Enhancing Functionalities in Nanocomposites for Effective Dye Removal from Wastewater: Isothermal, Kinetic and Thermodynamic Aspects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Spectrophotometric Analysis
2.3. Preparation of Bentonite
2.4. Preparation of Composite Materials
2.5. Adsorption Study
2.6. Electrocoagulation
Estimation of Absorbance and TDS
2.7. Optimization of Initial Dye Concentration
Adsorption Isotherms
2.8. Optimization of Contact Time
2.9. Optimization of Temperature
2.10. Characterization of Adsorbents
3. Results and Discussion
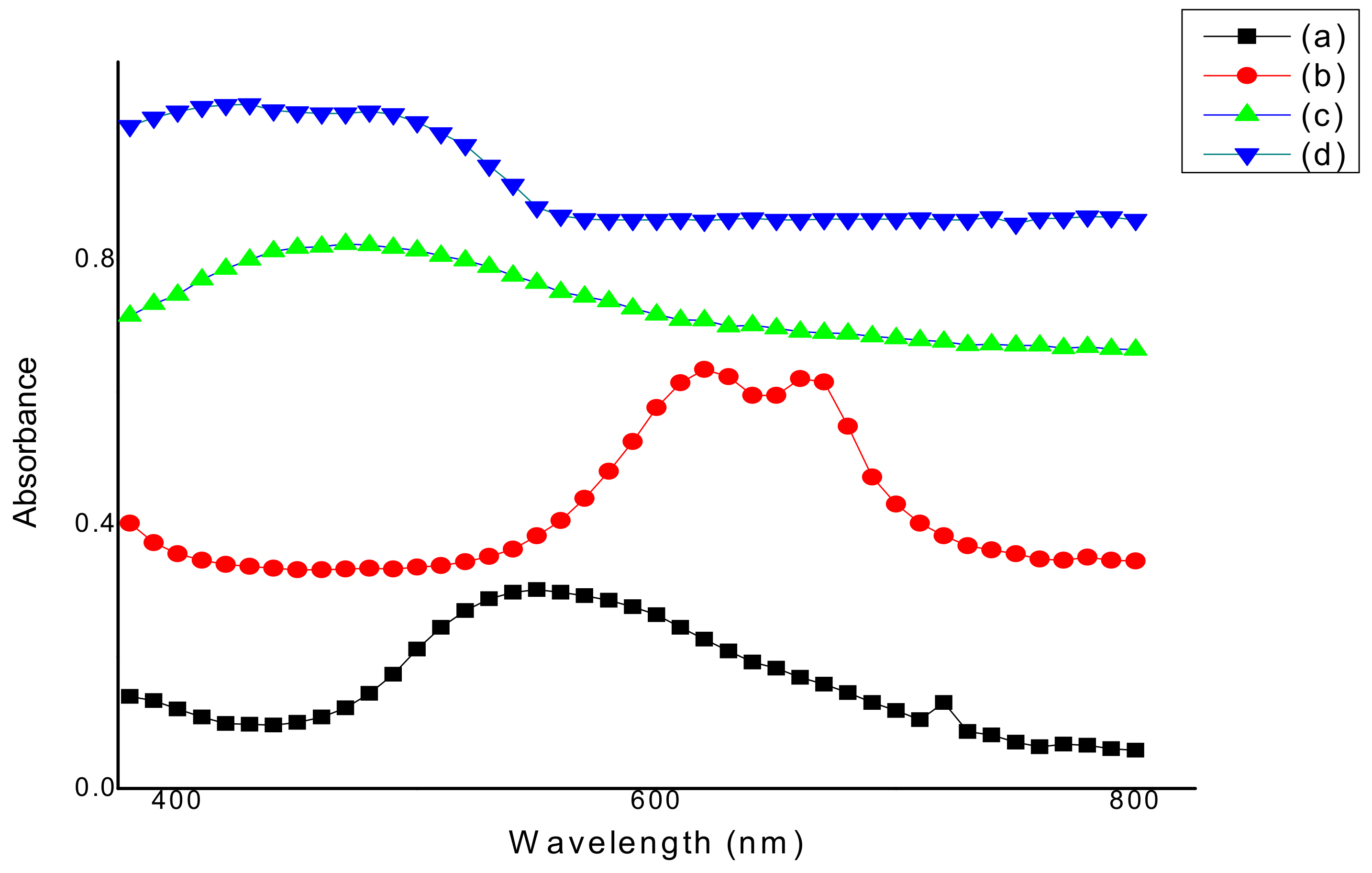
3.1. Determination of Maximum Wavelength (λmax)
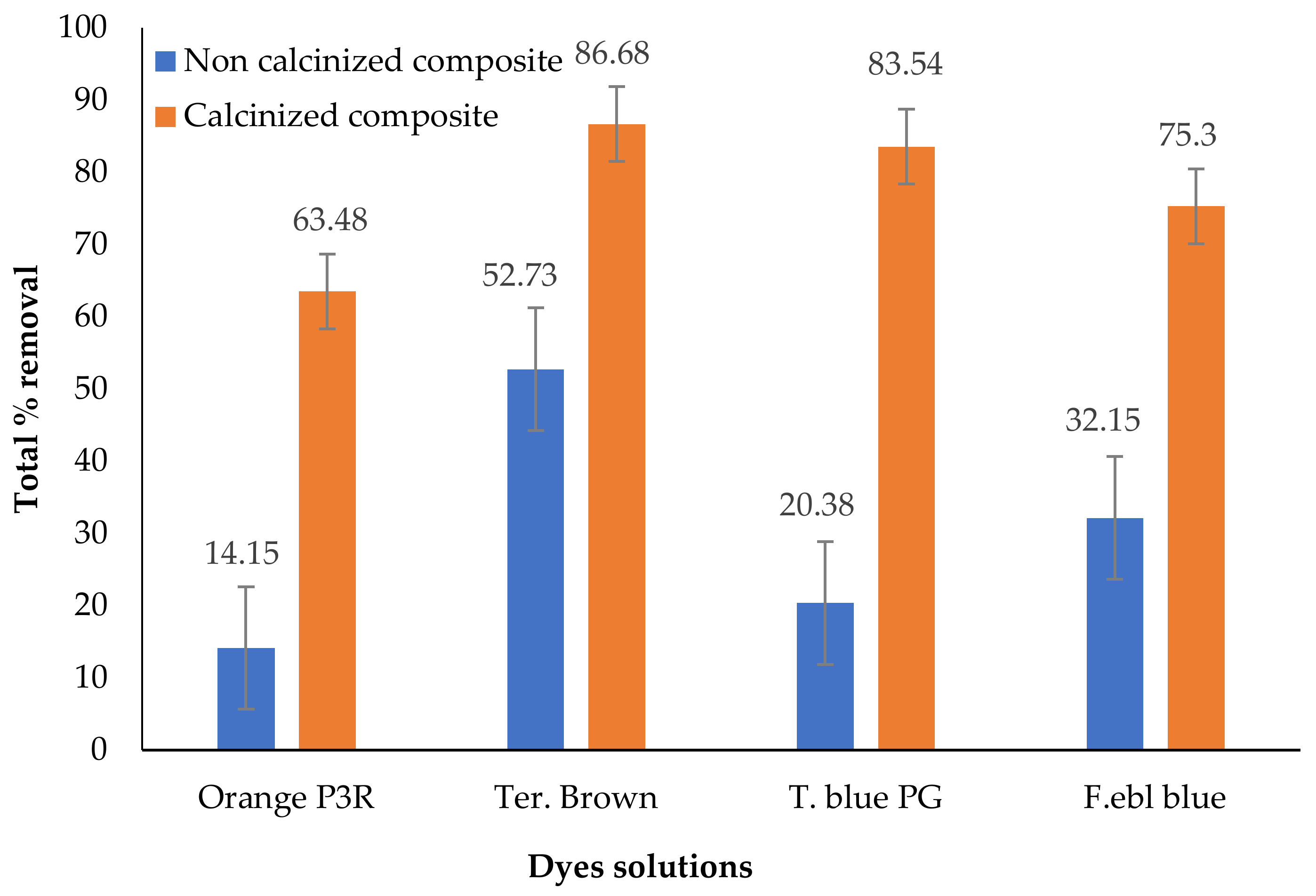
3.2. Effect of Calcination on Adsorption Capacity
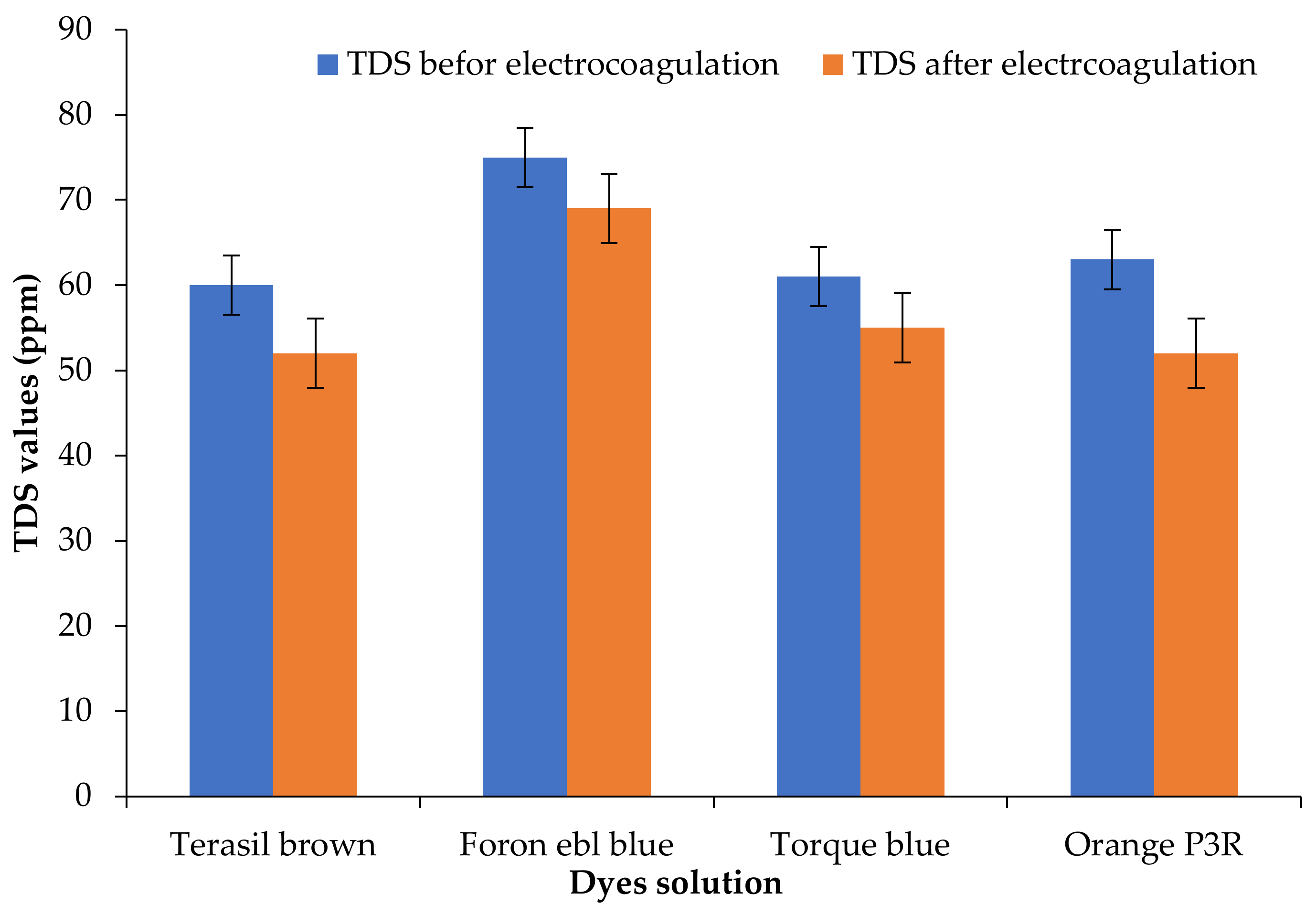
3.3. Effect of Electrocoagulation-Adsorption on Total Dissolved Solids (TDS)
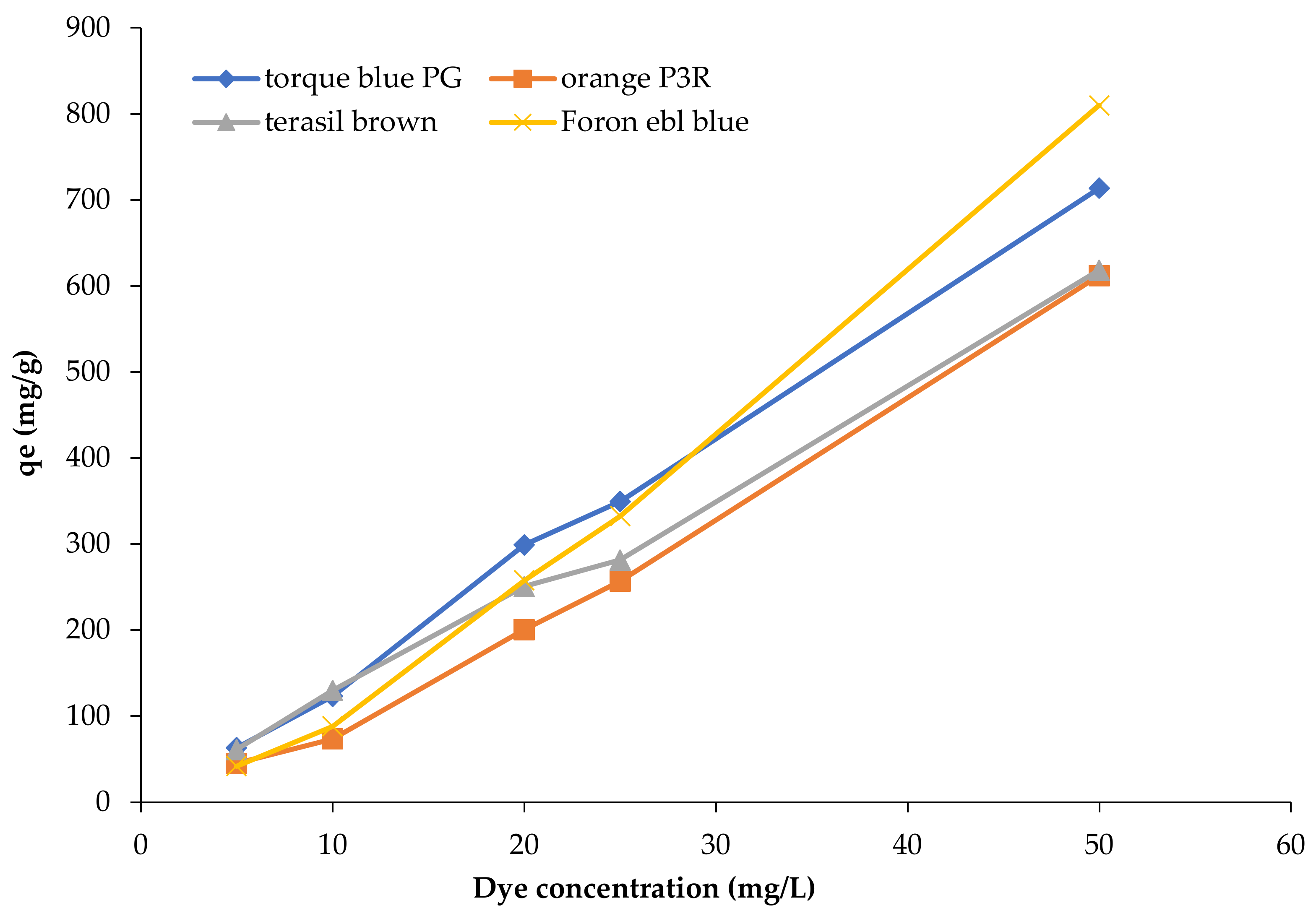
3.4. Effect of Initial Dye Concentration
3.5. Adsorption Modelling
3.6. Effect of Time Contact
3.7. Kinetic Studies
3.8. Thermodynamic Studies
3.9. SEM Analysis
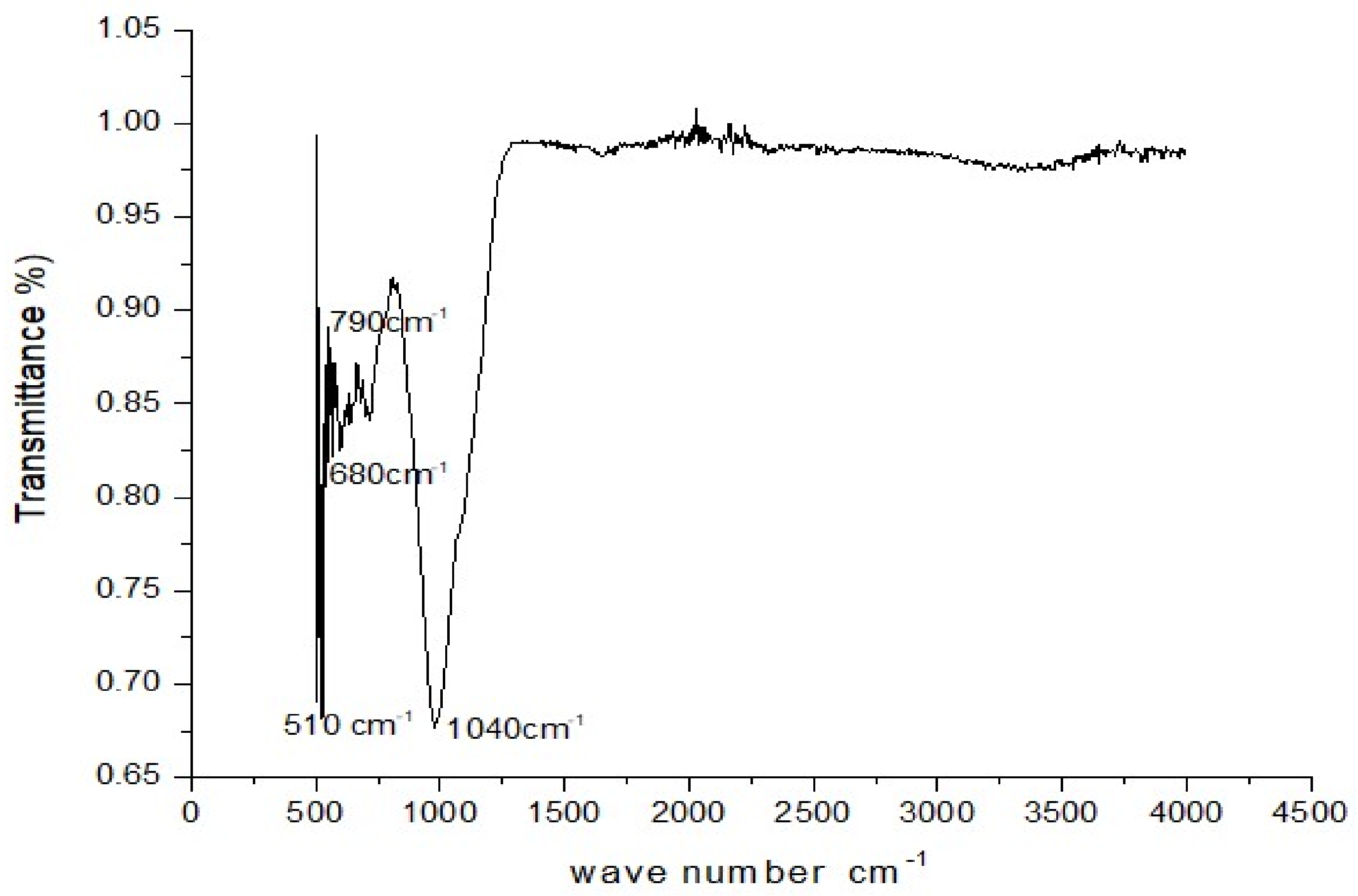
3.10. FTIR Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Buonocore, E.; Mellino, S.; De Angelis, G.; Liu, G.; Ulgiati, S. Life Cycle Assessment Indicators of Urban Wastewater and Sewage Sludge Treatment. Ecol. Indic. 2018, 94, 13–23. [Google Scholar] [CrossRef] [Green Version]
- Chopra, A.K.; Pathak, C. Accumulation of Heavy Metals in The Vegetables Grown in Wastewater Irrigated Areas of Dehradun, India with Reference to Human Health Risk. Environ. Monit. Assess. 2015, 187, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.; McMullan, G.; Marchant, R.; Nigam, P. Remediation of Dyes in Textile Effluent: A Critical Review on Current Treatment Technologies with A Proposed Alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Chu, W. Dye Removal from Textile Dye Wastewater Using Recycled Alum Sludge. Water Res. 2001, 35, 3147–3152. [Google Scholar] [CrossRef]
- Vakili, M.; Rafatullah, M.; Salamatinia, B.; Abdullah, A.Z.; Ibrahim, M.H.; Tan, K.B.; Gholami, Z.; Amouzgar, P. Application of Chitosan and Its Derivatives as Adsorbents for Dye Removal from Water and Wastewater: A Review. Carbohydr. Polym. 2014, 113, 115–130. [Google Scholar] [CrossRef]
- Ramesh, S.; Kim, H.S.; Sivasamy, A.; Kim, J.H. Synthesis of Octa (Maleimidophenyl) Silsesquioxane–Sio2/Tio2 Hybrid Nanocomposites: Adsorption Behavior for the Removal of an Organic Methylene Blue Dye and Antimicrobial Activity Against Pathogens. Polym. Plast. Technol. Eng. 2018, 57, 185–195. [Google Scholar] [CrossRef]
- Pandit, P.; Basu, S. Removal of Organic Dyes from Water by Liquid–Liquid Extraction Using Reverse Micelles. J. Colloid Interface Sci. 2002, 245, 208–214. [Google Scholar] [CrossRef]
