Effects of Chlorpyrifos Ethyl on Cholinesterase and Growth of Silver Barb (Barbonymus gonionotus)
Abstract
:1. Introduction
2. Methodology
2.1. Test Animals
2.2. Pesticide
2.3. Experimental Design
2.3.1. Acute Toxicity Testing
2.3.2. Effect of Sub-Acute Concentrations of Chlorpyrifos Ethyl on the Brain Cholinesterase Activity of Silver Barb
- Sample preparation
- Cholinesterase assay
2.3.3. Effect of Sub-Acute Concentrations of Chlorpyrifos Ethyl on the Growth of Silver Barb
2.3.4. Calculation
2.3.5. Data Analysis
3. Results and Discussion
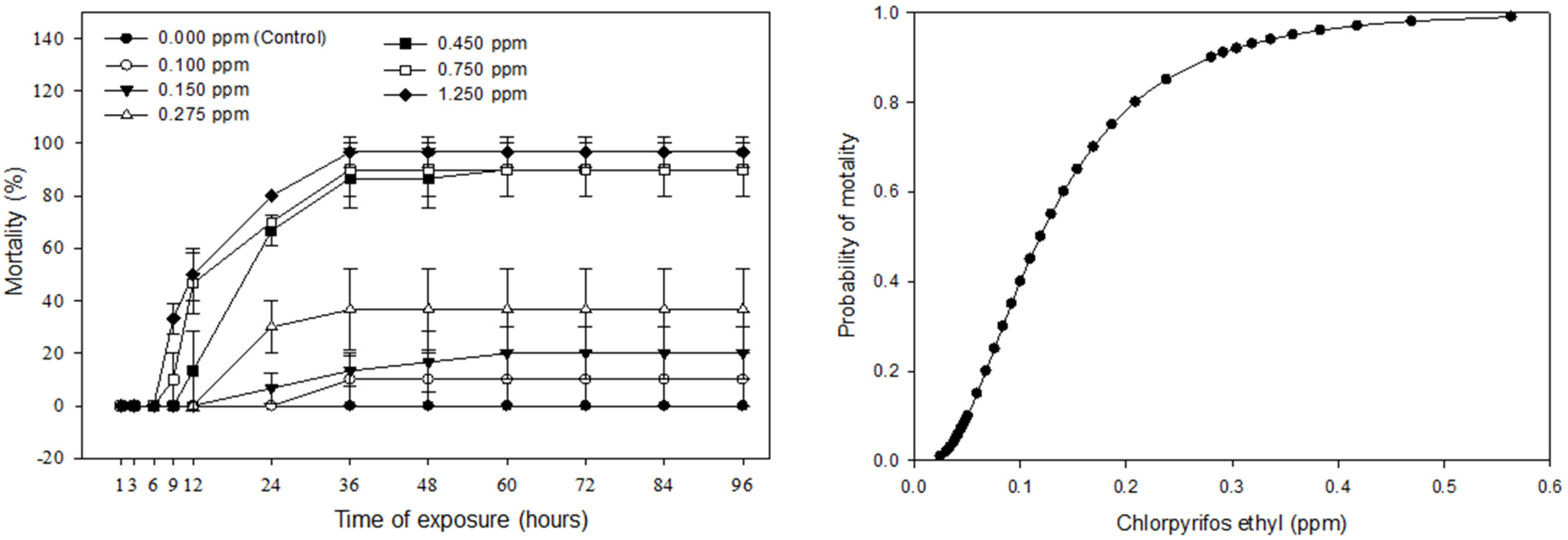
3.1. The LC50-96 h of Chlorpyrifos Ethyl for Fingerling Silver Barb
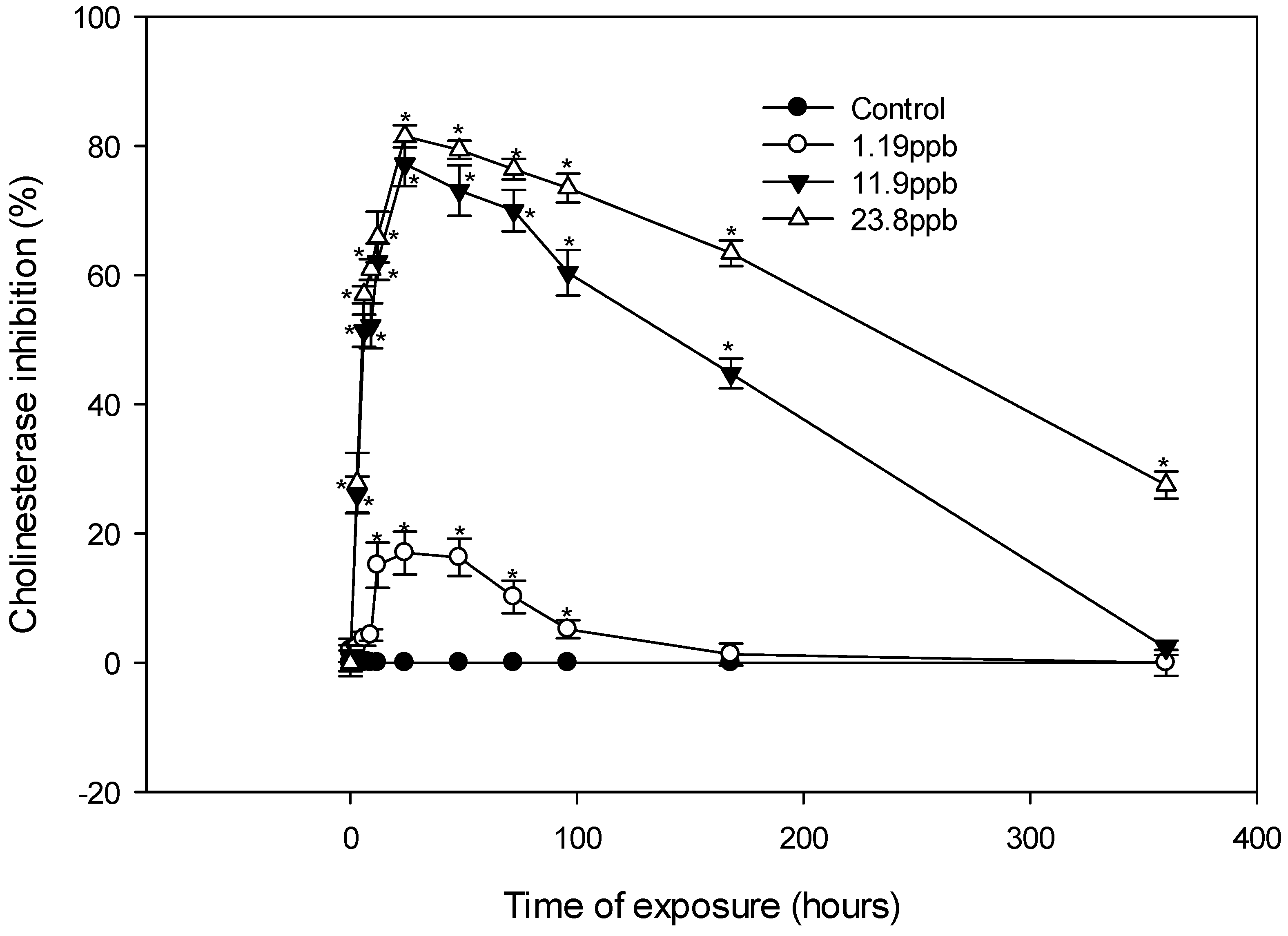
3.2. Effects of Sub-Acute Concentrations of Chlorpyrifos Ethyl on ChE Activity
3.3. Effects of Sub-Acute Chlorpyrifos Ethyl on the Growth Performance of Silver Barb
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Berg, H. Pesticide use in rice and rice-fish farms in the Mekong Delta, Vietnam. Crop Prot. 2001, 20, 897–905. [Google Scholar] [CrossRef]
- Berg, H.; Tam, N.T. Use of pesticides and attitude to pest management strategies among rice and rice-fish farmers in the Mekong Delta, Vietnam. Int. J. Pest Manag. 2012, 58, 153–164. [Google Scholar] [CrossRef]
- Toan, N.V.; Cong, N.V. Present status of pesticide uses in rice cultivation in the Mekong Delta. J. Nat. Resour. Environ. 2018, 5, 26–30. [Google Scholar]
- MARD (Ministry of Agriculture and Rural Development). List of Pesticides Permitted, Restricted, and Banned for Use; MARD: Hanoi, Vietnam, 9 September 2020; Circular No. 10/2020/TT-BNNPTNT. (In Vietnamese)
- Tam, N.T.; Berg, H.; Laureus, J.; Cong, N.V.; Tedengren, M. Effects of Sequential Applications of Bassa 50EC (Fenobucarb) and Vitashield 40EC (Chlorpyrifos ethyl) on Acetylcholinesterase Activity in Climbing Perch (Anabas testudineus) Cultured in Rice Fields in the Mekong Delta, Vietnam. Bull. Environ. Contam. Toxicol. 2016, 97, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Stenersen, J. Chemical Pesticides. Mode of Action and Toxicity; CRC Press: Boca Raton, FL, USA; London, UK, 2004; pp. 90–113. [Google Scholar]
