Drinking Water Standards and Their Implementation—A Critical Assessment
Abstract
1. Introduction
2. Contaminant Categories—Regulated Parameters
- Index organisms and model organisms, suggestive of pathogen presence and behavior, respectively, such as Clostridium perfringens, an index for enteric viruses.
3. Assessment of Standards and Regulated Parameters
3.1. Chemical Contaminants
3.1.1. Overview of Standards and Regulations
3.1.2. Contaminants of Emerging Concern (CEC)
- Statutory DW guideline values;
- Provisional guideline values based on recent toxicity data in absence of DW guidelines;
- Generic DW target values in absence of toxicity data.
3.1.3. Per- and Poly-fluoroalkyl Substances (PFAS)
3.1.4. Disinfection Byproducts (DBP)
3.2. Microbiological Contaminants
3.2.1. Indicator Microorganisms
- New infections resulting from changes in, or evolution of, existing organisms.
- Known infections spreading to new geographic areas or populations.
- Previously unrecognized infections appearing in areas undergoing ecologic transformation.
- Old infections re-emerging as a result of antibiotic resistance in known agents or breakdowns in public health measures.
- Which viruses could be used as biological markers;
- Which volume or amount of sample is ideal for virus detection;
- Which method is best for concentrating viruses from different matrices;
- When the use of genome detection or infectivity properties is more appropriate.
3.2.2. Antibiotic-Resistant Bacteria and Antibiotic-Resistance Genes (ARG)
3.3. Radiological Contaminants
“…it is not appropriate at this time to revise any of the NPDWRs covered under the Phase Rules or Radionuclide Rules. These NPDWRs were determined not to be candidates for revision for one or more of the following reasons: There was no new information to suggest possible changes in MCLG/MCL; new information did not present a meaningful opportunity for health risk reduction or cost savings while maintaining/improving public health protection; or there was an ongoing or pending regulatory action” [108].
4. Assessment of Standards Implementation
4.1. Monitoring Mode of Surveillance/Sampling
- The identification of the hazards associated with the catchment areas for abstraction points;
- A possibility for the water supplier to adapt monitoring to the main risks and to take the necessary measures to manage the risks identified in the supply chain from the abstraction, treatment, storage, and distribution of water;
- An assessment of the potential risks stemming from domestic DS, such as Legionella or lead.
“…If a parameter is not detected, water suppliers should be able to decrease the monitoring frequency or to stop monitoring that parameter altogether. Risk assessment and risk management of the supply system should be carried out for most parameters…This Directive mainly sets provisions on monitoring frequency for the purposes of compliance checks, with only limited provisions on monitoring for operational purposes…Such additional monitoring should be performed at the discretion of water suppliers. In that regard, water suppliers could refer to the WHO Guidelines and Water Safety Plan Manual.” [6].
- Groundwater systems: a minimum of one sample at every entry point to the DS, which is representative of each well after treatment, beginning in the initial compliance period.
- Surface water systems: a minimum of one sample at every entry point to the DS after any application of treatment or in the DS at a point, which is representative of each source after treatment in the initial compliance period.
- If a system draws water from more than one source and the sources are combined before distribution, the system must sample at an entry point to the DS during periods of normal operating conditions (i.e., when water is representative of all sources used).
4.2. Accepted Analytical Techniques
5. Discussion
- Identifying the characteristics of source water and determining its required treatment.
- Evaluating the performance/efficacy of DW treatment plants.
- Monitoring the (mostly undesirable and/or unforeseen) processes taking place in the DW DS (including the detection of accidental or intentional release of harmful substances).
- Insufficient or contradictory data regarding the contaminant’s presence and persistence in DW.
- Drawbacks in the available analytical methods/procedures for the identification and quantification of contaminants, including sampling protocols and lack of standardization thereof (e.g., large volumes of water samples required for some contaminant classes).
- The significant cost of monitoring (e.g., for specialized analytical equipment and personnel and extensive water supply risk assessment associated with the WSP approach).
- The measured quality parameters;
- The frequency of sampling;
- The points of compliance;
- The specifications of the employed/approved analytical methods.
6. Conclusions
- Completeness. There are serious deficiencies regarding inclusion in the standards mainly of chemical (CECs, DBPs, PFAS, perchlorate, and Cr (VI)) and microbiological contaminants (enteric viruses, Pseudomonas, Cryptosporidium, Giardia, Legionella, ARB/ARGs, and enveloped viruses).
- Adequacy. The most serious issues are related to the microbiological contaminants regarding the representativeness of indices, currently employed not for individual contaminants but for entire classes (e.g., bacteriophages as indicators of enteric viruses). Significant uncertainties are also identified regarding the stipulated limit values for several chemical contaminants.
- Implementation/monitoring. The importance of this aspect regarding drinking water quality is obvious, considering the variety of contamination sources in the distribution network, i.e., between the DW treatment plant and consumers taps. It is, therefore, understandable and appropriate that emphasis is placed on the regulations to adequately monitor DW quality. However, regarding effective monitoring, there are major issues and concerns due to the inherent deficiencies of the presently available/approved analytical techniques for determining chemical and, in particular, microbiological contaminants. Such deficiencies are related to the required sampling protocols (necessitating large sample volumes, adequacy of sensor networks, and sampling location/frequency) and in particular to the considerably long analysis time (of several hours) that essentially determines the response time of the entire monitoring system. Therefore, the time to implement such protocols is at present too long, rendering the system inefficient and incapable to cope with problems requiring relatively fast system response (e.g., unforeseen events, such as accidents and natural disasters). Moreover, the long analysis time (and delayed response) essentially neutralizes the benefits resulting from the recent progress made in fast signal acquisition/transmission and data collection and management systems.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Appendix A
| Contaminant | Inclusion in EU, USEPA, WHO Standards | Standards | Measurement Technique | Comments-Assessment | |
|---|---|---|---|---|---|
| Chemical Limit values or MCLs of chemical parameters | EU Directive 2020/2184 | Yes | Annex I, Part B |
| General comments:
|
| USEPA NPDWR | Yes | § 141.11 and § 141.50-51 |
| ||
| WHO Guidelines | Yes | Table 8.10, 8.13, 8.16 & Table A3.3/Annex 3 | Table A4.1-4 of Annex 4 a | ||
| CECs | EU Directive 2020/2184 | Pesticides b | 0.1 μg/L b | Pesticides: The performance characteristics for individual pesticides are given as an indication (Table A1 of Part B Annex III). Values for the uncertainty of measurement as low as 30% can be achieved for several pesticides, while higher values up to 80 % may be allowed for a number of pesticides. PAHs: the performance characteristics displayed in Table A1 of Part B, Annex III, apply to individual substances, specified at 25% of the parametric value. | |
