Aggregation Kinetics and Mechanism of Humic Acid Reaction with Cs+ and Co2+ Metal Ions Using Batch Techniques
Abstract
:1. Introduction
2. Materials and Methods
2.1. HA Solution and Reagents
2.2. DLS, EPM, EEM and FT-IR Spectroscopic Measurements
2.3. Aggregation Kinetics and Attachment Efficiency (α) Calculation
3. Results and Discussion
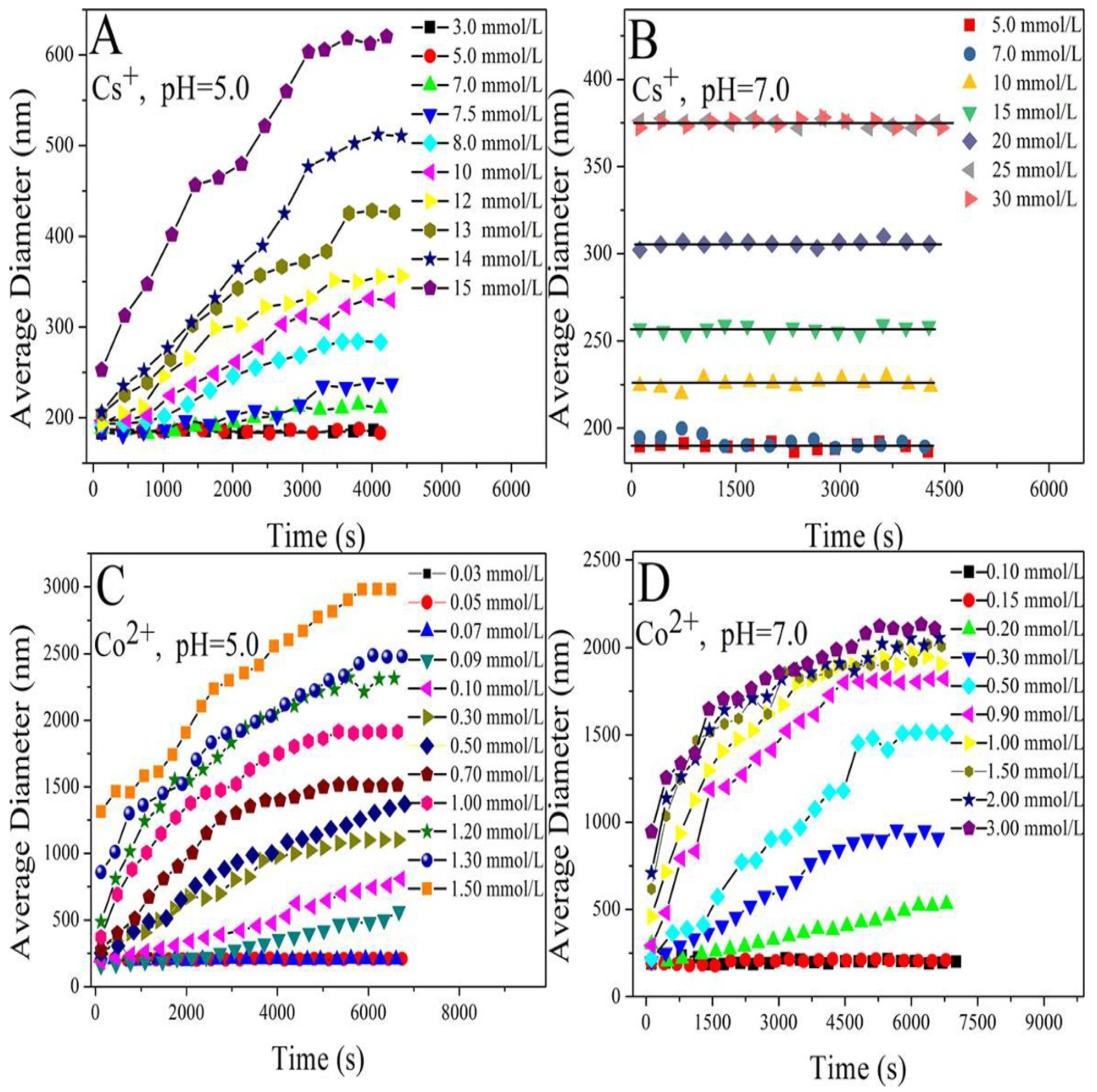
3.1. Aggregation Kinetics of HA in the Presence of Cs+ or Co2+ Metal Ions
3.2. EPM and Attachment Efficiency Studies
3.3. FT-IR Spectral Analysis
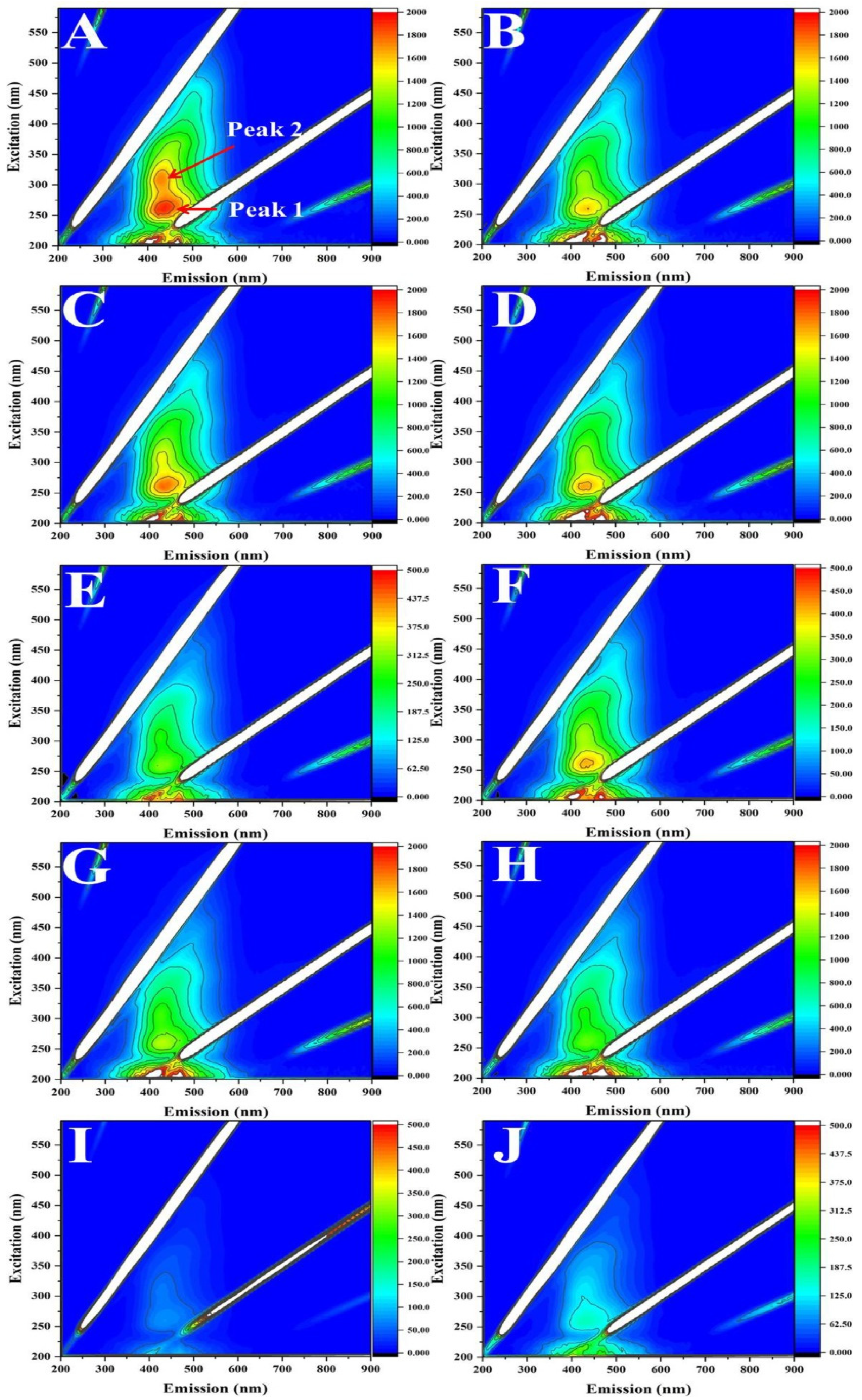
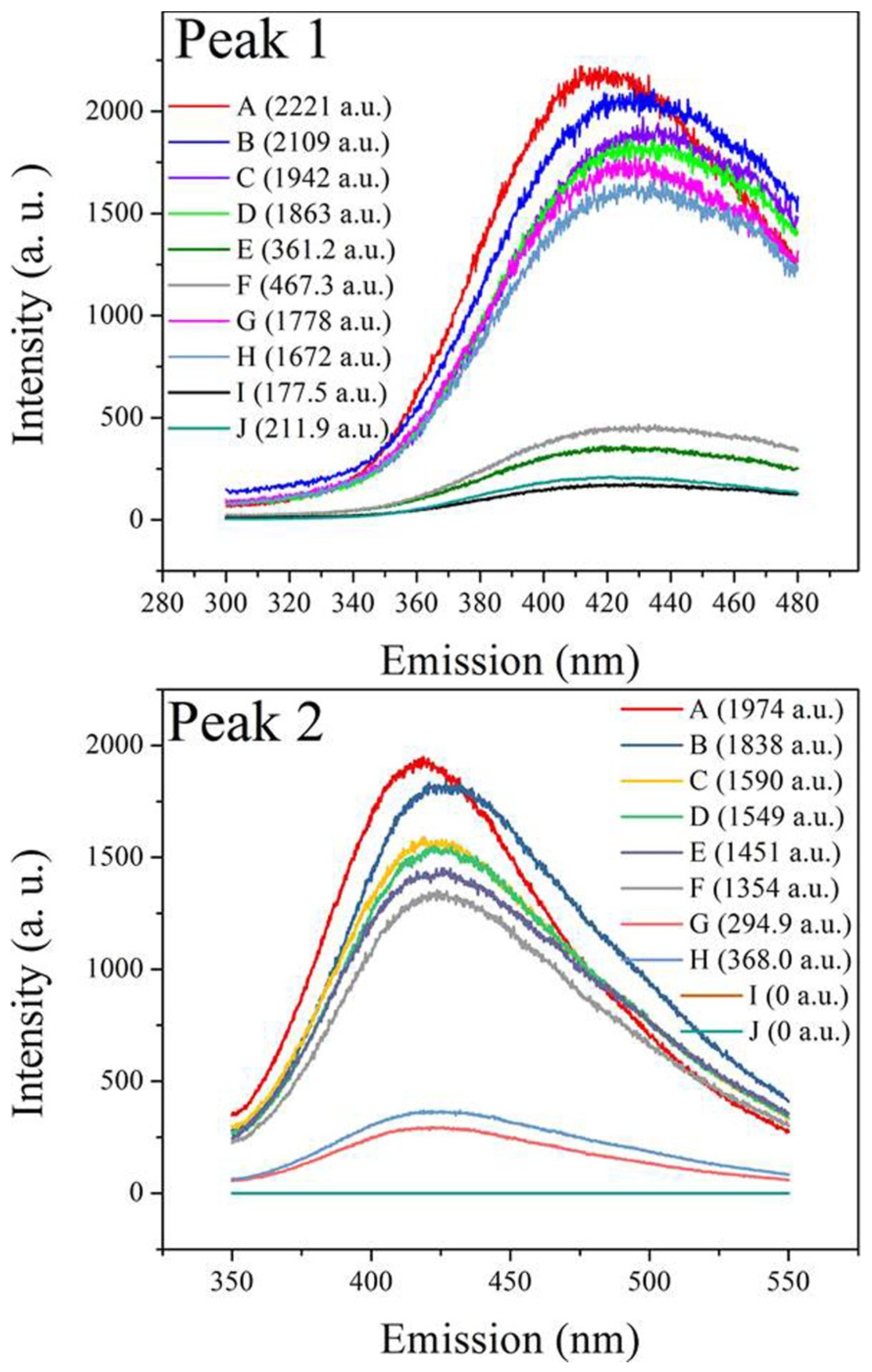
3.4. Fluorescence EEM Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Illés, E.; Tombácz, E. The role of variable surface charge and surface complexation in the adsorption of humic acid on magnetite. Colloids Surf. A 2003, 230, 99–109. [Google Scholar] [CrossRef]
- Mao, J.; Fang, X.; Schmidt-Rohr, K.; Carmo, A.M.; Hundal, L.S.; Thompson, M.L. Molecular-scale heterogeneity of humic acid in particle-size fractions of two Iowa soils. Geoderma 2007, 140, 17–29. [Google Scholar] [CrossRef]
- Shirshova, L.T.; Ghabbour, E.A.; Davies, G. Spectroscopic characterization of humic acid fractions isolated from soil using different extraction procedures. Geoderma 2006, 133, 204–216. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, L.; Yin, H.; Jin, S.; Liu, F.; Chen, H. Mechanism study of humic acid functional groups for Cr(VI) retention: Two-dimensional FTIR and 13C CP/MAS NMR correlation spectroscopic analysis. Environ. Pollut. 2017, 225, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Puebla, R.A.; Garrido, J.J. Effect of pH on the aggregation of a gray humic acid in colloidal and solid states. Chemosphere 2005, 59, 659–667. [Google Scholar] [CrossRef]
- Terashima, M.; Fukushima, M.; Tanaka, S. Influence of pH on the surface activity of humic acid: Micelle-like aggregate formation and interfacial adsorption. Colloids Surf. A 2004, 247, 77–83. [Google Scholar] [CrossRef]
