Performance of Reverse Electrodialysis System for Salinity Gradient Energy Generation by Using a Commercial Ion Exchange Membrane Pair with Homogeneous Bulk Structure
Abstract
1. Introduction
2. Materials and Methods
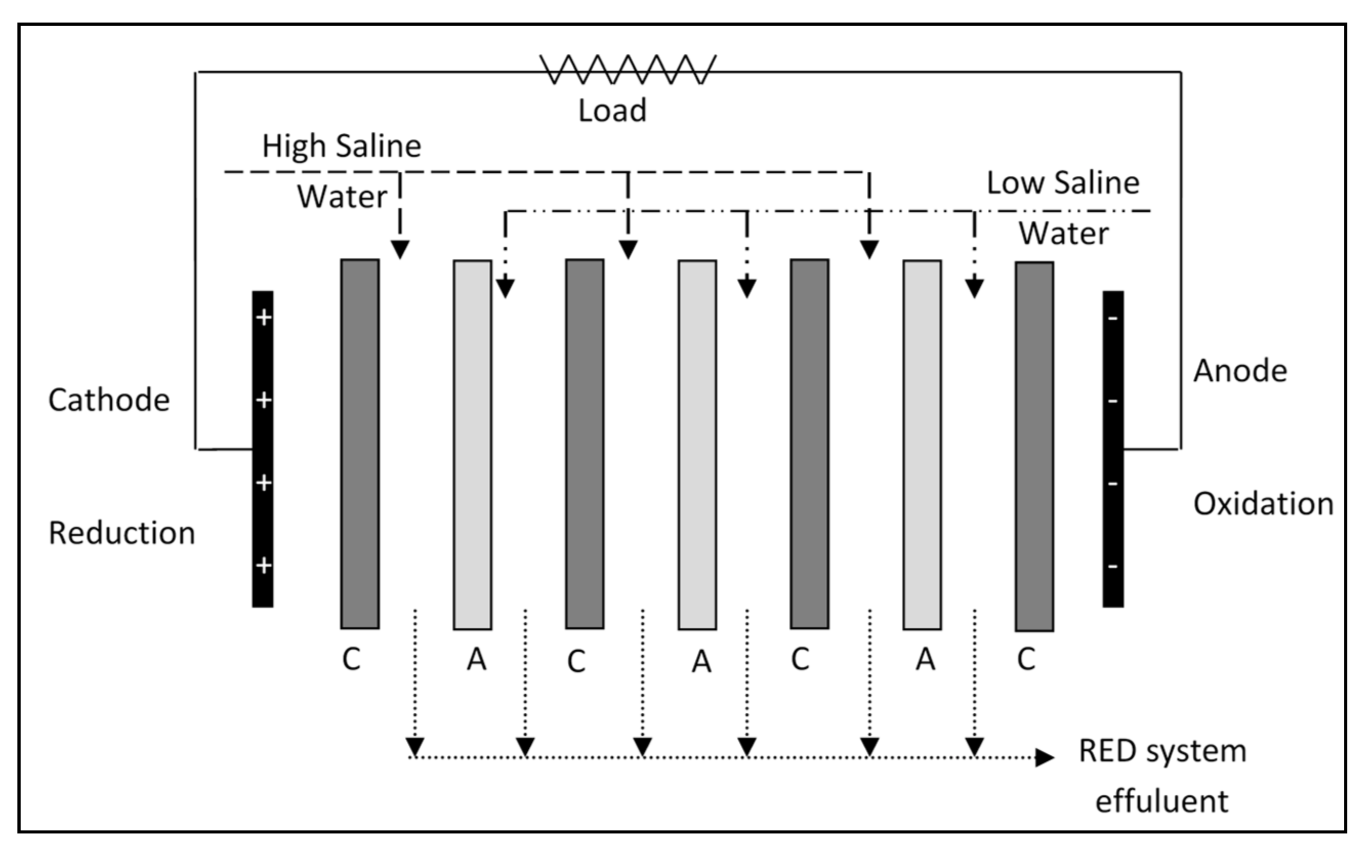
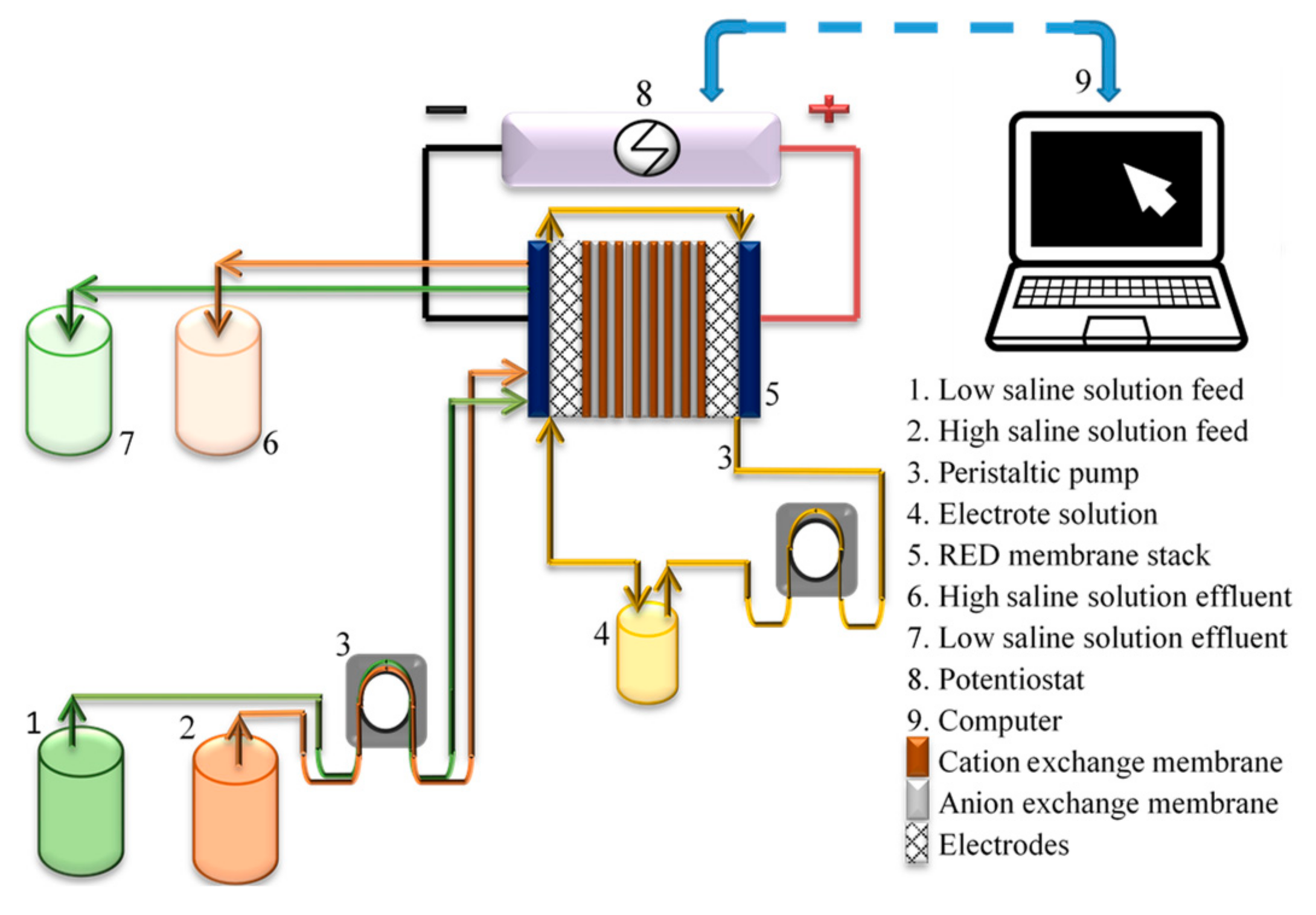
2.1. RED System
2.2. RED Tests
2.3. Parametric RED Studies with Fujifilm Membranes
2.4. Blank Tests
2.5. Calculations
3. Results and Discussion
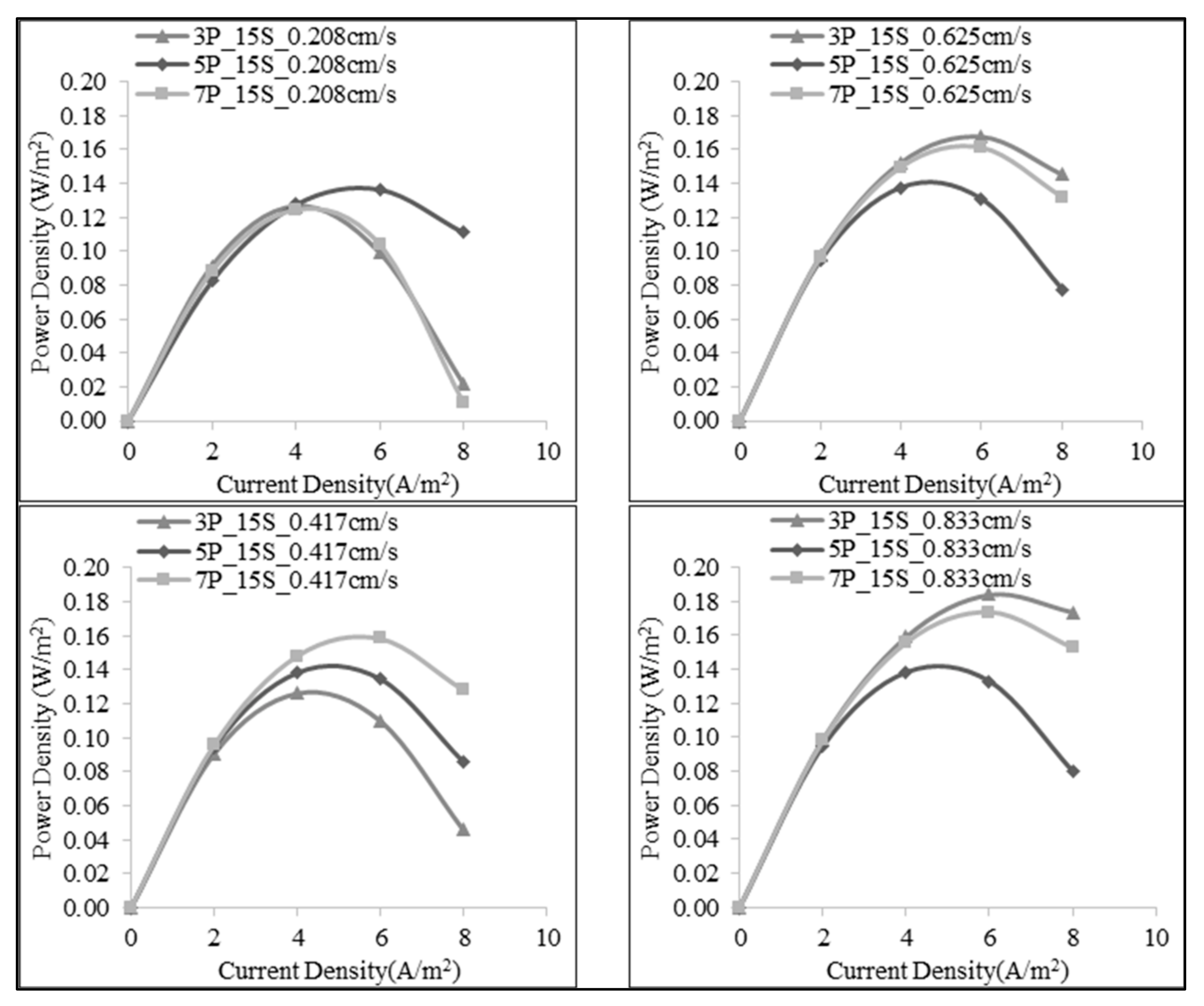
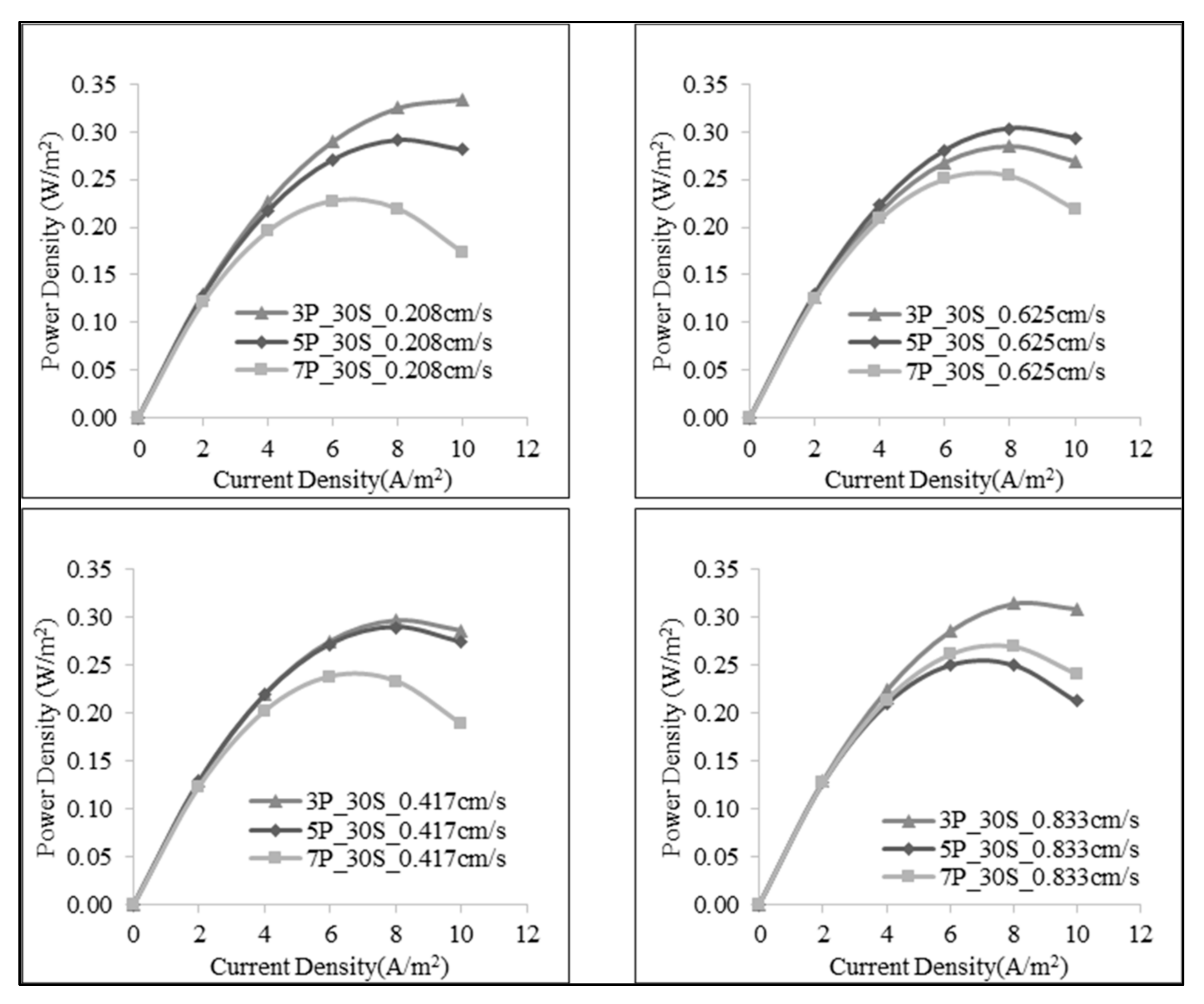
