Experimental Study of the Quantitative Impact of Flow Turbulence on Algal Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Algae
2.2. Experimental Apparatus and Methods
2.3. Statistical Analysis and Software
3. Results
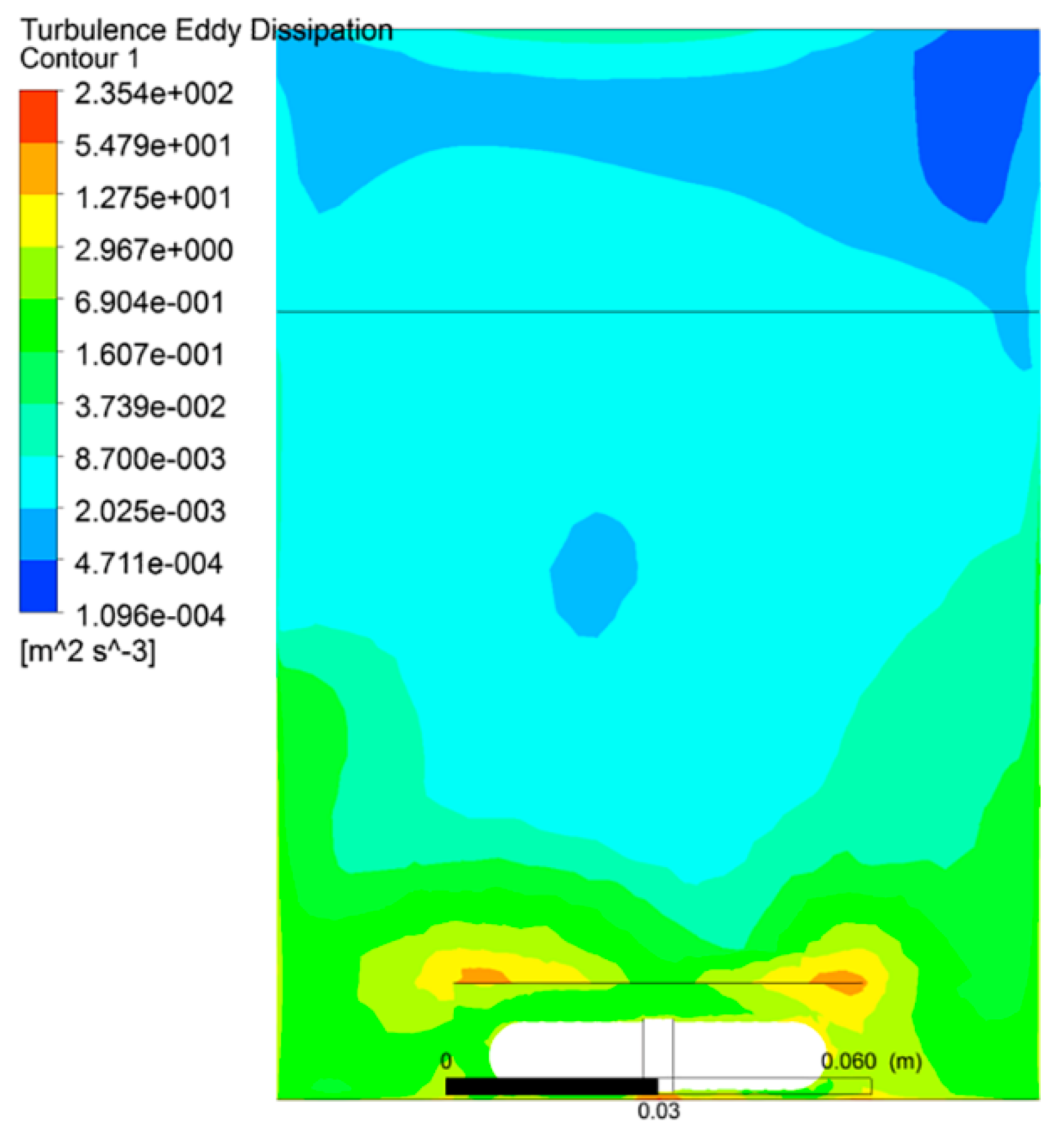
3.1. The Distribution of Turbulent Intensity
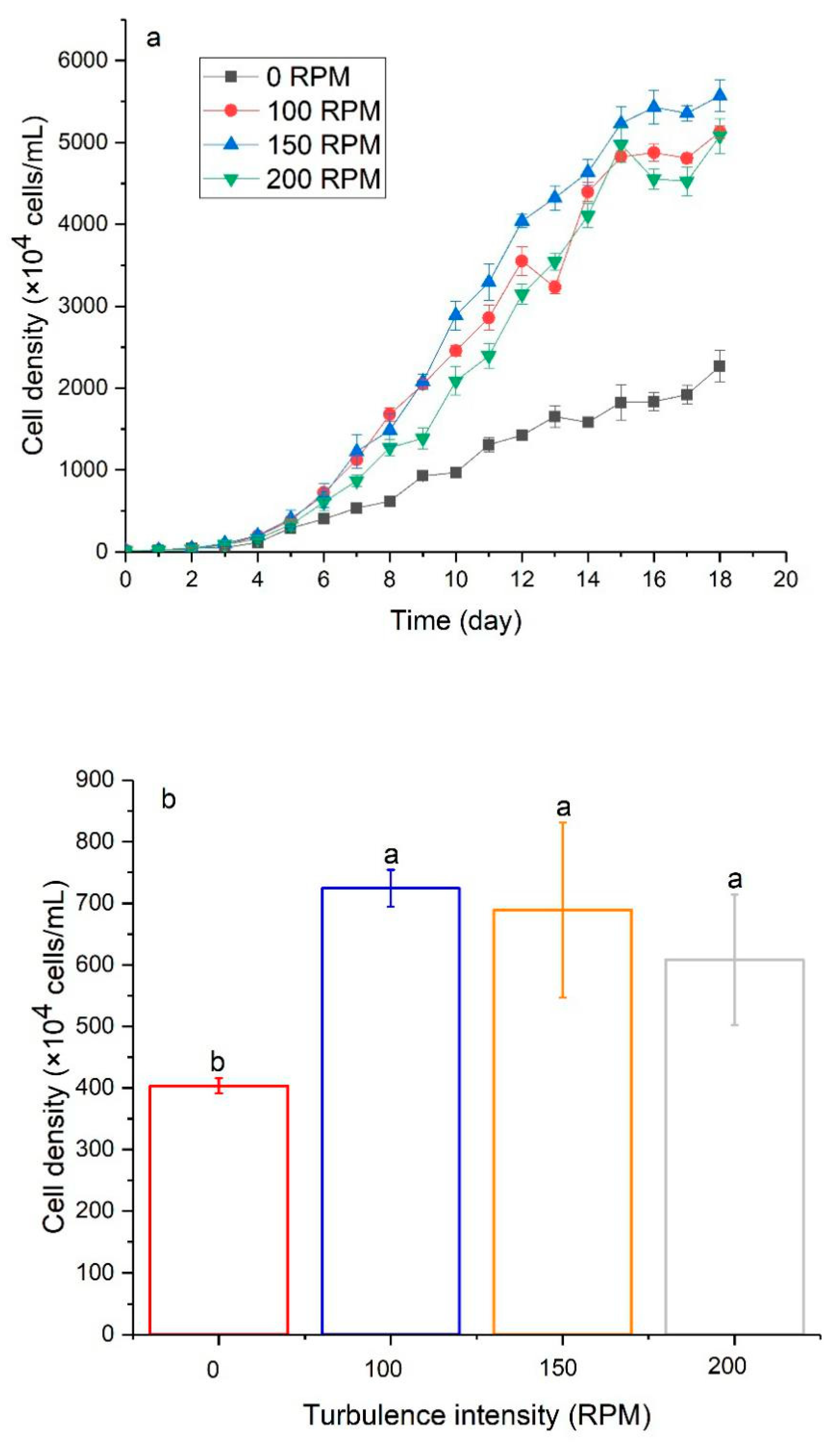
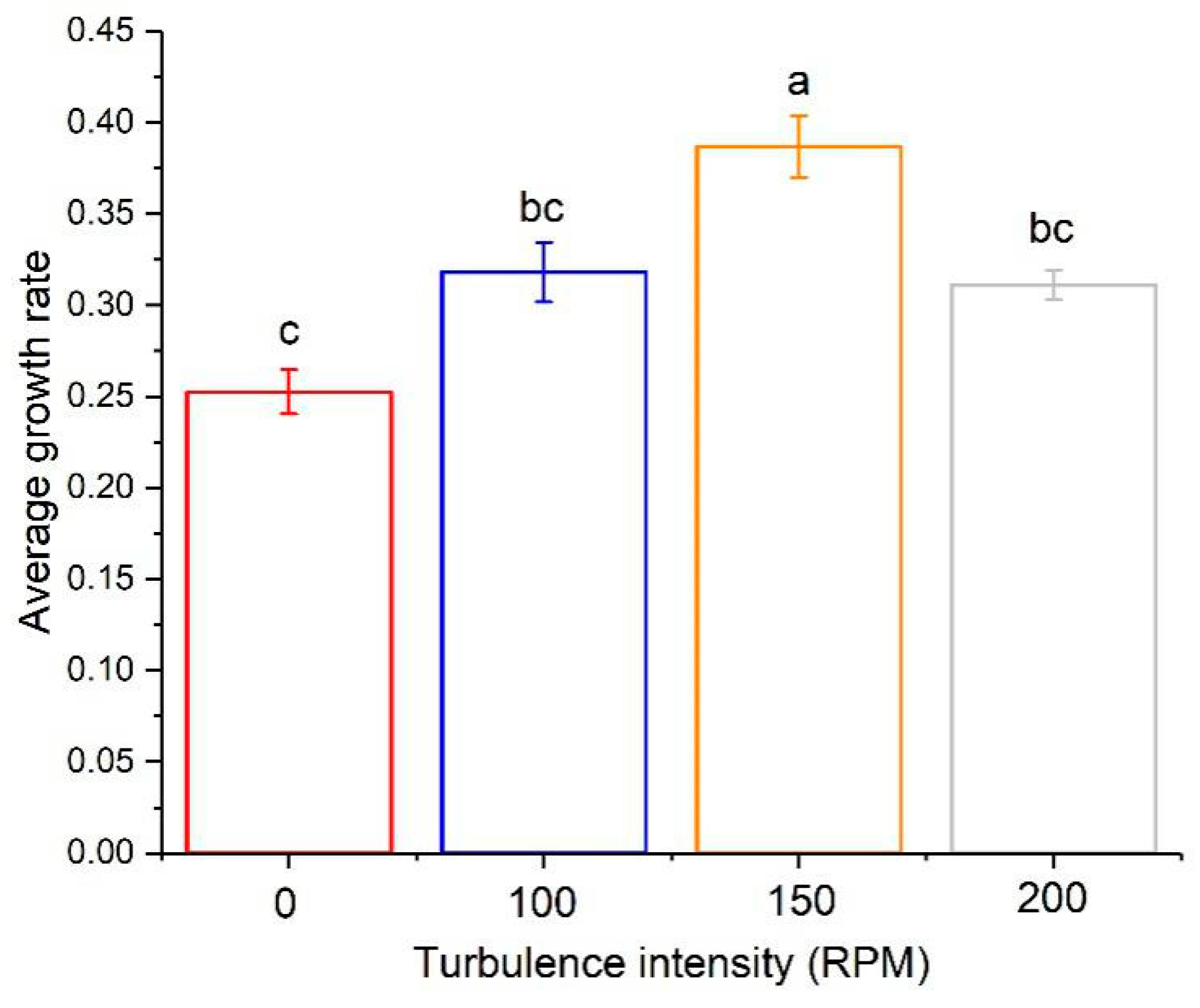
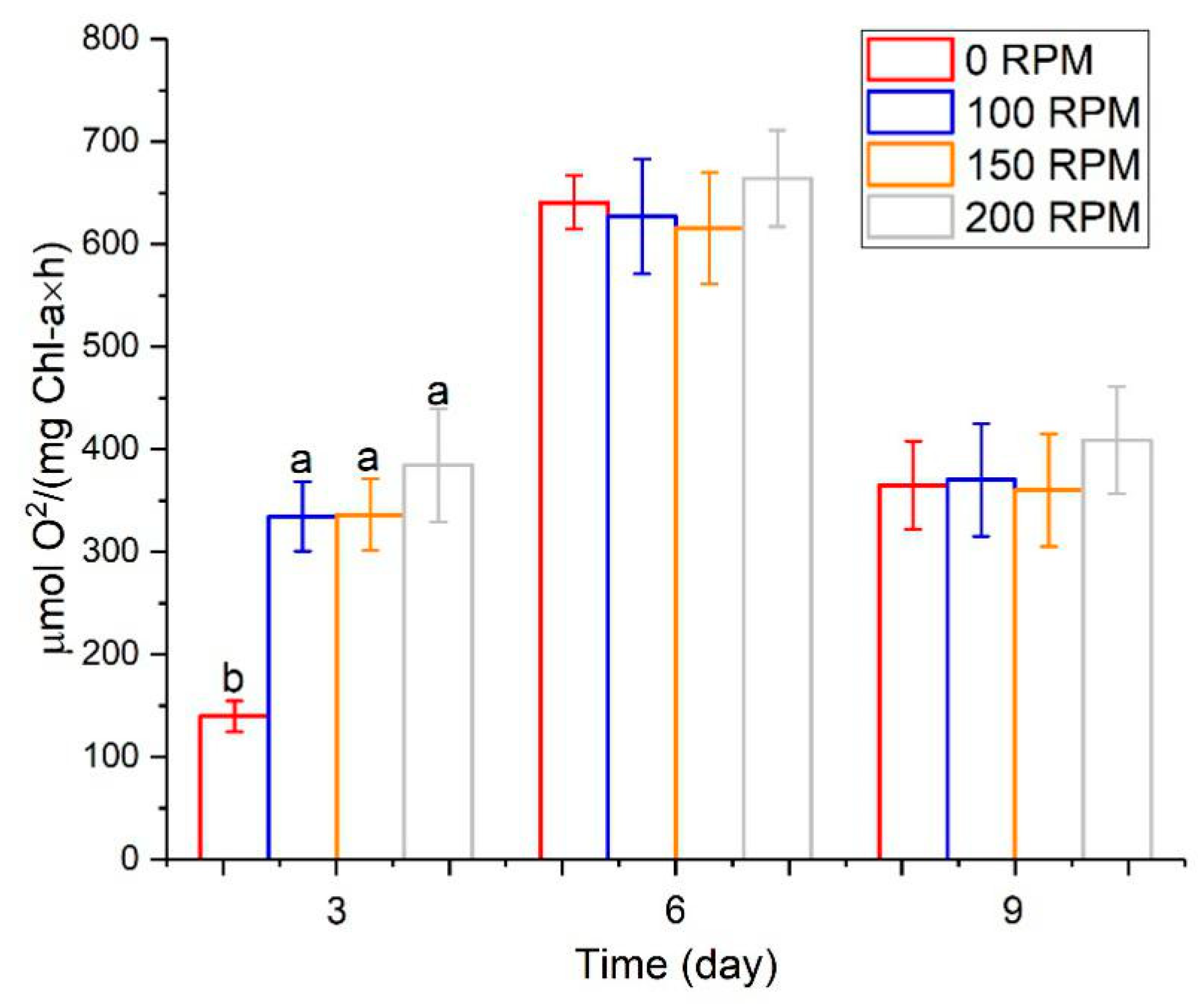
3.2. The Effect of Turbulence on Algal Growth
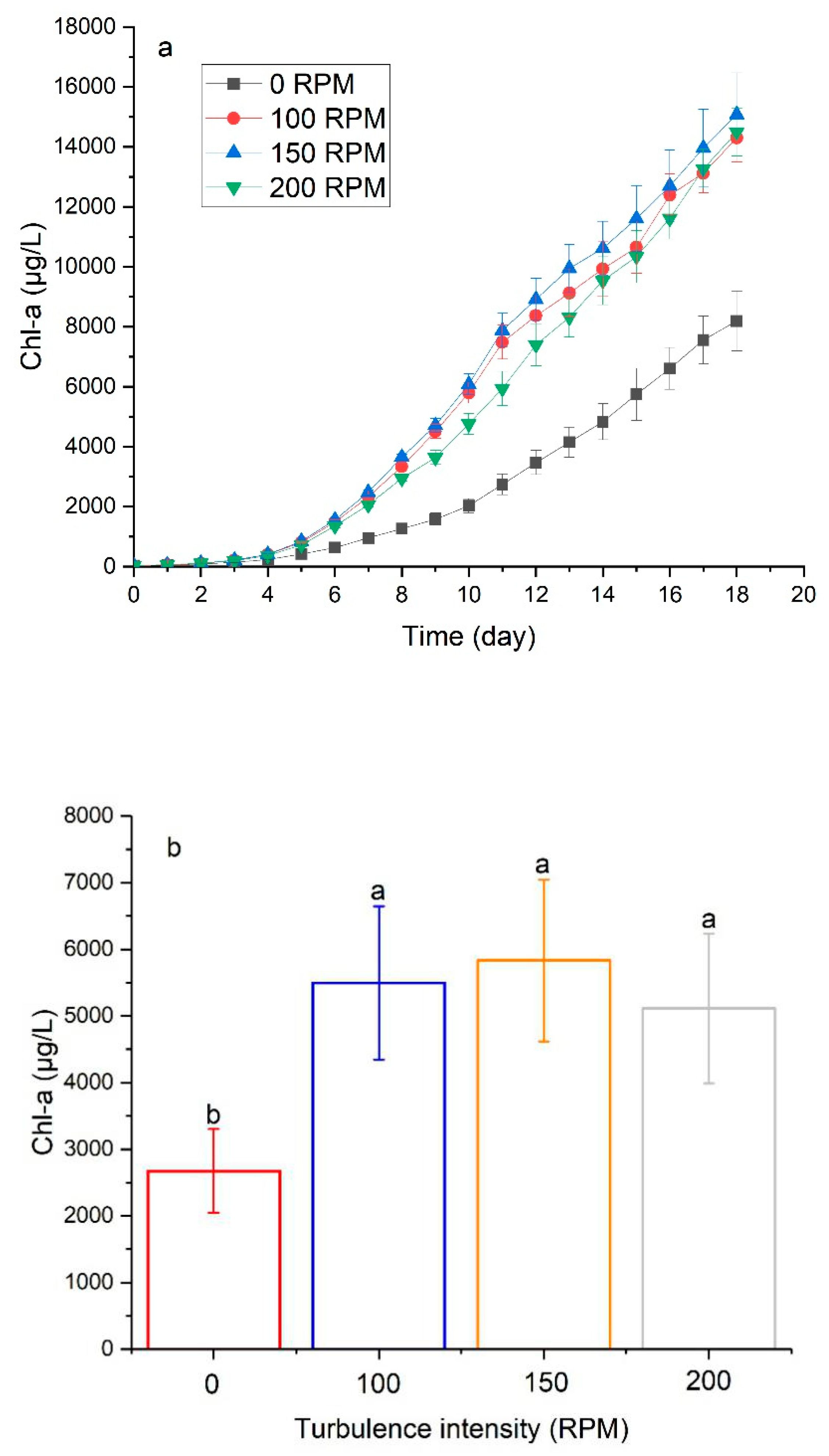
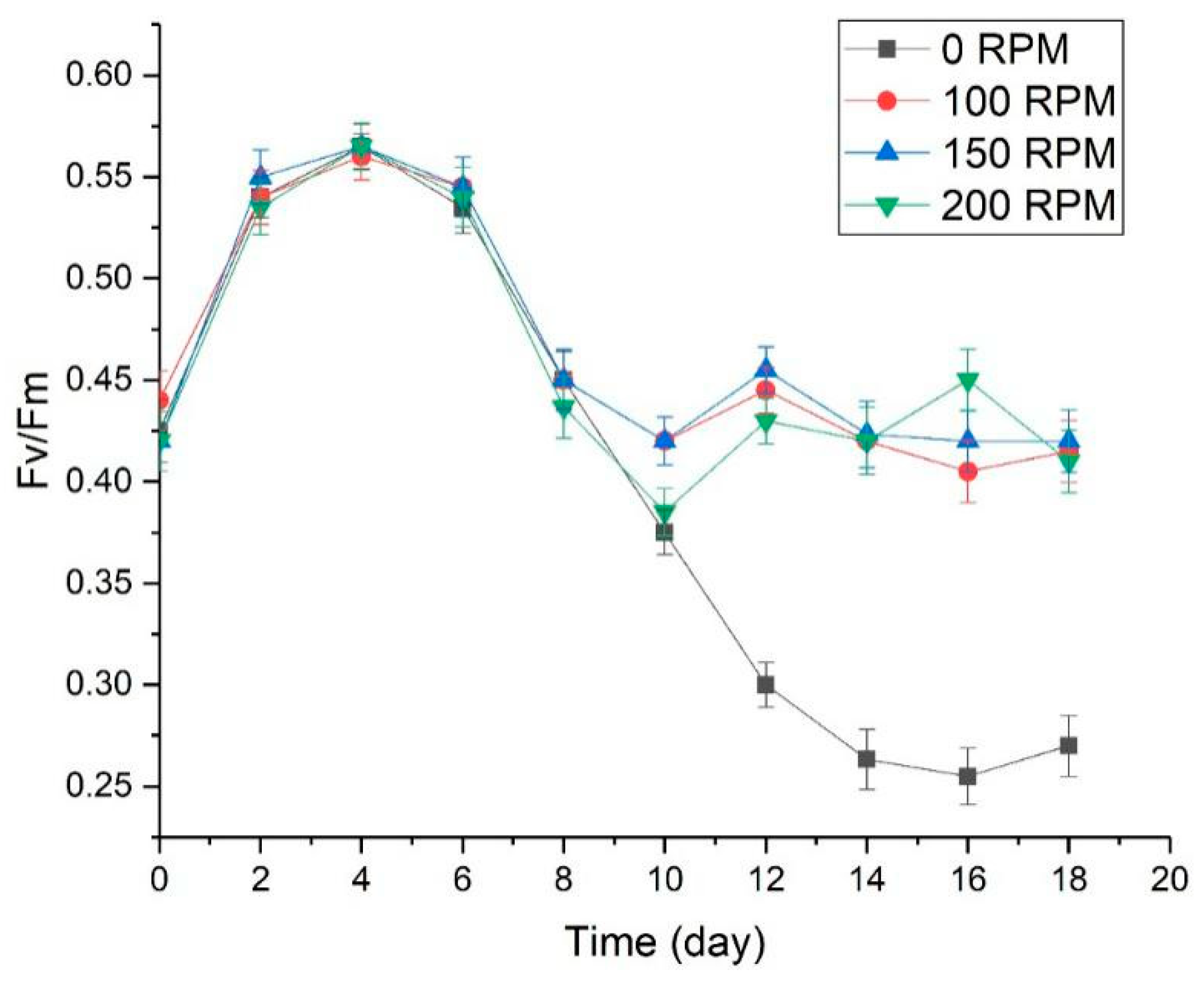
3.3. The Effect of Turbulence on Physiological Characteristics
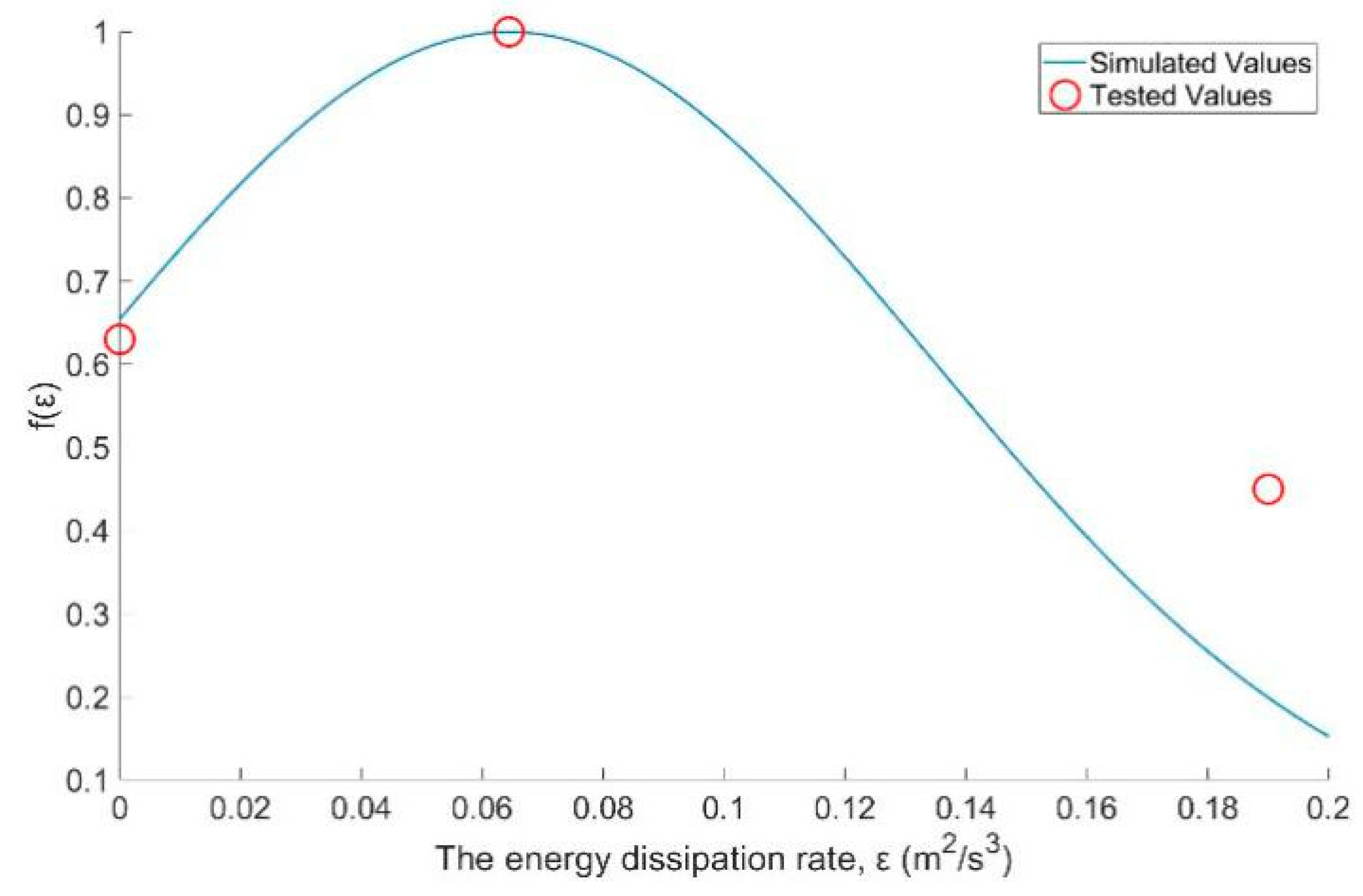
3.4. The Turbulence Effect Exponential Function
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xiao, M.; Li, M.; Reynolds, C.S. Colony formation in the cyanobacteriumMicrocystis. Biol. Rev. 2018, 93, 1399–1420. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Zhu, G.; Gao, G.; Zhang, Y.; Li, W.; Paerl, H.W.; Carmichael, W.W. A Drinking Water Crisis in Lake Taihu, China: Linkage to Climatic Variability and Lake Management. Environ. Manag. 