Photo-Catalytic Remediation of Pesticides in Wastewater Using UV/TiO2
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Samples
2.2. Standards and Reagents
2.3. Sample Remediation by UV Photolysis (UV)
2.4. Samples Extraction and Cleanup by QuEChERS
2.5. Analysis by Triple-Quadrupole Gas Chromatography Mass Spectrometry (GCMSMSTSQ 8000/SRM)
2.6. QAQC Strategies and Method Performance
3. Results and Discussion
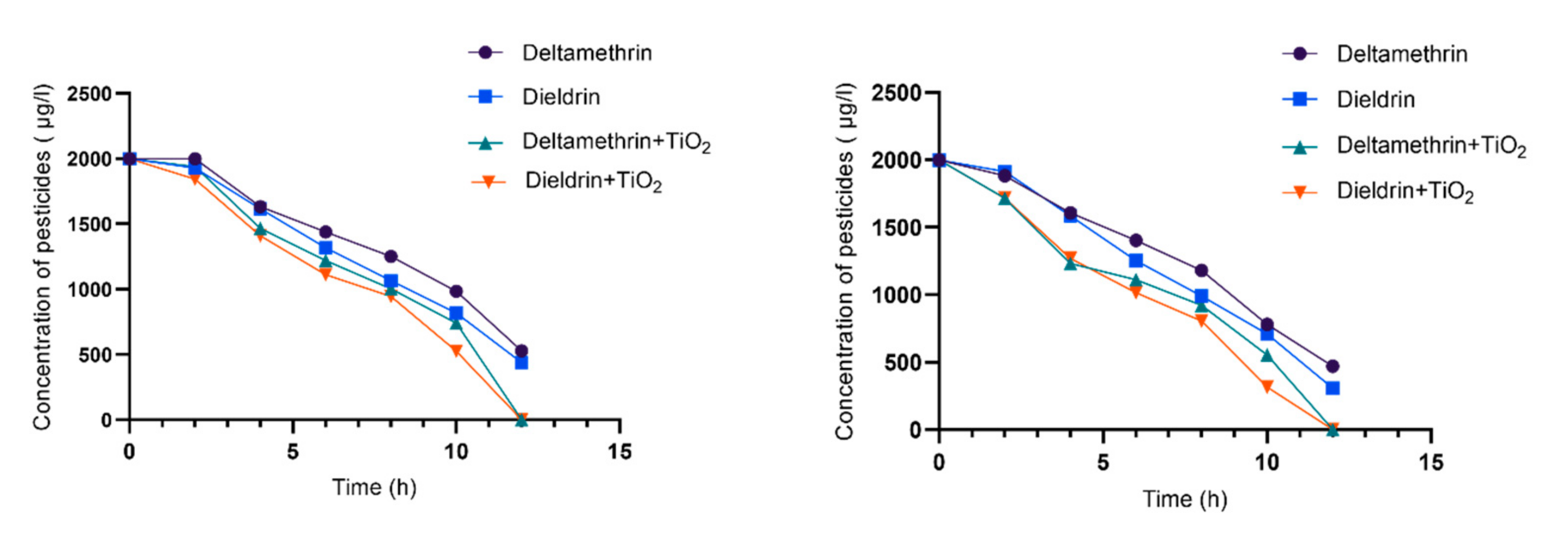
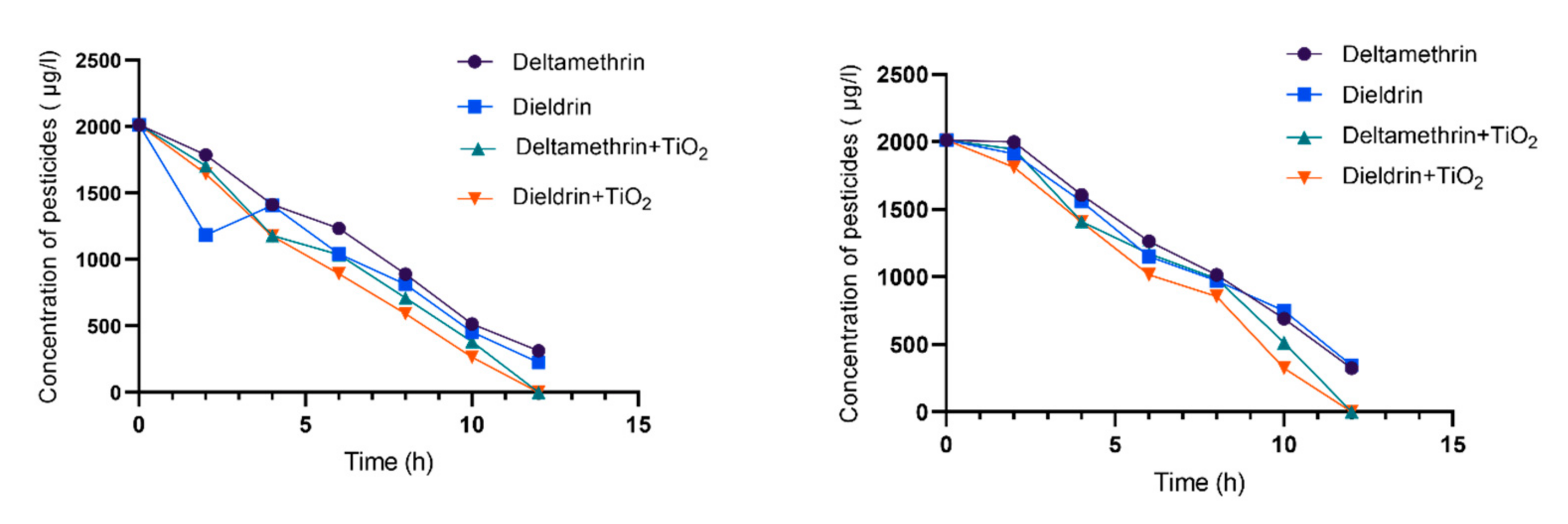
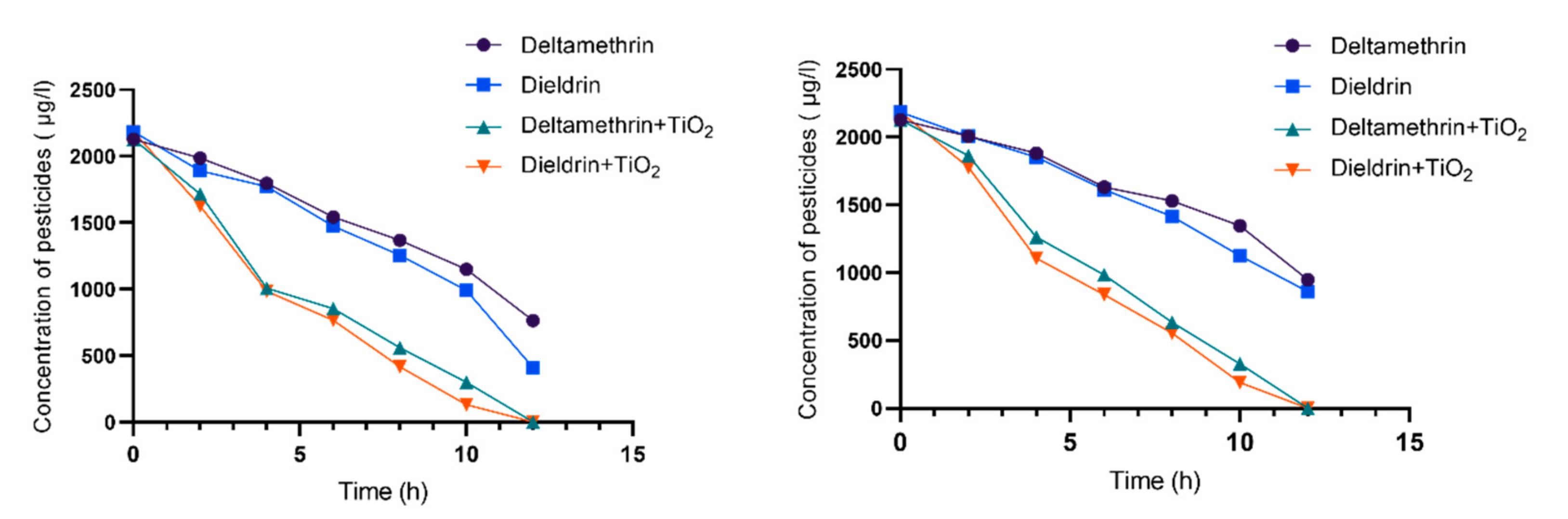
3.1. Photolysis Process
3.2. Photocatalysis Process
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cesco, M.; Lucini, L.; Miras-Moreno, B.; Borruso, L.; Mimmo, T.; Pii, Y.; Puglisi, E.; Spini, G.; Taskin, E.; Tiziani, R.; et al. The hidden effects of agrochemicals on plant metabolism and root-associated microorganisms. Plant Sci. 2021, 311, 111012. [Google Scholar] [CrossRef]
- WHO. Health Topics: Pesticides. 2020. Available online: https://www.who.int/topics/pesticides/en/ (accessed on 1 February 2020).
