Abstract
The article deals with improving the sewage systems of small remote settlements in the Arctic climate to prevent industrial pollution of the environment. Preliminary monitoring of the wastewater temperature and quality was carried out. From the laboratory results of the experimental modelling, such as the coagulation, sedimentation, filtration, oxidation, and wastewater chemical precipitation, we developed a substitute for the traditional biological stage of sewage treatment with physicochemical methods. A real sewage water sample, taken for this study from a septic tank in a remote northern village, can be considered as a distinctive feature of the experiment. The best results obtained according to the scheme with the chemical precipitation of nitrogen compounds are demonstrated. In this case, the chemical oxygen demand (COD) index was reduced by 96.6%, the ammonium ions concentration by 99.5%, and the suspended solids concentration by 99.9%. A technological scheme for low-productivity wastewater treatment plants (WWTPs) in northern settlements is proposed. A comparison of the proposed scheme and the traditional schemes in terms of efficiency and cost was carried out. Further study and implementation of recommended methods of sewage treatment at existing and newly designed stations is expected to improve the effective operation of these treatment plants in arctic conditions.
1. Introduction
A functional sewage system is pivotal for the health of the population. The refinement of small northern settlements is an urgent strategic direction for the development of the Russian Arctic. Currently, more than 60% of the settlements in the Yamalo-Nenets Autonomous District (YNAD) either do not have their own sewage treatment facilities (wastewater treatment plants (WWTPs)) or have very low-efficiency plants that do not treat wastewater to the required standards. A distinctive feature of the small northern settlements that affects the quality of wastewater treatment is a decentralized sewage system. The wastewater is accumulated in septic tanks. Then, it is pumped out by sewer trucks and transported to a WWTP [1,2].
Sewage stations widely used for domestic wastewater treatment apply biological treatment as the main method [3], which is based on the vital activity of microorganisms (activated sludge and biofilms), for which organic wastewater pollution is a nutrient [4].
The tendency of unsatisfactory performance of biological treatment can be found not only in the territory of the Russian Federation but also in the territory of foreign countries, such as Greenland [5], the Canadian Arctic [6,7], the state of Alaska (USA) [8], Northern Norway, Finland, and others. Research revealed that the concentration of pollutants in the conditions of a decentralized wastewater disposal system was two to three times higher in the obtained water compared with in samples from a centralized sewage in other regions [9].
Severe climatic conditions, remoteness, the inaccessibility of small settlements, a lack of the required resources and communication routes, a shortage of qualified personnel, unequal inflow of sewage waters, and high pollutant concentrations were the main reasons for low WWTP efficiency. As shown in [2], small standard sewage biotreatment plants (with a capacity of 10–250 m3/day) can reduce the concentrations of suspended solids by 80–95%, of organic matter by 60–85%, and of ammonia nitrogen by only 40–70%. The lower the capacity of the station, the more difficult it is to achieve the required cleaning effect. In addition, high-energy expenses and operational difficulties accompany biological wastewater treatment in arctic conditions [3,7].
All the above-mentioned problems have spurred research on alternative physicochemical methods that could partly or fully replace the traditional stage of biological treatment on WWTP, providing the required cleaning effects on organic contaminants (biochemical oxygen demand BOD, COD) and nutrients (nitrogen compounds).
For instance, as a potential solution to this problem, a bioelectrochemical anaerobic reactor, which allows a high level of wastewater treatment results at any temperature [10], and at the same time obtaining biogas, was developed in Canada [11].
In Greenland and Denmark, the use of preliminary coagulation is considered [12], as well as the use of oxidizing agents (for example, peracetic acid) in the disinfection of wastewater [13].
In the state of Alaska (USA), it was suggested to implement a closed water supply cycle with wastewater reuse for irrigation and domestic purposes. Such a scheme includes primary treatment, a membrane bioreactor, electrocoagulation, physicochemical oxidation, and filtration [14]. This scheme was able to significantly reduce the COD-index to 0.7 mg/L. This scheme proved to be quite competitive. The cost of its implementation was $ 3 per 1 m3 of water, which was only 13% higher than traditional schemes [15].
One of the widely studied methods of ‘non-biological’ removal of organic substances are the oxidative methods, both as by separate reagents (e.g., active chlorine), and by a combination of different methods: photocatalytic processes [16], Fenton’s process (a mixture of ferrous salt and hydrogen peroxide at pH 2.8–4) [17,18,19], the “Peroxone” process (ozonation in the presence of hydrogen peroxide) [20,21], ozonation in the presence of UV with addition of oxidants and various catalysts [22], the “Carbozone” process (ozonation + activated carbon) [23], the “Sonozon” process (ultrasound + ozone) [24], wet oxidation (WO), wet air oxidation (WAO) and supercritical water oxidation (SCWO) [25,26,27]. The efficiency of organic matter reduction by such methods can reach about 80–99%. However, these processes are accompanied by two main problems: corrosion and the formation of poorly soluble deposits in the reactor [25,26,27].
Many schemes using electrocoagulation and an electrolyzer have been proposed, with the advantages of a low environmental impact, flexibility of use, safety, selectivity, and higher profitability [28,29,30,31]. However, the purification effects for COD did not exceed 70%, and the ammonia nitrogen indices were no more than 50% [31].
Over the past few decades, Russian scientists have also explored the possibility of using physicochemical stations instead of biological ones for small settlements [32,33,34,35]. Methods of sedimentation, coagulation, and the chemical destruction of organic contaminants are used as the main stages in these schemes. Despite the fact that all physicochemical methods are devoid of the disadvantages of biological treatment, such as sensitivity to fluctuations in concentrations and low temperatures, and the need for a uniform supply of wastewater, widespread implementation of the above methods is problematic. This is due to the corrosion of pipelines and tanks, the requirement for determination of the oxidation parameters and doses of reagents in each case, large size, and relatively low treatment efficiency. It is possible to reduce the COD load by no more than 90% and for ammonium nitrogen, the possible decrease is no more than 70% [32].
The aim of this study was to determine the effectiveness of replacement of the biological treatment stage by physicochemical methods at small-sized WWTPs in arctic conditions. The object of the study was domestic wastewater samples taken from septic tanks in a remote settlement in the YNAD (Arctic zone of Russia). The use of real wastewater in the experiment helped to consider the influence of all components and to bring the expected results closer to real practical situations. The subject of the research was the change in characteristic indicators of the wastewater quality (pH, suspended solids—SS, ammonium ions—NH4+, chemical oxygen demand—COD, and temperature) at all treatment stages in the laboratory modelling process.
Based on the experimental work presented, we propose a technological scheme for sewage treatment plants. We conducted a preliminary feasibility analysis of the new scheme.
2. Materials and Methods
2.1. Preliminary Study of the Initial Wastewater Temperature and Quality
The temperature and quality of wastewater in the sewage system of small northern settlements were monitored for a number of years (2010–2015, Industrial University of Tyumen). The optimum water temperature is one of the main factors affecting the biological oxidation intensity. An initial wastewater temperature of at least 16 degrees Celsius is essential for normal biological treatment processes [32,33,34,35]. Research proved that, in aerotanks, bio-oxidation occurs most actively at a water temperature of 20–30 degrees Celsius and above [4].
