Fishways as Downstream Routes in Small Hydropower Plants: Experiences with a Potamodromous Cyprinid
Abstract
1. Introduction
2. Materials and Methods
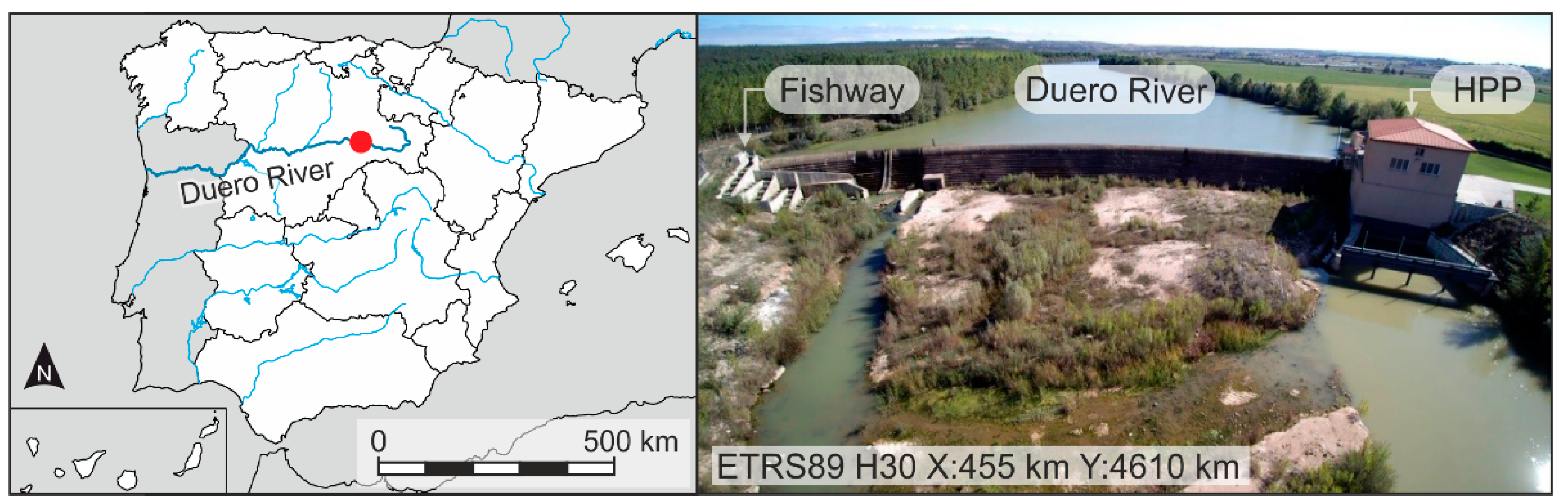
2.1. Study Site
2.2. Fish Collection and Tagging
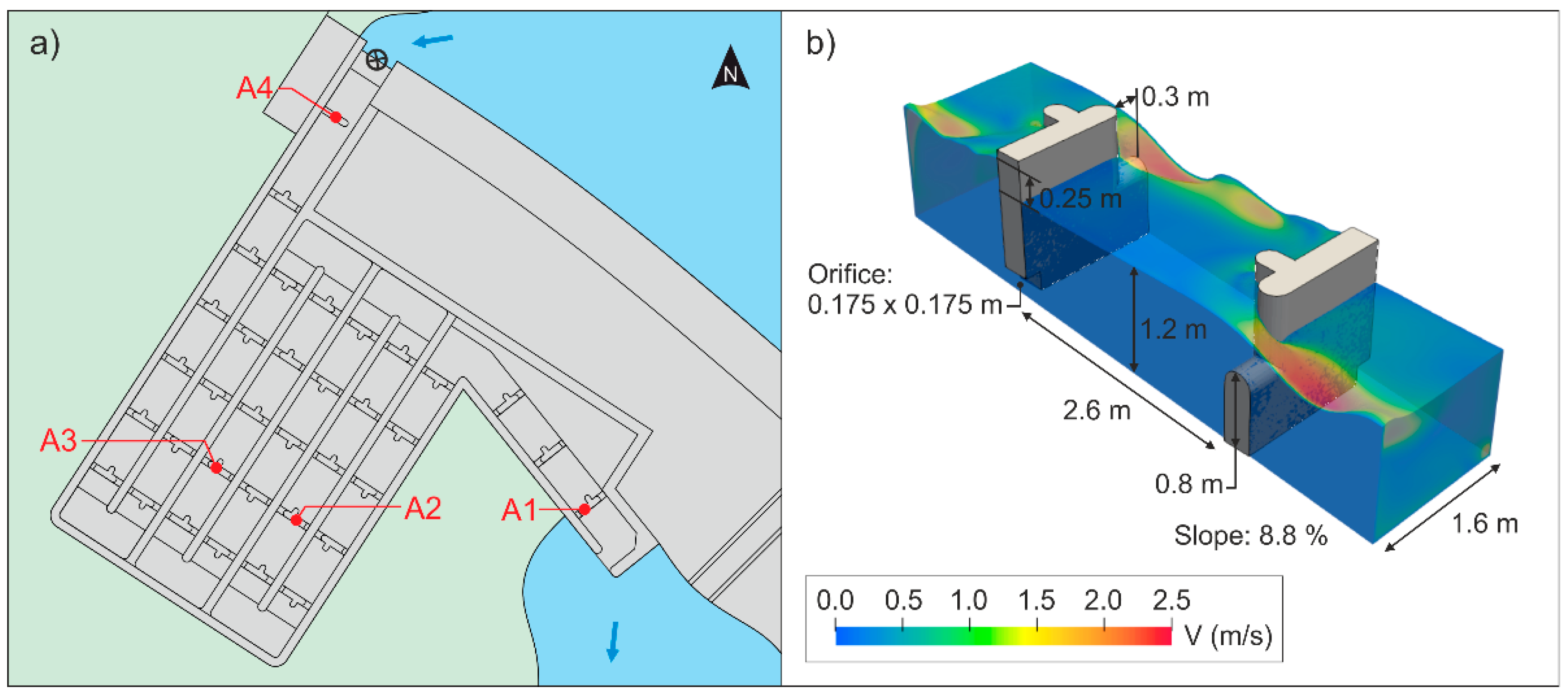
2.3. Data Collection
- (1)
- For each fish, only records of the same year of the release date were considered (as the experiments lasted two years, recaptures in different years were possible).
