Close Association between Stream Water Quality and Fluorescence Properties of Dissolved Organic Matter in Agriculture-Dominated Watersheds
Abstract
:1. Introduction
2. Materials and Methods
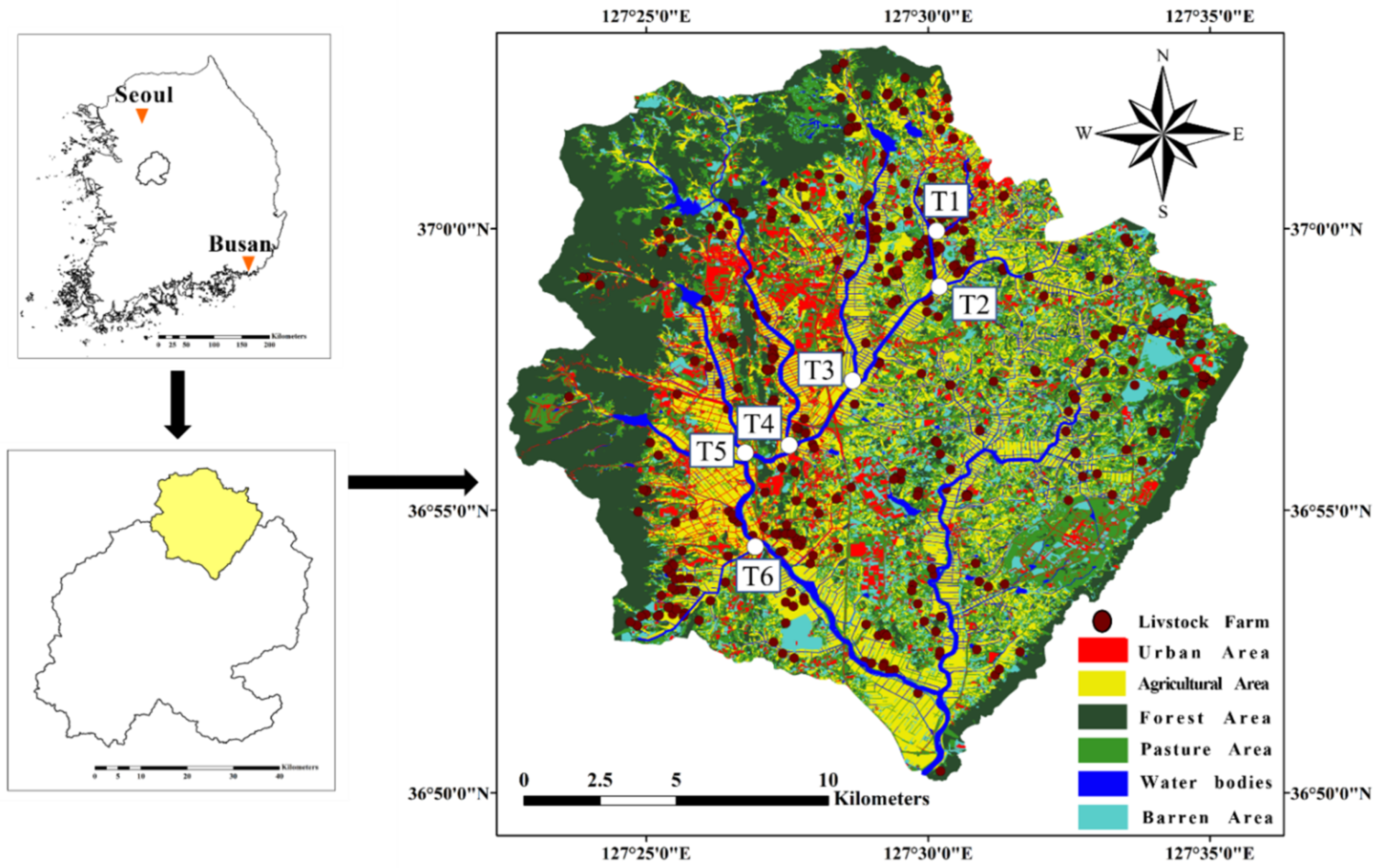
2.1. Study Area and Sampling Sites
2.2. Flow Rate and Water Quality Measurements
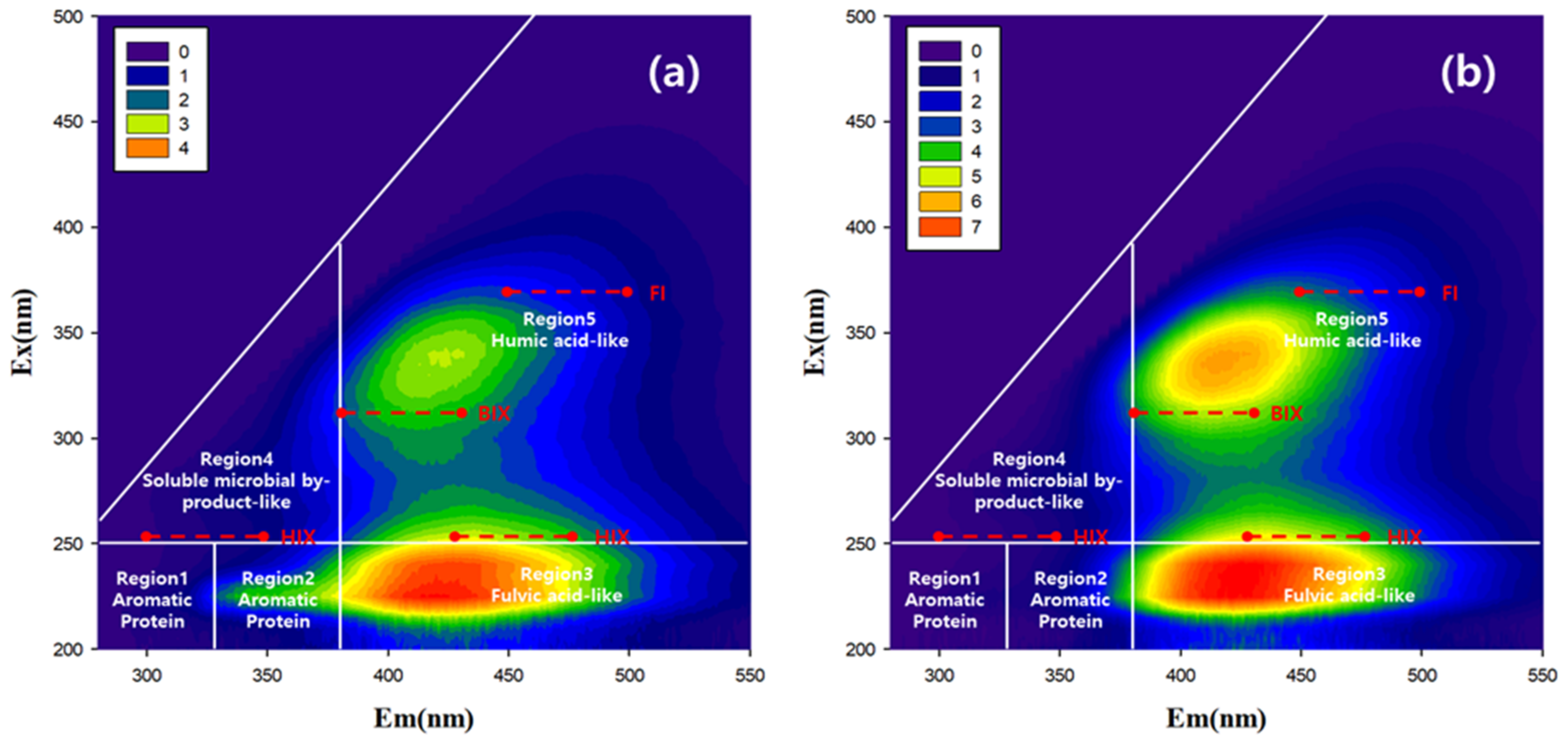
2.3. Absorbance and Fluorescence Measurements—Fluorescence Spectroscopy and UV Absorbance Analysis
2.4. Statistical Analysis
3. Results and Discussion
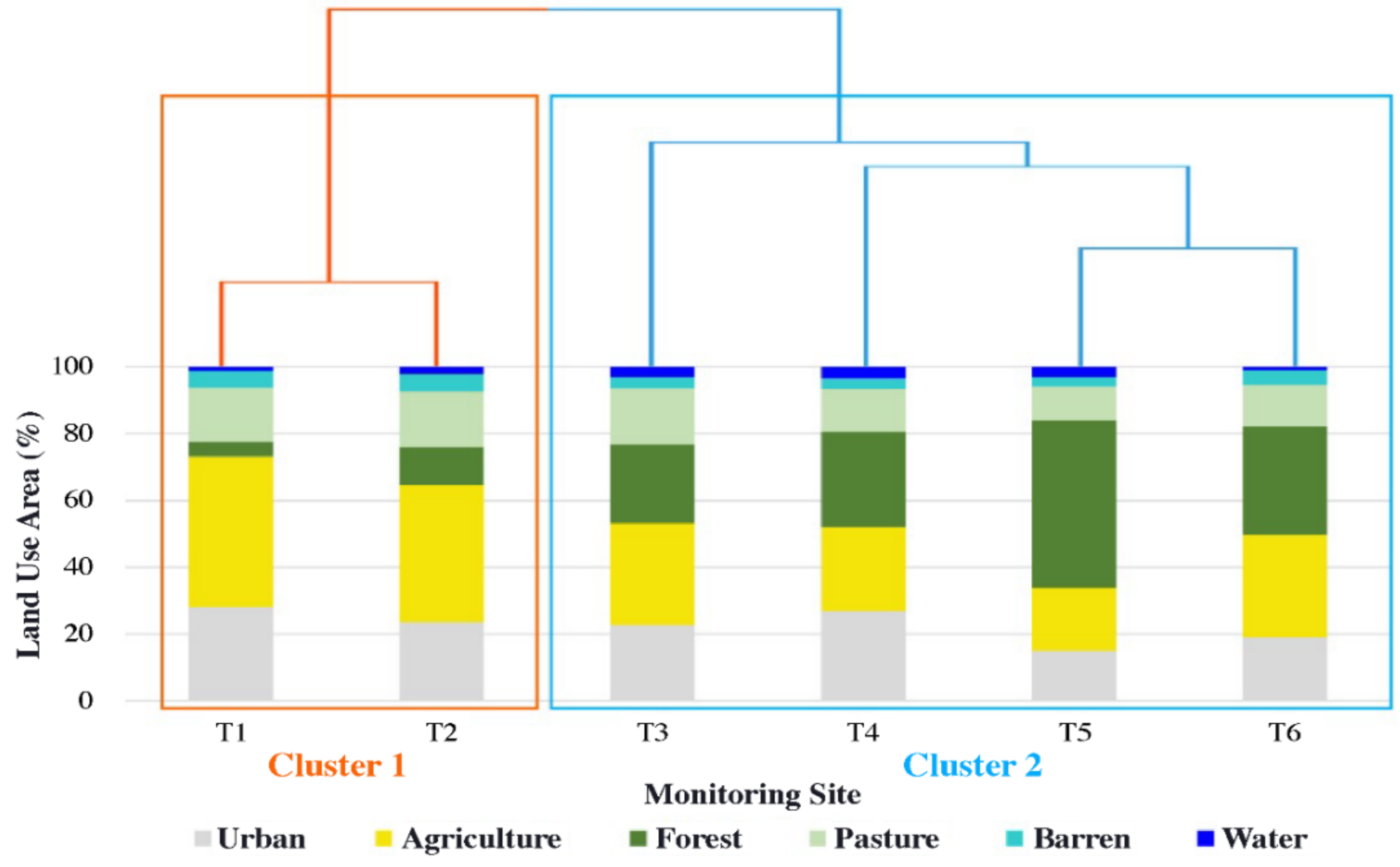
