Promotion of Growth and Physiological Characteristics in Water-Stressed Triticum aestivum in Relation to Foliar-Application of Salicylic Acid
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Growth Attributes
3.2. Photosynthetic Pigments
3.3. Osmolyte Accumulation
3.4. Oxidative Stress Attributes
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Anjorin:, A.; Leblebici, E.; Schürr, A. 20 years of triple graph grammars: A roadmap for future research. Electron. Commun. EASST 2016, 73. [Google Scholar] [CrossRef]
- Li, P.; Li, Y.-J.; Zhang, F.-J.; Zhang, G.-Z.; Jiang, X.-Y.; Yu, H.-M.; Hou, B.-K. The Arabidopsis UDP-glycosyltransferases UGT79B2 and UGT79B3, contribute to cold, salt and drought stress tolerance via modulating anthocyanin accumulation. Plant J. 2017, 89, 85–103. [Google Scholar] [CrossRef]
- Hussain, H.A.; Men, S.; Hussain, S.; Zhang, Q.; Ashraf, U.; Anjum, S.A.; Ali, I.; Wang, L. Maize Tolerance against Drought and Chilling Stresses Varied with Root Morphology and Antioxidative Defense System. Plants 2020, 9, 720. [Google Scholar] [CrossRef]
- Fathi, A.; Tari, D.B. Effect of drought stress and its mechanism in plants. Int. J. Life Sci. 2016, 10, 1–6. [Google Scholar] [CrossRef]
- Hussain, S.; Hussain, S.; Qadir, T.; Khaliq, A.; Ashraf, U.; Parveen, A.; Saqib, M.; Rafiq, M. Drought stress in plants: An overview on implications, tolerance mechanisms and agronomic mitigation strategies. Plant Sci. Today 2019, 6, 389–402. [Google Scholar] [CrossRef]
- Ahmad, H.T.; Hussain, A.; Aimen, A.; Jamshaid, M.U.; Ditta, A.; Asghar, H.N.; Zahir, Z.A. Improving resilience against drought stress among crop plants through inoculation of plant growth-promoting rhizobacteria. In Harsh Environment and Plant Resilience: Molecular and Functional Aspects, 1st ed.; Husen, A., Jawaid, M., Eds.; Springer: Cham, Switzerland, 2021; Volume 1, pp. 387–408. [Google Scholar] [CrossRef]
- Akram, N.A.; Waseem, M.; Ameen, R.; Ashraf, M. Trehalose pretreatment induces drought tolerance in radish (Raphanus sativus L.) plants: Some key physio-biochemical traits. Acta Physiol. Plant. 2016, 38, 1–10. [Google Scholar] [CrossRef]
- Cao, P.; Lu, C.; Yu, Z. Historical nitrogen fertilizer use in agricultural ecosystems of the contiguous United States during 1850–2015: Application rate, timing, and fertilizer types. Earth Syst. Sci. Data 2018, 10, 969–984. [Google Scholar] [CrossRef]
- Raza, G.; Ali, K.; Ashraf, M.Y.; Mansoor, S.; Javid, M.; Asad, S. Overexpression of an H+−PPase gene from Arabidopsis in sugarcane improvesdrought tolerance, plant growth, and photosynthetic responses. Turk. J. Biol. 2016, 40, 109–119. [Google Scholar] [CrossRef]
- Parveen, A.; Liu, W.; Hussain, S.; Asghar, J.; Perveen, S.; Xiong, Y. Silicon Priming Regulates Morpho-Physiological Growth and Oxidative Metabolism in Maize under Drought Stress. Plants 2019, 8, 431. [Google Scholar] [CrossRef]
