Potential Use of the Benthic Foraminifers Bulimina denudata and Eggerelloides advenus in Marine Sediment Toxicity Testing
Abstract
1. Introduction
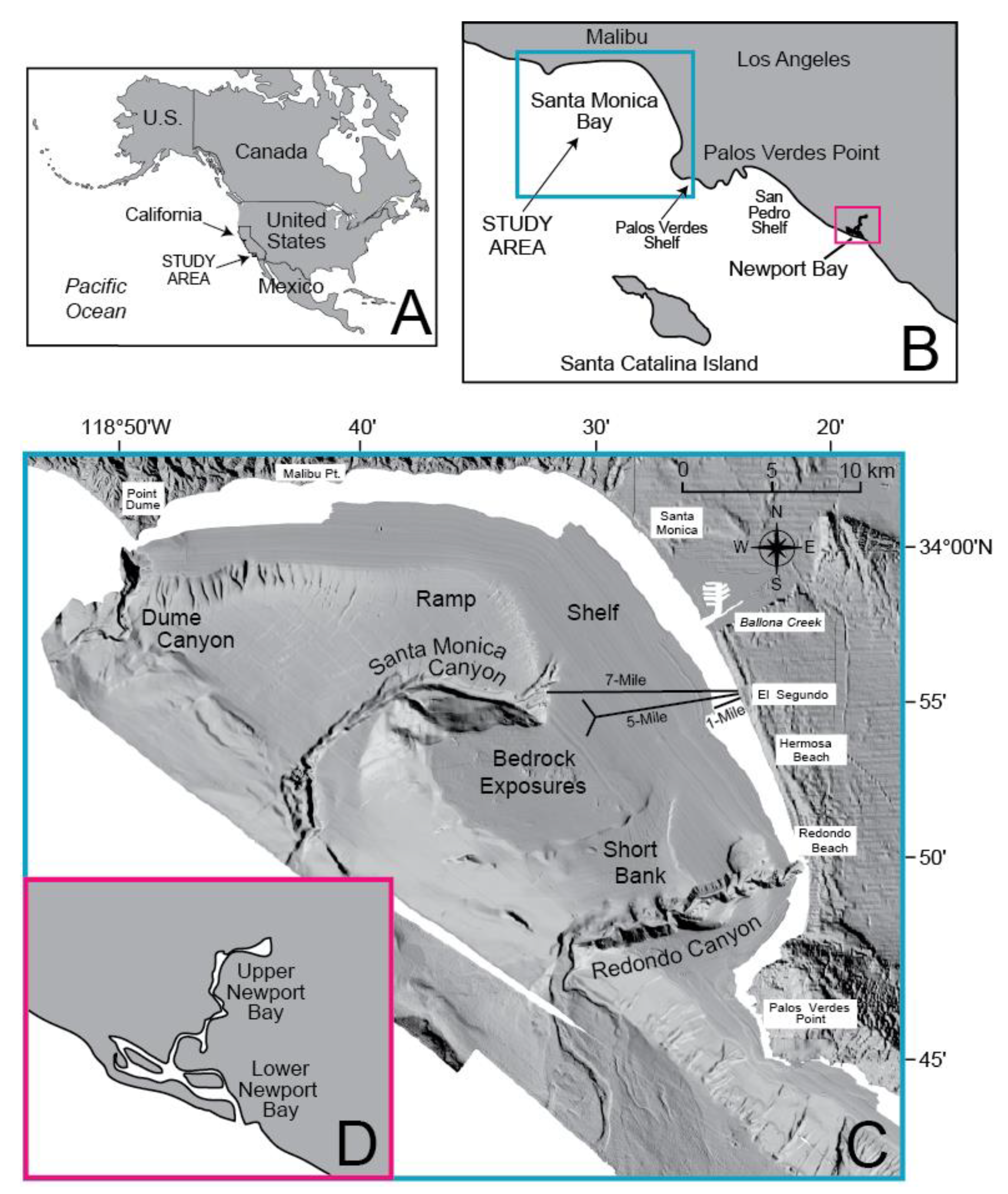
2. Setting
3. Materials and Methods
3.1. Field Sampling
3.2. Chronology
3.3. Grain Size
3.4. Foraminifera
3.5. Macrofaunal Toxicity Testing
3.6. Chemical Analysis
3.7. Data Analysis
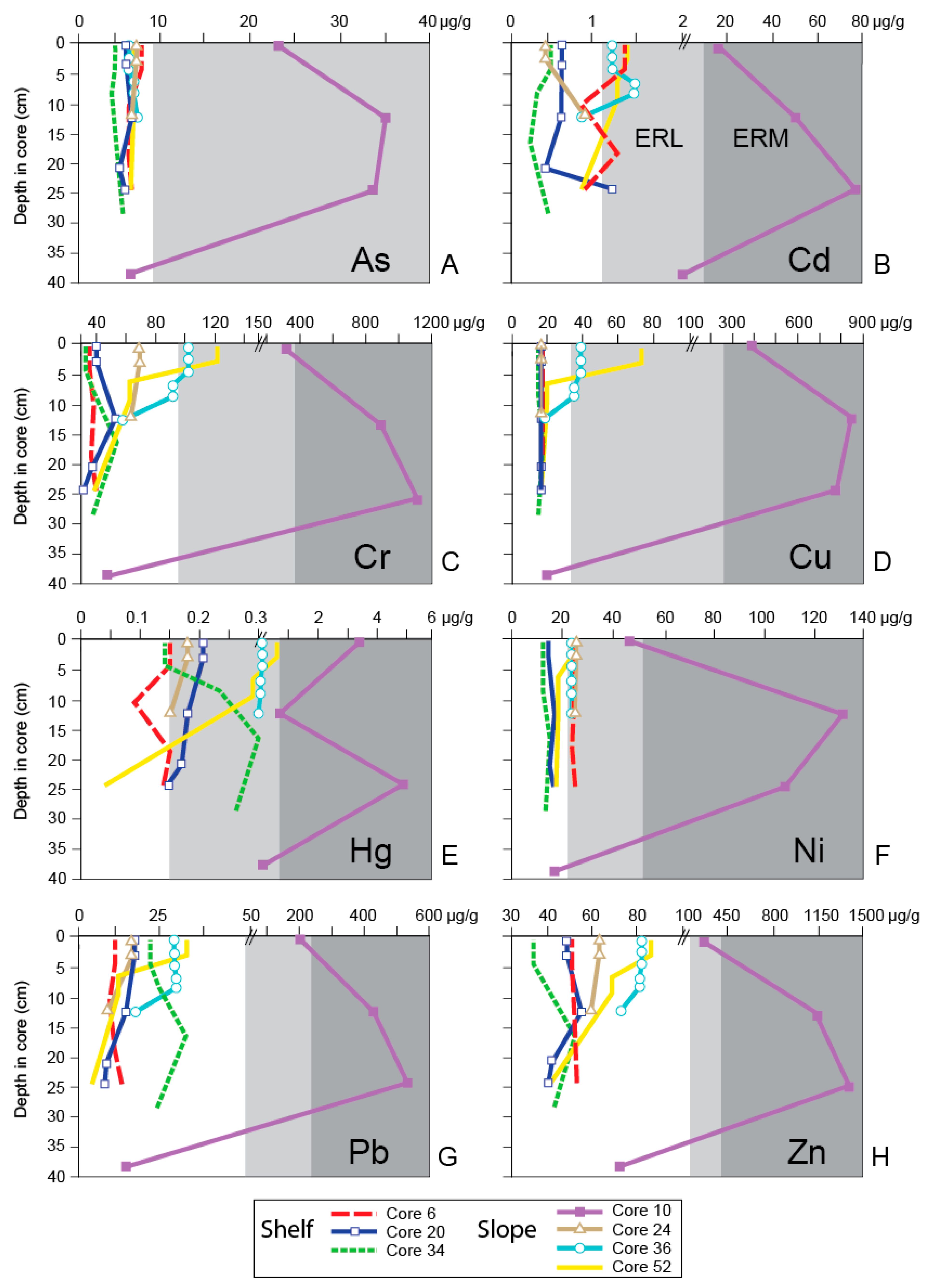
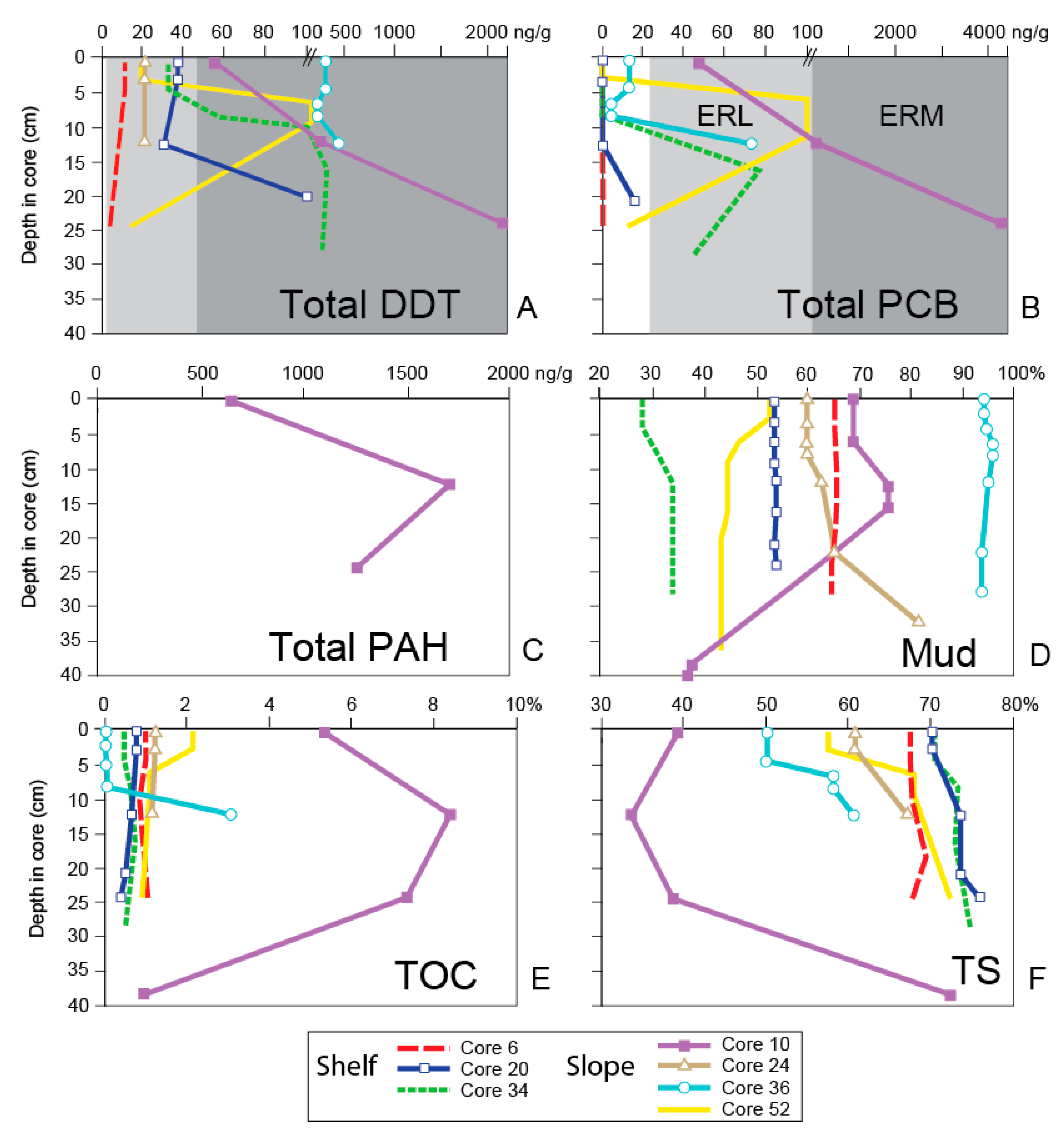
4. Results
5. Discussion
5.1. General Trends of Toxicity in Santa Monica Bay
5.2. Temporal Trends of Toxicity in Santa Monica Bay
5.3. Bulimina denudata and Eggerelloides advenus
5.4. Macrofaunal Toxicity Measures and Foraminiferal Abundance
5.5. Use of Foraminifera as Toxicity Measures
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| No., Station, Sample Interval (cm) | Mud Content (%) | % Sea Urchin Fertilization | % Amphipod Survival | % Bulimina denudata | % Eggerelloides advenus | % TS | % TOC | Unionized Ammonia (mg/L) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (1) 4, 0–2 | 24.75 | 115 | 91 | 1 | 23 | 73.5 | 0.185 | 0.136 | ||||||
| (2) 6, 0–2 | 65.52 | 98 | <68> | 6 | 46 | 67.6 | 0.990 | 0.072 | ||||||
| (3) 6, 4–5 | 65.52 | 2 | 66 | 67.6 | 0.990 | 0.072 | ||||||||
| (4) 6, 10–11 | 65.99 | 94 | <71> | 21 | 2 | 68 | 0.858 | |||||||
| (5) 6, 14.5–15.5 | 65.99 | 97 | <78> | 3 | 69 | |||||||||
| (6) 6, 18–19 | 65.52 | 98 | 94 | 17 | <0.5 | 69.5 | 0.016 | |||||||
| (7) 6, 24–25 | 65.05 | 99 | <71> | 12 | 28 | 67.9 | 1.065 | 0.013 | ||||||
| (8) 6, 28–29 | 65.05 | 100 | <73> | 8 | 1 | |||||||||
| (9) 10, 0–2 | 69.31 | <28> | 97 | 28 | 7 | 39.1 | 5.351 | 0.020 | ||||||
| (10) 10, 6–7 | 69.31 | <8> | <57> | VR | VR | |||||||||
| (11) 10, 12–13 | 75.93 | <18> | <68> | 7 | 3 | 33.6 | 8.393 | 0.008 | ||||||
| (12) 10, 15.5–16.5 | 75.93 | <15> | <50> | VR | VR | |||||||||
| (13) 10, 24–25 | 62.68 | <11> | <55> | 7 | <0.5 | 38.7 | 7.349 | 0.041 | ||||||
| (14) 10, 38–39 | 39.94 | <52> | <73> | 8 | 1 | 72.7 | 0.907 | 0.024 | ||||||
| (15) 10, 44–45 | 35.30 | 5 | 1 | |||||||||||
| (16) 16, 0–2 | 80.97 | 102 | 82 | 32 | 2 | 50.5 | 2.616 | 0.000 | ||||||
| (17) 18, 0–2 | 26.65 | 119 | 100 | 32 | 16 | 72.7 | 0.501 | 0.104 | ||||||
| (18) 20, 0–2 | 53.69 | 100 | 92 | 8 | 47 | 70.3 | 0.771 | 0.102 | ||||||
| (19) 20, 2.5–3.5 | 53.69 | 10 | 53 | 70.3 | 0.771 | 0.102 | ||||||||
| (20) 20, 6–7 | 53.69 | 112 | 94 | 36 | 22 | |||||||||
| (21) 20, 9–10 | 54.23 | 18 | 35 | |||||||||||
| (22) 20, 12–13 | 54.23 | 99 | <71> | 47 | 3 | 73.6 | 0.653 | 0.012 | ||||||
| (23) 20, 16–17 | 53.83 | 118 | 97 | 16 | 26 | |||||||||
| (24) 20, 20–22 | 53.83 | 102 | <73> | 40 | 4 | 73.9 | 0.507 | 0.003 | ||||||
| (25) 20, 24–25 | 54.33 | 95 | 94 | 11 | 3 | 76.1 | 0.399 | 0.003 | ||||||
| (26) 22, 0–2 | 47.35 | 118 | 83 | 17 | 22 | 72.6 | 0.649 | 0.028 | ||||||
| (27) 24, 0–2 | 60.29 | 111 | 100 | 27 | 6 | 60.8 | 1.224 | 0.031 | ||||||
| (28) 24, 2.5–3.5 | 60.29 | 26 | 9 | 60.8 | 1.224 | 0.031 | ||||||||
| (29) 24, 6–7 | 60.29 | 107 | 106 | 42 | 1 | |||||||||
| (30) 24, 7.5–8.5 | 60.29 | 25 | 7 | |||||||||||
| (31) 24, 12–13 | 63.02 | 115 | 102 | 33 | 2 | 67.2 | 1.154 | 0.011 | ||||||
| (32) 24, 22–23 | 65.72 | 118 | <74> | 17 | 1 | |||||||||
| (33) 24, 32–33 | 81.71 | 5 | 0 | |||||||||||
| (34) 28, 0–2 | 39.46 | 101 | 86 | 4 | 1 | 62.7 | 1.421 | 0.014 | ||||||
| (35) 30, 0–2 | 111 | 106 | 16 | 3 | 69.2 | 0.988 | 0.018 | |||||||
| (36) 33, 0–2 | 68.78 | 99 | 86 | 27 | 11 | 66.6 | 0.724 | 0.032 | ||||||
| (37) 34, 0–2 | 28.47 | 101 | 88 | 21 | 20 | 70.5 | 0.459 | 0.023 | ||||||
| (38) 34, 1–2 | 28.47 | 15 | 24 | 70.5 | 0.459 | 0.023 | ||||||||
| (39) 34, 3–4 | 28.47 | 23 | 21 | 70.5 | 0.459 | 0.023 | ||||||||
| (40) 34, 4–5 | 28.47 | 36 | 4 | 70.5 | 0.459 | 0.023 | ||||||||
| (41) 34, 8–9 | 31.67 | 31 | 104 | 40 | 2 | 73.4 | 0.634 | 0.006 | ||||||
| (42) 34, 12–13 | 34.15 | 101 | 93 | 17 | 21 | |||||||||
| (43) 34, 16–17 | 34.15 | 102 | 96 | 32 | 4 | 73 | 0.736 | 0.025 | ||||||
| (44) 34, 20–21 | 34.15 | 102 | 104 | 21 | 16 | |||||||||
| (45) 34, 22–23 | 34.30 | 99 | <73> | 18 | 11 | |||||||||
| (46) 34, 28–29 | 34.30 | 102 | 84 | 24 | 6 | 74.9 | 0.516 | 0.007 | ||||||
| (47) 36, 0–2 | 94.84 | 117 | <38> | 22 | 8 | 50.2 | 0.017 | 0.002 | ||||||
| (48) 36, 2–3 | 94.84 | 29 | 6 | 50.2 | 0.017 | 0.002 | ||||||||
| (49) 36, 4–5 | 94.84 | 43 | 2 | 50.2 | 0.017 | 0.002 | ||||||||
| (50) 36, 6.5–7 | 96.01 | 20 | 10 | 58.3 | 0.062 | 0.002 | ||||||||
| (51) 36, 8–9 | 96.01 | 113 | 108 | 30 | 1 | 58.3 | 0.062 | 0.002 | ||||||
| (52) 36, 12–13 | 95.62 | 113 | 111 | 25 | 1 | 60.9 | 3.076 | 0.005 | ||||||
| (53) 36, 22–23 | 93.70 | 110 | 91 | 5 | <0.5 | |||||||||
| (54) 36, 27–29 | 93.70 | 117 | <74> | 3 | 1 | |||||||||
| (55) 42, 0–2 | 62.15 | 101 | 96 | 20 | 31 | 66.1 | 0.783 | |||||||
| (56) 44, 0–2 | 66.48 | 95 | 84 | 14 | 36 | 67.7 | 1.076 | 0.003 | ||||||
| (57) 48, 0–2 | 56.59 | 118 | 97 | 9 | 9 | 57.5 | 1.547 | 0.069 | ||||||
| (58) 49, 0–2 | 42.36 | 117 | 85 | 4 | 40 | 72.4 | 0.588 | 0.011 | ||||||
| (59) 50, 0–2 | 13.64 | 101 | 93 | 21 | 24 | 71.8 | 0.702 | 0.011 | ||||||
| (60) 51, 0–2 | 34.87 | 102 | 82 | 12 | 2 | 60.2 | 1.812 | 0.004 | ||||||
| (61) 52, 0–2 | 52.93 | 117 | <74> | 11 | 2 | 57.6 | 2.144 | 0.018 | ||||||
| (62) 52, 2.7–3.7 | 52.93 | 10 | 2 | 57.6 | 2.144 | 0.018 | ||||||||
| (63) 52, 6–7 | 46.96 | 115 | 108 | 16 | 0 | 68 | 1.081 | 0.020 | ||||||
| (64) 52, 9–10 | 44.79 | 5 | <0.5 | 68 | 1.081 | 0.020 | ||||||||
| (65) 52, 10–11 | 44.79 | 114 | 89 | 6 | 0 | |||||||||
| (66) 52, 16–17 | 44.79 | 111 | <72> | 3 | 1 | |||||||||