3.1. Land Use/Cover Similarity and Site Clustering
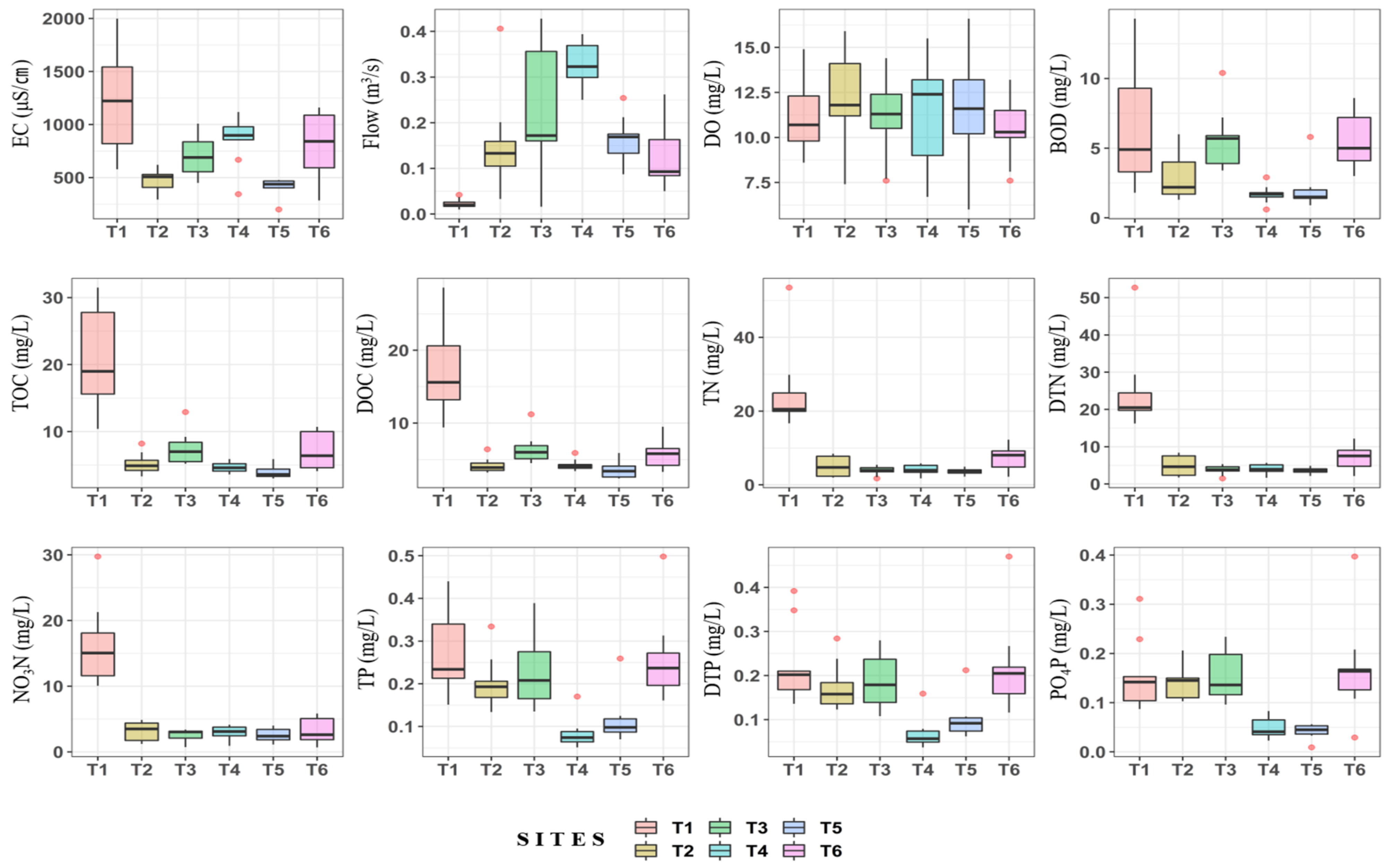
3.2. Comparison of Water Quality for Different Tributaries
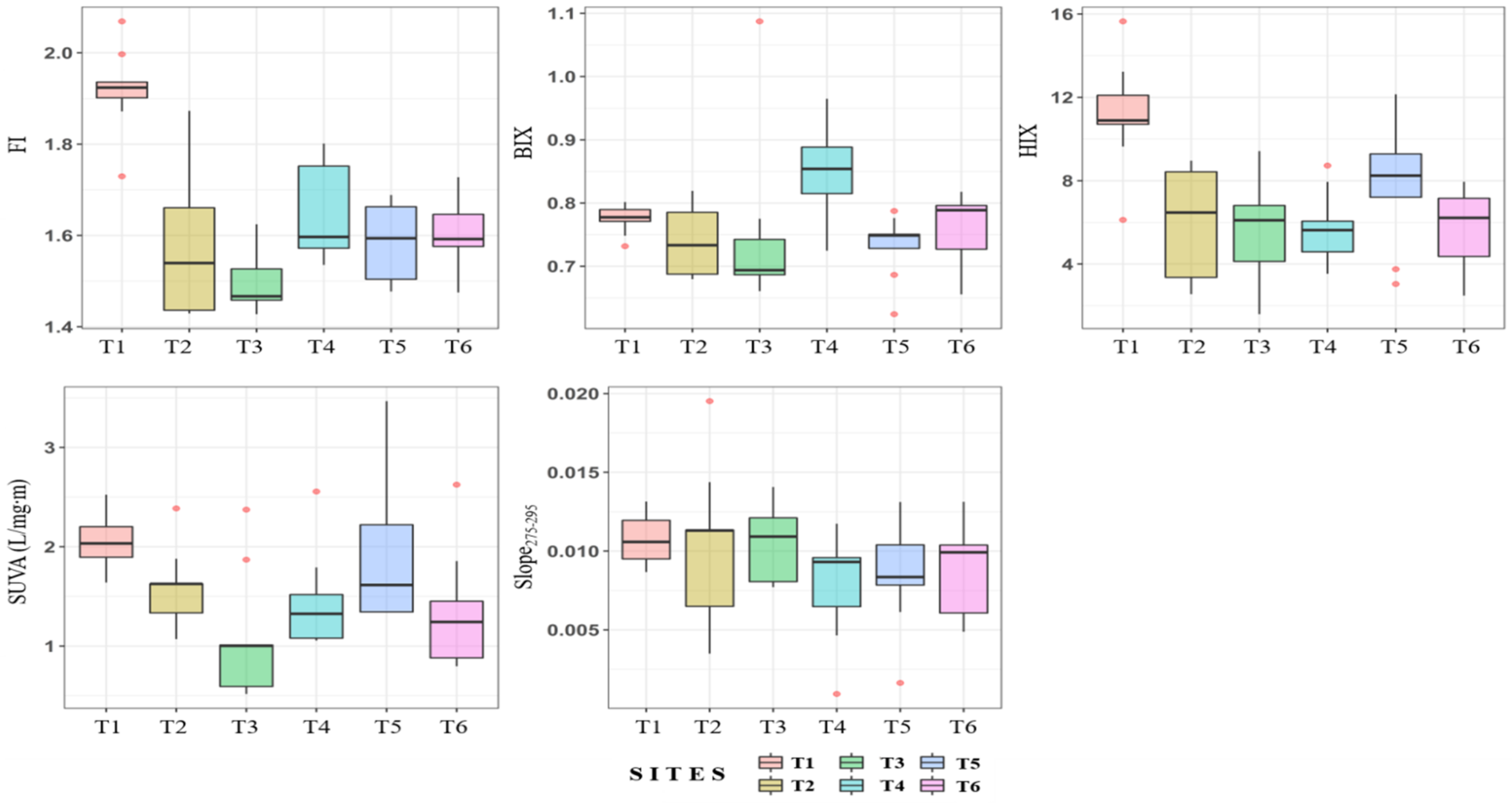
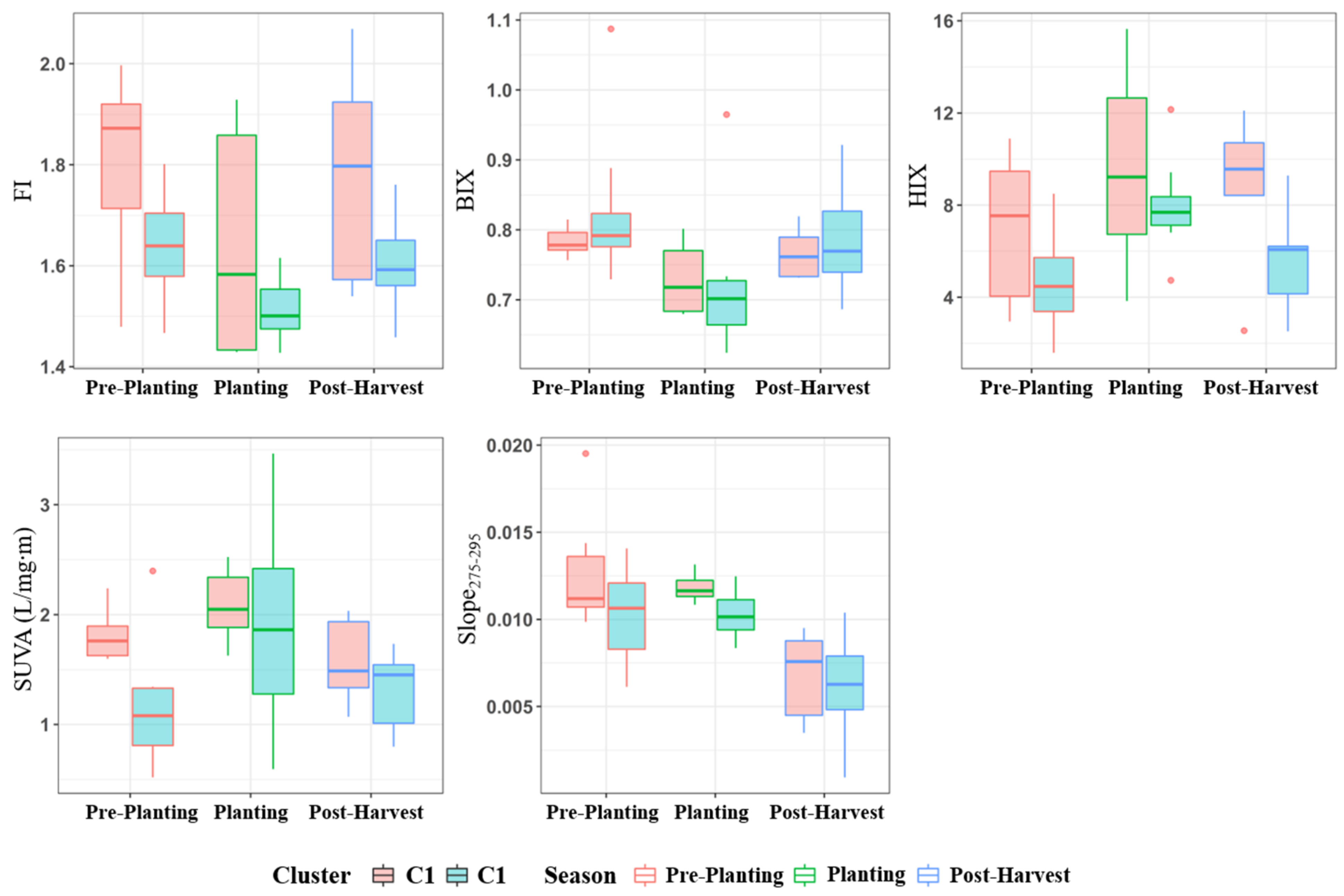
3.3. Comparison of Fluorescence and Spectroscopic Indices for Different Tributaries
3.4. Understanding of Pollutants Source using Fluorescence and Spectroschpic Characteristics
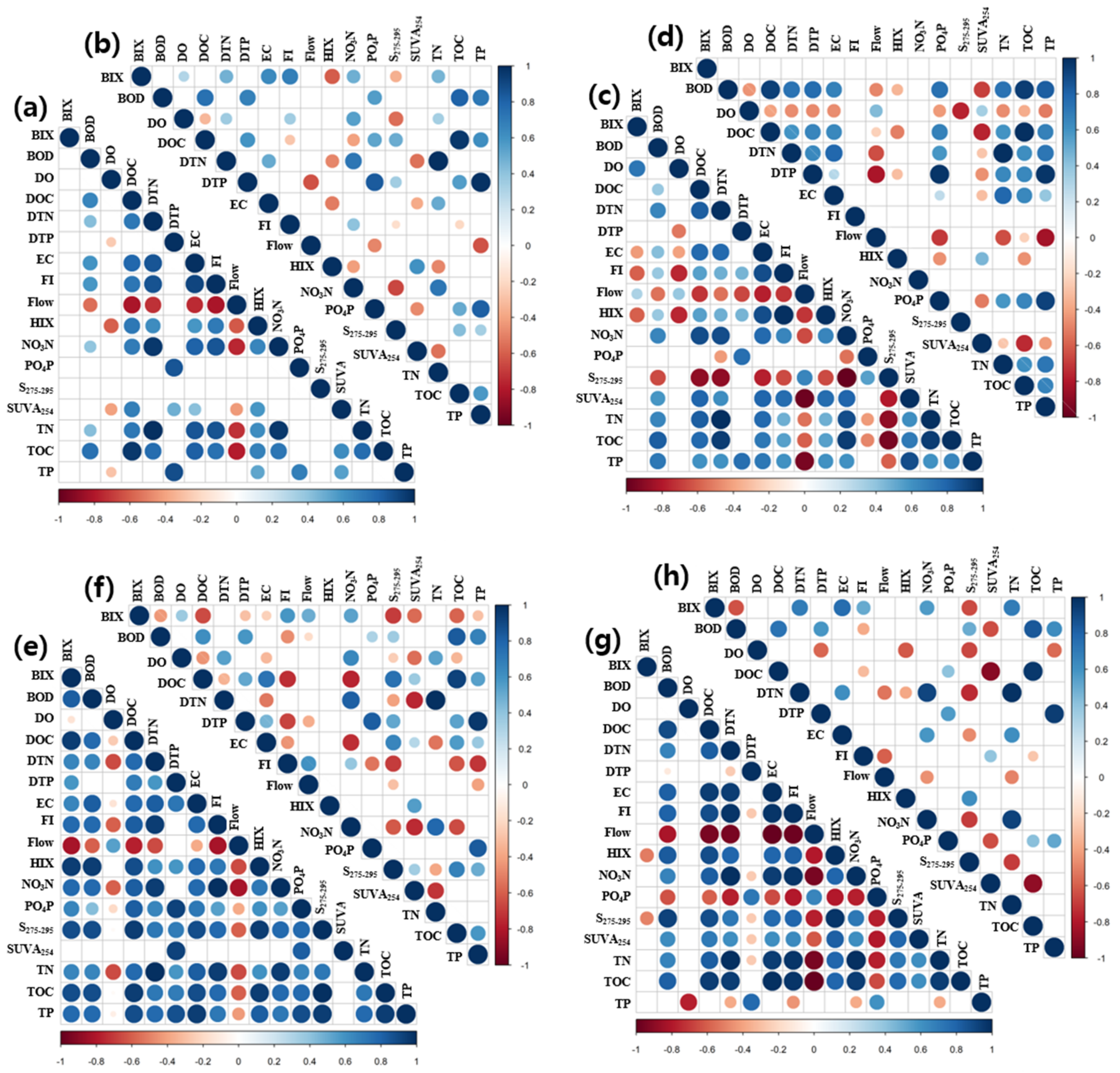
3.5. Correlation Analysis between Water Quality Parameters and Spectroscopic Indices for Different Groups According to Land Use and Farming Season
