Nitrate Removal from Actual Wastewater by Coupling Sulfur-Based Autotrophic and Heterotrophic Denitrification under Different Influent Concentrations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Wastewater
2.2. Experimental Apparatus and Operation
2.3. Measurements of Physicochemical Properties
2.4. Analysis of Microbial Community Dynamic
3. Result and Discussion
3.1. Wastewater Treatment Performance
3.1.1. Start-Up of the Coupling System under Mainstream Conditions
3.1.2. Nitrogen Removal Performances under Different Trophic Conditions
3.1.3. Nitrogen Removal Efficiencies under Different Pollution Loads
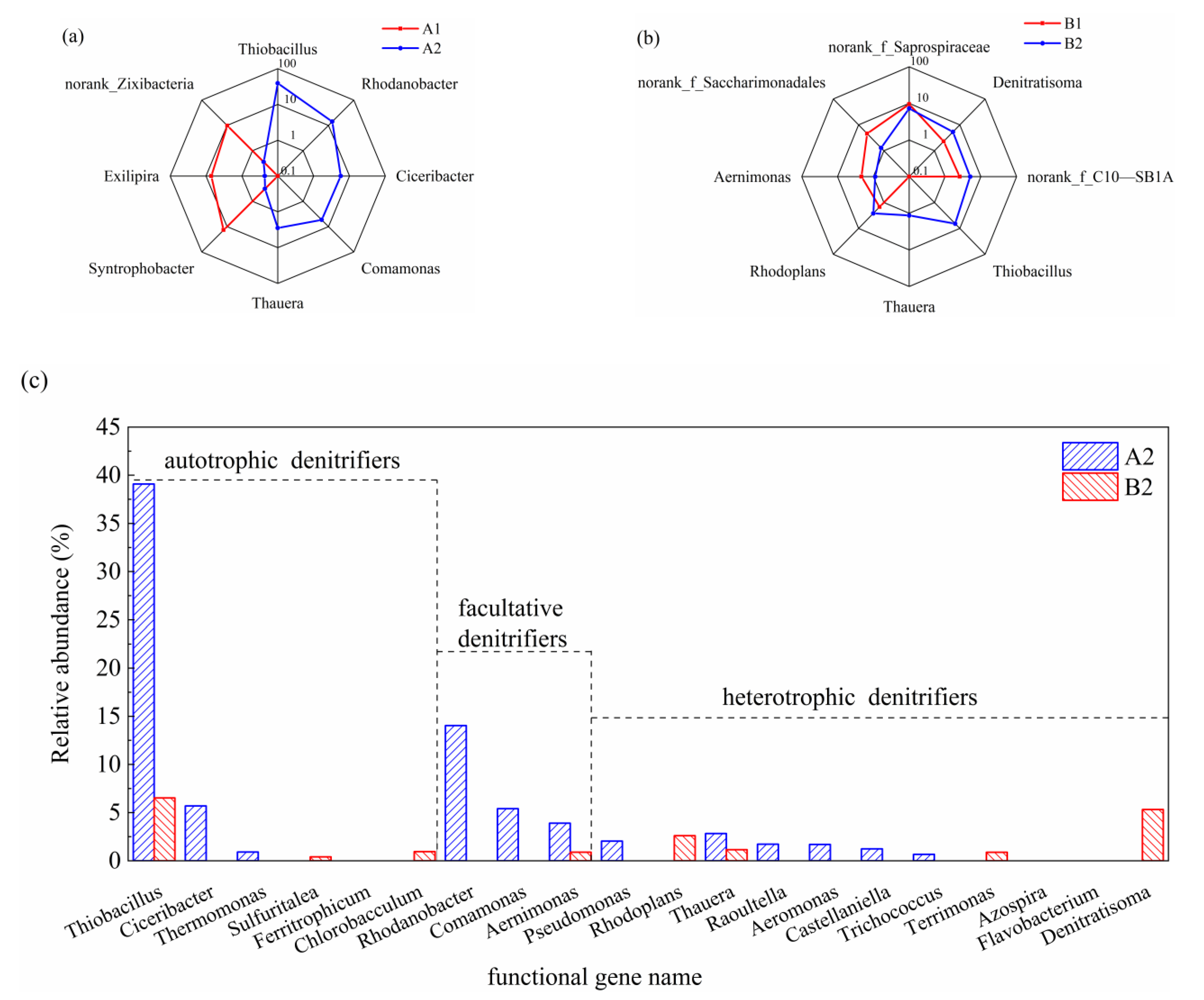
3.2. Microbial Community Analysis
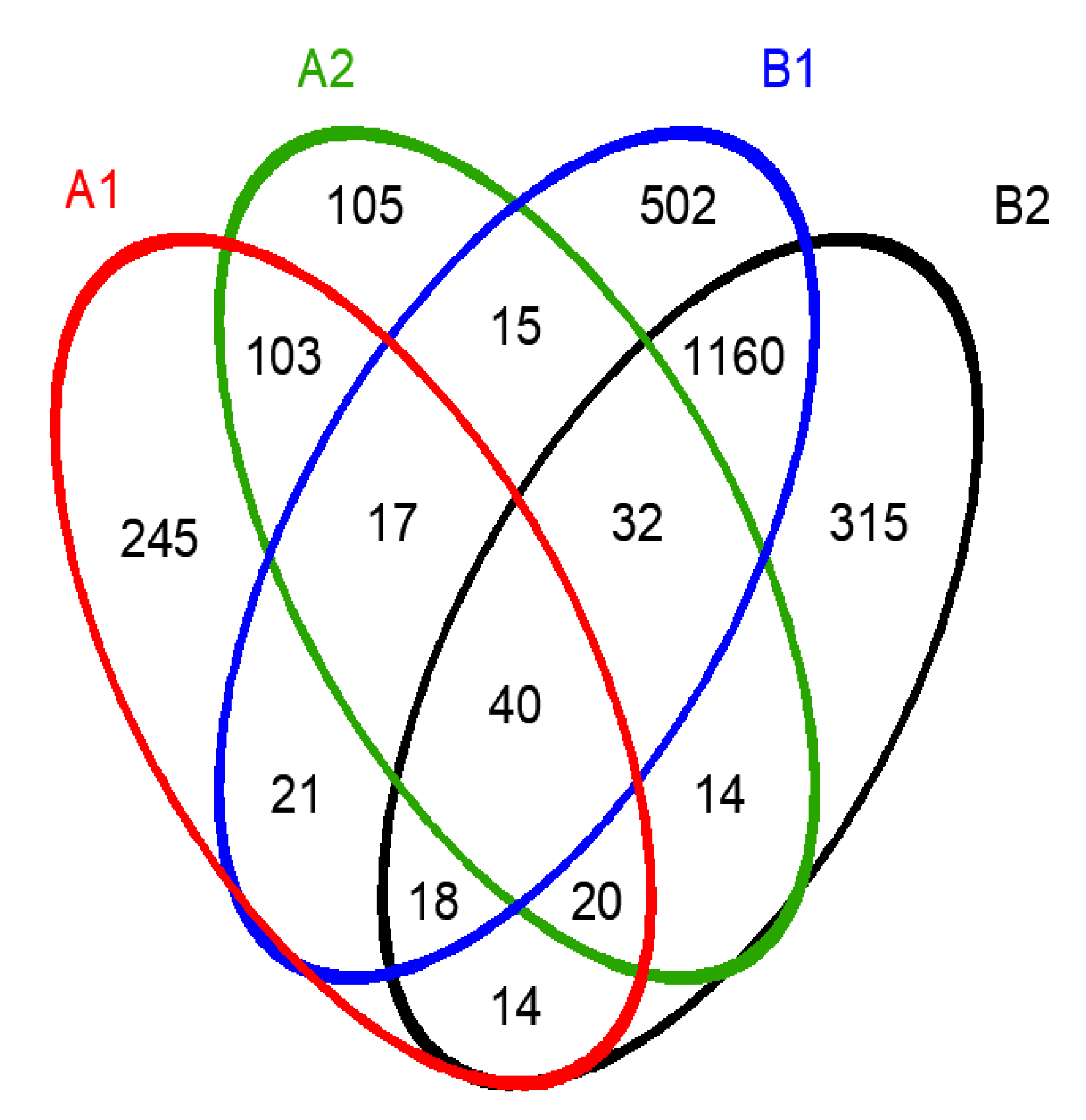
3.2.1. The Diversity and Richness of the Microbial Community
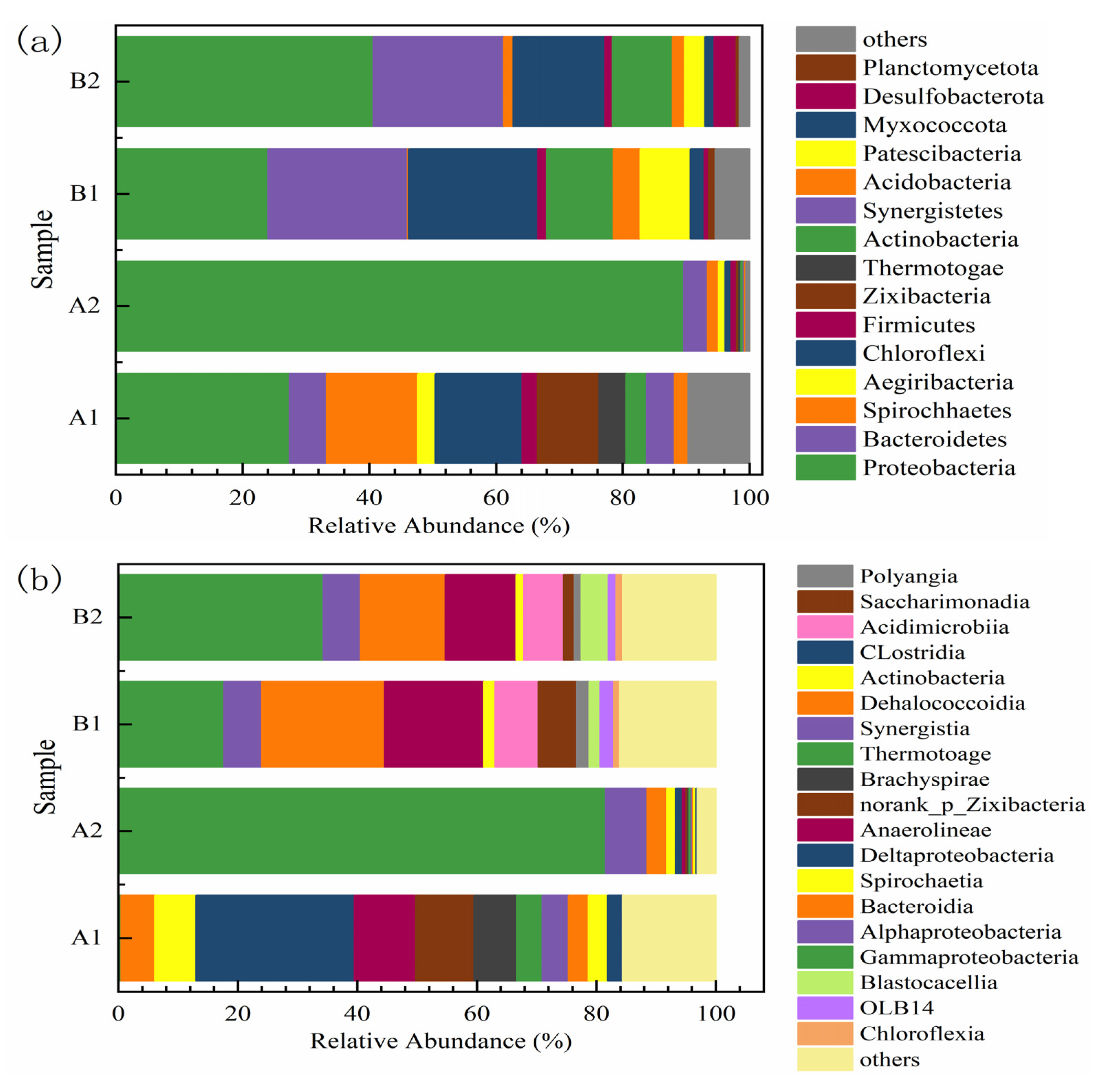
3.2.2. Changes in Microbial Community Structure
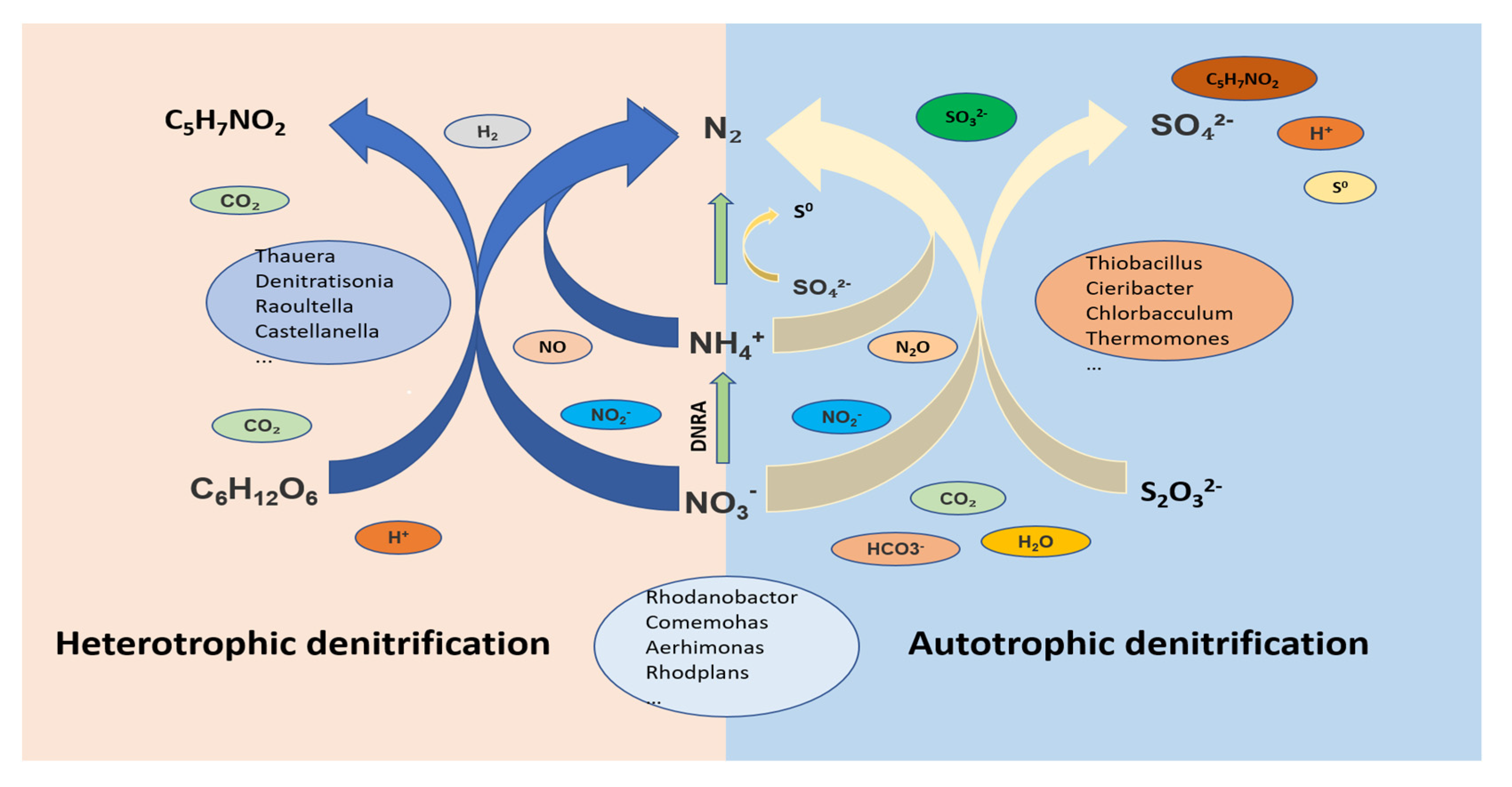
3.3. Substance Conversion Paths and Mechanisms Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, S.; Su, J.; Zheng, Z.; Yang, S. Denitrification strategies of strain ysf15 in response to carbon scarcity: Based on organic nitrogen, soluble microbial products and extracellular polymeric substances. Bioresour Technol. 2020, 314, 123733. [Google Scholar] [CrossRef]
- Rajanayaka, C.; Weir, J.; Barkle, G. Assessing changes in nitrogen contamination in groundwater using water aging: Waikato River, New Zealand. J. Contam. Hydrol. 2020, 234, 103686. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, R.; Dong, L.; Li, W.; Li, M.; Wu, H. Simultaneous removal of nitrogen and dimethyl phthalate from low-carbon wastewaters by using intermittently-aerated constructed wetlands. J. Hazard. Mater. 2021, 404, 124130. [Google Scholar] [CrossRef]
