Preliminary Assessment of Ecological Status of the Siversky Donets River Basin (Ukraine) Based on Phytoplankton Parameters and Its Verification by Other Biological Data
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling Strategy
2.3. Ecological Analysis Based on Species Composition and Quantitative Characteristics of Phytoplankton
2.4. Statistical Data Analysis
3. Results
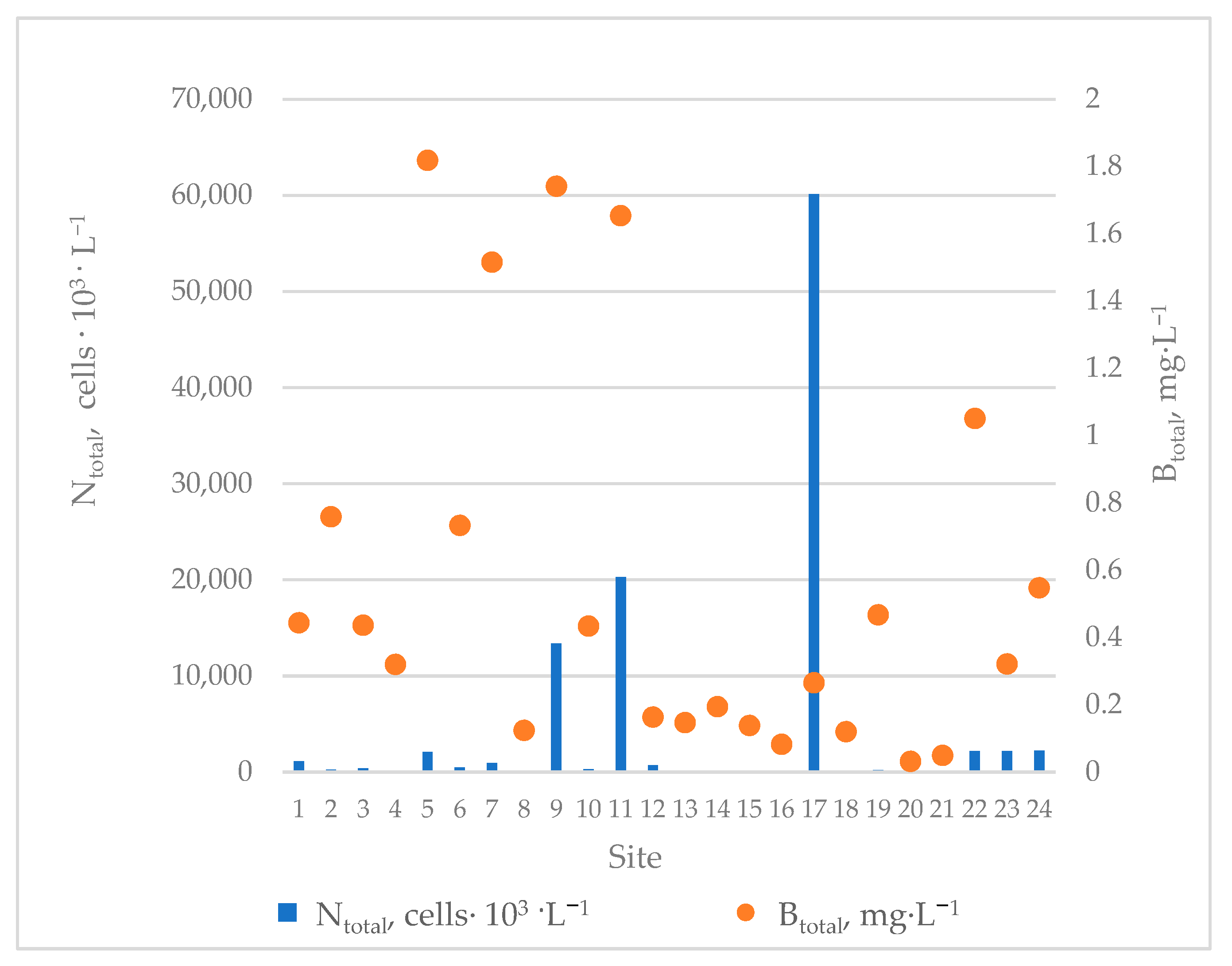
3.1. Physicochemical Characteristics of the Sampling Sites
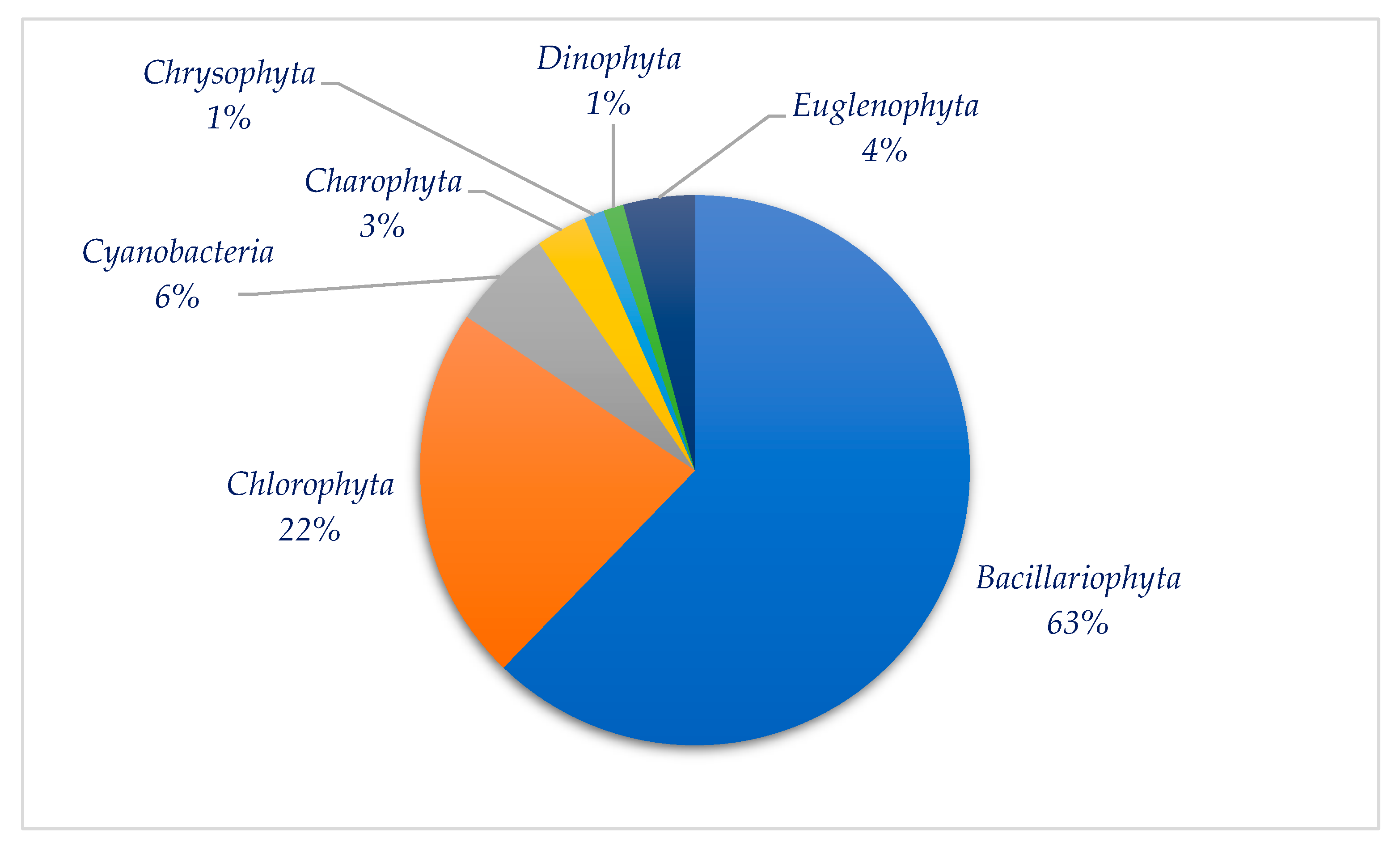
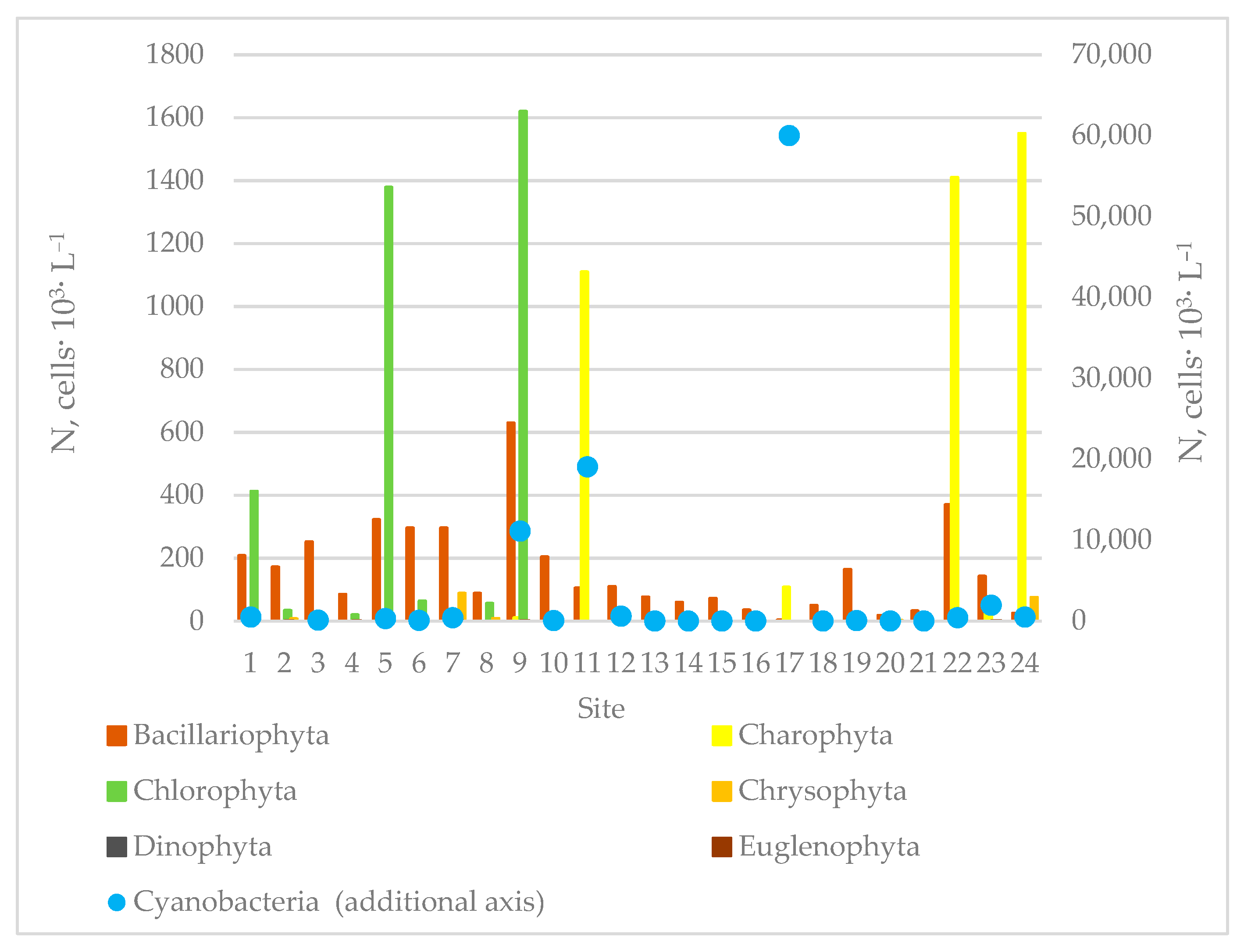
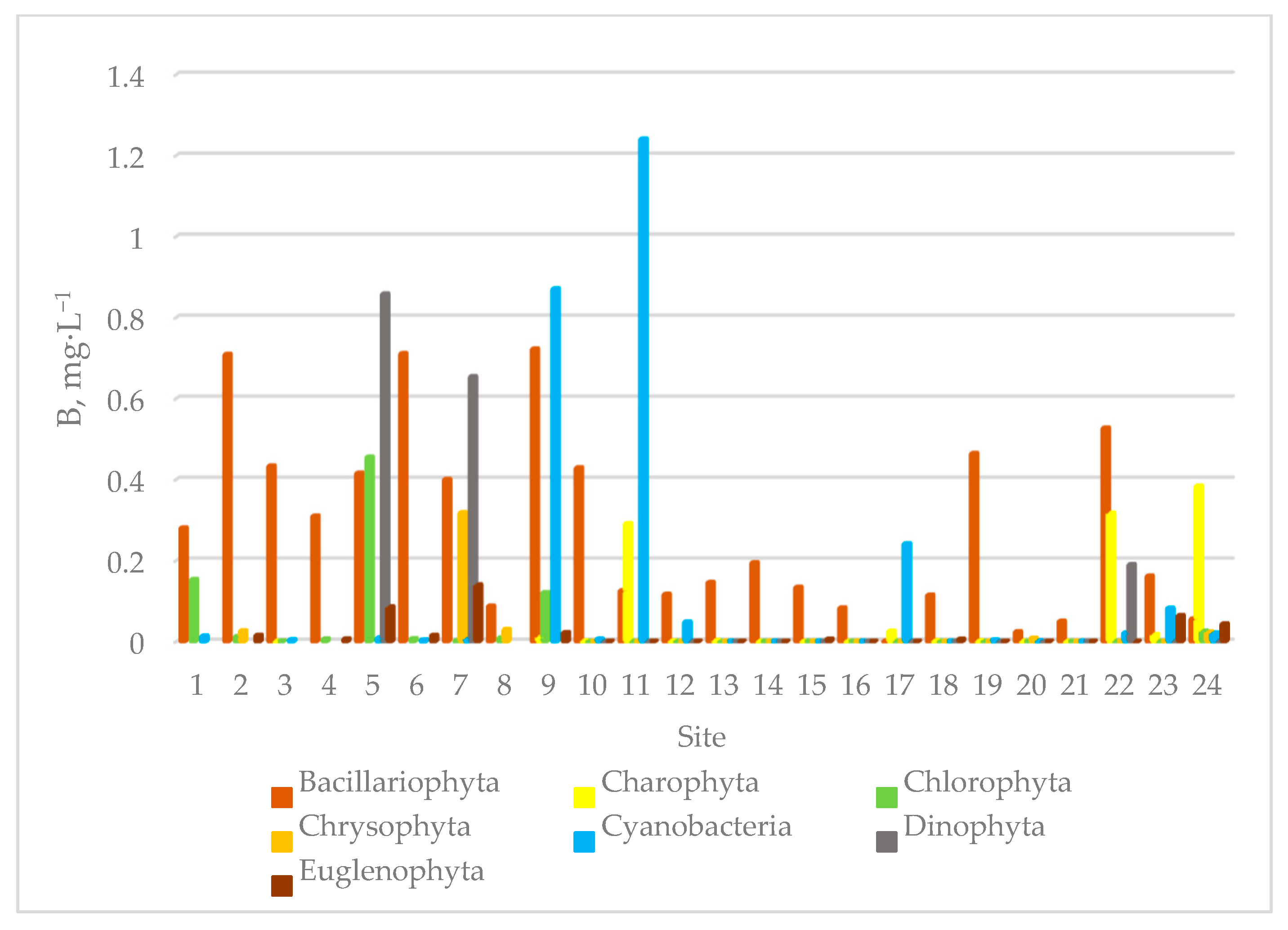
3.2. Species Composition of Algae in Sampling Sites of the Siversky Donets River Basin
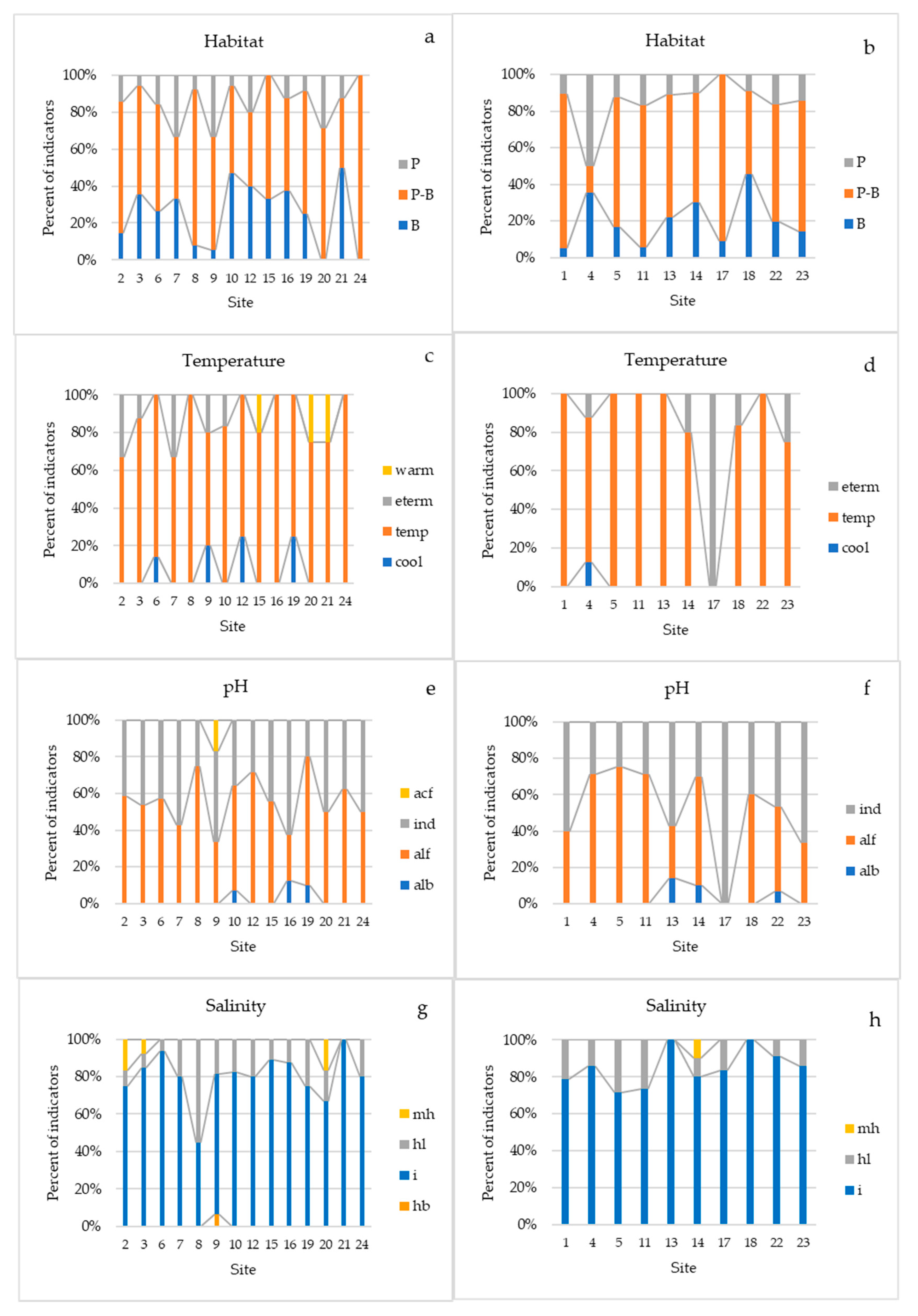
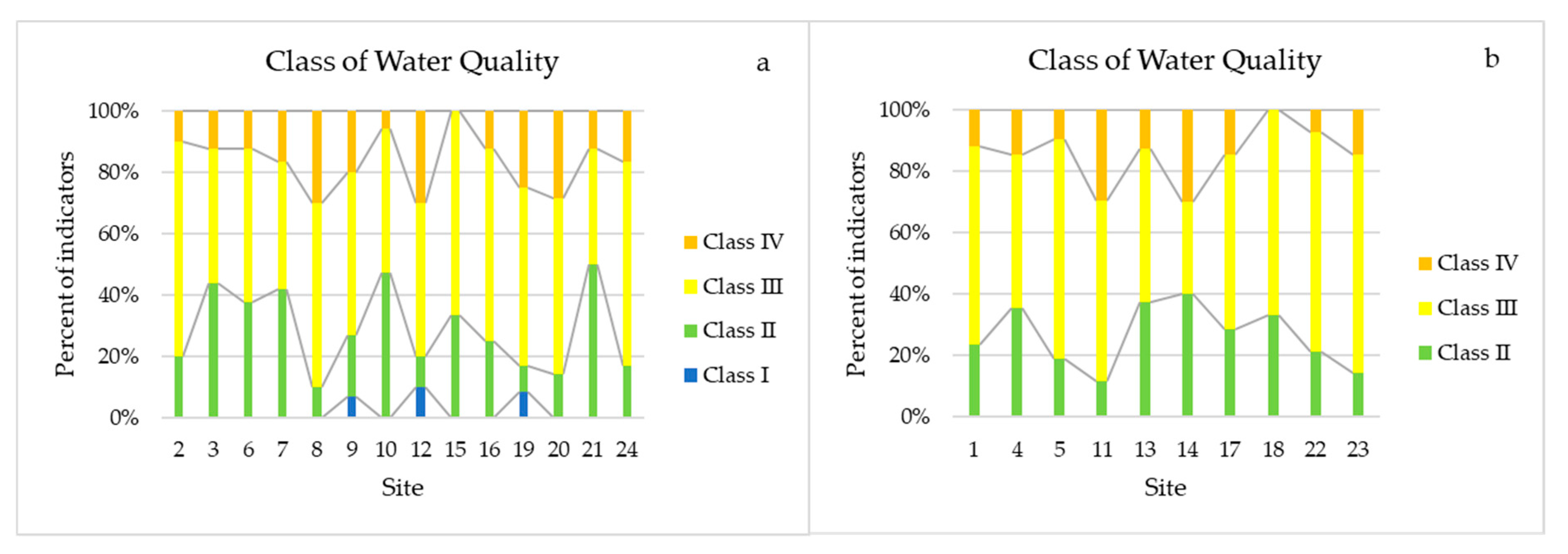
3.3. Ecological Analysis Based on Species Composition and Indicator Characteristics of Phytoplankton
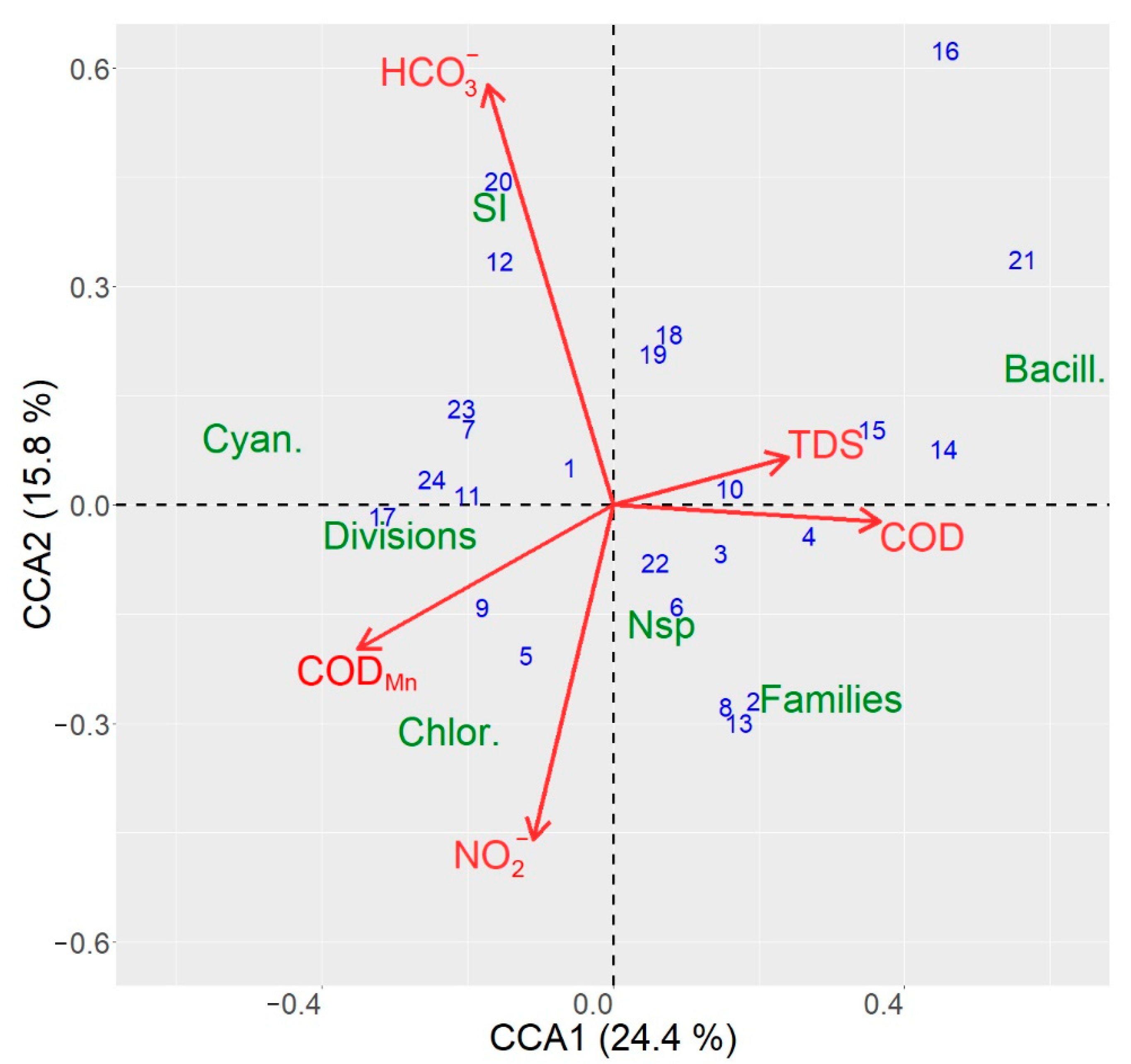
3.4. CCA Ordination Results for the Sampling Sites in the Siversky Donets River Basin
4. Discussion
4.1. Chemical Contamination in the Sampling Sites
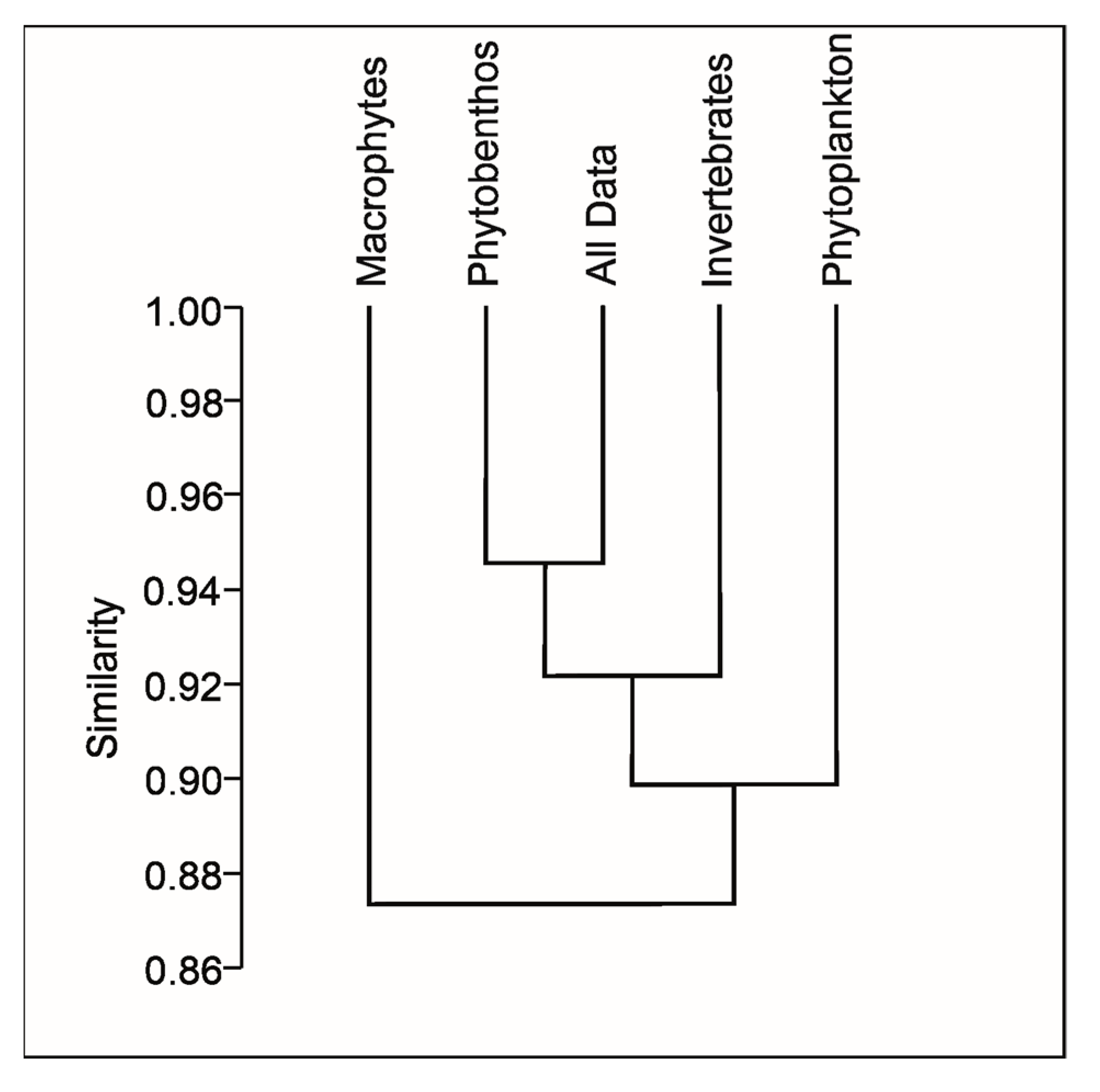
4.2. Characteristics of the Phytoplankton as a Biological Quality Element (BQE)
4.3. Analysis of Species-Environment Relationships and Identification of Sites with the Highest Water Quality
