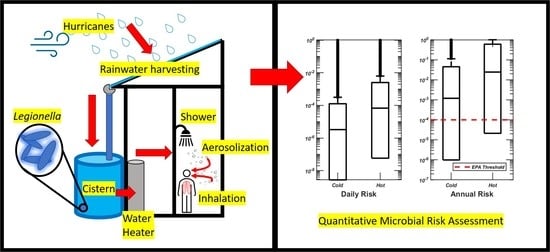
Assessing the Risk of Legionella Infection through Showering with Untreated Rain Cistern Water in a Tropical Environment
Abstract
1. Introduction
2. Materials and Methods
2.1. Household Survey
2.2. Risk Assessment
2.2.1. Hazard Identification
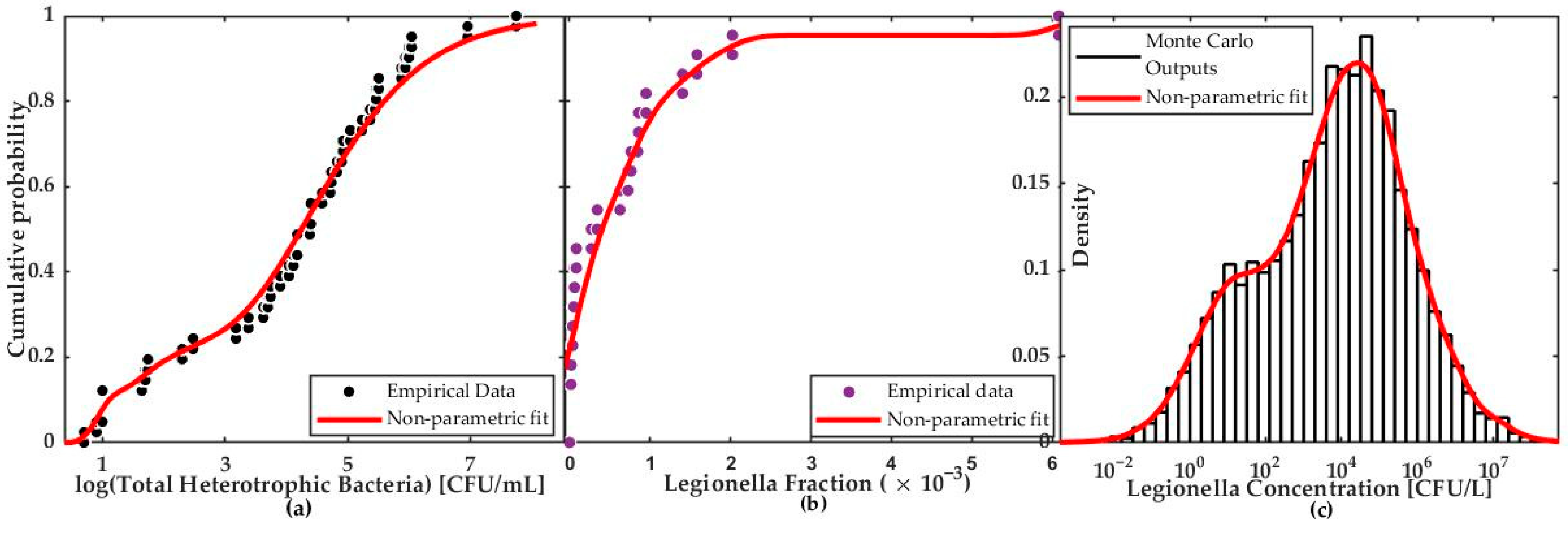
2.2.2. Exposure Assessment
2.2.3. Dose–Response Assessment
2.2.4. Risk Characterization
3. Results
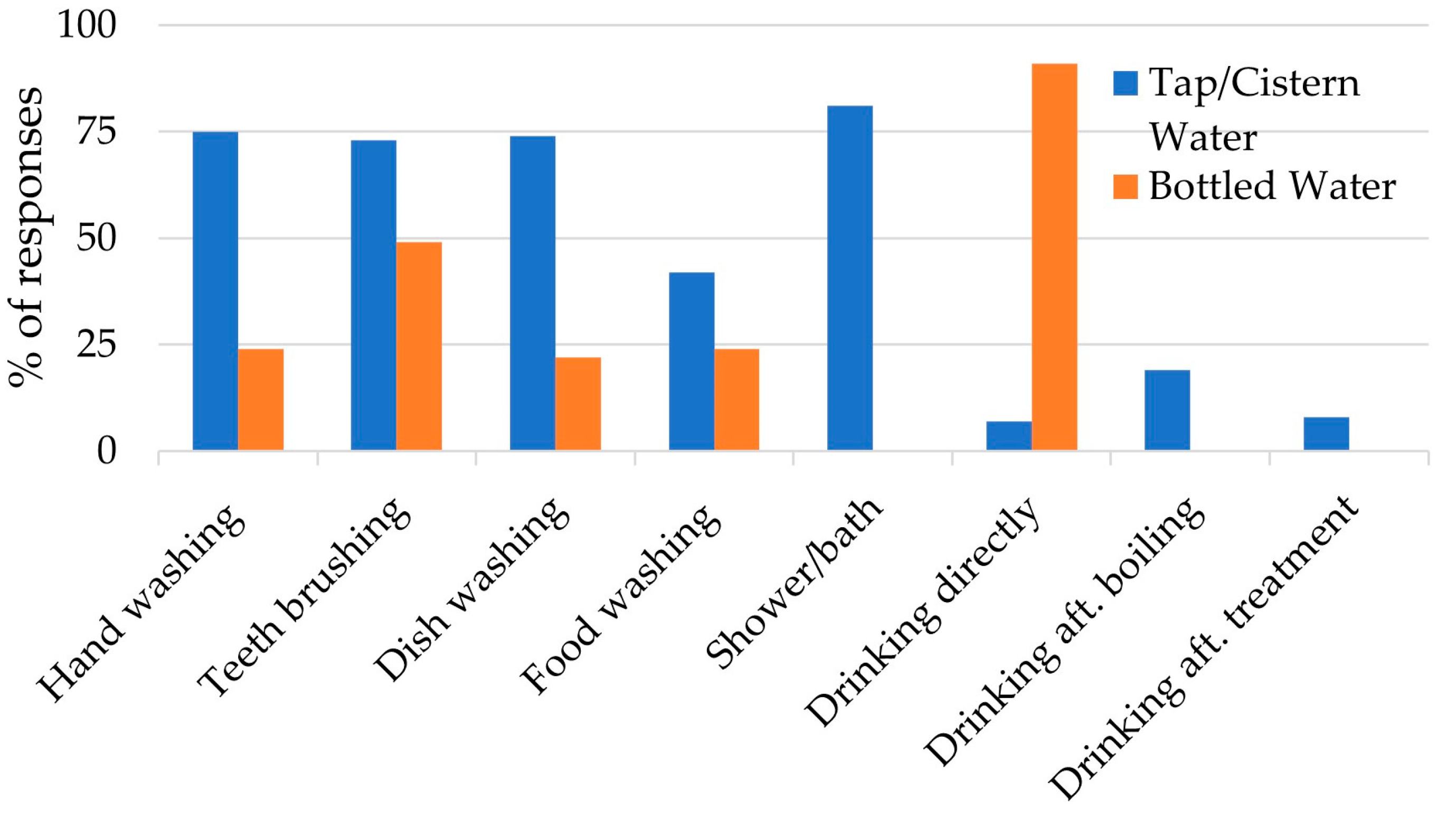
3.1. Household Survey