- Richards, S.L.; Wilkins, K.A.; Swarbreck, S.M.; Anderson, A.A.; Habib, N.; Smith, A.G.; McAinsh, M.; Davies, J.M. The hydroxyl radical in plants: From seed to seed. J. Exp. Bot. 2015, 66, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Zhou, Y.; Liu, M. Nitric oxide participates in the regulation of the ascorbate-glutathione cycle by exogenous jasmonic acid in the leaves of wheat seedlings under drought stress. Protoplasma 2015, 252, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M. Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol. Adv. 2009, 27, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Habib, N.; Akram, M.S.; Javed, M.T.; Azeem, M.; Ali, Q.; Shaheen, H.L.; Ashraf, M. Nitric Oxide Regulated Improvement in Growth and Yield of Rice Plants Grown Under Salinity Stress: Antioxidant Defense System. Appl. Ecol. Environ. Res. 2016, 14, 91–105. [Google Scholar] [CrossRef]
- Wu, D.; Chu, H.; Jia, L.; Chen, K.; Zhao, L. A feedback inhibition between nitric oxide and hydrogen peroxide in the heat shock pathway in Arabidopsis seedlings. Plant Growth Regul. 2014, 75, 503–509. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Ali, Q.; Ali, S.; Iqbal, N.; Javed, M.T.; Rizwan, M.; Khaliq, R.; Shahid, S.; Perveen, R.; Alamri, S.A.; Alyemeni, M.N.; et al. Alpha-tocopherol fertigation confers growth physio-biochemical and qualitative yield enhancement in field grown water deficit wheat (Triticum aestivum L.). Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Posmyk, M.M.; Bałabusta, M.; Wieczorek, M.; Sliwinska, E.; Janas, K.M. Melatonin applied to cucumber (Cucumis sativus L.) seeds improves germination during chilling stress. J. Pineal Res. 2009, 46, 214–223. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Rasheed, R.; Hussain, I.; Iqbal, M.; Haider, M.Z.; Parveen, S.; Sajid, M.A. Hydrogen peroxide modulates antioxidant system and nutrient relation in maize (Zea mays L.) under water-deficit conditions. Arch. Agron. Soil Sci. 2015, 61, 507–523. [Google Scholar] [CrossRef]
- Ali, Q.; Daud, M.; Haider, M.Z.; Ali, S.; Rizwan, M.; Aslam, N.; Noman, A.; Iqbal, N.; Shahzad, F.; Deeba, F.; et al. Seed priming by sodium nitroprusside improves salt tolerance in wheat (Triticum aestivum L.) by enhancing physiological and biochemical parameters. Plant Physiol. Biochem. 2017, 119, 50–58. [Google Scholar] [CrossRef]
- Caverzan, A.; Casassola, A.; Brammer, S.P. Antioxidant responses of wheat plants under stress. Genet. Mol. Biol. 2016, 39, 1–6. [Google Scholar] [CrossRef]
- Din, J.; Khan, S.U.; Ali, I.; Gurmani, A.R. Physiological and agronomic response of canola varieties to drought stress. J. Anim. Plant Sci. 2011, 21, 78–82. [Google Scholar]
- Shahzad, H.; Ullah, S.; Iqbal, M.; Bilal, H.M.; Shah, G.M.; Ahmad, S.; Zakir, A.; Ditta, A.; Farooqi, M.A.; Ahmad, I. Salinity types and level-based effects on the growth, physiology and nutrient contents of maize (Zea mays). Ital. J. Agron. 2019, 14, 199–207. [Google Scholar] [CrossRef]
- Shafiq, S.; Akram, N.A.; Ashraf, M.; Arshad, A. Synergistic effects of drought and ascorbic acid on growth, mineral nutrients and oxidative defense system in canola (Brassica napus L.) plants. Acta Physiol. Plant. 2014, 36, 1539–1553. [Google Scholar] [CrossRef]
- Mafakheri, A.; Siosemardeh, A.; Bahramnejad, B.; Struik, P.C.; Sohrabi, Y. Effect of drought stress on yield, proline and chlorophyll contents in three chickpea cultivars. Aust. J. Crop Sci. 2010, 4, 580–585. [Google Scholar]