| Pesticides Total c | 0.5 μg/L c | ||||
| PAHs d | 0.1 μg/L d | ||||
| USEPA NPDWR | A few EDCs and PPCPs in the CCL 4, not in enforceable standards |
| |||
| WHO Guidelines | Focus on pharmac/cals; no values | Pesticides: Table A4.5 of Annex 4 | |||
| PFAS | EU Directive 2020/2184 | Total e | 0.50 μg/L e | By 12 January 2024, the EU Commission shall establish technical guidelines regarding methods of analysis including detection limits, parametric values, and frequency of sampling. |
|
| Sum of PFAS f | 0.10 μg/L f | ||||
| USEPA NPDWR | No federal enforceable standards | Although they have been systematically detected in more than 25 states, there are no federal enforceable standards [57] | |||
| WHO Guidelines | No values | ||||
| Perchlorate | EU Regulation 2020/749 for chlorate residues in food and DW | Maximum residual level (MRL) | 0.01 mg/kg | [34] | |
| USEPA NPDWR | Unregulated | [38] | |||
| WHO Guidelines | Yes | 0.07 mg/L | |||
| DBP | EU Directive 2020/2184 | HAAs g | 60 μg/L g | ||
| THM (Total) h | 100 μg/L h | THM: the performance characteristics displayed in Table A1 of Part B, Annex III, apply to individual substances, specified at 25% of the parametric value. | |||
| USEPA NPDWR | Bromodichloro-methane, bromoform, bromate | 0 mg/L | § 141.131 and https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P100WD1L.txt (accessed on 8 September 2020) | ||
| chlorite, | 0.8 mg/L | ||||
| chloroform, | 0.07 mg/L | ||||
| dibromochloromethane, | 0.06 mg/L | ||||
| dichloroacetic acid | 0 mg/L | ||||
| Monochloroacetic acid | 0.07 mg/L | ||||
| Trichloroacetic acid | 0.02 mg/L | ||||
| WHO Guidelines | HAAs, THMs included in EU and USEPA regulations | Table 8.16 and Table A3.3 of Annex 3 | |||
| Microbiological Limit values, MCLGs and treatment techniques (TT) i for indicator pathogens | EU Directive 2020/2184 | Yes | Annex I, Parts A and C | Annex III, Part A | General comments: |
| USEPA NPDWR | Yes | § 141.52 | § 141.21 | ||
| WHO Guidelines | Yes | Table 7.5 and 7.10 | Table 7.11 with ISO standards for detection and quantification of fecal indicator organisms in water | ||
| Enteric viruses | EU Directive 2020/2184 | Not included |
| ||
| USEPA NPDWR | No MCL but MCLG or treatment technique (TT) i | Zero and 99.99% removal/inactivation | Not specified | ||
| WHO Guidelines | No value | ||||
| Pseudomonas | EU Directive 2020/2184 | Not included | Detected in treated water samples with free residual chlorine of 0.2–2.0 mg/L [79], confirming this species’ resistance to conventional water treatment processes [80] | ||
| USEPA NPDWR | No MCL but MCLG or TT i | Not specified | |||
| WHO Guidelines | No value | ||||
| Cryptosporidium | EU Directive 2020/2184 | Not included |
| ||
| USEPA NPDWR | No MCL but MCLG or TT i | Zero and 99% removal for filtration j | § 141.704 l | ||
| WHO Guidelines | No value | ||||
| Giardia lamblia | EU Directive 2020/2184 | Not included | |||
| USEPA NPDWR | No MCL but MCLG or TT i | Zero and 99.9% removal/inactivation | Not specified | ||
| WHO Guidelines | No value | ||||
| Legionella | EU Directive 2020/2184 | Limit value in the case of a risk assessment that indicates Legionella’s monitoring | <1000 CFU/L | In accordance with EN ISO 11731 For risk-based verification monitoring and to complement culture methods, methods such as ISO/TS 12869, rapid culture methods, non-culture-based methods, and molecular-based methods (in particular, qPCR), can be used. | |
| USEPA NPDWR | No MCL but MCLG or TT i | Zero k | Not specified |
| |
| WHO Guidelines | No value | ||||
| ARB and ARGs | EU Directive 2020/2184 | Not included | |||
| USEPA NPDWR | Not included | ||||
| WHO Guidelines | Not included | ||||
| Radiological Limit values or MCLs of radiological indicators | EU Directive 2013/51/EURATOM | Yes | Annex I | §3, Annex III | General comments: |
| USEPA NPDWR | Yes | § 141.66 | § 141.25 and https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P100WD57.txt (accessed on 11 October 2020) | ||
| WHO Guidelines | Yes | Table 9.2, Box 9.4 and Table A6.1, Annex 5 | |||
| Abbreviation | Definition |
|---|---|
| 2,3,7,8-TCDD | 2,3,7,8-Tetrachlorodibenzodioxin |
| AdVs | Adenoviruses |
| AI | Artificial intelligence |
| AR | Antibiotic resistance |
| ARB | Antibiotic-resistant bacteria |
| ARGs | Antibiotic-resistant genes |
| CCA | Chromogenic coliform agar |
| CCL | Contaminant Candidate List |
| CDC | Centers for Disease Control and Prevention |
| CECs | Contaminants of emerging concern |
| COVID-19 | Coronavirus disease 2019 |
| CUPs | Current-use pesticides |
| CWS | Contamination warning system |
| DBPs | Disinfection byproducts |
| DOM | Dissolved organic matter |
| DPR | Direct potable reuse |
| DS | Distribution system |
| DW | Drinking water |
| DWD | Drinking Water Directive |
| E. coli | Escherichia coli |
| EBOV | Ebola virus |
| EC | Effective concentration |
| EDCs | Endocrine-disrupting compounds |
| EQS | Environmental quality standards |
| EWG | Environmental Working Group |
| GC/MS | Gas chromatography/mass spectrometry |
| HAAs | Haloacetic acids |
| HACCP | Hazard Assessment and Critical Control Points |
| HALs | Haloacetaldehydes |
| HRGC/HRMS | High-resolution (HR) gas chromatography/HR mass spectrometry |
| ICCs | Industrial and commercial compounds |
| ID | Indicative dose |
| ISO | International Organization for Standardization |
| LCR | Lead and Copper Rule |
| LLE | Liquid–liquid extraction |
| MCLs | Maximum contaminant levels |
| MCs | Microcystins |
| MERS-CoV | Middle East respiratory syndrome coronavirus |
| MPN | Most probable number |
| NOM | Natural organic matter |
| NPDWR | National Primary Drinking Water Regulations |
| NSFWQI | National Science Foundation Water Quality Index |
| ORP | Oxidation reduction potential |
| PAF | Population attributable fraction |
| PAHs | Polyaromatic hydrocarbons |
| PFAS | Per- and poly-fluoroalkyl substances |
| PFOA | Perfluorooctanoic acid |
| PFOS | Perfluorooctane sulfonic acid |
| PhACs | Pharmaceuticals |
| PPCPs | Personal care products |
| PWS | Public water system |
| qPCR | Quantitative polymerase chain reaction |
| R&D | Research & development |
| REMdb | Radioactivity Environmental Monitoring database |
| RNA | Ribonucleic acid |
| RTCR | Revised Total Coliform Rule |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| SDWA | Safe Drinking Water Act |
| SWTR | Surface Water Treatment Rule |
| TCR | Total Coliform Rule |
| THMs | Trihalomethanes |
| TOC | Total organic carbon |
| UCMR | Unregulated Contaminant Monitoring Regulation |
| USEPA | United States Environmental Protection Agency |
| UV | Ultraviolet |
| WDS | Water distribution system |
| WHO | World Health Organization |
| WQI | Water quality index |
| WRD | Water-related disease |
| WSP | Water safety plan |
| WWTPs | Waste water treatment plants |
References
- Ashbolt, N.J. Microbial Contamination of Drinking Water and Human Health from Community Water Systems. Curr. Environ. Health Rep. 2015, 2, 95–106. [Google Scholar] [CrossRef]
- Tsoukalas, D.S.; Tsitsifli, S. A Critical Evaluation of Water Safety Plans (WSPs) and HACCP Implementation in Water Utilities. Proceedings 2018, 2, 600. [Google Scholar] [CrossRef]
- Curnin, S.; Brooks, B. Making waves: How do we prepare for the next drinking water disaster? Water Res. 2020, 185, 116277. [Google Scholar] [CrossRef] [PubMed]
- International Trade Center (ITC). Difference Between Standards and Technical Regulations. Available online: https://www.intracen.org/Part-3-Difference-between-standards-and-technical-regulations/ (accessed on 4 November 2020).
