1. Introduction
Nearly 90% of the world population has access to electricity, while in sub-Saharan Africa only 47% of the population has access, and as many as 548 million people lack electricity. Globally, 85% of the people currently without electricity live in rural areas [
1]. Rural electrification, defined as the percentage of the rural population with access to electricity, is a crucial part of socio-economic development [
2]. The seventh UN Sustainable Development Goal, to ensure access to affordable, reliable, sustainable and modern energy for all, reverberates the importance of access to energy for sustainable development [
3]. In Mozambique, with a total population of about 30 million, 31% of the population has access to electricity. Rural areas host about 70% of the country’s total population, of which only 8% have access to use of electricity for lighting [
4]. The primary source of energy in rural Mozambique is biomass, in the form of fuelwood, which is mainly used by households for heating and cooking purposes [
5,
6]. The national grid, including off-grid facilities, gives 30% of the country’s population potential access to the grid. The overwhelming majority of rural households and most rural schools, health centers and administrative posts do not have access to electricity [
5]. The limited extent of grid electricity is due to the high costs of expanding power grids to distant and isolated areas where the inhabitants are mostly poor, giving little return on invested capital in the rural areas [
5]. Local off-grid energy systems form an essential part of the electrification of these communities. Use of local biomass for energy conversion instead of imported fossil fuels could be more sustainable from an environmental, social and economic point of view.
This study was conducted in Inhambane Province, which is a province in southern Mozambique, and which has the third highest production of sweet cassava (
Manihot spp.) among all the provinces in the country [
7]. Apart from cassava, there are several agro-industries processing coconut wood (
Cocos nucifera) and jambolan wood (
Syzygium cumini) in Inhambane. Cassava is an important crop in Mozambique, used for food, flour and beer brewing. Inhambane has many palm trees and is a producer of coconut. Palm trees affected by diseases are felled to take advantage of the logs for wood. Wood from coconut and jambolan is used for construction material. The size of the agro-industries in the province ranges from small community-based businesses to a few large-scale industries. Firewood is the primary source of energy for both households and local business activities. Processing of cassava, coconut and jambolan wood generates large amounts of agro-waste. About 1.5 million tons of cassava peels are generated and discarded annually in Mozambique [
8]. Agro-waste causes environmental problems, allowing microbes and germs to thrive [
9]. It is therefore important to find alternative uses for agro-waste, and thereby decrease its aggregation in the local environment. To turn the biomass into energy could be one alternative. Ref. [
10] determined the high heat value of cassava waste to be between 16.59 and 18.75 MJ/kg. Studies conducted by [
11,
12,
13] have shown that the biogas yield of cassava makes it a suitable energy crop. The biogas yields from cassava peels were determined by [
14] to be the highest at 0.61 m
3 kg
−1 volatile solids (VS), which is comparable to other agricultural residues such as rice straw, wheat straw and coffee pulp. There are more than 100 varieties of cassava grown in Mozambique. The biomethane potential of the varieties of sweet cassava grown in Inhambane is still not very well known.
It is commonly known that woody biomass is not highly biodegradable due to its lignocellulose content. In this study, sawdust, a fine-grained power, was analyzed to see if that would increase the biochemical methane potential (BMP) compared with coarser woody biomass.
Several studies conducted during the last decade have identified lignocellulosic biomass as a potential substrate for biogas production [
15,
16,
17,
18]. Ref. [
16] showed that sawdust from wood processing mills could produce significant volumes of biomethane. Ref. [
17] concluded that there is significant variation in the biomethane potential of different woody species. The biomethane production is highly dependent on the concentration of lignin in the substrate [
18,
19]. Pre-treatment of the sawdust, by breaking the shield of lignin that protects the cellulose and hemicellulose, is essential to increase biomethane production [
15,
16].
In this context, this study investigated the biomethane potential of cassava peels from the four most common varieties of cassava grown in Inhambane Province, Chinhembue, Cussi, Cizangara and Nhassumbi. Agro-wastes from coconut wood and jambolan wood processing were also analyzed to give a complete analysis of the most significant sources of agro-waste in the province.
