A Comprehensive Overview of the Hydrochemical Characteristics of Precipitation across the Middle East
Abstract
1. Introduction
2. The Geography and Climatology of the Middle East
3. Materials and Methods
4. Results and Discussion
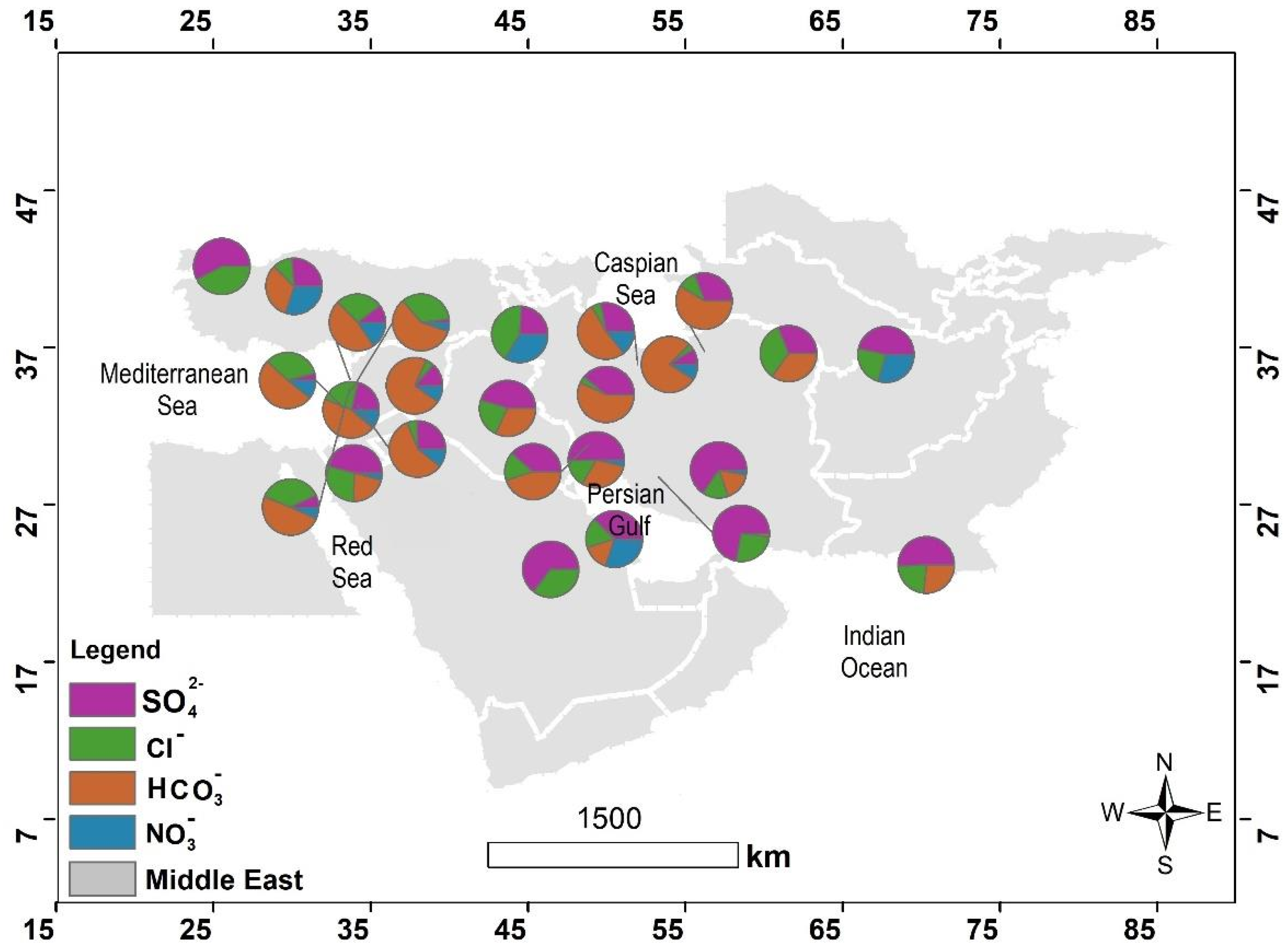
4.1. The Hydrochemical Characteristics of Precipitation across the Middle East
4.2. The Origins of the Major Ions in Precipitation in the Middle East
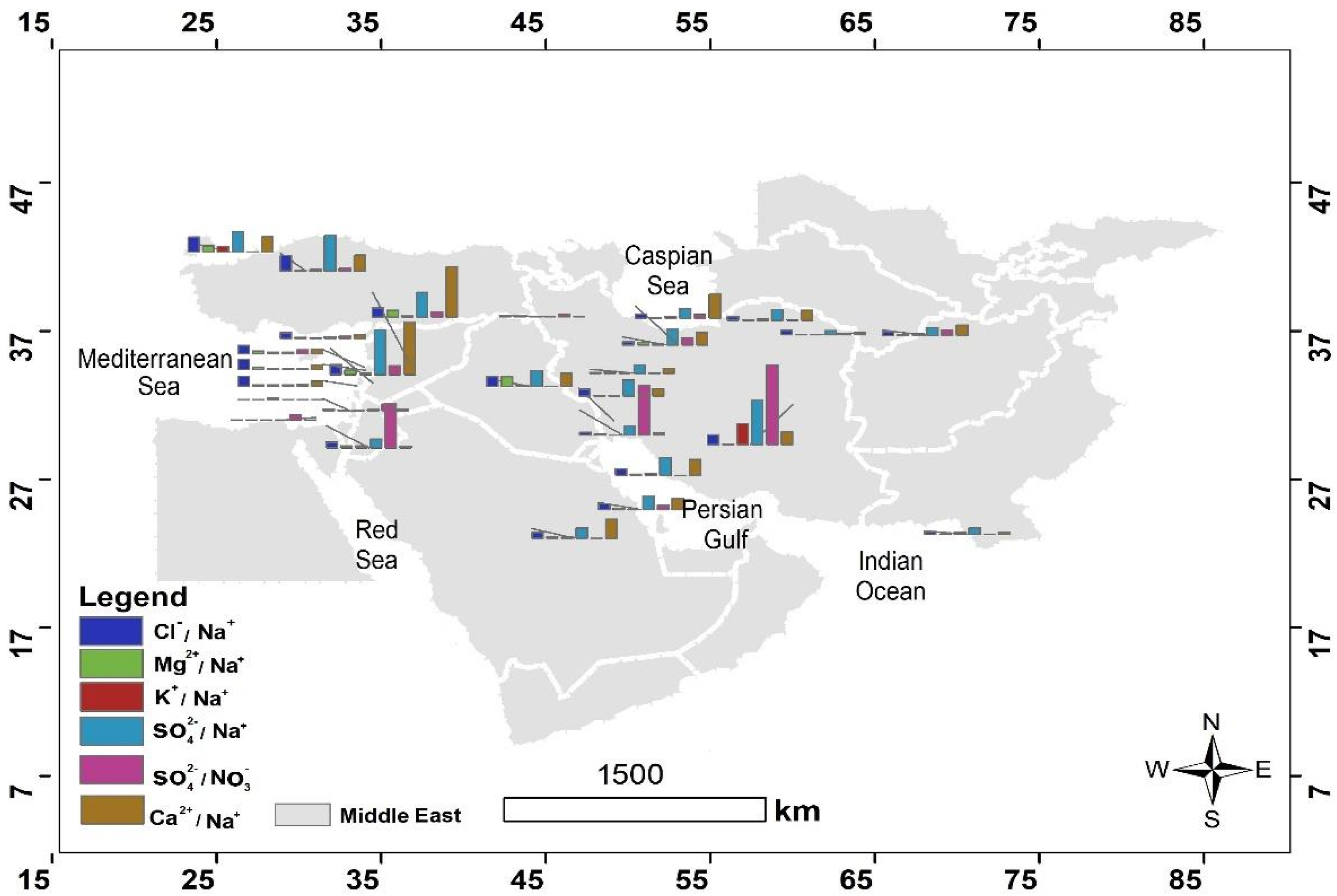
4.2.1. The Sea Salt and Nonsea Salt Contributions to the Middle East Precipitation
4.2.2. Calculating the Enrichment Factor in the Middle East Precipitation
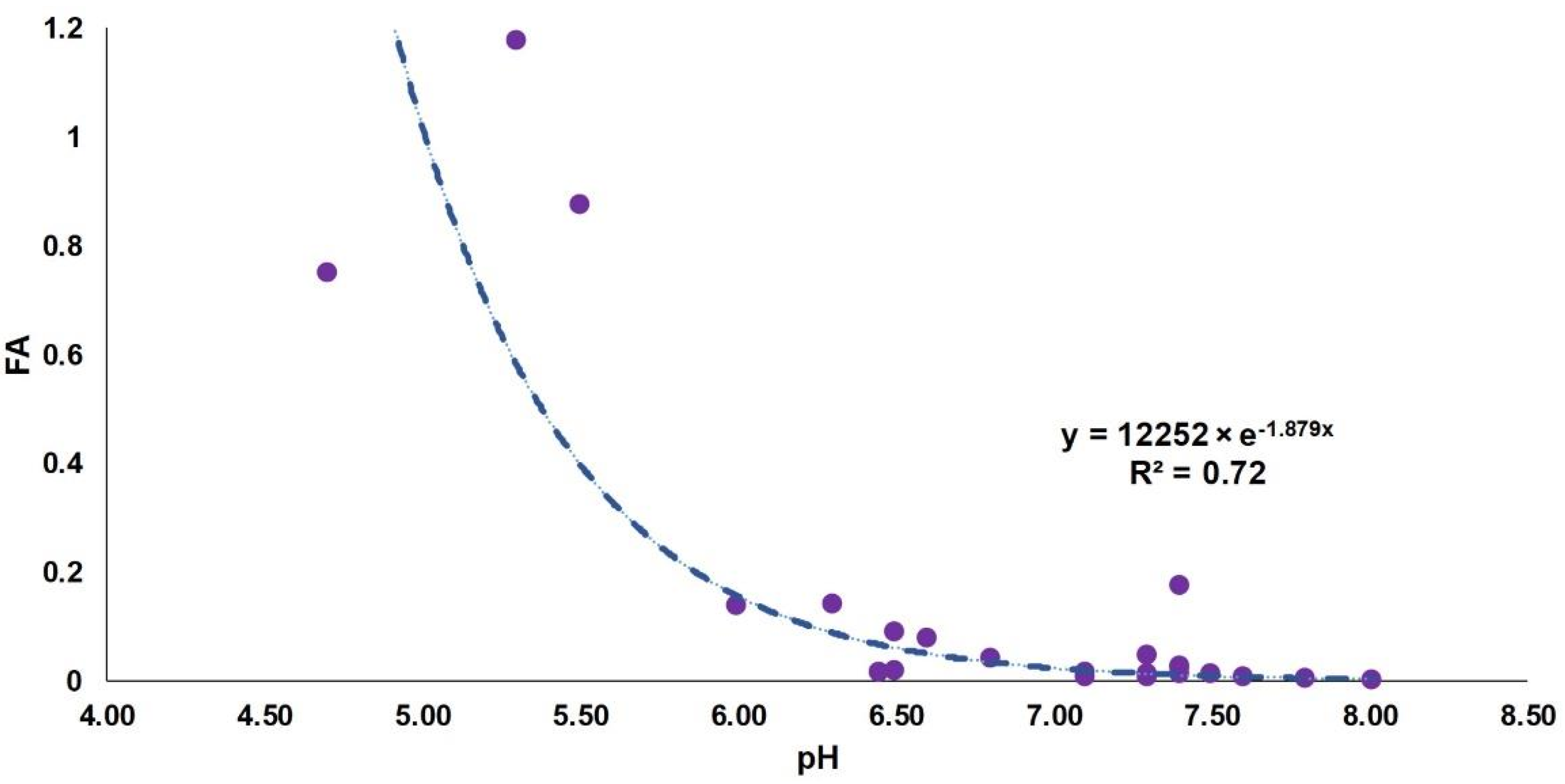
4.2.3. The Acidity and Neutralizing Capacity of the Middle East Precipitation
