Finding Nano: Challenges Involved in Monitoring the Presence and Fate of Engineered Titanium Dioxide Nanoparticles in Aquatic Environments
Abstract
:1. Introduction
2. Distribution and Fate of Nano-TiO2 in the Environment
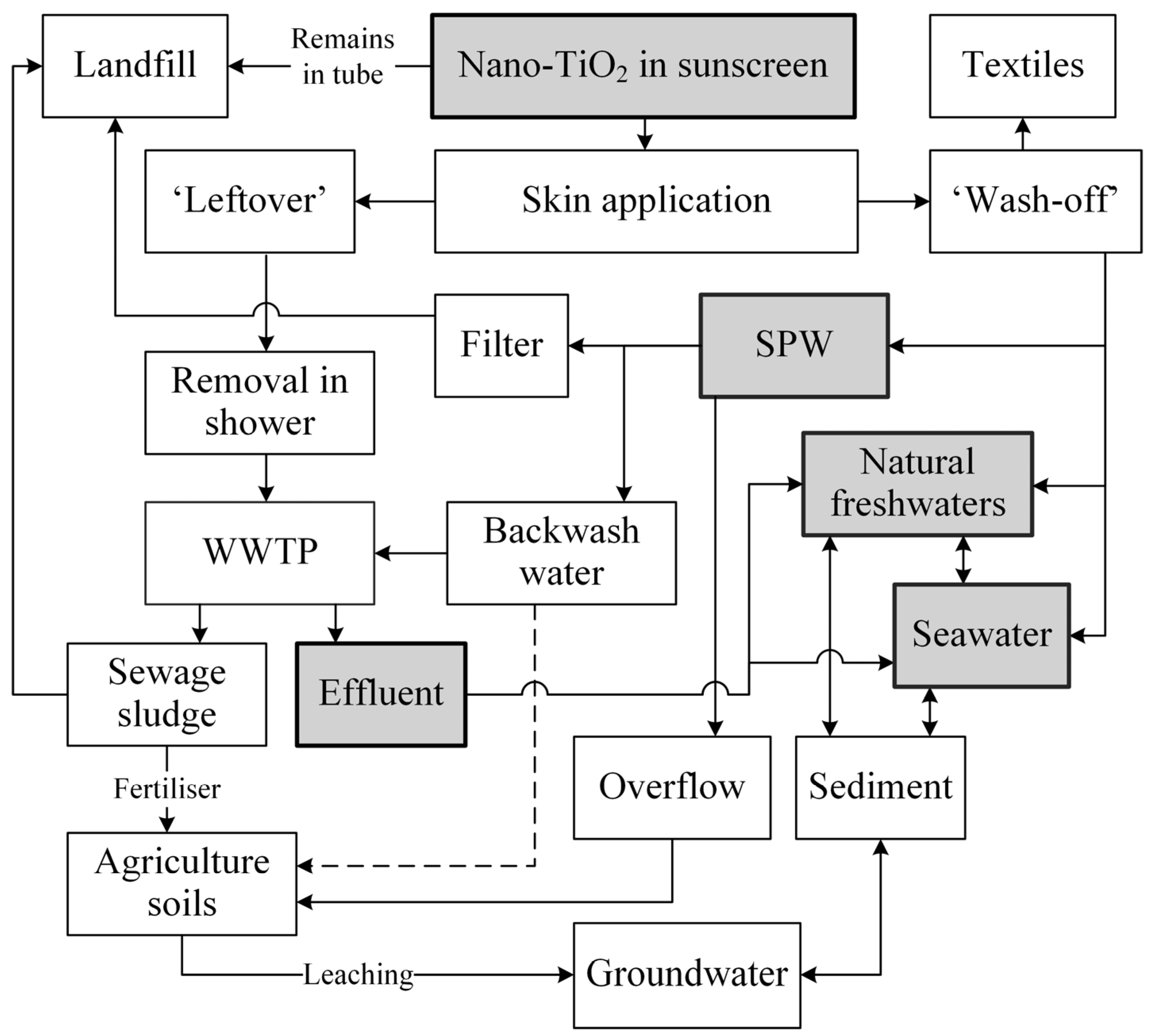
2.1. Exposure Pathways to the Environment
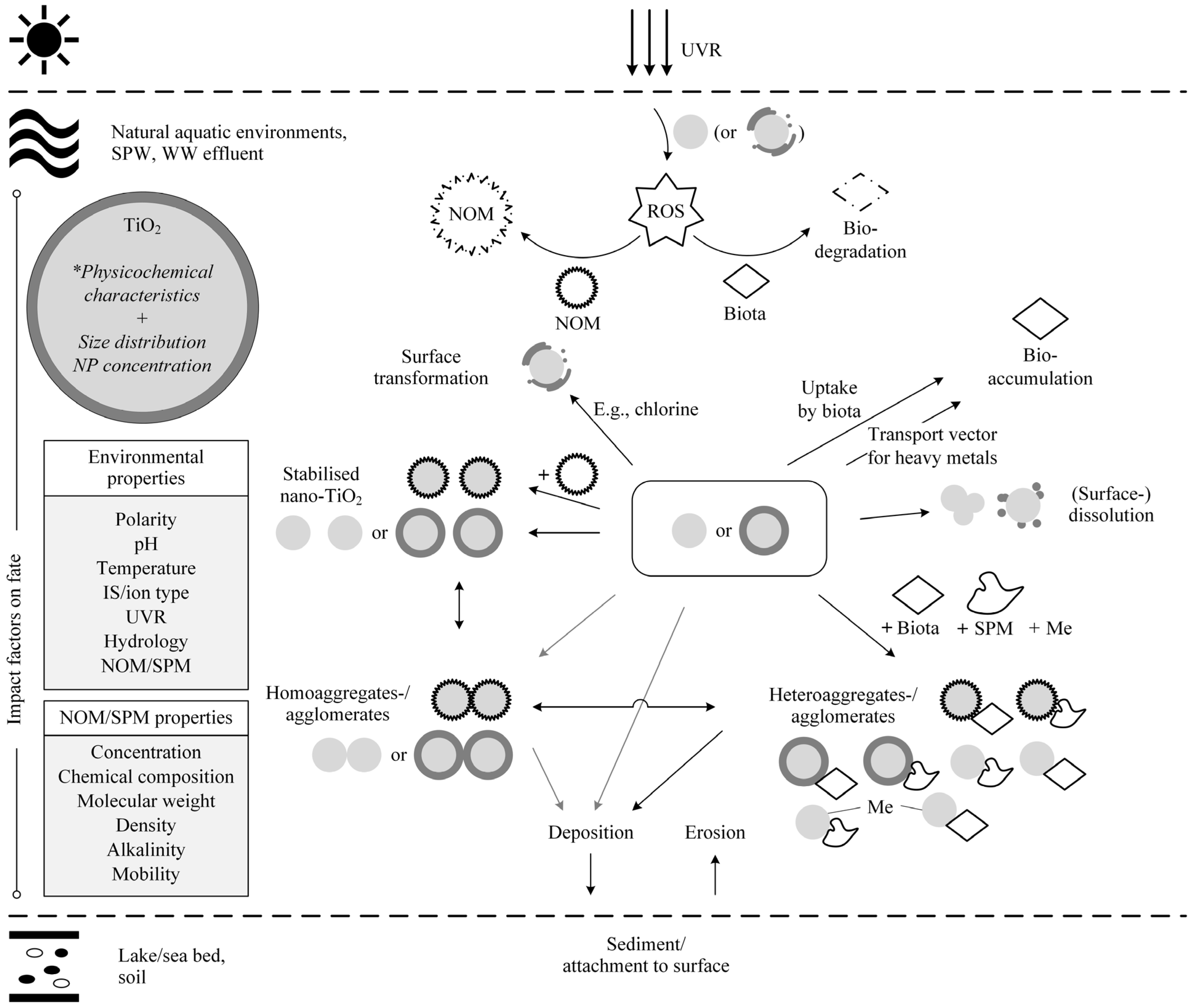
2.2. Behaviour and Fate of Nano-TiO2
2.2.1. The Pivotal Role of Nano-TiO2 Characteristics
2.2.2. The Influence of Environmental Conditions
2.2.3. Experimentally Observed Behaviour and Fate Processes
| Matrix | Exposure | Predominant Processes | Implication | Reference |
|---|---|---|---|---|
| Sunscreen | Oxybenzone, avobenzone, octyl methoxycinnamate | Photodegradation of organic UV filters | Joint toxicity | [87,88] |
| Acrylates/C10-30 alkyl acrylate cross-polymer (stabilising agent) | Adsorption of the polymer, decrease IEP from 6.3 to <5 | Electrostatic NP stabilization, enhanced NP mobility at environmentally relevant pH values (6–9), reduced filtration efficiency | [81] | |
| EtOH, UVR | Partial degradation of the hydrogen dimethicone coating due to the dissolving ability of EtOH | Photodegradation of the organic and subjacent layer: Al(OH)3 | [62] | |
| Hydroxy acids (Salicylic/citric acid) | Biomolecular chelation of Ti4+ | Induced solubilization by chelation | [8] | |
| Additional NPs in formulation | Higher phototoxicity | [14] | ||
| SPW | Calcium sulphate dihydrate (CaSO4 • 2 H2O) | Adsorption of Ca2+ increased zeta potential | Reduced colloidal stability | [65] |
| Chlorine | Surface transformation, redistribution of the Al(OH)3 protective layer | Reduced agglomeration, formation of porous aggregates, depletion of Al(OH)3 coating from the nano-TiO2 surface | [65] | |
| Surface transformation, redistribution of the Al(OH)3 protective layer | Depletion of Al(OH)3 coating from the nano-TiO2 surface, photocatalytic formation of hydroxyl radicals (•OH) | [12,64] | ||
| Potassium cetyl phosphate (C16H34KO4P, PCP) and chlorine | Degradation of PCP releases PO43− ions, subsequent formation of AlPO4 precipitates | Acceleration of protective coating depletion | [65] | |
| Change of physicochemical properties, e.g., smaller particles, more spherical | [64] | |||
| Dissolution, dominance of dissolved Ti species | [89] | |||
| Natural aquatic environment | UVR, seawater | Photocatalytic reactions | Formation of hydrogen peroxide (H2O2) | [90] |
| Shear forces (induced by light, sonication) | Partial disagglomeration | Enhanced NP mobility | [91] | |
| Higher ambient temperature | Hydrodynamic diameter increases | Enhanced NP sedimentation | [92] | |
| UVR + dissolved organic matter (DOM) (fresh-/seawater) | Photocatalytic halogenation of DOM | Formation of organobromine/-iodine compounds | [93] | |
| Natural clay colloids (SPM) (1) pH < IEP (2) pH > IEP | (1) Heteroaggregation (attractive electrostatic forces) (2) Weak affinity between NPs—SPM (both negatively charged) | Enhanced (1) NP aggregation (2) NP stabilisation | [86] | |
| HA (NOM) (pH < IEP) | Adsorption of macromolecules (1) at a low HA conc.: NP surface charge neutralisation; (2) at a high HA conc.: NP surface charge inversion | Enhanced (1) NP aggregation (2) NP stabilisation | [94] | |
| HA (NOM) (pH ≈ IEP) | Adsorption of macromolecules | Induced partial disaggregation | [94] | |
| HA, FA (NOM) (pH > IEP) | (1) IS < CCC: Adsorption of macromolecules (electrosteric repulsion) (2) IS > CCC: EDL compression + NOM entanglement | Enhanced (1) NP stabilisation (2) NP aggregation | [83] | |
| NOM (various) | Adsorption of macromolecules (1) low NOM conc.: interparticle bridging; (2) high NOM conc.: repulsive steric interactions | (1) Flocculation (dependent on NOM type, sometimes salt addition required) (2) steric NP stabilisation | [63] | |
| NOM | Adsorption of macromolecules | Photosensitiser AND ROS quencher (dominant) | [85] | |
| NaCl (Seawater) + Algae | Adsorption NPs—microalgae (note: NPs—NPs) | Intensified heteroagglomeration with increase in IS (note: homo-agglomeration is not affected by IS) | [95] | |
| NaCl (Seawater) (IS > CCC) | Aggregation | Induced NP aggregation | [96] | |
| UVR, seawater | Release of dissolved trace metals and inorganic nutrients | Greater bioavailability, bioaccumulation | [97] | |
| Siderophore-producing organisms | Biomolecular chelation of Ti4+ | Bioaccumulation, induced NP solubility | [8] | |
| Presence of Cu | Transport vector for heavy metals, Cu2+ absorption on TiO2 surface | Bioaccumulation of Cu | [98] | |
| Presence of Cd, As | Transport vector for heavy metals, Cd2+, AsO43- absorption on TiO2 surface | Bioaccumulation of Cd, As | [99] | |
| Aged TiO2 under UVR (in presence of Cd2+) | Production of singlet oxygen (1O2) | Higher sensitivity of cells to Cd | [100] |