- Eddleston, M.; Bateman, D.N. Pesticides. Medicine 2012, 40, 147–150. [Google Scholar] [CrossRef]
- Eddleston, M. Pesticides. Medicine 2016, 44, 193–196. [Google Scholar] [CrossRef]
- Verhaelen, K.; Bouwknegt, M.; Rutjes, S.; Husman, A. Persistence of human norovirus in reconstituted pesticides- Pesticide application as a possible source of viruses in fresh produce chains. Int. J. Food Microbiol. 2013, 160, 323–328. [Google Scholar] [CrossRef]
- Cooper, J.; Dobson, H. The benefits of pesticides to mankind and the environment. Crop Prod. 2007, 26, 1337–1348. [Google Scholar] [CrossRef]
- Codex. Code of Hygienic Practice for Fresh Fruits and Vegetables; (Revised 2010 (New Annex III for Fresh Leafy Vegetables)). 2003. Available online: https://www.ifsh.iit.edu/sites/ifsh/files/departments/ssa/pdfs/codex2003_053e.pdf (accessed on 21 October 2021).
- Oller, I.; Malato, S.; Sánchez-Peérez, J.A.; Maldonado, M.I.; Gasso, R. Detoxification of wastewater containing five common pesticides by solar AOPs–biological coupled system. Catal. Today 2007, 129, 69–78. [Google Scholar] [CrossRef]
- Begum, G. Carbofuran insecticide induced biochemical lalterations in liver and muscle tissues of the fish Clarias batrachus (Linnaeus) and recovery response. Aquat. Toxicol. 2004, 66, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, A.; Malik, R.; Li, J.; Zhan, G. Levels, distribution profile, and risk assessment of polychlorinated biphenyls (PCBs) in water and sediment from two tributaries of the River Chenab, Pakistan. Environ. Sci. Pollut. Res. 2014, 21, 7847–7855. [Google Scholar] [CrossRef]
- Fantke, P.; Friedrich, R.; Jolliet, O. Health impact and damage cost assessment of pesticides in Europe. Environ. Int. 2012, 49, 9–17. [Google Scholar] [CrossRef]
- Costa, L. The neurotoxicity of organochlorine and pyrethroid pesticides (Chapter 9). Handb. Clin. Neurol. 2015, 131, 135–148. [Google Scholar] [CrossRef]
- Hatcher, J.; Richardson, J.; Guillot, T.; McCormack, A.; Di Monte, A.; Jones, D.; Pennell, K.; Miller, G. Dieldrin exposure induces oxidative damage in the mouse nigrostriatal dopamine system. Exp. Neurol. 2007, 204, 619–630. [Google Scholar] [CrossRef] [Green Version]
- Sava, V.; Velasquez, A.; Song, S.; Sanchez-Ramos, J. Dieldrin Elicits a Widespread DNA Repair and Antioxidative Response in Mouse Brain. J. Biochem. Mol. Toxicol. 2007, 21, 3. [Google Scholar] [CrossRef]
- Hashimoto, Y. Dieldrin Residue in the Soil and Cucumber from Agricultural Field in Tokyo. J. Pestic. Sci. 2005, 30, 397–402. [Google Scholar] [CrossRef] [Green Version]
- Maldonado-Reyes, A.; Montero-Ocampo, C.; Solorza-Feria, O. Remediation of drinking water contaminated with arsenic by electro-removal process using different metal electrodes. Environ. Monit. 2007, 9, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Saqib, T.; Naqvi, S.; Siddiqui, P.; Azmi, M. Detection of pesticide residues in muscles, liver and fat of 3 species of Labeo found in Kalri and Haleji lakes. J. Environ. Biol. 2005, 26, 433–438. [Google Scholar]
