A Case Study of the Effects of Management Interventions on the Phosphorus Dynamics at a Coastal, Eutrophic, Caribbean lagoon (Laguna Cartagena, Puerto Rico)
Abstract
1. Introduction
2. Materials and Methods
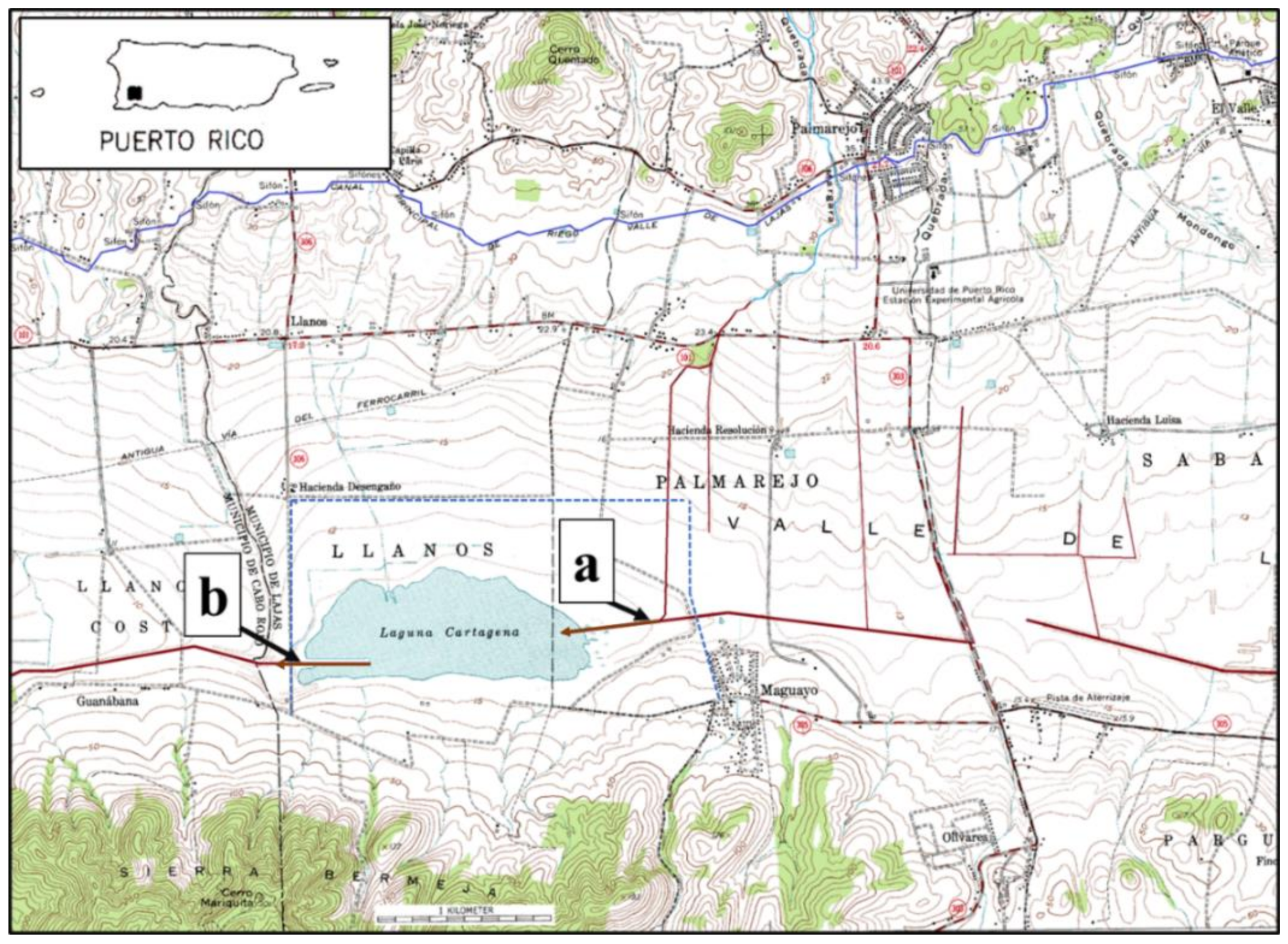
2.1. Description of the Study Site
2.2. Sampling and Processing
2.3. Statistical Analyses
3. Results
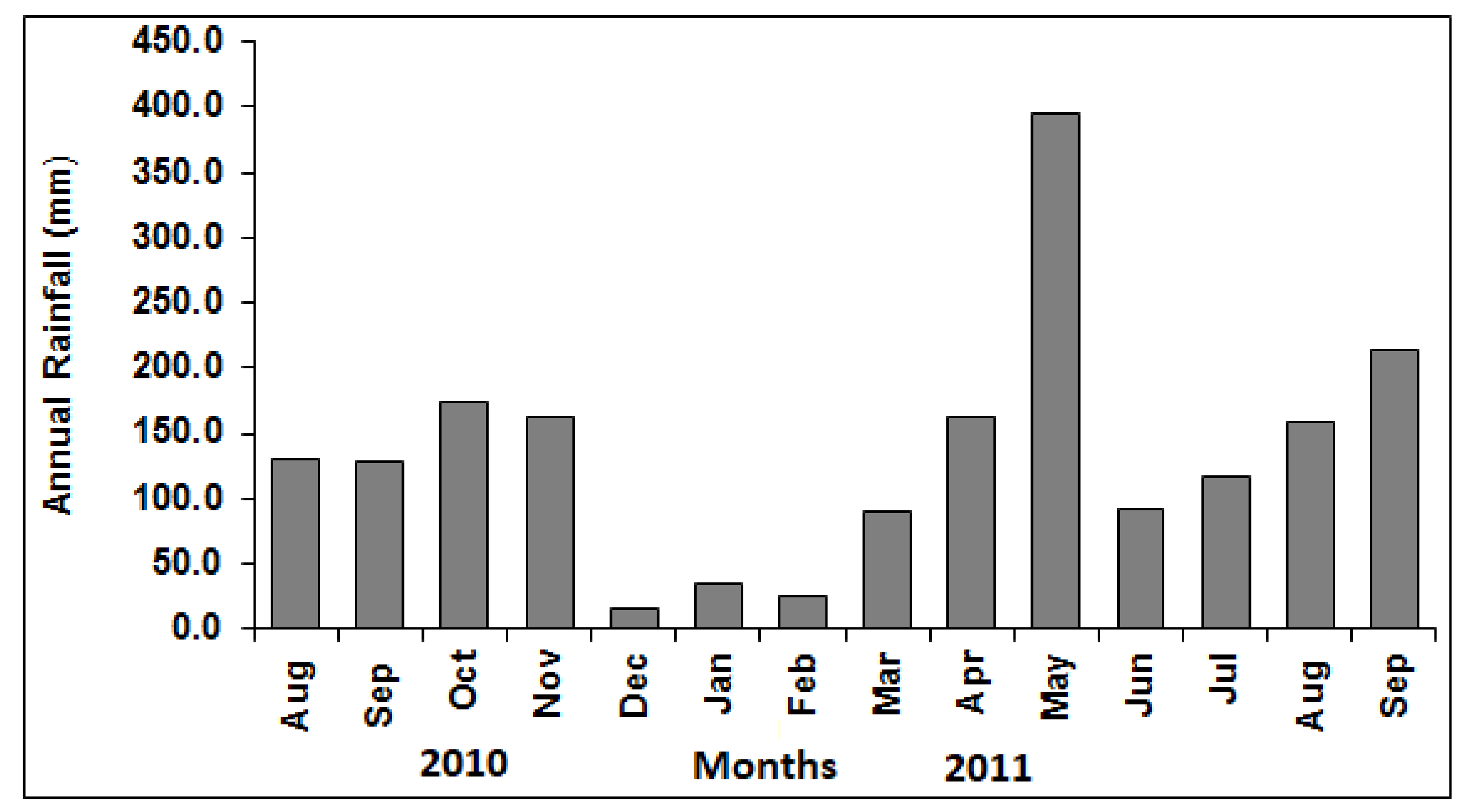
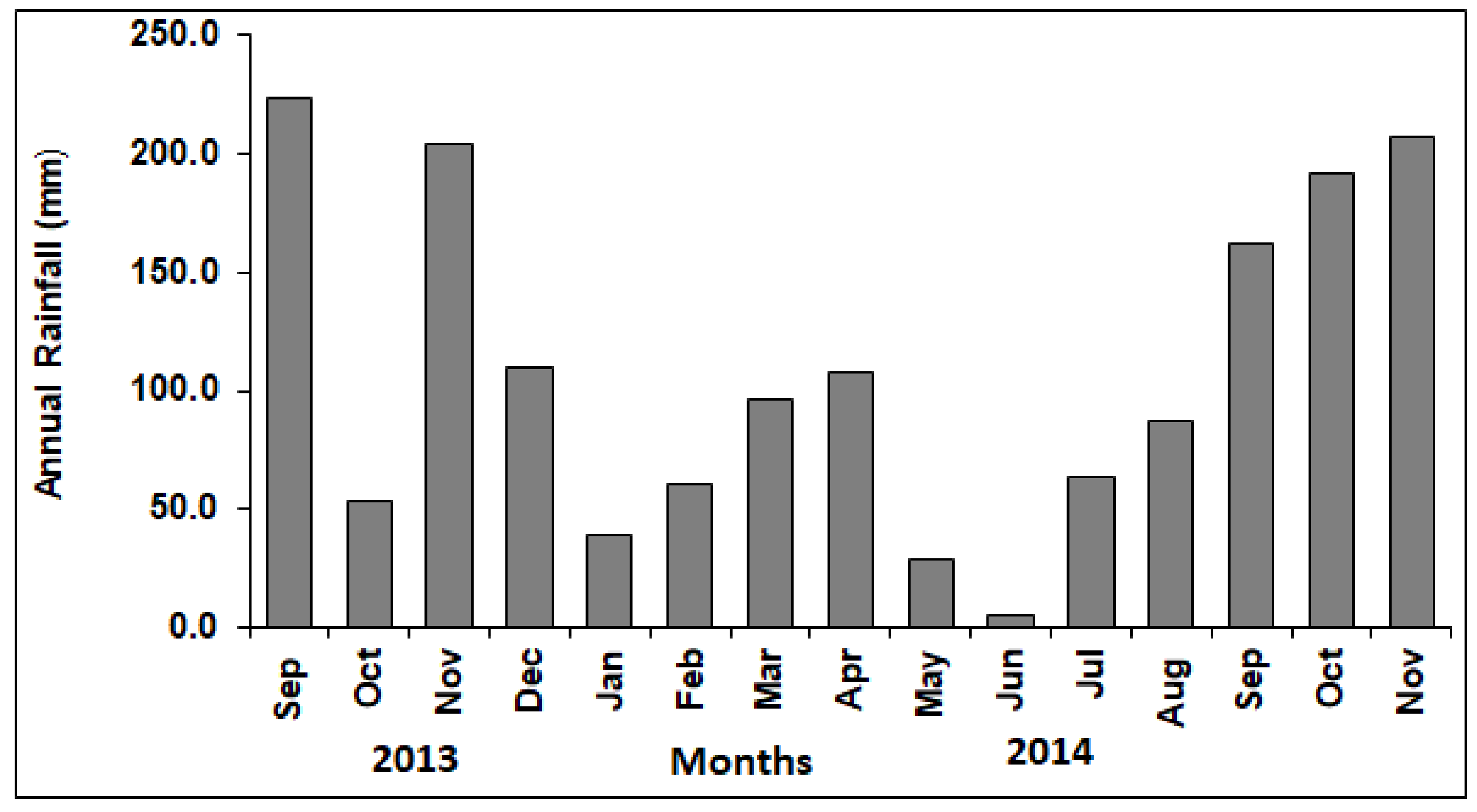
3.1. Rainfall Conditions
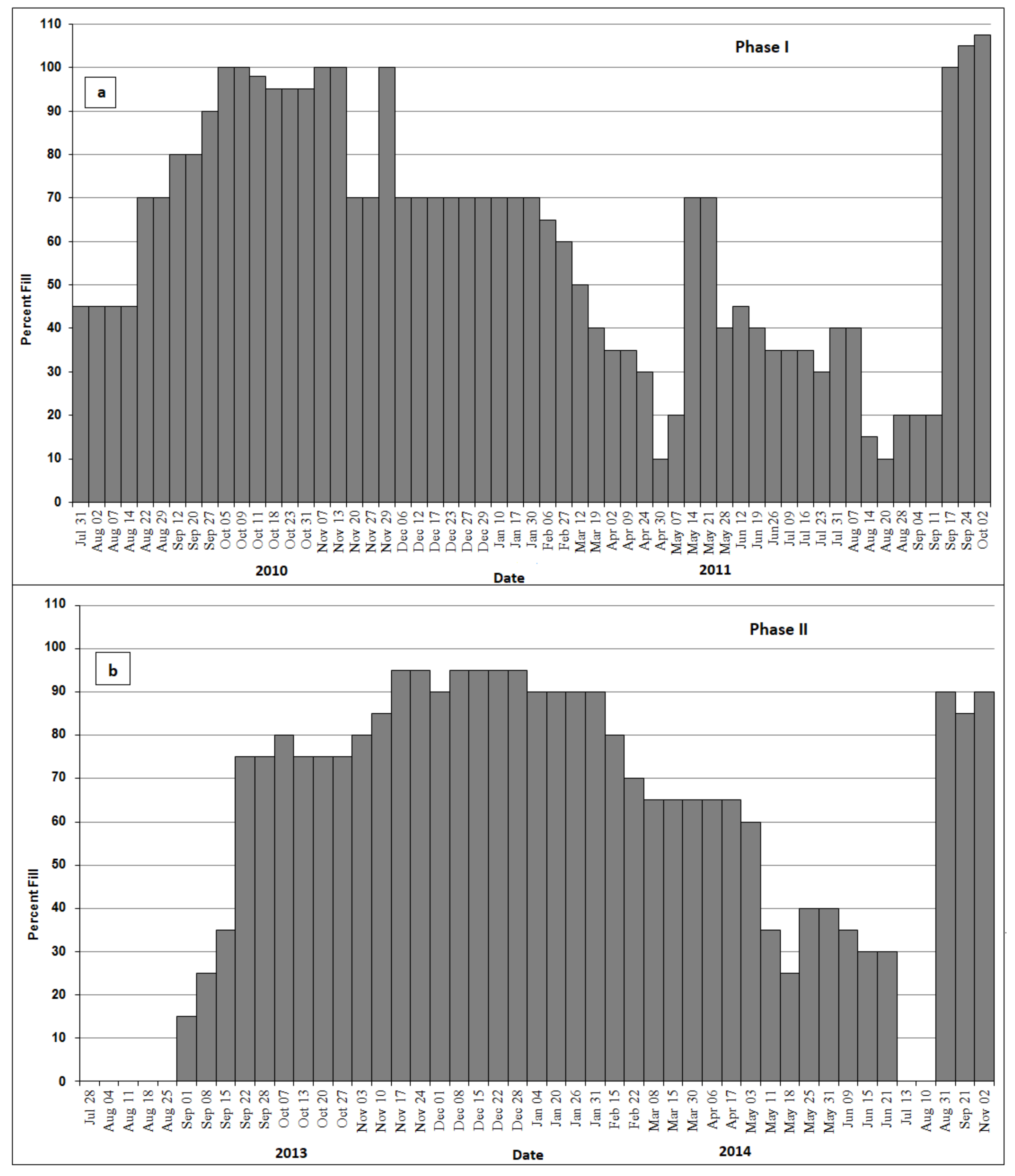
3.2. Stage Fluctuations
3.3. Dissolved Oxygen, pH, Total Dissolved Solids, Temperature, Turbidity, and Nitrogen
3.4. Analysis from Consolidated Bottom Substrate and Flocculence Associated with Floating Islands
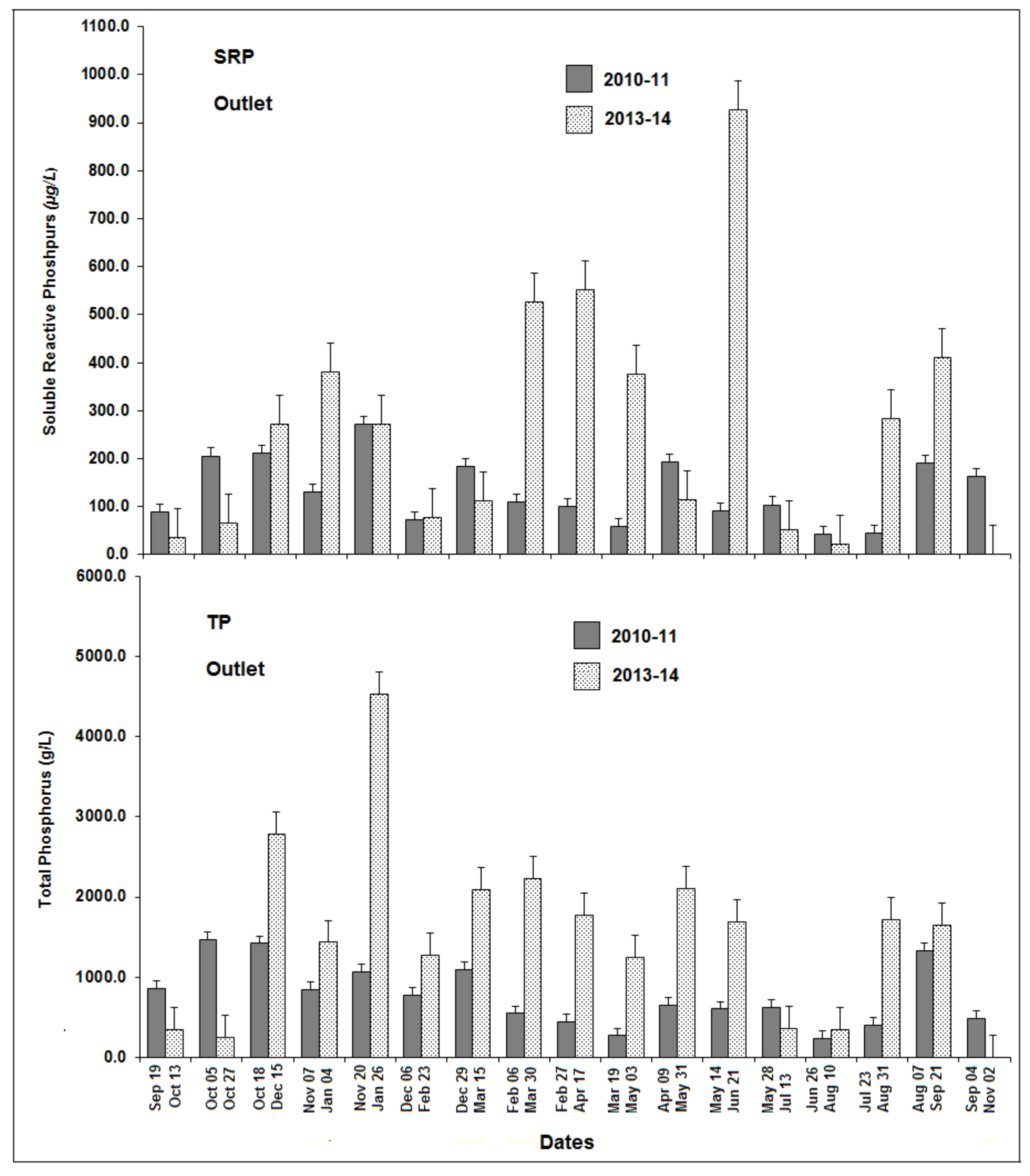
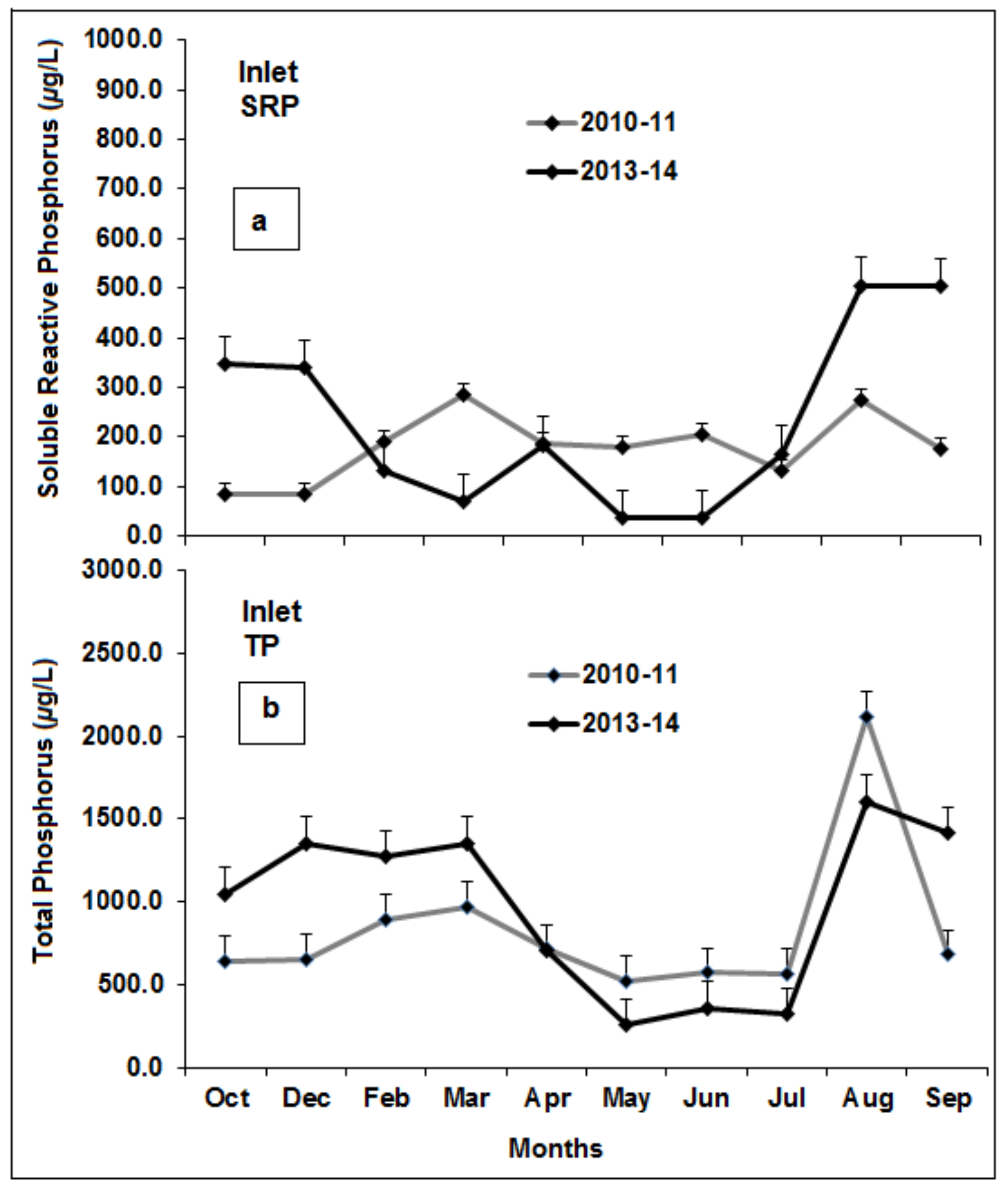
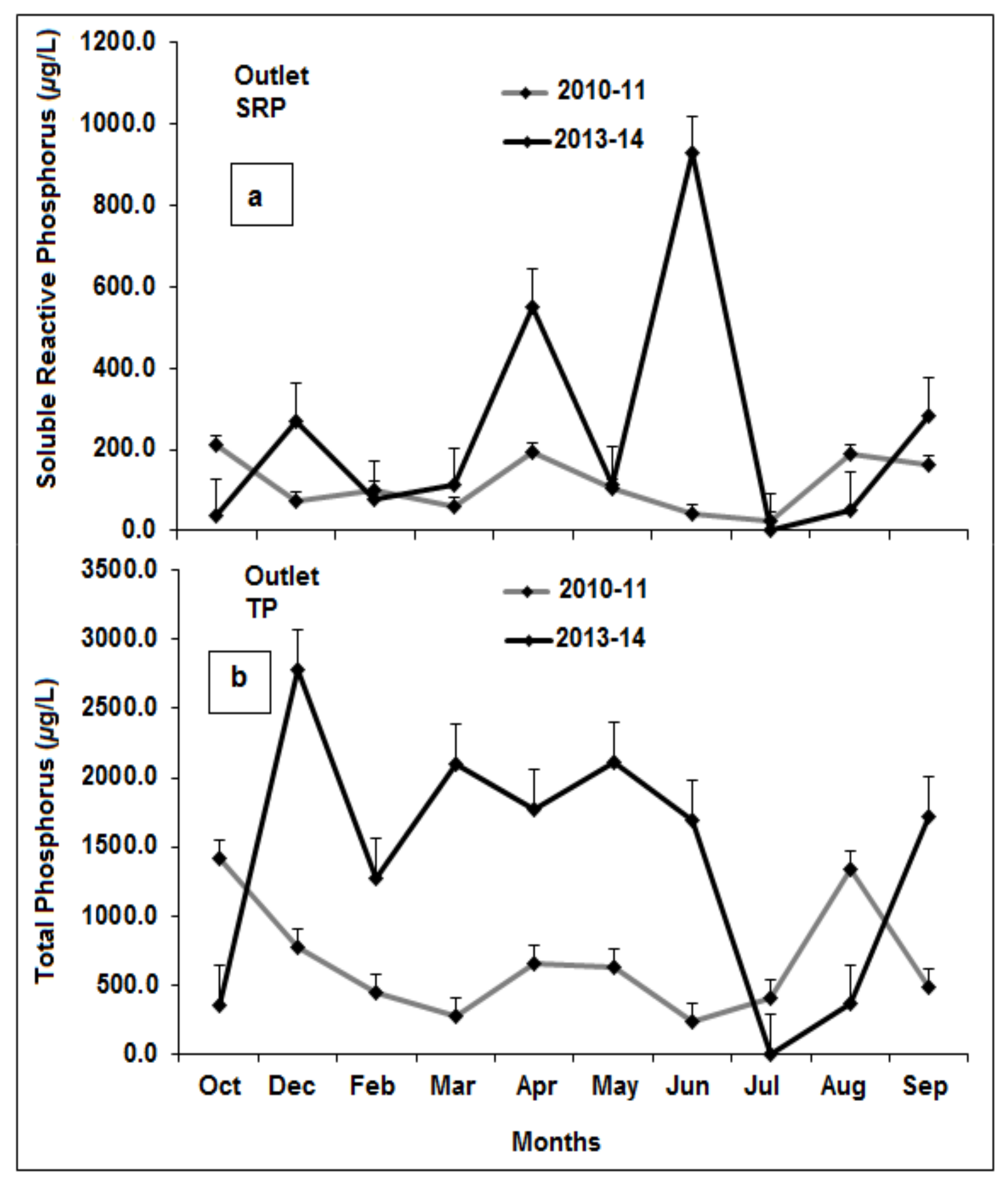
3.5. Phosphorus Source/Sink Dynamics
3.6. Source/Sink Dynamics under High Versus Low Rainfall and Water Level Fluctuations
3.7. Comparison of Phase One and Phase Two P Concentrations
4. Discussion
4.1. Phosphorus Inputs and Outlets Before and After Intervention Measures
4.2. Role of Precipitation and Water Level Changes in P Sink and Source Dynamics
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mesa, L.; Mayora, G.; Saigo, M.; Giri, F. Nutrient Dynamics in wetlands of the Middle Paraná River subjected to rotational cattle management. Wetlands 2015, 35, 1117–1125. [Google Scholar] [CrossRef]
