Non-Equilibrium Uranium as an Indicator of Global Climate Variations—The World Ocean and Large Lakes
Abstract
1. Introduction
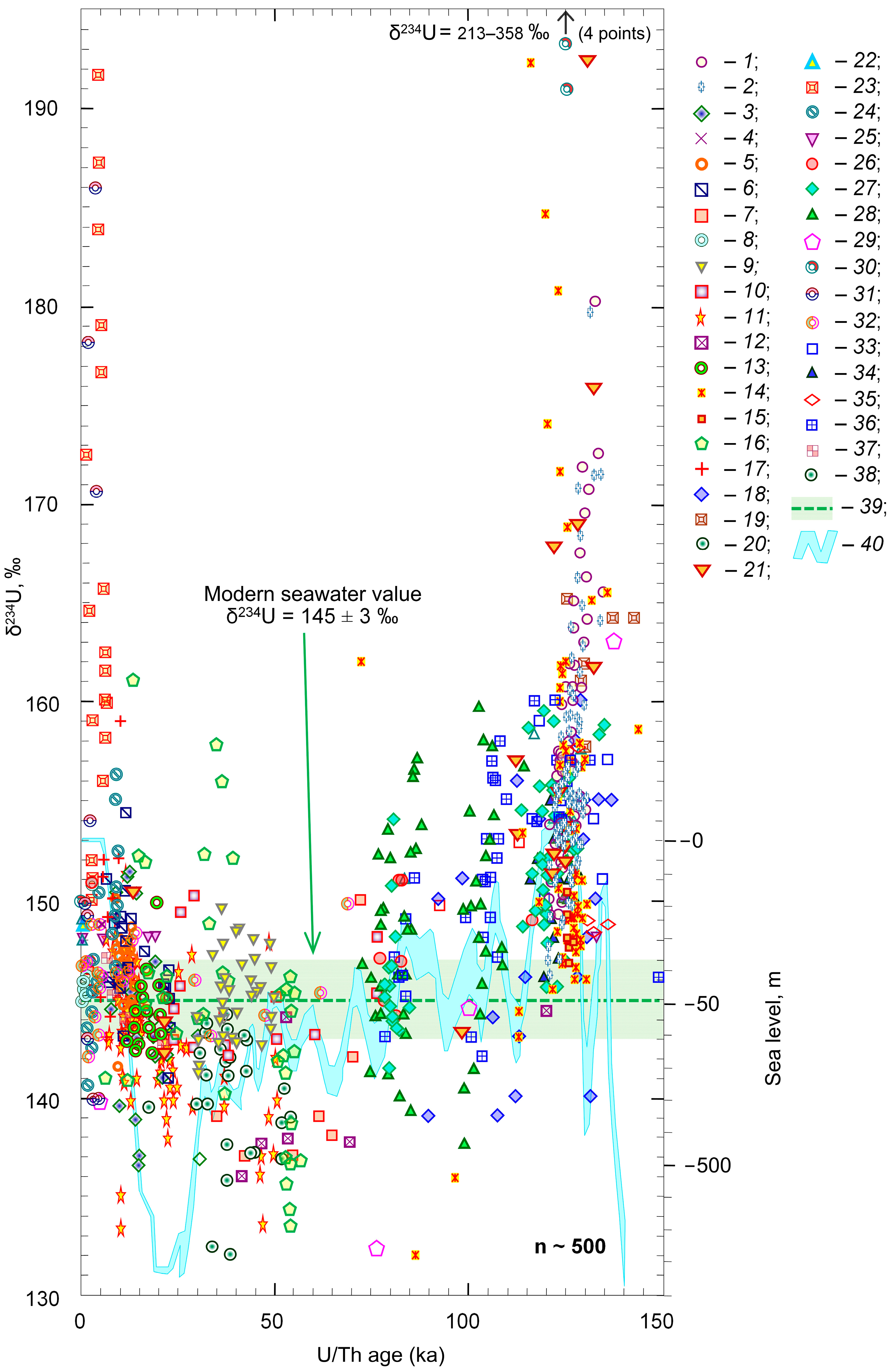
2. World Ocean
- Why is the loss of 234Th recoil nuclei by fine-grained particles small at the continental stage of transport? In the zone of finely dispersed rocks on the continents, Yedoma (Arctic), loess, and black earth (middle latitudes), whose origin is associated with the wind spread of dusty material during the glacial epochs, a significant violation of the radioactive equilibrium in the 238U chain is not observed in surface and shallow groundwater.
- Suppose that the increase in the amount of 234Th recoil nuclei is, in fact, due to an increase in the supply of dust to the ocean during cooling periods [33,34]. However, both dust and thorium in the chemical compounds have a very short average residence time in ocean water [35,36,37] and are quickly deposited in bottom sediments. What mechanism ensures the reverse flow of uranium from the pore water of sediments into ocean water? Oxidizing conditions favoring the migration of uranium in water-soluble forms are observed only in the thin surface layer of bottom sediments. The presence of organic material, for example, in the form of microbial mats and/or a stream of dead organisms, transforms the environmental conditions into an area of chemical reduction. The diffuse transfer of uranium from pore water into free water is hampered by the accumulation of new portions of sediments, with the exception of the oceanic abyssal, where the accumulation is greatly slowed down. In some cases, when an excess of uranium 234U (234U/238U > 1.14) is observed in the pore water, occasionally, none is recorded for the abyssal [10,13,14,15].