- World Health Organization (WHO). Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- European Union (EU). Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the quality of water intended for human consumption. Off. J. Eur. Union 2020, 435, 1–62. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Safe Drinking Water Act Amendments of 1996. Authenticated US Gov. Inf. 1996, 110, 1613–1693. [Google Scholar]
- Peyravi, M.; Jahanshahi, M.; Tourani, H. Analytical methods of water pollutants detection. In Inorganic Pollutants in Water; Elsevier: Amsterdam, The Netherlands, 2020; pp. 97–113. [Google Scholar]
- Cuerda-Correa, E.M.; Alexandre-Franco, M.F.; Fernández-González, C. Advanced Oxidation Processes for the Removal of Antibiotics from Water. An Overview. Water 2020, 12, 102. [Google Scholar] [CrossRef]
- Da Luz, N.; Kumpel, E. Evaluating the impact of sampling design on drinking water quality monitoring program outcomes. Water Res. 2020, 185, 116217. [Google Scholar] [CrossRef] [PubMed]
- Karabelas, A.J.; Plakas, K.V. Membrane Treatment of Potable Water for Pesticides Removal. In Herbicides, Theory and Applications; Soloneski, S., Larramendy, M.L., Eds.; InTech: Rijeka, Croatia, 2011; pp. 369–408. [Google Scholar]
- Khan, J.A.; Sayed, M.; Khan, S.; Shah, N.S.; Dionysiou, D.D.; Boczkaj, G. Advanced Oxidation Processes for the Treatment of Contaminants of Emerging Concern. In Contaminants of Emerging Concern in Water and Wastewater—Advanced Treatment Processes; Hernandez-Maldonado, A., Blaney, L., Eds.; Butterworth-Heinemann: Oxford, UK, 2020; pp. 299–365. [Google Scholar]
- Plakas, K.V.; Karabelas, A.J. Electro-Fenton Applications in the Water Industry. In Electro-Fenton Applications; Springer: Singapore, 2018; pp. 343–378. [Google Scholar]
- Plakas, K.V.; Sarasidis, V.C.; Patsios, S.I.; Lambropoulou, D.A.; Karabelas, A.J. Novel pilot scale continuous photocatalytic membrane reactor for removal of organic micropollutants from water. Chem. Eng. J. 2016, 304, 335–343. [Google Scholar] [CrossRef]
- Pandey, P.K.; Kass, P.H.; Soupir, M.L.; Biswas, S.; Singh, V.P. Contamination of water resources by pathogenic bacteria. AMB Express 2014, 4, 1–16. [Google Scholar] [CrossRef]
- Kulinkina, A.V.; Shinee, E.; Herrador, B.R.G.; Nygard, K.; Schmoll, O. The Situation of Water-Related Infectious Diseases in the Pan-European Region; WHO and UNECE: Copenhagen, Denmark, 2016. [Google Scholar]
- Jiménez, A.; Saikia, P.; Giné, R.; Avello, P.; Leten, J.; Liss Lymer, B.; Schneider, K.; Ward, R. Unpacking Water Governance: A Framework for Practitioners. Water 2020, 12, 827. [Google Scholar] [CrossRef]
- Hrudey, E.J.; Hrudey, S.E. Common themes contributing to recent drinking water disease outbreaks in affluent nations. Water Supply 2019, 19, 1767–1777. [Google Scholar] [CrossRef]
- Gunnarsdottir, M.J.; Gardarsson, S.M.; Figueras, M.J.; Puigdomenech, C.; Juarez, R.; Saucedo, G.; Arnedo, M.J.; Santos, R.; Monteiro, S.; Avery, L.; et al. Water safety plan enhancements with improved drinking water quality detection techniques. Sci. Total Environ. 2020, 698, 134185. [Google Scholar] [CrossRef]
- Said, A.; Stevens, D.K.; Sehlke, G. An innovative index for evaluating water quality in streams. Environ. Manag. 2004, 34, 406–414. [Google Scholar] [CrossRef]
- Noori, R.; Berndtsson, R.; Hosseinzadeh, M.; Adamowski, J.F.; Abyaneh, M.R. A critical review on the application of the National Sanitation Foundation Water Quality Index. Environ. Pollut. 2019, 244, 575–587. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Code of Federal Regulations. Title 40-Protection of the Environment. Chapter I. Environmental Protection Agency Subchapter D. Water Programs Part 141. National Primary Drinking Water Regulations. Available online: https://www.epa.gov/sites/default/files/2015-11/documents/howepargulates_cfr-2003-title40-vol20-part141_0.pdf (accessed on 21 May 2020).
- Pontius, F.W. Update on USEPA’s drinking water regulations. AWWA 2003, 95, 57–68. [Google Scholar] [CrossRef]
- Russo, F.; Nemer, L.; Martuzzi, M.; Zambon, F. Keeping our Water Clean: The Case of Water Contamination in the Veneto Region, Italy; WHO Regional Office for Europe Italy: Venice, Italy, 2017. [Google Scholar]
- Freeman, J.L. Human Health Concerns of Drinking Water Chemical Contaminants in Eastern India. In Proceedings of the Sustainable Solution With Appropriate Technological Development and Innovation (SWADIN), Kharagpur, India, 3–6 March 2013; pp. 22–27. [Google Scholar]
- Karabelas, A.J.; Plakas, K.V.; Solomou, E.S.; Drossou, V.; Sarigiannis, D.A. Impact of European legislation on marketed pesticides—A view from the standpoint of health impact assessment studies. Environ. Int. 2009, 35, 1096–1107. [Google Scholar] [CrossRef]
- Gray, N.F. Pathogen Control in Drinking Water. In Microbiology of Waterborne Diseases; Elsevier: Amsterdam, The Netherlands, 2014; pp. 537–569. [Google Scholar]
- Razzolini, M.T.P.; Breternitz, B.S.; Kuchkarian, B.; Bastos, V.K. Cryptosporidium and Giardia in urban wastewater: A challenge to overcome. Environ. Pollut. 2020, 257, 113545. [Google Scholar] [CrossRef] [PubMed]
- Ashbolt, N.J.; Grabow, W.O.K.; Snozzi, M. Indicators of microbial water quality. In Water Quality: Guidelines, Standards and Health; World Health Organization: London, UK, 2001; pp. 289–316. [Google Scholar]
- Lin, J.; Ganesh, A. Water quality indicators: Bacteria, coliphages, enteric viruses. Int J. Environ. Health Res. 2013, 23, 484–506. [Google Scholar] [CrossRef] [PubMed]
- Nuccetelli, C.; Rusconi, R.; Forte, M. Radioactivity in drinking water: Regulations, monitoring results and radiation protection issues. Ann. Ist. Super Sanita 2012, 48, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Weinhold, B. Unknown Quantity. Regulating Radionuclides in Tap Water. Environ. Health Perspect. 2012, 120, A350–A356. [Google Scholar] [CrossRef] [PubMed]
- McCasland, M.; Trautmann, N.M.; Porter, K.S.; Wagenet, R.J. Nitrate: Health Effects in Drinking Water. Available online: http://psep.cce.cornell.edu/facts-slides-self/facts/nit-heef-grw85.aspx (accessed on 19 August 2020).
- European Commission (EC). Commission Regulation (EU) 2020/749 of 4 June 2020 amending Annex III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for chlorate in or on certain products. Off. J. Eur. Union 2020, 178, 7–20. [Google Scholar]
- Heo, J.; Kim, S.; Her, N.; Park, C.M.; Yu, M.; Yoon, Y. Removal of contaminants of emerging concern by FO, RO, and UF membranes in water and wastewater. In Contaminants of Emerging Concern in Water and Wastewater; Elsevier: Amsterdam, The Netherlands, 2020; pp. 139–176. [Google Scholar]
- USEPA. Revisions to the Unregulated Contaminant Monitoring Regulation (UCMR 3) for Public Water Systems 77 FR 26072 2012, 26072–26101. Available online: https://www.federalregister.gov/documents/2012/05/02/2012-9978/revisions-to-the-unregulated-contaminant-monitoring-regulation-ucmr-3-for-public-water-systems (accessed on 6 October 2020).
- Richardson, S.D.; Plewa, M.J. To regulate or not to regulate? What to do with more toxic disinfection by-products? J. Environ. Chem. Eng. 2020, 8, 103939. [Google Scholar] [CrossRef]
- USEPA. Drinking Water: Final Action on Perchlorate. Fed. Regist. 2020, 85, 43990–44002. Available online: https://www.federalregister.gov/documents/2020/07/21/2020-13462/drinking-water-final-action-on-perchlorate (accessed on 11 September 2020).