- Mesa, L.M.; Maldini, C.; Mayora, G.; Saigo, M.; Marchese, M.; Giri, F. Decomposition of cattle manure and colonization by macroinvertebrates in sediment of the Middle Paraná River. J. Soils Sediments 2016, 16, 2319–2325. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Gosselink, J.G. Wetland, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 477–501. [Google Scholar]
- Baustian, J.J.; Kowalski, K.P.; Czayka, A. Using turbidity measurements to estimate total phosphorus and sediment flux in a Great Lakes coastal wetland. Wetlands 2018, 38, 1059–1065. [Google Scholar] [CrossRef]
- González-Rivas, E.J.; Roldán-Pérez, G.; Tundisi, J.G.; Vammen, K.; Örmeci, B.; Forde, M. Eutrophication: A growing problem in the America and the Caribbean. Braz. J. Biol. 2020, 80, 388–389. [Google Scholar]
- Spring, D.A.; Croft, L.; Bond, N.R.; Cunningham, S.C.; Mac Nally, R.; Kompas, T. Institutional impediments to conservation of freshwater dependent ecosystems. Sci. Total Environ. 2018, 621, 407–416. [Google Scholar] [CrossRef]
- Paredes-Gutiérrez, M.; Torres-Velásquez, C.B.; Sánchez-Colón, Y.M.; Schaffner-Gibbs, F.C. Two mathematical approaches to study the phosphorus eutrophication of a wetland in Puerto Rico. Inge Cuc 2019, 15, 63–76. [Google Scholar] [CrossRef]
- Tong, Y.; Zhang, W.; Wang, X.; Couture, R.M.; Larssen, T.; Zhao, Y.; Li, J.; Liang, H.; Liu, X.; Bu, X.; et al. Decline in Chinese lake phosphorus concentration accompanied by shift in sources since 2006. Nat. Geosci. 2017, 10, 507–511. [Google Scholar] [CrossRef]
- De Jesús Crespo, R.; Lázaro, P.M.; Yee, S.H. Linking wetland ecosystem services to vector-borne disease: Dengue fever in the San Juan Bay Estuary, Puerto Rico. Wetlands 2018, 39, 1281–1293. [Google Scholar] [CrossRef]
