Species-Specific Abundance Response of Montane Stream Fishes to Drought-Induced Variation in Streamflow
Abstract
:1. Introduction
2. Materials and Methods
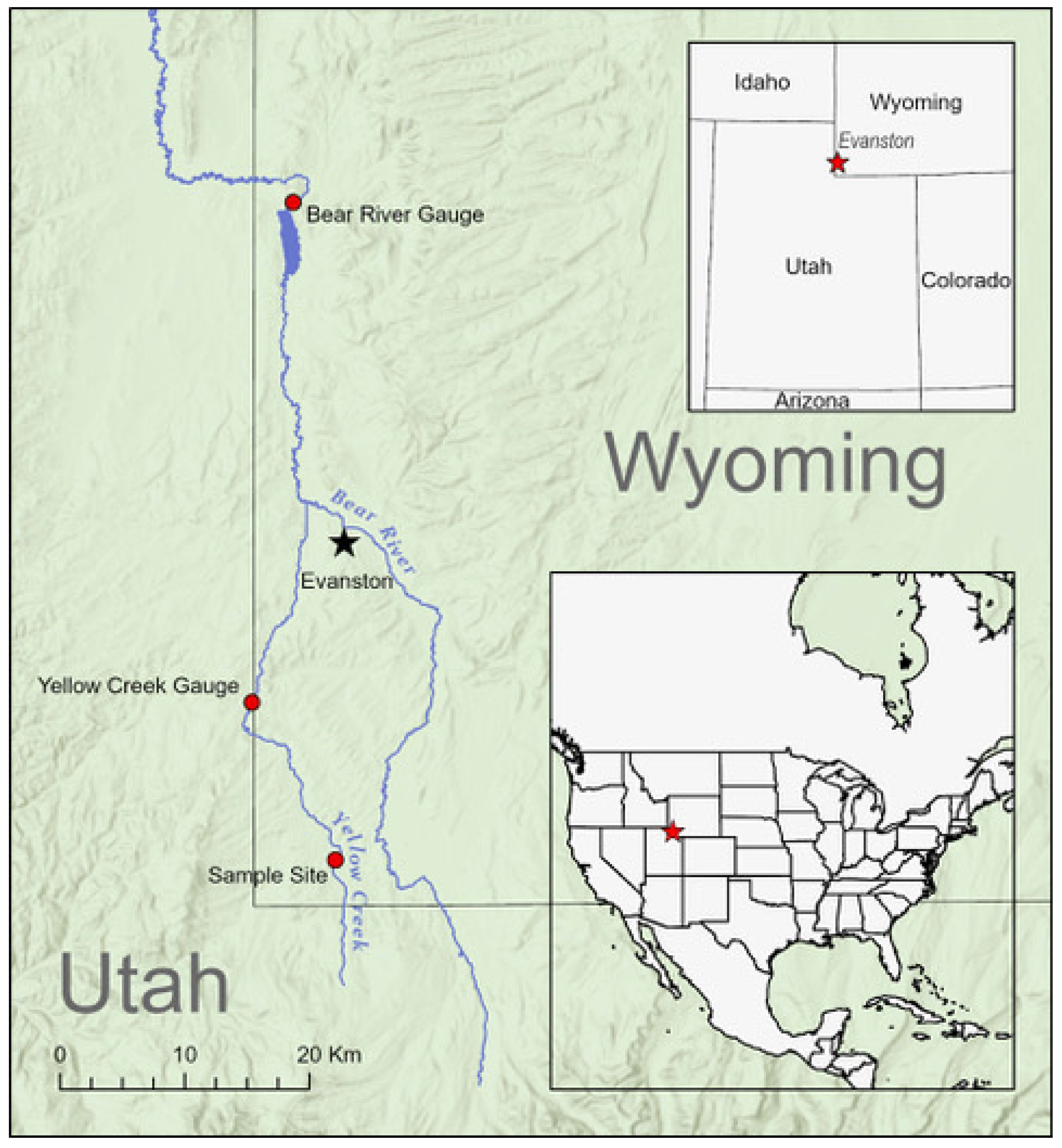
2.1. Study System
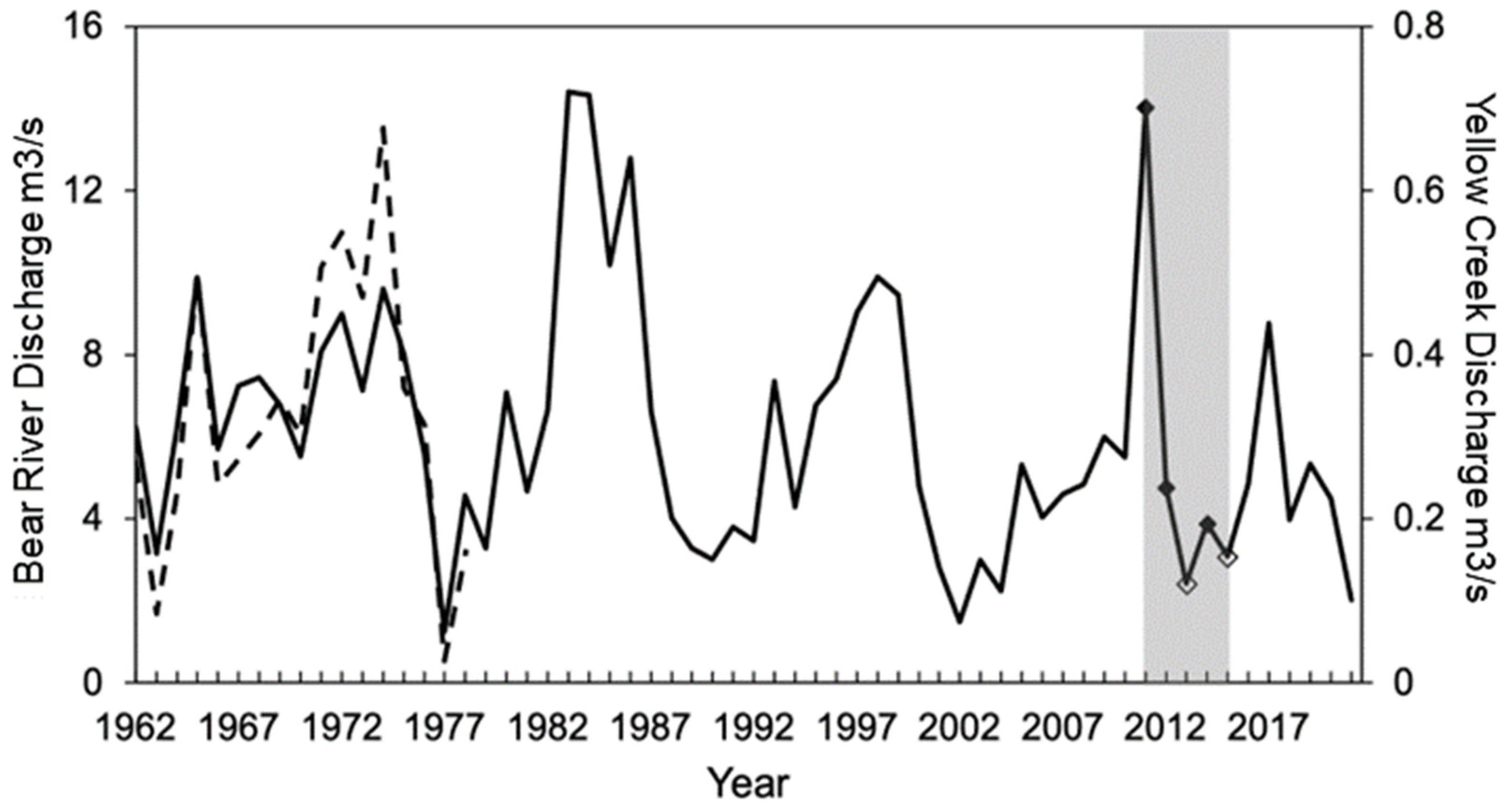
2.2. Streamflow Characterization
2.3. Fish Population Abundance
2.4. Analysis
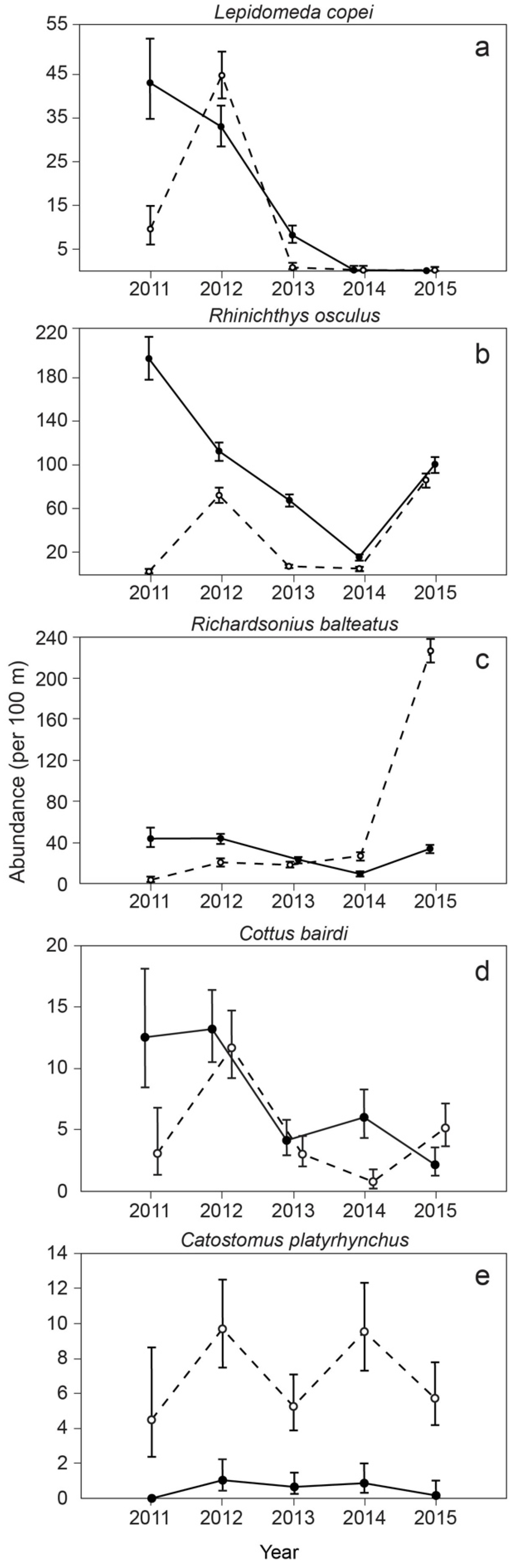
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barnett, T.P.; Adam, J.C.; Lettenmaier, D.P. Potential impacts of a warming climate on water availability in snow-dominated regions. Nature 2005, 438, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Mote, P.W.; Hamlet, A.F.; Clark, M.P.; Lettenmaier, D.P. Declining mountain snowpack in western North America. Bull. Am. Meteorol. Soc. 2005, 86, 39–50. [Google Scholar] [CrossRef]
- Hamlet, A.F.; Mote, P.W.; Clark, M.P.; Lettenmaier, D.P. Effects of temperature and precipitation variability on snowpack trends in the Western United States. J. Clim. 2005, 18, 4545–4561. [Google Scholar] [CrossRef]
- Safeeq, M.; Shukla, S.; Arismendi, I.; Grant, G.E.; Lewis, S.L.; Nolin, A. Influence of winter season climate variability on snow-precipitation ratio in the western United States. Int. J. Climatol. 2015, 36, 3175–3190. [Google Scholar] [CrossRef]
- Rhoades, A.M.; Ullrich, P.A.; Zarzycki, C.M. Projecting 21st Century snowpack trends in Western USA mountains using variable-resolution CESM. Clim. Dyn. 2017, 50, 261–288. [Google Scholar] [CrossRef]
- Serreze, M.C.; Clark, M.P.; Armstrong, R.L.; McGinnis, D.A.; Pulwarty, R.S. Characteristics of the Western United States snowpack from snowpack telemetry (SNOTEL) data. Water Resour. Res. 1999, 35, 2145–2160. [Google Scholar] [CrossRef] [Green Version]
- Julander, R.P.; Clayton, J.A. Determining the proportion of streamflow that is generated by cold season processes versus summer rainfall in Utah, USA. J. Hydrol. Reg. Stud. 2018, 17, 36–46. [Google Scholar] [CrossRef]