2. Methods
2.1. Study Area
This study was conducted in the districts of Inharrime and Jangamo, in Inhambane Province, southern Mozambique (
Figure 1). The districts of Inharrime and Jangamo have populations of 123,605 and 105,478 inhabitants, respectively [
20]. In these districts, agriculture, livestock, tourism, industry, commerce and fishing are the main economic activities. About 90% of the population practices subsistence agriculture, producing food crops in the form of maize, cassava, peanuts, nhemba beans, jugo beans and vegetables. In addition to food crops, many also produce cash crops, such as citrus fruit, pineapple, piri-piri, cashew nut and mango. Agricultural production is characterized by low production and productivity rates due to rudimentary production technologies, mainly working the land manually, and in some cases using animal traction, with limited ability to practice irrigated agriculture. The local government is promoting small industries, supporting small producers and associations in small units for processing agricultural products, mainly cassava. At this moment, the study area has an industrial cassava processing unit located in the Inharrime district; small micro- and small-scale cassava processing units in Inharrime and Jangamo; and small micro- and small-scale processing units for coconut wood and jambolan wood (sawmills) in Jangamo.
2.2. Test Substrates
The substrates used in this study are agro-wastes from the processing of cassava (peels, fiber and pressed fiber) and sawdust from coconut wood and jambolan wood. Cassava peels of the four most common varieties of cassava grown in Inhambane Province, Chinhembue, Cussi, Cizangara and Nhassumbi, were collected. The cassava fiber is a residue from the mixing and processing of the four varieties of cassava.
In the Inharrime district, agro-waste from cassava was collected from the industrial cassava processing unit of the DADTCO Mandioca Mozambique company, and from the small-scale producers of tapioca and flour in the Josina Machel association. In the Jangamo district, agro-waste from cassava was collected from the Licaca Agricultural Cooperative, and samples of sawdust from coconut and jambolan wood were collected from small-scale sawmills in the district.
After collection, the samples were vacuum-packed and stored at 4 °C in a freezer until analysis in the laboratory. The excessively large organic fraction particles of cassava peels and the unpressed cassava fibers were ground and shredded. All particles were <10 mm in any dimension. After preparation, substrate samples were stored at 4 °C for five days in a freezer. All samples analyzed were frozen. The thawing of frozen samples was performed at 4 °C, followed by carefully increasing the sample temperature to ambient conditions before use in the BMP test. According to [
21], thawing may affect the BMP of some substrates. All substrates were frozen, so it can be assumed that thawing affected BMP equally for all samples.
2.3. Collection and Pre-Treatment
All substrates were analyzed by Eurofins (Eurofins Food & Feed Testing, Lidköping, Sweden) for their macromolecular composition (following NMKL 173 for ash, NMKL 160 mod. for lipids, NMKL 6:2003 for proteins, and carbohydrates were calculated following EU 1169/2011). Macromolecular composition was used to calculate the theoretical maximum biochemical methane potential (BMP) with a BMP of 414, 534 and 1010 NmL
CH4 g
VS−1 for carbohydrates, proteins and lipids, respectively, assuming 100% degradation without use of substrate for microbial biomass production [
22]. Total solids (TS) and volatile solids (VS) were determined in triplicate by drying at 105 °C and igniting at 550 °C according to DIN EN 12879 and DIN EN 12880.
Microcrystalline cellulose (CEL) (CAS Number 9004-34-6, Sigma-Aldrich Chemie GmbH, Buchs, Switzerland) was used as a positive control in the BMP test to validate the BMP results obtained.
2.4. BMP Measurement
BMP tests were carried out in triplicate at 36 °C with blanks (inoculum only) and CEL as a positive control. A mixture of digestate (1:1 based on volume) from two full-scale anaerobic digestion plants (food waste digester operated by Växtkraft in Västerås, Sweden and sewage sludge digester operated by Mälarenergi in Västerås, Sweden) was used as inoculum (TS: 3.3% of fresh matter (FM), VS: 59.9% of TS) in order to supply a diverse methanogenic community. The digestates were sieved (<4 mm), mixed and pre-incubated at the test condition temperature for five days before the BMP test start.