- Dong, H.; Xu, L.; Mao, Y.; Wang, Y.; Duan, S.; Lian, J.; Li, J.; Yu, J.; Qiang, Z. Effective abatement of 29 pesticides in full-scale advanced treatment processes of drinking water: From concentration to human exposure risk. J. Hazard. Mater. 2021, 403, 123986. [Google Scholar] [CrossRef]
- Krzeminski, P.; Tomei, M.C.; Karaolia, P.; Langenhoff, A.; Almeida, C.M.R.; Felis, E.; Gritten, F.; Andersen, H.R.; Fernandes, T.; Manaia, C.M.; et al. Performance of secondary wastewater treatment methods for the removal of contaminants of emerging concern implicated in crop uptake and antibiotic resistance spread: A review. Sci Total Environ. 2019, 648, 1052–1081. [Google Scholar] [CrossRef]
- Memon, F.A.; Savic, D.; Johns, M.B.; Sadr, S.M.K.; Visanji, Z. Optimising wastewater treatment solutions for the removal of contaminants of emerging concern (CECs): A case study for application in India. J. Hydroinform. 2020, 22, 93–110. [Google Scholar] [CrossRef]
- Kim, S.; Chu, K.H.; Al-Hamadani, Y.A.J.; Park, C.M.; Jang, M.; Kim, D.-H.; Yu, M.; Heo, J.; Yoon, Y. Removal of contaminants of emerging concern by membranes in water and wastewater: A review. Chem. Eng. J. 2018, 335, 896–914. [Google Scholar] [CrossRef]
- Vidal-Dorsch, D.E.; Bay, S.M.; Maruya, K.; Snyder, S.A.; Trenholm, R.A.; Vanderford, B.J. Contaminants of emerging concern in municipal wastewater effluents and marine receiving water. Environ. Toxicol. Chem. 2012, 31, 2674–2682. [Google Scholar] [CrossRef]
- Troger, R.; Ren, H.; Yin, D.; Postigo, C.; Nguyen, P.D.; Baduel, C.; Golovko, O.; Been, F.; Joerss, H.; Boleda, M.R.; et al. What’s in the water? Target and suspect screening of contaminants of emerging concern in raw water and drinking water from Europe and Asia. Water Res. 2021, 198, 117099. [Google Scholar] [CrossRef]
- Baken, K.A.; Sjerps, R.M.A.; Schriks, M.; van Wezel, A.P. Toxicological risk assessment and prioritization of drinking water relevant contaminants of emerging concern. Environ. Int. 2018, 118, 293–303. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA) Chemical Contaminants—CCL 4. Available online: https://www.epa.gov/ccl/chemical-contaminants-ccl-4 (accessed on 10 October 2020).
- United States Environmental Protection Agency (USEPA) Microbial Contaminants—CCL 4. Available online: https://www.epa.gov/ccl/microbial-contaminants-ccl-4 (accessed on 12 September 2020).
- California Water Boards (CWB) Drinking Water Programs. The Division of Drinking Water (DDW) Regulates Public Drinking Water Systems. Available online: https://www.waterboards.ca.gov/drinking_water/programs/ (accessed on 16 November 2020).
- European Union (EU). Directive 2000/60/EC of the european parliament and of the council of 23 October 2000 establishing a framework for Community action in the field of water policy. Ammended by Commission Directive 2014/101/EU of 30 October 2014. Off. J. Eur. Communities 2014, 327, 1–93. [Google Scholar]
- Kreuzinger, N.; Murphy, D.; Greene, E. Continuous Increase of CECs in the Anthroposphere as a Stressor for Water Resources. Available online: http://www.waterjpi.eu/implementation/thematic-activities/water-jpi-knowledge-hub-1/jpi-khcec_january_2020_stakeholderbrief.pdf (accessed on 18 December 2020).
- European Commission (EC). Communication from the Commission to the European Parliament, the Council and the European Economic and Social Committee. European Union Strategic Approach to Pharmaceuticals in the Environment. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52019DC0128&from=EN (accessed on 24 June 2021).
- Kondor, A.C.; Molnár, É.; Vancsik, A.; Filep, T.; Szeberényi, J.; Szabó, L.; Maász, G.; Pirger, Z.; Weiperth, A.; Ferincz, Á.; et al. Occurrence and health risk assessment of pharmaceutically active compounds in riverbank filtrated drinking water. J. Water Process. Eng. 2021, 41, 102039. [Google Scholar] [CrossRef]
- Menger, R.F.; Funk, E.; Henry, C.S.; Borch, T. Sensors for detecting per- and polyfluoroalkyl substances (PFAS): A critical review of development challenges, current sensors, and commercialization obstacles. Chem. Eng. J. 2021, 417, 129133. [Google Scholar] [CrossRef]
- Kwiatkowski, C.F.; Andrews, D.Q.; Birnbaum, L.S.; Bruton, T.A.; DeWitt, J.C.; Knappe, D.R.U.; Maffini, M.V.; Miller, M.F.; Pelch, K.E.; Reade, A.; et al. Scientific Basis for Managing PFAS as a Chemical Class. Environ. Sci. Technol. Lett. 2020, 7, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.Y.; Ekpe, O.D.; Lee, H.J.; Oh, J.E. Perfluoroalkyl substances and pharmaceuticals removal in full-scale drinking water treatment plants. J. Hazard. Mater. 2020, 400, 123235. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. PFAS in Drinking Water and Self-Caught Fish—Risk Management. Available online: https://www.livsmedelsverket.se/en/production-control-and-trade/drinking-water-production-and-control/t#New_limit_values_for_PFAS_in_drinking_water (accessed on 25 June 2021).
- Hogue, C. Why limiting PFAS in drinking water is a challenge in the US. Chem. Eng. News 2020, 98. Available online: https://cen.acs.org/environment/persistent-pollutants/limiting-PFAS-drinking-water-challenge/98/i27 (accessed on 10 August 2020).
- USEPA. Drinking Water Health Advisories for PFOA and PFOS. Available online: https://www.epa.gov/ground-water-and-drinking-water/drinking-water-health-advisories-pfoa-and-pfos (accessed on 20 August 2020).
- Andrews, D. EWG Proposes PFAS Standards That Fully Protect Children’s Health. Available online: https://www.ewg.org/research/ewg-proposes-pfas-standards-fully-protect-children-s-health (accessed on 27 October 2020).
- Coalition of Trade Associations (CTA) RE: Comments on Preliminary PFOS and PFOA Regulatory Determination Under the Safe Drinking Water Act (EPA–HQ–OW–2019–0583; FRL–10005–88–OW). Available online: https://saferchemicals.org/wp-content/uploads/2021/01/pfas-reg_det_comments_6102020.pdf (accessed on 5 September 2020).