- Muttappa, K.; Reddy, H.R.V.; Padmanabha, A.; Shridhar, B.; Prabhudeva, K.N.; Basavaraju, Y.; Gangadhar, G.; Rajanna, K.B.; Chethan, N. Chlorpyrifos induced histological changes in the liver of tilapia (Oreochromis mossambicus). Int. J. Recent Sci. Res. 2015, 6, 5819–5823. [Google Scholar]
- Ogueji, E.O. Comparative acute toxicity of chlorpyrifos-ethyl (organophosphate) and lambda-cyhalothrin (Pyrethroid) to the African catfish (C. gariepinus) using some biochemical parameters. Glob. J. Pure Appl. Sci. 2008, 14, 263–269. [Google Scholar] [CrossRef]
- Khoa, T.T.; Huong, T.T.T. Classification of Fresh Water Fish of the Mekong River Delta, Vietnam; Cantho University Press: Cantho City, Vietnam, 1993. (In Vietnamese) [Google Scholar]
- Rothuis, A.J.; Nhan, D.K.; Richter, C.J.J.; Ollevier, F. Rice with fish culture in the semi-deep waters of the Mekong Delta, Vietnam: A socio-economical survey. Aquac. Res. 1998, 29, 47–571. [Google Scholar] [CrossRef]
- Cong, N.V.; Phuong, N.T.; Bayley, M. Sensitivity of brain cholinesterase activity to diazinon (Basudin 50EC) and fenobucarb (Bassa 50EC) insecticides in the air-breathing fish Channa striata (Bloch, 1793). Environ. Toxicol. Chem. 2006, 25, 1418–1425. [Google Scholar] [CrossRef] [PubMed]
- Cong, N.V.; Phuong, N.T.; Bayley, M. Effects of repeated exposure of diazinon on cholinesterase activity and growth in snakehead (Channa striata). Ecotoxicol. Environ. Saf. 2009, 72, 699–703. [Google Scholar] [CrossRef] [PubMed]
- APHA. Standard Methods for the Examination of Water and WastWater, 15th ed.; American Public Health Association: Washington, DC, USA, 1998; p. 1134. [Google Scholar]