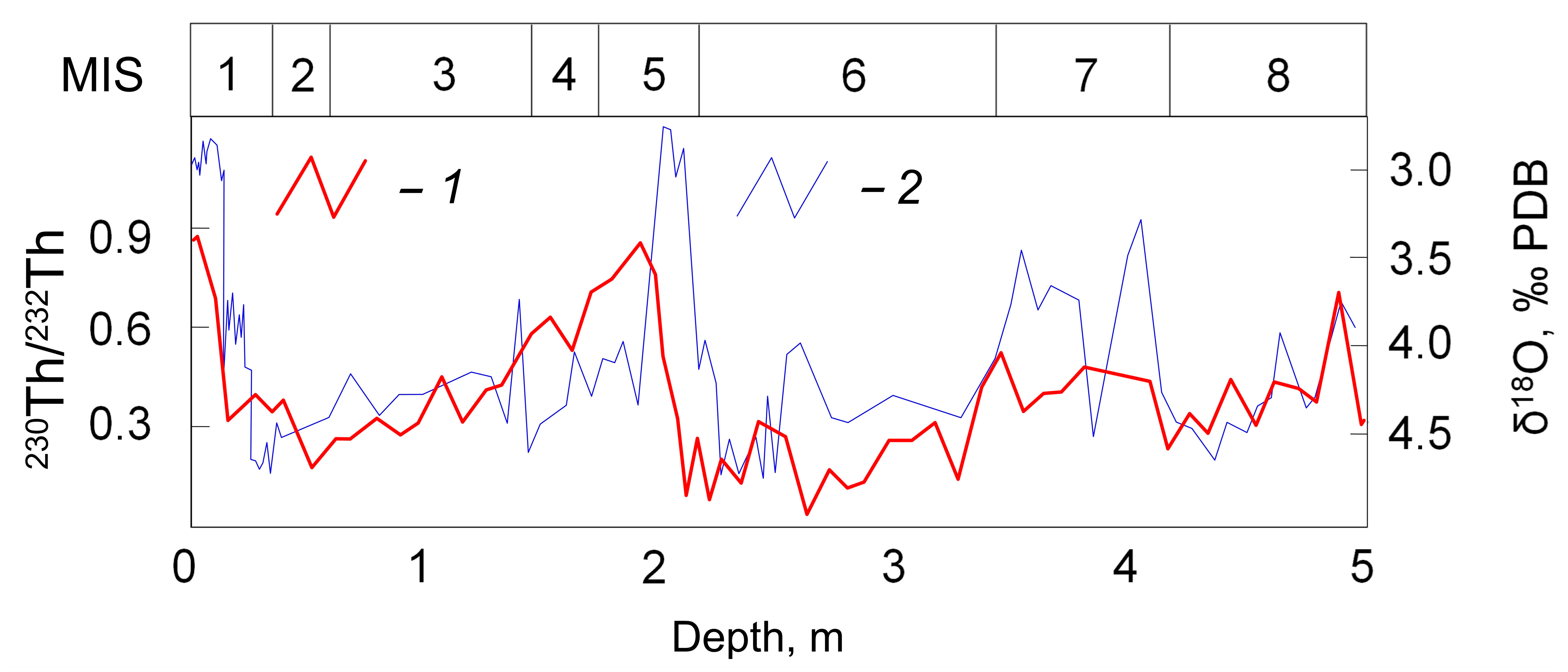
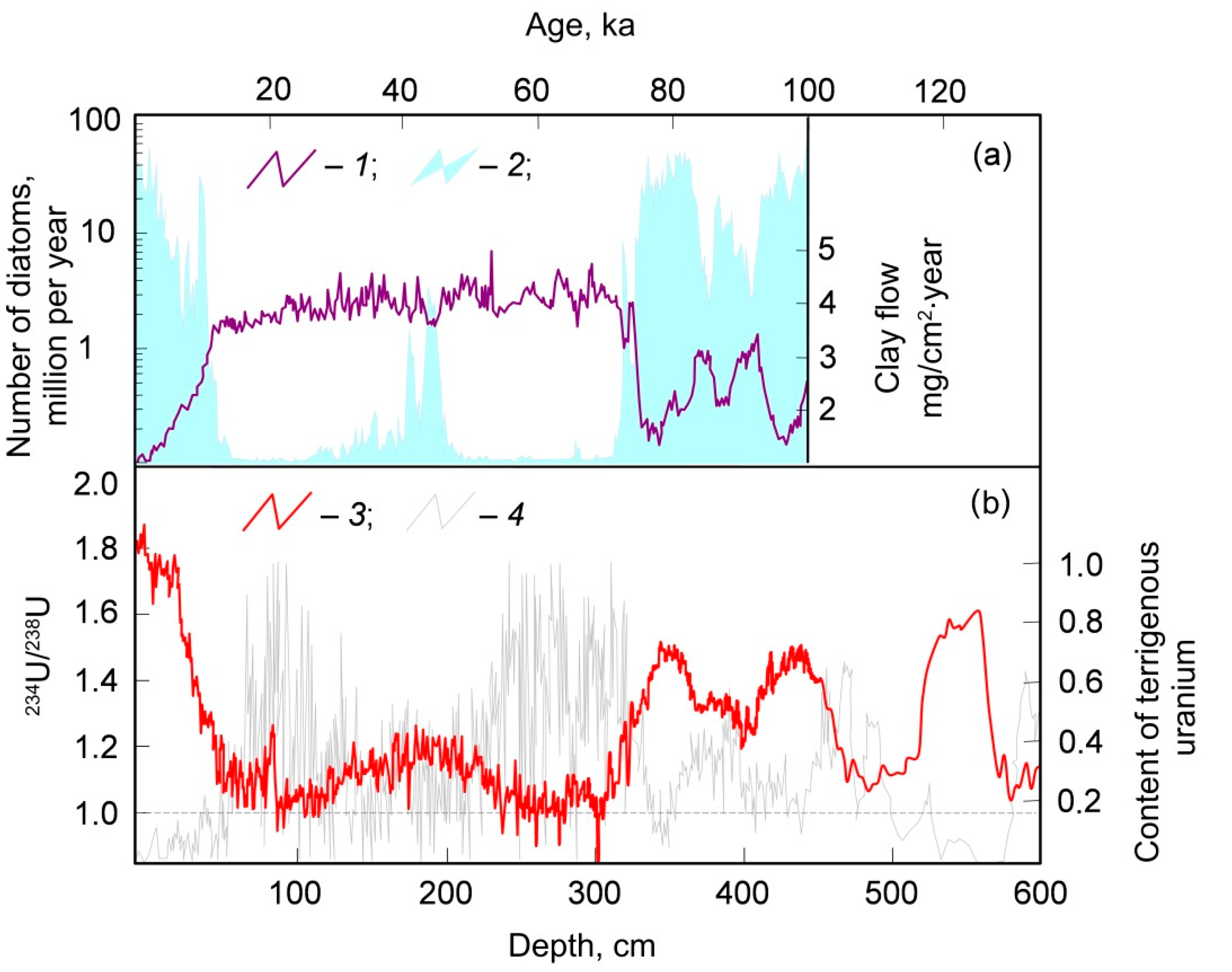
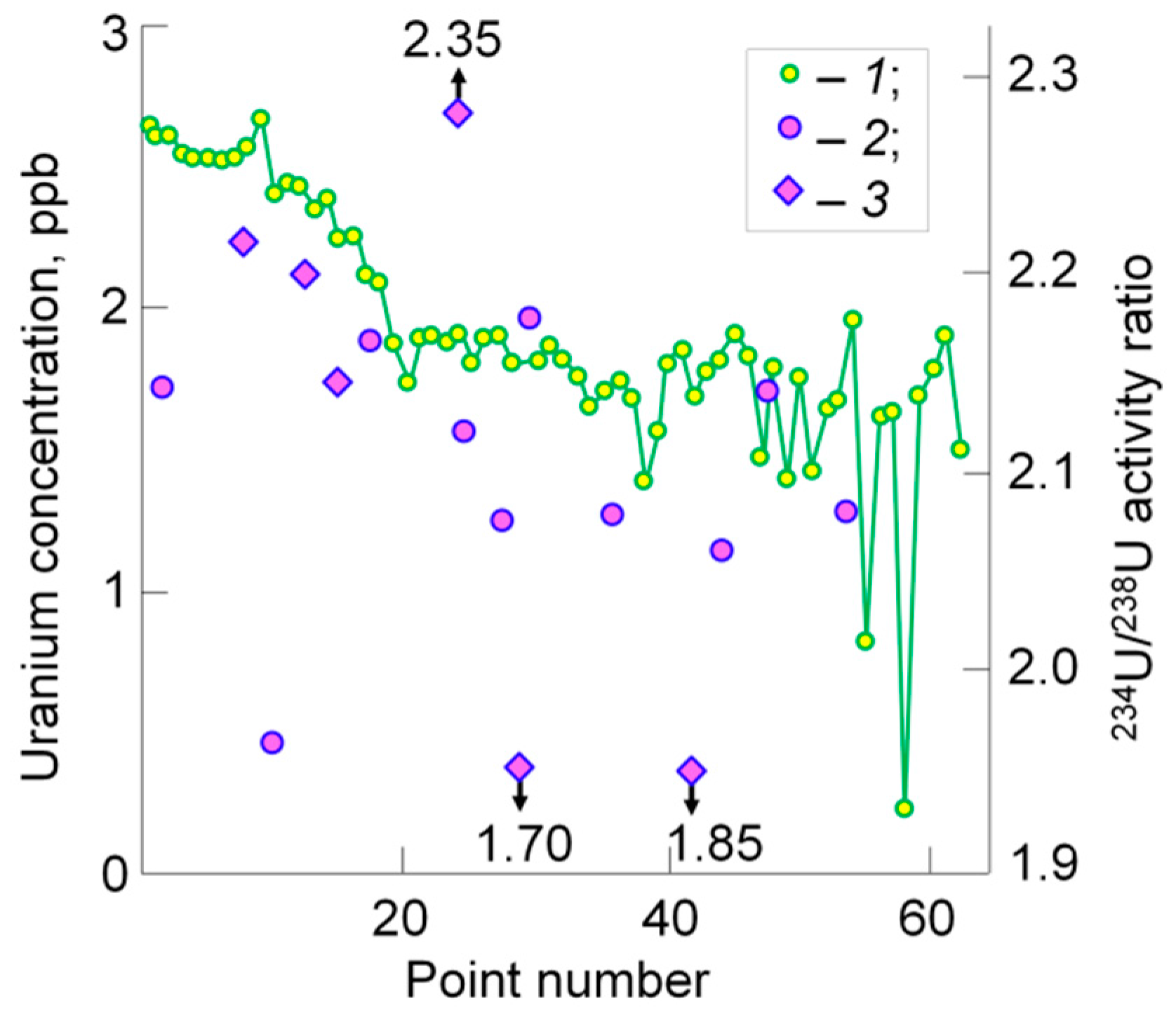
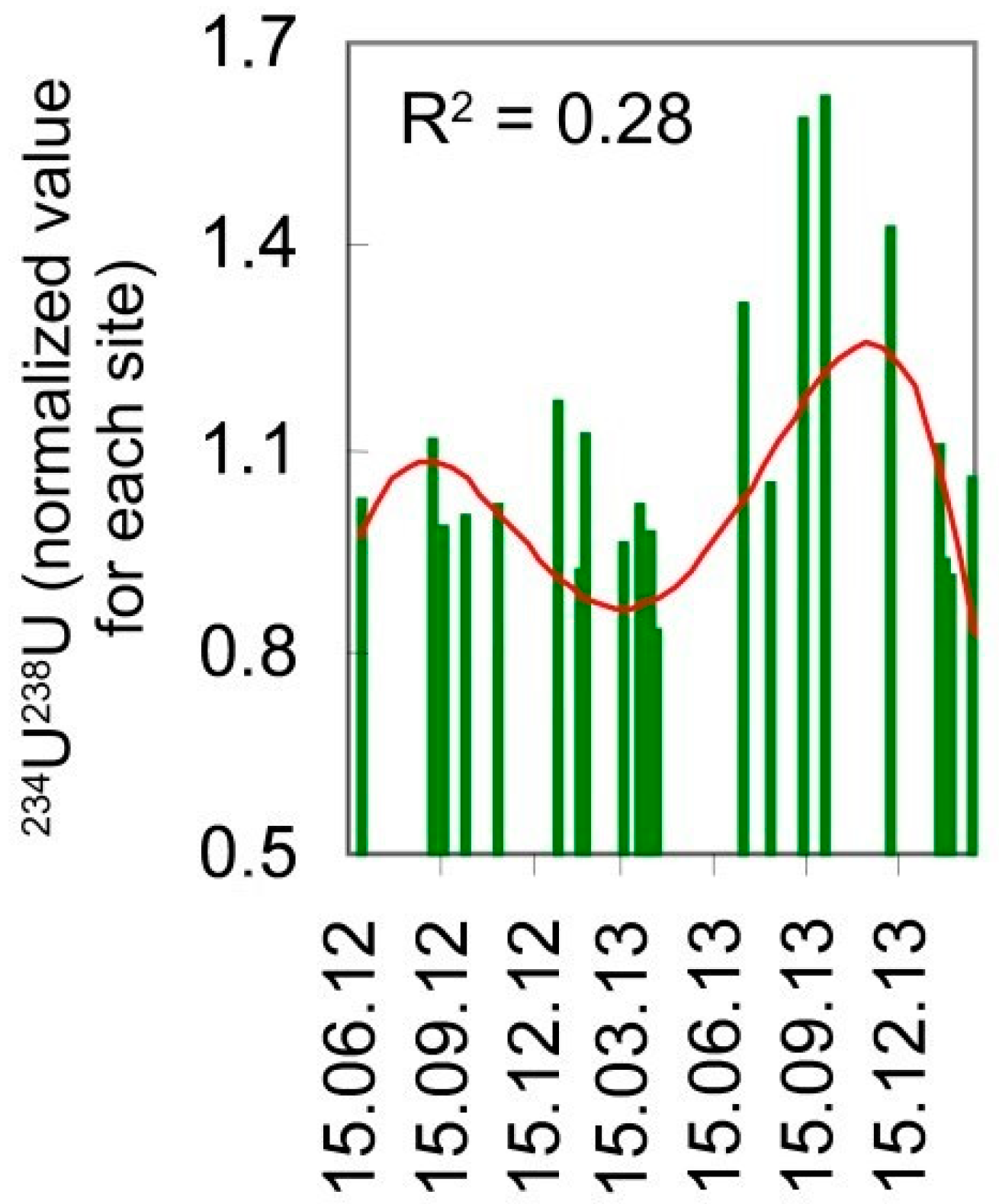
3. Continental Freshwater Bodies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chalov, P.I. Study of U(II)/U(I) Ratios in Some Natural Objects. Ph.D. Thesis, Academy of Sciences of the KazakhSSR, Alma-Ata, USSR, 1954. [Google Scholar]
- Chalov, P.I. U234/U238 isotopic ratio in some secondary minerals. Geochemistry 1959, 2, 165–170. [Google Scholar]
- Chalov, P.I. Isotopic Fractionation of Natural Uranium; Ilim: Frunze, Russia, 1975. [Google Scholar]
- Cherdyncev, V.V. Uran-234; Atomizdat: Moscow, Russia, 1967. [Google Scholar]
- Cherdyntsev, V.V. Isotopic Composition of Radioelements in Natural Objects and Their Importance in Geochronology; Institute of Precambrian Geology and Geochronology: Moscow, Russia, 1955. [Google Scholar]
- Polyakov, V.A. Study of Changes in Hydrochemistry and Groundwater Resources of Coastal Water Intakes in Estonia from Isotope Data; GIDEK: Zvenigorod, Russia, 1991. [Google Scholar]
- Arndt, M.F.; West, L. A Study of the Factors Affecting the Gross Alpha Measurement, and a Radiochemical Analysis of Some Groundwater Samples from the State of Wisconsin Exhibiting an Elevated Gross Alpha Activity. Wisconsin Groundwater Management Practice Monitoring Project, DNR-176. 2004. Available online: https://wri.wisc.edu/wp-content/uploads/FinalWR02R009.pdf (accessed on 1 November 2021).
