Figure 1.
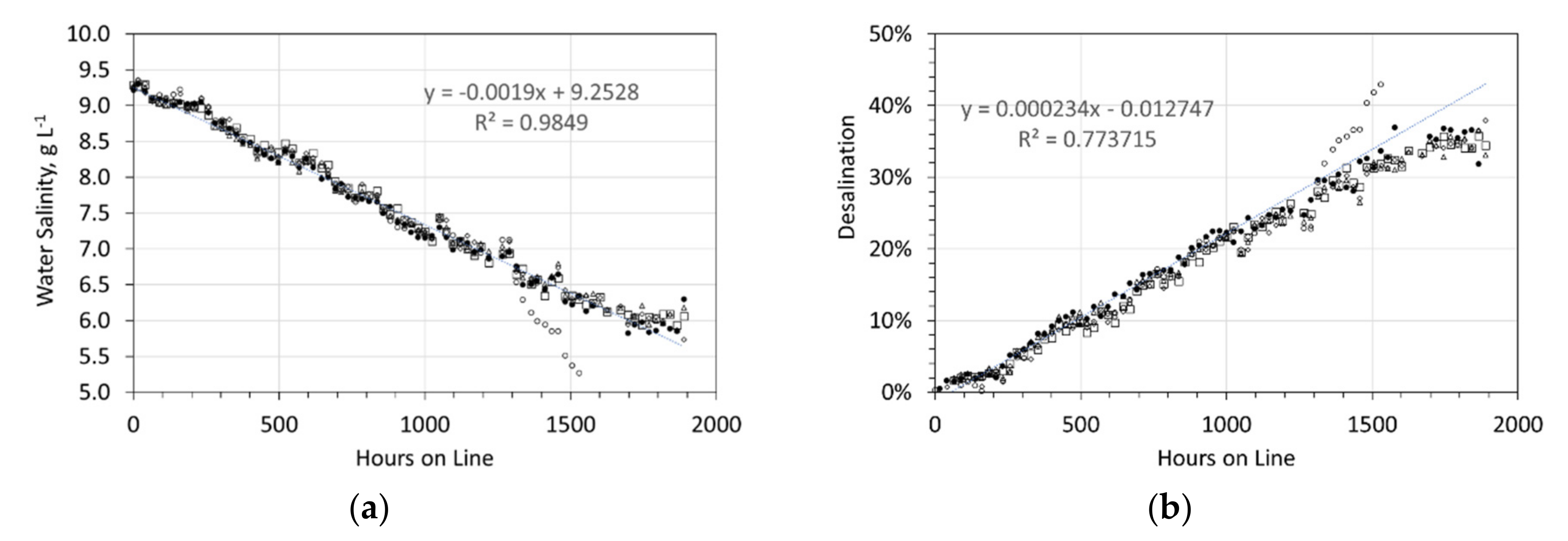
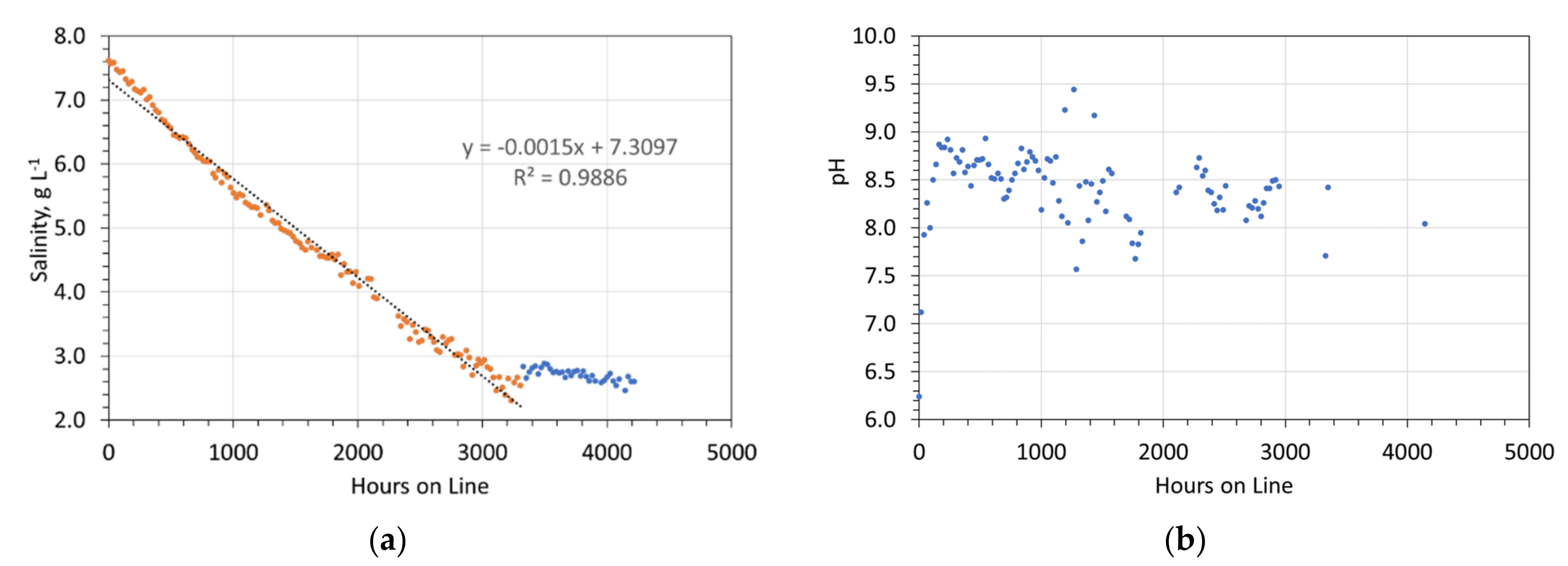
Desalination associated with the ZVI Pellets. Feed water salinity = 9.24 g NaCl L−1. Five 1 L, static water bodies, 8 cm water depth, with an air-water contact. Each water body, contained 5 × 15 mm O.D. Cu0 pellets produced using 8.8 g NaCl L−1 in the ZVI Reactor. The pellets contained about 25 g L−1 of ZVI Pellet Material + about 25 g L−1 of Cu0 sheathing. (a) Salinity vs. hours on line; (b) Desalination vs. hours on line.
Figure 1.
Desalination associated with the ZVI Pellets. Feed water salinity = 9.24 g NaCl L−1. Five 1 L, static water bodies, 8 cm water depth, with an air-water contact. Each water body, contained 5 × 15 mm O.D. Cu0 pellets produced using 8.8 g NaCl L−1 in the ZVI Reactor. The pellets contained about 25 g L−1 of ZVI Pellet Material + about 25 g L−1 of Cu0 sheathing. (a) Salinity vs. hours on line; (b) Desalination vs. hours on line.
Figure 2.
Conceptual Process Design Reactor Train Framework used to construct the desalination pellets.
Figure 2.
Conceptual Process Design Reactor Train Framework used to construct the desalination pellets.
Figure 3.
Process flow diagram for the manufacture of ZVI desalination pellets: P = pressure; F = flow rate. Black flow lines are gas. Blue flow lines are recycled water, which was abstracted from the product gas. The Halite Reactor is a fixed bed/packed bed reactor, containing granular halite, which can be supplemented with ZVI. The ZVI Reactor is an aqueous bubble column reactor, containing a static ZVI bed located below the gas distributor.
Figure 3.
Process flow diagram for the manufacture of ZVI desalination pellets: P = pressure; F = flow rate. Black flow lines are gas. Blue flow lines are recycled water, which was abstracted from the product gas. The Halite Reactor is a fixed bed/packed bed reactor, containing granular halite, which can be supplemented with ZVI. The ZVI Reactor is an aqueous bubble column reactor, containing a static ZVI bed located below the gas distributor.
Figure 4.
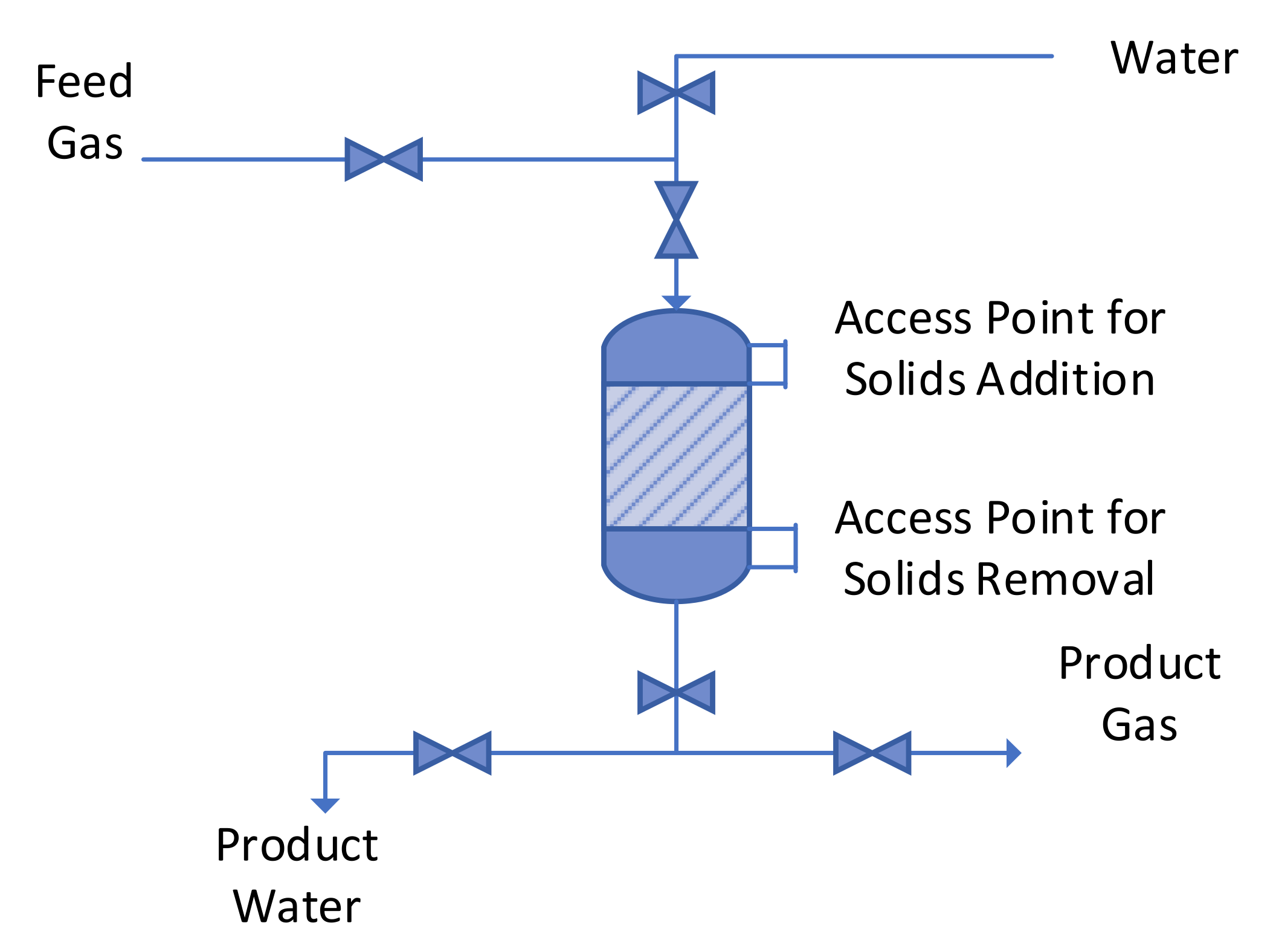
Halite Reactor showing access points for solids addition and removal, and access points for water used to wash and clean the reactor.