- Al-Wabel, M.; El-Saeid, M.H.; Usman, A.R.; Al-Turki, A.M.; Ahmad, M.; Hassanin, A.S.; El-Naggar, A.H.; Alenazi, K.K. Identification, Quantification, and Toxicity of PCDDs and PCDFs in Soils from Industrial Areas in the Central and Eastern Regions of Saudi Arabia. Bull. Environ. Contam. Toxicol. 2016, 96, 622–629. [Google Scholar] [CrossRef]
- El-Saeid, M.; Al-Dosari, S. Monitoring of pesticide residues in Riyadh dates by SFE, MSE, SFC, and GC techniques. Arab. J. Chem. 2010, 3, 179–186. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, T.; Rafatullah, M.; Ghazali, A.; Sulaiman, O.; Hashim, R.; Ahmad, A. Removal of pesticides from water and wastewater by different adsorbents: A review. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2010, 28, 231–271. [Google Scholar] [CrossRef]
- Ali, I.; Gupta, V.K. Advances in water treatment by adsorption technology. Nat. Protocol. 2006, 1, 2661–2667. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Khan, T.A.; Asim, M. Removal of arsenic from water by electrocoagulation and electrodialysis techniques. Sepn. Purif. Rev. 2011, 40, 25–42. [Google Scholar] [CrossRef]
- Ali, I.; Khan, T.; Asim, M. Removal of arsenate from groundwater by electrocoagulation method. Environ. Sci. Pollut. Res. 2012, 19, 1668–1676. [Google Scholar] [CrossRef]
- Ali, I.; Basheer, A.; Mbianda, X.; Burakov, A.; Galunin, E.; Burakova, I.; Mkrtchyan, E.; Tkachev, A.; Grachev, V. Graphene based adsorbents for remediation of noxious pollutants from wastewater. Environ. Int. 2019, 127, 160–180. [Google Scholar] [CrossRef]
- Saleh, I.; Zouari, N.; Al-Ghouti, M. Removal of pesticides from water and wastewater: Chemical, physical and biological treatment approaches. Environ. Technol. Innov. 2020, 19, 101029. [Google Scholar] [CrossRef]
- Oturan, M.; Aaron, J. Advanced Oxidation Processes in Water/Wastewater Treatment: Principles and Applications. A Review. Crit. Rev. Environ. Sci. Tech. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Z.; She, P.; Ding, F. Water removal from sludge in a horizontal electric field. Dry. Technol. 2001, 19, 627–638. [Google Scholar] [CrossRef]
- Baghirzade, B.; Yetis, U.; Dilek, F. Imidacloprid elimination by O3 and O3/UV: Kinetics study, matrix effect, and mechanism insight. Environ. Sci. Pollut. Res. 2021, 28, 24535–24551. [Google Scholar] [CrossRef] [PubMed]
- Shifu, C.; Gengyu, C. Photocatalytic degradation of organophosphorus pesticides using floating photocatalyst TiO2.SiO2/beads by sunlight. Sol. Energy 2005, 79, 1–9. [Google Scholar] [CrossRef]
- Zhang, R.; Yang, Y.; Huang, C.; Zhao, L.; Sum, P. Kinetics and modeling of sulfonamide antibiotic degradation in wastewater and human urine by UV/H2O2 and UV/PDS. Water Res. 2016, 103, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Cassano, D.; Zapata, A.; Brunetti, G.; Del Moro, G.; Di Iaconi, C.; Oller, I.; Malato, S.; Mascolo, G. Comparison of several combined/integrated biological-AOPs setups for the treatment of municipal landfill leachate: Minimization of operating costs and effluent toxicity. Chem. Eng. J. 2011, 172, 250–257. [Google Scholar] [CrossRef]
