1. Introduction
Drinking water quality has a significant impact on consumers’ health. Although water treatments have increased the safety of water, the drinking water quality remains susceptible to microbial and chemical contaminants during transport, storage, and handling, as well as in dispensing devices such as bottled water dispensers [
1]. In recent years, there has been a worldwide increase of water consumption derived from different sources besides tap or bottled water for drinking use [
2]. Considering the large request for water, one of the alternative sources is drinking microfiltered water from dispensers (or coolers), which has been mainly introduced in office buildings, companies, or commercial sites [
3]. In Italy, in the last 12 months, during the pandemic period, 82.7% of the Italian population drank tap water (treated and untreated), with a growth rate of over 5%, compared to 2020. In 32.6% of Italian families, there is at least one water filtration system, which allows treating tap water and obtaining water with better organoleptic characteristics [
4]. In this context, the use of these new devices that supply drinking water at point of use become more widespread, especially since they are able to offer more advantages, such us avoiding transport, reservoirs, or plastic containers, in addition to reducing the impact of plastic pollution.
It is known that the quality of drinking water is an important public health issue, and the Microfiltered Water Dispensers (MWDs) are often adopted as devices able to improve water organoleptic characteristics and are easy to use and to maintain [
5]. MWDs are devices directly connected to a municipal water supply and they often may include a CO
2 system for sparkling water or cooling systems for cold water. An activated carbon filtration system incorporated in the device improves the treatment of drinking water; sometimes an Ag
+ coating may be present. The activated carbon filter is used for a bacteriostatic effect and to reduce tastes and odors or remove organic and inorganic contaminants [
6,
7]. Furthermore, the use of activated carbon filters could amplify the microbiological contamination at point of use, especially in the presence of water stagnation [
8,
9]. The filtration system is sometimes associated with an UV lamp, installed inside MWDs or immediately before the water is dispensed, at the output nozzle. The UV lamp is able to remove airborne or surface microorganisms [
10,
11].
Considering the MWDs’ pipelines and their location in several sites, other than the improper use by consumers, opportunistic and pathogenic bacteria (
Pseudomonas,
Legionella, and
Mycobacterium species) that grow in water-flowing equipment can form an adherent biofilm, in which the bacteria will survive, becoming more resistant to antimicrobial agents and increasing the water contamination [
12,
13,
14,
15].
For these reasons, an adequate maintenance and disinfection protocol should be implemented on MWDs. Several studies have discussed the use of oxidants’ agents, such as peracetic acid and hydrogen peroxide, to reduce the microbiological contamination and to assure the quality of resulting water [
9,
16]. The disinfection products need to be certified as food-grade products to preserve the health of consumers; at the same time, it is recommended to avoid the continuous use of aggressive disinfectants, such as chlorine disinfectants, that could damage the devices, often characterized by plastic material causing a decay of the material, which can dissolve into the water flow and reach the consumers. Another aspect regards the continuous disinfection treatment that can lead to a bacterial species’ selection with consequent increase in bacterial resistance and inefficient disinfection procedures [
17,
18].
However, concerns have been raised about the quality of the water dispensed from MWDs due to the non-compliant maintenance procedures that could lead to a potential risk of uncontrolled bacterial contamination and the consequence of waterborne infectious diseases, especially as regards the sensitive and immunocompromised population [
5]. Multiple studies show that the contamination of MWDs is caused by missed or improper cleaning. Yamaguchi et al. in 2007 reported a major contamination of mineral water dispensers, other than tap water, in terms of yeasts, fungi, and total coliforms present [
19]. In an another study, from 2014, the heterotrophic bacterial load was shown above the acceptable limits in drinking water, significantly higher in water dispenser samples than tap water samples [
20]. Moniruzzaman et al. 2011 carried out a study about the microbiological contamination (heterotrophic bacteria,
Escherichia coli,
Pseudomonas sp., and
Salmonella sp.) in water samples dispensed in road-side restaurants compared to water samples from the bottle before dispensing [
21]. In Italy, Liguori et al. 2010 and Sacchetti et al. 2014 conducted studies to compare the microbial quality of water dispensers and municipal tap water [
2,
9]. Research conducted in Ajman (UAE) to investigate the microbial quality of drinking water distributed through water dispenser machines located in schools and universities displayed more than 50% of water samples positive for
Pseudomonas aeruginosa (
P. aeruginosa).
P. aeruginosa is considered an opportunistic pathogen, and its occurrence in drinking water can pose a serious health risk to children and immunocompromised individuals [
22].
Such findings indicate that the companies and customers should be informed about the proper use and correct maintenance of MWDs, and this helps to keep them free from microbiological contamination [
23].
In order to avoid or to reduce the risk of acquiring water-related infections, World Health Organization (WHO) Guidelines for Drinking-Water Quality and European Directive and subsequent amendments on the quality of water intended for human consumption have been introduced in some countries [
24,
25,
26,
27,
28]. In Italy, established criteria exist for human consumption of water, including water coming from MWDs [
11,
29]. According to the European Community Directive Guidelines, the microbiological quality of water is required to be tested for typical indicator bacteria, such as heterotrophic plate counts (HPCs) at 36 °C and 22 °C, and to be free from any pathogenic bacteria, such as
Enterococcus spp.,
P. aeruginosa,
Escherichia coli (E. coli),
Staphylococcus aureus (
S. aureus), and
Clostridium perfringens (C. perfringens), as well as chemical contaminants.