- Jarvie, H.P.; Johnson, L.T.; Sharpley, A.N.; Smith, D.R.; Baker, D.B.; Bruuselma, T.W.; Confesor, R. Increased soluble phosphorus loads to Lake Erie: Unintended consequences of conservation practices? J. Environ. Qual. 2017, 46, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Vollenweider, R.A. Advances in defining critical loading levels for phosphorus in lake eutrophication. Mem. Inst. Ital. Idrobiol. 1976, 33, 53–83. [Google Scholar]
- Vollenweider, R.A. Scientific Fundamentals of the Lutrophication of Lakes and Flowing Waters, with Particular Reference to Phosphorus and Nitrogen as Factors in Eutrophication; Report No. DAS/CSI/68.27; Organization for Economic Cooperation and Development (OECD): Paris, France, 1968. [Google Scholar]
- Vollenweider, R.A. Input-output models, with special reference to the phosphorus loading concept in limnology. Schweiz. Z. Hydrol. 1975, 37, 53–84. [Google Scholar]
- Ngatia, L.; Taylor, R. Phosphorus Eutrophication and Mitigation Strategies. In Phosphorus—Recovery and Recycling; Zhang, T., Ed.; IntechOpen: London, UK, 2019; ISBN 9781838810221. [Google Scholar]
- Schindler, D.W. The dilemma of controlling cultural eutrophication of lakes. Proc. R. Soc. B Biol. Sci. 2012, 279, 4322–4333. [Google Scholar] [CrossRef] [PubMed]
- Smolders, A.J.P.; Lamers, L.P.M.; Lucassen, E.C.H.E.T.; Van Der Velde, G.; Roelofs, J.G.M. Internal eutrophication: How it works and what to do about it—A review. Chem. Ecol. 2006, 22, 93–111. [Google Scholar] [CrossRef]
- Song, K.; Burgin, A.J. Eutrophication amplifies biological control on internal phosphorus loading in agricultural reservoirs. Ecosystems 2017, 20, 1483–1493. [Google Scholar] [CrossRef]