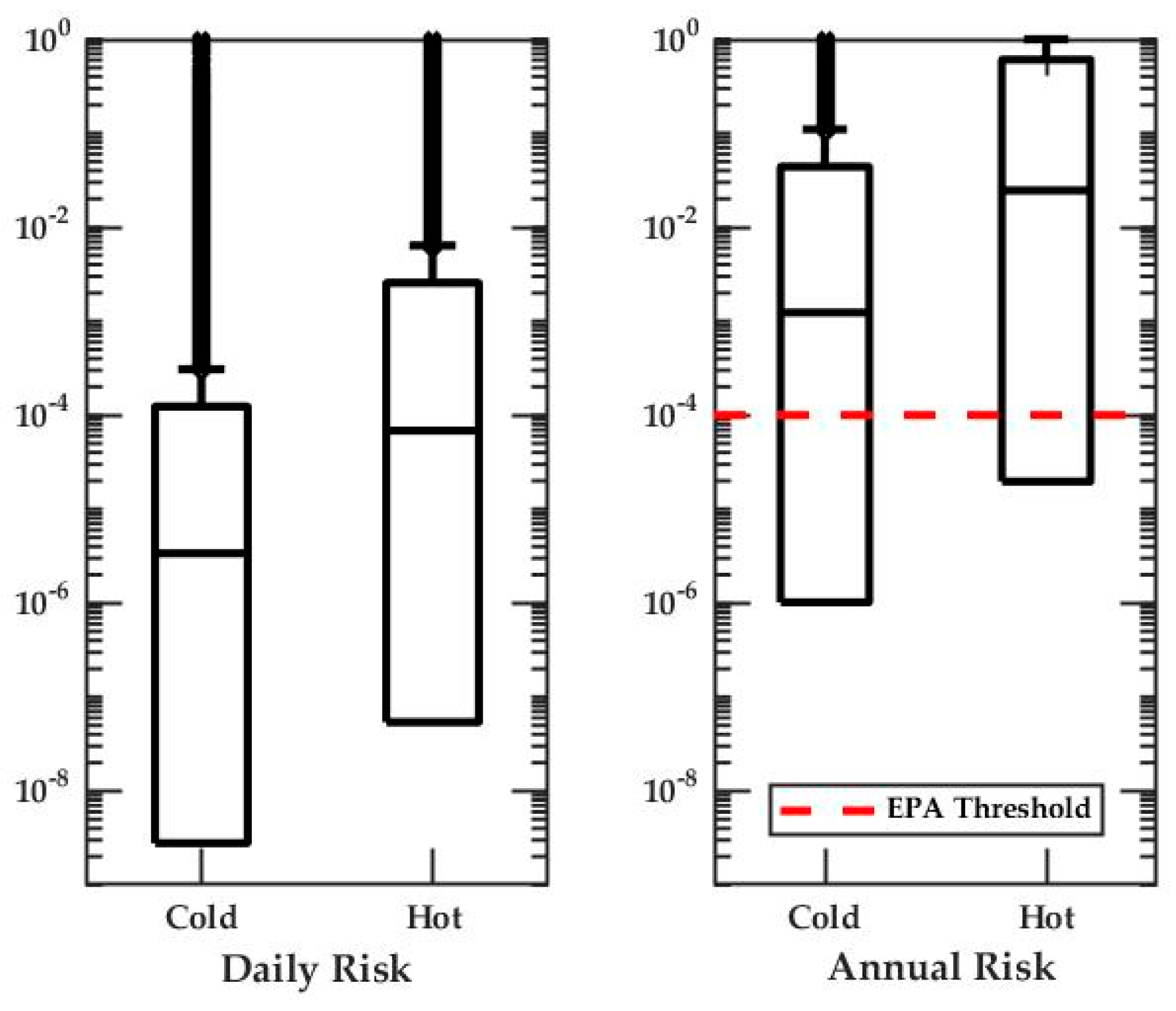
3.2. Quantitative Microbial Risk Assessment
4. Discussion
4.1. Legionella Risk Post Hurricanes
4.2. Risk Perception and Risk Management
5. Conclusions
- The estimated Legionella annual risks exceed the EPA guideline of 10−4 pppy.