- Katagi, T. Direct photolysis mechanism of pesticides in water. J. Pestic. Sci. 2018, 43, 57–72. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, K.; Kawanaka, Y.; Yun, S.J.; Oyaizu, H. Bioremediation of the organochlorine pesticides, dieldrin and endrin, and their occurrence in the environment. Appl. Microbiol. Biotechnol. 2009, 84, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Ismail, B.S.; Mazlinda, M.; Tayeb, M.A. The Persistence of Deltamethrin in Malaysian Agricultural Soils. Sains Malays. 2015, 44, 83–89. [Google Scholar] [CrossRef]
- Lehtola, M.J.; Miettinen, I.T.; Vartiainen, T.; Rantakokko, P.; Hirvonen, A.; Martikainen, P.J. Impact of UV disinfection on microbiallyavailable phosphorus, organic carbon, and microbial growth in drinking water. Water Res. 2003, 37, 1064–1070. [Google Scholar] [CrossRef]
- Li, X.; Cai, M.; Wang, L.; Niu, F.; Yang, D.; Zhang, G. Evaluation survey of microbial disinfection methods in UV-LED water treatment systems. Sci. Total. Environ. 2019, 659, 1415–1427. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Lombraña, J.I.; Ma De Luis, A.; Varona, F. UV/H2O2 chemical oxidation for high loaded effluents: A degradation kinetic study of las surfactant wastewaters. Environ. Technol. 2008, 247, 903–911. [Google Scholar] [CrossRef]
- El-Saeid, M.; Al-Turki, A.; Nadeem, M.; Hassanin, A.; Al-Wabel, M. Photolysis degradation of polyaromatic hydrocarbons (PAHs) on surface sandy soil. Environ. Sci. Pollut. Res. 2015, 22, 9603–9616. [Google Scholar] [CrossRef]
- EL-Saeid, M.H.; Alotaibi, M.O.; Alshabanat, M.; AL-Anazy, M.M.; Alharbi, K.R.; Altowyan, A.S. Impact of Photolysis and TiO2 on Pesticides Degradation in Wastewater. Water 2021, 13, 655. [Google Scholar] [CrossRef]
- Aaron, J.; Oturan, M. New photochemical and electrochemical methods for the degradation of pesticides in aqueous media. Turk. J. Chem. 2001, 25, 509–520. [Google Scholar]
- Shayeghi, M.; Dehghani, M.H.; Alimohammadi, M.; Goodini, K. Using Ultraviolet Irradiation for Removal of Malathion Pesticide in Water. J. Arthropod-Borne Dis. 2012, 6, 45–53. [Google Scholar] [PubMed]
- Tariq, S.R.; Ahmed, D.; Farooq, A.; Rasheed, S.; Mansoor, M. Photodegradation of bifenthrin and deltamethrin—effect of copper amendment and solvent system. Environ. Monit. Assess. 2017, 189, 71. [Google Scholar] [CrossRef]
- Daneshvar, N.; Hejazi, M.; Rangarangy, B.; Khataee, A. Photocatalytic Degradation of an Organophosphorus Pesticide Phosalone in Aqueous Suspensions of Titanium Dioxide. J. Environ. Sci. Health Part B 2004, 39, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Chen, J.; Shao, J.; Chen, C.; Zhao, H.; Hao, C. Important role of reaction field in photodegradation of deca-bromodiphenyl ether: Theoretical and experimental investigations of solvent effects. Chemosphere 2009, 76, 1486–1490. [Google Scholar] [CrossRef] [PubMed]
- Saqib, N.; Adnan, R.; Shah, I. A mini-review on rare earth metal-doped TiO2 for photocatalytic remediation of wastewater. Environ. Sci. Pollut. Res. 2016, 23, 15941–15951. [Google Scholar] [CrossRef]