The results of long-term observations showed that the wastewater temperature in remote northern settlements does not meet the requirements for more than 9 months a year. The water temperature in the small-size sewage systems did not exceed 13 degrees from April to May. Similar results were obtained in September and October. In winter, the temperature of wastewater entering the treatment station from sewage systems was 6–7 degrees Celsius. The temperature of water in the septic tank was 2–4 degrees Celsius in the cold season. This is one of the main reasons for unsatisfactory biological wastewater treatment [2,6,8,9,32].
Wastewater quality analysis was carried out on samples taken from septic tanks, in which the concentrations of the pollutants were averaged. The study was based on commonly accepted standard indicators for domestic wastewater. Comparing the obtained results (2014) with the standard ones, the concentrations in septic tanks clearly exceeded the permissible standards: 3.6 times for suspended solids, 2.7 times for BOD, 2.4 times for COD, and four times for ammonium ions. The anionic surfactants, petroleum products, and other quality indices were normal (Supplementary Materials).
In 2020, wastewater samples were taken from septic tanks of a small settlement located in the territory of the Yamalo-Nenets Autonomous District in permafrost conditions. On the basis of a preliminary study, the optimal water temperature was determined to be not higher than 5 degrees Celsius, and the four most problematic indicators (pH, suspended solids, COD, and ammonium ions) were selected, according to which, laboratory control and monitoring of wastewater treatment processes were carried out.
The initial composition of the wastewater samples, the average values, and measurement errors are presented in Table 1.

Table 1.
Initial wastewater composition (2020).
The study shows that the quality of initial wastewater samples corresponded to the typical quality of wastewater from septic tanks in remote northern settlements. The following stages of physicochemical wastewater treatment were considered: the first stage was reagent coagulation, the second stage was chemical oxidation of the organic matters, and the third stage was tertiary treatment from the ammonium ions.
2.2. Sampling and Standards
Wastewater samples, planned for testing at the physicochemical stage of treatment, were taken from septic tanks into five-liter bottles to be further delivered to the laboratory in a thermostat to maintain the temperature conditions. In these tanks, the concentration of pollutants was averaged for 1–3 days.
The characteristic indicators of the wastewater quality (pH, suspended solids—SS, ammonium ions—NH4+, chemical oxygen demand—COD, and temperature) at all treatment stages in the laboratory modelling process were identified. These indicators are the most difficult to extract from domestic wastewater using reagents. Standard methods and modern devices to determine the water quality indicators were used (Table 2).

Table 2.
Standard methods and devices.
The error rate of the presented results did not exceed 15%, which is normal for the standards.
The pH-index was determined using the potentiometric method: 30 cm3 of the test sample was taken into a measuring beaker. Then, the pH-meter electrode was immersed into the sample.
The concentration of suspended solids was determined by the gravimetric method by measuring the mass of solids remaining on the paper filter after drying at a temperature of 105 degrees Celsius to constant mass, in the volume of the sample taken for analysis.
COD determination was carried out photometrically. Glass vials were filled with 3 cm3 of a mixture of potassium dichromate K2Cr2O7 and silver sulfate Ag2SO4 dissolved in sulfuric acid H2SO4, 0.2 cm3 of the catalyst of mercury sulfate HgSO4, and 2 cm3 of the test sample. After that, the contents of the vial were maintained for two hours at a temperature of 150 degrees Celsius in a Thermion thermoreactor, and then, after cooling, they were photometrically measured on a Fluorat-0.2 M (liquid analyzer).
The concentration of ammonium ions (NH4+) was determined photometrically on a PE 5400VI spectrophotometer. To determine the concentration of ammonium ions, 1 cm3 of potassium-sodium tartrate KNaC4H4O6·4H2O and Nessler’s reagent K2[HgI4]·2H2O was added to 50 cm3 of the sample. Then, the sample was stirred, and, after 10 min, the concentration of NH4+ was measured in cuvettes with an absorbing layer thickness of 50 mm at a wavelength of 425 mm.
2.3. First Stage of Modeling Treatment—Reagent Coagulation
At the first stage, which is the reagent coagulation, a reagent wastewater treatment was performed to coagulate the suspended solids and part of the organic matters. Municipal wastewater from a septic tank was tested for the effectiveness of reagent coagulation using several coagulants: aluminum sulfate Al2(SO4)·18H2O (SA), aluminum oxychloride Al2(OH)5Cl (OA), aluminum polyoxychloride—Aqua-Aurat-30 (AA), ferric chloride FeCl3, and ferrous sulfate FeSO4. The initial wastewater was placed in cylinders with a volume of 500 cm3, into which various doses of coagulants were added, starting from 30 mg/L and gradually escalated to 50, 90, and 200 mg/L.
A dose of the coagulant was visually selected starting from 30 mg/L and gradually escalated to the meanings of 50, 90 and 200 mg/L. A mixture of sample water and reagents was intensively stirred with a glass rod for 2 min. Within 20 min, precipitation of the formed flakes occurred, and, at that instant, the sample was filtered through a sand filter (layer: h = 10 cm). Quartz sand with a fraction of 0.8–2 mm was used, and the filtration rate was 5 m/h. The experiments were carried out at a temperature of 4–5 degrees Celsius, which corresponds to the temperature of real wastewater in Arctic regions.
2.4. Second Stage of Modeling Treatment—Chemical Oxidation
At the second stage, the water samples were subjected to deep chemical oxidation. Various oxidants were tested: potassium permanganate (KMnO4) and Anolyte. Samples of wastewater after coagulation and filtration were placed in a measuring beaker with a volume of 500 cm3, into which various doses of oxidizing reagents (from 10 to 50 mg/L) were introduced. Mechanical filtration of the effluent was carried out on a sand filter (layer: h = 10 cm) after oxidation. The doses of the oxidizing reagents ranged from 10 to 50 mg/L. The time of wastewater contact with the oxidizing agent in all cases was 20 min. The chemical oxidation was carried out in a thermostat at a temperature of 5 degrees Celsius.
2.5. Third Stage of Modeling Treatment—Chemical Precipitation
In the third phase, the laboratory studies included the chemical precipitation of the nitrogen compounds [36]. This method was chosen as an option for reducing the concentration of ammonium ions. Tertiary wastewater treatment was accomplished by adding phosphate ions and magnesium ions into the water, followed by the sedimentation of magnesium-ammonium-phosphate in an alkaline medium (pH ≈ 9) according to the following mechanism:
MgCl2 + NH4Cl + Na2HPO4 + NaOH + 5H2O = MgNH4PO4∙6H2O↓ + 3NaCl
The reagents were fed in the molar ratio Mg: MgNH4PO4 to obtain magnesium-ammonium-phosphate MgNH4PO4∙6H2O. Chemical precipitation of ammonium ions was carried out in measuring beakers with a volume of 500 cm3, into which the reagents were added in a molar ratio according to the reaction equation. After mixing the reagents with water with a glass rod, the water was placed in a thermostat for settling during 10 min.