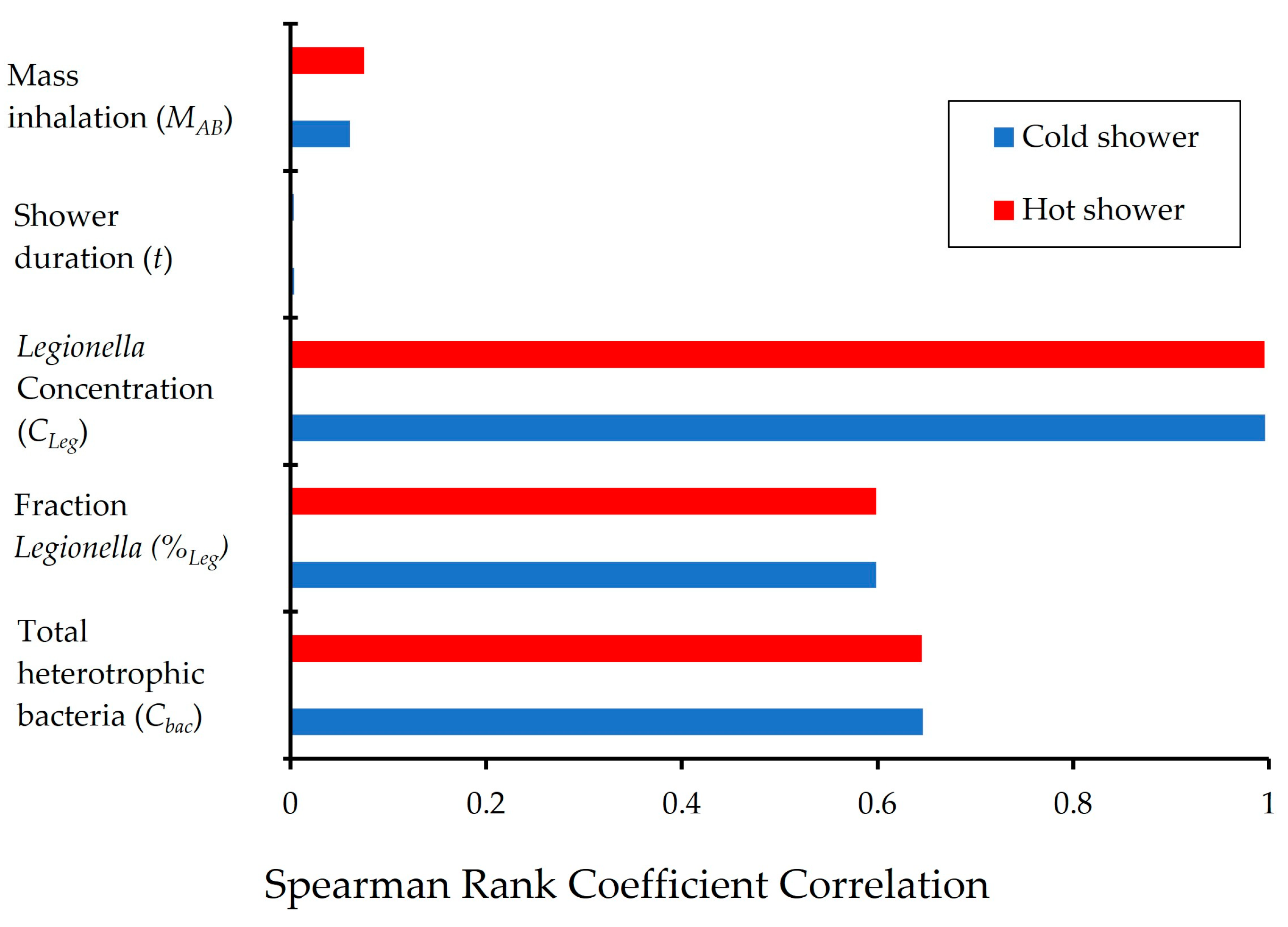
- The model outputs are sensitive to the concentration of Legionella in individual cisterns, which was highly variable and uncertain due to unevenness in the cistern water management on the island.
- There is a disparity between perceived risk and QMRA estimated risk of cistern water, suggesting the Legionella risk associated with the shower water is not apparent to the local residents.
- Moreover, the perception of water safety is divided by income group. Most people in the high-income group considered their water “safe”, while people in the low-income group only considered their water “somewhat safe”.
- Both income groups believed that the government could have done more to help them understand the water quality and water safety at the time of natural disaster. A fact-based public education program should be developed to bring residents onboard to manage the cistern water quality collaboratively.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abdulla, F.A.; Al-Shareef, A.W. Roof rainwater harvesting systems for household water supply in Jordan. Desalination 2009, 243, 195–207. [Google Scholar] [CrossRef]
- Ahmed, W.; Brandes, H.; Gyawali, P.; Sidhu, J.P.S.; Toze, S. Opportunistic pathogens in roof-captured rainwater samples, determined using quantitative PCR. Water Res. 2014, 53, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.Y.; Hamilton, A.J.; Jiang, S.C. Assessment of public health risk associated with viral contamination in harvested urban stormwater for domestic applications. Sci. Total Environ. 2015, 523, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Declerck, P.; Behets, J.; Margineanu, A.; van Hoef, V.; De Keersmaecker, B.; Ollevier, F. Replication of Legionella pneumophila in biofilms of water distribution pipes. Microbiol. Res. 2009, 164, 593–603. [Google Scholar] [CrossRef]
- Shah, P.; Barskey, A.; Binder, A.; Edens, C.; Lee, S.; Smith, J.; Schrag, S.; Whitney, C.; Cooley, L. Legionnaires’ Disease Surveillance Summary Report, United States. Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/legionella/health-depts/surv-reporting/2014-15-surv-report-508.pdf (accessed on 19 March 2021).
