Development, Synthesis and Characterization of Tannin/Bentonite-Derived Biochar for Water and Wastewater Treatment from Methylene Blue
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
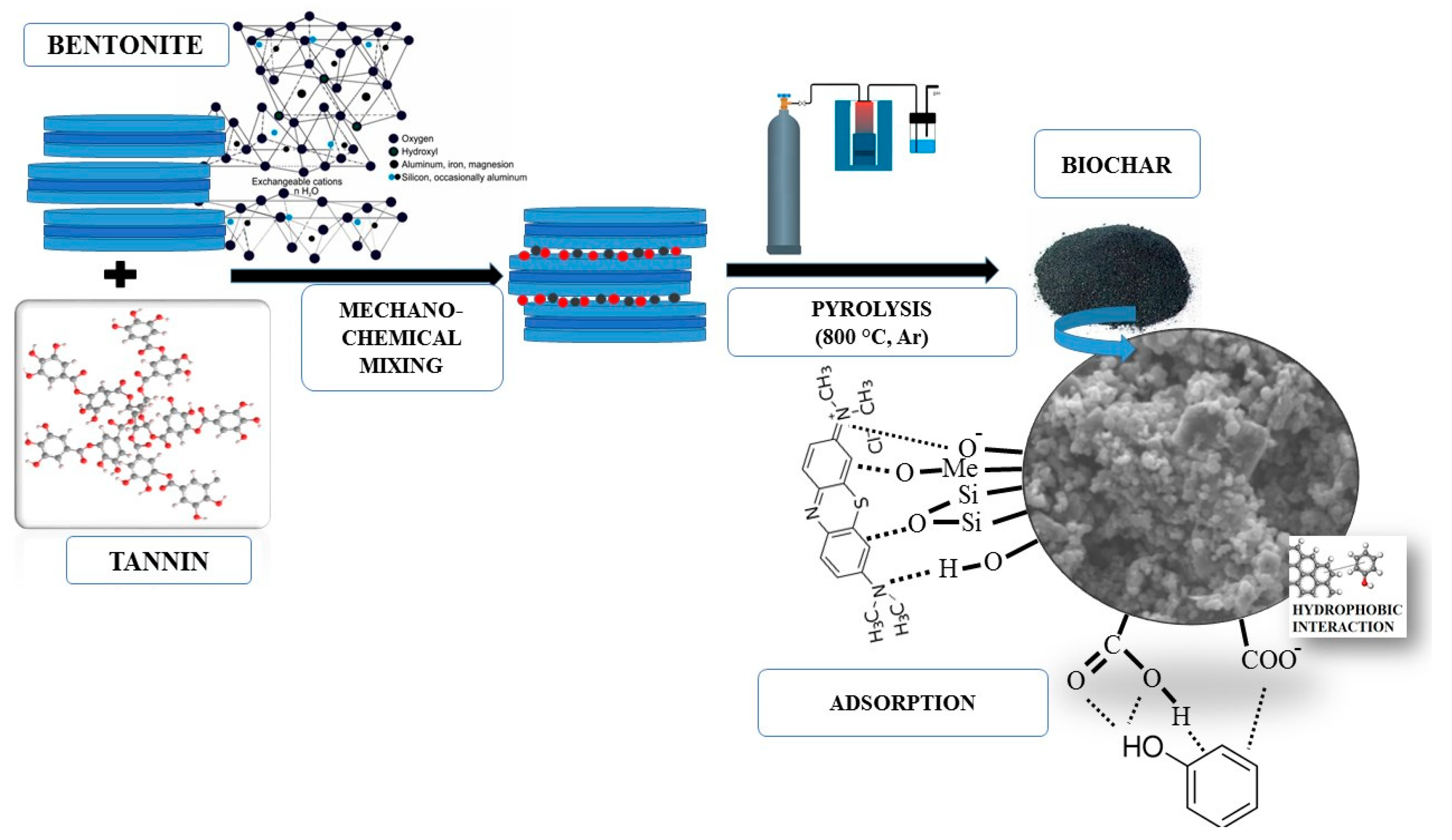
2.2. Materials Preparation
2.3. Methods
3. Results
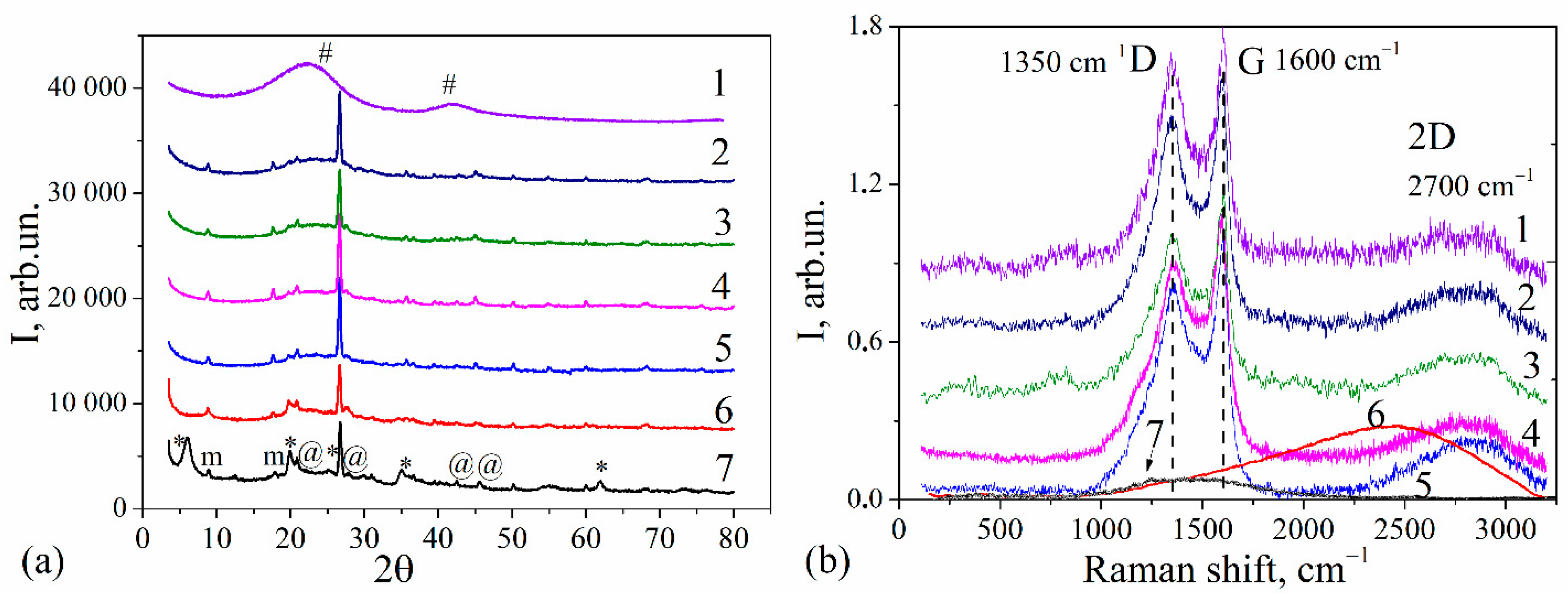
3.1. Structural Characteristics
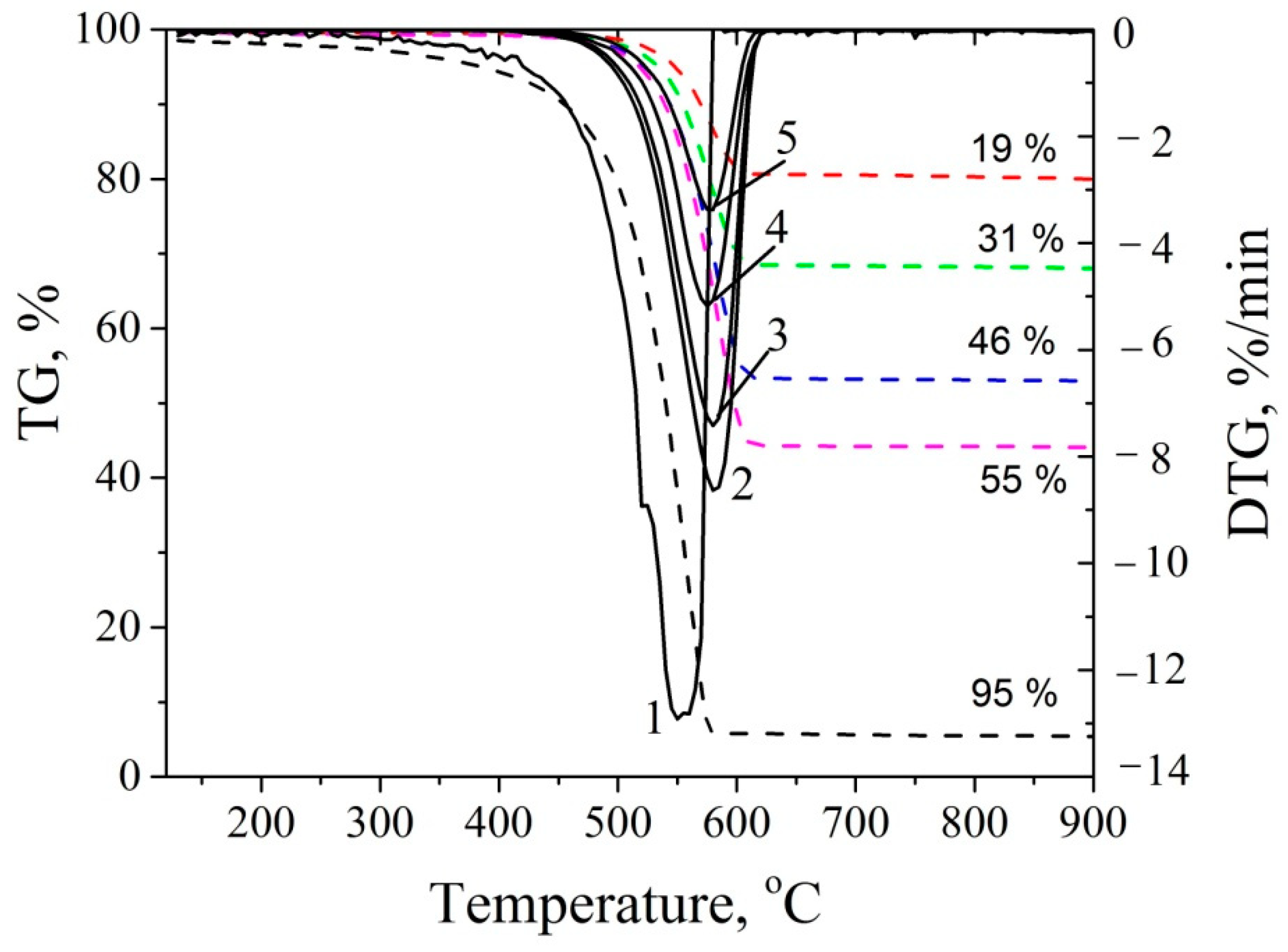
3.2. Thermal Properties
3.3. Morphological Properties
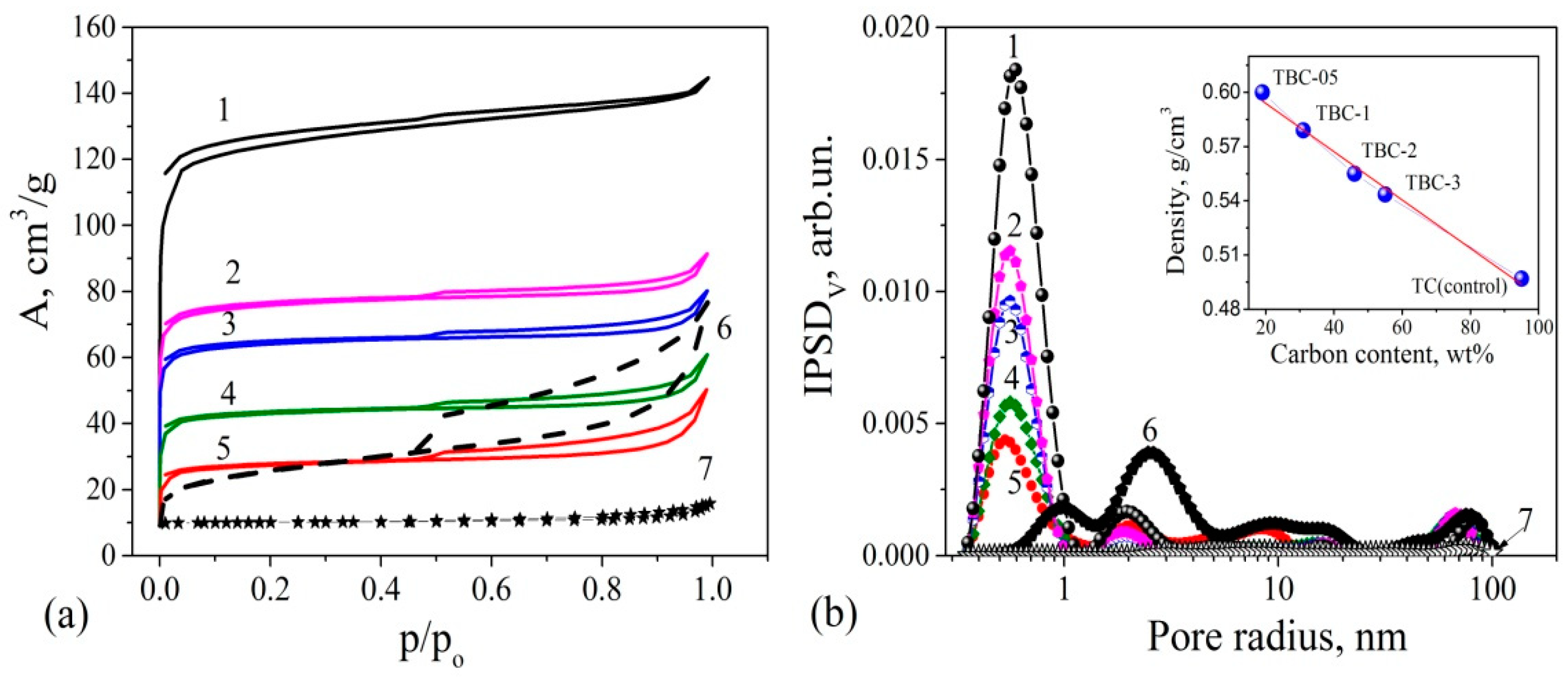
3.4. Porosity and Acid–Base Properties
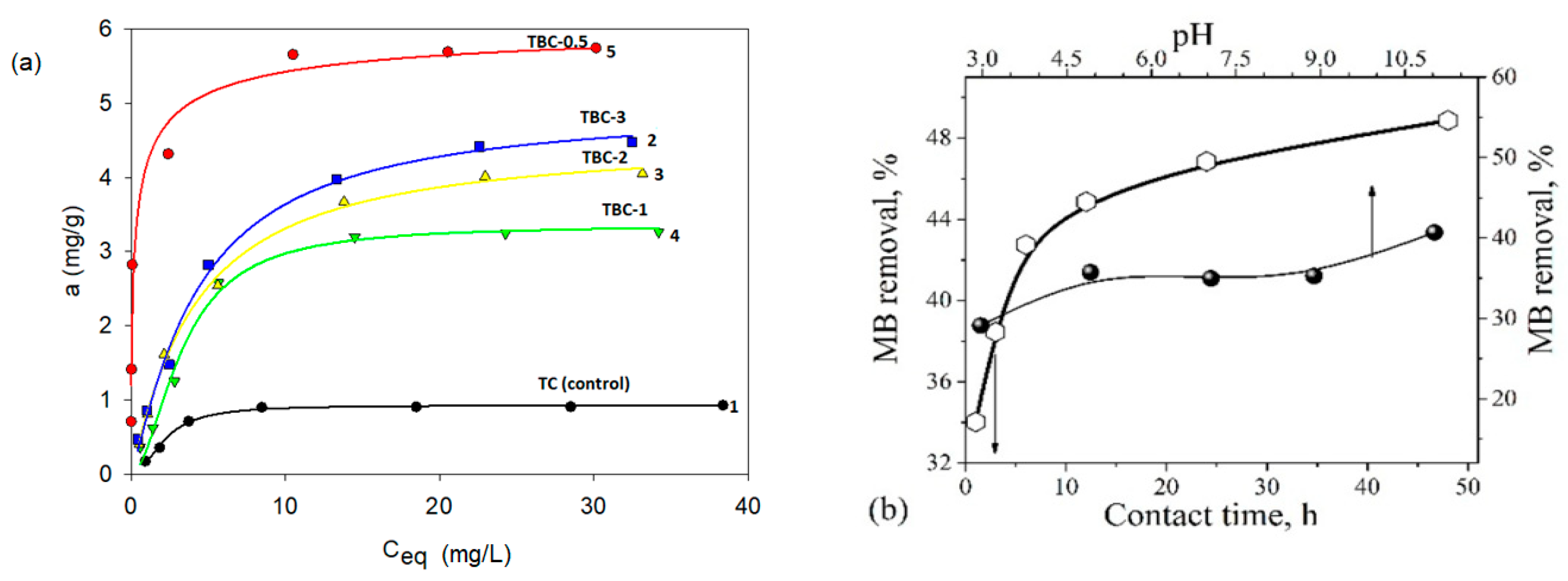
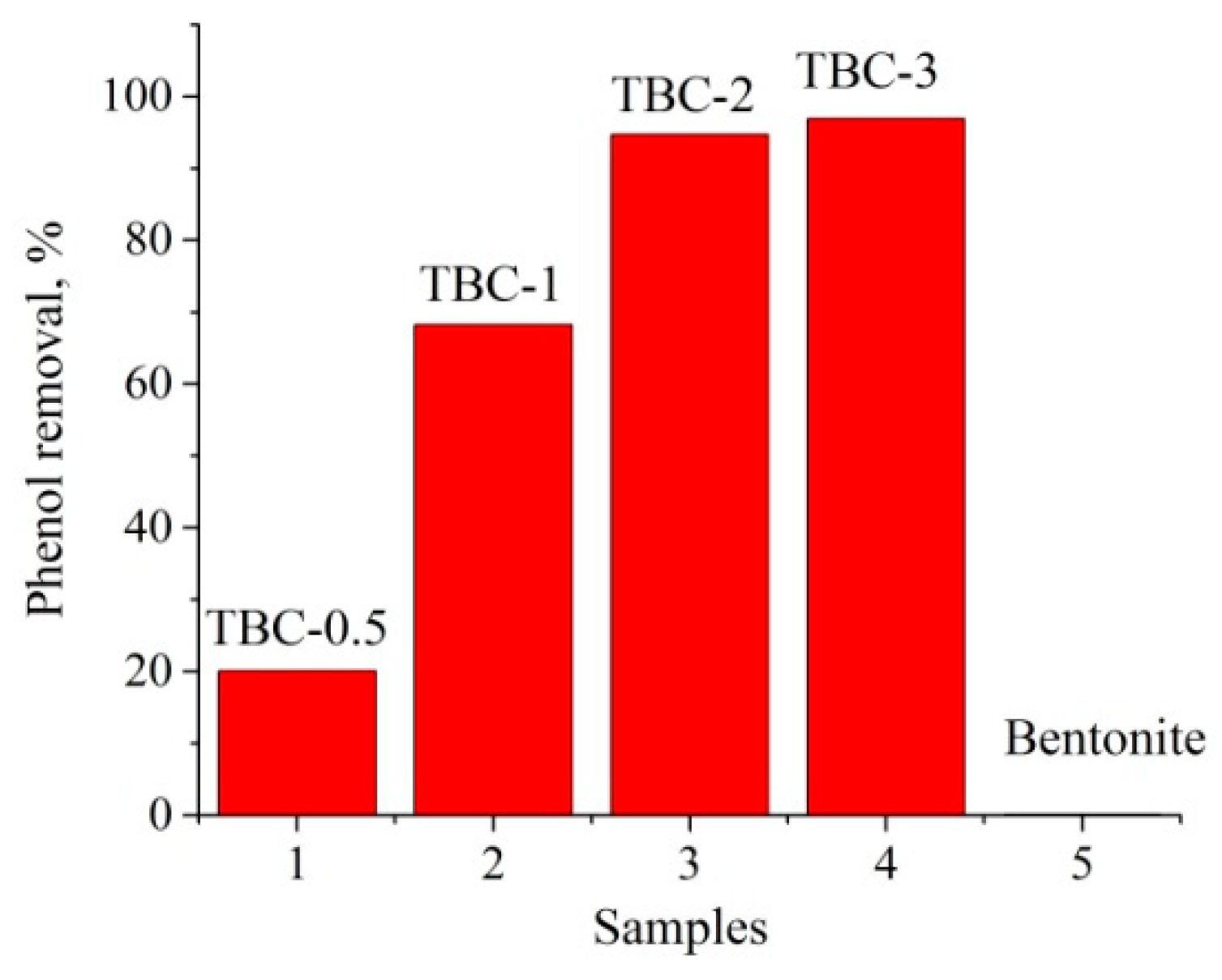
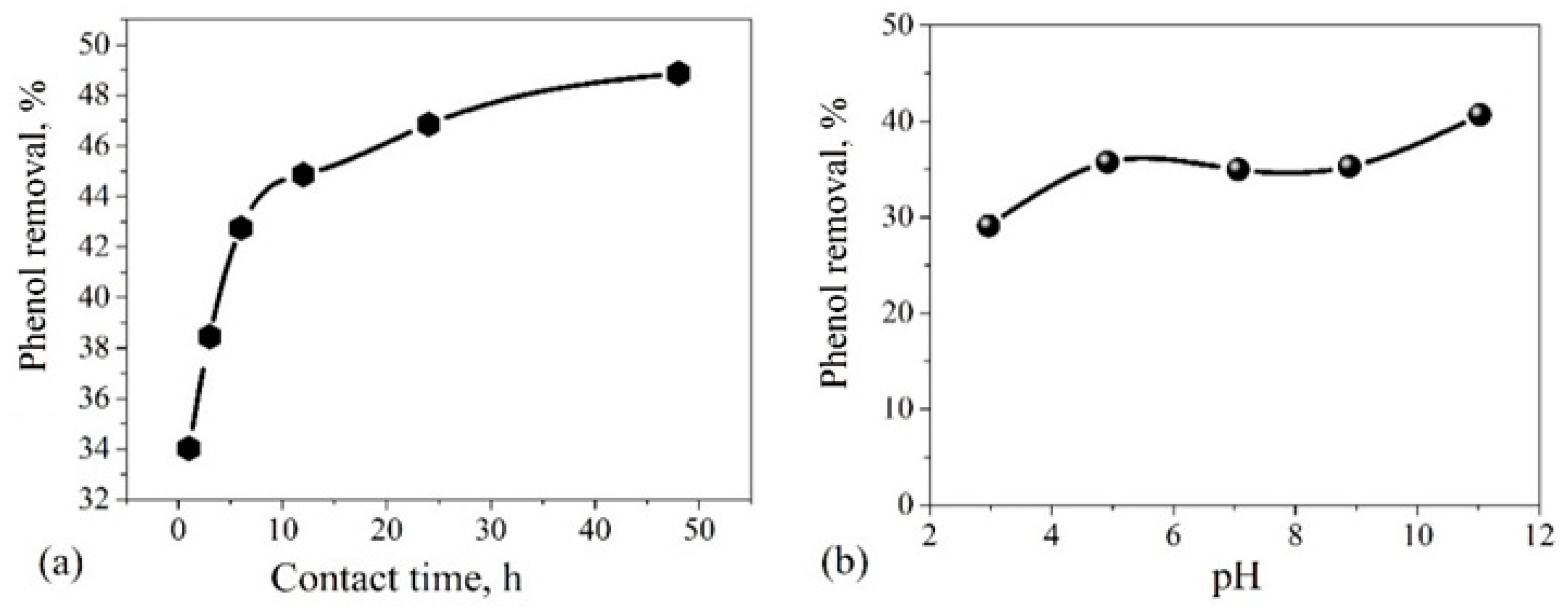
3.5. Adsorption Characteristics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Youcef, L.D.; Belaroui, L.S.; López-Galindo, A. Adsorption of a cationic methylene blue dye on an Algerian palygorskite. Appl. Clay Sci. 2019, 179, 105145. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; et al. Review on Methylene Blue: Its Properties, Uses, Toxicity and Photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Deng, H.; He, H.; Li, W.; Abbas, T.; Liu, Z. Characterization of amphoteric bentonite-loaded magnetic biochar and its adsorption properties for Cu2+ and tetracycline. PeerJ 2022, 10, e13030. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.H.; Ahmad, A.L.; Latiff, K.N.A. Adsorption of basic dye (methylene blue) onto activated carbon prepared from rattansaw dust. Dye. Pigment. 2007, 75, 143–149. [Google Scholar] [CrossRef]
- Bosacka, A.; Zienkiewicz-Strzalka, M.; Wasilewska, M.; Derylo-Marczewska, A.; Podkościelna, B. Physicochemical and Adsorption Characteristics of Divinylbenzene-co-Triethoxyvinylsilane Microspheres as Materials for the Removal of Organic Compounds. Molecules 2021, 8, 2396. [Google Scholar] [CrossRef] [PubMed]