- Su, X.; Hu, J.; Zhang, J.; Liu, H.; Yan, C.; Xu, J.; Ma, Y.; Song, J. Investigating the adsorption behavior and mechanisms of insoluble Humic acid/starch composite microspheres for metal ions from water. Colloids Surf. A 2021, 610, 125672. [Google Scholar] [CrossRef]
- Lian, X.; Liao, S.; Yang, Y.; Zhang, X.; Wang, Y. Effect of pH or metal ions on the oil/water interfacial behavior of humic acid based surfactant. Langmuir 2020, 36, 10838–10845. [Google Scholar] [CrossRef]
- Salehi, E.; Madaeni, S.S. Adsorption of humic acid onto ultrafiltration membranes in the presence of protein and metal ions. Desalination 2010, 263, 139–145. [Google Scholar] [CrossRef]
- Rupiasih, N.N.; Sumadiyasa, M.; Ratnawati, A.A. Study of the removal of humic acid, organic pollutant by water hyacinth plant from aquatic environment and its effect on pH, chlorophyll content and degradation. Asian J. Water Environ. Pollut. 2013, 10, 1–9. [Google Scholar]
- Zhang, J.; Wang, S.; Wang, Q.; Wang, N.; Li, C.; Wang, L. First determination of Cu adsorption on soil humin. Environ. Chem. Lett. 2016, 11, 41–46. [Google Scholar] [CrossRef]
- Zhu, Q.; Wu, Y.; Zeng, J.; Zhang, T.; Lin, X. Influence of organic amendments used for benz[a]anthracene remediation in a farmland soil: Pollutant distribution and bacterial changes. J. Soils Sediments 2020, 20, 32–41. [Google Scholar] [CrossRef]
- Kalinichev, A.G.; Iskrenova-Tchoukova, E.; Ahn, W.; Clark, M.M.; Krikpatrick, R.J. Effect of Ca2+ on supramolecular aggregation of natural organic matter in aqueous solutions: A comparison of molecular modeling approaches. Geoderma 2011, 169, 27–32. [Google Scholar] [CrossRef]
- Omar, F.T.; Aziz, H.A.; Stoll, S. Aggregation and disaggregation of ZnO nanoparticles: Influence of pH and adsorption of Suwannee River humic acid. Sci. Total Environ. 2014, 468–469, 195–201. [Google Scholar] [CrossRef]
