After the Storm: Fate and Leaching of Particulate Nitrogen (PN) in the Fluvial Network and the Influence of Watershed Sources and Moisture Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Incubation Setup and Sampling
2.2. Pore Water and Sediment Analysis and Microbial Gene Quantification
2.3. Data Analysis and Comparisons Performed
3. Results
3.1. Moisture Variation for Incubation Regimes
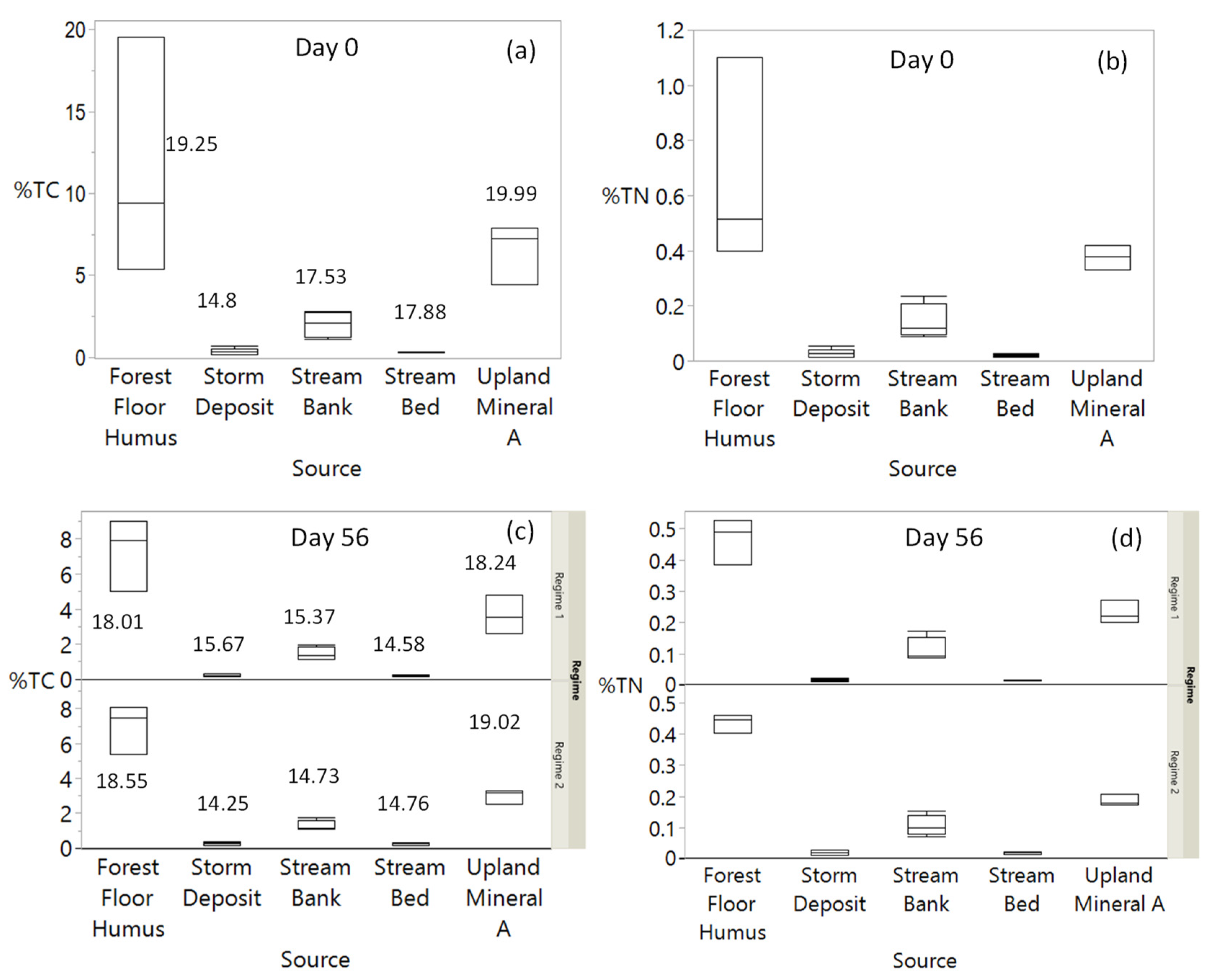
3.2. Changes in Sediment %TC and TN
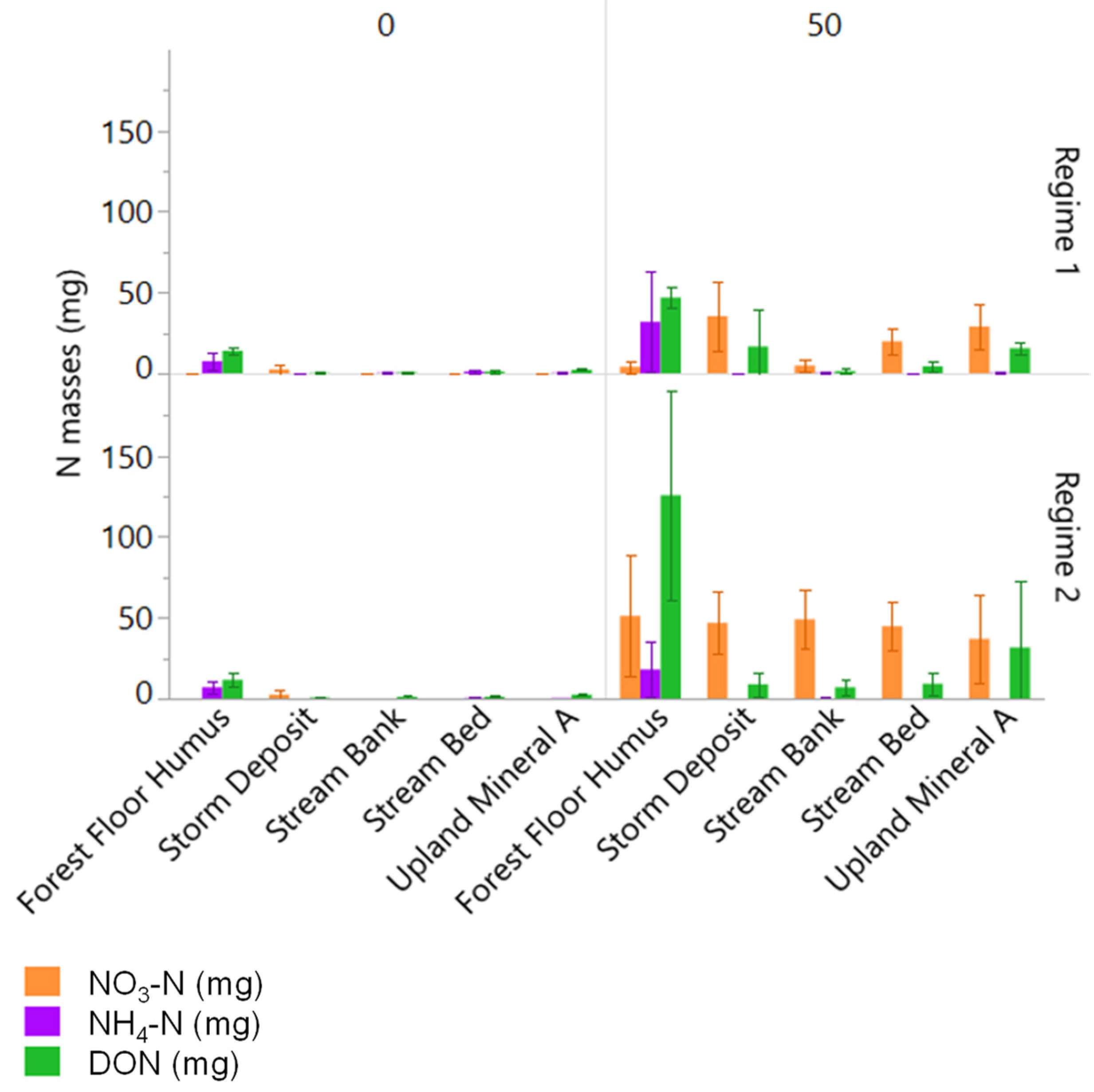
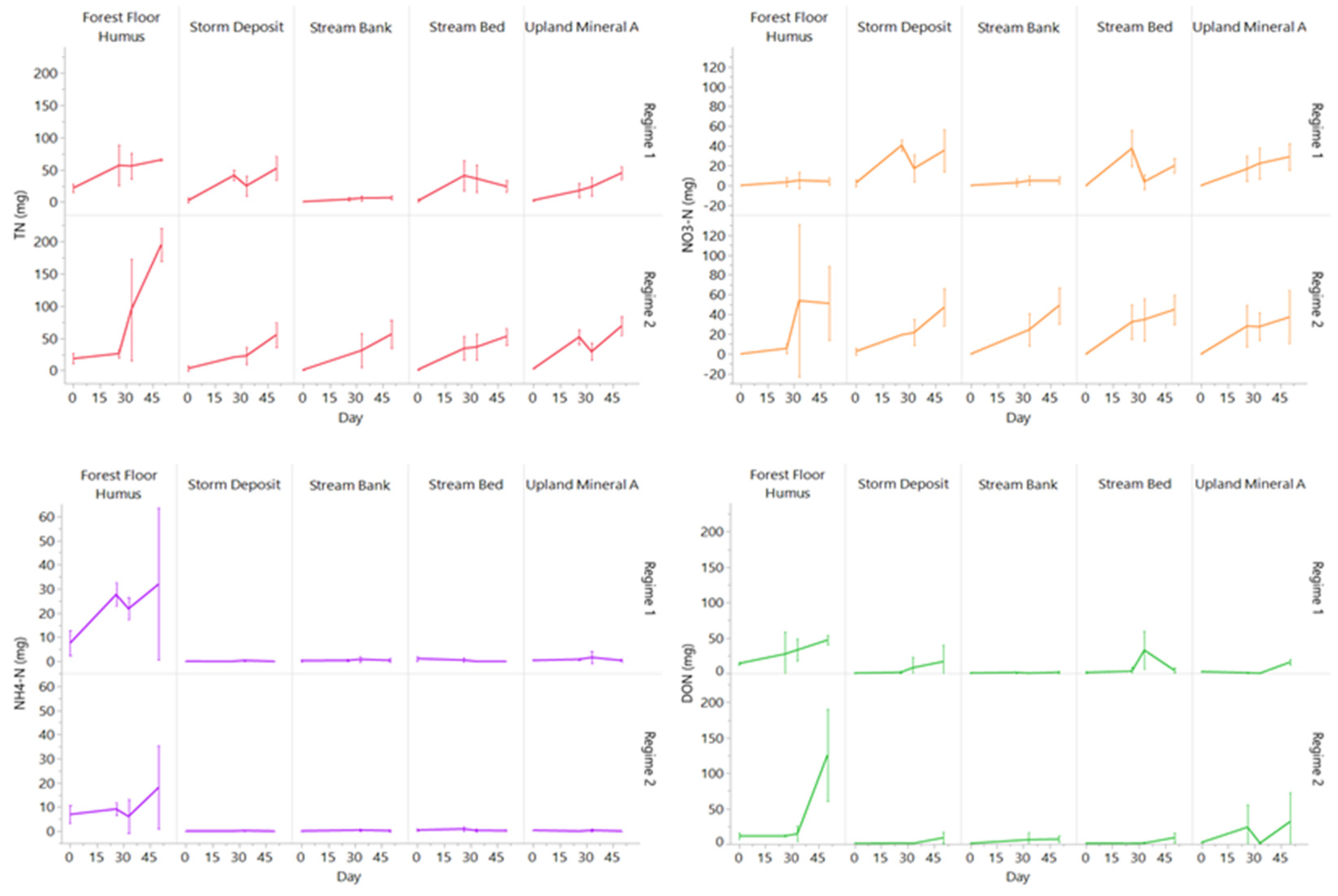
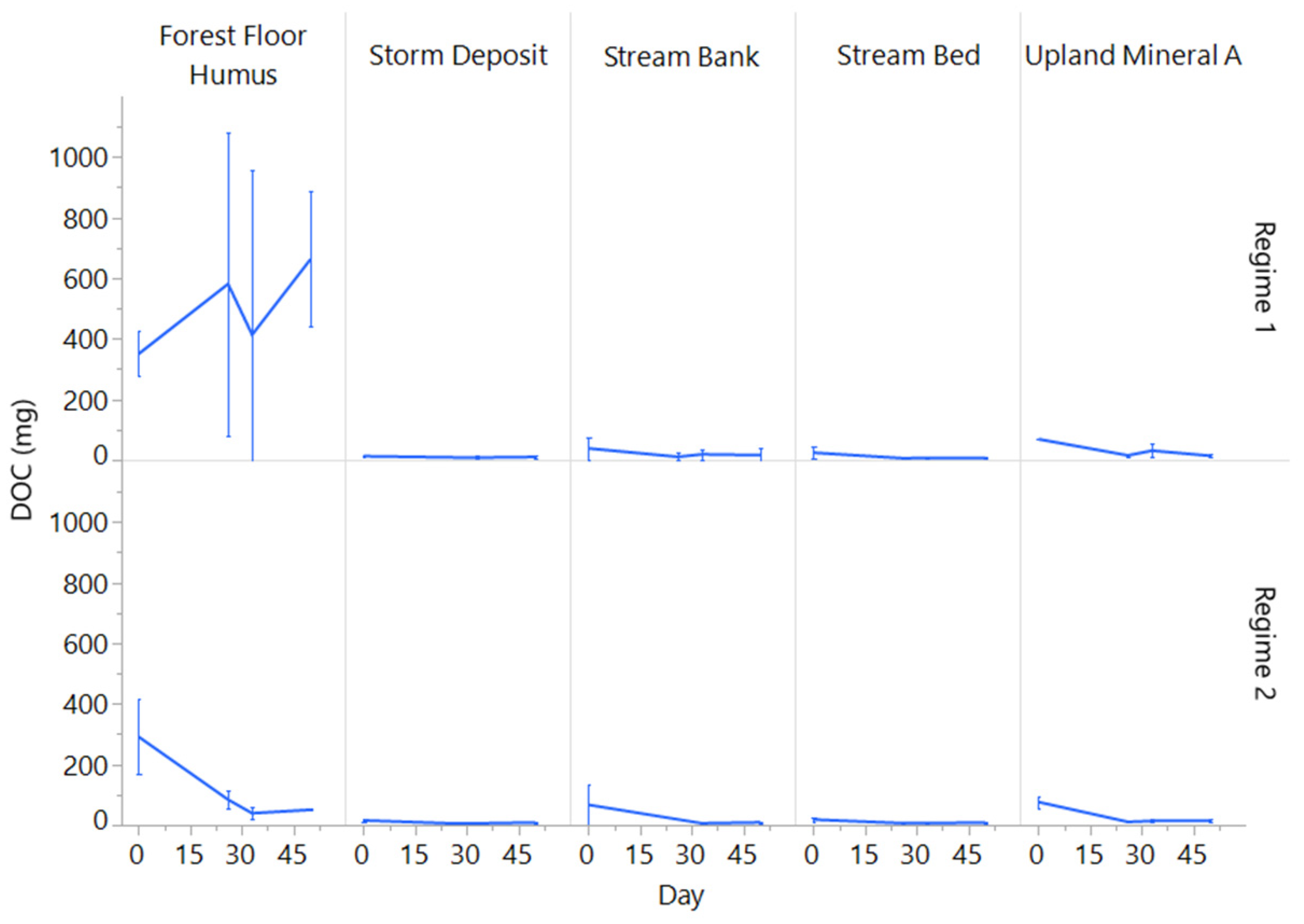
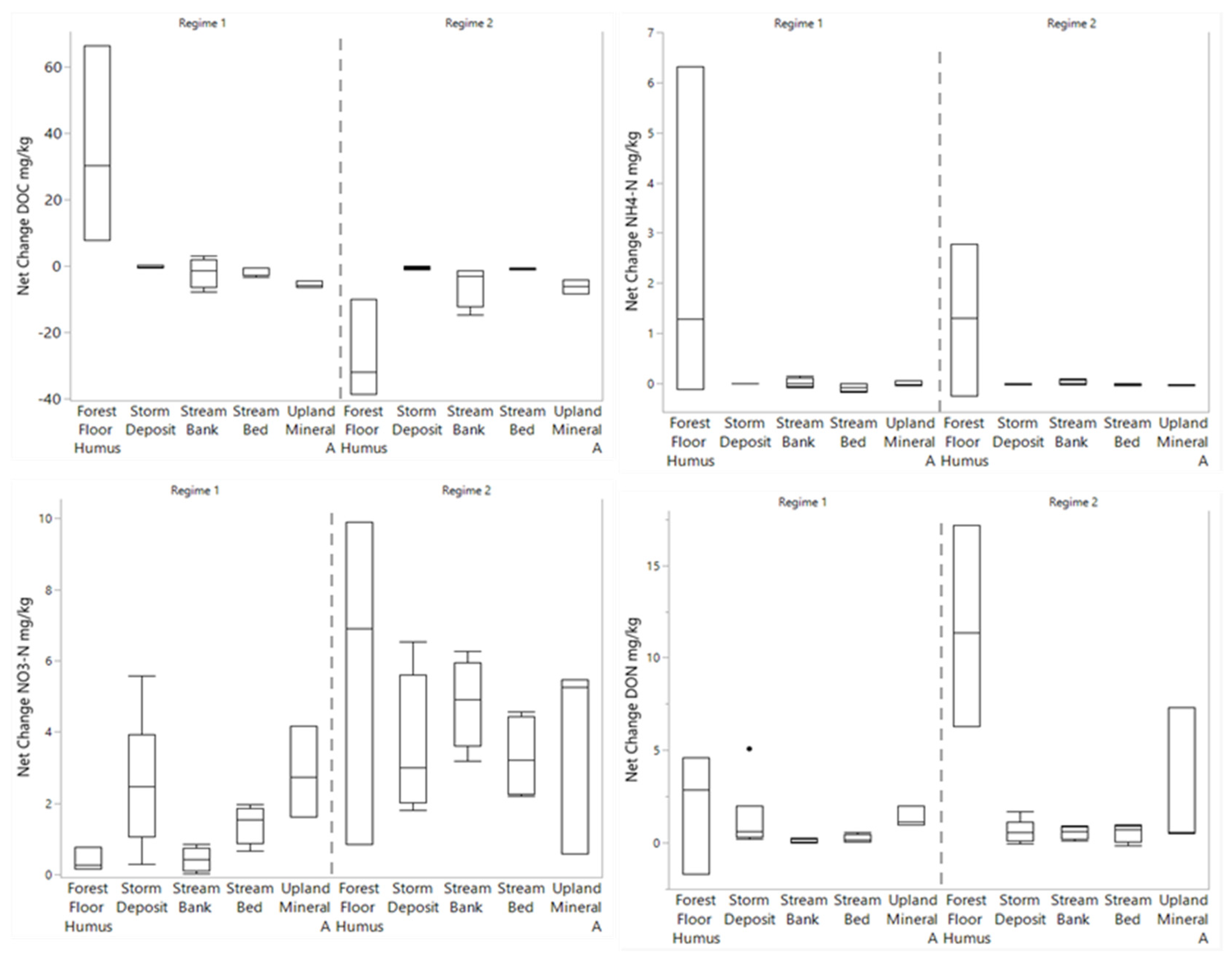
3.3. Changes in Porewater N and C
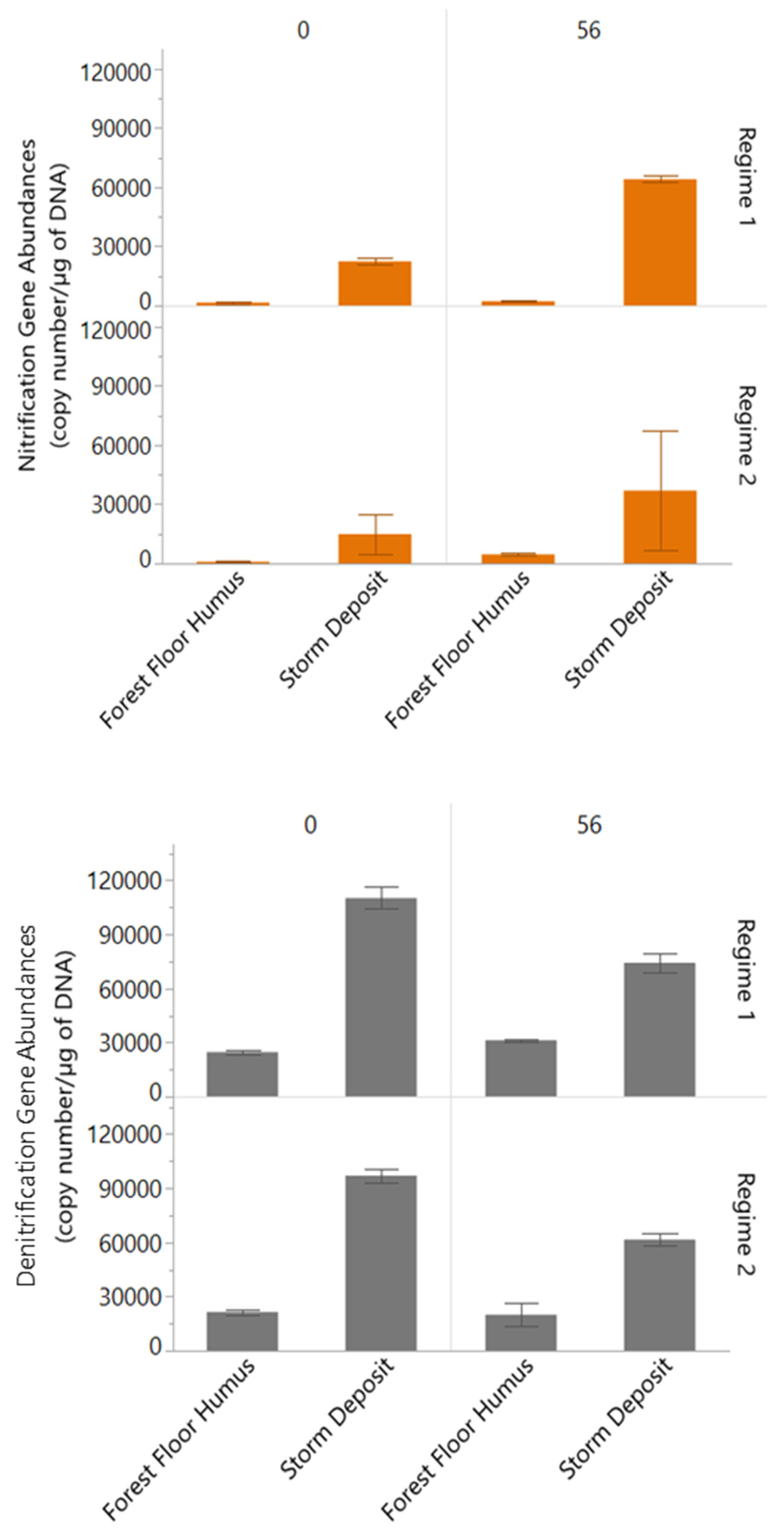
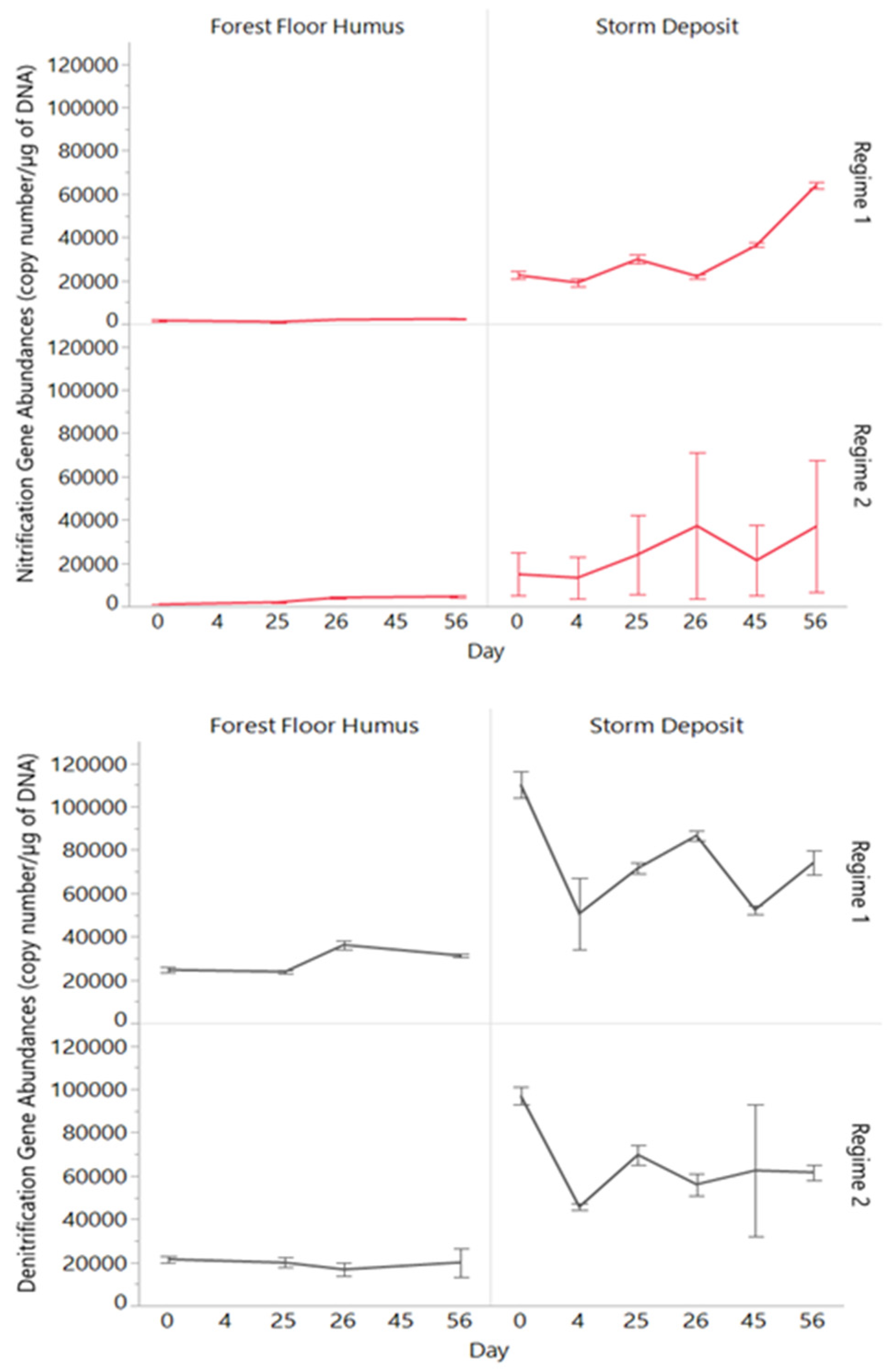
3.4. Nitrification and Denitrification Gene Abundances
4. Discussion
4.1. Role of Sources and Moisture Conditions for Processing and Leaching of PN
4.2. Broader Environmental Implications and Caveats
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Howarth, R.; Swaney, D.; Billen, G.; Garnier, J.; Hong, B.; Humborg, C.; Johnes, P.; Mörth, C.; Marino, R. Nitrogen fluxes from the landscape are controlled by net anthropogenic nitrogen inputs and by climate. Front. Ecol. Environ. 2012, 10, 37–43. [Google Scholar] [CrossRef] [Green Version]
- Baron, J.; Hall, E.; Nolan, B.; Finlay, J.; Bernhardt, E.; Harrison, J.; Chan, F.; Boyer, E. The interactive effects of excess reactive nitrogen and climate change on aquatic ecosystems and water resources of the United States. Biogeochemistry 2013, 114, 71–92. [Google Scholar] [CrossRef]
- Alvarez-Cobelas, M.; Angeler, D.G.; Sánchez-Carrillo, S.; Almendros, G. Worldwide view of organic carbon export from catchments. Biogeochemistry 2010, 107, 275–293. [Google Scholar] [CrossRef]
- Bertani, I.; Bhatt, G.; Shenk, G.W.; Linker, L.C. Quantifying the Response of Nitrogen Speciation to Hydrology in the Chesapeake Bay Watershed Using a Multilevel Modeling Approach. J. Am. Water Resour. Assoc. 2021. [Google Scholar] [CrossRef]
- Lusk, M.G.; Toor, G.S.; Inglett, P.W. Organic nitrogen in residential stormwater runoff: Implications for stormwater management in urban watersheds. Sci. Total Environ. 2020, 707, 135962. [Google Scholar] [CrossRef] [PubMed]