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ward, J.V.; Tockner, K.; Schiemer, F. Biodiversity of floodplain ecosystems: Ecotones and connectivity. Regul. Rivers Res. Manag. 1999, 15, 125–139. [Google Scholar] [CrossRef]
- Afanasyev, S.A. Forming of Hydrobiota of the River Systems in the Territory of Ukraine in View of History of Hydrographic Net. Hydrobiol. J. 2015, 51, 3–12. [Google Scholar] [CrossRef]
- Popa, G.-O.; Curtean-Bănăduc, A.; Bănăduc, D.; Florescu, I.E.; Burcea, A.; Dudu, A.; Georgescu, S.E.; Costache, M. Molecular Markers Reveal Reduced Genetic Diversity in Romanian Populations of Brown Trout, Salmo trutta L., 1758 (Salmonidae). Acta Zool. Bulg. 2016, 68, 399–406. [Google Scholar]
- Arthington, A.H.; Pusey, B.J. Flow restoration and protection in Australian rivers. River Res. Appl. 2003, 19, 377–395. [Google Scholar] [CrossRef]
- Boon, P.J. The development of integrated methods for assessing river conservation value. Hydrobiologia 2000, 149, 413–428. [Google Scholar] [CrossRef]
- Garcia, A.M.; Vieira, J.P.; Winemiller, K.O.; Raseira, M.B. Spatiotemporal variation in shallow-water freshwater fish distribution and abundance in a large subtropical coastal lagoon. Environ. Biol. Fishes 2003, 68, 215–228. [Google Scholar] [CrossRef]