- (2)
- For each fish, a fishway location occurred if the fish was recorded first, and at least once in the uppermost antenna (antenna 4). If there was more than one location registered by fish, only the earliest one was considered.
- (3)
- Location time of the fishway was defined as the time between the release and the first detection in the uppermost antenna (antenna 4).
- (4)
- For each fish, a fishway entry occurred if the fish was recorded first in the uppermost antenna (antenna 4) and then in antenna 3 or 2. If there was more than one entry by fish, only the first one was considered.
- (5)
- A success in the fishway downstream passage was defined for fish with a first record in antenna 4, a last record in antenna 1, and at least one intermediate record in antennas 3 and/or 2.
- (6)
- The downstream transit time was only calculated for the fish with a successful downstream passage. If there were more than one success by fish, the one with the lowest downstream transit time was considered.
- (7)
- Downstream transit time was defined as the time between the last detection in the uppermost antenna (antenna 4) and the first detection in the lowermost antenna (antenna 1).
- (8)
- As complementary information, downstream passage through turbines or spillways was assigned to the fish with (1) first records in the lowermost antenna (i.e., they passed downstream through the turbines or spillways, and they came back to the fishway for upstream migration), or (2) first records in the uppermost antenna and subsequent records in lowermost antenna with an elapsed time between them of at least one day (i.e., fish tried to enter the fishway but exited, then passed through turbines or spillways, and then, came back to the fishway for upstream migration).
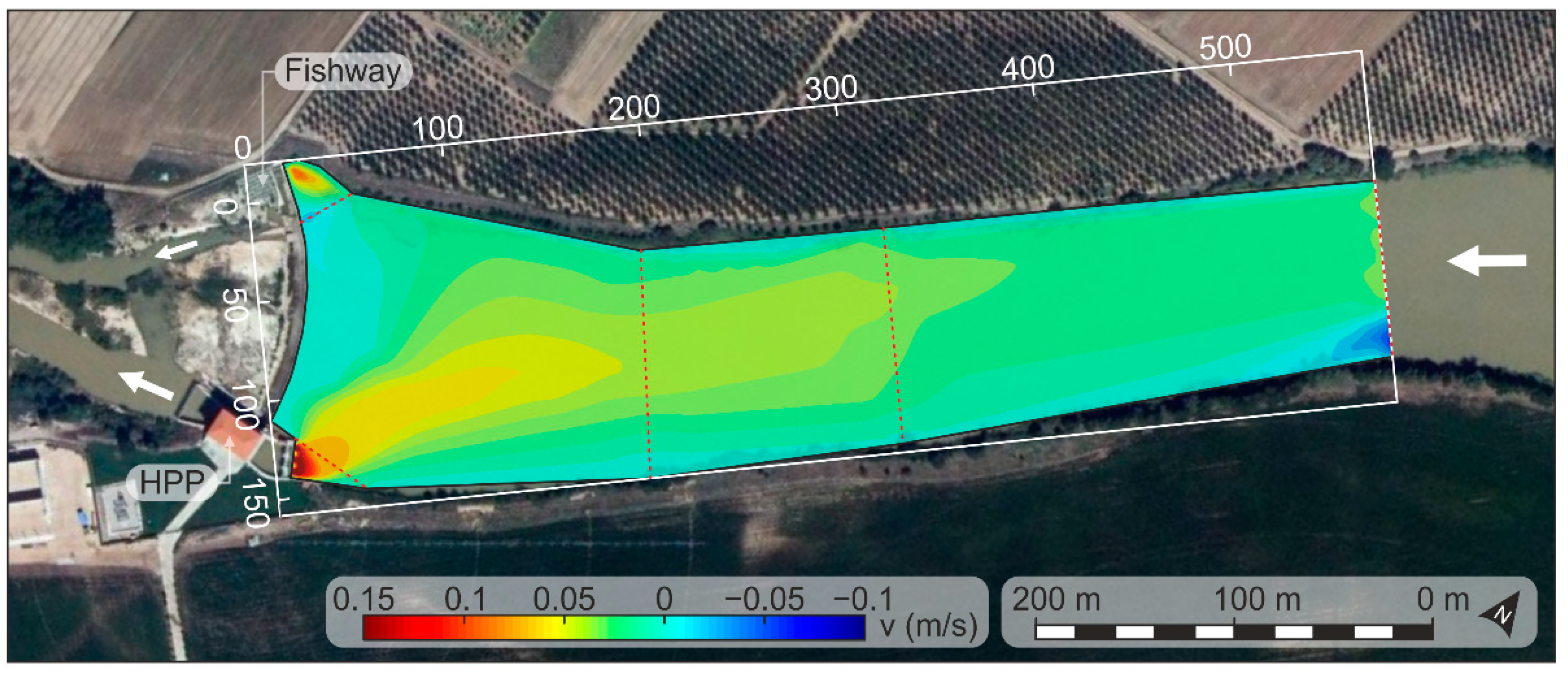