- Ku, T.-L. An evaluation of the U234/U238method as a tool for dating pelagic sediments. J. Geophys. Res. Space Phys. 1965, 70, 3457–3474. [Google Scholar] [CrossRef]
- Ku, T.-L.; Mathieu, G.G.; Knauss, K. Uranium in Open Ocean: Concentration and isotopic composition. Deep. Sea Res. 1977, 24, 1005–1017. [Google Scholar] [CrossRef]
- Cochran, J.K.; Krishnaswami, S. Radium, thorium, uranium, and 210 Pb in deep-sea sediments and sediment pore waters from the North Equatorial Pacific. Am. J. Sci. 1980, 280, 849–889. [Google Scholar] [CrossRef]
- Ivanovich, M.; Harmon, R.S. Uranium-Series Disequilibrium; Clarendon Press: Oxford, UK, 1992. [Google Scholar]
- Russell, A.D.; Edwards, R.L.; Hoff, J.A.; McCorkle, D.; Sayles, F. Sediment source of 234U suggested by d234U in North Pacific pore waters. Fall Meet. Suppl. EOS 1994, 75, 332. [Google Scholar]
- Russell, A.D.; Emerson, S.; Nelson, B.K.; Erez, J.; Lea, D.W. Uranium in foraminiferal calcite as a recorder of seawater uranium concentrations. Geochim. Cosmochim. Acta 1994, 58, 671–681. [Google Scholar] [CrossRef]
- Henderson, G.M.; Burton, K.W. Using (234U/238U) to assess diffusion rates of isotope tracers in ferromanganese crusts. Earth Planet. Sci. Lett. 1999, 170, 169–179. [Google Scholar] [CrossRef]
- Henderson, G.M.; Slowey, N.C.; Haddad, G.A. Fluid flow through carbonate platforms: Constraints from 234U/238U and Cl− in Bahamas pore-waters. Earth Planet. Sci. Lett. 1999, 169, 99–111. [Google Scholar] [CrossRef]
- Calsteren, V.; Thomas, L. Uranium-series dating applications in natural environmental science. Earth-Sci. Rev. 2006, 75, 155–175. [Google Scholar] [CrossRef]
- Tokarev, I.V. Using isotope data (δ2H, δ18O, 234U/238U) in studying the processes of permafrost degradation as a result of long-term climate variations. J. Min. Inst. 2008, 176, 191–195. [Google Scholar]
- Tokarev, I.V. Non-equilibrium uranium (234U/238U) of water bodies and climatic variations. 1. Oceanic reservoir. Geochem. Int. 2021, in press. [Google Scholar]
- Tokarev, I.V.; Zubkov, A.A.; Rumynin, V.G.; Polyakov, V.A.; Kuznetsov VYu Maksimov, F.E. Origin of high 234U/238U ratio in post-permafrost aquifers. In Uranium in the Environment (Mining Impact and Consequences); Merkel, B.J., Hasche-Berger, A., Eds.; Taylor & Francis: London, UK, 2006; pp. 847–856. [Google Scholar]
- Tokarev, I.; Zubkov, A.A.; Rumynin, V.G.; Pozdnyakov, S.P.; Polyakov, V.A.; Kuznetsov, V.Y. Assessment of the long-term safety of radioactive waste disposal: 1. Paleoreconstruction of groundwater formation conditions. Water Resour. 2009, 36, 206–213. [Google Scholar] [CrossRef]
- Tokarev, I.; Zubkov, A.A.; Rumynin, V.G.; Pozdnyakov, S.P. Assessment of the long-term safety of radioactive waste disposal: 2. Isotopic study of water exchange in a multilayer system. Water Resour. 2009, 36, 345–356. [Google Scholar] [CrossRef]
- Chen, J.H.; Edwards, R.L.; Wasserburg, G.J. 238U, 234U and 232Th in seawater. Earth Planet. Sci. Lett. 1986, 80, 241–251. [Google Scholar] [CrossRef]
- Henderson, G.M.; Anderson, R.F. The U-series toolbox for paleoceanography. Rev. Mineral. Geochem. 2003, 52, 493–532. [Google Scholar] [CrossRef]
- Chabaux, F.; Riotte, J.; Dequincey, O. U-Th-Ra Fractionation during Weathering and River Transport. Rev. Miner. Geochem. 2003, 52, 533–576. [Google Scholar] [CrossRef]
- Dunk, R.; Mills, R.; Jenkins, W. A reevaluation of the oceanic uranium budget for the Holocene. Chem. Geol. 2002, 190, 45–67. [Google Scholar] [CrossRef]
- Cheng, H.; Adkins, J.; Edwards, R.; Boyle, E.A. U-Th dating of deep-sea corals. Geochim. Cosmochim. Acta 2000, 64, 2401–2416. [Google Scholar] [CrossRef]
- Robinson, L.F.; Belshaw, N.S.; Henderson, G.M. U and Th isotopes in seawater and modern carbonates from the Bahamas. Geochim. Cosmochim. Acta 2004, 68, 1777–1789. [Google Scholar] [CrossRef]
- Henderson, G.M. Seawater (234U/238U) during the last 800 thousand years. Earth Planet. Sci. Lett. 2002, 199, 97–110. [Google Scholar] [CrossRef]
- Kronfeld, J.; Vogel, J. Uranium isotopes in surface waters from southern Africa. Earth Planet. Sci. Lett. 1991, 105, 191–195. [Google Scholar] [CrossRef]
- Kronfeld, J. Uranium deposition and Th-234 alpha-recoil: An explanation for extreme U-234/U-238 fractionation within the trinity aquifer. Earth Planet. Sci. Lett. 1974, 21, 327–330. [Google Scholar] [CrossRef]
- Kronfeld, J.; Gradsztajn, E.; Müller, H.; Radin, J.; Yaniv, A.; Zach, R. Excess 234U: An aging effect in confined waters. Earth Planet. Sci. Lett. 1975, 27, 342–345. [Google Scholar] [CrossRef]
- Esat, T.M.; Yokoyama, Y. Correlated Uranium and Sea-Level Fluctuations in Late Quaternary Oceans. J. Goldschmidt Conf. Abstr. 2000, 5, 387–388. [Google Scholar]
- Muhs, D.R. The geologic records of dust in the Quaternary. Aeolian Res. 2013, 9, 3–48. [Google Scholar] [CrossRef]
- Rea, D.K. The paleoclimatic record provided by eolian deposition in the deep sea: The geologic history of wind. Rev. Geophys. 1994, 32, 159–195. [Google Scholar] [CrossRef]
- Anderson, R.F.; Cheng, H.; Edwards, R.L.; Fleisher, M.Q.; Hayes, C.T.; Huang, K.-F.; Kadko, D.; Lam, P.J.; Landing, W.M.; Lao, Y.; et al. How well can we quantify dust deposition to the ocean? Phil. Trans. R. Soc. A 2016, 374, 20150285. [Google Scholar] [CrossRef] [PubMed]
- Broecker, W.; Kaufman, A.; Trier, R. The residence time of thorium in surface sea water and its implications regarding the rate of reactive pollutants. Earth Planet. Sci. Lett. 1973, 20, 35–44. [Google Scholar] [CrossRef]
- Moore, W.S. The thorium isotope content of ocean water. Earth Planet. Sci. Lett. 1981, 53, 419–426. [Google Scholar] [CrossRef]
