Invertebrate and Microbial Response to Hyporheic Restoration of an Urban Stream
Abstract
1. Introduction
2. Materials and Methods
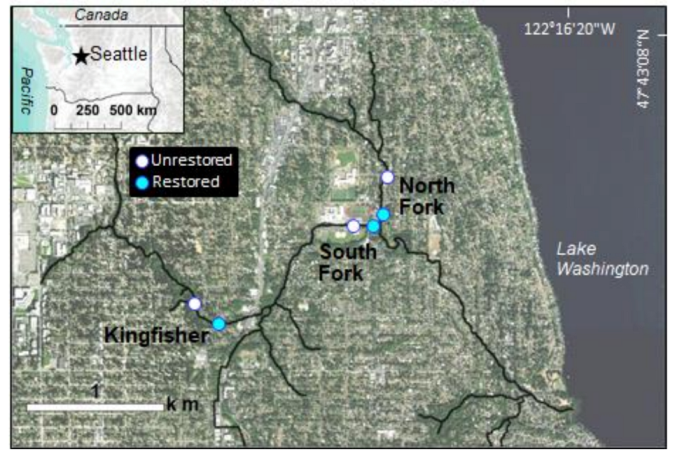
2.1. Study Region
2.2. Experimental Design
2.3. Sample Parameters
2.3.1. Environmental Covariates
2.3.2. Microbes
2.3.3. Invertebrates
2.4. Data Analyses
3. Results
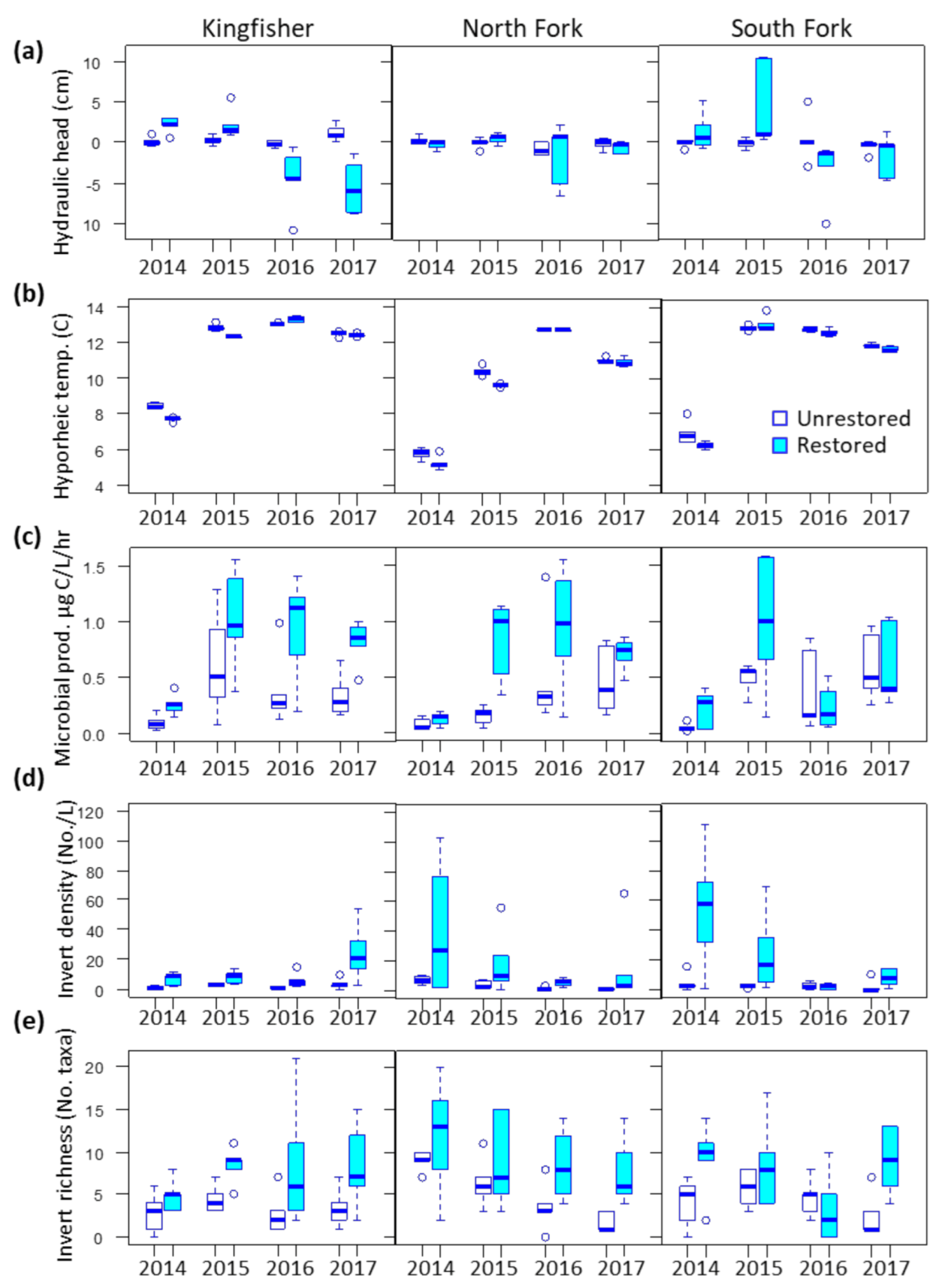
3.1. Environmental Covariates
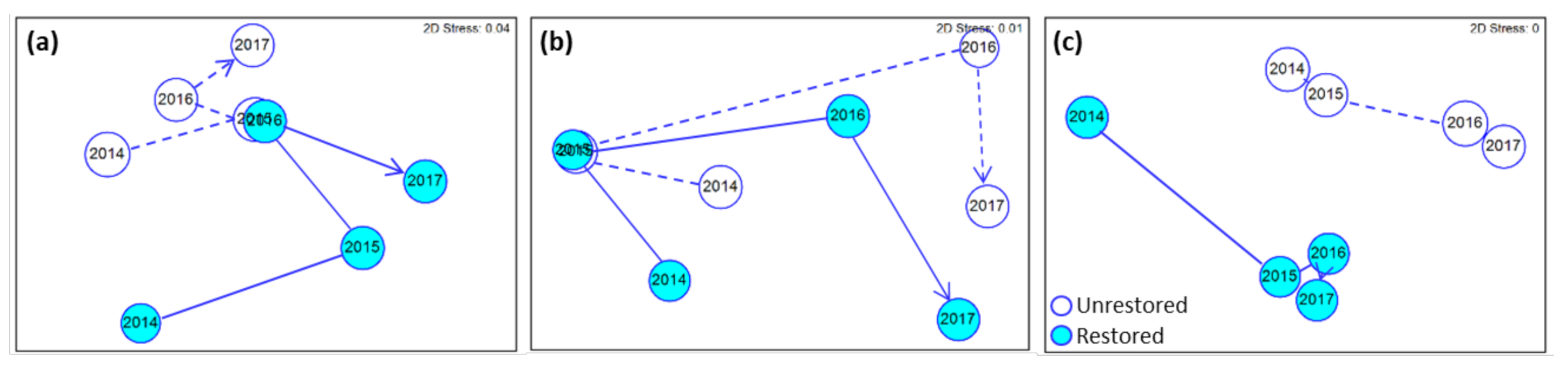
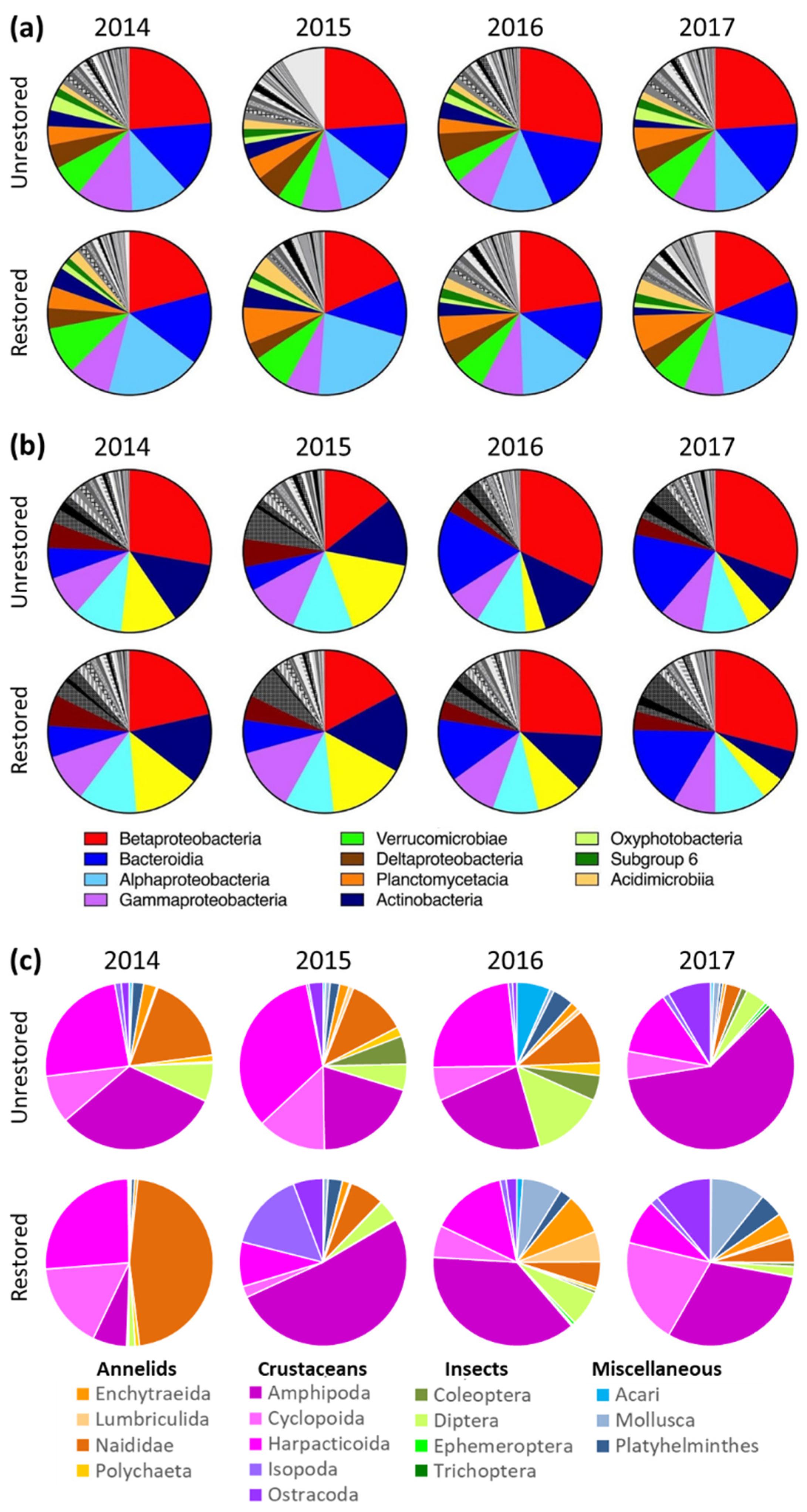
3.2. Microbes
3.3. Invertebrates
3.4. Relationship between Response Variables
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walsh, C.J.; Booth, D.B.; Burns, M.J.; Fletcher, T.D.; Hale, R.L.; Hoang, L.N.; Livingston, G.; Rippy, M.A.; Roy, A.H.; Scoggins, M. Principles for urban stormwater management to protect stream ecosystems. Freshw. Sci. 2016, 35, 398–411. [Google Scholar] [CrossRef]
- Levin, P.S.; Howe, E.R.; Robertson, J.C. Impacts of stormwater on coastal ecosystems: The need to match the scales of management objectives and solutions. Philos. Trans. R. Soc. B 2020, 375, 20190460. [Google Scholar] [CrossRef]
- Shuster, W.D.; Bonta, J.; Thurston, H.; Warnemuende, E.; Smith, D. Impacts of impervious surface on watershed hydrology: A review. Urban Water J. 2005, 2, 263–275. [Google Scholar] [CrossRef]
- Elmore, A.J.; Kaushal, S.S. Disappearing headwaters: Patterns of stream burial due to urbanization. Front. Ecol. Environ. 2008, 6, 308–312. [Google Scholar] [CrossRef]
- McGrane, S.J. Impacts of urbanisation on hydrological and water quality dynamics, and urban water management: A review. Hydrol. Sci. J. 2016, 61, 2295–2311. [Google Scholar] [CrossRef]
- Tian, Z.; Zhao, H.; Peter, K.T.; Gonzalez, M.; Wetzel, J.; Wu, C.; Hu, X.; Prat, J.; Mudrock, E.; Hettinger, R. A ubiquitous tire rubber–derived chemical induces acute mortality in coho salmon. Science 2020, 371, 185–189. [Google Scholar] [CrossRef]
- Walsh, C.J.; Roy, A.H.; Feminella, J.W.; Cottingham, P.D.; Groffman, P.M.; Morgan, R.P. The urban stream syndrome: Current knowledge and the search for a cure. J. N. Am. Benthol. Soc. 2005, 24, 706–723. [Google Scholar] [CrossRef]
- Dhakal, K.P.; Chevalier, L.R. Urban stormwater governance: The need for a paradigm shift. Environ. Manag. 2016, 57, 1112–1124. [Google Scholar] [CrossRef]
- Prudencio, L.; Null, S.E. Stormwater management and ecosystem services: A review. Environ. Res. Lett. 2018, 13, 033002. [Google Scholar] [CrossRef]
