Abstract
Wetland treatment systems are used extensively across the world to mitigate surface runoff. While wetland treatment for nitrogen mitigation has been comprehensively reviewed, the implications of common-use pesticides and antibiotics on nitrogen reduction remain relatively unreviewed. Therefore, this review seeks to comprehensively assess the removal of commonly used pesticides and antibiotics and their implications for nitrogen removal in wetland treatment systems receiving non-point source runoff from urban and agricultural landscapes. A total of 181 primary studies were identified spanning 37 countries. Most of the reviewed publications studied pesticides (n = 153) entering wetlands systems, while antibiotics (n = 29) had fewer publications. Even fewer publications reviewed the impact of influent mixtures on nitrogen removal processes in wetlands (n = 16). Removal efficiencies for antibiotics (35–100%), pesticides (−619–100%), and nitrate-nitrogen (−113–100%) varied widely across the studies, with pesticides and antibiotics impacting microbial communities, the presence and type of vegetation, timing, and hydrology in wetland ecosystems. However, implications for the nitrogen cycle were dependent on the specific emerging contaminant present. A significant knowledge gap remains in how wetland treatment systems are used to treat non-point source mixtures that contain nutrients, pesticides, and antibiotics, resulting in an unknown regarding nitrogen removal efficiency as runoff contaminant mixtures evolve.
1. Introduction
Water quality degradation in rivers and streams across the globe is becoming a major concern, especially as stresses from climate change increase [1]. The leading cause for surface water impairments in the United States is due to non-point source pollution [2]. Non-point sources include urban, agricultural, and construction runoff [3], which often contain nutrients, suspended sediment, pesticides, antibiotics, and other pharmaceuticals, fecal coliform, and metals [3,4,5,6]. Nutrient contamination, such as nitrate-N (NO3-N) from fertilizer applications, is persistent in rivers and is the most common form globally of chemical contamination in groundwater, resulting in NO3-N being a major cause of drinking water impairment across the United States and the globe [7,8,9,10,11]. In addition to nutrients, chemically derived common-use pesticide (CUPs) applications throughout the world have increased by 71% from 1990 to 2017, with 11% used for non-agricultural application in the United States [12,13]. The United States Geological Survey (USGS) recently reported that urban pesticides (e.g., 4-Hydroxychlorothalonil, azoxystrobin, carbendazim, propiconazole, 2,4-D, diuron, prometon, triclopyr, fipronil, imidacloprid) accounted for 83% of the pesticides found in urban sites where the mixture complexity and potential toxicity of the pesticides increased with increasing urbanization [14]. Additionally, most antibiotics used in human and veterinary medicine (e.g., macrolides, sulfonamides, trimethoprim, quinolones) are excreted via urine and feces. Antibiotics used by humans often enter wastewater treatment systems where removal efficiencies ranged from 15.8–78.4% from 2010 to 2019 [15]. Veterinary antibiotic residues in livestock products result in 24,000,000–72,000,000 kg of antibiotic residue being introduced into agricultural soil and downstream aquatic environments from manure applications annually [16]. While CUPs and antibiotics are important for agricultural productivity to support an ever-increasing global population, these chemicals have become ubiquitous in waterways worldwide, resulting in significant effects on agroecosystem food webs (e.g., honeybee colony collapse) [17] and human health (e.g., reproductive and development disruption, carcinogens, antibiotic-resistant genes) [18,19].
Wetland treatment systems, including natural and constructed wetlands, are now being used extensively across the United States and the world to treat non-point source pollution. This is due to their cost-effectiveness and low energy consumption compared to other surface water treatment methods (e.g., coagulation, membrane filtration, ion exchange) [20,21,22]. Wetlands utilize plants, soils, and associated microbial assemblages to remove pollutants through biodegradation, substrate adsorption, and plant uptake [23,24]. These wetland treatment processes take place in different biogeochemical compartments of the wetland (e.g., aqueous, sediment, detritus), which have separate roles as either sinks or sources [25]. However, there are limitations to wetland treatment systems including the requirement of large areas and the high variability in performance, as design and operation can be challenging because of the complexity of environmental processes, changing influent concentrations, seasonal changes in weather, and system clogging [22].
The use of wetlands as a treatment approach for NO3-N is well known [11,26,27,28,29]; however, N removal performance is significantly impacted by wetland design, operation, and internal and external environmental factors (e.g., microbial communities, plants, type and concentration of contaminants) [30]. Specifically, contaminants of emerging concerns (CECs), which include CUPs and veterinary antibiotics, in surface waters have only recently started to be investigated for their impact on ecologically important processes (e.g., NO2-N oxidizing bacteria population, NH4+-N oxidizing bacteria population) [31,32]. Exposure to specific CUPs (e.g., imidacloprid) in runoff waters and exposure to antibiotic residues contained in livestock manure and wastewater (e.g., tetracycline, lincomycin, chlortetracycline, sulfamethazine) may influence the population structure of denitrifying bacteria communities [33,34,35,36], and thus, the activity of microbial denitrification in adjacent wetland treatment systems.
Therefore, this review sought to comprehensively assess past reports related to CUPs and antibiotic treatment in wetlands along with the implications of runoff mixtures for the nitrogen (N) cycle and specifically NO3-N removal in wetland treatment systems receiving runoff from urban and agricultural landscapes. The objectives of this review were to: 1. Evaluate the current scientific status of the topic, 2. Quantify the scale and type of wetlands used to treat runoff mixtures, 3. Identify the source and mixture of antibiotics and CUPs reported to be entering wetlands along with removal mechanisms, and 4. Review implications to N removal processes (e.g., denitrification, plant uptake) in the presence of antibiotics and/or CUPs.
2. Materials and Methods
This review was designed to better understand the current state of knowledge on the implications of runoff mixtures to the N cycle in wetland treatment systems (specifically the removal of NO3-N). The review was completed by identifying the methodology and major results about nutrient (N) and CEC (pesticides and antibiotics) removal efficiencies along with implications to the N cycle. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) standard was used for analyzing and reporting [37]. Herein, studies used in this review are referred to as primary studies.
2.1. Eligibility Criteria
This review sought to include all searchable studies that have researched the ability of wetlands to treat runoff from urban and agricultural landscapes. The primary studies eligible for inclusion in this review needed to meet the following criteria: (1) the study performed analysis on the ability of wetlands to treat CUPs and/or antibiotics from non-point source pollution; (2) the study was from a peer-reviewed journal.
2.2. Identification of Records
The primary studies were obtained from searching Web of Science and SCOPUS databases. A search string was determined through an iterative process to identify studies using any of the search terms in the title, abstract, and/or keywords. The final search string used was the following: (treatment wetland AND antibiotic OR treatment wetland AND pesticide OR constructed wetland AND antibiotic OR constructed wetland AND pesticide). The search was further limited to full-text peer-reviewed journal articles available in English. The official search ended in July 2021 and resulted in 1224 studies meeting the criteria.
Limitations of the search included evaluating only English studies, inevitably underrepresenting the sample of collected studies by excluding those that may have met all other search criteria but were not available in English. Additionally, the keywords used were important to define papers retrieved in the database search; however, it was inevitable that the keywords did not entirely encompass all relevant papers even with careful consideration and several iterations of keywords. These are inherent limitations to all reviews; thus, even though the search was performed systematically, the primary studies may still be biased. However, the limitations do not necessarily take away from the importance and applicability of the findings.
2.3. Screening and Coding of Studies Based upon Eligibility Criteria
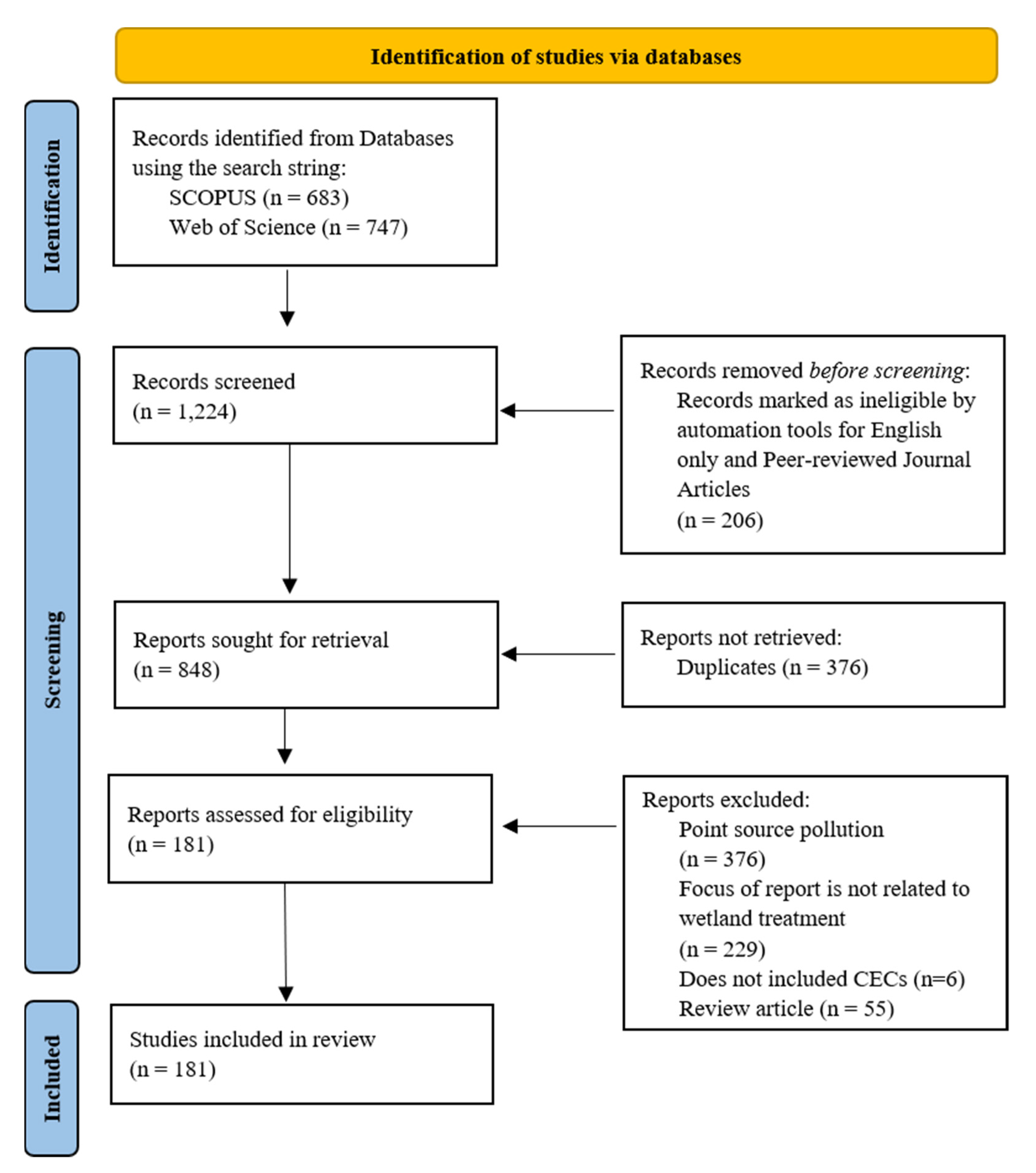
After the initial database search, a three-phase process was used to screen primary studies that met all eligibility criteria (Figure 1) [38]. Using Rayann [39], one coder independently read the title and abstract for all the studies obtained from the initial database search after duplicates were removed. One coder was used throughout the process to maintain consistency and reduce bias; however, if there was any uncertainty regarding the inclusion of a primary study, a second coder was sought for a final decision.
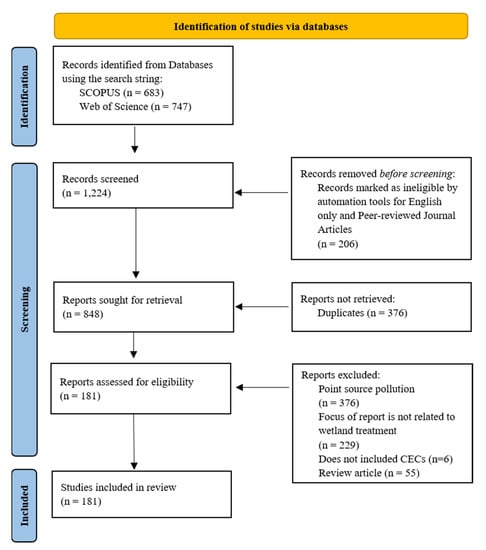
Figure 1.
PRISMA flow diagram detailing the review process (adapted from [38]).
Each study was evaluated to ensure it pertained to wetland treatment of non-point sources. For example, Zheng et al. (2021) was removed from further analysis because the authors simulated domestic sewage waste (point source pollution) to study the removal efficiency of sulfamethoxazole and nitrogen in wetland mesocosms [40]. Additionally, Maryniuk et al. (2016) considered how high-contaminant loads in wetlands impact largemouth bass [41], whereas Sen et al. (2019) focused on the impact of crows that roosted in constructed wetlands and their ability to be carriers of E. coli [42]. Neither of these studies quantified or identified wetland contaminant removal efficiencies or N cycle pathways, so these studies were removed from further analysis. Studies were also removed if only nutrient removal was analyzed without the presence of pesticides or antibiotics. For example, Hussain et al. (2011) analyzed N removal in the presence of antibiotics; thus, the study was retained for further analysis even though the antibiotic removal potential was not specified [43]; however, Jia et al. (2021) investigated N removal and poly-3-hydroxybutyrate-co-3hydroxyvalerate/polylactic acid removal in the absence of any pesticides or antibiotics, so this study was removed from further analysis [44]. Review articles were also excluded from the analysis, given that new research on the topic was not conducted; rather, a summary of the existing research was reviewed and/or meta-analysis was completed. From this, there were 181 primary studies used for further analysis [3,16,23,25,30,32,43,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181,182,183,184,185,186,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207,208,209,210,211,212,213,214,215,216,217,218].
The final step of the screening process was to code the primary studies using a common coding schema (Table 1). Key information was coded, including the publication year, location of the study, the scale of the wetland system, number and type of contaminant(s) of concern, removal mechanisms studied, source of runoff, and removal efficiencies. Concerns on correct coding were resolved by the second coder, which included defining wetland systems (e.g., excluding vegetated ditches and riparian buffers), non-point source identification, and identifying removal mechanisms. Throughout the screening and coding process, coders met weekly to check in on progress and to resolve any issues that arose.

Table 1.
Coding schema used for each primary study.
3. Results
Following the screening, 181 primary studies were identified, with 153 on CUP removal, 29 on antibiotic removal, and 31 on a combination of contaminants. Primary study publication dates ranged from 1995 to 2021, with a linear increase in primary studies on the topics over the last 15 years. The primary studies were drawn from 37 countries.