3. An Analytical Challenge
- Reduction of polyatomic interferencesNew approaches in pre-treatments should be developed that can sufficiently digest the TiO2 sample without the use of acids that generate polyatomic spectral overlaps or require heavy safety measures like HF. Further developments in CCT technology could also reduce spectral interferences.
- Reduction of SDLSolely Hadioui et al. [116] have reached SDL as low as 19−44 nm for TiO2 in rainwater and 27 nm for TiO2 in SPW (Table 3). A low SDL is vital for detecting and accurately differentiating between small particles and the dissolved form [109,128]. If the resolution is too low, statements about the dominant form (i.e., dissolved or particulate) are not conclusive. Furthermore, adequately low SDLs are a prerequisite for effectively using (SP-)ICP-MS as a multi-element analysis [118].
- Distinction between NNPs and ENPs using multi-element analysisThe advanced development of the (SP-)ICP-MS technique is required to effectively distinguish between TiO2 inputs from natural and anthropogenic sources in various matrices. A promising approach is the calculation of elemental ratios on an SP basis [118]. However, this technique is still in its infancy. Moreover, a unique fingerprint of TiO2 ENPs or NNPs should be identified to conclusively distinguish between the two [107].
- Evaluating the coating degradation using multi-element analysisThe multi-element analysis could also be used to examine the potential deterioration of the particle coating. For example, Yamanaka et al. [117] have shown that Al and Ti (matrix: sunscreen), as well as Zn and Ti (matrix: SPW), can quantitatively and simultaneously be analysed using SP-ICP-MS. They verified the outcome by comparing the concentration results of the multi-element SP-ICP-MS analysis with the total metal concentration of microwave-digested samples. Future studies should investigate the suitability of this multi-element analysis for observing variations in the elemental composition on an SP basis.
4. Modelling as a Complementary Method to Analytics
- The differentiation between the form (e.g., single, aggregated, or agglomerated) of nano-TiO2 at the time of entry into the environment. A few models attempt to consider the transformation state; however, they merely manage to distinguish between pristine, dissolved, transformed (only core material), matrix-embedded, and product-embedded [147] or consider only certain situations, such as an aggregated form for particles originating from WW [146].
- The impact of manufactured and natural coatings on the fate and behaviour of nano-TiO2 [148].
- Heteroaggregation/-agglomeration processes, which are not explicitly expressed as a function of environmental (e.g., NOM, pH, IS) and nano-TiO2 properties (e.g., PSD, shape, crystallinity, coating) but rather indirectly included as part of the attachment efficiency/factor description [144,146]. Recently, the theoretical background and different experimental approaches used to study heteroaggregation have been thoroughly reviewed to at least improve the accuracy of attachment efficiency values of ENPs in natural surface waters [149].
- Environmental and nano-TiO2 properties have not been comprehensively ranked in terms of their relevancy in determining the behaviour in complex systems. Meesters et al. [150] attempted to identify the most important physicochemical properties affecting the environmental fate in their previously developed SB4N model [143]. However, available models have not yet been successful in explaining the complex systems’ behaviours adequately due to an oversimplification of the modelling assumptions.
- To forecast the ecological risks of silver (Ag) NP exposure in aquatic environments (FINE model). The model integrates modules describing particle behaviour, exposure potential, hazard, and risk and was developed using expert knowledge [154].
- To predict the hazard potential and biological effects of five ENMs within the framework of a human health RA. The developed BN uses data obtained from the literature and expert elicitations and includes selected physicochemical ENM properties, human exposure routes, and biological effects [155].
- To assess environmental multimedia distributions of ENMs (BN-nanoExpo) based on simulated data derived from a previously developed multimedia fate and transport model (MendNano) [156].
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- The Skin Cancer Foundation. Skin Cancer Facts & Statistics. 2020. Available online: https://www.skincancer.org/skin-cancer-data.information/skin-cancer-facts/ (accessed on 5 July 2019).
- WHO. Cancer. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 12 October 2020).