- Yang, B.; Li, S. Green infrastructure design for stormwater runoff and water quality: Empirical evidence from large watershed-scale community developments. Water 2013, 5, 2038–2057. [Google Scholar] [CrossRef]
- McIntyre, J.; Davis, J.; Hinman, C.; Macneale, K.; Anulacion, B.; Scholz, N.; Stark, J. Soil bioretention protects juvenile salmon and their prey from the toxic impacts of urban stormwater runoff. Chemosphere 2015, 132, 213–219. [Google Scholar] [CrossRef]
- Ward, J.; Tockner, K.; Schiemer, F. Biodiversity of floodplain river ecosystems: Ecotones and connectivity1. River Res. Appl. 1999, 15, 125–139. [Google Scholar] [CrossRef]
- Opperman, J.J.; Luster, R.; McKenney, B.A.; Roberts, M.; Meadows, A.W. Ecologically functional floodplains: Connectivity, flow regime, and scale 1. JAWRA J. Am. Water Resour. Assoc. 2010, 46, 211–226. [Google Scholar] [CrossRef]
- Dixon, S.J.; Sear, D.A.; Odoni, N.A.; Sykes, T.; Lane, S.N. The effects of river restoration on catchment scale flood risk and flood hydrology. Earth Surface Process. Landf. 2016, 41, 997–1008. [Google Scholar] [CrossRef]
- Ahilan, S.; Guan, M.; Sleigh, A.; Wright, N.; Chang, H. The influence of floodplain restoration on flow and sediment dynamics in an urban river. J. Flood Risk Manag. 2018, 11, S986–S1001. [Google Scholar] [CrossRef]
- O’Donnell, E.C.; Thorne, C.R.; Yeakley, J.A.; Chan, F.K.S. Sustainable flood risk and stormwater management in blue-green cities; an interdisciplinary case study in Portland, Oregon. JAWRA J. Am. Water Resour. Assoc. 2020, 56, 757–775. [Google Scholar] [CrossRef]
- Beechie, T.J.; Sear, D.A.; Olden, J.D.; Pess, G.R.; Buffington, J.M.; Moir, H.; Roni, P.; Pollock, M.M. Process-based principles for restoring river ecosystems. BioScience 2010, 60, 209–222. [Google Scholar] [CrossRef]
- Vietz, G.J.; Rutherfurd, I.D.; Fletcher, T.D.; Walsh, C.J. Thinking outside the channel: Challenges and opportunities for protection and restoration of stream morphology in urbanizing catchments. Landsc. Urban Plan 2016, 145, 34–44. [Google Scholar] [CrossRef]
- Wohl, E. Connectivity in rivers. Progress Phys. Geogr. 2017, 41, 345–362. [Google Scholar] [CrossRef]
- Orghidan, T. Ein neuer Lebensraum des unterirdischen Wassers, der hyporheische Biotop. Arch. Hydrobiol. 1959, 55, 392–414. [Google Scholar]
- Boulton, A.J.; Findlay, S.; Marmonier, P.; Stanley, E.H.; Valett, H.M. The functional significance of the hyporheic zone in streams and rivers. Annu. Rev. Ecol. Syst. 1998, 29, 59–81. [Google Scholar] [CrossRef]