- Gou, W.; Tian, L.; Ruan, Z.; Zheng, P.; Chen, F.; Zhang, L.; Cui, Z.; Zheng, P.; Li, Z.; Gao, M.; et al. Accumulation of choline and glycinebetaine and drought stress tolerance induced in maize (Zea mays) by three plant growth promoting rhizobacteria (PGPR) strains. Pak. J. Bot. 2015, 47, 581–586. [Google Scholar]
- Ali, Q.; Anwar, F.; Ashraf, M.; Saari, N.; Perveen, R. Ameliorating Effects of Exogenously Applied Proline on Seed Composition, Seed Oil Quality and Oil Antioxidant Activity of Maize (Zea mays L.) under Drought Stress. Int. J. Mol. Sci. 2013, 14, 818–835. [Google Scholar] [CrossRef]
- Noman, A.; Ali, Q.; Maqsood, J.; Iqbal, N.; Javed, M.T.; Rasool, N.; Naseem, J. Deciphering physio-biochemical, yield, and nutritional quality attributes of water-stressed radish (Raphanus sativus L.) plants grown from Zn-Lys primed seeds. Chemosphere 2018, 195, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Hatzig, S.; Zaharia, L.I.; Abrams, S.; Hohmann, M.; Legoahec, L.; Bouchereau, A.; Nesi, N.; Snowdon, R.J. Early Osmotic Adjustment Responses in Drought-Resistant and Drought-Sensitive Oilseed Rape. J. Integr. Plant Biol. 2014, 56, 797–809. [Google Scholar] [CrossRef]
- Amin, A.A.; Rashad, E.-S.M.; Gharib, F.A. Changes in morphological, physiological and reproductive characters of wheat plants as affected by foliar application with salicylic acid and ascorbic acid. Aust. J. Basic Appl. Sci. 2008, 2, 252–261. [Google Scholar]
- Khan, W.; Prithiviraj, B.; Smith, D.L. Photosynthetic responses of corn and soybean to foliar application of salic-ylates. J. Plant Physiol. 2003, 160, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Shakirova, F.M.; Sakhabutdinova, A.R.; Bezrukova, M.V.A.; Fatkhutdinova, R.; Fatkhutdinova, D.R. Changes in the hormonal status of wheat seedlings induced by salicylic acid and salinity. Plant Sci. 2003, 164, 317–322. [Google Scholar] [CrossRef]
- Alvarez, M.E. Salicylic acid in the machinery of hypersensitive cell death and disease resistance. In Programmed Cell Death in Higher Plants; Lam, E., Fukuda, H., Greenberg, J., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 185–198. [Google Scholar]
- Singh, B.; Usha, K. Salicylic acid induced physiological and biochemical changes in wheat seedlings under water stress. Plant Growth Regul. 2003, 39, 137–141. [Google Scholar] [CrossRef]
- Metwally, A.; Finkemeier, I.; Georgi, M.; Dietz, K.-J. Salicylic Acid Alleviates the Cadmium Toxicity in Barley Seedlings. Plant Physiol. 2003, 132, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Borsani, O.; Valpuesta, V.; Botella, M.A. Evidence for a Role of Salicylic Acid in the Oxidative Damage Generated by NaCl and Osmotic Stress in Arabidopsis Seedlings. Plant Physiol. 2001, 126, 1024–1030. [Google Scholar] [CrossRef] [PubMed]
- Mazen, A. Accumulation of Four Metals in Tissues of Corchorus olitorius and Possible Mechanisms of Their Tolerance. Biol. Plant. 2004, 48, 267–272. [Google Scholar] [CrossRef]
- Costa, R.C.L.; Lobato, A.K.S.; Silveira, J.A.G.; Laughinghouse, I.V.H.D. ABA-mediated proline synthesis in cowpea leaves exposed to water deficiency and rehydration. Turk. J. Agric. For. 2011, 35, 309–317. [Google Scholar]
- Arnon, D. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Cakmak, I.; Horst, W.J. Effect of aluminium on lipid peroxidation, superoxide dismutase, catalase, and peroxidase activities in root tips of soybean (Glycine max). Physiol. Plant. 1991, 83, 463–468. [Google Scholar] [CrossRef]
