Removal of Chromium (VI) from the Steel Mill Effluents Using the Chemically Modified Leaves of Pteris vittata as Adsorbent
Abstract
:1. Introduction
2. Methods and Materials
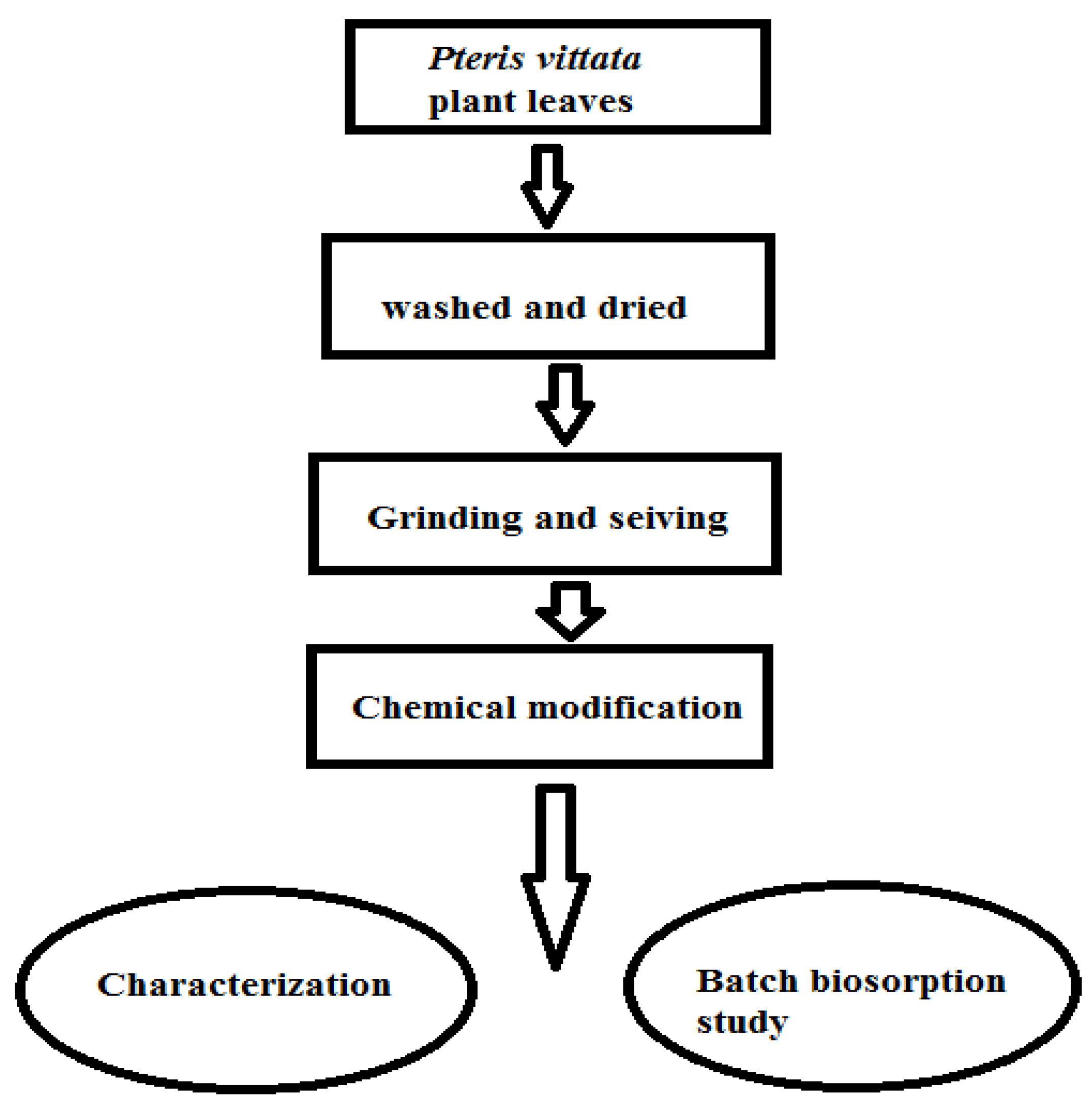
2.1. Preparation of Adsorbent
2.2. Chemical Modification of Biosorbent
2.3. Characterization of Biosorbent
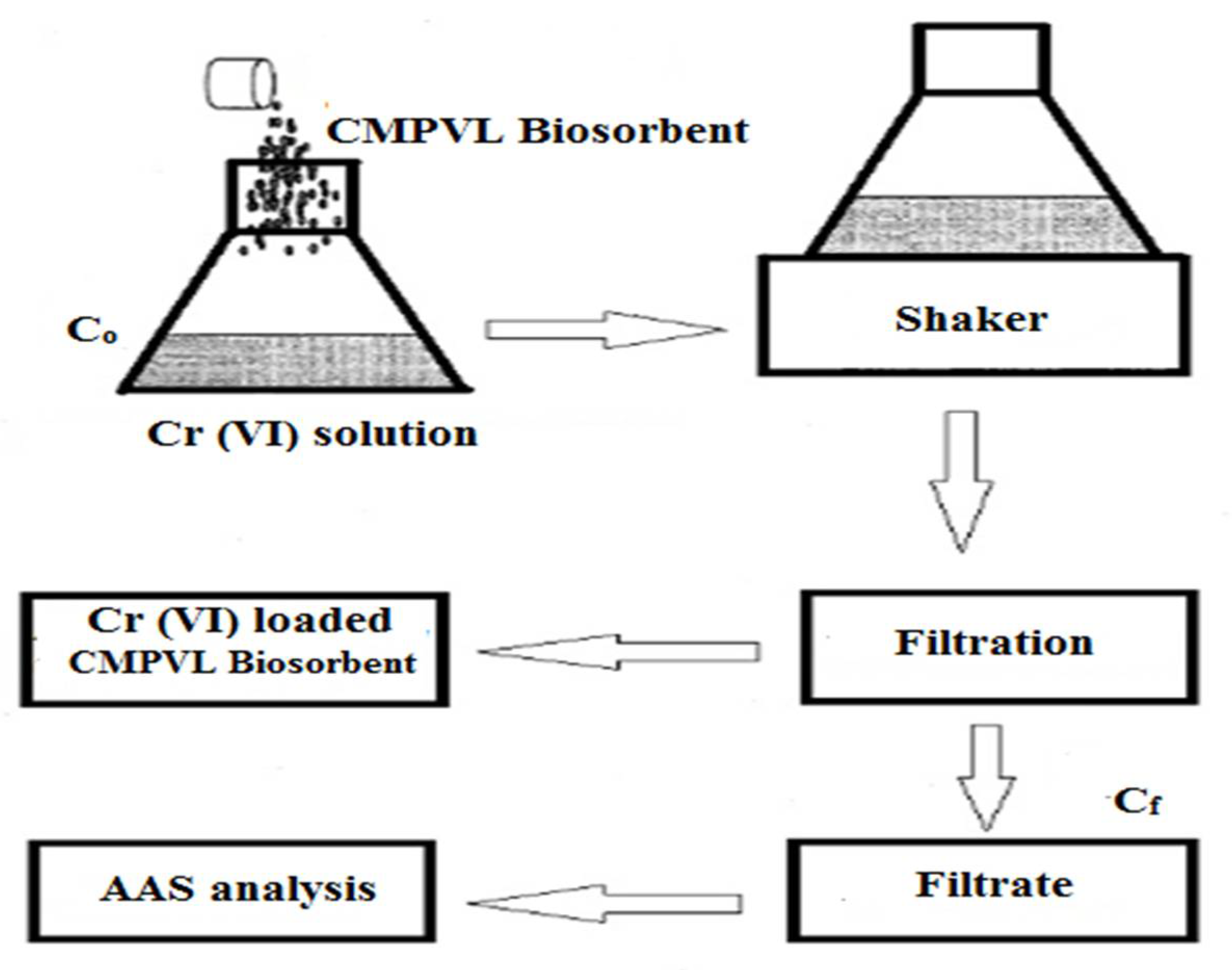
2.4. Batch Adsorption Experiment
2.5. Effect of Operating Physicochemical Parameters on the Sorption Process
2.6. Kinetic Study
3. Result and Discussion
3.1. Characterization of CMPVL
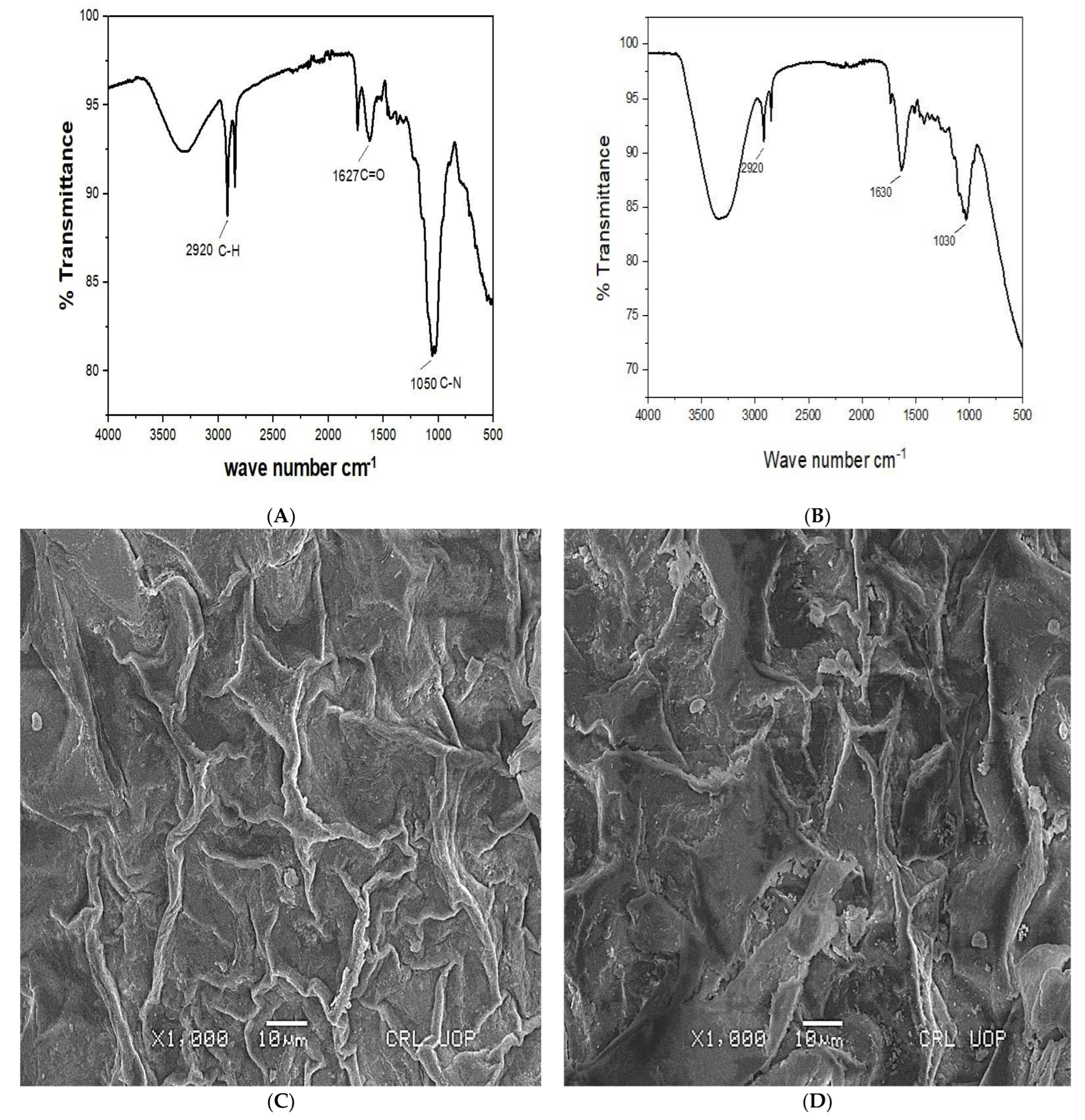
3.1.1. FTIR Spectra of Unloaded and Loaded Cr (VI) CMPVL
3.1.2. Surface Area and Pore Volume
3.1.3. SEM Analysis
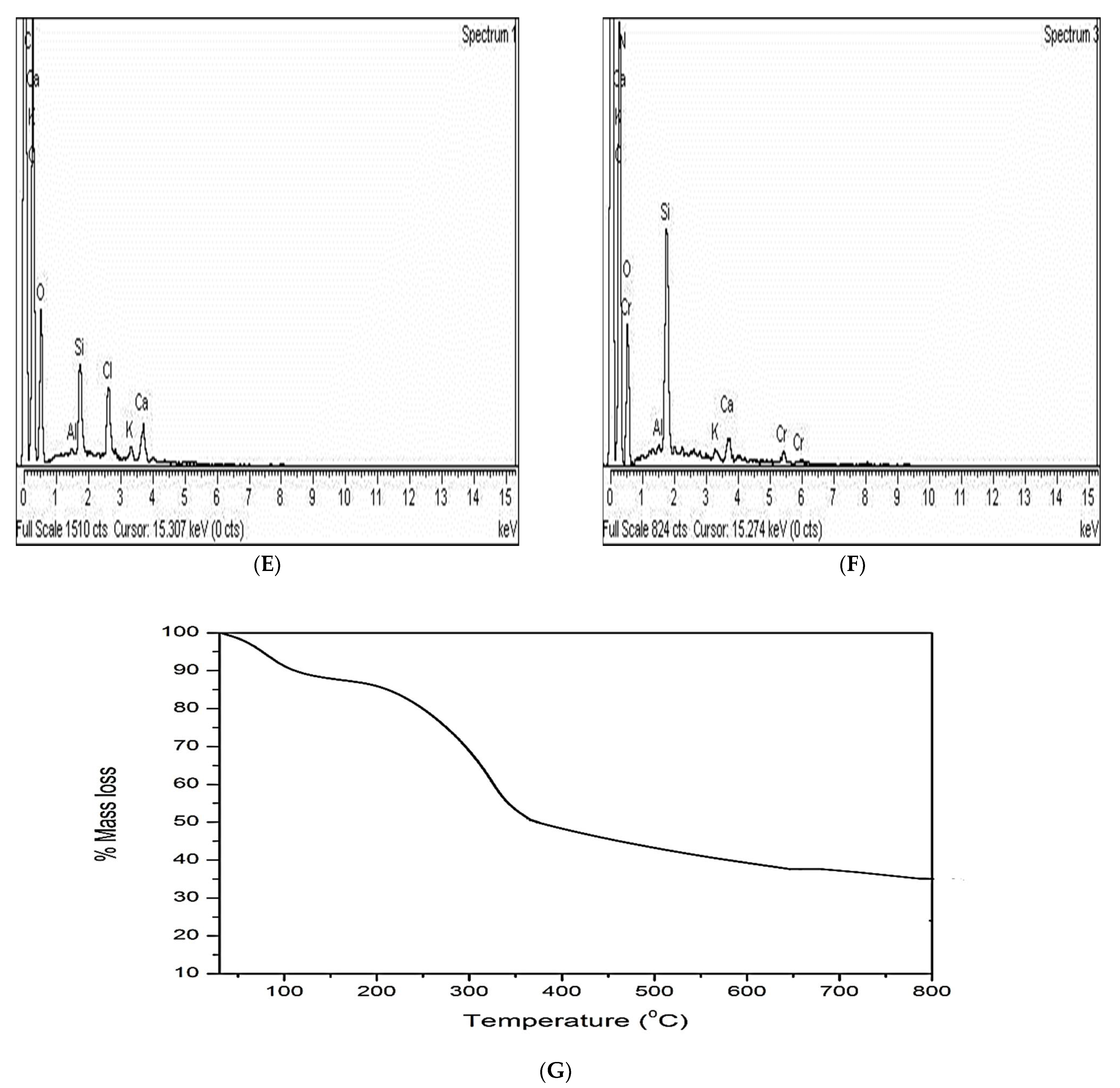
3.1.4. EDX Analysis
3.1.5. Thermal Gravimetric Analysis
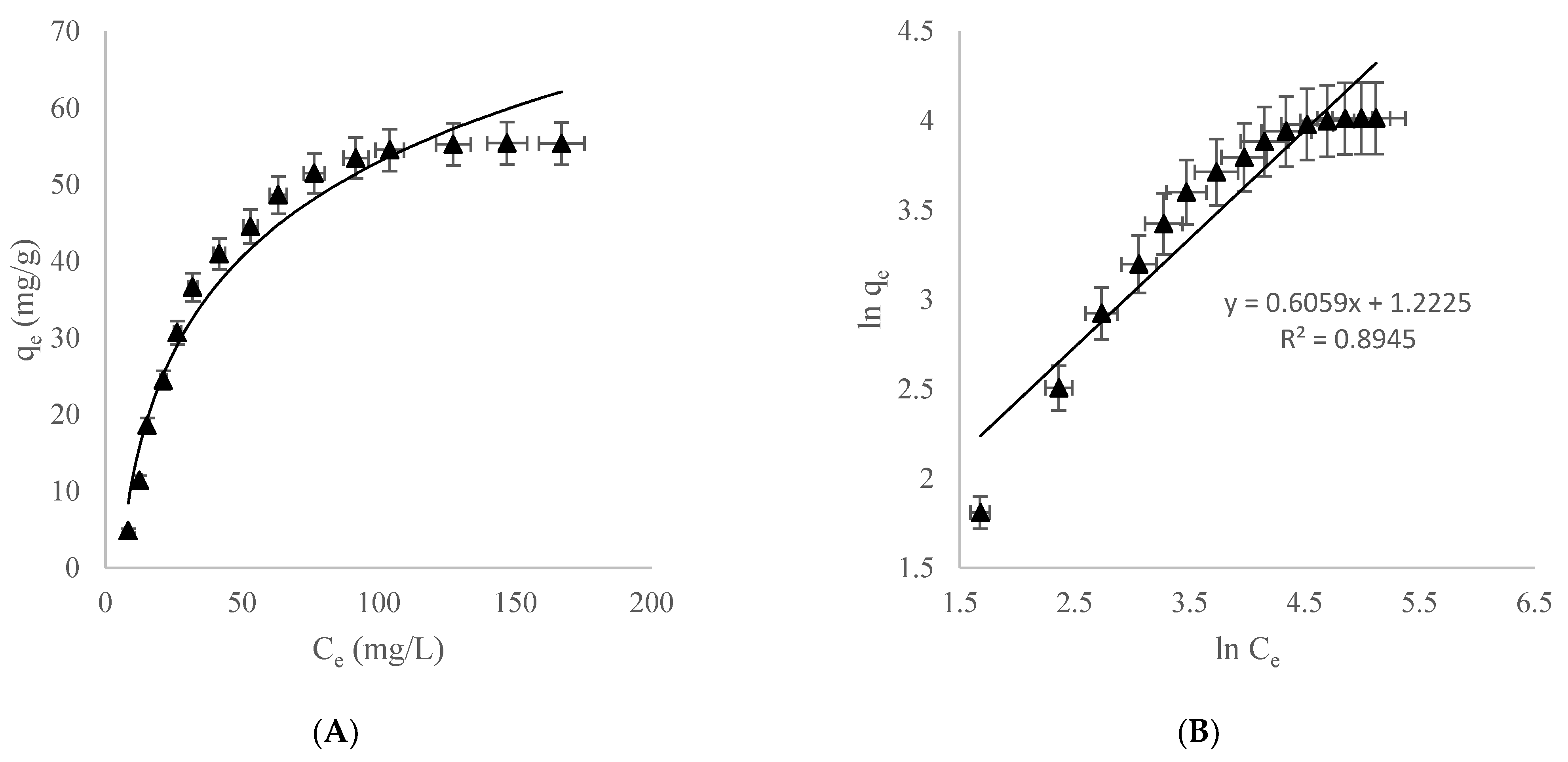
3.2. Adsorption Isothermal Studies
3.2.1. Freundlich Adsorption Isotherm
3.2.2. Langmuir Adsorption Isotherm
3.2.3. Temkin Adsorption Isotherm
3.2.4. Jovanovic Adsorption Isotherm
3.2.5. Harkins—Jura Adsorption Isotherm
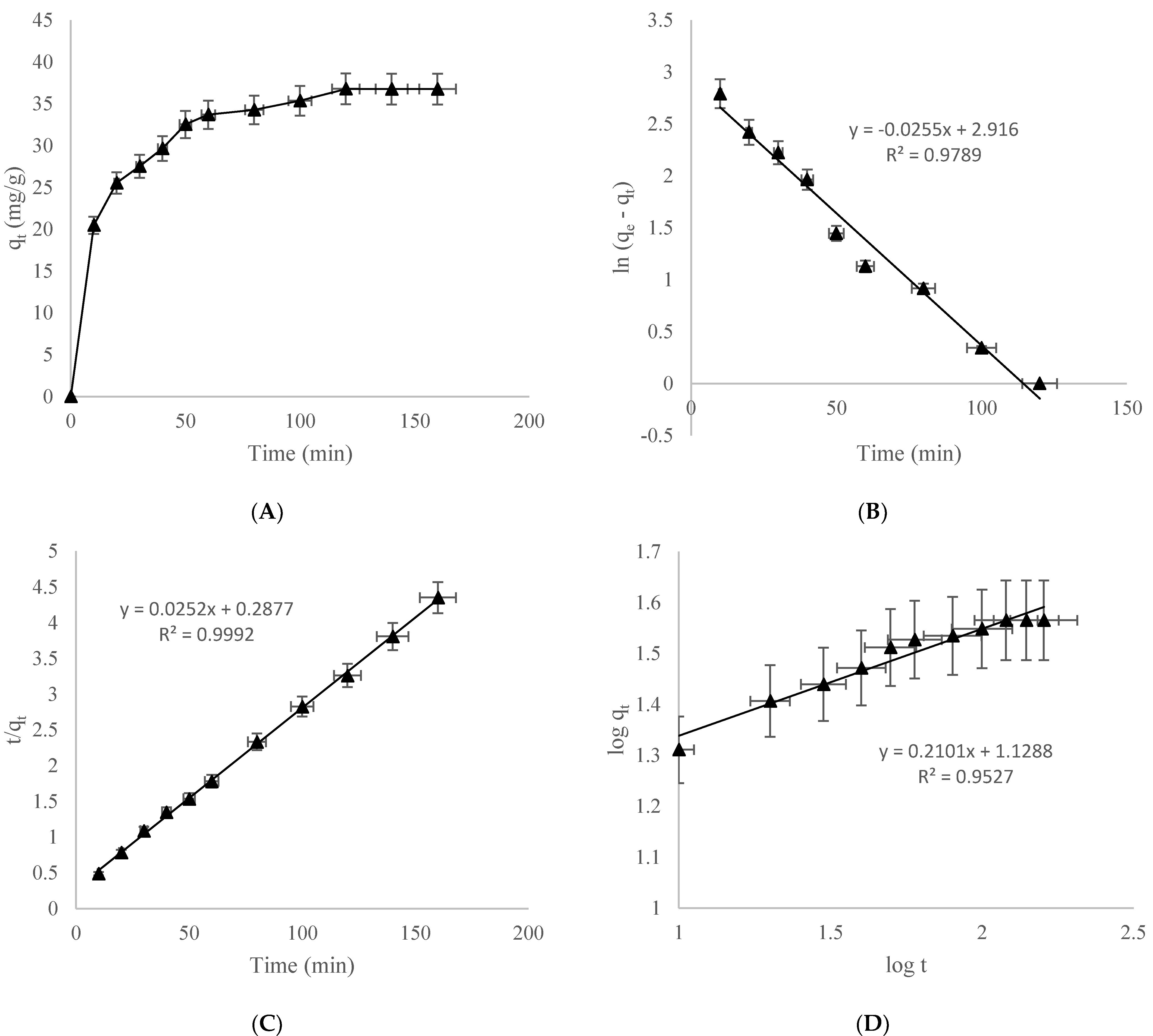
3.3. Impact of Contact Time and Kinetic Study
3.3.1. Pseudo-First-Order Kinetic Model
3.3.2. Pseudo-Second-Order Kinetic Model
3.3.3. Power Function Kinetic Model
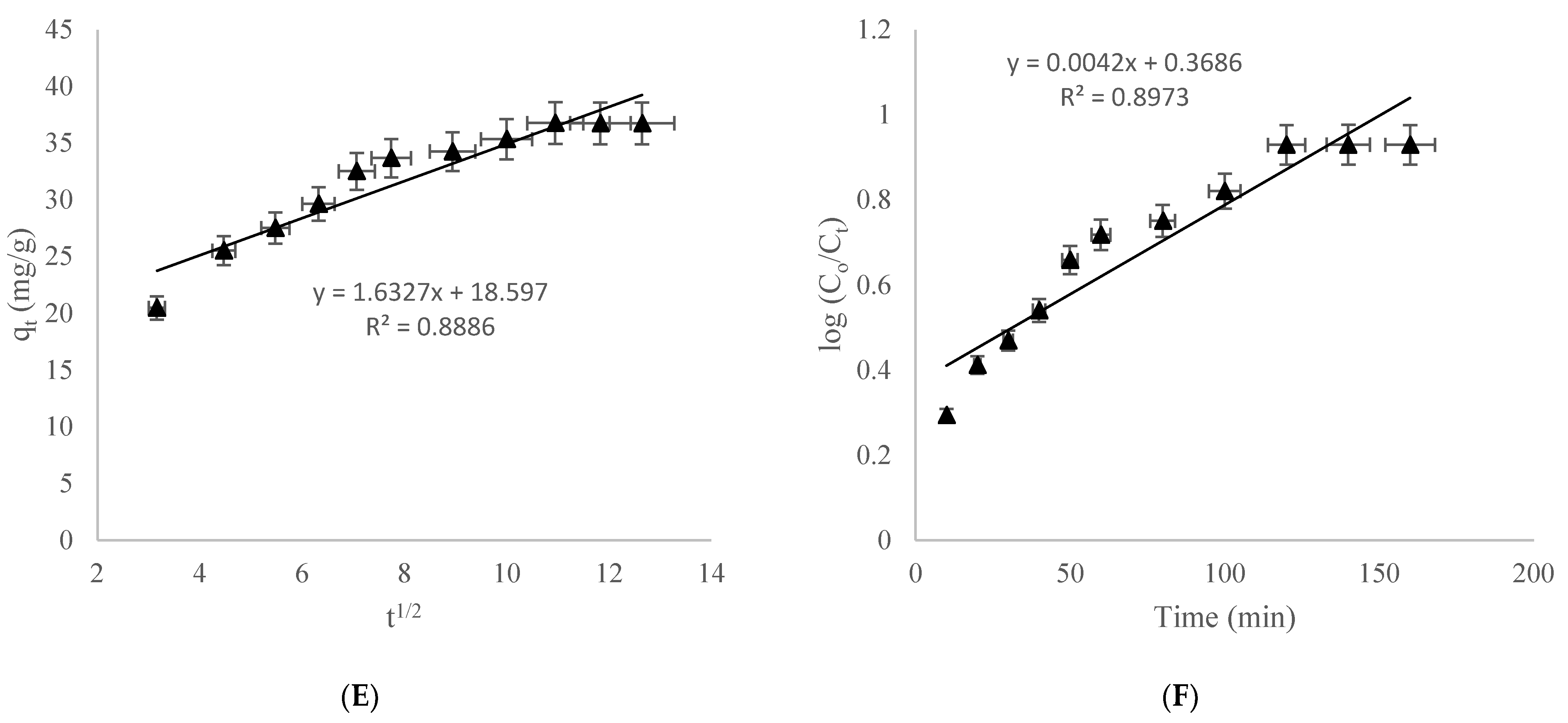
3.3.4. Intraparticle Kinetic Model
3.3.5. Natarajan–Khalaf Kinetic Model
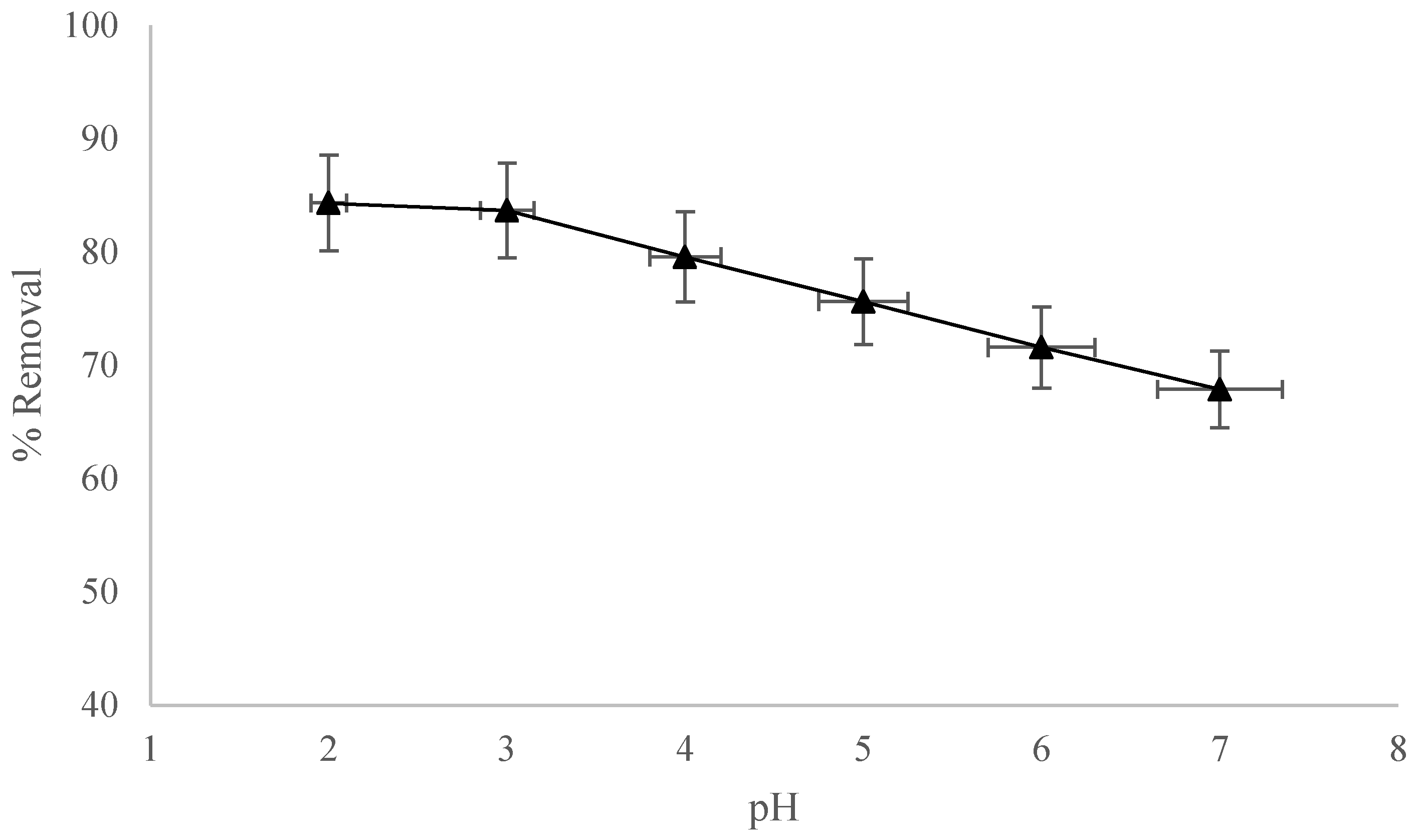
3.4. pH Analysis
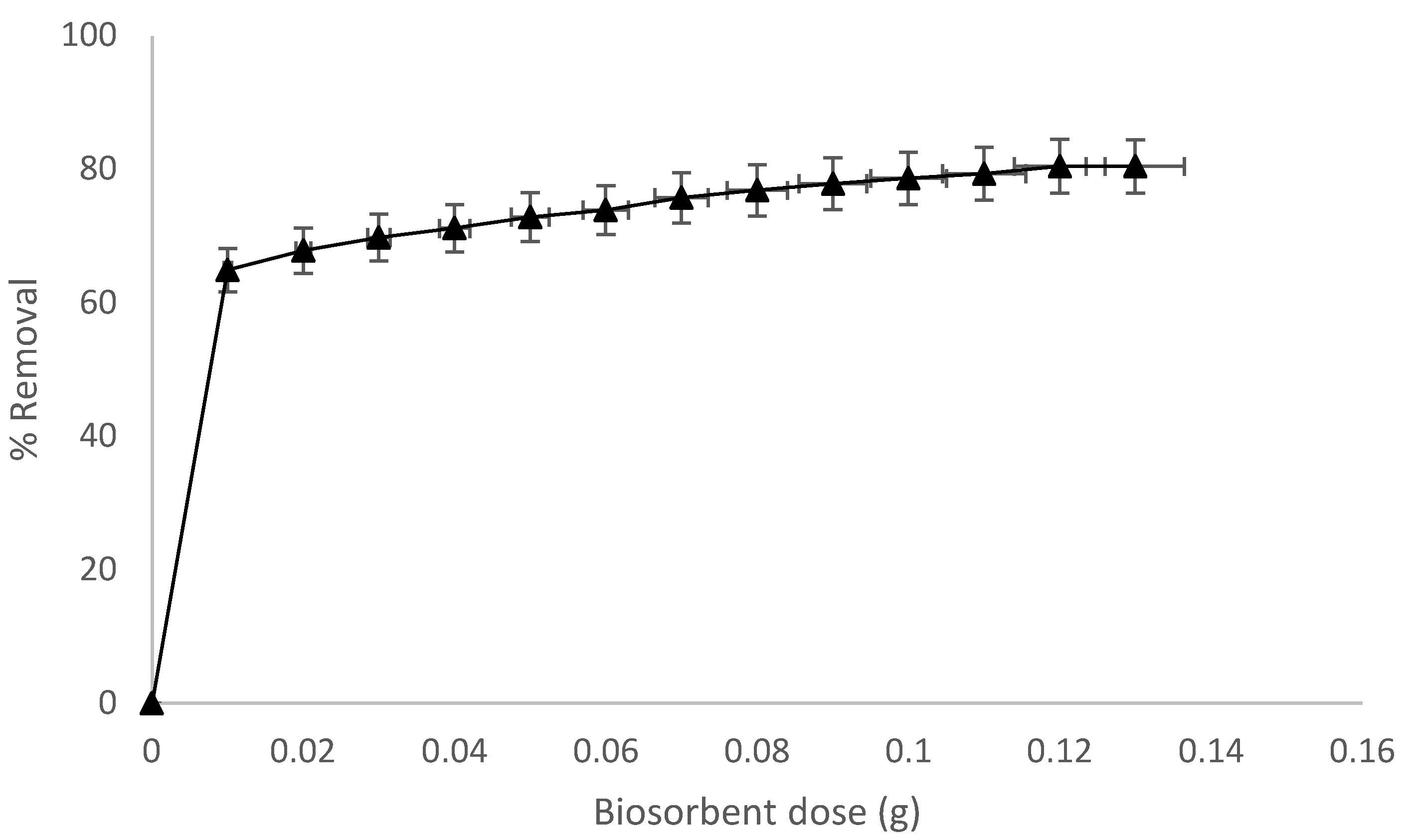
3.5. Biosorbent Dose Effect
3.6. Adsorption Thermodynamics
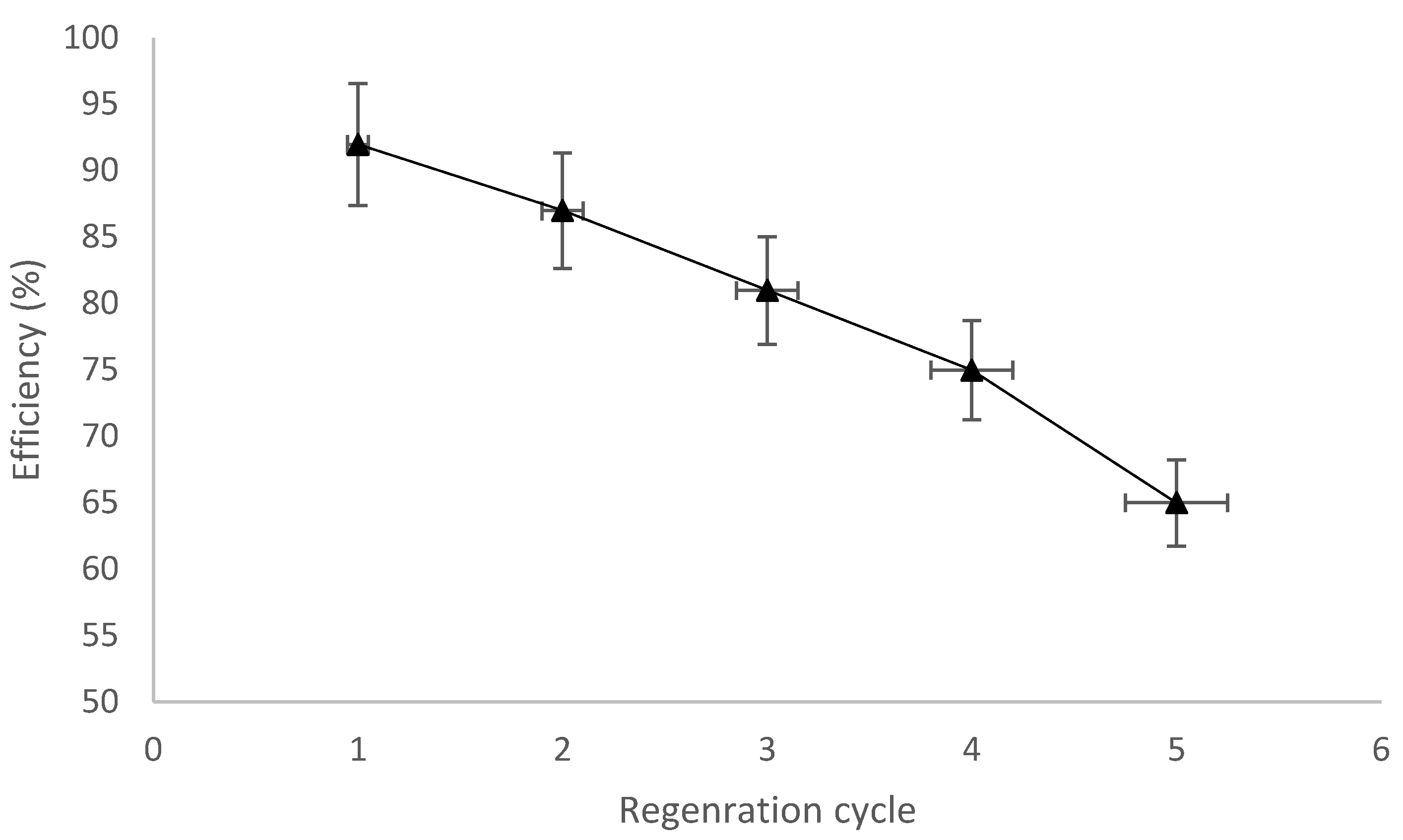
3.7. Regenration of CMPVL Biosorbent
3.8. Comparison of Adsorption Capacities of CMPVL with the Literature