| (67) 52, 20–21 | 43.62 | 117 | 83 | 3 | 1 | |||||||||
| (68) 52, 24–25 | 43.62 | 115 | <72> | 2 | 0 | 72.5 | 0.910 | |||||||
| (69) 52, 32–33 | 43.62 | 118 | <62> | 4 | 0 | |||||||||
| (70) 52–36–37 | 43.62 | 118 | 94 | 2 | 0 | |||||||||
| (71) 55, 0–2 | 32.55 | 100 | 106 | 7 | 14 | 68.1 | 0.707 | 0.021 | ||||||
| Trace Metals (µg/g dry) | Trace Organic Compounds (ng/g dry) | |||||||||||||
| No. | As | Be | Cd | Cr | Cu | Pb | Hg | Ni | Ag | Zn | Fe | Total DDT | Total PCBs | Total PAHs |
| (1) | 7.2 | 0.286 | 1.11 | 18.2 | <17 | 10.3 | 0.046 | 12 | <2.6 | 35.1 | 12,136 | (3.23) | 0.00 | 0.00 |
| (2) | 7.17 | 0.544 | (1.34) | 34.9 | <17 | 10.36 | (0.151) | (24.7) | <2.6 | 54.6 | 19,822 | (10.96) | 0.00 | 0.00 |
| (3) | 7.17 | 0.544 | (1.34) | 34.9 | <17 | 10.36 | (0.151) | (24.7) | <2.6 | 54.6 | 19,822 | (10.96) | 0.00 | 0.00 |
| (4) | 5.7 | 0.56 | 0.84 | 37.7 | <17 | 8.8 | 0.09 | (24.3) | <2.6 | 55.4 | 19,138 | |||
| (5) | ||||||||||||||
| (6) | 5.71 | 0.55 | (1.26) | 36.1 | <17 | 10.2 | 0.15 | (23.7) | <2.6 | 56.3 | 18,993 | |||
| (7) | 6.0 | 0.57 | 0.87 | 39.5 | <17 | 12.4 | 0.14 | (25.0) | <2.6 | 56.7 | 19,081 | (3.74) | 0.00 | 0.00 |
| (8) | ||||||||||||||
| (9) | (22.71) | 0.611 | [16.09] | (306.9) | [373.4] | (202.6) | [3.606] | (46.6) | [30.18] | (327.4) | 20,000 | [54.90] | (47.42) | [660] |
| (10) | ||||||||||||||
| (11) | (34.82) | 0.78 | [64.58] | (881) | [845.24] | [428.6] | [0.976] | [131.3] | [57.44] | [1148.8] | 17,857 | [236.93] | [285.06] | [1710] |
| (12) | ||||||||||||||
| (13) | (33.59) | 0.938 | [78.29] | [1113.7] | [782.95] | [532.3] | [5.065] | [108.3] | [59.43] | [1403.1] | 18,708 | [2148.74] | [4311.29] | [1263] |
| (14) | 5.7 | 0.51 | (2.59) | 47.0 | 18.4 | 12.9 | 0.38 | 16.8 | <2.6 | 74.3 | 16,374 | |||
| (15) | ||||||||||||||
| (16) | 6.7 | 0.59 | (1.60) | (155.5) | (100.6) | 37.6 | 0.70 | (28.7) | [12.3] | 101.0 | 20,398 | (28.08) | 0.00 | 0.00 |
| (17) | 5.16 | 0.38 | 1.09 | 41.3 | <17 | 14.3 | 0.122 | 14.7 | (2.96) | 49.8 | 17,607 | (18.80) | 0.00 | 0.00 |
| (18) | 5.39 | 0.395 | 0.61 | 39.4 | <17 | 16.1 | (0.206) | 14.2 | <2.6 | 52.6 | 15,932 | (37.03) | 0.00 | 0.00 |
| (19) | 5.39 | 0.395 | 0.61 | 39.4 | <17 | 16.1 | (0.206) | 14.2 | <2.6 | 52.6 | 15,932 | (37.03) | 0.00 | 0.00 |
| (20) | ||||||||||||||
| (21) | ||||||||||||||
| (22) | 6.0 | 0.50 | 0.60 | 52.6 | <17 | 13.5 | 0.18 | 16.6 | <2.6 | 58.6 | 16,438 | (30.08) | 0.00 | 0.00 |
| (23) | ||||||||||||||
| (24) | 4.7 | 0.43 | <0.42 | 36.1 | <17 | 7.9 | 0.17 | 14.8 | <2.6 | 46.7 | 15,271 | [106.92] | 16.22 | |
| (25) | 5.23 | 0.402 | (1.21) | 30.8 | <17 | 7.36 | 0.145 | 16.4 | <2.6 | 45.1 | 15,375 | |||
| (26) | 5.17 | 0.413 | 0.89 | 40.9 | <17 | 16.4 | (0.167) | 14.3 | <2.6 | 53.3 | 18,320 | (6.72) | 0.00 | 0.00 |
| (27) | 6.6 | 0.61 | <0.42 | 69.1 | <17 | 15.6 | 0.18 | (25.7) | <2.6 | 65.8 | 23,089 | (20.86) | 0.00 | 0.00 |
| (28) | 6.6 | 0.61 | <0.42 | 69.1 | <17 | 15.6 | 0.18 | (25.7) | <2.6 | 65.8 | 23,089 | (20.86) | 0.00 | 0.00 |
| (29) | ||||||||||||||
| (30) | ||||||||||||||
| (31) | 6.0 | 0.63 | 0.89 | 62.7 | <17 | 8.2 | 0.15 | (24.4) | <2.6 | 62.5 | 22,238 | (20.61) | 0.00 | 0.00 |
| (32) | ||||||||||||||
| (33) | ||||||||||||||
| (34) | (9.06) | 1.005 | <0.42 | (101.4) | 22.17 | 26 | (0.207) | 14.8 | <2.6 | 65.1 | 36,204 | [69.86] | 3.55 | 0.00 |
| (35) | 5.92 | 0.733 | (1.46) | 66.3 | 20.66 | 14.5 | (0.198) | 14.6 | <2.6 | 60.4 | 18,931 | [71.19] | 5.80 | 0.00 |
| (36) | 4.88 | 0.435 | 1.02 | 50.6 | <17 | 19.5 | (0.251) | 14 | <2.6 | 52.9 | 18,018 | [81.99] | 0.00 | 0.00 |
| (37) | 4.09 | 0.301 | 0.48 | 32.3 | <17 | 20.7 | 0.142 | 12.2 | <2.6 | 38.9 | 11,532 | (32.28) | 0.00 | 0.00 |
| (38) | 4.09 | 0.301 | 0.48 | 32.3 | <17 | 20.7 | 0.142 | 12.2 | <2.6 | 38.9 | 11,532 | (32.28) | 0.00 | 0.00 |
| (39) | 4.09 | 0.301 | 0.48 | 32.3 | <17 | 20.7 | 0.142 | 12.2 | <2.6 | 38.9 | 11,532 | (32.28) | 0.00 | 0.00 |
| (40) | 4.09 | 0.301 | 0.48 | 32.3 | <17 | 20.7 | 0.142 | 12.2 | <2.6 | 38.9 | 11,532 | (32.28) | 0.00 | 0.00 |
| (41) | 3.81 | 0.332 | 0.32 | 38.6 | <17 | 23.3 | (0.234) | 12.4 | <2.6 | 44.3 | 12,384 | [56.88] | 0.00 | 0.00 |
| (42) | ||||||||||||||
| (43) | 4.27 | 0.375 | 0.24 | 53.3 | 17.95 | 31.1 | 0.3 | 14.9 | <2.6 | 55.8 | 13,151 | [305.36] | (76.64) | |
| (44) | ||||||||||||||
| (45) | ||||||||||||||
| (46) | 4.95 | 0.326 | 0.45 | 36.7 | <17 | 22.7 | (0.262) | 13 | <2.6 | 47.5 | 12,270 | [257.17] | (44.83) | |
| (47) | 5.8 | 0.66 | 1.20 | (102.0) | (38.8) | 27.7 | 0.42 | (23.9) | <2.6 | 83.1 | 22,958 | [289.51] | 13.28 | 0.00 |
| (48) | 5.8 | 0.66 | 1.20 | (102.0) | (38.8) | 27.7 | 0.42 | (23.9) | <2.6 | 83.1 | 22,958 | [289.51] | 13.28 | 0.00 |
| (49) | 5.8 | 0.66 | 1.20 | (102.0) | (38.8) | 27.7 | 0.42 | (23.9) | <2.6 | 83.1 | 22,958 | [289.51] | 13.28 | 0.00 |
| (50) | 6.2 | 0.63 | (1.46) | (91.3) | (35.5) | 28.3 | 0.37 | (23.2) | <2.6 | 82.3 | 20,763 | [199.55] | 4.46 | 0.00 |
| (51) | 6.2 | 0.63 | (1.46) | (91.3) | (35.5) | 28.3 | 0.37 | (23.2) | <2.6 | 82.3 | 20,763 | [199.55] | 4.46 | 0.00 |
| (52) | 6.7 | 0.720 | 0.85 | 57.5 | <17 | 16.4 | 0.31 | (23.5) | <2.6 | 74.2 | 22,502 | [439.00] | (72.85) | |
| (53) | ||||||||||||||
| (54) | ||||||||||||||
| (55) | 5.7 | 0.401 | 0.74 | 43.1 | <17 | 16.5 | (0.274) | 14.8 | <2.6 | 57.2 | 15,885 | (35.14) | 0.00 | 0.00 |
| (56) | 5.88 | 0.437 | 0.97 | 47.3 | <17 | 17.6 | (0.312) | 15.5 | <2.6 | 61.2 | 16,839 | (43.74) | 6.79 | 0.00 |
| (57) | 8.0 | 0.61 | 1.15 | 72.7 | <17 | 27.5 | 0.39 | 19.5 | <2.6 | 73.2 | 18,203 | [106.96] | 2.41 | 0.00 |