- Do, J.L.; Friščić, T. Mechanochemistry: A Force of Synthesis. ACS Cent. Sci. 2017, 3, 13–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- AbdEl-Magied, M.O.; Hassan, A.M.A.; Gad, H.M.H.; Mohammaden, T.F.; Youssef, M.A.M. Removal of nickel (II) ions from aqueous solutions using modified activated carbon: Akinetic and equilibrium study. J. Dispers. Sci. Technol. 2018, 39, 862–873. [Google Scholar] [CrossRef]
- Kumar, A.; Lingfa, P. Sodium bentonite and kaolin clays: Comparative study on their FT-IR, XRF, and XRD. Mater. Today Proc. 2020, 22, 737–742. [Google Scholar] [CrossRef]
- Xu, C.; De, S.; Balu, A.M.; Ojeda, M.; Luque, R. Mechanochemical synthesis of advanced nanomaterials for catalytic applications. Chem. Commun. 2015, 51, 6698–6713. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, T. Mechanochemical synthesis of metal oxide nanoparticles. Commun. Chem. 2021, 4, 143. [Google Scholar] [CrossRef]
- Krupska, T.; Golovan, A.; Lupashku, T.; Povar, I.; Spinu, O.; Kartel, M.; Turov, V. The nanocomposite system based on tannin and methylsilica for the activation of development of seeds. Dopov. Nac. Akad. Nauk. 2017, 10, 83–90. [Google Scholar] [CrossRef]
- Arbenz, A.; Avérous, L. Chemical modification of tannins to elaborate aromatic biobased macromolecular architectures. Green Chem. 2015, 17, 2626–2646. [Google Scholar] [CrossRef] [Green Version]
- Kozakevych, R.; Bolbukh, Y.; Lupascu, L.; Lupascu, T.; Tertykh, V. Polymeric Composite Films with Controlled Release ofNatural Antioxidant Enoxil. In NANO 2017 Nanochemistry, Biotechnology, Nanomaterials, and Their Applications; Springer Proceedings in Physics; Fesenko, O., Yatsenko, L., Eds.; Springer: Cham, Switzerland, 2017; Volume 214, pp. 149–164. [Google Scholar]
- Hintz, T.; Matthews, K.K.; Di, R. The Use of Plant Antimicrobial Compounds for Food Preservation. BioMed Res. Int. 2015, 2015, 246264. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Martín, J.; Beltrán-Heredia, C.; Solera-Hernández, C. Surface water and wastewater treatment using a new tannin-based coagulant. J. Environ. Manag. 2010, 91, 2051–2058. [Google Scholar] [CrossRef]