2.4. Hydraulic Characterization
2.5. Data Analysis
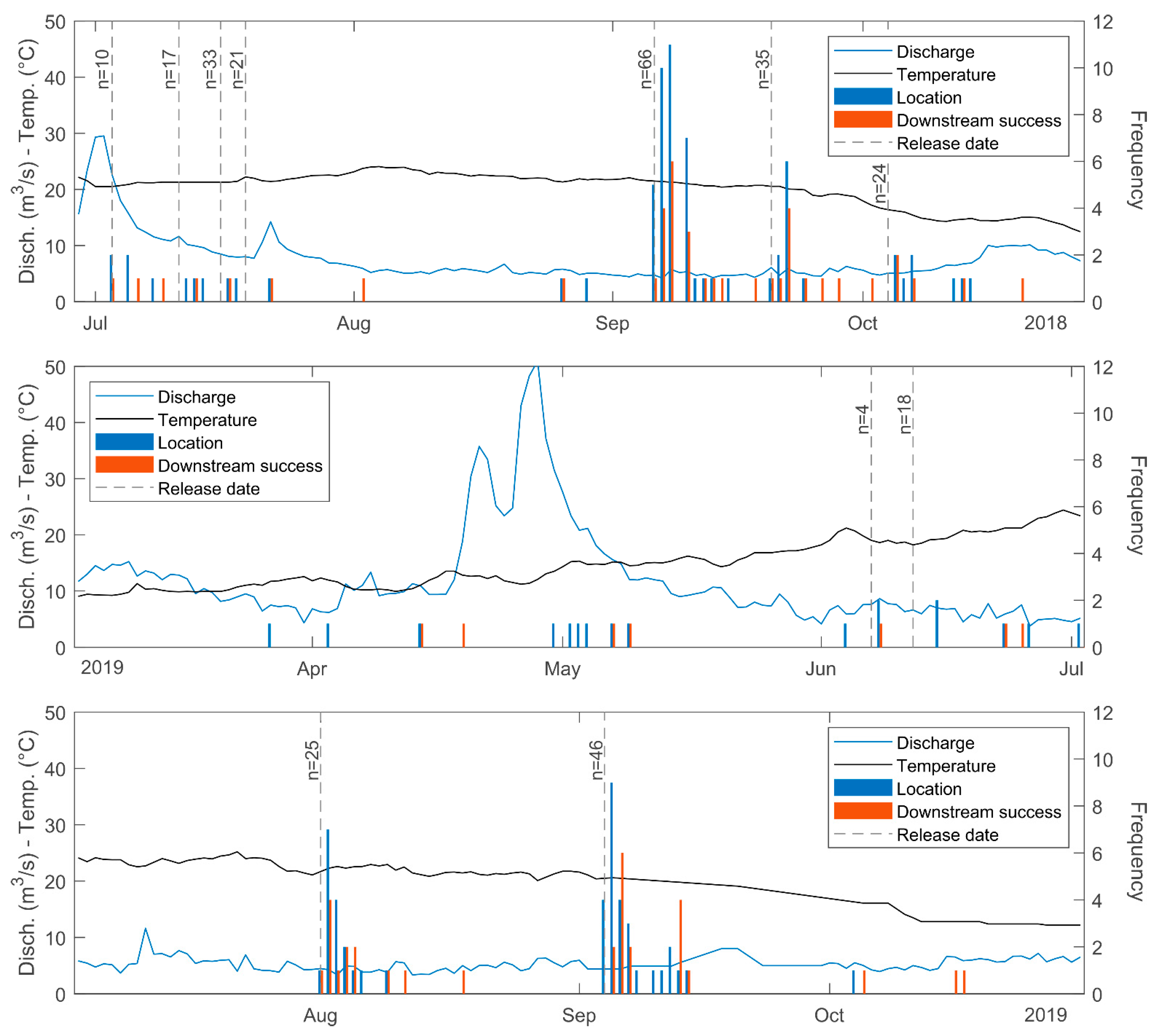
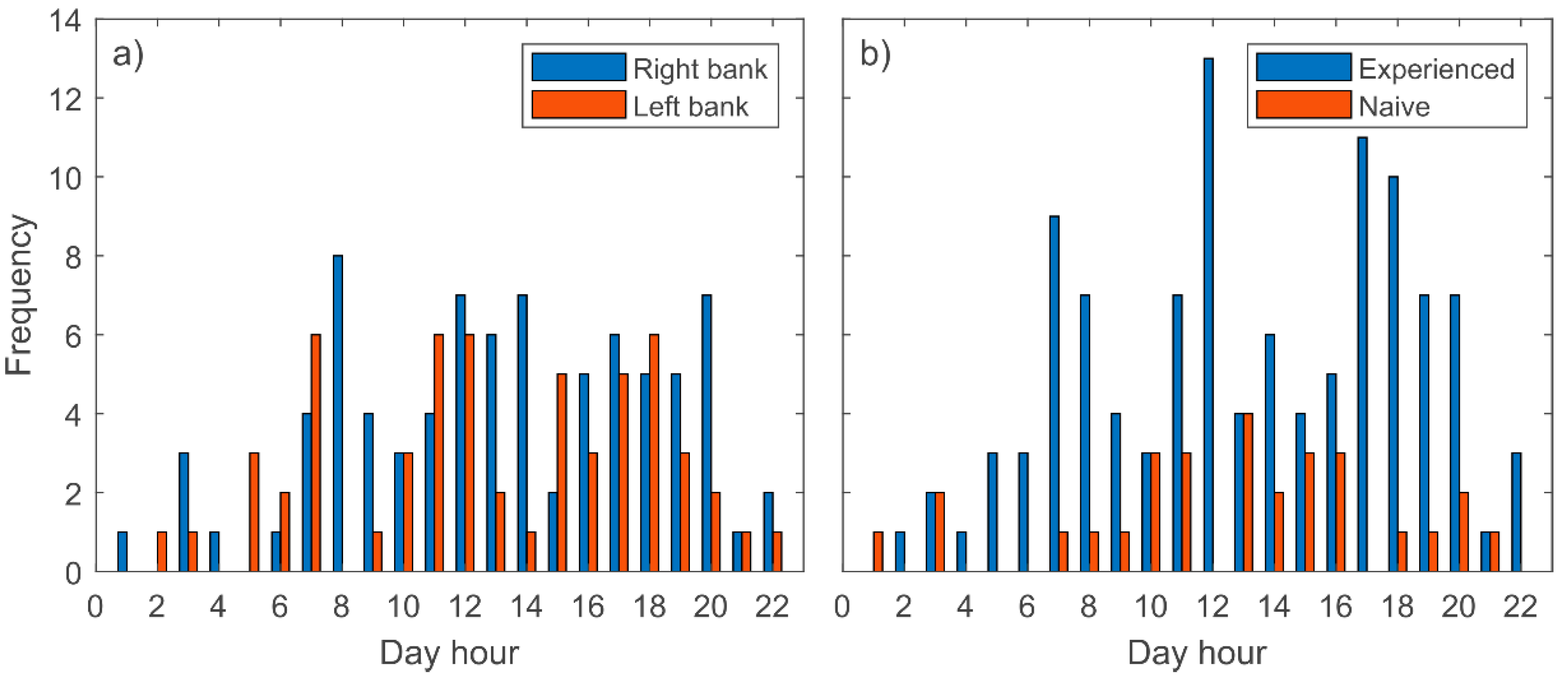
3. Results
4. Discussion
5. Summary and Conclusions
- A considerable percentage of barbel used the fishway for downstream movements, despite the low ratio between discharge and velocity fields at the turbines and the fishway, as well as the great distance between them.
- The distribution of fish along the cross-section affects the selection of passage routes in reservoirs with low water velocity and without clear waterways up to the turbine intakes.
- Previous experience of barbels affects their ability to locate the fishway.
- Barbels perform downstream movements from spring to autumn, and mainly during daylight.
- Fishways may serve as a complementary route in run-of-the-river small HPP with similar characteristics to the study site.