2010, 45, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, R.P.; Sinha, R.P.; Incharoensakdi, A. The cyanotoxin-microcystins: Current overview. Rev. Environ. Sci. Bio/Technol. 2014, 13, 215–249. [Google Scholar] [CrossRef]
- Yang, X.; Wu, X.; Hao, H.-L.; He, Z.-L. Mechanisms and assessment of water eutrophication. J. Zhejiang Univ. Sci. B 2008, 9, 197–209. [Google Scholar] [CrossRef]
- Stone, R. China Aims to Turn Tide Against Toxic Lake Pollution. Science 2011, 333, 1210–1211. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, Z.; Yu, Y.; Shi, X. Interannual and Seasonal Shift between Microcystis and Dolichospermum: A 7-Year Investigation in Lake Chaohu, China. Water 2020, 12, 1978. [Google Scholar] [CrossRef]
- Garcia, A.; Sámano, M.L.; Juanes, J.A.; Medina, R.; Revilla, J.A.; Álvarez, C. Assessment of the effects of a port expansion on algae appearance in a costal bay through mathematical modelling. Application to San Lorenzo Bay (North Spain). Ecol. Model. 2010, 221, 1413–1426. [Google Scholar] [CrossRef]
- Ouyang, P.; Wang, C.; Wang, P.; Gan, X.; Wang, X.; Yang, C. Effects of Mixed Allelochemicals on the Growth of Microcystis Aeruginosa, Microcystin Production, Extracellular Polymeric Substances, and Water Quality. Water 2020, 12, 1861. [Google Scholar] [CrossRef]
- Ding, L.; Wu, J.Q.; Pang, Y.; Li, L.; Gao, G.; Hu, D.W. Simulation study on algal dynamics based on ecological flume experiment in Taihu Lake, China. Ecol. Eng. 2007, 31, 200–206. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Eutrophication: Time to Adjust Expectations Response. Science 2009, 324, 724–725. [Google Scholar] [CrossRef]
- Zhang, H.P.; Zhu, Y.P.; Li, F.P.; Chen, L. Nutrients in the wet deposition of Shanghai and ecological impacts. Phys. Chem. Earth. 2011, 36, 407–410. [Google Scholar] [CrossRef]
- Sullivan, J.M.; Swift, E.; Donaghay, P.L.; Rines, J.E. Small-scale turbulence affects the division rate and morphology of two red-tide dinoflagellates. Harmful Algae 2003, 2, 183–199. [Google Scholar] [CrossRef]