- Afanasyev, S.O.; Lietitska, O.; Marushevska, O. River re-naturalisation in the Tisza River basin after forest cutting activities. Acta Zool. Bulg. 2014, 7, 57–62. [Google Scholar]
- Sipkay, C.; Kiss, K.T.; Drégelyi-Kiss, Á.; Farkas, E.; Hufnagel, L. Analysis of climate change scenarios based on the modelling of the seasonal dynamics of phyto-plankton in the Danube. Hidrológiai Közlöny 2009, 89, 56–59. (In Hungarian) [Google Scholar]
- Dokulil, M.T.; Donabaum, U. Phytoplankton of the Danube River: Composition and Long-Term Dynamics. Acta Zool. Bulg. Suppl. 2014, 7, 147–152. [Google Scholar]
- Abonyi, A.; Ács, É.; Hidas, A.; Grigorszky, I.; Várbíró, G.; Borics, G.; Kiss, K.T. Functional diversity of phytoplankton highlights long-term gradual regime shift in the middle section of the Danube River due to global warming, human impacts and oligotrophication. Freshw. Biol. 2018, 63, 456–472. [Google Scholar] [CrossRef] [Green Version]
- Trábert, Z.; Duleba, M.; Bíró, T.; Dobosy, P.; Földi, A.; Hidas, A.; Kiss, K.T.; Óvári, M.; Takács, A.; Várbíró, G.; et al. Effect of Land Use on the Benthic Diatom Community of the Danube River in the Region of Budapest. Water 2020, 12, 479. [Google Scholar] [CrossRef] [Green Version]
