Enhancement of Power Generation and Organic Removal in Double Anode Chamber Designed Dual-Chamber Microbial Fuel Cell (DAC-DCMFC)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Practical Techniques
2.2.2. Analytical Techniques
2.2.3. Data Calculation
3. Results
3.1. Seeding and Feeding Characteristics
3.2. Parameters Changes
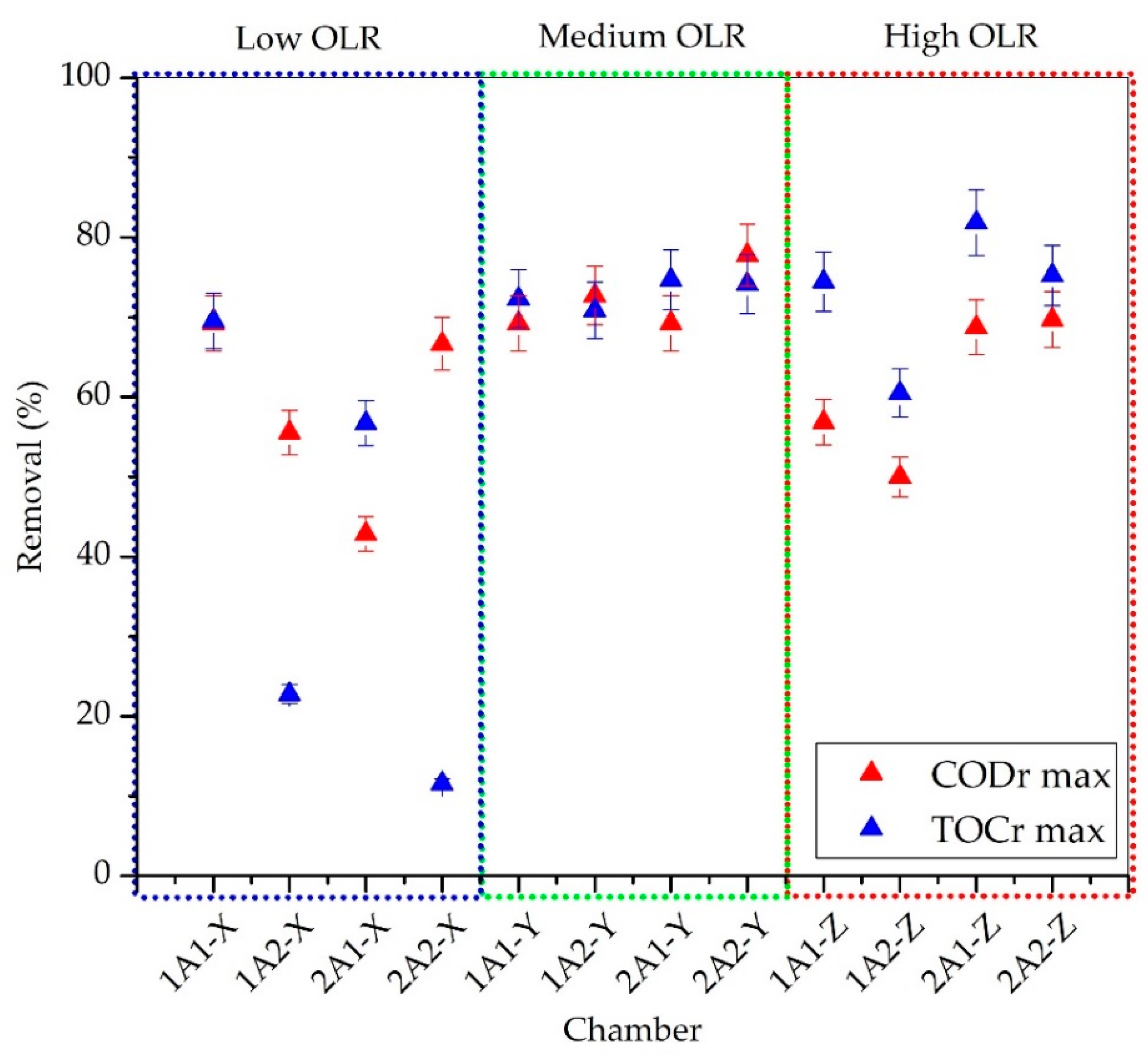
3.3. COD and TOC Removal
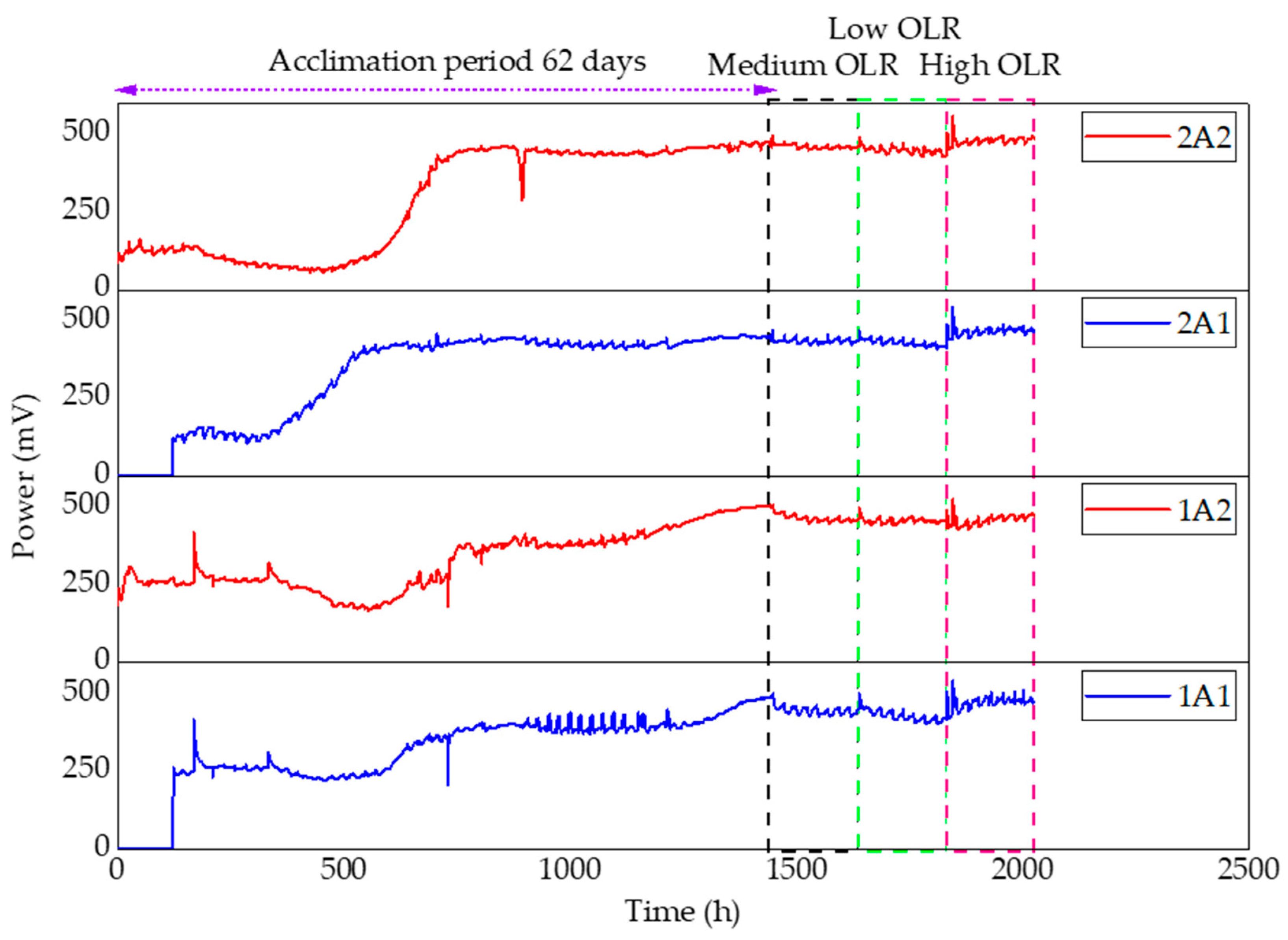
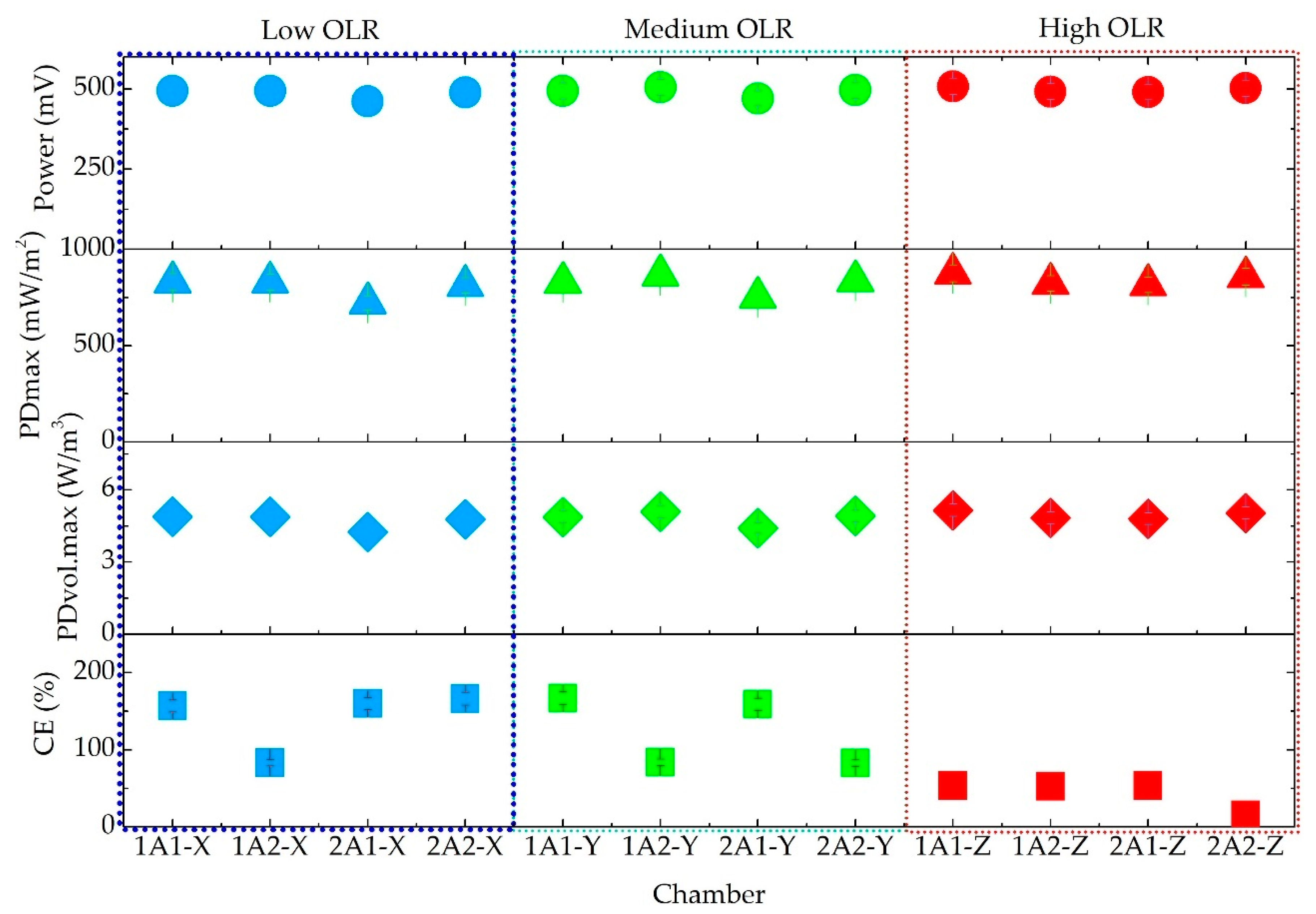
3.4. Power Production
4. Discussion
4.1. Conceptualization
4.2. Seeding and Feeding
4.3. Factors Affecting the Performance
4.4. Power Production Performance
4.5. Cost Consideration
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Samudro, H.; Mangkoedihardjo, S. Indoor phytoremediation using decorative plants: An overview of application principles. J. Phytol. 2021, 13, 28–32. [Google Scholar] [CrossRef]
- Samudro, G.; Mangkoedihardjo, S. Mixed plant operations for phytoremediation in polluted environments—A critical review. J. Phytol. 2020, 12, 99–103. [Google Scholar] [CrossRef]
- Syvitski, J.; Waters, C.N.; Day, J.; Milliman, J.D.; Summerhayes, C.; Steffen, W.; Zalasiewicz, J.; Cearreta, A.; Gałuszka, A.; Hajdas, I.; et al. Extraordinary human energy consumption and resultant geological impacts beginning around 1950 CE initiated the proposed Anthropocene Epoch. Commun. Earth Environ. 2020, 1, 1–13. [Google Scholar] [CrossRef]
- Ahsan, M.A.; Santiago, A.R.P.; Nair, A.N.; Weller, J.M.; Sanad, M.F.; Valles-Rosales, D.J.; Chan, C.K.; Sreenivasan, S.; Noveron, J.C. Metal-Organic frameworks-derived multifunctional carbon encapsulated metallic nanocatalysts for catalytic peroxymonosulfate activation and electrochemical hydrogen generation. Mol. Catal. 2020, 498, 111241. [Google Scholar] [CrossRef]
- Ahsan, M.A.; Imam, M.A.; Santiago, A.R.P.; Rodriguez, A.; Alvarado-Tenorio, B.; Bernal, R.; Luque, R.; Noveron, J.C. Spent tea leaves templated synthesis of highly active and durable cobalt-based trifunctional versatile electrocatalysts for hydrogen and oxygen evolution and oxygen reduction reactions. Green Chem. 2020, 22, 6967–6980. [Google Scholar] [CrossRef]
- Dominguez, N.; Torres, B.; Barrera, L.A.; Rincon, J.E.; Lin, Y.; Chianelli, R.R.; Ahsan, M.A.; Noveron, J.C. Bimetallic CoMoS Composite Anchored to Biocarbon Fibers as a High-Capacity Anode for Li-Ion Batteries. ACS Omega 2018, 3, 10243–10249. [Google Scholar] [CrossRef] [PubMed]
- Gajda, I.; Greenman, J.; Ieropoulos, I.A. Recent advancements in real-world microbial fuel cell applications. Curr. Opin. Electrochem. 2018, 11, 78–83. [Google Scholar] [CrossRef]
- Fadzli, F.S.; Bhawani, S.A.; Rania, E.A.M. Microbial Fuel Cell: Recent Developments in Organic Substrate Use and Bacterial Electrode Interaction. J. Chem. 2021, 2021, 4570388. [Google Scholar] [CrossRef]
- Chen, H.; Simoska, O.; Lim, K.; Grattieri, M.; Yuan, M.; Dong, F.; Lee, Y.S.; Beaver, K.; Weliwatte, S.; Gaffney, E.M.; et al. Fundamentals, Applications, and Future Directions of Bioelectrocatalysis. Chem. Rev. 2020, 120, 12903–12993. [Google Scholar] [CrossRef]
- Ahmed, I.; Zia, M.A.; Afzal, H.; Ahmed, S.; Ahmad, M.; Akram, Z.; Sher, F.; Iqbal, H.M.N. Socio-economic and environmental impacts of biomass valorisation: A strategic drive for sustainable bioeconomy. Sustainability 2021, 13, 4200. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Bolognesi, S.; Cecconet, D. Sustainable, decentralized sanitation and reuse with hybrid nature-based systems. Water 2021, 13, 1583. [Google Scholar] [CrossRef]