- Thiruvenkatachari, R.; Vigneswaran, S.; Shik, M. A review on UV/TiO2 photocatalytic oxidation process. Korean J. Chem. Eng. 2008, 25, 64–72. [Google Scholar] [CrossRef]
- Mecha, A.C.; Chollom, M.N. Photocatalytic ozonation of wastewater: A review. Environ. Chem. Lett. 2020, 18, 1491–1507. [Google Scholar] [CrossRef]
- Ramos-Delgado, N.A.; Gracia-Pinilla, M.A.; Maya-Trevino, L.; Hinojosa-Reyes, L.; Guzman-Mar, J.L.; Hernández-Ramírez, A. Solar photocatalytic activity of TiO2 modified with WO3 on the degradation of an organophosphorus pesticide. J. Hazard. Mater. 2013, 263, 36–44. [Google Scholar] [CrossRef]
- Yavg, V. Photocatalytic Degradation of Selected Organophosphorus Pesticides Using Titanium Dioxide and UV Light. In Titanium Dioxide: Material for a Sustainable Environment; Additional information is available at the end of the chapter; Petsas, A.S., Vagi, M.C., Eds.; IntechOpen: London, UK, 2018; p. 241. [Google Scholar] [CrossRef] [Green Version]
- Montañez, J.; Gómez, S.; Santiago, A.; Pierella, L. TiO2 Supported on HZSM-11 Zeolite as Efficient Catalyst for the Photodegradation of Chlorobenzoic Acids. J. Braz. Chem. Soc. 2015, 26, 1191–1200. [Google Scholar] [CrossRef]
- Burrows, H.; Canle, M.; Santaballa, J.; Steenken, S. Reaction pathways and mechanisms of photodegradation of pesticides. J. Photochem. Photobiol. B Biol. 2002, 67, 71–108. [Google Scholar] [CrossRef] [Green Version]
- Konstantinou, I.; Albanis, T. Photocatalytic transformation of pesticides in aqueous titanium dioxide suspensions using artificial and solar light: Intermediates and degradation pathways. Appl. Catal. B Environ. 2003, 42, 319–335. [Google Scholar] [CrossRef]
- Liu, S.; Liu, G.; Feng, Q. Al-doped TiO2 mesoporous materials: Synthesis and photodegradation properties. J. Porous Mater. 2010, 17, 197–206. [Google Scholar] [CrossRef]
- Liu, X.; Zhan, Y.; Zhang, Z.; Pan, L.; Hu, L.; Liu, K.; Zhou, X.; Bai, L. Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2. Catalysts 2019, 9, 294. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, A.T.; Juang, R. Photocatalytic degradation of p-chlorophenol by hybrid H2O2 and TiO2 in aqueous suspensions under UV irradiation. J. Environ. Manag. 2015, 147, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Sarkouhi, M.; Shamsipur, M.; Hassan, J. Metal ion promoted degradation mechanism of chlorpyrifos and phoxim. Arab. J. Chem. 2016, 9, 43–47. [Google Scholar] [CrossRef] [Green Version]
- Miguel, N.; Ormad, M.P.; Mosteo, R.; Overlleiro, J. Photocatalytic Degradation of Pesticides in Natural Water: Effect of Hydrogen Peroxide. Int. J. Photoenergy 2012, 2012, 371714. [Google Scholar] [CrossRef]
- Pelizzetti, E. Concluding remarks on heterogeneous solar photocatalysis. Sol. Energy Mater. Sol. Cells 1995, 38, 453–457. [Google Scholar] [CrossRef]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Mousset, E.; Dionysiou, D.D. Photoelectrochemical reactors for treatment of water and wastewater: A review. Environ. Chem. Lett. 2020, 18, 1301–1318. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Z. Degradation of six typical pesticides in water by VUV/UV/chlorine process: Evaluation of the synergistic effect. Water Res. 2019, 161, 439–447. [Google Scholar] [CrossRef]