- Wukhan, O.O. Integral assessment of surface water quality of the Siversky Donets basin. Hydrol. Hydrochem. Hydroecol. 2010, 20, 111–125. (In Ukrainian) [Google Scholar]
- Yakovliev, Y.; Chumachenko, S. With contributions from HD staff. In Ecological Threats in Donbas, Ukraine. Assessment of Ecological Hazards in Donbas Impacted by the Armed Conflict in Eastern Ukraine; Centre for Humanitarian Dialogue: Geneva, Switzerland, 2017; pp. 1–64. [Google Scholar]
- Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02000L0060-20140101 (accessed on 27 September 2021).
- Afanasyev, S.O. Problems and Progress of Investigations of Hydroecosystems’ Ecological State in View of Implementation of EU Environmental Directives in Ukraine. Hydrobiol. J. 2019, 55, 3–17. [Google Scholar] [CrossRef]
- Mischke, U.; Behrendt, H.; Köhler, J.; Opitz, D. Überarbeiteter Endbericht zum LAWA-Vorhaben: Entwicklung eines Bewertungsverfahrens für Fließgewässer mittels Phytoplankton zur Umsetzung der EU-Wasserrahmenrichtlinie; Report; Berlin, Germany; 2005. pp. 1–70. Available online: http://www.gewaesser-bewertung.de/files/mischke_praxistestbericht_06.pdf (accessed on 25 September 2021). (In German).