- Verma, P.; Daverey, A.; Kumar, A.; Arunachalam, K. Microbial Fuel Cell—A Sustainable Approach for Simultaneous Wastewater Treatment and Energy Recovery. J. Water Process. Eng. 2021, 40, 101768. [Google Scholar] [CrossRef]
- Flimban, S.G.A.; Ismail, I.M.I.; Kim, T.; Oh, S.-E. Overview of recent advancements in the microbial fuel cell from fundamentals to applications: Design, major elements, and scalability. Energies 2019, 12, 3390. [Google Scholar] [CrossRef]
- Paucar, N.E.; Sato, C. Microbial Fuel Cell for Energy Production, Nutrient Removal and Recovery from Wastewater: A Review. Processes 2021, 9, 1318. [Google Scholar] [CrossRef]
- Igboamalu, T.E.; Bezuidenhout, N.; Matsena, M.T.; Chirwa, E.M.N. Microbial fuel cell power output and growth: Effect of pH on anaerobic microbe consortium. Chem. Eng. Trans. 2019, 76, 1381–1386. [Google Scholar]
- Potrykus, S.; León-Fernández, L.F.; Nieznański, J.; Karkosiński, D.; Fernandez-Morales, F.J. The influence of external load on the performance of microbial fuel cells. Energies 2021, 14, 612. [Google Scholar] [CrossRef]
- Lawson, K.; Rossi, R.; Regan, J.M.; Logan, B.E. Impact of cathodic electron acceptor on microbial fuel cell internal resistance. Bioresour. Technol. 2020, 316, 123919. [Google Scholar] [CrossRef]
- Rossi, R.; Logan, B.E. Unraveling the contributions of internal resistance components in two-chamber microbial fuel cells using the electrode potential slope analysis. Electrochim. Acta 2020, 348, 136291. [Google Scholar] [CrossRef]
- Waheeb, H.A.; Al-Alalawy, A.F. Innovative microbial fuel cell design for investigation of cathode chamber effect and electricity generation enhancement. AIP Conf. Proc. 2020, 2213, 020212. [Google Scholar]
- Gajda, I.; Obata, O.; Greenman, J.; Ieropoulos, I.A. Electroosmotically generated disinfectant from urine as a by-product of electricity in microbial fuel cell for the inactivation of pathogenic species. Scientific Reports. 2020, 10, 5533. [Google Scholar] [CrossRef] [PubMed]
- Nath, D.; Ghangrekar, M.M. Plant secondary metabolites induced electron flux in microbial fuel cell: Investigation from laboratory-to-field scale. Scientific Reports. 2020, 10, 17185. [Google Scholar] [CrossRef]
- Ren, J.; Li, N.; Du, M.; Zhang, Y.; Hao, C.; Hu, R. Study on the effect of synergy effect between the mixed cultures on the power generation of microbial fuel cells. Bioengineered 2021, 12, 844–854. [Google Scholar] [CrossRef]
- Rajesh, P.P.; Noori, M.T.; Ghangrekar, M.M. Improving Performance of Microbial Fuel Cell by Using Polyaniline-Coated Carbon–Felt Anode. J. Hazardous Toxic Radioact. Waste 2020, 24, 04020024. [Google Scholar] [CrossRef]
- Jingyu, H.; Miwornunyuie, N.; Ewusi-Mensah, D.; Koomson, D.A. Assessing the factors influencing the performance of constructed wetland-microbial fuel cell integration. Water Sci. Technol. 2020, 81, 631–643. [Google Scholar] [CrossRef]
- Chakraborty, I.; Das, S.; Dubey, B.K.; Ghangrekar, M.M. Novel low cost proton exchange membrane made from sulphonated biochar for application in microbial fuel cells. Mater. Chem. Phys. 2020, 239, 122025. [Google Scholar] [CrossRef]