Figure 4.
Halite Reactor showing access points for solids addition and removal, and access points for water used to wash and clean the reactor.
Figure 5.
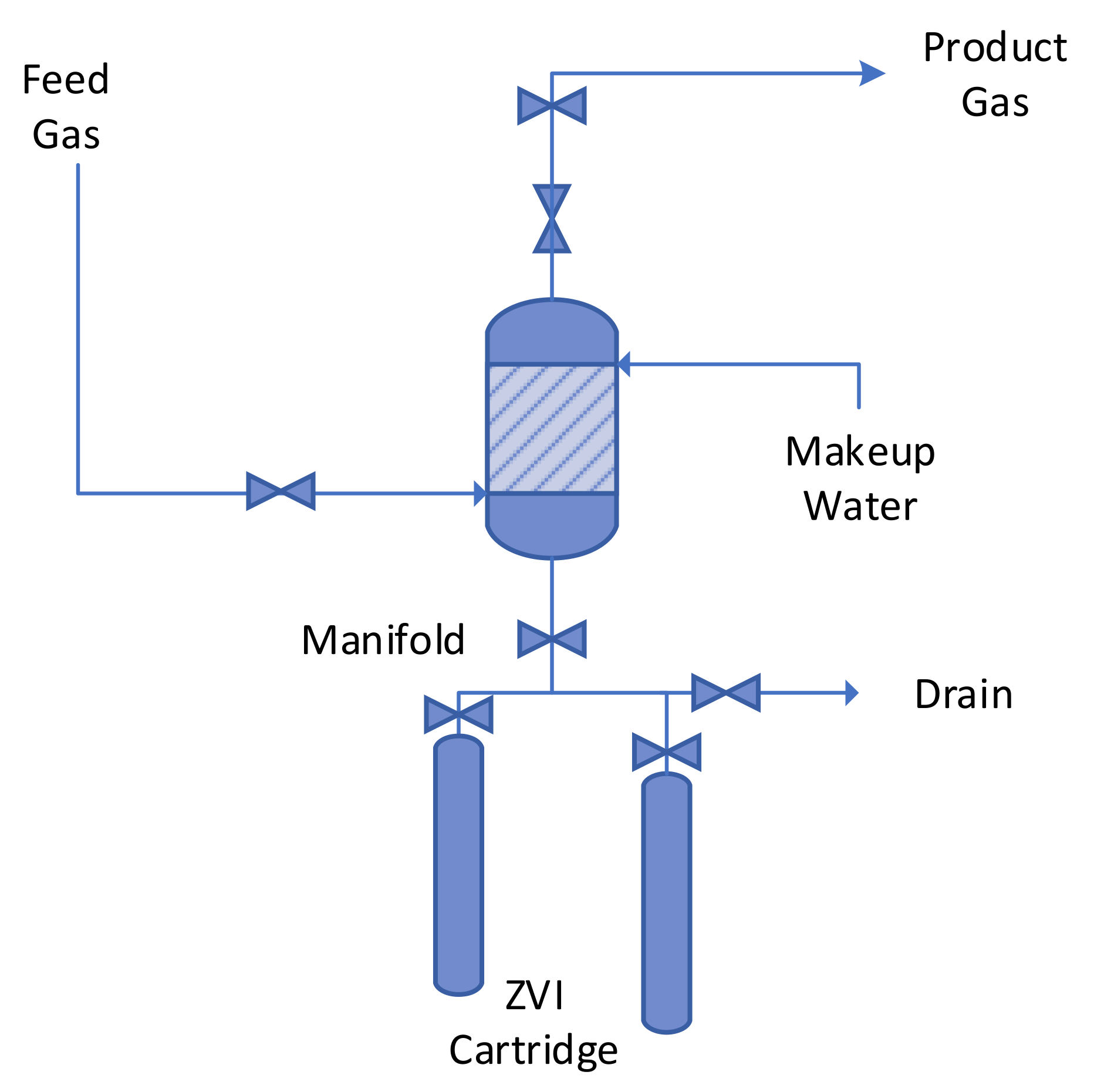
ZVI Reactor Process Flow Structure.
Figure 5.
ZVI Reactor Process Flow Structure.
Figure 6.
Example set of desalination pellets.
Figure 6.
Example set of desalination pellets.
Figure 7.
Detail of an iron fibre-coated with a [n-Fe(b)]p@[n-urea]a polymer catalyst. (a) transmitted light; (b) false colour. Field of view 0.4 mm.
Figure 7.
Detail of an iron fibre-coated with a [n-Fe(b)]p@[n-urea]a polymer catalyst. (a) transmitted light; (b) false colour. Field of view 0.4 mm.
Figure 8.
Photomicrograph of a freshly manufactured pellet. Polished surface, reflected light. Field of view = 0.1 mm. White areas are polished Fe0 metal. Brown/black areas show porosity coated in n-Fe-polymer@n-C0 polymer. The cross-sectional porosity is about 80%.
Figure 8.
Photomicrograph of a freshly manufactured pellet. Polished surface, reflected light. Field of view = 0.1 mm. White areas are polished Fe0 metal. Brown/black areas show porosity coated in n-Fe-polymer@n-C0 polymer. The cross-sectional porosity is about 80%.
Figure 9.
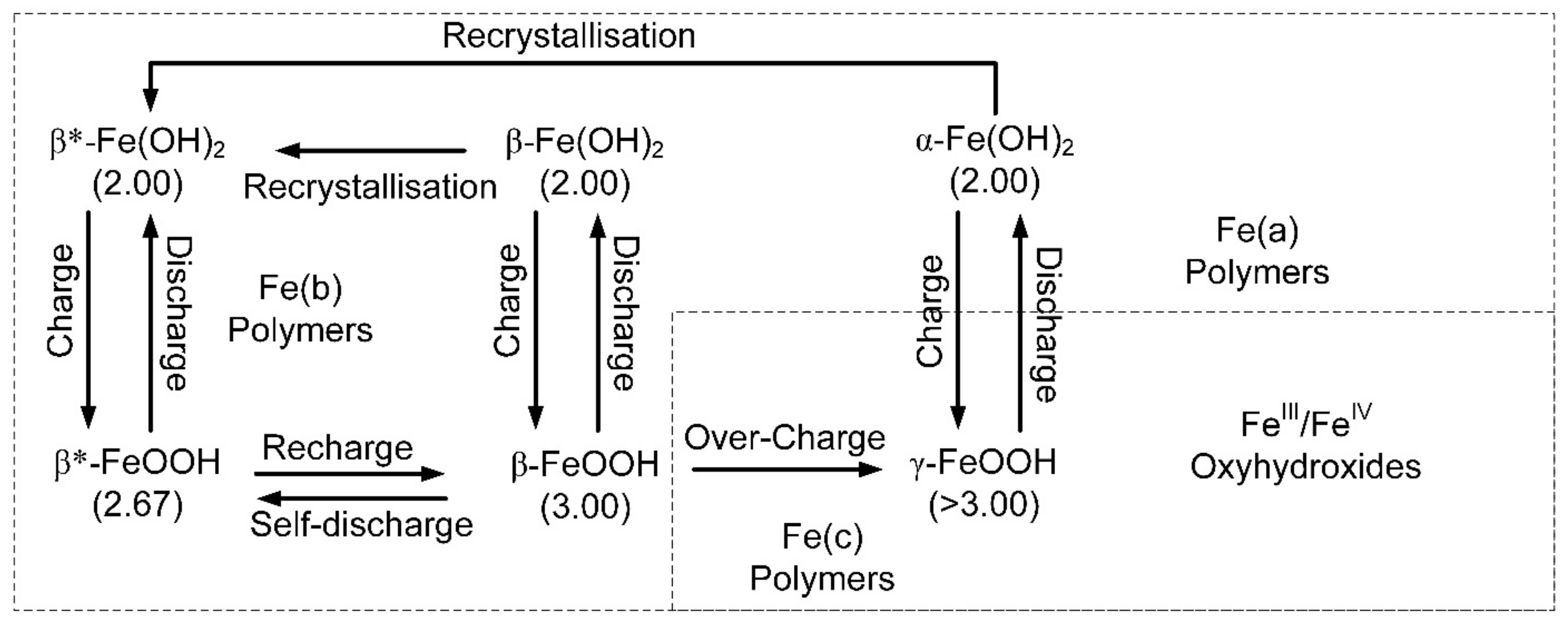
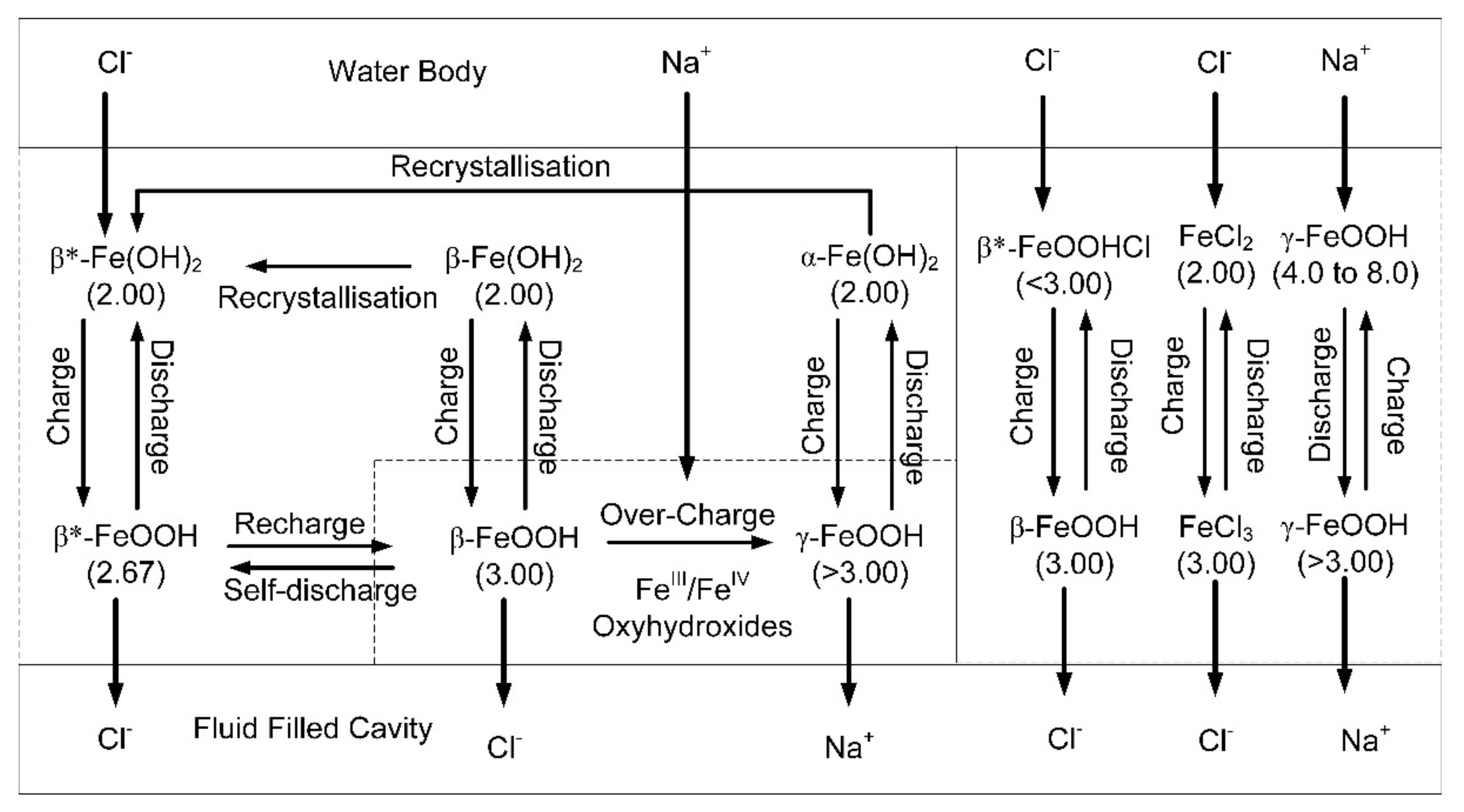
Simplified, electrochemical model associated with the Fe-polymers. Charge indicates directions, where the change is an increase in the oxidation number of the iron. The terminology and charge relationships shown are for a natural Hydrogen-Iron Battery. The β* notation indicates that the polymer has an oxidation number and ion charge, which can vary in response to changing environmental conditions. The electrochemical terminology used here follows the recommendations of Reference [
52].