Although the impact of water bacterial contamination on human health is widely recognized, few studies have been performed on MWDs’ contamination and the role of WSP introduction in their management, as far as monitoring, hygienic quality level, and safety for consumers.
Based on our previous work [
30], where the water supplied by MWDs showed a high contamination by indicator as well as pathogenic bacteria, this research presents the results of a study conducted for 5 years, from 2017 to 2021, to monitor the water quality supplied by MWDs, installed in an Italian metalworking industrial site. The aim of the study was to assess, in a long-term period, the effectiveness of a specific Water Safety Plan (WSP), based on maintenance and sanitization procedures, developed to improve the quality and safety of drinking water supplied by MWDs. Moreover, the monitoring was continued during the lockdown period that occurred for the global SARS-CoV-2 pandemic in 2020, to test the efficacy of measures implemented also in an emergency period.
2. Materials and Methods
2.1. Microfiltered Water Dispensers’ Characteristics
The MWDs involved in the study are considered “point of use”, meaning that they are connected to a main potable water network (
Figure 1).
A dedicated pipeline containing a removable filter with a pore size of 1 mm was used to remove particulate matter that could be delivered from the municipal water. The main water flowed through an internal activated carbon filter with a pore size of 0.5 µm, which reduced tastes, odors, and various organic and inorganic substances (e.g., clays, chlorine, etc.), as well as potentially disinfectant chemicals used in municipal water. The MWDs showed an approximate capacity of 20–28 L/h and were provided by a single, low-pressure UV lamp at the output water supply point. The lamp consisted of a small, glass tube encircled with a power of 1.5 W. The MWDs provided water at room temperature, sparkling water (by carbon dioxide gas cylinder), and hot water, via a circular supply point (nozzle). The water was supplied by a single nozzle located at the base of the dispenser, which had a capacity of 30 cL. The technical characteristics of MWDs were provided in the manufacturer’s manuals and conformed to the Italian directives [
11].
The MWDs involved in this study were installed in several industrial sites (e.g., coffee break areas, offices, recreational points, etc.) and the intended use was especially to provide drinking water in common or private spaces, not catering use, as admitted. Moreover, in Italy, it is mandatory to notify the presence and use of MWDs to the Public Health Authorities [
31] only when these devices are in use in hotels, canteens, restaurants, and cafés; therefore, their presence in other facilities often is underestimated, with a lack of authority control.
2.2. Maintenance and Sanitation Procedures Developed
An established program of routine and extraordinary maintenance procedures of MWDs is considered a key factor to prevent bacterial growth and the colonization of MWDs’ pipelines or the point of use, to guarantee water quality and safety for consumers.
The results obtained in our previous study [
30], where two different typologies of MWDs (Type A and Type B) were compared to test their performance on drinking water treatment, demonstrated how the MWDs called Type B showed a higher microbial contamination for all parameters tested. Considering the number of these devices located in the company (
n = 57), a specific WSP was developed according to the Company’s environmental heath safety office.
Following the manufacturer’s instructions, the MWDs were subjected to a follow-up study from 2017 to 2021, to evaluate the WSP effectiveness, through water quality supply monitoring.
The WSP consisted of various maintenance and sanitation procedures, as well as the water quality assessment.
In the following bullet points are reported the activities undertaken:
Daily external cleaning of MWDs’ surfaces;
Daily cleaning of the external water supply (point of connection to municipal water pipelines);
Monthly checking and cleaning of mechanical filters on the water supply;
Monthly functional checking and cleaning of the cooling condenser;
Weekly emptying of the water collection tray and removal of any residues;
Replacement of the UV lamp, at the supply point, every 6000 h (6–8 months) of working;
Replacement of the internal activated carbon filter, every 6 months;
Replacement of the carbon dioxide gas cylinder (non-rechargeable), every 120 L of supplied water;
Daily cleaning of the water dispensing nozzles;
Flushing the input and output water supplies, for 5 minutes, after the weekends or the holidays;
Descaling procedure by recirculation of an acid solution (1–3% v/v for 30 min), certified as food-grade product, in respect to enforced legislations, every 6 months;
Disinfection procedure on the internal components by recirculation in the MWDs’ pipelines of a hydrogen peroxide solution (3% v/v for 30 min), every 6 months, every time the water filter is changed, after a long inoperative period, or/and after non-compliance of microbiological results;
MWDs’ water sampling performed every 6 months, after non-compliance results, or after disinfection procedures were undertaken.
Given that the chemical products used for this procedure contained corrosive acid and alkaline substances, it was important to provide the staff involved with protective equipment (e.g., gloves, visor, etc.) in addition to considering the disinfection concentration, contact time, and the volume of water supplied.
A qualified and professional staff were identified and informed regarding the chemical and microbiological risk, in order to safely perform MWDs’ functional and maintenance protocols. Moreover, each of above bullet points were recorded on a maintenance register, validated by the Maintenance office.