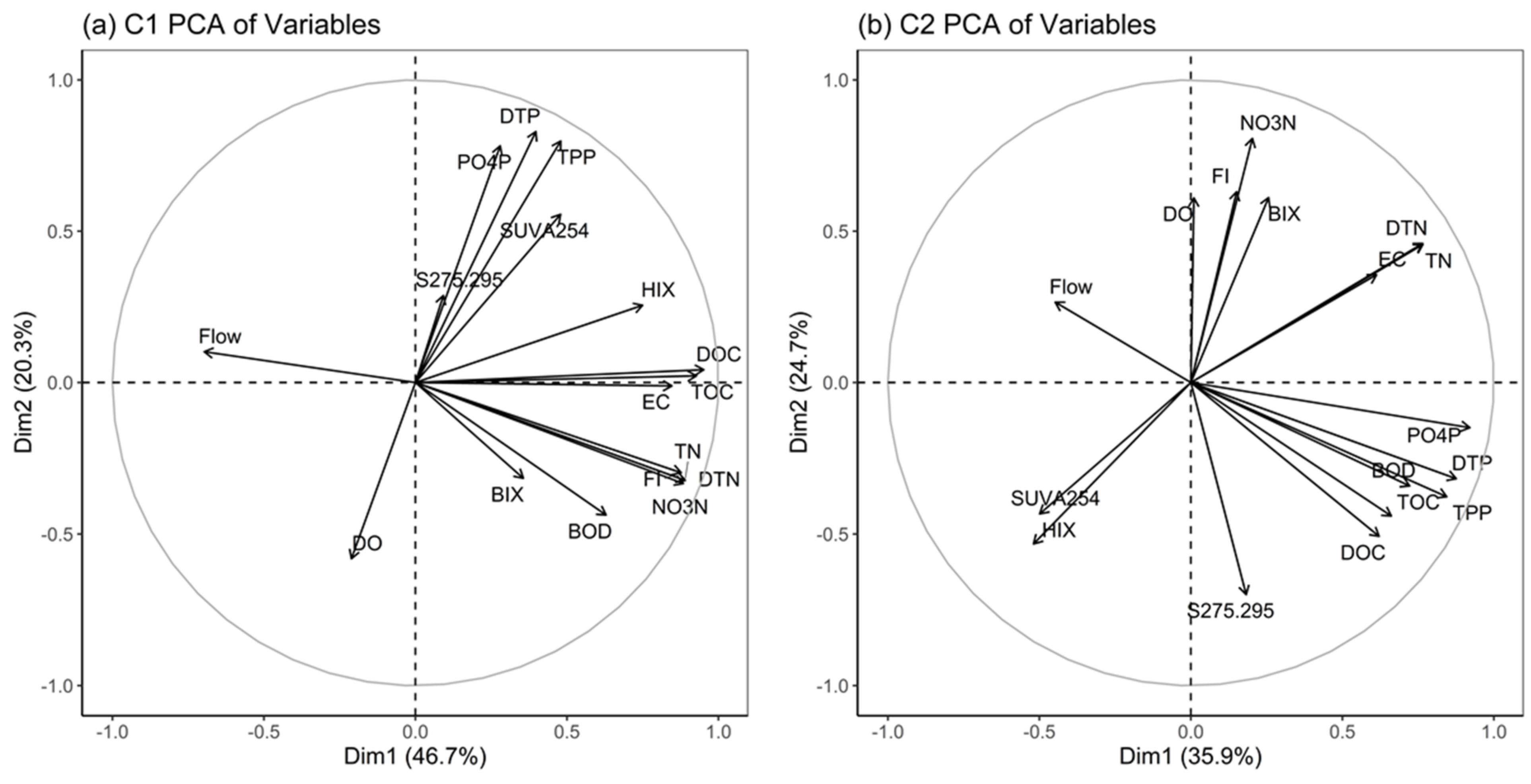
3.6. Principal Component Analysis Results of the Two Clusters
4. Conclusions
- (1)
- The stream (T1) where both FI and HIX were high, unlike general streams, was identified, and its EEM fluorescence patterns were analyzed. The fluorescence pattern of T1 was similar to that of the swine manure wastewater treatment plant. The method used in this study can be used in other watersheds to explore the effects of streams polluted by livestock effluent.
- (2)
- On land when using clustering analysis, the Miho upstream watershed was largely divided into watersheds (C1) with much livestock and many agricultural activities and other watersheds (C2). Furthermore, water quality data analysis by the agricultural activity period in each cluster revealed that most water pollution indices were high in areas where livestock farms were located, and the proportion of agricultural area was high.
- (3)
- Analysis of the correlations between fluorescence characteristics and general water quality parameters according to land use clustering revealed that the cluster with a large proportion of agricultural area showed high correlations of fluorescence characteristics and organic matter indices with nitrate ions. This suggests that the degree of nitrogenous and organic matter pollution can be determined using the fluorescence characteristics of watersheds with a large agricultural area proportion (over 40%, in this study). Therefore, it is possible to monitor water quality in various watersheds using clustering and correlation analysis.