- Khadhraoui, M.; Trabelsi, H.; Ksibi, M.; Bouguerra, S.; Elleuch, B. Discoloration and Detoxicification of a Congo Red Dye Solution by Means of Ozone Treatment for a Possible Water Reuse. J. Hazard. Mater. 2009, 161, 974–981. [Google Scholar] [CrossRef]
- Sarkheil, H.; Tavakoli, J.; Behnood, R. Oil By-Product Removal from Aqueous Solution Using Sugarcane Bagasse as Absorbent. Int. J. Emerg. Sci. Eng. 2014, 2, 48–52. [Google Scholar]
- Castañeda-Díaz, J.; Pavón-Silva, T.; Gutiérrez-Segura, E.E.; Colín-Cruz, A. Electrocoagulation-Adsorption to Remove Anionic and Cationic Dyes from Aqueous Solution by PV-Energy. J. Chem. 2017, 2017, 5184590. [Google Scholar] [CrossRef] [Green Version]
- Brillas, E.; Martínez-Huitle, C.A. Decontamination of Wastewaters Containing Synthetic Organic Dyes by Electrochemical Methods. An Updated Review. Appl. Catal. B Environ. 2015, 166, 603–643. [Google Scholar] [CrossRef]
- Mollah, M.Y.; Morkovsky, P.; Gomes, J.A.; Kesmez, M.; Parga, J.; Cocke, D.L. Fundamentals, Present and Future Perspectives of Electrocoagulation. J. Hazard. Mater. 2004, 114, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Vimonses, V.; Lei, S.; Jin, B.; Chow, C.W.; Saint, C. Adsorption of Congo Red by Three Australian Kaolins. Appl. Clay Sci. 2009, 43, 465–472. [Google Scholar] [CrossRef]
- Queiroga, L.N.; Pereira, M.B.; Silva, L.S.; Filho, E.C.S.; Santos, I.M.; Fonseca, M.G.; Georgelin, T.; Jaber, M. Microwave ben-tonite silylation for dye removal: Influence of the solvent. Appl. Clay Sci. 2019, 168, 478–487. [Google Scholar] [CrossRef]
- Mahmoodi, N.M.; Saffar-Dastgerdi, M.H. Zeolite Nanoparticle as a Superior Adsorbent with High Capacity: Synthesis, Surface Modification and Pollutant Adsorption Ability from Wastewater. Microchem. J. 2018, 145, 74–83. [Google Scholar] [CrossRef]
- Zhang, G.; Qu, J.; Liu, H.; Liu, R.; Wu, R. Preparation and Evaluation of a Novel Fe–Mn Binary Oxide Adsorbent for Effective Arsenite Removal. Water Res. 2007, 41, 1921–1928. [Google Scholar] [CrossRef]
- Şen, F.; Demirbaş, Ö.; Çalımlı, M.H.; Aygün, A.; Alma, M.H.; Nas, M.S. The Dye Removal from Aqueous Solution Using Polymer Composite Films. Appl. Water Sci. 2018, 8, 206. [Google Scholar] [CrossRef] [Green Version]
- Aljeboree, A.M.; Alshirifi, A.N.; Alkaim, A.F. Kinetics and Equilibrium Study for the Adsorption of Textile Dyes on Coconut Shell Activated Carbon. Arab. J. Chem. 2017, 10, S3381–S3393. [Google Scholar] [CrossRef] [Green Version]
- Pereira, I.D.S.; Silva, V.C.; Neto, J.F.D.; Neves, G.A.; Ferreira, H.C.; Menezes, R. Influence of the Purification of Bentonite Clay from New Deposits in the State of Paraíba-Brazil for Use in Water-Based Drilling Fluids. Cerâmica 2018, 64, 538–546. [Google Scholar] [CrossRef]