2.3. Water Samples’ Collection and Microbiological Parameters
The study was conducted from 2017 to 2021, as part of a surveillance program based on the previous WSP started in 2017 and still in place in the Italian company. This made it possible to monitor, over time, the microbiological contamination of the MWDs and to correlate it to the temperatures detected during the sampling, in relation to the adopted protocols based on ordinary and extraordinary maintenance procedures.
Following the WSP established in the industrial site in 2017, according to the WHO and the Italian regulation of drinking water for human consumption, the MWDs were monitored to analyze the drinking water quality every 6 months [
25,
28].
For each MWD, 500 mL of water at room temperature were collected from the output water supply point. The water sample was collected in the same way the water was delivered to the consumer, without flushing, and collected in sterile polytetrafluoroethylene (PTFE) bottles, according to EN ISO 19458 [
32]. Microbiological analyses included the detection of typical indicator bacteria, HPCs at 36 °C and 22 °C, and pathogenic bacteria,
Enterococcus spp.,
P. aeruginosa,
E. coli,
S. aureus, and
C. perfringens, in accordance with the Italian regulations for drinking water and reference standard methods for each microbiological parameter [
25].
The HPCs’ analysis was performed according to UNI EN ISO 6222:2001 [
33], using the standard plate method on tryptic glucose yeast agar (Plate Count Agar, PCA; Biolife, Milan, Italy).
Analysis of
Enterococcus spp. was carried out using the standard membrane filter technique according to ISO 7899-2:2000 [
34], using Slanetz Bartley Medium (Enterococcus Agar) (Thermo Fisher Scientific, Diagnostics, Ltd., Basingstoke, UK).
P. aeruginosa analysis was carried out according to UNI EN ISO 16266:2008 standard membrane filter technique [
35], using
Pseudomonas C-N Selective Agar (Cetrimide Agar) (Thermo Fisher Scientific, Diagnostics, Ltd., Basingstoke, UK).
The
E. coli bacteria were analyzed using the standard membrane filter technique according to UNI EN ISO 9308-1:2017 [
36], using Chromogenic Coliform Agar (ISO) (Thermo Fisher Scientific, Diagnostics, Ltd., Basingstoke, UK).
S. aureus contamination was detected using the standard membrane filter technique according to ISO 16140-2:2016 [
37], using Brilliance™ Staph 24 Agar (Thermo Fisher Scientific, Diagnostics, Ltd., Basingstoke, UK).
Finally, analysis of
C. perfringens was conducted according to UNI EN ISO 14189:2016 [
38], using m-CP Selective Agar (Thermo Fisher Scientific, Diagnostics, Ltd., Basingstoke, UK).
Suspected colonies grown on the different selective media were sub-cultured on Tryptic Soy Agar (TSA) (Biolife, Milan, Italy) and identified using a Crystal Enteric/Non-Fermenter ID kit (Crystal E/NF) or BBL Crystal Gram Positive ID kit (Crystal GP), both produced by Becton Dickinson (Cockeysville, MD, USA), according to the manufacturer’s instructions [
39].
The reference values for drinking water were the following: for HPCs at 36 °C and 22 °C, less than 20 and 100 colony forming units (cfu) per 1 mL of seeded water, respectively. The samples under the regulation limits were considered negative [
25].
Enterococcus spp.,
P. aeruginosa,
E. coli,
S. aureus, and
C. perfringens should not be detected in 100 mL of filtered water.
Samples’ collection and microbiological analyses were conducted by our research group, at Bologna University.
2.4. Statistical Analysis
All statistical analyses were performed using R software (version 4.0.5, “Shake and Throw” R Foundation for Statistical Computing, Vienna, Austria). DataMaid R package was used to generate a baseline descriptive analysis, including frequency, median, mode, and interquartile range values on numeric and categorical variables [
40]. A Shapiro–Wilk test evaluated if the data of temperature and HPCs’ variables came from a normally distributed population. Spearman’s rho rank test was calculated to detect a possible correlation between temperature and HPCs. Graphical representations of HPCs’ contamination trend during the years were elaborated with ggplot2 [
41].
A Wilcoxon–Mann–Whitney test was used to compare the two groups of HPCs at 36 °C and 22 °C values above and below the 25 °C temperature value established by the Guidelines for Drinking-Water Quality. The same test was used to compare the heterotrophic bacteria contamination detected from the beginning (2017) to the current data (2021) of the drinking water surveillance program, and to compare contamination levels pre-COVID (2019) and post-COVD (2021) lockdown periods, included in this study.
Bacteriological data were expressed as mean contamination ± standard deviation (SD) of positive samples. The p-values (p) < 0.05 were considered statistically significant.
3. Results
Considering the contamination found in the previous study [
30], in relation to HPCs at 36 °C and 22 °C,
P. aeruginosa and other pathogenic bacteria,
Table 1 displays the status of MWDs’ contamination at the beginning of the study (2017) and the mean contamination found during the period of the study.