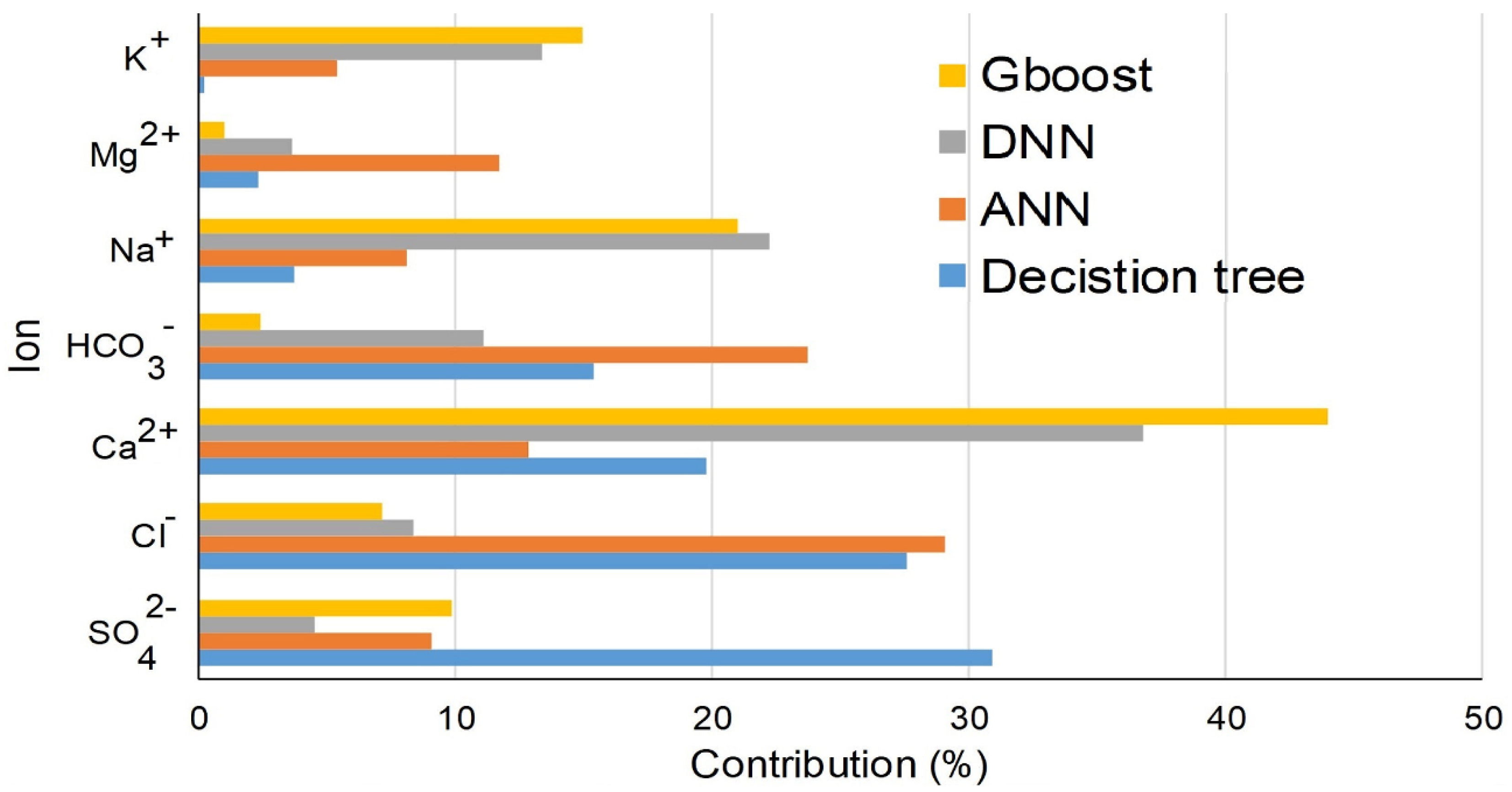
4.3. Applying Statistical and Machine Learning Techniques in Hydrochemical Studies of the Middle East Precipitation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bozorg-Haddad, O.; Zolghadr-Asli, B.; Sarzaeim, P.; Aboutalebi, M.; Chu, X.; Loáiciga, H.A. Evaluation of water shortage crisis in the Middle East and possible remedies. J. Water Supply Res. Technol. 2019, 69, 85–98. [Google Scholar] [CrossRef]
- Baconi, T. Testing the Water: How Water Scarcity Could Destabilise the Middle East and North Africa. 2018. Available online: https://ecfr.eu/publication/how_water_scarcity_could_destabilise_the_middle_east_and_north_africa/ (accessed on 11 July 2022).