- Borics, G.; Várbíró, G.; Grigorszky, I.; Krasznai, E.; Szabó, S.; Kiss, K.T. A new evaluation of potamo-plankton for the assessmant of the ecological status of rivers. Arch. Hydrobiol. Suppl. 2007, 161, 465–486. [Google Scholar]
- Dokulil, M.; Donabaum, U. 8 Phytoplankton. In Joint Danube Survey 3, A Comprehensive Analysis of Danube Water QualityICPDR/International Commission for the Protection of the Danube River; ICPDR—International Commission for the Protection of the Danube River: Vienna, Austria, 2015; pp. 119–125. [Google Scholar]
- Thomas, M.K.; Kremer, C.T.; Litchman, E. Environment and evolutionary history determine the global biogeography of phytoplankton temperature traits. Glob. Ecol. Biogeogr. 2016, 25, 75–86. [Google Scholar] [CrossRef]
- Bolgovics, Á.; Várbíró, G.; Ács, É.; Trábert, Z.; Kiss, K.T.; Pozderka, V.; Görgényi, J.; Boda, P.; Lukács, B.-A.; Nagy-László, Z.; et al. Phytoplankton of rhithral rivers: Its origin, diversity and possible use for quality-assessment. Ecol. Indic. 2017, 81, 587–596. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.S.; Seo, I.W.; Baek, D. Seasonally varying effects of environmental factors on phytoplankton abundance in the regulated rivers. Sci. Rep. 2019, 9, 9266. [Google Scholar] [CrossRef]
- Rusanov, A.G.; Bíró, T.; Kiss, K.T.; Buczkó, K.; Grigorszky, I.; Hidas, A.; Duleba, M.; Trábert, Z.; Földi, A.; Ács, É. Relative importance of climate and spatial processes in shaping species composition, functional structure and beta diversity of phytoplankton in a large river. Sci. Total Environ. 2021, 807, 150891. [Google Scholar] [CrossRef]
- Silva, K.R.P.; Barbosa, J.E.L.; Santana, L.M.; Barbosa, L.G. Phytoplankton functional groups in shallow aquatic ecosystems from the semiarid region of Brazil Grupos funcionais fitoplanctônicos em ecossistemas aquáticos rasos na região semiárida do Brasil. Acta Limnol. Bras. 2021, 33, e24. [Google Scholar] [CrossRef]
- Stanković, I.; Udovič, M.G.; Borics, G. Phytoplankton. In Joint Danube Survey 4. Scientific Report. A Shared Analysis of the Danube River; Liška, I., Wagner, F., Sengl, M., Deutsch, K., Slobodník, J., Paunović, M., Eds.; ICPDR—International Commission for the Protection of the Danube River: Vienna, Austria, 2021; pp. 73–82. [Google Scholar]
- Vasenko, O.G.; Hrytsenko, A.V.; Каrаbаsh, G.O.; Stankevych, P.P. Siversky Donets: Water and Ecological Atlas; Hrytsenko, A.V., Vasenko, O.G., Eds.; Raider: Kharkiv, Ukraine, 2006; pp. 1–188. (In Ukrainian) [Google Scholar]
- Vishnevsky, V.I. Rivers and Water Bodies in Ukraine. Status and Exploration; Vipol: Kyiv, Ukraine, 2000; pp. 1–367. (In Ukrainian) [Google Scholar]
- Hrytsenko, A.V.; Vasenko, O.G.; Kolisnyk, A.V. Modern Ecological Status of the Ukrianian part of the Siversky Donets River (Expeditionary Research); Grytsenko, A.V., Vasenko, O.G., Eds.; Коntrast: Kharkiv, Ukraine, 2011; pp. 1–340. (In Ukrainian) [Google Scholar]
- Illies, J. (Ed.) Limnofauna Europaea. In A Checklist of the Animals Inhabiting European Inland Waters, with an Account of Their Distribution and Ecology, 2nd ed.; Gustav Fischer Verlag: Stuttgart, Germany, 1978; pp. 1–552. [Google Scholar]
- Osadchy, V.I.; Nabyvanets, B.Y.; Osadcha, N.M.; Nabyvanets, Y.B. Hydrochemical Reference Book. Surface Waters of Ukraine. Hydrochemical Calculations; Methods of Analysis; Nika-Center: Kyiv, Ukraine, 2008; pp. 1–655. (In Ukrainian) [Google Scholar]
- Krammer, K. Bacillariophyceae 1. Naviculaceae. In Süßwasserflora von Mitteleuropa; Gustav Fischer Verlag: Stuttgart, Germany, 1986; pp. 1–876. [Google Scholar]
- Krammer, K. Bacillariophyceae. 4. Achnanthaceae. Kritische Erganzungen zu Navicula (Lineolatae) und Gomphonema. In Süßwasserflora von Mitteleuropa; Gustav Fischer Verlag: Stuttgart, Germany, 1991; pp. 1–437. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae. 3. Centrales, Fragilariaceae, Eunotiaceae. In Süßwasserflora von Mitteleuropa; Gustav Fischer Verlag: Stuttgart, Germany, 1991; pp. 1–600. [Google Scholar]
- Komárek, J. Cyanoprokaryota 3. Teil/3rd Part: Heterocystous genera. In Süsswasserflora von Mitteleuropa; Budel, B., Gärtner, G., Krienitz, L., Schagerl, M., Eds.; Springer Spektrum: Heidelberg, Germany, 2013; pp. 1–1130. [Google Scholar]
- Komarek, J.; Anagnostidis, K. Cyanoprokaryota. 1. Chroococcales. In Susswasserflora von Mirreleuropa; Bd. 19/1; Gustav Vischer Verlag: Jena, Germany, 1998; pp. 1–548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota. Teil 2. Oscillatoriales. In Süßwasserflora von Mitteleuropa; Bd 19/2; Elsevier: München, Germany, 2005; pp. 1–759. [Google Scholar]
- Krammer, K. The Genus Pinnularia. In Diatoms of Europe; A.R.G. Gantner Verlag: Königstein, Germany, 2000; Volume 1, pp. 1–703. [Google Scholar]
- Krammer, K. Cymbella. In Diatoms of Europe; A.R.G. Gantner Verlag, K.-G.: Ruggell, Liechtenstein, 2002; Volume 3, pp. 1–584. [Google Scholar]
- Krammer, K. Cymbopleura, Delicata, Navicymbula, Gomphocymbellopsis, Afrocymbella. In Diatoms of Europe; A.R.G. Gantner Verlag, K.-G.: Ruggell, Liechtenstein, 2003; Volume 4, pp. 1–530. [Google Scholar]
- Vetrova, Z.I. Euglenophyta. In Flora of Algae of the Continental Water Bodies of the Ukrainian SSR. 1; Naukova Dumka: Kiev, Ukraine, 1986; pp. 1–348. (In Russian) [Google Scholar]
- Tsarenko, P.M. Short Identification Book of Chlorococcales of the Ukraine SSR; Naukova Dumka: Kiev, Ukraine, 1990; pp. 1–208. (In Russian) [Google Scholar]
- Vetrova, Z.I. Euglenophyta. Order 1. Euglenales, Family 2. Euglenaceae. Genus 1. Trachelomonas. Group II. Genus 2. Strombomonas. Genus 3. Euglena. In Flora of Algae of the Continental Water Bodies of Ukraine. 1; Naukova Dumka: Kiev, Ukraine, 1993; pp. 1–260. (In Russian) [Google Scholar]
- Krammer, K. Die cymbelloiden Diatomeen. Eine Monographie der weltweit bekannten Taxa. 1. Allegmeins und Encyonema Part. In Bibliotheca Diatomologica; J. Cramer: Berlin/Stuttgart, Germany, 1997; pp. 1–382. [Google Scholar]
- Palamar-Mordvintseva, G.M. Desmidiales. Gonatozygaceae, Peniaceae, Closteriaceae, Desmidiaceae. In Flora of Algae of the Continental Water Bodies of Ukraine; Fasc. 1, Part 1; Akademperiodyka Press: Kiev, Ukraine, 2003; pp. 1–355. (In Ukrainian) [Google Scholar]
- Vetrova, Z.I. Euglenofitovye vodorosli. In Flora of Algae of the Continental Water Bodies of Ukraine. 2; Lileia Press: Kiev, Ukraine, 2004; pp. 1–272. (In Russian) [Google Scholar]
- Palamar-Mordvintseva, G.M. Desmidiales. In Flora of Algae of the Continental Water Bodies of Ukraine. Fasc. 1, Pars 2; Akademperiodyka Press: Kyiv, Ukraine, 2005; pp. 1–573. (In Ukrainian) [Google Scholar]
- Kovalenko, O.V. Cyanoprokaryota. Ordo Chroococcales. In Flora of Algae of Ukraine. 1. Fasc. 1; VELES: Kyiv, Ukraine, 2009; pp. 1–397. (In Ukrainian) [Google Scholar]
- Palamar-Mordvintseva, G.M.; Petlovany, O.A. Streptophyta. Familia Mesotaeniaceae. In Flora of Algae of Ukraine. 12. Fasc. 1; VELES: Kyiv, Ukraine, 2009; pp. 1–158. (In Ukrainian) [Google Scholar]
- Lange-Bertalot, H.; Hofmann, G.; Werum, M.; Cantonati, M. Freshwater Benthic Diatoms of Central Europe: Over 800 Common Species Used in Ecological Assessment; Koeltz Botanical Books: Schmitten-Oberreifenberg, Germany, 2017; pp. 1–942. [Google Scholar]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. World-Wide Electronic Publication, National University of Ireland, Galway. 2021. Available online: http://www.algaebase.org (accessed on 24 September 2021).
- Tsarenko, P.M.; Wasser, S.P.; Nevo, E. (Eds.) Chlorophyta, V.3. In Algae of Ukraine: Diversity, Nomenclature, Taxonomy, Ecology and Geography.–Koeltz Scientific Books; Gantner Verlag: Ruggel, Liechtenstein, 2011; pp. 1–511. [Google Scholar]
- Tsarenko, P.M.; Wasser, S.P.; Nevo, E. (Eds.) Bacillariophyta, V.2. In Algae of Ukraine: Diversity, Nomenclature, Taxonomy, Ecology and Geography.–Koeltz Scientific Books; Gantner Verlag: Ruggel, Liechtenstein, 2009; pp. 1–413. [Google Scholar]
- Tsarenko, P.M.; Wasser, S.P.; Nevo, E. (Eds.) Charophyta, V.4. In Algae of Ukraine: Diversity, Nomenclature, Taxonomy, Ecology and Geography. Koeltz Scientific Books; Gantner Verlag: Ruggell, Liechtenstein, 2014; pp. 1–703. [Google Scholar]
- Tsarenko, P.M.; Wasser, S.P.; Nevo, E. (Eds.) Cyanoprokaryota, Euglenophyta, Chrysophyta, Xanthophyta, Raphidophyta, Dinophyta, Cryptophyta, Glaucocystophyta and Rhodophyta. V.1. In Algae of Ukraine: Diversity, Nomenclature, Taxonomy, Ecology and Geography; Gantner Verlag: Ruggell, Liechtenstein, 2006; pp. 1–713. [Google Scholar]
- Hillebrand, H.; Dürselen, C.D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Barinova, S.S.; Belous, Y.P.; Tsarenko, P.M. Algal Indication of Water Bodies in Ukraine; Haifa University Press: Haifa, Israel; Kiev, Ukraine, 2019; pp. 1–367. (In Russian) [Google Scholar]
- Romanenko, V.D.; Zhukynsky, V.M.; Oksiyuk, O.P.; Yatsyk, A.V.; Chernyavska, A.D.; Vasenko, O.G.; Vernichenko, G.A. Procedure of the Determination and Use of Ecological Norms of the Quality of the Surface Waters of Land and Estuaries of Ukraine; Minekoresursiv Ukrayiny Press: Kyiv, Ukraine, 2001; pp. 1–48. (In Ukrainian) [Google Scholar]
- Bilous, O.; Barinova, S.; Klochenko, P. The role of phytoplankton in the ecological assessment of the Southern Bug River middle reaches (Ukraine). Fundam. Appl. Limnol. 2014, 184, 277–295. [Google Scholar] [CrossRef]
- Tsarenko, P.M.; Bilous, O.P.; Kryvosheia-Zakharova, O.M.; Lilitska, H.H.; Barinova, S. Diversity of Algae and Cyanobacteria and Bioindication Characteristics of the Alpine Lake Nesamovyte (Eastern Carpathians, Ukraine) from 100 Years Ago to the Present. Diversity 2021, 13, 256. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 1–989. [Google Scholar]
- Oksanen, F.J.; Blanchet, G.; Friendly, U.M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R package Version 2.4-3. 2017. Available online: https://CRAN.R-project.org/package=vegan (accessed on 29 September 2021).