In detail, a total of 477 water samples were collected from 57 MWDs and analyzed for HPCs at 36 °C and 22 °C and for pathogenic bacteria, Enterococcus spp., P. aeruginosa, E. coli, S. aureus, and C. perfringens.
The overall results showed that the samples were more contaminated by HPCs at 36 °C and 22 °C; instead,
P. aeruginosa was detected in 44/477 (9.2%) samples. Total and fecal coliforms,
E. coli,
S. aureus, and
C. perfringens were not detected in any of the investigated samples. The MWDs’ mean contamination found, shown in
Table 1, displayed a continuous decrease during the years of the study. The HPCs at 36 °C and 22 °C data were studied to understand the trend of bacteriological contamination. The analysis described below was not performed for other pathogenic parameters found (e.g.,
P. aeruginosa), due to the sample size of data being too small for statistical reporting and for other microbiological parameters never detected in MWDs during the period.
The positive samples were analyzed with respect to the reference values prescribed by the Italian regulations.
Regarding HPCs at 36 °C and 22 °C, the following microbiological profiles were recorded: a total of 187/477 (39.2%) samples, with a range of contamination of 21–14,940 cfu/mL, and 94/477 (19.7%) samples, with a range of contamination of 104–10,960 cfu/mL, exceeded the regulation limit values of 20 cfu/mL and 100 cfu/mL, respectively, for HPCs at 36 °C and 22 °C.
The contamination found was studied also in relation to the temperature measured, considering the temperature as one of the factors that could influence the bacterial growth. According to the Guidelines for Drinking-Water Quality, the reference water temperature was between the range of 12–20 °C, with a limit not to exceed set at 25 °C [
25,
27,
28,
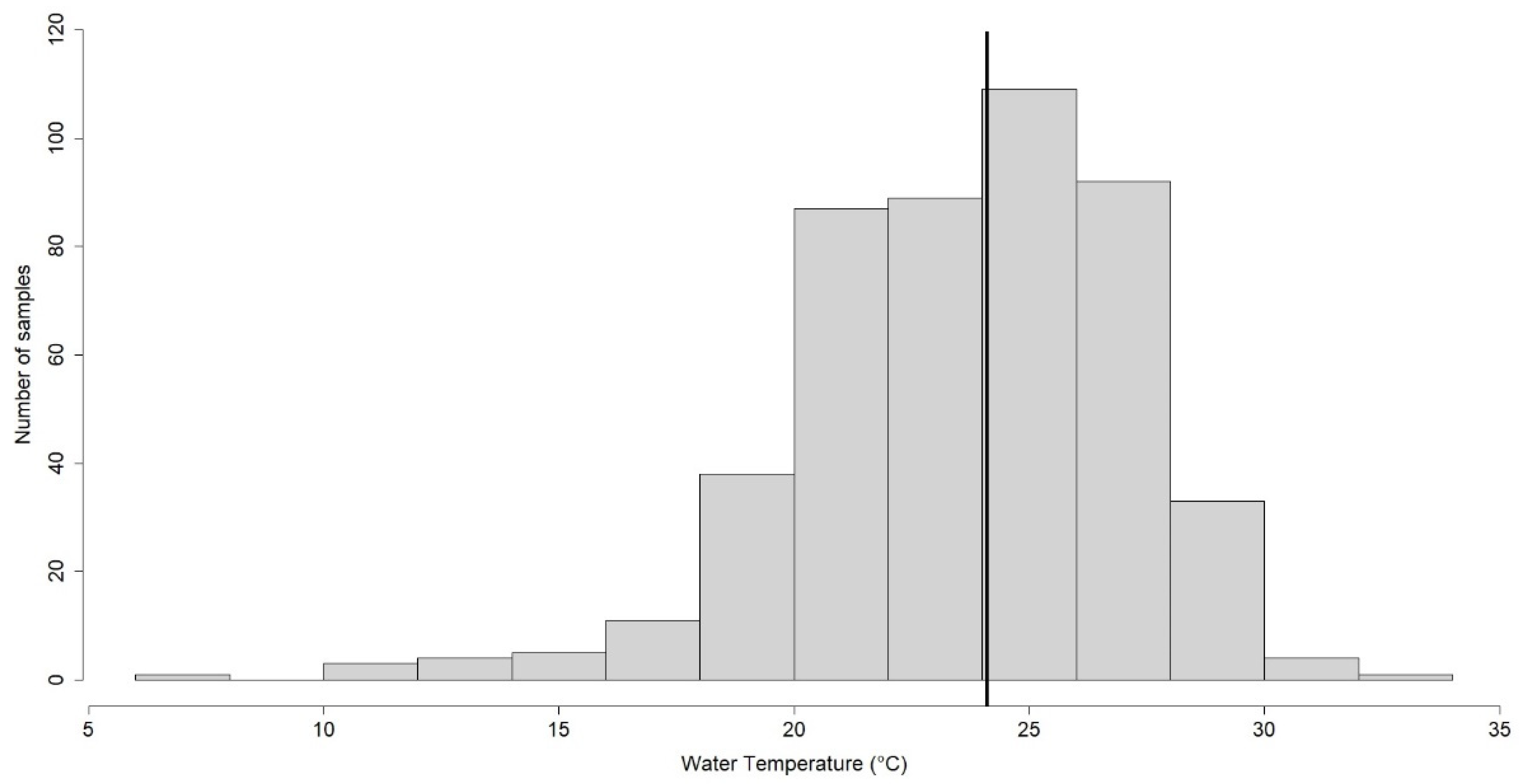
42]. In particular, in our study, 292/477 (61.2%) samples showed temperature values ≤ 25° C and 185/477 (38.8%) samples showed values > 25° C. The samples showed a minimum value measured of 7.2 °C and a maximum of 32.4 °C, with a median value of 24.1 °C.