- Araghi, A.; Mousavi-Baygi, M.; Adamowski, J.; Martinez, C. Association between three prominent climatic teleconnections and precipitation in Iran using wavelet coherence. Int. J. Clim. 2016, 37, 2809–2830. [Google Scholar] [CrossRef]
- Balling, R.C.; Kiany, M.S.K.; Roy, S.S.; Khoshhal, J. Trends in Extreme Precipitation Indices in Iran: 1951–2007. Adv. Meteorol. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Rahimzadeh, F.; Asgari, A.; Fattahi, E. Variability of extreme temperature and precipitation in Iran during recent decades. Int. J. Clim. 2008, 29, 329–343. [Google Scholar] [CrossRef]
- Pourasghar, F.; Tozuka, T.; Jahanbakhsh, S.; Sarraf, B.S.; Ghaemi, H.; Yamagata, T. The interannual precipitation variability in the southern part of Iran as linked to large-scale climate modes. Clim. Dyn. 2012, 39, 2329–2341. [Google Scholar] [CrossRef]
- Khalili, A.; Rahimi, J. High-resolution spatiotemporal distribution of precipitation in Iran: A comparative study with three global-precipitation datasets. Arch. Meteorol. Geophys. Bioclimatol. Ser. B 2013, 118, 211–221. [Google Scholar] [CrossRef]
- Javari, M. Trend and Homogeneity Analysis of Precipitation in Iran. Climate 2016, 4, 44. [Google Scholar] [CrossRef]
- Raziei, T.; Mofidi, A.; dos Santos, J.C.A.; Bordi, I. Spatial patterns and regimes of daily precipitation in Iran in relation to large-scale atmospheric circulation. Int. J. Clim. 2011, 32, 1226–1237. [Google Scholar] [CrossRef]
- Al-Khashman, O.A. Ionic composition of wet precipitation in the Petra Region, Jordan. Atmos. Res. 2005, 78, 1–12. [Google Scholar] [CrossRef]
- Salameh, E.; Rimawi, O. Hydrochemistry of precipitation of northern Jordan. Int. J. Environ. Stud. 1988, 32, 203–216. [Google Scholar] [CrossRef]
- Shamsi, A.; Karami, G.; Taheri, A.; Hunkeler, D. Hydrochemistry and stable isotopes study of the precipitation at Haraz Basin, north of Iran. Geopersia 2019, 10, 23–39. [Google Scholar] [CrossRef]
- Zhai, Y.; Wang, J.; Zhang, Y.; Teng, Y.; Zuo, R.; Huan, H. Hydrochemical and isotopic investigation of atmospheric precipitation in Beijing, China. Sci. Total Environ. 2013, 456–457, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, M.; Pajooheshfar, S.P. Acid Rain Examination and Chemical Composition of Atmospheric Precipitation in Tehran, Iran. Environ. Asia 2012, 5, 39–47. [Google Scholar] [CrossRef]
- Okuda, T.; Iwase, T.; Ueda, H.; Suda, Y.; Tanaka, S.; Dokiya, Y.; Fushimi, K.; Hosoe, M. Long-term trend of chemical constituents in precipitation in Tokyo metropolitan area, Japan, from 1990 to 2002. Sci. Total Environ. 2005, 339, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Vong, R.J.; Larson, T.V.; Covert, D.S.; Waggoner, A.P. Measurement and modeling of Western Washington precipitation chemistry. Water Air Soil Pollut. 1985, 26, 71–84. [Google Scholar] [CrossRef]
- De Mello, W. Precipitation chemistry in the coast of the Metropolitan Region of Rio de Janeiro, Brazil. Environ. Pollut. 2001, 114, 235–242. [Google Scholar] [CrossRef]
- Ayers, G.P.; Gillett, R.W. Some observations on the acidity and composition of rainwater in Sydney, Australia, during the summer of 1980?81. J. Atmos. Chem. 1984, 2, 25–46. [Google Scholar] [CrossRef]
- Ali, K.; Momin, G.; Tiwari, S.; Safai, P.; Chate, D.; Rao, P. Fog and precipitation chemistry at Delhi, North India. Atmos. Environ. 2004, 38, 4215–4222. [Google Scholar] [CrossRef]
- Miller, N.H. The Composition of Rain-Water Collected in the Hebrides in Iceland; With Special Reference to Theamount of Nitrogen as Ammonia and as Nitrates. Scott. Met. Soc. 1913, 16, 141–158. Available online: https://repository.rothamsted.ac.uk/item/97w54/the-composition-of-rainwater-collected-in-the-hebrides-in-iceland-with-special-reference-to-the-amount-of-nitrogen-as-ammonia-and-as-nitrates (accessed on 11 July 2022).
- Schemenauer, R.S.; Cereceda, P. Monsoon cloudwater chemistry on the Arabian Peninsula. Atmos. Environ. Part. A Gen. Top. 1992, 26, 1583–1587. [Google Scholar] [CrossRef]
- Kattan, Z. Chemical and environmental isotope study of precipitation in Syria. J. Arid Environ. 1997, 35, 601–615. [Google Scholar] [CrossRef]
- Keresztesi, Á.; Birsan, M.-V.; Nita, I.-A.; Bodor, Z.; Szép, R. Assessing the neutralisation, wet deposition and source contributions of the precipitation chemistry over Europe during 2000–2017. Environ. Sci. Eur. 2019, 31, 50. [Google Scholar] [CrossRef]
- Abdemanafi, D.; Meshkatee, A.-H.; Hajjam, S.; Vazifedoust, M. The study of relation between aerosols and quality of rain water over Tehran during 2012–2013 years. J. Environ. Sci. Tech. 2017, 19, 15–30. [Google Scholar]
- Saad, Z.; Slim, K.; Ghaddar, A.; Nasreddine, M.; Kattan, Z. Chemical composition of rain water in Lebanon. J. Eur. Hydrol. 2000, 31, 223–238. [Google Scholar] [CrossRef]
- Naimabadi, A.; Shirmardi, M.; Maleki, H.; Teymouri, P.; Goudarzi, G.; Shahsavani, A.; Sorooshian, A.; Babaei, A.A.; Mehrabi, N.; Baneshi, M.M.; et al. On the chemical nature of precipitation in a populated Middle Eastern Region (Ahvaz, Iran) with diverse sources. Ecotoxicol. Environ. Saf. 2018, 163, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Alkhashman, O. Study of chemical composition in wet atmospheric precipitation in Eshidiya area, Jordan. Atmos. Environ. 2005, 39, 6175–6183. [Google Scholar] [CrossRef]
- Ghadimi, F.; Ghomi, M.; Ranjbar, M.; Hajati, A. Sources of contamination in rainwater by major and heavy elements in Arak, Iran. J. Water Sci. Res. 2013, 5, 67–84. [Google Scholar]
- Fabbian, D.; de Dear, R.; Lellyett, S.C. Application of Artificial Neural Network Forecasts to Predict Fog at Canberra International Airport. Weather Forecast 2007, 22, 372–381. [Google Scholar] [CrossRef]
- Rumelhart, D.E.; Hinton, G.E.; McClelland, J.L. A general framework for Parallel Distributed Processing. In Parallel Distributed Processing; The MIT Press: Cambridge, MA, USA, 1986; p. 517. [Google Scholar]
- Hall, T.; Brooks, H.E.; Doswell, C.A., III. Precipitation Forecasting Using a Neural Network. Weather Forecast 1999, 14, 338–345. [Google Scholar] [CrossRef]
- Mislan; Haviluddin; Hardwinarto, S.; Sumaryono; Aipassa, M. Rainfall Monthly Prediction Based on Artificial Neural Network: A Case Study in Tenggarong Station, East Kalimantan—Indonesia. Procedia Comput. Sci. 2015, 59, 142–151. [Google Scholar] [CrossRef]
- Moustris, K.P.; Larissi, I.K.; Nastos, P.T.; Paliatsos, A.G. Precipitation Forecast Using Artificial Neural Networks in Specific Regions of Greece. Water Resour. Manag. 2011, 25, 1979–1993. [Google Scholar] [CrossRef]
- Kang, J.; Wang, H.; Yuan, F.; Wang, Z.; Huang, J.; Qiu, T. Prediction of Precipitation Based on Recurrent Neural Networks in Jingdezhen, Jiangxi Province, China. Atmosphere 2020, 11, 246. [Google Scholar] [CrossRef]
- Ogunrinde, A.T.; Oguntunde, P.G.; Fasinmirin, J.T.; Akinwumiju, A.S. Application of artificial neural network for forecasting standardized precipitation and evapotranspiration index: A case study of Nigeria. Eng. Rep. 2020, 2. [Google Scholar] [CrossRef]
- Bączkiewicz, A.; Wątróbski, J.; Sałabun, W.; Kołodziejczyk, J. An ANN Model Trained on Regional Data in the Prediction of Particular Weather Conditions. Appl. Sci. 2021, 11, 4757. [Google Scholar] [CrossRef]
