/poly (ethylene oxide)/permutit electrospun nanofibers. New J. Chem. 2018, 42, 17740–17749. [Google Scholar] [CrossRef]
- Wang, M.; Li, X.; Zhang, T.; Deng, L.; Li, P.; Wang, X.; Hsiao, B.S. Eco-friendly poly (acrylic acid)-sodium alginate nanofibrous hydrogel: A multifunctional platform for superior removal of Cu (II) and sustainable catalytic applications. Colloids Surf. A 2018, 558, 228–241. [Google Scholar] [CrossRef]
- Jiang, M.; Han, T.; Wang, J.; Shao, L.; Qi, C.; Zhang, X.M.; Liu, C.; Liu, X. Removal of heavy metal chromium using cross-linked chitosan composite nanofiber mats. Int. J. Biol. Macromol. 2018, 120, 213–221. [Google Scholar] [CrossRef]
- Zhang, S.; Shi, Q.; Christodoulatos, C.; Meng, X. Lead and cadmium adsorption by electrospun PVA/PAA nanofibers: Batch, spectroscopic, and modeling study. Chemosphere 2019, 233, 405–413. [Google Scholar] [CrossRef]
- Aquino, R.; Tolentino, M.; Amen, S.; Arceo, M.; Dolojan, M.; Basilia, B. Preparation of cellulose acetate blended with chitosan nanostructured membrane via electrospinning for Cd2+ adsorption in artificial wastewater. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; p. 012137. [Google Scholar]
- Zhang, S.; Shi, Q.; Christodoulatos, C.; Korfiatis, G.; Meng, X. Adsorptive filtration of lead by electrospun PVA/PAA nanofiber membranes in a fixed-bed column. Chem. Eng. J. 2019, 370, 1262–1273. [Google Scholar] [CrossRef]
- Zhang, S.; Shi, Q.; Korfiatis, G.; Christodoulatos, C.; Wang, H.; Meng, X. Chromate removal by electrospun PVA/PEI nanofibers: Adsorption, reduction, and effects of co-existing ions. Chem. Eng. J. 2020, 387, 124179. [Google Scholar] [CrossRef]
- Wu, C.; Wang, H.; Wei, Z.; Li, C.; Luo, Z. Polydopamine-mediated surface functionalization of electrospun nanofibrous membranes: Preparation, characterization and their adsorption properties towards heavy metal ions. Appl. Surf. Sci. 2015, 346, 207–215. [Google Scholar] [CrossRef]
- Min, M.; Shen, L.; Hong, G.; Zhu, M.; Zhang, Y.; Wang, X.; Chen, Y.; Hsiao, B.S. Micro-nano structure poly (ether sulfones)/poly (ethyleneimine) nanofibrous affinity membranes for adsorption of anionic dyes and heavy metal ions in aqueous solution. Chem. Eng. J. 2012, 197, 88–100. [Google Scholar] [CrossRef]
- Wang, X.; Min, M.; Liu, Z.; Yang, Y.; Zhou, Z.; Zhu, M.; Chen, Y.; Hsiao, B.S. Poly (ethyleneimine) nanofibrous affinity membrane fabricated via one step wet-electrospinning from poly (vinyl alcohol)-doped poly (ethyleneimine) solution system and its application. J. Membr. Sci. 2011, 379, 191–199. [Google Scholar] [CrossRef]
- Alcaraz-Espinoza, J.J.; Chávez-Guajardo, A.E.; Medina-Llamas, J.C.; Andrade, C.A.; De Melo, C.P. Hierarchical composite polyaniline–(electrospun polystyrene) fibers applied to heavy metal remediation. ACS Appl. Mater. Interfaces 2015, 7, 7231–7240. [Google Scholar] [CrossRef]
- Bornillo, K.A.S.; Kim, S.; Choi, H. Cu (II) removal using electrospun dual-responsive polyethersulfone-poly (dimethyl amino) ethyl methacrylate (PES-PDMAEMA) blend nanofibers. Chemosphere 2020, 242, 125287. [Google Scholar] [CrossRef] [PubMed]
- Zang, L.; Lin, R.; Dou, T.; Ma, J.; Sun, L. Electrospun superhydrophilic membranes for effective removal of Pb (II) from water. Nanoscale Adv. 2019, 1, 389–394. [Google Scholar] [CrossRef] [Green Version]
- Zhu, F.; Zheng, Y.-M.; Zhang, B.-G.; Dai, Y.-R. A critical review on the electrospun nanofibrous membranes for the adsorption of heavy metals in water treatment. J. Hazard. Mater. 2021, 401, 123608. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Song, X.; Zhang, Q.; Zhai, T. Electrospun polyindole nanofibers as a nano-adsorbent for heavy metal ions adsorption for wastewater treatment. Fibers Polym. 2017, 18, 502–513. [Google Scholar] [CrossRef]
- Wang, J.; Pan, K.; He, Q.; Cao, B. Polyacrylonitrile/polypyrrole core/shell nanofiber mat for the removal of hexavalent chromium from aqueous solution. J. Hazard. Mater. 2013, 244, 121–129. [Google Scholar] [CrossRef]
- Mokhena, T.C.; Jacobs, N.V.; Luyt, A. Electrospun alginate nanofibres as potential bio-sorption agent of heavy metals in water treatment. Express Polym. Lett. 2017, 11, 652–663. [Google Scholar] [CrossRef]