- Diana, M.; Felipe-Sotelo, M.; Bond, T. Disinfection byproducts potentially responsible for the association between chlorinated drinking water and bladder cancer: A review. Water Res. 2019, 162, 492–504. [Google Scholar] [CrossRef]
- Ding, S.; Deng, Y.; Bond, T.; Fang, C.; Cao, Z.; Chu, W. Disinfection byproduct formation during drinking water treatment and distribution: A review of unintended effects of engineering agents and materials. Water Res. 2019, 160, 313–329. [Google Scholar] [CrossRef] [PubMed]
- Evlampidou, I.; Font-Ribera, L.; Rojas-Rueda, D.; Gracia-Lavedan, E.; Costet, N.; Pearce, N.; Vineis, P.; Jaakkola, J.J.K.; Delloye, F.; Makris, K.C.; et al. Trihalomethanes in Drinking Water and Bladder Cancer Burden in the European Union. Environ. Health Perspect. 2020, 128, 17001. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Chen, S.; Long, L.; Bu, Y.; Xu, H.; Chen, B.; Krasner, S. A one-year long survey of temporal disinfection byproducts variations in a consumer’s tap and their removals by a point-of-use facility. Water Res. 2019, 159, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Plewa, M.J.; Wagner, E.D.; Richardson, S.D. TIC-Tox: A preliminary discussion on identifying the forcing agents of DBP-mediated toxicity of disinfected water. J. Environ. Sci. 2017, 58, 208–216. [Google Scholar] [CrossRef]
- Leclerc, H.; Edberg, S.; Pierzo, V.; Delattre, J.M. Bacteriophages as indicators of enteric viruses and public health risk in groundwaters. J. Appl. Microbiol. 2000, 88, 5–21. [Google Scholar] [CrossRef]
- Payment, P.; Berte, A.; Prevost, M.; Menard, B.; Barbeau, B. Occurrence of pathogenic microorganisms in the Saint Lawrence River (Canada) and comparison of health risks for populations using it as their source of drinking water. Can. J. Microbiol 2000, 46, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Verani, M.; Federigi, I.; Donzelli, G.; Cioni, L.; Carducci, A. Human adenoviruses as waterborne index pathogens and their use for Quantitative Microbial Risk Assessment. Sci. Total Environ. 2019, 651, 1469–1475. [Google Scholar] [CrossRef]
- Hai, F.; Riley, T.; Shawkat, S.; Magram, S.; Yamamoto, K. Removal of Pathogens by Membrane Bioreactors: A Review of the Mechanisms, Influencing Factors and Reduction in Chemical Disinfectant Dosing. Water 2014, 6, 3603–3630. [Google Scholar] [CrossRef]
- Bonadonna, L.; Briancesco, R.; Suffredini, E.; Coccia, A.; Della Libera, S.; Carducci, A.; Verani, M.; Federigi, I.; Iaconelli, M.; Bonanno Ferraro, G.; et al. Enteric viruses, somatic coliphages and Vibrio species in marine bathing and non-bathing waters in Italy. Mar. Pollut. Bull. 2019, 149, 110570. [Google Scholar] [CrossRef]
- Ye, Y.; Ellenberg, R.M.; Graham, K.E.; Wigginton, K.R. Survivability, Partitioning, and Recovery of Enveloped Viruses in Untreated Municipal Wastewater. Environ. Sci. Technol. 2016, 50, 5077–5085. [Google Scholar] [CrossRef]
- Allard, A.; Vantarakis, A.; Meschke, J.S.; Girones, R. Adenoviruses. In Water and Sanitation for the 21st Century: Health and Microbiological Aspects of Excreta and Wastewater Management (Global Water Pathogen Project); Meschke, J.S., Girones, R., Eds.; Michigan State University: Lansing, MI, USA, 2019. [Google Scholar]
- Jurzik, L.; Hamza, I.A.; Puchert, W.; Uberla, K.; Wilhelm, M. Chemical and microbiological parameters as possible indicators for human enteric viruses in surface water. Int. J. Hyg. Environ. Health 2010, 213, 210–216. [Google Scholar] [CrossRef]
- Truchado, P.; Garre, A.; Gil, M.I.; Simon-Andreu, P.J.; Sanchez, G.; Allende, A. Monitoring of human enteric virus and coliphages throughout water reuse system of wastewater treatment plants to irrigation endpoint of leafy greens. Sci. Total Environ. 2021, 782, 146837. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). Guidelines for Water Reuse (EPA/600/R-12/618). Available online: https://www.epa.gov/sites/default/files/2019-08/documents/2012-guidelines-water-reuse.pdf (accessed on 29 August 2020).
- Mena, K.D.; Gerba, C.P. Risk assessment of Pseudomonas aeruginosa in water. Rev. Environ. Contam. Toxicol. 2009, 201, 71–115. [Google Scholar] [CrossRef] [PubMed]
- Mores Rall, V.L.; Alves Dantas, S.T.; Nogueira Nascentes, G.A.; Pereira, V.B.R.; da Silva Ruiz, L.; Garbelotti, M.; Arantes Stancari, R.C.; Anversa, L. Pseudomonas aeruginosa in public water supply. Water Pract. Technol. 2019, 14, 732–737. [Google Scholar] [CrossRef]
- Guerra, N.M.M.; Otenio, M.H.; Silva, M.E.Z.; Guilhermetti, M.; Nakamura, C.V.; Ueda-Nakamura, T.; Filho, B.P.D. Ocorrência de Pseudomonas aeruginosa em água potável (Occurrence of Pseudomonas aeruginosa in potable water). Acta Sci. Biol. Sci. 2006, 28, 13–18. [Google Scholar] [CrossRef][Green Version]
- Buffer, J.; Williams, M.; Medeiros, L.; LeJeune, J. Parasites: Cryptosporidium parvum, Cyclospora, and Giardia lamblia; College of Food, Agricultural, and Environmental Sciences, The Ohio State University: Columbus, OH, USA. Available online: https://ohioline.osu.edu/factsheet/HYG-5577 (accessed on 22 August 2020).
- Kondo Nakada, L.Y.; Urbano Dos Santos, L.; Guimaraes, J.R. Pre-ozonation of surface water: An effective water treatment process to reduce the risk of infection by Giardia in drinking water. Environ. Pollut. 2020, 266, 115144. [Google Scholar] [CrossRef] [PubMed]
- Kumar, T.; Abd Majid, M.A.; Onichandran, S.; Jaturas, N.; Andiappan, H.; Salibay, C.C.; Tabo, H.A.; Tabo, N.; Dungca, J.Z.; Tangpong, J.; et al. Presence of Cryptosporidium parvum and Giardia lamblia in water samples from Southeast Asia: Towards an integrated water detection system. Infect. Dis. Poverty 2016, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Betancourt, W.Q.; Rose, J.B. Drinking water treatment processes for removal of Cryptosporidium and Giardia. Vet. Parasitol. 2004, 126, 219–234. [Google Scholar] [CrossRef]
- Silva, K.J.S.; Sabogal-Paz, L.P. Cryptosporidium spp. and Giardia spp. (oo)cysts as target-organisms in sanitation and environmental monitoring: A review in microscopy-based viability assays. Water Res. 2020, 189, 116590. [Google Scholar] [CrossRef]
- Araujo, M.; Sueiro, R.A.; Gomez, M.J.; Garrido, M.J. Enumeration of Clostridium perfringens spores in groundwater samples: Comparison of six culture media. J. Microbiol. Methods 2004, 57, 175–180. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. Management of Legionella in Water Systems; National Academies Press: Washington, DC, USA, 2019. [Google Scholar]
- Wang, H.; Edwards, M.; Falkinham, J.O., 3rd; Pruden, A. Molecular survey of the occurrence of Legionella spp., Mycobacterium spp., Pseudomonas aeruginosa, and amoeba hosts in two chloraminated drinking water distribution systems. Appl. Environ. Microbiol. 2012, 78, 6285–6294. [Google Scholar] [CrossRef]
- Lu, J.; Struewing, I.; Vereen, E.; Kirby, A.E.; Levy, K.; Moe, C.; Ashbolt, N. Molecular Detection of Legionella spp. and their associations with Mycobacterium spp., Pseudomonas aeruginosa and amoeba hosts in a drinking water distribution system. J. Appl. Microbiol. 2016, 120, 509–521. [Google Scholar] [CrossRef]
- Naddeo, V.; Liu, H. Editorial Perspectives: 2019 novel coronavirus (SARS-CoV-2): What is its fate in urban water cycle and how can the water research community respond? Environ. Sci. Water Res. Technol. 2020, 6, 1213–1216. [Google Scholar] [CrossRef]
- Weber, D.J.; Rutala, W.A.; Fischer, W.A.; Kanamori, H.; Sickbert-Bennett, E.E. Emerging infectious diseases: Focus on infection control issues for novel coronaviruses (Severe Acute Respiratory Syndrome-CoV and Middle East Respiratory Syndrome-CoV), hemorrhagic fever viruses (Lassa and Ebola), and highly pathogenic avian influenza viruses, A(H5N1) and A(H7N9). Am. J. Infect. Control 2016, 44, e91–e100. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). EID Journal Background and Goals. “Emerging” Infectious Diseases. Available online: https://wwwnc.cdc.gov/eid/page/background-goals (accessed on 30 April 2020).