- Zabbah, I.; Roshani, A.R.; Khafage, A. Prediction of monthly rainfall using artificial neural network mixture approach, Case Study: Torbat-e Heydariyeh. J. Earth Space Phys. 2018, 44, 115–126. [Google Scholar] [CrossRef]
- Yen, M.-H.; Liu, D.-W.; Hsin, Y.-C.; Lin, C.-E.; Chen, C.-C. Application of the deep learning for the prediction of rainfall in Southern Taiwan. Sci. Rep. 2019, 9, 12774. [Google Scholar] [CrossRef]
- Weesakul, U.; Chaiyasarn, K.; Mahat, S. Long-term rainfall forecasting using deep neural network coupling with input variables selection technique: A case study of Ping River Basin, Thailand. Eng. Appl. Sci. Res. 2021, 48, 209–220. [Google Scholar]
- Das, S.; Chakraborty, R.; Maitra, A. A random forest algorithm for nowcasting of intense precipitation events. Adv. Space Res. 2017, 60, 1271–1282. [Google Scholar] [CrossRef]
- Wolfensberger, D.; Gabella, M.; Boscacci, M.; Germann, U.; Berne, A. RainForest: A random forest algorithm for quantitative precipitation estimation over Switzerland. Atmos. Meas. Tech. 2021, 14, 3169–3193. [Google Scholar] [CrossRef]
- Ortiz-García, E.G.; Salcedo-Sanz, S.; Casanova-Mateo, C. Accurate precipitation prediction with support vector classifiers: A study including novel predictive variables and observational data. Atmos. Res. 2014, 139, 128–136. [Google Scholar] [CrossRef]
- Du, J.; Liu, Y.; Yu, Y.; Yan, W. A Prediction of Precipitation Data Based on Support Vector Machine and Particle Swarm Optimization (PSO-SVM) Algorithms. Algorithms 2017, 10, 57. [Google Scholar] [CrossRef]
- Zhang, J.; Yao, Y.; Cao, N. Prediction of whether precipitation based on decision tree. J. Geomat. 2017, 42, 107–109. [Google Scholar] [CrossRef]
- Monego, V.S.; Anochi, J.A.; Velho, H.F.D.C. South America Seasonal Precipitation Prediction by Gradient-Boosting Machine-Learning Approach. Atmosphere 2022, 13, 243. [Google Scholar] [CrossRef]
- Beck, H.E.; Zimmermann, N.E.; McVicar, T.R.; Vergopolan, N.; Berg, A.; Wood, E.F. Present and future Köppen-Geiger climate classification maps at 1-km resolution. Sci. Data. 2018, 5, 180214. [Google Scholar] [CrossRef]
- Mohebi, A. Studying the parameters influenceing precipitation variations across Middle East. Water Resour. 2012, 15, 205–209. [Google Scholar]
- Heydarizad, M.; Gimeno, L.; Sorí, R.; Minaei, F.; Mayvan, J.E. The Stable Isotope Characteristics of Precipitation in the Middle East Highlighting the Link between the Köppen Climate Classifications and the δ18O and δ2H Values of Precipitation. Water 2021, 13, 2397. [Google Scholar] [CrossRef]
- Alijani, B. Iran Climatology, 5th ed.; Payam Nour Publication: Tehran, Iran, 2000; p. 230. [Google Scholar]
- Drumond, A.; Taboada, E.; Nieto, R.; Gimeno, L.; Vicente-Serrano, S.M.; Ignacio López-Moreno, J. Lagrangian analysis of the present-day sources of moisture for major ice-core sites. Earth Syst. Dynam. 2016, 7, 549–558. [Google Scholar] [CrossRef]
- Heydarizad, M. Meteoric Water Lines of Iran for Various Precipitation Sources. Ph.D. Thesis, Shiraz University, Shiraz, Iran, 2018. [Google Scholar]
- Heydarizad, M.; Raeisi, E.; Sori, R.; Gimeno, L. The Identification of Iran’s Moisture Sources Using a Lagrangian Particle Dispersion Model. Atmosphere 2018, 9, 408. [Google Scholar] [CrossRef]
- Dee, D.P.; Uppala, S.M.; Simmons, A.J.; Berrisford, P.; Poli, P.; Kobayashi, S.; Andrae, U.; Balmaseda, M.A.; Balsamo, G.; Bauer, P.; et al. The ERA-Interim reanalysis: Configuration and performance of the data assimilation system. Q. J. R. Meteorol. Soc. 2011, 137, 553–597. [Google Scholar] [CrossRef]
- Moore, F.; Attar, A. Rainwater and the resulting runoff chemistry in Shiraz city, southwest Iran. Int. J. Environ. Stud. 2011, 68, 703–717. [Google Scholar] [CrossRef]
- Mehr, M.R.; Keshavarzi, B.; Sorooshian, A. Influence of natural and urban emissions on rainwater chemistry at a southwestern Iran coastal site. Sci. Total Environ. 2019, 668, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Mirhosseini, M.; Shahabpour, J.; Behrouz, M. Hydrochemical charcterstic of acidic rain in Sarcheshmeh area in Kerman province. Scinece J. 2008, 19, 74–83. [Google Scholar]
- Behravesh, F.; Mahmoudi Gharaee, M.H.; Ghasem zadeh, F.; Avaz Moghadam, S. Quality assessment of precipitation and run off in Mashhad city. In Proceedings of the the 2nd Iranian Rainwater Catchment Systems Association; Iranian Rainwater Catchment Systems Association: Mashhad, Iran, 2019; pp. 1–9. [Google Scholar]
- Kazemi, G.A.; Ichiyanagi, K.; Shimada, J. Isotopic characteristics, chemical composition and salinization of atmospheric precipitation in Shahrood, northeastern Iran. Environ. Earth Sci. 2014, 73, 361–374. [Google Scholar] [CrossRef]
- Hajizadeh, Y.; Nemati, S.; Aghapour, A.A.; Abdolahnejad, A.; Mohammadi, A.; Panahi, H.; Moghanjooghi, S.M.; Nikoonahad, A. Influence of Air Pollution on Chemical Quality of Wet Atmospheric Deposition: A Case Study in Urmia, Iran. Iran. J. Heal. Saf. Environ. 2017, 5, 904–910. Available online: https://www.researchgate.net/publication/320629105_Influence_of_Air_Pollution_on_Chemical_Quality_of_Wet_Atmospheric_Deposition_a_Case_Study_in_Urmia_Iran (accessed on 11 July 2022).
- Michelsen, N.; Reshid, M.; Siebert, C.; Schulz, S.; Knöller, K.; Weise, S.M.; Rausch, R.; Al-Saud, M.; Schüth, C. Isotopic and chemical composition of precipitation in Riyadh, Saudi Arabia. Chem. Geol. 2015, 413, 51–62. [Google Scholar] [CrossRef]
- Precipitation Quality and Related Atmospheric Chemistry over the Greater Damietta Area-Egypt. Res. J. Environ. Sci. 2008, 2, 252–265. [CrossRef][Green Version]
- Masood, S.S.; Saied, S.; Siddique, A.; Mohiuddin, S.; Hussain, M.M.; Khan, M.K.; Khwaja, H.A. nfluence of urban–coastal activities on organic acids and major ionchemistry of wet precipitation at a metropolis in Pakistan. Arab. J. Geosci. 2018, 11, 802. [Google Scholar] [CrossRef]
- Ghanem, M.; Shalash, I.; Al-Rimmawi, H. Hydrochemical Characteristics of Rainwater in Ramallah District. Asian J. Earth Sci. 2010, 3, 11–19. [Google Scholar] [CrossRef]
- Al Charideh, A.R.; Zakhem, B.A. Distribution of tritium and stable isotopes in precipitation in Syria. Hydrol. Sci. J. 2010, 55, 832–843. [Google Scholar] [CrossRef]
- Ghaleb, N.; Salman, A.; Qassim, S.; Ebrahim, A.; Saleh, M. Quality Assessment of Rainwater from Rainwater Harvesting Systems in Jerdab, Kingdom of Bahrain. Int. J. Eng. Adv. Res. Technol. 2018, 4, 1–4. [Google Scholar]
- Topçu, S.; Incecik, S.; Atimtay, A.T. Chemical composition of rainwater at EMEP station in Ankara, Turkey. Atmos. Res. 2002, 65, 77–92. [Google Scholar] [CrossRef]
- Başak, B.; Alagha, O. The chemical composition of rainwater over Büyükçekmece Lake, Istanbul. Atmos. Res. 2004, 71, 275–288. [Google Scholar] [CrossRef]
- AL-Adili, A.; Sadeq, J. Hydrochemical study of rain water in Baghdad city, Iraq. Eng. Tech. 2008, 26, 408. [Google Scholar]
- Banks, D. A Hydrogeological Atlas of Faryab Province, Northern Afghanistan. 2014. Available online: https://eprints.gla.ac.uk/107735/ (accessed on 11 July 2022).