| (58) | 4.1 | 0.304 | 0.86 | 27.1 | <17 | 29.3 | (0.196) | 11.5 | <2.6 | 46.4 | 12,983 | (25.32) | 4.44 | (103) |
| (59) | 4.21 | 0.286 | (1.36) | 38.7 | 19.08 | 9.6 | (0.322) | 10 | 0 | 50 | 12,033 | (21.24) | 3.20 | 0.00 |
| (60) | 6.08 | 0.374 | (3.46) | (116.1) | (78.24) | 39 | 0.62 | 19.4 | [5.7] | 86.4 | 16,611 | (22.60) | 6.53 | 0.00 |
| (61) | 6.1 | 0.64 | (1.37) | (121.5) | (72.6) | 31.4 | 0.90 | (23.4) | [6.3] | 87.0 | 21,995 | (18.82) | 0.00 | 0.00 |
| (62) | 6.1 | 0.64 | (1.37) | (121.5) | (72.6) | 31.4 | 0.90 | (23.4) | [6.3] | 87.0 | 21,995 | (18.82) | 0.00 | 0.00 |
| (63) | 6.2 | 0.66 | (1.3) | 62.4 | 20.3 | 11.3 | 0.29 | 18.4 | <2.6 | 70.9 | 20,921 | [141.43] | (113.78) | |
| (64) | 6.2 | 0.66 | (1.3) | 62.4 | 20.3 | 11.3 | 0.29 | 18.4 | <2.6 | 70.9 | 20,921 | [141.43] | (113.78) | |
| (65) | ||||||||||||||
| (66) | ||||||||||||||
| (67) | ||||||||||||||
| (68) | 5.9 | 0.61 | 0.84 | 37.9 | <17 | 3.7 | <0.04 | 17.1 | <2.6 | 45.8 | 20,629 | (13.77) | 12.28 | |
| (69) | ||||||||||||||
| (70) | ||||||||||||||
| (71) | 3.64 | 0.340 | (1.76) | 53.3 | 31.1 | 15.1 | (0.370) | 13.2 | (3.7) | 63.1 | 13,377 | (11.49) | 1.32 | 0.00 |
References
- Anderson, J.W.; Bay, S.M.; Thompson, B.E. Characteristics and effects of contaminated sediments from Southern California. Final Report to California State Water Resources Control Board. In Proceedings of the OCEANS, Westminster, CA, USA, 18–21 September 1988. [Google Scholar]
- Borja, A.; Bald, J.; Franco, J.; Larreta, J.; Muxika, I.; Reville, M.; Rodríguez, J.G.; Solaun, O.; Uriarte, A.; Valencia, V. Using multiple ecosystem components, in assessing ecological status in Spanish (Basque Country) Atlantic marine waters. Mar. Pollut. Bull. 2009, 59, 54–64. [Google Scholar] [CrossRef]
- Borja, A.; Galparsoro, I.; Irigoien, X.; Iriondo, A.; Menchaca, I.; Muxika, I.; Pascual, M.; Quincoces, I.; Revilla, M.; Germán Rodríquez, J.; et al. Implementation of the European Marine Strategy Framework Directive: A methodological approach for the assessment of environmental status, from the Basque Country (Bay of Biscay). Mar. Pollut. Bull. 2011, 62, 889–904. [Google Scholar] [CrossRef] [PubMed]
- Deets, G.B.; Cash, C.L. Soft-bottom macrofaunal communities in Santa Monica Bay: Phylogenetic approaches toward the elucidation of fine-scale biotic pattern and its physicochemical explanans. In Earth Science in the Urban Ocean: The Southern California Continental Borderland; Lee, H.J., Normark, W.R., Eds.; Special Paper 454; Geological Society of America: Boulder, CO, USA, 2009; pp. 457–481. [Google Scholar] [CrossRef]
- Greenstein, D.; Bay, S.; Jirik, A.; Brown, J.; Alexander, C. Toxicity assessment of sediment cores from Santa Monica Bay, California. Mar. Environ. Res. 2003, 56, 277–297. [Google Scholar] [CrossRef]
- Bay, S.M.; Wiborg, L.; Greenstein, D.J.; Haring, N.; Pottios, C.; Stransky, C.; Schiff, K. Southern California Bight 2013 Regional Monitoring Program: Volume 1. Sediment Toxicity; Southern California Coastal Water Research Project: Westminster, CA, USA, 2015. [Google Scholar]
- Parks, A.N.; Greenstein, D.J.; McLaughlin, K.; Schiff, K. Southern California Bight 2018 Regional Monitoring Program: Volume I. Sediment Toxicity; Southern California Coastal Water Research Project: Westminster, CA, USA, 2020. [Google Scholar]
- Alve, E. Benthic foraminifera response to estuarine pollution: A review. J. Foraminifer. Res. 1995, 25, 190–203. [Google Scholar] [CrossRef]
- Bandy, O.L.; Ingle, J.C., Jr.; Resig, J.M. Foraminifera, Los Angeles County Outfall area, California. Limnol. Oceanogr. 1964, 9, 124–137. [Google Scholar] [CrossRef]
- Bandy, O.L.; Ingle, J.C., Jr.; Resig, J.M. Foraminiferal trends, Laguna Beach Outfall area, California. Limnol. Oceanogr. 1964, 9, 112–123. [Google Scholar] [CrossRef]
- Bandy, O.L.; Ingle, J.C., Jr.; Resig, J.M. Facies trends, San Pedro Bay, California. Bull. Geol. Soc. Am. 1964, 75, 403–424. [Google Scholar] [CrossRef]
- Bandy, O.L.; Ingle, J.C., Jr.; Resig, J.M. Foraminiferal trends, Hyperion Outfall, California. Limnol. Oceanogr. 1965, 10, 314–332. [Google Scholar] [CrossRef]
- Bandy, O.L.; Ingle, J.C., Jr.; Resig, J.M. Modification of foraminiferal distribution by the Orange County Outfall, California. Ocean Sci. Ocean Eng. Mar. Technol. Soc. Trans. 1965, 9, 54–76. [Google Scholar]
- Bouchet, V.M.P.; Alve, E.; Rygg, B.; Telford, R.J. Benthic foraminifera provide a promising tool for ecological quality assessment of marine waters. Ecol. Indic. 2012, 23, 66–75. [Google Scholar] [CrossRef]
- Bouchet, V.M.P.; Goberville, E.; Frontalini, F. Benthic foraminifera to assess Ecological Quality Statuses in Italian transitional waters. Ecol. Indic. 2018, 84, 130–139. [Google Scholar] [CrossRef]
- Gómez-León, A.; Rodríguez-Figueroa, G.M.; Shumilin, E.; Carreño, A.L.; Sánchez, A. Abundance and distribution of benthic foraminifera as indicators of the quality of the sedimentary environment in a subtropical lagoon, Gulf of California. Mar. Pollut. Bull. 2018, 130, 31–39. [Google Scholar] [CrossRef]
- Martínez-Colón, M.; Hallock, P.; Green-Ruíz, C. Strategies for using shallow-water benthic foraminifers as bioindicators of potentially toxic elements: A review. J. Foraminifer. Res. 2009, 39, 278–299. [Google Scholar] [CrossRef]
- Martínez-Colón, M.; Hallock, P.; Green-Ruíz, C.R.; Smoak, J.M. Temporal variability in potentially toxic elements (PTE’s) and benthic Foraminifera in an estuarine environment in Puerto Rico. Micropaleontology 2017, 63, 357–381. [Google Scholar]