- Fischer, H.; Kloep, F.; Wilzcek, S.; Pusch, M.T. A river’s liver—Microbial processes within the hyporheic zone of a large lowland river. Biogeochemistry 2005, 76, 349–371. [Google Scholar] [CrossRef]
- Hancock, P.J.; Boulton, A.J.; Humphreys, W.F. Aquifers and hyporheic zones: Towards an ecological understanding of groundwater. Hydrogeol. J. 2005, 13, 98–111. [Google Scholar] [CrossRef]
- Lewandowski, J.; Arnon, S.; Banks, E.; Batelaan, O.; Betterle, A.; Broecker, T.; Coll Mora, C.; Drummond, J.; Gaona, J.; Galloway, J.; et al. Is the Hyporheic Zone Relevant beyond the Scientific Community? Water 2019, 11, 2230. [Google Scholar] [CrossRef]
- Robertson, A.L.; Wood, P.J. Ecology of the hyporheic zone: Origins, current knowledge and future directions. Fundam. Appl. Limnol. 2010, 176, 279–289. [Google Scholar] [CrossRef]
- Vorste, R.V.; Corti, R.; Sagouis, A.; Datry, T. Invertebrate communities in gravel-bed, braided rivers are highly resilient to flow intermittence. Freshw. Sci. 2016, 35, 164–177. [Google Scholar] [CrossRef]
- Stanford, J.A.; Ward, J.V. The Hyporheic Habitat of River Ecosystems. Nature 1988, 335, 64–66. [Google Scholar] [CrossRef]
- Gilbert, J.; Doleolivier, M.J.; Marmonier, P.; Vervier, P. Surface Water-Groundwater Ecotones. Ecol. Manag. Aquat. Terr. Ecotones 1990, 4, 199–225. [Google Scholar]
- Boulton, A.J. Hyporheic rehabilitation in rivers: Restoring vertical connectivity. Freshw. Biol. 2007, 52, 632–650. [Google Scholar] [CrossRef]
- Kasahara, T.; Datry, T.; Mutz, M.; Boulton, A.J. Treating causes not symptoms: Restoration of surface-groundwater interactions in rivers. Mar. Freshw. Res. 2009, 60, 976–981. [Google Scholar] [CrossRef]
- Hester, E.T.; Gooseff, M.N. Moving Beyond the Banks: Hyporheic Restoration Is Fundamental to Restoring Ecological Services and Functions of Streams. Environ. Sci. Technol. 2010, 44, 1521–1525. [Google Scholar] [CrossRef]
- Mendoza-Lera, C.; Datry, T. Relating hydraulic conductivity and hyporheic zone biogeochemical processing to conserve and restore river ecosystem services. Sci. Total Environ. 2017, 579, 1815–1821. [Google Scholar] [CrossRef]
- Magliozzi, C.; Coro, G.; Grabowski, R.C.; Packman, A.I.; Krause, S. A multiscale statistical method to identify potential areas of hyporheic exchange for river restoration planning. Environ. Model. Softw. 2019, 111, 311–323. [Google Scholar] [CrossRef]
- Herzog, S.P.; Higgins, C.P.; McCray, J.E. Engineered Streambeds for Induced Hyporheic Flow: Enhanced Removal of Nutrients, Pathogens, and Metals from Urban Streams. J. Environ. Eng. 2015, 142, 04015053. [Google Scholar] [CrossRef]