Glass bottles with a total volume of 1100 mL were used with an active volume of 700 mL. The inoculum/substrate ratio (ISR) was 2:1 in all bottles. A trace element and vitamin solution (according to the recommendations by [
23]) was added to all samples. The bottles were sealed with butyl caps and aluminum screw tops, and the headspaces were flushed with a mixture of N
2 and CO
2 (35 vol%).
The bottles were mixed manually once per day before measurement. The duration of the test was at least until the net CH
4 production (CH
4 production of blanks subtracted from total CH
4 production in the sample) dropped below 1% of the cumulative production for 3 consecutive days. The BMP was accordingly reported as BMP1% net 3d as suggested by [
24].
Methane content was measured using gas chromatography (Agilent 490 Micro-GC, Da Vinci Laboratory Solutions, Rotterdam, Netherlands). The gas chromatograph was equipped with two channels (CH1: packed column (10 m MS5Acolumn), thermal conductivity detector, carrier gas Argon; CH2: 1 m COX column, thermal conductivity detector, carrier gas hydrogen) and externally calibrated to detect CH4, CO2, O2, N2 and H2. Samples were taken from the bottle headspace with a syringe and injected manually into the sample line. The measured methane content was normalized (sum of CO2 and CH4 to 100 vol%).
Two measurement methods (manometric with gas chromatography and gravimetric with gas chromatography) were compared in order to validate the measurement data.
For the manometric measurement, headspace pressure was measured (GMH3155, Greisinger, Remscheid, Germany) for each sampling point before venting the biogas produced until ambient pressure was reached (after taking a sample for GC analysis). The gravimetric method was based on measurement of mass loss in the bottles [
25]. Mass of the bottles (BP3100S, Sartorius, Göttingen, Germany) was measured for each sampling point before biogas was vented to ambient air pressure and a sample was taken for GC measurement.
The web application Online Biogas App (OBA) based on the biogas package in the programming software R [
22] was used to calculate the BMP from all measurement data as recommended by [
24]. All gas volumes were standardized to dry volume at 0 °C and 101.325 kPa. A statistical analysis (
t-test) was performed to compare the variance between the two different methods tested. Corresponding daily mean BMP values based on triplicates for each substrate for both methods were compared with an F-test and a
t-test. The tests were validated using the most recently recommended criteria [
24] based on CEL BMP mean and the relative standard deviation among the triplicates for mean BMP (RSD
B):
2.5. Electricity Production
Electricity production was estimated based on the potential amount of CH
4 that could be produced with amounts available in the regions per day, the energy content of CH
4 (10kWh/Nm
3 CH
4) and the electricity efficiency for a power unit. The amount of CH
4 per day was estimated by multiplying the mass of substrate available per day with TS and VS content, BMP value and degradability obtained from the experimental results obtained in this work. An electricity efficiency of 35% was used for the power unit [
26].
3. Results
Substrate characteristics are summarized in
Table 1. All measurements were made by Eurofin. For all parameters, an uncertainty of 10% was given by the lab. All substrates used in this study showed a high organic content (>90% of the total solids). Cassava peel samples had the highest water content (72–76% of fresh matter) and the lowest organic content. Differences between the four different cassava varieties were rather low. Chinhembue cassava peels (CAS1) however showed the highest content in carbohydrates and lowest protein content. More extensive analysis of cassava peels has been published elsewhere [
27] showing that carbohydrate content was between 63% and 85% TS, as obtained in this study, and both cellulose and lignin were present with 14% and 11–14% TS, respectively.
Pressed mixed cassava fiber had a high dry matter content (88% of fresh matter), and both fiber substrates had very low content in proteins and lipids.