- Martínez-Colón, M.; Hallock, P.; Green-Ruíz, C.R.; Smoak, J.M. Benthic foraminifera as bioindicators of potentially toxic element (PTE) pollution: Torrecillas lagoon (San Juan Bay Estuary), Puerto Rico. Ecol. Indic. 2018, 89, 516–527. [Google Scholar] [CrossRef]
- Martins, V.; Ferreira da Silva, E.; Sequeira, C.; Rocha, F.; Duarte, A.C. Evaluation of the ecological effects of heavy metals on the assemblages of benthic foraminifera of the canals of Aveiro (Portugal). Estuar. Coast. Shelf Sci. 2010, 87, 93–304. [Google Scholar] [CrossRef]
- Martins, M.V.A.; Silva, F.; Laut, L.L.M.; Frontalini, F.; Clemente, I.M.M.M.; Miranda, P.; Figueira, R.; Sousa, S.H.M.; Dias, J.M.A. Response of benthic foraminifera to organic matter quantity and quality and bioavailable concentrations of metals in Aveiro Lagoon (Portugal). PLoS ONE 2015, 10, e0118077. [Google Scholar] [CrossRef]
- Martins, M.V.A.; Helali, M.A.; Zaaboub, N.; Omrane, I.B.-B.; Frontalini, F.; Reis, D.; Portela, H.; Clemente, I.M.M.M.; Nogueira, L.; Pereira, E.; et al. Organic matter quantity and quality, metals availability and foraminifera assemblages as environmental proxy applied to the Bizerte Lagoon (Tunisia). Mar. Pollut. Bull. 2016, 105, 161–179. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.V.A.; Hohenegger, J.; Martínez-Colón, M.; Frontalini, F.; Bergamashi, S.; Laut, L.; Belart, P.; Mahiques, M.; Pereira, E.; Rodrigues, R.; et al. Ecological quality status of the NE sector of the Guanabara Bay (Brazil): A case of living benthic foraminiferal resilience. Mar. Pollut. Bull. 2020, 158, 111449. [Google Scholar] [CrossRef]
- Resig, J.M. Foraminiferal ecology around ocean outfalls off Southern California. In Waste Disposal in the Marine Environment; Pearson, E.A., Ed.; Pergamon: London, UK, 1960; pp. 104–121. [Google Scholar]
- Resig, J.M. Chapter 7: Foraminifera. In An Oceanographic and Biological Survey of the Southern California Mainland Shelf; California State Water Quality Control Board: Sacramento, CA, USA, 1965; Volume 27, pp. 214–219; Appendix pp. 340–350. [Google Scholar]
- Romano, E.; Bergamin, L. Foraminifera and marine pollution. Mar. Pollut. Bull. 2009, 59, 219–368. [Google Scholar] [CrossRef]
- Yanko, V.; Kronfeld, J.; Flexer, A. Response of benthic foraminifera to various pollution sources: Implications for pollution monitoring. J. Foraminifer. Res. 1994, 24, 1–17. [Google Scholar] [CrossRef]
- Yanko, V.; Arnold, A.J.; Parker, W.C. Effects of marine pollution on benthic foraminifera. In Modern Foraminifera; Sen Gupta, B.K., Ed.; Kluwer Academic Publishers: London, UK, 1999; pp. 217–235. [Google Scholar]
- Watkins, J.G. Foraminiferal ecology around the Orange County, California, ocean sewer outfall. Micropaleontology 1961, 7, 199–206. [Google Scholar] [CrossRef]
- Zalesny, E.R. Foraminiferal ecology of Santa Monica Bay, California. Micropaleontology 1959, 5, 101–126. [Google Scholar] [CrossRef]
- Borja, A.; Franco, J.; Perez, V. A marine biotic index to establish the ecological quality of soft-bottom benthos within European estuarine and coastal environments. Mar. Pollut. Bull. 2000, 40, 1100–1114. [Google Scholar] [CrossRef]
- Dauvin, J.C.; Alizier, S.; Rolet, C.; Bakalem, A.; Bellan, G.; Gesteira, J.G.; Grimes, S.; De-La-Ossa-Carretero, J.; Del-Pilar-Ruso, Y. Response of different benthic indices to diverse human pressures. Ecol. Indic. 2012, 12, 143–153. [Google Scholar] [CrossRef]
- Hyland, J.; Balthis, L.; Karakassis, I.; Magni, P.; Petrov, A.; Shine, J.; Vestergaard, O.; Warwick, R. Organic carbon content of sediments as an indicator of stress in the marine benthos. Mar. Ecol. Prog. Ser. 2005, 295, 91–103. [Google Scholar] [CrossRef]
- Pearson, T.H.; Rosenberg, R. Macrobenthic succession in relation to organic enrichment and pollution in the marine environment. Annu. Rev. Oceanogr. Mar. Biol. 1978, 16, 229–311. [Google Scholar]
- Smith, R.W.; Bergen, M.; Weisberg, S.B.; Cadien, D.; Dalkey, A.; Montagne, D.; Stull, J.K.; Velarde, R.G. Benthic response index for assessing infaunal communities on the southern California mainland shelf. Ecol. Appl. 2001, 11, 1073–1087. [Google Scholar] [CrossRef]
- Lipps, J.H. Biotic interactions in benthic foraminifera. In Biotic Interactions in Recent and Fossil Benthic Communities; Trevesz, M.J.S., McCall, P.L., Eds.; Plenum Press: New York, NY, USA, 1983; pp. 331–376. [Google Scholar]
- Lipps, J.H.; Valentine, J.W. The role of foraminifera in the trophic structure of marine communities. Lethaia 1970, 3, 279–286. [Google Scholar] [CrossRef]
- Buzas, M.A. Foraminifera as prey for benthic deposit feeders: Results of predator exclusion experiments. J. Mar. Res. 1978, 36, 617–625. [Google Scholar]
- Lankford, R.R. Distribution and ecology of foraminifera from east Mississippi Delta margin. Am. Assoc. Pet. Geol. Bull. 1959, 43, 2068–2099. [Google Scholar]
- Sen Gupta, B.K. The benthonic foraminifera of the Tail of the Grand Banks. Micropaleontology 1971, 17, 69–98. [Google Scholar] [CrossRef]
- Buzas, M.A.; Culver, S.J. Species diversity and dispersal of foraminifera. BioScience 1991, 41, 483–489. [Google Scholar] [CrossRef]
- Anderson, O.R. Comparative Protozoology: Ecology, Physiology, Life History; Springer: Berlin/Heidelberg, Germany, 1988. [Google Scholar]
- Haynes, J.R. Foraminifera; John Wiley and Sons: New York, NY, USA, 1981. [Google Scholar]
- Lee, J.J.; Anderson, O.R. Biology of Foraminifera; Academic Press: London, UK, 1991. [Google Scholar]
- Southern California Bight Regional Monitoring Program Assessment Reports. Available online: http://www.sccwrp.org/Documents/BightDocuments.aspx (accessed on 12 January 2021).