- Dong, S.; Cai, W.; Xia, J.; Sheng, L.; Wang, W.; Liu, H. Aggregation kinetics of fragmental PET nanoparticles in aqueous environment: Complex roles of electrolytes, pH and humic acid. Environ. Pollut. 2021, 268, 115828. [Google Scholar] [CrossRef] [PubMed]
- Dasher, D.; Hanson, W.; Read, S.; Faller, S.; Efurd, W.; Kelley, J.; Patrick, R. An assessment of the reported leakage of anthropogenic radionuclides from the underground nuclear test sites at Amchitka Island, Alaska, USA to the surface environment. J. Environ. Radioact. 2002, 60, 165–187. [Google Scholar] [CrossRef]
- Diansky, N.A.; Morozov, E.G.; Fomin, V.V.; Frey, D.I. Spread of pollution from a bottom source in the Norwegian sea. Izv. Atmos. Ocean. Phys. 2021, 57, 197–207. [Google Scholar] [CrossRef]
- Sheth, Y.; Dharaskar, S.; Khalid, M.; Sonawane, S. An environment friendly approach for heavy metal removal from industrial wastewater using chitosan based biosorbent: A review. Sustain. Energy Technol. Assess. 2021, 43, 100951. [Google Scholar] [CrossRef]
- Basu, H.; Singhal, R.K.; Pimple, M.V.; Kumar, A.; Reddy, A.V.R. Association and migration of uranium and thorium with silica colloidal particles in saturated subsurface zone. J. Radioanal. Nucl. Chem. 2015, 303, 2283–2290. [Google Scholar] [CrossRef]
- Aba, A.; Al-Boloushi, O.; Ismaeel, A.; Al-Tamimi, S. Migration behavior of radiostrontium and radiocesium in arid-region soil. Chemosphere 2021, 281, 130953. [Google Scholar] [CrossRef]
- Chawla, F.; Steinmann, P.; Pfeifer, H.R.; Froidevaux, P. Atmospheric deposition and migration of artificial radionuclides in Alpine soils (Val Piora, Switzerland) compared to the distribution of selected major and trace elements. Sci. Total Environ. 2010, 3, 3292–3302. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Liang, B.; Fang, Z.; Xie, Y.; Tsang, P. Effect of humic acid and transition metal ions on the debromination of decabromodiphenyl by nano zero-valent iron: Kinetics and mechanisms. J. Nanopart. Res. 2014, 16, 2786–2799. [Google Scholar] [CrossRef]