- Ma, F.-F.; Zhang, D.; Zhang, N.; Huang, T.; Wang, Y. Polydopamine-assisted deposition of polypyrrole on electrospun poly (vinylidene fluoride) nanofibers for bidirectional removal of cation and anion dyes. Chem. Eng. J. 2018, 354, 432–444. [Google Scholar] [CrossRef]
- Cai, Z.-J.; Yang, H.-Z.; Xu, Y.; Wang, C.-K. Preparation of Polyindole Nanofibers and Their Cadium Ion Adsorption Performance. Acta Polym. Sin. 2015, 5, 581–588. [Google Scholar]
- Ma, F.-F.; Zhang, N.; Wei, X.; Yang, J.-H.; Wang, Y.; Zhou, Z.-W. Blend-electrospun poly (vinylidene fluoride)/polydopamine membranes: Self-polymerization of dopamine and the excellent adsorption/separation abilities. J. Mater. Chem. A 2017, 5, 14430–14443. [Google Scholar] [CrossRef]
- Huang, M.; Tu, H.; Chen, J.; Liu, R.; Liang, Z.; Jiang, L.; Shi, X.; Du, Y.; Deng, H. Chitosan-rectorite nanospheres embedded aminated polyacrylonitrile nanofibers via shoulder-to-shoulder electrospinning and electrospraying for enhanced heavy metal removal. Appl. Surf. Sci. 2018, 437, 294–303. [Google Scholar] [CrossRef]
- Hong, G.; Li, X.; Shen, L.; Wang, M.; Wang, C.; Yu, X.; Wang, X. High recovery of lead ions from aminated polyacrylonitrile nanofibrous affinity membranes with micro/nano structure. J. Hazard. Mater. 2015, 295, 161–169. [Google Scholar] [CrossRef]
- Li, Y.; Wen, Y.; Wang, L.; He, J.; Al-Deyab, S.S.; El-Newehy, M.; Yu, J.; Ding, B. Simultaneous visual detection and removal of lead (II) ions with pyromellitic dianhydride-grafted cellulose nanofibrous membranes. J. Mater. Chem. A 2015, 3, 18180–18189. [Google Scholar] [CrossRef]
- Yang, D.; Li, L.; Chen, B.; Shi, S.; Nie, J.; Ma, G. Functionalized chitosan electrospun nanofiber membranes for heavy-metal removal. Polymer 2019, 163, 74–85. [Google Scholar] [CrossRef]
- Xiang, T.; Zhang, Z.; Liu, H.; Yin, Z.; Li, L.; Liu, X. Characterization of cellulose-based electrospun nanofiber membrane and its adsorptive behaviours using Cu (II), Cd (II), Pb (II) as models. Sci. China Chem. 2013, 56, 567–575. [Google Scholar] [CrossRef]
- Yang, R.; Aubrecht, K.B.; Ma, H.; Wang, R.; Grubbs, R.B.; Hsiao, B.S.; Chu, B. Thiol-modified cellulose nanofibrous composite membranes for chromium (VI) and lead (II) adsorption. Polymer 2014, 55, 1167–1176. [Google Scholar] [CrossRef]
- Brandes, R.; Belosinschi, D.; Brouillette, F.; Chabot, B. A new electrospun chitosan/phosphorylated nanocellulose biosorbent for the removal of cadmium ions from aqueous solutions. J. Environ. Chem. Eng. 2019, 7, 103477. [Google Scholar] [CrossRef]
- Feng, Q.; Wu, D.; Zhao, Y.; Wei, A.; Wei, Q.; Fong, H. Electrospun AOPAN/RC blend nanofiber membrane for efficient removal of heavy metal ions from water. J. Hazard. Mater. 2018, 344, 819–828. [Google Scholar] [CrossRef]
- Fang, Y.; Liu, X.; Wu, X.; Tao, X.; Fei, W. Electrospun polyurethane/phytic acid nanofibrous membrane for high efficient removal of heavy metal ions. Environ. Technol. 2021, 42, 1053–1060. [Google Scholar] [CrossRef]
- Mohamadi, Z.; Abdolmaleki, A. Heavy metal remediation via poly (3, 4-ethylene dioxythiophene) deposition onto neat and sulfonated nonwoven poly (ether sulfone). J. Ind. Eng. Chem. 2017, 55, 164–172. [Google Scholar] [CrossRef]
- Casado-Coterillo, C. Mixed Matrix Membranes; Multidisciplinary Digital Publishing Institute: Basel, Switzerland, 2019. [Google Scholar]
- Lim, Y.J.; Goh, K.; Lai, G.S.; Ng, C.Y.; Torres, J.; Wang, R. Fast water transport through biomimetic reverse osmosis membranes embedded with peptide-attached (pR)-pillar [5] arenes water channels. J. Membr. Sci. 2021, 628, 119276. [Google Scholar] [CrossRef]
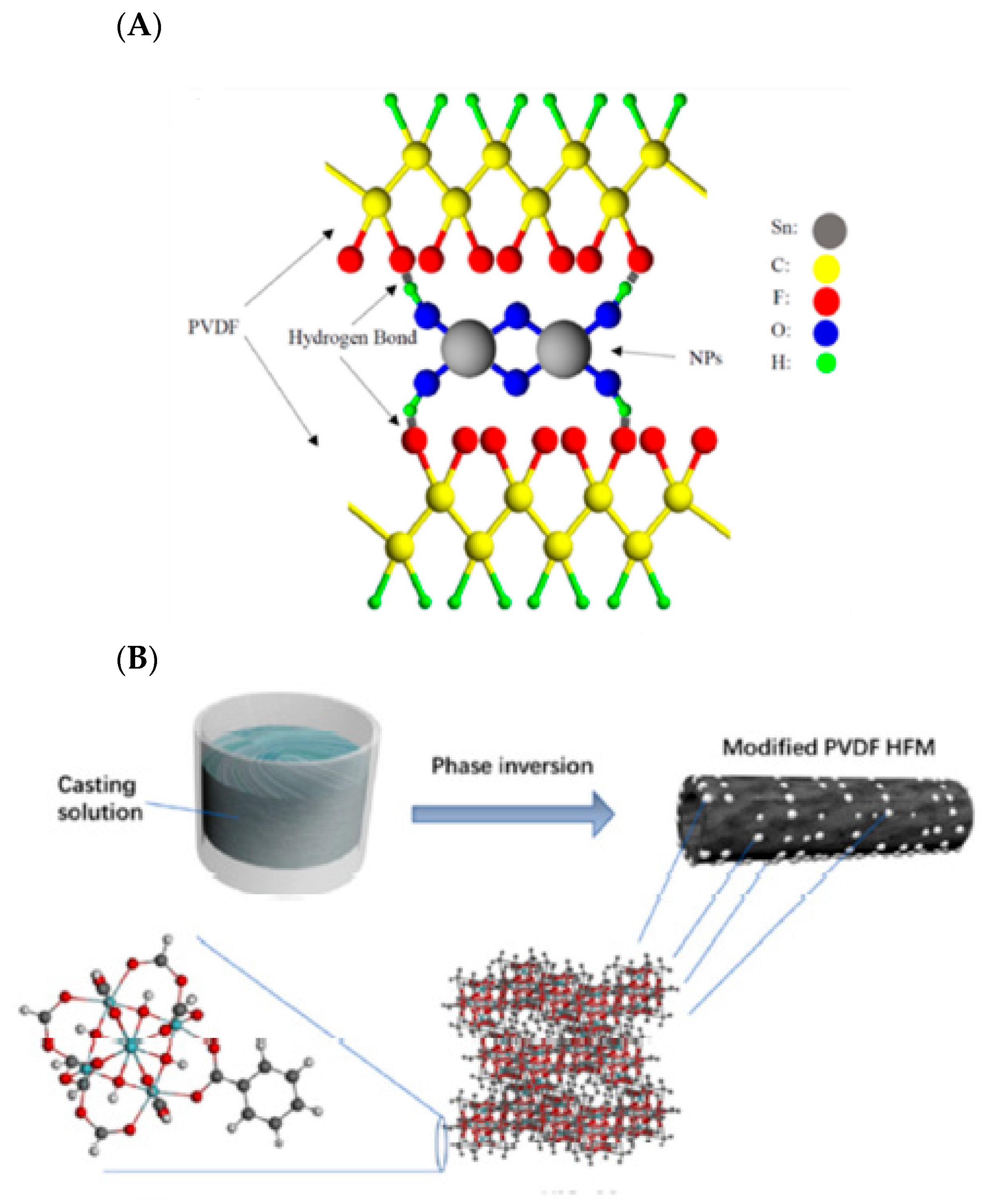
- Ibrahim, Y.; Naddeo, V.; Banat, F.; Hasan, S.W. Preparation of novel polyvinylidene fluoride (PVDF)-Tin (IV) oxide (SnO2) ion exchange mixed matrix membranes for the removal of heavy metals from aqueous solutions. Sep. Purif. Technol. 2020, 250, 117250. [Google Scholar] [CrossRef]
- Marino, T.; Russo, F.; Rezzouk, L.; Bouzid, A.; Figoli, A. PES-kaolin mixed matrix membranes for arsenic removal from water. Membranes 2017, 7, 57. [Google Scholar] [CrossRef]