- Das, T.; Pierce, D.W.; Cayan, D.R.; Vano, J.A.; Lettenmaier, D.P. The importance of warm season warming to western US streamflow changes. Geophys. Res. Lett. 2011, 38, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Milly, P.C.; Dunne, K.A.; Vecchia, A.V. Global pattern of trends in streamflow and water availability in a changing climate. Nature 2005, 438, 347–350. [Google Scholar] [CrossRef]
- Reynolds, L.V.; Shafroth, P.B.; LeRoy Poff, N. Modeled intermittency risk for small streams in the upper Colorado River basin under climate change. J. Hydrol. 2015, 523, 768–780. [Google Scholar] [CrossRef]
- Cayan, D.R.; Kammerdiener, S.A.; Dettinger, M.D.; Caprio, J.M.; Peterson, D.H. Changes in the onset of spring in the Western United States. Bull. Am. Meteorol. Soc. 2001, 82, 399–416. [Google Scholar] [CrossRef] [Green Version]
- Berghuijs, W.R.; Woods, R.A.; Hrachowitz, M. A precipitation shift from snow towards rain leads to a decrease in streamflow. Nat. Clim. Chang. 2014, 4, 583–586. [Google Scholar] [CrossRef] [Green Version]
- Bêche, L.A.; Connors, P.G.; Resh, V.H.; Merenlender, A.M. Resilience of fishes and invertebrates to prolonged drought in two California streams. Ecography 2009, 32, 778–788. [Google Scholar] [CrossRef]
- Keaton, M.; Haney, D.; Andersen, C.B. Impact of drought upon fish assemblage structure in two South Carolina Piedmont streams. Hydrobiologia 2005, 545, 209–223. [Google Scholar] [CrossRef]
- Magalhaes, M.F.; Beja, P.; Schlosser, I.J.; Collares-Pereira, M.J. Effects of multi-year droughts on fish assemblages of seasonally drying Mediterranean streams. Freshw. Biol. 2007, 52, 1494–1510. [Google Scholar] [CrossRef]
- Magoulick, D.D.; Kobza, R.M. The role of refugia for fishes during drought: A review and synthesis. Freshw. Biol. 2003, 48, 1186–1198. [Google Scholar] [CrossRef]
- Matthews, W.J.; Marsh-Matthews, E. Effects of drought on fish across axes of space, time and ecological complexity. Freshw. Biol. 2003, 48, 1232–1253. [Google Scholar] [CrossRef]