Figure 9.
Simplified, electrochemical model associated with the Fe-polymers. Charge indicates directions, where the change is an increase in the oxidation number of the iron. The terminology and charge relationships shown are for a natural Hydrogen-Iron Battery. The β* notation indicates that the polymer has an oxidation number and ion charge, which can vary in response to changing environmental conditions. The electrochemical terminology used here follows the recommendations of Reference [
52].
Figure 10.
Simplified electrochemical model showing how the Fe-polymers transport Na+ and Cl− ions from the water body into dead-end pores. The β* notation indicates that the polymer has an oxidation number and ion charge, which can vary in response to changing environmental conditions.
Figure 10.
Simplified electrochemical model showing how the Fe-polymers transport Na+ and Cl− ions from the water body into dead-end pores. The β* notation indicates that the polymer has an oxidation number and ion charge, which can vary in response to changing environmental conditions.
Figure 11.
Example reflected light, polished cross section through a desalination pellet following operation. Dead-end porosity is infilled with NaCl (white). Field of view = 1 mm.
Figure 11.
Example reflected light, polished cross section through a desalination pellet following operation. Dead-end porosity is infilled with NaCl (white). Field of view = 1 mm.
Figure 12.
Example of desalination using a desalination pellet. Note a linear reduction in salinity with time (brown dots) followed by a period with no salinity change (blue dots). Reaction Details: 5 pellets containing 27.12 g desalination catalyst, produced using 40 g NaCl L−1 in the ZVI Reactor; Feed water salinity = 7.6 g NaCl L−1; Reactor size 1 L; temperature range during operation = 0 to 14 °C. (a) Salinity vs. Hours on line; (b) pH vs. Hours on line.
Figure 12.
Example of desalination using a desalination pellet. Note a linear reduction in salinity with time (brown dots) followed by a period with no salinity change (blue dots). Reaction Details: 5 pellets containing 27.12 g desalination catalyst, produced using 40 g NaCl L−1 in the ZVI Reactor; Feed water salinity = 7.6 g NaCl L−1; Reactor size 1 L; temperature range during operation = 0 to 14 °C. (a) Salinity vs. Hours on line; (b) pH vs. Hours on line.
Figure 13.
Example relationship for the expected desalination as a function of time. Determined for feed water with a salinity of 5 g L−1. Red line = 90% probability of a greater desalination. Blue line = mean outcome; black line = 10% probability of a greater desalination. Water containing 20–30 g ZVI treatment product L−1 is expected to cease desalinating after 1500–2000 h. Water containing 50–60 g ZVI treatment product L−1 is expected to cease desalinating after 2500–4000 h.
Figure 13.
Example relationship for the expected desalination as a function of time. Determined for feed water with a salinity of 5 g L−1. Red line = 90% probability of a greater desalination. Blue line = mean outcome; black line = 10% probability of a greater desalination. Water containing 20–30 g ZVI treatment product L−1 is expected to cease desalinating after 1500–2000 h. Water containing 50–60 g ZVI treatment product L−1 is expected to cease desalinating after 2500–4000 h.
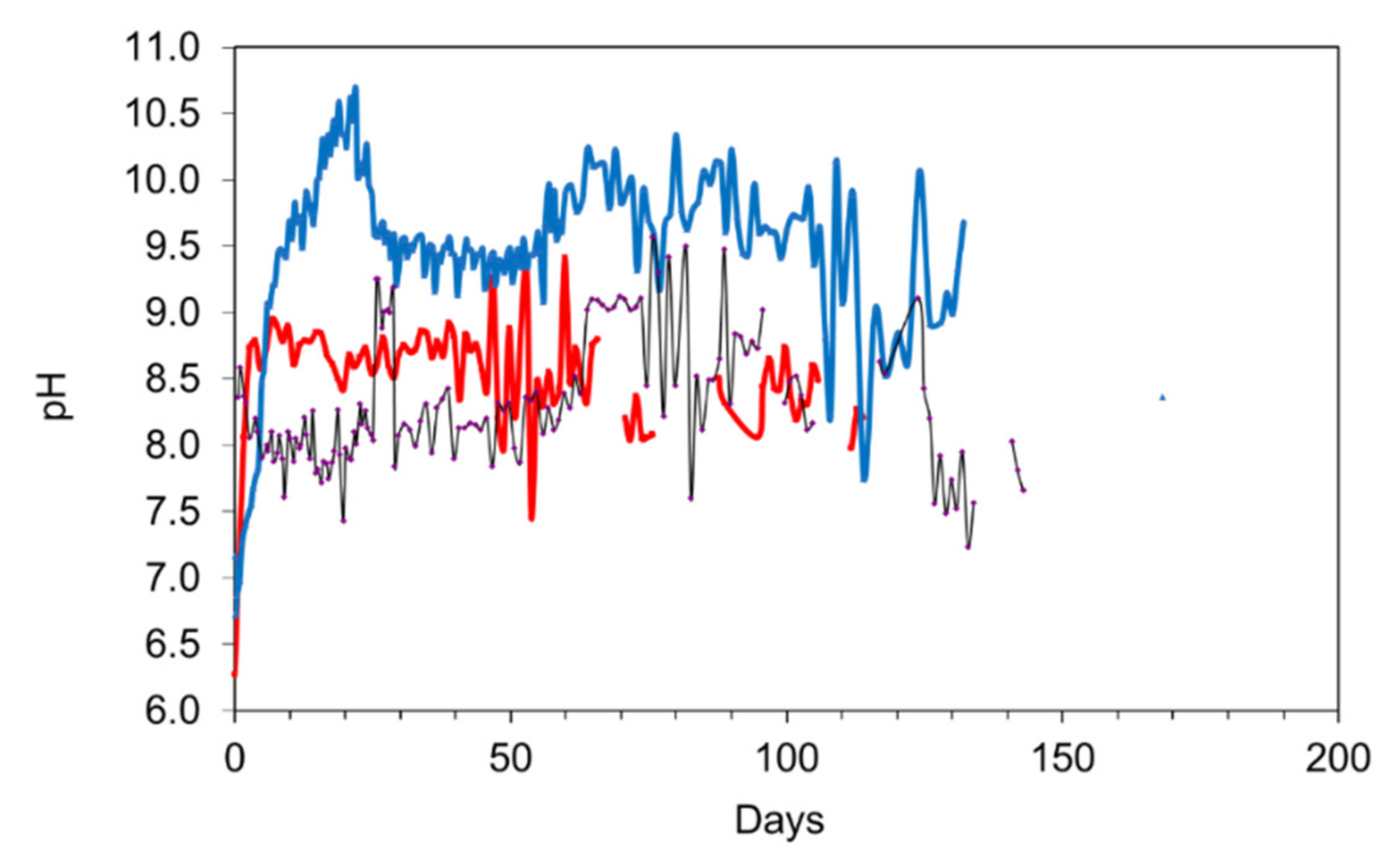
Figure 14.
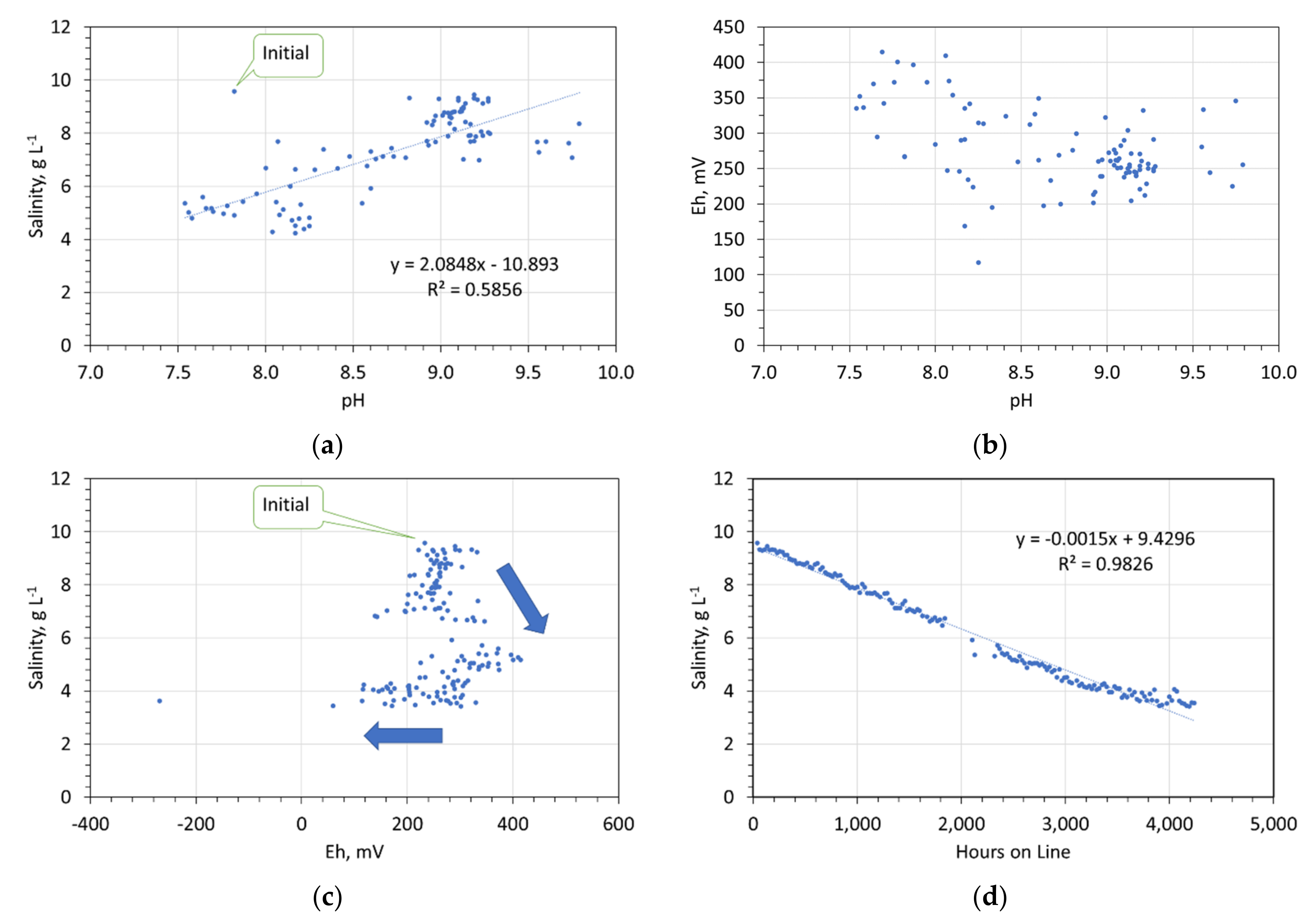
Redox controls on desalination. 47.5 g desalination pellet L−1. (a) Salinity vs. pH; (b) Eh vs. pH; (c) Salinity vs. Eh; (d) salinity vs. hours on line.