- Al-Momani, I. Trace elements in atmospheric precipitation at Northern Jordan measured by ICP-MS: Acidity and possible sources. Atmos. Environ. 2003, 37, 4507–4515. [Google Scholar] [CrossRef]
- Kulshrestha, U.; Sarkar, A.; Srivastava, S.; Parashar, D. Investigation into atmospheric deposition through precipitation studies at New Delhi (India). Atmos. Environ. 1996, 30, 4149–4154. [Google Scholar] [CrossRef]
- Arsene, C.; Olariu, R.I.; Mihalopoulos, N. Chemical composition of rainwater in the northeastern Romania, Iasi region (2003–2006). Atmos. Environ. 2007, 41, 9452–9467. [Google Scholar] [CrossRef]
- Rao, P.; Tiwari, S.; Matwale, J.; Pervez, S.; Tunved, P.; Safai, P.; Srivastava, A.; Bisht, D.; Singh, S.; Hopke, P. Sources of chemical species in rainwater during monsoon and non-monsoonal periods over two mega cities in India and dominant source region of secondary aerosols. Atmos. Environ. 2016, 146, 90–99. [Google Scholar] [CrossRef]
- Kulshrestha, U.; Kulshrestha, M.J.; Sekar, R.; Sastry, G.; Vairamani, M. Chemical characteristics of rainwater at an urban site of south-central India. Atmos. Environ. 2003, 37, 3019–3026. [Google Scholar] [CrossRef]
- Chen, H.-Y.; Hsu, L.-F.; Huang, S.-Z.; Zheng, L. Assessment of the Components and Sources of Acid Deposition in Northeast Asia: A Case Study of the Coastal and Metropolitan Cities in Northern Taiwan. Atmosphere 2020, 11, 983. [Google Scholar] [CrossRef]
- Bisht, D.S.; Srivastava, A.; Joshi, H.; Ram, K.; Singh, N.; Naja, M.; Tiwari, S. Chemical characterization of rainwater at a high-altitude site “Nainital” in the central Himalayas, India. Environ. Sci. Pollut. Res. 2016, 24, 3959–3969. [Google Scholar] [CrossRef]
- Anatolaki, C.; Tsitouridou, R. Relationship between acidity and ionic composition of wet precipitation: A two years study at an urban site, Thessaloniki, Greece. Atmos. Res. 2009, 92, 100–113. [Google Scholar] [CrossRef]
- Jolliffe, I.T.; Cadima, J. Principal component analysis: A review and recent developments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150202. [Google Scholar] [CrossRef] [PubMed]
- R Core Team R: A Language and Environment for Statistical Computing. 2018. Available online: https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing (accessed on 11 July 2022).
- Xu, Z.; Wu, Y.; Liu, W.-J.; Liang, C.-S.; Ji, J.; Zhao, T.; Zhang, X. Chemical composition of rainwater and the acid neutralizing effect at Beijing and Chizhou city, China. Atmos. Res. 2015, 164–165, 278–285. [Google Scholar] [CrossRef]
- Huang, K.; Zhuang, G.; Xu, C.; Wang, Y.; Tang, A. The chemistry of the severe acidic precipitation in Shanghai, China. Atmos. Res. 2008, 89, 149–160. [Google Scholar] [CrossRef]
- Tiwari, S.; Hopke, P.K.; Thimmaiah, D.; Dumka, U.C.; Srivastava, A.K.; Bisht, D.S.; Rao, P.S.; Chate, D.M.; Srivastava, M.K.; Tripathi, S.N. Nature and Sources of Ionic Species in Precipitation across the Indo-Gangetic Plains, India. Aerosol Air Qual. Res. 2016, 16, 943–957. [Google Scholar] [CrossRef]
- Sahin, M.; Incecik, S.; Topcu, S.; Yildirim, A. Analysis of Atmospheric Conditions during Air Pollution Episodes in Ankara, Turkey. J. Air Waste Manag. Assoc. 2001, 51, 972–982. [Google Scholar] [CrossRef]
- Ali-Mohamed, A.Y. Estimation of inorganic particulate matter in the atmosphere of Isa Town, Bahrain, by dry deposition. Atmos. Environ. Part. B Urban. Atmos. 1991, 25, 397–405. [Google Scholar] [CrossRef]
- Ali-Mohamed, A. Estimation of inorganic particulate matter in atmospheres of villages in Bahrain, by dry fall. Atmos. Environ. 1995, 29, 1519–1529. [Google Scholar] [CrossRef]
- Madany, I.M.; Akhter, M.S.; Al Jowder, O. The correlations between heavy metals in residential indoor dust and outdoor street dust in Bahrain. Environ. Int. 1994, 20, 483–492. [Google Scholar] [CrossRef]
- Mohammadzadeh, H.; Heydarizad, M. A conceptual model for water resources circulation patterns in Andarokh-Kardeh region (NE, Iran). Geochemistry 2020, 80, 125593. [Google Scholar] [CrossRef]
- Ayati, F.; Yavuz, F.; Noghreyan, M.; Haroni, H.A.; Yavuz, R. Chemical characteristics and composition of hydrothermal biotite from the Dalli porphyry copper prospect, Arak, central province of Iran. Miner. Pet. 2008, 94, 107–122. [Google Scholar] [CrossRef]
- Anil, I.; Alagha, O.; Blaisi, N.I.; Mohamed, I.A.; Barghouthi, M.H.; Manzar, M.S. Source Identification of Episodic Rain Pollutants by New Approach: Combining Satellite Observations and Backward Air Mass Trajectories. Aerosol Air Qual. Res. 2019, 9, 2827–2843. [Google Scholar] [CrossRef]
- Négrel, P.; Roy, S. Chemistry of rainwater in the Massif Central (France): A strontium isotope and major element study. Appl. Geochem. 1998, 13, 941–952. [Google Scholar] [CrossRef]
- Kattan, Z. Factors affecting the chemical composition of precipitation in Syria. Environ. Sci. Pollut. Res. 2020, 27, 28408–28428. [Google Scholar] [CrossRef]
- Lu, X.; Li, L.Y.; Li, N.; Yang, G.; Luo, D.; Chen, J. Chemical characteristics of spring rainwater of Xi’an city, NW China. Atmos. Environ. 2011, 45, 5058–5063. [Google Scholar] [CrossRef]
- Wang, Y.; Zhuang, G.; Zhang, X.; Huang, K.; Xu, C.; Tang, A.; Chen, J.; An, Z. The ion chemistry, seasonal cycle, and sources of PM2.5 and TSP aerosol in Shanghai. Atmos. Environ. 2006, 40, 2935–2952. [Google Scholar] [CrossRef]
- Vet, R.; Artz, R.S.; Carou, S.; Shaw, M.; Ro, C.-U.; Aas, W.; Baker, A.; Bowersox, V.C.; Dentener, F.; Galy-Lacaux, C.; et al. A global assessment of precipitation chemistry and deposition of sulfur, nitrogen, sea salt, base cations, organic acids, acidity and pH, and phosphorus. Atmos. Environ. 2014, 93, 3–100. [Google Scholar] [CrossRef]
- Tuncer, B.; Bayar, B.; Yeşilyurt, C.; Tuncel, G. Ionic composition of precipitation at the Central Anatolia (Turkey). Atmos. Environ. 2001, 35, 5989–6002. [Google Scholar] [CrossRef]
- Yatkin, S.; Adali, M.; Bayram, A. A study on the precipitation in Izmir, Turkey: Chemical composition and source apportionment by receptor models. J. Atmos. Chem. 2015, 73, 241–259. [Google Scholar] [CrossRef]
- Szép, R.; Bodor, Z.; Miklóssy, I.; Nita, I.-A.; Oprea, O.A.; Keresztesi, A. Influence of peat fires on the rainwater chemistry in intra-mountain basins with specific atmospheric circulations (Eastern Carpathians, Romania). Sci. Total Environ. 2018, 647, 275–289. [Google Scholar] [CrossRef]
- Xiao, J. Chemical composition and source identification of rainwater constituents at an urban site in Xi’an. Environ. Earth Sci. 2016, 75, 209. [Google Scholar] [CrossRef]
- Wu, Y.; Xu, Z.; Liu, W.; Zhao, T.; Zhang, X.; Jiang, H.; Yu, C.; Zhou, L.; Zhou, X. Chemical compositions of precipitation at three non-urban sites of Hebei Province, North China: Influence of terrestrial sources on ionic composition. Atmos. Res. 2016, 181, 115–123. [Google Scholar] [CrossRef]
- Balasubramanian, R.; Victor, T.; Chun, N. Chemical and Statistical Analysis of Precipitation in Singapore. Water Air Soil Pollut. 2001, 130, 451–456. [Google Scholar] [CrossRef]
- Seinfeld, J. Atmospheric Chemistry and Physics of Air Pollution, 1st ed.; Wiley-Interscience: Cambridge, UK, 1986; Available online: https://pubs.acs.org/doi/pdf/10.1021/es00151a602 (accessed on 11 July 2022).