- Bar-Matthews, M.; Wasserburg, G.; Chen, J. Diagenesis of fossil coral skeletons: Correlation between trace elements, textures, and 234U238U. Geochim. Cosmochim. Acta 1993, 57, 257–276. [Google Scholar] [CrossRef]
- Thompson, W.G.; Curran, H.A.; Wilson, M.A.; White, B. Sea-level oscillations during the last interglacial highstand recorded by Bahamas corals. Nat. Geosci. 2011, 4, 684–687. [Google Scholar] [CrossRef]
- Bard, E.; Hamelin, B.; Fairbanks, R.G. U-Th ages obtained by mass spectrometry in corals from Barbados: Sea level during the past 130,000 years. Nature 1990, 346, 456–458. [Google Scholar] [CrossRef]
- Bard, E.; Fairbanks, R.G.; Hamelin, B.; Zindler, A.; Hoang, C.T. Uranium-234 anomalies in corals older than 150,000 years. Geochim. Cosmochim. Acta 1991, 55, 2385–2390. [Google Scholar] [CrossRef]
- Bard, E.; Hamelin, B.; Arnold, M.; Montaggioni, L.; Cabioch, G.; Faure, G.; Rougerie, F. Deglacial sea-level record from Tahiti corals and the timing of global meltwater discharge. Nature 1996, 382, 241–244. [Google Scholar] [CrossRef]
- Cabioch, G.; Ayliffe, L.K. Raised Coral Terraces at Malakula, Vanuatu, Southwest Pacific, Indicate High Sea Level During Marine Isotope Stage 3. Quat. Res. 2001, 56, 357–365. [Google Scholar] [CrossRef]
- Chappell, J.; Omura, A.; Esat, T.; McCulloch, M.; Pandolfi, J.; Ota, Y.; Pillans, B. Reconcilitation of late Quaternary sea levels derived from coral terraces at Huon Peninsula with deep sea oxygene isotope records. Earth Planet. Sci. Lett. 1996, 141, 227–236. [Google Scholar] [CrossRef]
- Copard, K.; Colin, C.; Henderson, G.; Scholten, J.; Douville, E.; Sicre, M.-A.; Frank, N. Late Holocene intermediate water variability in the northeastern Atlantic as recorded by deep-sea corals. Earth Planet. Sci. Lett. 2012, 313-314, 34–44. [Google Scholar] [CrossRef]
- Chiu, T.; Fairbanks, R.G.; Mortlock, R.A.; Bloom, A.L. Extending the radiocarbon calibration beyond 26,000 years before present using fossil corals. Quat. Sci. Rev. 2005, 24, 1797–1808. [Google Scholar] [CrossRef][Green Version]
- Cutler, K.; Edwards, R.; Taylor, F.; Cheng, H.; Adkins, J.; Gallup, C.; Cutler, P.; Burr, G.; Bloom, A. Rapid sea-level fall and deep-ocean temperature change since the last interglacial period. Earth Planet. Sci. Lett. 2003, 206, 253–271. [Google Scholar] [CrossRef]
- Cutler, K.B.; Gray, S.C.; Burr, G.S.; Edwards, R.L.; Taylor, F.W.; Cabioch, G.; Beck, J.W.; Cheng, H.; Moore, J. Radiocarbon Calibration and Comparison to 50 Kyr BP with Paired 14C and 230Th Dating of Corals from Vanuatu and Papua New Guinea. Radiocarbon 2004, 46, 1127–1160. [Google Scholar] [CrossRef]
- Dia, A.N.; Cohen, A.S.; O’Nions, R.K.; Shackleton, N.J. Seawater Sr isotope variation over the past 300 kyr and influence of global climate cycles. Nature 1992, 356, 786–788. [Google Scholar] [CrossRef]
- Douarin, M.; Elliot, M.; Noble, S.R.; Sinclair, D.; Henry, L.-A.; Long, D.; Moreton, S.G.; Roberts, J.M. Growth of north-east Atlantic cold-water coral reefs and mounds during the Holocene: A high resolution U-series and 14C chronology. Earth Planet. Sci. Lett. 2013, 375, 176–187. [Google Scholar] [CrossRef]
- Dutton, A.; Webster, J.M.; Zwartz, D.; Lambeck, K.; Wohlfarth, B. Tropical tales of polar ice: Evidence of Last Interglacial polar ice sheet retreat recorded by fossil reefs of the granitic Seychelles islands. Quat. Sci. Rev. 2015, 107, 182–196. [Google Scholar] [CrossRef]
- Dutton, A.; Rubin, K.; McLean, N.; Bowring, J.; Bard, E.; Edwards, R.; Henderson, G.; Reid, M.; Richards, D.; Sims, K.; et al. Data reporting standards for publication of U-series data for geochronology and timescale assessment in the earth sciences. Quat. Geochronol. 2017, 39, 142–149. [Google Scholar] [CrossRef]
- Eisele, M.H. The Long-Term Development of Cold-Water Coral Mounds in the NE-Atlantic. Ph.D. Thesis, University of Bremen, Bremen, Germany, 2010. [Google Scholar]
- Eisenhauer, A.; Wasserburg, G.; Chen, J.; Bonani, G.; Collins, L.; Zhu, Z.; Wyrwoll, K. Holocene sea-level determination relative to the Australian continent: U/Th (TIMS) and 14C (AMS) dating of coral cores from the Abrolhos Islands. Earth Planet. Sci. Lett. 1993, 114, 529–547. [Google Scholar] [CrossRef]
- Esat, T.M.; McCulloch, M.T.; Chappell, J.; Pillans, B.; Omura, A. Rapid Fluctuations in Sea Level Recorded at Huon Peninsula During the Penultimate Deglaciation. Science 1999, 283, 197–201. [Google Scholar] [CrossRef]
- Esat, T.; Yokoyama, Y. Variability in the uranium isotopic composition of the oceans over glacial–interglacial timescales. Geochim. Cosmochim. Acta 2006, 70, 4140–4150. [Google Scholar] [CrossRef]
- Hamelin, B.; Bard, E.; Zindler, A.; Fairbanks, R.G. 234U/238U mass spectrometry of corals: How accurate is the U-Th age of the last interglacial period? Earth Planet. Sci. Lett. 1991, 106, 169–180. [Google Scholar] [CrossRef]
- Gallup, C.D.; Edwards, R.L.; Johnson, R.G. The timing of high sea levels over the past 200,000 years. Science 1994, 263, 796–800. [Google Scholar] [CrossRef]
- Gutjahr, M.; Vance, D.; Hoffmann, D.L.; Hillenbrand, C.-D.; Foster, G.; Rae, J.; Kuhn, G. Structural limitations in deriving accurate U-series ages from calcitic cold-water corals contrast with robust coral radiocarbon and Mg/Ca systematics. Chem. Geol. 2013, 355, 69–87. [Google Scholar] [CrossRef]
- Correa, M.L.; Montagna, P.; Joseph, N.; Rüggeberg, A.; Fietzke, J.; Flögel, S.; Dorschel, B.; Goldstein, S.; Wheeler, A.; Freiwald, A. Preboreal onset of cold-water coral growth beyond the Arctic Circle revealed by coupled radiocarbon and U-series dating and neodymium isotopes. Quat. Sci. Rev. 2012, 34, 24–43. [Google Scholar] [CrossRef]