- Velikova, V.; Yordanov, I.; Edreva, A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants: Protective role of exogenous polyamines. Plant Sci. 2000, 151, 59–66. [Google Scholar] [CrossRef]
- Yang, G.; Rhodes, D.; Joly, R. Effects of High Temperature on Membrane Stability and Chlorophyll Fluorescence in Glycinebetaine-Deficient and Glycinebetaine-Containing Maize Lines. Funct. Plant Biol. 1996, 23, 437–443. [Google Scholar] [CrossRef]
- Julkunen-Tiitto, R. Phenolic constituents in the leaves of northern willows: Methods for the analysis of certain phenolics. J. Agric. Food Chem. 1985, 33, 213–217. [Google Scholar] [CrossRef]
- Mukherjee, S.P.; Choudhuri, M.A. Implications of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol. Plant. 1983, 58, 166–170. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef]
- Chance, B.; Maehly, A. (136) Assay of catalases and peroxidases. Methods Enzym. 1955, 2, 764–775. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef]
- Abid, M.; Ali, S.; Qi, L.K.; Zahoor, R.; Tian, Z.; Jiang, D.; Snider, J.L.; Dai, T. Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Skirycz, A.; Inzé, D. More from less: Plant growth under limited water. Curr. Opin. Biotechnol. 2010, 21, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Bramley, H.; Turner, N.C.; Turner, D.W.; Tyerman, S.D. Roles of Morphology, Anatomy, and Aquaporins in Determining Contrasting Hydraulic Behavior of Roots. Plant Physiol. 2009, 150, 348–364. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cang, Z.; Jiao, F.; Bai, X.; Zhang, D.; Zhai, R. Influence of drought stress on photosynthetic characteristics and protective enzymes of potato at seedling stage. J. Saudi Soc. Agric. Sci. 2017, 16, 82–88. [Google Scholar] [CrossRef]
- El-Bahay, M.M. Metabolic changes, Phytohormonal Level, and activities of certain related enzymes associated with growth of presoaked Lupine seeds in salicylic and gallic acids. Bull. Fac. Sci. Assiut. Univ. 2002, 31, 259–270. [Google Scholar]
- Shehata, S.; Ibrahim, S.; Zaghlool, S. Physiological response of flag leaf and ears of maize plant induced by foliar application of kinetin, KIN and acetyl salicylic acid, ASA. Ann. Agric. Sci. 2001, 46, 435–449. [Google Scholar]
- Iqbal, M.; Ashraf, M. Wheat seed priming in relation to salt tolerance: Growth, yield and levels of free salicylic acid and polyamines. Ann. Bot. Fenn. 2006, 43, 250–259. [Google Scholar]
- Afzal, I.; Basara, S.M.A.; Faooq, M.; Nawaz, A. Alleviation of salinity stress in spring wheat by hormonal priming with ABA, salicylic acid and ascorbic acid. Int. J. Agric. Biol. 2006, 8, 23–28. [Google Scholar]
- Idrees, M.; Khan, M.M.A.; Aftab, T.; Naeem, M.; Hashmi, N. Salicylic acid-induced physiological and biochemical changes in lemongrass varieties under water stress. J. Plant Interact. 2010, 5, 293–303. [Google Scholar] [CrossRef]
- Ashraf, M.; Harris, P.J.C. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Amirjani, M.R.; Mahdiyeh, M. Antioxidative and biochemical responses of wheat to drought stress. J. Agric. Biol. Sci. 2013, 8, 291–301. [Google Scholar]