- Arnott, R.N.; Cherif, M.; Bryant, L.D.; Wain, D.J. Artificially generated turbulence: A review of phycological nanocosm, microcosm, and mesocosm experiments. Hydrobiologia 2021, 1–31. [Google Scholar] [CrossRef]
- Song, Y.; Zhang, L.-L.; Li, J.; Chen, M.; Zhang, Y.-W. Mechanism of the influence of hydrodynamics on Microcystis aeruginosa, a dominant bloom species in reservoirs. Sci. Total Environ. 2018, 636, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Hondzo, M.; Lyn, D. Quantified small-scale turbulence inhibits the growth of a green alga. Freshw. Biol. 1999, 41, 51–61. [Google Scholar] [CrossRef]
- Warnaars, T.A.; Hondzo, M. Small-scale fluid motion mediates growth and nutrient uptake of Selenastrum capricornutum. Freshw. Biol. 2006, 51, 999–1015. [Google Scholar] [CrossRef]
- Hondzo, M.M.; Kapur, A.; Lembi, C.A. The effect of small-scale fluid motion on the green alga Scenedesmus quadricauda. Hydrobiologia 1998, 364, 225–235. [Google Scholar] [CrossRef]
- Mitsuhashi, S.; Hosaka, K.; Tomonaga, E.; Muramatsu, H.; Tanishita, K. Effects of Shear-Flow on Photosynthesis in a Dilute Suspension of Microalgae. Appl. Microbiol. Biot. 1995, 42, 744–749. [Google Scholar] [CrossRef]
- Hondzo, M.; Warnaars, T.A. Coupled Effects of Small-Scale Turbulence and Phytoplankton Biomass in a Small Stratified Lake. J. Environ. Eng. 2008, 134, 954–960. [Google Scholar] [CrossRef]
- Missaghi, S.; Hondzo, M.; Sun, C.; Guala, M. Influence of fluid motion on growth and vertical distribution of cyanobacterium Microcystis aeruginosa. Aquat. Ecol. 2016, 50, 639–652. [Google Scholar] [CrossRef]
- Li, J.-X.; Yu, X.-Z.; Xing, Z.-G. Eutrophication model for the branch of Three-Gorges Reservoir. Adv. Water Sci. 2005, 16, 777. [Google Scholar]
- Long, T.-Y.; Wu, L.; Meng, G.-H.; Guo, W.-H. Numerical simulation for impacts of hydrodynamic conditions on algae growth in Chongqing Section of Jialing River, China. Ecol. Model. 2011, 222, 112–119. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Xia, K.; Yang, R.; Liu, X. Flow-disturbance considered simulation for algae growth in a river-lake system. Ecohydrology 2015, 9, 601–609. [Google Scholar] [CrossRef]