- Katz, B.G. Nitrogen Overload: Environmental Degradation, Ramifications, and Economic Costs; John Wiley & Sons: Hoboken, NJ, USA, 2020; p. 272. [Google Scholar]
- Dhillon, G.S.; Inamdar, S. Extreme storms and changes in particulate and dissolved organic carbon in runoff: Entering uncharted waters? Geophys. Res. Lett. 2013, 40, 1322–1327. [Google Scholar] [CrossRef]
- Inamdar, S.; Dhillon, G.; Singh, S.; Parr, T.; Qin, Z. Particulate nitrogen exports in stream runoff exceed dissolved nitrogen forms during large tropical storms in a temperate, headwater, forested watershed. J. Geophys. Res. Biogeosci. 2015, 120, 1548–1566. [Google Scholar] [CrossRef] [Green Version]
- Taylor, P.G.; Wieder, W.R.; Weintraub, S.; Cohen, S.; Cleveland, C.C.; Townsend, A.R. Organic forms dominate hydrologic nitrogen export from a lowland tropical watershed. Ecology 2015, 96, 1229–1241. [Google Scholar] [CrossRef] [Green Version]
- Lloret, E.; Dessert, C.; Pastor, L.; Lajeunesse, E.; Crispi, O.; Gaillardet, J.; Benedetti, M.F. Dynamic of particulate and dissolved organic carbon in small volcanic mountainous tropical watersheds. Chem. Geol. 2013, 351, 229–244. [Google Scholar] [CrossRef]
- Gao, Q.; Tao, Z.; Yao, G.; Ding, J.; Liu, Z.; Liu, K. Elemental and isotopic signatures of particulate organic carbon in the Zengjiang River, southern China. Hydrol. Process. 2007, 21, 1318–1327. [Google Scholar] [CrossRef]
- Gomez, B.; Baisden, W.T.; Rogers, K.M. Variable composition of particle-bound organic carbon in steepland river systems. J. Geophys. Res. Earth Surf. 2010, 115. [Google Scholar] [CrossRef]
- Jung, B.; Lee, H.; Jeong, J.; Owen, J.; Kim, B.; Meusburger, K.; Alewell, C.; Gebauer, G.; Shope, C.; Park, J. Storm pulses and varying sources of hydrologic carbon export from a mountainous watershed. J. Hydrol. 2012, 440–441, 90–101. [Google Scholar] [CrossRef]
- Johnson, E.R.; Inamdar, S.; Kan, J.; Vargas, R. Particulate Organic Matter Composition in Stream Runoff Following Large Storms: Role of POM Sources, Particle Size, and Event Characteristics. J. Geophys. Res. Biogeosci. 2018, 123, 660–675. [Google Scholar] [CrossRef]