The resulting theoretical maximum biochemical methane potential (BMP) was similar for all samples, ranging from 415 to 457 NmL
CH4 g
VS−1 (
Table 2) due to the high carbohydrate content in all samples. The validation criteria for the positive control cellulose (CEL) as suggested by [
24] (see method section) were used to validate the BMP results obtained. Gravimetric BMP measurement was performed in addition to check the accuracy of manometric BMP measurement. The CEL BMP1% net 3d was for both measurement methods within the suggested BMP range (340 and 395 NmL
CH4 g
VS−1), with a test duration of 18 days until net methane production of 3 consecutive days was lower than 1% of the cumulative production. Degradability was 83% and 84% for CEL manometric and CEL gravimetric. These values are in line with measurements of microbial yields that suggest that 85% of the theoretical maximum BMP should be obtained for complete cellulose degradation without degradation of microbial biomass [
24]. Relative standard deviation of the mean was for both methods lower than the suggested 6% (3.2% and 2.2% for CEL manometric and CEL gravimetric, respectively). The BMP measurements presented in this study can accordingly be validated following the current recommendations as suggested by the research community. In the following, results for the manometric measurements are presented; however, gravimetric measurements were used to check the accuracy of the measurement method as suggested by [
24]. Except for one sample (SD2), where the BMP value was lower for the gravimetric method, the BMP values obtained with the manometric method were lower. The longer test duration that was observed for the gravimetric method to reach the BMP1% net 3d criteria had only a minor impact on that difference. However, statistical analysis showed that the variances of the two sample populations were equal (with 95% confidence) and differences between the methods are not statistically significant.
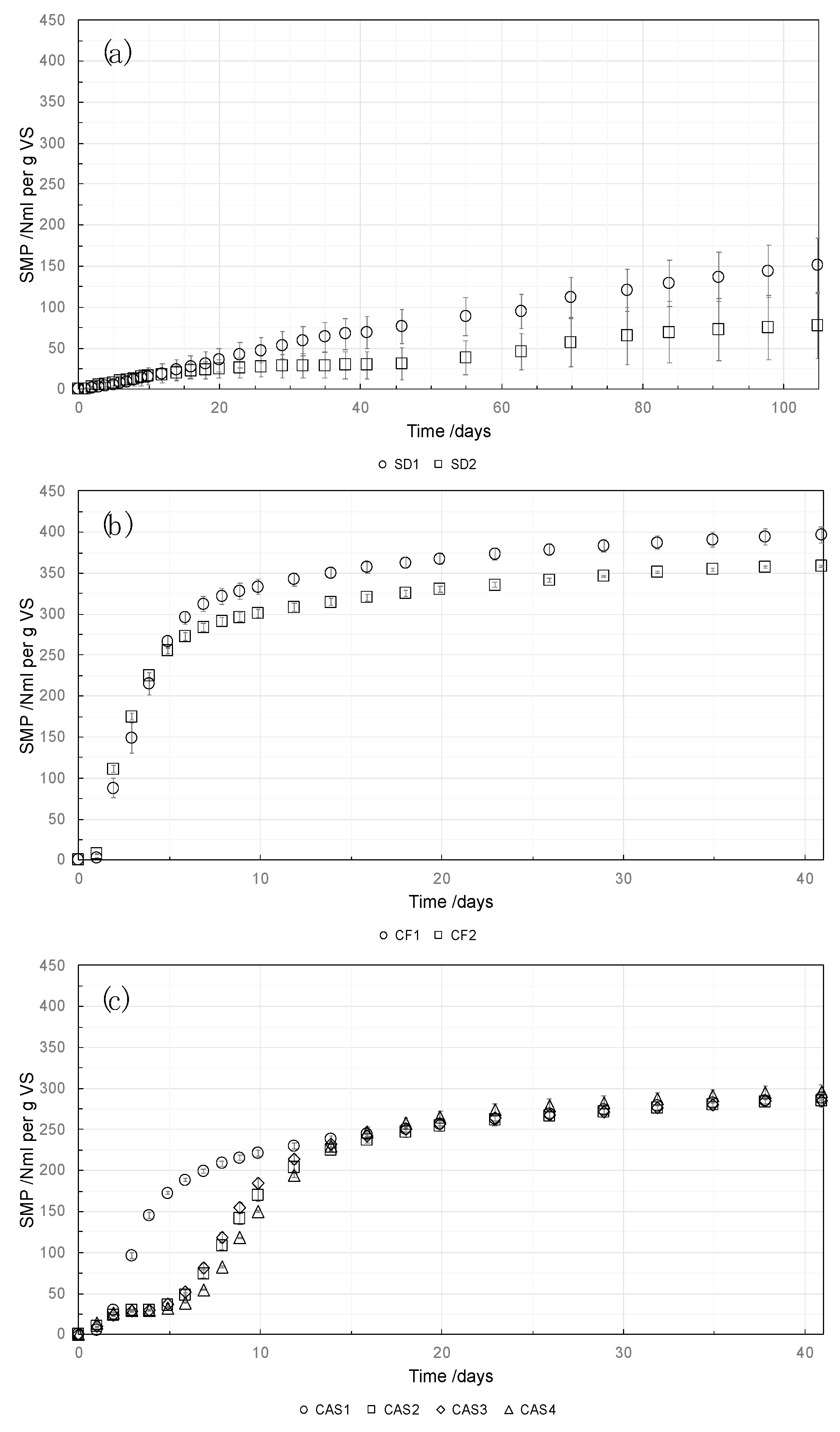
The three different substrate groups (sawdust, cassava peels and cassava fiber) showed large difference in BMP values (manometric measurement) between the different groups but similar behavior within those groups. Sawdust showed a considerably low BMP (129 and 73 NmL
CH4 g
VS−1 for SD1 and SD2) with a high relative standard deviation (>10%, see [
21]). Both sample groups (SD1 and SD2) had one extreme outlier, whereas the other two samples were similar. A statistical test to eliminate outliers was not done due to the generally low BMP value and the resulting lack of suitability of the substrate for anaerobic digestion, as could be confirmed by the low degradability (29% and 18% for SD1 and SD2) and long test duration (91 and 98 days for SD1 and SD2). All other samples in both measurement methods showed very low relative standard deviation (0.6–4.0%).