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Dingle, H.; Drake, V.A. What is migration? Bioscience 2007, 57, 113–121. [Google Scholar] [CrossRef]
- Lucas, M.C.; Baras, E.; Thom, T.J.; Duncan, A.; Slavík, O. Migration of Freshwater Fishes; Wiley Online Library: Oxford, UK, 2001. [Google Scholar]
- Brodersen, J.; Nilsson, P.A.; Hansson, L.-A.; Skov, C.; Brönmark, C. Condition-dependent individual decision-making determines cyprinid partial migration. Ecology 2008, 89, 1195–1200. [Google Scholar] [CrossRef]
- Chapman, B.B.; Skov, C.; Hulthén, K.; Brodersen, J.; Nilsson, P.A.; Hansson, L.; Brönmark, C. Partial migration in fishes: Definitions, methodologies and taxonomic distribution. J. Fish. Biol. 2012, 81, 479–499. [Google Scholar] [CrossRef] [PubMed]
- Ovidio, M.; Parkinson, D.; Philippart, J.-C.; Baras, E.; Guillaume, N.; Hallot, E.; Petit, F.; Philippart, J.-C.; Aparicio, E.; Sostoa, A.D. Multiyear homing and fidelity to residence areas by individual barbel (Barbus barbus). Belg. J. Zool. 2007, 137, 183–190. [Google Scholar]
- Britton, J.R.; Pegg, J. Ecology of European barbel Barbus barbus: Implications for river, fishery, and conservation management. Rev. Fish. Sci. 2011, 19, 321–330. [Google Scholar] [CrossRef]
- García-Vega, A.; Fuentes-Pérez, J.F.; Bravo-Córdoba, F.J.; Ruiz-Legazpi, J.; Valbuena-Castro, J.; Sanz-Ronda, F.J. Pre-reproductive movements of potamodromous cyprinids in the Iberian Peninsula: When environmental variability meets semipermeable barriers. Hydrobiologia 2021. [Google Scholar] [CrossRef]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes; Publications Kottelat: Cornol, Switzerland, 2007; Volume 2008, ISBN 978-2-8399-0298-4. [Google Scholar]
- Collares-Pereira, M.J.; Martins, M.J.; Pires, A.M.; Geraldes, A.M.; Coelho, M.M. Feeding behaviour of Barbus bocagei assessed under a spatio-temporal approach. Folia Zool. 1996, 45, 65–76. [Google Scholar]
- Rodriguez-Ruiz, A.; Granado-Lorencio, C. Spawning period and migration of three species of cyprinids in a stream with Mediterranean regimen (SW Spain). J. Fish. Biol. 1992, 41, 545–556. [Google Scholar] [CrossRef]
- Herrera, M.; Hernando, J.A.; Fernández-Delgado, C.; Bellido, M. Age, growth and reproduction of the barbel, Barbus sclateri (Günther, 1868), in a first-order stream in southern Spain. J. Fish. Biol. 1988, 33, 371–381. [Google Scholar] [CrossRef]
- Aparicio, E.; De Sostoa, A. Pattern of movements of adult Barbus haasi in a small Mediterranean stream. J. Fish. Biol. 1999, 55, 1086–1095. [Google Scholar] [CrossRef]
- Baras, E.; Cherry, B. Seasonal activities of female barbel Barbus barbus (L.) in the River Ourthe (Southern Belgium), as revealed by radio tracking. Aquat. Living Resour. 1990, 3, 283–294. [Google Scholar] [CrossRef]
- Baras, E.; Nindaba, J. Diel dynamics of habitat use by riverine young-of-the-year Barbus barbus and Chondrostoma nasus (Cyprinidae). Arch. Hydrobiol. 1999, 146, 431–448. [Google Scholar] [CrossRef]
- Roberts, C.G.; Britton, J.R. Trophic interactions in a lowland river fish community invaded by European barbel Barbus barbus (Actinopterygii, Cyprinidae). Hydrobiologia 2018, 819, 259–273. [Google Scholar] [CrossRef]
- Baras, E. Thermal related variations of seasonal and daily spawning periodicity in Barbus barbus. J. Fish. Biol. 1995, 46, 915–917. [Google Scholar] [CrossRef]
- Penaz, M.; Barus, V.; Prokes, M.; Homolka, M. Movements of barbel, Barbus barbus (Pisces: Cyprinidae). Folia Zool. 2002, 51, 55–66. [Google Scholar]
- Philippart, J.-C.; Baras, E. Comparison of Tagging and Tracking Studies to Estimate Mobility and Home-Range in Barbus barbus. In Underwater Biotelemetry. Proceedings of the First Conference and Workshop on Fish Telemetry in Europe; Université de Liège: Liège, Belgium, 1996; pp. 3–12. [Google Scholar]
- Benitez, J.; Ovidio, M. The influence of environmental factors on the upstream movements of rheophilic cyprinids according to their position in a river basin. Ecol. Freshw. Fish. 2018, 27, 660–671. [Google Scholar] [CrossRef]
- Pringle, C. What is hydrologic connectivity and why is it ecologically important? Hydrol. Process. 2003, 17, 2685–2689. [Google Scholar] [CrossRef]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Nilsson, C.; Reidy, C.A.; Dynesius, M.; Revenga, C. Fragmentation and flow regulation of the world’s large river systems. Science 2005, 308, 405–408. [Google Scholar] [CrossRef]
- Clay, C.H. Design of Fishways and Other Fish Facilities; CRC Press: Ottawa, ON, Canada, 1995; ISBN 1566701112. [Google Scholar]
- FAO/DVWK. Fish Passes: Design, Dimensions, and Monitoring; FAO: Rome, Italy, 2002; ISBN 9251048940. [Google Scholar]
- Larinier, M.; Porcher, J.P.; Travade, F.; Gosset, C. Passes à Poissons: Expertise, Conception des Ouvrages de Franchissement; CSP: Paris, France, 1994. [Google Scholar]