- Vu, T.T.; Kim, H.; Tran, V.K.; Vu, H.D.; Hoang, T.X.; Han, J.W.; Choi, Y.H.; Jang, K.S.; Choi, G.J.; Kim, J.-C. Antibacterial activity of tannins isolated from Sapium baccatumextractanduse for control of tomato bacterial wilt. PLoS ONE 2017, 12, e0181499. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Liu, P. Composition and biological activities of hydrolysable tannins of fruits of phyllanthusemblica. J. Agric. Food Chem. 2014, 62, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Gonta, A.; Lupascu, T.; Timbaliuc, N.; Meghea, A. Obtaining and characterizing modified tannins by physical-chemical methods. Open Chem. 2014, 12, 757–762. [Google Scholar] [CrossRef]
- Ismadji, S.; Tong, D.S.; Soetaredjo, F.E.; Ayucitra, A.; Yu, W.H.; Zhou, C.H. Bentonite hydrochar composite for removal of ammonium from Koifishtank. Appl. Clay Sci. 2016, 119, 146–154. [Google Scholar] [CrossRef]
- Available online: https://forum.md/ru/741258 (accessed on 30 May 2022).
- Aprianti, N.; Faizal, M.; Said, M.; Nasir, S. Catalytic gasification of oil palm empty fruit bunch by using Indonesian bentonite as the catalyst. J. Appl. Eng. Sci. 2021, 2, 334–343. [Google Scholar] [CrossRef]
- Moosavi, M. Bentonite clay as a natural remedy: A brief review. Bentonite Clay A Nat. Rem. A Brief Rev. 2017, 46, 1176–1183. [Google Scholar]
- Dou, G.; Goldfarb, J.L. In situ upgrading of pyrolysis biofuels by bentonite clay with simultaneous production of heterogeneous adsorbents for watertreatment. J. Appl. Eng. Sci. 2017, 195, 273–283. [Google Scholar]