- Wang, H.; Chen, N.; Feng, C.; Deng, Y. Insights into heterotrophic denitrification diversity in wastewater treatment systems: Progress and future prospects based on different carbon sources. Sci. Total Environment. 2021, 780, 146521. [Google Scholar] [CrossRef] [PubMed]
- Ucar, D.; Capua, F.D.; Yücel, A. Effect of nitrogen loading on denitrification, denitritation and filtration performances of membrane bioreactors fed with biogenic and chemical elemental sulfur. Chem. Eng. J. 2021, 419, 129514. [Google Scholar] [CrossRef]
- Qiu, T.; Liu, L.L.; Gao, M.; Zhang, L.; Tursun, H.; Wang, X. Effects of Solid-Phase Denitrification on the Nitrate Removal and Bacterial Community Structure in Recirculating Aquaculture System. Biodegradation 2016, 27, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Yu, H.Q. Denitrification with non-organic electron donor for treating low C/N ratio wastewaters. Bioresource Technol. 2020, 299, 122686. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Nan, C.H.; Tong, S.; Liu, Y.; Feng, C. Sulfur autotrophic denitrification(SAD)driven by homogeneous composite particles containing CaCO3-type kitchen waste for groundwater remediation. Chemosphere 2018, 212, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liu, X.; Dong, X.; Wang, Z. Sulfur-based autotrophic denitrification from the micro-polluted water. J. Environ. Sci. 2016, 44, 180–188. [Google Scholar] [CrossRef]
- Cui, Y.X.; Biswal, B.K.; Guo, G.; Deng., Y.F.; Wu, D. Biological nitrogen removal from wastewater using sulphur-driven autotrophic denitrification. Appl. Microbiol. Biotechnol. 2019, 103, 6023–6039. [Google Scholar] [CrossRef]