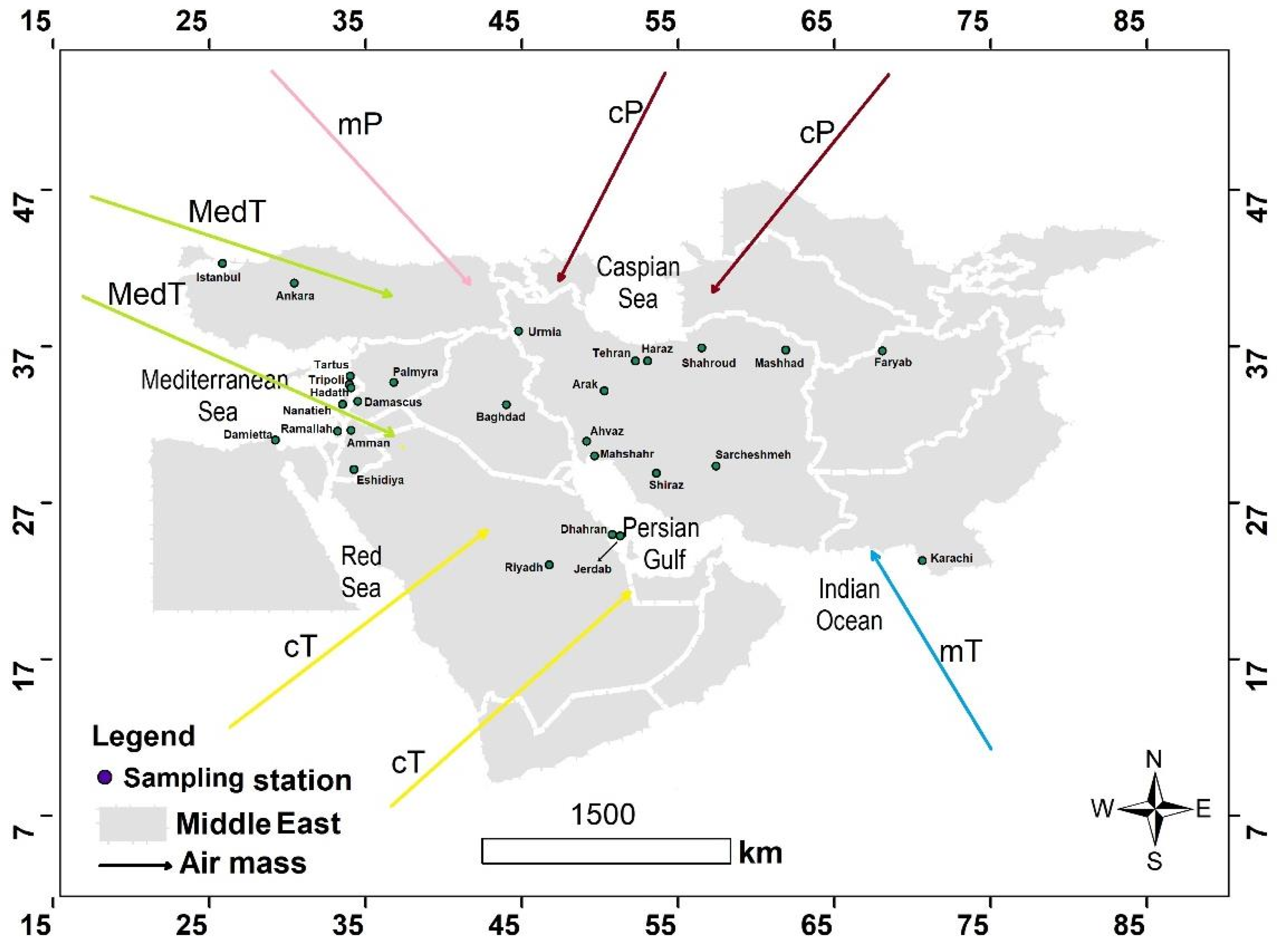
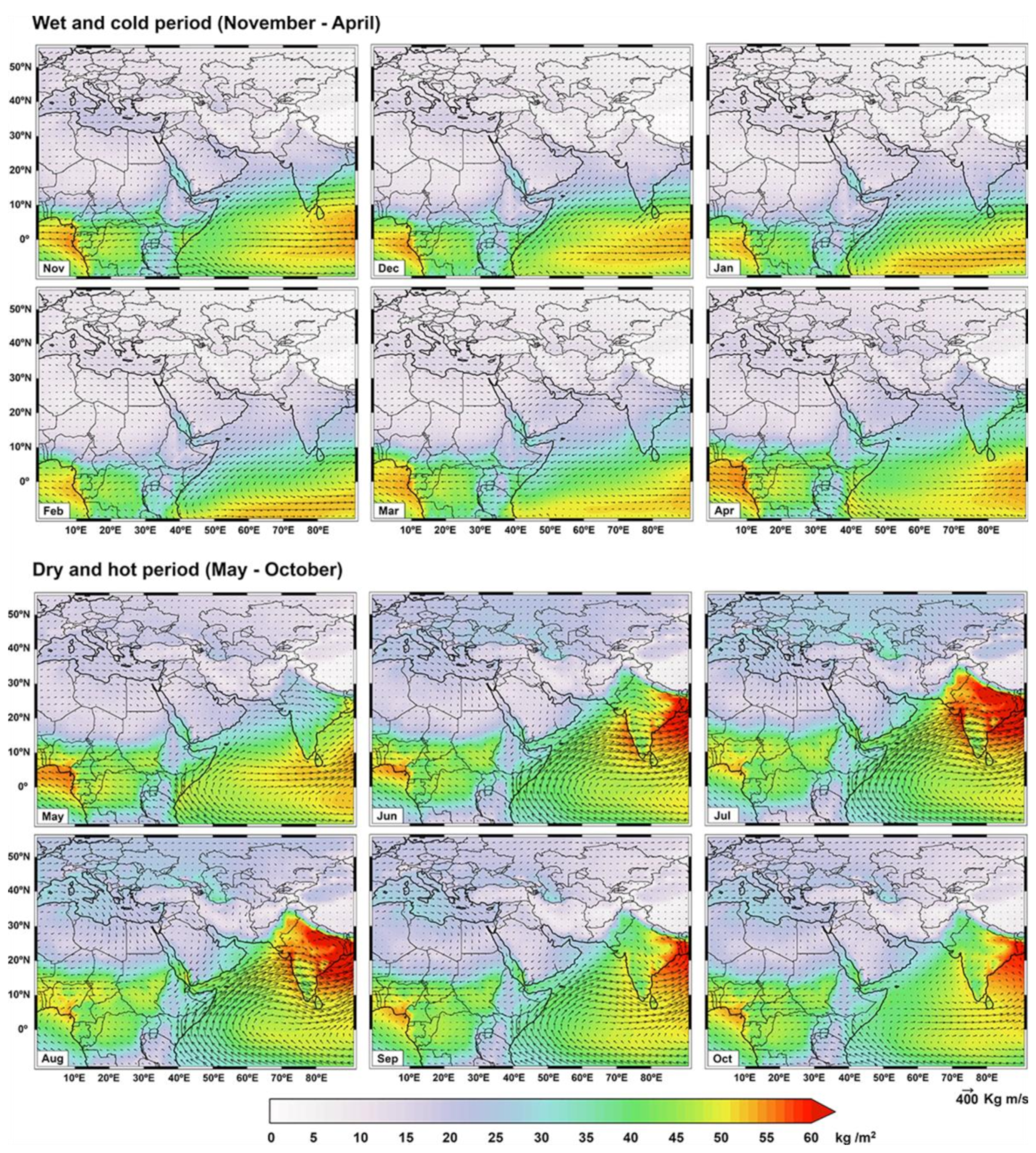
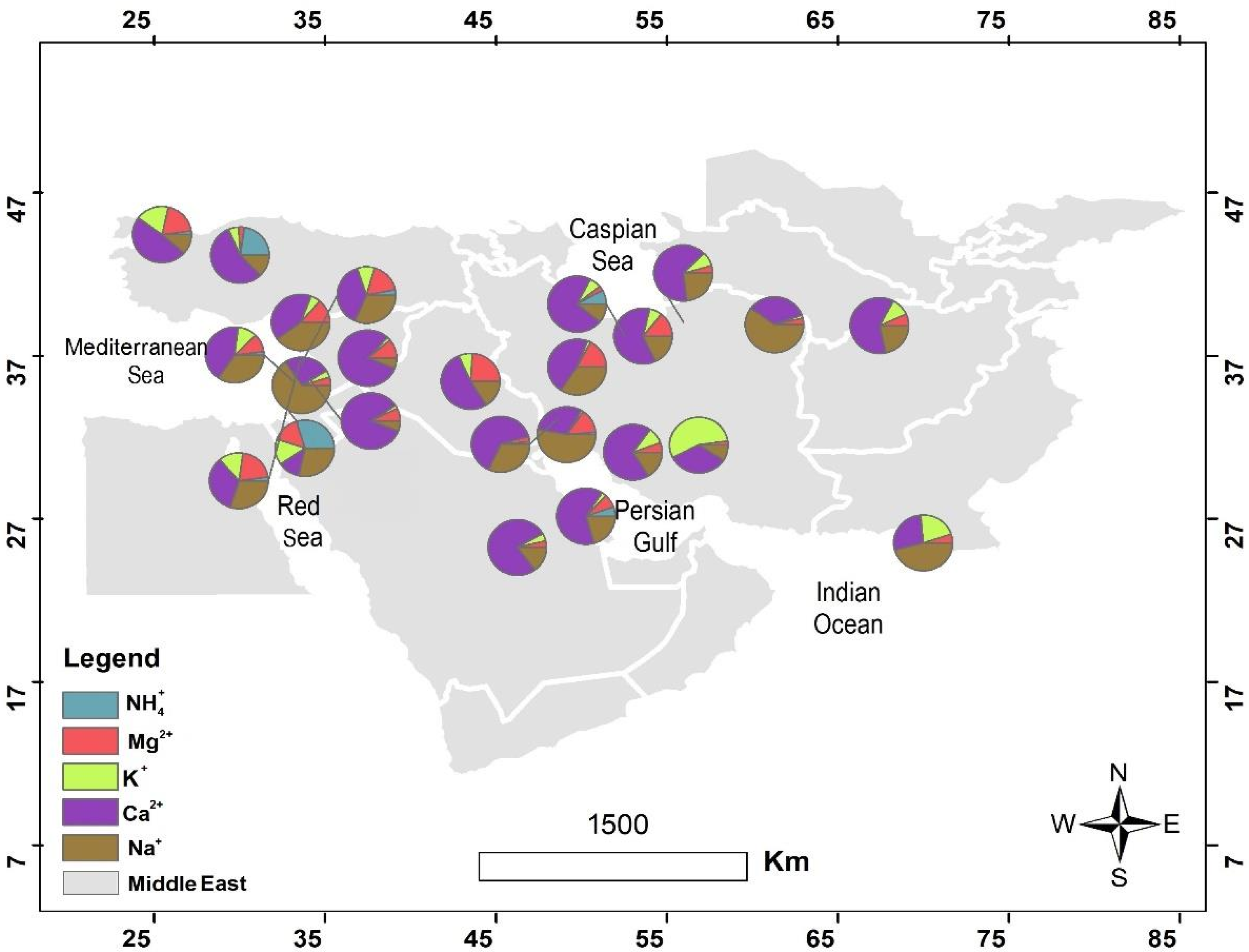

| Row | Station | Sea Salt Fraction (SSF) | Non Sea Salt Fraction (NSSF) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca2+ | K+ | Mg2+ | SO42− | Cl− | HCO3− | Ca2+ | K+ | Mg2+ | SO42− | Cl− | HCO3− | ||
| Continental stations | |||||||||||||
| 1 | Shiraz | 0.11 | 0.11 | 0.35 | 0.75 | 5.25 | 0.03 | 12.49 | 1.59 | 0.66 | 13.45 | −0.15 | 0.33 |
| 2 | Ahvaz | 0.32 | 0.31 | 0.99 | 2.09 | 14.68 | 0.08 | 15.83 | −0.31 | −0.21 | 33.02 | 0.21 | 40.62 |
| 3 | Shahroud | 0.31 | 0.3 | 0.95 | 2.01 | 14.11 | 0.08 | 21.59 | 2.5 | 0.45 | 20.99 | −6.11 | 44.12 |
| 4 | Haraz | 0.07 | 0.07 | 0.23 | 0.48 | 3.4 | 0.02 | 12.35 | 1.02 | 0.28 | 4.41 | −1.21 | 43.79 |
| 5 | Sarcheshmeh | 0.06 | 0.06 | 0.2 | 0.42 | 2.93 | 0.02 | 5.68 | 9.29 | 0.18 | 19.39 | 1.56 | 5.01 |
| 6 | Tehran | 0.11 | 0.11 | 0.34 | 0.72 | 5.08 | 0.03 | 9.39 | 1.01 | 1.81 | 11.6 | −2.61 | 23.02 |
| 7 | Mashhad | 0.94 | 0.91 | 2.92 | 6.15 | 43.3 | 0.25 | 12.82 | −0.21 | −1.46 | 19.87 | −14.98 | 28.97 |
| 8 | Arak | 0.75 | 0.73 | 2.33 | 4.92 | 34.6 | 0.2 | 24.97 | 0.29 | 7.35 | 38.19 | −30.17 | 62.27 |
| 9 | Urmia | _ | _ | _ | _ | _ | _ | _ | _ | _ | 2.34 | 4.04 | _ |
| 10 | Riyadh | 0.24 | 0.23 | 0.74 | 1.57 | 11.04 | 0.06 | 31.26 | 1.57 | 0.76 | 15.73 | −1.54 | −0.06 |
| 11 | Eshidiya | 0.15 | 0.15 | 0.47 | 0.99 | 6.96 | 0.04 | 1.47 | 1.81 | 1.53 | 8.24 | −1.13 | 4.27 |
| 12 | Ankara | 0.48 | 0.46 | 1.48 | 3.13 | 22.04 | 0.13 | 52.42 | 4.97 | 1.43 | 116.97 | 29.72 | 146.27 |
| 13 | Faryab | 1.09 | 0.55 | 0.57 | 0.31 | 2.9 | _ | 6.99 | 1.15 | 0.23 | 5.19 | −0.10 | _ |
| 14 | Hadath | 0.29 | 0.28 | 0.89 | 1.87 | 13.17 | 0.08 | 8.58 | 1.87 | 3.21 | −0.79 | 6.71 | 34.09 |
| 15 | Nabatieh | 0.28 | 0.27 | 0.87 | 1.84 | 12.96 | 0.08 | 8.56 | 1.9 | 1.29 | 0.43 | 6.15 | 28.7 |
| 16 | Palmyra | 0.08 | 0.08 | 0.26 | 0.56 | 3.91 | 0.02 | 29.25 | 0.75 | 3.74 | 13.77 | 1.42 | 77.31 |
| 17 | Damascus | 0.06 | 0.06 | 0.2 | 0.42 | 2.93 | 0.02 | 22.94 | 0.44 | 1.8 | 19.33 | 1.57 | 45.73 |
| 18 | Amman | 0.56 | 0.54 | 1.74 | 3.66 | 25.79 | 0.15 | 5.07 | 0.33 | −0.75 | 1.53 | −20.06 | 10.95 |
| 19 | Baghdad | 2.5 | 1.24 | 1.29 | 0.71 | 6.61 | _ | 17.95 | 2.57 | 7.11 | 51.9 | 19.59 | _ |
| 20 | Ramallah | 0.43 | 0.42 | 1.34 | 2.82 | 19.85 | 0.11 | −0.43 | 4.56 | −1.34 | −2.82 | −19.85 | −0.11 |
| 21 | Jerdab | _ | _ | _ | _ | _ | _ | _ | _ | _ | _ | _ | _ |
| 22 | Tartus | 0.32 | 0.31 | 0.99 | 2.08 | 14.66 | 0.08 | 8.08 | 0.79 | 1.81 | 3.12 | −0.66 | 24.12 |