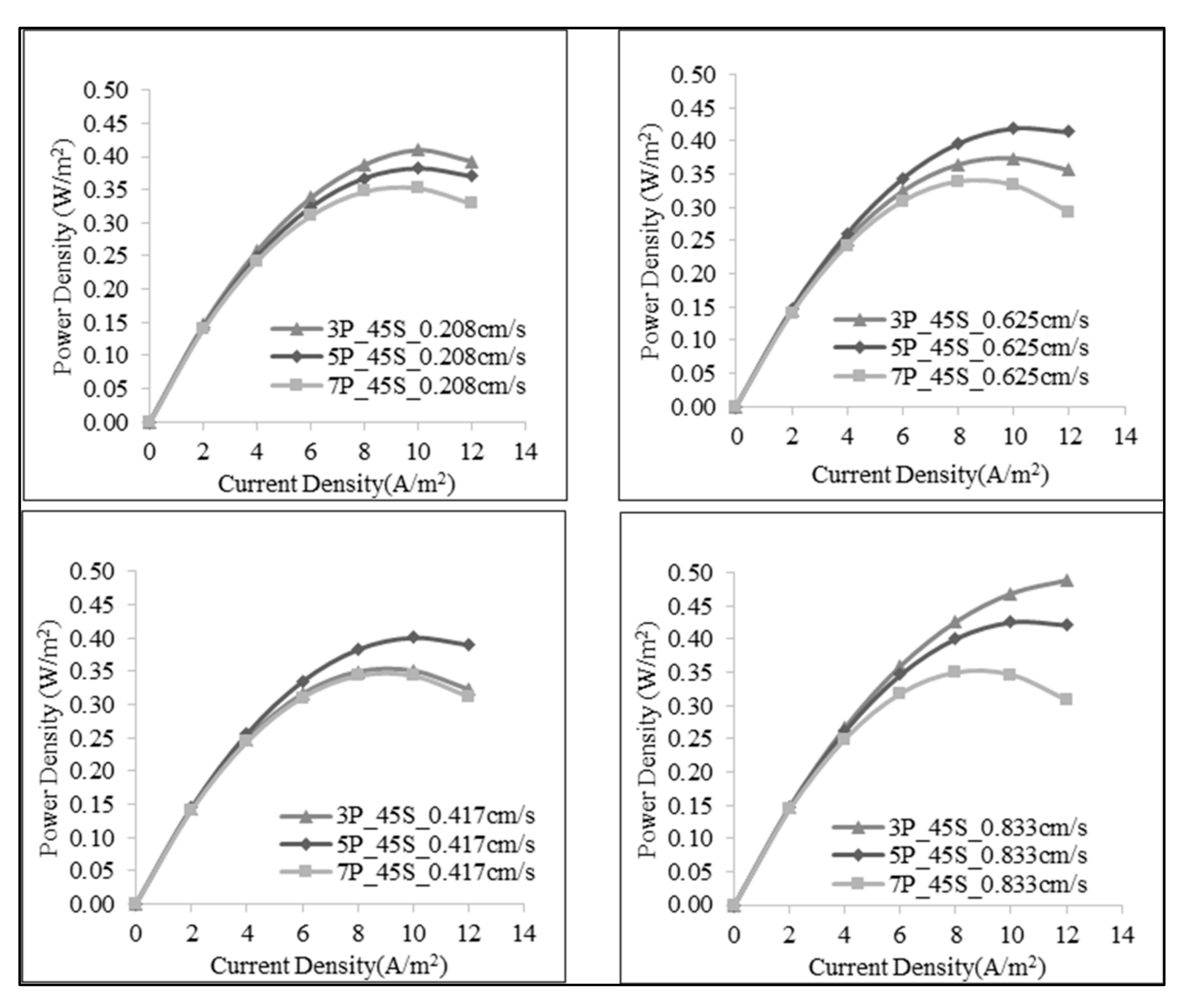
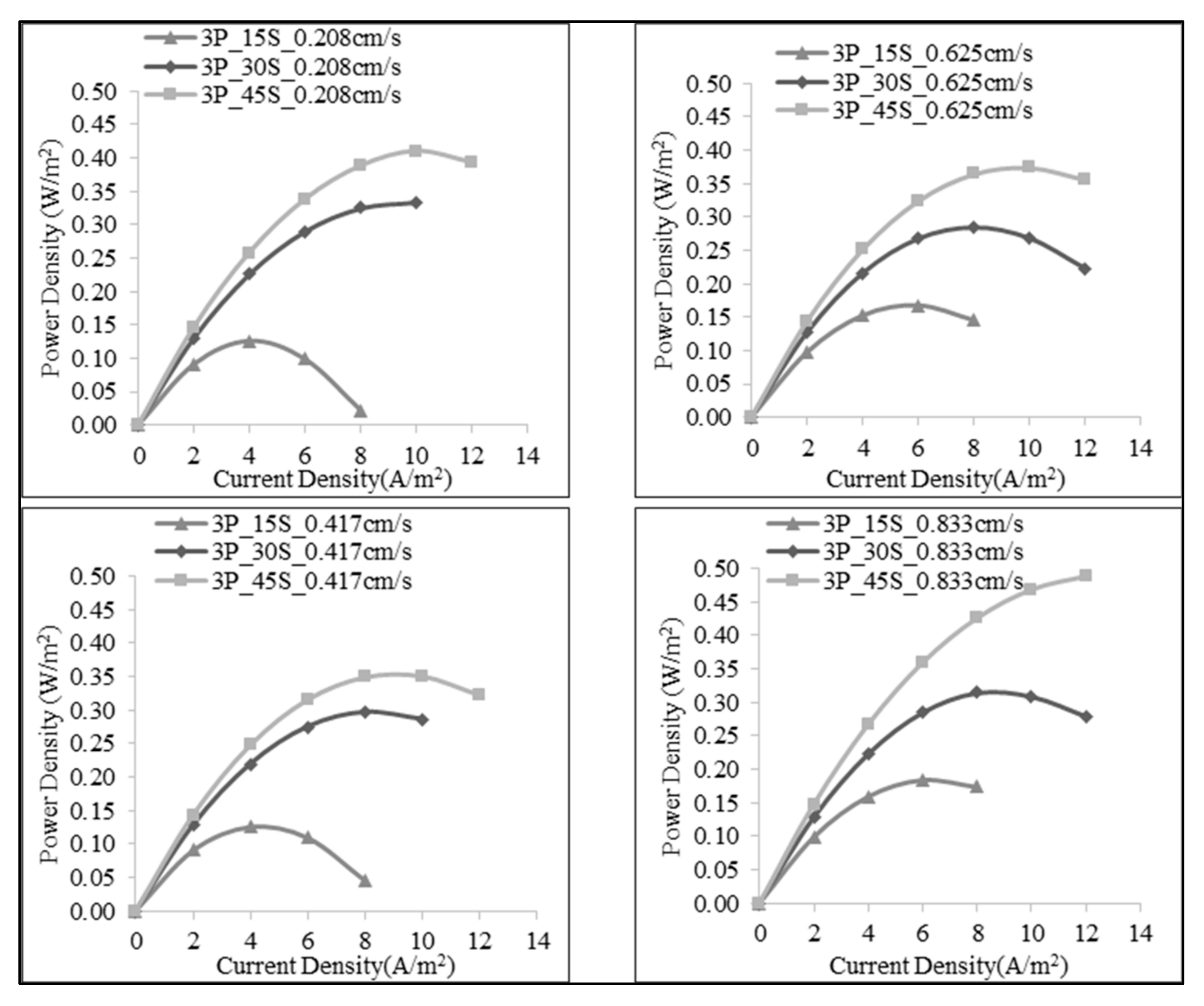
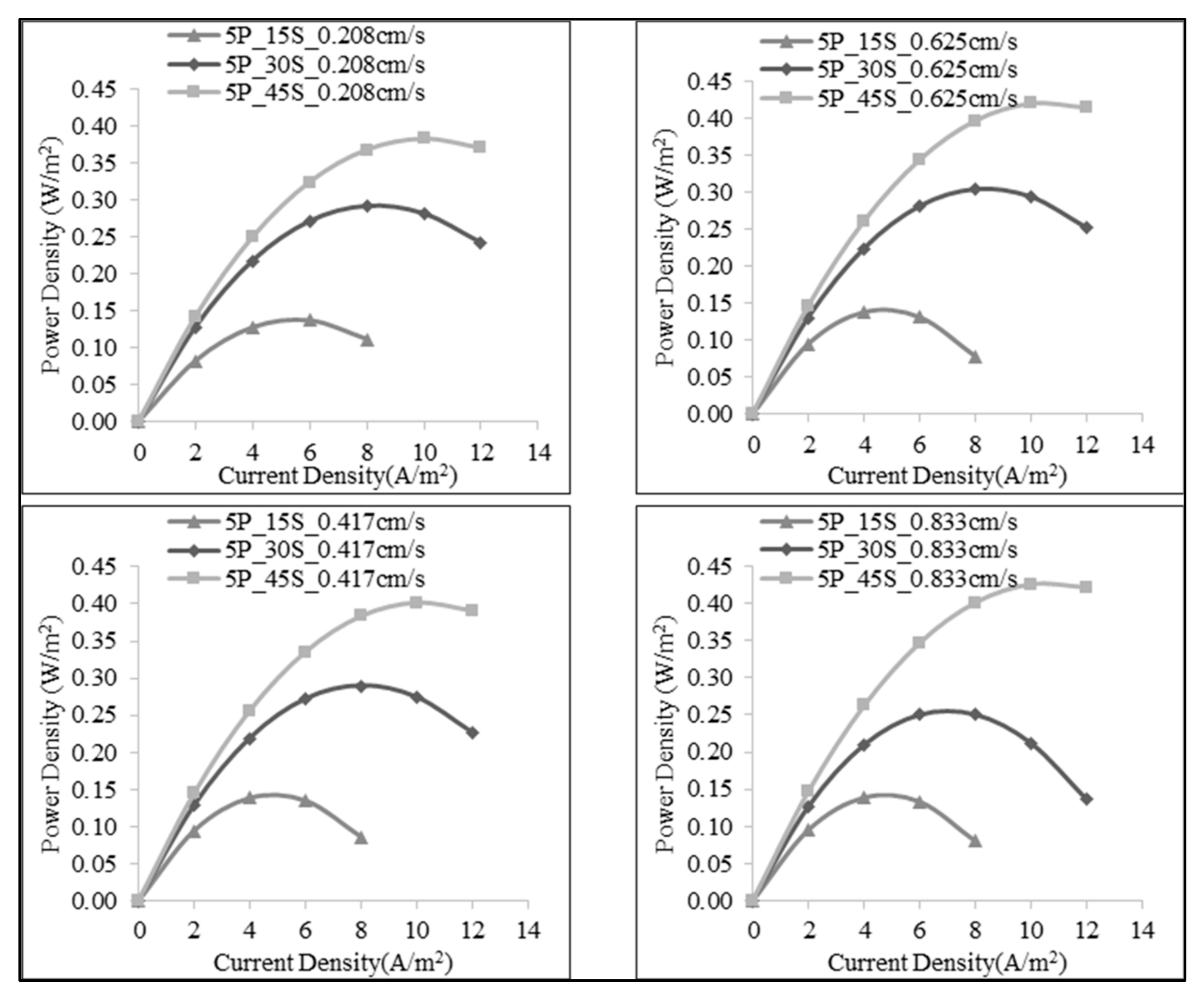
3.1. Effect of Number of the Membrane Pairs on SGE Production
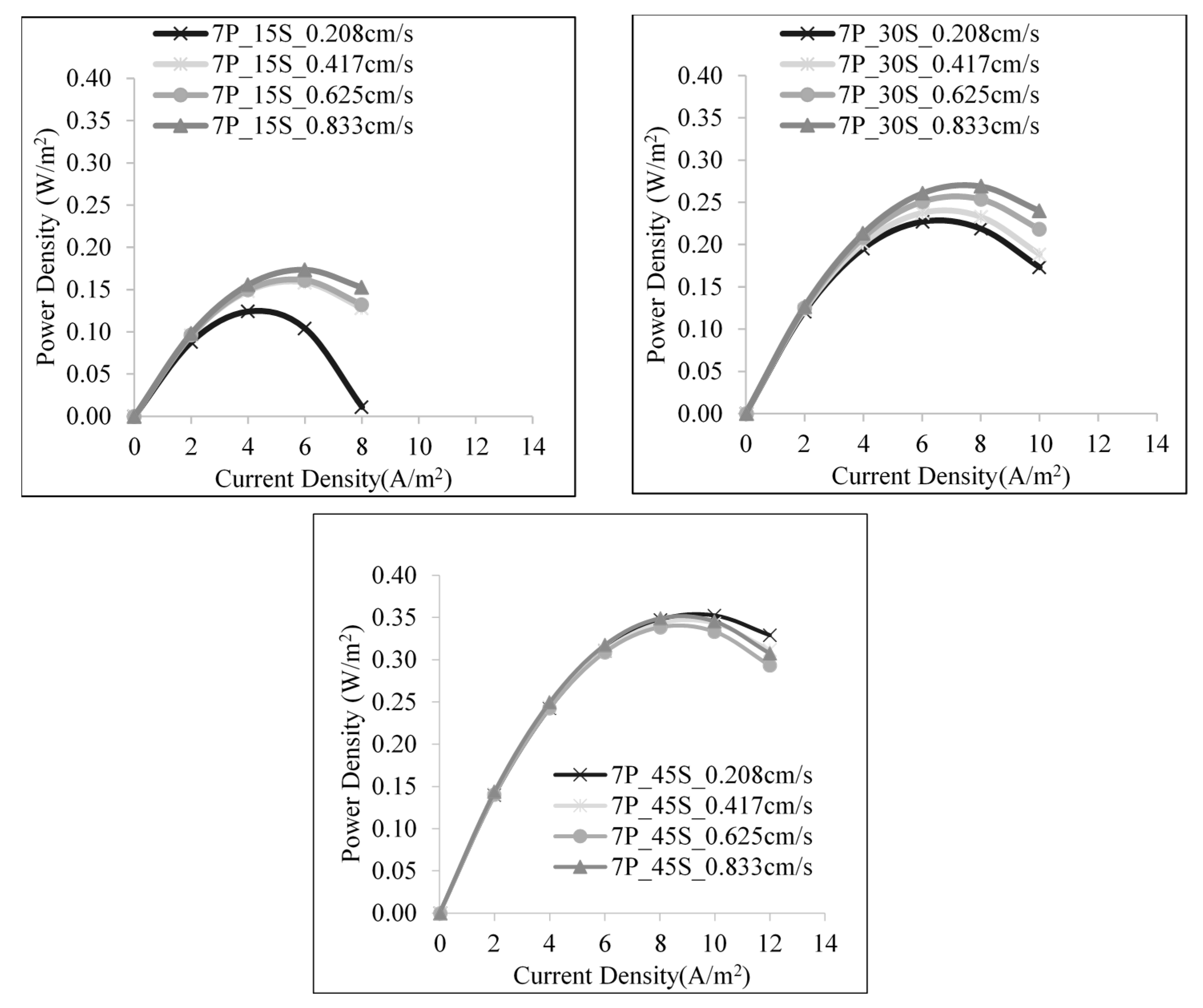
3.2. Effect of Linear Flow Rate of Feed Solutions on SGE Production
3.3. Effect of Salt Ratio on SGE Production
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nijmeijer, K.; Metz, S. Chapter 5: Salinity Gradient Energy. In Sustainability Science and Engineering; Elsevier B.V.: Amsterdam, The Netherlands, 2010; Volume 2, pp. 95–139. [Google Scholar]