- Aysla Costa De Oliveira, M.; D’Epifanio, A.; Ohnuki, H.; Mecheri, B. Platinum group metal-free catalysts for oxygen reduction reaction: Applications in microbial fuel cells. Catalysts 2020, 10, 475. [Google Scholar] [CrossRef]
- You, J.; Greenman, J.; Ieropoulos, I. Novel Analytical Microbial Fuel Cell Design for Rapid in Situ Optimisation of Dilution Rate and Substrate Supply Rate, by Flow, Volume Control and Anode Placement. Energies 2018, 11, 2377. [Google Scholar] [CrossRef]
- Singh, A.; Kaushik, A. Sustained energy production from wastewater in microbial fuel cell: Effect of inoculum sources, electrode spacing and working volume. 3 Biotech. 2021, 11, 334. [Google Scholar] [CrossRef] [PubMed]
- Flimban, S.G.A.; Hassan, S.H.A.; Rahman, M.M.; Oh, S.E. The effect of Nafion membrane fouling on the power generation of a microbial fuel cell. Int. J. Hydrogen Energy 2020, 45, 13643–13651. [Google Scholar] [CrossRef]
- Shabani, M.; Younesi, H.; Pontié, M.; Rahimpour, A.; Rahimnejad, M.; Zinatizadeh, A.A. A critical review on recent proton exchange membranes applied in microbial fuel cells for renewable energy recovery. J. Clean. Prod. 2020, 264, 121446. [Google Scholar] [CrossRef]
- Ieropoulos, I.; Winfield, J.; Greenman, J. Effects of flow-rate, inoculum and time on the internal resistance of microbial fuel cells. Bioresour. Technol. 2010, 101, 3520–3525. [Google Scholar] [CrossRef]
- Krieg, T.; Wood, J.A.; Mangold, K.M.; Holtmann, D. Mass transport limitations in microbial fuel cells: Impact of flow configurations. Biochem. Eng. J. 2018, 138, 172–178. [Google Scholar] [CrossRef]
- Santos, J.B.C.; de Barros, V.V.S.; Linares, J.J. The Hydraulic Retention Time as a Key Parameter for the Performance of a Cyclically Fed Glycerol-Based Microbial Fuel Cell from Biodiesel. J. Electrochem. Soc. 2017, 164, H3001–H3006. [Google Scholar] [CrossRef]
- Tominaga, M.; Ototani, S.; Darmawan, R. The effect of connection type in series and parallel on electric power generation of mud microbial fuel cell. AIP Conf. Proc. 2020, 2230, 030002. [Google Scholar]
- Miller, A.; Singh, L.; Wang, L.; Liu, H. Linking internal resistance with design and operation decisions in microbial electrolysis cells. Environ. Int. 2019, 126, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Aelterman, P.; Rabaey, K.; Clauwert, P.; Verstraete, W. Microbial fuel cells for wastewater treatment. Biotechnol. Biomim. Civ. Eng. 2006, 54, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Clauwaert, P.; Aelterman, P.; Pham, T.H.; De Schamphelaire, L.; Carballa, M.; Rabaey, K.; Verstraete, W. Minimizing losses in bio-electrochemical systems: The road to applications. Appl. Microbiol. Biotechnol. 2008, 79, 901–913. [Google Scholar] [CrossRef]
- Nandy, A.; Kundu, P.P. Configurations of microbial fuel cells. In Progress and Recent Trends in Microbial Fuel Cells; Elsevier: Amsterdam, The Netherlands, 2018; pp. 25–45. [Google Scholar]
- Minutillo, M.; Nastro, R.A.; Di Micco, S.; Jannelli, E.; Cioffi, R.; Di Giuseppe, M. Performance Assessment of Multi-Electrodes Reactors for Scaling-up Microbial Fuel Cells. E3S Web Conf. 2020, 197, 08020. [Google Scholar] [CrossRef]