- Ludwig, K.R.; Szabo, B.J.; Moore, J.G.; Simmons, K.R. Crustal subsidence rate off Hawaii determined from 234U/238U ages of drowned coral reefs. Geology 1991, 19, 171–174. [Google Scholar] [CrossRef]
- Ludwig, K.R.; Muhs, D.R.; Simmons, K.R.; Halley, R.B.; Shinn, E.A. Sea-level records at ~80 ka from tectonically stable platforms: Florida and Bermuda. Geology 1996, 24, 211–214. [Google Scholar] [CrossRef]
- Muhs, D.R.; Simmons, K.R.; Steinke, B. Timing and warmth of the Last Interglacial period: New U-series evidence from Hawaii and Bermuda and a new fossil compilation for North America. Quat. Sci. Rev. 2002, 21, 1355–1383. [Google Scholar] [CrossRef]
- Potter, E.-K.; Esat, T.; Schellmann, G.; Radtke, U.; Lambeck, K.; McCulloch, M. Suborbital-period sea-level oscillations during marine isotope substages 5a and 5c. Earth Planet. Sci. Lett. 2004, 225, 191–204. [Google Scholar] [CrossRef]
- Potter, E.-K.; Stirling, C.H.; Wiechert, U.H.; Halliday, A.N.; Spötl, C. Uranium-series dating of corals in situ using laser-ablation MC-ICPMS. Int. J. Mass Spectrom. 2005, 240, 27–35. [Google Scholar] [CrossRef]
- Roberts, G.P.; Houghton, S.L.; Underwood, C.; Papanikolaou, I.; Cowie, P.A.; Van Calsteren, P.; Wigley, T.; Cooper, F.J.; McArthur, J.M. Localization of Quaternary slip rates in an active rift in 105years: An example from central Greece constrained by234U-230Th coral dates from uplifted paleoshorelines. J. Geophys. Res. Space Phys. 2009, 114, B1046. [Google Scholar] [CrossRef]
- Shaked, Y.; Agnon, A.; Lazar, B.; Marco, S.; Avner, U.; Stein, M. Large earthquakes kill coral reefs at the north-west Gulf of Aqaba. Terra Nova 2004, 16, 133–138. [Google Scholar] [CrossRef]
- Shen, C.-C.; Li, K.-S.; Sieh, K.; Natawidjaja, D.; Cheng, H.; Wang, X.; Edwards, R.L.; Lam, D.D.; Hsieh, Y.-T.; Fan, T.-Y.; et al. Variation of initial 230Th/232Th and limits of high precision U–Th dating of shallow-water corals. Geochim. Cosmochim. Acta 2008, 72, 4201–4223. [Google Scholar] [CrossRef]
- Stein, M.; Wasserburg, G.; Aharon, P.; Chen, J.; Zhu, Z.; Bloom, A.; Chappell, J. TIMS U-series dating and stable isotopes of the last interglacial event in Papua New Guinea. Geochim. Cosmochim. Acta 1993, 57, 2541–2554. [Google Scholar] [CrossRef]
- Stirling, C.; Esat, T.; Lambeck, K.; McCulloch, M. Timing and duration of the Last Interglacial: Evidence for a restricted interval of widespread coral reef growth. Earth Planet. Sci. Lett. 1998, 160, 745–762. [Google Scholar] [CrossRef]
- Thomas, A.L.; Fujita, K.; Iryu, Y.; Bard, E.; Cabioch, G.; Camoin, G.; Cole, J.E.; Deschamps, P.; Durand, N.; Hamelin, B.; et al. Assessing subsidence rates and paleo water-depths for Tahiti reefs using U-Th chronology of altered corals. Mar. Geol. 2012, 295-298, 86–94. [Google Scholar] [CrossRef]
- Thompson, W.G.; Spiegelman, M.W.; Goldstein, S.L.; Speed, R.C. An open-system model for U-series age determinations of fossil corals. Earth Planet. Sci. Lett. 2003, 210, 365–381. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Esat, T.M. Long term variations of uranium isotopes and radiocarbon in the surface seawater recorded in corals. In Global Environmental Change in the Ocean and on Land; Shiyomi, M., Kawahata, H., Koizumi, H., Tsuda, A., Awaya, Y., Eds.; TERRAPUB: Tokyo, Japan, 2004; pp. 279–309. [Google Scholar]
- Yokoyama, Y.; Esat, T.; Lambeck, K. Coupled climate and sea-level changes deduced from Huon Peninsula coral terraces of the last ice age. Earth Planet. Sci. Lett. 2001, 193, 579–587. [Google Scholar] [CrossRef]
- Spratt, R.M.; Lisiecki, L.E. A Late Pleistocene sea level stack. Clim. Past. 2016, 12, 1079–1092. [Google Scholar] [CrossRef]
- Zykov, S.B.; Kiselev, G.P.; Zykova, E.N. New data on the uranium-isotopic composition of the Barents Sea water. In Radioactivity and Radioactive Elements in the Human Environment; Rikhvanov, L.P., Ed.; Tomsk State University: Tomsk, Tussai, 2013; pp. 214–217. [Google Scholar]
- Scholten, J.; Botz, R.; Paetsch, H.; Stoffers, P.; Weinelt, M. High-resolution uranium-series dating of Norwegian-Greenland Sea sediments: 230Th vs. δ18O stratigraphy. Mar. Geol. 1994, 121, 77–85. [Google Scholar] [CrossRef]
- Bezrukova, E.V. Paleogeography of the Baikal Region in the Late Glacial and Holocene; Nauka: Novosibirsk, Russia, 1999. [Google Scholar]
- Biskaborn, B.K.; Smith, S.L.; Noetzli, J.; Matthes, H.; Vieira, G.; Streletskiy, D.A.; Schoeneich, P.; Romanovsky, V.E.; Lewkowicz, A.G.; Abramov, A.; et al. Permafrost is warming at a global scale. Nat. Commun. 2019, 10, 264. [Google Scholar] [CrossRef]
- Afanasiev, A.H. Water Resources and Water Balance of the Lake Baikal; Nauka: Novosibirsk, Russia, 1976. [Google Scholar]
- Karabanov, E.B.; Prokopenko, A.A.; Kuzmin, M.I.; Williams, D.F.; Gvozdkov, A.N.; Kerber, E.V. Glaciation and interglacials of Siberia—Paleoclimatic record from Lake Baikal and its correlation with the West Siberian stratigraphy (Brunhes era of direct polarity). Russ. Geol. Geophys. 2001, 42, 48–63. [Google Scholar]
- Kuzmin, M.I.; Bychinsky, V.A.; Kerber, E.V.; Oshchepkova, A.V.; Goreglyad, A.V.; Ivanov, E.V. Chemical composition of sediments of deep-water Baikal wells as a basis for reconstruction of climate and environmental changes. Russ. Geol. Geophys. 2014, 55, 3–22. [Google Scholar]
- Stolpovskaya, V.N.; Solotchina, E.P.; Zhdanova, A.N. Quantitative IR spectroscopic analysis of non-clay minerals from the bottom sediments of Lakes Baikal and Hovsgol (in the context of paleoclimatic reconstructions). Russ. Geol. Geophys. 2006, 47, 778–788. [Google Scholar]