- Chapman, P.M.; McDonald, B.G. Using the Sediment Quality Triad (Sqt) in ecological risk assessment. In Small-Scale Freshwater Toxicity Investigations; Blaise, C., Férard, J.F., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 305–329. [Google Scholar]
- Riba, I.; Forja, J.M.; Gómez-Parra, A.; DelValls, T.A. Sediment quality in littoral regions of the Gulf of Cádiz: A triad approach to address the influence of mining activities. Environ. Pollut. 2004, 132, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Borja, A.; Elliott, M.; Andersen, J.H.; Berg, T.; Carstensen, J.; Halpern, B.S.; Heiskanen, A.-S.; Korpinen, S.; Stewart Lowndes, J.S.; Martin, G.; et al. Overview of integrative assessment of marine systems: The ecosystem approach in practice. Front. Mar. Sci. 2016, 3. [Google Scholar] [CrossRef]
- McGann, M. Review of impacts of contaminated sediment on microfaunal communities in the Southern California Bight. In Earth Science in the Urban Ocean: The Southern California Continental Borderland; Lee, H.J., Normark, W.R., Eds.; Special Paper 454; Geological Society of America: Boulder, CO, USA, 2009; pp. 413–455. [Google Scholar]
- McGann, M.; Alexander, C.R.; Bay, S.M. Response of benthic foraminifers to sewage discharge and remediation in Santa Monica Bay, California. Mar. Environ. Res. 2003, 56, 299–342. [Google Scholar] [CrossRef]
- Stott, L.D.; Hayden, T.P.; Griffith, J. Benthic foraminifera at the Los Angeles County Whites Point outfall revisited. J. Foraminifer Res. 1996, 26, 357–368. [Google Scholar] [CrossRef]
- Murray, J.W. Ecology and Paleoecology of Benthic Foraminifera; John Wiley and Sons: New York, NY, USA, 1991. [Google Scholar]
- Oki, K. Vertical distribution of benthic foraminifera and mercury in cores from the south Yatsushiro Sea, Kyushu, Japan. In Proceedings of the Third International Congress on Environmental Micropaleontology, Microbiology, and Meiobenthology, Calgary, AB, Canada, 1–6 September 2002; Yanko-Hombach, V., Ed.; The International Society of Environmental Micropaleontology, Microbiology, and Meiobenthology: Winnipeg, MB, Canada, 2002; pp. 154–156. [Google Scholar]
- Rifardi, R. Distribution of sediment, benthic foraminifera and mercury in the South Yatsushiro Sea, Kyushu, Japan. J. Coast. Dev. 2008, 11, 104–112. [Google Scholar]
- Hayward, W.B.; Grenfell, H.R.; Sabaaa, A.T.; Cartera, R.; Cochran, U.; Lipps, J.H.; Shaned, P.R.; Morleya, M.S. Micropaleontological evidence of large earthquakes in the past 7200 years in southern Hawke’s Bay, New Zealand. Quat. Sci. Rev. 2006, 25, 1186–1207. [Google Scholar] [CrossRef]
- Lutze, G.F.; Colbourn, W.T. Recent benthic foraminifera from the continental margin of Northwest Africa, community structure and distribution. Mar. Micropaleontol. 1984, 8, 361–401. [Google Scholar] [CrossRef]
- Mojtahid, M.; Jorissen, R.; Durrieu, J.; Galgani, F.; Howa, H.; Redois, F.; Camps, R. Benthic foraminifera as bioindicators of drill cutting disposal in tropical east Atlantic outer shelf environments. Mar. Micropaleontol. 2006, 61, 58–75. [Google Scholar] [CrossRef]
- Debenay, J.P.; Redois, F. Distribution of the twenty-seven dominant species of shelf benthic foraminifers on the continental shelf, north of Dakar (Senegal). Mar. Micropaleontol. 1997, 29, 237–255. [Google Scholar] [CrossRef]
- Bates, J.M.; Spencer, R.S. Modification of foraminiferal trends by the Chesapeake-Elizabeth sewage outfall, Virginia Beach, Virginia. J. Foraminifer. Res. 1979, 9, 125–140. [Google Scholar] [CrossRef]
- Clark, R.B. Effects of aquaculture outfall on benthonic foraminifera in Clam Bay, Nova Scotia. Marit. Sediments 1971, 7, 76–84. [Google Scholar] [CrossRef]
- Schafer, C.T.; Cole, F.E. Distribution of benthonic foraminifera: Their use in delimiting local nearshore environments, Offshore Geology of Canada, Eastern Canada. Geol. Sur. Can. 1974, 74–30, 103–108. [Google Scholar]
- Schafer, C.T.; Young, J.A. Experiments on mobility and transportability of some nearshore benthonic foraminifera species. Geol. Sur. Can. 1977, 77, 27–31. [Google Scholar]
- Schafer, C.T. Foraminiferal colonization of an offshore dump site in Chaleur Bay, New Brunswick, Canada. J. Foraminifer. Res. 1982, 12, 317–326. [Google Scholar] [CrossRef]
- Dojiri, M. Chapter 1, Introduction. In Marine Monitoring in Santa Monica Bay: Biennial Assessment Report for the Period January 1995 through December 1996; City of Los Angeles, Department of Public Works, Bureau of Sanitation, Environmental Monitoring Division: Los Angeles, CA, USA, 1997; pp. 1–10. [Google Scholar]
- Alexander, C.R.; Venherm, C. Modern sedimentary processes in the Santa Monica, California continental margin: Sediment accumulation, mixing and budget. Mar. Environ. Res. 2003, 56, 177–204. [Google Scholar] [CrossRef]
- Alexander, C.R.; Smith, R.; Schropp, S.; Calder, S.; Windom, H. The historical record of metal enrichment in two Florida estuaries. Estuaries 1993, 16, 627–637. [Google Scholar] [CrossRef]
- Johannessen, S.C.; Macdonald, R.W. There is no 1954 in that core! Interpreting sedimentation rates and contaminant trends in marine sediment cores. Mar. Pollut. Bull. 2012, 64, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Alexander, C.; Lee, H. Sediment accumulation on the Southern California Bight continental margin during the twentieth century. In Earth Science in the Urban Ocean: The Southern California Continental Borderland; Lee, H.J., Normark, W.R., Eds.; Special Paper 454; Geological Society of America: Boulder, CO, USA, 2009; pp. 69–87. [Google Scholar]
- Drexler, J.Z.; Fuller, C.C.; Archfield, S. The approaching obsolescence of 137 Cs dating of wetland soils in North America. Quat. Sci. Rev. 2018, 199, 83–96. [Google Scholar] [CrossRef]
- Wang, J.; Baskaran, M.; Nierdermiller, J. Mobility of 137Cs in freshwater lakes: A mass balance and diffusion study of Lake St. Clair, southeast Michigan. Geochem. Cosmochim. Acta 2017, 218, 323–342. [Google Scholar] [CrossRef]
- Santschi, P.H.; Guo, L.; Asbill, S.; Allison, M.; Kepple, A.B.; Wen, L.-S. Accumulation rates and sources of sediments and organic carbon on the Palos Verdes shelf based on radioisotopic tracers (137Cs, 239,240 Pu, 210 Pb, 234Th, 238U and 14C). Mar. Chem. 2001, 73, 125–152. [Google Scholar] [CrossRef]
- Leonard-Pingel, J.S.; Kidwell, S.M.; Tomašových, A.; Alexander, C.R.; Cadien, D.B. Gauging benthic recovery from 20th century pollution on the southern California continental shelf using bivalves from sediment cores. Mar. Ecol. Prog. Ser. 2019, 615, 101–199. [Google Scholar] [CrossRef]
- Tomašových, A.; Kidwell, S.M.; Alexander, C.R.; Kaufman, D.S. Millennial-scale age offsets within fossil assemblages: Result of bioturbation below the taphonomic active zone and out-of-phase production. Paleocean. Paleoclim. 2019, 34, 954–977. [Google Scholar] [CrossRef]
- Zeng, E.Y.; Venkatesan, M.I. Dispersion of sediment DDTs in the coastal ocean off southern California. Sci. Total Environ. 1999, 229, 195–208. [Google Scholar] [CrossRef]
- Eganhouse, R.P.; Pontolillo, J. Depositional history of organic contaminants on the Palos Verdes Shelf, California. Mar. Chem. 2000, 70, 317–338. [Google Scholar] [CrossRef]
- Gambrell, R.P.; Reddy, C.N.; Collard, V.; Green, G.; Patrick, W.H. The recovery of DDT, kepone, and permethrin added to soil and sediment suspensions incubated under controlled redox potential and pH conditions. J. Water Pollut. Control Fed. 1984, 56, 174–182. [Google Scholar]
- Chiu, T.C.; Yen, J.H.; Liu, T.L.; Wang, Y.S. Anaeroic degradation of the organochlorine pesticides DDT and heptachlor in river sediment of Taiwan. Bull. Environ. Contam. Toxicol. 2004, 72, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Eggen, T.; Majcherczyk, A. Effects of zero-valent iron (Fe0) and temperature on the transformation of DDT and its metabolites in lake sediment. Chemosphere 2006, 62, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Connor, M.S.; Davis, J.A.; Leatherbarrow, J.; Greenfield, B.K.; Gunther, A.; Hardin, D.; Mumley, T.; Oram, J.J.; Werme, C. The slow recovery of San Francisco Bay from the legacy of organochlorine pesticides. Environ. Res. 2007, 105, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Eganhouse, R.P.; Pontolillo, J. Assessment of 1-Chloro-4-[2,2-Dichloro-1-(4- Chlorophenyl)Ethenyl]Benzene (DDE) Transformation Rates on the Palos Verdes Shelf; CA: US Geological Survey Open-File Report, 1362, 119; U.S. Geological Survey: Richmond, VA, USA, 2007; 119p.
- Bay, S.M.; Zeng, E.Y.; Lorenson, T.D.; Tran, K.; Alexander, C. Temporal and spatial distributions of contaminants in sediments of Santa Monica Bay, California. Mar. Environ. Res. 2003, 56, 255–276. [Google Scholar] [CrossRef]
- EPA (U.S. Environmental Protection Agency). EPA Bans PCB Manufacture; Phases Out Uses. 1979. Available online: https://archive.epa.gov/epa/aboutepa/epa-bans-pcb-manufacture-phases-out-uses.html (accessed on 22 February 2021).
- Tomašových, A.; Kidwell, S.M. Nineteenth-century collapse of a benthic marine ecosystem on the open continental shelf. Proc. R. Soc. B 2017, 284, 20170328. [Google Scholar] [CrossRef]
- usSEABED. Pacific Offshore Surficial-Sediment Data Release, Data Catalog, USGS Cruise S297SC. 2006. Available online: https://pubs.usgs.gov/ds/2006/182/data_cata.html (accessed on 12 January 2021).
- American Society for Testing and Materials. Standard guide for conducting 10-day static sediment toxicity tests with marine and estuarine amphipods. In 1996 Annual Book of ASTM Standards; American Society for Testing and Materials: West Conshohocken, PA, USA, 1996; pp. 769–794. [Google Scholar]
- U.S. Environmental Protection Agency. Methods for Assessing the Toxicity of Sediment-Associated Contaminants with Estuarine and Marine Amphipods; EPA-600/R-94/025; U.S. Environmental Protection Agency: Washington, DC, USA, 1994.