- Hamed, M.S.; Majdi, H.S.; Hasan, B.O. Effect of Electrode Material and Hydrodynamics on the Produced Current in Double Chamber Microbial Fuel Cells. ACS Omega 2020, 5, 10339–10348. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, X.; Zhang, Q.; Li, C. Microbial Fuel Cells: Nanomaterials Based on Anode and Their Application. Energy Technol. 2020, 8, 2000206. [Google Scholar] [CrossRef]
- Frattini, D.; Karunakaran, G.; Cho, E.-B.; Kwon, Y. Sustainable Syntheses and Sources of Nanomaterials for Microbial Fuel/Electrolysis Cell Applications: An Overview of Recent Progress. Processes 2021, 9, 1221. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.M.; Rafatullah, M.; Chua, Y.S.; Ahmad, A.; Umar, K. Recent advances in anodes for microbial fuel cells: An overview. Materials 2020, 13, 2078. [Google Scholar] [CrossRef]
- Fatima, M.; Kiros, Y.; Farooq, R.; Lindström, R.W. Low-Cost Single Chamber MFC Integrated With Novel Lignin-Based Carbon Fiber Felt Bioanode for Treatment of Recalcitrant Azo Dye. Front. Energy Res. 2021, 9, 672817. [Google Scholar] [CrossRef]
- Koók, L.; Dörgő, G.; Bakonyi, P.; Rózsenberszki, T.; Nemestóthy, N.; Bélafi-Bakó, K.; Abonyi, J. Directions of membrane separator development for microbial fuel cells: A retrospective analysis using frequent itemset mining and descriptive statistical approach. J. Power Sources 2020, 478, 229014. [Google Scholar] [CrossRef]
- Obileke, K.C.; Onyeaka, H.; Meyer, E.L.; Nwokolo, N. Microbial fuel cells, a renewable energy technology for bio-electricity generation: A mini-review. Electrochem. Commun. 2021, 125, 107003. [Google Scholar] [CrossRef]
- Kim, M.H.; Iwuchukwu, I.J.; Wang, Y.; Shin, D.; Sanseverino, J.; Frymier, P. An analysis of the performance of an anaerobic dual anode-chambered microbial fuel cell. J. Power Sources 2011, 196, 1909–1914. [Google Scholar] [CrossRef]
- Samsudeen, N.; Sharma, A.; Radhakrishnan, T.K.; Matheswaran, M. Performance investigation of multi-chamber microbial fuel cell: An alternative approach for scale up system. J. Renew. Sustain. Energy 2015, 7, 043101. [Google Scholar] [CrossRef]
- Abbassi, R.; Yadav, A.K.; Khan, F.; Garaniya, V. Integrated Microbial Fuel Cells for Wastewater Treatment, 1st ed.; Butterworth-Heinemann: Chennai, India, 2020. [Google Scholar]
- Hemalatha, M.; Shanthi Sravan, J.; Venkata Mohan, S. Self-Induced Bioelectro-Potential Influence on Sulfate Removal and Desalination in Microbial Fuel Cell; Elsevier: Amsterdam, The Netherlands, 2020; Volume 309. [Google Scholar]
- Zhou, W.; Imai, T.; Ukita, M.; Sekine, M.; Higuchi, T. Triggering forces for anaerobic granulation in UASB reactors. Process. Biochem. 2006, 41, 36–43. [Google Scholar] [CrossRef]
- Zhou, W.; Imai, T.; Ukita, M.; Li, F.; Yuasa, A. Effect of loading rate on the granulation process and granular activity in a bench scale UASB reactor. Bioresour. Technol. 2007, 98, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Qaiser, A.A.; Huda, N.U.; Saeed, A. Heterogeneous ion exchange membranes based on thermoplastic polyurethane (TPU): Effect of PSS/DVB resin on morphology and electrodialysis. RSC Adv. 2020, 10, 3029–3039. [Google Scholar] [CrossRef]
- Ortiz-Martínez, V.M.; Gajda, I.; Salar-García, M.J.; Greenman, J.; Hernández-Fernández, F.J.; Ieropoulos, I. Study of the effects of ionic liquid-modified cathodes and ceramic separators on MFC performance. Chem. Eng. J. 2016, 291, 317–324. [Google Scholar] [CrossRef]