Figure 14.
Redox controls on desalination. 47.5 g desalination pellet L−1. (a) Salinity vs. pH; (b) Eh vs. pH; (c) Salinity vs. Eh; (d) salinity vs. hours on line.
Figure 15.
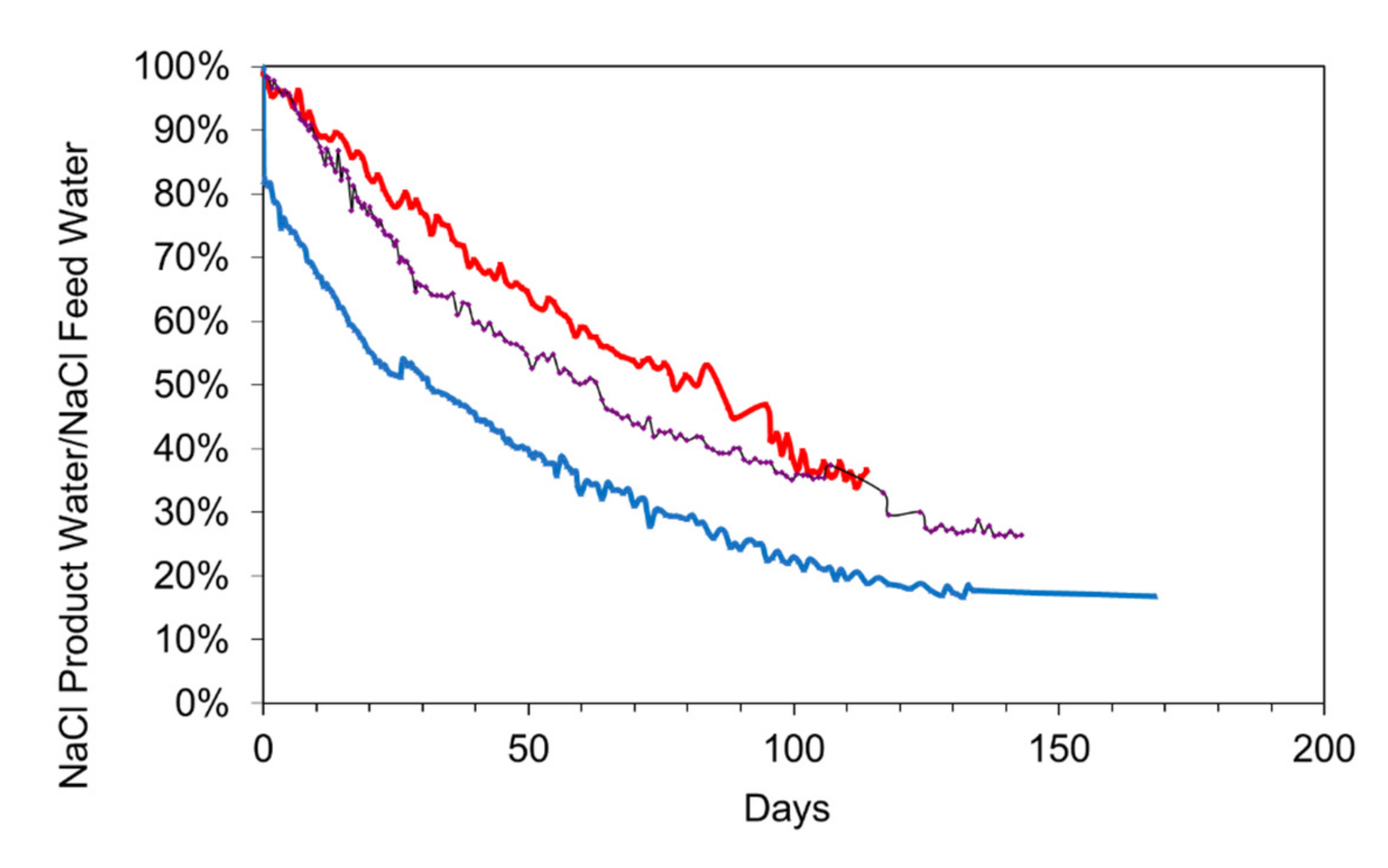
Comparison of three different materials for the desalination of static saline water: EC (electrical conductivity) = 15.2 mScm−1; Salinity = 7.52 g L−1 for the desalination pellet; EC (electrical conductivity) = 24.9 mScm−1; Salinity = 13.44 g L−1 for the n-Fe0; Temperature = 0.2 to 14 °C; water volume = 1 L). Red = Desalination pellet, 18 g L−1; black = 1 g L−1 n-Fe0; Blue = 25 g L−1 n-Fe0. The 25 g L−1 n-Fe0 displayed a 20% instantaneous desalination associated with the formation of Fe polymers.
Figure 15.
Comparison of three different materials for the desalination of static saline water: EC (electrical conductivity) = 15.2 mScm−1; Salinity = 7.52 g L−1 for the desalination pellet; EC (electrical conductivity) = 24.9 mScm−1; Salinity = 13.44 g L−1 for the n-Fe0; Temperature = 0.2 to 14 °C; water volume = 1 L). Red = Desalination pellet, 18 g L−1; black = 1 g L−1 n-Fe0; Blue = 25 g L−1 n-Fe0. The 25 g L−1 n-Fe0 displayed a 20% instantaneous desalination associated with the formation of Fe polymers.
Figure 16.
Comparison of three different materials for the desalination of saline water: Salinity = 7.52 g L−1; Temperature over the reaction period = 0.2 to 14 °C; water volume = 1 L. Red = Desalination pellet, 18 g L−1; black = 1 g L−1 n-Fe0; Blue = 25 g L−1 n-Fe0.
Figure 16.
Comparison of three different materials for the desalination of saline water: Salinity = 7.52 g L−1; Temperature over the reaction period = 0.2 to 14 °C; water volume = 1 L. Red = Desalination pellet, 18 g L−1; black = 1 g L−1 n-Fe0; Blue = 25 g L−1 n-Fe0.
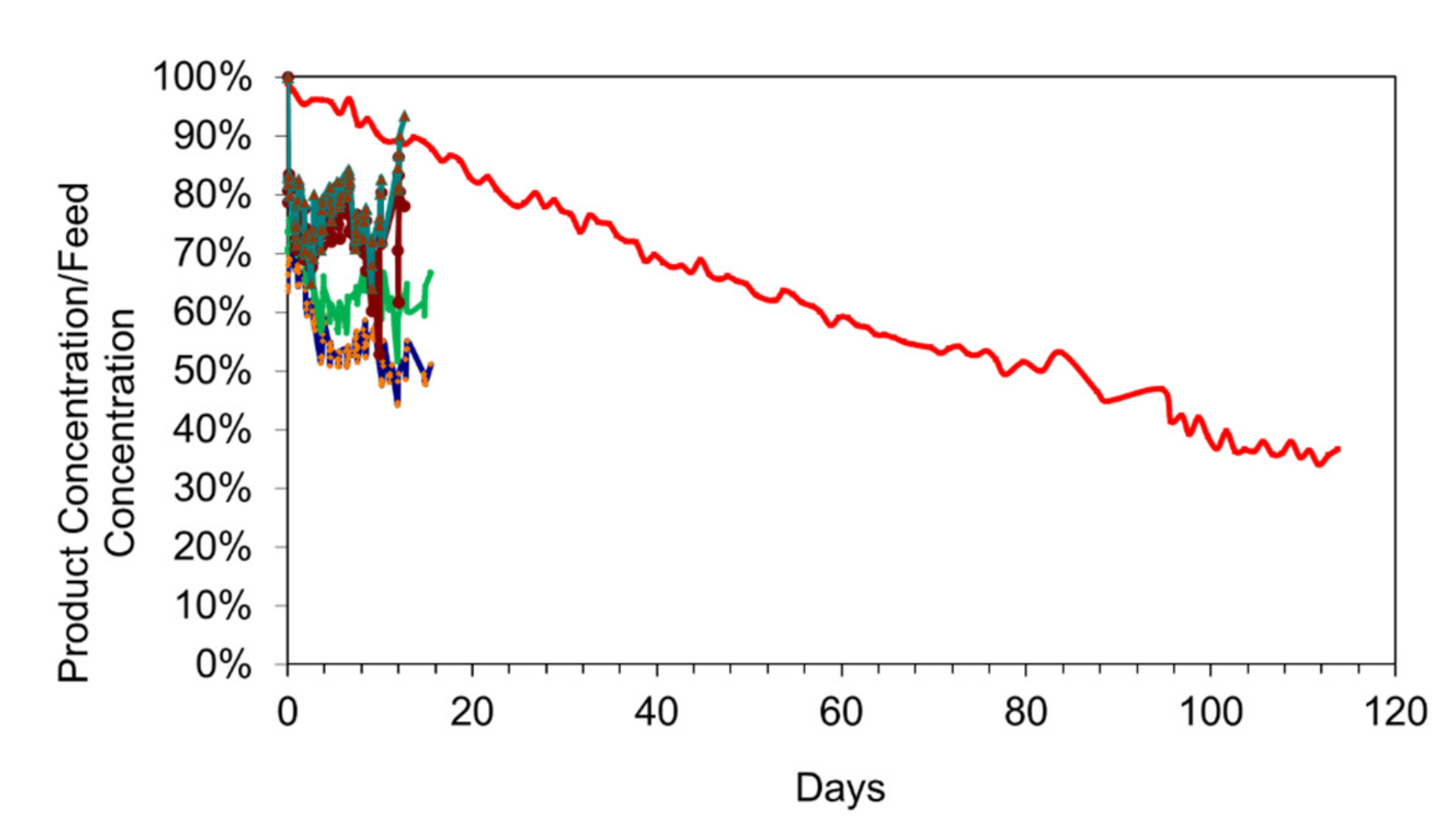
Figure 17.
Comparison of different materials for the desalination pellet construction: Salinity = 7.52 g L−1; Temperature = 0.2 to 14 °C; water volume = 1 L. Red = Desalination pellet, m-Fe0: n-Fe-polymer@n-C0, 18 g L−1; Salinity = 7.52 g L−1; Other colours: desalination pellet: unsupported n-Fe-polymer@n-R-CxHyOz polymer (four different polymer combinations; Blue, green, brown, buff = different n-Fe-polymer@n-gallic acid polymers; light blue: n-Fe-polymer@n-urea polymers); Each polymer contains 2 g Fe L−1; Feed water seawater salinity for the organic polymers = 32 g L−1. These polymer examples, show an almost instantaneous, 20% to 35% desalination after the polymer is added to the water.