- Rowland, R.; Inamdar, S.; Parr, T. Evolution of particulate organic matter (POM) along a headwater drainage: Role of sources, particle size class, and storm magnitude. Biogeochemistry 2017, 133, 181–200. [Google Scholar] [CrossRef]
- Coynel, A.; Etcheber, H.; Abril, G.; Maneux, E.; Dumas, J.; Hurtrez, J. Contribution of Small Mountainous Rivers to Particulate Organic Carbon Input in the Bay of Biscay. Biogeochemistry 2005, 74, 151–171. [Google Scholar] [CrossRef]
- Oeurng, C.; Sauvage, S.; Coynel, A.; Maneux, E.; Etcheber, H.; Sánchez-Pérez, J. Fluvial transport of suspended sediment and organic carbon during flood events in a large agricultural catchment in southwest France. Hydrol. Process. 2011, 25, 2365–2378. [Google Scholar] [CrossRef] [Green Version]
- Vidon, P.; Allan, C.; Burns, D.; Duval, T.P.; Gurwick, N.; Inamdar, S.; Lowrance, R.; Okay, J.; Scott, D.; Sebestyen, S. Hot Spots and Hot Moments in Riparian Zones: Potential for Improved Water Quality Management1. J. Am. Water Resour. Assoc. 2010, 46, 278–298. [Google Scholar] [CrossRef]
- McClain, M.E.; Boyer, E.W.; Dent, C.L.; Gergel, S.E.; Grimm, N.B.; Groffman, P.M.; Hart, S.C.; Harvey, J.W.; Johnston, C.A.; Mayorga, E.; et al. Biogeochemical Hot Spots and Hot Moments at the Interface of Terrestrial and Aquatic Ecosystems. Ecosystems 2003, 6, 301–312. [Google Scholar] [CrossRef]
- Bernhardt, E.S.; Likens, G.E.; Hall, R.O.; Buso, D.C.; Fisher, S.G.; Burton, T.M.; Meyer, J.L.; McDowell, W.H.; Mayer, M.S.; Bowden, W.B.; et al. Can’t See the Forest for the Stream? In-stream Processing and Terrestrial Nitrogen Exports. BioScience 2005, 55, 219–230. [Google Scholar] [CrossRef]
- Tank, J.L.; Rosi-Marshall, E.J.; Griffiths, N.A.; Entrekin, S.A.; Stephen, M.L. A review of allochthonous organic matter dynamics and metabolism in streams. J. N. Am. Benthol. Soc. 2010, 29, 118–146. [Google Scholar] [CrossRef] [Green Version]