- Won Lee, H.; Seok Lee, Y.; Kim, J.; Jae Lim, K.; Hyun Choi, J. Contribution of internal nutrients loading on the water quality of a reservoir. Water 2019, 11, 1409. [Google Scholar]
- Kinsman-Costello, L. Effects of Water Level Fluctuations on Phosphorus, Iron, Sulfur, and Nitrogen Cycling in Shallow Freshwater Ecosystems. Ph.D. Thesis, Michigan State University, East Lansing, MI, USA, 2012. [Google Scholar]
- Kinsman-Costello, L.; O’Brien, J.M.; Hamilton, S.K. Natural stressor in uncontaminated sediments of shallow freshwaters: The prevalence of sulfide, ammonia, and reduced iron. Environ. Toxicol. Chem. 2015, 34, 467–479. [Google Scholar] [CrossRef]
- U.S. Fish and Wildlife Service. Laguna Cartagena Wildlife Refuge: Comprehensive Conservation Plan and Environment Assessment; USFWS: Washington, DC, USA, 2011. [Google Scholar]
- Schaffner, F.C. Accelerated terrestrialization of a subtropical lagoon: The role of agency mismanagement. In Proceedings of the 32nd Annual Conference on Ecosystems Creation and Restoration, Tampa, FL, USA, 27–28 October 2005. [Google Scholar]
- Sánchez-Colón, Y.M. Identifying nonpoint sources of phosphorus (P) and nitrogen (N) pollution and dynamics, internal eutrophication and anoxia variability at a tropical freshwater wetland (Laguna Cartagena, Puerto Rico). Ph.D. Thesis, Universidad del Turabo, Gurabo, PR, USA, 2015. [Google Scholar]
- Sánchez-Colón, Y.M. Effect of water level fluctuations and rainfall on phosphorus release and binding at a tropical freshwater wetland (Laguna Cartagena, PR). Master’s Thesis, Universidad del Turabo, Gurabo, PR, USA, 2012. [Google Scholar]