- (4)
- The PCA results for C1 and C2 showed that C1 was greatly affected by pollutants due to livestock farming and agricultural activities (the first component). The PCA results for C2 showed that there was less influence of anthropogenic sources than in C1. In addition, the first component of C1, FI and HIX occupied a high weight which confirms that the influence of FDOM was greater in watersheds with a great deal of livestock farming and agricultural activities.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hur, J.; Lee, B.M.; Shin, H.S. Microbial degradation of dissolved organic matter (DOM) and its influence on phenanthrene-DOM interactions. Chemosphere 2011, 85, 1360–1367. [Google Scholar] [CrossRef] [PubMed]
- Gondar, D.; Thacker, S.A.; Tipping, E.; Baker, A. Functional variability of dissolved organic matter from the surface water of a productive lake. Water Res. 2008, 42, 81–90. [Google Scholar] [CrossRef] [Green Version]
- Leloup, M.; Nicolau, R.; Pallier, V.; Yéprémian, C.; Feuillade-Cathalifaud, G. Organic matter produced by algae and cyanobacteria: Quantitative and qualitative characterization. J. Environ. Sci. 2013, 25, 1089–1097. [Google Scholar] [CrossRef]
- Lee, Y.K.; Hur, J. Using two-dimensional correlation size exclusion chromatography (2D-CoSEC) to explore the size-dependent heterogeneity of humic substances for copper binding. Environ. Pollut. 2017, 227, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Hudson, P.K.; Gibson, E.R.; Young, M.A.; Kleiber, P.D.; Grassian, V.H. A newly designed and constructed instrument for coupled infrared extinction and size distribution measurements of aerosols. Aerosol Sci. Technol. 2007, 41, 701–710. [Google Scholar] [CrossRef] [Green Version]
- McKnight, D.M.; Boyer, E.W.; Westerhoff, P.K.; Doran, P.T.; Kulbe, T.; Andersen, D. Spectrofluorometric characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol. Oceanogr. 2001, 46, 38–48. [Google Scholar] [CrossRef]
- Chen, M.; Price, R.M.; Yamashita, Y.; Jaffé, R. Comparative study of dissolved organic matter from groundwater and surface water in the Florida coastal Everglades using multi-dimensional spectrofluorometry combined with multivariate statistics. Appl. Geochem. 2010, 25, 872–880. [Google Scholar] [CrossRef]
- Hoppe-Jones, C.; Dickenson, E.R.; Drewes, J.E. The role of microbial adaptation and biodegradable dissolved organic carbon on the attenuation of trace organic chemicals during groundwater recharge. Sci. Total Environ. 2012, 437, 137–144. [Google Scholar] [CrossRef]
- McDowell, W.H.; Zsolnay, A.; Aitkenhead-Peterson, J.A.; Gregorich, E.G.; Jones, D.L.; Jödemann, D.; Kalbitz, K.; Marschner, B.; Schwesig, D. A comparison of methods to determine the biodegradable dissolved organic carbon from different terrestrial sources. Soil Biol. Biochem. 2006, 38, 1933–1942. [Google Scholar] [CrossRef]
- Mermillod-Blondin, F.; Simon, L.; Maazouzi, C.; Foulquier, A.; Delolme, C.; Marmonier, P. Dynamics of dissolved organic carbon (DOC) through stormwater basins designed for groundwater recharge in urban area: Assessment of retention efficiency. Water Res. 2015, 81, 27–37. [Google Scholar] [CrossRef]
- Liu, W.X.; He, W.; Wu, J.Y.; Wu, W.J.; Xu, F.L. Effects of fluorescent dissolved organic matters (FDOMs) on perfluoroalkyl acids (PFAAs) in lake and river water. Sci. Total Environ. 2019, 666, 598–607. [Google Scholar] [CrossRef]
- Kalbitz, K.; Schmerwitz, J.; Schwesig, D.; Matzner, E. Biodegradation of soil-derived dissolved organic matter as related to its properties. Geoderma. 2003, 113, 273–291. [Google Scholar] [CrossRef]
- Leenheer, J.A.; Croué, J.-P. Peer reviewed: Characterizing aquatic dissolved organic matter. Environ. Sci. Technol. 2003, 37, 18A–26A. [Google Scholar] [CrossRef] [Green Version]
- Baker, A. Fluorescence excitation− emission matrix characterization of some sewage-impacted rivers. Environ. Sci. Technol. 2001, 35, 948–953. [Google Scholar] [CrossRef]
- Henderson, R.K.; Baker, A.; Murphy, K.R.; Hambly, A.; Stuetz, R.M.; Khan, S.J. Fluorescence as a potential monitoring tool for recycled water systems: A review. Water Res. 2009, 43, 863–881. [Google Scholar] [CrossRef]
- Peiris, R.H.; Budman, H.; Moresoli, C.; Legge, R.L. Identification of humic acid-like and fulvic acid-like natural organic matter in river water using fluorescence spectroscopy. Water Sci. Technol. 2011, 63, 2427–2433. [Google Scholar] [CrossRef]
- Henderson, R.K.; Baker, A.; Murphy, K.R.; Hambly, A.; Stuetz, R.M.; Khan, S.J. Properties of fluorescent dissolved organic matter in the Gironde Estuary. Org. Geochem. 2009, 40, 706–719. [Google Scholar]
- Zhang, X.; Li, B.; Deng, J.; Qin, B.; Wells, M.; Tefsen, B. Regional-scale investigation of dissolved organic matter and lead binding in a large impacted lake with a focus on environmental risk assessment. Water Res. 2020, 172, 115478. [Google Scholar] [CrossRef]
- Zhao, Y.; Song, K.; Wen, Z.; Fang, C.; Shang, Y.; Lv, L. Evaluation of CDOM sources and their links with water quality in the lakes of Northeast China using fluorescence spectroscopy. J. Hydrol. 2017, 550, 80–91. [Google Scholar] [CrossRef]
- Bernal, S.; Butturini, A.; Sabater, F. Variability of DOC and nitrate responses to storms in a small Mediterranean forested catchment. Hydrol. Earth Syst. Sci. 2002, 6, 1031–1041. [Google Scholar] [CrossRef] [Green Version]
- Molinero, J.; Burke, R.A. Effects of land use on dissolved organic matter biogeochemistry in piedmont headwater streams of the Southeastern United States. Hydrobiologia 2009, 635, 289–308. [Google Scholar] [CrossRef]
- Cohen, E.; Levy, G.J.; Borisover, M. Fluorescent components of organic matter in wastewater: Efficacy and selectivity of the water treatment. Water Res. 2014, 55, 323–334. [Google Scholar] [CrossRef]
- Zhao, Y.; Song, K.; Li, S.; Ma, J.; Wen, Z. Characterization of CDOM from urban waters in Northern-Northeastern China using excitation-emission matrix fluorescence and parallel factor analysis. Environ. Sci. Pollut. Res. Int. 2016, 23, 15381–15394. [Google Scholar] [CrossRef]
- Hur, J.W.; Kim, K.H.; Lee, J.J. Calculation (Computation) of Habitat Suitability Index for Swimming Fish Species Living in Miho Stream in Geum River Water System. Ecol. Resilient Infrastruct. 2021, 8, 9–21. [Google Scholar]
- ME, Framework Act on Environmental Policy in Korea. 2020. Available online: https://law.go.kr (accessed on 25 March 2022).