- Driver, L.J.; Hoeinghaus, D.J. Spatiotemporal dynamics of intermittent stream fish metacommunities in response to prolonged drought and reconnectivity. Mar. Freshw. Res. 2016, 67, 1667. [Google Scholar] [CrossRef]
- Oberdorff, T.; Hugueny, B.; Vigneron, T. Is assemblage variability related to environmental variability? An answer for riverine fish. Oikos 2001, 93, 419–428. [Google Scholar] [CrossRef]
- Ross, S.T.; Matthews, W.J.; Echelle, A.A. Persistence of Stream Fish Assemblages: Effects of Environmental Change. Am. Nat. 1985, 126, 24–40. [Google Scholar] [CrossRef]
- Meijer, C.G.; Warburton, H.J.; Harding, J.S.; McIntosh, A.R. Shifts in population size structure for a drying-tolerant fish in response to extreme drought. Austral. Ecol. 2019, 44, 658–667. [Google Scholar] [CrossRef]
- Arthington, A.H.; Balcombe, S.R. Extreme flow variability and the ‘boom and bust’ ecology of fish in arid-zone floodplain rivers: A case history with implications for environmental flows, conservation and management. Ecohydrology 2011, 4, 708–720. [Google Scholar] [CrossRef] [Green Version]
- Williams, J.E.; Haak, A.L.; Neville, H.M.; Colyer, W.T. Potential consequences of climate change to persistence of cutthroat trout populations. N. Am. J. Fish. Manag. 2009, 29, 533–548. [Google Scholar] [CrossRef]
- Pease, A.A.; Paukert, C.P. Potential impacts of climate change on growth and prey consumption of stream-dwelling smallmouth bass in the central United States. Ecol. Freshw. Fish 2014, 23, 336–346. [Google Scholar] [CrossRef]
- Wesner, J.S.; Belk, M.C. Variation in the trophic position of common stream fishes and its relationship to the presence of a rare fish, northern leatherside chub (Lepidomeda copei). Ecol. Freshw. Fish 2015, 24, 234–241. [Google Scholar] [CrossRef]
- Meador, M.R.; Cuffney, T.F.; Gurtz, M.E. Methods for Sampling Fish Communities as Part of the National Water-Quality Assessment Program (No. 93-104); US Geological Survey: Raleigh, NC, USA, 1993. [Google Scholar]