- Peterson, B.J.; Wollheim, W.M.; Mulholland, P.J.; Webster, J.R.; Meyer, J.L.; Tank, J.L.; Martí, E.; Bowden, W.B.; Valett, H.M.; Hershey, A.E.; et al. Control of Nitrogen Export from Watersheds by Headwater Streams. Science 2001, 292, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Seitzinger, S.; Harrison, J.A.; Böhlke, J.K.; Bouwman, A.F.; Lowrance, R.; Peterson, B.; Tobias, C.; Drecht, G.V. Denitrification Across Landscapes and Waterscapes: A Synthesis. Ecol. Appl. 2006, 16, 2064–2090. [Google Scholar] [CrossRef] [Green Version]
- Bernhardt, E.S.; Likens, G.E.; Buso, D.C.; Driscoll, C.T. In-stream uptake dampens effects of major forest disturbance on watershednitrogen export. Proc. Natl. Acad. Sci. USA 2003, 100, 10304–10308. [Google Scholar] [CrossRef] [Green Version]
- Triska, F.J.; Duff, J.H.; Avanzino, R.J. Patterns of hydrological exchange and nutrient transformation in the hyporheic zone of a gravel-bottom stream: Examining terrestrial—Aquatic linkages. Freshw. Biol. 1993, 29, 259–274. [Google Scholar] [CrossRef]
- Storey, R.G.; Fulthorpe, R.R.; Williams, D.D. Perspectives and predictions on the microbial ecology of the hyporheic zone. Freshw. Biol. 1999, 41, 119–130. [Google Scholar] [CrossRef] [Green Version]
- Mulholland, P.J. The Importance of In-stream Uptake for Regulating Stream Concentrations and Outputs of N And P from a Forested Watershed: Evidence from Long-term Chemistry Records for Walker Branch Watershed. Biogeochemistry 2004, 70, 403–426. [Google Scholar] [CrossRef]
- Birch, H.F. The effect of soil drying on humus decomposition and nitrogen availability. Plant Soil 1958, 10, 9–31. [Google Scholar] [CrossRef]
- Cui, M.; Caldwell, M.M. A large ephemeral release of nitrogen upon wetting of dry soil and corresponding root responses in the field. Plant Soil 1997, 191, 291–299. [Google Scholar] [CrossRef]
- Jarvis, P.; Rey, A.; Petsikos, C.; Wingate, L.; Rayment, M.; Pereira, J.; Banza, J.; David, J.; Miglietta, F.; Borghetti, M.; et al. Drying and wetting of Mediterranean soils stimulates decomposition and carbon dioxide emission: The “Birch effect”. Tree Physiol. 2007, 27, 929–940. [Google Scholar] [CrossRef] [Green Version]
- Franzluebbers, K.; Weaver, R.W.; Juo, A.S.R.; Franzluebbers, A.J. Carbon and nitrogen mineralization from cowpea plants part decomposing in moist and in repeatedly dried and wetted soil. Soil Biol. Biochem. 1994, 26, 1379–1387. [Google Scholar] [CrossRef]