- Sternik, D.; Galaburda, M.; Bogatyrov, V.M.; Gun’Ko, V.M. Influence of the Synthesis Method on the Structural Characteristics of Novel Hybrid Adsorbents Based on Bentonite. Colloids Interfaces 2019, 3, 18. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Chen, B. Sorption of naphthalene and 1-naphthol by biochars of orange peels with different pyrolytic temperatures. Chemosphere 2009, 76, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Uchimiya, M.; Wartelle, L.H.; Lima, I.M.; Klasson, K.T. Sorption of Deisopropylatrazine on Broiler Litter Biochars. J. Agric. Food Chem. 2009, 58, 12350–12356. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Islam, T.; Li, Y.; Cheng, H. Biochars and engineered biochars for water and soil remediation: A review. Sustainability 2021, 13, 9932. [Google Scholar] [CrossRef]
- Yahaya Bermakai, M.; Farahin Jafri, N.; Abdul Hadi, N. Biomass activated carbon properties through activation method forsupercapacitor- a mini-review. Sustainability 2021, 9, 8–13. [Google Scholar]
- Kumar, A.; Jena, H.M. Preparation and characterization of high surface area activated carbon from Fox nut (Euryaleferox) shell by chemical activation with H3PO4. Results Phys. 2016, 6, 651–658. [Google Scholar] [CrossRef] [Green Version]
- Ambroz, F.; Macdonald, T.J.; Martis, V.; Parkin, I.P. Evaluation of the BET Theory for the Characterization of Meso and Microporous MOFs. Small Methods 2018, 2, 1800173. [Google Scholar] [CrossRef] [Green Version]
- Gun’ko, V.M.; Mikhalovsky, S.V. Evaluation of slitlike porosity of carbon adsorbents. Carbon 2004, 42, 843–849. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Raman spectroscopy of amorphous, nanostructured, diamond-like carbon, and nanodiamond. Philos. Trans. R. Soc. Lond. A Math. Phys. Eng. Sci. 2004, 362, 2477–2512. [Google Scholar] [CrossRef]
- Fang, Y.; Lv, Y.; Che, R.; Wu, H.; Zhang, X.; Gu, D.; Zheng, G.; Zhao, D. Two-Dimensional Mesoporous Carbon Nanosheets and Their Derived Graphene Nanosheets: Synthesis and Efficient Lithium Ion Storage. J. Am. Chem. Soc. 2013, 135, 1524–1530. [Google Scholar] [CrossRef] [PubMed]