- Touach, N.; Ortiz-Martínez, V.M.; Salar-García, M.J.; Benzaouak, A.; Hernández-Fernández, F.; de los Ríos, A.P.; Labjar, N.; Louki, S.; El Mahi, M.; Lotfi, E.M. Influence of the preparation method of MnO2-based cathodes on the performance of single-chamber MFCs using wastewater. Sep. Purif. Technol. 2016, 171, 174–181. [Google Scholar] [CrossRef]
- Liu, S.H.; Fu, S.H.; Chen, C.Y.; Lin, C.W. Enhanced processing of exhaust gas and power generation by connecting mini-tubular microbial fuel cells in series with a biotrickling filter. Renew. Energy 2020, 156, 342–348. [Google Scholar] [CrossRef]
- Marassi, R.J.; Queiroz, L.G.; Silva, D.C.V.R.; da Silva, F.T.; Silva, G.C.; de Paiva, T.C.B. Performance and toxicity assessment of an up-flow tubular microbial fuel cell during long-term operation with high-strength dairy wastewater. J. Clean. Prod. 2020, 259, 120882. [Google Scholar] [CrossRef]
- Stoll, Z.A.; Dolfing, J.; Xu, P. Minimum performance requirements for microbial fuel cells to achieve energy-neutral wastewater treatment. Water 2018, 10, 243. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- Kloch, M.; Toczylowska-Maminska, R. Toward optimization of wood industry wastewater treatment in microbial fuel cells-mixed wastewaters approach. Energies 2020, 13, 263. [Google Scholar] [CrossRef]
- Logan, B.E. Microbial Fuel Cells; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar]
- Tanaka, S.; Kobayashi, T.; Kamiyama, K.I.; Bildan, M.L.N. Effects of thermochemical pretreatment on the anaerobic digestion of waste activated sludge. Water Sci. Technol. 1997, 35, 209–215. [Google Scholar] [CrossRef]
- Prashanth, S.; Kumar, P.; Mehrotra, I. Anaerobic Degradability: Effect of Particulate COD. J. Environ. Eng. 2006, 132, 4. [Google Scholar] [CrossRef]
- Gözde Tuğba Köksoy. Investigation of Digester F/M Ratio as a Parameter to Affect Sludge Minimization and Gas Production of Ultrasonically Treated Sludge. Master’s Thesis, Middle East Technical University, Ankara, Turkey, 2009. [Google Scholar]
- Fedorovich, V.; Knighton, M.C.; Pagaling, E.; Ward, F.B.; Free, A.; Goryanin, I. Novel electrochemically active bacterium phylogenetically related to Arcobacter butzleri, isolated from a microbial fuel cell. Appl. Environ. Microbiol. 2009, 75, 7326–7334. [Google Scholar] [CrossRef]
- Toh, H.; Sharma, V.K.; Oshima, K.; Kondo, S.; Hattori, M.; Ward, F.B.; Free, A.; Taylor, T.D. Complete genome sequences of Arcobacter butzleri ED-1 and Arcobacter sp. Strain L, both isolated from a microbial fuel cell. J. Bacteriol. 2011, 193, 6411–6412. [Google Scholar] [CrossRef][Green Version]
- Pereira-Medrano, A.G.; Knighton, M.; Fowler, G.J.S.; Ler, Z.Y.; Pham, T.K.; Ow, S.Y.; Free, A.; Ward, B.; Wright, P.C. Quantitative proteomic analysis of the exoelectrogenic bacterium Arcobacter butzleri ED-1 reveals increased abundance of a flagellin protein under anaerobic growth on an insoluble electrode. J. Proteom. 2013, 78, 197–210. [Google Scholar] [CrossRef]
- Szydlowski, L.; Lan, T.C.T.; Shibata, N.; Goryanin, I. Metabolic engineering of a novel strain of electrogenic bacterium Arcobacter butzleri to create a platform for single analyte detection using a microbial fuel cell. Enzym. Microb. Technol. 2020, 139, 109564. [Google Scholar] [CrossRef]