- Šmilauer, P.; Lepš, J. Using Canoco 5. In Multivariate Analysis of Ecological Data Using CANOCO 5; Cambridge University Press: Cambridge, UK, 2014; pp. 15–38. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F. Canonical Correspondence Analysis: A new eigenvector technique for multivariate direct gradient analysis. Ecology 1986, 67, 1167–1179. [Google Scholar] [CrossRef] [Green Version]
- Svirenko, D.O. Dniprovske vodoskhovyshche. (Vol. 1–5). Fitoplankton vodoskhovyshcha i yoho zatok. Visnyk Dnipropetr. Hidrobiolohichnoi Stantsii 1938, 4, 1. (In Ukrainian) [Google Scholar]
- Rajwa-Kuligiewicz, A.; Bialik, R.J.; Rowiński, P.M. Dissolved oxygen and water temperature dynamics in lowland rivers over various timescales. J. Hydrol. Hydromech. 2015, 63, 353–363. [Google Scholar] [CrossRef] [Green Version]
- Wukhan, O.O.; Osadcha, N.M. Characteristics of the oxygen regime of surface waters of the basin of the Seversky Donets. Res. Pap. UkrNDGMI 2010, 259, 199–216. (In Ukrainian) [Google Scholar]
- Widodo, T.; Budiastuti, M.; Komariah, K. Water quality and pollution index in Grenjeng River, Boyolali Regency, Indonesia Caraka Tani. J. Sustain. Agric. 2019, 34, 150–161. [Google Scholar]
- Vasenko, O.G.; Lungu, M.L.; Ilievska, Y.A.; Klimov, O.V. Comprehensive Expeditionary Research of the Ecological Condition of Water Bodies of the Udy River Basin (Siversky Donets Sub-Basin); Ryder Publishing House: Kharkiv, Ukraine, 2006; pp. 1–154. (In Ukrainian) [Google Scholar]
- Wukhan, O.O.; Osadchy, V.I.; Osadcha, N.M.; Manchenko, A.P. Features of formation of chemical composition of surface waters of the Siversky Donets basin. Sci. Work. UHMI 2002, 250, 262–279. (In Ukrainian) [Google Scholar]
- Dedić, A.; Gerhardt, A.; Kelly, M.G.; Stanić-Koštroman, S.; Šiljeg, M.; Kalamujić, S.B.; Kamberović, J.; Mateljak, Z.; Pešić, V.; Vučković, I.; et al. Innovative methods and approaches for WFD: Ideas to fill knowledge gaps in science and policy. Water Solut. 2020, 3, 30–42. [Google Scholar]
- Lyaluk, N.M.; Petraeva, M.Y. Algae in the plankton of the medium reaches of the Siversky Donets river (Ukraine). Algologiya 2014, 24, 380–384. (In Russian) [Google Scholar]
- Kiss, K.T. Phytoplankton studies in the Szigetköz Section of the Danube during 1981–1982. Arch. Hydrobiol. Suppl. 78 Algol. Stud. 1987, 47, 247–273. [Google Scholar]
- Kiss, K.T.; Genkal, S.I. Phytoplankton of the Danube’s Reservoirs from Germany to Hungary. 31; Limnologische Berichte Donau 1996, Bd.I-Wissenschaftliche Referate; Konferenz IAD: Baja/Ungarn, Hungary, 1996; pp. 133–148. [Google Scholar]
- Lee, H.-J.; Park, H.-K.; Cheon, S.-U. Effects of Weir Construction on Phytoplankton Assemblages and Water Quality in a Large River System. Int. J. Environ. Res. Public Health 2018, 15, 2348. [Google Scholar] [CrossRef] [Green Version]
- Oseji, O.F.; Fan, C.; Chigbu, P. Composition and Dynamics of Phytoplankton in the Coastal Bays of Maryland, USA, Revealed by Microscopic Counts and Diagnostic Pigments Analyses. Water 2019, 11, 368. [Google Scholar] [CrossRef] [Green Version]
- Sirenko, L.A.; Gavrylenko, M.Y. Water “Bloom” and Eutrophication; Naukova Dumka: Kyiv, Ukraine, 1978; pp. 1–232. (In Russian) [Google Scholar]
- Kiss, K.T.; Genkal, S.I. Winter blooms of centric diatoms in the River Danube and in its side-arms near Budapest (Hungary). Hydrobiologia 1993, 269, 317–325. [Google Scholar] [CrossRef]
- Li, H.; Chen, H.; Gu, X.; Mao, Z.; Zeng, Q.; Ding, H. Dynamics of Cyanobacteria and Related Environmental Drivers in Freshwater Bodies Affected by Mitten Crab Culturing: A Study of Lake Guchenghu, China. Water 2019, 11, 2468. [Google Scholar] [CrossRef] [Green Version]
- Singh, M.; Lodha, P.; Singh, G.P.; Singh, R. Studies on diatom diversity in response to abiotic factors in Mawatha lake of Jaipur, Rajasthan. Int. J. Life Sci. Pharma Res. 2011, 1, 29–37. [Google Scholar]
- Li, R.; Xu, Q.J.; Zhang, G.S.; Cheng, X.Y. Effects of various total dissolved solids (TDS) on the growth phytoplankton. Res. Env. Sci. 2013, 26, 1072–1078. [Google Scholar]
- Sui, F.; Zang, S.; Fan, Y.; Ye, H. Effects of Different Saline-Alkaline Conditions on the Characteristics of Phytoplankton Communities in the Lakes of Songnen Plain, China. PLoS ONE 2016, 11, e0164734. [Google Scholar] [CrossRef] [Green Version]



| Site | Coordinates | Type of Waterbody | Water Tempera-ture, °C | pH | Dissolved Oxygen (DO), mg·L−1 | DO, % | Conductivity, mS cm−1 | Salinity, g·L−1 (ppt) | Total Dissolved Solids, ppm | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | N 49°58′16.4″ | E 37°51′07.8″ | UA_R_16_XL_1_Si | 26.0 | 6.53 | 5.7 | 67.4 | 749 | 0.44 | 375 |
| 2 | N 49°16′15.6″ | E 39°35′16.4″ | UA_R_16_M_1_Ca | 23.8 | 6.18 | 6.8 | 80 | 2240 | 1.18 | 1621 |
| 3 | N 49°35′27.4″ | E 38°12′08.5″ | UA_R_16_L_1_Si | 23.0 | 6.29 | 5.9 | 69 | 1388 | 0.87 | 696 |
| 4 | N 49°32′52.4″ | E 38°09′34.2″ | UA_R_16_M_1_Si | 23.2 | 6.2 | 7.2 | 74.6 | 2000 | 1.04 | 763 |
| 5 | N 48°52′55.3″ | E 37°54′04.3″ | UA_R_16_XL_1_Si | 23.6 | 6.48 | 6.5 | 83.2 | 1292 | 0.88 | 649 |
| 6 | N 48°58′19.6″ | E 37°58′51.8″ | UA_R_16_M_1_Si | 23.0 | 6.47 | 6.5 | 74 | 2380 | 1.26 | 1190 |
| 7 | N 48°25′46.4″ | E 37°40′31.5″ | UA_R_16_M_1_Si | 26.5 | 6.07 | 6.3 | 74.1 | 4800 | 2.65 | 2410 |
| 8 | N 49°53′09.5″ | E 36°43′18.5″ | UA_R_16_S_1_Si | 25.9 | 6.28 | 4.9 | 61.9 | 782 | 0.47 | 392 |
| 9 | N 49°47′36.1″ | E 36°19′49.2″ | UA_R_16_M_1_Si | 24.5 | 6.22 | 7.6 | 81 | 1313 | 0.8 | 657 |
| 10 | N 48°25′21.4″ | E 37°40′47.5″ | UA_R_16_M_1_Si | 20.8 | 7.88 | 14.3 | 155 | 5660 | 3.17 | 2830 |
| 11 | N 48°57′59.0″ | E 37°36′53.0″ | UA_R_16_XL_1_Si | 22.8 | 8.06 | 7.0 | 85 | 832 | 0.51 | 416 |
| 12 | N 48°55′10.9″ | E 39°37′45.1″ | UA_R_16_S_1_Ca | 13.8 | 7.75 | 11.6 | 117 | 2590 | 1.37 | 1290 |