- Habib, N.; Ali, Q.; Ali, S.; Javed, M.T.; Haider, M.Z.; Perveen, R.; Shahid, M.R.; Rizwan, M.; Abdel-Daim, M.M.; Elkelish, A.; et al. Use of Nitric Oxide and Hydrogen Peroxide for Better Yield of Wheat (Triticum aestivum L.) under Water Deficit Conditions: Growth, Osmoregulation, and Antioxidative Defense Mechanism. Plants 2020, 9, 285. [Google Scholar] [CrossRef]
- Naz, H.; Akram, N.A.; Ashraf, M. Impact of ascorbic acid on growth and some physiological attributes of cucumber (Cucumis sativus) plants under water-deficit conditions. Pak. J. Bot. 2016, 48, 877–883. [Google Scholar]
- Akram, N.A.; Iqbal, M.; Muhammad, A.; Ashraf, M.; Al-Qurainy, F.; Shafiq, S. Aminolevulinic acid and nitric oxide regulate oxidative defense and secondary metabolisms in canola (Brassica napus L.) under drought stress. Protoplasma 2018, 255, 163–174. [Google Scholar] [CrossRef]
- Razzaq, H.; Tahir, M.H.N.; Sadaqat, H.A.; Sadia, B. Screening of sunflower (Helianthus annus L.) accessions under drought stress conditions, an experimental assay. J. Soil Sci. Plant Nutr. 2017, 17, 662–671. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Y. Activation of salicylic acid–induced protein kinase, a mitogen-activated protein kinase, induces multiple defense responses in tobacco. Plant Cell 2001, 13, 1877–1889. [Google Scholar]
- El-Tayeb, M.S. Response of barley grains to the interactive e. ect of salinity and salicylic acid. Plant Growth Regul. 2005, 45, 215–224. [Google Scholar] [CrossRef]
- Idrees, M.; Naeem, M.; Aftab, T.; Khan, M.M.A.; Moinuddin. Salicylic acid mitigates salinity stress by improving antioxidant defence system and enhances vincristine and vinblastine alkaloids production in periwinkle [Catharanthus roseus (L.) G. Don]. Acta Physiol. Plant. 2011, 33, 987–999. [Google Scholar] [CrossRef]
- Habib, N.; Ashraf, M.; Ali, Q.; Perveen, R. Response of salt stressed okra (Abelmoschus esculentus Moench) plants to foliar-applied glycine betaine and glycine betaine containing sugarbeet extract. S. Afr. J. Bot. 2012, 83, 151–158. [Google Scholar] [CrossRef]
- Ahmad, M.S.A.; Javed, F.; Ashraf, M. Iso-osmotic effect of NaCl and PEG on growth, cations and free proline accumulation in callus tissue of two indica rice (Oryza sativa L.) genotypes. Plant Growth Regul. 2007, 53, 53–63. [Google Scholar] [CrossRef]
- Damghan, I.R. Exogenous application of brassinosteroid alleviates drought-induced oxidative stress in Lycopersicon esculentum L. Gen. Appl. Plant Physiol. 2009, 35, 22–34. [Google Scholar]
- Zhang, J.; Jia, W.; Yang, J.; Ismail, A.M. Role of ABA in integrating plant responses to drought and salt stresses. Field Crop. Res. 2006, 97, 111–119. [Google Scholar] [CrossRef]
- Umebese, C.; Olatimilehin, T.; Ogunsusi, T. Salicylic Acid Protects Nitrate Reductase Activity, Growth and Proline in Ama-Ranth and Tomato Plants during Water Deficit. Am. J. Agric. Biol. Sci. 2009, 4, 224–229. [Google Scholar] [CrossRef][Green Version]
- Amin, A.A.; Rashad, M.; El-Abagy, H.M.H. Physiological effect of indole-3-butyric acid and salicylic acid on growth, yield and chemical constituents of onion plants. J. Appl. Sci. Res. 2007, 3, 1554–1563. [Google Scholar]
- Bakry, B.A.; El-Hariri, D.M.; Sadak, M.S.; El-Bassiouny, H.M.S. Drought stress mitigation by foliar application of salicylic acid in two linseed varieties grown under newly reclaimed sandy soil. J. Appl. Sci. Res. 2012, 8, 3503–3514. [Google Scholar]