- Tabbakh, T.; Volkov, A.; Wakefield, M.; Dobbinson, S. Implementation of the SunSmart program and population sun protection behaviour in Melbourne, Australia: Results from cross-sectional summer surveys from 1987 to 2017. PLoS Med. 2019, 16, e1002932. [Google Scholar] [CrossRef]
- Adler, B.L.; DeLeo, V.A. Sunscreen safety: A Review of recent studies on humans and the environment. Curr. Dermatol. Rep. 2020, 9, 1–9. [Google Scholar] [CrossRef]
- Sobek, A.; Bejgarn, S.; Rudén, C.; Molander, L.; Breitholtz, M. In the shadow of the cosmetic directive—Inconsistencies in EU environmental hazard classification requirements for UV-filters. Sci. Total. Environ. 2013, 461–462, 706–711. [Google Scholar] [CrossRef]
- IARC. CAS No. 13463-67-7 Titanium Dioxide. 12/12/2019 ed.; International Agency for Research on Cancer: Lyon, France, 2010; Volume 93. [Google Scholar]
- Zhang, X.; Li, W.; Yang, Z. Toxicology of nanosized titanium dioxide: An update. Arch. Toxicol. 2015, 89, 2207–2217. [Google Scholar] [CrossRef]
- Sharma, S.; Sharma, R.K.; Gaur, K.; Torres, J.F.C.; Loza-Rosas, S.A.; Torres, A.; Saxena, M.; Julin, M.; Tinoco, A.D. Fueling a hot debate on the application of TiO2 nanoparticles in sunscreen. Materials 2019, 12, 2317. [Google Scholar] [CrossRef] [Green Version]
- Fenoglio, I.; Ponti, J.; Alloa, E.; Ghiazza, M.; Corazzari, I.; Capomaccio, R.; Rembges, D.; Oliaro-Bosso, S.; Rossi, F. Singlet oxygen plays a key role in the toxicity and DNA damage caused by nanometric TiO2 in human keratinocytes. Nanoscale 2013, 5, 6567–6576. [Google Scholar] [CrossRef]
- Mu, Q.; Jiang, G.; Chen, L.; Zhou, H.; Fourches, D.; Tropsha, A.; Yan, B. Chemical basis of interactions between engineered nanoparticles and biological systems. Chem. Rev. 2014, 114, 7740–7781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egambaram, O.P.; Pillai, S.K.; Ray, S.S. Materials science challenges in skin UV protection: A review. Photochem. Photobiol. 2020, 96, 779–797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Abed, S.R.; Virkutyte, J.; Ortenzio, J.N.R.; McCarrick, R.M.; Degn, L.L.; Zucker, R.; Coates, N.H.; Childs, K.; Ma, H.; Diamond, S.; et al. Environmental aging alters Al(OH)3 coating of TiO2 nanoparticles enhancing their photocatalytic and phototoxic activities. Environ. Sci. Nano 2016, 3, 593–601. [Google Scholar] [CrossRef] [Green Version]
- Auffan, M.; Pedeutour, M.; Rose, J.; Masion, A.; Ziarelli, F.; Borschneck, D.; Chanéac, C.; Botta, C.; Chaurand, P.; Labille, J.; et al. Structural degradation at the surface of a TiO2-based nanomaterial used in cosmetics. Environ. Sci. Technol. 2010, 44, 2689–2694. [Google Scholar] [CrossRef] [PubMed]
- Donia, D.T.; Carbone, M. Fate of the nanoparticles in environmental cycles. Int. J. Environ. Sci. Technol. 2018, 16, 583–600. [Google Scholar] [CrossRef]
- Minetto, D.; Libralato, G.; Ghirardini, A.V. Ecotoxicity of engineered TiO2 nanoparticles to saltwater organisms: An overview. Environ. Int. 2014, 66, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Picado, A.; Paixão, S.M.; Moita, L.; Silva, L.; Diniz, M.S.; Lourenço, J.; Peres, I.; Castro, L.; Correia, J.B.; Pereira, J.; et al. A multi-integrated approach on toxicity effects of engineered TiO2 nanoparticles. Front. Environ. Sci. Eng. 2015, 9, 793–803. [Google Scholar] [CrossRef] [Green Version]
- Scown, T.M.; Van Aerle, R.; Tyler, C.R. Review: Do engineered nanoparticles pose a significant threat to the aquatic environment? Crit. Rev. Toxicol. 2010, 40, 653–670. [Google Scholar] [CrossRef]
- De La Vega, A.C.S.; Cruz-Alcalde, A.; Mazón, C.S.; Martí, C.B.; Diaz-Cruz, M.S. Nano-TiO2 phototoxicity in fresh and seawater: Daphnia magna and Artemia sp. as proxies. Water 2020, 13, 55. [Google Scholar] [CrossRef]
- Barone, A.N.; Hayes, C.E.; Kerr, J.J.; Lee, R.C.; Flaherty, D.B. Acute toxicity testing of TiO2-based vs. oxybenzone-based sunscreens on clownfish (Amphiprion ocellaris). Environ. Sci. Pollut. Res. 2019, 26, 14513–14520. [Google Scholar] [CrossRef] [PubMed]
- Fouqueray, M.; Dufils, B.; Vollat, B.; Chaurand, P.; Botta, C.; Abacci, K.; Labille, J.; Rose, J.; Garric, J. Effects of aged TiO2 nanomaterial from sunscreen on Daphnia magna exposed by dietary route. Environ. Pollut. 2012, 163, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Hamzeh, M.; Dodard, S.; Zhao, Y.H.; Sunahara, G.I. Effects of TiO2 nanoparticles on ROS production and growth inhibition using freshwater green algae pre-exposed to UV irradiation. Environ. Toxicol. Pharmacol. 2015, 39, 1074–1080. [Google Scholar] [CrossRef] [PubMed]
- ChemSafetyPro. How to Calculate Predicted No-Effect Concentration (PNEC). Available online: https://www.chemsafetypro.com/Topics/CRA/How_to_Calculate_Predicted_No-Effect_Concentration_(PNEC).html (accessed on 16 July 2020).
- Musee, N. Simulated environmental risk estimation of engineered nanomaterials: A case of cosmetics in Johannesburg City. Hum. Exp. Toxicol. 2011, 30, 1181–1195. [Google Scholar] [CrossRef]
- Coll, C.; Notter, D.; Gottschalk, F.; Sun, T.; Som, C.; Nowack, B. Probabilistic environmental risk assessment of five nanomaterials (nano-TiO2, nano-Ag, nano-ZnO, CNT, and fullerenes). Nanotoxicology 2015, 10, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Slijkerman, D.; Onderzoeksformatie, I.; Keur, M. Sunscreen Ecoproducts: Product Claims, Potential Effects and Environmental Risks of Applied UV Filters; Wageningen University and Research: Den Helder, The Netherlands, 2018. [Google Scholar]
- Lead, J.R.; Batley, G.E.; Alvarez, P.J.J.; Croteau, M.-N.; Handy, R.D.; McLaughlin, M.J.; Judy, J.D.; Schirmer, K. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects-An updated review. Environ. Toxicol. Chem. 2018, 37, 2029–2063. [Google Scholar] [CrossRef] [PubMed]
- Baalousha, M.; Cornelis, G.; Kuhlbusch, T.A.J.; Lynch, I.; Nickel, C.; Peijnenburg, W.; Brink, N.W.V.D. Modeling nanomaterial fate and uptake in the environment: Current knowledge and future trends. Environ. Sci. Nano 2016, 3, 323–345. [Google Scholar] [CrossRef]
- Sun, T.Y.; Gottschalk, F.; Hungerbühler, K.; Nowack, B. Comprehensive probabilistic modelling of environmental emissions of engineered nanomaterials. Environ. Pollut. 2014, 185, 69–76. [Google Scholar] [CrossRef]
- Number of Products, vs. Titanium Dioxide; Nanodatabase: DTU Environment, the Danish Ecological Council and Danish Consumer Council, 2020.
- Jeon, S.-K.; Kim, E.-J.; Lee, J.; Lee, S. Potential risks of TiO2 and ZnO nanoparticles released from sunscreens into outdoor swimming pools. J. Hazard. Mater. 2016, 317, 312–318. [Google Scholar] [CrossRef]
- Choi, S.; Johnston, M.; Wang, G.-S.; Huang, C. A seasonal observation on the distribution of engineered nanoparticles in municipal wastewater treatment systems exemplified by TiO2 and ZnO. Sci. Total. Environ. 2018, 625, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- EPA, U. Nanomaterial Case Studies: Nanoscale Titanium Dioxide in Water Treatment and in Topical Sunscreen (Final); US Environmental Protection Agency Research Triangle Park: Washington, DC, USA, 2010.
- Wiechers, J.W.; Musee, N. Engineered inorganic nanoparticles and cosmetics: Facts, issues, knowledge gaps and challenges. J. Biomed. Nanotechnol. 2010, 6, 408–431. [Google Scholar] [CrossRef]
- Cornelis, G.; Hund-Rinke, K.; Kuhlbusch, T.; Van den Brink, N.; Nickel, C. Fate and bioavailability of engineered nanoparticles in soils: A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2720–2764. [Google Scholar] [CrossRef]
- Dulger, M.; Sakallioglu, T.; Temizel, I.; Demirel, B.; Copty, N.; Onay, T.; Uyguner-Demirel, C.; Karanfil, T. Leaching potential of nano-scale titanium dioxide in fresh municipal solid waste. Chemosphere 2016, 144, 1567–1572. [Google Scholar] [CrossRef] [PubMed]
- Morgan, R.; Swimming Pool Ownership Increases in Australia. Roy Morgan Research: 2018. Available online: http://www.roymorgan.com/findings/7811-australian-swimming-pool-ownership-september-2018-201811230555 (accessed on 27 May 2020).