- Chebykin, E.P. Express Mass Spectrometric Determination of Elements and Natural Isotopes of Uranium and Thorium in Sediments of Lake Baikal for Their Dating and Deciphering of Paleoclimate Parameters. Ph.D. Thesis, Limnological Institute, Irkutsk, Russia, 2006. [Google Scholar]
- Chebykin, E.; Goldberg, E.; Kulikova, N. Elemental composition of suspended particles from the surface waters of Lake Baikal in the zone affected by the Selenga River. Russ. Geol. Geophys. 2010, 51, 1126–1132. [Google Scholar] [CrossRef]
- Carter, S.J.; Colman, S.M. Biogenic silica in Lake Baikal sediments: Results from 1990–1992 American cores. J. Great Lakes Res. 1994, 20, 751–760. [Google Scholar] [CrossRef]
- Colman, S.M.; Jones, G.A.; Rubin, M.; King, J.W.; Peck, J.A.; Orem, W.H. AMS radiocarbon analyses from Lake Baikal, Siberia: Challenges of dating sediments from a large, oligotrophic lake. Quat. Sci. Rev. 1996, 15, 669–684. [Google Scholar] [CrossRef]
- Chebykin, E.P.; Edgington, D.N.; Grachev, M.A.; Zheleznyakova, T.O.; Vorobyova, S.S.; Kulikova, N.S.; Azarova, I.N.; Khlystov, O.M.; Goldberg, E.L. Abrupt increase in precipitation and weathering of soils in East Siberia coincident with the end of the last glaciation (15 cal kyr BP). Earth Planet. Sci. Lett. 2002, 200, 167–175. [Google Scholar] [CrossRef]
- Demory, F.; Oberhänsli, H.; Nowaczyk, N.R.; Gottschalk, M.; Wirth, R.; Naumann, R. Detrital input and early diagenesis in sediments from Lake Baikal revealed by rock magnetism. Glob. Planet. Chang. 2005, 46, 145–166. [Google Scholar] [CrossRef]
- Edgington, D.; Robbins, J.; Colman, S.; Orlandini, K.; Gustin, M.-P. Uranium-series disequilibrium, sedimentation, diatom frustules, and paleoclimate change in Lake Baikal. Earth Planet. Sci. Lett. 1996, 142, 29–42. [Google Scholar] [CrossRef]
- Fagel, N.; Thamó-Bózsó, E.; Heim, B. Mineralogical signatures of Lake Baikal sediments: Sources of sediment supplies through Late Quaternary. Sediment. Geol. 2007, 194, 37–59. [Google Scholar] [CrossRef]
- Goldberg, E.; Phedorin, M.; Grachev, M.; Bobrov, V.; Dolbnya, I.; Khlystov, O.; Levina, O.; Ziborova, G. Geochemical signals of orbital forcing in the records of paleoclimates found in the sediments of Lake Baikal. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2000, 448, 384–393. [Google Scholar] [CrossRef]
- Goldberg, E.; Grachev, M.; Chebykin, E.; Phedorin, M.; Kalugin, I.; Khlystov, O.; Zolotarev, K. Scanning SRXF analysis and isotopes of uranium series from bottom sediments of Siberian lakes for high-resolution climate reconstructions. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2005, 543, 250–254. [Google Scholar] [CrossRef]
- Goldberg, E.; Chebykin, E.; Zhuchenko, N.; Vorobyeva, S.S.; Stepanova, O.; Khlystov, O.; Ivanov, E.; Weinberg, E.; Gvozdkov, A. Uranium isotopes as proxies of the environmental history of the Lake Baikal watershed (East Siberia) during the past 150ka. Palaeogeogr. Palaeoclim. Palaeoecol. 2010, 294, 16–29. [Google Scholar] [CrossRef]
- Grachev, M.A.; Vorobyova, S.S.; Likhoshway, E.V.; Goldberg, E.L.; Ziborova, G.A.; Levina, O.V.; Khlystov, O.M. A high resolution diatom record of the palaeoclimates of East Siberia for the last 2.5 my from Lake Baikal. Quat. Sci. Rev. 1998, 17, 1101–1106. [Google Scholar] [CrossRef]
- Horiuchi, K.; Minoura, K.; Hoshino, K.; Oda, B.; Nakamura, T.; Kawai, T. Palaeoenvironmental history of Lake Baikal during the last 23000 years. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2000, 157, 95–108. [Google Scholar] [CrossRef]
- Kashiwaya, K. Long Continental Records from Lake Baikal; Springer: Tokyo, Japan, 2003. [Google Scholar]
- Khursevich, G.K.; Karabanov, E.B.; Prokopenko, A.A.; Williams, D.F.; Kuzmin, M.I.; Fedenya, S.A.; Gvozdkov, A.A. Insolation regime in Siberia as a major factor controlling diatom production in Lake Baikal during the past 800,000 years. Quat. Int. 2001, 80-81, 47–58. [Google Scholar] [CrossRef]
- Kuzmin, M.I.; Karabanov, E.B.; Kawai, T.; Williams, D.; Bychinsky, V.A.; Kerber, E.V.; Kravchinsky, V.A.; Bezrukova, E.V.; Prokopenko, A.A.; Geletti, V.F.; et al. Deep drilling on Lake Baikal: Main results. Russ. Geol. Geoph. 2001, 42, 3–28. [Google Scholar]
- Morgenstern, U.; Ditchburn, R.G.; Vologina, E.G.; Sturm, M. 32Si dating of sediments from Lake Baikal. J. Paleolimnol. 2013, 50, 345–352. [Google Scholar] [CrossRef]
- Och, L.M.; Müller, B.; Wichser, A.; Ulrich, A.; Vologina, E.G.; Sturm, M. Rare earth elements in the sediments of Lake Baikal. Chem. Geol. 2014, 376, 61–75. [Google Scholar] [CrossRef]
- Prokopenko, A.A.; Karabanov, E.B.; Williams, D.F.; Kuzmin, M.I.; Shackleton, N.J.; Crowhurst, S.J.; Peck, J.A.; Gvozdkov, A.N.; King, J.W. Biogenic Silica Record of the Lake Baikal Response to Climatic Forcing during the Brunhes. Quat. Res. 2001, 55, 123–132. [Google Scholar] [CrossRef]
- Shchetnikov, A.A.; Bezrukova, E.V.; Maksimov, F.E.; Kuznetsov, V.Y.; Filinov, I.A. Environmental and climate reconstructions of the Fore-Baikal area during MIS 5-1: Multiproxy record from terrestrial sediments of the Ust-Oda section (Siberia, Russia). J. Asian Earth Sci. 2016, 129, 220–230. [Google Scholar] [CrossRef]
- Sklyarova, O.; Sklyarov, E.; Och, L.; Pastukhov, M.; Zagorulko, N. Rare earth elements in tributaries of Lake Baikal (Siberia, Russia). Appl. Geochem. 2017, 82, 164–176. [Google Scholar] [CrossRef]
- Sturm, M.; Vologina, E.G.; Vorob’Eva, S.S. Holocene and Late Glacial sedimentation near steep slopes in southern Lake Baikal. J. Limnol. 2016, 75, 24–35. [Google Scholar] [CrossRef][Green Version]
- Tarasov, P.; Bezrukova, E.; Karabanov, E.; Nakagawa, T.; Wagner, M.; Kulagina, N.; Letunova, P.; Abzaeva, A.; Granoszewski, W.; Riedel, F. Vegetation and climate dynamics during the Holocene and Eemian interglacials derived from Lake Baikal pollen records. Palaeogeogr. Palaeoclim. Palaeoecol. 2007, 252, 440–457. [Google Scholar] [CrossRef]