- Du, R.; He, L.; Li, P.; Zhao, G. Polydopamine-Modified Al2O3/Polyurethane Composites with Largely Improved Thermal and Mechanical Properties. Materials 2020, 13, 1772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, X.; Sun, S.; Zhou, L.; Miao, Z.; Zhang, X.; Su, Z.; Wei, G. Removing Metal Ions from Water with Graphene–Bovine Serum Albumin Hybrid Membrane. Nanomaterials 2019, 9, 276. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Yang, P. Protein-based Separation Membranes: State of the Art and Future Trends. Adv. Energy Sustain. Res. 2021, 2, 2100008. [Google Scholar] [CrossRef]
- Abdullah, N.; Gohari, R.; Yusof, N.; Ismail, A.; Juhana, J.; Lau, W.; Matsuura, T. Polysulfone/hydrous ferric oxide ultrafiltration mixed matrix membrane: Preparation, characterization and its adsorptive removal of lead (II) from aqueous solution. Chem. Eng. J. 2016, 289, 28–37. [Google Scholar] [CrossRef]
- Yurekli, Y. Removal of heavy metals in wastewater by using zeolite nano-particles impregnated polysulfone membranes. J. Hazard. Mater. 2016, 309, 53–64. [Google Scholar] [CrossRef]
- Mukherjee, R.; Bhunia, P.; De, S. Impact of graphene oxide on removal of heavy metals using mixed matrix membrane. Chem. Eng. J. 2016, 292, 284–297. [Google Scholar] [CrossRef]
- Balkanloo, P.G.; Mahmoudian, M.; Hosseinzadeh, M.T. A comparative study between MMT-Fe3O4/PES, MMT-HBE/PES, and MMT-acid activated/PES mixed matrix membranes. Chem. Eng. J. 2020, 396, 125188. [Google Scholar] [CrossRef]
- Gohari, R.J.; Lau, W.; Matsuura, T.; Halakoo, E.; Ismail, A. Adsorptive removal of Pb (II) from aqueous solution by novel PES/HMO ultrafiltration mixed matrix membrane. Sep. Purif. Technol. 2013, 120, 59–68. [Google Scholar] [CrossRef]
- Gohari, R.J.; Lau, W.; Matsuura, T.; Ismail, A. Fabrication and characterization of novel PES/Fe–Mn binary oxide UF mixed matrix membrane for adsorptive removal of As (III) from contaminated water solution. Sep. Purif. Technol. 2013, 118, 64–72. [Google Scholar] [CrossRef]
- Tetala, K.K.; Stamatialis, D.F. Mixed matrix membranes for efficient adsorption of copper ions from aqueous solutions. Sep. Purif. Technol. 2013, 104, 214–220. [Google Scholar] [CrossRef]
- Abdulkarem, E.; Ibrahim, Y.; Kumar, M.; Arafat, H.A.; Naddeo, V.; Banat, F.; Hasan, S.W. Polyvinylidene fluoride (PVDF)-α-zirconium phosphate (α-ZrP) nanoparticles based mixed matrix membranes for removal of heavy metal ions. Chemosphere 2021, 267, 128896. [Google Scholar] [CrossRef]
- Shahrin, S.; Lau, W.-J.; Goh, P.-S.; Ismail, A.F.; Jaafar, J. Adsorptive mixed matrix membrane incorporating graphene oxide-manganese ferrite (GMF) hybrid nanomaterial for efficient As (V) ions removal. Compos. Part B Eng. 2019, 175, 107150. [Google Scholar] [CrossRef]
- Sherugar, P.; Naik, N.S.; Padaki, M.; Nayak, V.; Gangadharan, A.; Nadig, A.R.; Deon, S. Fabrication of zinc doped aluminium oxide/polysulfone mixed matrix membranes for enhanced antifouling property and heavy metal removal. Chemosphere 2021, 275, 130024. [Google Scholar] [CrossRef]
- Gebru, K.A.; Das, C. Removal of chromium (VI) ions from aqueous solutions using amine-impregnated TiO2 nanoparticles modified cellulose acetate membranes. Chemosphere 2018, 191, 673–684. [Google Scholar] [CrossRef]
- Mukherjee, R.; Bhunia, P.; De, S. Long term filtration modelling and scaling up of mixed matrix ultrafiltration hollow fiber membrane: A case study of chromium (VI) removal. J. Membr. Sci. 2019, 570, 204–214. [Google Scholar] [CrossRef]
- Marjani, A.; Nakhjiri, A.T.; Adimi, M.; Jirandehi, H.F.; Shirazian, S. Effect of graphene oxide on modifying polyethersulfone membrane performance and its application in wastewater treatment. Sci. Rep. 2020, 10, 2049. [Google Scholar] [CrossRef]
- Lim, Y.J.; Lee, S.M.; Wang, R.; Lee, J. Emerging materials to prepare mixed matrix membranes for pollutant removal in water. Membranes 2021, 11, 508. [Google Scholar] [CrossRef]
- Akbari, A.; Sheath, P.; Martin, S.T.; Shinde, D.B.; Shaibani, M.; Banerjee, P.C.; Tkacz, R.; Bhattacharyya, D.; Majumder, M. Large-area graphene-based nanofiltration membranes by shear alignment of discotic nematic liquid crystals of graphene oxide. Nat. Commun. 2016, 7, 10891. [Google Scholar] [CrossRef]
- Wan, P.; Yuan, M.; Yu, X.; Zhang, Z.; Deng, B. Arsenate removal by reactive mixed matrix PVDF hollow fiber membranes with UIO-66 metal organic frameworks. Chem. Eng. J 2020, 382, 122921. [Google Scholar] [CrossRef]