- Bay, S.M.; Greenstein, D.J.; Jirik, A.W.; Brow, J.S. Southern California Bight 1994 Pilot Project: VI. Sediment Toxicity; Southern California Coastal Water Research Project: Westminster, CA, USA, 1998. [Google Scholar]
- Long, E.R.; Field, L.J.; MacDonald, D.D. Predicting toxicity in marine sediments with numerical sediment quality guidelines. Environ. Toxicol. Chem. 1998, 17, 714–727. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities. An Approach to Statistical Analysis and Interpretation, 2nd ed.; Primer-E. Ltd.: Plymouth, UK, 2001. [Google Scholar]
- Clarke, K.R.; Ainsworth, M. A method of linking multivariate community structure to environmental variables. Mar. Ecol. Prog. Ser. 1993, 92, 205–219. [Google Scholar] [CrossRef]
- Somerfield, P.J.; Clarke, K.R. Inverse analysis in non-parametric multivariate analyses: Distinguishing groups of associated species which covary coherently across samples. J. Exper. Mar. Biol. Ecol. 2013, 449, 261–273. [Google Scholar] [CrossRef]
- Long, E.R.; MacDonald, D.D.; Smith, S.L.; Calder, F.D. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ. Manag. 1995, 19, 81–97. [Google Scholar] [CrossRef]
- Swartz, R.C.; Cole, F.A.; Lamberson, J.O.; Ferraro, S.P.; Schults, D.W.; Deben, W.A.; Lee II, H.; Ozretich, R.J. Sediment toxicity, contamination and amphipod abundance at a DDT- and Dieldrin-contaminated site in San Francisco Bay. Environ. Toxicol. Chem. 1994, 13, 949–962. [Google Scholar] [CrossRef]
- Raco-Rands, V.E.; Steinberger, A. Characteristics of effluents from large municipal wastewater treatment facilities in 1997. In Southern California Coastal Water Research Project Annual Report 1999–2000; Weisberg, S.B., Elmore, D., Eds.; Southern California Coastal Water Research Project: Westminster, CA, USA, 2000; pp. 28–44. [Google Scholar]
- Axtmann, E.V.; Luoma, S.N. Large-scale distribution of metal contamination in the fine-grained sediments of the Clark Fork River, Montana, U.S.A. Appl. Geochem. 1991, 6, 75–88. [Google Scholar] [CrossRef]
- Owen, M.R. Phosphorus Contamination and Storage in Bottom Sediments of the James River arm of Table Rock Lake, Southwest Missouri. Ph.D. Thesis, Southwest Missouri State University, Springfield, MI, USA, 2003. [Google Scholar]
- Stone, M.; Droppo, I.G. In-channel surficial fine-grained sediment laminae. Part II: Chemical characteristics and implications of contaminant transport in fluvial systems. Hydrol. Process. 1994, 8, 113–124. [Google Scholar] [CrossRef]
- Sommerfield, C.K.; Lee, H.J. Magnitude and variability of Holocene sediment accumulation in Santa Monica Bay, California. Mar. Environ. Res. 2003, 56, 151–176. [Google Scholar] [CrossRef]
- Inman, D.L.; Jenkins, S.A. Climate change and the episodicity of sediment flux of small California rivers. J. Geol. 1999, 107, 251–270. [Google Scholar] [CrossRef]
- Noble, M.A.; Ryan, H.F.; Wiberg, P.L. The dynamics of subtidal poleward flows over a narrow continental shelf, Palos Verdes, CA. Cont. Shelf Res. 2003, 22, 923–944. [Google Scholar] [CrossRef]
- Lee, H.J.; Sherwood, C.R.; Drake, D.E.; Edwards, B.D.; Wong, F.; Hamer, M. Spatial and temporal distribution of contaminated, effluent-affected sediment on the Palos Verdes margin, Southern California. Cont. Shelf Res. 2003, 22, 859–880. [Google Scholar] [CrossRef]
- Carnahan, E.A.; Hoare, A.M.; Hallock, P.; Lidz, B.H.; Reich, C.D. Foraminiferal assemblages in Biscayne Bay, Florida, USA: Responses to urban and agricultural influence in a subtropical estuary. Mar. Pollut. Bull. 2009, 59, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Bay, S.M.; Greenstein, D.J.; Ranasinghe, J.A.; Diehl, D.W.; Fetscher, A.E. Sediment Quality Assessment Draft Technical Support Manual; Technical Report 582; Southern California Coastal Water Research Project: Costa Mesa, CA, USA, 2009. [Google Scholar]
- Chapman, P.M.; Ho, K.T.; Munns, W.R., Jr.; Solomon, K.; Weinstein, M.P. Issues in sediment toxicity and ecological risk assessment. Mar. Pollut. Bull. 2002, 44, 271–278. [Google Scholar] [CrossRef]
- Long, E.R.; Hong, C.B.; Severn, C.G. Relationships between acute sediment toxicity in laboratory tests and abundance and diversity of benthic infauna in marine sediments: A review. Environ. Toxicol. Chem. 2001, 20, 46–60. [Google Scholar] [CrossRef]
- Swartz, R.C.; Cole, F.A.; Schults, D.W.; DeBen, W.A. Ecological changes on the Palos Verdes Shelf near a large sewage outfall: 1980–1983. Mar. Ecol. Prog. Ser. 1986, 31, 1–13. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. Standard test method for measuring the toxicity of sediment-associated contaminants with estuarine and marine invertebrates. In 2014 Annual Book of ASTM Standards; ASTM International (ASTM): West Conshohocken, PA, USA, 2014; pp. 444–505. [Google Scholar]
- California Environmental Protection Agency. Overview of Freshwater and Marine Toxicity Tests: A Technical Tool for Ecological Risk Assessment; Ecotoxicology Unit Reproductive and Cancer Hazard Assessment Section, Office of Environmental Health Hazard Assessment: Sacramento, CA, USA, 2004; 147p.
- Mearns, A.J.; Word, J.Q. Forecasting Effects of Sewage Solids on Marine Benthic Communities; EPA/600/J-82/331 (NTIS PB83223792); U.S. Environmental Protection Agency: Washington, DC, USA, 1982.
- Kim, C.B. A Systematic Study of Marine Gammaridean Amphipoda from Korea. Ph.D. Thesis, Seoul National University, Seoul, Korea, 1991. [Google Scholar]
- Moore, P.G. A new species in the genus Grandidierella Coutière (Crustacea: Amphipoda) from an Australian solar salt-work. J. Nat. Hist. 1986, 20, 1393–1399. [Google Scholar] [CrossRef]
- Nipper, M.G.; Greenstein, D.J.; Bay, S.M. Short- and long-term sediment toxicity test methods with the amphipod Grandidierella japonica. Environ. Toxicol. Chem. 1989, 8, 1191–1200. [Google Scholar] [CrossRef]
- Boese, B.L.; Lamberson, J.O.; Swartz, R.C.; Ozretich, R.J. Photoinduced toxicity of fluoranthene to seven marine benthic crustaceans. Arch. Environ. Contam. Toxicol. 1997, 32, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Kohn, N.P.; Word, J.Q.; Niyogi, D.K.; Ross, L.T.; Dillon, T.; Moore, D.W. Acute toxicity of Ammonia to four species of marine amphipod. Mar. Environ. Res. 1994, 38, 1–15. [Google Scholar] [CrossRef]
- Lee, J.S.; Lee, K.T.; Park, G.S. Acute toxicity of heavy metals, tributyltin, ammonia, and polycyclic aromatic hydrocarbons to benthic amphipod Grandidierella japonica. Ocean Sci. J. 2005, 40, 61–66. [Google Scholar] [CrossRef]
- Lamberson, J.; Swartz, R.; Ozretich, R. Field validation of acute and chronic marine amphipod sediment toxicity tests with Grandidierella japonica. In Proceedings of the 17th SETAC Annual Meeting, Washington, DC, USA, 17–21 November 1996. [Google Scholar]
- Anderson, B.S.; Hunt, J.W.; Phillips, B.M.; Fairey, R.J.; Oakden, J.M.; Puckett, H.M.; Stephenson, M.; Tjeerdema, R.S.; Long, E.R.; Wilson, C.J.; et al. Sediment quality in Los Angeles Harbor: A triad assessment. Environ. Toxicol. Chem. 2001, 20, 359–370. [Google Scholar] [CrossRef]
- Greenstein, D.; Bay, S.M. Selection of Methods for Assessing Sediment Toxicity in California Bays and Estuaries; Annual Report; Southern California Coastal Water Research Project: Costa Mesa, CA, USA, 2011; pp. 119–136.
- Bay, S.M.; Lapota, D.; Anderson, J.; Armstrong, J.; Mikel, T.; Jirik, A.; Asato, S. Southern California Bight 1998 Regional Monitoring Program: IV. Sediment Toxicity; Southern California Coastal Water Research Project: Westminster, CA, USA, 2000. [Google Scholar]
- Anderson, B.; Hunt, J.; Tudor, S.; Newman, J.; Tjeerdema, R.; Fairy, R.; Oakden, J.; Bretz, C.; Wilson, C.; LaCaro, F.; et al. Chemistry, Toxicity and Benthic Community Conditions in Sediments of Selected Southern California Bays and Estuaries; Report to California State Water Resources Control Board; California State Water Resources Control Board: Sacramento, CA, USA, 1997.
- Lewis, M.A.; Campbell, J.G.; Harris, P.S.; Dantin, D.D.; Quarles, R.L.; Chancy, C.A. Field characterization of potential reference sediments in the Gulf of Mexico: Chemical and biological quality. Soil Sediment Contam. 2006, 15, 1–20. [Google Scholar] [CrossRef]
- McGee, B.L.; Fisher, D.J.; Yonkos, L.T.; Ziegler, G.P.; Turley, S. Assessment of sediment contamination, acute toxicity, and population viability of the estuarine amphipod Leptocheirus plumulosus in Baltimore Harbor, Maryland, USA. Environ. Toxico. Chem. 1999, 18, 2151–2160. [Google Scholar] [CrossRef]
- Podlesińska, W.; Dąborska, H. Amphipods in estuarine and marine quality assessment—A review. Oceanologia 2019, 61, 179–196. [Google Scholar] [CrossRef]
- Jonczyk, E.; Gilron, G.; Zajdlik, B. Sea urchin fertilization assay: An evaluation of assumptions related to sample salinity adjustment and use of natural and synthetic marine waters for testing. Environ. Toxicol. Chem. 2001, 20, 804–809. [Google Scholar] [CrossRef]
- Bay, S.M.; Greenstein, D.J.; Jacobe, M.; Barton, C.; Sakamoto, K.; Young, D.; Ritter, K.J.; Schiff, K.C. Southern California Bight 2008 Regional Monitoring Program: I. Sediment Toxicity; Southern California Coastal Water Research Project: Costa Mesa, CA, USA, 2011. [Google Scholar]
- U.S. Environmental Protection Agency. Short-Term Methods for Estimating the Chronic Toxicity of Effluents and Receiving Waters to West Coast Marine and Estuarine Organisms; Office of Research and Development EPA/600/R- 95/136; U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1995.