3.1. Bibliometric Source Overview of the Primary Studies
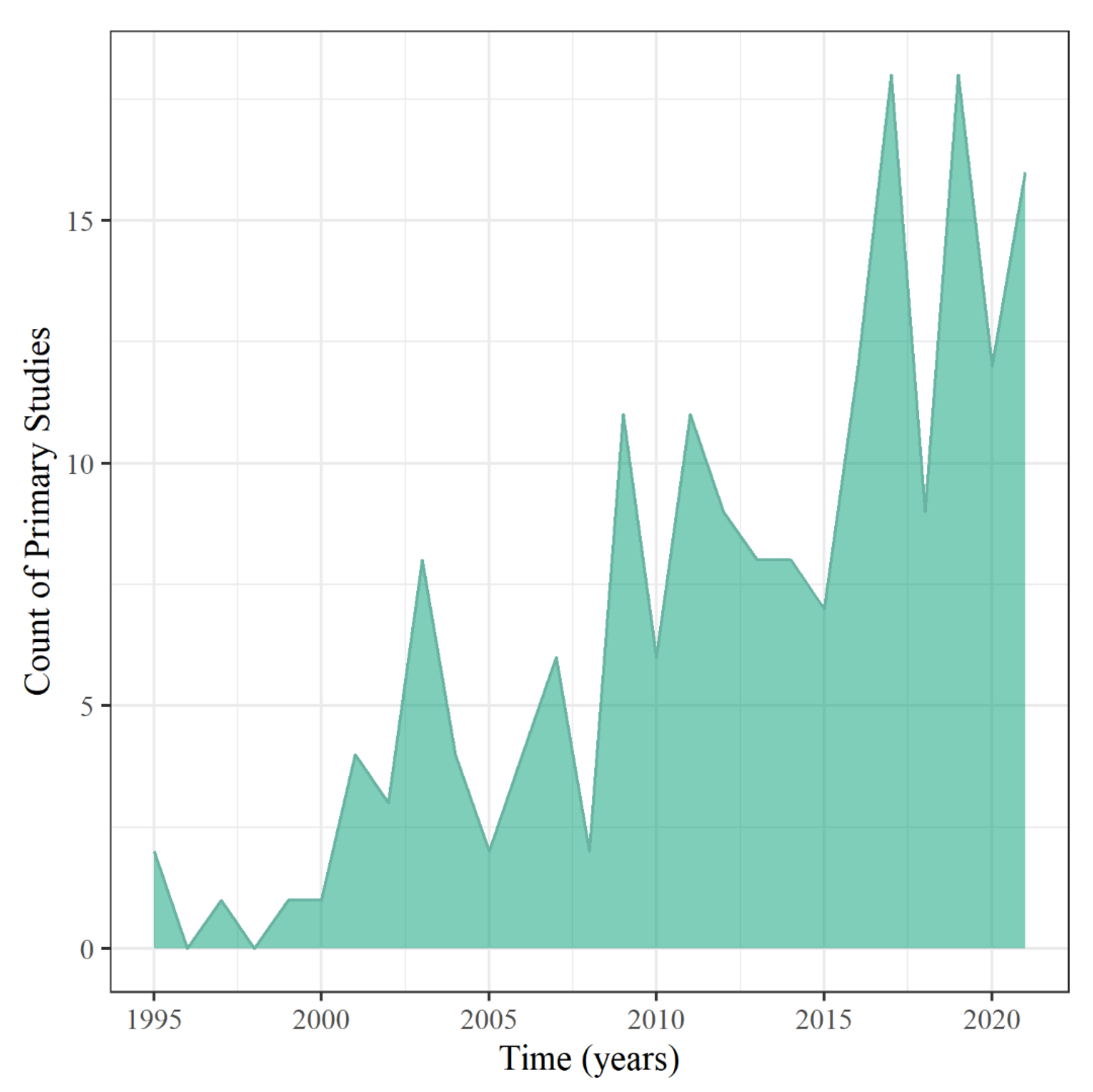
Publication information, such as geography, was analyzed to provide insight into the annual scientific production growth of research about wetland treatment systems of runoff mixtures to assess Objective 1. Annual scientific production growth on the topic was approximately 29%, showing that this research area is growing, with the peak year of production being 2019 (Figure 2). The decline in publications in 2020 and 2021 could be due to delayed publications from the COVID-19 pandemic. However, 2021 was close behind with 16 publications, even without a complete representative sample of the year, since the studies were collected up to July of 2021. These results demonstrate that research about CUP and antibiotic treatment from wetland systems is growing as more of these contaminants are introduced into the environment and being detected in downstream best management practices and waterways.
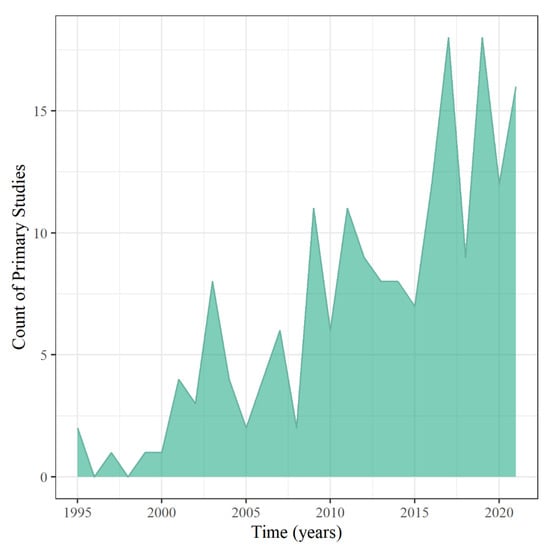
Figure 2.
Annual scientific production of primary studies.
The publication sources for the primary studies were indicative of the interdisciplinary nature, broad application, and relevance of the topic, dependent on region. The top publication sources for the primary studies included Chemosphere (n = 25), Science of the Total Environment (n = 16), and Ecological Engineering (n = 16; Table 2). Table 2 only reports the top five sources. However, 61 total sources were identified. These findings indicate that wetland treatment systems treating non-point source runoff mixtures have broad applicability to a variety of journal sources, which ranged from chemistry, environmental hazards and contaminations, engineering, biology, and ecology-focused journals.

Table 2.
Results by publication source for the top 5 sources of the primary studies.
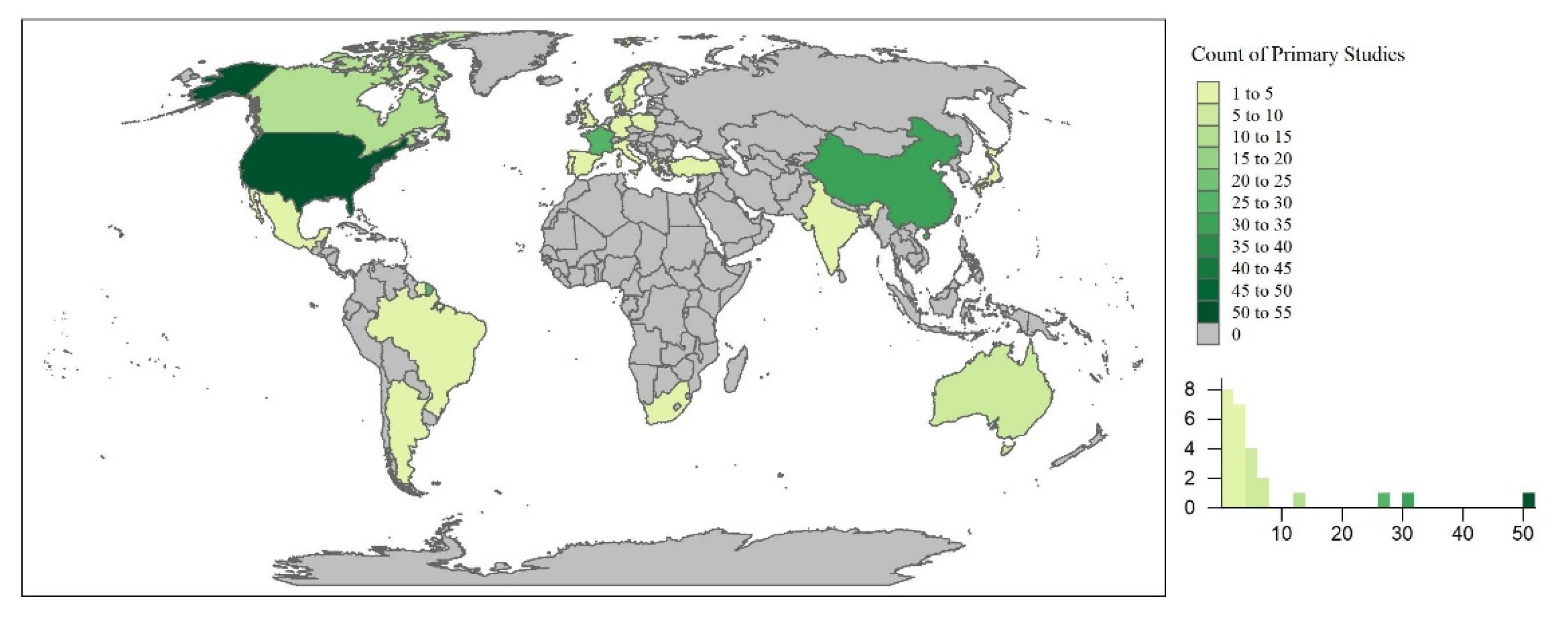
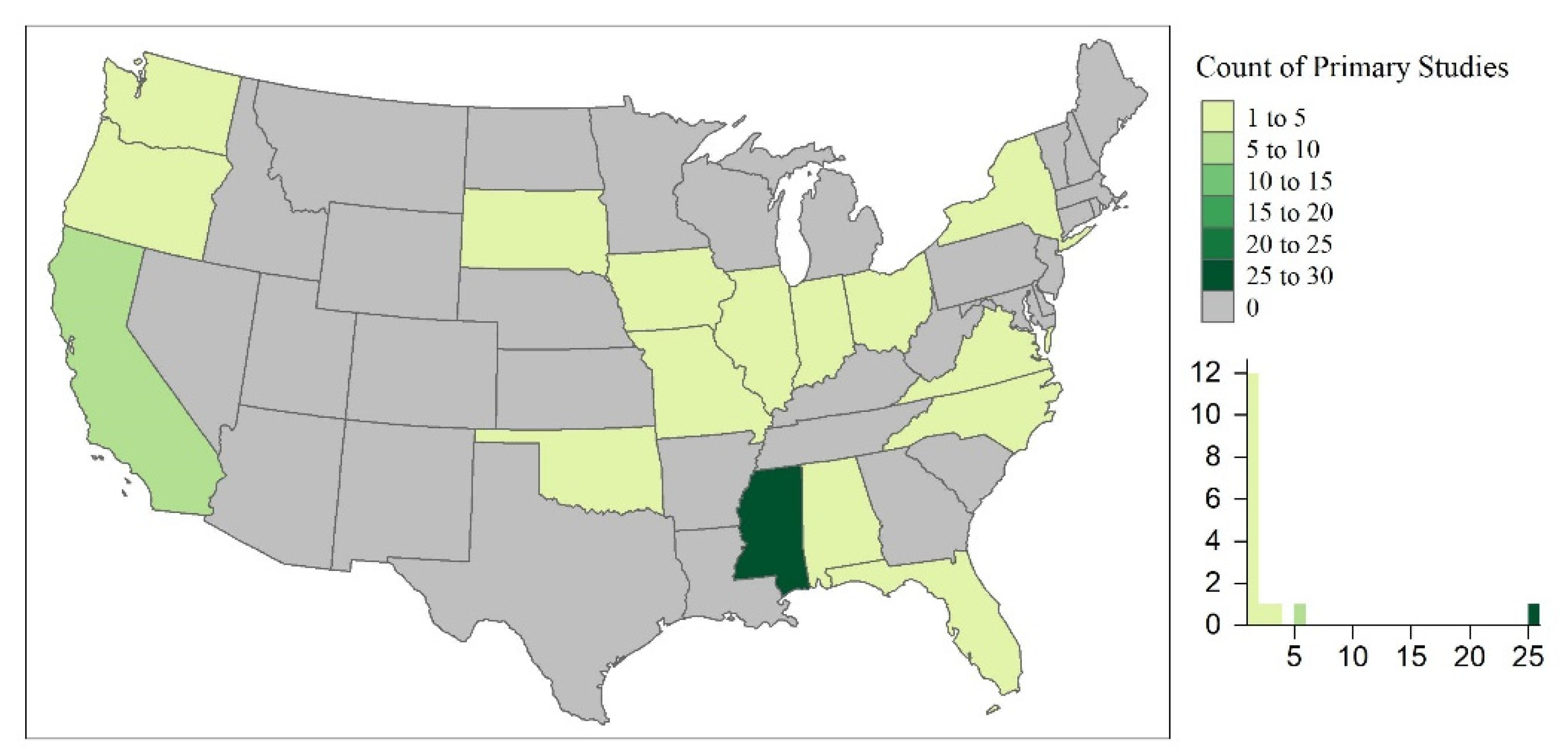
The primary studies took place throughout the world, occurring in 37 countries. Countries with the highest number of primary studies included the United States (U.S.; n = 52), China (n = 30), and France (n = 26; Figure 3). This could undoubtedly be due to the limitation of the review to English journals. However, this could also be indicative of water quality concerns and policies limiting the use of emerging contaminants in different regions of the world. For example, the U.S. has approved the use of pesticides and antibiotics (e.g., phorate, dicrotophos, tribufos, oxytetracycline, streptomycin) for outdoor agricultural use, of which many have been banned or phased out by the European Union, Brazil, and China [219]. In the U.S., the Mississippi River Basin is the largest, most intensively farmed region with phorate, dictophos, tirubfos, and oxytetracycline primarily applied to land in the Southeastern region of the United States [220]. This is represented in Figure 4, where the state of Mississippi was the location of 48% of the primary studies in the U.S. Furthermore, these studies were further analyzed based upon experimental design and implications to wetland treatment processes in the sections below.
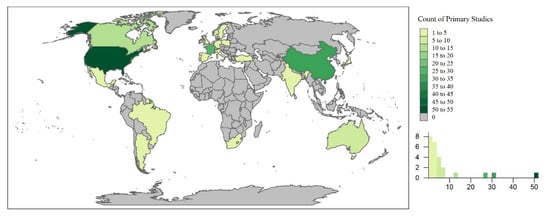
Figure 3.
The spatial distribution of the primary studies across the world.
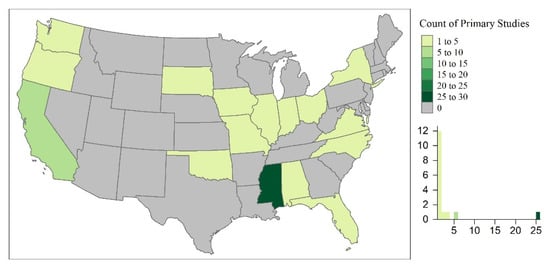
Figure 4.
The spatial distribution of the primary studies within the continental United States (U.S.).
3.2. Scale and Type of Wetlands Used
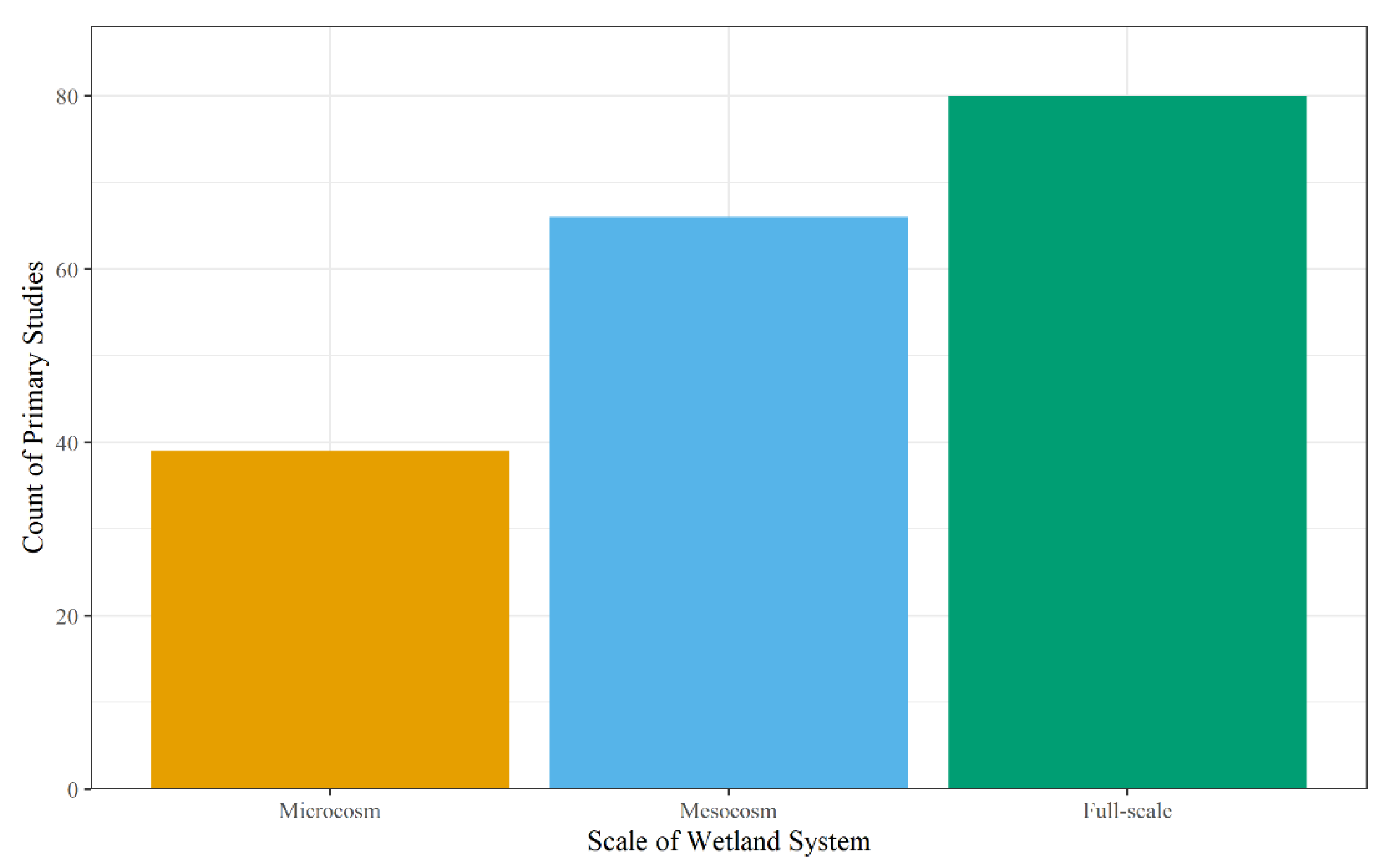
The scale and type of wetland system used for analysis within the primary studies were coded to determine the scope of the studies along with their impact on the study results to assess Objective 2. The scale has the potential to impact removal efficiencies of contaminants by increasing the number of variables in the system, moving from a controlled environment to a natural environment, and introducing artificial impacts (i.e., wall-effects in microcosm and mesocosm experiments) as studies progress from microcosm to full-scale [221,222]. Microcosm studies utilize repeatable small-scale systems to understand a specific biogeochemical process (e.g., sorption, phytoremediation, microbial activity) in a controlled environment [222,223]. On the other hand, full-scale systems include major processes in the natural environment; however, these studies are harder to replicate due to land and cost constraints [221]. Mesocosm studies are a more cost-effective tool to study contaminant removal in wetlands compared to a full-scale system, while still providing useful predictions using treatment replicates and controls to study wetland efficacy in controlled and natural environments [221,224]. As for the primary studies, 43% studied full-scale systems, 37% studied mesocosms, and 21% studied microcosms (Figure 5), with only two studies investigating more than one scale [53,76].