- Warsinger, D.M.; Chakraborty, S.; Tow, E.W.; Plumlee, M.H.; Bellona, C.; Loutatidou, S.; Karimi, L.; Mikelonis, A.M.; Achilli, A.; Ghassemi, A.; et al. A review of polymeric membranes and processes for potable water reuse. Prog. Polym. Sci. 2018, 81, 209–237. [Google Scholar] [CrossRef]
- Wang, C.; Lee, M.; Liu, X.; Wang, B.; Chen, J.P.; Li, K. A metal-organic framework/alpha-alumina composite with a novel geometry for enhanced adsorptive separation. Chem. Commun. 2016, 52, 8869–8872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valenzano, L.; Civalleri, B.; Chavan, S.; Bordiga, S.; Nilsen, M.H.; Jakobsen, S.; Lillerud, K.P.; Lamberti, C. Disclosing the complex structure of UiO-66 metal organic framework: A synergic combination of experiment and theory. Chem. Mater. 2011, 23, 1700–1718. [Google Scholar] [CrossRef]
- Liu, T.Y.; Yuan, H.G.; Liu, Y.Y.; Ren, D.; Su, Y.C.; Wang, X. Metal-organic framework nanocomposite thin films with interfacial bindings and self-standing robustness for high water flux and enhanced ion selectivity. ACS Nano 2018, 12, 9253–9265. [Google Scholar] [CrossRef] [PubMed]
- Seoane, B.; Coronas, J.; Gascon, I.; Benavides, M.E.; Karvan, O.; Caro, J.; Kapteijn, F.; Gascon, J. Metal–organic framework based mixed matrix membranes: A solution for highly efficient CO2 capture? Chem. Soc. Rev. 2015, 44, 2421–2454. [Google Scholar] [CrossRef] [PubMed]
- Bruno, R.; Mon, M.; Escamilla, P.; Ferrando-Soria, J.; Esposito, E.; Fuoco, A.; Monteleone, M.; Jansen, J.C.; Elliani, R.; Tagarelli, A. Bioinspired Metal-Organic Frameworks in Mixed Matrix Membranes for Efficient Static/Dynamic Removal of Mercury from Water. Adv. Funct. Mater. 2021, 31, 2008499. [Google Scholar] [CrossRef]
- Shayegan, H.; Ali, G.A.M.; Safarifard, V. Amide-Functionalized Metal–Organic Framework for High Efciency and Fast Removal of Pb(II) from Aqueous Solution. J. Inorg. Organomet. Polym. 2020, 30, 3170–3178. [Google Scholar] [CrossRef]
- Wen, J.; Fang, Y.; Zeng, G. Progress and prospect of adsorptive removal of heavy metal ions from aqueous solution using metal-organic frameworks: A review of studies from the last decade. Chemosphere 2018, 201, 627–643. [Google Scholar] [CrossRef]
- Shayegan, H.; Ali, G.A.M.; Safarifard, V. Recent Progress in the Removal of Heavy Metal Ions from Water Using Metal-Organic Frameworks. ChemistrySelect 2020, 5, 124–146. [Google Scholar] [CrossRef]
- Zhou, H.-C.; Kitagawa, S. Metal–Organic Frameworks (MOFs). Chem. Soc. Rev. 2014, 43, 5415–5418. [Google Scholar] [CrossRef]
- Kobielska, P.A.; Howarth, A.J.; Farha, O.K.; Nayak, S. Metal–organic frameworks for heavy metal removal from water. Coord. Chem. Rev. 2018, 358, 92–107. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, G.; Dai, H.; Yang, M.; Fu, Y.; Ying, Y.; Li, Y. Biomineralization-mimetic preparation of hybrid membranes with ultra-high loading of pristine metal–organic frameworks grown on silk nanofibers for hazard collection in water. J. Mater. Chem. A 2018, 6, 3402–3413. [Google Scholar] [CrossRef]
- Efome, J.E.; Rana, D.; Matsuuraa, T.; Lan, C.Q. Metal–organic frameworks supported on nanofibers to remove heavy metals. J. Mater. Chem. A 2018, 6, 4550–4555. [Google Scholar] [CrossRef]









| Element to Be Removed | Element Type | Source | Limit of Acceptability (mg/L) | Effect on Public Health [18,19] | Refs. |
|---|---|---|---|---|---|
| Arsenic | Metalloid | In groundwater, where there are sulfide mineral deposits and sedimentary deposits deriving from volcanic rocks, the concentrations can be significantly elevated. | 10 | Long-term exposure to arsenic in drinking water is causally related to increased risks of cancer in the skin, lungs, bladder, and kidney, as well as other skin changes, such as hyperkeratosis and pigmentation changes. | [20] |
| Boron | Metalloid | Groundwaters, seawater, wastewaters | 2400 | Animals suffer from kidney failure and reproductive system and nervous system diseases while plants can wither and eventually die when exposed to excess amounts of boron. | [21,22] |