- Nischwitz, V.; Goenaga-Infante, H. Improved sample preparation and quality control for the characterisation of titanium dioxide nanoparticles in sunscreens using flow field flow fractionation on-line with inductively coupled plasma mass spectrometry. J. Anal. At. Spectrom. 2012, 27, 1084–1092. [Google Scholar] [CrossRef]
- Dan, Y.; Shi, H.; Stephan, C.; Liang, X. Rapid analysis of titanium dioxide nanoparticles in sunscreens using single particle inductively coupled plasma-mass spectrometry. Microchem. J. 2015, 122, 119–126. [Google Scholar] [CrossRef]
- Popov, A.P.; Lademann, J.; Priezzhev, A.V.; Myllylä, R.A. Effect of size of TiO2 nanoparticles embedded into stratum corneum on ultraviolet-A and ultraviolet-B sun-blocking properties of the skin. J. Biomed. Opt. 2005, 10, 064037. [Google Scholar] [CrossRef]
- Walter, D. Primary Particles—Agglomerates—Aggregates. In Nanomaterials; Wiley: Bonn, Germany, 2013; pp. 9–24. [Google Scholar]
- Lin, X.; Li, J.; Ma, S.; Liu, G.; Yang, K.; Tong, M.; Lin, D. Toxicity of TiO2 nanoparticles to escherichia coli: Effects of particle size, crystal phase and water chemistry. PLoS ONE 2014, 9, e110247. [Google Scholar] [CrossRef] [Green Version]
- Solaiman, S.M.; Algie, J.; Bakand, S.; Sluyter, R.; Sencadas, V.; Lerch, M.; Huang, X.-F.; Konstantinov, K.; Barker, P.J. Nano-sunscreens—A double-edged sword in protecting consumers from harm: Viewing Australian regulatory policies through the lenses of the European Union. Crit. Rev. Toxicol. 2019, 49, 122–139. [Google Scholar] [CrossRef] [Green Version]
- Pelclova, D.; Navratil, T.; Kacerova, T.; Zamostna, B.; Fenclova, Z.; Vlckova, S.; Kacer, P. NanoTiO2 sunscreen does not prevent systemic oxidative stress caused by UV radiation and a minor amount of NanoTiO2 is absorbed in humans. Nanomater. 2019, 9, 888. [Google Scholar] [CrossRef] [Green Version]
- Almquist, C.B.; Biswas, P. Role of synthesis method and particle size of nanostructured TiO2 on its photoactivity. J. Catal. 2002, 212, 145–156. [Google Scholar] [CrossRef]
- Philippe, A.; Košík, J.; Welle, A.; Guigner, J.-M.; Clemens, O.; Schaumann, G.E. Extraction and characterization methods for titanium dioxide nanoparticles from commercialized sunscreens. Environ. Sci. Nano 2017, 5, 191–202. [Google Scholar] [CrossRef]
- Bairi, V.G.; Lim, J.-H.; Fong, A.; Linder, S.W. Size characterization of metal oxide nanoparticles in commercial sunscreen products. J. Nanoparticle Res. 2017, 19, 256. [Google Scholar] [CrossRef]
- Robertson, T.A.; Sanchez, W.Y.; Roberts, M.S. Are commercially available nanoparticles safe when applied to the skin? J. Biomed. Nanotechnol. 2010, 6, 452–468. [Google Scholar] [CrossRef] [PubMed]
- Catalano, R.; Labille, J.; Gaglio, D.; Alijagic, A.; Napodano, E.; Slomberg, D.; Campos, A.; Pinsino, A. Safety evaluation of TiO2 nanoparticle-based sunscreen UV filters on the development and the immunological state of the sea urchin Paracentrotus Lividus. Nanomaterials 2020, 10, 2102. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, I.-L.; Huang, Y.-J. Effects of various physicochemical characteristics on the toxicities of ZnO and TiO2 nanoparticles toward human lung epithelial cells. Sci. Total. Environ. 2011, 409, 1219–1228. [Google Scholar] [CrossRef]
- Lu, P.; Fang, S.; Cheng, W.; Huang, S.; Cheng, H. Characterization of titanium dioxide and zinc oxide nanoparticles in sunscreen powder by comparing different measurement methods. J. Food Drug Anal. 2018, 26, 1192–1200. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Hwang, H.-M. Engineered TiO2 Nanoparticles: Their Fate and Effects in Natural Aquatic Environments; Nova Science Publishers Inc.: Jackson, MS, USA, 2014; pp. 1–20. [Google Scholar]
- Johnston, H.J.; Hutchison, G.R.; Christensen, F.M.; Peters, S.; Hankin, S.; Stone, V. Identification of the mechanisms that drive the toxicity of TiO2 particulates: The contribution of physicochemical characteristics. Part. Fibre Toxicol. 2009, 6, 33. [Google Scholar] [CrossRef]
- Corinaldesi, C.; Marcellini, F.; Nepote, E.; Damiani, E.; Danovaro, R. Impact of inorganic UV filters contained in sunscreen products on tropical stony corals (Acropora spp.). Sci. Total Environ. 2018, 637–638, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Borm, P.J.; Robbins, D.; Haubold, S.; Kuhlbusch, T.; Fissan, H.; Donaldson, K.; Schins, R.; Stone, V.; Kreyling, W.; Lademann, J.; et al. The potential risks of nanomaterials: A review carried out for ECETOC. Part. Fibre Toxicol. 2006, 3, 11. [Google Scholar] [CrossRef] [Green Version]
- Wakefield, G.; Lipscomb, S.; Holland, E.; Knowland, J. The effects of manganese doping on UVA absorption and free radical generation of micronised titanium dioxide and its consequences for the photostability of UVA absorbing organic sunscreen components. Photochem. Photobiol. Sci. 2004, 3, 648–652. [Google Scholar] [CrossRef]
- Park, B.; Martin, A.P.; Harris, C.; Guest, R.; Whittingham, A.; Jenkinson, P. Preliminary in vitro investigation of the potential health effects of Optisol™, a nanoparticulate manganese modified titanium dioxide UV-filter used in certain sunscreen products. Nanotoxicology 2009, 3, 73–90. [Google Scholar] [CrossRef]
- Labille, J.; Catalano, R.; Slomberg, D.; Motellier, S.; Pinsino, A.; Hennebert, P.; Santaella, C.; Bartolomei, V. Assessing sunscreen lifecycle to minimize environmental risk posed by nanoparticulate UV-filters—A review for safer-by-design products. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Jacobs, J.F.; Van De Poel, I.; Osseweijer, P. Sunscreens with titanium dioxide (TiO2) nano-particles: A societal experiment. NanoEthics 2010, 4, 103–113. [Google Scholar] [CrossRef] [Green Version]
- Osterwalder, U.; Sohn, M.; Herzog, B. Global state of sunscreens. Photodermatol. Photoimmunol. Photomed. 2014, 30, 62–80. [Google Scholar] [CrossRef] [Green Version]
- Rossano, M.; Hucher, N.; Picard, C.; Colletta, D.; Le Foll, F.; Grisel, M. Effects of aging on structure and stability of TiO2 nanoparticle-containing oil-in-water emulsions. Int. J. Pharm. 2014, 461, 89–96. [Google Scholar] [CrossRef]
- Dréno, B.; Alexis, A.; Chuberre, B.; Marinovich, M. Safety of titanium dioxide nanoparticles in cosmetics. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 34–46. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Xiang, Q.; Wu, Z.; Shan, G.; Zhu, L. Depletion of double-layer coated nano-TiO2 and generation of reactive oxygen species in the presence of ethanol under simulated solar irradiation. NanoImpact 2018, 11, 164–169. [Google Scholar] [CrossRef]
- Labille, J.; Feng, J.; Botta, C.; Borschneck, D.; Sammut, M.; Cabie, M.; Auffan, M.; Rose, J.; Bottero, J.-Y. Aging of TiO2 nanocomposites used in sunscreen. Dispersion and fate of the degradation products in aqueous environment. Environ. Pollut. 2010, 158, 3482–3489. [Google Scholar] [CrossRef]
- Virkutyte, J.; Al-Abed, S.R. Statistical evaluation of potential damage to the Al(OH)3 layer on nTiO2 particles in the presence of swimming pool and seawater. J. Nanoparticle Res. 2012, 14, 787. [Google Scholar] [CrossRef]
- Virkutyte, J.; Al-Abed, S.R.; Dionysiou, D.D. Depletion of the protective aluminum hydroxide coating in TiO2-based sunscreens by swimming pool water ingredients. Chem. Eng. J. 2012, 191, 95–103. [Google Scholar] [CrossRef]
- Wallis, L.K.; Diamond, S.A.; Ma, H.; Hoff, D.J.; Al-Abed, S.R.; Li, S. Chronic TiO2 nanoparticle exposure to a benthic organism, Hyalella azteca: Impact of solar UV radiation and material surface coatings on toxicity. Sci. Total. Environ. 2014, 499, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Morsella, M.; D’Alessandro, N.; Lanterna, A.E.; Scaiano, J.C. Improving the sunscreen properties of TiO2 through an understanding of its catalytic properties. ACS Omega 2016, 1, 464–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, J.; Li, L.; Qian, Y.; Lou, H.; Yang, D.; Qiu, X. Facile and green preparation of high UV-blocking lignin/titanium dioxide nanocomposites for developing natural sunscreens. Ind. Eng. Chem. Res. 2018, 57, 15740–15748. [Google Scholar] [CrossRef]