- Karl, T.R.; Melillo, J.M.; Peterson, T.C. Global Climate Change Impacts in the United States; Cambridge University Press: Cambridge, UK, 2009; p. 188. [Google Scholar]
- Blackmer, G.C. Preliminary Bedrock Geologic Map of a Portion of the Wilmington 30- by 60-Minute Quadrangle, Southeastern Pennsylvania. 2005. Available online: http://maps.dcnr.pa.gov/publications/Default.aspx?id=94 (accessed on 9 November 2021).
- Krieg, C.S. The Fate of Particulate Nitrogen in Fluvial Systems. Master’s Thesis, University of Delaware, Newark, DE, USA, 2018. [Google Scholar]
- Francis, C.A.; Roberts, K.J.; Beman, J.M.; Santoro, A.E.; Oakley, B.B. Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. Proc. Natl. Acad. Sci. USA 2005, 102, 14683–14688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rotthauwe, J.H.; Witzel, K.P.; Liesack, W. The ammonia monooxygenase structural gene amoA as a functional marker: Molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl. Env. Microbiol. 1997, 63, 4704–4712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Braker, G.; Fesefeldt, A.; Witzel, K.P. Development of PCR primer systems for amplification of nitrite reductase genes (nirK and nirS) to detect denitrifying bacteria in environmental samples. Appl. Env. Microbiol. 1998, 64, 3769–3775. [Google Scholar] [CrossRef] [Green Version]
- Mosier, A.C.; Francis, C.A. Denitrifier abundance and activity across the San Francisco Bay estuary. Environ. Microbiol. Rep. 2010, 2, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Stelzer, R.S.; Scott, J.T.; Bartsch, L.A. Buried particulate organic carbon stimulates denitrification and nitrate retention in stream sediments at the groundwater–surface water interface. Freshw. Sci. 2015, 34, 161–171. [Google Scholar] [CrossRef]
- Thamdrup, B. New Pathways and Processes in the Global Nitrogen Cycle. Annu. Rev. Ecol. Evol. Syst. 2012, 43, 407–428. [Google Scholar] [CrossRef]