- Sánchez-Colón, Y.M.; Schaffner, F.C. The dynamics of total and soluble reactive phosphorus in a seasonal eutrophic, tropical freshwater. Ambientis 2017, 2, 2–7. [Google Scholar]
- APHA American Public Health Association; American Water Works Association; Water Environment Federation. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; Joint Editorial Board: Baltimore, MD, USA, 2017; p. 1546. [Google Scholar]
- Bostic, E.M.; White, J.R. Soil phosphorus and vegetation influence on wetland phosphorus release after simulated drought. Soil Sci. Soc. Am. J. 2007, 71, 238–244. [Google Scholar] [CrossRef]
- Pant, H.K.; Reddy, K.R. Hydrologic influence on stability of organic phosphorus in wetland detritus. J. Environ. Qual. 2001, 30, 368–674. [Google Scholar] [CrossRef]
- Kinsman-Costello, L.; O’Brien, J.; Hamilton, S.K. Re-flooding a historically drained wetland leads to rapid sediment phosphorus release. Ecosystems 2014, 17, 641–656. [Google Scholar] [CrossRef]
- Correll, D.L. Phosphorus: A rate limiting nutrient in surface waters. Poult. Sci. J. 1999, 78, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R.; Fisher, M.M.; Wang, Y.; White, J.R.; James, R.T. Potential effects of sediment dredging on internal phosphorus loading in a shallow, subtropical lake. Lake Reserv. Manag. 2007, 23, 27–38. [Google Scholar] [CrossRef]
- Banaszuk, P.; Wysocka-Czubaszek, A.; Kamocki, A.K. Internal eutrophication of restored peatland stream: The role of bed sediments. Ecol. Eng. 2011, 37, 260–268. [Google Scholar] [CrossRef]