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Jeyaseelan, C.; Gupta, A. Green tea leaves as a natural adsorbent for the removal of Cr (VI) from aqueous solutions. Air Soil Water Res. 2016, 9, ASWR-S35227. [Google Scholar] [CrossRef]
- Shirzad, S.M.; Samarghandi, M.; Azizian, S.; Kim, W.; Lee, S. The removal of hexavalent chromium from aqueous solutions using modified holly sawdust: Equilibrium and kinetics studies. Environ. Eng. Res. 2011, 16, 55–60. [Google Scholar]
- Zhou, J.; Wu, P.; Dang, Z.; Zhu, N.; Li, P.; Wu, J.; Wang, X. Polymeric Fe/Zr pillared montmorillonite for the removal of Cr (VI) from aqueous solutions. Chem. Eng. J. 2010, 162, 1035–1044. [Google Scholar] [CrossRef]
- Yasuda, M.; Miwa, A.; Kitagawa, M. Morphometric studies of renal lesions in itai-itai disease: Chronic cadmium nephropathy. Nephron 1995, 69, 14–19. [Google Scholar] [CrossRef]
- Kadirvelu, K.; Thamaraiselvi, K.; Namasivayam, C. Removal of heavy metals from industrial wastewaters by adsorption onto activated carbon prepared from an agricultural solid waste. Bioresour. Technol. 2001, 76, 63–65. [Google Scholar] [CrossRef]
- Wilbur, S.; Abadin, H.; Fay, M.; Yu, D.; Tencza, B.; Ingerman, L.; Klotzbach, J.; James, S. Toxicological Profile for Chromium; Agency for Toxic Substances and Disease Registry (US): Atlanta, GA, USA, 2012. [Google Scholar] [PubMed]
- Akram, M.; Bhatti, H.N.; Iqbal, M.; Noreen, S.; Sadaf, S. Biocomposite efficiency for Cr (VI) adsorption: Kinetic, equilibrium and thermodynamics studies. J. Environ. Chem. Eng. 2017, 5, 400–411. [Google Scholar] [CrossRef]