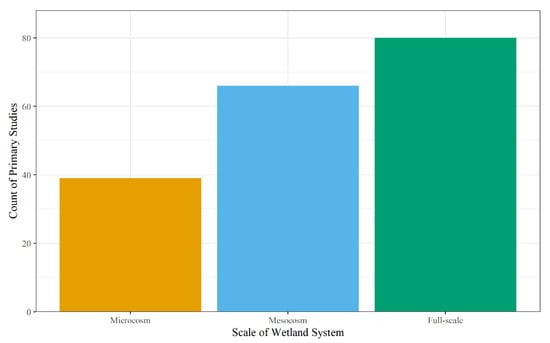
Figure 5.
Count of the scale used in the primary studies for the wetland system.
The primary studies that showed high removal efficiencies (greater than 75%) were larger for mesocosm studies. Of the studies that analyzed the mesocosm scale, 68% had at least one contaminant removed at an efficiency greater than or equal to 75% [16,23,45,48,53,54,55,58,59,67,69,70,76,79,86,102,104,105,112,113,114,124,144,151,154,161,163,169,170,171,175,178,179,180,182,183,185,192,193,195,201,202,203,209,210,217,218,219]. Additionally, 54% of microcosm studies resulted in high removal efficiencies [30,60,76,78,83,90,94,97,119,129,131,143,146,164,168,177,181,188,194,200,207] and 59% of field studies resulted in high removal efficiencies [3,46,50,53,62,63,68,74,77,80,82,84,85,87,92,95,99,103,106,107,117,119,121,122,126,128,133,135,139,141,142,147,150,156,159,160,173,174,184,186,187,189,191,196,206,208,212]. However, efficiencies between studies for specific contaminants and/or classes were not able to undergo statistical evaluation due to few studies having the same contaminant and/or class evaluated.
The type of wetland system was another principal factor to consider because the type of system impacts removal efficiencies, dependent on the contaminant of interest [225]. Wetlands are often defined based upon hydrology (e.g., free-water surface and subsurface flow) along with vegetation (e.g., emergent, submerged, floating, and free-floating) [226]. In this review, 19 different wetland systems were identified throughout the primary studies. These ranged from natural systems to different types of constructed systems including agricultural field practices (e.g., rice fields and wetland buffers). Natural wetlands were defined as wetlands that existed naturally in the environment. These included, but were not limited to, floodplains, prairie potholes, depressions, salt marshes, and ephemeral wetlands. Constructed wetland systems included horizontal flow wetlands, subsurface flow wetlands, man-made reservoirs (e.g., ponds, lakes, lagoons, dugouts, and storage dams), free-water surface wetlands, vertical flow wetlands, wetlands in a series, floating treatment wetlands, stormwater basins, and recirculating wetlands. Wetlands used as an agricultural field practice included rice fields, which acted as temporary wetlands with some of the same species as temporary ponds [227]. Additionally, wetland buffers were defined as a wetland system constructed near agricultural fields to remove contaminants in the water before entering receiving waterbodies.
The most common wetland systems identified were horizontal subsurface flow constructed wetlands (n = 42; Table 3). However, while horizontal subsurface flow constructed wetlands were the most common wetland system reviewed, removal efficiencies for CUPs and antibiotics varied widely from 0% norlurazon removal at the field-scale [61] to 100% imidacloprid removal at the mesocosm scale [210]. Additionally, natural wetland systems and reservoirs were analyzed in 15% and 14% of the primary studies, respectively. Both wetlands were used to treat a variety of contaminants (e.g., atrazine, metolachlor, chlorpyrifos, clothianidin, endosulfan, permethrin, prosulfocarb, fluometuron, isoproturon) in full-scale systems. Removal efficiencies ranged from 10% (clothianidin) [187] to 100% (permethrin) [77] for natural wetlands and 0% (prosulfocarb) [128] to 100% (isoproturon) [126] for reservoirs. The least studied systems were depressions, ephemeral wetlands, wetland buffers, recirculating constructed wetlands, and salt marshes each appearing only once throughout the primary studies. However, six studies investigated the ability of rice fields to mitigate nutrients, pesticides, and antibiotics as a wetland system in Mississippi, U.S. [63,160,174], India [56], China [115], and Spain [103] with high removal efficiencies (58–100%) for several different contaminants (e.g., diazinon, benthocarb, carbofuran, atrazine, permethrin, NH4+-N, NO3-N, nitrate-N).

Table 3.
Results by wetland type analyzed in the primary studies.
3.3. Source and Mixture of Contaminant Types Entering Wetlands
Specific design approaches used in the primary studies were analyzed to identify study length (e.g., days, years), type of water analyzed (e.g., urban runoff, rural runoff), and contaminants evaluated to address Objective 3. This was completed to determine which methodological approaches were used most often throughout the primary studies, along with attempting to understand the impact of seasonality, type of landscape runoff, contaminant type, the presence of mixtures of contaminants, and wetland plant type in the primary study results. The length of the study was important to identify the impact of time on wetland treatment. Water type provided insight into runoff from urban and agricultural landscapes and into which landscapes have been studied the most. The specific contaminants studied and the presence of mixtures were coded to determine the most common contaminants and how the contaminant (CUPs and/or antibiotics) impacted NO3-N removal efficiencies. Finally, the type of wetland plants recorded in the primary studies assisted with identifying which wetland plants were most commonly used and if there was an impact on the removal efficiencies of different contaminants based on planting plan.
The length of the study was coded in five different categories: hours, days, weeks, months, and years (Table 4). Hours were defined as any study that took place less than 24 h. Only one study fell into this category, which evaluated removal efficiencies of permethrin in a mesocosm study with four different wetland plant species (i.e., Leersia oryzoides, Typha latifolia, Sparaganium americanum, Thalia dealbata) over 12 h [54]. The second category, days, was defined as an experiment that lasted between 1 to 7 days, which included 13% of the primary studies. The category of weeks was then defined as an experiment that was between 7 to 30 days, with months defined as an experiment that took place between 30 to 365 days, and years being an experiment that was longer than 365 days.

Table 4.
Results by the length of the study analyzed by the primary studies.
Months were the most common length of study (53%), and weeks were the second most commonly evaluated (26%). Most of the microcosm studies (67%) addressed periods shorter than months, while a majority of the mesocosm studies (64%) used longer periods. Additionally, several contaminants (e.g., methyl-parathion, malathion, endosulfan, chlorpyrifos, diazinon, tetracycline) were removed within a relatively short period (i.e., less than 10 days) [144,168,171,174,177]. However, other studies found accumulation of contaminants (e.g., pyrethroid, 2,4-MCPA, glyphosate, propoxycarbazone-Na, NO3-N) [68,114,126] with recommendations that the long-term impact on wetlands needs to be further investigated.
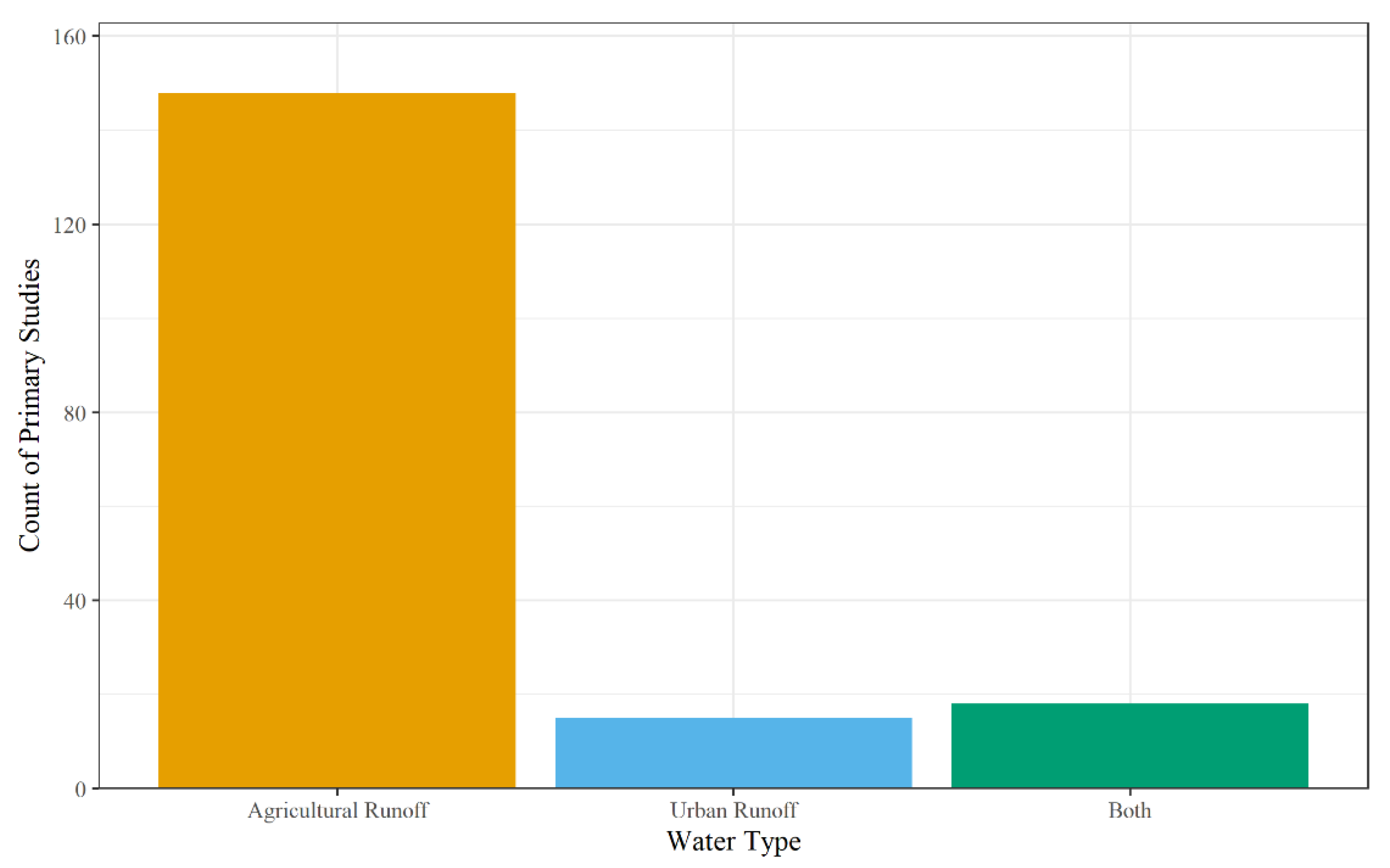
The runoff from landscapes defined the water type and included agricultural runoff, urban runoff, both agriculture and urban runoff, or nature reserve. This was coded based on land cover in the watershed for field-scale systems. For mesocosm and microcosm experiments, the primary study either stated where the water was collected from, or the type and concentration of contaminants used for synthetic water. For example, Birch et al. (2004) studied a wetland located in an urban watershed in Sydney, Australia to determine the removal efficiencies of organochlorine pesticides, polycyclic aromatic hydrocarbons, trace metals, nitrogen, phosphorous, and stormwater effluent [213]. On the other hand, Butkovskyi et al. (2021) used synthetic wastewater to simulate agricultural runoff by applying tap water, fertilizer solution, and different types of pesticides (bentazone, MCPA, metalaxyl, propiconazole, pencycuron, imidacloprid) to potted microcosms containing Phalaris arundinacea sp. Larsa [181]. For the primary studies, the most common water type was from agricultural runoff (n = 148), with both agriculture and urban runoff encompassing 18 studies, and urban runoff alone assessed in 15 studies (Figure 6). Studies investigating urban and agricultural runoff were either not specific about the water type and instead reported contaminant(s) applied in upstream regions (e.g., sulfonamides used to treat both human and animal infections [76,90,170], or the field site for the primary studies’ watershed contained both urban and agricultural runoff [74,80]. Finally, nature reserve was the least studied landscape (n = 1); Tsui et al. (2008) assessed glyphosate concentrations in the Mia Po Nature Reserve in Hong Kong [87].
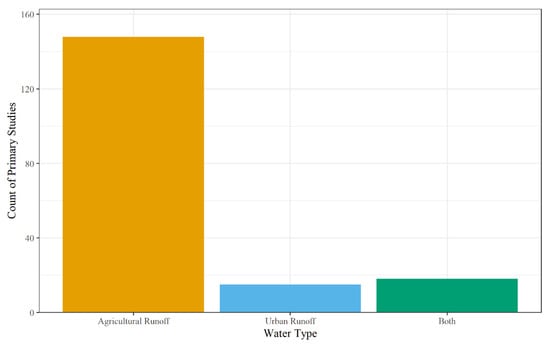
Figure 6.
Count of non-point source water used by primary studies.
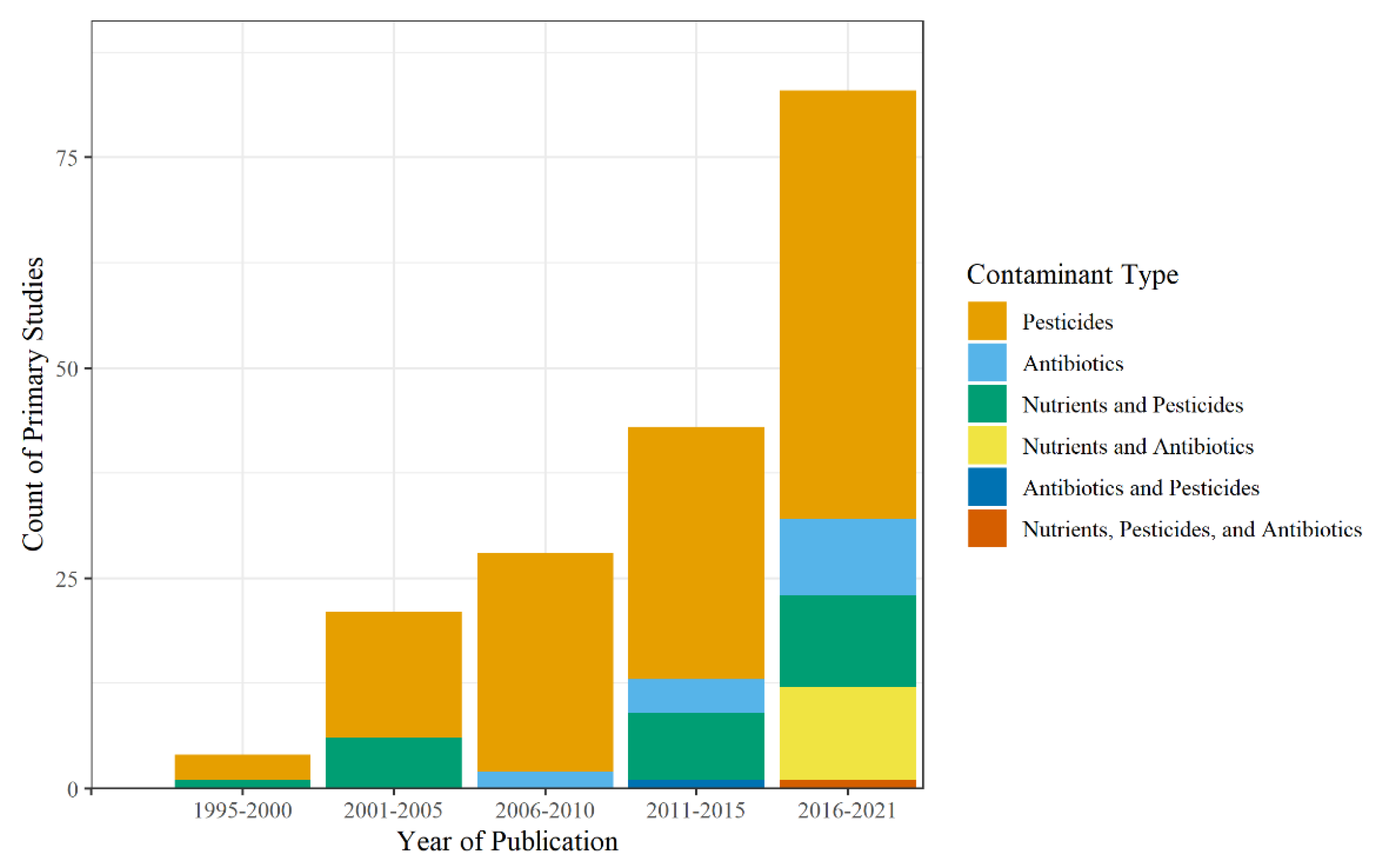
Seven different types of contaminants were studied: nutrients, pesticides, antibiotics, other pharmaceuticals besides antibiotics, metals, minerals, and industrial by-products (Table S1). Pesticides included herbicides, insecticides, and fungicides and were the most common type of contaminant analyzed, appearing 556 times throughout the primary studies with peak study counts occurring between 2016 and 2021 (Figure 7). The most common pesticides studied were atrazine (n = 25), chlorpyrifos (n = 22), s-metolachlor (n = 21), alachlor (n = 16), and isoproturon (n = 14), along with 158 other pesticides analyzed. Atrazine is a commonly used herbicide, introduced in 1985, and is mainly used for agricultural landscapes while also being used on residential lawns and golf courses, particularly in the Southeastern United States [228]. Additionally, chlorpyrifos, an insecticide introduced in 1965, and s-metolachlor, an herbicide introduced in 1976, are used for agricultural and non-agricultural landscapes, while alachlor and isoproturon are herbicides mainly used for agriculture.