- Van Deventer, J.S. Microcomputer Software System for Generating Population Statistics from Electrofishing Data: User's Guide for Microfish 3.0; US Department of Agriculture, Forest Service, Intermountain Research Station: Ogden, UT, USA, 1989; Volume 254. [Google Scholar]
- Hopper, G.W.; Gido, K.B.; Pennock, C.A.; Hedden, S.C.; Frenette, B.D.; Barts, N.; Bruckerhoff, L.A. Nowhere to swim: Interspecific responses of prairie stream fishes in isolated pools during severe drought. Aquat. Sci. 2020, 82, 1–15. [Google Scholar] [CrossRef]
- Propst, D.L.; Gid, K.B.; Stefferud, J.A. Natural flow regimes, nonnative fishes, and native fish persistence in arid-land river systems. Ecol. Appl. 2008, 18, 1236–1252. [Google Scholar] [CrossRef] [Green Version]
- Resh, V.H.; Bêche, L.A.; Lawrence, J.E.; Mazor, R.D.; McElravy, E.P.; O’Dowd, A.P.; Carlson, S.M. Long-term population and community patterns of benthic macroinvertebrates and fishes in Northern California Mediterranean-climate streams. Hydrobiologia 2013, 719, 93–118. [Google Scholar] [CrossRef]
- Kiernan, J.D.; Moyle, P.B.; Crain, P.K. Restoring native fish assemblages to a regulated California stream using the natural flow regime concept. Ecol. Appl. 2012, 22, 1472–1482. [Google Scholar] [CrossRef]
- Team, U.W.A.P.J. Utah Wildlife Action Plan: A Plan for Managing Native Wildlife Species and Their Habitats to Help Prevent Listing under the Endangered Species Act; Utah Division of Wildlife Resources: Salt Lake City, UT, USA, 2015. [Google Scholar]
- Wesner, J.S.; Belk, M.C. Habitat relationships among biodiversity indicators and co-occurring species in a freshwater fish community. Anim. Conserv. 2012, 155, 445–456. [Google Scholar] [CrossRef]
- Billman, E.J.; Wagner, E.J.; Arndt, R.E.; VanDyke, E. Optimal temperatures for growth and upper thermal tolerance of juvenile northern leatherside chub. West. N. Am. Nat. 2008, 68, 463–474. [Google Scholar] [CrossRef]
- Mas-Martí, E.; García-Berthou, E.; Sabater, S.; Tomanova, S.; Munoz, I. Comparing fish assemblages and trophic ecology of permanent and intermittent reaches in a Mediterranean stream. In Global Change and River Ecosystems—Implications for Structure, Function and Ecosystem Services; Springer: Dordrecht, The Netherlands, 2010; pp. 167–180. [Google Scholar]