- Ke, T.; Li, L.; Rajavel, K.; Wang, Z.; Lin, D. A multi-method analysis of the interaction between humic acids and heavy metal ions. J. Environ. Sci. Health. A 2018, 53, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.K.; Jain, A.; Kumar, S.; Tomar, B.S.; Tomar, R.; Manchanda, V.K.; Ramanathan, S. Role of magnetite and humic acid in radionuclide migration in the environment. J. Contam. Hydrol. 2009, 106, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Zaccone, C.; Cocozza, C.; Cheburkin, A.K.; Shotyk, W.; Miano, T.M. Enrichment and depletion of major and trace elements, and radionuclides in ombrotrophic raw peat and corresponding humic acid. Geoderma 2007, 141, 235–246. [Google Scholar] [CrossRef]
- Volkov, I.V.; Polyakov, E.V. Interaction of humic acid with microelements/radionuclides in sorption systems. Radiochemistry 2020, 62, 141–160. [Google Scholar] [CrossRef]
- Chruszcz, M.; Potrzebowski, W.; Zimmerman, M.D.; Grabowski, M.; Zheng, H.; Lasota, P.; Minor, W. Analysis of solvent content and oligomeric states in protein crystals-does symmetry matter. Protein. Sci. 2008, 17, 623–632. [Google Scholar] [CrossRef]
- Malikov, D.A.; Goryacheva, T.A.; Novikov, A.P.; Platonov, V.V.; Treityak, R.Z. Adsorption of actinides on chemically modified humic acids. Geochem. Int. 2012, 50, 1032–1037. [Google Scholar] [CrossRef]
- Ibrahim, A.M.; Ali, I.M.; Zakaria, E.S.; EI-Naggar, I.M. Adsorption and kinetic performance of Cs+, Co2+, and Ce4+ radionuclides on zirconium vanadate as a cation exchanger. Radiochemistry 2013, 55, 581–588. [Google Scholar] [CrossRef]
- Mahmoud, M.R.; Rashad, G.M.; Metwally, E.; Saad, E.A.; Elewa, A.M. Adsorption removal of 134Cs+, 60Co2+ and 152+154Eu3+ radionuclides from aqueous solutions using sepiolite: Single and multicomponent systems. Appl. Clay Sci. 2017, 141, 72–80. [Google Scholar] [CrossRef]