- Sergeeva, A.V.; Zhitova, E.S.; Nuzhdaev, A.A.; Zolotarev, A.A.; Bocharov, V.N.; Ismagilova, R.M. Infrared and Raman Spectroscopy of Ammonio voltaite,(NH4)2Fe2+5Fe3+3Al(SO4)12(H2O)18. Minerals 2020, 10, 781. [Google Scholar] [CrossRef]
- Reddy, T.R.; Kaneko, S.; Endo, T.; Reddy, S.L. Spectroscopic Characterization of Bentonite. J. Lasers Opt. Photonics 2017, 4, 171. [Google Scholar]
- Koswojo, R.; Utomo, R.P.; Ju, Y.H.; Ayucitra, A.; Soetaredjo, F.E.; Sunarso, J.; Ismadji, S. AcidGreen 25 removal from wastewater by organo-bentonite fromPacitan. Appl. Clay Sci. 2010, 48, 81–86. [Google Scholar] [CrossRef]
- Mierzwa-Hersztek, M.; Gondek, K.; Nawrocka, A.; Pińkowska, H.; Bajda, T.; Stanek-Tarkowska, J.; Szostek, M. FT-IR analysis and the content of phenolic compounds in exogenous organic matter produced from plant biomass. J. Elem. 2019, 24, 879–896. [Google Scholar] [CrossRef]
- Tomczyk, A.; Sokolowska, Z.; Boguta, P. Biochar physicochemical properties: Pyrolysis temperature and feedstock in deffects. Rev. Environ. Sci. Bio/Technol. 2020, 19, 191–215. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Na, T.; Wang, X. Effect of temperature on the structural and physicochemical properties of biochar with apple treebranches as feed stock material. Energies 2017, 10, 1293. [Google Scholar] [CrossRef] [Green Version]
- Blachnio, M.; Derylo-Marczewska, A.; Charmas, B.; Zienkiewicz-Strzalka, M.; Bogatyrov, V.; Galaburda, M. Activated Carbon from Agricultural Wastes for Adsorption of Organic Pollutants. Molecules 2020, 25, 5105. [Google Scholar] [CrossRef]
- Zhu, Y.; Yi, B.; Yuan, Q.; Wu, Y.; Wang, M.; Yan, S. Removal of methylene blue from aqueous solution by cattlemanure-derived low temperature biochar. Molecules 2018, 8, 19917–19929. [Google Scholar]