- Bakke, P.D.; Hrachovec, M.; Lynch, K.D. Hyporheic Process Restoration: Design and Performance of an Engineered Streambed. Water 2020, 12, 425. [Google Scholar] [CrossRef]
- Peter, K.T.; Herzog, S.; Tian, Z.Y.; Wu, C.; McCray, J.E.; Lynch, K.; Kolodziej, E.P. Evaluating emerging organic contaminant removal in an engineered hyporheic zone using high resolution mass spectrometry. Water Res. 2019, 150, 140–152. [Google Scholar] [CrossRef]
- Crispell, J.K.; Endreny, T.A. Hyporheic exchange flow around constructed in-channel structures and implications for restoration design. Hydrol. Process. 2009, 23, 1158–1168. [Google Scholar] [CrossRef]
- Knust, A.E.; Warwick, J.J. Using a fluctuating tracer to estimate hyporheic exchange in restored and unrestored reaches of the Truckee River, Nevada, USA. Hydrol. Process. 2009, 23, 1119–1130. [Google Scholar] [CrossRef]
- Peralta-Maraver, I.; Perkins, D.M.; Thompson, M.S.; Fussmann, K.; Reiss, J.; Robertson, A.L. Comparing biotic drivers of litter breakdown across stream compartments. J. Anim. Ecol. 2019, 88, 1146–1157. [Google Scholar] [CrossRef]
- Weatherill, J.J.; Atashgahi, S.; Schneidewind, U.; Krause, S.; Ullah, S.; Cassidy, N.; Rivett, M.O. Natural attenuation of chlorinated ethenes in hyporheic zones: A review of key biogeochemical processes and in-situ transformation potential. Water Res. 2018, 128, 362–382. [Google Scholar] [CrossRef]
- Lawrence, J.E.; Skold, M.E.; Hussain, F.A.; Silverman, D.R.; Resh, V.H.; Sedlak, D.L.; Luthy, R.G.; McCray, J.E. Hyporheic Zone in Urban Streams: A Review and Opportunities for Enhancing Water Quality and Improving Aquatic Habitat by Active Management. Environ. Eng. Sci. 2013, 30, 480–501. [Google Scholar] [CrossRef]
- Grimm, N.B.; Baxter, C.V.; Crenshaw, C.L. Surface-subsurface interactions in streams. In Methods in Stream Ecology; Hauer, F.R., Lamberti, G.A., Eds.; Academic Press: San Diego, CA, USA, 2007; pp. 761–782. [Google Scholar]
- Drummond, J.; Aubeneau, A.; Packman, A. Stochastic modeling of fine particulate organic carbon dynamics in rivers. Water Resour. Res. 2014, 50, 4341–4356. [Google Scholar] [CrossRef]
- City of Seattle. State of the Waters. In Volume I: Seattle Watercourses; City of Seattle: Seattle, WA, USA, 2007; Volume I, p. 262. Available online: https://www.seattle.gov/util/cs/groups/public/@spu/@conservation/documents/webcontent/spu01_003413.pdf (accessed on 4 November 2020).