- Wang, L.; Wang, L.; Ye, X.; Li, W.; Ren, X.; Sheng, G.; Yu, H.; Wang, X. Coagulation kinetics of humic aggregates in mono- and di-valent electrolyte solutions. Environ. Sci. Technol. 2013, 47, 5042–5049. [Google Scholar] [CrossRef] [PubMed]
- Huynh, K.A.; Chen, K.L. Aggregation kinetics of citrate and polyvinylpyrrolidone coated silver nanoparticles in monovalent and divalent electrolyte solutions. Environ. Sci. Technol. 2011, 45, 5564–5571. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Xing, S.; Gong, Y.; Miyajima, T. Characterization of humic acids by two-dimensional correlation fluorescence spectroscopy. J. Mol. Struct. 2008, 883–884, 155–159. [Google Scholar] [CrossRef]
- Yang, K.; Chen, B.; Zhu, X.; Xing, B. Aggregation, adsorption, and morphological transformation of graphene oxide in aqueous solutions containing different metal caitons. Environ. Sci. Technol. 2016, 50, 11066–11075. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.B.; Pfefferle, L.D.; Elimelech, M. Influence of biomacromolecules and humic acid on the aggregation kinetics of single-walled carbon nanotubes. Environ. Sci. Technol. 2010, 44, 2412–2418. [Google Scholar] [CrossRef]
- Baalousha, M.; Motelica-Heino, M.; Coustumer, P.L. Conformation and size of humic substances: Effects of major cation concentration and type, pH, salinity, and residence time. Colloids Surf. A Physicochem. Eng. Aspects 2006, 272, 48–55. [Google Scholar] [CrossRef]
- Wei, X.; Pan, D.; Xu, Z.; Xian, D.; Li, X.; Tan, Z.; Liu, C.; Wu, W. Colloidal stability and correlated migration of illite in the aquatic environment: The roles of pH, temperature, multiple cations and humic acid. Sci. Total Environ. 2021, 768, 144174. [Google Scholar] [CrossRef]
- Christl, I.; Milne, C.J.; Kinniburgh, D.G.; Kretzschmar, R. Relating ion binding by fulvic and humic acids to chemical composition and molecular size. 2. Metal binding. Environ. Sci. Technol. 2001, 25, 2515–2517. [Google Scholar]