After chemical precipitation, the water was filtered through a layer (h = 10 cm) of activated carbon (sorption filtration). All laboratory measurements were carried out in three parallel experiments to determine the average values of the indicator and the errors of the direct and indirect measurements. Figure 1 shows the scheme of the process order. The laboratory modeling results of the processes of wastewater treatment are presented in Section 3.1, Section 3.2 and Section 3.3.
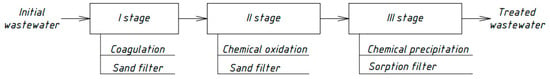
Figure 1.
Scheme of the laboratory modeling processes.
3. Results
3.1. Study of the Wastewater Quality after the First Stage of Treatment
At the first stage, coagulation of suspended solids with coagulants was performed. The best result among aluminum-based coagulants was shown by aluminum polyoxychloride—Aqua-Aurat-30 (AA). An active process of coagulation at a dose of 50 mg/L was observed. To control the quality of processing, the following indicators were selected: pH-index, suspended solids (SS), and chemical oxygen demand (COD) (Table 3). Each indicator was measured at least three times. After that, the average value and measurement error were determined.

Table 3.
The initial wastewater parameters and the course and results of the laboratory studies.
The use of iron-containing coagulants (ferric chloride FeCl3 and ferrous sulfate FeSO4) at doses of 50 and 90 mg/L presented no visible flakes for observations during a 20-min treatment. With the addition of 120 mg/L of ferric chloride, stable flocs began to form; to form stable flocs while using ferrous sulfate, 120 mg/L of coagulant was added, and the sample was simultaneously acidified to pH = 4.74.
Further study of the coagulation process with the use of iron-containing coagulants was not continued due to economic expediency. Table 3 shows the course and results of the laboratory studies.
Based on the obtained results, we determined that the best COD purification effect was achieved with the use of Aqua-Aurat 30 (the optimal dose was 50 mg/L), and, at the same time, the COD reduction effect was 64.3%. The concentration of the suspended solids decreased by about 96%. However, ammonium ions were not practically removed from the wastewater. After the reagent coagulation stage, it is necessary to settle and filter the samples in order to remove the reaction products from the water.
A flocculant PAA (polyacrylamide, a dose of 1 mg/L) inclusion contributed to an additional decrease by about 3–6% in COD, by about 70–75% in the concentration of nitrates, and by 96.5–98% in suspended solids.
Thus, at the first stage of reagent coagulation (with 50 mg/L of Aqua-Aurat 30 and 1 mg/L of polyacrylamide), it was possible to reduce the concentrations of suspended solids by 96.9% and the COD by 70.1%.
3.2. Study of the Wastewater Quality after the Second Treatment Stage
At the second stage, the water samples were subjected to deep chemical oxidation. Two oxidants were tested: potassium permanganate (KMnO4) and Anolyte. Table 4 shows the best experimental results of the chemical oxidation method on the wastewater samples after coagulation by reagents. To control the wastewater treatment quality, the following indicators were selected: pH-index, suspended solids (SS), chemical oxygen demand (COD), and ammonium ions (NH4+) (Table 4). Each indicator was measured at least three times. After that, the average value and measurement error were determined.

Table 4.
The initial wastewater parameters and the course and results of the laboratory oxidation studies (2020).
Potassium permanganate (10 mg/L) proved to be the most effective oxidizing agent at 20 min contact. The decrease in the COD with this oxidation method, along with a subsequent mechanical filtration, was 91.4%. The concentration of suspended solids in the purified water was 2.1 mg/L, and the water treatment effect was 99.7%. A maximal decrease of the concentration of ammonium ions with the selected oxidation method was 67.7%. After such an oxidation, the pH-index was 7.93. The only limitation for the wide scale of this oxidizer reagent is that it is a precursor used in the illicit manufacture of drugs and is subject to careful control when used.
3.3. Study of the Wastewater Quality after the Third Treatment Stage
As a result of the experiment at the third stage (chemical precipitation plus sorption filtration), the total efficiency of removal of ammonium ions from the wastewater was 99.5%. In this case, a transition of pH-index in the alkaline side was observed. The common results of the study on wastewater treatment by physicochemical methods are presented in Table 5.

Table 5.
Research results for physicochemical methods of wastewater treatment (2020).
According to the results of the third stage, organic nitrogen compounds were decreased by 99.5% through chemical precipitation. At the same time, suspended solids were almost completely removed (by 99.9%), and organic pollution in COD was significantly reduced (by 96.6%). The pH index changed from 7.36 to 8.82.
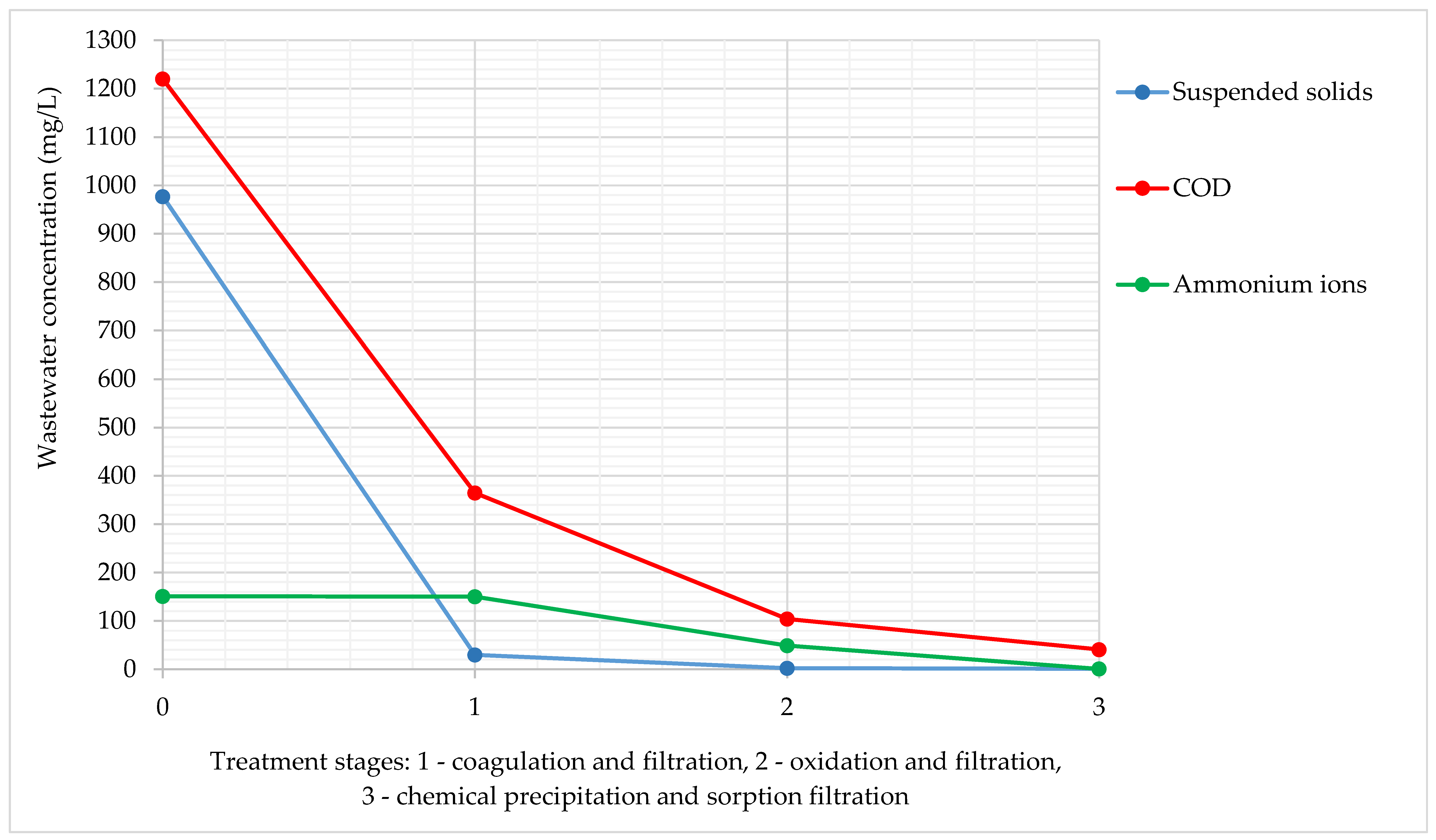
Figure 2 shows the results of the study of wastewater concentration at different physicochemical treatment stages.