- Alberti, M.; Booth, D.; Hill, K.; Coburn, B.; Avolio, C.; Coe, S.; Spirandelli, D. The impact of urban patterns on aquatic ecosystems: An empirical analysis in Puget lowland sub-basins. Landsc. Urban Plan. 2007, 80, 345–361. [Google Scholar] [CrossRef]
- Brett, M.T.; Arhonditsis, G.B.; Mueller, S.E.; Hartley, D.M.; Frodge, J.D.; Funke, D.E. Non-point-source impacts on stream nutrient concentrations along a forest to urban gradient. Environ. Manag. 2005, 35, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Reidy, C. Variability of Hyporheic Zones in Puget Sound Lowland Streams; University of Washington: Seattle, WA, USA, 2004. [Google Scholar]
- Scholz, N.L.; Myers, M.S.; McCarthy, S.G.; Labenia, J.S.; McIntyre, J.K.; Ylitalo, G.M.; Rhodes, L.D.; Laetz, C.A.; Stehr, C.M.; French, B.L.; et al. Recurrent Die-Offs of Adult Coho Salmon Returning to Spawn in Puget Sound Lowland Urban Streams. PLoS ONE 2011, 6, e28013. [Google Scholar] [CrossRef]
- Morley, S.A.; Karr, J.R. Assessing and restoring the health of urban streams in the Puget Sound basin. Conserv. Biol. 2002, 16, 1498–1509. [Google Scholar] [CrossRef]
- Puget Sound Stream Benthos. Available online: https://pugetsoundstreambenthos.org/ (accessed on 11 April 2020).
- Valderrama, J.C. The simultaneous analysis of total nitrogen and total phosphorus in natural waters. Mar. Chem. 1981, 10, 109–122. [Google Scholar] [CrossRef]
- Scientific Committee on Oceanic Research. Protocols for the Joint Global Ocean Flux Study (JGOFS) Core Measurements; Intergovernmental Oceanographic Commission: Paris, France, 1994; Volume 29, p. 170. [Google Scholar]
- Hambrook Berkman, J.A.; Canova, M.G. Algal Biomass Indicators (ver.1.0): U.S. Geological Survey Techniques of Water Resources Investigations; Book 9, Chapter A7, Section 7.4; U.S. Geological Survey: Reston, VA, USA, 2007. [CrossRef]
- Longnecker, K.; Sherr, B.F.; Sherr, E.B. Variation in cell-specific rates of leucine and thymidine incorporation by marine bacteria with high and with low nucleic acid content off the Oregon coast. Aquat. Microb. Ecol. 2006, 43, 113–125. [Google Scholar] [CrossRef]
- Sherr, E.B.; Sherr, B.F.; Longnecker, K. Distribution of bacterial abundance and cell-specific nucleic acid content in the Northeast Pacific Ocean. Deep Sea Res. Part 1 2006, 53, 713–725. [Google Scholar] [CrossRef]
- Green, M.R.; Sambrook, J. Isolation of High-Molecular-Weight DNA from Suspension Cultures of Mammalian Cells Using Proteinase K and Phenol. Cold Spring Harb. Protoc. 2018, 2018. [Google Scholar] [CrossRef]
- Illumina. 16S Metagenomic Sequencing Library Preparation; Illumina: San Diego, CA, USA, 2013. [Google Scholar]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Author Correction: Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 1091. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, P.; Parfrey, L.W.; Yarza, P.; Gerken, J.; Pruesse, E.; Quast, C.; Schweer, T.; Peplies, J.; Ludwig, W.; Glockner, F.O. The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks. Nucleic Acids Res. 2014, 42, D643–D648. [Google Scholar] [CrossRef]
- Pruesse, E.; Peplies, J.; Glöckner, F.O. SINA: Accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics 2012, 28, 1823–1829. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User Manual/Tutorial; PRIMER-E: Plymouth, UK, 2006. [Google Scholar]
- Leavy, T.R.; Bakke, P.; Peters, R.J.; Morley, S.A. Influence of Habitat Complexity and Floodplain Reconnection Projects on the Physical and Biological Conditions of Seattle Urban Streams: Pre Project Monitoring, 2005–2009; U.S. Fish and Wildlife Service, Western Washington Fish and Wildlife Office: Lacey, WA, USA, 2010.
- Morley, S.A.; Hall, J.E.; Chamberlin, J.W.; Hanson, K. Thornton Creek Restoration: Baseline Project Effectiveness Monitoring, 2012–2013; Report of the National Marine Fisheries Service to the City of Seattle; Department of Public Utilities: Seattle, WA, USA, 2013.