- Piccolo, A.; Conte, P.; Cozzolono, A. Effects of mineral and monocarboxylic acids on the molecular association of dissolved humic substances. Eur. J. Soil Sci. 1999, 50, 687–694. [Google Scholar] [CrossRef]
- Saleh, N.B.; Pfefferle, L.D.; Elimelech, M. Aggregation kinetics of multiwalled carbon nanotubes in aquatic systems: Measurements and environmental implications. Environ. Sci. Technol. 2008, 42, 7963–7969. [Google Scholar] [CrossRef]
- Liu, X.; Wazne, M.; Chou, T.; Xiao, R.; Xu, S. Influence of Ca2+ and Suwannee river humic acid on aggregation of silicon nanoparticles in aqueous media. Water Res. 2011, 45, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Lee, S. Effect of seepage velocity on the attachment efficiency of TiO2 nanoparticles in porous media. J. Hazard. Mater. 2014, 279, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Sheng, A.; Liu, F.; Shi, L.; Liu, J. Aggregation kinetics of hematite particles in the presence of outer membrane cytochrome Omca of Shewanella oneidenesis MR-1. Environ. Sci. Technol. 2016, 50, 11016–11024. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.; Okamura, J.; Shinkai, S. Colloidal nature of single-walled carbon nanotubes in electrolyte solution: The Schulze-Hardy rule. Langmuir 2001, 17, 7172–7173. [Google Scholar] [CrossRef]
- Li, J.; Zhao, Z.; Song, Y.; You, Y.; Li, J.; Cheng, X. Synthesis of Mg(II) doped ferrihydrite-humic acid coprecipitation and its Pb(II)/Cd(II) ion sorption mechanism. Chin. Chem. Lett. 2021, 32, 3231–3236. [Google Scholar] [CrossRef]
- Alvarez-Puebla, R.A.; Garrido, J.J.; Valenzuela-Calahorro, C.; Goulet, P.J.G. Retention and induced aggregation of Co(II) on a humic substances: Sorption isotherms, infrared absorption, and molecular modeling. Surf. Sci. 2005, 575, 136–146. [Google Scholar] [CrossRef]