Figure 2 shows how most samples were centered around the median value of 24.1 °C, a temperature value close to the reference limit threshold of 25 °C [
28].
The HPCs’ contamination found (for both 36 °C and 22 °C) were separated into two groups based on the temperature values above and below the 25 °C temperature threshold, and compared with each other. In both cases there was no significant difference between the groups with values above and below 25 °C, with a p = 0.56 and p = 0.18 for HPCs at 36 °C and 22 °C, respectively.
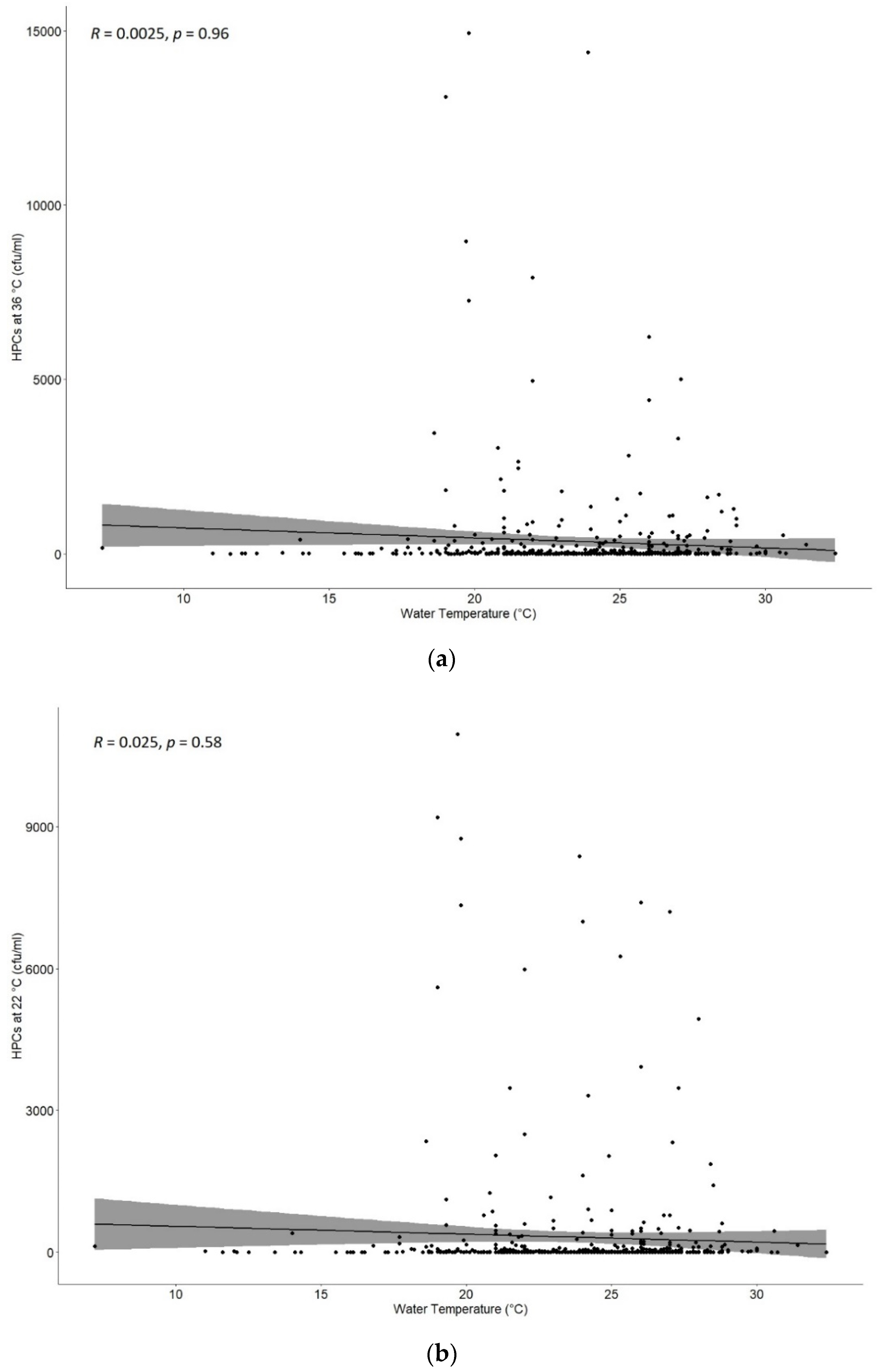
Moreover, data were evaluated to find a possible correlation between HPCs at 36 °C and 22 °C and the temperature of samples measured, as shown in
Figure 3a,b. There was no significant correlation with a
p = 0.96 and
p = 0.58 for HPCs at 36 °C and 22 °C, respectively.