- Fields, B.S.; Benson, R.F.; Besser, R.E. Legionella and Legionnaire’s disease: 25 Years of investigation. J. Clin. Microbiol. 1982, 16, 697–699. [Google Scholar]
- Abu Khweek, A.; Amer, A.O. Factors mediating environmental biofilm formation by Legionella pneumophila. Front. Cell. Infection Microbiol. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Zolnikov, T.R. A Humanitarian crisis: Lessons learned from hurricane Irma. Am. J. Public Health 2017, 108, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.C.; Han, M.; Chandrasekaran, S.; Fang, Y.; Kellogg, C.A. Assessing the water quality impacts of two Category-5 hurricanes on St. Thomas, Virgin Islands. Water Res. 2020, 171. [Google Scholar] [CrossRef]
- WAPA Issues Precautionary Boil Water Advisory. Available online: https://viconsortium.com/VIC/?p=57057 (accessed on 10 June 2019).
- The St. Thomas Source. WAPA Issues Boil Notice for Potable Water Customers. Available online: https://stthomassource.com/content/2017/10/11/wapa-issues-boil-notice-for-potable-water-customers/ (accessed on 4 September 2020).
- RStudio Team. RStudio: Integrated Development for R. Available online: http://www.rstudio.com/ (accessed on 1 November 2020).
- National Research Council. Risk assessment in the federal government. In Risk Assessment in the Federal Government. Available online: https://www.nap.edu/catalog/366/risk-assessment-in-the-federal-government-managing-the-process (accessed on 1 January 2021).
- Benedict, K.M.; Reses, H.; Vigar, M.; Roth, D.M.; Roberts, V.A.; Mattioli, M.; Cooley, L.A.; Hilborn, E.D.; Wade, T.J.; Fullerton, K.E.; et al. Surveillance for waterborne disease outbreaks associated with drinking water—United States, 2013–2014. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1216–1221. [Google Scholar] [CrossRef]
- Broadhead, A.N.; Negron-Alvira, A.; Baez, L.A.; Hazen, T.C.; Canoy, M.J. Occurrence of Legionella species in tropical rain water cisterns. Caribb. J. Sci. 1988, 24, 71–73. [Google Scholar]
- Simmons, G.; Jury, S.; Thornley, C.; Harte, D.; Mohiuddin, J.; Taylor, M. A Legionnaires’ disease outbreak: A water blaster and roof-collected rainwater systems. Water Res. 2008, 42, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Diederen, B.M.W. Legionella spp. and Legionnaires’ disease. J. Infect. 2008, 56, 1–12. [Google Scholar] [CrossRef] [PubMed]
- 9215 Heterotrophic Plate Count, Standard Methods for the Examination of Water and Wastewater. Available online: https://www.standardmethods.org/doi/full/10.2105/SMWW.2882.188 (accessed on 15 February 2021).