| Cadmium | (Heavy) metal | Cadmium is released to the environment in wastewater from steel and plastic industries, batteries, impurities in the zinc of galvanized pipes and solders and some metal fittings, etc. Diffuse pollution is caused by contamination from fertilizers and local air pollution. Food is the main source of daily exposure to cadmium. | 3 | The kidney is the main target organ for cadmium toxicity. Cadmium has a long biological half-life in humans of 10–35 years. There is evidence that cadmium is carcinogenic by the inhalation route. However, there is no evidence of carcinogenicity by the oral route and no clear evidence for the genotoxicity of cadmium. | [20] |
| Copper | (heavy) metal | Copper is used to make pipes, valves, and fittings and is present in alloys and coatings. The primary source most often is the corrosion of interior copper plumbing. | 2 | Gastrointestinal effects | [20] |
| Iron | (heavy) metal | Anaerobic groundwater may contain ferrous iron at concentrations up to several milligrams per liter. Iron may also be present in drinking water as a result of the use of iron coagulants or the corrosion of steel and cast iron pipes during water distribution. | No health-based guideline value is proposed | Iron is an essential element in human nutrition, particularly in the (II) oxidation state. | [20] |
| Lead | (heavy) metal | Lead is rarely present in tap water as a result of its dissolution from natural sources. Rather, its presence is primarily from corrosive water effects on household plumbing systems containing lead in pipes, solder, fittings, or the service connections to homes. | 10 | Exposure to lead is associated with a wide range of effects, including various neurodevelopmental effects, mortality, impaired renal function, hypertension, impaired fertility, and adverse pregnancy outcomes. | [20] |
| Manganese | (heavy) metal | Manganese is naturally occurring in many surface water and groundwater sources, particularly in anaerobic or low oxidation conditions, and this is the most important source of drinking water. | At levels exceeding 100 mg L−1, it causes an undesirable taste | Not of health concern at levels found in drinking water | [20] |
| Mercury | (heavy) metal | Electrolytic production of chlorine, in electrical appliances, in dental amalgams and as a raw material for various mercury compounds. | 6 | Hemorrhagic gastritis and colitis with the ultimate damage to the kidney. | [20] |
| Molybdenum | (heavy) metal | Molybdenum is found naturally in soil and is used in the manufacture of special steels and in the production of tungsten and pigments. Molybdenum compounds are used as lubricant additives and in agriculture to prevent molybdenum deficiency in crops. | Although Mo is an essential element in the human body, WHO standards recommend that it does not exceed 70 mg L−1 in potable water | - | |
| Nickel | (heavy) metal | Where there is heavy pollution, areas in which nickel that occurs naturally in groundwater is mobilized or where there is use of certain types of kettles, of non-resistant material in wells, or of water that has come into contact with nickel or chromium-plated taps, the nickel contribution from water may be significant. | 70 | Possibly carcinogenic | [20] |
| Zinc | (heavy) metal | High concentrations in tap water can be observed as a result of dissolution of zinc from pipes. | Not of health concern at levels found in drinking-water | May affect acceptability of drinking-water |
| Entry # | Process | Description | Advantages | Disadvantages |
|---|---|---|---|---|
| 1 | Chemical precipitation | Chemicals (calcium hydroxide and sodium hydroxide in hydroxide precipitation, iron sulfide in sulfide precipitation) react with heavy metals ions to form insoluble precipitates, which can be separated from the water by sedimentation or filtration | Relatively simple and inexpensive | • Large volumes of relatively low-density sludge with possible problems of dewatering and disposal. • amphoteric characteristics of metals hydroxides. • inhibition of metal hydroxide precipitation due to possible presence of complexing agents in the wastewater. Moreover, in the case of sulfide precipitation, evolution of toxic H2S fumes in acidic conditions can occur and formation of colloidal precipitates can cause problems in either settling or filtration processes. |