- DeCastro, M.L.F.A.; Abad, M.L.B.; Sumalinog, D.A.G.; Abarca, R.R.M.; Paoprasert, P.; Luna, M.D.G.d. Adsorption of Methylene Blue dye and Cu(II) ions on EDTA-modified bentonite: Isotherm, kinetic and thermodynamic studies. Sustain. Environ. Res. 2018, 28, 197–205. [Google Scholar] [CrossRef]
- Derakhshan, Z.; Baghapour, M.A.; Ranjbar, M.; Faramarzian, M. Adsorption of Methylene Blue Dye from Aqueous Solutions by Modified Pumice Stone: Kinetics and Equilibrium Studies. Health Scope 2013, 3, 136–144. [Google Scholar] [CrossRef] [Green Version]
- Veliev, T.; Öztürk, S.; Veli, A.; Fatullayev, A.G. Application of Diffusion Model for Adsorption of Azo Reactive Dye on Pumice. Pol. J. Environ. Stud. 2006, 15, 345–353. [Google Scholar]
- Jawad, A.H.; Rashid, R.A.; Ishak, M.A.M.; Wilson, L.D. Adsorption of methylene blue onto activated carbon developed from biomass waste by H2SO4 activation: Kinetic, equilibrium and thermodynamic studies. Desalination Water Treat. 2016, 57, 25194–25206. [Google Scholar] [CrossRef]
- Vijayalakshmi, R.; Jobish, J. Adsorption of Methylene Blue onto Natural Rubber/Chitosan Blends. Int. J. Polym. Mater. 2011, 60, 766–775. [Google Scholar]
- Fatiha, M.; Belkacem, B. Adsorption of methylene blue from aqueous solutions using natural clay. J. Matter Environ. Sci. 2016, 7, 285–292. [Google Scholar]


| Sample | Component Ratio, g | |
|---|---|---|
| Tannin | Bentonite | |
| TC (control) | 10 | - |
| TBC-1 | 10 | 10 |
| TBC-2 | 20 | 10 |
| TBC-3 | 30 | 10 |
| TBC-0.5 | 6 | 10 |
| No. | Sample | PDF-2 Card No. | Phase | FWHM (cm−1) | Center (cm−1) | D/G Intensity Ratio (ID/IG) | ||
|---|---|---|---|---|---|---|---|---|
| D-Band | G-Band | D-Band | G-Band | |||||
| 1 | TC (control) | - | Amorphous carbon | 126.3 | 126.3 | 1340.4 | 1598.0 | 0.8 |
| 2 | TBC-1 | - 85–795 7–42 - | Montmorillonitedestr. Quartz Muscovite Amorphous carbon | 145.3 | 145.3 | 1354.8 | 1590.0 | 0.8 |