- Greenan, C.M.; Moorman, T.B.; Kaspar, T.C.; Parkin, T.B.; Jaynes, D.B. Comparing Carbon Substrates for Denitrification of Subsurface Drainage Water. J. Environ. Qual. 2006, 35, 824–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnes, R.T.; Smith, R.L.; Aiken, G.R. Linkages between denitrification and dissolved organic matter quality, Boulder Creek watershed, Colorado. J. Geophys. Res. Biogeosci. 2012, 117. [Google Scholar] [CrossRef] [Green Version]
- Stelzer, R.; Thad Scott, J.; Bartsch, L.; Parr, T. Particulate organic matter quality influences nitrate retention and denitrification in stream sediments: Evidence from a carbon burial experiment. Biogeochemistry 2014, 119, 387–402. [Google Scholar] [CrossRef]
- Yoshimura, C.; Fujii, M.; Omura, T.; Tockner, K. Instream release of dissolved organic matter from coarse and fine particulate organic matter of different origins. Biogeochemistry 2010, 100, 151–165. [Google Scholar] [CrossRef]
- Follett, R.F. Chapter 2-Transformation and Transport Processes of Nitrogen in Agricultural Systems. In Nitrogen in the Environment, 2nd ed.; Hatfield, J.L., Follett, R.F., Eds.; Academic Press: San Diego, CA, USA, 2008; pp. 19–50. [Google Scholar]
- Utomo, W.H.; Dexter, A.R. Changes in soil aggregate water stability induced by wetting and drying cycles in non-saturated soil. J. Soil Sci. 1982, 33, 623–637. [Google Scholar] [CrossRef]
- Denef, K.; Six, J.; Paustian, K.; Merckx, R. Importance of macroaggregate dynamics in controlling soil carbon stabilization: Short-term effects of physical disturbance induced by dry–wet cycles. Soil Biol. Biochem. 2001, 33, 2145–2153. [Google Scholar] [CrossRef]
- Bottner, P. Response of microbial biomass to alternate moist and dry conditions in a soil incubated with 14C- and 15N-labelled plant material. Soil Biol. Biochem. 1985, 17, 329–337. [Google Scholar] [CrossRef]
- Stark, J.M.; Firestone, M.K. Mechanisms for Soil Moisture Effects on Activity of Nitrifying Bacteria. Appl. Environ. Microbiol. 1995, 61, 218–221. [Google Scholar] [CrossRef] [Green Version]
- Fierer, N.; Schimel, J.P. Effects of drying–rewetting frequency on soil carbon and nitrogen transformations. Soil Biol. Biochem. 2002, 34, 777–787. [Google Scholar] [CrossRef]