| 2 | Adsorption | Heavy metal ions are directly separated from wastewater by adsorption on activated carbon, low-cost adsorbents, bio-adsorbents | Effective, economic, flexible in design and operation | Weak selectivity, waste product |
| 3 | Ion exchange | Solution containing heavy metals ions passes through a cation column allowing the exchange with the hydrogen ions on the resin | High treatment capacity, high removal efficiency | Careful control of variables such as pH, temperature, initial metal concentration, contact time, and ionic charge of the wastewater is mandatory |
| 4 | Ion flotation | Metal ions made hydrophobic by use of surfactants are removed by air bubbles | Ease of operation, low cost, and suitable for treating large volumes of dilute aqueous solutions | Careful control of variables such as pH, temperature, and initial metal concentration |
| 5 | Membrane processes | A membrane acts as a selective barrier in pressure-driven membrane operations and electrodialysis or only as barrier in liquid membranes operations allowing the separation of metal ions | High separation efficiency, easy to scale up, environmentally friendly, energy saving, no phase changing, and easy integration with other traditional processes | Careful selection of the membrane materials to address the challenges related to membrane lifetime, fouling, and selectivity reduction in the case of strong acidic, alkaline, and/or oxidizing feed solutions |
| Entry # | Membrane Process | Membrane Type | Driving Force | Mechanism of Separation | Remarks | Selected Studies |
|---|---|---|---|---|---|---|
| 1 | Reverse Osmosis (RO) | dense | Pressure difference | solution-diffusion | The process can be performant in removing low levels of heavy metals. Usually, it is coupled to other removal operations in a hybrid–integrated process. | [56,57,58,59,60,61,62,63,64,65,66] |
| 2 | Nanofiltration (NF) | porous charged (1–10 nm) | Pressure difference | Donnan exclusion | Compared to RO, NF is an energy-saving process and is very attractive for effective removal of heavy metal ions. | [67,68,69] |
| 3 | Forward osmosis (FO) | dense | Osmotic pressure difference | solution-diffusion | Differently, from RO, FO can be applied to treat high-salinity wastewater with the challenging objective to eliminate heavy metals here contained. However, FO suffers from concentration polarization. | [70,71] |
| 4 | Electrodialysis (ED) | ion-exchange membranes | Electric potential difference | Ion exclusion | ED can only be cost-effective if applied integrated with other treatment processes. Electrodeionization (EDI) can be effectively used in removal of metal ions from dilute solutions. | [72,73,74,75,76,77,78,79,80] |
| 5 | Membrane Capacitive DeIonization (MCDI) | ion-exchange membranes | Electrical potential | Ion exclusion | The electrostatic adsorption mechanism leads to low adsorption selectivities between different ion types with a similar charge. | [81,82] |
| 6 | Polymer-enhanced ultrafiltration (PEUF) | porous (0.01–0.1 μm) | Pressure difference | size exclusion | PEUF is a combination of selective binding of target metal ions to a water-soluble polymer followed by ultrafiltration. | [83,84,85,86,87,88,89] |
| 7 | Membrane bioreactors employing UF membranes (MBRs) | porous (0.01–0.1 μm) | Pressure difference | size exclusion | Removal efficiencies are lower than other membrane processes and depend on Mixed Liquor Suspended Solids (MLSS) concentration. Therefore, new concepts such as the ion-exchange membrane bioreactor (IEMB) process were developed to achieve high metal ions removal. | [90,91,92] |
| 8 | Osmotic membrane bioreactors (OMBRs) | dense | Osmotic pressure difference | solution-diffusion | OMBR is a low membrane fouling technology with high capability of nutrient removals and rejections of monovalent ions and thus can be considered promising for industrial wastewater containing heavy metals. | [93] |
| 9 | Liquid membranes (LMs) | porous (0.1–10 μm) | Concentration difference | solution-diffusion | LMs can be efficiently used for wastewater treatment. However, instability problems over time are one of the main shortcomings. | [94,95,96] |