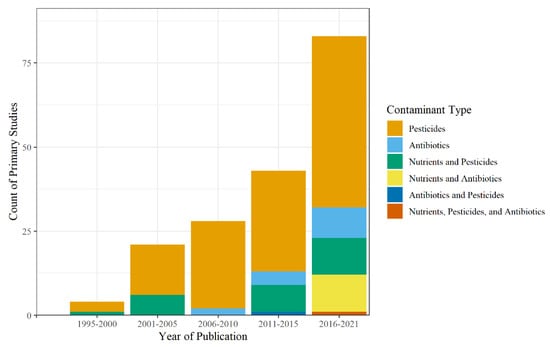
Figure 7.
Count of contaminant type and mixture by year.
Antibiotics appeared throughout the primary studies 60 times, starting in 2007, with peak counts of primary studies (nantibiotics = 9, nnutrients and antibiotics = 11, nnutrients, pesticides, and antibiotics = 1) between 2016 and 2021. The most common antibiotics studied were tetracycline (n = 11), sulfamethoxazole (n = 5), monensin (n = 3), narasin (n = 3), and ciprofloxacin (n = 5), out of a total of 29 primary studies. Of these, narasin and monensin are used as veterinary antibiotics and were found primarily in agricultural runoff, whereas sulfamethoxazole, tetracycline, and ciprofloxacin are used more for humans and were found in urban runoff.
Additional contaminants found in the primary studies included other pharmaceuticals, metals, minerals, and industrial byproducts. Other pharmaceuticals included any pharmaceutical that was not considered an antibiotic (e.g., carbamazepine, caffeine, diclofenac, fluoxetine, naproxen, ibuprofen). Industrial byproducts were defined as contaminants that are commonly used in industry such as fragrances (e.g., cashmeran, dihydrojasmonate), plastic production (e.g., bisphenol A, di-n-butyl phthalate), dyes (e.g., uranine), corrosion inhibitors (e.g., benzotriazole), and de-icing fluids (e.g., 5-methyl-1H-benzotriazole). While these contaminants were not the focus of the review, they were observed in the primary studies and are often found in runoff from urban and agricultural landscapes [80,99,101,103,112,137,151,213].
There was a total of 9 nutrients analyzed throughout the primary studies, with the most common being TN (n = 29), NO3-N (n = 15), NH4+-N (n = 12), total phosphorus (TP, n = 10), and nitrite-N (NO2-N; n = 4). Other nutrients found in the primary studies were orthophosphate, sulfate, chloride, and urea. As for the presence of mixtures, only 30 primary studies looked at contaminant mixtures. Nutrient and pesticide mixtures were the most common mixtures studied, peaking between 2016 and 2021 with 11 primary studies. Only one study in 2020 considered a runoff mixture containing antibiotics, nutrients, pesticides, and other pharmaceuticals [103]. Of these studies that looked at mixtures, only 16 specifically analyzed the impact on nitrogen removal in the presence of different contaminants [23,30,58,59,67,97,102,103,112,113,114,120,131,169,179,181,201]. Thus, the data shows that only recently have some of these contaminants started to be studied individually, in the case of antibiotics, but also as mixtures. This has resulted in the limitation of this review to systematically analyze the implications of wetland design parameters and mechanisms on N removal processes.
3.4. Removal Mechanisms and Efficiencies
Removal mechanisms were coded based upon the specific processes that were the focus of the primary study to further address Objective 3. Fifteen removal mechanisms were evaluated (Table 5). Varying mechanisms provide insight into the impact on contaminant removal efficiencies, particularly the impact on nitrogen removal processes. Design parameters included size, depth, and aspect ratio of the wetland in addition to the impact of timing (the removal efficiencies over time from contaminant exposure) and space (distance from wetland inlet). If a specific process was not listed and instead removal efficiency of contaminants was analyzed for the wetland system as a whole, then “holistic approach” was coded.

Table 5.
Results by removal mechanisms studied for impact on removal efficiencies of contaminants by primary studies.
For the primary studies, the most common removal mechanisms studied were biological processes such as phytoremediation (n = 90), sorption (n = 69), and microbial activity (n = 43). Phytoremediation was coded if the primary study looked at the impact of vegetated vs. non-vegetated systems [55,161,175], or analyzed the plant roots [23,119,127,180,210], leaves [127,210], and stems [119,210] for contaminant concentrations. The type of wetland plant used throughout the primary studies helps to identify which wetland plants were most common and if there was an impact on the removal efficiencies for different contaminants dependent on plant species. There were 92 genera of plants identified, with Typha spp. being the most common, comprising 14% of the primary studies, and Phragmites spp. used in 11% of the studies (Table 6). It is important to note that not all studies used wetland plants [43,83,89] or identified the specific plant species within the wetland system [148,159,179]. Also, not all primary studies that specified the plant species present in the wetland system analyzed the impact of phytoremediation as a removal mechanism [21,173,205]. For the studies that did investigate phytoremediation, vegetated systems increased removal efficiencies compared to non-vegetated systems [51,55,66,95,154,161,175,181,217], with mature plants out-performing younger ones [158].

Table 6.
Results of the top 10 plant genus used by the primary studies.
As for the impact of the type of plant on removal efficiencies, Lv et al. (2016) concluded that Typha latifolia, Phragmites australis, Iris pseudacorus, and Juncus effusus were all able to take up and metabolize imazalil and tebuconazole with removal efficiencies between 46–96% and 25–41%, respectively [78]. Additionally, Tang et al. (2019) concluded that there were no significant differences in planted systems (Cyperus alternifolius, Canna indica, Iris pseudacorus, Juncus effusus, and Typha orientalis) and that plants with high biomass and transpiration were able to accelerate the removal of chlorpyrifos and conventional pollutants with removal efficiencies between 94–98% [86]. On the other hand, some primary studies found that specific species outperformed others, with Lemna minor having high removal efficiencies for dimethomorph (17%) and pyrimethanil (12%) compared to Spirodela polyrhiza (11–15%) [111]. Additionally, Phalaris arundinacea was better at up taking dicamba, dimethoate, trifloxystrobin, metamitron, and tebuconazole (mean removal of 4%) compared to Typha latifolia (2%) [95], Eleocharis mutata retained less imidacloprid in the plant material and roots (0.5%) compared to Nymphaea amazonum (78.9%) [210], and Pontederia cordata reduced greater amounts of azoxystrobin (51.7%) compared to Juncus effusus (24.9%) and Silene latifolia (28.7%), while Silene latifolia was the best at removing imidacloprid (79.3%) [154].
Sorption to wetland media was a removal mechanism of focus for 18% of the primary studies (Table 5). This included studies that compared different media types [67,104,170] or analyzed the amount of contaminant sorbed to the wetland media by determining the concentrations of contaminants [74,87,135,170,229] or determining sorption isotherm coefficients for the media [137,196,198]. The different media types found in the primary studies included biochar [59,170,179,192], straw [133,134], compost [170], different types of soil [67,145,196,197], gravel [104,161,229], pebbles [179], zeolite [59,114,147,218], and cobbles [104].
Soil types and properties (e.g., organic matter content, porosity, structure, moisture content, electrical conductivity) are important components of wetland systems and have been found to impact microbial communities, N cycling, and vegetation growth [229]. However, these soil properties are dependent upon wetland type (natural vs. constructed wetlands) and age. For example, newly developed, constructed wetlands may have to overcome soil compaction, resulting in decreased porosity and redox potential, which in turn impacts N cycling [230]. Overall, the type of contaminants present has been reported to impact sorption processes due to competing cations [87] and differences between highly vs. weakly sorbing contaminants [54]. Additionally, sedimentation or sorption was not the primary removal mechanism in wetland systems with the presence of multiple removal mechanisms (e.g., increased hydraulic retention time, vegetation, microbial activity); instead, it enhanced contaminant removal [67,74,84,115,135,196,218]. For example, Uddin et al. (2019) found electrical conductivity, total organic carbon, and total nitrogen in the soil significantly impacted microbial richness and diversity [115].
Microbial activity is another important removal mechanism in wetland systems because microbial communities facilitate water treatment through metabolic actions (e.g., anabolism, catabolism) [231]. Microbial communities are mainly found in the rhizospheres, the biofilms around the media, and the water. For the primary studies, 11% focused on microbial activity as a specific removal mechanism. The increased microbial activity enhanced the removal capabilities for CUPS and antibiotics [59,60], with microbial degradation being a leading mechanism for removal [194]. However, the microbial communities were impacted by the substrate [59,194], the type and concentration of contaminants present [58,114,179], and the physicochemical properties of the water [179]. In particular, Lu et al. (2021) reported the presence of sulfamethoxazole improved interactions for denitrifying bacteria, but also decreased network complexity and microbial interaction on the whole molecular network, thus altering the community structure of nitrogen-transforming microorganisms [114]. Yuan et al. (2020) observed the addition of Mn ore impacted microbial diversity, causing increased removal potential for antibiotics (ciprofloxacin hydrochloride and sulfamethazine), TN, NH4+-N, and NO3-N [59].
3.5. The Impact of CUPs and Antibiotics on Nitrogen Removal
Overall, 31 primary studies analyzed and reported contaminant removal efficiencies for runoff mixtures containing nitrogen, pesticides, and/or antibiotics to address Objective 4 (Table S2). The removal efficiencies for each contaminant varied widely, with total nitrogen ranging from 5% to 99% removal and a mean of 58%. Ammonia-N had a similar range of 7% to 100% and a mean of 75% removal. Nitrate-N removal varied widely from −113% to 98%; however, only one primary study found an accumulation of NO3-N, which was attributed to the amount of zeolite used in the mesocosm cells [114]. As for the CUPs and antibiotics studied, the removal efficiencies varied from −619% to 100% and 35% to 100% for pesticides and antibiotics respectively, with negative removal efficiencies being attributed to runoff and remobilization of pesticides in full-scale systems.
The location of the wetlands for these 31 primary studies with the highest removal efficiencies of TN and NO3-N occurred in the United States [67,99], while the highest removal efficiencies of NH4-N and pesticides occurred in the United States and Greece [77,99,218]. In contrast, the highest efficiencies of antibiotic removal occurred in China [58]. However, the variation in removal efficiencies reported throughout the primary studies was likely due to the wide range of CUPS and antibiotics studied, the type of wetland systems (e.g., scale, wetland type, plant type), and the climatic conditions of the wetland analyzed. For example, Lu et al. (2021) reported that removal efficiencies of NH4+-N were most affected by temperature, rather than the concentration of contaminants, with increasing removal efficiencies occurring at higher temperatures and with increased contact time [113]. For the 31 primary studies identified as analyzing runoff mixtures containing nitrogen, CUPs, and/or antibiotics, only two studies analyzed natural wetland systems and found relatively high removal efficiencies (43–100%) for TN, NO3-N, NH4+-N, atrazine, S-metolachlor, and Permethrin [77,184]. The lowest removal efficiencies for TN and NH4+-N were found with full-scale horizontal flow constructed wetlands [3,213], while the lowest removal efficiencies for NO3-N and antibiotics were found with mesocosm vertical flow constructed wetlands [114]. In contrast, the lowest removal efficiencies for pesticides were associated with a full-scale wetland buffer [126]. Despite these external climatic and wetland design parameters, the leading cause for N-removal disruptions is thought to be due to contaminant mixture type and concentration.
Several primary studies reported the impact of CUPs’ and antibiotics’ presence on nitrogen removal efficiencies. Of these, seven studies reported that the specific contaminant analyzed decreased nitrogen removal [30,58,67,113,114,169]. The impact of CUPS and antibiotics on nutrient removal was mainly attributed to a decline in the microbial communities responsible for nutrient metabolism and degradation [30,114,131,169], where the presence of plants and type of plant species [23,102,120,201], saturated vs. unsaturated conditions [120,201], weather conditions [201], and timing [48,169] also played a role in N removal in the presence of CUPS or antibiotics. Ohore et al. (2021) reported that the presence of tetracycline decreased nitrogen removal initially, but observed an increase in total N removal with an increasing number of days due to the degradation of antibiotics in the wetland system [169]. Additionally, Tong et al. (2019) observed the presence of plants protected the microbial communities, limiting ofloxacin’s ability to negatively impact NO3-N and NH4+-N removal [30].
However, the presence of CUPs and antibiotics has also been observed not to affect nitrogen removal in wetland treatment systems [23,158,201], and in some cases, the CUPs or antibiotics increased N removal [43,131]. These increases in N removal are presumed to be due to an increase in microbial community diversity and richness in the presence of CUPS or antibiotics [23,171]. Yu et al. (2019) observed that tetracycline had a slightly negative effect on nitrogen removal (91% to 71%); however, the presence of copper and tetracycline led to higher microbial richness with increases in microbial variations [171]. In contrast, Lu et al. (2021) observed that the presence of sulfamethoxazole had positive influences on denitrifying bacteria interactions, but reduced the network complexity and microbial interactions in the wetland mesocosms [114].
Runoff mixtures not only impact N removal efficiencies, but also impact the wetlands’ ability to remove CUPs and antibiotics. Recent investigations on the impact of N on CUPs indicated that nitrifying bacteria can also degrade certain pesticides (e.g., metribuzin, imazalil, tebuconazole) [59,102,120]. However, in some cases, the presence of nutrients decreased CUP or antibiotic removal efficiencies in the wetland [171,205]. For example, Matamoros et al. (2020) reported both the presence of nutrients impacted CECs removal and that the presence of other CECs (caffeine, tributyl phosphate, 5TTri, bisphenol A, benzotriazole, carbamazepine, diclofenac, ibuprofen, lorazepam, naproxen, oxazepam, primidone, and triclosan) can reduce pesticide (sulfonyl 104, alachlor, bentazone, chlorpyrifos, DEET, molinate, oxadiazone, propanil, tebuconazole, and MCPA) removal in rice fields [103].
4. Conclusions
This review sought to analyze 181 primary studies that used wetlands of varying scales to treat runoff from urban and agricultural landscapes; this was accomplished by assessing the bibliometric information of the primary studies, identifying wetland scale and type, quantifying the source and mixture of CUPs and antibiotics entering wetlands, identifying mechanisms of removal used to treat runoff mixtures, and analyzing the implications of these mixtures to nitrogen removal processes (e.g., denitrification, plant uptake). While the scientific production of wetland treatment systems used to treat runoff has increased by 29%, of the primary studies reviewed, only 16 wetland treatment systems received urban runoff, while 82% of the studies used wetlands to treat agricultural runoff. Additionally, antibiotics and runoff mixtures have only recently begun to be studied. This has resulted in only 31 of the primary studies evaluating removal efficiencies in the presence of runoff mixtures, with only one study analyzing mixtures containing nutrients, antibiotics, and pesticides.