- Micale, G.; Cipollina, A.; Tamburini, A. Chapter 1: Salinity gradient energy. In Sustainable Energy from Salinity Gradients; Elsevier B.V.: Amsterdam, The Netherlands, 2016; pp. 1–18. [Google Scholar]
- Schaetzle, O.; Buisman, C.J.N. Salinity Gradient Energy: Current State and New Trends. Engineering 2015, 1, 164–166. [Google Scholar] [CrossRef]
- Kempener, R.; Neumann, F. Ocean Energy Technology Brief. IRENA 2014, 2, 1–26. [Google Scholar]
- Hong, J.G.; Gao, H.; Gan, L.; Tong, X.; Xiao, C.; Liu, S.; Zhang, B.; Chen, Y. Chapter 13: Nanocomposite and nanostructured ion exchange membrane in salinity gradient power generation using reverse electrodialysis. In Advanced Nanomaterials for Membrane Synthesis and Its Applications; Elsevier B.V.: Amsterdam, The Netherlands, 2019; pp. 295–316. [Google Scholar]
- Zhijun, J.; Baoguo, W.; Shiqiang, S.; Yongsheng, F. Blue energy: Current Technologies for sustainable power generation from water salinity gradient. Renew. Sust. Energ. Rev. 2014, 31, 91–100. [Google Scholar]
- Logan, B.; Elimelech, M. Membrane-based processes for sustainable power generation using water. Nature 2012, 488, 313–319. [Google Scholar] [CrossRef]
- Skråmestø, Ø.S.Ø.; Skilhagen, S.S.E.; Nielsen, W.K.W. Power Production Based on Osmotic Pressure. Waterpower XVI 2009, 1–9. Available online: https://www.statkraft.com/globalassets/old-contains-the-old-folder-structure/documents/waterpower_xvi_-_power_production_based_on_osmotic_pressure_tcm21-4795.pdf (accessed on 5 March 2021).