- R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E: Plymouth, UK, 2008. [Google Scholar]
- Puget Sound Ecosystem Monitoring Program Marine Waters Workgroup. Puget Sound Marine Waters 2017 Overview; Moore, S.K., Wold, R., Stark, K., Bos, J., Williams, P., Hamel, N., Kim, S., Brown, A., Krembs, C., Newton, J., Eds.; Available online: https://www.psp.wa.gov/PSmarinewatersoverview.php (accessed on 11 April 2020).
- Gaudes, A.; Artigas, J.; Romani, A.; Sabater, S.; Munoz, I. Contribution of microbial and invertebrate communities to leaf litter colonization in a Mediterranean stream. J. N. Am. Benthol. Soc. 2009, 28, 34–43. [Google Scholar] [CrossRef]
- Krause, S.; Hannah, D.M.; Fleckenstein, J.H.; Heppell, C.M.; Kaeser, D.; Pickup, R.; Pinay, G.; Robertson, A.L.; Wood, P.J. Inter-disciplinary perspectives on processes in the hyporheic zone. Ecohydrology 2011, 4, 481–499. [Google Scholar] [CrossRef]
- Story, A.; Moore, R.D.; Macdonald, J.S. Stream temperatures in two shaded reaches below cutblocks and logging roads: Downstream cooling linked to subsurface hydrology. Can. J. For. Res. 2003, 33, 1383–1396. [Google Scholar] [CrossRef]
- Hannah, D.M.; Malcolm, I.A.; Soulsby, C.; Youngson, A.F. Heat exchanges and temperatures within a salmon spawning stream in the cairngorms, Scotland: Seasonal and sub-seasonal dynamics. River Res. Appl. 2004, 20, 635–652. [Google Scholar] [CrossRef]
- Storey, R.G.; Fulthorpe, R.R.; Williams, D.D. Perspectives and predictions on the microbial ecology of the hyporheic zone. Freshw. Biol. 1999, 41, 119–130. [Google Scholar] [CrossRef]
- Peralta-Maraver, I.; Reiss, J.; Robertson, A.L. Interplay of hydrology, community ecology and pollutant attenuation in the hyporheic zone. Sci. Total Environ. 2018, 610, 267–275. [Google Scholar] [CrossRef]
- Duff, J.; Triska, F. Nitrogen Biogeochemistry and Surface–Subsurface Exchange in Streams. In Streams and Ground Waters; Academic Press: San Diego, CA, USA, 2000; pp. 197–220. [Google Scholar] [CrossRef]
- Merill, L.; Tonjes, D.J. A Review of the Hyporheic Zone, Stream Restoration, and Means to Enhance Denitrification. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2337–2379. [Google Scholar] [CrossRef]
- Ohta, Y.; Nishi, S.; Hasegawa, R.; Hatada, Y. Combination of six enzymes of a marine Novosphingobium converts the stereoisomers of β-O-4 lignin model dimers into the respective monomers. Sci. Rep. 2015, 5, 15105. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Fu, H.-Y.; Lee, T.-H.; Shih, C.-J.; Huang, L.; Wang, Y.-S.; Ismail, W.; Chiang, Y.-R. Estrogen degraders and estrogen degradation pathway identified in an activated sludge. Appl. Environ. Microb. 2018, 84. [Google Scholar] [CrossRef]
- Wagner, K.; Bengtsson, M.M.; Besemer, K.; Sieczko, A.; Burns, N.R.; Herberg, E.R.; Battin, T.J. Functional and Structural Responses of Hyporheic Biofilms to Varying Sources of Dissolved Organic Matter. Appl. Environ. Microb. 2014, 80, 6004–6012. [Google Scholar] [CrossRef]
- DeLong, E.F.; Franks, D.G.; Alldredge, A.L. Phylogenetic diversity of aggregate-attached vs. free-living marine bacterial assemblages. Limnol. Oceanogr. 1993, 38, 924–934. [Google Scholar] [CrossRef]
- Fuchsman, C.A.; Staley, J.T.; Oakley, B.B.; Kirkpatrick, J.B.; Murray, J.W. Free-living and aggregate-associated Planctomycetes in the Black Sea. FEMS Microbiol. Ecol. 2012, 80, 402–416. [Google Scholar] [CrossRef] [PubMed]