- Anderson, B.S.; Hunt, J.W.; Hester, H.; Phillips, B.M. Assessment of sediment toxicity at the sediment-water interface. In Techniques in Aquatic Toxicology; Ostrander, G.K., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1996; pp. 609–624. [Google Scholar]
- Jorissen, F.J.; Wittling, I.; Peypouquet, J.P.; Rabouille, C.; Relexans, J.C. Live benthic foraminiferal faunas off Cape Blanc, NW Africa; community structure and microhabitats. Deep Sea Res. Part I 1998, 45, 2157–2188. [Google Scholar] [CrossRef]
- Bergin, F.; Kucuksezgin, F.; Uluturhan, E.; Barut, I.F.; Meric, E.; Avsar, N.; Nazik, A. The response of benthic foraminifera and Ostracoda to heavy metal pollution in Gulf of Izmir (Eastern Aegean Sea). Estuar. Coast. Shelf Sci. 2006, 66, 368–386. [Google Scholar] [CrossRef]
- Becker, D.S.; Ginn, T.C. Effects of storage time on toxicity of sediments from Puget Sound, Washington. Environ. Toxicol. Chem. 1995, 14, 829–835. [Google Scholar] [CrossRef]
- Surface Water Ambient Monitoring Program. Measurement Quality Objectives for Acute and Chronic Marine Sediment Toxicity Test Methods. Available online: https://www.waterboards.ca.gov/water_issues/programs/swamp/swamp_iq/docs/marine_sediment_tox_mqo.pdf) (accessed on 16 January 2021).
- Geffard, O.; His, E.; Budzinski, H.; Chiffoleau, J.F.; Coynel, A.; Etcheber, H. Effects of storage method and duration on the toxicity of marine sediments to embryos of Crassostrea gigas oysters. Environ. Pollut. 2004, 129, 457–465. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency-U.S. Army Corps of Engineers. In Evaluation of Dredged Material Proposed for Discharge in Waters of the US—Testing Manual; EPA-823-B-98–004; US Environmental Protection Agency/Army Corps of Engineers: Washington, DC, USA, 1998. [Google Scholar]
- Sen Gupta, B.K.; Machain-Castillo, M.L. Benthic foraminifera in oxygen-poor habitats. Mar. Micropaleontol. 1993, 20, 183–201. [Google Scholar] [CrossRef]
- Martin, R.E. Taphonomy and temporal resolution of foraminiferal assemblages. In Modern Foraminifera; Sen Gupta, B.K., Ed.; Kluwer Academic Publishers: London, UK, 1999; pp. 281–298. [Google Scholar]
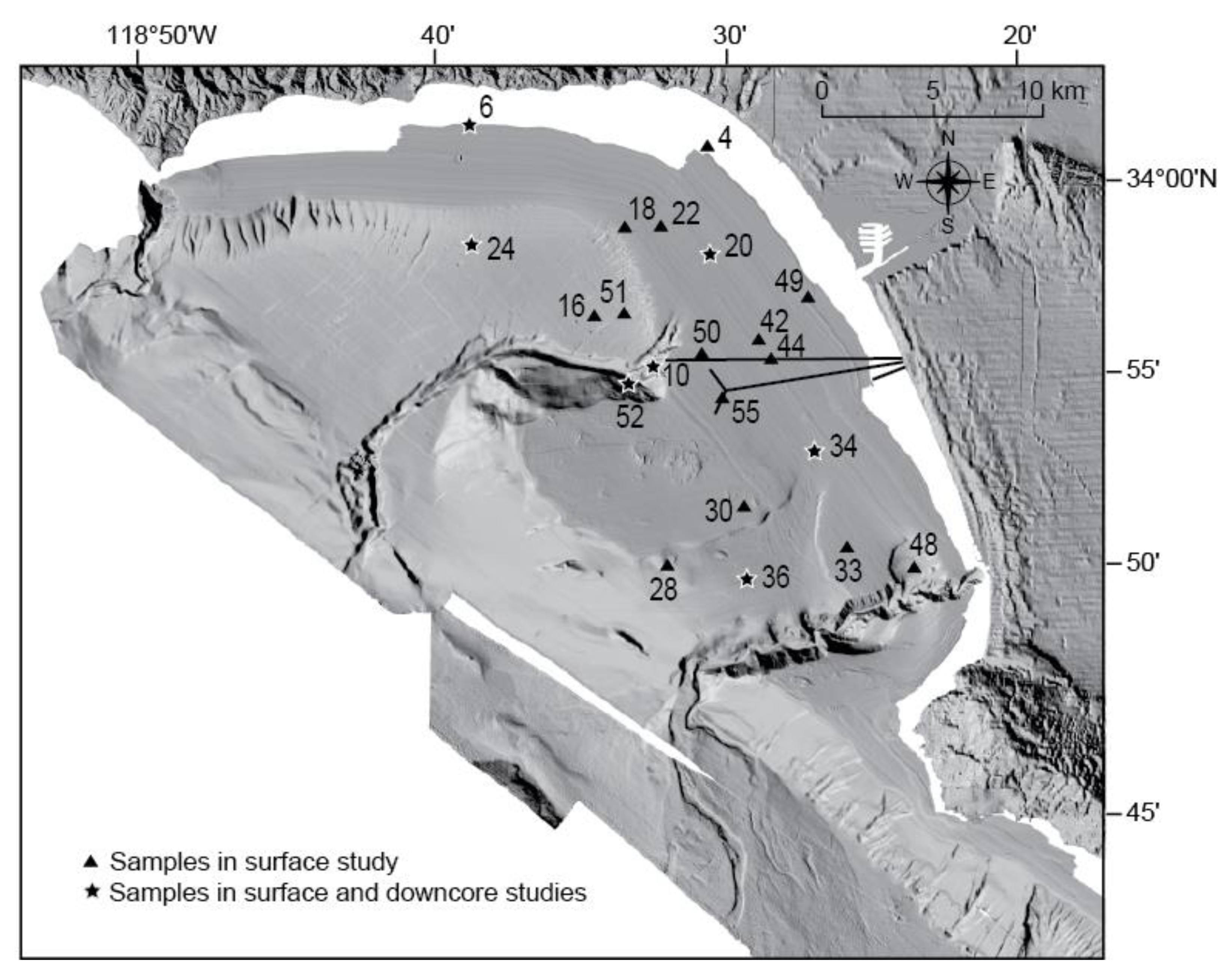
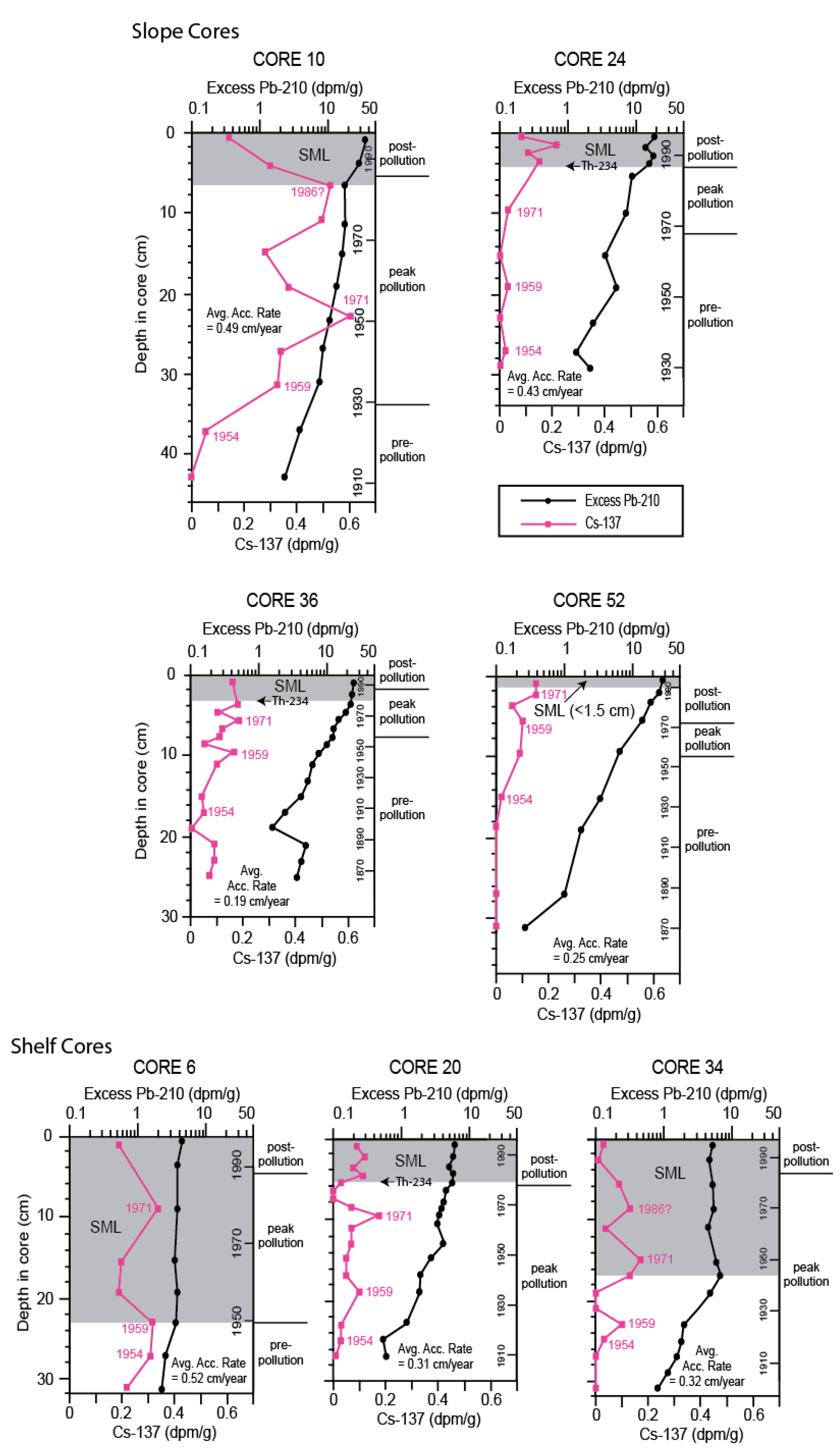
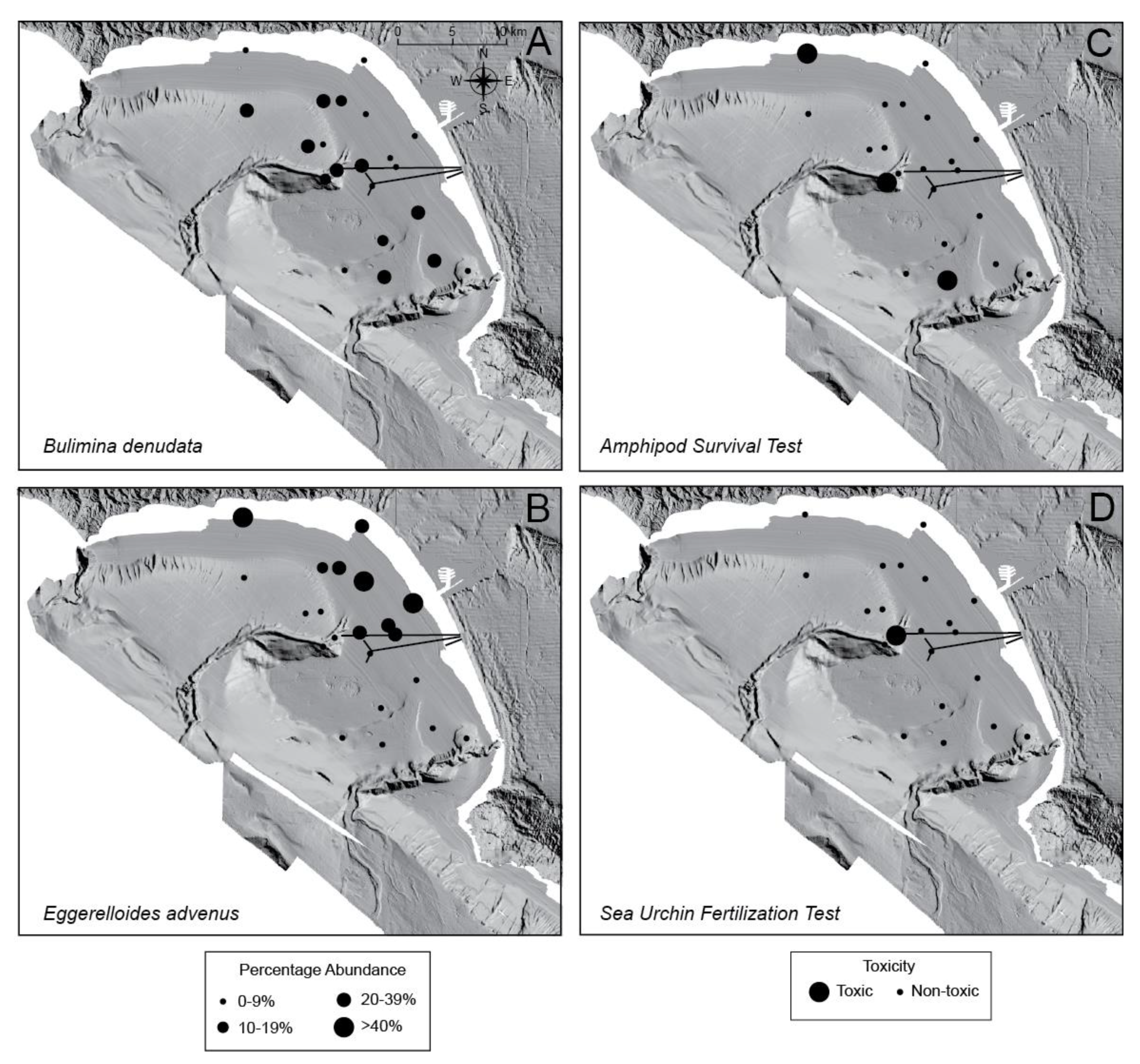


| Core | Latitude (°N) | Longitude (°W) | Water Depth (m) |
|---|---|---|---|
| 4 | 34.01350 | 118.53405 | 16 |
| 6 | 34.02117 | 118.65417 | 2 |
| 10 | 33.92133 | 118.56017 | 139 |
| 16 | 33.94183 | 118.58983 | 180 |
| 18 | 33.97933 | 118.57517 | 60 |
| 20 | 33.96883 | 118.53217 | 47 |
| 22 | 33.98017 | 118.55683 | 50 |
| 24 | 33.97117 | 118.65150 | 182 |
| 28 | 33.83850 | 118.55133 | 108 |
| 30 | 33.86333 | 118.51367 | 65 |
| 33 | 33.84683 | 118.46083 | 78 |
| 34 | 33.88667 | 118.47800 | 53 |
| 36 | 33.83333 | 118.51133 | 125 |
| 42 | 33.93333 | 118.50667 | 47 |
| 44 | 33.92483 | 118.50050 | 46 |
| 48 | 33.83850 | 118.42700 | 134 |
| 49 | 33.95033 | 118.48233 | 28 |
| 50 | 33.92783 | 118.53500 | 60 |
| 51 | 33.94283 | 118.57483 | 144 |
| 52 | 33.91383 | 118.57233 | 181 |
| 55 | 33.90900 | 118.52517 | 60 |
| Sediment Chemistry Analyte | Sediment Quality Guidelines | ||
|---|---|---|---|
| ERL Value (µg/g Dry Weight) | ERM Value (µg/g Dry Weight) | ||
| Trace Metals | Arsenic | 8.2 | 70 |
| Cadmium | 1.2 | 9.6 | |
| Chromium | 81 | 370 | |
| Copper | 34 | 270 | |
| Lead | 46.7 | 218 | |
| Mercury | 0.15 | 0.71 | |
| Nickel | 20.9 | 51.6 | |
| Silver | 1 | 3.7 | |
| Zinc | 150 | 410 | |
| Trace Organic Compounds | Total PAHs | 4022 | 44,792 |
| Total DDT | 1.58 | 46.1 | |
| Total PCBs | 22.7 | 180 | |
| Macrofaunal Toxicity Test | ||||||
|---|---|---|---|---|---|---|
| Samples | Number of Samples | Benthic Foraminiferal Species | Sea Urchin Fertilization | Amphipod Survival | ||
| rs | ps | rs | ps | |||
| Surface & Downcore | 67 | Bulimina denudata | −0.015 | >0.5 | 0.263 | [0.032] |
| Eggerelloides advenus | −0.252 | [0.040] | 0.022 | >0.5 | ||
| Surface & Downcore (except core 20) | 59 | Bulimina denudata | −0.028 | >0.5 | 0.330 | [0.011] |
| Eggerelloides advenus | −0.292 | [0.025] | −0.028 | >0.5 | ||