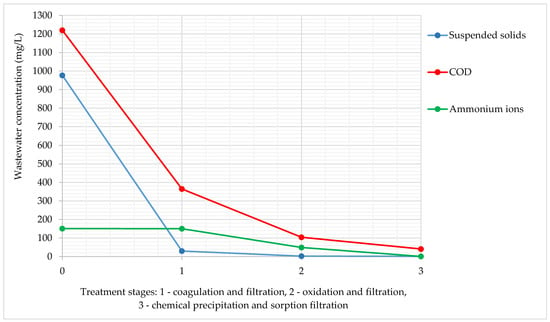
Figure 2.
Changes of wastewater concentration at different treatment stages during experiments in the laboratory.
The suspended solids were practically removed at the first stage of treatment. The COD gradually decreased and reached the required value only after the third stage. The ammonium ion concentration began to decrease only after the second stage, and after the third stage it was fully extracted.
4. Discussion
In the process of the experimental treatment of wastewater samples taken from the sewage system in the remote northern settlement, it was possible to achieve good quality after three stages of physicochemical treatment. The wastewater temperature did not exceed 5 degrees Celsius. The organic nitrogen compounds were decreased by 99.5%, the suspended solids were almost completely removed (by 99.9%), and the COD value was significantly reduced (by 96.6%). The achieved results allow replacing traditional biological treatment at the WWTPs with physical-chemical treatments (processes of coagulation, oxidation, and chemical precipitation).
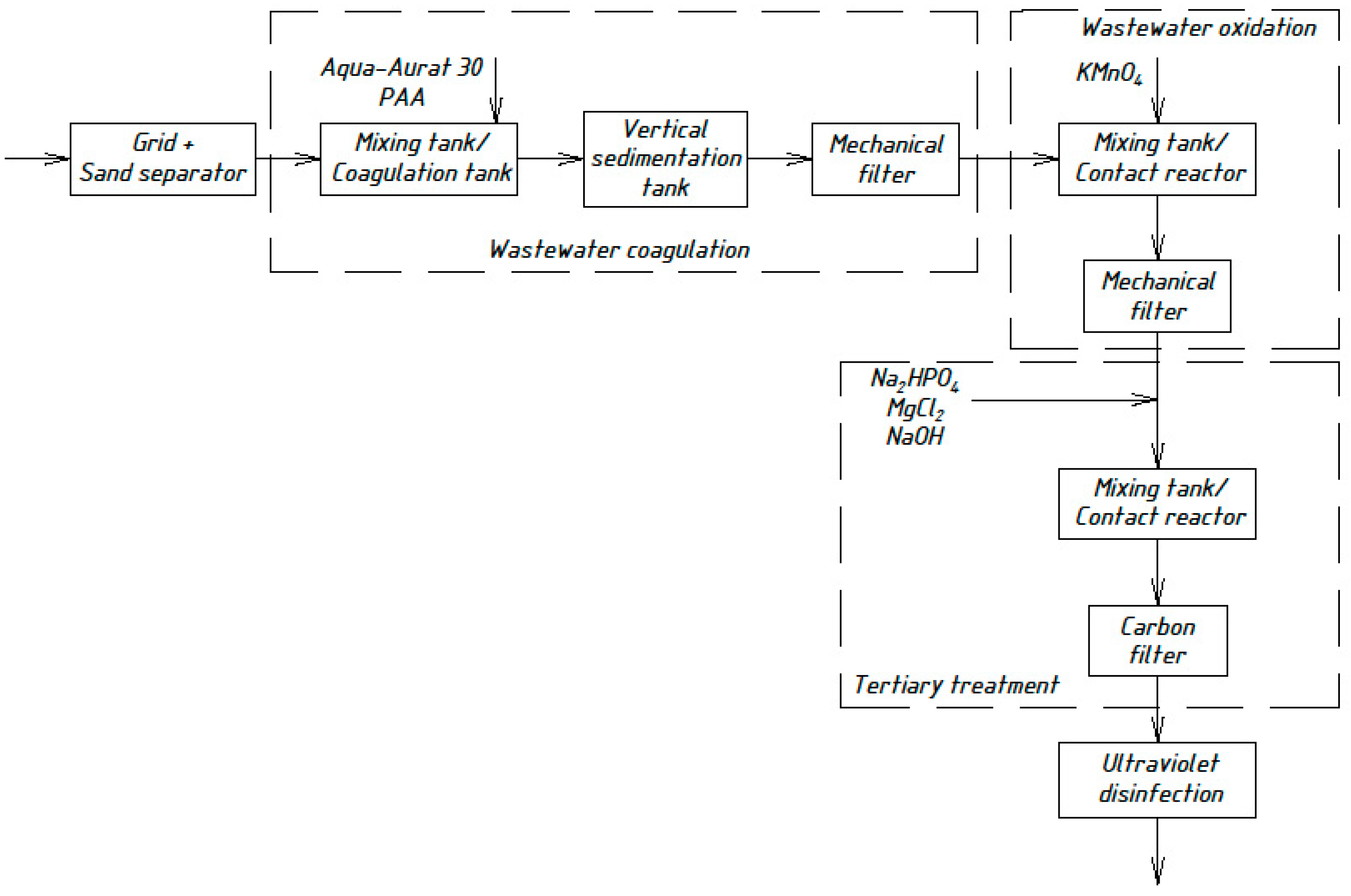
Based on the experimental work presented, a technological scheme for sewage treatment plants with a capacity of up to 1000 m3 per day is proposed (Figure 3).