| 23 | Dhahran | 0.12 | 0.11 | 0.36 | 0.77 | 5.41 | 0.03 | 9.15 | 0.28 | 0.73 | 10.4 | −0.33 | 4.45 |
| 24 | Istanbul | 0.07 | 0.06 | 0.21 | 0.43 | 3.06 | 0.02 | 7.11 | 2.65 | 2.73 | 8.97 | 3.88 | −0.02 |
| 25 | Tripoli | 0.26 | 0.26 | 0.82 | 1.74 | 12.21 | 0.07 | 7.45 | 2.8 | 3.85 | 1.19 | 2.79 | 19.93 |
| 26 | Damietta | _ | _ | _ | _ | _ | _ | _ | _ | _ | 143.54 | 37.13 | _ |
| 27 | Karachi | 0.44 | 0.42 | 1.36 | 2.86 | 20.14 | 0.12 | 6.23 | 4.74 | −0.18 | 16.21 | −11.56 | 9.89 |
| 28 | Mahshahr | 0.92 | 0.89 | 2.85 | 6.01 | 42.27 | 0.24 | 12.61 | −0.30 | 3.27 | 52.15 | −23.95 | 33.83 |
| Row | Station | Ca2+ | K+ | Mg2+ | SO42− | Cl− | HCO3− |
|---|---|---|---|---|---|---|---|
| 1 | Shiraz | 110.73 | 15.43 | 2.86 | 19.04 | 0.97 | 11.85 |
| 2 | Ahvaz | 50.75 | _ | 0.79 | 16.84 | 1.01 | 479.04 |
| 3 | Shahroud | 71.56 | 9.45 | 1.47 | 11.47 | 0.57 | 540.91 |
| 4 | Haraz | 168.37 | 15.26 | 2.23 | 10.12 | 0.64 | 2224.4 |
| 5 | Sarcheshmeh | 90.3 | 151.88 | 1.91 | 47.55 | 1.53 | 296.38 |
| 6 | Tehran | 86.16 | 10.49 | 6.28 | 17.05 | 0.49 | 783 |
| 7 | Mashhad | 14.65 | 0.77 | 0.5 | 4.23 | 0.65 | 116.56 |
| 8 | Arak | 34.28 | 1.4 | 4.15 | 8.77 | 0.13 | 311.87 |
| 9 | Urmia | _ | _ | _ | _ | _ | _ |
| 10 | Riyadh | 131.61 | 7.77 | 2.02 | 11.03 | 0.86 | _ |
| 11 | Eshidiya | 10.75 | 13.37 | 4.26 | 9.33 | 0.84 | 106.84 |
| 12 | Ankara | 110.69 | 11.73 | 1.96 | 38.35 | 2.35 | 1147.34 |
| 13 | Faryab | 64.69 | 22.01 | 1.41 | 17.6 | 0.96 | _ |
| 14 | Hadath | 31.05 | 7.77 | 4.62 | 0.58 | 1.51 | 448.03 |
| 15 | Nabatieh | 31.47 | 7.98 | 2.47 | 1.23 | 1.47 | 383.68 |
| 16 | Palmyra | 346.06 | 10.11 | 15.19 | 25.8 | 1.36 | 3417.36 |
| 17 | Damascus | 361.83 | 8.12 | 10.13 | 47.41 | 1.54 | 2695.71 |
| 18 | Amman | 10.07 | 1.61 | 0.57 | 1.42 | 0.22 | 74.35 |
| 19 | Baghdad | 72.73 | 21.72 | 6.49 | 72.84 | 3.96 | _ |
| 20 | Ramallah | _ | 11.95 | _ | _ | _ | _ |
| Coastal stations | |||||||
| 21 | Jerdab | _ | _ | _ | _ | _ | _ |
| 22 | Tartus | 26.43 | 3.57 | 2.84 | 2.5 | 0.96 | 285.19 |
| 23 | Dhahran | 79.01 | 3.43 | 2.99 | 14.53 | 0.94 | 143.02 |
| 24 | Istanbul | 108.28 | 42.2 | 14.27 | 21.63 | 2.27 | _ |
| 25 | Tripoli | 29.11 | 11.93 | 5.68 | 1.69 | 1.23 | 282.83 |
| 26 | Damietta | _ | _ | _ | _ | _ | _ |
| 27 | Karachi | 15.27 | 12.2 | 0.87 | 6.66 | 0.43 | 85.85 |
| 28 | Mahshahr | 14.76 | 0.66 | 2.15 | 9.68 | 0.43 | 139.22 |
| Row | Station | Ca2+ | K+ | Mg2+ |
|---|---|---|---|---|
| Continental stations | ||||
| 1 | Shiraz | _ | _ | _ |
| 2 | Ahvaz | _ | _ | _ |