- Crabtree, K.D.; Ruskin, R.H.; Shaw, S.B.; Rose, J.B. The Detection of cryptosporidium oocysts and giardia cysts in cistern water in the U.S. Virgin Islands. Water Res. 1996, 30, 211. [Google Scholar] [CrossRef]
- Hamilton, K.A.; Ahmed, W.; Palmer, A.; Sidhu, J.P.S.; Hodgers, L.; Toze, S.; Haas, C.N. Public health implications of Acanthamoeba and multiple potential opportunistic pathogens in roof-harvested rainwater tanks. Environ. Res. 2016, 150, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Rogers, J.; Dowsett, A.B.; Dennis, P.J.; Lee, J.V.; Keevil, C.W. Influence of temperature and plumbing material selection on biofilm formation and growth of Legionella pneumophila in a model potable water system containing complex Microbial Flora. Appl. Environ. Microbiol. 1994, 60, 1590. [Google Scholar] [CrossRef]
- Copenhaver, A.M.; Casson, C.N.; Nguyen, H.T.; Fung, T.C.; Duda, M.M.; Roy, C.R.; Shin, S. Alveolar macrophages and neutrophils are the primary reservoirs for Legionella pneumophila and mediate cytosolic surveillance of type IV secretion. Infect. Immunity 2014, 82, 4325–4336. [Google Scholar] [CrossRef]
- Bollin, G.E.; Plouffe, J.F.; Para, M.F.; Hackman, B. Aerosols containing Legionella pneumophila generated by shower heads and hot-water faucets. Appl. Environ. Microbiol. 1985, 50, 1128–1131. [Google Scholar] [CrossRef]
- Feazel, L.M.; Baumgartner, L.K.; Peterson, K.L.; Frank, D.N.; Harris, J.K.; Pace, N.R. Opportunistic pathogens enriched in showerhead biofilms. Proc. Natl. Acad. Sci. USA 2009, 106, 16393–16398. [Google Scholar] [CrossRef]
- Zhou, Y.; Benson, J.M.; Irvin, C.; Irshad, H.; Cheng, Y.S. Particle size distribution and inhalation dose of shower water under selected operating conditions. Inhalation Toxicol. 2007, 19, 333–342. [Google Scholar] [CrossRef]
- Cervero-Aragó, S.; Rodríguez-Martínez, S.; Puertas-Bennasar, A.; Araujo, R.M. Effect of common drinking water disinfectants, chlorine and heat, on free Legionella and amoebae-associated Legionella. PLoS ONE 2015, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- DeOreo, W.B.; Mayer, P.W.; Dziegielewski, B.; Kiefer, J. Residential End Uses of Water. Water Res. Found. 2016, 2, 9. [Google Scholar]
- Armstrong, T.W.; Haas, C.N. A quantitative microbial risk assessment model for Legionnaires’ disease: Animal model selection and dose-response modeling. Risk Anal. 2007, 27, 1581–1596. [Google Scholar] [CrossRef] [PubMed]
- Berendt, R.F.; Young, H.W.; Allen, R.G.; Knutsen, G.L. Dose-response of guinea pigs experimentally infected with aerosols of Legionella pneumophila. J. Infect. Dis. 1980, 141, 186–192. [Google Scholar] [CrossRef] [PubMed]
- U.S. Census Bureau, 2010 Census, U.S. Virgin Islands. Available online: https://www.census.gov/prod/cen2010/doc/dpsfvi.pdf (accessed on 4 September 2020).
- Kim, T.; Lye, D.; Donohue, M.; Mistry, J.H.; Pfaller, S.; Vesper, S.; Kirisits, M.J. Harvested rainwater quality before and after treatment and distribution in residential systems. J. Am. Water Works Assn. 2016, 108, E571–E584. [Google Scholar] [CrossRef]
- Ahmed, W.; Gardner, T.; Toze, S. Microbiological quality of roof-harvested rainwater and health risks: A review. J. Environ. Qual. 2010, 40, 13. [Google Scholar] [CrossRef]
- Hamilton, K.A.; Ahmed, W.; Toze, S.; Haas, C.N. Human health risks for Legionella and Mycobacterium avium complex (MAC) from potable and non-potable uses of roof-harvested rainwater. Water Res. 2017, 119, 288–303. [Google Scholar] [CrossRef]
- Lee, J.V.; Lai, S.; Exner, M.; Lenz, J.; Gaia, V.; Casati, S.; Hartemann, P.; Luck, C.; Pangon, B.; Ricci, M.L.; et al. An international trial of quantitative PCR for monitoring Legionella in artificial water systems. J. Appl. Microbiol. 2011, 110, 1032–1044. [Google Scholar] [CrossRef] [PubMed]
- Scaturro, M.; Buffoni, M.; Girolamo, A.; Cristino, S.; Girolamini, L.; Mazzotta, M.; Bucci Sabattini, M.A.; Zaccaro, C.M.; Chetti, L.; Laboratory, M.A.N.; et al. Performance of Legiolert test vs. ISO 11731 to confirm Legionella pneumophila contamination in potable water samples. Pathogens 2020, 9, 690. [Google Scholar] [CrossRef]
- De Giglio, O.; Diella, G.; Trerotoli, P.; Consonni, M.; Palermo, R.; Tesauro, M.; Laganà, P.; Serio, G.; Montagna, M.T. Legionella detection in water networks as per ISO 11731:2017: Can different filter pore sizes and direct placement on culture media influence laboratory results? Int. J. Environ. Res. Public Health 2020, 17, 2077. [Google Scholar] [CrossRef]
- Ditommaso, S.; Gentile, M.; Giacomuzzi, M.; Zotti, C.M. Recovery of Legionella species from water samples using an internal method based on ISO 11731: Suggestions for revision and implementation. Diagn. Microbiol. Infect. Dis. 2011, 70, 200–206. [Google Scholar] [CrossRef]
- ISO 11731:2017. Available online: https://www.iso.org/standard/61782.html (accessed on 15 February 2021).