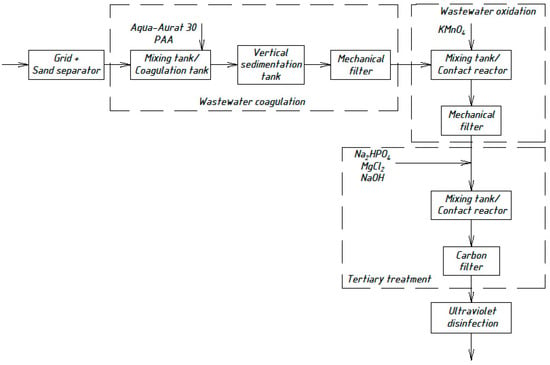
Figure 3.
Wastewater treatment scheme with the chemical precipitation of organic nitrogen compounds.
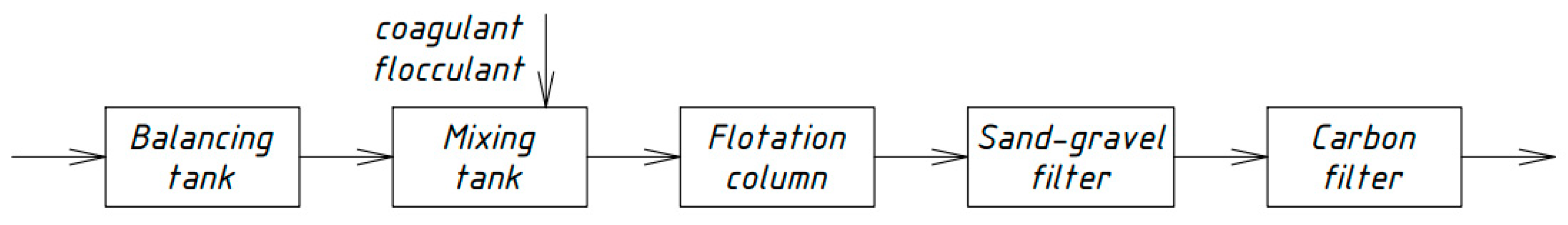
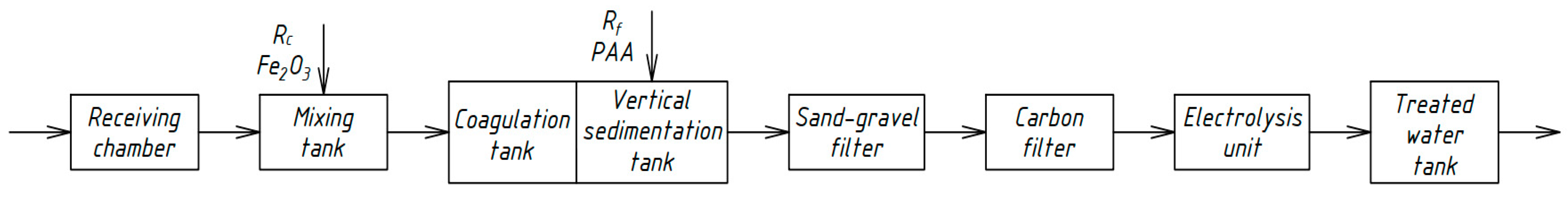
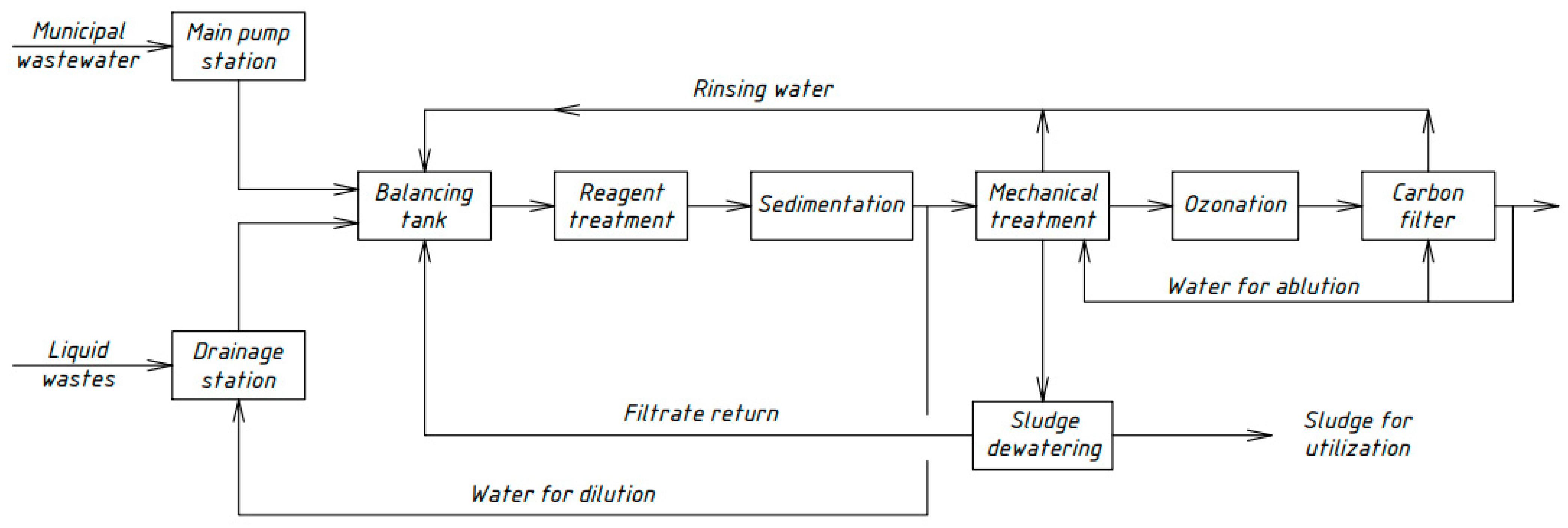
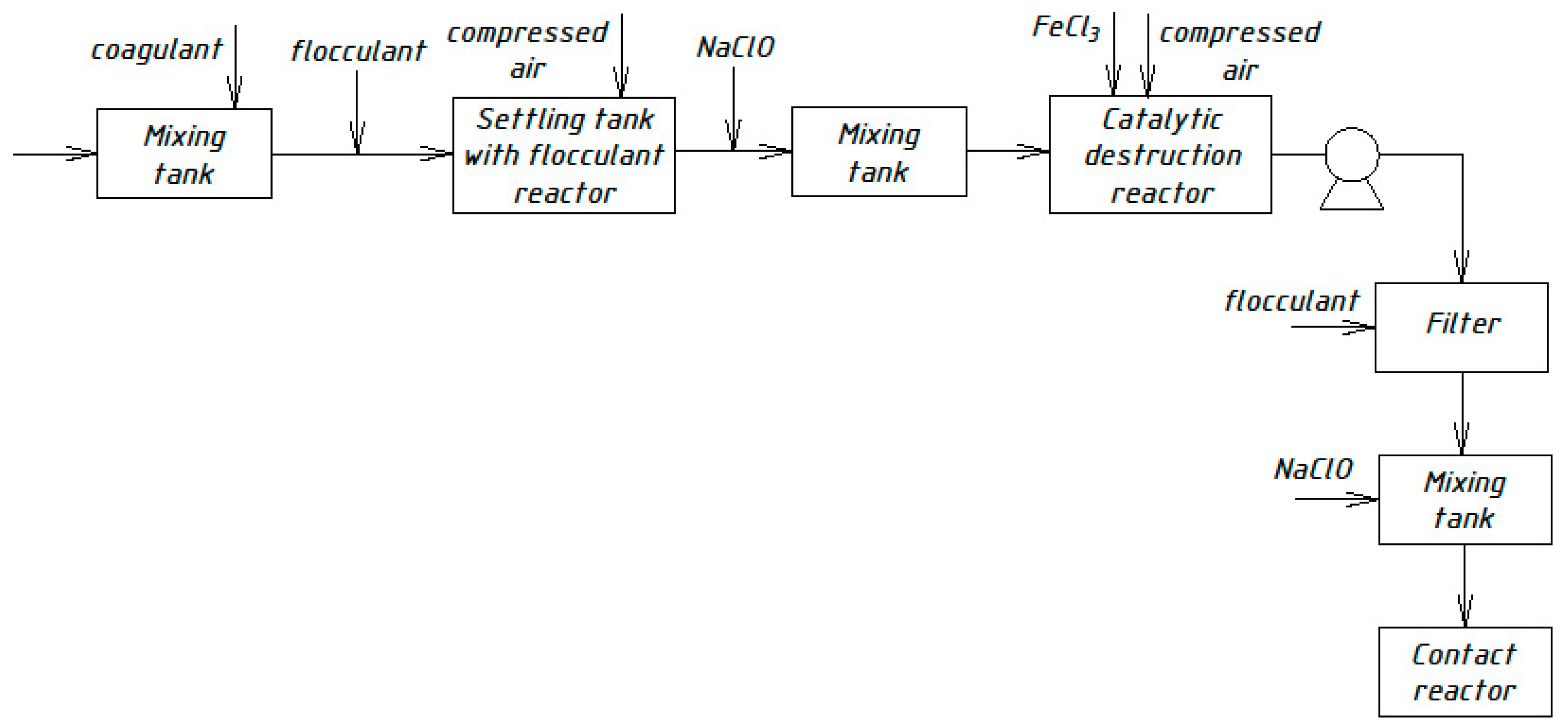
Similar schemes for the physicochemical treatment of domestic wastewater [33,34,35] were developed by Russian scientists in different periods. Figure 4, Figure 5, Figure 6 and Figure 7 show the technological schemes of physical-chemical wastewater treatment based on the data of Russian authors [33,34,35].
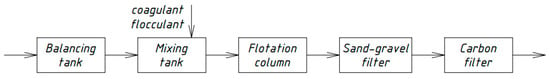
Figure 4.
Wastewater treatment scheme with a flotation column.
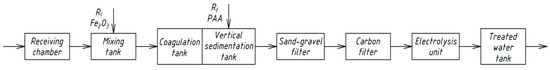
Figure 5.
Wastewater treatment scheme with an ozonation unit.
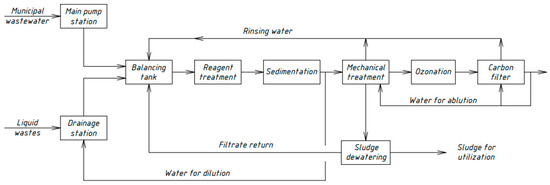
Figure 6.
Wastewater treatment scheme with an ozonation unit.
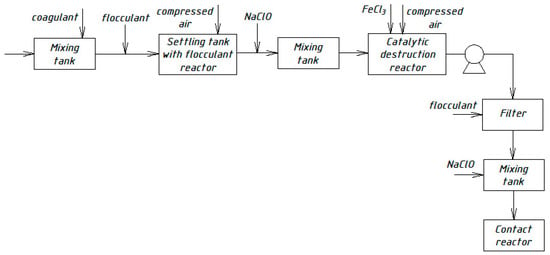
Figure 7.
Wastewater treatment scheme with a catalytic destruction reactor.
All previously proposed schemes for the physicochemical treatment of wastewater (Figure 4, Figure 5, Figure 6 and Figure 7) were based on the main stages: coagulation for the almost complete removal of suspended solids, the destruction of organic substances through the oxidation process, and intermediate stages of filtration and settling. In all these options, it is almost completely possible to achieve a decrease in the concentration of suspended solids—at least 99%.
The most common disadvantage of these schemes (Figure 4, Figure 5, Figure 6 and Figure 7) is low treatment effects for organic pollutants. The purification effects for COD do not exceed 90% compared with the required 95–98%, and ammonia nitrogen indices are no more than 70%, while at least 95% is required [33,34,35]. The difference in the decrease in COD concentrations between the schemes in Figure 4, Figure 5, Figure 6 and Figure 7 and the scheme in Figure 3 may be explained by the use of various reagents, as well as methods of destruction of organics. At the same time, the authors managed to achieve a greater decrease in COD (by 96.6%) in laboratory studies due to the use of a stronger oxidant KMnO4.