- Bartoszek, L.; Tomaszek, J.A. Phosphorus distribution in the bottom sediments of the Solina-Myczkowce Reservoirs. Environ. Prot. Eng. 2007, 33, 25–32. [Google Scholar]
- Dieter, D.; Herzog, C.; Hupfer, M. Effects of drying on phosphorus uptake in re-flooded lake sediments. Environ. Sci. Pollut. Res. 2015, 22, 17065–17081. [Google Scholar] [CrossRef]
- Jing, L.; Liu, X.; Bai, S.; Wu, C.; Ao, H.; Liu, J. Effects of sediment dredging on internal phosphorus: A comparative field study focused on iron and phosphorus forms in sediments. Ecol. Eng. 2015, 82, 267–271. [Google Scholar] [CrossRef]
- Yu, J.; Ding, S.; Zhong, J.; Fan, C.; Chen, Q.; Yin, H.; Zhang, L.; Zhang, Y. Evaluations of simulated dredging to control internal phosphorus release from sediments: Focused on phosphorus transfer and resupply across the sediment—Water interface. Sci. Total Environ. 2017, 592, 662–673. [Google Scholar] [CrossRef]
- Chang, M.; Cui, J.; Lin, J.; Ding, S.; Gong, M.; Ren, M.; Tsang, D.C.W. Successful control of internal phosphorus loading after sediment dredging for 6 years: A field assessment using high-resolution sampling techniques. Sci. Total Environ. 2018, 616, 927–936. [Google Scholar]
- White, J.R.; Gardner, L.M.; Sees, M.; Corstanje, R. The short term effects of prescribed burning on biomass removal and the release of nitrogen and phosphorus in a treatment wetland. J. Environ. Qual. 2008, 37, 2386–2391. [Google Scholar] [CrossRef]