- Shandilya, N.; Capron, I. Safer-by-design hybrid nanostructures: An alternative to conventional titanium dioxide UV filters in skin care products. RSC Adv. 2017, 7, 20430–20439. [Google Scholar] [CrossRef] [Green Version]
- Son, H.Y.; Koo, B.I.; Lee, J.B.; Kim, K.R.; Kim, W.; Jang, J.; Yoon, M.S.; Cho, J.-W.; Nam, Y.S. Tannin-titanium oxide multilayer as a photochemically suppressed ultraviolet filter. ACS Appl. Mater. Interfaces 2018, 10, 27344–27354. [Google Scholar] [CrossRef]
- Battistin, M.; Dissette, V.; Bonetto, A.; Durini, E.; Manfredini, S.; Marcomini, A.; Casagrande, E.; Brunetta, A.; Ziosi, P.; Molesini, S.; et al. A new approach to UV protection by direct surface functionalization of TiO2 with the antioxidant polyphenol dihydroxyphenyl benzimidazole carboxylic acid. Nanomater. 2020, 10, 231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Åhlén, M.; Cheung, O.; Strømme, M. Amorphous mesoporous magnesium carbonate as a functional support for UV-blocking semiconductor nanoparticles for cosmetic applications. ACS Omega 2019, 4, 4429–4436. [Google Scholar] [CrossRef]
- Bernstein, E.F.; Sarkas, H.W.; Ba, P.B.; Bouche, D. Beyond sun protection factor: An approach to environmental protection with novel mineral coatings in a vehicle containing a blend of skincare ingredients. J. Cosmet. Dermatol. 2020, 19, 407–415. [Google Scholar] [CrossRef] [Green Version]
- Borrás, M.C.; Sluyter, R.; Barker, P.J.; Konstantinov, K.; Bakand, S. Y2O3 decorated TiO2 nanoparticles: Enhanced UV attenuation and suppressed photocatalytic activity with promise for cosmetic and sunscreen applications. J. Photochem. Photobiol. B: Biol. 2020, 207, 111883. [Google Scholar] [CrossRef]
- Morlando, A.; Borrás, M.C.; Rehman, Y.; Bakand, S.; Barker, P.; Sluyter, R.; Konstantinov, K. Development of CeO2 nanodot encrusted TiO2 nanoparticles with reduced photocatalytic activity and increased biocompatibility towards a human keratinocyte cell line. J. Mater. Chem. B 2020, 8, 4016–4028. [Google Scholar] [CrossRef] [PubMed]
- Derjaguin, B.; Landau, L. Theory of the stability of strongly charged lyophobic sols and of the adhesion of strongly charged particles in solutions of electrolytes. Prog. Surf. Sci. 1993, 43, 30–59. [Google Scholar] [CrossRef]
- Overbeek, T.; Verwey, E. Theory of the Stability of Lyophobic Colloids: The Interaction of Sol Particles Having An Electric Double Layer; Elsevier: Amsterdam, The Netherlands, 1948. [Google Scholar]
- Beck, R.; Guterres, S.; Pohlmann, A. Nanocosmetics and Nanomedicines; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J.; ProQuest, E. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Wiley: New York, NY, USA, 1996. [Google Scholar]
- Nobbmann, U. Nanomaterial Isoelectric Points IEPs; Malvern Panalytical Ltd: Malvern, UK, 2017. [Google Scholar]
- Englehart, J.; Lyon, B.A.; Abriola, L.M.; Becker, M.D.; Wang, Y.; Pennell, K.D. Influence of a polymer sunscreen additive on the transport and retention of titanium dioxide nanoparticles in water-saturated porous media. Environ. Sci. Nano 2015, 3, 157–168. [Google Scholar] [CrossRef]
- Zhang, C.; Lohwacharin, J.; Takizawa, S. Properties of residual titanium dioxide nanoparticles after extended periods of mixing and settling in synthetic and natural waters. Sci. Rep. 2017, 7, 9943. [Google Scholar] [CrossRef] [Green Version]
- Luo, M.; Huang, Y.; Zhu, M.; Tang, Y.-N.; Ren, T.; Ren, J.; Wang, H.; Li, F. Properties of different natural organic matter influence the adsorption and aggregation behavior of TiO2 nanoparticles. J. Saudi Chem. Soc. 2018, 22, 146–154. [Google Scholar] [CrossRef]
- Slomberg, D.L.; Ollivier, P.; Miche, H.; Angeletti, B.; Bruchet, A.; Philibert, M.; Brant, J.; Labille, J. Nanoparticle stability in lake water shaped by natural organic matter properties and presence of particulate matter. Sci. Total. Environ. 2019, 656, 338–346. [Google Scholar] [CrossRef]
- Li, S.; Ma, H.; Wallis, L.K.; Etterson, M.A.; Riley, B.; Hoff, D.J.; Diamond, S.A. Impact of natural organic matter on particle behavior and phototoxicity of titanium dioxide nanoparticles. Sci. Total. Environ. 2016, 542, 324–333. [Google Scholar] [CrossRef]
- Labille, J.; Harns, C.; Bottero, J.-Y.; Brant, J. Heteroaggregation of titanium dioxide nanoparticles with natural clay colloids. Environ. Sci. Technol. 2015, 49, 6608–6616. [Google Scholar] [CrossRef]
- De La Vega, A.C.S.; Molins-Delgado, D.; Barceló, D.; Díaz-Cruz, M.S. Nanosized titanium dioxide UV filter increases mixture toxicity when combined with parabens. Ecotoxicol. Environ. Saf. 2019, 184, 109565. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kim, M.; Im, N.; Park, S. Photolysis of the organic UV filter, avobenzone, combined with octyl methoxycinnamate by nano-TiO2 composites. J. Photochem. Photobiol. B: Biol. 2015, 149, 196–203. [Google Scholar] [CrossRef]
- Holbrook, R.D.; Motabar, D.; Quiñones, O.; Stanford, B.; Vanderford, B.; Moss, D. Titanium distribution in swimming pool water is dominated by dissolved species. Environ. Pollut. 2013, 181, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Quiles, D.; Tovar-Sánchez, A. Sunscreens as a source of hydrogen peroxide production in coastal waters. Environ. Sci. Technol. 2014, 48, 9037–9042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, D.; Bennett, S.W.; Keller, A.A. Increased mobility of metal oxide nanoparticles due to photo and thermal induced disagglomeration. PLoS ONE 2012, 7, e37363. [Google Scholar] [CrossRef] [Green Version]
- Lv, X.; Tao, J.; Chen, B.; Zhu, X. Roles of temperature and flow velocity on the mobility of nano-sized titanium dioxide in natural waters. Sci. Total. Environ. 2016, 565, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Yin, Y.; Wang, J.; Cao, D.; Liu, J. Formation of organobromine and organoiodine compounds by engineered TiO2 nanoparticle-induced photohalogenation of dissolved organic matter in environmental waters. Sci. Total. Environ. 2018, 631–632, 158–168. [Google Scholar] [CrossRef]
- Loosli, F.; Le Coustumer, P.; Stoll, S. TiO2 nanoparticles aggregation and disaggregation in presence of alginate and Suwannee River humic acids. pH and concentration effects on nanoparticle stability. Water Res. 2013, 47, 6052–6063. [Google Scholar] [CrossRef] [PubMed]
- Sendra, M.; Yeste, M.; Gatica, J.; Moreno-Garrido, I.; Blasco, J. Homoagglomeration and heteroagglomeration of TiO2, in nanoparticle and bulk form, onto freshwater and marine microalgae. Sci. Total. Environ. 2017, 592, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Botta, C.; Labille, J.; Auffan, M.; Borschneck, D.; Miche, H.; Cabié, M.; Masion, A.; Rose, J.; Bottero, J.-Y. TiO2-based nanoparticles released in water from commercialized sunscreens in a life-cycle perspective: Structures and quantities. Environ. Pollut. 2011, 159, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Romero, A.; Ruiz-Gutiérrez, G.; Viguri, J.R.; Tovar-Sánchez, A. Sunscreens as a new source of metals and nutrients to coastal waters. Environ. Sci. Technol. 2019, 53, 10177–10187. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Cui, M.; Liu, H.; Wang, C.; Shi, Z.; Tan, C.; Yang, X. Nano-TiO2 enhances the toxicity of copper in natural water to daphnia magna. Environ. Pollut. 2011, 159, 729–734. [Google Scholar] [CrossRef]
- Skocaj, M.; Filipic, M.; Petkovic, J.; Novak, S. Titanium dioxide in our everyday life; is it safe? Radiol. Oncol. 2011, 45, 227–247. [Google Scholar] [CrossRef] [Green Version]
- Santaella, C.; Allainmat, B.; Simonet, F.; Chanéac, C.; Labille, J.; Auffan, M.; Rose, J.; Achouak, W. Aged TiO2-based nanocomposite used in sunscreens produces singlet oxygen under long-wave UV and sensitizesescherichia colito cadmium. Environ. Sci. Technol. 2014, 48, 5245–5253. [Google Scholar] [CrossRef]
- Li, L.; Sillanpää, M.; Risto, M. Influences of water properties on the aggregation and deposition of engineered titanium dioxide nanoparticles in natural waters. Environ. Pollut. 2016, 219, 132–138. [Google Scholar] [CrossRef]