- Zheng, C.; Zheng, H.; Wang, Y.; Wang, Y.; Qu, W.; An, Q.; Liu, Y. Synthesis of novel modified magnetic chitosan particles and their adsorption performance toward Cr (VI). Bioresour. Technol. 2018, 267, 1–8. [Google Scholar] [CrossRef]
- Ye, Z.; Yin, X.; Chen, L.; He, X.; Lin, Z.; Liu, C.; Ning, S.; Wang, X.; Wei, Y. An integrated process for removal and recovery of Cr (VI) from electroplating wastewater by ion exchange and reduction–precipitation based on a silica-supported pyridine resin. J. Clean. Prod. 2019, 236, 117631. [Google Scholar] [CrossRef]
- Yang, X.; Liu, L.; Zhang, M.; Tan, W.; Qiu, G.; Zheng, L. Improved removal capacity of magnetite for Cr (VI) by electrochemical reduction. J. Hazard. Mater. 2019, 374, 26–34. [Google Scholar] [CrossRef]
- Kretschmer, I.; Senn, A.M.; Meichtry, J.M.; Custo, G.; Halac, E.B.; Dillert, R.; Bahnemann, D.W.; Litter, M.I. Photocatalytic reduction of Cr (VI) on hematite nanoparticles in the presence of oxalate and citrate. Appl. Catal. B Environ. 2019, 242, 218–226. [Google Scholar] [CrossRef]
- Jin, W.; Du, H.; Zheng, S.; Zhang, Y. Electrochemical processes for the environmental remediation of toxic Cr (VI): A review. Electrochim. Acta 2016, 191, 1044–1055. [Google Scholar] [CrossRef]
- Muthusaravanan, S.; Sivarajasekar, N.; Vivek, J.; Paramasivan, T.; Naushad, M.; Prakashmaran, J.; Gayathri, V.; Al-Duaij, O.K. Phytoremediation of heavy metals: Mechanisms, methods and enhancements. Environ. Chem. Lett. 2018, 16, 1339–1359. [Google Scholar] [CrossRef]
- Kausar, A.; MacKinnon, G.; Alharthi, A.; Hargreaves, J.; Bhatti, H.N.; Iqbal, M. A green approach for the removal of Sr (II) from aqueous media: Kinetics, isotherms and thermodynamic studies. J. Mol. Liq. 2018, 257, 164–172. [Google Scholar] [CrossRef]