- Wang, Y. Solar Photocatalytic Degradation of Eight Commercial Dyes in TiO2 Suspension. Water Res. 2000, 34, 990–994. [Google Scholar] [CrossRef]
- Amin, M.T.; Alazba, A.A.; Shafiq, M. Adsorptive Removal of Reactive Black 5 from Wastewater Using Bentonite Clay: Isotherms, Kinetics and Thermodynamics. Sustainability 2015, 7, 15302–15318. [Google Scholar] [CrossRef] [Green Version]
- Dada, A.; Olalekan, A.; Olatunya, A.; Dada, O. Langmuir, Freundlich, Temkin and Dubinin–Radushkevich Isotherms Studies of Equilibrium Sorption of Zn2+ Unto Phosphoric Acid Modified Rice Husk. IOSR J. Appl. Chem. 2012, 3, 38–45. [Google Scholar]
- Khalil, M.; Hanif, M.A.; Rashid, U.; Ahmad, J.; Alsalme, A.; Tsubota, T. Low-Cost Novel Nano-Constructed Granite Com-Posites for Removal of Hazardous Terasil Dye from Wastewater. Environ. Sci. Pollut. Res. 2022, 1–19. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G.; Wase, D.A.J.; Forster, C.F. Study of the Sorption of Divalent Metal Ions on to Peat. Adsorpt. Sci. Technol. 2000, 18, 639–650. [Google Scholar] [CrossRef]
- Vimonses, V.; Lei, S.; Jin, B.; Chow, C.; Saint, C. Kinetic Study and Equilibrium Isotherm Analysis of Congo Red Adsorption by Clay Materials. Chem. Eng. J. 2009, 148, 354–364. [Google Scholar] [CrossRef]
- Zhao, X.; Hardin, I.R. HPLC and Spectrophotometric Analysis of Biodegradation of Azo Dyes by Pleurotus Ostreatus. Dye. Pigment. 2007, 73, 322–325. [Google Scholar] [CrossRef]
- Mahmoud, M.; Farah, J.Y.; Farrag, T.E. Enhanced removal of Methylene Blue by Electrocoagulation Using Iron Electrodes. Egypt. J. Pet. 2013, 22, 211–216. [Google Scholar] [CrossRef] [Green Version]
- Rejeb, R.; Antonissen, G.; De Boevre, M.; Detavernier, C.; Van de Velde, M.; De Saeger, S.; Ducatelle, R.; Ayed, M.H.; Ghorbal, A. Calcination Enhances the Aflatoxin and Zearalenone Binding Efficiency of a Tunisian Clay. Toxins 2019, 11, 602. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Guo, Y.; Tang, D.; Liu, Y.; Wang, X.; Li, P.; Wang, G. Sol-Gel Synthesis of New Znfe2o4/Na-Bentonite Composites for Simultaneous Oxidation of Rhb and Reduction of Cr (VI) Under Visible Light Irradiation. J. Alloy. Compd. 2019, 781, 1101–1109. [Google Scholar] [CrossRef]
- dos Santos, F.R.; de Oliveira Bruno, H.C.; Melgar, L.Z. Use of Bentonite Calcined Clay as An Adsorbent: Equilibrium and Ther-Modynamic Study of Rhodamine B Adsorption in Aqueous Solution. Environ. Sci. Pollut. Res. 2019, 26, 28622–28632. [Google Scholar] [CrossRef]
- Islam, A.; Guha, A.K. Removal of pH, TDS and Color from Textile Effluent by Using Coagulants and Aquatic/Non-Aquatic Plants as Adsorbents. Resour. Environ. 2013, 3, 101–114. [Google Scholar]