- Nannini, M.A.; Belk, M.C. Antipredator responses of two native stream fishes to an introduced predator: Does similarity in morphology predict similarity in behavioral response? Ecol. Freshw. Fish 2006, 15, 453–463. [Google Scholar] [CrossRef]
- Schultz, L.D.; Bertrand, K.N.; Graeb, B.D. Factors from multiple scales influence the distribution and abundance of an imperiled fish—mountain sucker in the Black Hills of South Dakota, USA. Environ. Biol. Fishes 2016, 99, 3–14. [Google Scholar] [CrossRef]
- Boguski, D.A.; Watkinson, D.A. Information in Support of a Recovery Potential Assessment of Mountain Sucker (Catostomus Platyrhynchus), Milk River Populations (Designatable Unit 2); Canadian Science Advisory Secretariat: Winnipeg, MB, Canada, 2013. [Google Scholar]
| Source | DF | Chi-Square | Pr > Chi-Sqr |
|---|---|---|---|
| Year | 4 | 195.76 | <0.0001 |
| Life stage | 1 | 0.65 | 0.4192 |
| Species | 4 | 423.72 | <0.0001 |
| Year * Life stage | 4 | 32.12 | <0.0001 |
| Year * Species | 16 | 1336.13 | <0.0001 |
| Species * Life stage | 4 | 326.62 | <0.0001 |
| Year * Species * Life stage | 16 | 378.19 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larsen, N.E.; Simkins, R.M.; Wesner, J.S.; Tuckfield, R.C.; Belk, M.C. Species-Specific Abundance Response of Montane Stream Fishes to Drought-Induced Variation in Streamflow. Water 2022, 14, 2467. https://doi.org/10.3390/w14162467
Larsen NE, Simkins RM, Wesner JS, Tuckfield RC, Belk MC. Species-Specific Abundance Response of Montane Stream Fishes to Drought-Induced Variation in Streamflow. Water. 2022; 14(16):2467. https://doi.org/10.3390/w14162467
Chicago/Turabian StyleLarsen, Noah E., Richard M. Simkins, Jeff S. Wesner, R. Cary Tuckfield, and Mark C. Belk. 2022. "Species-Specific Abundance Response of Montane Stream Fishes to Drought-Induced Variation in Streamflow" Water 14, no. 16: 2467. https://doi.org/10.3390/w14162467