Figure 17.
Comparison of different materials for the desalination pellet construction: Salinity = 7.52 g L−1; Temperature = 0.2 to 14 °C; water volume = 1 L. Red = Desalination pellet, m-Fe0: n-Fe-polymer@n-C0, 18 g L−1; Salinity = 7.52 g L−1; Other colours: desalination pellet: unsupported n-Fe-polymer@n-R-CxHyOz polymer (four different polymer combinations; Blue, green, brown, buff = different n-Fe-polymer@n-gallic acid polymers; light blue: n-Fe-polymer@n-urea polymers); Each polymer contains 2 g Fe L−1; Feed water seawater salinity for the organic polymers = 32 g L−1. These polymer examples, show an almost instantaneous, 20% to 35% desalination after the polymer is added to the water.
Figure 18.
Process flow diagram for a desalination plant using entrained, unsupported, n-Fe-polymer catalyst.
Figure 18.
Process flow diagram for a desalination plant using entrained, unsupported, n-Fe-polymer catalyst.
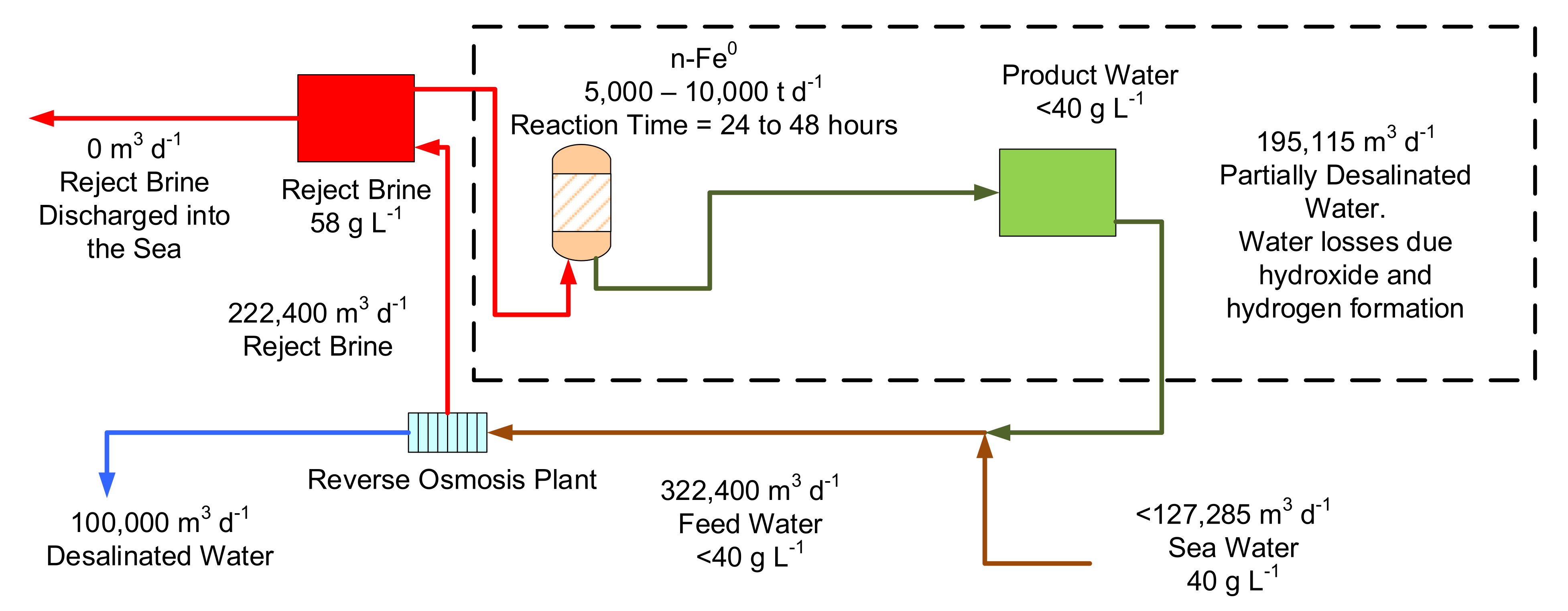
Figure 19.
Conceptual design for a zero-waste discharge conventional desalination plant based on 2016 technology, where the n-Fe
0 was not reusable. n-Fe
0 requirements are reduced, if the n-Fe
0 can be reused over a prolonged period. n-Fe
0 reaction profile as shown in
Figure 14, requiring a 24-to-48-h reaction time, and ZVI reactors containing 222,400 to 444,800 m
3 of water.
Figure 19.
Conceptual design for a zero-waste discharge conventional desalination plant based on 2016 technology, where the n-Fe
0 was not reusable. n-Fe
0 requirements are reduced, if the n-Fe
0 can be reused over a prolonged period. n-Fe
0 reaction profile as shown in
Figure 14, requiring a 24-to-48-h reaction time, and ZVI reactors containing 222,400 to 444,800 m
3 of water.
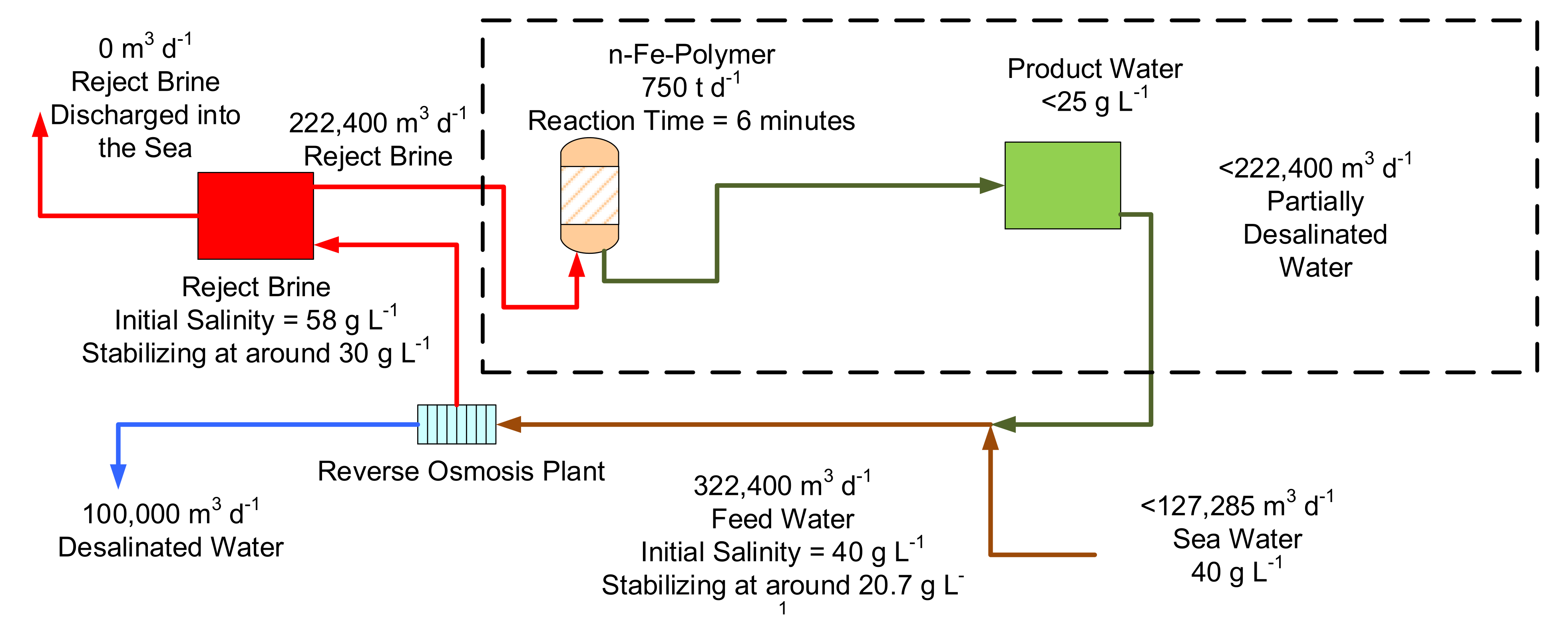
Figure 20.
Conceptual design for a zero-waste discharge conventional desalination plant based on 2022 technology, using a recycled n-Fe-polymer to process the reject brine. The required n-Fe-polymer reactor size is around 1000 m3. This figure illustrates what might be possible if the experimental results can be transferred into a commercial environment at a future date.
Figure 20.
Conceptual design for a zero-waste discharge conventional desalination plant based on 2022 technology, using a recycled n-Fe-polymer to process the reject brine. The required n-Fe-polymer reactor size is around 1000 m3. This figure illustrates what might be possible if the experimental results can be transferred into a commercial environment at a future date.
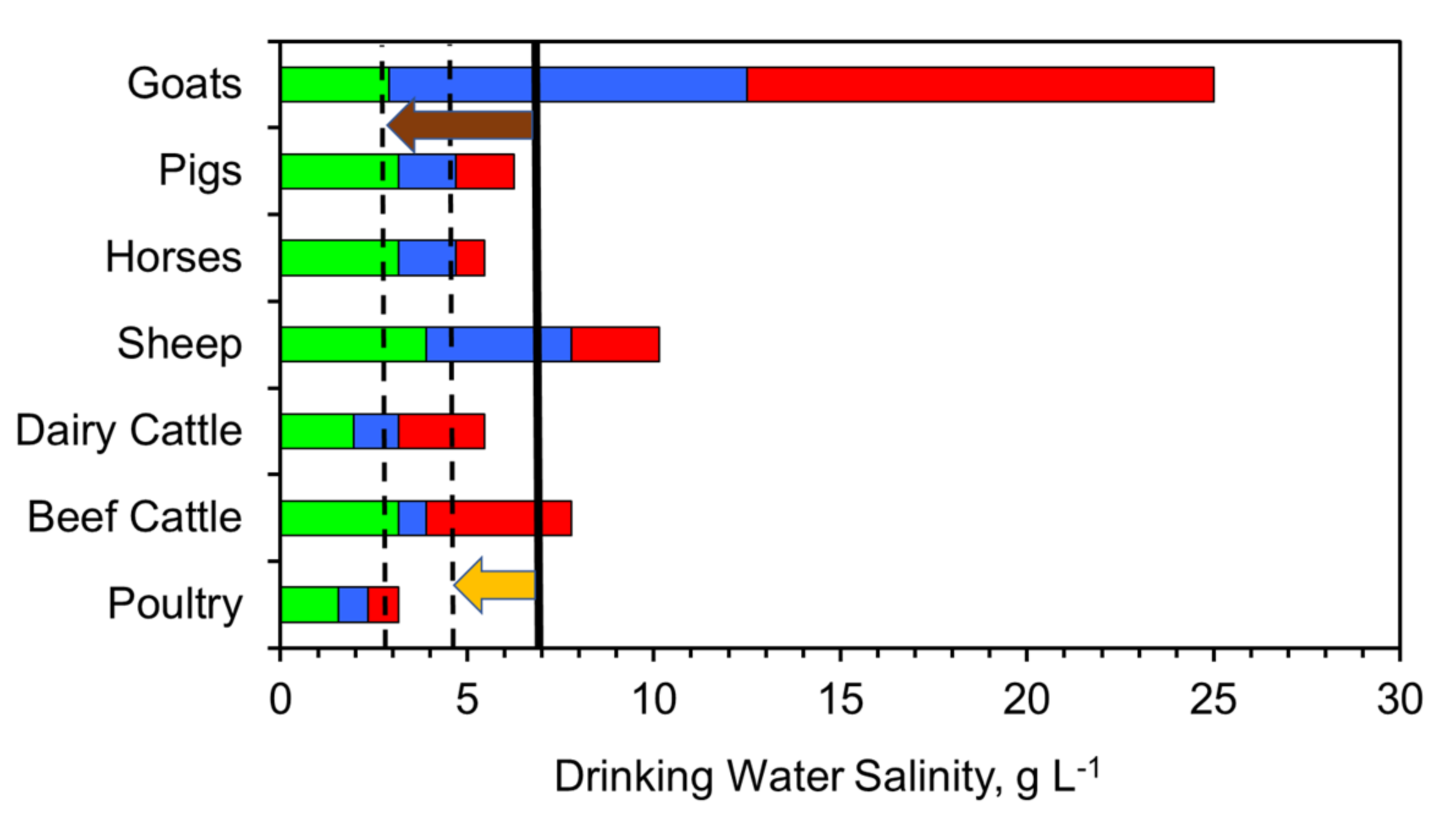
Figure 21.