- Casanova, L.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Survival of surrogate coronaviruses in water. Water Res. 2009, 43, 1893–1898. [Google Scholar] [CrossRef]
- Wigginton, K.R.; Boehm, A.B. Environmental Engineers and Scientists Have Important Roles to Play in Stemming Outbreaks and Pandemics Caused by Enveloped Viruses. Environ. Sci. Technol. 2020, 54, 3736–3739. [Google Scholar] [CrossRef]
- Wolf-Baca, M.; Siedlecka, A. Detection of pathogenic bacteria in hot tap water using the qPCR method: Preliminary research. SN Appl. Sci. 2019, 1, 1–9. [Google Scholar] [CrossRef]
- WHO; UNICEF. Water, Sanitation, Hygiene, and Waste Management for the COVID-19 Virus: Interim Guidance. Available online: https://apps.who.int/iris/bitstream/handle/10665/331499/WHO-2019-nCoV-IPC_WASH-2020.2-eng.pdf?sequence=1&isAllowed=y (accessed on 3 February 2021).
- Gundy, P.M.; Gerba, C.P.; Pepper, I.L. Survival of Coronaviruses in Water and Wastewater. Food Environ. Virol. 2008, 1, 10–14. [Google Scholar] [CrossRef]
- Langone, M.; Petta, L.; Cellamare, C.M.; Ferraris, M.; Guzzinati, R.; Mattioli, D.; Sabia, G. SARS-CoV-2 in water services: Presence and impacts. Environ. Pollut. 2021, 268, 115806. [Google Scholar] [CrossRef] [PubMed]
- Karkman, A.; Do, T.T.; Walsh, F.; Virta, M.P.J. Antibiotic-Resistance Genes in Waste Water. Trends Microbiol. 2018, 26, 220–228. [Google Scholar] [CrossRef]
- Rizzo, L.; Manaia, C.; Merlin, C.; Schwartz, T.; Dagot, C.; Ploy, M.C.; Michael, I.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for antibiotic resistant bacteria and genes spread into the environment: A review. Sci. Total Environ. 2013, 447, 345–360. [Google Scholar] [CrossRef]
- Xu, L.; Zhou, Z.; Zhu, L.; Han, Y.; Lin, Z.; Feng, W.; Liu, Y.; Shuai, X.; Chen, H. Antibiotic resistance genes and microcystins in a drinking water treatment plant. Environ. Pollut. 2020, 258, 113718. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Infographic: Antibiotic resistance—An Increasing Threat to Human Health. Available online: https://www.ecdc.europa.eu/en/publications-data/infographic-antibiotic-resistance-increasing-threat-human-health (accessed on 30 April 2020).
- Sanganyado, E.; Gwenzi, W. Antibiotic resistance in drinking water systems: Occurrence, removal, and human health risks. Sci. Total Environ. 2019, 669, 785–797. [Google Scholar] [CrossRef]
- Chen, J.; Li, W.; Zhang, J.; Qi, W.; Li, Y.; Chen, S.; Zhou, W. Prevalence of antibiotic resistance genes in drinking water and biofilms: The correlation with the microbial community and opportunistic pathogens. Chemosphere 2020, 259, 127483. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, T.; Jiang, L.; Luo, Y.; Yao, S.; Zhang, D.; Lin, K.; Cui, C. Occurrence and reduction of antibiotic resistance genes in conventional and advanced drinking water treatment processes. Sci. Total Environ. 2019, 669, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Su, H.C.; Liu, Y.S.; Pan, C.G.; Chen, J.; He, L.Y.; Ying, G.G. Persistence of antibiotic resistance genes and bacterial community changes in drinking water treatment system: From drinking water source to tap water. Sci. Total Environ. 2018, 616–617, 453–461. [Google Scholar] [CrossRef]
- European Union (EU). Directive 2013/51/Euratom. Requirements for the protection of the health of the general public with regard to radioactive substances in water intended for human consumption. Off. J. Eur. Union 2013, 296, 12–21. [Google Scholar]
- European Union (EU). Council Regulation (Euratom) 2016/52 of 15 January 2016 laying down maximum permitted levels of radioactive contamination of food and feed following a nuclear accident or any other case of radiological emergency, and repealing Regulation (Euratom) No 3954/87 and Commission Regulations (Euratom) No 944/89 and (Euratom) No 770/90. Off. J. Eur. Union 2016, 327, 2–11. [Google Scholar]
- Grande, S.; Risica, S. Radionuclides in drinking water: The recent legislative requirements of the European Union. J. Radiol. Prot. 2015, 35, 1–19. [Google Scholar] [CrossRef] [PubMed][Green Version]
- United States Environmental Protection Agency (USEPA) National Primary Drinking Water Regulations; Announcement of the Results of EPA’s Review of Existing Drinking Water Standards and Request for Public Comment and/or Information on Related Issues. Fed. Regist. 2017, 82, 3518–3551.
- New Zealand Ministry of Health (NZMH) Radiological Compliance. In Drinking Water Guidelines-Water Quality Management for New Zealand, 3rd ed.; Ministry of Health: Wellington, New Zealand, 2019.
- Munter, R. Technology for the removal of radionuclides from natural water and waste management: State of the art. Proc. Est. Acad. Sci. 2013, 62, 122–132. [Google Scholar] [CrossRef]
- Korak, J.; Arias-Paic, M. Monitoring Strategies for Direct Reuse of Reclaimed Water; U.S. Department of the Interior, Bureau of Reclamation, Research and Development Office: Denver, CO, USA, 2016.
- International Organization for Standardization (ISO) 24512:2007. Activities Relating to Drinking Water and Wastewater Services—Guidelines for the Management of Drinking Water Utilities and for the Assessment of Drinking Water Services. Available online: https://www.iso.org/standard/37248.html (accessed on 21 August 2020).
- International Organization for Standardization (ISO). Standards by ISO/TC 224. Service Activities Relating to Drinking Water Supply, Wastewater and Stormwater Systems. Available online: https://www.iso.org/committee/299764/x/catalogue/ (accessed on 16 April 2021).
- United States Environmental Protection Agency (USEPA). Revised Total Coliform Rule: A Quick Reference Guide. Available online: https://www.epa.gov/dwreginfo/revised-total-coliform-rule-and-total-coliform-rule (accessed on 5 October 2020).