- Adam, V.; Loyaux-Lawniczak, S.; Quaranta, G. Characterization of engineered TiO2 nanomaterials in a life cycle and risk assessments perspective. Environ. Sci. Pollut. Res. 2015, 22, 11175–11192. [Google Scholar] [CrossRef]
- Mozhayeva, D.; Engelhard, C. A critical review of single particle inductively coupled plasma mass spectrometry—A step towards an ideal method for nanomaterial characterization. J. Anal. At. Spectrom. 2019, 35, 1740–1783. [Google Scholar] [CrossRef] [Green Version]
- Krause, B.; Meyer, T.; Sieg, H.; Kästner, C.; Reichardt, P.; Tentschert, J.; Jungnickel, H.; Estrela-Lopis, I.; Burel, A.; Chevance, S.; et al. Characterization of aluminum, aluminum oxide and titanium dioxide nanomaterials using a combination of methods for particle surface and size analysis. RSC Adv. 2018, 8, 14377–14388. [Google Scholar] [CrossRef] [Green Version]
- Wilbur, S.; Yamanaka, M.; Sannac, S. Characterization of Nanoparticles in Aqueous Samples by ICP-MS.; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2017. [Google Scholar]
- Bustos, A.R.M.; Winchester, M.R. Single-particle-ICP-MS advances. Anal. Bioanal. Chem. 2016, 408, 5051–5052. [Google Scholar] [CrossRef] [Green Version]
- Gondikas, A.; Von Der Kammer, F.; Kaegi, R.; Borovinskaya, O.; Neubauer, E.; Navratilova, J.; Praetorius, A.; Cornelis, G.; Hofmann, T. Where is the nano? Analytical approaches for the detection and quantification of TiO2 engineered nanoparticles in surface waters. Environ. Sci. Nano 2017, 5, 313–326. [Google Scholar] [CrossRef]
- Gondikas, A.P.; Von Der Kammer, F.; Reed, R.B.; Wagner, S.; Ranville, J.F.; Hofmann, T. Release of TiO2 nanoparticles from sunscreens into surface waters: A one-year survey at the old danube recreational lake. Environ. Sci. Technol. 2014, 48, 5415–5422. [Google Scholar] [CrossRef]
- Vidmar, J.; Milačič, R.; Ščančar, J. Sizing and simultaneous quantification of nanoscale titanium dioxide and a dissolved titanium form by single particle inductively coupled plasma mass spectrometry. Microchem. J. 2017, 132, 391–400. [Google Scholar] [CrossRef]
- Peters, R.J.; van Bemmel, G.; Milani, N.B.; Hertog, G.C.D.; Undas, A.K.; van der Lee, M.; Bouwmeester, H. Detection of nanoparticles in Dutch surface waters. Sci. Total. Environ. 2018, 621, 210–218. [Google Scholar] [CrossRef]
- Reed, R.B.; Martin, D.P.; Bednar, A.J.; Montaño, M.D.; Westerhoff, P.; Ranville, J.F. Multi-day diurnal measurements of Ti-containing nanoparticle and organic sunscreen chemical release during recreational use of a natural surface water. Environ. Sci. Nano 2017, 4, 69–77. [Google Scholar] [CrossRef]
- Rand, L.N.; Bi, Y.; Poustie, A.; Bednar, A.J.; Hanigan, D.J.; Westerhoff, P.; Ranville, J.F. Quantifying temporal and geographic variation in sunscreen and mineralogic titanium-containing nanoparticles in three recreational rivers. Sci. Total. Environ. 2020, 743, 140845. [Google Scholar] [CrossRef]
- Wang, J.; Nabi, M.; Mohanty, S.K.; Afrooz, A.N.; Cantando, E.; Aich, N.; Baalousha, M. Detection and quantification of engineered particles in urban runoff. Chemosphere 2020, 248, 126070. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, A.K.; Reed, R.B.; Lee, S.; Bi, X.; Hanigan, D.; Yang, Y.; Ranville, J.F.; Herckes, P.; Westerhoff, P. Detection and sizing of Ti-containing particles in recreational waters using single particle ICP-MS. Bull. Environ. Contam. Toxicol. 2018, 100, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Labille, J.; Slomberg, D.; Catalano, R.; Robert, S.; Apers-Tremelo, M.-L.; Boudenne, J.-L.; Manasfi, T.; Radakovitch, O. Assessing UV filter inputs into beach waters during recreational activity: A field study of three French Mediterranean beaches from consumer survey to water analysis. Sci. Total. Environ. 2020, 706, 136010. [Google Scholar] [CrossRef] [PubMed]
- Hadioui, M.; Knapp, G.; Azimzada, A.; Jreije, I.; Frechette-Viens, L.; Wilkinson, K.J. Lowering the Size Detection Limits of Ag and TiO2 Nanoparticles by Single Particle ICP-MS. Anal. Chem. 2019, 91, 13275–13284. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, M.; Itagaki, T.; Wilbur, S. Measuring Multiple Elements in Nanoparticles Using spICP-MS; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2018. [Google Scholar]
- Loosli, F.; Wang, J.; Rothenberg, S.; Bizimis, M.; Winkler, C.; Borovinskaya, O.; Flamigni, L.; Baalousha, M. Sewage spills are a major source of titanium dioxide engineered (nano)-particle release into the environment. Environ. Sci. Nano 2019, 6, 763–777. [Google Scholar] [CrossRef]
- Khosravi, K.; Hoque, M.E.; Dimock, B.; Hintelmann, H.; Metcalfe, C.D. A novel approach for determining total titanium from titanium dioxide nanoparticles suspended in water and biosolids by digestion with ammonium persulfate. Anal. Chim. Acta 2012, 713, 86–91. [Google Scholar] [CrossRef]
- Markus, A.; Krystek, P.; Tromp, P.; Parsons, J.; Roex, E.; De Voogt, P.; Laane, R. Determination of metal-based nanoparticles in the river Dommel in the Netherlands via ultrafiltration, HR-ICP-MS and SEM. Sci. Total. Environ. 2018, 631-632, 485–495. [Google Scholar] [CrossRef]
- Neale, P.A.; Jämting, Å.K.; O’Malley, E.; Herrmann, J.; Escher, B.I. Behaviour of titanium dioxide and zinc oxide nanoparticles in the presence of wastewater-derived organic matter and implications for algal toxicity. Environ. Sci. Nano 2015, 2, 86–93. [Google Scholar] [CrossRef] [Green Version]
- Mudunkotuwa, I.A.; Anthony, T.R.; Grassian, V.H.; Peters, T.M. Accurate quantification of TiO2 nanoparticles collected on air filters using a microwave-assisted acid digestion method. J. Occup. Environ. Hyg. 2016, 13, 30–39. [Google Scholar] [CrossRef] [Green Version]
- May, T.W.; Wiedmeyer, R.H. A table of polyatomic interferences in ICP-MS. At. Spectrosc. Norwalk Conn. 1998, 19, 150–155. [Google Scholar]
- Raeve, P.; Bianga, J. Fast and Accurate Absolute-Quantification of Proteins and Antibodies Using Isotope Dilution-Triple Quadrupole ICP-MS; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2015. [Google Scholar]
- Wills, J.D.; Lindemann, T. Analysis of Sulfuric Acid Using a Single Set of Operating Conditions with HR-ICP-MS.; Thermo Fisher Scientific Inc.: Waltham, MA, USA, 2016. [Google Scholar]
- Yamanaka, M.; Yamanaka, K.; Itagaki, T.; Wilbur, S.; McCurdy, E. Automated, High Sensitivity Analysis of Single Nanoparticles Using the Agilent 7900 ICP-MS with Single Nanoparticle Application Module; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2015. [Google Scholar]
- Watkins, P.S.; Castellon, B.T.; Tseng, C.; Wright, M.V.; Matson, C.W.; Cobb, G.P. Validation of a sulfuric acid digestion method for inductively coupled plasma mass spectrometry quantification of TiO2 nanoparticles. Bull. Environ. Contam. Toxicol. 2018, 100, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Tharaud, M.; Gondikas, A.P.; Benedetti, M.F.; Von Der Kammer, F.; Hofmann, T.; Cornelis, G. TiO2 nanomaterial detection in calcium rich matrices by spICPMS. A matter of resolution and treatment. J. Anal. At. Spectrom. 2017, 32, 1400–1411. [Google Scholar] [CrossRef]
- Fabricius, A.-L.; Duester, L.; Meermann, B.; Ternes, T.A. ICP-MS-based characterization of inorganic nanoparticles—Sample preparation and off-line fractionation strategies. Anal. Bioanal. Chem. 2014, 406, 467–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.; Bi, X.; Reed, R.B.; Ranville, J.F.; Herckes, P.; Westerhoff, P. Nanoparticle size detection limits by single particle ICP-MS for 40 elements. Environ. Sci. Technol. 2014, 48, 10291–10300. [Google Scholar] [CrossRef] [PubMed]
- Bocca, B.; Caimi, S.; Senofonte, O.; Alimonti, A.; Petrucci, F. ICP-MS based methods to characterize nanoparticles of TiO2 and ZnO in sunscreens with focus on regulatory and safety issues. Sci. Total. Environ. 2018, 630, 922–930. [Google Scholar] [CrossRef]
- Lange, T.; Schneider, P.; Schymura, S.; Franke, K. The fate of anthropogenic nanoparticles, nTiO2 and nCeO2, in waste water treatment. Water 2020, 12, 2509. [Google Scholar] [CrossRef]
- Thomas, R. Practical Guide to ICP-MS: A Tutorial for Beginners; CRC Press LLC: Baton Rouge, LA, USA, 2013. [Google Scholar]