- Ai, Y.; Zhao, C.; Sun, L.; Wang, X.; Liang, L. Coagulation mechanism of humic acid in metal ions solution under different pH conditions: A molecular dynamic simulation. Sci. Total Environ. 2021, 702, 135072. [Google Scholar] [CrossRef]
- Liao, Z.; Zhao, Z.; Zhu, J.; Chen, H. Complexation characteristics between Cu(II) ions and dissolved organic matter in combined sewer overflows: Implications for the removal of heavy metals by enhanced coagulation. Chemosphere 2021, 265, 129023. [Google Scholar] [CrossRef]
- Engebretson, R.R.; Wanaruszka, R.V. Kinetic aspects of cation-enhanced aggregation in aqueous humic acids. Environ. Sci. Technol. 1998, 32, 488–493. [Google Scholar] [CrossRef]
- Boguta, P.; D’Orazio, V.; Senesi, N.; Sokolowska, Z.; Szewczuk-Karpisz, K. Insight into the interaction mechanism of iron ions with soil humic acids. The effect of the pH and chemical properties of humic acids. J. Environ. Manag. 2019, 245, 367–374. [Google Scholar] [CrossRef]
- Tan, L.; Tan, X.; Mei, H.; Ai, Y.; Sun, L.; Zhao, G.; Hayat, T.; Alsaedi, A.; Chen, C.; Wang, X. Coagulation behavior of humic acid in aqueous solutions containing Cs+, Sr2+ and Eu3+: DLS, EEM and MD simulations. Environ. Pollut. 2018, 236, 835–843. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, L.; Wang, Y.; Wang, S.; Wu, C.; Li, D.; Chen, Y.; Wang, H. Aggregation Kinetics and Mechanism of Humic Acid Reaction with Cs+ and Co2+ Metal Ions Using Batch Techniques. Water 2022, 14, 2619. https://doi.org/10.3390/w14172619
Tan L, Wang Y, Wang S, Wu C, Li D, Chen Y, Wang H. Aggregation Kinetics and Mechanism of Humic Acid Reaction with Cs+ and Co2+ Metal Ions Using Batch Techniques. Water. 2022; 14(17):2619. https://doi.org/10.3390/w14172619
Chicago/Turabian StyleTan, Liqiang, Yuxiang Wang, Song Wang, Caijin Wu, Dong Li, Yisheng Chen, and Haocheng Wang. 2022. "Aggregation Kinetics and Mechanism of Humic Acid Reaction with Cs+ and Co2+ Metal Ions Using Batch Techniques" Water 14, no. 17: 2619. https://doi.org/10.3390/w14172619