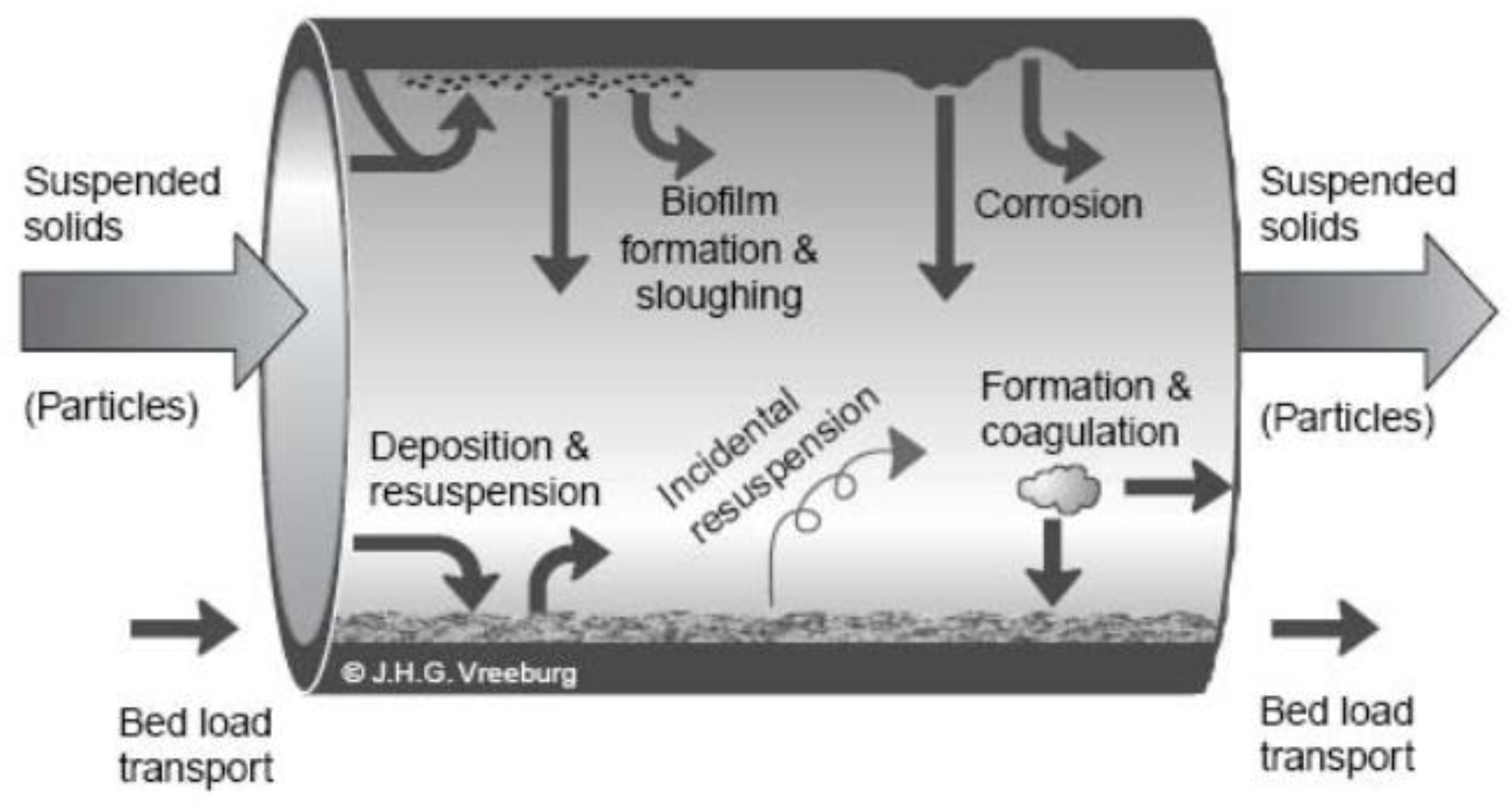
- Rubulis, J.; Verberk, J.; Vreeburg, J.; Gruškevica, K.; Juhna, T. Chemical and Microbial Composition of Loose Deposits in Drinking Water Distribution Systems. In Environmental Engineering: The 7th International Conference: Selected Papers. Vol. 2: Water Engineering. Energy for Buildings; Vilnius, Lithuania, 22–23 May 2008, Technika: Vilnius, Lithuania, 2008; pp. 695–702. [Google Scholar]
- Khorshidi, M.S.; Nikoo, M.R.; Sadegh, M. Optimal and objective placement of sensors in water distribution systems using information theory. Water Res. 2018, 143, 218–228. [Google Scholar] [CrossRef]
- Zhang, Q.; Zheng, F.; Kapelan, Z.; Savic, D.; He, G.; Ma, Y. Assessing the global resilience of water quality sensor placement strategies within water distribution systems. Water Res. 2020, 172, 115527. [Google Scholar] [CrossRef]
- Anonymous. Emerging home and hospital waterborne pathogens. Perspect. Public Health 2018, 138, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Lytle, D.A.; Sorg, T.; Wang, L.; Chen, A. The accumulation of radioactive contaminants in drinking water distribution systems. Water Res. 2014, 50, 396–407. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Lead and Copper Rule: A Quick Reference Guide. Available online: https://nepis.epa.gov/Exe/ZyNET.exe/60001N8P.TXT?ZyActionD=ZyDocument&Client=EPA&Index=2006+Thru+2010&Docs=&Query=&Time=&EndTime=&SearchMethod=1&TocRestrict=n&Toc=&TocEntry=&QField=&QFieldYear=&QFieldMonth=&QFieldDay=&IntQFieldOp=0&ExtQFieldOp=0&XmlQuery=&File=D%3A%5Czyfiles%5CIndex%20Data%5C06thru10%5CTxt%5C00000015%5C60001N8P.txt&User=ANONYMOUS&Password=anonymous&SortMethod=h%7C-&MaximumDocuments=1&FuzzyDegree=0&ImageQuality=r75g8/r75g8/x150y150g16/i425&Display=hpfr&DefSeekPage=x&SearchBack=ZyActionL&Back=ZyActionS&BackDesc=Results%20page&MaximumPages=1&ZyEntry=1&SeekPage=x&ZyPURL (accessed on 20 September 2020).
- Grayman, W.M. Contamination of Water Distribution Systems. In Proceedings of the International Seminar on Nuclear War and Planetary Emergencies—45th Session, Erice, Italy, 19–24 August 2012. [Google Scholar]
- Rutkowski, T.A.; Prokopiuk, F. Identification of the Contamination Source Location in the Drinking Water Distribution System Based on the Neural Network Classifier. IFAC Pap. 2018, 51, 15–22. [Google Scholar] [CrossRef]
- VEOLIA. AQUAVISTA™ Digital Services. Available online: https://www.veoliawatertechnologies.com/en/aquavista (accessed on 11 September 2020).
- Carminati, M.; Stefanelli, V.; Ferrari, G.; Sampietro, M.; Turolla, A.; Rossi, M.M.A.; Malavasi, S.; Antonelli, M.; Pifferi, V.; Falciola, L. Smart Pipe: A Miniaturized Sensor Platform for Real-Time Monitoring of Drinking Water Quality. In Proceedings of the IEEE Workshop on Environmental, Energy, and Structural Monitoring Systems (EESMS), Milan, Italy, 24–25 July 2017. [Google Scholar]
- Storey, M.V.; van der Gaag, B.; Burns, B.P. Advances in on-line drinking water quality monitoring and early warning systems. Water Res. 2011, 45, 741–747. [Google Scholar] [CrossRef]
- Anonymous akwaMetric. Smart Metering 4.0 for Water Quality Monitoring. Available online: https://www.iagua.es/market/grupo-mejoras/akwametric (accessed on 16 November 2020).
- Anonymous Smart Water 4.0. Water Quality Monitoring, IAgua, Smart Water Magazine. Available online: https://smartwatermagazine.com/news/grupo-mejoras/smart-water-40-water-quality-monitoring (accessed on 15 September 2020).
- International Organization for Standardization (ISO) 9308-1:2014. Water Quality—Enumeration of Escherichia coli and Coliform Bacteria—Part 1: Membrane Filtration Method for Waters with Low Bacterial Background Flora. Available online: https://www.iso.org/standard/55832.html (accessed on 27 August 2020).
- International Organization for Standardization (ISO) 9308-2:2012. Water Quality—Enumeration of Escherichia coli and Coliform Bacteria—Part 2: Most Probable Number Method. Available online: https://www.iso.org/standard/52246.html (accessed on 27 August 2020).
- Rhodes, E.R.; Hamilton, D.W.; See, M.J.; Wymer, L. Evaluation of hollow-fiber ultrafiltration primary concentration of pathogens and secondary concentration of viruses from water. J. Virol. Methods 2011, 176, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Sobiech-Matura, K.; Máté, B.; Altzitzoglou, T. Radioactivity monitoring in foodstuff and drinking water—An overview of performance of EU laboratories based on interlaboratory comparisons. Food Control. 2017, 72, 225–231. [Google Scholar] [CrossRef]
- Spellman, F.R. Handbook of Water and Wastewater Treatment Plant. Operations, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Szabo, J.; Hall, J. On-line Water Quality Monitoring for Drinking Water Contamination. In Comprehensive Water Quality and Purification; Elsevier: Amsterdam, The Netherlands, 2014; Volume 2, pp. 266–282. [Google Scholar]
- Metcalfe, C.; Hoque, M.E.; Sultana, T.; Murray, C.; Helm, P.; Kleywegt, S. Monitoring for contaminants of emerging concern in drinking water using POCIS passive samplers. Environ. Sci. Process. Impacts 2014, 16, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Petrie, B.; Barden, R.; Kasprzyk-Hordern, B. A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015, 72, 3–27. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (USEPA). Approved Drinking Water Analytical Methods. Available online: https://www.epa.gov/dwanalyticalmethods/approved-drinking-water-analytical-methods (accessed on 8 September 2020).