- DWA. Fish Protection Technologies and Downstream Fishways. Dimensioning, Design, Effectiveness, Inspection; DWA German Association for Water, Wastewater and Waste: Hennef, Germany, 2005. [Google Scholar]
- USBR. Fish Protection at Water Diversions. A Guide for Planning and Designing Fish. Exclusion Facilities; U.S. Department of Interior Bureau of Reclamation: Denver, CO, USA, 2006. [Google Scholar]
- Larinier, M.; Travade, F. Downstream migration: Problems and facilities. Bull. Français Pêche Piscic. 2002, 181–207. [Google Scholar] [CrossRef]
- Williams, J.G.; Armstrong, G.; Katopodis, C.; Larinier, M.; Travade, F. Thinking like a fish: A key ingredient for development of effective fish passage facilities at river obstructions. River Res. Applic. 2012, 28, 407–417. [Google Scholar] [CrossRef]
- Pelicice, F.M.; Pompeu, P.S.; Agostinho, A.A. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish Fish. 2015, 16, 697–715. [Google Scholar] [CrossRef]
- Coutant, C.C.; Whitney, R.R. Fish behavior in relation to passage through hydropower turbines: A review. Trans. Am. Fish. Soc. 2000, 129, 351–380. [Google Scholar] [CrossRef]
- Calles, O.; Greenberg, L. Connectivity is a two-way street. The need for a holistic aprroach to fish passage problems in regultated rivers. River Res. Appl. 2009, 25, 1268–1286. [Google Scholar] [CrossRef]
- Čada, G.F. The development of advanced hydroelectric turbines to improve fish passage survival. Fisheries 2001, 26, 14–23. [Google Scholar] [CrossRef]
- EPRI. Evaluation of Fish. Injury and Mortality Associated with Hydrokinetic Turbines; Electric Power Research Institute: Palo Alto, CA, USA, 2011. [Google Scholar]
- Pedersen, M.I.; Jepsen, N.; Aarestrup, K.; Koed, A.; Pedersen, S.; Økland, F. Loss of European silver eel passing a hydropower station. J. Appl. Ichthyol. 2012, 28, 189–193. [Google Scholar] [CrossRef]
- Wertheimer, R.H. Evaluation of a surface flow bypass system for steelhead kelt passage at Bonneville Dam, Washington. N. Am. J. Fish. Manag. 2007, 27, 21–29. [Google Scholar] [CrossRef]
- Schilt, C.R. Developing fish passage and protection at hydropower dams. Appl. Anim. Behav. Sci. 2007, 104, 295–325. [Google Scholar] [CrossRef]
- Kraabøl, M.; Johnsen, S.I.; Museth, J.; Sandlund, O.T. Conserving iteroparous fish stocks in regulated rivers: The need for a broader perspective! Fish. Manag. Ecol. 2009, 16, 337–340. [Google Scholar] [CrossRef]
- Celestino, L.F.; Sanz-Ronda, F.J.; Miranda, L.E.; Makrakis, M.C.; Dias, J.H.P.; Makrakis, S. Bidirectional connectivity via fish ladders in a large Neotropical river. River Res. Appl. 2019, 35, 236–246. [Google Scholar] [CrossRef]
- Pelicice, F.M.; Pompeu, P.S.; Agostinho, A.A. Fish conservation must go beyond the concrete: A comment on Celestino et al. (2019). River Res. Appl. 2020, 36, 1373–1376. [Google Scholar] [CrossRef]
- Ordeix, M.; González, G.; Sanz-Ronda, F.J.; Santos, J.M. Restoring Fish Migration in the Rivers of the Iberian Peninsula. In From Sea to Source 2. Protection and Restoration of Fish Migration in Rivers Worldwide; Brink, K., Gough, P., Royte, J., Schollema, P.P., Wanningen, H., Eds.; World Fish Migration Foundation: Groningen, The Netherlands, 2018. [Google Scholar]
- Belletti, B.; Leaniz, C.G.D.; Jones, J.; Bizzi, S.; Börger, L.; Segura, G.; Castelletti, A.; Van de Bund, W.; Aarestrup, K.; Barry, J. More than one million barriers fragment Europe’s rivers. Nature 2020, 588, 436–441. [Google Scholar] [CrossRef]
- Mayor, B.; Rodríguez-Muñoz, I.; Villarroya, F.; Montero, E.; López-Gunn, E. The role of large and small scale hydropower for energy and water security in the Spanish Duero Basin. Sustainability 2017, 9, 1807. [Google Scholar] [CrossRef]
- Gasith, A.; Resh, V.H. Streams in Mediterranean climate regions: Abiotic influences and biotic responses to predictable seasonal events. Annu. Rev. Ecol. Syst. 1999, 30, 51–81. [Google Scholar] [CrossRef]
- Tánago, M.D.G.; Martínez-Fernández, V.; Jalón, D.D.G. Diagnosing problems produced by flow regulation and other disturbances in Southern European Rivers: The Porma and Curueño Rivers (Duero Basin, NW Spain). Aquat. Sci. 2016, 78, 121–133. [Google Scholar] [CrossRef]
- Sanz-Ronda, F.J.; Bravo-Córdoba, F.J.; Sánchez-Pérez, A.; García-Vega, A.; Valbuena-Castro, J.; Fernandes-Celestino, L.; Torralva, M.; Oliva-Paterna, F.J. Passage performance of technical pool-type fishways for potamodromous cyprinids: Novel experiences in semiarid environments. Water 2019, 11, 2362. [Google Scholar] [CrossRef]
- CHD. Inventory of River Obstacles in the Spanish part of the Duero River Basin. Information System “Mírame IDEDuero.” Confederación Hidrográfica del Duero. Resources. Available online: http://www.mirame.chduero.es/DMADuero_09/webPresas/presaGlobal.faces (accessed on 10 February 2021).