- Liu, H.; Zeng, W.; Fan, Z.; Li, J. Effect of iron on enhanced nitrogen removal from wastewater by sulfur autotrophic denitrification coupled to heterotrophic denitrification under different substrate ratios. Chem. Eng. J. 2021, 421, 129828. [Google Scholar] [CrossRef]
- Wang, S.S.; Yi, H.C.; Zhang, H. Sulfur autotrophic denitrification filter and heterotrophic denitrification filter: Comparison on denitrification performance, hydrodynamic characteristics and operating cost. Environ. Res. 2021, 197, 111029. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Feng, C.; Hu, W. Woodchip-sulfur based heterotrophic and autotrophic denitrification(WSHAD) process for nitrate contaminated water remediation. Water Res. 2016, 89, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Cheng, L.; Wua, B.C. Short term performance and microbial community of a sulfide-based denitrification and Anammox coupling system at different N/S ratios. Bioresour. Technol. 2019, 294, 122130. [Google Scholar]
- Du, S.; Ya, T.; Zhang, M.; Zhu, M.; Li, N.; Liu, S.; Wang, X. Distinct microbial communities and their networks in an anammox coupled with sulfur autotrophic/ mixotrophic denitrification system. Environ. Pollut. 2020, 262, 114190. [Google Scholar] [CrossRef]
- Erkan, S.; Adem, K. Heterotrophic and elemental-sulfur-based autotrophic denitrification processes for simultaneous nitrate and Cr(VI) reduction. Water Res. 2014, 50, 278–286. [Google Scholar]
- Oberoi, A.S.; Huang, H.; Khanal, S.K.; Sun, L.; Lu, H. Electron distribution in sulfur-driven autotrophic denitrification under different electron donor and acceptor feeding schemes. Chem. Eng. J. 2021, 404, 126486. [Google Scholar] [CrossRef]