- Kausar, A.; Bhatti, H.N.; Iqbal, M.; Ashraf, A. Batch versus column modes for the adsorption of radioactive metal onto rice husk waste: Conditions optimization through response surface methodology. Water Sci. Technol. 2017, 76, 1035–1043. [Google Scholar] [CrossRef]
- Azzaz, A.A.; Jellali, S.; Assadi, A.A.; Bousselmi, L. Chemical treatment of orange tree sawdust for a cationic dye enhancement removal from aqueous solutions: Kinetic, equilibrium and thermodynamic studies. Desalination Water Treat. 2016, 57, 22107–22119. [Google Scholar] [CrossRef]
- Azzaz, A.A.; Jellali, S.; Akrout, H.; Assadi, A.A.; Bousselmi, L. Optimization of a cationic dye removal by a chemically modified agriculture by-product using response surface methodology: Biomasses characterization and adsorption properties. Environ. Sci. Pollut. Res. 2017, 24, 9831–9846. [Google Scholar] [CrossRef]
- La, D.D.; Nguyen-Tri, P.; Le, K.H.; Nguyen, P.T.; Nguyen, M.D.B.; Vo, A.T.; Nguyen, D.D. Effects of antibacterial ZnO nanoparticles on the performance of a chitosan/gum arabic edible coating for post-harvest banana preservation. Prog. Org. Coat. 2021, 151, 106057. [Google Scholar] [CrossRef]
- Zhu, S.; Xia, M.; Chu, Y.; Khan, M.A.; Lei, W.; Wang, F.; Wang, A. Adsorption and desorption of Pb (II) on l-lysine modified montmorillonite and the simulation of interlayer structure. Appl. Clay Sci. 2019, 169, 40–47. [Google Scholar] [CrossRef]