- Rusdianasari, R.; Bow, Y.; Yuniar, Y. Treatment of Traditional Cloth Wastewater by Electrocoagulation Using Aluminum Elec-Trodes. Int. J. Adv. Sci. Eng. Inf. Technol. 2014, 4, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.S.; Ramalingam, S.; Senthamarai, C.; Niranjanaa, M.; Vijayalakshmi, P.; Sivanesan, S. Adsorption of Dye from Aqueous Solution by Cashew Nutshell: Studies on Equilibrium Isotherm, Kinetics and Thermodynamics of Interactions. Desalination 2010, 261, 52–60. [Google Scholar] [CrossRef]
- Ozdes, D.; Duran, C.; Senturk, H.B.; Avan, H.; Bicer, B. Kinetics, Thermodynamics, and Equilibrium Evaluation of Adsorptive Removal of Methylene Blue onto Natural Illitic Clay Mineral. Desalin. Water Treat. 2013, 52, 208–218. [Google Scholar] [CrossRef]
- Shamsudin, M.S.; Azha, S.F.; Shahadat, M.; Ismail, S. Cellulose/Bentonite-Zeolite Composite Adsorbent Material Coating for Treatment of N-Based Antiseptic Cationic Dye from Water. J. Water Process. Eng. 2019, 29, 100764. [Google Scholar] [CrossRef]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Kinetics, Isotherms and Thermodynamic Modeling of Liquid Phase Adsorption of Rhodamine B Dye onto Raphia Hookerie Fruit Epicarp. Water Resour. Ind. 2016, 15, 14–27. [Google Scholar] [CrossRef] [Green Version]
- Arivoli, S.; Henkuzhali, M. Kinetic, Mechanistic, Thermodynamic and Equilibrium Studies on the Adsorption of Rhodamine B by Acid Activated Low-Cost Carbon. E J. Chem. 2008, 5, 187–200. [Google Scholar] [CrossRef] [Green Version]
- Foo, K.Y.; Hameed, B.H. Insights into the Modeling of Adsorption Isotherm Systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Loukidou, M.X.; Zouboulis, A.I.; Karapantsios, T.D.; Matis, K.A. Equilibrium and Kinetic Modeling of Chromium (VI) Biosorp-Tion by Aeromonas Caviae. Colloids Surf. A Physicochem. Eng. Aspects. 2004, 242, 93–104. [Google Scholar] [CrossRef]
- Bhatti, H.N.; Noreen, S.; Tahir, N.; Ilyas, S.; Siddiqua, U.H. Equilibrium, Thermodynamic and Kinetic Studies for Biosorption of Terasil Brown 2RFL From Contaminated Water Using Economical Biomaterial. Mediterr. J. Chem. 2015, 4, 239–251. [Google Scholar] [CrossRef]
- Sun, Q.; Hu, X.; Zheng, S.; Sun, Z.; Liu, S.; Li, H. Influence of Calcination Temperature on the Structural, Adsorption and Pho-Tocatalytic Properties of Tio2 Nanoparticles Supported on Natural Zeolite. Powder Technol. 2015, 274, 88–97. [Google Scholar] [CrossRef]
- Liu, Q.; Li, T.; Zhang, S.; Qu, L.; Ren, B. Optimization and Evaluation of Alkali-Pretreated Paeonia Ostii Seed Coats as Adsorbent for the Removal of Mb from Aqueous Solution. Pol. J. Chem. Technol. 2018, 20, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Özcan, A.S.; Erdem, B.; Özcan, A. Adsorption of Acid Blue 193 from Aqueous Solutions Onto BTMA-Bentonite. Colloids Surf. A Physicochem. Eng. Asp. 2005, 266, 73–81. [Google Scholar] [CrossRef]