- Alvarez-Silva, O.A.; Osorio, A.F.; Winter, C. Practical global salinity gradient energy potential. Renew. Sustain. Energy Rev. 2016, 60, 1387–1395. [Google Scholar] [CrossRef]
- Post, J.W. Blue Energy: Electricity Production from Salinity Gradients by Reverse Electrodialysis. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2009. [Google Scholar]
- Bijmans, M.F.M.; Burheim, O.S.; Bryjak, M.; Delgado, A.; Hack, P.; Tenisson, S.; Hamelers, H.V.M. CAPMIX—Deploying capacitors for salt gradient power. Energy Procedia 2012, 20, 108–115. [Google Scholar] [CrossRef]
- Gómez-Coma, L.; Ortiz-Martínez, V.M.; Fallanza, M.; Ortiz, A.; Ibañez, R.; Ortiz, I. Blue energy for sustainable water reclamation in WWTPs. J. Water Pro. Eng. 2020, 101020. [Google Scholar] [CrossRef]
- Bryjak, M.; Kabay, N.; Güler, E.; Tomaszewska, B. Concept for energy harvesting from the salinity gradient on the basis of geothermal water. WEENTECH Proc. Energy 2018, 4, 88–96. [Google Scholar]
- Hong, J.G.; Park, T.W.; Dhadake, Y. Property evaluation of custom-made ion exchange membranes for electrochemical performance in reverse electrodialysis application. J. Electroanal. Chem. 2019, 850, 1–8. [Google Scholar] [CrossRef]
- Choi, J.; Yang, S.C.; Jeong, N.J.; Kim, H.; Kim, W.S. Fabrication of an Anion-Exchange Membrane by Pore-Filling Using Catechol−1,4-Diazabicyclo-[2,2,2] octane Coating and Its Application to Reverse Electrodialysis. Langmuir 2018, 34, 10837–10846. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.C.; Choi, Y.W.; Choi, J.; Jeong, N.; Kim, H.; Jeong, H.; Byeon, S.Y.; Yoon, H.; Kim, Y.H. Green fabrication of pore-filling anion exchange membranes using R2R processing. J. Membr. Sci. 2019, 584, 181–190. [Google Scholar] [CrossRef]
- Shah, B.G.; Shahi, V.K.; Thampy, S.K.; Rangarajan, R.; Ghosh, P.K. Comparative studies on performance of interpolymer and heterogeneous ion-exchange membranes for water desalination by electrodialysis. Desalination 2005, 172, 257–265. [Google Scholar] [CrossRef]
- Smolinska-Kempisty, K.; Siekierka, A.; Bryjak, M. Interpolymer ion exchange membranes for CapMix process. Desalination 2020, 482, 114384. [Google Scholar] [CrossRef]
- Fujifilm. Ion Exchange Membranes. Available online: https://www.fujifilm.eu/eu/products/industrial-products/membrane-technology/ion-exchange-membranes#content0 (accessed on 17 February 2021).
- Vecino, X.; Reig, M.; Gibert, O.; Valderrama, C.; Cortina, J.L. Integration of monopolar and bipolar electrodialysis processes for tartaric acid recovery from residues of the winery industry. ACS Sus. Chem. Eng. 2020, 8, 13387–13399. [Google Scholar] [CrossRef]
- Bhadja, V.; Makwana, B.S.; Maiti, S.; Sharma, S.; Chatterjee, U. Comparative efficacy study of different types of ion exchange membranes for production of ultrapure water via electrodeionization. Ind. Eng. Chem. Res. 2015, 54, 10974–10982. [Google Scholar] [CrossRef]
- Sarapulova, V.; Shkorkina, I.; Mareev, S.; Pismenskaya, N.; Kononenko, N.; Larchet, C.; Nikonenko, V. Transport characteristics of fujifilm ion-exchange membranes as compared to homogeneous membranes AMX and CMX and to heterogeneous membranes MK-40 and MA-41. Membranes 2019, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Ahdab, Y.D.; Rehman, D.; Schücking, G.; Barbosa, M.; Lienhard, J.H. Treating Irrigation Water Using High-Performance Membranes for Monovalent Selective Electrodialysis. ACS EST Water 2020, 1, 117–124. [Google Scholar] [CrossRef]
- Rijnaarts, T.; Huerta, E.; van Baak, W.; Nijmeijer, K. Effect of divalent cations on RED performance and cation exchange membrane selection to enhance power densities. Environ. Sci. Technol. 2017, 51, 13028–13035. [Google Scholar] [CrossRef]
- Moreno, J.; Grasman, S.; van Engelen, R.; Nijmeijer, K. Upscaling reverse electrodialysis. Environ. Sci. Technol. 2018, 52, 10856–10863. [Google Scholar] [CrossRef]
- Veerman, J.; de Jong, R.M.; Saakes, M.; Metz, S.J.; Harmsen, G.J. Reverse electrodialysis: Comparison of six commercial membrane pairs on the thermodynamic efficiency and power density. J. Membr. Sci. 2009, 343, 7–15. [Google Scholar] [CrossRef]
- Güler, E. Anion Exchange Membrane Design for Reverse Electrodialysis. Ph.D. Thesis, University of Twente, Enschede, The Netherlands, 2013. [Google Scholar]
- Fujifilm, 2020, Ion Exchange Membranes for Water Purification. Available online: https://www.fujifilmmembranes.com/images/IEM_brochure_1_1_final_small_size.pdf (accessed on 13 July 2020).