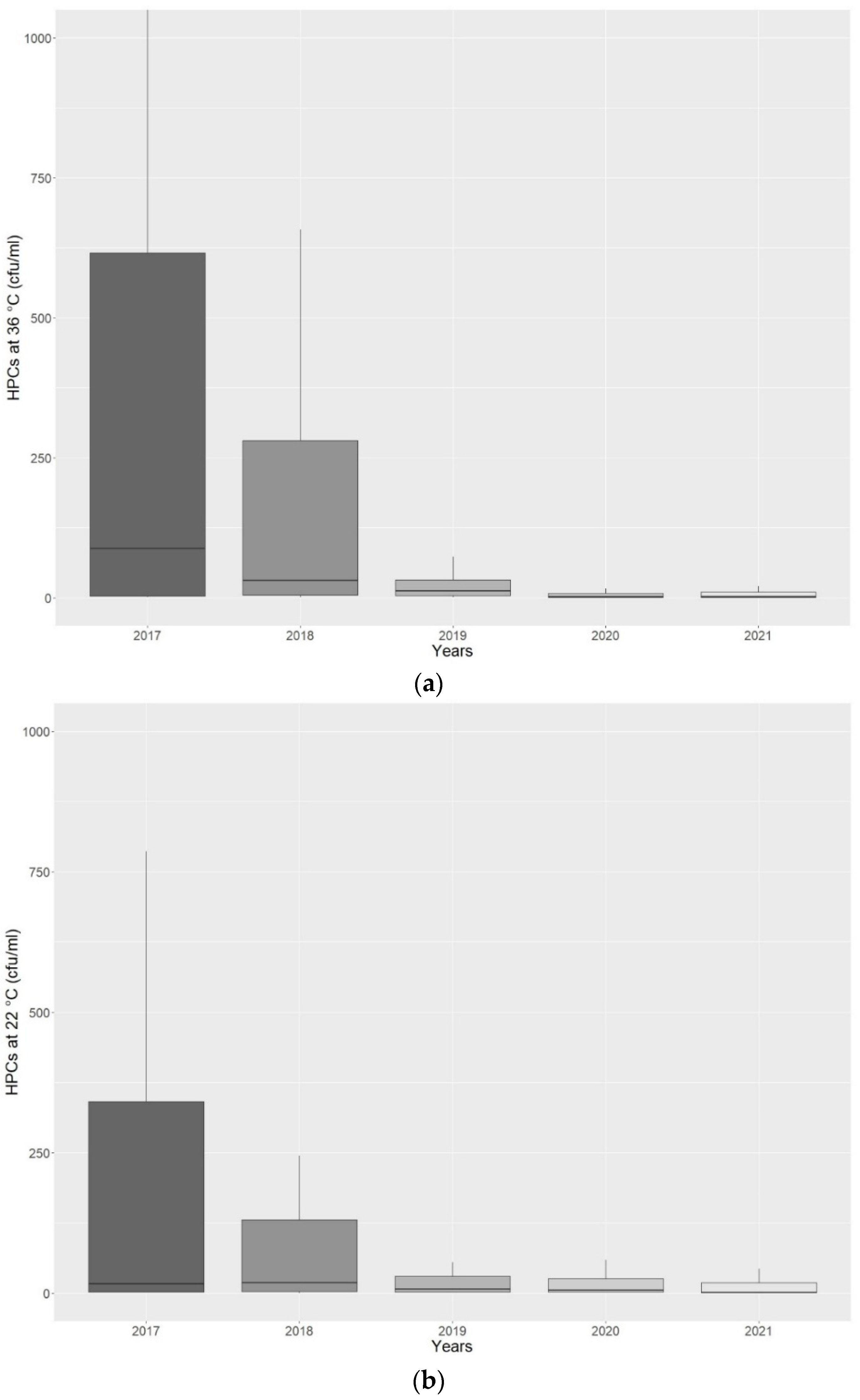
Considering the period of analysis, from 2017 to 2021, to assess the compliance of monitoring and maintenance measures undertaken, we studied the microbiological contamination trend of HPCs at 36 °C and 22 °C (
Figure 4a,b).
Figure 4a,b show how the continuous water quality monitoring and the MWDs’ maintenance, according to International Regulations and Guidelines, led to a decrease in bacterial contamination. The same contamination trend was observed in terms of percentage of positive samples that exceeded the regulation limits. We found 68/109 (62.4%) for HPCs at 36 °C and 36/109 (33.0%) for HPCs at 22 °C positive samples that exceeded the regulation limits in 2017, compared to 11/57 (19.3%) and 5/57 (8.8%) in 2021.
The value of mean contamination ± SD found were 1014.99 ± 2637.63 cfu/mL and 686.25 ± 1952.55 cfu/mL for HPCs at 36 °C and 22 °C, respectively, in 2017, compared to 30.02 ± 77.62 cfu/mL and 91.75 ± 469.23 cfu/mL for HPCs at 36 °C and 22 °C, respectively, in 2021.