- IUCN. The IUCN Red List of Threatened Species. Version 2020-International Union for Conservation of Nature and Natural Resources. Available online: www.iucnredlist.org (accessed on 10 February 2021).
- Illies, J.; Botoseanu, L. Problèmes et méthodes de la classification et de la-zonation écologique des eaux courantes, considérées surtout-du point de vue faunistique. Mitt. Int. Ver. Theor. Amgewandte Limnol. 1963, 12, 1–57. [Google Scholar] [CrossRef]
- Rosgen, D.L.; Silvey, H.L. Applied River Morphology; Wildland Hydrology: Pagosa Springs, CO, USA, 1996; Volume 1481. [Google Scholar]
- Larinier, M. Pool fishways, pre-barrages and natural bypass channels. Bull. Fr. Pêche Piscic. 2002, 364, 54–82. [Google Scholar] [CrossRef]
- Ruiz-Legazpi, J.; Sanz-Ronda, F.J.; Bravo-Córdoba, F.J.; Fuentes-Pérez, J.F.; Castro-Santos, T. Influence of environmental and biometric factors on the swimming capacity of the Iberian barbel (Luciobarbus bocagei Steindachner, 1864), an endemic potamodromous cyprinid of the Iberian Peninsula. Limnetica 2018, 37, 251–265. [Google Scholar] [CrossRef]
- Bravo-Córdoba, F.J.; Valbuena-Castro, J.; García-Vega, A.; Fuentes-Pérez, J.F.; Ruiz-Legazpi, J.; Sanz-Ronda, F.J. Fish passage assessment in stepped fishways: Passage success and transit time as standardized metrics. Ecol. Eng. 2021, 162. [Google Scholar] [CrossRef]
- Britto, S.G.D.C.; Carvalho, E.D. Reproductive migration of fish and movement in a series of reservoirs in the Upper Parana River basin, Brazil. Fish. Manag. Ecol. 2013, 20, 426–433. [Google Scholar] [CrossRef]
- Havn, T.B.; Sæther, S.A.; Thorstad, E.B.; Teichert, M.A.K.; Heermann, L.; Diserud, O.H.; Borcherding, J.; Tambets, M.; Økland, F. Downstream migration of Atlantic salmon smolts past a low head hydropower station equippped with Archimedes screw and Francis turbines. Ecol. Eng. 2017, 105, 262–275. [Google Scholar] [CrossRef]
- Cooke, S.J.; Hinch, S.G. Improving the reliability of fishway attraction and passage efficiency estimates to inform fishway engineering, science, and practice. Ecol. Eng. 2013, 58, 123–132. [Google Scholar] [CrossRef]
- Bravo-Córdoba, F.J.; Sanz-Ronda, F.J.; Ruiz-Legazpi, J.; Celestino, L.F.; Makrakis, S. Fishway with two entrance branches: Understanding its performance for potamodromous Mediterranean barbels. Fish. Manag. Ecol. 2018, 25, 12–21. [Google Scholar] [CrossRef]
- Baras, E. Etude des stratégies d’occupation du temps et de l’espace chez le barbeau fluviatile, Barbus barbus L. Cah. D’éthologie 1992, 12, 125–442. [Google Scholar]
- Bravo-Córdoba, F.J.; Sanz-Ronda, F.J.; Navas-Pariente, A.; Valbuena-Castro, J. Comportamiento del barbo ibérico (Luciobarbus bocagei) en una escala de peces de vertedero sumergido con orificio de fondo. Cuad. Soc. Española Cienc. For. 2019, 231–240. [Google Scholar] [CrossRef]
- FIThydro. Deliverable 2.2 Working Basis of Solutions, Models, Tools and Devices and Identification of Their Application Range on a Regional and OverAll Level to Attain Self-Sustained Fish Populations; Fishfriendly Innovative Technologies for Hydropower: Munich, Germany, 2019. [Google Scholar]
- Havn, T.B.; Thorstad, E.B.; Borcherding, J.; Heermann, L.; Teichert, M.A.K.; Ingendahl, D.; Tambets, M.; Sæther, S.A.; Økland, F. Impacts of a weir and power station on downstream migrating Atlantic salmon smolts in a German river. River Res. Appl. 2020, 36, 784–796. [Google Scholar] [CrossRef]

| Experience | Release Bank | n | Fork Length (mm) | |