- Laborda, F.; Bolea, E.; Jiménez-Lamana, J. Single particle inductively coupled plasma mass spectrometry: A powerful tool for nanoanalysis. Anal. Chem. 2014, 86, 2270–2278. [Google Scholar] [CrossRef]
- Merrifield, R.C.; Stephan, C.; Lead, J.R. Single-particle inductively coupled plasma mass spectroscopy analysis of size and number concentration in mixtures of monometallic and bimetallic (core-shell) nanoparticles. Talanta 2017, 162, 130–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azimzada, A.; Farner, J.M.; Jreije, I.; Hadioui, M.; Liu-Kang, C.; Tufenkji, N.; Shaw, P.; Wilkinson, K.J. Single- and multi-element quantification and characterization of TiO2 nanoparticles released from outdoor stains and paints. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Sun, T.Y.; Bornhöft, N.A.; Hungerbuehler, K.; Nowack, B. Dynamic Probabilistic Modeling of environmental emissions of engineered nanomaterials. Environ. Sci. Technol. 2016, 50, 4701–4711. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.Y.; Mitrano, D.M.; Bornhöft, N.A.; Scheringer, M.; Hungerbühler, K.; Nowack, B. Envisioning nano release dynamics in a changing world: Using dynamic probabilistic modeling to assess future environmental emissions of engineered nanomaterials. Environ. Sci. Technol. 2017, 51, 2854–2863. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sonderer, T.; Scholz, R.W.; Nowack, B. Modeled Environmental concentrations of engineered nanomaterials (TiO2, ZnO, Ag, CNT, fullerenes) for different regions. Environ. Sci. Technol. 2009, 43, 9216–9222. [Google Scholar] [CrossRef]
- Gottschalk, F.; Lassen, C.; Kjoelholt, J.; Christensen, F.; Nowack, B. Modeling flows and concentrations of nine engineered nanomaterials in the danish environment. Int. J. Environ. Res. Public Health 2015, 12, 5581–5602. [Google Scholar] [CrossRef] [Green Version]
- Meesters, J.A.J.; Koelmans, A.A.; Quik, J.T.K.; Hendriks, A.J.; Van De Meent, D. Multimedia modeling of engineered nanoparticles with SimpleBox4nano: Model definition and evaluation. Environ. Sci. Technol. 2014, 48, 5726–5736. [Google Scholar] [CrossRef] [PubMed]
- Markus, A.; Parsons, J.; Roex, E.; De Voogt, P.; Laane, R. Modeling aggregation and sedimentation of nanoparticles in the aquatic environment. Sci. Total. Environ. 2015, 506-507, 323–329. [Google Scholar] [CrossRef]
- Meesters, J.A.J.; Quik, J.T.K.; Koelmans, A.A.; Hendriks, A.J.; Van De Meent, D. Multimedia environmental fate and speciation of engineered nanoparticles: A probabilistic modeling approach. Environ. Sci. Nano 2016, 3, 715–727. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.H.; Cohen, Y. Multimedia environmental distribution of engineered nanomaterials. Environ. Sci. Technol. 2014, 48, 3281–3292. [Google Scholar] [CrossRef]
- Garner, K.L.; Suh, S.; Keller, A.A. Assessing the risk of engineered nanomaterials in the environment: Development and application of the nanoFate Model. Environ. Sci. Technol. 2017, 51, 5541–5551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sani-Kast, N.; Scheringer, M.; Slomberg, D.; Labille, J.; Praetorius, A.; Ollivier, P.; Hungerbühler, K. Addressing the complexity of water chemistry in environmental fate modeling for engineered nanoparticles. Sci. Total. Environ. 2015, 535, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Adam, V.; Caballero-Guzman, A.; Nowack, B. Considering the forms of released engineered nanomaterials in probabilistic material flow analysis. Environ. Pollut. 2018, 243, 17–27. [Google Scholar] [CrossRef]
- Dale, A.L.; Casman, E.A.; Lowry, G.V.; Lead, J.R.; Viparelli, E.; Baalousha, M. Modeling nanomaterial environmental fate in aquatic systems. Environ. Sci. Technol. 2015, 49, 2587–2593. [Google Scholar] [CrossRef]
- Praetorius, A.; Badetti, E.; Brunelli, A.; Clavier, A.; Gallego-Urrea, J.A.; Gondikas, A.; Hassellöv, M.; Hofmann, T.; Mackevica, A.; Marcomini, A.; et al. Strategies for determining heteroaggregation attachment efficiencies of engineered nanoparticles in aquatic environments. Environ. Sci. Nano 2020, 7, 351–367. [Google Scholar] [CrossRef] [Green Version]
- Meesters, J.A.J.; Peijnenburg, W.J.G.M.; Hendriks, A.J.; Van De Meent, D.; Quik, J.T.K.; Hendriks, J. A model sensitivity analysis to determine the most important physicochemical properties driving environmental fate and exposure of engineered nanoparticles. Environ. Sci. Nano 2019, 6, 2049–2060. [Google Scholar] [CrossRef]
- Praetorius, A.; Scheringer, M.; Hungerbühler, K. Development of environmental fate models for engineered nanoparticles—A case study of TiO2 nanoparticles in the Rhine River. Environ. Sci. Technol. 2012, 46, 6705–6713. [Google Scholar] [CrossRef]
- Markus, A.; Parsons, J.; Roex, E.; De Voogt, P.; Laane, R. Modelling the transport of engineered metallic nanoparticles in the river Rhine. Water Res. 2016, 91, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.H.; Pollino, C.A. Good practice in Bayesian network modelling. Environ. Model. Softw. 2012, 37, 134–145. [Google Scholar] [CrossRef]
- Money, E.S.; Reckhow, K.H.; Wiesner, M.R. The use of Bayesian networks for nanoparticle risk forecasting: Model formulation and baseline evaluation. Sci. Total. Environ. 2012, 426, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Marvin, H.J.P.; Bouzembrak, Y.; Janssen, E.M.; Van Der Zande, M.; Murphy, F.; Sheehan, B.; Mullins, M.; Bouwmeester, H. Application of Bayesian networks for hazard ranking of nanomaterials to support human health risk assessment. Nanotoxicology 2017, 11, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Liu, H.; Liu, R.; Cohen, Y. Bayesian network as a support tool for rapid query of the environmental multimedia distribution of nanomaterials. Nanoscale 2017, 9, 4162–4174. [Google Scholar] [CrossRef]
- Sahin, O.; Stewart, R.A.; Faivre, G.; Ware, D.; Tomlinson, R.; Mackey, B. Spatial Bayesian network for predicting sea level rise induced coastal erosion in a small Pacific Island. J. Environ. Manag. 2019, 238, 341–351. [Google Scholar] [CrossRef]
- Global Market Research Database: Products by Ingredient/Sun Care/World Euromonitor International; Passport: London, UK, 2014–2019.
- Schaumann, G.E.; Philippe, A.; Bundschuh, M.; Metreveli, G.; Klitzke, S.; Rakcheev, D.; Grün, A.; Kumahor, S.K.; Kuhn, M.; Baumann, T.; et al. Understanding the fate and biological effects of Ag- and TiO2-nanoparticles in the environment: The quest for advanced analytics and interdisciplinary concepts. Sci. Total. Environ. 2015, 535, 3–19. [Google Scholar] [CrossRef] [PubMed]
| Surface Treatment | Benefits | Reference | |
|---|---|---|---|
 R: Chemically coated lignosulfonate | Lignin/TiO2 nanocomposites Previous study with similar approach: Encapsulation of TiO2 by different types of lignin [67] | Good dispersibility; high sun protection factor (SPF) | [68] |
 CNC: Cellulose nanocrystals | Organic/TiO2 hybrid nanostructures TiO2 NPs grafted onto cellulose nanocrystals (CNC) | High SPF; in o/w: replaces surfactant in formulation | [69] |
 TA: Tannic acid p-MS: porous polymer microspheres | Tannin/TiO2 multilayers on p-MS Deposition on porous polymethylmethacrylate (PMMA) microspheres Layer-by-layer assembly of TiO2 and TA via ligand-to-metal complexation | Reduced ROS; high SPF | [70] |
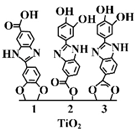 | Surface functionalisation with Oxisol Oxisol: Polyphenol dihydroxyphenyl benzimidazole carboxylic acid Addition mechanism of Oxisol to TiO2 Steric stabilisation likely | Reduced ROS; reduced photocatalytic activity; high SPF | [71] |
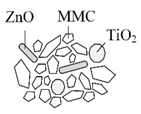 MMC: Mesoporous magnesium carbonate cluster | MMC-TiO2-ZnO Incorporation of TiO2 and ZnO into an amorphous MMC structure (+ dimethicone layer) | Comparable SPF; no photocatalytic activity | [72] |
 | TiO2 multifunctional coating Dense and homogeneous encapsulation | Improved esthetic feel; reduced photocatalytic activity; high SPF | [73] |
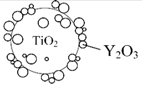 Y2O3: Yttrium oxide | Y2O3/TiO2 nanocomposites Deposition of Y2O3 onto TiO2 Uniform Y2O3 coating | Enhanced UV attenuation; reduced photocatalytic activity; biocompatible | [74] |