- Lopez-Alvarez, B.; Villegas-Guzman, P.; Peñuela, G.A.; Torres-Palma, R.A. Degradation of a Toxic Mixture of the Pesticides Carbofuran and Iprodione by UV/H2O2: Evaluation of Parameters and Implications of the Degradation Pathways on the Synergistic Effects. Water Air Soil Pollut. 2016, 227, 215. [Google Scholar] [CrossRef]
- Konstantinou, I.K.; Zarkadis, A.K.; Albanis, T.A. Photodegradation of Selected Herbicides in Various Natural Waters and Soils under Environmental Conditions. J. Environ. Qual. 2001, 30, 121–130. [Google Scholar] [CrossRef]
| Pesticides | Molecular Formula | Molecular Weight | Solubility | Type | Effective against |
|---|---|---|---|---|---|
| Dieldrin | C12H8Cl6O | 380.895 g/mol | soluble in water | organochlorine insecticide | controlling locusts, vectors, tropical disease, and termites |
| Deltamethrin | C22H19Br2NO3 | 505.206 g/mol | soluble in water | pyrethroid ester pesticide | controlling malaria vectors |
| GC Trace Ultra Conditions | TSQ Quantum MS/MS Conditions | ||
|---|---|---|---|
| Column | TR-Pesticide 30 m × 0.25 mm × 0.25 μm | Ionization mode | EI |
| Injector | Splitless | Electron energy | 70 eV |
| Injected volume | 1 μL | Emission current | 50 μA |
| Injector temperature | 225 °C | Q1/Q3 resolution | 0.7 u (FWHM) |
| Carrier gas | Helium, 1.2 mL/min | Collision gas | Argon |
| Oven program | 80 °C hold 1 min 15 °C/min to 160 °C hold 1 min 2.2 °C/min to 230 °C hold 1 min 5 °C/min to 290 °C hold 5 min, Run time: 57.15 min | Operating mode | Selected reaction monitoring (SRM) |
| Transfer line temperature | 280 °C | Collision gas pressure | 1 mTorr |
| Polarity | Positive | ||
| Name | RT min | Mass | Product Mass | Collision Energy m/z | LOQ ng/ml | LOD ng/mL | r2 | Recovery % | SD |
|---|---|---|---|---|---|---|---|---|---|
| Deltamethrin | 21.4 | 176 | 124 | 9 | 3.6 | 1.2 | 0.8034 | 102.4 | 8.3 |
| Dieldrin | 30.5 | 279 | 243 | 10 | 7.9 | 5.3 | 0.9486 | 105.5 | 7.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
EL-Saeid, M.H.; Alotaibi, M.O.; Alshabanat, M.; Alharbi, K.; Altowyan, A.S.; Al-Anazy, M. Photo-Catalytic Remediation of Pesticides in Wastewater Using UV/TiO2. Water 2021, 13, 3080. https://doi.org/10.3390/w13213080
EL-Saeid MH, Alotaibi MO, Alshabanat M, Alharbi K, Altowyan AS, Al-Anazy M. Photo-Catalytic Remediation of Pesticides in Wastewater Using UV/TiO2. Water. 2021; 13(21):3080. https://doi.org/10.3390/w13213080
Chicago/Turabian StyleEL-Saeid, Mohamed H., Modhi O. Alotaibi, Mashael Alshabanat, Khadiga Alharbi, Abeer S. Altowyan, and Murefah Al-Anazy. 2021. "Photo-Catalytic Remediation of Pesticides in Wastewater Using UV/TiO2" Water 13, no. 21: 3080. https://doi.org/10.3390/w13213080
APA StyleEL-Saeid, M. H., Alotaibi, M. O., Alshabanat, M., Alharbi, K., Altowyan, A. S., & Al-Anazy, M. (2021). Photo-Catalytic Remediation of Pesticides in Wastewater Using UV/TiO2. Water, 13(21), 3080. https://doi.org/10.3390/w13213080