- Peters, F.; Marrase, C. Effects of turbulence on plankton: An overview of experimental evidence and some theoretical considerations. Mar. Ecol. Prog. Ser. 2000, 205, 291–306. [Google Scholar] [CrossRef]
- Sukhodolov, A.; Thiele, M.; Bungartz, H. Turbulence structure in a river reach with sand bed. Water Resour. Res. 1998, 34, 1317–1334. [Google Scholar] [CrossRef]
- Hondzo, M.; Haider, Z. Boundary mixing in a small stratified lake. Water Resour. Res. 2004, 40. [Google Scholar] [CrossRef]
- Li, N. Relationship between CO2 Concentration and Growth of Microcystis Aerugionsa. Master’s Thesis, Harbin Institute of Technology, Harbin, China, 2010. [Google Scholar]
- Liu, M.; Ma, J.; Kang, L.; Wei, Y.; He, Q.; Hu, X.; Li, H. Strong turbulence benefits toxic and colonial cyanobacteria in water: A potential way of climate change impact on the expansion of Harmful Algal Blooms. Sci. Total Environ. 2019, 670, 613–622. [Google Scholar] [CrossRef]
- KarpBoss, L.; Boss, E.; Jumars, P.A. Nutrient fluxes to planktonic osmotrophs in the presence of fluid motion. Oceanogr. Mar. Biol. 1996, 34, 71–107. [Google Scholar]
- Bolli, L.; Llaveria, G.; Garces, E.; Guadayol, O.; van Lenning, K.; Peters, F.; Berdalet, E. Modulation of ecdysal cyst and toxin dynamics of two Alexandrium (Dinophyceae) species under small-scale turbulence. Biogeosciences 2007, 4, 559–567. [Google Scholar] [CrossRef]
- Sournia, A. Form and Function in Marine-Phytoplankton. Biol. Rev. 1982, 57, 347–394. [Google Scholar] [CrossRef]
| Component | Content | |
|---|---|---|
| 1 | NaNO3 | 1.5 g/L |
| 2 | K2HPO4 | 0.04 g/L |
| 3 | MgSO4·7H2O | 0.075 g/L |
| 4 | CaCl2·2H2O | 0.036 g/L |
| 5 | Citric acid | 0.006 g/L |
| 6 | Ferric ammonium citrate | 0.006 g/L |
| 7 | EDTANa2 | 0.001 g/L |
| 8 | Na2CO3 | 0.02 g/L |
| 9 | A5 (Trace metal solution) | 1 mL/L |
| The Energy Dissipation Rate ε/(m2/s3) | |||
|---|---|---|---|
| Rotor Speed n/RPM | Average | Maximum | Minimum |