- Qian, J.; Zhang, M.; Pei, X.; Zhang, Z.; Niu, J.; Liu, Y. A novel integrated thiosulfate-driven denitritation (TDD) and anaerobic ammonia oxidation (anammox) process for biological nitrogen removal. Biochem. Eng. J. 2018, 139, 68–73. [Google Scholar] [CrossRef]
- Qian, J.; Wei, L.; Wu, Y.; Wang, Q.; Fu, X.; Zhang, X.; Pei, X. A comparative study on denitrifying sludge granulation with different electron donors: Sulfide, thiosulfate and organics. Chemosphere 2017, 186, 322–330. [Google Scholar] [CrossRef]
- Yuan, Y.; Li, X.; Li, B. Autotrophic nitrogen removal characteristics of PN-anammox process enhanced by sulfur autotrophic denitrification under mainstream conditions. Bioresour. Technol. 2020, 316, 123926. [Google Scholar] [CrossRef]
- WE Federation; APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Han, F.; Zhang, M.; Shang, H.; Liu, Z.; Zhou, W. Microbial community succession, species interactions and metabolic pathways of sulfur-based autotrophic denitrification system in organic-limited nitrate wastewater. Bioresour. Technol. 2020, 315, 123826. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, W.; Wan, D.; Qu, J. Study of a combined heterotrophic and sulfur autotrophic denitrification technology for removal of nitrate in water. J. Hazard. Mater. 2009, 169, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, S.H. Enhancement of nitrate removal in constructed wetlands utilizing a combined autotrophic and heterotrophic denitrification technology for treating hydroponic wastewater containing high nitrate and low organic carbon concentrations. Agric. Water Manag. 2015, 162, 1–14. [Google Scholar] [CrossRef]