The largest BMP values were obtained for the two substrates within the cassava fiber group (363 and 326 NmLCH4 gVS−1 for CF1 and CF2) with a high degradability (86% and 78% for CF1 and CF2) and short test duration of 18 days.
For all samples within the cassava peel group (CAS1–4), a similar, medium BMP value was obtained. BMP and degradability were between 263 and 279 NmL
CH4 g
VS−1 and 58% to 62%, respectively, with a test duration of 23 to 26 days. Interestingly, a difference in the kinetics could be observed for one of the samples compared with the remaining three (
Figure 2). The latter showed a retarded methane production in the first 6 days of the experiment.
Based on the availability of agro-waste generated by the cassava producers included in this study and the amount of biomethane produced in our laboratory experiment, we estimate how much electricity can be generated. In the DADTCO Mandioca Mozambique company, the results reveal that from 40,331 kg of CF2 and 8892 kg of cassava peels generated/day, 30,892 and 1196 kWh/day can be produced, respectively. In the Josina Machel association, 40 kg of CF1 generated/day can produce 20 kWh/day, and 200 kg of cassava peels generated/day can produce 27 kWh/day. In the Licaca Agricultural Cooperative, 30 kg of CF1 generated/day can produce 15 kWh/day, and 300 kg of cassava peels generated/day can produce 40 kWh/day. The calculations show that the agro-waste generated by the cassava producers in this study could have a potential of around 32,190 kWh/day.
4. Discussion
Cellulose BMP between 340 and 395 NmL
CH4 g
VS−1 and RSD
B ≤ 6%, with a duration of at least 1% net 3 d (by substrate), makes the tests valid [
24]. The low percentage (cellulose BMP RSD
B ≤ 6%) indicates that the dispersion of the results of the biomethane potentials of the triplicates (three bottles used for cellulose in the experiment) is small, which validates the results of the BMP tests.
According to
Table 2, coconut sawdust (SD1) has a higher theoretical maximum BMP than Jambalan sawdust (SD2). The reason for this is due to the higher contents of carbohydrates, lipids and proteins as shown in
Table 1. A similar result was shown by [
16]. Furthermore, in
Table 2 cassava peels have higher theoretical maximum BMP than cassava fiber. This is due to the higher content of lipids and proteins in the cassava peels as seen in
Table 1.