- Ellman, G.L.; Courtney, D.; Anderdres, V.J.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharm. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Parfitt, C.H. Pesticide and Industrial Chemical Residues. In Official Methods of Analysis of AOAC International, 17th ed.; Horwitz, W., Ed.; Vol 1—Agrochemicals; Contaminants; Drugs; Association of Analytical Communities International: Gaithersburg, MD, USA, 2000; pp. 22–23. [Google Scholar]
- Finney, D.J. Probit Analysis, 3rd ed.; Cambridge University Press: London, UK, 1971; pp. 20–49. [Google Scholar]
- Cong, N.V.; Phuong, N.T.; Bayley, M. Effects of Sublethal Concentrations of Diazinon on Surfacing and Hanging Behaviors of Snakehead (Channa striata). Fish. Sci. 2008, 74, 1330–1332. [Google Scholar] [CrossRef]
- Arunachalam, S.; Palanichamy, S. Sublethal Effects of Carbaryl on Surfacing Behaviour and Food Utilization in the Air-Breathing Fish, Macropodus cupanus. Physiol. Behav. 1982, 29, 23–27. [Google Scholar] [CrossRef]
- Fulton, M.H.; Key, P.B. Acetylcholinesterase inhibition in estuarine fish and invertabrates as an indicator of Organophosphorus insecticide exposure and effects. Environ. Toxicol. Chem. 2001, 20, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Beauvais, S.L.; Jones, S.B.; Parris, J.T.; Brewer, S.K.; Little, E.E. Cholinergic and Behavioral Neurotoxicity of Carbaryl and Cadmium to Larval Rainbow Trout (Oncorhynchus mykiss). Ecotoxicol. Environ. Saf. 2001, 49, 84–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brewer, S.K.; Little, E.E.; DeLonay, A.J.; Beauvais, S.L.; Jones, S.B.; Ellersieck, M.R. Behavioral Dysfunctions Correlate to Altered Physiology in Rainbow Trout (Oncorynchus mykiss) Exposed to Cholinesterase-Inhibiting Chemicals. Arch. Environ. Contam. Toxicol. 2001, 40, 70–76. [Google Scholar] [PubMed]
- Roex, E.W.M.; Vries, E.D.; van Gestel, C.A.M. Sensitivity of the zebrafish (Danio rerio) early life stage test for compounds with different modes of action. Environ. Poll. 2002, 120, 355–362. [Google Scholar] [CrossRef]