| 3 | TBC-2 | - 85–795 7–42 - | Montmorillonitedestr. Quartz Muscovite Amorphous carbon | 155.6 | 154.8 | 1349.6 | 1600.2 | 0.8 |
| 4 | TBC-3 | - 85–795 7–42 - | Montmorillonitedestr. Quartz Muscovite Amorphous carbon | 144.0 | 144.0 | 1340.8 | 1600.2 | 0.8 |
| 5 | TBC-0.5 | - 85–795 7–42 - | Montmorillonitedestr. Quartz Muscovite Amorphous carbon | 145.3 | 145.3 | 1352.9 | 1600.2 | 0.9 |
| 6 | Bentonite | 3–19 85–795 7–42 | Montmorillonite Quartz Muscovite | - | - | - | - | - |
| 7 | Bentonite (pyrolyzed) | - 85–795 7–42 | Montmorillonitedestr. Quartz Muscovite | - | - | - | - | - |
| No. | Sample | SBET (m2/g) | Smicro, m2/g | Smeso, m2/g | Smacro, m2/g | Vp (cm3/g) | Vmicro (cm3/g) | Vmeso (cm3/g) | Vmacro (cm3/g) | Vmicro/Vp | Vmeso/Vp | pHPZC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TBC-0.5 | 86 | 75.2 | 9.0 | - | 0.08 | 0.05 | 0.03 | 0.00 | 0.61 | 0.39 | 8.55 |
| 2 | TBC-1 | 128 | 121.1 | 6.2 | 0.2 | 0.09 | 0.06 | 0.02 | 0.01 | 0.72 | 0.20 | 6.41 |
| 3 | TBC-2 | 194 | 188.2 | 5.4 | 0.2 | 0.13 | 0.10 | 0.02 | 0.01 | 0.80 | 0.13 | 6.39 |
| 4 | TBC-3 | 230 | 223.4 | 6.2 | 0.2 | 0.14 | 0.11 | 0.02 | 0.01 | 0.81 | 0.12 | 5.84 |
| 5 | TC(control) | 380 | 368.5 | 10.8 | 0.1 | 0.23 | 0.20 | 0.02 | 0.01 | 0.86 | 0.10 | 6.95 |
| 6 | Bentonite | 86 | 28.3 | 57.0 | 0.6 | 0.12 | 0.01 | 0.08 | 0.02 | 0.08 | 0.70 | 7.90 |
| 7 | Bentonite pyrolyzed | 3 | 0.6 | 1.9 | 0.2 | 0.01 | - | 0.01 | 0.00 | 0.59 | 0.39 | 8.42 |
| No. | Sample | Langmuir Isotherm | ||
|---|---|---|---|---|
| R2 | KL (L/mg) | am (mg/g) | ||
| 1 | TC (control) | 0.994 | 0.39 | 1.00 |
| 2 | TBC-0.5 | 0.999 | 3.61 | 5.78 |
| 3 | TBC-1 | 0.983 | 0.19 | 3.90 |
| 4 | TBC-2 | 0.996 | 0.21 | 4.74 |
| 5 | TBC-3 | 0.995 | 0.20 | 5.26 |
| Freundlich Isotherm | ||||
| No. | Sample | R2 | KF(mg/g) | n |
| 1 | TC (control) | 0.783 | 0.28 | 2.50 |