In
Table 1, it is shown that there is a difference in total solids content between pressed mixed cassava fiber (CF2) and mixed cassava fiber (CF1). Pressed mixed cassava fiber is waste from the mechanical processing of cassava collected after pressing at the DADTCO Mandioca Mozambique company, while mixed cassava fiber (CF1) is waste from manual processing, with higher moisture content, collected in the Josina Machel association and Licaca Agricultural Cooperative. Thus, due to the pressing, which removes more of the liquids, the pressed mixed cassava fiber has a higher total solids content than mixed cassava fiber. Further, the lower content of volatile solids, carbohydrates and lipids in pressed mixed cassava fiber (CF2) compared with mixed cassava fiber (CF1) is likely due to probable losses in mechanical processing. Therefore, samples of pressed mixed cassava fiber (CF2) have lower theoretical maximum BMP and mean BMP than mixed cassava fiber (
Table 2).
The high carbohydrate content of cassava peel from the composition analysis could partly be attributed to the peeling process (manual peeling step), which normally leaves some amount of the flesh that contains starch as well as the cellulose [
28] (
Table 1). For cassava fiber, the mechanical or manual processing of cassava also leaves some amount of the flesh that contains starch as well as the cellulose content, which raises the carbohydrate content (
Table 1).
The total content of proteins and lipids in the cassava peel and cassava fiber could be due to the variety of cassava as well as the cultivar, the environment and several other factors (
Table 1). For the cassava fiber in particular, it also depends on the type of cassava processing.
During fieldwork, it was observed that the industrial process involves filtration and decantation, while the manual process involves pressing and sieving of the fibers. Similar processes have been described by [
29]. Based on the results presented in
Table 1, the industrial process seems to remove more of the proteins and lipids from the fiber compared with the manual process.
In
Figure 2, it can be seen that cassava fiber has a higher biomethane potential compared with peels and sawdust. Furthermore, cassava fiber follows the expected biomethane production over time. Biomethane yields of 86% and 78% of the theoretical maximum in the manometric analysis, and 91% and 85% of the theoretical maximum in the gravimetric analysis, were obtained in cassava fibers. The higher biomethane yield from cassava fiber compared with peels and sawdust is likely due to the higher content of carbohydrates and less lignin compared with the peels and sawdust, as can be seen in
Table 1. A similar relationship has been reported by [
30] showing that lignin acts as structural barrier and also affects the hydrolysis by unproductive binding of enzymes to lignin.
Cassava peels have less biomethane potential compared with fibers and higher compared with sawdust (
Figure 2). Biomethane yields ranging from 58% to 62% of the theoretical maximum in the manometric analysis, and 62% to 67% of the theoretical maximum in the gravimetric analysis, were obtained in the four varieties of cassava peel (
Table 2). The composition of the cassava peel is made up of residual starch, cellulose, hemicellulose and lignin [
28,
31]. In this context, the slow start for three of the four cassava peel samples could be as a result of the high level of lignin content (
Figure 2). Analyses of the chemical composition of cassava peels gave the following variation in the lignin content: 1.92% [
28], 7.52% [
32], 8.4% [
31]. A high lignin content inhibits initial hydrolysis of substrates [
30]. This explains why cassava peels have a lower biomethane potential than cassava fibers and higher than sawdust.
On the other hand, during anaerobic degradation of cassava peel, its composition (high starch content, high carbon to nitrogen ratio, presence of cyanogenic glucosides) usually results in excess acid production, nitrogen deficiency and the release of cyanide, which is highly toxic to the methanogenic community [
14].