- Khan, Q.; Zahoor, M.; Salman, S.M.; Wahab, M.; Khan, F.A.; Gulfam, N.; Zekker, I. Removal of Iron (II) from Effluents of Steel Mills Using Chemically Modified Pterisvittata Plant Leaves Utilizing the Idea of Phytoremediation. Water 2022, 14, 2004. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über die adsorption in lösungen. Z. Für Phys. Chem. 1907, 57, 385–470. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Shahbeig, H.; Bagheri, N.; Ghorbanian, S.A.; Hallajisani, A.; Poorkarimi, S. A new adsorption isotherm model of aqueous solutions on granular activated carbon. World J. Model. Simul. 2013, 9, 243–254. [Google Scholar]
- Samarghandi, M.; Hadi, M.; Moayedi, S.; BARJESTEH, A.F. Two-parameter isotherms of methyl orange sorption by pinecone derived activated carbon. J. Environ. Health Sci. Eng. 2009, 6, 285–294. [Google Scholar]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Ramesh, S.; Rameshbabu, N.; Gandhimathi, R.; Srikanth Kumar, M.; Nidheesh, P. Adsorptive removal of Pb (II) from aqueous solution using nano-sized hydroxyapatite. Appl. Water Sci. 2013, 3, 105–113. [Google Scholar] [CrossRef]
- McKay, G.; Ho, Y.; Ng, J. Biosorption of copper from waste waters: A review. Sep. Purif. Methods 1999, 28, 87–125. [Google Scholar] [CrossRef]
- Simonin, J.-P. On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef]
- Terpinc, P.; Bezjak, M.; Abramovič, H. A kinetic model for evaluation of the antioxidant activity of several rosemary extracts. Food Chem. 2009, 115, 740–744. [Google Scholar] [CrossRef]
- Srivastava, S.; Tyagi, R.; Pant, N. Adsorption of heavy metal ions on carbonaceous material developed from the waste slurry generated in local fertilizer plants. Water Res. 1989, 23, 1161–1165. [Google Scholar] [CrossRef]
- Weber, W.J., Jr.; Morris, J.C. Kinetics of adsorption on carbon from solution. J. Sanit. Eng. Div. 1963, 89, 31–59. [Google Scholar] [CrossRef]