| 2 | TBC-0.5 | 0.847 | 3.03 | 4.21 |
| 3 | TBC-1 | 0.907 | 0.60 | 1.77 |
| 4 | TBC-2 | 0.928 | 0.78 | 1.83 |
| 5 | TBC-3 | 0.957 | 0.87 | 1.85 |
| No. | Sample | Langmuir Isotherm | |||
|---|---|---|---|---|---|
| R2 | KL (L/mg) | am (mg/g) | Literature Source | ||
| 1 | EDTA modified bentonite | 0.998 | 3.45 | 160.00 | [43] |
| 2 | Modified Pumice Stone | 0.992 | 0.13 | 15.87 | [44] |
| 3 | Pumice | 0.975 | - | 0.02 | [45] |
| 4 | SAC | 0.991 | 0.14 | 126.60 | [46] |
| 5 | NR90CS10 blend | - | 0.94 | 1.12 | [47] |
| 6 | Natural clay | - | - | 4.98–50.00 | [48] |
| Freundlich Isotherm | |||||
| No. | Sample | R2 | KF(mg/g) | n | Literature Source |
| 1 | EDTA modified bentonite | 0.774 | 84.8 | 3.61 | [43] |
| 2 | Modified Pumice Stone | 0.999 | 3.45 | 2.18 | [44] |
| 3 | Pumice | 0.986 | - | 1.47 | [45] |
| 4 | SAC | 0.987 | 41.7 | 4.76 | [46] |
| 5 | NR90CS10 blend | - | 0.47 | 2.32 | [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galaburda, M.; Bosacka, A.; Sternik, D.; Bogatyrov, V.; Oranska, O.; Gun’ko, V.; Deryło-Marczewska, A. Development, Synthesis and Characterization of Tannin/Bentonite-Derived Biochar for Water and Wastewater Treatment from Methylene Blue. Water 2022, 14, 2407. https://doi.org/10.3390/w14152407
Galaburda M, Bosacka A, Sternik D, Bogatyrov V, Oranska O, Gun’ko V, Deryło-Marczewska A. Development, Synthesis and Characterization of Tannin/Bentonite-Derived Biochar for Water and Wastewater Treatment from Methylene Blue. Water. 2022; 14(15):2407. https://doi.org/10.3390/w14152407
Chicago/Turabian StyleGalaburda, Mariia, Alicja Bosacka, Dariusz Sternik, Viktor Bogatyrov, Olena Oranska, Volodymyr Gun’ko, and Anna Deryło-Marczewska. 2022. "Development, Synthesis and Characterization of Tannin/Bentonite-Derived Biochar for Water and Wastewater Treatment from Methylene Blue" Water 14, no. 15: 2407. https://doi.org/10.3390/w14152407