- El Tayeb, M.A.; Ahmed, N.L. Response of wheat cultivars to drought and salicylic acid. American-Eurasian J. Agron. 2010, 3, 1–7. [Google Scholar]
- Ali, Q.; Javed, M.T.; Noman, A.; Haider, M.Z.; Waseem, M.; Iqbal, N.; Waseem, M.; Shah, M.S.; Shahzad, F.; Perveen, R. Assessment of drought tolerance in mung bean cultivars/lines as depicted by the activities of germination enzymes, seedling’s antioxidative potential and nutrient acquisition. Arch. Agron. Soil Sci. 2018, 64, 84–102. [Google Scholar] [CrossRef]
- Noctor, G.; Foyer, C.H. Ascorbate and glutathione: Keeping active oxygen under control. Ann. Rev. Plant Biol. 1998, 49, 249–279. [Google Scholar] [CrossRef]
- Hamurcu, M.; Demiral, T.; Calik, M.; Avsaroglu, Z.Z.; Celik, O.; Hakki, E.E.; Topal, A.; Gezgin, S.; Bell, R.W. Effect of nitric oxide on the tolerance mechanism of bread wheat genotypes under drought stress. J. Biotechnol. 2014, 185, S33. [Google Scholar] [CrossRef]
- Li, D.-M.; Zhang, J.; Sun, W.-J.; Li, Q.; Dai, A.-H.; Bai, J.-G. 5-Aminolevulinic acid pretreatment mitigates drought stress of cucumber leaves through altering antioxidant enzyme activity. Sci. Hortic. 2011, 130, 820–828. [Google Scholar] [CrossRef]
- De Carvalho, M.H.C.; Brunet, J.; Bazin, J.; Kranner, I.; d’Arcy-Lameta, A.; Zuily-Fodil, Y.; Contour-Ansel, D. Homoglutathione synthetase and glutathione synthetase in drought-stressed cowpea leaves: Ex-pression patterns and accumulation of low-molecular-weight thiols. J. Plant Physiol. 2010, 167, 480–487. [Google Scholar] [CrossRef]
- Noreen, Z.; Ashraf, M. Assessment of variation in antioxidative defense system in salt-treated pea (Pisum sativum) cultivars and its putative use as salinity tolerance markers. J. Plant Physiol. 2009, 166, 1764–1774. [Google Scholar] [CrossRef]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Scandalios, J.G. Oxidative Stress and the Molecular Biology of Antioxidant Defenses; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1997. [Google Scholar]
- Smirnoff, N. Tansley Review No. 52. The role of active oxygen in the response of plants to water deficit and desiccation. New Phytol. 1993, 125, 27–58. [Google Scholar] [CrossRef]
- Bowler, C.; Montagu, M.V.; Inze, D. Superoxide dismutase and stress tolerance. Annu. Rev. Plant Biol. 1992, 43, 83–116. [Google Scholar] [CrossRef]
- Manivannan, P.; Jaleel, C.A.; Kishorekumar, A.; Sankar, B.; Somasundaram, R.; Sridharan, R.; Panneerselvam, R. Changes in antioxidant metabolism of Vigna unguiculata (L.) Walp. by propiconazole under water deficit stress. Coll. Surfaces B Biointerfaces 2007, 57, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Noctor, G.; Veljovic-Jovanovic, S.; Foyer, C.H. Peroxide processing in photosynthesis: Antioxidant coupling and redox signalling. Philos. Trans. R. Soc. B Biol. Sci. 2000, 355, 1465–1475. [Google Scholar] [CrossRef] [PubMed]
- Fornazier, R.F.; Ferreira, R.R.; Pereira, G.J.G.; Molina, S.M.G.; Smith, R.J.; Lea, P.J.; Azevedo, R.A. Cadmium stress in sugar cane callus cultures: Effect on antioxidant enzymes. Plant Cell Tissue Organ Cult. 2002, 71, 125–131. [Google Scholar] [CrossRef]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef] [PubMed]