Tolerance of livestock to salinity. Green = safe; blue = suitable; green = poor. Black line = average saline water salinity of 6.9 g L−1; Dashed black line = average partially desalinated water salinity with 30% desalination, and 60% desalination; Orange arrow indicates the salinity removed by 30% partial desalination. Brown arrow indicates the salinity removed by 60% partial desalination.
Figure 21.
Tolerance of livestock to salinity. Green = safe; blue = suitable; green = poor. Black line = average saline water salinity of 6.9 g L−1; Dashed black line = average partially desalinated water salinity with 30% desalination, and 60% desalination; Orange arrow indicates the salinity removed by 30% partial desalination. Brown arrow indicates the salinity removed by 60% partial desalination.
Figure 22.
Salinity tolerance of four livestock grass crops. Solid black line = Expected soil salinity if it is irrigated with saline irrigation; Dotted black line = Expected soil salinity if it is irrigated with partially desalinated water; Blue arrow indicates the change associated with 30% desalination; Dotted brown line = Expected soil salinity if it is irrigated with partially desalinated water, Brown arrow indicates the change associated with 60% desalination.
Figure 22.
Salinity tolerance of four livestock grass crops. Solid black line = Expected soil salinity if it is irrigated with saline irrigation; Dotted black line = Expected soil salinity if it is irrigated with partially desalinated water; Blue arrow indicates the change associated with 30% desalination; Dotted brown line = Expected soil salinity if it is irrigated with partially desalinated water, Brown arrow indicates the change associated with 60% desalination.
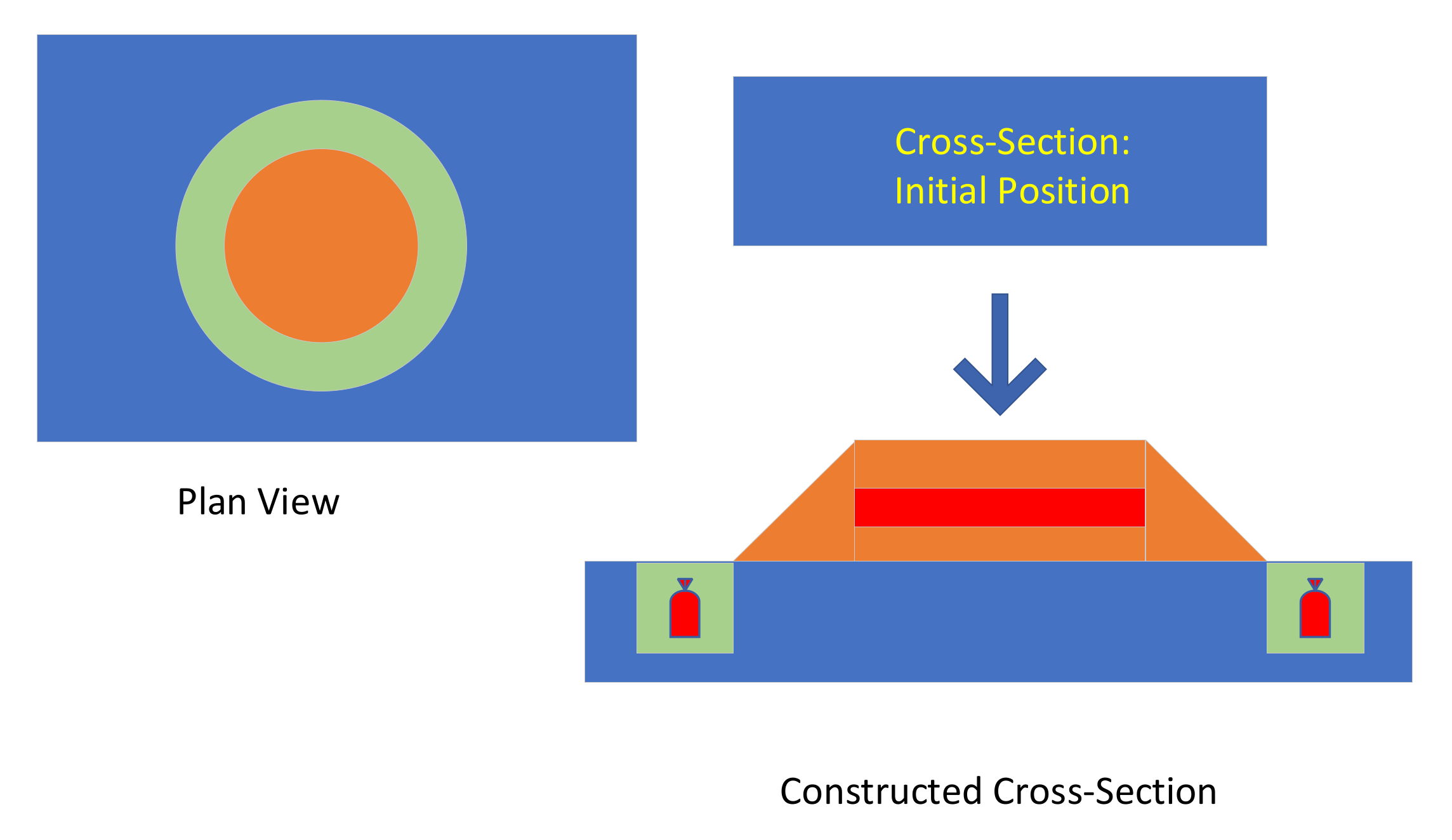
Figure 23.
Raised Field Irrigation Scheme. A raised mound is created (brown), surrounded by a ditch (green). ZVI desalination pellet placement locations are shown in red.
Figure 23.
Raised Field Irrigation Scheme. A raised mound is created (brown), surrounded by a ditch (green). ZVI desalination pellet placement locations are shown in red.
Figure 24.
Raised Field Irrigation Scheme-Soil Water Salinity vs. depth below ground surface. Black line = no irrigation; Blue line is irrigation with 14 g NaCl L−1; Red and Green lines are the expected soil salinity range following irrigation with 14 g NaCl L−1, where the ZVI desalination treatment product placed at about 0.7 m depth within the raised field.
Figure 24.
Raised Field Irrigation Scheme-Soil Water Salinity vs. depth below ground surface. Black line = no irrigation; Blue line is irrigation with 14 g NaCl L−1; Red and Green lines are the expected soil salinity range following irrigation with 14 g NaCl L−1, where the ZVI desalination treatment product placed at about 0.7 m depth within the raised field.
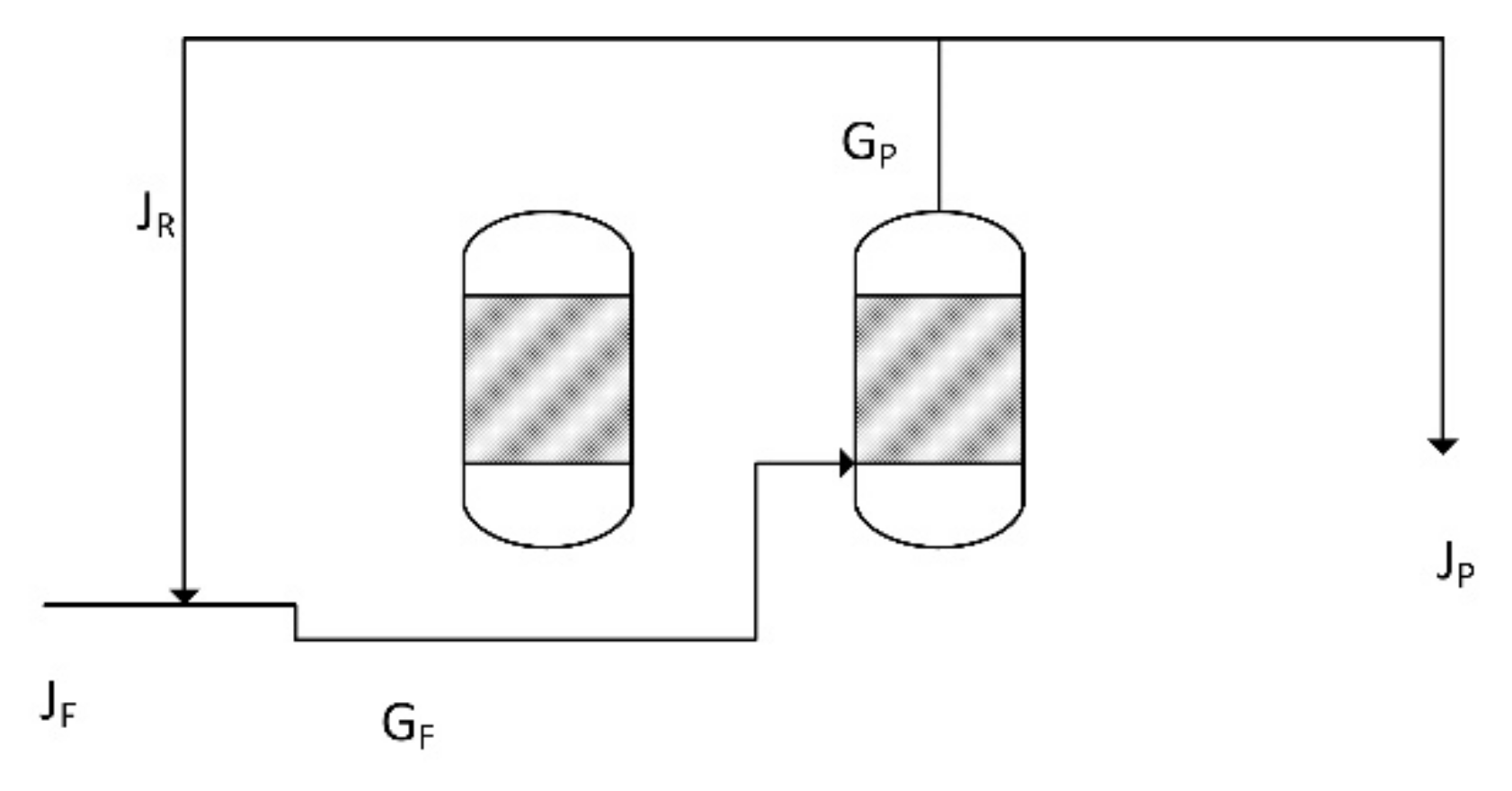
Figure 25.