The impacts on CUP (−619–100%), antibiotic (35–100%), and N (TN = 5–99%, NO3-N= −113–98%, NH4-N = 7–100%) removal efficiencies varied greatly over the primary studies. Variations were likely due to the wide range of different types of CUPs (n = 556) and antibiotics (n = 60) analyzed. Additionally, the location, scale of the wetland system (microcosm, mesocosm, field-scale), presence and type of plants, the substrate used, weather conditions, wetland type, and design parameters all impacted the efficacy of the wetland system for water quality treatment. However, the largest impact on nitrogen removal was the shift in microbial community diversity and richness in the presence of CECs; however, this shift was seen to both increase and decrease microbial communities depending on the contaminant mixture, showing a lack of understanding of how microbial communities are impacted by different types and mixtures of CUPs and antibiotics. In addition, long-term exposure analyses of wetland treatment processes and efficacy were limited.
Throughout the primary studies, a lack of uniform reporting on wetland removal performance appeared, with a portion of the studies reporting removal efficiencies, defined as the difference between the influent and effluent mean concentrations over the influent concentration, while others reported removal loads, defined as a mass balance removal rate. Some primary studies also reported on sorption isotherms, microbial populations, or toxicity. In addition, the wide variety of different classes of CUPs and antibiotics analyzed throughout the primary studies made it difficult to perform further statistical analysis to compare wetland type, scale, length of study, and removal mechanisms with removal efficiencies. A significant knowledge gap remains in urban wetland treatment systems, which are continuing to become more important as urban centers continue to grow. Even more, a significant knowledge gap remains in how wetland treatment systems are used to treat non-point source mixtures containing antibiotics and CUPs, resulting in a significant unknown regarding nitrogen removal efficiency in wetland systems as contaminant runoff mixtures and mechanisms for treatment evolve.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/w13243631/s1, Table S1: Contaminant type and appearances in the primary studies. Table S2: Contaminant removal efficiencies for the primary studies that analyzed both nitrogen and pesticide and/or antibiotics in wetland treatment systems.
Author Contributions
Conceptualization, T.L.M. and E.R.N.; methodology, E.R.N. and T.L.M.; validation, E.R.N. and T.L.M.; formal analysis, E.R.N.; investigation, E.R.N.; data curation, E.R.N.; writing—original draft preparation, E.R.N.; writing—review and editing, E.R.N. and T.L.M.; visualization, E.R.N.; supervision, T.L.M.; project administration, T.L.M.; funding acquisition, T.L.M. All authors have read and agreed to the published version of the manuscript.
Funding
This material is based upon work supported by the National Science Foundation under Grant No. (2042761). This project was also supported with funds by the United States Hatch multistate capacity funding grant (W-4045). Any opinions, findings, and conclusions, or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation or United States Department of Agriculture.
Informed Consent Statement
Not applicable.
Acknowledgments
A special thanks to Mathew Russel, Dayana Nayelly Rodriguez Jimenez, Jacob Richardson, Kyra Sigler, and William Rud for their support and encouragement throughout the project.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Murdoch, P.S.; Baron, J.S.; Miller, T.L. Potential Effects of Climate Change on Surface-Water Quality in North America. JAWRA 2000, 36, 347–366. [Google Scholar] [CrossRef]
- EPA. Protecting Water Quality from Agricultural Runoff. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi/P10039OH.PDF?Dockey=P10039OH.PDF (accessed on 9 September 2021).
- White, K.D.; Meyers, A.L. Above par storm-water management. Civ. Eng. 1997, 67, 50. [Google Scholar]
- Panthi, S.; Sapkota, A.R.; Raspanti, G.; Allard, S.M.; Bui, A.; Craddock, H.A.; Murray, R.; Zhu, L.; East, C.; Handy, E.; et al. Pharmaceuticals, herbicides, and disinfectants in agricultural water sources. Environ. Res. 2019, 174, 1–8. [Google Scholar] [CrossRef]
- Jaimes-Correa, J.C.; Snow, D.D.; Bartelt-Hunt, S.L. Seasonal occurrence of antibiotics and a beta agonist in an agriculturally-intensive watershed. Environ. Pol. 2015, 205, 87–96. [Google Scholar] [CrossRef]
- Vymazal, J.; Brezinova, T. The use of constructed wetlands for removal of pesticides from agricultural runoff and drainage: A review. Environ. Int. 2015, 75, 11–20. [Google Scholar] [CrossRef]
- Dwivedi, D.; Mohanty, B. Hot Spots and Persistence of Nitrate in Aquifers Across Scales. Entropy 2016, 18, 25. [Google Scholar] [CrossRef]
- Molenat, J.; Gascuel-Odoux, C.; Ruiz, L.; Gruau, G. Role of water table dynamics on stream nitrate export and concentration in agricultural headwater catchment (France). J. Hydrol. 2008, 348, 363–378. [Google Scholar] [CrossRef]
- Mittelstet, A.R.; Gilmore, T.E.; Messer, T.; Rudnick, D.R.; Heatherly, T. Evaluation of selected watershed characteristics to identify best management practices to reduce Nebraskan nitrate loads from Nebraska to the Mississippi/Atchafalaya River basin. Agric. Ecosyst. Environ. 2019, 277, 1–10. [Google Scholar] [CrossRef]
- Katz, B.G.; Berndt, M.P.; Crandall, C.A. Factors affecting the movement and persistence of nitrate and pesticides in the surficial and upper Floridan aquifers in two agricultural areas in the southeastern United States. Environ. Earth Sci. 2013, 71, 2779–2795. [Google Scholar] [CrossRef]
- Karpuzcu, M.E.; Stringfellow, W.T. Kinetics of nitrate removal in wetlands receiving agricultural drainage. Ecol. Eng. 2012, 42, 295–303. [Google Scholar] [CrossRef]
- Roser, M. Pesticides—Our World in Data. Available online: https://ourworldindata.org/pesticides (accessed on 9 September 2021).
- Atwood, D.; Paisley-Jones, C. Pesticides Industry Sales and Usage, 2008–2012 Market Estimates. Available online: https://www.epa.gov/pesticides/pesticides-industry-sales-and-usage-2008-2012-market-estimates (accessed on 9 September 2021).
- Nowell, L.H.; Moran, P.W.; Bexfield, L.M.; Mahler, B.J.; Van Metre, P.C.; Bradley, P.M.; Schmidt, T.S.; Button, D.T.; Qi, S.L. Is there an urban pesticide signature? Urban streams in five U.S. regions share common dissolved-phase pesticides but differ in predicted aquatic toxicity. Sci. Total Environ. 2021, 793, 148453. [Google Scholar] [CrossRef]
- Wang, J.; Chu, L.; Wojnarovits, L.; Takacs, E. Occurrence and fate of antibiotics, antibiotic resistant genes (ARGs) and antibiotic resistant bacteria (ARB) in municipal wastewater treatment plant: An overview. Sci. Total Environ. 2020, 744, 140997. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Xie, H.J.; Mu, Y.; Xu, X.; Zhang, J.; Liu, C.; Liang, S.; Ngo, H.H.; Guo, W.; Xu, J.; et al. Bioremediation of endosulfan in laboratory-scale constructed wetlands: Effect of bioaugmentation and biostimulation. Environ. Sci. Pol. Res. 2014, 21, 12827–12835. [Google Scholar] [CrossRef] [PubMed]
- Chauzat, M.P.; Faucon, J.P. Pesticide residues in beeswax samples collected from honey bee colonies (Apis mellifera L.) in France. Pest Manag. Sci. 2007, 63, 1100–1106. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.C.; Dubetz, C.; Palace, V.P. Neonicotinoids in the Canadian aquatic environment: A literature review on current use products with a focus on fate, exposure, and biological effects. Sci. Total Environ. 2015, 505, 409–422. [Google Scholar] [CrossRef]
- Ben, Y.; Fu, C.; Hu, M.; Liu, L.; Wong, M.H.; Zheng, C. Human health risk assessment of antibiotic resistance associated with antibiotic residues in the environment: A review. Environ. Res. 2019, 169, 483–493. [Google Scholar] [CrossRef]
- Trepel, M. Assessing the cost-effectiveness of the water purification function of wetlands for environmental planning. Ecol. Complex. 2010, 7, 320–326. [Google Scholar] [CrossRef]
- Weerakoon, G.M.P.R.; Jinadasa, K.B.S.N.; Herath, G.B.B.; Mowjood, M.I.M.; Ng, W.J. Applicability of constructed wetlands for water quality improvement in a tea estate catchment: The Pussellawa case study. Water 2018, 10, 332. [Google Scholar] [CrossRef]
- Fitch, M.W. Constructed Wetlands. Compr. Water Qual. Purif. 2014, 3, 268–295. [Google Scholar] [CrossRef]
- Tong, X.N.; Wang, X.Z.; He, X.J.; Wang, Z.; Li, W.X. Effects of antibiotics on microbial community structure and microbial functions in constructed wetlands treated with artificial root exudates. Environ. Sci. Process. Impacts 2020, 22, 217–226. [Google Scholar] [CrossRef]
- Qasaimeh, A.; AlSharie, H.; Masoud, T. A Review on Constructed Wetlands Components and Heavy Metal Removal from Wastewater. J. Environ. Prot. 2015, 6, 710–718. [Google Scholar] [CrossRef]
- Moore, M.T.; Schulz, R.; Cooper, C.M.; Smith, S.; Rodgers, J.H., Jr. Mitigation of chlorpyrifos runoff using constructed wetlands. Chemosphere 2002, 46, 827–835. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Tanner, C.C.; Hally, V.M.; Gibbs, M.M. Nitrogen spiraling in subsurface-flow constructed wetlands: Implications for treatment response. Ecol. Eng. 2005, 25, 365–381. [Google Scholar] [CrossRef]
- Borin, M.; Tocchetto, D. Five year water and nitrogen balance for a constructed surface flow wetland treating agricultural drainage waters. Sci. Total Environ. 2007, 380, 38–47. [Google Scholar] [CrossRef]
- Uwimana, A.; van Dam, A.A.; Irvine, K. Effects of conversion of wetlands to rice and fish farming on water quality in valley bottoms of the Migina catchment, southern Rwanda. Ecol. Eng. 2018, 125, 76–86. [Google Scholar] [CrossRef]
- Bachand, P.A.M.; Horne, A.J. Denitrification in constructed free-water surface wetlands: I. very high nitrate removal rates in a macrocosm study. Ecol. Eng. 2000, 14, 9–15. [Google Scholar] [CrossRef]
- Tong, X.; Wang, X.; He, X.; Xu, K.; Mao, F. Effects of ofloxacin on nitrogen removal and microbial community structure in constructed wetland. Sci. Total Environ. 2019, 656, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Pontius, F.W. Emerging Contaminants in Water: Detection, Treatment, and Regulation. Water 2021, 13, 1470. [Google Scholar] [CrossRef]
- Stoler, A.; Walker, B.; Hintz, W.; Jones, D.; Lind, L.; Mattes, B.; Schuler, M.; Relyea, R. Combined Effects of Road Salt and an Insecticide on Wetland Communities. Environ. Toxicol. Chem. 2017, 36, 771–779. [Google Scholar] [CrossRef]
- Tu, C.M. Effect of five insecticides on microbial and enzymatic activities in sandy soil. J. Environ. Sci. Health, Part B 1995, 30, 289–306. [Google Scholar] [CrossRef]
- Shan, J.; Yang, P.; Rahman, M.M.; Shang, X.; Yan, X. Tetracycline and sulfamethazine alter dissimilatory nitrate reduction processes and increase N2O release in rice fields. Environ. Pol. 2018, 242, 788–796. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, W.; Tang, L.; Heenan, M.; Xu, Z. Effects of nitrification inhibitor and herbicides on nitrification, nitrite and nitrate consumptions and nitrous oxide emission in an Australian sugarcane soil. Biol. Fertil. Soils 2018, 54, 697–706. [Google Scholar] [CrossRef]
- Robson, S.V.; Rosi, E.J.; Richmond, E.K.; Grace, M.R. Environmental concentrations of pharmaceuticals alter metabolism, denitrification, and diatom assemblages in artificial streams. Freshw. Sci. 2020, 39, 256–267. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Zheng, Y.; Liu, Y.; Qu, M.; Hao, M.; Yang, D.; Yang, Q.; Wang, X.C.; Dzakpasu, M. Fate of an antibiotic and its effects on nitrogen transformation functional bacteria in integrated vertical flow constructed wetlands. Chem. Eng. J. 2021, 417, 129272. [Google Scholar] [CrossRef]
- Martyniuk, C.J.; Doperalski, N.J.; Prucha, M.S.; Zhang, J.L.; Kroll, K.J.; Conrow, R.; Barber, D.S.; Denslow, N.D. High contaminant loads in Lake Apopka’s riparian wetland disrupt gene networks involved in reproduction and immune function in largemouth bass. Comp. Biochem. Physiol. Part D Genom. Proteom. 2016, 19, 140–150. [Google Scholar] [CrossRef]
- Sen, K.; Berglund, T.; Soares, M.A.; Taheri, B.; Ma, Y.; Khalil, L.; Fridge, M.; Lu, J.; Turner, R.J. Antibiotic resistance of E. coli Isolated from a Constructed Wetland Dominated by a Crow Roost, with Emphasis on ESBL and AmpC Containing E. coli. Front. Microbiol. 2019, 10, 1034. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.A.; Prasher, S.O.; Chenier, M.; Arya, G. Removal of nitrate-N by antibiotic exposed bacterial isolates from constructed wetlands. World J. Microbiol. Biotechnol. 2011, 27, 2061–2069. [Google Scholar] [CrossRef]
- Jia, L.; Sun, H.; Zhou, Q.; Zhao, L.; Wu, W. Pilot-scale two-stage constructed wetlands based on novel solid carbon for rural wastewater treatment in southern China: Enhanced nitrogen removal and mechanism. J. Environ. Manag. 2021, 292, 112750. [Google Scholar] [CrossRef]
- Moore, M.T.; Rodgers, J.H., Jr.; Smith, S., Jr.; Cooper, C.M. Mitigation of metolachlor-associated agricultural runoff using constructed wetlands in Mississippi, USA. Agric. Ecosyst. Environ. 2001, 84, 169–176. [Google Scholar] [CrossRef]
- Schultz, R.; Colletti, J.; Isenhart, T.; Simpkins, W.; Mize, C.; Thompson, M. Design and placement of a multi-species riparian buffer strip system. Agroforest. Syst. 1995, 29, 201–226. [Google Scholar] [CrossRef]
- Runes, H.; Jenkins, J.; Bottomley, P. Atrazine degradation by bioaugmented sediment from constructed wetlands. Appl. Microbiol. Biotechnol. 2001, 57, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Chao, H.; Chen, K. Treatment of Organic Matter and Tetracycline in Water by Using Constructed Wetlands and Photocatalysis. Appl. Sci. 2019, 9, 2680. [Google Scholar] [CrossRef]
- Cedillo-Herrera, C.I.G.; Roé-Sosa, A.; Pat-Espadas, A.M.; Ramírez, K.; Rochín-Medina, J.; Amabilis-Sosa, L.E. Efficient malathion removal in constructed wetlands coupled to UV/H2O2 pretreatment. Appl. Sci. 2020, 10, 5036. [Google Scholar] [CrossRef]
- Bouldin, J.; Farris, J.; Moore, M.; Smith, S.; Cooper, C. Assessment of diazinon toxicity in sediment and water of constructed wetlands using deployed Corbicula fluminea and laboratory testing. Arch. Environ. Contam. Toxicol. 2007, 53, 174–182. [Google Scholar] [CrossRef]