- Ramulu, N.; Thirumurugan, V.; Krishnaveni, S.; Rajajeyaganthan, R. Adsorption of Rhodamine-B dye from an aqueous solution by biomass pine apple peel: Kinetics, equilibrium and thermodynamic studies. SOJ Mater. Sci. Eng. 2016, 4, 1–9. [Google Scholar]
- Tiwari, D.; Promod, K.; Mishra, A.; Singh, R.; Srivastav, R. Removal of toxic metals from electroplating industries(effect of pH on removal by adsorption). Indian J. Environ. Health 1989, 31, 120–124. [Google Scholar]
- Saravanane, R.; Sundararajan, T.; Reddy, S.S. Efficiency of chemically modified low cost adsorbents for the removal of heavy metals from waste water: A comparative study. Indian J. Environ. Health 2002, 44, 78–87. [Google Scholar]
- Rao, M.; Parwate, A.; Bhole, A. Removal of Cr6+ and Ni2+ from aqueous solution using bagasse and fly ash. Waste Manag. 2002, 22, 821–830. [Google Scholar] [CrossRef]
- Gupta, S.; Babu, B. Removal of toxic metal Cr (VI) from aqueous solutions using sawdust as adsorbent: Equilibrium, kinetics and regeneration studies. Chem. Eng. J. 2009, 150, 352–365. [Google Scholar] [CrossRef]
- Gritti, F.; Guiochon, G. Adsorption mechanisms and effect of temperature in reversed-phase liquid chromatography. Meaning of the classical Van’t Hoff plot in chromatography. Anal. Chem. 2006, 78, 4642–4653. [Google Scholar] [CrossRef]
- Qiu, S.; Wu, L.; Pan, X.; Zhang, L.; Chen, H.; Gao, C. Preparation and properties of functionalized carbon nanotube/PSF blend ultrafiltration membranes. J. Membr. Sci. 2009, 342, 165–172. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Venkateswarlu, K.; Lee, Y.B.; Naidu, R. Potential of Melaleuca diosmifolia leaf as a low-cost adsorbent for hexavalent chromium removal from contaminated water bodies. Process Saf. Environ. Prot. 2016, 100, 173–182. [Google Scholar] [CrossRef]