| Surface | 21 | Bulimina denudata | 0.012 | >0.5 | 0.129 | >0.5 |
| Eggerelloides advenus | −0.222 | 0.334 | 0.012 | >0.5 | ||
| Downcore | 46 | Bulimina denudata | −0.037 | >0.5 | 0.275 | 0.064 |
| Eggerelloides advenus | −0.340 | [0.021] | −0.010 | >0.5 | ||
| Downcore (except core 20) | 39 | Bulimina denudata | −0.041 | >0.5 | 0.350 | [0.029] |
| Eggerelloides advenus | −0.437 | [0.005] | −0.083 | >0.5 | ||
| Surface & Downcore (49 Samples) | Surface (21 Samples) | |||||||
| Bulimina denudata | Eggerelloides advenus | Bulimina denudata | Eggerelloides advenus | |||||
| Sediment Constituent | rs | ps | rs | ps | rs | ps | rs | ps |
| As | −0.296 | [0.039] | −0.285 | [0.048] | −0.120 | >0.5 | −0.438 | [0.047] |
| Be | −0.068 | >0.5 | −0.558 | [<0.001] | 0.154 | >0.5 | −0.592 | [0.005] |
| Cd | −0.326 | [0.022] | −0.217 | 0.134 | 0.127 | >0.5 | −0.334 | 0.139 |
| Cr | 0.126 | 0.387 | −0.436 | [0.002] | 0.362 | 0.107 | −0.832 | [<0.001] |
| Pb | 0.115 | 0.431 | −0.070 | >0.5 | 0.159 | 0.490 | −0.501 | [0.021] |
| Hg | 0.016 | >0.5 | −0.274 | 0.057 | 0.209 | 0.362 | −0.481 | [0.027] |
| Ni | 0.036 | >0.5 | −0.345 | [0.015] | 0.344 | 0.127 | −0.479 | [0.028] |
| Zn | 0.006 | >0.5 | −0.442 | [0.001] | 0.237 | 0.302 | −0.704 | [<0.001] |
| Fe | 0.025 | >0.5 | −0.426 | [0.002] | 0.230 | 0.316 | −0.631 | [0.002] |
| Total DDT | 0.294 | [0.041] | −0.183 | 0.209 | 0.217 | 0.346 | −0.257 | 0.260 |
| Total PCBs | −0.029 | >0.5 | −0.364 | [0.010] | −0.063 | >0.5 | −0.221 | 0.337 |
| Unionized Ammonia | −0.317 | [0.026] | 0.357 | [0.012] | −0.146 | >0.5 | 0.266 | 0.243 |
| TOC | −0.289 | [0.044] | −0.234 | 0.106 | 0.075 | >0.5 | −0.521 | [0.015] |
| % TS | 0.023 | >0.5 | 0.181 | 0.213 | −0.282 | 0.216 | 0.611 | [0.003] |
| % Mud | 0.116 | 0.354 | −0.113 | 0.354 | 0.370 | 0.108 | −0.143 | >0.5 |
| Downcore (28 Samples) | Downcore (Except Core 20) (24 Samples) | |||||||
| Bulimina denudata | Eggerelloides advenus | Bulimina denudata | Eggerelloides advenus | |||||
| Sediment Constituent | rs | ps | rs | ps | rs | ps | rs | ps |
| As | −0.388 | [0.041] | −0.258 | 0.185 | −0.452 | [0.026] | −0.203 | 0.341 |
| Be | −0.242 | 0.215 | −0.501 | [0.007] | −0.197 | 0.356 | −0.477 | [0.018] |
| Cd | −0.560 | [0.002] | −0.375 | [0.049] | −0.521 | [0.009] | −0.307 | 0.145 |
| Cr | −0.0004 | >0.5 | −0.308 | 0.111 | 0.008 | >0.5 | −0.271 | 0.201 |
| Pb | 0.132 | >0.5 | 0.131 | >0.5 | 0.204 | 0.338 | 0.184 | 0.389 |
| Hg | −0.019 | >0.5 | −0.242 | 0.215 | −0.001 | >0.5 | −0.212 | 0.321 |
| Ni | −0.252 | 0.195 | −0.167 | 0.396 | −0.215 | 0.312 | 0.002 | >0.5 |
| Zn | −0.130 | >0.5 | −0.352 | 0.066 | −0.137 | >0.5 | −0.306 | 0.146 |
| Fe | −0.042 | >0.5 | −0.319 | 0.098 | 0.088 | >0.5 | −0.257 | 0.225 |
| Total DDT | 0.270 | 0.164 | −0.050 | >0.5 | 0.304 | 0.180 | −0.315 | 0.164 |
| Total PCBs | −0.093 | >0.5 | −0.375 | [0.049] | −0.212 | 0.356 | −0.552 | [0.010] |
| Unionized Ammonia | −0.407 | [0.032] | 0.181 | 0.356 | −0.443 | [0.039] | 0.131 | >0.5 |
| TOC | −0.437 | [0.020] | −0.217 | 0.268 | −0.488 | [0.018] | −0.337 | 0.116 |
| % TS | 0.173 | 0.379 | 0.098 | >0.5 | 0.072 | >0.5 | 0.014 | >0.5 |
| % Mud | 0.022 | >0.5 | −0.077 | >0.5 | 0.019 | >0.5 | −0.101 | .0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGann, M. Potential Use of the Benthic Foraminifers Bulimina denudata and Eggerelloides advenus in Marine Sediment Toxicity Testing. Water 2021, 13, 775. https://doi.org/10.3390/w13060775
McGann M. Potential Use of the Benthic Foraminifers Bulimina denudata and Eggerelloides advenus in Marine Sediment Toxicity Testing. Water. 2021; 13(6):775. https://doi.org/10.3390/w13060775
Chicago/Turabian StyleMcGann, Mary. 2021. "Potential Use of the Benthic Foraminifers Bulimina denudata and Eggerelloides advenus in Marine Sediment Toxicity Testing" Water 13, no. 6: 775. https://doi.org/10.3390/w13060775
APA StyleMcGann, M. (2021). Potential Use of the Benthic Foraminifers Bulimina denudata and Eggerelloides advenus in Marine Sediment Toxicity Testing. Water, 13(6), 775. https://doi.org/10.3390/w13060775