- Fierer, N.; Schimel, J.P.; Holden, P.A. Influence of Drying-Rewetting Frequency on Soil Bacterial Community Structure. Microb. Ecol. 2003, 45, 63–71. [Google Scholar] [CrossRef]
- Borken, W.; Matzner, E. Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Glob. Chang. Biol. 2009, 15, 808–824. [Google Scholar] [CrossRef]
- Franzluebbers, A.J.; Haney, R.L.; Honeycutt, C.W.; Schomberg, H.H.; Hons, F.M. Flush of Carbon Dioxide Following Rewetting of Dried Soil Relates to Active Organic Pools. Soil Sci. Soc. Am. J. 2000, 64, 613–623. [Google Scholar] [CrossRef]
- Kim, D.-G.; Vargas, R.; Bond-Lamberty, B.; Turetsky, M.R. Effects of soil rewetting and thawing on soil gas fluxes: A review of current literature and suggestions for future research. Biogeosciences 2012, 9, 2459–2483. [Google Scholar] [CrossRef] [Green Version]
- Tant, C.J.; Rosemond, A.D.; First, M.R. Stream nutrient enrichment has a greater effect on coarse than on fine benthic organic matter. Freshw. Sci. 2013, 32, 1111–1121. [Google Scholar] [CrossRef] [Green Version]
- Guenet, B.; Danger, M.; Abbadie, L.; Lacroix, G. Priming effect: Bridging the gap between terrestrial and aquatic ecology. Ecology 2010, 91, 2850–2861. [Google Scholar] [CrossRef]
- Guenet, B.; Danger, M.; Harrault, L.; Allard, B.; Jauset-alcala, M.; Bardoux, G.; Benest, D.; Abbadie, L.; Lacroix, G. Fast mineralization of land-born C in inland waters: First experimental evidences of aquatic priming effect. Hydrobiologia 2014, 721, 35–44. [Google Scholar] [CrossRef]
- Chesapeake Bay Program. Chesapeake Bay TMDL Executive Summary; Chesapeake Bay Program Office: Annapolis, MD, USA, 2018. [Google Scholar]
- Baird, D.; Ulanowicz, R.E.; Boynton, W.R. Seasonal Nitrogen Dynamics in Chesapeake Bay: A Network Approach. Estuar. Coast. Shelf Sci. 1995, 41, 137–162. [Google Scholar] [CrossRef]
- Bronk, D.A.; Glibert, P.M.; Malone, T.C.; Banahan, S.; Sahlsten, E. Inorganic and organic nitrogen cycling in Chesapeake Bay: Autotrophic versus heterotrophic processes and relationships to carbon flux. Aquat. Microb. Ecol. 1998, 15, 177–189. [Google Scholar] [CrossRef]