 CeO2: Cerium dioxide | CeO2/TiO2 nanocomposites CeO2 nanodot encrusted TiO2 NPs | Reduced photocatalytic activity; biocompatible | [75] |
| Matrix | Sample Collection | Pre-Treatment | Analysis | Reference | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Location | Period | Filtration | Transport, Storage, Time Until Tested | Acid Digestion | Microwave-Assisted Acid Digestion | Fusion | Technology | SP Mode | Multi- Element | Mentioned Interferences | Gas | Dwell Time (ms) | Measured Isotopes | TiO2 SDL (nm) | |||
| Freshwater (lake) | WS, 1 m depth | 12 mos. | Unfiltered | PE bottles, 7 °C, <16 mos. | Triple Quad (CCT), TOF | × | Al, Fe, Mn, Pb, V | 48Ca | NH3, He | 3, 4 | 81 | [107] | |||||
| AWI | 0.8 µm CA membrane | HNO3, HF, H2O2, HCl | |||||||||||||||
| Freshwater (lake) | WS, 1 m depth | 12 mos. | Unfiltered | Quad (TRA) | × | Al, V, Ga, Y, Nb, Eu, Ho, Er, Tm, Yb, Ta | 48Ca | 10 | 47Ti | 130 | [108] | ||||||
| 0.2 µm CA membrane | HNO3, HF, H2O2, HCl | ||||||||||||||||
| Freshwater (river) | 0.45 µm | PE bottles, immediately | Quad (TRA) | × | 48Ca | Ar, He, no gas | 3, 5, 10 | 47Ti | 128 rutile, 44 anatase | [109] | |||||||
| HNO3, HF, HCl | |||||||||||||||||
| Freshwater (river) | WS | PE, glass bottles, 4 °C, <24 h | Quad | × | 48Ca | 3 | 48Ti | 100 | [110] | ||||||||
| Freshwater (creek) | WS | Hourly, over 72 h | PP centrifuge tubes, 4 °C | Quad (TRA), SF | × | Al, Fe | 48Ca, 32S16O | 3 | 49Ti | 79 (SF = 42) | [111] | ||||||
| HDPE bottles + HNO3, 4 °C | |||||||||||||||||
| Freshwater (river) | 10 cm depth | At least twice daily, 3−7 days | HDPE bottles, refrigerated, + HNO3, <24 h | Quad | × | 0.1 | 49Ti | 48 | [112] | ||||||||
| 0.02 µm PP | |||||||||||||||||
| Freshwater (urban runoff) | HDPE bottles, 4 °C, <24 h | H2O2, HF, HNO3 | Quad | Nb | 47Ti, 93Nb | [113] | |||||||||||
| Saltwater (river), SPW | WS | Over 9 h | 0.7 µm | Amber bottles, cooled, <7 days | Quad (TRA) | × | 10 | 148 river, 173 SPW | [114] | ||||||||
| Saltwater (ocean) | WS, 40 cm depth | 3× in 24 h | TFF: >0.5 µm, 0.02 < x < 0.5 µm | HDPE bottles, 4 °C | HNO3, HF, H2O2 | Quad | Zn, Al, Fe, V | [115] | |||||||||
| Freshwater (rain), SPW, Milli-Q water | 30 cm depth (SPW) | Once | FEP tubes, 4 °C | SF, Quad | × | Ar, N2 | 0.05, 0.1 | 49Ti | 19.2 wet/12.1 dry aerosol, 19−44 urban waters, 27 SPW | [116] | |||||||
| Freshwater (tap water), SPW, sand filter BW | 5 cm depth (SPW) | Twice daily, 3 mos. | Spiral wound (SPW)/ membrane filter (BW) + TFF | Dechlorinated, PTFE bottles, refrigerated, <24 h | HF | Quad (CCT) | 48Ca | He | 150, 300 | 48Ti, 49Ti | [89] | ||||||
| SPW | Quad (TRA) | × | Zn, Al | 0.1 | 47Ti, 27Al, 66Zn | [117] | |||||||||||
| Freshwater (creek) after sewage spill | Monthly, 4 mos. | HDPE bottles, then PP tubes, 4 °C | TOF | × | Various (Al, V, Ga, Y, Nb, Ba, W, Zr, Yb, Ta, Zn, Fe, Mn) | 47Ti, 48Ti, 93Nb | 100, (40 UPW) | [118] | |||||||||
| H2O2, HF, HNO3, 110 °C | SF | ||||||||||||||||
| Untreated/ treated WW, liquid biosolids | WWTP (Canada) | 24 h period | 0.22 µm nylon | Biosolid sample frozen, <48 h | (a) HNO3, HF, HCl | (b) Ammonium persulfate | Quad (CCT) | 32S16O | 49Ti | [119] | |||||||
| Freshwater (river), WW influent/ effluent, sludge | 50 cm depth | Once | 0.45 µm nylon | + selective UF (450, 200, 100, 20 nm) | PS bottles, 5 °C, then PE tubes | HNO3, HF, 70 °C (overnight) | SF | [120] | |||||||||
| Unfiltered + HNO3 | |||||||||||||||||
| WW effluent | WWTP (Australia) | 24 h period | 1.6 µm glass fibre + 0.45 µm nylon, 0.22 µm PES | 4 °C, stored at pH = 2, <8 mos. | × | Quad | [121] | ||||||||||
| WW influent/ effluent, sludge, sediment | WWTP (USA) | Monthly, 12 mos. | 0.2 µm CA membrane | 4 °C, <3 days | H2O2, HNO3, H2SO4 | [31] | |||||||||||
| Matrix | Pre-Treatment | Analysis | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Acid Digestion | Microwave-Assisted Acid Digestion | Technology | SP- Mode | Multi- Element | Mentioned Interferences | Gas | Dwell Time (ms) | Measured Isotopes | TiO2 SDL (nm) | ||
| UPW | Quad (TRA) | × | 0.1 | [126] | |||||||
| H2SO4, HNO3 | Quad (CCT) | Ar, He | 46−50Ti | [127] | |||||||
| SF, tandem Quad | × | 48Ca, 32S16O, 36Ar12C | O2, He +NH3 | 3, 5 | 47−49Ti, 48Ca, 63TiNH | 23−47 | [128] | ||||
| (a) H2SO4 | (b) H2SO4 | Quad (CCT) | He | 47Ti | [129] | ||||||
| DI water | Quad (TRA) | × | Ar | 10 | 47,49Ti | 91−95 | [130] | ||||
| SL dispersed in 1% Triton X-100 | Quad (KED) | × | Zn | He | 5 | 48Ti, 64Zn | 25 | [131] | |||
| Quad | × | 48Ca, 32S16O | 0.1 | 48Ti | 27−29 | [38] | |||||
| synthetic WW, sludge | HCl, HNO3 | Quad | Ar | 48Ti | [132] | ||||||
| × | 150 | ||||||||||
| Model | Technique | System Specification | Fate Processes | Reference | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MFA | EFM | Multimedia | Probabilistic Model | Mechanistic Model | Data Type; Source | Source of Nano-TiO2 | TiO2 Removal, Sinks | Nanoparticle Characteristics | Media Characteristics | Environmental Compartments | Space and Time Considerations | Transport Processes | Transformation Processes | |
| Release model; distribution model based on [28] | × | PV, SPC, PR (dt), TC; experiments, expert opinion | Temporal (in-use stock)/final sinks (soil, landfill, sediment) | Air, water, soil, ST soil, sediment, technical compartments | Temporally resolved; spatially averaged (EU) | [137] | ||||||||
| Based on [28] | Based on [151] | × | × | Single input source (steady state): STP effluent, derived from previous MFA + PPr, ESP, HD | PSD, Assumption: Aggregated form | SPM (type, conc., size), constant within one river section | Water, sediment | Water chemistry effects spatially resolved (Rhône River, France) | Sedimentation, resuspension, advection | Heteroaggregation | [146] | |||
| × | × | PV, PC, PR (LC), TC; experiments, assumptions | Various (e.g., cosmetics, plastics, paint, coating) | Soil, sediment | Distinction: Anatase/rutile | Air, water, soil, ST soil, sediment, technical compartments | Spatiotemporally averaged (Denmark) | Sedimentation | [140] | |||||
| × SimpleBox4nano, SB4N | × | × | × | PV, SPC, PR (LC), PPr, ESP; literature, models, experiments, DLVO variables | Various (e.g., cosmetics, electronics, coatings, paints) | Transport processes | Physicochemical properties (e.g., PSD) | E.g., PS, ζ of natural particles in colloidal or particulate form | Air, water, soil, sediment | Spatiotemporally averaged | Sedimentation (incl. burial), deposition, resuspension, erosion, advection | Heteroaggregation | [143] | |
| × | Based on [142] | HD, PU, input scenarios: WW (treated/untreated), sludge run-off | WWTP | Distinction: free/unattached particles, homo- or heteroaggregates | SPM (conc.) | Water, sediment | Spatiotemporally averaged (Rhine River, Germany, Netherlands) | Sedimentation, resuspension, advection | Homo-/heteroaggregation | [152] | ||||
| × NanoFATE | × | HD, PR (dt) (LC), ESP (dt) | PS, distinction: free particles or small homoaggregates, heteroaggregates | Water chemistry, soil properties (e.g., pH) | Air, water, soil, ST soil, sediment | Temporally resolved, limitation: spatial resolution (San Francisco Bay, USA) | Sedimentation (incl. burial), deposition, resuspension, soil run-off/leaching, advection | Heteroaggregation | [145] | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heilgeist, S.; Sekine, R.; Sahin, O.; Stewart, R.A. Finding Nano: Challenges Involved in Monitoring the Presence and Fate of Engineered Titanium Dioxide Nanoparticles in Aquatic Environments. Water 2021, 13, 734. https://doi.org/10.3390/w13050734
Heilgeist S, Sekine R, Sahin O, Stewart RA. Finding Nano: Challenges Involved in Monitoring the Presence and Fate of Engineered Titanium Dioxide Nanoparticles in Aquatic Environments. Water. 2021; 13(5):734. https://doi.org/10.3390/w13050734
Chicago/Turabian StyleHeilgeist, Simone, Ryo Sekine, Oz Sahin, and Rodney A. Stewart. 2021. "Finding Nano: Challenges Involved in Monitoring the Presence and Fate of Engineered Titanium Dioxide Nanoparticles in Aquatic Environments" Water 13, no. 5: 734. https://doi.org/10.3390/w13050734