It was possible to reduce ammonium nitrogen by 99.5% due only to two-stage treatment (oxidation and chemical precipitation), while only oxidation reduced the concentration of ammonium ions by only 67.7%, which was not sufficient to achieve the required effects. Despite all the advantages and disadvantages of the physicochemical wastewater treatment schemes proposed by the Russians, they all require experimental verification and improvement, especially when implemented in severe conditions.
The technological scheme (Figure 3) is quite suitable for further improvement, detailed study, and practical implementation in real WWTPs in the Arctic. A feasibility study and comparison of the options in terms of energy and resources savings are required. Despite the fact that the new scheme is more complex, it is more efficient.
A treatment plant (WWTP) with a capacity of 250 m3 per day was selected for a preliminary comparison of the technical and economic feasibility. Two variants of the WWTP technological scheme were considered: (1) biological treatment (BIO) and (2) physicochemical treatment (PC). The biological treatment plant includes a preliminary treatment unit, a primary sedimentation tank, an aeration tank–nitrifier–denitrifier, a membrane sludge separator, a tertiary-treatment filter, and an installation for UV disinfection.
The design for the physicochemical treatment plant was adopted according to the above scheme (Figure 3). The results of the preliminary calculations (2020), which exclude the costs of sludge disposal, showed that capital investments for manufacturing, transportation, and construction, and installation works in the Yamalo-Nenets Autonomous District amounted to €1,144,000 and €625,000 for biological and physicochemical stations, respectively.