- Lizotte, R.E., Jr.; Moore, M.T.; Locke, M.A.; Kröger, R. Role of vegetation in a constructed wetland on nutrient-pesticide mixture toxicity to Hyalella azteca. Arch. Environ. Contam. Toxicol. 2011, 60, 261–271. [Google Scholar] [CrossRef]
- Lizotte, R.; Testa, S.; Locke, M.; Steinriede, R. Responses of Phytoplankton and Hyalella azteca to Agrichemical Mixtures in a Constructed Wetland Mesocosm. Arch. Environ. Contam. Toxicol. 2013, 65, 474–485. [Google Scholar] [CrossRef]
- Mazanti, L.; Rice, C.; Bialek, K.; Sparling, D.; Stevenson, C.; Johnson, W.E.; Kangas, P.; Rheinstein, J. Aqueous-phase disappearance of atrazine, metolachlor, and chlorpyrifos in laboratory aquaria and outdoor macrocosms. Arch. Environ. Contam. Toxicol. 2003, 44, 67–76. [Google Scholar] [CrossRef]
- Moore, M.T.; Kröger, R.; Cooper, C.M.; Smith, S., Jr. Ability of four emergent macrophytes to remediate permethrin in mesocosm experiments. Arch. Environ. Contam. Toxicol. 2009, 57, 282–288. [Google Scholar] [CrossRef]
- Schulz, R.; Moore, M.T.; Bennett, E.R.; Milam, C.D.; Bouldin, J.L.; Farris, J.L.; Smith, S., Jr.; Cooper, C.M. Acute toxicity of methyl-parathion in wetland mesocosms: Assessing the influence of aquatic plants using laboratory testing with Hyalella azteca. Arch. Environ. Contam. Toxicol. 2003, 45, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Dash, N.P.; Kumar, A.; Kaushik, M.S.; Abraham, G.; Singh, P.K. Agrochemicals influencing nitrogenase, biomass of N2-fixing cyanobacteria and yield of rice in wetland cultivation. Biocatal. Agric. Biotechnol. 2017, 9, 28–34. [Google Scholar] [CrossRef]
- Guo, X.; Wang, P.; Li, Y.; Zhong, H.; Li, P.; Zhang, C.; Zhao, T. Effect of copper on the removal of tetracycline from water by Myriophyllum aquaticum: Performance and mechanisms. Bioresour. Technol. 2019, 291, 121916. [Google Scholar] [CrossRef] [PubMed]
- Ohore, O.E.; Zhang, S.; Guo, S.; Addo, F.G.; Manirakiza, B.; Zhang, W. Ciprofloxacin increased abundance of antibiotic resistance genes and shaped microbial community in epiphytic biofilm on Vallisneria spiralis in mesocosmic wetland. Bioresour. Technol. 2021, 323, 124574. [Google Scholar] [CrossRef]
- Yuan, Y.; Yang, B.; Wang, H.; Lai, X.; Li, F.; Salam, M.M.A.; Pan, F.; Zhao, Y. The simultaneous antibiotics and nitrogen removal in vertical flow constructed wetlands: Effects of substrates and responses of microbial functions. Bioresour. Technol. 2020, 310, 123419. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Bai, S.; Li, C.; Yang, J.; Ma, F. Bioaugmentation of atrazine removal in constructed wetland: Performance, microbial dynamics, and environmental impacts. Bioresour. Technol. 2019, 289, 121618. [Google Scholar] [CrossRef]
- Wilson, P.; Lu, H.; Lin, Y. Norflurazon and Simazine Removal from Surface Water Using a Constructed Wetland. Bull. Environ. Contam. Toxicol. 2011, 87, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.; Kroger, R.; Locke, M.; Lizotte, R.; Testa, S.; Cooper, C. Diazinon and Permethrin Mitigation Across a Grass-Wetland Buffer. Bull. Environ. Contam. Toxicol. 2014, 93, 574–579. [Google Scholar] [CrossRef]
- Moore, M.; Lizotte, R.; Kroger, R. Efficiency of Experimental Rice (Oryza sativa L.) Fields in Mitigating Diazinon Runoff Toxicity to Hyalella azteca. Bull. Environ. Contam. Toxicol. 2009, 82, 777–780. [Google Scholar] [CrossRef]
- Moore, M.T.; Lizotte, R.E., Jr.; Smith, S., Jr. Responses of Hyalella azteca to a pyrethroid mixture in a constructed wetland. Bull. Environ. Contam. Toxicol. 2007, 78, 245–248. [Google Scholar] [CrossRef]
- Gikas, G.; Vryzas, Z.; Tsihrintzis, V. S-metolachlor herbicide removal in pilot-scale horizontal subsurface flow constructed wetlands. Chem. Eng. J. 2018, 339, 108–116. [Google Scholar] [CrossRef]
- Gikas, G.D.; Pérez-Villanueva, M.; Tsioras, M.; Alexoudis, C.; Pérez-Rojas, G.; Masís-Mora, M.; Lizano-Fallas, V.; Rodríguez-Rodríguez, C.E.; Vryzas, Z.; Tsihrintzis, V.A. Low-cost approaches for the removal of terbuthylazine from agricultural wastewater: Constructed wetlands and biopurification system. Chem. Eng. J. 2018, 335, 647–656. [Google Scholar] [CrossRef]
- Lizotte, R.E., Jr.; Locke, M.A.; Testa Iii, S. Influence of varying nutrient and pesticide mixtures on abatement efficiency using a vegetated free water surface constructed wetland mesocosm. Chem. Ecol. 2014, 30, 280–294. [Google Scholar] [CrossRef]
- Budd, R.; O’Geen, A.; Goh, K.S.; Bondarenko, S.; Gan, J. Removal mechanisms and fate of insecticides in constructed wetlands. Chemosphere 2011, 83, 1581–1587. [Google Scholar] [CrossRef] [PubMed]
- Caldelas, C.; Gurí, R.; Araus, J.; Sorolla, A. Effect of ZnO nanoparticles on Zn, Cu, and Pb dissolution in a green bioretention system for urban stormwater remediation. Chemosphere 2021, 282, 131045. [Google Scholar] [CrossRef]
- Cheng, S.; Vidakovic-Cifrek, Ž.; Grosse, W.; Karrenbrock, F. Xenobiotics removal from polluted water by a multifunctional constructed wetland. Chemosphere 2002, 48, 415–418. [Google Scholar] [CrossRef]
- Dosnon-Olette, R.; Trotel-Aziz, P.; Couderchet, M.; Eullaffroy, P. Fungicides and herbicide removal in Scenedesmus cell suspensions. Chemosphere 2010, 79, 117–123. [Google Scholar] [CrossRef]
- Elsayed, O.; Maillard, E.; Vuilleumier, S.; Nijenhuis, I.; Richnow, H.; Imfeld, G. Using compound-specific isotope analysis to assess the degradation of chloroacetanilide herbicides in lab-scale wetlands. Chemosphere 2014, 99, 89–95. [Google Scholar] [CrossRef]
- Gebremariam, S.Y.; Beutel, M.W. Effects of drain-fill cycling on chlorpyrifos mineralization in wetland sediment-water microcosms. Chemosphere 2010, 78, 1337–1341. [Google Scholar] [CrossRef]
- Imfeld, G.; Lefrancq, M.; Maillard, E.; Payraudeau, S. Transport and attenuation of dissolved glyphosate and AMPA in a stormwater wetland. Chemosphere 2013, 90, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Kuchta, S.L.; Cessna, A.J. Fate of lincomycin in snowmelt runoff from manure-amended pasture. Chemosphere 2009, 76, 439–446. [Google Scholar] [CrossRef]
- Kurade, M.; Xiong, J.; Govindwar, S.; Roh, H.; Saratale, G.; Jeon, B.; Lim, H. Uptake and biodegradation of emerging contaminant sulfamethoxazole from aqueous phase using Ipomoea aquatica. Chemosphere 2019, 225, 696–704. [Google Scholar] [CrossRef]
- Lizotte, R.; Shields, F.; Murdock, J.; Knight, S. Responses of Hyalella azteca and phytoplankton to a simulated agricultural runoff event in a managed backwater wetland. Chemosphere 2012, 87, 684–691. [Google Scholar] [CrossRef]
- Lv, T.; Zhang, Y.; Casas, M.E.; Carvalho, P.N.; Arias, C.A.; Bester, K.; Brix, H. Phytoremediation of imazalil and tebuconazole by four emergent wetland plant species in hydroponic medium. Chemosphere 2016, 148, 459–466. [Google Scholar] [CrossRef]
- Maillard, E.; Lange, J.; Schreiber, S.; Dollinger, J.; Herbstritt, B.; Millet, M.; Imfeld, G. Dissipation of hydrological tracers and the herbicide S-metolachlor in batch and continuous-flow wetlands. Chemosphere 2016, 144, 2489–2496. [Google Scholar] [CrossRef] [PubMed]
- Matamoros, V.; Arias, C.A.; Nguyen, L.X.; Salvadó, V.; Brix, H. Occurrence and behavior of emerging contaminants in surface water and a restored wetland. Chemosphere 2012, 88, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Page, D.; Dillon, P.; Mueller, J.; Bartkow, M. Quantification of herbicide removal in a constructed wetland using passive samplers and composite water quality monitoring. Chemosphere 2010, 81, 394–399. [Google Scholar] [CrossRef]
- Passeport, E.; Tournebize, J.; Chaumont, C.; Guenne, A.; Coquet, Y. Pesticide contamination interception strategy and removal efficiency in forest buffer and artificial wetland in a tile-drained agricultural watershed. Chemosphere 2013, 91, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Liébana, J.A.; ElGouzi, S.; Peña, A. Laboratory persistence in soil of thiacloprid, pendimethalin and fenarimol incubated with treated wastewater and dissolved organic matter solutions. Contribution of soil biota. Chemosphere 2017, 181, 508–517. [Google Scholar] [CrossRef]
- Rose, M.T.; Crossan, A.N.; Kennedy, I.R. The effect of vegetation on pesticide dissipation from ponded treatment wetlands: Quantification using a simple model. Chemosphere 2008, 72, 999–1005. [Google Scholar] [CrossRef]
- Rose, M.T.; Sanchez-Bayo, F.; Crossan, A.N.; Kennedy, I.R. Pesticide removal from cotton farm tailwater by a pilot-scale ponded wetland. Chemosphere 2006, 63, 1849–1858. [Google Scholar] [CrossRef]
- Tang, X.Y.; Yang, Y.; McBride, M.B.; Tao, R.; Dai, Y.N.; Zhang, X.M. Removal of chlorpyrifos in recirculating vertical flow constructed wetlands with five wetland plant species. Chemosphere 2019, 216, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Tsui, M.; Chu, L.M. Environmental fate and non-target impact of glyphosate-based herbicide (Roundup®) in a subtropical wetland. Chemosphere 2008, 71, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Vandermeeren, P.; Baken, S.; Vanderstukken, R.; Diels, J.; Springael, D. Impact of dry-wet and freeze-thaw events on pesticide mineralizing populations and their activity in wetland ecosystems: A microcosm study. Chemosphere 2016, 146, 85–93. [Google Scholar] [CrossRef]
- Vilas-Boas, J.A.; Arenas-Sánchez, A.; Vighi, M.; Romo, S.; Brink, P.J.V.D.; Dias, R.J.P.; Rico, A. Multiple stressors in Mediterranean coastal wetland ecosystems: Influence of salinity and an insecticide on zooplankton communities under different temperature conditions. Chemosphere 2021, 269, 129381. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.; Te, S.H.; Reinhard, M.; Yang, Y.; Chen, H.; He, Y.; Gin, K.Y.H. Biotransformation of Sulfluramid (N-ethyl perfluorooctane sulfonamide) and dynamics of associated rhizospheric microbial community in microcosms of wetland plants. Chemosphere 2018, 211, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Kanjilal, T.; Bhattacharjee, C.; Datta, S. Utilization of S. aureusstrain 502A in biodegradation of insecticide acetamiprid from wetland wastewater. Desalination Water Treat. 2015, 57, 13190–13206. [Google Scholar] [CrossRef]
- Lizotte, R.; Shields, F.; Knight, S.; Bryant, C. Efficiency of a modified backwater wetland in trapping a pesticide mixture. Ecohydrology 2009, 2, 287–293. [Google Scholar] [CrossRef]
- Bundschuh, M.; Elsaesser, D.; Stang, C.; Schulz, R. Mitigation of fungicide pollution in detention ponds and vegetated ditches within a vine-growing area in Germany. Ecol. Eng. 2016, 89, 121–130. [Google Scholar] [CrossRef]
- Douglass, J.F.; Radosevich, M.; Tuovinen, O.H. Molecular analysis of atrazine-degrading bacteria and catabolic genes in the water column and sediment of a created wetland in an agricultural/urban watershed. Ecol. Eng. 2016, 83, 405–412. [Google Scholar] [CrossRef]
- Elsaesser, D.; Blankenberg, A.-G.B.; Geist, A.; Mæhlum, T.; Schulz, R. Assessing the influence of vegetation on reduction of pesticide concentration in experimental surface flow constructed wetlands: Application of the toxic units approach. Ecol. Eng. 2011, 37, 955–962. [Google Scholar] [CrossRef]
- Gaullier, C.; Baran, N.; Dousset, S.; Devau, N.; Billet, D.; Kitzinger, G.; Coisy, E. Wetland hydrodynamics and mitigation of pesticides and their metabolites at pilot-scale. Ecol. Eng. 2019, 136, 185–192. [Google Scholar] [CrossRef]
- Hongbin, L.U.; Wang, H.; Shaoyong, L.U.; Jiaxin, L.I.; Wang, T. Response mechanism of typical wetland plants and removal of water pollutants under different levofloxacin concentration. Ecol. Eng. 2020, 158, 106023. [Google Scholar] [CrossRef]
- Hussain, S.A.; Prasher, S.O.; Patel, R.M. Removal of ionophoric antibiotics in free water surface constructed wetlands. Ecol. Eng. 2012, 41, 13–21. [Google Scholar] [CrossRef]
- Kohler, E.A.; Poole, V.L.; Reicher, Z.J.; Turco, R.F. Nutrient, metal, and pesticide removal during storm and nonstorm events by a constructed wetland on an urban golf course. Ecol. Eng. 2004, 23, 285–298. [Google Scholar] [CrossRef]
- Krone-Davis, P.; Watson, F.; Los Huertos, M.; Starner, K. Assessing pesticide reduction in constructed wetlands using a tanks-in-series model within a Bayesian framework. Ecol. Eng. 2013, 57, 342–352. [Google Scholar] [CrossRef]
- Lebrun, J.D.; Ayrault, S.; Drouet, A.; Bordier, L.; Fechner, L.C.; Uher, E.; Chaumont, C.; Tournebize, J. Ecodynamics and bioavailability of metal contaminants in a constructed wetland within an agricultural drained catchment. Ecol. Eng. 2019, 136, 108–117. [Google Scholar] [CrossRef]
- Lv, T.; Zhang, Y.; Carvalho, P.N.; Zhang, L.; Button, M.; Arias, C.A.; Weber, K.P.; Brix, H. Microbial community metabolic function in constructed wetland mesocosms treating the pesticides imazalil and tebuconazole. Ecol. Eng. 2017, 98, 378–387. [Google Scholar] [CrossRef]
- Matamoros, V.; Caiola, N.; Rosales, V.; Hernández, O.; Ibáñez, C. The role of rice fields and constructed wetlands as a source and a sink of pesticides and contaminants of emerging concern: Full-scale evaluation. Ecol. Eng. 2020, 156, 105971. [Google Scholar] [CrossRef]
- Papaevangelou, V.A.; Gikas, G.D.; Vryzas, Z.; Tsihrintzis, V.A. Treatment of agricultural equipment rinsing water containing a fungicide in pilot-scale horizontal subsurface flow constructed wetlands. Ecol. Eng. 2017, 101, 193–200. [Google Scholar] [CrossRef]
- Romain, V.; Sylvie, D.; David, B. Water residence time and pesticide removal in pilot-scale wetlands. Ecol. Eng. 2015, 85, 76–84. [Google Scholar] [CrossRef]
- Schulz, R.; Peall, S.K.C.; Hugo, C.; Krause, V. Concentration, load and toxicity of spraydrift-borne azinphos-methyl at the inlet and outlet of a constructed wetland. Ecol. Eng. 2001, 18, 239–245. [Google Scholar] [CrossRef]
- Tournebize, J.; Passeport, E.; Chaumont, C.; Fesneau, C.; Guenne, A.; Vincent, B. Pesticide de-contamination of surface waters as a wetland ecosystem service in agricultural landscapes. Ecol. Eng. 2013, 56, 51–59. [Google Scholar] [CrossRef]
- Yu, X.; Zhen, S.; Zheng, M.; Ma, X.; Wang, G.; Zou, Y. Herbicide accumulations in the Xingkai lake area and the use of restored wetland for agricultural drainage treatment. Ecol. Eng. 2018, 120, 260–265. [Google Scholar] [CrossRef]
- Alvord, H.; Kadlec, R. Atrazine fate and transport in the Des Plaines Wetlands. Ecol. Model. 1996, 90, 97–107. [Google Scholar] [CrossRef]