- ME, Environmental Geographic Information Service(EGIS). 2020. Available online: https://egis.me.go.kr/main.do (accessed on 13 November 2020).
- Hur, M.; Lee, I.; Tak, B.M.; Lee, H.J.; Yu, J.J.; Cheon, S.U.; Kim, B.S. Temporal shifts in cyanobacterial communities at different sites on the Nakdong River in Korea. Water Res. 2013, 47, 6973–6982. [Google Scholar] [CrossRef] [PubMed]
- Weishaar, J.L.; Aiken, G.R.; Bergamaschi, B.A.; Fram, M.S.; Fujii, R.; Mopper, K. Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ. Sci. Technol. 2003, 37, 4702–4708. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.R.; Stubbins, A.; Ritchie, J.D.; Minor, E.C.; Kieber, D.J.; Mopper, K. Erratum: Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnol. Oceanogr. 2009, 54, 1023. [Google Scholar]
- Søndergaard, M.; Stedmon, C.A.; Borch, N.H. Fate of terrigenous dissolved organic matter (DOM) in estuaries: Aggregation and bioavailability. Ophelia 2003, 57, 161–176. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, D.; Tang, Q.; Xu, H.; Huang, S.; Shang, D.; Liu, R. Water quality assessment and source identification of the Shuangji River (China) using multivariate statistical methods. PLoS ONE 2021, 16, e0245525. [Google Scholar] [CrossRef]
- de Andrade Costa, D.; Soares de Azevedo, J.P.; Dos Santos, M.A.; dos Santos Facchetti Vinhaes Assumpção, R. Water quality assessment based on multivariate statistics and water quality index of a strategic river in the Brazilian Atlantic Forest. Sci. Rep. 2020, 10, 22038. [Google Scholar] [CrossRef]
- Singh, S.; Dutta, S.; Inamdar, S. Land application of poultry manure and its influence on spectrofluorometric characteristics of dissolved organic matter. Agric. Ecosyst. Environ. 2014, 193, 25–36. [Google Scholar] [CrossRef]
- Cirpka, O.A.; Fienen, M.N.; Hofer, M.; Hoehn, E.; Tessarini, A.; Kipfer, R.; Kitanidis, P.K. Analyzing bank filtration by deconvoluting time series of electric conductivity. Ground Water 2007, 45, 318–328. [Google Scholar] [CrossRef]
- Vogt, T.; Hoehn, E.; Schneider, P.; Freund, A.; Schirmer, M.; Cirpka, O.A. Fluctuations of electrical conductivity as a natural tracer for bank filtration in a losing stream. Adv. Water Res. 2010, 33, 1296–1308. [Google Scholar] [CrossRef]
- Lazcano, C.; Zhu-Barker, X.; Decock, C. Effects of Organic Fertilizers on the Soil Microorganisms Responsible for N2O Emissions: A Review. Microorganisms 2021, 9, 983. [Google Scholar] [CrossRef]
- Torres-Martínez, J.A.; Mora, A.; Mahlknecht, J.; Daesslé, L.W.; Cervantes-Avilés, P.A.; Ledesma-Ruiz, R. Estimation of nitrate pollution sources and transformations in groundwater of an intensive livestock-agricultural area (Comarca Lagunera), combining major ions, stable isotopes and MixSIAR model. Environ. Pollut. 2021, 269, 115445. [Google Scholar] [CrossRef]
- Duan, S.; Banger, K.; Toor, G.S. Evidence of phosphate mining and Agriculture influence on Concentrations, forms, and ratios of nitrogen and phosphorus in a Florida River. Water 2021, 13, 1064. [Google Scholar] [CrossRef]
- Boyer, E.W.; Goodale, C.L.; Jaworski, N.A.; Howarth, R.W. Anthropogenic nitrogen sources and relationships to riverine nitrogen export in the northeastern USA. Biogeochemistry 2002, 57, 137–169. [Google Scholar] [CrossRef]
- Russell, M.J.; Weller, D.E.; Jordan, T.E.; Sigwart, K.J.; Sullivan, K.J. Net anthropogenic phosphorus inputs: Spatial and temporal variability in the Chesapeake Bay region. Biogeochemistry 2008, 88, 285–304. [Google Scholar] [CrossRef]
- Cory, R.M.; McKnight, D.M. Fluorescence spectroscopy reveals ubiquitous presence of oxidized and reduced quinones in dissolved organic matter. Environ. Sci. Technol. 2005, 39, 8142–8149. [Google Scholar] [CrossRef]
- Parlanti, E.; Wörz, K.; Geoffroy, L.; Lamotte, M. Dissolved organic matter fluorescence spectroscopy as a tool to estimate biological activity in a coastal zone submitted to anthropogenic inputs. Org. Geochem. 2000, 31, 1765–1781. [Google Scholar] [CrossRef]
- Birdwell, J.E.; Engel, A.S. Characterization of dissolved organic matter in cave and spring waters using UV–Vis absorbance and fluorescence spectroscopy. Org. Geochem. 2010, 41, 270–280. [Google Scholar] [CrossRef]
- Zsolnay, A.; Baigar, E.; Jimenez, M.; Steinweg, B.; Saccomandi, F. Differentiating with fluorescence spectroscopy the sources of dissolved organic matter in soils subjected to drying. Chemosphere 1999, 38, 45–50. [Google Scholar] [CrossRef]
- Chen, W.; Westerhoff, P.; Leenheer, J.A.; Booksh, K. Fluorescence excitation—Emission matrix regional integration to quantify spectra for dissolved organic matter. Environ. Sci. Techonl. 2003, 37, 5701–5710. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Zheng, P.; Ding, A.; Zhang, M.; Abbas, G.; Li, W. Source analysis of organic matter in swine wastewater after anaerobic digestion with EEM-PARAFAC. Envrion. Sci. Pollut. Res. Int. 2017, 24, 6770–6778. [Google Scholar] [CrossRef]
- Du, X.; Gu, L.P.; Wang, T.T.; Kou, H.J.; Sun, Y. The relationship between the molecular composition of dissolved organic matter and bioavailability of digestate during anaerobic digestion process: Characteristics, transformation and the key molecular interval. Bioresour. Technol. 2021, 342, 125958. [Google Scholar] [CrossRef]