- Howarth, R.W.; Billen, G.; Swaney, D.; Townsend, A.; Jaworski, N.; Lajtha, K.; Downing, J.A.; Elmgren, R.; Caraco, N.; Jordan, T.; et al. Regional Nitrogen Budgets and Riverine N & P Fluxes for the Drainages to the North Atlantic Ocean: Natural and Human Influences. Biogeochemistry 1996, 35, 75–139. [Google Scholar]
- Boynton, W.R.; Garber, J.H.; Summers, R.; Kemp, W.M. Inputs, Transformations, and Transport of Nitrogen and Phosphorus in Chesapeake Bay and Selected Tributaries. Estuaries 1995, 18, 285–314. [Google Scholar] [CrossRef]
- Mbabazi, J.; Inoue, T.; Yokota, K.; Saga, M. Variability of particulate bioavailable phosphorus, particulate organic carbon and nitrogen in agricultural and urban rivers. J. Environ. Chem. Eng. 2019, 7, 103086. [Google Scholar] [CrossRef]
- Jani, J.; Yang, Y.; Lusk, M.G.; Toor, G.S. Composition of nitrogen in urban residential stormwater runoff: Concentrations, loads, and source characterization of nitrate and organic nitrogen. PLoS ONE 2020, 15, e0229715. [Google Scholar] [CrossRef] [PubMed] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krieg, C.; Johnson, E.; Peck, E.; Kan, J.; Inamdar, S. After the Storm: Fate and Leaching of Particulate Nitrogen (PN) in the Fluvial Network and the Influence of Watershed Sources and Moisture Conditions. Water 2021, 13, 3182. https://doi.org/10.3390/w13223182
Krieg C, Johnson E, Peck E, Kan J, Inamdar S. After the Storm: Fate and Leaching of Particulate Nitrogen (PN) in the Fluvial Network and the Influence of Watershed Sources and Moisture Conditions. Water. 2021; 13(22):3182. https://doi.org/10.3390/w13223182
Chicago/Turabian StyleKrieg, Chelsea, Erin Johnson, Erin Peck, Jinjun Kan, and Shreeram Inamdar. 2021. "After the Storm: Fate and Leaching of Particulate Nitrogen (PN) in the Fluvial Network and the Influence of Watershed Sources and Moisture Conditions" Water 13, no. 22: 3182. https://doi.org/10.3390/w13223182
APA StyleKrieg, C., Johnson, E., Peck, E., Kan, J., & Inamdar, S. (2021). After the Storm: Fate and Leaching of Particulate Nitrogen (PN) in the Fluvial Network and the Influence of Watershed Sources and Moisture Conditions. Water, 13(22), 3182. https://doi.org/10.3390/w13223182