- Milenkovski, S.; Bååth, E.; Lindgren, P.E.; Berglund, O. Toxicity of fungicides to natural bacterial communities in wetland water and sediment measured using leucine incorporation and potential denitrification. Ecotoxicology 2010, 19, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Dosnon-Olette, R.; Couderchet, M.; Eullaffroy, P. Phytoremediation of fungicides by aquatic macrophytes: Toxicity and removal rate. Ecotoxicol. Environ. Saf. 2009, 72, 2096–2101. [Google Scholar] [CrossRef]
- Guo, X.; Zhong, H.; Li, P.; Zhang, C. Microbial communities responded to tetracyclines and Cu(II) in constructed wetlands microcosms with Myriophyllum aquaticum. Ecotoxicol. Environ. Saf. 2020, 205, 111362. [Google Scholar] [CrossRef]
- Lu, H.; Wang, T.; Lu, S.; Liu, H.; Wang, H.; Li, C.; Liu, X.; Guo, X.; Zhao, X.; Liu, F. Performance and bacterial community dynamics of hydroponically grown Iris pseudacorus L. during the treatment of antibiotic-enriched wastewater at low/normal temperature. Ecotoxicol. Environ. Saf. 2021, 213, 111997. [Google Scholar] [CrossRef]
- Lu, S.; Zhang, Y.; Liu, X.; Xu, J.; Liu, Y.; Guo, W.; Liu, X.; Chen, J. Effects of sulfamethoxazole on nitrogen removal and molecular ecological network in integrated vertical-flow constructed wetland. Ecotoxicol. Environ. Saf. 2021, 219, 112292. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.; Chen, J.; Qiao, X.; Tian, R.; Arafat, Y.; Yang, X. Bacterial community variations in paddy soils induced by application of veterinary antibiotics in plant-soil systems. Ecotoxicol. Environ. Saf. 2019, 167, 44–53. [Google Scholar] [CrossRef]
- Ramos, A.; Whelan, M.J.; Guymer, I.; Villa, R.; Jefferson, B. On the potential of on-line free-surface constructed wetlands for attenuating pesticide losses from agricultural land to surface waters. Environ. Chem. 2019, 16, 563–576. [Google Scholar] [CrossRef]
- Cryder, Z.; Wolf, D.; Carlan, C.; Gan, J. Removal of urban-use insecticides in a large-scale constructed wetland. Environ. Pollut. 2021, 268, 115586. [Google Scholar] [CrossRef]
- Guo, X.; Mu, Q.; Zhong, H.; Li, P.; Zhang, C.; Wei, D.; Zhao, T. Rapid removal of tetracycline by Myriophyllum aquaticum: Evaluation of the role and mechanisms of adsorption. Environ. Pollut. 2019, 254, 113101. [Google Scholar] [CrossRef]
- Lv, T.; Carvalho, P.N.; Casas, M.E.; Bollmann, U.E.; Arias, C.A.; Brix, H.; Bester, K. Enantioselective uptake, translocation and degradation of the chiral pesticides tebuconazole and imazalil by Phragmites australis. Environ. Pollut. 2017, 229, 362–370. [Google Scholar] [CrossRef]
- Lyu, T.; Zhang, L.; Xu, X.; Arias, C.A.; Brix, H.; Carvalho, P.N. Removal of the pesticide tebuconazole in constructed wetlands: Design comparison, influencing factors and modelling. Environ. Pollut. 2018, 233, 71–80. [Google Scholar] [CrossRef]
- Moore, M.T.; Bennett, E.R.; Cooper, C.M.; Smith, S., Jr.; Farris, J.L.; Drouillard, K.G.; Schulz, R. Influence of vegetation in mitigation of methyl parathion runoff. Environ. Pollut. 2006, 142, 288–294. [Google Scholar] [CrossRef]
- Moore, M.T.; Cooper, C.M.; Smith, S.; Cullum, R.F.; Knight, S.S.; Locke, M.A.; Bennett, E.R. Mitigation of two pyrethroid insecticides in a Mississippi Delta constructed wetland. Environ. Pollut. 2009, 157, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.T.; Rodgers, J.H., Jr.; Cooper, C.M.; Smith, S., Jr. Constructed wetlands for mitigation of atrazine-associated agricultural runoff. Environ. Pollut. 2000, 110, 393–399. [Google Scholar] [CrossRef]
- Sherrard, R.M.; Bearr, J.S.; Murray-Gulde, C.L.; Rodgers, J.H., Jr.; Shah, Y.T. Feasibility of constructed wetlands for removing chlorothalonil and chlorpyrifos from aqueous mixtures. Environ. Pollut. 2004, 127, 385–394. [Google Scholar] [CrossRef]
- Stoler, A.; Mattes, B.; Hintz, W.; Jones, D.; Lind, L.; Schuler, M.; Relyea, R. Effects of a common insecticide on wetland communities with varying quality of leaf litter inputs. Environ. Pollut. 2017, 226, 452–462. [Google Scholar] [CrossRef]
- Vallée, R.; Dousset, S.; Schott, F.X.; Pallez, C.; Ortar, A.; Cherrier, R.; Munoz, J.F.; Benoît, M. Do constructed wetlands in grass strips reduce water contamination from drained fields? Environ. Pollut. 2015, 207, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, N.; Zalidis, G. The Use of Typha latifolia L. in Constructed Wetland Microcosms for the Remediation of Herbicide Terbuthylazine. Environ. Process 2019, 6, 985–1003. [Google Scholar] [CrossRef]
- Gaillard, J.; Thomas, M.; Lazartigues, A.; Bonnefille, B.; Pallez, C.; Dauchy, X.; Feidt, C.; Banas, D. Potential of barrage fish ponds for the mitigation of pesticide pollution in streams. Environ. Sci. Pollut. Res. 2016, 23, 23–35. [Google Scholar] [CrossRef]
- Gaullier, C.; Dousset, S.; Billet, D.; Baran, N. Is pesticide sorption by constructed wetland sediments governed by water level and water dynamics? Environ. Sci. Pollut. Res. 2018, 25, 14324–14335. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Liang, J.; Gong, J.; Song, B.; Yang, Z.; Fang, S.; Zhang, P.; Cao, W.; Li, J.; Luo, Y. The effects of biochar/compost for adsorption behaviors of sulfamethoxazole in amended wetland soil. Environ. Sci. Pollut. Res. 2021, 28, 49289–49301. [Google Scholar] [CrossRef]
- Tong, X.; Wang, X.; He, X.; Sui, Y.; Shen, J.; Feng, J. Effects of antibiotics on nitrogen uptake of four wetland plant species grown under hydroponic culture. Environ. Sci. Pollut. Res. 2019, 26, 10621–10630. [Google Scholar] [CrossRef]
- Ulrich, U.; Lange, J.; Pfannerstill, M.; Loose, L.; Fohrer, N. Hydrological tracers, the herbicide metazachlor and its transformation products in a retention pond during transient flow conditions. Environ. Sci. Pollut. Res. 2019, 26, 26706–26720. [Google Scholar] [CrossRef] [PubMed]
- Vallée, R.; Dousset, S.; Billet, D. Influence of substrate water saturation on pesticide dissipation in constructed wetlands. Environ. Sci. Pollut. Res. 2016, 23, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Vallée, R.; Dousset, S.; Billet, D.; Benoit, M. Sorption of selected pesticides on soils, sediment and straw from a constructed agricultural drainage ditch or pond. Environ. Sci. Pollut. Res. 2014, 21, 4895–4905. [Google Scholar] [CrossRef] [PubMed]
- Budd, R.; O’Geen, A.; Goh, K.S.; Bondarenko, S.; Gan, J. Efficacy of constructed wetlands in pesticide removal from tailwaters in the central valley, California. Environ. Sci. Technol. 2009, 43, 2925–2930. [Google Scholar] [CrossRef] [PubMed]
- Carley, L.N.; Panchagavi, R.; Song, X.; Davenport, S.; Bergemann, C.M.; McCumber, A.W.; Gunsch, C.K.; Simonin, M. Long-Term Effects of Copper Nanopesticides on Soil and Sediment Community Diversity in Two Outdoor Mesocosm Experiments. Environ. Sci. Technol. 2020, 54, 8878–8889. [Google Scholar] [CrossRef] [PubMed]
- Jeppe, K.; Kellar, C.; Marshall, S.; Colombo, V.; Sinclair, G.; Pettigrove, V. Bifenthrin Causes Toxicity in Urban Stormwater Wetlands: Field and Laboratory Assessment Using Austrochiltonia (Amphipoda). Environ. Sci. Technol. 2017, 51, 7254–7262. [Google Scholar] [CrossRef] [PubMed]
- Kuechle, K.J.; Webb, E.B.; Mengel, D.; Main, A.R.; Webb, L. Factors Influencing Neonicotinoid Insecticide Concentrations in Floodplain Wetland Sediments across Missouri. Environ. Sci. Technol. 2019, 53, 10591–10600. [Google Scholar] [CrossRef]
- Maillard, E.; Imfeld, G. Pesticide Mass Budget in a Stormwater Wetland. Environ. Sci. Technol. 2014, 48, 8603–8611. [Google Scholar] [CrossRef]
- Main, A.; Michel, N.; Headley, J.; Peru, K.; Morrissey, C. Ecological and Landscape Drivers of Neonicotinoid Insecticide Detections and Concentrations in Canada’s Prairie Wetlands. Environ. Sci. Technol. 2015, 49, 8367–8376. [Google Scholar] [CrossRef]
- Schulz, R.; Hahn, C.; Bennett, E.R.; Dabrowski, J.M.; Thiere, G.; Peall, S.K.C. Fate and effects of azinphos-methyl in a flow-through wetland in South Africa. Environ. Sci. Technol. 2003, 37, 2139–2144. [Google Scholar] [CrossRef]
- Schulz, R.; Peall, S.K.C. Effectiveness of a constructed wetland for retention of nonpoint-source pesticide pollution in the Lourens River catchment, South Africa. Environ. Sci. Technol. 2001, 35, 422–426. [Google Scholar] [CrossRef]
- Bouldin, J.L.; Farris, J.L.; Moore, M.T.; Smith, S., Jr.; Stephens, W.W.; Cooper, C.M. Evaluated fate and effects of atrazine and lambda-cyhalothrin in vegetated and unvegetated microcosms. Environ. Toxicol. 2005, 20, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Milam, C.D.; Bouldin, J.L.; Farris, J.L.; Schulz, R.; Moore, M.T.; Bennett, E.R.; Cooper, C.M.; Smith, S., Jr. Evaluating acute toxicity of methyl parathion application in constructed wetland mesocosms. Environ. Toxicol. 2004, 19, 471–479. [Google Scholar] [CrossRef]
- Passeport, E.; Benoit, P.; Bergheaud, V.; Coquet, Y.; Tournebize, J. Selected pesticides adsorption and desorption in substrates from artificial wetland and forest buffer. Environ. Toxicol. Chem. 2011, 30, 1669–1676. [Google Scholar] [CrossRef]
- Thomas, K.; Hand, L. Assessing the potential for algae and macrophytes to degrade crop protection products in aquatic ecosystems. Environ. Toxicol. Chem. 2011, 30, 622–631. [Google Scholar] [CrossRef]
- Pappalardo, S.; Otto, S.; Gasparini, V.; Zanin, G.; Borin, M. Mitigation of herbicide runoff as an ecosystem service from a constructed surface flow wetland. Hydrobiologia 2016, 774, 193–202. [Google Scholar] [CrossRef]
- Greiwe, J.; Olsson, O.; Kummerer, K.; Lange, J. Pesticide peak concentration reduction in a small vegetated treatment system controlled by chemograph shape. Hydrol. Earth Syst. Sci. 2021, 25, 497–509. [Google Scholar] [CrossRef]
- Blankenberg, A.-G.B.; Braskerud, B.; Haarstad, K. Pesticide retention in two small constructed wetlands: Treating non-point source pollution from agriculture runoff. Int. J. Environ. Anal. Chem. 2006, 86, 225–231. [Google Scholar] [CrossRef]
- Maillard, E.; Payraudeau, S.; Ortiz, F.; Imfeld, G. Removal of dissolved pesticide mixtures by a stormwater wetland receiving runoff from a vineyard catchment: An inter-annual comparison. Int. J. Environ. Anal. Chem. 2011, 92, 979–994. [Google Scholar] [CrossRef]
- Turcios, A.E.; Hielscher, M.; Duarte, B.; Fonseca, V.F.; Caçador, I.; Papenbrock, J. Screening of emerging pollutants (Eps) in estuarine water and phytoremediation capacity of tripolium pannonicum under controlled conditions. Int. J. Environ. Res. Public Health 2021, 18, 943. [Google Scholar] [CrossRef] [PubMed]
- Barchanska, H.; Plonka, J.; Jaros, A.; Ostrowska, A. Potential application of Pistia stratiotes for the phytoremediation of mesotrione and its degradation products from water. Int. J. Phytoremed. 2019, 21, 1090–1097. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Xiao, J.; Xiao, H.; Zhang, L.; Wu, Z. Phytoremediation of triazophos by Canna indica Linn. in a hydroponic system. Int. J. Phytoremed. 2007, 9, 453–463. [Google Scholar] [CrossRef] [PubMed]
- McKnight, A.M.; Gannon, T.W.; Yelverton, F. Phytoremediation of azoxystrobin and imidacloprid by wetland plant species Juncus effusus, Pontederia cordata and Sagittaria latifolia. Int. J. Phytoremed. 2021, 1–9. [Google Scholar] [CrossRef]
- Mercado-Borrayo, B.; Heydrich, S.; Perez, I.; Quiroz, M.; Hill, C. Organophosphorus and Organochlorine Pesticides Bioaccumulation by Eichhornia crassipes in Irrigation Canals in an Urban Agricultural System. Int. J. Phytoremed. 2015, 17, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, J.; Thomas, M.; Iuretig, A.; Pallez, C.; Feidt, C.; Dauchy, X.; Banas, D. Barrage fishponds: Reduction of pesticide concentration peaks and associated risk of adverse ecological effects in headwater streams. J. Environ. Manag. 2016, 169, 261–271. [Google Scholar] [CrossRef]
- Gaullier, C.; Dousset, S.; Baran, N.; Kitzinger, G.; Coureau, C. Influence of hydrodynamics on the water pathway and spatial distribution of pesticide and metabolite concentrations in constructed wetlands. J. Environ. Manag. 2020, 270. [Google Scholar] [CrossRef] [PubMed]
- Knuteson, S.; Whitwell, T.; Klaine, S. Influence of plant age and size on simazine toxicity and uptake. J. Environ. Qual. 2002, 31, 2096–2103. [Google Scholar] [CrossRef] [PubMed]
- McMaine, J.T.; Vogel, J.R.; Belden, J.B.; Schnelle, M.A.; Morrison, S.A.; Brown, G.O. Field studies of pollutant removal from nursery and greenhouse runoff by constructed wetlands. J. Environ. Qual. 2020, 49, 106–118. [Google Scholar] [CrossRef]
- Moore, M.T.; Locke, M.A.; Cullum, R.F. Expanding wetland mitigation: Can rice fields remediate pesticides in agricultural runoff? J. Environ. Qual. 2018, 47, 1564–1571. [Google Scholar] [CrossRef]
- Stearman, G.K.; George, D.B.; Carlson, K.; Lansford, S. Pesticide removal from container nursery runoff in constructed wetland cells. J. Environ. Qual. 2003, 32, 1548–1556. [Google Scholar] [CrossRef]
- Sura, S.; Waiser, M.; Tumber, V.; Lawrence, J.; Cessna, A.; Glozier, N. Effects of Glyphosate and Two Herbicide Mixtures on Microbial Communities in Prairie Wetland Ecosystems: A Mesocosm Approach. J. Environ. Qual. 2012, 41, 732–743. [Google Scholar] [CrossRef]
- Casas-Zapata, J.C.; Ríos, K.; Florville-Alejandre, T.R.; Morató, J.; Peñuela, G. Influence of chlorothalonil on the removal of organic matter in horizontal subsurface flow constructed wetlands. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2013, 48, 122–132. [Google Scholar] [CrossRef][Green Version]
- Friesen-Pankratz, B.; Doebel, C.; Farenhorst, A.; Goldsborough, L.G. Interactions between algae (Selenastrum capricornutum) and pesticides: Implications for managing constructed wetlands for pesticide removal. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2003, 38, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Verma, B.; Headley, J.; Robarts, R. Behaviour and fate of tetracycline in river and wetland waters on the Canadian Northern Great Plains. J. Environ. Sci. Health Part A 2007, 42, 109–117. [Google Scholar] [CrossRef]
- Munoz, A.; Trevisan, M.; Capri, E. Sorption and photodegradation of chlorpyrifos on riparian and aquatic macrophytes. J. Environ. Sci. Health Part B 2009, 44, 7–12. [Google Scholar] [CrossRef]
- Moore, M.T.; Locke, M.A.; Kröger, R. Mitigation of atrazine, S-metolachlor, and diazinon using common emergent aquatic vegetation. J. Environ. Sci. 2017, 56, 114–121. [Google Scholar] [CrossRef]