- Kerdnaimongkol, K.; Bhatia, A.; Joly, R.J.; Woodson, W.R. Oxidative Stress and Diurnal Variation in Chilling Sensitivity of Tomato Seedlings. J. Am. Soc. Hortic. Sci. 1997, 122, 485–490. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Gopi, R.; Manivannan, P.; Panneerselvam, R. Antioxidative potentials as a protective mechanism in Catharanthus roseus (L.) G. Don. plants under salinity stress. Turk. J. Bot. 2007, 31, 245–251. [Google Scholar]
- Cirulis, J.T.; Scott, J.A.; Ross, G.M. Management of oxidative stress by microalgae. Can. J. Physiol. Pharmacol. 2013, 91, 15–21. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Li, J.-L.; Ma, X.-R. Exogenous hydrogen peroxide enhanced the thermotolerance of Festuca arundinacea and Lolium perenne by increasing the antioxidative capacity. Acta Physiol. Plant. 2014, 36, 2915–2924. [Google Scholar] [CrossRef]
- Lima, A.L.S.; DaMatta, F.M.; Pinheiro, H.A.; Totola, M.R.; Loureiro, M.E. Photochemical responses and oxidative stress in two clones of Coffea canephora under water deficit conditions. Environ. Exp. Bot. 2002, 47, 239–247. [Google Scholar] [CrossRef]
- Yadav, N.; Sharma, S.; Samir, P. Research, Reactive oxygen species, oxidative stress and ROS scavenging system in plants. J. Chem. Pharm. Res. 2016, 8, 595–604. [Google Scholar]
- Shafiq, S.; Akram, N.A.; Ashraf, M. Does exogenously-applied trehalose alter oxidative defense system in the edible part of radish (Raphanus sativus L.) under water-deficit conditions? Sci. Hortic. 2015, 185, 68–75. [Google Scholar] [CrossRef]
- Després, C.; Fobert, P.R. In Vivo biochemical characterization of transcription factors regulating plant defense response to disease. Can. J. Plant Pathol. 2006, 28, 3–15. [Google Scholar] [CrossRef]
- Aftab, T.; Khan, M.M.A.; Da Silva, J.A.T.; Idrees, M.; Naeem, M.; Moinuddin. Role of Salicylic Acid in Promoting Salt Stress Tolerance and Enhanced Artemisinin Production in Artemisia annua L. J. Plant Growth Regul. 2011, 30, 425–435. [Google Scholar] [CrossRef]
- Khan, M.N.; Khan, Z.; Luo, T.; Liu, J.; Rizwan, M.; Zhang, J.; Xu, Z.; Wu, H.; Hu, L. Seed priming with gibberellic acid and melatonin in rapeseed: Consequences for improving yield and seed quality under drought and non-stress conditions. Ind. Crop. Prod. 2020, 156, 112850. [Google Scholar] [CrossRef]
- Matysiak, K.; Siatkowski, I.; Kierzek, R.; Kowalska, J.; Krawczyk, R. Effect of foliar applied acetyl salicilic acid on wheat (Triticum aestivum L.) under field conditions. Agronomy 2020, 10, 1918. [Google Scholar] [CrossRef]
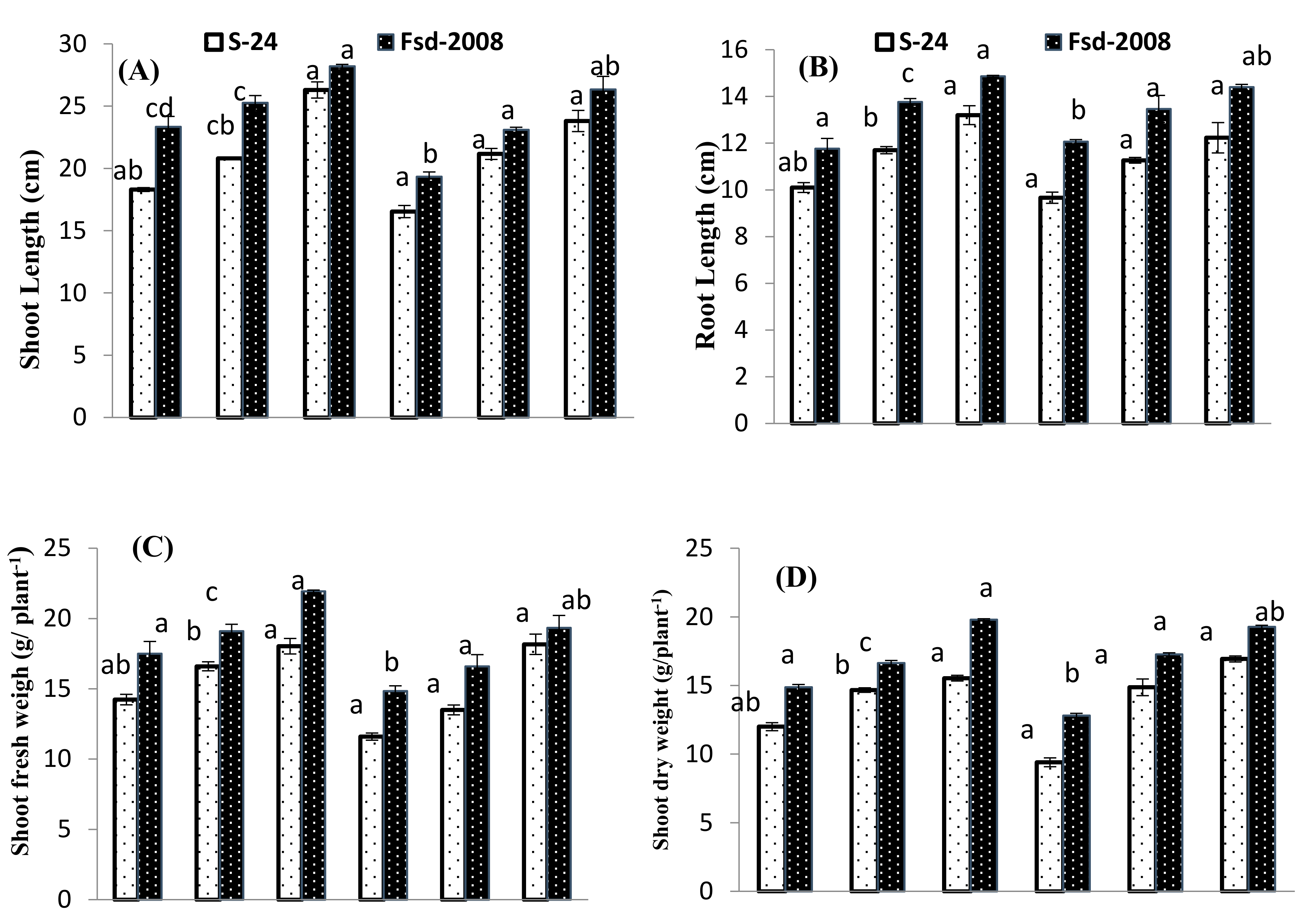


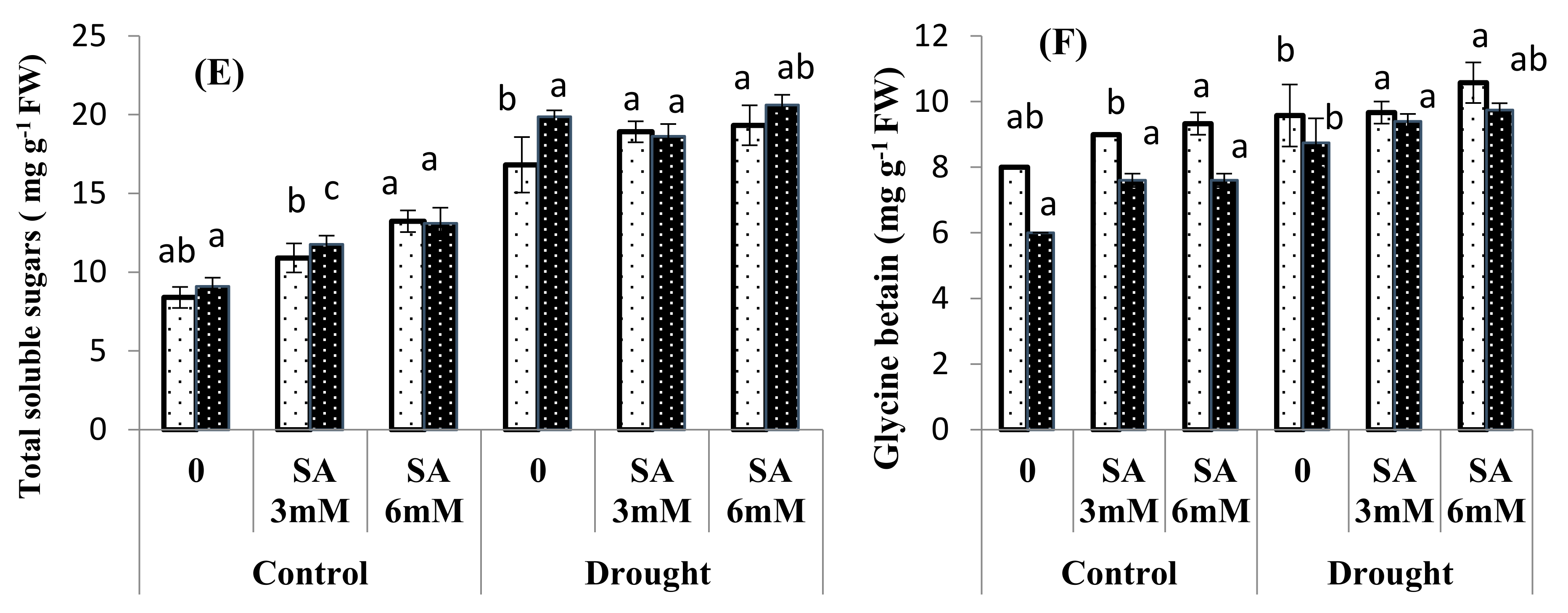
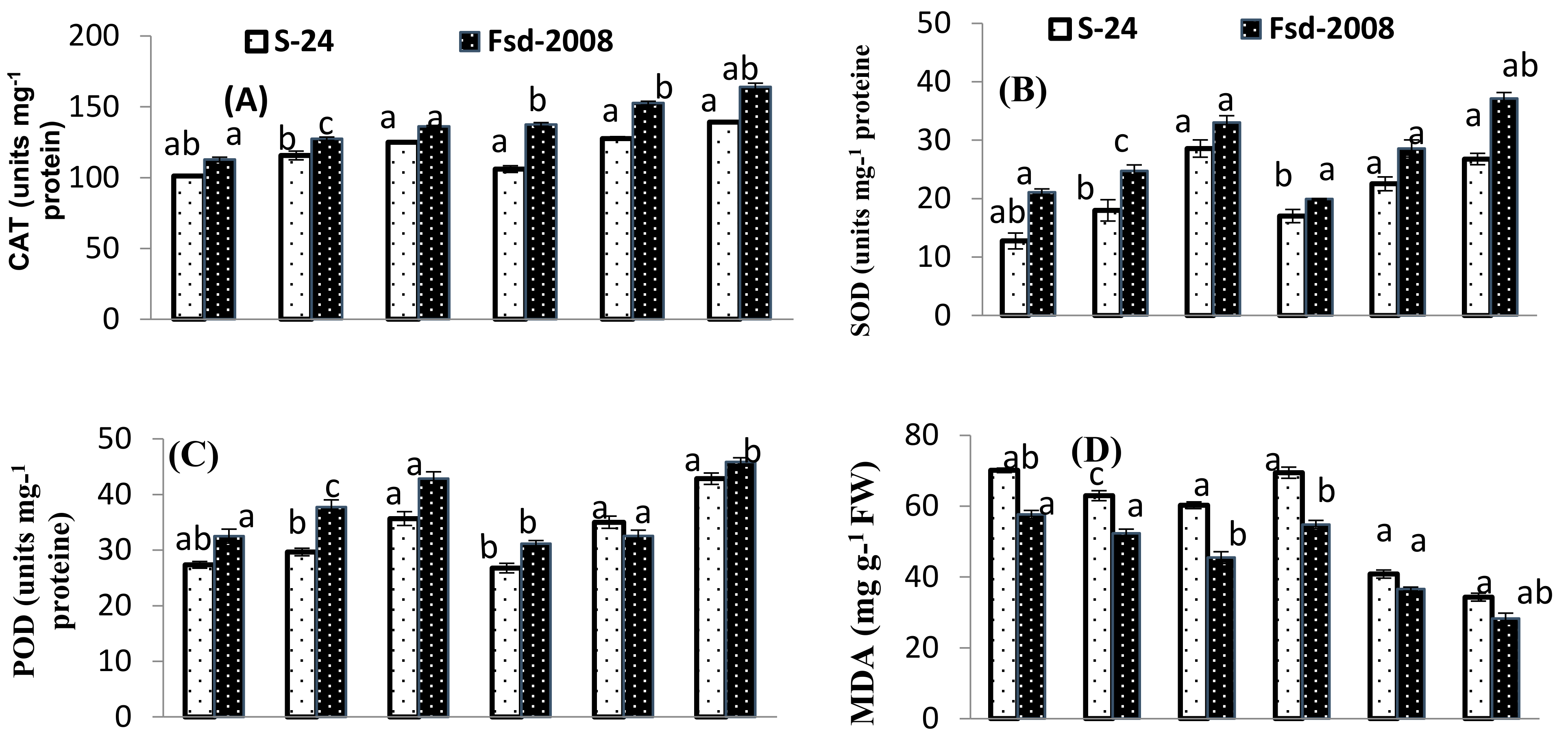
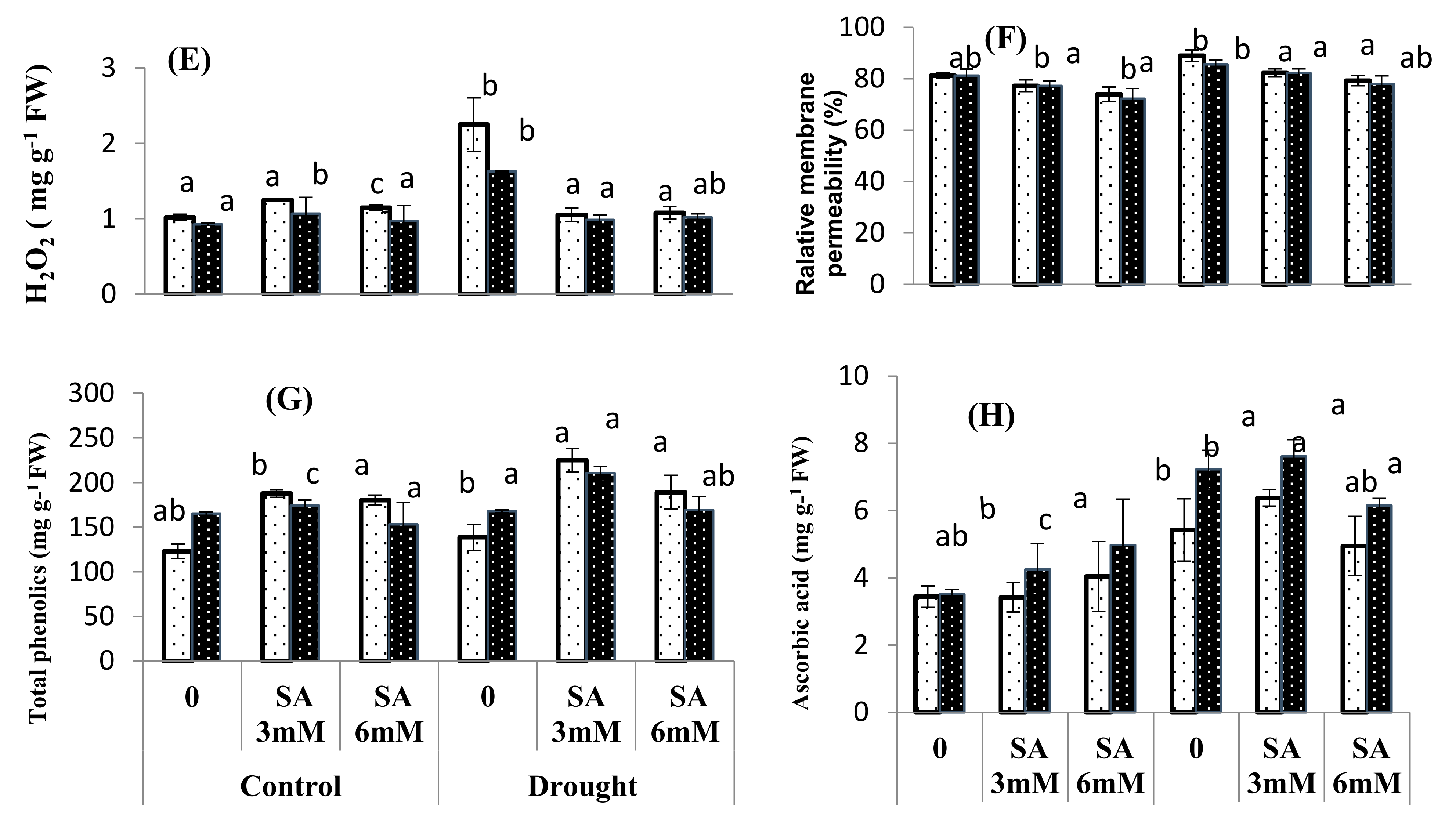
| Source of Variance | df | SL | RL | SFW | SDW | RFW | RDW | Chl. a | Chl. b | Carotenoids | MDA |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Varieties (V) | 1 | 87.11 *** | 37.01 *** | 73.67 *** | 102.35 *** | 9.45 *** | 3.17 *** | 7.40 *** | 41.14 *** | 0.08 *** | 983.28 *** |
| Drought Stress (S) | 1 | 35.60 *** | 22.2 *** | 44.67 *** | 8.900 *** | 0.51 *** | 2..87 *** | 13.04 *** | 122.57 *** | 0.04 *** | 1787.99 *** |