- Park, S.; Seon, J.; Byun, I.; Cho, S.; Park, T.; Lee, T. Comparison of nitrogen removal and microbial distribution in wastewater treatment process under different electron donor conditions. Bioresour. Technol. 2010, 101, 2988–2995. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Feng, C.; Xi, B.; Chen, N.; Jiang, Y.; Zhao, Y.; Li, M.; Dang, Q.; Zhao, B. Nitrate removal efficiency of a mixotrophic denitrification wall for nitrate-polluted groundwater in situ remediation. Ecol. Eng. 2017, 106, 523–531. [Google Scholar] [CrossRef]
- Dan, C.; Kai, Y.; Li, W.; Hong, Y.W. Microbial community and metabolism activity in a bioelectrochemical denitrification system under long-term presence of p-nitrophenol, Bioresour. Technol. 2016, 218, 189–195. [Google Scholar]
- 30Sorokin, D.Y.; Kuenen, J.; Jetten, M. Denitrification at extremely alkaline condi-tions in obligately autotrophic alkaliphilic sulfur-oxidizing bacterium Thioalkalivibrio denitrificans. Arch. Microbiol. 2001, 175, 101. [Google Scholar]
- Zhu, T.-T.; Cheng, H.-Y.; Yang, L.-H.; Su, S.-G.; Wang, H.-C.; Wang, S.-S.; Wang, A.-J. Coupled sulfur and iron (II) carbonate-driven autotrophic denitrification for significantly enhanced nitrate removal. Environ. Sci. Technol. 2018, 53, 1545–1554. [Google Scholar] [CrossRef]
- Yang, Y.; Gerrity, S.; Collins, G.; Chen, T.; Li, R.; Xie, S.; Zhan, X. Enrichment and characterization of autotrophic Thiobacillus denitrifiers from anaerobic sludge for nitrate removal. Process Biochem. 2018, 68, 165–170. [Google Scholar] [CrossRef]
- Zhou, W.; Li, Y.; Liu, X.; He, S.; Huang, J.C. Comparison of microbial communities in different sulfur-based autotrophic denitrification reactors. Appl. Microbiol. Biotechnol. 2017, 101, 447–453. [Google Scholar] [CrossRef]