- Vimonses, V.; Jin, B.; Chow, C.W. Insight into Removal Kinetic and Mechanisms of Anionic Dye by Calcined Clay Materials and Lime. J. Hazard. Mater. 2010, 177, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Shikuku, V.O.; Zanella, R.; Kowenje, C.O.; Donato, F.F.; Bandeira, N.M.; Prestes, O.D. Single and Binary Adsorption of Sul-Fonamide Antibiotics onto Iron-Modified Clay: Linear and Nonlinear Isotherms, Kinetics, Thermodynamics, And Mechanistic Studies. Appl. Water Sci. 2018, 8, 175. [Google Scholar] [CrossRef] [Green Version]
- Singh, K.P.; Mohan, D.; Sinha, S.; Tondon, G.; Gosh, D. Color removal from wastewater using low-cost activated carbon de-rived from agricultural waste material. Ind. Eng. Chem. Res. 2003, 42, 1965–1976. [Google Scholar] [CrossRef]
- Akl, M.; Youssef, A.; Al-Awadhi, M. Adsorption of Acid Dyes onto Bentonite and Surfactant-Modified Bentonite. J. Anal. Bioanal. Technol. 2013, 4, 3–7. [Google Scholar]
- Hua, J. Synthesis and Characterization of Bentonite Based Inorgano–Organo-Composites and Their Performances for Removing Arsenic from Water. Appl. Clay Sci. 2015, 114, 239–246. [Google Scholar] [CrossRef]
- Abdullahi, S.; Audu, A. Comparative Analysis on Chemical Composition of Bentonite Clays Obtained from Ashaka and Tango Deposits in Gombe State, Nigeria. ChemSearch J. 2017, 8, 35–40. [Google Scholar]
- Cótica, L.; Freitas, V.; Santos, I.; Barabach, M.; Anaissi, F.; Miyahara, R.; Sarvezuk, P. Cobalt-Modified Brazilian Bentonites: Preparation, Characterisation, and Thermal Stability. Appl. Clay Sci. 2010, 51, 187–191. [Google Scholar] [CrossRef] [Green Version]
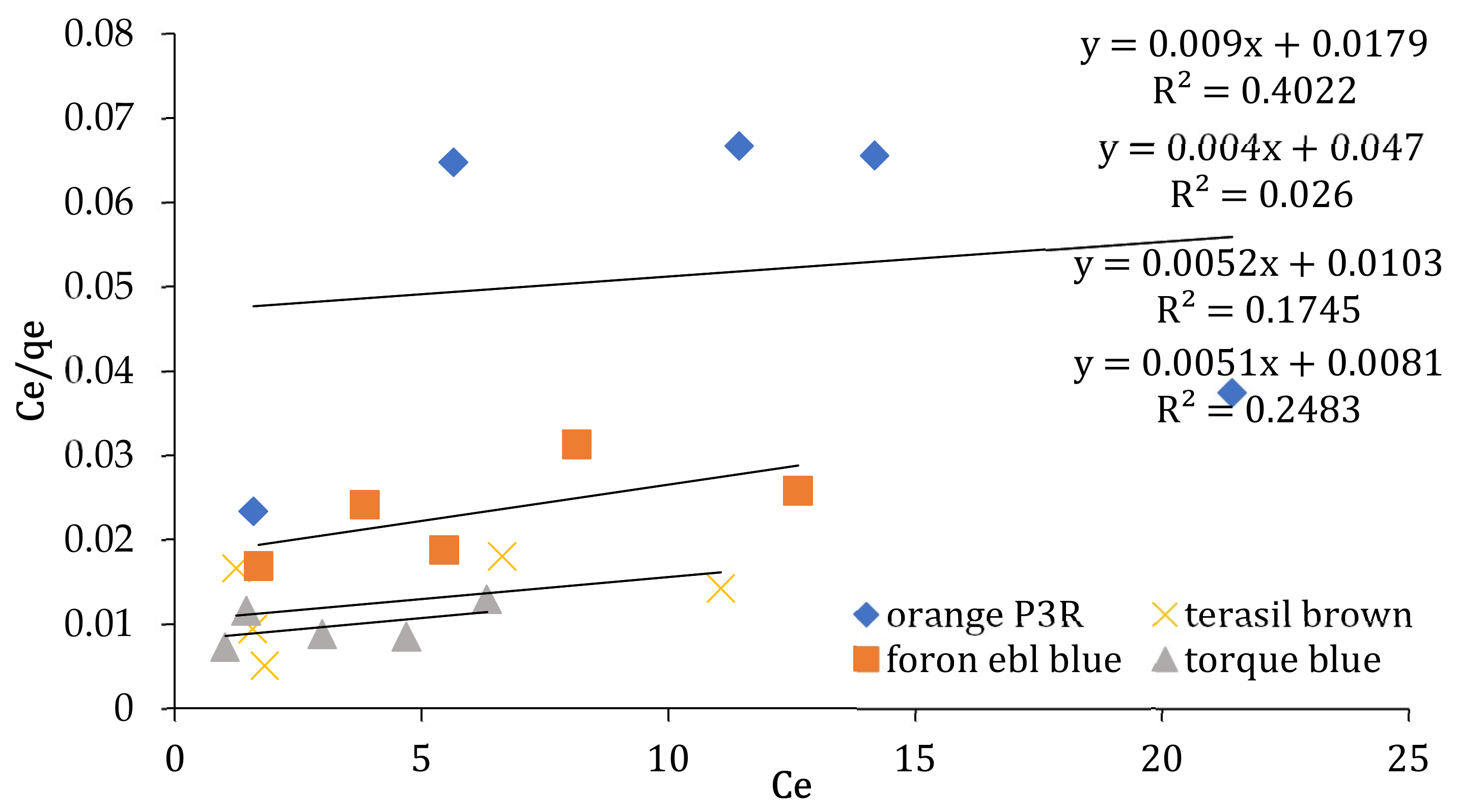
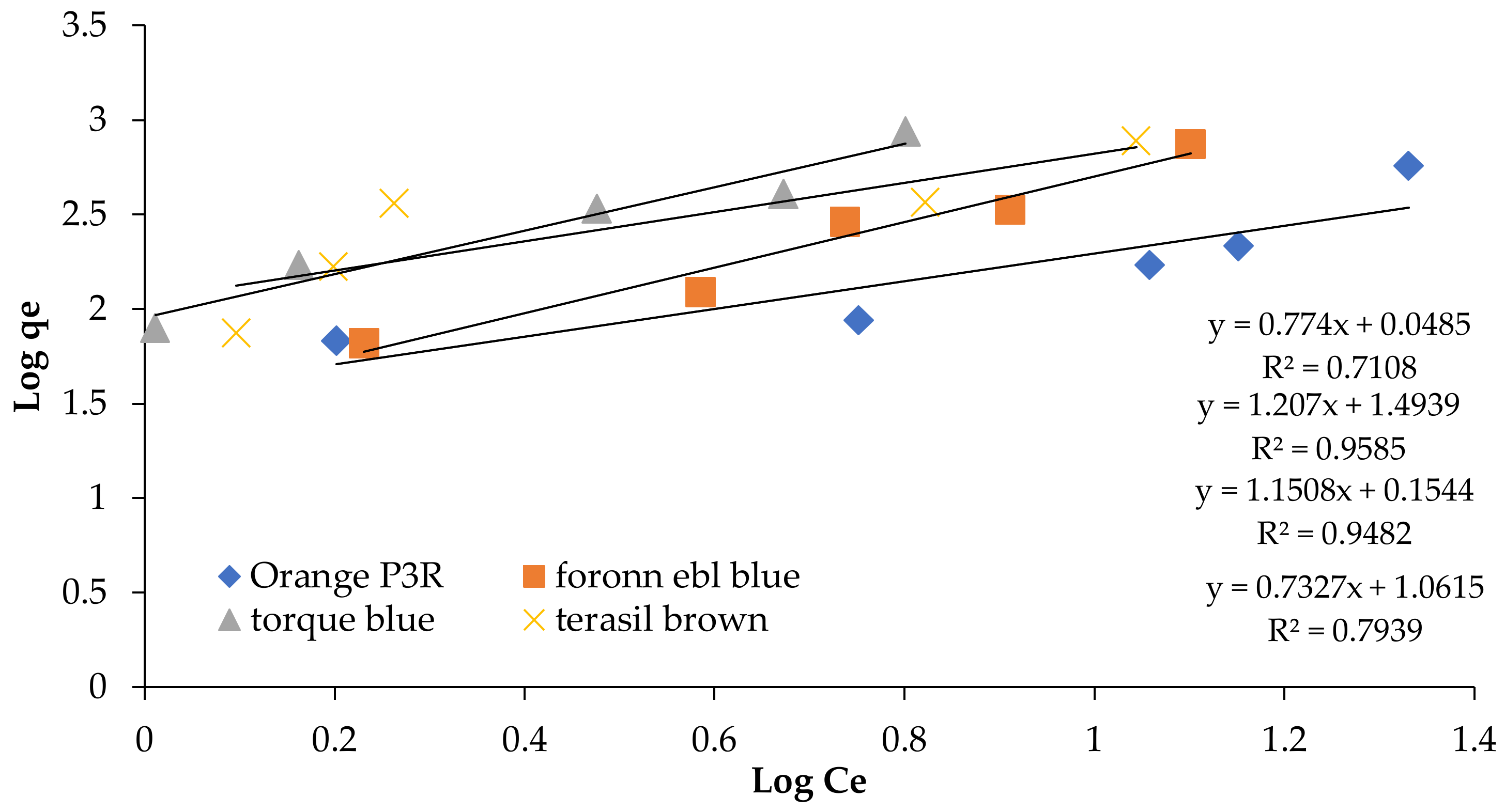
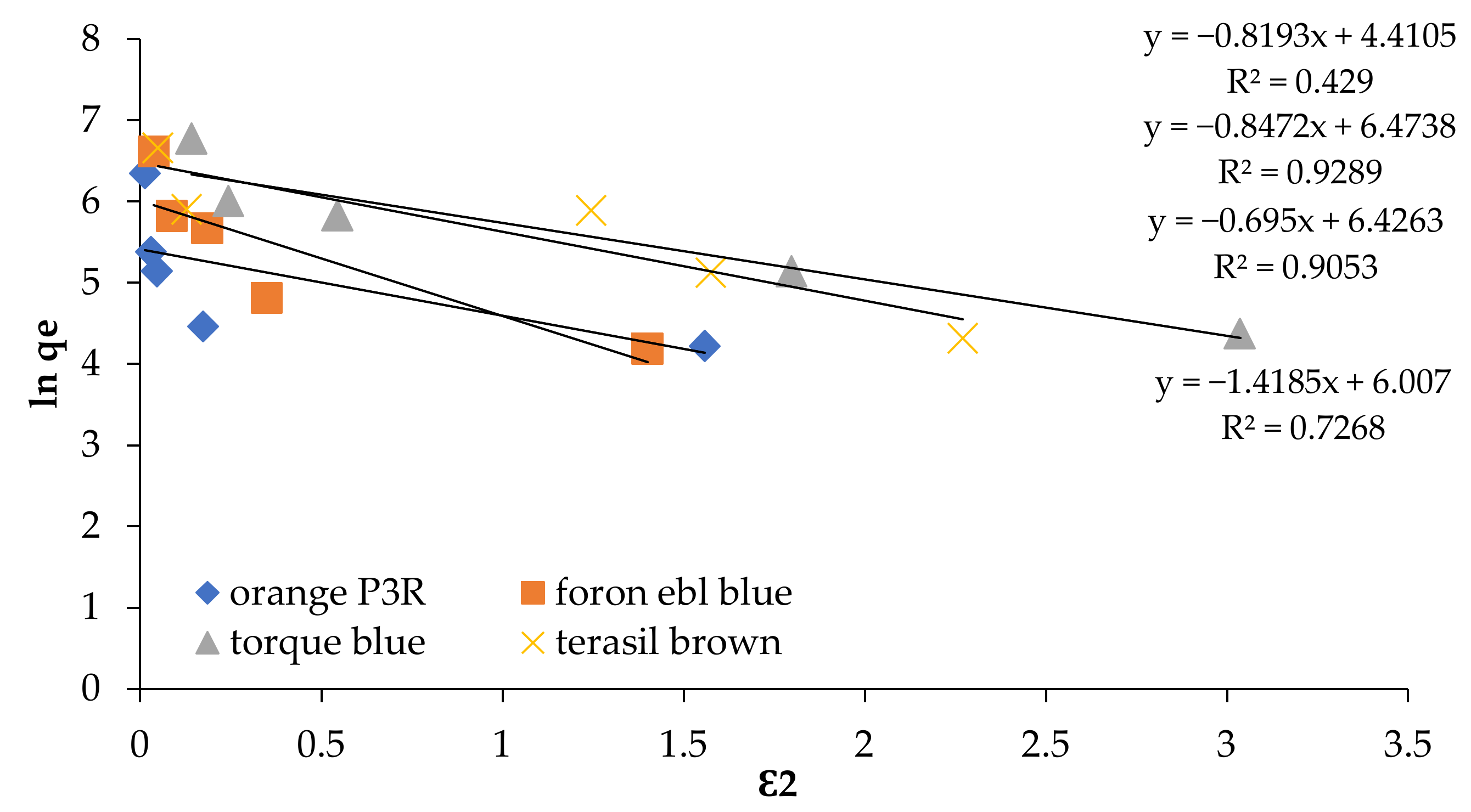
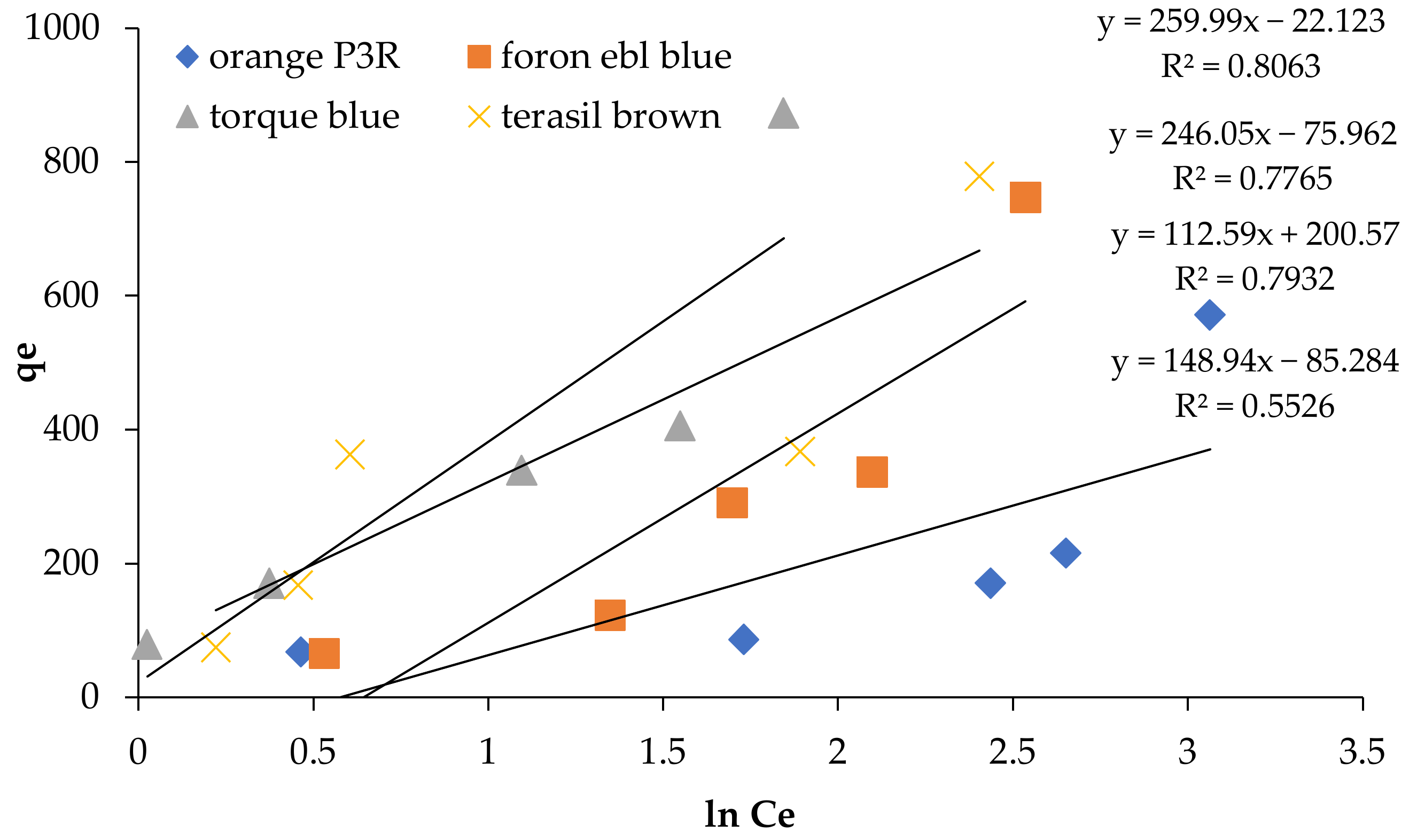
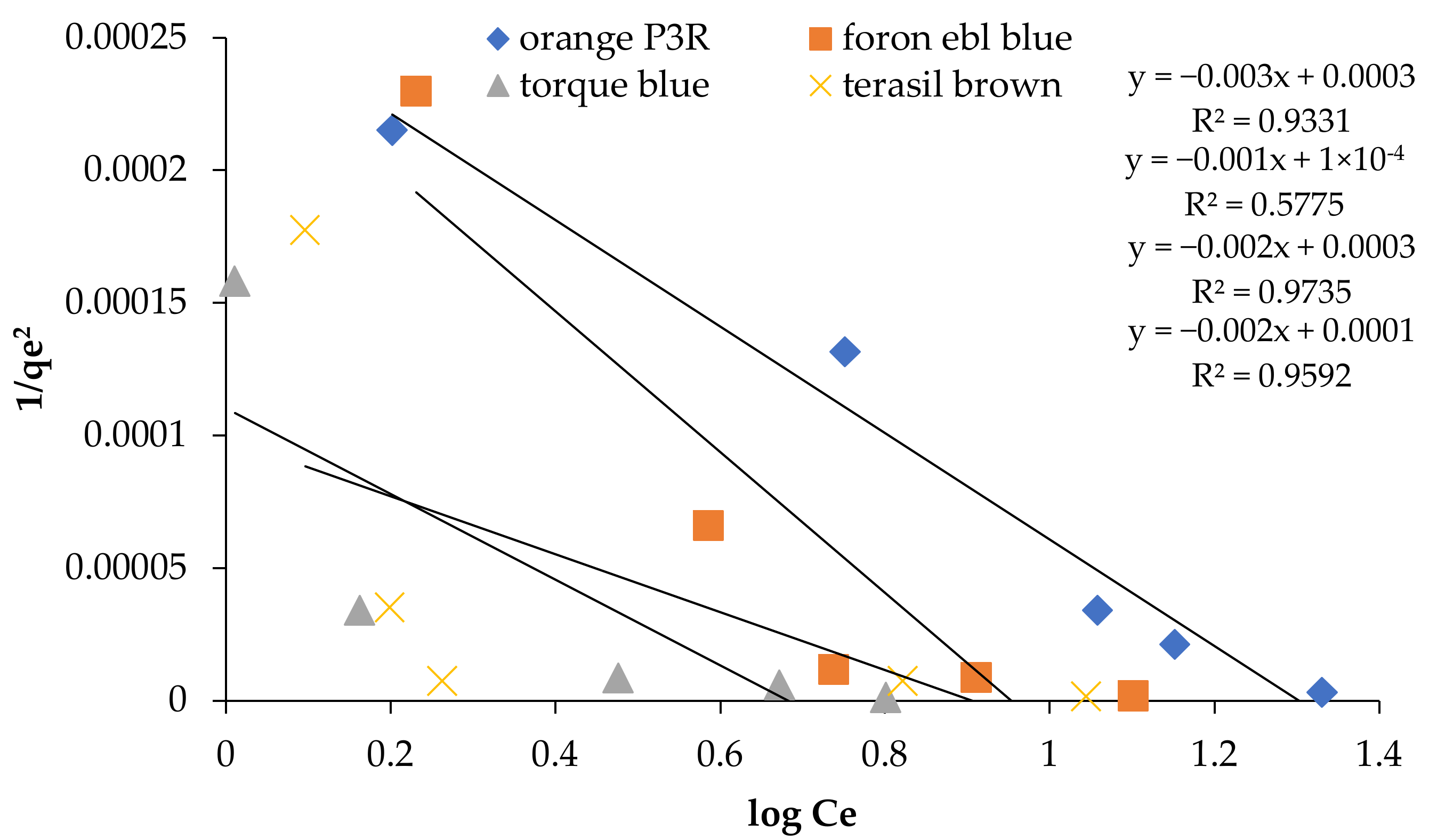
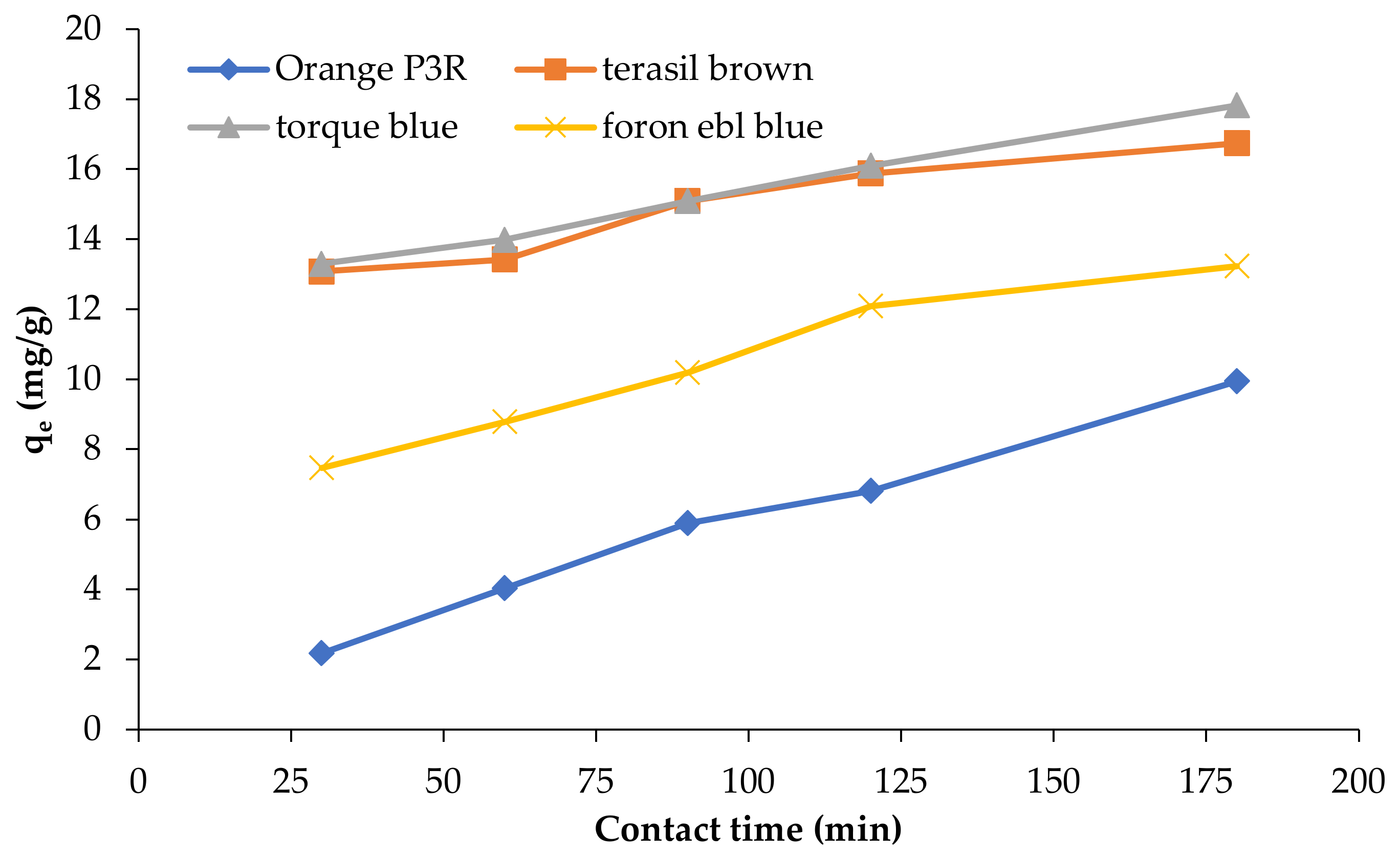

| Isotherm Models | Foron Ebl Blue Dye | Terasil Brown Dye | Orange P3R | Torque Blue PG Dye |
|---|---|---|---|---|
| Langmuir | ||||
| qmax (mg/g) (cal) | 111.11 | 112.30 | 150 | 104.07 |
| qmax(exp) | 747.4408 | 778.688 | 571.688 | 873.4896 |
| KL (L/mg) | 0.0179 | 0.0103 | 0.047 | 0.0081 |
| R2 | 0.4022 | 0.1745 | 0.026 | 0.2483 |
| Fruendlich | ||||
| qmax (cal) (mg/g) | 95.509 | 192.623 | 180.517 | 196.55 |
| qmax (exp) | 747.44 | 788.688 | 571.688 | 878.48 |
| R2 | 0.9585 | 0.7108 | 0.7939 | 0.9482 |
| Kf (L/mg) | 3.18 | 1.118 | 0.037 | 1.426 |
| N | 2.075 | 2.109 | 1.3648 | 0.868 |
| Dubinin-Radushkevich | ||||
| qo (mg/g) cal | 149.54 | 647.36 | 123.74 | 617.88 |
| qo (exp) | 747.44 | 788.688 | 571.688 | 878.48 |
| β (mol2/J2) | 1.4185 | 0.8472 | 0.8193 | 0.695 |
| R2 | 0.7268 | 0.9289 | 0.4297 | 0.9053 |
| E (KJ/mol) | 0.419 | 0.5432 | 0.55239 | 0.5997 |
| Temkin isotherm | ||||
| B | 112.59 | 146.04 | 148.94 | 159.99 |
| R2 | 0.7932 | 0.7765 | 0.5526 | 0.8063 |
| At (L/g) | 0.5774 | 1.1753 | 0.5545 | 2.464 |