- Havens, K.E.; James, R.T.; East, T.L.; Smith, V.H. N:P ratios, light limitation, and cyanobacterial dominance in a subtropical lake impacted by non-point source nutrient pollution. Environ. Pollut. 2003, 122, 379–390. [Google Scholar] [CrossRef]
- Li, X.; Guo, M.; Duan, X.; Zhao, J.; Hua, Y.; Zhou, Y.; Liu, G.; Dionysiou, D.D. Distribution of organic phosphorus species in sediment profiles of shallow lakes and its effect on photo-release of phosphate during sediment resuspension. Environ. Int. 2019, 130, 104916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Cheng, P.; Zhong, B.; Wand, D. Total phosphorus release from bottom sediments in flowing water. J. Hydrodyn. Ser. B 2012, 24, 589–594. [Google Scholar] [CrossRef]
- Xiao, Y.; Chang, H.; Yu, W.; Ll, Z. Effects of water flow on the uptake of phosphorus by sediments: An experimental investigation. J. Hydrodyn. Ser. B 2016, 28, 329–332. [Google Scholar] [CrossRef]
- Smith, R.L.; Smith, T.M. Ecología, 6th ed.; Pearson Education: Madrid, Spain, 2007; pp. 704–776. [Google Scholar]
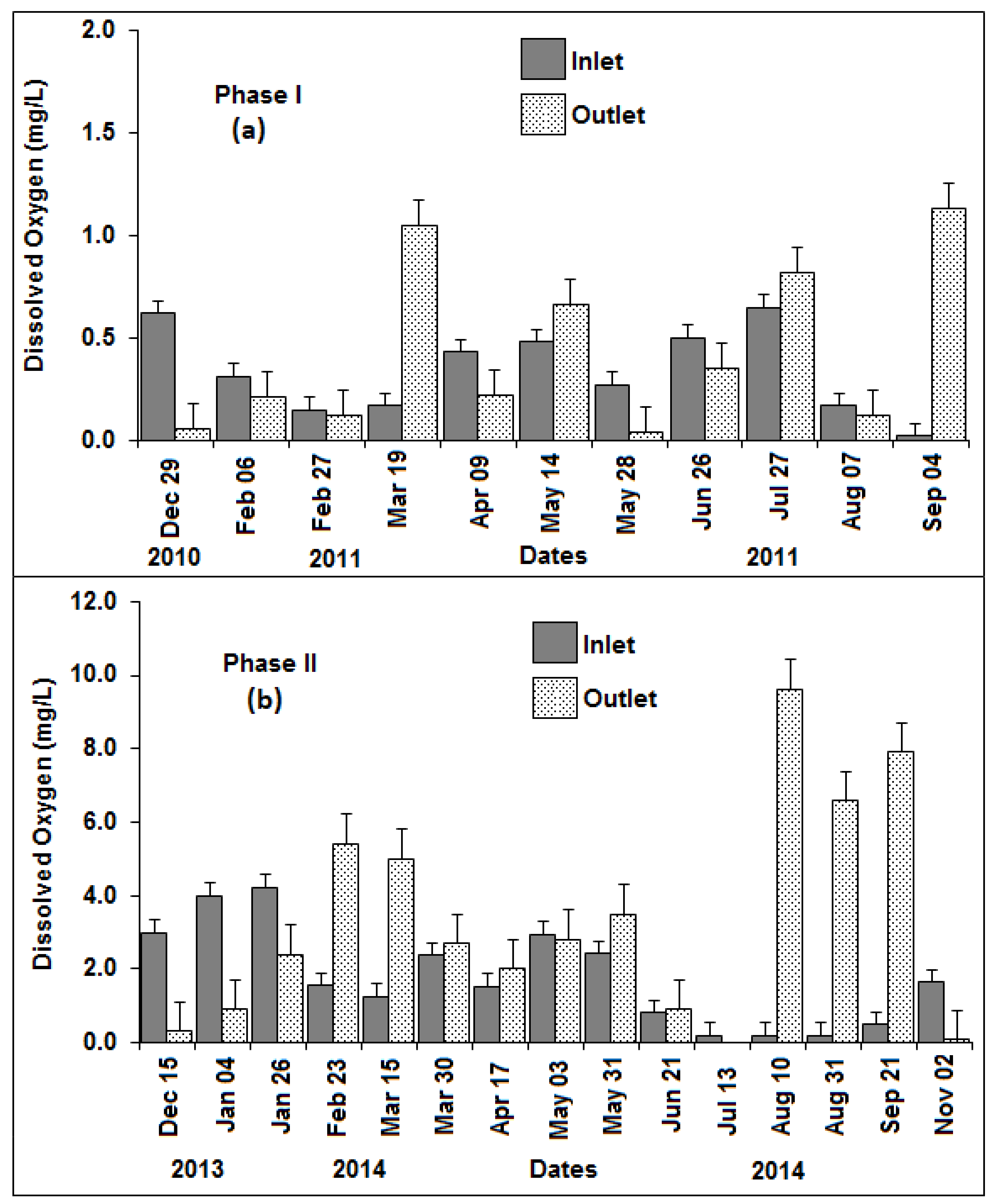
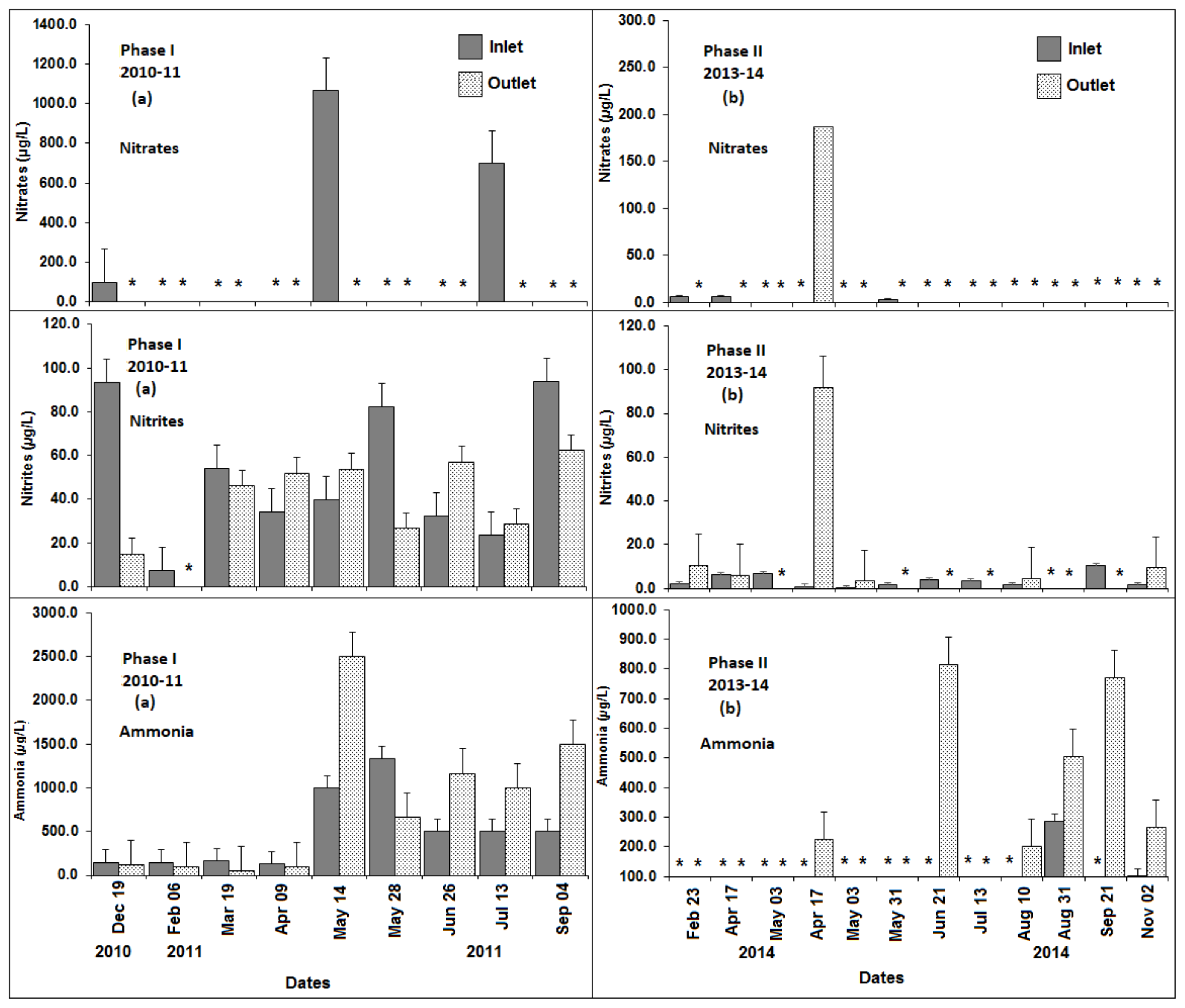
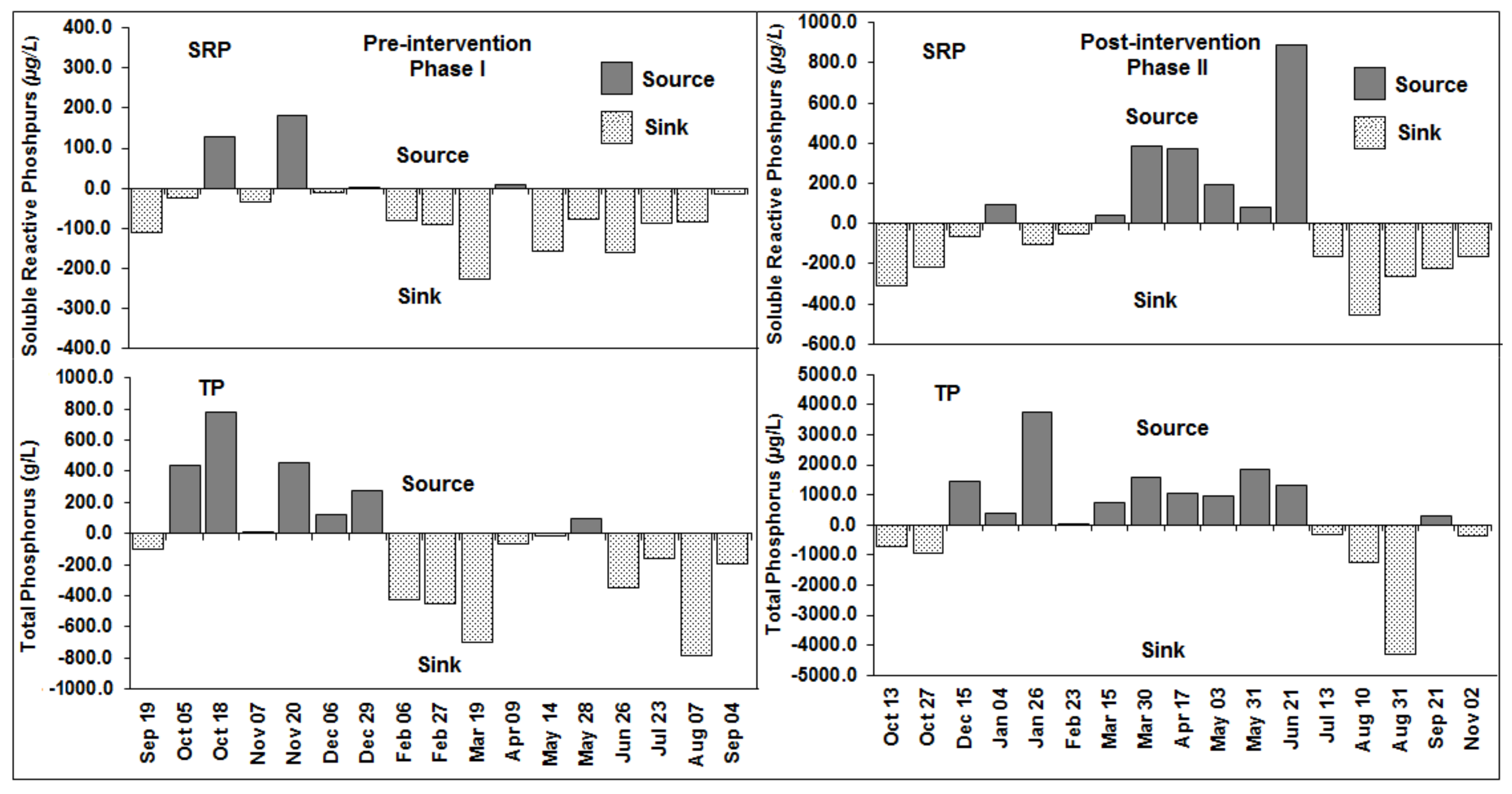
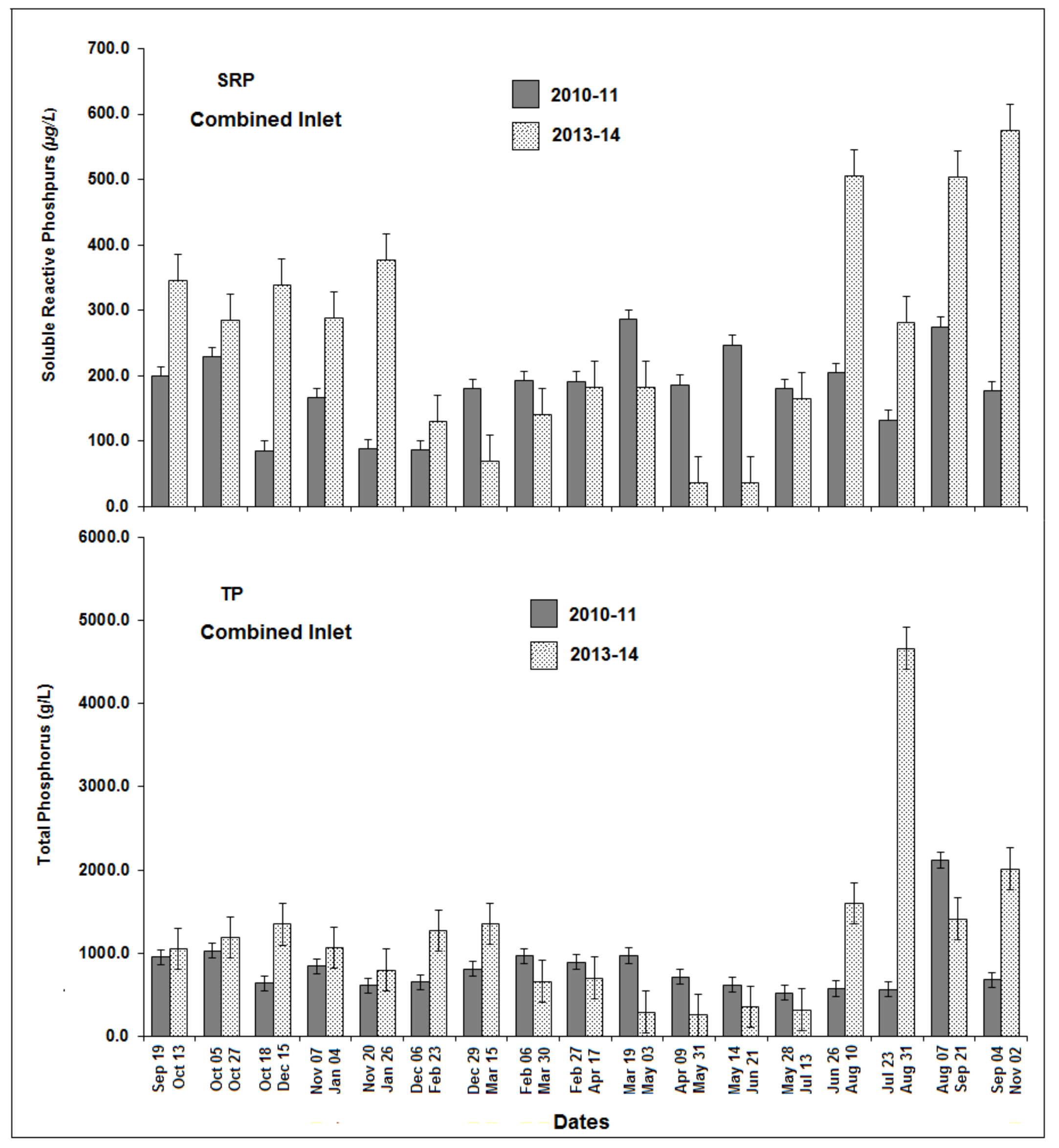
| Location | DO (mg/L) | pH | TDS (mg/L) | Temp (°C) | Turbidity (cm) * |
|---|---|---|---|---|---|
| Lagoon Inlet | |||||
| 2010–2011 | 0.34 ± 0.20 | 7.1 ± 0.6 | 325 ± 157 | 25.5 ± 2.1 | 49.0 ± 25 |
| 2013–2014 | 1.80 ± 1.4 | 7.2 ± 0.3 | 255 ± 65 | 26.5 ± 1.9 | 68.9 ± 27 |
| Lagoon Outlet | |||||
| 2010–2011 | 0.43 ± 0.40 | 6.9 ± 0.5 | 243 ± 93 | 27.2 ± 3.0 | 33.6 ± 13 |
| 2013–2014 | 3.59 ± 2.9 | 7.0 ± 1.2 | 459 ± 203 | 27.6 ± 2.7 | 40.0 ± 19 |