- Matamoros, V.; Rodriguez, Y. Batch vs continuous-feeding operational mode for the removal of pesticides from agricultural run-off by microalgae systems: A laboratory scale study. J. Hazard. Mater. 2016, 309, 126–132. [Google Scholar] [CrossRef]
- Ohore, O.; Zhang, S.; Guo, S.; Manirakiza, B.; Addo, F.; Zhang, W. The fate of tetracycline in vegetated mesocosmic wetlands and its impact on the water quality and epiphytic microbes. J. Hazard. Mater. 2021, 417, 126148. [Google Scholar] [CrossRef]
- Tang, X.; Liu, H.; Naïla, R.S.L.; Dai, Y.; Zhang, X.; Tam, N.F.Y.; Xiong, C.; Yang, Y. Irrigation using hybrid constructed wetland treated domestic sewage: Uptake of phthalic acid esters and antibiotics by Ipomoea aquatica forssk. J. Hazard. Mater. 2021, 405, 124025. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhu, H.; Yan, B.; Xu, Y.; Banuelos, G.; Shutes, B.; Wen, H.; Cheng, R. Removal of chlorpyrifos and its hydrolytic metabolite 3,5,6-trichloro-2-pyridinol in constructed wetland mesocosms under soda saline-alkaline conditions: Effectiveness and influencing factors. J. Hazard. Mater. 2019, 373, 67–74. [Google Scholar] [CrossRef]
- Challis, J.K.; Carlson, J.C.; Friesen, K.J.; Hanson, M.L.; Wong, C.S. Aquatic photochemistry of the sulfonamide antibiotic sulfapyridine. J. Photochem. Photobiol. A Chem. 2013, 262, 14–21. [Google Scholar] [CrossRef]
- Bois, P.; Huguenot, D.; Norini, M.P.; Haque, M.F.U.; Vuilleumier, S.; Lebeau, T. Herbicide degradation and copper complexation by bacterial mixed cultures from a vineyard stormwater basin. J. Soils Sed. 2011, 11, 860–873. [Google Scholar] [CrossRef]
- Moore, M.T.; Kröger, R.; Cooper, C.M.; Cullum, R.F.; Smith, S., Jr.; Locke, M.A. Diazinon reduction and partitioning between water, sediment and vegetation in stormwater runoff mitigation through rice fields. Pest Manag. Sci. 2009, 65, 1182–1188. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, B.; Xu, D.; Zhang, D.; He, F.; Zhou, Q.; Wu, Z. Mitigation of wastewater-borne chlorpyrifos in constructed wetlands: The role of vegetation on partitioning. Pol. J. Environ. Stud. 2017, 26, 347–354. [Google Scholar] [CrossRef]
- Wang, C.; Liu, B.; Xu, D.; Zhang, L.; He, F.; Zhou, Q.; Wu, Z. Mitigation of wastewater-borne chorpyrifos in constructed wetlands: An ecological suitability assessment by macrophyte and microbial responses. Pol. J. Environ. Stud. 2017, 26, 1279–1287. [Google Scholar] [CrossRef]
- Topal, M.; Obek, E.; Senel, G.; Topal, E. Phytoremediation of Tetracycline and Degradation Products from Aqueous Solutions. Pollution 2018, 4, 471–480. [Google Scholar] [CrossRef]
- De Souza, T.D.; Borges, A.C.; de Matos, A.T.; Mounteer, A.H.; de Queiroz, M.E.L.R. Removal of chlorpyrifos insecticide in constructed wetlands with different plant species. Rev. Bras. Eng. Agric. Ambient. 2017, 21, 878–883. [Google Scholar] [CrossRef]
- Sha, N.-Q.; Wang, G.-H.; Li, Y.-H.; Bai, S.-Y. Removal of abamectin and conventional pollutants in vertical flow constructed wetlands with Fe-modified biochar. RSC Adv. 2020, 10, 44171–44182. [Google Scholar] [CrossRef]
- Agudelo, C.R.M.; Jaramillo, M.L.; Peñuela, G. Comparison of the removal of chlorpyrifos and dissolved organic carbon in horizontal sub-surface and surface flow wetlands. Sci. Total Environ. 2012, 431, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Butkovskyi, A.; Jing, Y.; Bergheim, H.; Lazar, D.; Gulyaeva, K.; Odenmarck, S.R.; Norli, H.R.; Nowak, K.M.; Miltner, A.; Kästner, M.; et al. Retention and distribution of pesticides in planted filter microcosms designed for treatment of agricultural surface runoff. Sci. Total Environ. 2021, 778, 146114. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, Y.; Vymazal, J.; Kule, L.; Koželuh, M. Dynamics of chloroacetanilide herbicides in various types of mesocosm wetlands. Sci. Total Environ. 2017, 577, 386–394. [Google Scholar] [CrossRef]
- Elsayed, O.F.; Maillard, E.; Vuilleumier, S.; Imfeld, G. Bacterial communities in batch and continuous-flow wetlands treating the herbicide S-metolachlor. Sci. Total Environ. 2014, 499, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Lizotte, R.; Shields, F.; Murdock, J.; Kroger, R.; Knight, S. Mitigating agrichemicals from an artificial runoff event using a managed riverine wetland. Sci. Total Environ. 2012, 427, 427–428. [Google Scholar] [CrossRef] [PubMed]
- Lobson, C.; Luong, K.; Seburn, D.; White, M.; Hann, B.; Prosser, R.; Wong, C.; Hanson, M. Fate of thiamethoxam in mesocosms and response of the zooplankton community. Sci. Total Environ. 2018, 637, 637–638. [Google Scholar] [CrossRef] [PubMed]
- Maillard, E.; Payraudeau, S.; Faivre, E.; Gregoire, C.; Gangloff, S.; Imfeld, G. Removal of pesticide mixtures in a stormwater wetland collecting runoff from a vineyard catchment. Sci. Total Environ. 2011, 409, 2317–2324. [Google Scholar] [CrossRef] [PubMed]
- Main, A.; Fehr, J.; Liber, K.; Headley, J.; Peru, K.; Morrissey, C. Reduction of neonicotinoid insecticide residues in Prairie wetlands by common wetland plants. Sci. Total Environ. 2017, 579, 1193–1202. [Google Scholar] [CrossRef]
- Sahin, C.; Karpuzcu, M.E. Mitigation of organophosphate pesticide pollution in agricultural watersheds. Sci. Total Environ. 2020, 710, 136261. [Google Scholar] [CrossRef]
- Solis, M.; Cappelletti, N.; Bonetto, C.; Franco, M.; Fanelli, S.; Amalvy, J.; Mugni, H. Attenuation of insecticide impact by a small wetland in a stream draining a horticultural basin in Argentina. Sci. Total Environ. 2021, 785, 147317. [Google Scholar] [CrossRef]
- Sura, S.; Waiser, M.; Tumber, V.; Farenhorst, A. Effects of herbicide mixture on microbial communities in prairie wetland ecosystems: A whole wetland approach. Sci. Total Environ. 2012, 435, 435–436. [Google Scholar] [CrossRef]
- Sura, S.; Waiser, M.; Tumber, V.; Raina-Fulton, R.; Cessna, A. Effects of a herbicide mixture on primary and bacterial productivity in four prairie wetlands with varying salinities: An enclosure approach. Sci. Total Environ. 2015, 512, 512–513. [Google Scholar] [CrossRef]
- Tang, X.; Yang, Y.; Tao, R.; Chen, P.; Dai, Y.; Jin, C.; Feng, X. Fate of mixed pesticides in an integrated recirculating constructed wetland (IRCW). Sci. Total Environ. 2016, 571, 935–942. [Google Scholar] [CrossRef]
- Wu, J.; Feng, Y.; Dai, Y.; Cui, N.; Anderson, B.; Cheng, S. Biological mechanisms associated with triazophos (TAP) removal by horizontal subsurface flow constructed wetlands (HSFCW). Sci. Total Environ. 2016, 553, 13–19. [Google Scholar] [CrossRef]
- Zhu, H.; Yu, X.; Xu, Y.; Yan, B.; Banuelos, G.; Shutes, B.; Wen, Z. Removal of chlorpyrifos and its hydrolytic metabolite in microcosm-scale constructed wetlands under soda saline-alkaline condition: Mass balance and intensification strategies. Sci. Total Environ. 2021, 777, 145956. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, Z.; Wu, L.; Zhong, F.; Cui, N.; Dai, Y.; Cheng, S. Triazophos (TAP) removal in horizontal subsurface flow constructed wetlands (HSCWs) and its accumulation in plants and substrates. Sci. Rep. 2017, 7, 5468. [Google Scholar] [CrossRef]
- D’Angelo, E.; Reddy, K. Effect of aerobic and anaerobic conditions on chlorophenol sorption in wetland soils. Soil Sci. Soc. Am. J. 2003, 67, 787–794. [Google Scholar] [CrossRef]
- Passeport, E.; Tournebize, J.; Jankowfsky, S.; Prömse, B.; Chaumont, C.; Coquet, Y.; Lange, J. Artificial wetland and forest buffer zone: Hydraulic and tracer characterization. Vadose Zone J. 2010, 9, 73–84. [Google Scholar] [CrossRef]
- Lizotte, R.; Shields, F.; Testa, S. Effects of a Simulated Agricultural Runoff Event on Sediment Toxicity in a Managed Backwater Wetland. Water Air Soil Pollut. 2012, 223, 5375–5389. [Google Scholar] [CrossRef]
- Mauffrey, F.; Baccara, P.; Gruffaz, C.; Vuilleumier, S.; Imfeld, G. Bacterial Community Composition and Genes for Herbicide Degradation in a Stormwater Wetland Collecting Herbicide Runoff. Water Air Soil Pollut. 2017, 228, 452. [Google Scholar] [CrossRef]
- Bois, P.; Huguenot, D.; Jezequel, K.; Lollier, M.; Cornu, J.; Lebeau, T. Herbicide mitigation in microcosms simulating stormwater basins subject to polluted water inputs. Water Res. 2013, 47, 1123–1135. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Carvalho, P.N.; Zhang, L.; Zhang, Y.; Button, M.; Arias, C.A.; Weber, K.P.; Brix, H. Functionality of microbial communities in constructed wetlands used for pesticide remediation: Influence of system design and sampling strategy. Water Res. 2017, 110, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Lv, T.; Zhang, Y.; Zhang, L.; Carvalho, P.N.; Arias, C.A.; Brix, H. Removal of the pesticides imazalil and tebuconazole in saturated constructed wetland mesocosms. Water Res. 2016, 91, 126–136. [Google Scholar] [CrossRef]
- McKinlay, R.G.; Kasperek, K. Observations on decontamination of herbicide-polluted water by marsh plant systems. Water Res. 1999, 33, 505–511. [Google Scholar] [CrossRef]
- Borges, A.C.; Do Carmo Calijuri, M.; De Matos, A.T.; De Queiroz, M.E.L.R. Horizontal subsurface flow constructed wetlands for mitigation of ametryn-contaminated water. Water SA 2009, 35, 441–446. [Google Scholar] [CrossRef]
- Braskerud, B.C.; Haarstad, K. Screening the retention of thirteen pesticides in a small constructed wetland. Water Sci. Technol. 2003, 48, 267–274. [Google Scholar] [CrossRef]
- Haarstad, K.; Braskerud, B.C. Pesticide retention in the watershed and in a small constructed wetland treating diffuse pollution. Water Sci. Technol. 2005, 51, 143–150. [Google Scholar] [CrossRef][Green Version]
- Roseth, R.; Haarstad, K. Pesticide runoff from greenhouse production. Water Sci. Technol. 2010, 61, 1373–1381. [Google Scholar] [CrossRef]
- Sudo, M.; Nishino, M.; Okubo, T. Decrease in herbicide concentrations and affected factors in lagoons located around Lake Biwa. Water Sci. Technol. 2006, 53, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Lizotte, R.E., Jr.; Moore, M.T.; Locke, M.A.; Kröger, R. Effects of vegetation in mitigating the toxicity of pesticide mixtures in sediments of a wetland mesocosm. Water Air Soil Pollut. 2011, 220, 69–79. [Google Scholar] [CrossRef]
- Mahabali, S.; Spanoghe, P. Mitigation of two insecticides by wetland plants: Feasibility study for the treatment of agricultural runoff in suriname (South America). Water Air Soil Pollut. 2014, 225, 1771. [Google Scholar] [CrossRef]
- Moore, M.T.; Cooper, C.M.; Smith, S., Jr.; Cullum, R.F.; Knight, S.S.; Locke, M.A.; Bennett, E.R. Diazinon mitigation in constructed wetlands: Influence of vegetation. Water Air Soil Pollut. 2007, 184, 313–321. [Google Scholar] [CrossRef]
- Rose, M.T.; Crossan, A.N.; Kennedy, I.R. Dissipation of cotton pesticides from runoff water in glasshouse columns. Water Air Soil Pollut. 2007, 182, 207–218. [Google Scholar] [CrossRef]
- Birch, G.F.; Matthai, C.; Fazeli, M.S.; Suh, J. Efficiency of a constructed wetland in removing contaminants from stormwater. Wetlands 2004, 24, 459–466. [Google Scholar] [CrossRef]
- Henry, B.L.; Wesner, J.S.; Kerby, J.L. Cross-Ecosystem Effects of Agricultural Tile Drainage, Surface Runoff, and Selenium in the Prairie Pothole Region. Wetlands 2020, 40, 527–538. [Google Scholar] [CrossRef]
- Hussain, S.A.; Prasher, S.O. Understanding the sorption of ionophoric pharmaceuticals in a treatment wetland. Wetlands 2011, 31, 563–571. [Google Scholar] [CrossRef]
- Shan, A.; Wang, W.; Kang, K.J.; Hou, D.; Luo, J.; Wang, G.; Pan, M.; Feng, Y.; He, Z.; Yang, X. The Removal of Antibiotics in Relation to a Microbial Community in an Integrated Constructed Wetland for Tail Water Decontamination. Wetlands 2020, 40, 993–1004. [Google Scholar] [CrossRef]
- Schulz, R.; Moore, M.T.; Bennett, E.R.; Farris, J.L.; Smith, S., Jr.; Cooper, C.M. Methyl parathion toxicity in vegetated and nonvegetated wetland mesocosms. Environ. Toxicol. Chem. 2003, 22, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Berberidou, C.; Kitsiou, V.; Lambropoulou, D.A.; Antoniadis, A.; Ntonou, E.; Zalidis, G.C.; Poulios, I. Evaluation of an alternative method for wastewater treatment containing pesticides using solar photocatalytic oxidation and constructed wetlands. J. Environ. Manag. 2017, 195, 133–139. [Google Scholar] [CrossRef]
- Donley, N. The USA lags behind other agricultural nations in banning harmful pesticides. Environ. Health 2019, 18, 44. [Google Scholar] [CrossRef]
- Survey, U.S.G. Estimated Annual Agricultural Pesticide Use. Available online: https://water.usgs.gov/nawqa/pnsp/usage/maps/compound_listing.php (accessed on 24 September 2021).
- Ahn, C.; Mitsch, W.J. Scaling considerations of mesocosm wetlands in simulating large created freshwater marshes. Ecol. Eng. 2020, 18, 327–342. [Google Scholar] [CrossRef]
- Nally, R.M. Scaling artefacts in confinement experiments: A simulation model. Ecol. Model. 1997, 99, 229–245. [Google Scholar] [CrossRef]
- Pilson, M.; Nixon, S. Marine Microcosms in Ecological Research. In Department of Energy Symposium Series; U.S. Environmental Protection Agency: Washington, DC, USA, 1980; EPA/600/D-82/218 (NTIS PB82181900). [Google Scholar]
- Messer, T.; Burchell, M.; Bírgand, F. Comparison of Four Nitrate Removal Kinetic Models in Two Distinct Wetland Restoration Mesocosm Systems. Water 2017, 9, 517. [Google Scholar] [CrossRef]
- Parde, D.; Patwa, A.; Shukla, A.; Vijay, R.; Killedar, D.J.; Kumar, R. A review of constructed wetland on type, treatment and technology of wastewater. Environ. Technol. Innov. 2021, 21, 101261. [Google Scholar] [CrossRef]
- Vymazal, J. Constructed Wetlands for Wastewater Treatment. Water 2010, 2, 530–549. [Google Scholar] [CrossRef]
- Lawler, S. Rice fields as temporary wetlands. Israel J. Zool. 2001, 47, 513–528. [Google Scholar] [CrossRef]
- Agency, E.P. Atrazine. Available online: Epa.gov/ingredients-used-pesticide-products/atrazine (accessed on 12 October 2021).
- Wolf, K.L.; Ahn, C.; Noe, G.B. Development of soil properties and nitrogen cycling in created wetlands. Wetlands 2011, 31, 699–712. [Google Scholar] [CrossRef]
- Bruland, G.L.; Richardson, C.J. Spatial variability of soil properties in created, restored, and paired natural wetlands. Soil Sci. Soc. Am. J. 2005, 69, 273–284. [Google Scholar] [CrossRef]
- Weber, K. Microbial Community Assessment in Wetlands for Water Pollution Control: Past, Present, and Future Outlook. Water 2016, 8, 503. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).