Summary of the reactor recycle process flow. Two reactors are shown, a Halite Reactor and a ZVI Reactor. The product gas from the ZVI Reactor is recycled.
Figure 25.
Summary of the reactor recycle process flow. Two reactors are shown, a Halite Reactor and a ZVI Reactor. The product gas from the ZVI Reactor is recycled.
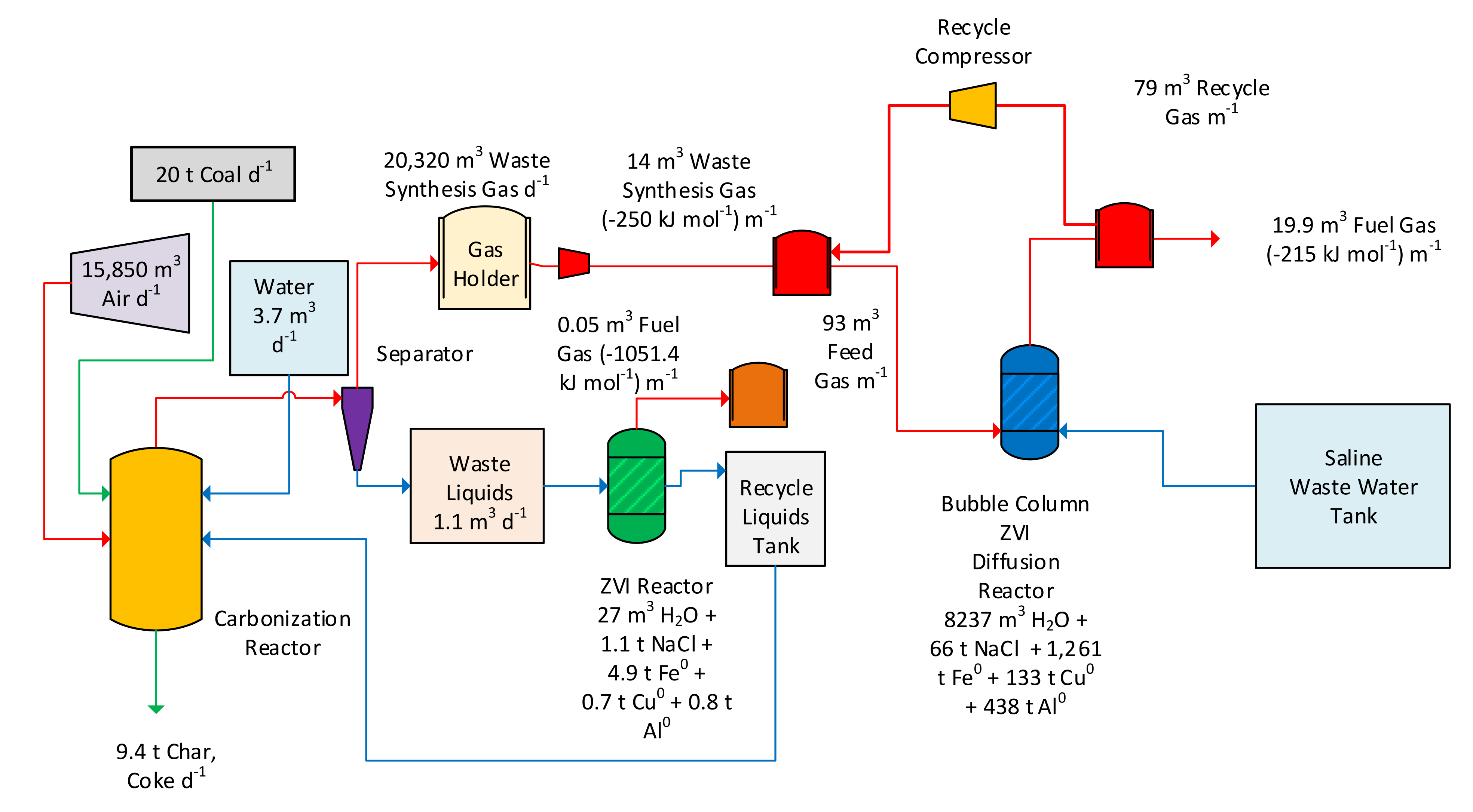
Figure 26.
Example process flow diagram for a plant manufacturing 16,000 t a−1 of desalination pellets.
Figure 26.
Example process flow diagram for a plant manufacturing 16,000 t a−1 of desalination pellets.
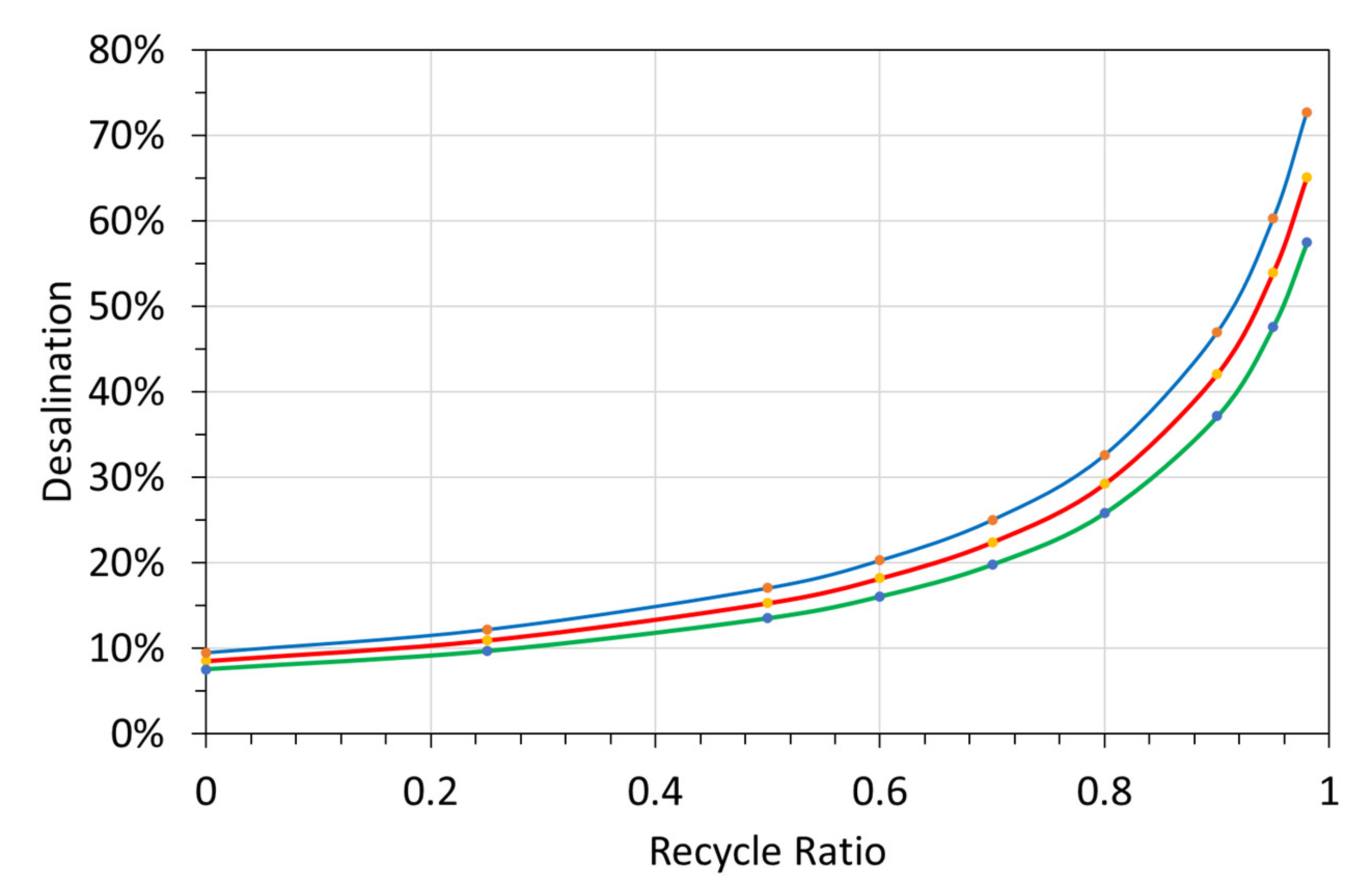
Figure 27.
Impact of recycle ratio in the ZVI reactor, on the desalination associated with the ZVI pellets, 50 g L−1. The n-C0 content of the ZVI pellets, may increase with increasing recycle ratio. The graph shows the results for a feed water with a salinity of 5 g L−1, and a reaction time of 500 h. Red = median outcome: Blue = upper quartile; green = lower quartile.
Figure 27.
Impact of recycle ratio in the ZVI reactor, on the desalination associated with the ZVI pellets, 50 g L−1. The n-C0 content of the ZVI pellets, may increase with increasing recycle ratio. The graph shows the results for a feed water with a salinity of 5 g L−1, and a reaction time of 500 h. Red = median outcome: Blue = upper quartile; green = lower quartile.
Table 1.
Halite and ZVI composition used. All amounts are in grams. Halite Reactor column length = 6 m, 15 mm O.D. ZVI Reactor water column length above the gas distributer = 8.3 m, 22 mm O.D.; Fe0 = zero valent iron; Al0 = zero valent aluminium; Cu0 = zero valent copper.
Table 1.
Halite and ZVI composition used. All amounts are in grams. Halite Reactor column length = 6 m, 15 mm O.D. ZVI Reactor water column length above the gas distributer = 8.3 m, 22 mm O.D.; Fe0 = zero valent iron; Al0 = zero valent aluminium; Cu0 = zero valent copper.
| | NaCl | Fe0 | Cu0 | Al0 | SiO2 | H2O |
|---|
| Halite Reactor | 322.7 | 0 | 0 | 0 | 176.2 | 0 |
| ZVI Reactor | 23.3 | 405.6 | 43.4 | 141.7 | 0 | 2650 |
Table 2.
Continuous flow trials decontaminating water containing Cl− ions and Na+ ions. Reactor volume = 1.5 L; Flow rate: Trials 1, 3 = 0.25 L m−1; Trial 4 = 0.2 L m−1; Trial 5 to 9 = 0.4 L m−1; Temperature = <20 °C; Product analysis was made following the processing of the initial 45 L of feed water. Reactor: 3 m long conduit (4 cm O.D.). Product + 24 hrs indicates the product water composition following 24 h in storage. Polymer details: (i) 1 g L−1 of reconstituted: Fe0: plus (ii) Fe(b)@n-C0 polymer + n-Fe(b)@polymer. Trial 1: 0.3 g L−1 n-Fe(b)-S-gallic acid polymer; Trial 2: 0.2 g L−1 n-Fe(b)-S-gallic acid polymer; Trial 3: 0.7 g L−1 n-Fe(b)@Ca(b) polymer; Trial 4: 1.5 g L−1 n-Fe(b)@Ca(b)@K(b) polymer; Trial 5: 0.7 g L−1 n-Fe(b)@Ca(b); Trial 5: 0.7 g L−1 n-Fe(b)@Ca(b); Trial 6: 1.2 g L−1 n-Fe(b)@Ca(b)@K(b) polymer; Trial 7: 1.2 g L−1 n-Fe(b)@Ca(b)@K(b)@organic polymer; Trial 8: 1.8 g L−1 n-Fe(b)@Ca(b)@K(b)@Mn@Zn@organic polymer; Trial 9: 1.8 g L−1 n-Fe(b)@Ca(b)@K(b)@Mn@Zn@organic polymer; Ion concentrations were measured using ion selective Na+ and Cl− electrodes.