| 10 | Membrane distillation (MD) | porous (0.1–10 μm) | Partial vapor pressure difference due to a temperature difference | vapor/liquid equilibrium of a liquid mixture occurs, therefore, the permeate composition is dependent on the partial pressure of respective components of the feed. | Comparison with pressure-driven membrane processes such as RO or NF suggests that with direct contact MD high metal ion removal efficiencies can be obtained. | [97,98] |
| 11 | Adsorptive membrane | Porous/dense | Pressure difference/Electrical potential | Adsorption (coordinative interactions, ion exchange, electrostatic adsorption, hydrogen bonding, specific surface bonding, and chelation). | All the advantages of adsorption (see Table 2) are combined with the ones of the membrane processes. | This review and references herein |
| Entry # | Adsorbent | Membrane | DM Type | Heavy Metals | Remarks | Ref. |
|---|---|---|---|---|---|---|
| 1 | Micro-sized granular ferric hydroxide (Fe(OH)3) (mGFH) | MF | Pre-deposited | As(V) | μGFH proved to be promising as emerging pre-depositing material for a DM filter | [132] |
| 2 | Micro-sized tetravalent manganese feroxyhyte (δ-Mn(IV) FeOOH) (mTMF) | MF | Pre-deposited | As(V) | μTMF proved to be promising as emerging pre-depositing material for a DM filter. | [132] |
| 3 | Polydopamine (PDA) nanoparticles | UF | Pre-deposited | Pb(II), Cd(II), Cu(II) | Significant enhancement of adsorption capacity is derived from the three-dimensional distribution of the adsorbent PDA on the cross section of UF membrane. | [133] |
| 4 | Hollow porous Zr(OH)x nanospheres (HPZNs) coated with PDA | UF | Pre-deposited | Pb(II) | Compared to the blend membrane, the pre-deposited DM showed 2.1-fold increase in the effective treatment volume for the treatment of Pb(II)-contaminated water from 100 ppb to below 10 ppb (WHO drinking water standard). | [134] |
| 5 | Graphene oxide/ PDA | UF | Pre-deposited | Pb(II) | Outstanding Pb(II) rejection can be attributed to chelation of amino groups on the PDA layer. | [135] |
| 6 | UiO-66-NH2 | UF | Pre-deposited | Cr(III)/Cr(VI) | Negligible leakage of the adsorbent UiO-66-NH2 owing to the successful cross-link of PEI. The membrane can be easily regenerated and reused. | [136] |
| Entry # | Type of Membrane | Heavy Metal Ions | Optimum Conditions/ Experimental Conditions | Adsorption | Ref. |
|---|---|---|---|---|---|
| mg g−1 | |||||
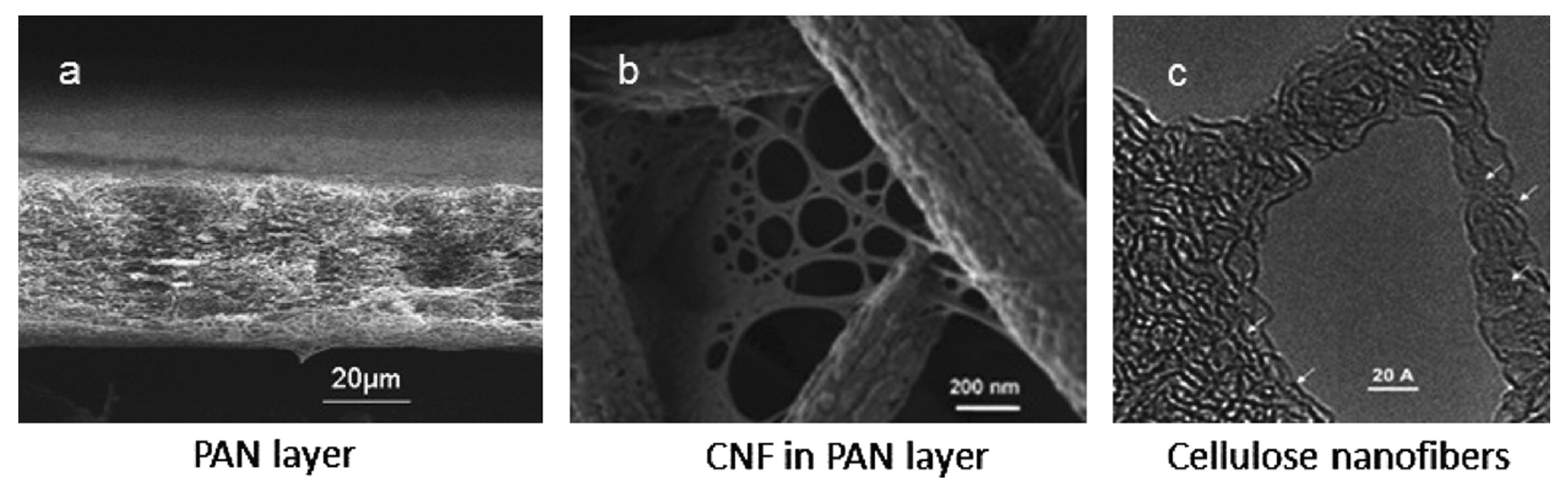
| 1 | Microfiltration nanofibrous (PVAm-CNF) | Cr(VI) | pH 4; initial conc. 50 ppm | 100 | [141] |
| 2 | Microfiltration nanofibrous (CNF) | Pb(II) | pH 6; initial conc. 50 ppm | 260 | [141] |
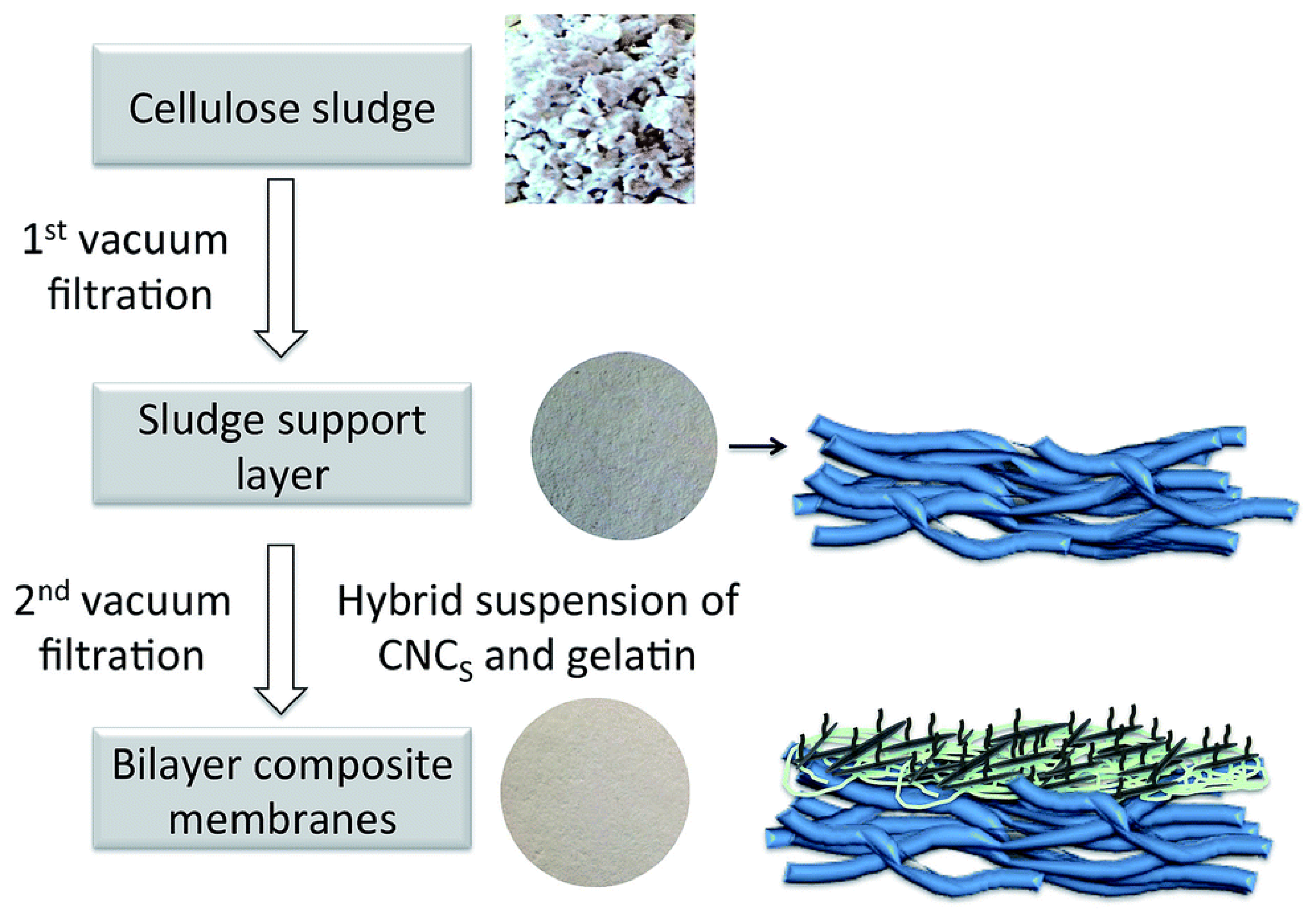
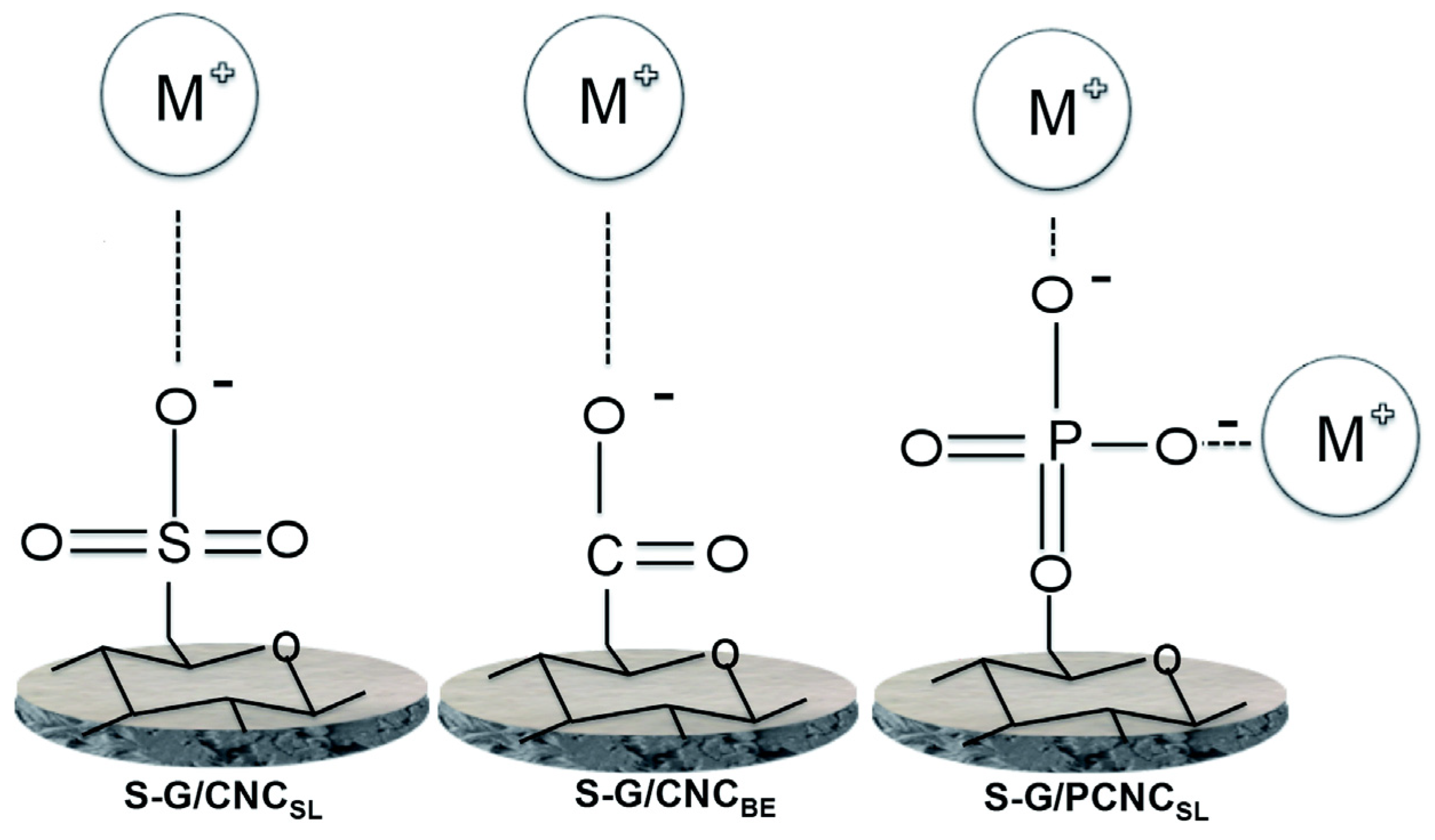
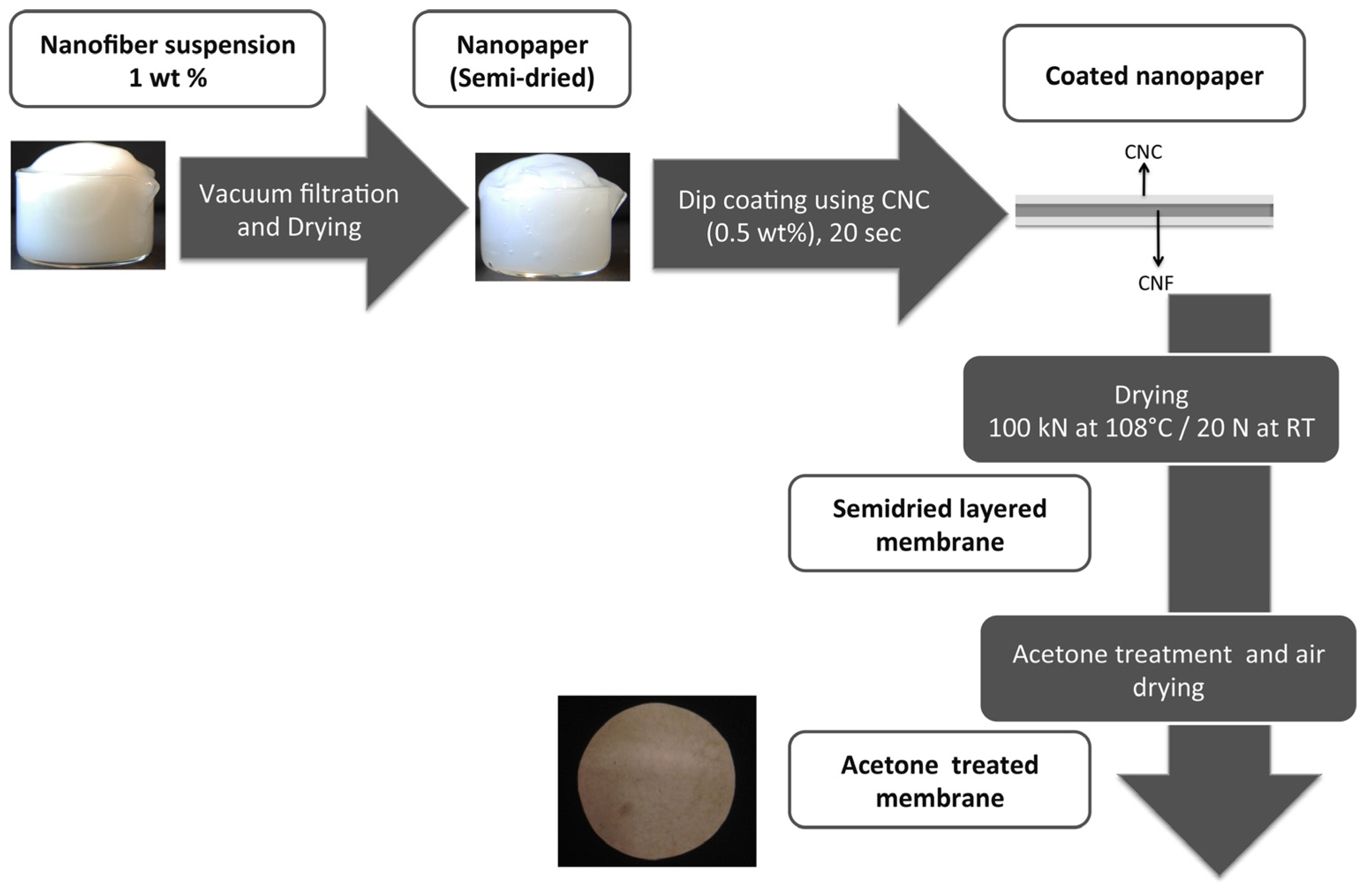
| 3 | Fully bio-based nanocellulose membrane (CNCSL) | Cu(II) * | pH 2.3; initial conc. 330 ppm | 9.6 | [142] |
| 4 | Fully bio-based nanocellulose membrane (CNCBE) | Cu(II) * | pH 2.3; initial conc. 330 ppm | 24 | [142] |
| 5 | Fully bio-based phosphorylated Nanocellulose membrane (PCNCSL) | Cu(II) * | pH 2.3; initial conc. 330 ppm | 79 | [142] |
| 6 | Fully bio-based nanocellulose membrane (CNCBE)_acetone treatment | Cu(II) * | pH 2.3 Initial conc. 330 pm | 33 | [143] |
| 7 | Fully bio-based nanocellulose membrane (CNCSL)_acetone treatment | Cu(II) * | pH 2.3; Initial conc. 330 ppm | 33 | [144] |
| 8 | Regenerated cellulose membrane grafted with PDMAEMA | Cu(II) | pH-; Initial conc. 25.6 ppm | 41.9 | [144] |
| 9 | Regenerated cellulose membrane grafted with PDMAEMA | Cu(II) | pH-; Initial conc. 76.8 ppm | 150 | [144] |
| 10 | Regenerated cellulose membrane grafted with PDMAEMA | Cu(II) | pH-; Initial conc. 128 ppm | 225 | [144] |
| Entry # | Cross-Linking Agent/Stabilizer | Heavy Metal Ion | Maximum Adsorption Capacity (mg g−1) | Ref. |
|---|---|---|---|---|
| 1 | PDA-βCD (cross-linking agent) | Pb(II) | 102 | [181] |
| 2 | PDA-PEI (cross-linking agent) | U(VI) | 531 | [184] |
| 3 | PVA (cross-linking agent) | Co(II), Ni(II), Cd(II) | 73, 62, 84 | [185] |
| 4 | Cellulose (cross-linking agent) | Co(II), Ni(II), Cu(II), Zn(II), Cd(II), Pb(II) | 16, 14, 27, 17, 27, 108 | [186] |
| 5 | CNTs (stabilizer) | Co(II), Ni(II), Cu(II), Zn(II), Cd(II), Pb(II) | 37, 40, 50, 42, 48, 98 | [187] |
| Membrane Materials | Target Pollutant | Adsorption Capacity (mg g−1) | Ref. |