- Saraceno, J.F.; Pellerin, B.A.; Downing, B.D.; Boss, E.; Bachand, P.A.; Bergamaschi, B.A. High-frequency in situ optical measurements during a storm event: Assessing relationships between dissolved organic matter, sediment concentrations, and hydrologic processes. J. Geophys. Res. 2009, 114, G4. [Google Scholar] [CrossRef]
- Nguyen, H.V.-M.; Hur, J.; Shin, H.-S. Changes in Spectroscopic and Molecular Weight Characteristics of Dissolved Organic Matter in a River During a Storm Event. Water Air Soil Pollut. 2010, 212, 395–406. [Google Scholar] [CrossRef]
- Nguyen, H.V.M.; Lee, M.H.; Hur, J.; Schlautman, M.A. Variations in spectroscopic characteristics and disinfection byproduct formation potentials of dissolved organic matter for two contrasting storm events. J. Hydrol. 2013, 481, 132–142. [Google Scholar] [CrossRef]
- Han, Z.; Xiao, M.; Yue, F.; Yi, Y.; Mostofa, K.M.G. Seasonal Variations of Dissolved Organic Matter by Fluorescent Analysis in a Typical River Catchment in Northern China. Water 2021, 13, 494. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, F.; Ghulam, A.; Trumbo, A.L.; Yang, J.; Ren, Y.; Jing, Y. Evaluation and estimation of surface water quality in an arid region based on EEM-PARAFAC and 3D fluorescence spectral index: A case study of the Ebinur Lake Watershed, China. Catena 2017, 155, 62–74. [Google Scholar] [CrossRef]
- Tang, J.; Li, X.; Cao, C.; Lin, M.; Qiu, Q.; Xu, Y.; Ren, Y. Compositional variety of dissolved organic matter and its correlation with water quality in peri-urban and urban river watersheds. Ecol. Indic. 2019, 104, 459–469. [Google Scholar] [CrossRef]
- Mouri, G.; Takizawa, S.; Oki, T. Spatial and temporal variation in nutrient parameters in stream water in a rural-urban catchment, Shikoku, Japan: Effects of land cover and human impact. J. Environ. Manag. 2011, 92, 1837–1848. [Google Scholar] [CrossRef]
- Dalzell, B.J.; King, J.Y.; Mulla, D.J.; Finlay, J.C.; Sands, G.R. Influence of subsurface drainage on quantity and quality of dissolved organic matter export from agricultural landscapes. J. Geophys. Res. Biogeosciences 2011, 116. [Google Scholar] [CrossRef]
- Hernes, P.J.; Spencer, R.G.; Dyda, R.Y.; Pellerin, B.A.; Bachand, P.A.; Bergamaschi, B.A. DOM composition in an agricultural watershed: Assessing patterns and variability in the context of spatial scales. Geochim. Cosmochim. Acta 2013, 121, 599–610. [Google Scholar] [CrossRef]
- Zeng, F.; Zuo, Z.; Mo, J.; Chen, C.; Yang, X.; Wang, J.; Wang, Y.; Zhao, Z.; Chen, T.; Li, Y.; et al. Runoff Losses in Nitrogen and Phosphorus From Paddy and Maize Cropping System: A Field Study in Dongjiang Basin, South China. Front. Plant Sci. 2021, 12, 1593. [Google Scholar] [CrossRef]
- Cui, N.; Cai, M.; Zhang, X.; Abdelhafez, A.A.; Zhou, L.; Sun, H.; Chen, G.; Zou, G.; Zhou, S. Runoff loss of nitrogen and phosphorus from a rice paddy field in the east of China: Effect of long-term chemical N fertilizer and organic manure applications. Glob. Ecol. Conserv. 2020, 22, e01011. [Google Scholar] [CrossRef]

| Site | Cows | Dairy Cows | Pigs |
|---|---|---|---|
| T1 | 1807 (384 heads/km2) | - | 14,282 (3039 heads/km2) |
| T2 | 1857 (95 heads/km2) | - | - |
| T3 | 2507 (74 heads/km2) | 61 (2 heads/km2) | 8184 (241 heads/km2) |
| T4 | 875 (38 heads/km2) | 421 (18 heads/km2) | 5468 (240 heads/km2) |
| T5 | 780 (26 heads/km2) | 54 (2 heads/km2) | 1550 (52 heads/km2) |
| T6 | 1059 (74 heads/km2) | 460 (32 heads/km2) | 6596 (461 heads/km2) |
| Total | 19,635 (69 heads/km2) | 2838 (10 heads/km2) | 121,171 (425 heads/km2) |
| Site | Urban Area (%) | Agricultural Area (%) | Forest Area (%) | Pasture Area (%) | Barren Area (%) | Water Bodies Area (%) | Watershed Area (km2) |
|---|---|---|---|---|---|---|---|
| T1 | 28.14 | 44.90 | 4.46 | 16.26 | 4.92 | 1.32 | 4.7 |
| T2 | 23.53 | 41 | 11.38 | 16.73 | 5.22 | 2.15 | 19.6 |
| T3 | 22.67 | 30.37 | 23.65 | 16.81 | 3.53 | 2.97 | 34.0 |
| T4 | 26.85 | 25.22 | 28.50 | 12.69 | 3.22 | 3.52 | 22.8 |
| T5 | 14.95 | 18.86 | 49.94 | 10.34 | 2.77 | 3.15 | 29.6 |
| T6 | 18.83 | 30.84 | 32.37 | 12.58 | 4.26 | 1.12 | 14.3 |
| Total | 14.11 | 34.91 | 25.79 | 16.94 | 5.33 | 2.92 | 285.3 |
| Index | Calculation | Description |
|---|---|---|
| Fluorescence index (FI) | Terrestrial DOM < 1.3–1.4 1.8–1.9 < algal–microbial DOM [6] | |
| Biological index (BIX) | Less autochthonous DOM < 0.6–0.8 0.8–1.0 < Autochthonous and newly generated sources [17] | |
| Humidification index (HIX) | Higher HIX indicates increasing humidification [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeon, P.; Cho, S.; Hur, J.; Mun, H.; Chae, M.; Cho, Y.; Seok, K.; Hong, S. Close Association between Stream Water Quality and Fluorescence Properties of Dissolved Organic Matter in Agriculture-Dominated Watersheds. Water 2022, 14, 2459. https://doi.org/10.3390/w14162459
Jeon P, Cho S, Hur J, Mun H, Chae M, Cho Y, Seok K, Hong S. Close Association between Stream Water Quality and Fluorescence Properties of Dissolved Organic Matter in Agriculture-Dominated Watersheds. Water. 2022; 14(16):2459. https://doi.org/10.3390/w14162459
Chicago/Turabian StyleJeon, Pilyong, Sohyun Cho, Jin Hur, Hyunsaing Mun, Minhee Chae, Yoonhae Cho, Kwangseol Seok, and Seonhwa Hong. 2022. "Close Association between Stream Water Quality and Fluorescence Properties of Dissolved Organic Matter in Agriculture-Dominated Watersheds" Water 14, no. 16: 2459. https://doi.org/10.3390/w14162459