- Güler, E.; Elizen, R.; Vermaas, D.A.; Saakes, M.; Nijmeijer, K. Performance-determining membrane properties in reverse electrodialysis. J. Membr. Sci. 2013, 446, 266–276. [Google Scholar] [CrossRef]
- Tedesco, M.; Cipollina, A.; Tamburini, A.; Micale, G. Towards 1 kW Power Production in a Reverse Electrodialysis Pilot Plant with Saline Waters and Concentrated Brines. J. Membr. Sci. 2017, 522, 226–236. [Google Scholar] [CrossRef]
- Long, R.; Li, B.; Liu, Z.; Liu, W. Reverse electrodialysis: Modelling and performance analysis based on multi-objective optimization. Energy 2018, 151, 1–10. [Google Scholar] [CrossRef]
- Long, R.; Li, B.; Liu, Z.; Liu, W. Performance analysis of reverse electrodialysis stacks: Channel geometry and flow rate optimization. Energy 2018, 158, 427–436. [Google Scholar] [CrossRef]
- Daniilidis, A.; Vermaas, D.A.; Herber, R.; Nijmeijer, K. Experimentally obtainable energy from mixing river water, seawater or brines with reverse electrodialysis. Renew. Energy 2014, 64, 123–131. [Google Scholar] [CrossRef]
| Membrane | Fujifilm Membrane Codes | Membrane Type | Polymer Matrix | Reinforcement | Dry Thickness (µm) | Water Uptake (wt %) | Ion Exchange Capacity (meq/g) | Electrical Resistance (Ω·cm2) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Cation Exchange Membranes | Type II CEM | Homogeneous Standard grade | Polyamide | 3D polyolefin fibers structure | 165 ± 5 | 25 ± 5 | 1.35 ± 0.05 | 2.97 | [20,21,22] |
| Type I CEM | 120 ± 5 | 29 ± 5 | 1.43 ± 0.05 | - | [22,24] | ||||
| Type X CEM | 125 ± 5 | 21 ± 5 | 1.67 ± 0.05 | - | [22] | ||||
| T0 CEM | - | - | - | - | [24] | ||||
| T1 CEM | Homogeneous multivalent (magnesium) permeable | 115 | - | 1.7 | [24,25] | ||||
| T1 CEM P150 | Homogeneous Profiled | 115/150 1 | - | - | 2.2 | [25] | |||
| Type 10 CEM | 125 | - | - | 2.3 | [25] | ||||
| Anion Exchange Membranes | Type II AEM | Homogeneous Standard grade | Polyamide | 3D polyolefin fibers structure | 165 ± 5 | 10 ± 5 | 1.08 ± 0.05 | 1.55 | [20,21,22] |
| Type I AEM | 120 ± 5 | 8 ± 2 | 1.50 ± 0.05 | - | [22,24,25] | ||||
| Type X AEM | 115 ± 5 | 23 ± 2 | 1.50 ± 0.05 | - | [22] | ||||
| Type 10 AEM | 125 | - | - | 1.5 | [25] |
| Parameter | Properties | |
|---|---|---|
| Active membrane area (electrode area) | 10 cm × 10 cm | |
| Electrodes (anode and cathode) | Ti/Ru-Ir alloyed mesh type (mesh 1.0, Area: 10 cm × 10 cm) | |
| Spacer thickness (μm) | 400 | |
| Volumetric flowrate of electrode rinse solution | 300 mL/min | |
| Composition of electrode rinse solution | 0.05 M K4Fe(CN)6/0.05 M K3Fe(CN)6 and 0.25 M NaCl mixture | |
| Concentrations of feed solutions (20.0 °C) | Low saline 1 g NaCl/L | High saline 15, 30 and 45 g NaCl/L |
| Flow velocities of feed solutions | 0.208; 0.417; 0.625; 0.833 cm/s | |
| Type | AEM Type 2 | CEM Type 2 |
|---|---|---|
| Homogeneous Anion Exchanger | Homogeneous Cation Exchanger | |
| Support material | Polyolefine | |
| Dry thickness (µm) | 160 | |
| Ion exchange capacity (mmol/g) | 1.08 ± 0.05 | 1.35 ± 0.05 |
| Electrical resistance (Ω·cm2) | 5 | 8 |
| Permselectivity (%) | 95 | 96 |
| Burst strength (kg/cm2) | 5.0 | 4.7 |
| pH range | 2–10 | 4–12 |
| Maximum temperature (°C) | 40 | |