Operating expenses were €204,000 and €136,000 for biological and physicochemical stations, respectively. Those costs take into account the price of maintenance, electricity, fuel, reagents, and others. The cost of treating one cubic meter of wastewater was €2.24 in the case of biological treatment, compared to €1.06 for physicochemical treatment. According to preliminary calculations, physicochemical methods of wastewater treatment are more profitable in the conditions of a remote village in Arctic climate.
It is necessary to make a forecast for the formation of secondary waste to determine the hazard class, to calculate the volumes obtained, and to propose the methods for their processing and disposal. According to preliminary calculations, while implementing this water purification scheme, the amount of formed sediments, with the account of reagents, is 1.4–2% of the total wastewater volume.
In this case, the moisture content of the primary sediment was 90–93%. After the process of mechanical dehydration to a moisture content of 70%, the volume of precipitation will be reduced by five to six times. The sediment has good moisture-releasing properties. Periodically exporting the sediments to the city of Salekhard (YNAD, Russia) for incineration in a special furnace is planned.
5. Conclusions
To summarize, based on a preliminary study of quality and temperature, we proposed a solution for an important problem regarding physicochemical wastewater treatment at WWTPs in small remote northern settlements, laboratory experimental modeling of the domestic wastewater treatment stages was carried out, and a technological scheme (Figure 3) was suggested.
In the course of the research, the stages of coagulation, filtration, oxidation, and chemical precipitation of wastewater pollution were studied. During the experiment, various conditions of observation, combinations of stages, and reagents were applied. The treatment efficiency was tested by the significant characteristics such as the pH, chemical oxygen demand (COD), concentration of suspended solids (SS), and ammonium ions (NH4+).
At the first stage, the best results of reducing the COD and suspended solids were obtained using the aluminum polyoxychloride Aqua-Aurat 30 at a dose of 50 mg/L in combination with a flocculant PAA at a dose of 1 mg/L. The best wastewater treatment effects were determined as 96% for SS and 64.3% for COD.
In the process of wastewater oxidation at the second stage, where the samples underwent preliminary sedimentation and coagulation, potassium permanganate at a dose of 10 mg/L turned out to be the most effective oxidizing agent. In this case, the concentration of COD decreased by 91.4%, NH4+ by 67.7%, and SS by 99.7%.
At the third stage of the chemical precipitation method of treatment, it was possible to reduce the concentration of NH4+ by 99.5%, SS by 99.9%, and COD by 96.6%. The temperature of the wastewater samples did not affect the processes significantly.
As a result, during the realization of the physicochemical treatment scheme (Figure 3) in laboratory conditions, it was possible to achieve much better effects of wastewater treatment than in the previously proposed schemes (Figure 4, Figure 5, Figure 6 and Figure 7). The preliminary economic calculations carried out by the authors showed that, when implementing the new scheme, the cost of the water purification process decreased by 2.1 times in comparison with the traditional biological treatment.
In the future, we plan to study the changes in water quality using other indicators, including phosphates, sulfates, chlorides, synthetic surfactants, petroleum products, and metal ions. In addition, studies on the residual products of chemical reactions are needed. Microbiological indicators according to the list of Russian standards will be suitable for monitoring after ultraviolet treatment. It is also necessary to analyze the secondary waste and determine the hazard class. The next stage is modelling of the technological equipment and design of a WWTP that is suitable for efficient operation in the Arctic.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/w13070919/s1, Figure S1: Efficiency of wastewater treatment at a biological WWTP depending on capacity (2010–2015), Figure S2: Seasonal variations of temperature in the initial wastewater according to the WWTP capacity (2014–2015), Figure S3: Diurnal water temperature variations at different treatment stages for WWTPs with a capacity of 20 m3 per day (2014), Figure S4: Seasonal changes of average monthly water temperature at different treatment stages for WWTPs with a capacity of 20 m3 per day (2014), Table S1: The study results of wastewater quality in the septic tanks (2014).
Author Contributions
Conceptualization, E.V.; methodology, E.V. and E.G.; formal analysis, E.V.; investigations, E.G. and E.V.; data curation, E.V. and E.G.; writing—original draft preparation, E.V. and E.G.; writing—review and editing, E.V. and E.G.; visualization, E.V. and E.G.; supervision, E.V.; project administration, E.V.; funding acquisition, E.V. All authors have read and agreed to the published version of the manuscript.
Funding
The article is published with the grant support of the Industrial University of Tyumen (Contract No. 4176/NPK, 10 December 2020).
Institutional Review Board Statement
The study did not involve humans or animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
Authors acknowledge any administrative and technical support given by Kropcheva S.N.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Glushchenko, E.; Vialkova, E.; Sidorenko, O.; Fugaeva, A. Physical-Chemical Wastewater Treatment in Arctic Conditions. E3S Web Conf. 2020, 157, 02014. [Google Scholar] [CrossRef]
- Vialkova, E.; Maksimova, S.; Zemlyanova, M.; Maksimov, L.; Vorotnikova, A. Integrated Design Approach to Small Sewage Systems in the Arctic Climate. Environ. Process. 2020, 7, 673–690. [Google Scholar] [CrossRef]
- Preisner, M. Surface Water Pollution by Untreated Municipal Wastewater Discharge Due to a Sewer Failure. Environ. Process. 2020, 7, 767–780. [Google Scholar] [CrossRef]
- Gerardi, M.H. Nitrification and Denitrification in the Activated Sludge Process; Wastewater Microbiology Series; Wiley-Interscience: New York, NY, USA, 2002; ISBN 9780471065081. [Google Scholar]
- Hendriksen, K.; Hoffmann, B. Greenlandic Water and Sanitation Systems—Identifying System Constellation and Challenges. Environ. Sci. Pollut. Res. 2018, 25, 32964–32974. [Google Scholar] [CrossRef]
- Daley, K.; Jamieson, R.; Rainham, D.; Truelstrup Hansen, L. Wastewater Treatment and Public Health in Nunavut: A Microbial Risk Assessment Framework for the Canadian Arctic. Environ. Sci. Pollut. Res. 2018, 25, 32860–32872. [Google Scholar] [CrossRef]