| Treatments (T) | 2 | 138.18 *** | 23.38 *** | 70.86 *** | 126.97 *** | 2.26 *** | 1.48 *** | 13.49 *** | 28.89 *** | 0.08 *** | 1392.31 *** |
| V × S | 1 | 4.27 * | 0.47ns | 1.17ns | 1.03 * | 0.07ns | 0.27 * | 0.38 * | 5.26 *** | 0.02 * | 42.09 ** |
| V × T | 2 | 2.19ns | 0.4ns | 0.39ns | 4.52 *** | 0.03ns | 0.01ns | 0.03ns | 2.49 *** | 7.83ns | 28.08 ** |
| S ×T | 2 | 3.03ns | 0.32ns | 2.24ns | 18.13 *** | 0.03ns | 0.04ns | 0.22ns | 0.03ns | 0.001 ** | 347.64 *** |
| V × S × T | 2 | 2.29ns | 0.07ns | 2.35ns | 5.64 *** | 0.01ns | 0.24ns | 0.01ns | 0.22 * | 8.13ns | 24.72 * |
| Error | 24 | 1.00 | 0.32 | 0.97 | 0.20 | 0.03 | 0.04 | 0.07 | 0.04 | 2.97 | 4.45 |
| Source of Variance | df | H2O2 | RMP | Phenolics | Ascorbic Acid | Glycine betaine | TSS | Proline | CAT | SOD | POD |
| Varieties (V) | 1 | 0.36 ** | 157.85ns | 274.09ns | 6.65 ** | 82.75 *** | 86.89 *** | 4.59 *** | 3374.62 *** | 376.62 *** | 160.94 *** |
| Drought Stress (S) | 1 | 1.33 *** | 157.85ns | 4042.84 ** | 75.23 *** | 182.61 *** | 393.56 *** | 10.88 *** | 2966.28 *** | 47.60 *** | 5.84 ** |
| Treatments (T) | 2 | 0.77 *** | 290.40 ** | 7836.06 ** | 2.59 ** | 86.32 *** | 122.65 *** | 38.28 *** | 2180.47 *** | 565.74 *** | 155.29 *** |
| V × S | 1 | 0.02ns | 1.17ns | 25.56ns | 0.93ns | 2.65ns | 10.79 * | 0.002ns | 547.68 *** | 0.01ns | 19.70 *** |
| V × T | 2 | 0.26 ** | 0.94ns | 4293.44 *** | 0.24ns | 1.11ns | 3.19ns | 1.56 *** | 11.12ns | 2.41ns | 1.52ns |
| S ×T | 2 | 1.26 *** | 0.94ns | 613.61ns | 0.01ns | 3.25ns | 3.50ns | 0.21 ** | 29.92 * | 8.12ns | 10.33 *** |
| V × S × T | 2 | 0.35 *** | 0.49ns | 57.12ns | 0.80ns | 0.66ns | 1.31ns | 2.87 *** | 10.07ns | 24.37 ** | 18.51 ** |
| Error | 24 | 0.03 | 588.81 | 434.98ns | 0.83 | 2.76 | 1.48 | 0.04 | 8.57 | 4.24 | 3.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parveen, A.; Arslan Ashraf, M.; Hussain, I.; Perveen, S.; Rasheed, R.; Mahmood, Q.; Hussain, S.; Ditta, A.; Hashem, A.; Al-Arjani, A.-B.F.; et al. Promotion of Growth and Physiological Characteristics in Water-Stressed Triticum aestivum in Relation to Foliar-Application of Salicylic Acid. Water 2021, 13, 1316. https://doi.org/10.3390/w13091316
Parveen A, Arslan Ashraf M, Hussain I, Perveen S, Rasheed R, Mahmood Q, Hussain S, Ditta A, Hashem A, Al-Arjani A-BF, et al. Promotion of Growth and Physiological Characteristics in Water-Stressed Triticum aestivum in Relation to Foliar-Application of Salicylic Acid. Water. 2021; 13(9):1316. https://doi.org/10.3390/w13091316
Chicago/Turabian StyleParveen, Abida, Muhammad Arslan Ashraf, Iqbal Hussain, Shagufta Perveen, Rizwan Rasheed, Qaisar Mahmood, Shahid Hussain, Allah Ditta, Abeer Hashem, Al-Bandari Fahad Al-Arjani, and et al. 2021. "Promotion of Growth and Physiological Characteristics in Water-Stressed Triticum aestivum in Relation to Foliar-Application of Salicylic Acid" Water 13, no. 9: 1316. https://doi.org/10.3390/w13091316
APA StyleParveen, A., Arslan Ashraf, M., Hussain, I., Perveen, S., Rasheed, R., Mahmood, Q., Hussain, S., Ditta, A., Hashem, A., Al-Arjani, A.-B. F., Alqarawi, A. A., & Abd Allah, E. F. (2021). Promotion of Growth and Physiological Characteristics in Water-Stressed Triticum aestivum in Relation to Foliar-Application of Salicylic Acid. Water, 13(9), 1316. https://doi.org/10.3390/w13091316