- Van Gylswyk, N.O.; Hippe, H.; Rainey, F.A. Schwartzia succinivorans gen. nov., sp. nov., another ruminal bacterium utilizing succinate as the sole energy source. Int. J. Syst. Bacteriol. 1997, 47, 155–159. [Google Scholar] [CrossRef]
- Pham, T.H.; Boon, N.; De Maeyer, K.; Höfte, M.; Rabaey, K.; Verstraete, W. Use of Pseudomonas species producing phenazine-based metabolites in the anodes of microbial fuel cells to improve electricity generation. Appl. Microbiol. Biotechnol. 2008, 80, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Verstraete, W. Microbial fuel cells: Novel biotechnology for energy generation. Trends Biotechnol. 2005, 23, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Hilton, C.L.; Mackey, B.M.; Hargreaves, A.J.; Forsythe, S.J. The recovery of Arcobacter butzleri NCTC 12481 from various temperature treatments. J. Appl. Microbiol. 2001, 91, 929–932. [Google Scholar] [CrossRef]
- Yan, D.; Song, X.; Weng, B.; Yu, Z.; Bi, W.; Wang, J. Bioelectricity generation from air-cathode microbial fuel cell connected to constructed wetland. Water Sci. Technol. 2018, 78, 1990–1996. [Google Scholar] [CrossRef]
- Ryckelynck, N.; Stecher III, H.A.S.; Reimers, C.E. Understanding the Anodic Mechanism of a Seafloor Fuel Cell: Interactions between Geochemistry and Microbial Activity. Biogeochemistry 2005, 76, 113–139. [Google Scholar] [CrossRef]
- Almatouq, A.; Babatunde, A.O. Concurrent phosphorus recovery and energy generation in mediator-less dual chamber microbial fuel cells: Mechanisms and influencing factors. Int. J. Environ. Res. Public Health 2016, 13, 375. [Google Scholar] [CrossRef] [PubMed]
- Miyahara, M.; Kouzuma, A.; Watanabe, K. Effects of NaCl concentration on anode microbes in microbial fuel cells. AMB Express 2015, 5, 34. [Google Scholar] [CrossRef]
- Grattieri, M.; Minteer, S.D. Microbial fuel cells in saline and hypersaline environments: Advancements, challenges and future perspectives. Bioelectrochemistry 2018, 120, 127–137. [Google Scholar] [CrossRef]
- Adekunle, A.; Raghavan, V.; Tartakovsky, B. On-line monitoring of heavy metals-related toxicity with a microbial fuel cell biosensor. Biosens. Bioelectron. 2019, 132, 382–390. [Google Scholar] [CrossRef]
- Sharma, P.; Pandey, A.K.; Kim, S.-H.; Singh, S.P.; Chaturvedi, P.; Varjani, S. Critical review on microbial community during in-situ bioremediation of heavy metals from industrial wastewater. Environ. Technol. Innov. 2021, 24, 101826. [Google Scholar] [CrossRef]
- Gustave, W.; Yuan, Z.; Liu, F.; Chen, Z. Mechanisms and challenges of microbial fuel cells for soil heavy metal(loid)s remediation. Sci. Total Environ. 2021, 756, 143865. [Google Scholar] [CrossRef]
- Puig, S.; Serra, M.; Coma, M.; Cabré, M.; Balaguer, M.D.; Colprim, J. Effect of pH on nutrient dynamics and electricity production using microbial fuel cells. Bioresour. Technol. 2010, 101, 9594–9599. [Google Scholar] [CrossRef]
- Liu, M.; Yuan, Y.; Zhang, L.X.; Zhuang, L.; Zhou, S.G.; Ni, J.R. Bioelectricity generation by a Gram-positive Corynebacterium sp. strain MFC03 under alkaline condition in microbial fuel cells. Bioresour. Technol. 2010, 101, 1807–1811. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, Y.; Wu, J. Enhancement of methane production in anaerobic digestion process: A review. Appl. Energy 2019, 240, 120–137. [Google Scholar] [CrossRef]