- Tam, N.T.; Berg, H.; Cong, N.V. Evaluation of the joint toxicity of chlorpyrifos ethyl and fenobucarb on climbing perch (Anabas testudineus) from rice fields in the Mekong Delta, Vietnam. Environ. Sci. Pollut. Res. 2018, 25, 13226–13234. [Google Scholar] [CrossRef] [PubMed]
| Chlorpyrifos Ethyl (ppm) | Temperature (°C) | pH | DO (mg/L) | |||
|---|---|---|---|---|---|---|
| Morning | Afternoon | Morning | Afternoon | Morning | Afternoon | |
| Control | 26.8 ± 0.3 | 28.6 ± 0.5 | 7.1 ± 0.1 | 7.1 ± 0.1 | 3.6 ± 0.7 | 2.8 ± 0.5 |
| 0.100 | 26.9 ± 0.3 | 28.5 ± 0.5 | 7.1 ± 0.0 | 7.1 ± 0.2 | 3.5 ± 0.5 | 3.1 ± 0.6 |
| 0.150 | 26.9 ± 0.3 | 28.7 ± 0.4 | 7.1 ± 0.1 | 7.1 ± 0.1 | 3.3 ± 0.4 | 3.0 ± 0.3 |
| 0.275 | 26.8 ± 0.3 | 28.9 ± 0.2 | 7.1 ± 0.0 | 7.1 ± 0.2 | 3.4 ± 0.3 | 3.2 ± 0.4 |
| 0.450 | 27.0 ± 0.1 | 28.4 ± 0.5 | 7.2 ± 0.1 | 7.1 ± 0.1 | 3.5 ± 1.3 | 3.0 ± 1.0 |
| 0.750 | 26.8 ± 0.3 | 28.5 ± 0.4 | 7.2 ± 0.1 | 7.1 ± 0.2 | 3.3 ± 1.1 | 3.2 ± 1.0 |
| 1.250 | 27.0 ± 0.2 | 28.6 ± 0.4 | 7.2 ± 0.1 | 7.2 ± 0.1 | 3.4 ± 1.2 | 3.4 ± 1.1 |
| Chlorpyrifos Ethyl (ppb) | Temperature (°C) | pH | DO (mg/L) | |||
|---|---|---|---|---|---|---|
| Morning | Afternoon | Morning | Afternoon | Morning | Afternoon | |
| Control | 28.1 ± 0.1 | 29.5 ± 0.1 | 7.1 ± 0.1 | 7.3 ± 0.1 | 4.0 ± 0.02 | 2.9 ± 0.03 |
| 1.19 | 28.2 ± 0.1 | 29.9 ± 0.1 | 7.1 ± 0.0 | 7.3 ± 0.2 | 3.9 ± 0.04 | 2.8 ± 0.02 |
| 11.9 | 28.1 ± 0.1 | 29.6 ± 0.1 | 7.1 ± 0.1 | 7.3 ± 0.1 | 3.8 ± 0.04 | 2.9 ± 0.02 |
| 23.8 | 28.3 ± 0.1 | 29.6 ± 0.1 | 7.1 ± 0.0 | 7.3 ± 0.2 | 3.8 ± 0.02 | 2.7 ± 0.02 |
| Treatment | Vitashield 40EC (ppb) | Actual Chlorpyrifos Ethyl (µg/L) |
|---|---|---|
| Control | Tap water | <MDL = 0.1 µg/L |
| 1% LC50 | 3.0 | 0.66 (0.58 ÷ 0.78) |
| 10% LC50 | 30.0 | 10.43 (9.60 ÷ 11.20) |
| 20% LC50 | 60.0 | 22.97 (22.10 ÷ 23.80) |
| Environmental Parameters | Time | Treatment | |||
|---|---|---|---|---|---|
| Control | 1% LC50-96 h | 10% LC50-96 h | 20% LC50-96 h | ||
| Temperature (°C) | M | 27.79 ± 0.12 | 27.72 ± 0.12 | 27.74 ± 0.12 | 27.77 ± 0.13 |