| 3 | Shahroud | _ | _ | _ |
| 4 | Haraz | 0.013 | 0.042 | 0.013 |
| 5 | Sarcheshmeh | 0.045 | 0.152 | 0.045 |
| 6 | Tehran | 0.016 | 0.048 | 0.016 |
| 7 | Mashhad | 0.952 | 0.047 | 1.61 |
| 8 | Arak | 0.665 | 0.025 | 1.115 |
| 9 | Urmia | _ | _ | _ |
| 10 | Riyadh | 0.153 | 0.471 | 0.146 |
| 11 | Eshidiya | 0.084 | 0.263 | 0.084 |
| 12 | Ankara | 0.003 | 0.011 | 0.003 |
| 13 | Faryab | 0.286 | 0.15 | 0.144 |
| 14 | Hadath | 0.061 | 0.187 | 0.059 |
| 15 | Nabatieh | 0.036 | 0.111 | 0.034 |
| 16 | Palmyra | 0.007 | 0.024 | 0.007 |
| 17 | Damascus | 0.007 | 0.023 | 0.007 |
| 18 | Amman | 0.087 | 0.269 | 0.084 |
| 19 | Baghdad | 3.521 | 1.817 | 1.746 |
| 20 | Ramallah | 0.02 | 0.062 | 0.019 |
| Coastal stations | ||||
| 21 | Jerdab | _ | _ | _ |
| 22 | Tartus | 0.032 | 0.098 | 0.031 |
| 23 | Dhahran | 0.012 | 0.037 | 0.011 |
| 24 | Istanbul | 0.163 | 0.488 | 0.14 |
| 25 | Tripoli | 0.059 | 0.185 | 0.059 |
| 26 | Damietta | _ | _ | _ |
| 27 | Karachi | 0.154 | 0.476 | 0.147 |
| 28 | Mahshahr | 0.089 | 0.276 | 0.086 |
| Element | Component | |
|---|---|---|
| 1 | 2 | |
| Ca2+ | 0.85 | −0.04 |
| Mg2+ | 0.34 | −0.61 |
| Na+ | 0.43 | −0.57 |
| K+ | 0.23 | 0.72 |
| SO42− | 0.94 | 0.03 |
| HCO3− | 0.88 | −0.06 |
| Cl− | 0.82 | −0.01 |
| pH | −0.21 | −0.81 |
| EC | 0.96 | 0.1 |
| TDS | 0.87 | 0.084 |
| Method | DNN | ANN | Decision Tree | Gradient Boosting (Gboost) | |
|---|---|---|---|---|---|
| Train set | R2 | 0.49 | 0.59 | 0.64 | 0.71 |
| Test set | R2 | 0.45 | 0.52 | 0.51 | 0.67 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heydarizad, M.; Gimeno, L.; Amiri, S.; Minaei, M.; Mohammadabadi, H.G. A Comprehensive Overview of the Hydrochemical Characteristics of Precipitation across the Middle East. Water 2022, 14, 2657. https://doi.org/10.3390/w14172657
Heydarizad M, Gimeno L, Amiri S, Minaei M, Mohammadabadi HG. A Comprehensive Overview of the Hydrochemical Characteristics of Precipitation across the Middle East. Water. 2022; 14(17):2657. https://doi.org/10.3390/w14172657
Chicago/Turabian StyleHeydarizad, Mojtaba, Luis Gimeno, Somayeh Amiri, Masoud Minaei, and Hamid Ghalibaf Mohammadabadi. 2022. "A Comprehensive Overview of the Hydrochemical Characteristics of Precipitation across the Middle East" Water 14, no. 17: 2657. https://doi.org/10.3390/w14172657
APA StyleHeydarizad, M., Gimeno, L., Amiri, S., Minaei, M., & Mohammadabadi, H. G. (2022). A Comprehensive Overview of the Hydrochemical Characteristics of Precipitation across the Middle East. Water, 14(17), 2657. https://doi.org/10.3390/w14172657