| Harkin Jura isotherm | ||||
| R2 | 0.9931 | 0.5775 | 0.9735 | 0.9592 |
| A | 333.3 | 703 | 412 | 500 |
| B | 0.099 | 0.1 | 0.15 | 0.5 |
| Isotherm Models | Foron Ebl Blue Dye | Terasil Brown Dye | Orange P3R | Torque Blue PG Dye |
|---|---|---|---|---|
| Langmuir | ||||
| qmax (mg/g)(cal) | 109.89 | 85 | 151.51 | 175.33 |
| qmax(exp) | 810.13 | 618.48 | 612.204 | 713.57 |
| KL (L/mg) | 0.0214 | 0.0206 | 0.0037 | 0.0167 |
| R2 | 0.8425 | 0.7665 | 0.5251 | 0.4608 |
| Fruendlich | ||||
| qmax (cal) (mg/g) | 125.81 | 136.3699 | 223.738 | 218.68 |
| qmax(exp) | 810.13 | 618.48 | 612.204 | 713.57 |
| R2 | 0.8932 | 0.9795 | 0.9402 | 0.9536 |
| Kf (L/mg) | 2.6503 | 5.156 | 8.994 | 3.0704 |
| N | 1.037 | 1.0548 | 0.738 | 0.828 |
| Dubinin-Radushkevich | ||||
| qo (mg/g)cal | 145.65 | 363.72 | 283.36 | 617.099 |
| qo (exp) | 810.13 | 618.48 | 612.204 | 713.57 |
| β (mol2/J2) | 4.6532 | 1.6845 | 3.21911 | 1.6933 |
| R2 | 0.7268 | 0.9287 | 0.7297 | 0.9143 |
| E (KJ/mol) | 0.2318 | 0.3852 | 0.2786 | 0.3842 |
| Temkin isotherm | ||||
| B | 130.83 | 117.83 | 162.4 | 114.67 |
| R2 | 0.7773 | 0.8543 | 0.7333 | 0.8907 |
| At (L/g) | 0.5125 | 0.4791 | 0.1972 | 0.0222 |
| Harkin Jura Isotherm | ||||
| R2 | 0.9411 | 0.713 | 0.9035 | 0.9386 |
| A | 416.66 | 500 | 333.3 | 166.67 |
| B | 0.916 | 0.15 | 0.1 | 0.116 |
| Dyes | Pseudo First Order Kinetics | Pseudo Second Order Kinetics | |||||
|---|---|---|---|---|---|---|---|
| qe (mg/g) Cal | K1,ads | R2 | qe (mg/g) exp | qe (mg/g) | K2,ads | R2 | |
| Electrocoagulation time intervals after 30 min | |||||||
| Foron ebl blue | 4.637565 | 0.05893 | 0.7957 | 12.1187 | 13.5318 | 0.022636 | 0.9918 |
| Terasil brown | 5.807644 | 0.06813 | 0.8063 | 18.0032 | 18.60784 | 0.020365 | 0.9964 |
| Orange P3R | 16.11759 | 0.08931 | 0.8752 | 12.70712 | 21.72727 | 0.00556 | 0.9525 |
| Torque blue | 2.736529 | 0.05639 | 0.89 | 17.9484 | 19.08397 | 0.024481 | 0.9957 |
| Electrocoagulation time intervals after 60 min | |||||||
| Foron ebl blue | 2.782915 | 0.04235 | 0.7365 | 11.617 | 12.42236 | 0.035351 | 0.9964 |
| Terasil brown | 5.82908 | 0.06169 | 0.877 | 19.068 | 19.40816 | 0.020867 | 0.9964 |
| Orange P3R | 11.55846 | 0.08356 | 0.8606 | 12.244 | 18.01802 | 0.007998 | 0.9322 |
| Torque blue | 3.624933 | 0.04880 | 0.8571 | 19.1793 | 20.048 | 0.031247 | 0.998 |
| Electrocoagulation time intervals after 90 min | |||||||
| Foron ebl blue | 1.695509 | 0.032228 | 0.3254 | 12.3336 | 12.87001 | 0.054284 | 0.9988 |
| Terasil brown | 2.200392 | 0.038213 | 0.5002 | 18.6022 | 19.19386 | 0.046535 | 0.9993 |
| Orange P3R | 10.65124 | 0.082181 | 0.8838 | 15.6011 | 19.37984 | 0.010362 | 0.9978 |
| Torque blue | 3.053515 | 0.046731 | 0.725 | 18.2904 | 19.08397 | 0.035842 | 0.9991 |
| Electrocoagulation time intervals after 120 min | |||||||