|---|---|---|---|---|
| Mean ± SD | Range | |||
| Experienced | Left | 104 | 149 ± 8 | 91–293 |
| Right | 81 | 144 ± 7 | 100–245 | |
| Naive | Left | 38 | 139 ± 13 | 80–235 |
| Right | 76 | 155 ± 15 | 77–420 | |
| Metric | Global | Experience | Release Bank | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Experienced | Naive | Left | Right | ||||||||||
| Release Bank | Global | Global | Release Bank | Experience | Global | Global | Experience | ||||||
| Left | Right | Left | Right | Experienced | Naive | Experienced | Naive | ||||||
| Location | 40.1% (120/299) | 34.6% (36/104) | 70.4% (57/81) | 50.3% (93/185) | 23.7% (27/114) | 26.3% (10/38) | 22.4% (17/76) | 34.6% (36/104) | 26.3% (10/38) | 32.4% (46/142) | 47.1% (74/157) | 70.4% (57/81) | 22.4% (17/76) |
| p < 0.001 | p < 0.001 | p = 0.640 | p = 0.350 | p = 0.009 | p < 0.001 | ||||||||
| Entry * | 34.5% (103/299) | 29.8% (31/104) | 67.9% (55/81) | 46.5% (86/185) | 14.9% (17/114) | 21.1% (8/38) | 11.8% (9/76) | 29.8% (31/104) | 21.1% (8/38) | 27.5% (39/142) | 40.8% (64/157) | 67.9% (55/81) | 11.8% (9/76) |
| p < 0.001 | p < 0.001 | p = 0.193 | p = 0.301 | p = 0.016 | p < 0.001 | ||||||||
| Downstream passage success | 24.8% (74/299) | 23.1% (24/104) | 46.9% (38/81) | 33.5% (62/185) | 10.5% (12/114) | 13.2% (5/38) | 9.2% (7/76) | 23.1% (24/104) | 13.2% (5/38) | 20.4% (29/142) | 28.7% (45/157) | 46.9% (38/81) | 9.2% (7/76) |
| p < 0.001 | p < 0.001 | p = 0.517 | p = 0.194 | p = 0.099 | p < 0.001 | ||||||||
| Location time (median) | 1.9 days (n = 120) | 1.9 days (n = 36) | 1.8 days (n = 57) | 1.9 days (n = 93) | 2.0 days (n = 27) | 2.3 days (n = 10) | 2.0 days (n = 17) | 1.9 days (n = 36) | 2.3 days (n = 10) | 1.9 days (n = 46) | 1.8 days (n = 74) | 1.8 days (n = 57) | 2.0 days (n = 17) |
| p = 0.303 | p = 0.843 | p = 0.744 | p = 0.947 | p = 0.267 | p = 0.898 | ||||||||
| Global | Experience | Release Bank | ||||
|---|---|---|---|---|---|---|
| Experienced | Naive | Left | Right | |||
| Downstream transit time | Median | 2.2 h (n = 74) | 2.2 h (n = 62) | 3.2 h (n = 12) | 1.5 h (n = 29) | 4.8 h (n = 45) |
| IQR | 1.0–8.7 h | 1.0–7.8 h | 0.8–9.3 h | 0.7–3.1 h | 1.2–9.9 h | |
| p-value | p = 0.988 | p = 0.011 | ||||
| Month (2020) | June | July | August | September | October |
|---|---|---|---|---|---|
| Downstream proportion | 58% (11/19) | 71% (12/17) | 35% (6/17) | 50% (21/42) | 100% (3/3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanz-Ronda, F.J.; Fuentes-Pérez, J.F.; García-Vega, A.; Bravo-Córdoba, F.J. Fishways as Downstream Routes in Small Hydropower Plants: Experiences with a Potamodromous Cyprinid. Water 2021, 13, 1041. https://doi.org/10.3390/w13081041
Sanz-Ronda FJ, Fuentes-Pérez JF, García-Vega A, Bravo-Córdoba FJ. Fishways as Downstream Routes in Small Hydropower Plants: Experiences with a Potamodromous Cyprinid. Water. 2021; 13(8):1041. https://doi.org/10.3390/w13081041
Chicago/Turabian StyleSanz-Ronda, Francisco Javier, Juan Francisco Fuentes-Pérez, Ana García-Vega, and Francisco Javier Bravo-Córdoba. 2021. "Fishways as Downstream Routes in Small Hydropower Plants: Experiences with a Potamodromous Cyprinid" Water 13, no. 8: 1041. https://doi.org/10.3390/w13081041
APA StyleSanz-Ronda, F. J., Fuentes-Pérez, J. F., García-Vega, A., & Bravo-Córdoba, F. J. (2021). Fishways as Downstream Routes in Small Hydropower Plants: Experiences with a Potamodromous Cyprinid. Water, 13(8), 1041. https://doi.org/10.3390/w13081041