- Tyszkowski, S.; Kaczmarek, H.; Słowiński, M.; Kozyreva, E.; Brykała, D.; Rybchenko, A.; Babicheva, V. Geology, permafrost, and lake level changes as factors initiating landslides on Olkhon Island (Lake Baikal, Siberia). Landslides 2014, 12, 573–583. [Google Scholar] [CrossRef]
- Watanabe, T.; Nakamura, T.; Nara, F.W.; Kakegawa, T.; Horiuchi, K.; Senda, R.; Oda, T.; Nishimura, M.; Matsumoto, G.I.; Kawai, T. High-time resolution AMS 14C data sets for Lake Baikal and Lake Hovsgol sediment cores: Changes in radiocarbon age and sedimentation rates during the transition from the last glacial to the Holocene. Quat. Int. 2009, 205, 12–20. [Google Scholar] [CrossRef]
- Fleisher, R.L.; Raabe, O.G. Recoiling alpha-emitting nuclei. Mechanisms for uranium-series disequilibrium. Geochim. Cosmochim. Acta 1978, 42, 973–978. [Google Scholar] [CrossRef]
- Hovsgol Drilling Project (HDP) Members. Sedimentary record from Lake Hovsgol, NW Mongolia: Results from the HDP-04 and HDP-06 drill cores. Quat. Int. 2009, 205, 21–37. [Google Scholar] [CrossRef]
- Katsuta, N.; Matsumoto, G.I.; Tani, Y.; Tani, E.; Murakami, T.; Kawakami, S.-I.; Nakamura, T.; Takano, M.; Matsumoto, E.; Abe, O.; et al. A higher moisture level in the early Holocene in northern Mongolia as evidenced from sediment records of Lake Hovsgol and Lake Erhel. Quat. Int. 2017, 455, 70–81. [Google Scholar] [CrossRef]
- Kim, B.; Cheong, D.; Lee, E. Paleoenvironmental changes in northern Mongolia during the last deglaciation revealed by trace element records in ostracods from Lake Hovsgol. Quat. Int. 2015, 384, 169–179. [Google Scholar] [CrossRef]
- Mochizukia, A.; Murata, T.; Hosoda, K.; Katano, T.; Tanaka, Y.; Mimura, T.; Mitamura, O.; Nakano, S.; Okazaki, Y.; Sugiyama, Y.; et al. Distributions and geochemical behaviors of oxyanion-forming trace elements and uranium in the Hövsgöl–Baikal–Yenisei water system of Mongolia and Russia. J. Geochem. Explor. 2018, 188, 123–136. [Google Scholar] [CrossRef]
- Orkhonselenge, A.; Krivonogov, S.K.; Mino, K.; Kashiwaya, K.; Safonova, I.Y.; Yamamoto, M.; Kashima, K.; Nakamura, T.; Kim, J.Y. Holocene sedimentary records from Lake Borsog at the eastern shore of Lake Hovsgol, Mongolia, and their paleoenvironmental implications. Quat. Int. 2013, 290–291, 95–109. [Google Scholar] [CrossRef]
- Oyunchimeg, T.; Chebykin, E.P. High-Performance Technique on the Base of ICP-MS for Obtaining High-Resolution Records of Climate-Sensitive Elements in Bottom Sediments of Lake Hovsgol (Mongolia). Chem. Sustain. Dev. 2009, 17, 97–110. [Google Scholar]
- Prokopenko, A.A.; Khursevich, G.K.; Bezrukova, E.V.; Kuzmin, M.I.; Boes, X.; Williams, D.F.; Fedenya, S.A.; Kulagina, N.V.; Letunova, P.P.; Abzaeva, A.A. Paleoenvironmental proxy records from Lake Hovsgol, Mongolia, and a Synthesis of Holocene Climate Change in the Lake Baikal watershed. Quat. Res. 2007, 68, 2–17. [Google Scholar] [CrossRef]
- Solotchina, E.P.; Prokopenko, A.A.; Kuzmin, M.I.; Solotchin, P.A.; Zhdanova, A.N. Climate signals in sediment mineralogy of Lake Baikal and Lake Hovsgol during the LGM-Holocene transition and the 1-Ma carbonate record from the HDP-04 drill core. Quat. Int. 2009, 205, 38–52. [Google Scholar] [CrossRef]
- Watanabe, T.; Minoura, K.; Nara, F.W.; Shichi, K.; Horiuchi, K.; Kakegawa, T.; Kawai, T. Last glacial to post glacial climate changes in continental Asia inferred from multi-proxy records (geochemistry, clay mineralogy, and paleontology) from Lake Hovsgol, northwest Mongolia. Glob. Planet. Chang. 2012, 88–89, 53–63. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, W.; Feng, Z.; Mischke, S.; Gao, X.; Gao, D.; Sun, F. Holocene hydrological and climatic change on the northern Mongolian Plateau based on multi-proxy records from Lake Gun Nuur. Palaeogeogr. Palaeoclim. Palaeoecol. 2012, 323–325, 75–86. [Google Scholar] [CrossRef]
- Vosel, Y.S. Geochemistry of Uranium in Modern Carbonate Deposits of Small Lakes: Forms of Occurrence and Isotopic Ratios 234U/238U. Ph.D. Thesis, Novosibirsk State University, Novosibirsk, Russia, 2015. [Google Scholar]
- Chebykin, E.P.; Sorokovikova, L.M.; Tomberg, I.V.; Vodneva, E.N.; Rasskazov, S.V.; Khodzher, T.V.; Grachev, M.A. Current state of the river. Selenga on the territory of Russia by main components and trace elements. Chem. Sustain. Dev. 2012, 20, 613–631. [Google Scholar]
- Rasskazov, S.V.; Chebykin, E.P.; Ilyasova, A.M.; Vodneva, E.N.; Chuvashova, I.S.; Bornyakov, S.A.; Seminsky, A.K.; Snopkov, S.V.; Chechel’nitsky, V.V.; Gileva, N.A. Creating the Kultuk polygon for earthquake prediction: Variations of (234U/238U) and 87Sr/86Sr in groundwater from active faults at the western shore of Lake Baikal. Geodyn. Tectonophys. 2015, 26, 519–554. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tokarev, I.; Yakovlev, E. Non-Equilibrium Uranium as an Indicator of Global Climate Variations—The World Ocean and Large Lakes. Water 2021, 13, 3514. https://doi.org/10.3390/w13243514
Tokarev I, Yakovlev E. Non-Equilibrium Uranium as an Indicator of Global Climate Variations—The World Ocean and Large Lakes. Water. 2021; 13(24):3514. https://doi.org/10.3390/w13243514
Chicago/Turabian StyleTokarev, Igor, and Evgeny Yakovlev. 2021. "Non-Equilibrium Uranium as an Indicator of Global Climate Variations—The World Ocean and Large Lakes" Water 13, no. 24: 3514. https://doi.org/10.3390/w13243514
APA StyleTokarev, I., & Yakovlev, E. (2021). Non-Equilibrium Uranium as an Indicator of Global Climate Variations—The World Ocean and Large Lakes. Water, 13(24), 3514. https://doi.org/10.3390/w13243514