| Foron ebl blue | 0.94973 | 0.019107 | 0.826 | 13.9457 | 14.28571 | 0.086855 | 0.9997 |
| Terasil brown | 3.423734 | 0.047191 | 0.8186 | 19.267 | 20.12072 | 0.056117 | 0.9978 |
| Orange P3R | 14.65548 | 0.091159 | 0.8787 | 16.7587 | 20.27171 | 0.007611 | 0.9979 |
| Torque blue | 3.053515 | 0.046731 | 0.8707 | 19.3845 | 20.20202 | 0.030037 | 0.9969 |
| Electrocoagulation time intervals after 180 min | |||||||
| Foron ebl blue | 2.875411 | 0.04558 | 0.6214 | 15.91606 | 16.72241 | 0.036117 | 0.9978 |
| Terasil brown | 2.293507 | 0.048342 | 0.3587 | 19.3344 | 20.08032 | 0.044437 | 0.9996 |
| Orange P3R | 8.340652 | 0.075275 | 0.8146 | 17.569 | 19.80198 | 0.014567 | 0.9908 |
| Torque blue | 1.663796 | 0.024631 | 0.4924 | 19.8632 | 20.28398 | 0.056828 | 0.999 |
| Dyes | Pseudo First Order Kinetics | Pseudo Second Order Kinetics | |||||
|---|---|---|---|---|---|---|---|
| qe (mg/g) Cal | K1,ads | R2 | qe (mg/g) exp | qe (mg/g) | K2,ads | R2 | |
| Electrocoagulation time intervals after 30 min | |||||||
| Foron ebl blue | 1.570724 | 0.035451 | 0.2292 | 12.9432 | 13.51351 | 0.056796 | 0.999 |
| Terasil brown | 6.757717 | 0.066298 | 0.8794 | 15.74016 | 17.51313 | 0.017227 | 0.993 |
| Orange P3R | 7.120328 | 0.070441 | 0.8355 | 8.2 | 13.2626 | 0.010422 | 0.9859 |
| Torque blue | 4.983105 | 0.063075 | 0.8043 | 12.4096 | 14.0647 | 0.020893 | 0.9945 |
| Electrocoagulation time intervals after 60 min | |||||||
| Foron ebl blue | 1.631924 | 0.030386 | 0.3308 | 13.588 | 14.08451 | 0.056654 | 0.9992 |
| Terasil brown | 5.556483 | 0.06952 | 0.7120 | 16.47232 | 18.11594 | 0.020705 | 0.9958 |
| Orange P3R | 10.03095 | 0.083102 | 0.8883 | 12.01256 | 18.58736 | 0.007411 | 0.9594 |
| Torque blue | 5.642869 | 0.065377 | 0.8424 | 13.9144 | 15.625 | 0.019432 | 0.993 |
| Electrocoagulation time intervals after 90 min | |||||||
| Foron ebl blue | 2.340453 | 0.035911 | 0.7134 | 14.412 | 15.01502 | 0.042644 | 0.9979 |
| Terasil brown | 6.907168 | 0.070671 | 0.8690 | 17.53728 | 20.37984 | 0.017332 | 0.993 |
| Orange P3R | 9.89464 | 0.079649 | 0.8693 | 12.24408 | 16.6113 | 0.009786 | 0.9786 |
| Torque blue | 5.735879 | 0.060312 | 0.8831 | 15.4872 | 19.89189 | 0.019987 | 0.9927 |
| Electrocoagulation time intervals after 120 min | |||||||
| Foron ebl blue | 1.588913 | 0.028545 | 0.3170 | 14.1256 | 14.59854 | 0.057211 | 0.9988 |
| Terasil brown | 5.663697 | 0.062845 | 0.6409 | 18.0032 | 19.41748 | 0.021089 | 0.9969 |
| Orange P3R | 13.03137 | 0.087246 | 0.8767 | 15.48536 | 22.66116 | 0.007715 | 0.9671 |
| Torque blue | 5.410034 | 0.062614 | 0.8932 | 16.64918 | 16.08318 | 0.021385 | 0.9962 |
| Electrocoagulation time intervals after 180 min | |||||||
| Foron ebl blue | 2.198872 | 0.048572 | 0.2993 | 15.7728 | 14.59854 | 0.032246 | 0.9988 |