- Ahadi, A.; Partoazar, A.; Abedi-Khorasgani, M.-H.; Shetab-Boushehri, S.V. Comparison of liquid-liquid extraction-thin layer chromatography with solid-phase extraction-high-performance thin layer chromatography in detection of urinary morphine. J. Biomed. Res. 2011, 25, 362–367. [Google Scholar] [CrossRef]
- Mahara, B.M.; Borossay, J.; Torkos, K. Liquid–Liquid Extraction for Sample Preparation prior to Gas Chromatography and Gas Chromatography–Mass Spectrometry Determination of Herbicide and Pesticide Compounds. Microchem. J. 1998, 58, 31–38. [Google Scholar] [CrossRef]
- Abrera, J.; Balentine, J.; Bell, K. Digital Solutions Enhance Source Control in Memphis. WaterWorld 2020, 36, 42–43. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Analytical Methods Approved for Compliance Monitoring under the Revised Total Coliform Rule. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P100WD73.txt (accessed on 14 September 2020).
- United States Environmental Protection Agency (USEPA). Pathogen Detection Systems 2017 ECTA EC TC Method- Mar 20 2017 version 2.0. Available online: https://www.regulations.gov/document?D=EPA-HQ-OW-2017-0284-0021 (accessed on 10 November 2020).
- Water Management Society (WMS) MPN (Legiolert™) Factsheet FINAL 1.0. Available online: https://www.wmsoc.org.uk/downloads/RMLG-Legiolert-Factsheet-Final-1.0.pdf (accessed on 10 November 2020).
- Hojris, B.; Christensen, S.C.; Albrechtsen, H.J.; Smith, C.; Dahlqvist, M. A novel, optical, on-line bacteria sensor for monitoring drinking water quality. Sci. Rep. 2016, 6, 23935. [Google Scholar] [CrossRef]
- Masaaki, K.; Samendra, P.S. Rapid Detection Technologies for Monitoring Microorganisms in Water. Biosens. J. 2014, 3, 1000109. [Google Scholar] [CrossRef]
- Simoes, J.; Dong, T. Continuous and Real-Time Detection of Drinking-Water Pathogens with a Low-Cost Fluorescent Optofluidic Sensor. Sensors 2018, 18, 2210. [Google Scholar] [CrossRef]
- Hossain, S.M.Z.; Mansour, N. Biosensors for on-line water quality monitoring—A review. Arab. J. Basic Appl. Sci. 2019, 26, 502–518. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, X.; Hao, Z.; Qu, K. Advances in online methods for monitoring microbial growth. Biosens. Bioelectron. 2019, 126, 433–447. [Google Scholar] [CrossRef]
- Mao, K.; Zhang, H.; Pan, Y.; Yang, Z. Biosensors for wastewater-based epidemiology for monitoring public health. Water Res. 2021, 191, 116787. [Google Scholar] [CrossRef] [PubMed]
- Hyun Chung, T.; Meshref, M.N.A.; Ranjan Dhar, B. A review and roadmap for developing microbial electrochemical cell-based biosensors for recalcitrant environmental contaminants, emphasis on aromatic compounds. Chem. Eng. J. 2021, 424, 130245. [Google Scholar] [CrossRef]
- Li, L.; Rong, S.; Wang, R.; Yu, S. Recent advances in artificial intelligence and machine learning for nonlinear relationship analysis and process control in drinking water treatment: A review. Chem. Eng. J. 2021, 405, 126673. [Google Scholar] [CrossRef]
- Song, M.; Yang, M.; Hao, J. Pathogenic Virus Detection by Optical Nanobiosensors. Cell Rep. Phys. Sci 2021, 2, 100288. [Google Scholar] [CrossRef] [PubMed]
| Source of Chemical Constituents | Examples of Sources |
|---|---|
| Naturally occurring | Rocks, soils, and the effects of the geological setting and climate; eutrophic water bodies (also influenced by sewage inputs and agricultural runoff) |
| Industrial sources and human dwellings | Mining (extractive industries) and manufacturing and processing industries, sewage (including a number of contaminants of emerging concern), solid wastes, urban runoff, fuel leakages |
| Agricultural activities | Manures, fertilizers, intensive animal practices, and pesticides |
| Water treatment or materials in contact with DW | Coagulants, disinfection byproducts (DBPs), piping materials |
| Pesticides used in water for public health | Larvicides used in the control of insect vectors of disease |
| Chemical | WHO Guideline Value, mg/L | EU DWD Limit Value, mg/L | USEPA NPDWR MCL, mg/L |
|---|---|---|---|
| Barium | 1.3 | - | 2 |
| Cadmium | 0.003 | 0.005 | 0.005 |
| Carbofuran | 0.007 | - | 0.040 |
| Carbon tetrachloride | 0.004 | - | 0.005 |
| Chlordane | 0.0002 | - | 0.002 |
| Chlorite | 0.7 | 0.25 | 1 |
| Chromium (total) * | 0.05 | 0.05 | 0.1 |
| 2,4-Dichloro- phenoxy-acetic acid (2,4-D) | 0.03 | - | 0.07 |
| Endrin | 0.0006 | - | 0.002 |
| Ethylbenzene | 0.3 | - | 0.7 |
| Fluoride | 1.5 | 1.5 | 4 |
| Lead | 0.01 | 0.005 | 0.015 |
| Methoxychlor | 0.02 | - | 0.04 |
| Selenium | 0.04 | 0.02 | 0.05 |
| Simazine | 0.002 | - | 0.004 |
| Styrene | 0.02 | - | 0.1 |
| Vinyl chloride | 0.0003 | 0.0005 | 0.002 |
| Xylenes | 0.5 | - | 10 |
| Category | Description |
|---|---|
| Optics (non-imaging) | Measuring bacterial growth by detecting changes in optical signals using photometers |
| Optics (imaging individual cell) | Measuring physiological, morphological, metabolic, or structural features of bacteria with cameras integrated with microscopy |
| Optics (imaging population) | Measuring population of bacteria in liquid with imaging |
| Electrochemistry (sensor) | Measuring bacterial growth by detecting changes in electro-chemical features of the electrodes and analytes |
| Electrochemistry (biosensor) | Measuring bacterial growth by monitoring changes in electrochemical features of cells or metabolites using bio-elements immobilized on electrodes |
| Electrochemistry (contactless sensor) | Measuring E. coli growth by detecting changes in conductivity with C4D or other contactless sensors |
| Microcalorimetry | Monitoring heat generation by growing bacterial cells |
| Resonant mass | Quantifying cell number by measuring changes in the mass of individual cells using a small channel of cantilever |
| Gene analysis | Measuring E. coli growth by detecting genes via augmentation with PCR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsaridou, C.; Karabelas, A.J. Drinking Water Standards and Their Implementation—A Critical Assessment. Water 2021, 13, 2918. https://doi.org/10.3390/w13202918
Tsaridou C, Karabelas AJ. Drinking Water Standards and Their Implementation—A Critical Assessment. Water. 2021; 13(20):2918. https://doi.org/10.3390/w13202918
Chicago/Turabian StyleTsaridou, Charikleia, and Anastasios J. Karabelas. 2021. "Drinking Water Standards and Their Implementation—A Critical Assessment" Water 13, no. 20: 2918. https://doi.org/10.3390/w13202918
APA StyleTsaridou, C., & Karabelas, A. J. (2021). Drinking Water Standards and Their Implementation—A Critical Assessment. Water, 13(20), 2918. https://doi.org/10.3390/w13202918