- Glöckner, F.O.; Kube, M.; Bauer, M.; Teeling, H.; Lombardot, T.; Ludwig, W.; Gade, D.; Beck, A.; Borzym, K.; Heitmann, K. Complete genome sequence of the marine planctomycete Pirellula sp. strain 1. Proc. Natl. Acad. Sci. USA 2003, 100, 8298–8303. [Google Scholar] [CrossRef]
- Zhu, P.; Ye, Y.; Pei, F.; Lu, K. Characterizing the structural diversity of a bacterial community associated with filter materials in recirculating aquaculture systems of Scortum barcoo. Can. J. Microbiol. 2012, 58, 303–310. [Google Scholar] [CrossRef]
- Sliva, L.; Williams, D.D. Responses of hyporheic meiofauna to habitat manipulation. Hydrobiologia 2005, 548, 217–232. [Google Scholar] [CrossRef]
- Boulton, A.J.; Datry, T.; Kasahara, T.; Mutz, M.; Stanford, J.A. Ecology and management of the hyporheic zone: Stream-groundwater interactions of running waters and their floodplains. J. N. Am. Benthol. Soc. 2010, 29, 26–40. [Google Scholar] [CrossRef]
- Descloux, S.; Datry, T.; Usseglio-Polatera, P. Trait-based structure of invertebrates along a gradient of sediment colmation: Benthos versus hyporheos responses. Sci. Total Environ. 2014, 466, 265–276. [Google Scholar] [CrossRef]
- Wagenhoff, A.; Olsen, D. Does large woody debris affect the hyporheic ecology of a small New Zealand pasture stream? N. Zeal. J. Mar. Freshw. 2014, 48, 547–559. [Google Scholar] [CrossRef]
- Magliozzi, C.; Usseglio-Polatera, P.; Meyer, A.; Grabowski, R.C. Functional traits of hyporheic and benthic invertebrates reveal importance of wood-driven geomorphological processes in rivers. Funct. Ecol. 2019, 33, 1758–1770. [Google Scholar] [CrossRef]
- Sundermann, A.; Stoll, S.; Haase, P. River restoration success depends on the species pool of the immediate surroundings. Ecol. Appl. 2011, 21, 1962–1971. [Google Scholar] [CrossRef] [PubMed]
- Hester, E.T.; Gooseff, M.N. Hyporheic restoration in streams and rivers. In Stream Restoration in Dynamic Fluvial Systems: Scientific Approaches, Analyses, and Tools; Simon, A., Bennett, S., Castro, J., Eds.; American Geophysical Union: Washington, DC, USA, 2011; Volume 194. [Google Scholar]
- Blakely, T.J.; Harding, J.S.; McIntosh, A.R.; Winterbourn, M.J. Barriers to the recovery of aquatic insect communities in urban streams. Freshw. Biol. 2006, 51, 1634–1645. [Google Scholar] [CrossRef]
- Morley, S.A.; Rhodes, L.D.; Baxter, A.E.; Goetz, G.W.; Wells, A.H.; Lynch, K.D. Invertebrate, Microbial, and Environmental Data from Surface and Hyporheic Waters of Urban and Forested Streams of the Cedar River-Lake Washington Watershed; NOAA Data Report NMFS-NWFSC-DR-2021; U.S. Department of Commerce: Washington, DC, USA, 2021. [CrossRef]

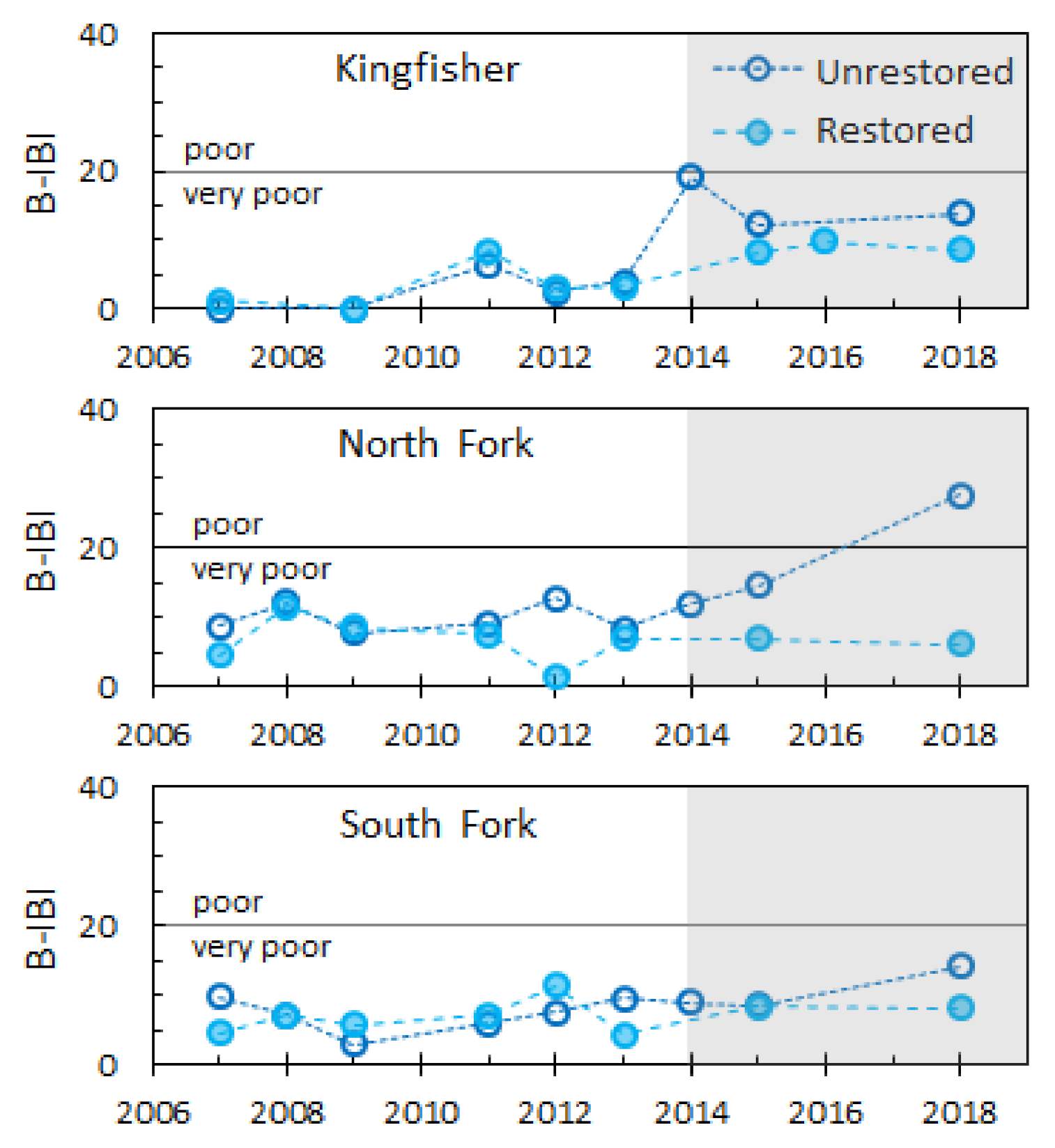
| Variable | Site | n | Reach | Year | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|---|---|---|
| Microbial diversity (H’) | KF | 40 | ns | 2014 a, 2015 a, 2016 b, 2017 c | - | - | - | - |
| NF | 39 | ns | 2015 a, 2014 ab, 2016 ab, 2017 c | - | - | - | - | |
| SF | 40 | U > R | 2015 a, 2014 ab, 2016 b, 2017 c | - | - | - | - | |
| Microbial richness (d) | KF | 40 | ns | 2014 a, 2015 a, 2017 b, 2016 c | - | - | - | - |
| NF | 39 | ns | 2015 a, 2014 ab, 2016 b, 2017 b | - | - | - | - | |
| SF | 40 | U > R | 2015 a, 2014 b, 2017 c, 2016 c | ns | ns | U > R | ns | |