- Salman, S.M.; Ali, A.; Khan, B.; Iqbal, M.; Alamzeb, M. Thermodynamic and kinetic insights into plant-mediated detoxification of lead, cadmium, and chromium from aqueous solutions by chemically modified Salvia moorcroftiana leaves. Environ. Sci. Pollut. Res. 2019, 26, 14339–14349. [Google Scholar] [CrossRef]
- Ponou, J.; Kim, J.; Wang, L.P.; Dodbiba, G.; Fujita, T. Sorption of Cr (VI) anions in aqueous solution using carbonized or dried pineapple leaves. Chem. Eng. J. 2011, 172, 906–913. [Google Scholar] [CrossRef]
- Mondal, N.K.; Samanta, A.; Roy, P.; Das, B. Optimization study of adsorption parameters for removal of Cr (VI) using Magnolia leaf biomass by response surface methodology. Sustain. Water Resour. Manag. 2019, 5, 1627–1639. [Google Scholar] [CrossRef]
- Rehman, R.; Anwar, J.; Mahmud, T.; Salman, M.; Saleem, M. Evaluation of batch biosorption of chromium (VI) from aqueous solution by chemically modified Polyalthialongifolia leaves. J. Chem. Soc. Pak 2011, 33, 846. [Google Scholar]


| Biosorbent | CMPVL |
|---|---|
| BET surface area (m2/g) | 73.28 |
| Pore volume (cc/g) | 0.82 |
| Adsorption Isotherm | Parameters | Values |
|---|---|---|
| Langmuir | qmax (mg/g) KL (L/mg) R2 | 66.6 0.03671 0.991 |
| Freundlich | KF (mg/g) n R2 | 3.394 1.653 0.894 |
| Temkin | β α b R2 | 16.57 4.197 136.98 0.968 |
| Jovanovich | KJ (L/g) qmax (mg/g) R2 | 0.009 18.32 0.546 |
| Harkins–Jura | AH (g2/L) BH (mg2/L) R2 | 5 4 0.834 |
| Kinetic Model | Parameters | Values |
|---|---|---|
| Pseudo-first-order | K1 (1/min) qe (mg/g) R2 | −0.025 18.467 0.978 |