| A | 28.74 ± 0.12 | 28.72 ± 0.12 | 28.72 ± 0.13 | 28.77 ± 0.13 | |
| pH | M | 7.50 ± 0.02 | 7.51 ± 0.02 | 7.52 ± 0.02 | 7.53 ± 0.02 |
| A | 7.44 ± 0.02 | 7.45 ± 0.02 | 7.47 ± 0.02 | 7.47 ± 0.02 | |
| DO (mg/L) | M | 7.19 ± 0.06 | 7.15 ± 0.07 | 7.21 ± 0.07 | 7.26 ± 0.06 |
| A | 6.69 ± 0.09 | 6.65 ± 0.11 | 6.75 ± 0.11 | 6.77 ± 0.1 | |
| N-NO2 (mg/L) | M | 0.308 ± 0.047 | 0.340 ± 0.049 | 0.347 ± 0.051 | 0.362 ± 0.049 |
| A | 0.254 ± 0.041 | 0.334 ± 0.044 | 0.388 ± 0.050 | 0.339 ± 0.050 | |
| N-NH4 (mg/L) | M | 0.232 ± 0.06 | 0.241 ± 0.052 | 0.206 ± 0.047 | 0.449 ± 0.13 |
| A | 0.241 ± 0.046 | 0.249 ± 0.046 | 0.317 ± 0.053 | 0.413 ± 0.103 | |
| Growth Parameters | Treatments | |||
|---|---|---|---|---|
| Control | 1% LC50-96 h | 10% LC50-96 h | 20% LC50-96 h | |
| FI (mg/g/d) | 20.3 ± 0.4 a | 20.4 ± 0.5 a | 21.2 ± 0.5 a | 22.1 ± 1.5 a |
| DWG (mg/d) | 95.2 ± 4.9 a | 92.8 ± 2.3 a | 75.5 ± 2.0 b | 60.6 ± 2.9 c |
| SGR (%/d) | 1.86 ± 0.04 a | 1.84 ± 0.01 a | 1.61 ± 0.02 b | 1.39 ± 0.03 c |
| FCR | 1.82 ± 0.06 a | 1.83 ± 0.03 a | 2.05 ± 0.05 b | 2.34 ± 0.22 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cong, N.V.; Danh, D.T.; Nam, T.S. Effects of Chlorpyrifos Ethyl on Cholinesterase and Growth of Silver Barb (Barbonymus gonionotus). Water 2021, 13, 2885. https://doi.org/10.3390/w13202885
Cong NV, Danh DT, Nam TS. Effects of Chlorpyrifos Ethyl on Cholinesterase and Growth of Silver Barb (Barbonymus gonionotus). Water. 2021; 13(20):2885. https://doi.org/10.3390/w13202885
Chicago/Turabian StyleCong, Nguyen Van, Dinh Thai Danh, and Tran Sy Nam. 2021. "Effects of Chlorpyrifos Ethyl on Cholinesterase and Growth of Silver Barb (Barbonymus gonionotus)" Water 13, no. 20: 2885. https://doi.org/10.3390/w13202885
APA StyleCong, N. V., Danh, D. T., & Nam, T. S. (2021). Effects of Chlorpyrifos Ethyl on Cholinesterase and Growth of Silver Barb (Barbonymus gonionotus). Water, 13(20), 2885. https://doi.org/10.3390/w13202885