- 35Deng, S.; Peng, S.; Xie, B.; Yang, X.; Sun, S.; Yao, H.; Li, D. Influence characteristics and mechanism of organic carbon on denitrification, N2O emission and NO2−accumulation in the iron [Fe(0)]-oxidizing supported autotrophic denitrification process. Chem. Eng. J. 2020, 393, 124736. [Google Scholar]
- Huang, Z.; Wei, Z.; Xiao, X.; Tang, M.; Li, B.; Zhang, X. Nitrification/denitrification shaped the mercury- oxidizing microbial community for simultaneous Hg0 and NO removal. Bioresour. Technol. 2019, 274, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ji, M.; Qiu, Z.; Liu, H.; Wang, J.; Li, J. Microbial population dynamics during sludge granulation in an anaerobic–aerobic biological phosphorus removal system. Bioresour. Technol. 2011, 102, 2474–2480. [Google Scholar] [CrossRef]
- Sun, S.; Liu, J.; Zhang, M. Thiosulfate-driven autotrophic and mixotrophic denitrification processes for secondary effluent treatment: Reducing sulfate production and nitrous oxide Emission, Bioresour. Technol. 2020, 300, 122651. [Google Scholar] [CrossRef]
- Zhang, R.C.; Xu, X.J.; Chen, C.; Xing, D.F.; Shao, B.; Liu, W.Z. Interactions of functional bacteria and their contributions to the performance in integrated autotrophic and heterotrophic denitrification. Water Res. 2018, 143, 355–366. [Google Scholar] [CrossRef]
- Llorens-Marès, T.; Yooseph, S.; Goll, J.; Hoffman, J.; Vila-Costa, M.; Borrego, C.M.; Casamayor, E.O. Connecting biodiversity and potential functional role in modern euxinic environments by microbial metagenomics. ISME J. 2015, 9, 1648–1661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, F.; Deng, Q.; Li, X.; Tang, M.; Ma, X.; Cao, X.; Song, C. Organic