- Legionnaires Disease Procedures Manual for Recovery. Available online: https://www.cdc.gov/legionella/labs/procedures-manual.html (accessed on 15 February 2021).

| Parameter Definition | Symbol | Point Estimate or Distribution | Unit | Source |
|---|---|---|---|---|
| Concentration of total heterotrophic bacteria measured in an untreated rainwater cistern | Cbac | Empirical distribution | CFU/mL | [19] |
| Percent of total bacteria DNA represented by Legionella | %Leg | Empirical distribution | unitless | [9] |
| Shower water temperature | Tcold | 24.5 | °C | This study |
| Thot | 43.5 | °C | ||
| Percent of shower water that is heated to 60 °C by a conventional water heater | %hot | 54 | unitless | This study Equation (3) |
| Density of water | ρhot | 991 | g/cm3 | This study |
| ρcold | 997 | g/cm3 | ||
| Thermal inactivation of Legionella at temperature T = 60°C | logT | 3 | unitless | [26] |
| Shower duration | t | Normal distribution (μ = 7.8, α = 0.02, left-truncated at zero) | min | [27] |
| Aerosol mass inhaled and deposited in the alveolar-bronchiolar region, hot shower | MAB-h | Uniform distribution U(0.036, 0.364) | mg/min | [25] |
| Aerosol mass inhaled and deposited in the alveolar-bronchiolar region, cold shower | MAB-c | Uniform distribution U(0.001, 0.008) | mg/min | [25] |
| Dose–response curve constant | k | 0.0599 | unitless | [28] |
| Number of exposures per year | n | 365 | per year | This study |
| 25th Percentile | Median | 75th Percentile | Unit | Source | |
|---|---|---|---|---|---|
| Legionella Concentration | 3.0 × 103 | 9.0 × 104 | 1.3 × 106 | Gene copies/L | [20] |
| 4.0 × 103 | 8.5 × 104 | 3.1 × 106 | |||
| 1.6 × 104 | 2.5 × 104 | 1.0 × 105 | [2] | ||
| 2.5 × 102 | 8.8 × 103 | 1.2 × 105 | CFU/L | This paper | |
| Annual Risk | 1.0 × 10−2 | pppy (per person per year) | [33] | ||
| 1.1 × 10−3 | 3.1 × 10−3 | [32] | |||
| 2.9 × 10−6 | 4.5 × 10−3 | 2.1 × 10−1 | This paper |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quon, H.; Allaire, M.; Jiang, S.C. Assessing the Risk of Legionella Infection through Showering with Untreated Rain Cistern Water in a Tropical Environment. Water 2021, 13, 889. https://doi.org/10.3390/w13070889
Quon H, Allaire M, Jiang SC. Assessing the Risk of Legionella Infection through Showering with Untreated Rain Cistern Water in a Tropical Environment. Water. 2021; 13(7):889. https://doi.org/10.3390/w13070889
Chicago/Turabian StyleQuon, Hunter, Maura Allaire, and Sunny C. Jiang. 2021. "Assessing the Risk of Legionella Infection through Showering with Untreated Rain Cistern Water in a Tropical Environment" Water 13, no. 7: 889. https://doi.org/10.3390/w13070889
APA StyleQuon, H., Allaire, M., & Jiang, S. C. (2021). Assessing the Risk of Legionella Infection through Showering with Untreated Rain Cistern Water in a Tropical Environment. Water, 13(7), 889. https://doi.org/10.3390/w13070889