| 100 | 7.40 × 10−3 | 4.58 | 3.78 × 10−5 |
| 150 | 6.44 × 10−2 | 15.82 | 4.36 × 10−5 |
| 200 | 0.19 | 44.31 | 3.29 × 10−4 |
| 300 | 0.62 | 155.51 | 1.74 × 10−3 |
| 400 | 1.50 | 377.35 | 4.18 × 10−3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Cui, Y.; Zhang, Y.; Xu, H.; Li, F. Experimental Study of the Quantitative Impact of Flow Turbulence on Algal Growth. Water 2021, 13, 659. https://doi.org/10.3390/w13050659
Zhang H, Cui Y, Zhang Y, Xu H, Li F. Experimental Study of the Quantitative Impact of Flow Turbulence on Algal Growth. Water. 2021; 13(5):659. https://doi.org/10.3390/w13050659
Chicago/Turabian StyleZhang, Haiping, Yafei Cui, Yuehong Zhang, Hanling Xu, and Feipeng Li. 2021. "Experimental Study of the Quantitative Impact of Flow Turbulence on Algal Growth" Water 13, no. 5: 659. https://doi.org/10.3390/w13050659
APA StyleZhang, H., Cui, Y., Zhang, Y., Xu, H., & Li, F. (2021). Experimental Study of the Quantitative Impact of Flow Turbulence on Algal Growth. Water, 13(5), 659. https://doi.org/10.3390/w13050659