| 13 | N 48°52′40.9″ | E 39°49′26.3″ | UA_R_16_L_1_Si | 18.2 | 7.85 | 10.1 | 108 | 2390 | 1.27 | 1190 |
| 14 | N 49°07′35.9″ | E 39°36′29.0″ | UA_R_16_L_1_Ca | 19.0 | 7.96 | 11.3 | 130 | 2070 | 1.09 | 1040 |
| 15 | N 49°17′20.2″ | E 40°06′22.1″ | UA_R_16_S_1_Ca | 12.0 | 7.68 | 7.5 | 68.4 | 2640 | 1.42 | 1330 |
| 16 | N 49°21′00.9″ | E 40°04′13.4″ | UA_R_16_S_1_Ca | 14.5 | 7.42 | 4.2 | 43 | 3170 | 1.72 | 1590 |
| 17 | N 49°22′32.1″ | E 38°55′57.3″ | UA_R_16_L_1_Si | 21.3 | 7.95 | 9.0 | 102 | 2040 | 1.08 | 1020 |
| 18 | N 48°59′40.2″ | E 38°29′25.1″ | UA_R_16_L_1_Si | 16.5 | 7.96 | 11.1 | 118 | 2250 | 1.2 | 1130 |
| 19 | N 48°49′57.5″ | E 38°21′18.6″ | UA_R_16_S_1_Ca | 16.5 | 7.91 | 9.8 | 93 | 2700 | 1.45 | 1350 |
| 20 | N 49°37′12.3″ | E 37°00′29.3″ | UA_R_16_M_1_Si | 16.4 | 7.68 | 7.4 | 84.3 | 2020 | 1.07 | 1010 |
| 21 | N 49°55′27.7″ | E 36°47′48.9″ | UA_R_16_M_1_Si | 16.3 | 7.73 | 5.8 | 60.4 | 980 | 0.59 | 485 |
| 22 | N 49°40′58.8″ | E 36°25′26.7″ | UA_R_16_XL_1_Si | 22.9 | 7.56 | 4.7 | 52.7 | 877 | 0.45 | 439 |
| 23 | N 49°44′37.8″ | E 36°09′04.5″ | UA_R_16_L_1_Si | 22.0 | 7.95 | 4.3 | 51.5 | 880 | 0.54 | 441 |
| 24 | N 49°48′45.0″ | E 35°44′44.0″ | UA_R_16_M_1_Si | 15.8 | 7.56 | 2.9 | 30.3 | 892 | 0.56 | 448 |
| Survey Area | N-NH4+, mg·L−1 | N-NO2−, mg·L−1 | N-NO3−, mg·L−1 | P-PO43−, mg·L−1 | CODMn, mg·L−1 | COD, mg O·L−1 | Hardness, mmol·L−1 | Ca2+, mg·L−1 | Mg2+, mg·L−1 | Cl−, mg·L−1 | SO42−, mg·L−1 | HCO3−, mg·L−1 | K+ + Na+, mg·L−1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Oskil river | 0.220 | 0.025 | 0.707 | 0.510 | 7.17 | 16.00 | 6.6 | 104.21 | 17.01 | 53.18 | 196 | 292.8 | 94.50 |
| 2 | Derkul river (near Bilovodsk village) | 0.112 | 0.011 | 0.258 | 0.090 | 9.12 | 56.00 | 13.6 | 190.38 | 49.82 | 350.96 | 288 | 298.9 | 180.00 |
| 3 | Krasna river (near Nyzhnya Duvanka village) | 0.145 | 0.011 | 0.257 | 0.204 | 9.45 | 41.60 | 9.6 | 128.26 | 38.88 | 148.89 | 260 | 341.6 | 140.50 |
| 4 | Krasna river (near Preobrazhennya village) | 0.246 | 0.002 | 0.043 | 0.210 | 9.78 | 54.40 | 12.2 | 160.32 | 51.03 | 209.16 | 296 | 366.0 | 146.75 |
| 5 | Siversky Donets river (near Kryva Luka village) | 0.163 | 0.025 | 0.212 | 0.473 | 10.11 | 35.20 | 8.6 | 112.22 | 36.45 | 159.53 | 224 | 268.4 | 124.25 |
| 6 | Zherebets river | 0.146 | 0.015 | 0.378 | 0.200 | 9.78 | 51.20 | 12.6 | 148.3 | 63.18 | 202.06 | 280 | 311.1 | 100.75 |
| 7 | Bychok river | 0.351 | 0.002 | 0.052 | 0.077 | 18.92 | 102.40 | 26.0 | 308.62 | 128.79 | 478.58 | 206 | 427.0 | 132.00 |
| 8 | Tetlyha river | 0.208 | 0.001 | 0.242 | 0.135 | 8.47 | 16.00 | 6.0 | 84.17 | 21.87 | 265.88 | 104 | 341.6 | 231.75 |
| 9 | Udy river | 14.725 | 0.761 | 0.184 | 2.292 | 14.35 | 22.40 | 7.6 | 104.21 | 29.16 | 88.62 | 32 | 372.1 | 145.75 |
| 10 | Bychok river | 0.220 | 0.002 | 0.046 | 0.101 | 14.08 | 188.77 | 30.0 | 360.72 | 145.8 | 808.26 | 112 | 427.0 | 53.25 |
| 11 | Siversky Donets river (near Mayaky village) | 0.259 | 0.015 | 0.544 | 0.326 | 9.60 | 20.64 | 5.7 | 64.13 | 30.38 | 93.94 | 164 | 219.6 | 99.25 |
| 12 | Chuhynka river | 0.077 | 0.016 | 3.561 | 0.182 | 5.76 | 44.24 | 16 | 256.51 | 38.88 | 152.44 | 348 | 384.3 | 46.25 |
| 13 | Derkul river (near Krasnyy Derkul village) | 0.158 | 0.003 | 0.050 | 0.189 | 10.88 | 94.37 | 15.2 | 148.3 | 94.77 | 421.86 | 252 | 378.2 | 203.75 |
| 14 | Derkul river (near Novoderkul village) | 0.079 | 0.008 | 0.558 | 0.053 | 6.72 | 97.32 | 13.4 | 204.41 | 38.88 | 386.41 | 228 | 335.5 | 193.75 |
| 15 | Cherepakha river | 0.145 | 0.013 | 1.164 | 0.127 | 9.28 | 123.88 | 18.5 | 286.57 | 51.03 | 584.93 | 180 | 359.9 | 191.25 |
| 16 | Milova river | 0.258 | 0.002 | 0.050 | 0.100 | 13.76 | 123.88 | 22.5 | 416.83 | 20.66 | 553.02 | 248 | 475.8 | 151.75 |
| 17 | Aidar river | 0.123 | 0.002 | 0.064 | 0.126 | 9.60 | 23.6 | 12.9 | 112.22 | 88.7 | 322.6 | 252 | 335.5 | 173.75 |
| 18 | Borova river | 0.059 | 0.007 | 0.634 | 0.096 | 7.36 | 47.19 | 14.8 | 210.42 | 52.25 | 265.88 | 308 | 317.2 | 108.00 |
| 19 | Bilenka river | 0.053 | 0.002 | 1.458 | 0.023 | 6.08 | 53.08 | 15.5 | 214.43 | 58.32 | 210.93 | 268 | 420.9 | 73.25 |
| 20 | Serednia Balakliika river | 0.140 | 0.002 | 0.147 | 0.100 | 8.96 | 23.59 | 9.5 | 80.16 | 66.83 | 53.18 | 264 | 555.1 | 165.00 |
| 21 | Velyka Babka river | 0.067 | 0.002 | 0.074 | 0.249 | 9.60 | 20.64 | 7.6 | 112.22 | 24.30 | 42.54 | 212 | 396.5 | 113.00 |
| 22 | Siversky Donets river (near Cheremushne village) | 4.685 | 0.355 | 1.665 | 0.984 | 10.88 | 29.49 | 5.8 | 74.15 | 25.52 | 81.54 | 140 | 317.2 | 115.50 |
| 23 | Mozh river (near Tymchenky village) | 0.274 | 0.004 | 0.049 | 0.147 | 10.88 | 29.49 | 7.7 | 98.2 | 34.02 | 60.26 | 52 | 515.5 | 88.25 |
| 24 | Mozh river (near Fedorivka village) | 0.252 | 0.005 | 0.059 | 0.624 | 14.4 | 41.29 | 7.4 | 90.18 | 35.24 | 63.81 | 50 | 509.4 | 94.75 |
| Sites | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | |
| Phylum (Division) | 3 | 4 | 4 | 3 | 5 | 4 | 6 | 3 | 5 | 3 | 3 | 3 | 2 | 1 | 2 | 1 | 3 | 3 | 3 | 3 | 1 | 4 | 4 | 6 |
| Class | 6 | 8 | 7 | 5 | 7 | 6 | 7 | 4 | 9 | 4 | 5 | 3 | 3 | 2 | 3 | 2 | 4 | 4 | 4 | 5 | 2 | 7 | 5 | 7 |
| Order | 11 | 14 | 10 | 10 | 13 | 9 | 10 | 7 | 12 | 9 | 10 | 8 | 8 | 7 | 7 | 5 | 12 | 8 | 9 | 7 | 5 | 15 | 8 | 8 |
| Family | 14 | 15 | 11 | 10 | 16 | 12 | 11 | 8 | 13 | 12 | 12 | 8 | 9 | 8 | 11 | 6 | 12 | 10 | 9 | 8 | 8 | 20 | 10 | 9 |
| Species | 24 | 19(20) | 19(20) | 14(15) | 25 | 21 | 14 | 16 | 20 | 18 | 19 | 12 | 10 | 10 | 12 | 9 | 12 | 11 | 13(14) | 10 | 10 | 35 | 11 | 13 |
| Species | Sites’ Number |
|---|---|
| Anagnostidinema amphibium (C. Agardh ex Gomont) Strunecký, Bohunická, J.R. Johansen & J. Komárek | 11 |
| Aphanizomenon flosaquae Ralfs ex Bornet & Flahault | 9, 11, 12 |
| Aphanocapsa planctonica (G.M. Smith) Komárek & Anagnostidis | 17 |