carbon quantity and quality jointly riggered the switch between dissimilatory nitrate reduction to ammonium (DNRA) and denitrification in biofilters. Chemosphere 2021, 280, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Moraes, B.S.; Souza, T.S.O.; Foresti, E. Effect of sulfide concentration on autotrophic denitrification from nitrate and nitrite in vertical fixed-bed reactor. Process Biochem. 2012, 47, 1395–1401. [Google Scholar] [CrossRef]
- Aalto, S.L.; Eero, A.; Tom, J.; Susanna, H. Autochthonous organic matter promotes DNRA and suppresses N2O production in sediments of the coastal Baltic Sea. Estuar. Coast. Shelf Sci. Vol. 2021, 255, 107369. [Google Scholar] [CrossRef]
- Liu, C.; Li, W.; Li, X.; Zhao, D.; Ma, B.; Wang, Y.; Lee, D.J. Nitrite accumulation in continuous-flow partial autotrophic denitrification reactor using sulfide as electron donor. Bioresour. Technol. 2017, 243, 1237–1240. [Google Scholar] [CrossRef]
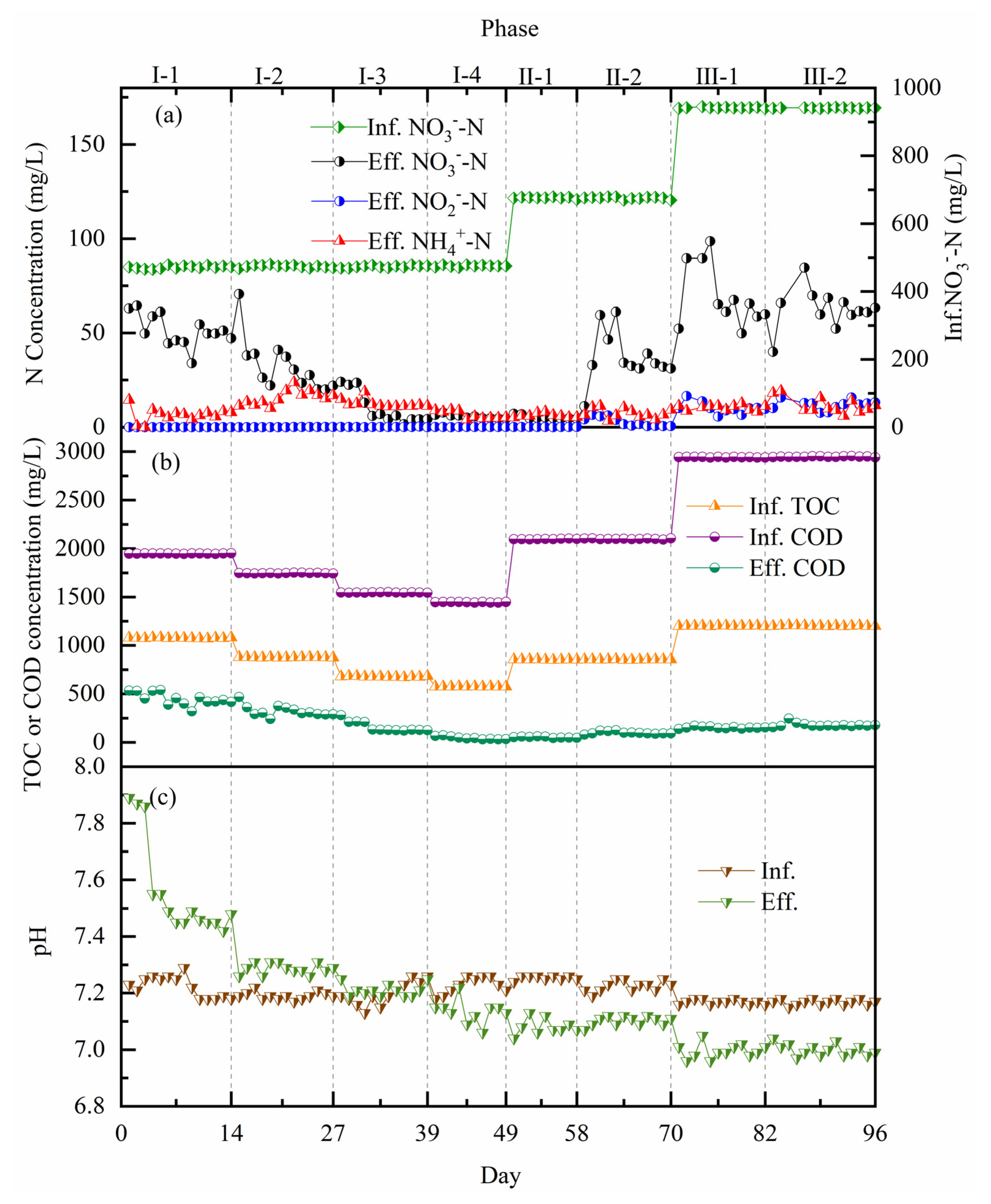
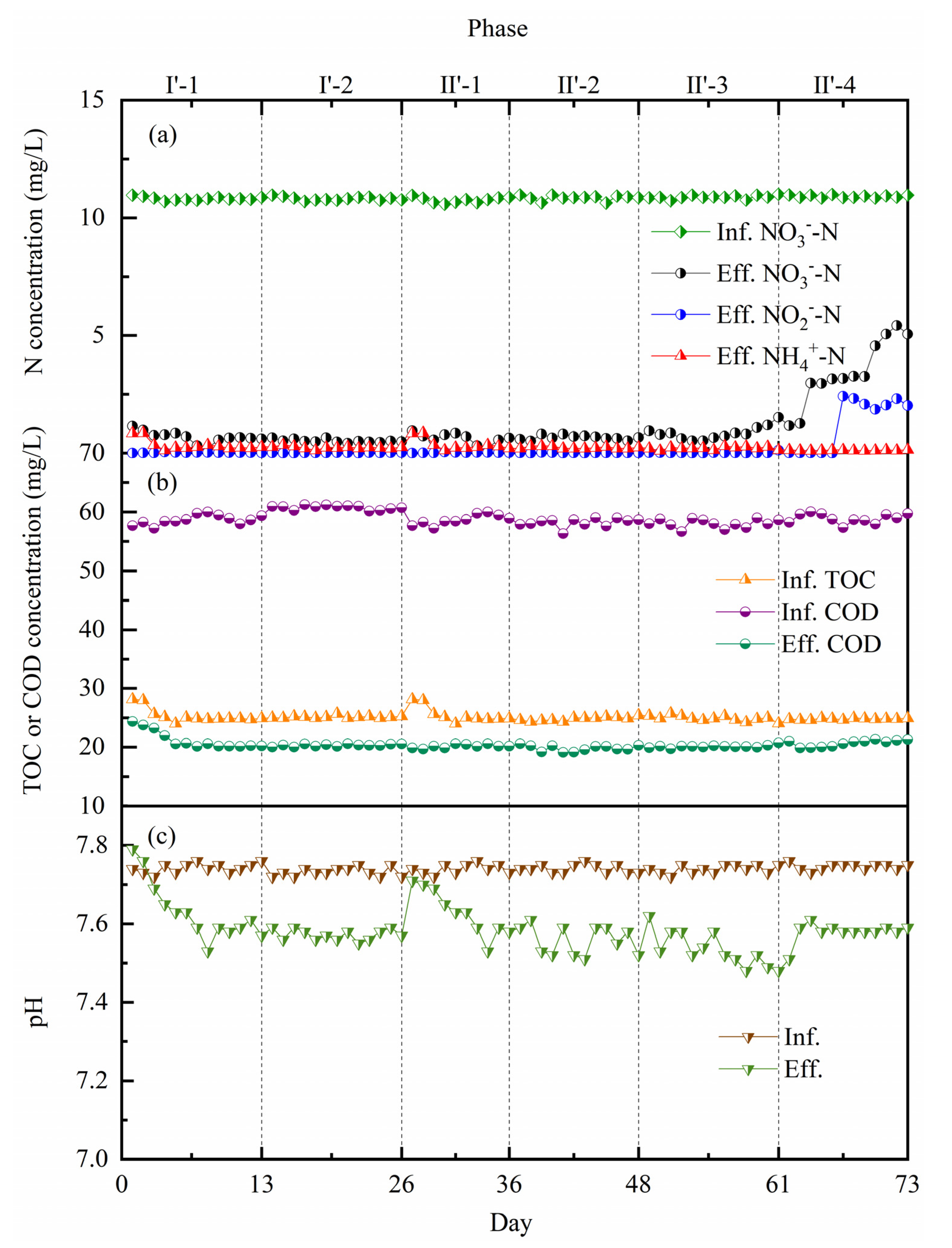
| Wastewater | COD (mg/L) | -N (mg/L) | -N (mg/L) | -N (mg/L) | TN (mg/L) | pH | (mg/L) |
|---|---|---|---|---|---|---|---|
| WH | 153.60 | 941.35 | 2.62 | 1.85 | 951.58 | 7.16 | 125.60 |
| WL | 24.58 | 10.95 | 0 | 0.45 | 11.59 | 7.73 | 99.80 |
| Reactor | Phase | Days (d) | -N Influent (mg/L) | HRT (h) | C/N/S |
|---|---|---|---|---|---|
| A | I-1 | 14 | 470 | 12 | 2.4/1/1.9 |
| I-2 | 13 | 470 | 12 | 1.9/1/1.9 | |
| I-3 | 12 | 470 | 12 | 1.5/1/1.9 | |
| I-4 | 10 | 470 | 12 | 1.3/1/1.9 | |
| II-1 | 9 | 670 | 12 | 1.3/1/1.9 | |
| II-2 | 12 | 670 | 9.6 | 1.3/1/1.9 | |
| III-1 | 12 | 940 | 8 | 1.3/1/1.9 | |
| III-2 | 14 | 940 | 6 | 1.3/1/1.9 | |
| B | I’-1 | 13 | 10.95 | 6 | 1.3/1/1.9 |