| Location | pH | %TKP | Pavailable (µg/L) | %TKN | NH3 (µg/L) | (µg/L) | (µg/L) | |
|---|---|---|---|---|---|---|---|---|
| Consolidate bottom substrate | ||||||||
| Center cattails | 2010–11 | 4.16 | 0.09 | 7750 | 0.39 | 220,000 | 34,000 | ND* |
| 2013–14 | 3.86 | 0.17 | 35,000 | 0.54 | 80,000 | ND* | ND* | |
| Flocculence associated with floating islands | ||||||||
| Center cattails | 2010–11 | 2.82 | 0.21 | 50,720 | 0.70 | 63,000 | 1000 | 1000 |
| 2013–14 | 4.30 | 0.08 | 13,000 | 0.98 | 163, 000 | ND* | ND* | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Colón, Y.M.; Schaffner, F.C. A Case Study of the Effects of Management Interventions on the Phosphorus Dynamics at a Coastal, Eutrophic, Caribbean lagoon (Laguna Cartagena, Puerto Rico). Water 2021, 13, 449. https://doi.org/10.3390/w13040449
Sánchez Colón YM, Schaffner FC. A Case Study of the Effects of Management Interventions on the Phosphorus Dynamics at a Coastal, Eutrophic, Caribbean lagoon (Laguna Cartagena, Puerto Rico). Water. 2021; 13(4):449. https://doi.org/10.3390/w13040449
Chicago/Turabian StyleSánchez Colón, Yashira Marie, and Fred Charles Schaffner. 2021. "A Case Study of the Effects of Management Interventions on the Phosphorus Dynamics at a Coastal, Eutrophic, Caribbean lagoon (Laguna Cartagena, Puerto Rico)" Water 13, no. 4: 449. https://doi.org/10.3390/w13040449
APA StyleSánchez Colón, Y. M., & Schaffner, F. C. (2021). A Case Study of the Effects of Management Interventions on the Phosphorus Dynamics at a Coastal, Eutrophic, Caribbean lagoon (Laguna Cartagena, Puerto Rico). Water, 13(4), 449. https://doi.org/10.3390/w13040449