| Aulacoseira granulata (Ehrenberg) Simonsen | 13, 15, 16, 18–20 |
| Cocconeis pediculus Ehrenberg | 2, 4, 6 |
| Cocconeis placentula Ehrenberg | 13, 14, 18 |
| Coelastrum pseudomicroporum Korshikov | 24 |
| Cyclotella meneghiniana Kützing | 3, 5, 7, 10 |
| Desmodesmus communis (E. Hegewald) E. Hegewald | 4 |
| Desmodesmus costato-granulatus (Skuja) E. Hegewald | 8, 22 |
| Dolichospermum flosaquae (Brébisson ex Bornet & Flahault) P. Wacklin, L. Hoffmann & J. Komárek | 10, 23 |
| Gomphonema constrictum Ehrenberg | 10 |
| Halamphora veneta (Kützing) Levkov | 14 |
| Lemmermannia triangularis (Chodat) C. Bock & Krienitz | 5 |
| Melosira varians C. Agardh | 2 |
| Microcystis viridis (A. Braun) Lemmermann | 5 |
| Nitzschia vermicularis (Kützing) Hantzsch | 8 |
| Oscillatoria limosa C. Agardh ex Gomont | 22, 24 |
| Oscillatoria planctonica Woloszynska | 1, 6 |
| Oscillatoria tenuis C. Agardh ex Gomont | 1, 7, 11, 12, 19, 20 |
| Oscillatoria ucrainica Vladimirova | 23 |
| Phormidium terebriforme (C. Agardh ex Gomont) Anagnostidis & Komárek | 3 |
| Raphidocelis sigmoidea Hindák | 8 |
| Rhoicosphenia abbreviata (C. Agardh) Lange-Bertalot | 21 |
| Stephanodiscus hantzschii Grunow | 21 |
| Ulnaria ulna (Nitzsch) Compère | 15 |
| Willea apiculata (Lemmermann) D.M. John, M.J. Wynne & P.M. Tsarenko | 1, 5 |
| Species | Division | Sites’ Number |
|---|---|---|
| Amphora pediculus (Kützing) Grunow | Bacillariophyta | 18 |
| Aphanizomenon flosaquae Ralfs ex Bornet & Flahault | Cyanobacteria | 9 |
| Aphanocapsa planctonica (G.M. Smith) Komárek & Anagnostidis | Cyanobacteria | 17 |
| Aulacoseira granulata (Ehrenberg) Simonsen | Bacillariophyta | 19, 20 |
| Campylodiscus noricus Ehrenberg ex Kützing | Bacillariophyta | 19 |
| Cocconeis pediculus Ehrenberg | Bacillariophyta | 2–4, 6, 10 |
| Cocconeis placentula Ehrenberg | Bacillariophyta | 1, 13, 18, 23, 24 |
| Cocconeis placentula var. euglypta (Ehrenberg) Grunow | Bacillariophyta | 6 |
| Coelastrum pseudomicroporum Korshikov | Chlorophyta | 24 |
| Cyclotella meneghiniana Kützing | Bacillariophyta | 5, 7, 10 |
| Diatoma tenuis C. Agardh | Bacillariophyta | 8 |
| Gymnodinium paradoxum A.J. Schilling | Dinophyta | 5 |
| Halamphora veneta (Kützing) Levkov | Bacillariophyta | 12, 14 |
| Mallomonas sp. | Ochrophyta | 7, 8, 20 |
| Melosira varians C. Agardh | Bacillariophyta | 2, 4 |
| Navicula recens (Lange-Bertalot) Lange-Bertalot | Bacillariophyta | 20 |
| Navicula vulpina Kützing | Bacillariophyta | 16 |
| Nitzschia acicularis (Kützing) W. Smith | Bacillariophyta | 12 |
| Nitzschia vermicularis (Kützing) Hantzsch | Bacillariophyta | 8 |
| Oscillatoria tenuis C. Agardh ex Gomont | Cyanobacteria | 11 |
| Oscillatoria ucrainica Vladimirova | Cyanobacteria | 23 |
| Peridiniopsis quadridens (F. Stein) Bourrelly | Dinophyta | 22 |
| Rhoicosphenia abbreviata (C. Agardh) Lange-Bertalot | Bacillariophyta | 21 |
| Sphaerocystis planctonica (Korshikov) Bourrelly | Chlorophyta | 1 |
| Surirella librile (Ehrenberg) Ehrenberg | Bacillariophyta | 14 |
| Trachelomonas volvocina (Ehrenberg) Ehrenberg | Euglenophyta | 23 |
| Ulnaria acus (Kützing) Aboal | Bacillariophyta | 13, 14, 16 |
| Ulnaria biceps (Kützing) Compère | Bacillariophyta | 15 |
| Ulnaria ulna (Nitzsch) Compère | Bacillariophyta | 10, 13, 15, 16, 18, 21 |
| Willea apiculata (Lemmermann) D.M. John, M.J. Wynne & P.M. Tsarenko | Chlorophyta | 24 |
| Environmental Variable | Variability Explained | F-Value | p-Value |
|---|---|---|---|
| CODMn | 8.6 | 2.70 | 0.04 |
| TDS | 8.1 | 2.54 | 0.05 |
| NO2− | 9.8 | 3.09 | 0.02 |
| HCO3− | 10.3 | 3.25 | 0.02 |
| COD | 9.2 | 2.90 | 0.03 |
| Variability Explained | F-Value | p-Value | |
|---|---|---|---|
| Axis 1 | 24.4 | 7.67 | 0.007 |
| Axis 2 | 15.8 | 4.97 | 0.015 |
| Distance from CCA Plot Origin | Bacillariophyta | Chlorophyta | Cyanobacteria | Site (See Table 2) |
|---|---|---|---|---|
| 0.47 | 12 | 10 | 2 | 1 |
| 0.57 | 16 | 16 | 2 | 22 |
| 1.05 | 16 | 1 | 1 | 10 |
| 1.09 | 15 | 2 | 1 | 3 |
| 1.16 | 15 | 4 | 1 | 6 |
| 1.33 | 5 | 10 | 4 | 11 |
| 1.47 | 12 | 1 | 1 | 19 |
| 1.55 | 7 | 1 | 2 | 7 |
| 1.57 | 5 | 8 | 4 | 9 |
| 1.59 | 9 | 1 | 0 | 18 |
| 1.61 | 7 | 1 | 2 | 23 |
| 1.64 | 7 | 14 | 2 | 5 |
| 1.70 | 1 | 8 | 1 | 24 |
| 1.93 | 13 | 1 | 0 | 4 |
| 2.15 | 1 | 10 | 1 | 17 |
| 2.29 | 10 | 5 | 0 | 8 |
| 2.32 | 8 | 2 | 0 | 13 |
| 2.37 | 13 | 4 | 0 | 2 |
| 2.53 | 11 | 0 | 0 | 15 |
| 2.59 | 9 | 1 | 2 | 12 |
| 3.25 | 10 | 0 | 0 | 14 |
| 3.29 | 8 | 0 | 1 | 20 |
| 4.68 | 10 | 0 | 0 | 21 |
| 5.56 | 9 | 0 | 0 | 16 |
| Correlation with distance: | −0.19 | −0.50 ** | −0.48 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bilous, O.; Afanasyev, S.; Lietytska, O.; Manturova, O.; Polishchuk, O.; Nezbrytska, I.; Pohorielova, M.; Barinova, S. Preliminary Assessment of Ecological Status of the Siversky Donets River Basin (Ukraine) Based on Phytoplankton Parameters and Its Verification by Other Biological Data. Water 2021, 13, 3368. https://doi.org/10.3390/w13233368
Bilous O, Afanasyev S, Lietytska O, Manturova O, Polishchuk O, Nezbrytska I, Pohorielova M, Barinova S. Preliminary Assessment of Ecological Status of the Siversky Donets River Basin (Ukraine) Based on Phytoplankton Parameters and Its Verification by Other Biological Data. Water. 2021; 13(23):3368. https://doi.org/10.3390/w13233368
Chicago/Turabian StyleBilous, Olena, Sergey Afanasyev, Olena Lietytska, Oksana Manturova, Oleksandr Polishchuk, Inna Nezbrytska, Maryna Pohorielova, and Sophia Barinova. 2021. "Preliminary Assessment of Ecological Status of the Siversky Donets River Basin (Ukraine) Based on Phytoplankton Parameters and Its Verification by Other Biological Data" Water 13, no. 23: 3368. https://doi.org/10.3390/w13233368