| Pseudo-second-order | K2 (1/min) qe (mg/g) R2 | 0.287 40 0.999 |
| Power function | α b R2 | 13.427 0.210 0.952 |
| Intra particle diffusion | Kdiff (mg/g min1/2) C R2 | 1.632 18.59 0.888 |
| Natarajan–Khalaf | KN (1/min) R2 | 9.2 × 10−3 0.897 |
| Parameters Values | Values |
|---|---|
| ΔH° (J/mol K) | −20.51 |
| ΔS° (J/mol K) | 67.09 |
| ΔG°(KJ/mol) | |
| 293 K | −19.67 |
| 303 K | −20.34 |
| 313 K | −21.019 |
| 323 K | −21.69 |
| 333 K | −22.36 |
| S.NO | Biosorbent | qmax (mgg−1) | References |
|---|---|---|---|
| 1 | This research work | 66.67 | |
| 2 | Melaleuca diosmifolia leaf | 62.5 | [40] |
| 3 | CMSML | 93.45 | [41] |
| 4 | Carbonized pineapple leaves | 18.77 | [42] |
| 5 | Magnolia leaf biomass | 12.3 | [43] |
| 6 | chemically modified Polyalthia longifolia leaves | 5.128 | [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, Q.; Zahoor, M.; Salman, S.M.; Wahab, M.; Talha, M.; Kamran, A.W. Removal of Chromium (VI) from the Steel Mill Effluents Using the Chemically Modified Leaves of Pteris vittata as Adsorbent. Water 2022, 14, 2599. https://doi.org/10.3390/w14172599
Khan Q, Zahoor M, Salman SM, Wahab M, Talha M, Kamran AW. Removal of Chromium (VI) from the Steel Mill Effluents Using the Chemically Modified Leaves of Pteris vittata as Adsorbent. Water. 2022; 14(17):2599. https://doi.org/10.3390/w14172599
Chicago/Turabian StyleKhan, Qaiser, Muhammad Zahoor, Syed Muhammad Salman, Muhammad Wahab, Muhammad Talha, and Abdul Waheed Kamran. 2022. "Removal of Chromium (VI) from the Steel Mill Effluents Using the Chemically Modified Leaves of Pteris vittata as Adsorbent" Water 14, no. 17: 2599. https://doi.org/10.3390/w14172599