These data are supported by the statistical analysis applied on HPCs at 36 °C and 22 °C detected in 2017 compared to 2021, when it was possible to assess a significant decrease of HPCs at 36 °C (p = 0.0000000001) and 22 °C (p = 0.000006) in 2021 compared to 2017.
The WSP implementation resulted in a 43.09% decrease for HPCs at 36 °C and 24.26% decrease for HPCs at 22 °C.
Finally, in order to verify the impact of the lockdown period, due to the global SARS-CoV-2 pandemic that occurred during 2020, which also affected the industrial company involved in this study, causing the closure of MWDs’ devices, we focused on the analysis of the data, both for HPCs at 36 °C and 22 °C, comparing the contamination found in the pre-COVID (year 2019) and post-COVID (year 2021) periods.
The extended period of inactivity did not affect the MDWs’ contamination, as evidenced by a significant decreasing trend of HPCs’ parameters, with a p-value of 0.000005 for 36 °C and 0.001 for 22 °C, respectively. Moreover, a decreasing number of positive samples over the directive limits was observed: 31/94 (33.0%) for HPCs at 36 °C and 9/94 (9.6%) for HPCs at 22 °C in 2019 compared to 11/57 (19.3%) and 5/57 (8.8%) in 2021.
Regarding the contamination found, it was possible to evaluate again a decrease, such as 39.05 ± 86.37 cfu/mL and 69.78 ± 347.89 cfu/mL for HPCs at 36 °C and 22 °C, respectively, in 2019 compared to 30.02 ± 77.62 cfu/mL and 91.75 ± 469.23 cfu/mL for HPCs at 36 °C and 22 °C, respectively, in 2021.
4. Discussion
The MWDs are increasingly used in the private or public areas, such as restaurants or companies, and are a serious problem for public health, concerning potential waterborne pathogens, due to the incorrect use by ordinary consumers and lack or failure of maintenance procedures.
The most relevant health-based target in relation to drinking water is to provide a good-quality water with no taste, odor, or microbiological contaminants. In order to prevent the introduction, growth, and spread of pathogenic bacteria, appropriate cleaning and sanitation procedures must be implemented, following a WSP, as suggested by the International regulation for drinking water quality [
25,
28].
Several studies have already demonstrated how the drinking water quality could change over time within MWDs [
1,
20,
22,
43]. According to these findings, the bacterial contamination found could be attributed to many factors, such as insufficient and inadequate maintenance of the dispenser outlets/nozzles, storage tanks, UV lamps, and manufacturer’s materials, in addition to the absence of knowledge about the correct cleaning procedures and routine functionality checks required to maintain MWDs’ hygiene and safety.
In this study, the role of indicator and common pathogenic bacteria was considered. The type of MWDs analyzed showed a poor quality of drinking water at the beginning of study, with a high risk for consumers. Starting from these contaminations and considering that the Company acquired these devices to definitively remove the use of plastic water bottles, a new WSP was introduced, according to Italian directives. Already in 2017 compared to the previous study of Girolamini et al. in 2019, the contamination by
Enterococcus spp.,
E. coli,
S. aureus, and
C. perfringens, disappeared, despite few samples positive for
P. aeruginosa. Interesting data were found for HPCs’ bacteria at 36 °C and 22 °C. Although, as suggested by WHO [
28], the HPCs’ bacteria were not a suitable criteria for estimating the quality of drinking water, they could be considered as a clean water indicator, and in our study they were used to check the maintenance and disinfection protocol applied over time. Several studies over time were in line with our results. The Authors clearly showed the main contamination found in devices used to supply microfiltered water was attributed to HPCs at 36 °C and 22 °C, followed by
P. aeruginosa and fungi [
2,
42].
Starting from the results obtained, to explain the level of HPCs found, two main factors were considered: the temperature values detected at MWDs’ output and the performance of continuous maintenance procedures applied in the industrial company from 2017 until today (2021).
The temperature is considered a relevant determinant to preserve water quality, since its changes can influence physical, chemical, and biological processes that occur in natural and artificial environments. Specifically, temperature can influence the dynamics of microorganisms promoting biofilm formation, which could be a reservoir of waterborne pathogens, guaranteeing their survival [
43,
44]. According to Semerjian et al. 2020 [
1], another risk factor is the MWDs’ location, such as placing them in humid environments or in facilities or rooms with poor ventilation. This factor could promote the bacteria growth due to the warming of MWDs, with a consequent increase of water temperatures supplied.
The WHO guidelines recommend that drinking water assumes a temperature value between 12 to 20 °C, with an alert limit of 25 °C; this limit must not be exceeded [
25,
27,
28].
The results obtained displayed how the MWDs showed a large range of temperature values (min. 7.2 °C–max. 32.4 °C), with a median of 24.1 °C, strictly close to 25 °C. In order to evaluate the impact of the temperature found on the HPCs’ contamination, two groups of data were selected, considering the MWDs with temperature above and below 25 °C. The comparison showed no significance differences between them. Considering that the majority of the detected temperatures fell near the threshold value, the result did not surprise.