| I’-2 | 13 | 10.95 | 6 | 1.3/1/2.4 | |
| II’-1 | 10 | 10.95 | 6 | 1.3/1/1.9 | |
| II’-2 | 12 | 10.95 | 4 | 1.3/1/1.9 | |
| II’-3 | 13 | 10.95 | 2 | 1.3/1/1.9 | |
| II’-4 | 12 | 10.95 | 1 | 1.3/1/1.9 |
| Sample A | OTU | Shannon | Simpson | Ace | Chao | Coverage (%) |
|---|---|---|---|---|---|---|
| A1 | 478 | 4.39 | 0.03 | 532.35 | 529.89 | 99.87 |
| A2 | 346 | 2.94 | 0.16 | 412.51 | 411.02 | 99.71 |
| B1 | 1805 | 6.02 | 0.006 | 1886.40 | 1866.98 | 99.54 |
| B2 | 1613 | 5.75 | 0.009 | 1823.32 | 1862.66 | 99.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, F.; Wang, S.; Zhang, X.; Qian, F.; Wang, Y.; Yin, Y. Nitrate Removal from Actual Wastewater by Coupling Sulfur-Based Autotrophic and Heterotrophic Denitrification under Different Influent Concentrations. Water 2021, 13, 2913. https://doi.org/10.3390/w13202913
Liu F, Wang S, Zhang X, Qian F, Wang Y, Yin Y. Nitrate Removal from Actual Wastewater by Coupling Sulfur-Based Autotrophic and Heterotrophic Denitrification under Different Influent Concentrations. Water. 2021; 13(20):2913. https://doi.org/10.3390/w13202913
Chicago/Turabian StyleLiu, Feng, Suqin Wang, Xuezhi Zhang, Feiyue Qian, Yaobing Wang, and Yao Yin. 2021. "Nitrate Removal from Actual Wastewater by Coupling Sulfur-Based Autotrophic and Heterotrophic Denitrification under Different Influent Concentrations" Water 13, no. 20: 2913. https://doi.org/10.3390/w13202913
APA StyleLiu, F., Wang, S., Zhang, X., Qian, F., Wang, Y., & Yin, Y. (2021). Nitrate Removal from Actual Wastewater by Coupling Sulfur-Based Autotrophic and Heterotrophic Denitrification under Different Influent Concentrations. Water, 13(20), 2913. https://doi.org/10.3390/w13202913