- Kaur, A.; Boghani, H.C.; Michie, I.; Dinsdale, R.M.; Guwy, A.J.; Premier, G.C. Inhibition of methane production in microbial fuel cells: Operating strategies which select electrogens over methanogens. Bioresour. Technol. 2014, 173, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, P.P.; Jadhav, D.A.; Ghangrekar, M.M. Improving performance of microbial fuel cell while controlling methanogenesis by Chaetoceros pretreatment of anodic inoculum. Bioresour. Technol. 2015, 180, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.K.; Yuan, L.; Liu, T.; Li, Z.H.; Zheng, X.; Sheng, G.P. Thermal/alkaline pretreatment of waste activated sludge combined with a microbial fuel cell operated at alkaline pH for efficient energy recovery. Appl. Energy 2020, 275, 115291. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhao, B.; Zhou, S.; Zhong, S.; Zhuang, L. Electrocatalytic activity of anodic biofilm responses to pH changes in microbial fuel cells. Bioresour. Technol. 2011, 102, 6887–6891. [Google Scholar] [CrossRef]
- Liao, Q.; Zhang, J.; Li, J.; Ye, D.; Zhu, X.; Zheng, J.; Zhang, B. Electricity generation and COD removal of microbial fuel cells (MFCs) operated with alkaline substrates. Int. J. Hydrogen Energy 2014, 39, 19349–19354. [Google Scholar] [CrossRef]
- Gonzalez del Campo, A.; Lobato, J.; Cañizares, P.; Rodrigo, M.A.; Fernandez Morales, F.J. Short-term effects of temperature and COD in a microbial fuel cell. Appl. Energy 2013, 101, 213–217. [Google Scholar] [CrossRef]
- Mateo, S.; Gonzalez Del Campo, A.; Lobato, J.; Rodrigo, M.; Cañizares, P.; Fernandez-Morales, F.J. Long-term effects of the transient COD concentration on the performance of microbial fuel cells. Biotechnol. Prog. 2016, 32, 883–890. [Google Scholar] [CrossRef]
- Mahmood, N.A.N.; Ghazali, N.F.; Ibrahim, K.A.; Ali, M.A. Anodic pH Evaluation on performance of power generation from palm oil empty fruit bunch (EFB) in dual chambered microbial fuel cell (MFC). Chem. Eng. Trans. 2017, 56, 1795–1800. [Google Scholar]
- López Velarde Santos, M.; Rodríguez Valadéz, F.J.; Mora Solís, V.; González Nava, C.; Cornejo Martell, A.J.; Hensel, O. Performance of a microbial fuel cell operated with vinasses using different cod concentrations. Rev. Int. Contam. Ambient. 2017, 33, 521–528. [Google Scholar] [CrossRef]
- Algar, C.K.; Howard, A.; Ward, C.; Wanger, G. Sediment microbial fuel cells as a barrier to sulfide accumulation and their potential for sediment remediation beneath aquaculture pens. Sci. Rep. 2020, 10, 13087. [Google Scholar] [CrossRef]
- Linares, R.V.; Domínguez-Maldonado, J.; Rodríguez-Leal, E.; Patrón, G.; Castillo-Hernández, A.; Miranda, A.; Romero, D.D.; Moreno-Cervera, R.; Camara-chale, G.; Borroto, C.G.; et al. Scale up of microbial fuel cell stack system for residential wastewater treatment in continuous mode operation. Water 2019, 11, 217. [Google Scholar] [CrossRef]
- Goto, Y.; Yoshida, N. Scaling up Microbial Fuel Cells for Treating Swine Wastewater. Water 2019, 11, 1803. [Google Scholar] [CrossRef]
- Scott, K.; Yu, E.H.; Ghangrekar, M.M.; Erable, B.; Duteanu, N.M. Biological and microbial fuel cells. Compr. Renew. Energy 2012, 4, 277–300. [Google Scholar]
- Cheng, S.; Liu, H.; Logan, B.E. Increased performance of single-chamber microbial fuel cells using an improved cathode structure. Electrochem. Commun. 2006, 8, 489–494. [Google Scholar] [CrossRef]