Table 2.
Continuous flow trials decontaminating water containing Cl− ions and Na+ ions. Reactor volume = 1.5 L; Flow rate: Trials 1, 3 = 0.25 L m−1; Trial 4 = 0.2 L m−1; Trial 5 to 9 = 0.4 L m−1; Temperature = <20 °C; Product analysis was made following the processing of the initial 45 L of feed water. Reactor: 3 m long conduit (4 cm O.D.). Product + 24 hrs indicates the product water composition following 24 h in storage. Polymer details: (i) 1 g L−1 of reconstituted: Fe0: plus (ii) Fe(b)@n-C0 polymer + n-Fe(b)@polymer. Trial 1: 0.3 g L−1 n-Fe(b)-S-gallic acid polymer; Trial 2: 0.2 g L−1 n-Fe(b)-S-gallic acid polymer; Trial 3: 0.7 g L−1 n-Fe(b)@Ca(b) polymer; Trial 4: 1.5 g L−1 n-Fe(b)@Ca(b)@K(b) polymer; Trial 5: 0.7 g L−1 n-Fe(b)@Ca(b); Trial 5: 0.7 g L−1 n-Fe(b)@Ca(b); Trial 6: 1.2 g L−1 n-Fe(b)@Ca(b)@K(b) polymer; Trial 7: 1.2 g L−1 n-Fe(b)@Ca(b)@K(b)@organic polymer; Trial 8: 1.8 g L−1 n-Fe(b)@Ca(b)@K(b)@Mn@Zn@organic polymer; Trial 9: 1.8 g L−1 n-Fe(b)@Ca(b)@K(b)@Mn@Zn@organic polymer; Ion concentrations were measured using ion selective Na+ and Cl− electrodes.
| Trial | Water | pH | Eh, mV | Temperature, C | Cl−, g L−1 | Na+, g L−1 |
|---|
| 1 | Feed | 9.41 | 374 | 5 | 4.86 | 14.33 |
| Product | 8.92 | 385 | 4.4 | 0.99 | 11.61 |
| 2 | Feed | 8.92 | 385 | 4.4 | 0.99 | 11.61 |
| Product | 8.86 | 399 | 4.9 | 0.51 | 10.65 |
| 3 | Feed | 7.97 | −26 | 9.3 | 146.15 | 6.22 |
| Product | 7.68 | 72 | 12.8 | 85.12 | 5.43 |
| 4 | Feed | 10.13 | 119 | 14.8 | 117.40 | 5.24 |
| Product | 7.68 | 298 | 15.6 | 106.35 | 5.19 |
| Product + 24 h | 7.68 | 327 | 11.2 | 74.73 | 5.17 |
| 5 | Feed | 9.17 | 254 | 10.3 | 2.45 | 1.79 |
| Product | 11.3 | −40 | 10.3 | 1.13 | 1.79 |
| 6 | Feed | 9.17 | 254 | 10.3 | 2.45 | 1.79 |
| Product | 11.7 | −41 | 9.9 | 1.79 | 1.68 |
| 7 | Feed | 9.06 | 215 | 10.3 | 7.89 | 6.03 |
| Product | 7.53 | 17 | 12.1 | 4.64 | 1.04 |
| 8 | Feed | 9.06 | 215 | 10.3 | 7.89 | 6.03 |
| Product | 12.25 | −137 | 14.1 | 1.79 | 0.92 |
| Product + 24 h | 12.58 | −53 | 11.9 | 1.39 | 0.97 |
| 9 | Feed | 9.1 | 232 | 10.6 | 9.02 | 4.76 |
| Product | 11.77 | −399 | 11.5 | 2.62 | 1.18 |
| Product + 24 h | 12.24 | −97 | 7.4 | 2.48 | 0.97 |
Table 3.
Expected Product Gas Composition, pellets manufactured and desalination water potential of the manufactured pellets. Assumptions: ZVI Reactors are placed in series where the product gas from one reactor is passed directly into the next downstream reactor. Manufactured volumes are based on a feed gas flow rate of 14 m3 m−1. The product gas volume from 9 reactors placed in series is 23.69 m3 m−1.
Table 3.
Expected Product Gas Composition, pellets manufactured and desalination water potential of the manufactured pellets. Assumptions: ZVI Reactors are placed in series where the product gas from one reactor is passed directly into the next downstream reactor. Manufactured volumes are based on a feed gas flow rate of 14 m3 m−1. The product gas volume from 9 reactors placed in series is 23.69 m3 m−1.
| Number of ZVI Reactors in Series | N2 | CO | CO2 | CH4 | H2 | Pellets Manufactured, t a−1 | Water Desalination Potential, m3 a−1 |
|---|
| 1 | 40.98% | 9.69% | 8.60% | 13.03% | 27.35% | 1700 | 68,000 |
| 2 | 37.48% | 8.05% | 8.83% | 10.38% | 34.97% | 3203 | 128,113 |
| 3 | 34.85% | 6.81% | 8.99% | 8.41% | 40.71% | 4532 | 181,274 |
| 4 | 32.81% | 5.82% | 9.12% | 6.90% | 45.15% | 5708 | 228,307 |
| 5 | 31.19% | 5.03% | 9.23% | 5.72% | 48.66% | 6748 | 269,936 |
| 6 | 29.89% | 4.38% | 9.31% | 4.77% | 51.50% | 7670 | 306,796 |
| 7 | 28.82% | 3.84% | 9.38% | 4.01% | 53.82% | 8486 | 339,449 |
| 8 | 27.94% | 3.39% | 9.44% | 3.39% | 55.75% | 9210 | 368,386 |
| 9 | 27.20% | 3.00% | 9.48% | 2.87% | 57.36% | 9851 | 394,041 |
| Feed | 46.03% | 11.97% | 8.33% | 16.79% | 16.88% | | |
Table 4.
Impact of recycle ratio on the stable average gas product output. Part of the gas product (recycle gas) is recycled from the ZVI Reactor to the ZVI Reactor (
Figure 24). Recycle Period = 480 min. All gas flow rates are standardised to the feed gas supplied from a Maclaurin retort, processing 20 t coal d
−1. The bulk of the removed carbon is retained in ZVI desalination pellets. Recycle ratio is the proportion of product gas which is recycled to the ZVI Reactor.
Table 4.
Impact of recycle ratio on the stable average gas product output. Part of the gas product (recycle gas) is recycled from the ZVI Reactor to the ZVI Reactor (
Figure 24). Recycle Period = 480 min. All gas flow rates are standardised to the feed gas supplied from a Maclaurin retort, processing 20 t coal d
−1. The bulk of the removed carbon is retained in ZVI desalination pellets. Recycle ratio is the proportion of product gas which is recycled to the ZVI Reactor.
| Recycle Ratio | N2 | CO | CO2 | CH4 | H2 | Product Gas, m3m−1 | Recycle Gas, m3m−1 | Carbon Removed |
|---|
| 98% | 23.73% | 1.03% | 9.53% | 1.03% | 63.09% | 27.15 | 1330.41 | 39.39% |
| 95% | 25.88% | 2.24% | 9.41% | 2.39% | 58.65% | 24.90 | 473.13 | 32.65% |
| 90% | 28.65% | 3.72% | 9.27% | 4.22% | 52.91% | 22.50 | 202.47 | 25.45% |
| 80% | 32.37% | 5.61% | 9.06% | 6.79% | 45.19% | 19.91 | 79.63 | 17.69% |
| 70% | 34.77% | 6.78% | 8.94% | 8.50% | 40.21% | 18.53 | 43.24 | 13.57% |
| 60% | 36.46% | 7.58% | 8.85% | 9.71% | 36.72% | 17.68 | 26.51 | 11.01% |
| 50% | 37.70% | 8.17% | 8.78% | 10.62% | 34.14% | 17.09 | 17.09 | 9.26% |
| 25% | 39.74% | 9.12% | 8.67% | 12.11% | 29.91% | 16.22 | 5.41 | 6.63% |
| 0% | 40.98% | 9.69% | 8.60% | 13.03% | 27.35% | 15.73 | 0.00 | 5.17% |
| Unprocessed | 46.03% | 11.97% | 8.33% | 16.79% | 16.88% | 14.00 | 0.00 | 0.00% |
Table 5.
Expected Pellets manufactured and desalination water potential of the manufactured pellets. Assumptions: A proportion of the product gas is recycled to the ZVI Reactor; Manufactured volumes are based on a feed gas flow rate of 14 m
3 m
−1 (
Table 4).
Table 5.
Expected Pellets manufactured and desalination water potential of the manufactured pellets. Assumptions: A proportion of the product gas is recycled to the ZVI Reactor; Manufactured volumes are based on a feed gas flow rate of 14 m
3 m
−1 (
Table 4).
| Recycle Ratio | Pellets Manufactured, t a−1 | Water Desalination Potential, m3 a−1 |
|---|
| 98% | 229,993 | 9,199,715 |
| 95% | 80,602 | 3,224,099 |
| 90% | 34,205 | 1,368,215 |
| 80% | 13,817 | 552,662 |
| 70% | 8046 | 321,822 |
| 60% | 5491 | 219,628 |
| 50% | 4096 | 163,836 |
| 25% | 2433 | 97,312 |
| 0% | 1700 | 68,000 |
Table 6.
Comparison of ZVI desalination pellet performance with the desalination performance of 50 nm Fe
0 (PVP (polyvinylpyrrolidone) coated) and steel wool (Fe
0:Fe(b)@ureapolymer);
as = particle surface area;
Pw = ZVI concentration. * = steel wool (
Figure 7), which is operated in a catalytic pressure-swing-adsorption-desorption diffusion reactor.
Table 6.
Comparison of ZVI desalination pellet performance with the desalination performance of 50 nm Fe
0 (PVP (polyvinylpyrrolidone) coated) and steel wool (Fe
0:Fe(b)@ureapolymer);
as = particle surface area;
Pw = ZVI concentration. * = steel wool (
Figure 7), which is operated in a catalytic pressure-swing-adsorption-desorption diffusion reactor.
| Reference | This Study | [79] | [13] | [13] |
|---|
| Fe0 Particle Size | 0.002 to 0.08 mm | 50 nm | Steel Wool | Steel Wool * |
| as, m2 g−1 | not measured | 20 | not measured | not measured |
| Pw, g L−1 | 25–50 | 25 | 1.67 | 1.67 |
| Reactor Size, L | 1 | 0.2 | 240 | 240 |
| Time, h, to Achieve 24.5% desalination | 1500 | 720 to 1400 | Not Achieved | 3 |
| Time, h, to Achieve 60% desalination | 3000 | 1400 to 2800 | Not Achieved | Not Achieved |
| Air flow, L h−1 kg Fe0 | 0 | 0 | 0 | 150 |
| Reaction Order | Zero | First | n/a | First or second |