Furthermore, no statistical correlation between HPCs’ values at 36 °C and 22 °C contamination and the temperature values was detected (p = 0.74 and p = 0.59, respectively).
The data obtained could be explained with the activities implemented with the WSP over the years in the Company, aimed to maintain a constant temperature under 25 °C, avoiding exceeding the limit proposed, according to the Guidelines. This aim was achieved through a strict control of flushing activities (every day) to avoid the water stagnation, in addition to the choice of the right MWDs’ location, preferring ventilated areas, far from the heating systems (e.g., fan coil units), and avoiding direct light exposure to prevent the device warming. All these factors, as suggested by Semerjian et al. in 2020, contribute to bacteria and fungi proliferation [
1].
The maintenance procedures allowed both keeping the temperature constant near the value established by law of 25 °C and decreasing HPCs’ contamination values at 36 °C and 22 °C over time, as demonstrated by data comparison of heterotrophic bacteria from 2017 and 2021.
Despite the low pathogenic impact of HPCs on human health, their presence in water highlights the need to implement maintenance protocols to avoid the biofilm growth. Especially in MWDs, considering their functional characteristics and the lower volume of water supply, it is important to realize ordinary maintenance procedures such as cleaning surfaces, flushing the water every day at the opening time, and the descaling procedures [
2,
23]. These ordinary measures, combined with the control of temperature, the maintenance of manufacturer’s characteristics, and the microbiological parameters’ proliferation, permit a rapid detection of health-based risks factors, avoiding the deterioration of water quality.
These considerations are also supported by the results reported during the recent global SARS-CoV-2 pandemic, in 2020. Despite the closure of the company and the closure of MWDs, the implementation of the maintenance and sanitation procedures, such as flushing at input and output points three times a week or daily cleaning of external surfaces, permitted preserving the water quality and avoiding the use of shock treatments by chemical disinfectants in MDWs’ water circuits. Indeed, the statistical data analysis from 2019 to 2021 underlined a further decrease of microbiological contamination, demonstrating how the only maintenance procedure without the use of continuous disinfection treatments was able to limit the risk of water contamination.
The MWDs involved in the study were supplied by the same municipal water, through a single pipeline, that, inside the Company, was divided into different lines. The different water qualities could be explained with the contamination of the last part of the pipeline (the point of connection with the distal outlets) and the different volumes of water supplies, but also by the consumer habits. In our experience, the MWDs located in the production area, especially in a metallurgical company, were more subjected to air pollution and use with dirty hands and poor hygienic conditions, with respect to devices located in the kitchen, canteens, or close to the offices. Based on our experience, especially during the last year, to reduce the plastic pollution, there was an increase in the use of water bottles that were directly filled from MWDs. During the filling, the contact between the water bottle and MWDs’ nozzles could contribute to the device contamination. Therefore, it was crucial to propose a training to the consumers on the proper use of MWDs. All the employers were trained regarding the need to regularly wash, empty, and dry the bottles, and to fill them avoiding contact with the MWDs’ nozzles, to minimize the cross-contamination risks.
Moreover, according to our WSP, the maintenance procedures based on flushing, descaling, and disinfection were performed by trained professionals and scheduled on the same days. This means that in a range of 2 days all devices were submitted to the same procedure in order to obtain the same sanitation level. All of these aspects were discussed also during the workers’ training program and strict monitoring procedures were implemented by the heath safety environment manager designed by the Company.
These considerations need also to be combined with the application of the manufacturer’s instructions. The MWDs should be regularly cleaned and the integrity of all components should be checked. The following aspects must be included in the maintenance and monitoring program: the cleaning of the outside of the device; the replacement of electrical components and power leads; checking the water connection; inspections of level indicators and control devices; cleaning of the water collection tray; cooling the condenser, water filters, and water dispenser spouts; and UV lamp maintenance and replacement. All these practices improve the hygienic conditions of MWDs, promoting safe and good drinking water quality.
5. Conclusions
The outcomes of this work prove the long-term effectiveness of the WSP followed by the company and the consumers, in addition to the role of a multidisciplinary approach established to assure the drinking water quality, according to the International and National directives. In addition, this study suggests a need to implement more rigorous microbiological monitoring to apply to MWDs’ environment, underestimated to date, despite the large diffusion of these devices in the communities.
Lack of awareness and knowledge on these issues, in addition to missing systematical control by users as well as by public health authorities, can lead to a high risk to human health, especially in hospitals and, in general, in all health-care settings, where the use of MWDs is already widespread and the presence of immunocompromised people could increase the risk of infections.
Moreover, these findings assume a relevant importance even across new scenarios: the higher request for plastic reduction, the introduction, starting from January 2021, of the new European Directive 2184/2020 on the quality of water intended for human consumption focused to implement a WSP approach in all facilities, and the introduction of new microbiological parameters in order to assure the water quality, such as, for example,
Legionella spp. [
45]. Considering the habitat, the pathogenic role, and the impact on public health demonstrated for these bacteria, the sanitation and maintenance procedures of MWDs will play a relevant role to preserve the water quality.