| Terasil brown | 5.430002 | 0.061694 | 0.8270 | 18.93504 | 19.28398 | 0.022101 | 0.9971 |
| Orange P3R | 9.088663 | 0.079879 | 0.8891 | 15.60112 | 18.45018 | 0.012665 | 0.9868 |
| Torque blue | 6.546362 | 0.067449 | 0.8396 | 18.01688 | 16.72387 | 0.018392 | 0.9957 |
| Dyes | T (Kelvin) | ∆G (kJmol−1) | ∆H (J/mol) | ∆S (J mol−1 K−1) | R2 |
|---|---|---|---|---|---|
| Orange P3R | 303 | 7.381 | −8.4638 | −24.3938 | 0.9509 |
| 308 | 7.503 | ||||
| 313 | 7.625 | ||||
| 318 | 7.747 | ||||
| 328 | 7.991 | ||||
| Torque blue PG | 303 | 7.029 | −8.8605 | −23.233 | 0.7805 |
| 308 | 7.145 | ||||
| 313 | 7.262 | ||||
| 318 | 7.378 | ||||
| 328 | 7.61 | ||||
| Terasil brown 2RFL | 303 | 3.38 | −4.619 | −11.18 | 0.6429 |
| 308 | 3.438 | ||||
| 313 | 3.494 | ||||
| 318 | 3.55 | ||||
| 328 | 3.662 | ||||
| Foron ebl blue | 303 | 2.063 | −3.1669 | −6.8205 | 0.6285 |
| 308 | 2.097 | ||||
| 313 | 2.131 | ||||
| 318 | 2.165 | ||||
| 328 | 2.23 |
| Dyes | T (Kelvin) | ∆G (kJmol−1) | ∆H (J/mol) | ∆S (J mol−1 K−1) | R2 |
|---|---|---|---|---|---|
| Orange P3R | 303 | 2.126 | −3.245 | −7.02973 | 0.9441 |
| 308 | 2.160 | ||||
| 313 | 2.196 | ||||
| 318 | 2.231 | ||||
| 328 | 2.302 | ||||
| Torque blue PG | 303 | 8.658 | −1.0704 | −28.5883 | 0.9244 |
| 308 | 8.801 | ||||
| 313 | 8.94 | ||||
| 318 | 9.087 | ||||
| 328 | 9.373 | ||||
| Terasil brown 2RFL | 303 | 3.791 | −5.5052 | −12.5358 | 0.6268 |
| 308 | 3.853 | ||||
| 313 | 3.916 | ||||
| 318 | 3.979 | ||||
| 328 | 4.1073 | ||||
| Foron ebl blue | 303 | 2.3405 | −3.8039 | −7.73755 | 0.5832 |
| 308 | 2.379 | ||||
| 313 | 2.417 | ||||
| 318 | 2.456 | ||||
| 328 | 2.533 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Javed, I.; Hanif, M.A.; Rashid, U.; Nadeem, F.; Alharthi, F.A.; Kazerooni, E.A. Enhancing Functionalities in Nanocomposites for Effective Dye Removal from Wastewater: Isothermal, Kinetic and Thermodynamic Aspects. Water 2022, 14, 2600. https://doi.org/10.3390/w14172600
Javed I, Hanif MA, Rashid U, Nadeem F, Alharthi FA, Kazerooni EA. Enhancing Functionalities in Nanocomposites for Effective Dye Removal from Wastewater: Isothermal, Kinetic and Thermodynamic Aspects. Water. 2022; 14(17):2600. https://doi.org/10.3390/w14172600
Chicago/Turabian StyleJaved, Ifrah, Muhammad Asif Hanif, Umer Rashid, Farwa Nadeem, Fahad A. Alharthi, and Elham Ahmed Kazerooni. 2022. "Enhancing Functionalities in Nanocomposites for Effective Dye Removal from Wastewater: Isothermal, Kinetic and Thermodynamic Aspects" Water 14, no. 17: 2600. https://doi.org/10.3390/w14172600
APA StyleJaved, I., Hanif, M. A., Rashid, U., Nadeem, F., Alharthi, F. A., & Kazerooni, E. A. (2022). Enhancing Functionalities in Nanocomposites for Effective Dye Removal from Wastewater: Isothermal, Kinetic and Thermodynamic Aspects. Water, 14(17), 2600. https://doi.org/10.3390/w14172600