- Ragush, C.M.; Schmidt, J.J.; Krkosek, W.H.; Gagnon, G.A.; Truelstrup-Hansen, L.; Jamieson, R.C. Performance of Municipal Waste Stabilization Ponds in the Canadian Arctic. Ecol. Eng. 2015, 83, 413–421. [Google Scholar] [CrossRef]
- Hickel, K.A.; Dotson, A.; Thomas, T.K.; Heavener, M.; Hébert, J.; Warren, J.A. The Search for an Alternative to Piped Water and Sewer Systems in the Alaskan Arctic. Environ. Sci. Pollut. Res. 2018, 25, 32873–32880. [Google Scholar] [CrossRef] [PubMed]
- Kallenborn, R.; Brorström-Lundén, E.; Reiersen, L.-O.; Wilson, S. Pharmaceuticals and Personal Care Products (PPCPs) in Arctic Environments: Indicator Contaminants for Assessing Local and Remote Anthropogenic Sources in a Pristine Ecosystem in Change. Environ. Sci. Pollut. Res. 2018, 25, 33001–33013. [Google Scholar] [CrossRef] [PubMed]
- LaBarge, N.; Yilmazel, Y.D.; Hong, P.-Y.; Logan, B.E. Effect of Pre-Acclimation of Granular Activated Carbon on Microbial Electrolysis Cell Startup and Performance. Bioelectrochemistry 2017, 113, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Tartakovsky, B.; Kleiner, Y.; Manuel, M.-F. Bioelectrochemical Anaerobic Sewage Treatment Technology for Arctic Communities. Environ. Sci. Pollut. Res. 2018, 25, 32844–32850. [Google Scholar] [CrossRef] [PubMed]
- Koivunen, J.; Heinonen-Tanski, H. Peracetic Acid (PAA) Disinfection of Primary, Secondary and Tertiary Treated Municipal Wastewaters. Water Res. 2005, 39, 4445–4453. [Google Scholar] [CrossRef] [PubMed]
- Chhetri, R.K.; Klupsch, E.; Andersen, H.R.; Jensen, P.E. Treatment of Arctic Wastewater by Chemical Coagulation, UV and Peracetic Acid Disinfection. Environ. Sci. Pollut. Res. 2018, 25, 32851–32859. [Google Scholar] [CrossRef]
- Wu, T.; Englehardt, J.D. Mineralizing Urban Net-Zero Water Treatment: Field Experience for Energy-Positive Water Management. Water Res. 2016, 106, 352–363. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Englehardt, J.D.; Guo, T.; Gassie, L.; Dotson, A. Applicability of Energy-Positive Net-Zero Water Management in Alaska: Technology Status and Case Study. Environ. Sci. Pollut. Res. 2018, 25, 33025–33037. [Google Scholar] [CrossRef]
- Chong, M.N.; Jin, B.; Chow, C.W.K.; Saint, C. Recent Developments in Photocatalytic Water Treatment Technology: A Review. Water Res. 2010, 44, 2997–3027. [Google Scholar] [CrossRef]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-Fenton Process and Related Electrochemical Technologies Based on Fenton’s Reaction Chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Dirany, A.; Sirés, I.; Oturan, N.; Oturan, M.A. Electrochemical Abatement of the Antibiotic Sulfamethoxazole from Water. Chemosphere 2010, 81, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Neafsey, K.; Zeng, X.; Lemley, A.T. Degradation of Sulfonamides in Aqueous Solution by Membrane Anodic Fenton Treatment. J. Agric. Food Chem. 2010, 58, 1068–1076. [Google Scholar] [CrossRef]
- Pisarenko, A.N.; Stanford, B.D.; Yan, D.; Gerrity, D.; Snyder, S.A. Effects of Ozone and Ozone/Peroxide on Trace Organic Contaminants and NDMA in Drinking Water and Water Reuse Applications. Water Res. 2012, 46, 316–326. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Canonica, S.; von Gunten, U. Efficiency and Energy Requirements for the Transformation of Organic Micropollutants by Ozone, O3/H2O2 and UV/H2O2. Water Res. 2011, 45, 3811–3822. [Google Scholar] [CrossRef] [PubMed]
- Trapido, M. Ozone-Based Advanced Oxidation Processes; Encyclopedia of Life Support Systems; 2008; pp. 1–17. Available online: http://www.eolss.net/sample-chapters/c07/e6-192-07a-00.pdf (accessed on 10 January 2021).
- Sánchez-Polo, M.; von Gunten, U.; Rivera-Utrilla, J. Efficiency of Activated Carbon to Transform Ozone into OH Radicals: Influence of Operational Parameters. Water Res. 2005, 39, 3189–3198. [Google Scholar] [CrossRef]
- Sangave, P.C.; Gogate, P.R.; Pandit, A.B. Ultrasound and Ozone Assisted Biological Degradation of Thermally Pretreated and Anaerobically Pretreated Distillery Wastewater. Chemosphere 2007, 68, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Blöcher, C.; Niewersch, C.; Melin, T. Phosphorus Recovery from Sewage Sludge with a Hybrid Process of Low Pressure Wet Oxidation and Nanofiltration. Water Res. 2012, 46, 2009–2019. [Google Scholar] [CrossRef]
- Loppinet-Serani, A.; Aymonier, C.; Cansell, F. Supercritical Water for Environmental Technologies. J. Chem. Technol. Biotechnol. 2010, 85, 583–589. [Google Scholar] [CrossRef]
- Xu, D.; Wang, S.; Tang, X.; Gong, Y.; Guo, Y.; Wang, Y.; Zhang, J. Design of the First Pilot Scale Plant of China for Supercritical Water Oxidation of Sewage Sludge. Chem. Eng. Res. Des. 2012, 90, 288–297. [Google Scholar] [CrossRef]
- Sirés, I.; Brillas, E. Remediation of Water Pollution Caused by Pharmaceutical Residues Based on Electrochemical Separation and Degradation Technologies: A Review. Environ. Int. 2012, 40, 212–229. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.; Pacheco, M.J.; Ciríaco, L.; Lopes, A. Anodic Oxidation of a Biologically Treated Leachate on a Boron-Doped Diamond Anode. J. Hazard. Mater. 2012, 199–200, 82–87. [Google Scholar] [CrossRef]
- Comninellis, C.; Kapalka, A.; Malato, S.; Parsons, S.A.; Poulios, I.; Mantzavinos, D. Advanced Oxidation Processes for Water Treatment: Advances and Trends for R&D. J. Chem. Technol. Biotechnol. 2008, 83, 769–776. [Google Scholar] [CrossRef]
- Harif, T.; Khai, M.; Adin, A. Electrocoagulation versus Chemical Coagulation: Coagulation/Flocculation Mechanisms and Resulting Floc Characteristics. Water Res. 2012, 46, 3177–3188. [Google Scholar] [CrossRef]
- Mochalov, I.P. Wastewater Treatment and Disinfection for Small Settlements (in Far North Conditions): Monograph, 20th revised ed.; DAR/VODGEO: Moscow, Russia, 2016; 466p. [Google Scholar]
- Trunova, N.A. Wastewater Treatment and Its Reuse in Cotton Industry. Ph.D. Thesis, Engineering Science, MICE, Moscow, Russia, 1984. [Google Scholar]
- Andreev, S.Y.; Isaev, A.M.; Kochergin, A.S. Development and Research of Combined Wastewater Treatment Technology of Small Settlements: Monograph; PGUAS: Penza, Russia, 2015; 120p. [Google Scholar]
- Nebukina, I.A.; Smirnova, N.N.; Rvachev, I.S. The Effect of Organic Compounds on Removal Efficiency of Ammonium Ions from Wastewater by Oxidation Method. Probl. Contemp. Sci. Pract. Vernadsky Univ. 2015, 2, 28–33. [Google Scholar] [CrossRef]
- Lobanov, S.A.; Poilov, V.Z. Treatment of Wastewater to Remove Ammonium Ions by Precipitation. Russ. J. Appl. Chem. 2006, 79, 1473–1477. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).