Several studies investigating cassava peel reported temporary inhibition due to acidification based on the high amount of available carbohydrates in the substrate [
27,
33]. This explanation is highly unlikely here when comparing the composition and kinetic behavior of the affected samples with samples from the cassava fiber group that contain higher amounts of carbohydrates and organic compounds in general, a higher BMP value and a higher daily production at the beginning of the experiment. The cassava peel samples showed a higher complexity in their composition with higher protein and lipid content, components that are known for lower hydrolysis rate constants [
34]. Though the lipid and protein contents were lower for CAS1 that did not show retarded degradation, carbohydrates as main components were present in all samples, and accordingly, the effect observed would have been expected at a later time after the degradation of carbohydrates has taken place. However, cassava roots, peels and leaves contain two cyanogenic glucosides, linamarin and lotaustralin [
35]. The enzymatic degradation of those compounds by the natural occurring linamarase in cassava peels liberates hydrogen cyanide (HCN). Cassava roots are classified as toxic or non-toxic based on the amount of cyanide in the roots, which is dependent on different factors such as plant age or access to water during production and further processing of the roots and peels [
35,
36,
37]. Cyanide levels of between 1 and 1500 ppm can be found in cassava peels [
28]. Addition of cyanide (as KCN or linamarin) showed temporary inhibition of methanogenesis at concentrations >6 mg L
−1 [
38]. Removal of cyanide was however observed in both non-enzymatic processes (e.g., complexation and volatilization) and the enzymatic reaction during anaerobic digestion. It is therefore most likely that samples CAS2–4 contained high levels of toxic cyanogenic glucosides, which caused a temporary inhibition of methanogenesis [
39]. Cyanide is a toxic compound that would affect/inhibit the population of microorganisms [
28]. As a consequence, the presence of cyanogenic glucosides is an important factor when considering cassava peels as a substrate for anaerobic digestion. Linamarin can be broken under high pressure, high temperature and the use of mineral acids [
35]. In addition, biological pre-treatment has been considered a cheap and simple method for reducing HCN [
40] as well as co-digestion with substrates that additionally balance out the high carbon to nitrogen ratio [
33]. This is a theory that could explain the inhibition, but no measurements of the amount of cyanide in the samples were performed to confirm this hypothesis.
Sawdust (SD1 and SD2), despite having high theoretical maximum BMP, has less biomethane potential (29% and 18% of the theoretical maximum in manometric analysis; 32% and 14% of the theoretical maximum in the gravimetric analysis, respectively) compared with fibers and peels (
Table 2), with a slow start to the methane production (
Figure 2). This phenomenon could be as a result of the high level of lignin content, which inhibits the hydrolysis of lignocellulosic biomass [
16,
28,
30].
To complete the BMP test of the sawdust, SD1 and SD2, took 91 and 98 days in the manometric analysis, and 105 and 98 days in the gravimetric analysis, respectively. The test took much longer compared with the BMP test of the cassava peels and cassava fiber. This is likely due to the high lignin content in the sawdust, which hinders sample degradability (Fd) (
Table 2). The negative effects of high lignin content on BMP have been shown by [
15,
16,
19].
The total estimated amount of electricity that can be produced from agro-waste at the DADTCO Mandioca Mozambique company is about 32,000 kWh/day. This electricity can provide a significant contribution to the processing of cassava at the company. For the Josina Machel association, the estimated amount of electricity that can be produced is in the range of 47 to 55 kWh/day. This electricity could be used for small-scale production of cassava at the association, and possibly for lighting. This energy would reduce the dependency on wood for the processing of cassava.
5. Conclusions
In this study, we investigated the biomethane potential of cassava peels from the four most common varieties of cassava grown in Inhambane Province, Chinhembue, Cussi, Cizangara and Nhassumbi. Agro-wastes from coconut wood and jambolan wood processing were also analyzed to give a complete analysis of the most significant sources of agro-waste in the province. Our results show that cassava fiber and cassava peel, which are considered to be waste, are viable resources for biomethane production. The results show that cassava fiber and cassava peels have a higher biomethane potential compared with sawdust, and are therefore more suitable for biomethane production. This is likely due to the higher lignin content of the sawdust, which inhibits the process.
The low biomethane potential of sawdust due to higher lignin content can be optimized in future work, e.g., by using substrate pre-treatment techniques. A study to investigate the effect of toxic cyanogenic glycosides and lignin on cassava peels, using pre-treatment techniques to enhance biogas yield, should be carried out. A study should also be carried out in the study area to investigate the effect on the biomethane yield when blending cassava peels with cassava fiber and animal waste.
According to the estimate of electricity production, biogas produced from cassava peels and cassava fibers is viable as a productive energy source for implementation in small and medium sized enterprises (SMEs) and the cassava industry, or some other use in the rural setting.
In this study, the BMP of sawdust was analyzed. The results show that the BMP is significantly lower for sawdust compared with cassava peels and fibers. Therefore, it would be more efficient to use another technique to turn this bio-waste into energy that could benefit local industries and households in rural Mozambique. Both pyrolysis and gasification are techniques that could be investigated.