|---|---|---|---|
| Chitosan-PVA | Cu(II) Ni(II) Cd(II) | 98.65 116.89 124.23 | [213] |
| Chitosan-cellulose | As(V) Pb(II) Cu(II) | 39.4 57.3 112.6 | [214] |
| Chitosan-polyethylene oxide | Ni(II) Cu(II) Ca(II) Pb(II) | 357.1 310.2 248.1 237.2 | [215] |
| Chitosan-poly (L-lactic acid) | Cu(II) | 111.66 | [216] |
| Chitosan-poly(ethylene oxide) | Cu(II) Zn(II) Pb(II) | 120 117 108 | [217] |
| Chitosan-PVA | Cu(II) | 90.3 | [218] |
| Chitosan-poly(ethylene oxide) | Ni(II) | 227.27 | [219] |
| Chitosan-poly(ethylene oxide) | Cr(VI) | 208 | [220] |
| Polyacrylic acid-sodium alginate | Cu(II) | 591.7 | [221] |
| Chitosan-sodium polyacrylate | Cr(VI) | 78.92 | [222] |
| Polyacrylic acid-PVA | Pb(II) Cd(II) | 159 102 | [223] |
| Chitosan-CA | Cd(II) | 110.48 | [224] |
| Polyacrylic acid-PVA | Pb(II) | 288 | [225] |
| Polyethyleneimine-PVA | Cr(VI) | 150 | [226] |
| Polyethyleneimine-PDA | Cu(II) | 33.59 | [227] |
| Poly(ether sulfones)-poly(ethyleneimine) | Cu(II) Cd(II) Pb(II) | 161.29 357.14 94.34 | [228] |
| Polyethyleneimine-PVA | Cu(II) Cd(II) Pb(II) | 67.16 116.94 90.03 | [229] |
| Polyaniline-polystyrene | Pb(II) Cu(II) Hg(II) Cd(II) Cr(VI) | 312 171 148 124 58 | [230] |
| Polyethersulfone-poly (dimethyl amine) ethyl methacrylate | Cu(II) | 161.3 | [231] |
| CA-polymethacrylic acid | Pb(II) | 146.21 | [232] |
| β-cyclodextrin-polyacrylate | Cu(II) Fe(II) | 82 219.5 | [233] |
| Polyindole | Cu(II) | 121.95 | [234] |
| PAN—polypyrrole | Cr(VI) | 74.91 | [235] |
| Poly(ethylene oxide) | Cu(II) | 15.6 | [236] |
| PDA-PVDF-polypyrrole | Cr(VI) | 126.7 | [237] |
| Polyindole | Cd(II) | 140.36 | [238] |
| PVDF-PDA | Cu(II) | 26.7 | [239] |
| PAN-Chitosan-Regenerated cellulose | Pb(II) | 500.95 | [240] |
| PAN-polyvinylpyrrolidone | Pb(II) | 1520 | [241] |
| Deacetylated cellulose-pyromellitic dianhydride | Pb(II) | 326.80 | [242] |
| Chitosan-poly(glycidyl methacrylate)-polyethylenimine | Cr(VI) Cu(II) Co(II) | 138.96 69.27 68.31 | [243] |
| CA-polyvinylpyrrolidone | Pb(II) Cu(II) Cd(II) | 30.96 19.63 34.70 | [244] |
| Cellulose nanofiber-PAN | Cr(VI) Pb(II) | 87.5 137.7 | [245] |
| Chitosan-poly(ethylene oxide) | Cd(II) | 232.55 | [246] |
| PAN-CA | Fe(III) Cu(II) Cd(II) | 418.32 272.64 126.56 | [247] |
| Polyurethane-phytic acid | Pb(II) | 136.52 | [248] |
| Poly(ether sulfone)-poly(3,4-ethylene dioxythiophene) | Pb(II) Cd(II) Cr(VI) | 656.42 315.55 418.86 | [249] |
| MMM (Filler/Polymer Matrix) | Removed Heavy Metal | Reported Removal Efficiency | Membrane Filtration Area (cm2) | Ref. |
|---|---|---|---|---|
| Hydrous ferric oxide NPs a/PSf | Pb(II) | 13.2 mg g−1 | - | [257] |
| Zeolite/PSf | Ni(II) | 122 mg g−1 | 13.4 | [258] |
| GO/PSf | Pb(II) Cu(II) Cd(II) Cr(II) | 79 mg g−1 75 mg g−1 68 mg g−1 154 mg g−1 | 4 | [259,260,261,262,263,264,265,266,267,268] |
| SnNPs b/PVDF | Pb(II) Cu(II) Zn(II) Cd(II) Ni(II) | 93.9% 92.8% 82.3% 70.7% 63.9% | 29.20 | [252] |
| HBE-MMTc/PES | Cu(II) Zn(II) Cd(II) Ni(II) | 46.8% 66.9% 39.1% 68.8% | - | [260] |
| HMO d/PES | Pb(II) | 204.1 mg g−1 | 12.56 | [261] |
| Fe-Mn binary Oxide/PES | As(III) | 87.5% | 12.56 | [262] |
| Chitosan beads/EVAL | Cu(II) | 225 mg g−1 | - | [263] |
| α-ZrP e/PVDF | Pb(II) Cu(II) Zn(II) Cd(II) Ni(II) | 91.2% 93.1% 44.2% 42.8% 44.4% | 1 | [264] |
| GMF f/PES | As(V) | 75.5 mg g−1 | 42 | [265] |
| Zn:-Al2O3 g/PSf | Pb(II) | 98% | - | [266] |
| Amine modified TiO2/CA | Cr(VI) | 99.6% | - | [267] |
| GO/PES | Cu(II) Zn(II) | Cu(II): 72% Zn(II): 87% | 3.73 | [269] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buonomenna, M.G.; Mousavi, S.M.; Hashemi, S.A.; Lai, C.W. Water Cleaning Adsorptive Membranes for Efficient Removal of Heavy Metals and Metalloids. Water 2022, 14, 2718. https://doi.org/10.3390/w14172718
Buonomenna MG, Mousavi SM, Hashemi SA, Lai CW. Water Cleaning Adsorptive Membranes for Efficient Removal of Heavy Metals and Metalloids. Water. 2022; 14(17):2718. https://doi.org/10.3390/w14172718
Chicago/Turabian StyleBuonomenna, Maria Giovanna, Seyyed Mojtaba Mousavi, Seyyed Alireza Hashemi, and Chin Wei Lai. 2022. "Water Cleaning Adsorptive Membranes for Efficient Removal of Heavy Metals and Metalloids" Water 14, no. 17: 2718. https://doi.org/10.3390/w14172718