| Application areas | Process water purification, wastewater minimization, obtaining tap water from saline water | |
| Number of Membrane Pairs | Salt Ratio (g:g) | Volumetric Flow Rate of Feed (mL/min) | Flow Velocity of Feed (cm/s) | Open Circuit Voltage (V) | Power (W) | Maximum Power Density (W/m2) |
|---|---|---|---|---|---|---|
| 3 | 1:15 | 30 | 0.208 | 0.353 | 0.008 | 0.126 |
| 60 | 0.417 | 0.356 | 0.008 | 0.126 | ||
| 90 | 0.625 | 0.353 | 0.024 | 0.393 | ||
| 120 | 0.833 | 0.352 | 0.024 | 0.393 | ||
| 1:30 | 30 | 0.208 | 0.441 | 0.040 | 0.668 | |
| 60 | 0.417 | 0.438 | 0.033 | 0.558 | ||
| 90 | 0.625 | 0.437 | 0.017 | 0.285 | ||
| 120 | 0.833 | 0.435 | 0.019 | 0.314 | ||
| 1:45 | 30 | 0.208 | 0.489 | 0.025 | 0.410 | |
| 60 | 0.417 | 0.484 | 0.021 | 0.351 | ||
| 90 | 0.625 | 0.484 | 0.022 | 0.374 | ||
| 120 | 0.833 | 0.484 | 0.029 | 0.488 | ||
| 5 | 1:15 | 50 | 0.208 | 0.505 | 0.014 | 0.137 |
| 100 | 0.417 | 0.596 | 0.014 | 0.138 | ||
| 150 | 0.625 | 0.600 | 0.014 | 0.137 | ||
| 200 | 0.833 | 0.602 | 0.014 | 0.138 | ||
| 1:30 | 50 | 0.208 | 0.738 | 0.029 | 0.291 | |
| 100 | 0.417 | 0.744 | 0.029 | 0.290 | ||
| 150 | 0.625 | 0.747 | 0.030 | 0.303 | ||
| 200 | 0.833 | 0.747 | 0.025 | 0.251 | ||
| 1:45 | 50 | 0.208 | 0.800 | 0.038 | 0.382 | |
| 100 | 0.417 | 0.810 | 0.040 | 0.401 | ||
| 150 | 0.625 | 0.813 | 0.042 | 0.420 | ||
| 200 | 0.833 | 0.814 | 0.043 | 0.426 | ||
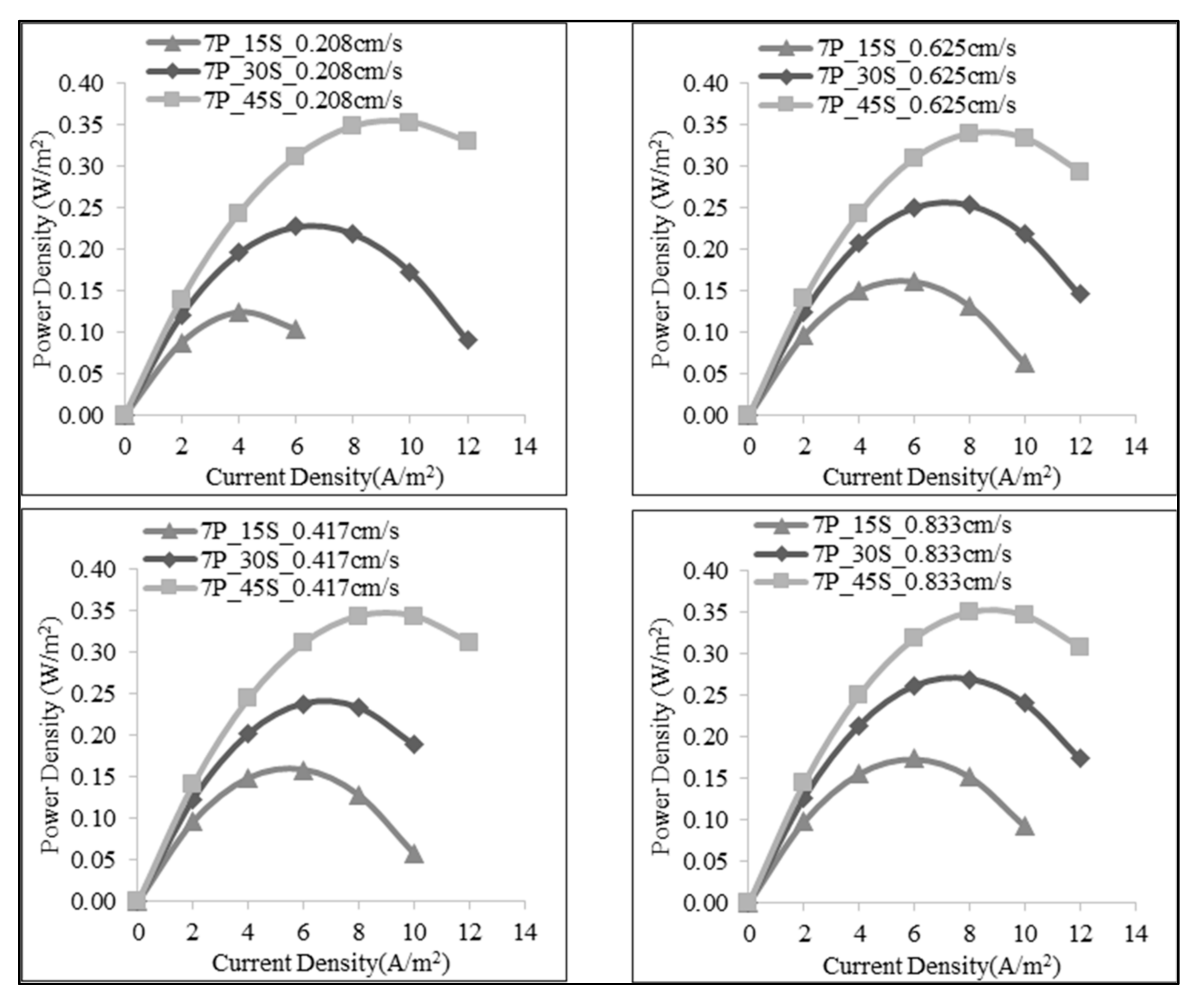
| 7 | 1:15 | 70 | 0.208 | 0.789 | 0.017 | 0.124 |
| 140 | 0.417 | 0.825 | 0.022 | 0.158 | ||
| 210 | 0.625 | 0.826 | 0.023 | 0.161 | ||
| 280 | 0.833 | 0.830 | 0.024 | 0.174 | ||
| 1:30 | 70 | 0.208 | 1.011 | 0.032 | 0.227 | |
| 140 | 0.417 | 1.018 | 0.033 | 0.238 | ||
| 210 | 0.625 | 1.023 | 0.036 | 0.254 | ||
| 280 | 0.833 | 1.027 | 0.038 | 0.269 | ||
| 1:45 | 70 | 0.208 | 1.106 | 0.049 | 0.352 | |
| 140 | 0.417 | 1.120 | 0.048 | 0.343 | ||
| 210 | 0.625 | 1.110 | 0.047 | 0.338 | ||
| 280 | 0.833 | 1.146 | 0.049 | 0.349 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altıok, E.; Kaya, T.Z.; Güler, E.; Kabay, N.; Bryjak, M. Performance of Reverse Electrodialysis System for Salinity Gradient Energy Generation by Using a Commercial Ion Exchange Membrane Pair with Homogeneous Bulk Structure. Water 2021, 13, 814. https://doi.org/10.3390/w13060814
Altıok E, Kaya TZ, Güler E, Kabay N, Bryjak M. Performance of Reverse Electrodialysis System for Salinity Gradient Energy Generation by Using a Commercial Ion Exchange Membrane Pair with Homogeneous Bulk Structure. Water. 2021; 13(6):814. https://doi.org/10.3390/w13060814
Chicago/Turabian StyleAltıok, Esra, Tuğçe Zeynep Kaya, Enver Güler, Nalan Kabay, and Marek Bryjak. 2021. "Performance of Reverse Electrodialysis System for Salinity Gradient Energy Generation by Using a Commercial Ion Exchange Membrane Pair with Homogeneous Bulk Structure" Water 13, no. 6: 814. https://doi.org/10.3390/w13060814
APA StyleAltıok, E., Kaya, T. Z., Güler, E., Kabay, N., & Bryjak, M. (2021). Performance of Reverse Electrodialysis System for Salinity Gradient Energy Generation by Using a Commercial Ion Exchange Membrane Pair with Homogeneous Bulk Structure. Water, 13(6), 814. https://doi.org/10.3390/w13060814