- Zhang, X.; He, W.; Yang, W.; Liu, J.; Wang, Q.; Liang, P.; Huang, X.; Logan, B.E. Diffusion layer characteristics for increasing the performance of activated carbon air cathodes in microbial fuel cells. Environ. Sci. Water Res. Technol. 2016, 2, 266–273. [Google Scholar] [CrossRef]
- Midyurova, B.; Nenov, V. Electricity Generation in Microbial Fuel Cells as a Function of Air: Cathode Configuration. Int. J. Altern. Fuels Energy 2017, 1, 9–13. [Google Scholar]
- Walter, X.A.; Greenman, J.; Ieropoulos, I. Binder materials for the cathodes applied to self-stratifying membraneless microbial fuel cell. Bioelectrochemistry 2018, 123, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, B.; An, J.; Lee, Y.S.; Chang, I.S. Development of anode zone using dual-anode system to reduce organic matter crossover in membraneless microbial fuel cells. Bioresour. Technol. 2016, 213, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Rozendal, R.A.; Hamelers, H.V.M.; Molenkamp, R.J.; Buisman, C.J.N. Performance of single chamber biocatalyzed electrolysis with different types of ion exchange membranes. Water Res. 2007, 41, 1984–1994. [Google Scholar] [CrossRef]
- Sleutels, T.H.J.A.; Darus, L.; Hamelers, H.V.M.; Buisman, C.J.N. Effect of operational parameters on Coulombic efficiency in bioelectrochemical systems. Bioresour. Technol. 2011, 102, 11172–11176. [Google Scholar] [CrossRef] [PubMed]
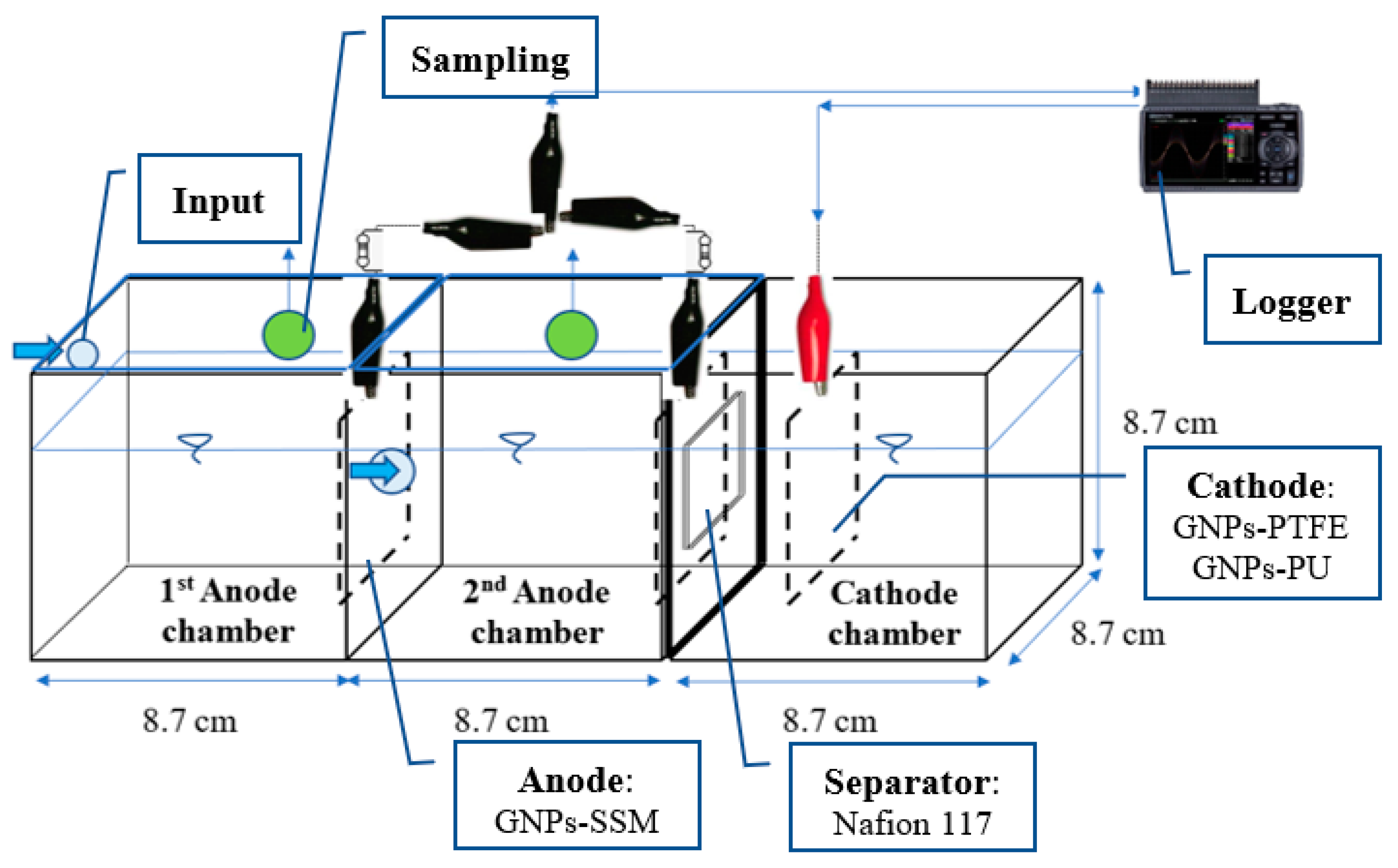
| Reactor Type | Anolyte | Anode | Separator | Catholyte | Cathode | |
|---|---|---|---|---|---|---|
| A1 | A2 | |||||
| 1 | Sucrose-based substrate | GNPs-SSM 30 mesh | GNPs-SSM 30 mesh | CEM (Nafion 117) | 2 g/L Na2CO3.1.5H2O2 | GNPs:PU 1:1(v/v)-SSM 30 mesh |
| 2 | Sucrose-based substrate | GNPs-SSM 30 mesh | GNPs-SSM 30 mesh | CEM (Nafion 117) | 2 g/L Na2CO3.1.5H2O2 | GNPs:PTFE 60%-SSM 30 mesh |
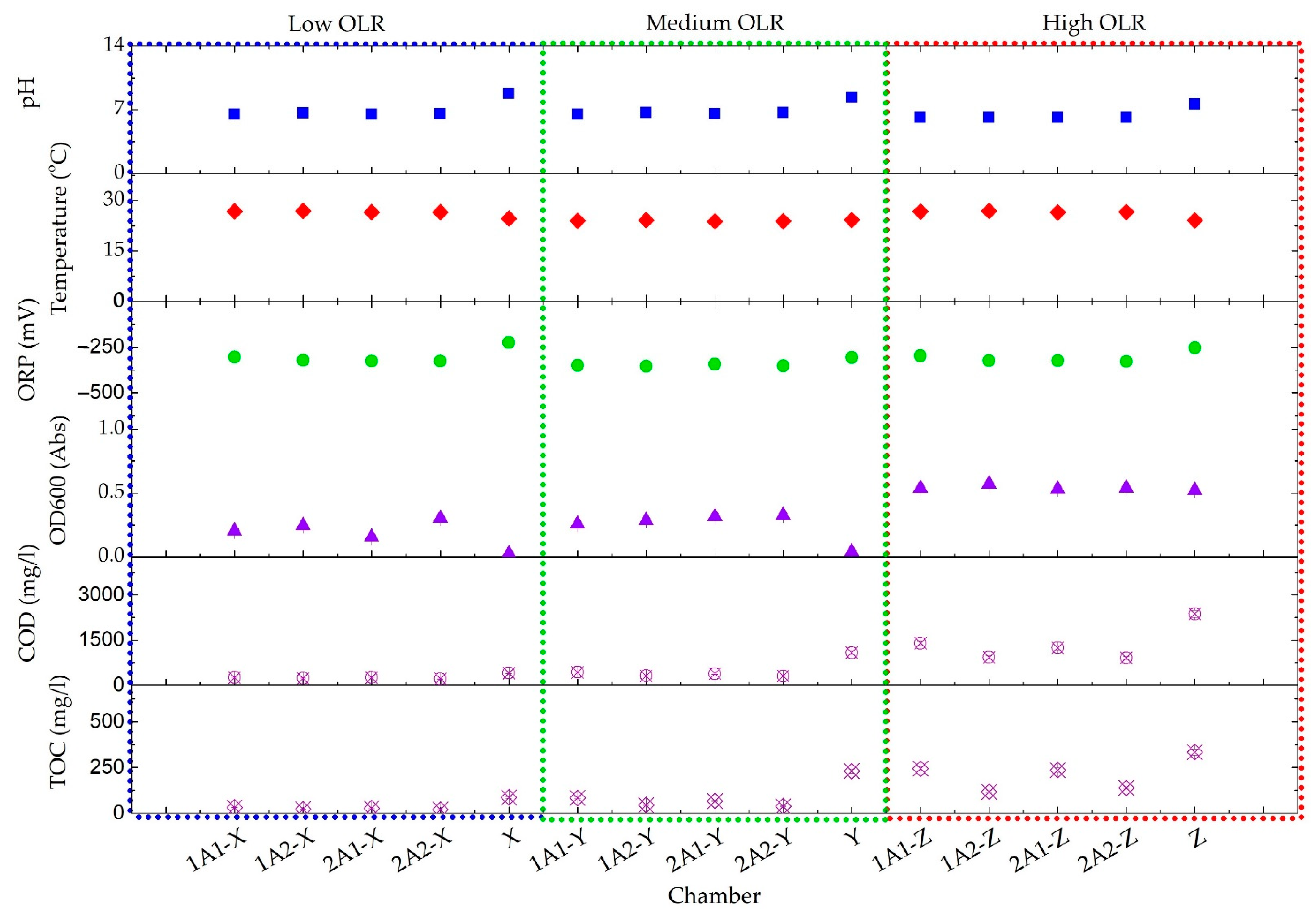
| Parameters | Low OLR | Medium OLR | High OLR |
|---|---|---|---|
| pH | 8.8 ± 0.09 | 8.3 ± 0.1 | 7.6 ± 0.01 |
| Temperature (°C) | 24.7 ± 1.09 | 24.2 ± 0.34 | 24.2 ± 0.12 |
| ORP (mV) | −226 ± 3.98 | −306 ± 3.98 | −255 ± 7.74 |
| OD600 (Abs) | 0.025 ± 0.007 | 0.04 ± 0.011 | 0.5 ± 0.29 |
| COD (mg/L) | 407 ± 68.89 | 1040 ± 160 | 2387 ± 205 |
| TOC (mg/L) | 87 ± 2.08 | 230 ± 4.65 | 724 ± 40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samudro, G.; Imai, T.; Hung, Y.-T. Enhancement of Power Generation and Organic Removal in Double Anode Chamber Designed Dual-Chamber Microbial Fuel Cell (DAC-DCMFC). Water 2021, 13, 2941. https://doi.org/10.3390/w13212941
Samudro G, Imai T, Hung Y-T. Enhancement of Power Generation and Organic Removal in Double Anode Chamber Designed Dual-Chamber Microbial Fuel Cell (DAC-DCMFC). Water. 2021; 13(21):2941. https://doi.org/10.3390/w13212941
Chicago/Turabian StyleSamudro, Ganjar, Tsuyoshi Imai, and Yung-Tse Hung. 2021. "Enhancement of Power Generation and Organic Removal in Double Anode Chamber Designed Dual-Chamber Microbial Fuel Cell (DAC-DCMFC)" Water 13, no. 21: 2941. https://doi.org/10.3390/w13212941
APA StyleSamudro, G., Imai, T., & Hung, Y.-T. (2021). Enhancement of Power Generation and Organic Removal in Double Anode Chamber Designed Dual-Chamber Microbial Fuel Cell (DAC-DCMFC). Water, 13(21), 2941. https://doi.org/10.3390/w13212941