| Microbial evenness (J) | KF | 40 | ns | 2016 a, 2014 b, 2015 b, 2017 b | - | - | - | - |
| NF | 39 | ns | ns | - | - | - | - | |
| SF | 40 | ns | 2016 a, 2014 ab, 2015 b, 2017 c | - | - | - | - | |
| Invertebrate density (No L−1) | KF | 40 | R > U | ns | - | - | - | - |
| NF | 40 | R > U | 2017 a, 2016 a, 2015 ab, 2014 b | - | - | - | - | |
| SF | 39 | R > U | ns | - | - | - | - | |
| Invertebrate richness (No. taxa) | KF | 40 | R > U | ns | - | - | - | - |
| NF | 40 | R > U | 2017 a, 2016 ab, 2015 ab, 2014 b | - | - | - | - | |
| SF | 39 | R > U | ns | R > U | ns | ns | R > U |
| Community | Site | n | Reach | Year | Pairwise Year Contrasts Contrasts | Reach Year | Pairwise Reach Contrasts | |||
|---|---|---|---|---|---|---|---|---|---|---|
| x Year | 2014 | 2015 | 2016 | 2017 | ||||||
| Microbes: particle- associated | KF | 40 | 0.001 | ns | - | 0.001 | 0.004 | ns | 0.010 | 0.009 |
| NF | 39 | 0.040 | 0.001 | 2014 a, 2015 b, 2016 c, 2017 d | 0.001 | 0.009 | 0.006 | ns | ns | |
| SF | 37 | 0.001 | ns | - | 0.006 | 0.017 | 0.024 | 0.027 | ns | |
| Microbes: planktonic | KF | 39 | 0.002 | 0.017 | 2014 a, 2015 b, 2016 c, 2017 d | 0.013 | 0.009 | ns | ns | 0.005 |
| NF | 38 | ns | 0.013 | 2014 a, 2015 b, 2016 c, 2017 d | ns | - | - | - | - | |
| SF | 39 | 0.028 | 0.001 | 2014 a, 2015 b, 2016 c, 2017 d | 0.001 | 0.018 | ns | 0.009 | ns | |
| Invertebrates | KF | 40 | 0.001 | 0.001 | 2014 a, 2015 b, 2016 bc, 2017 c | ns | - | - | - | - |
| NF | 40 | 0.002 | 0.001 | 2014 a, 2015 b, 2016 b, 2017 c | 0.022 | ns | ns | 0.009 | 0.023 | |
| SF | 39 | 0.007 | 0.012 | 2014 a, 2015 b, 2016 ab, 2017 b | ns | - | - | - | - | |
| Particle Microbes | Planktonic Microbes | Inverte- Brates | ||||
|---|---|---|---|---|---|---|
| Variables | U | R | U | R | U | R |
| Hydraulic head | ||||||
| Temperature | ||||||
| Conductivity | ||||||
| Dissolved oxygen | ||||||
| Dissolved oxygen (surface) | ||||||
| Ammonium | ||||||
| Nitrate | ||||||
| Nitrite | ||||||
| Total nitrogen | ||||||
| Silicate | ||||||
| Phosphate | ||||||
| Dissolved organic carbon | ||||||
| Particulate organic matter | ||||||
| Microbial production | ||||||
| Bacterial abundance | ||||||
| Model R2 | 0.26 | 0.38 | 0.42 | 0.45 | 0.15 | 0.18 |
| Spearman’s rho | 0.55 ** | 0.43 ** | 0.41 ** | 0.48 ** | 0.36 * | 0.22 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morley, S.A.; Rhodes, L.D.; Baxter, A.E.; Goetz, G.W.; Wells, A.H.; Lynch, K.D. Invertebrate and Microbial Response to Hyporheic Restoration of an Urban Stream. Water 2021, 13, 481. https://doi.org/10.3390/w13040481
Morley SA, Rhodes LD, Baxter AE, Goetz GW, Wells AH, Lynch KD. Invertebrate and Microbial Response to Hyporheic Restoration of an Urban Stream. Water. 2021; 13(4):481. https://doi.org/10.3390/w13040481
Chicago/Turabian StyleMorley, Sarah A., Linda D. Rhodes, Anne E. Baxter, Giles W. Goetz, Abigail H. Wells, and Katherine D. Lynch. 2021. "Invertebrate and Microbial Response to Hyporheic Restoration of an Urban Stream" Water 13, no. 4: 481. https://doi.org/10.3390/w13040481
APA StyleMorley, S. A., Rhodes, L. D., Baxter, A. E., Goetz, G. W., Wells, A. H., & Lynch, K. D. (2021). Invertebrate and Microbial Response to Hyporheic Restoration of an Urban Stream. Water, 13(4), 481. https://doi.org/10.3390/w13040481






