Phosphodiesterase-5 (PDE-5) Inhibitors as Emergent Environmental Contaminants: Advanced Remediation and Analytical Methods
Abstract
1. Introduction
2. Aim and Review Methodology
3. Source of PDE-5 Inhibitors in the Environment
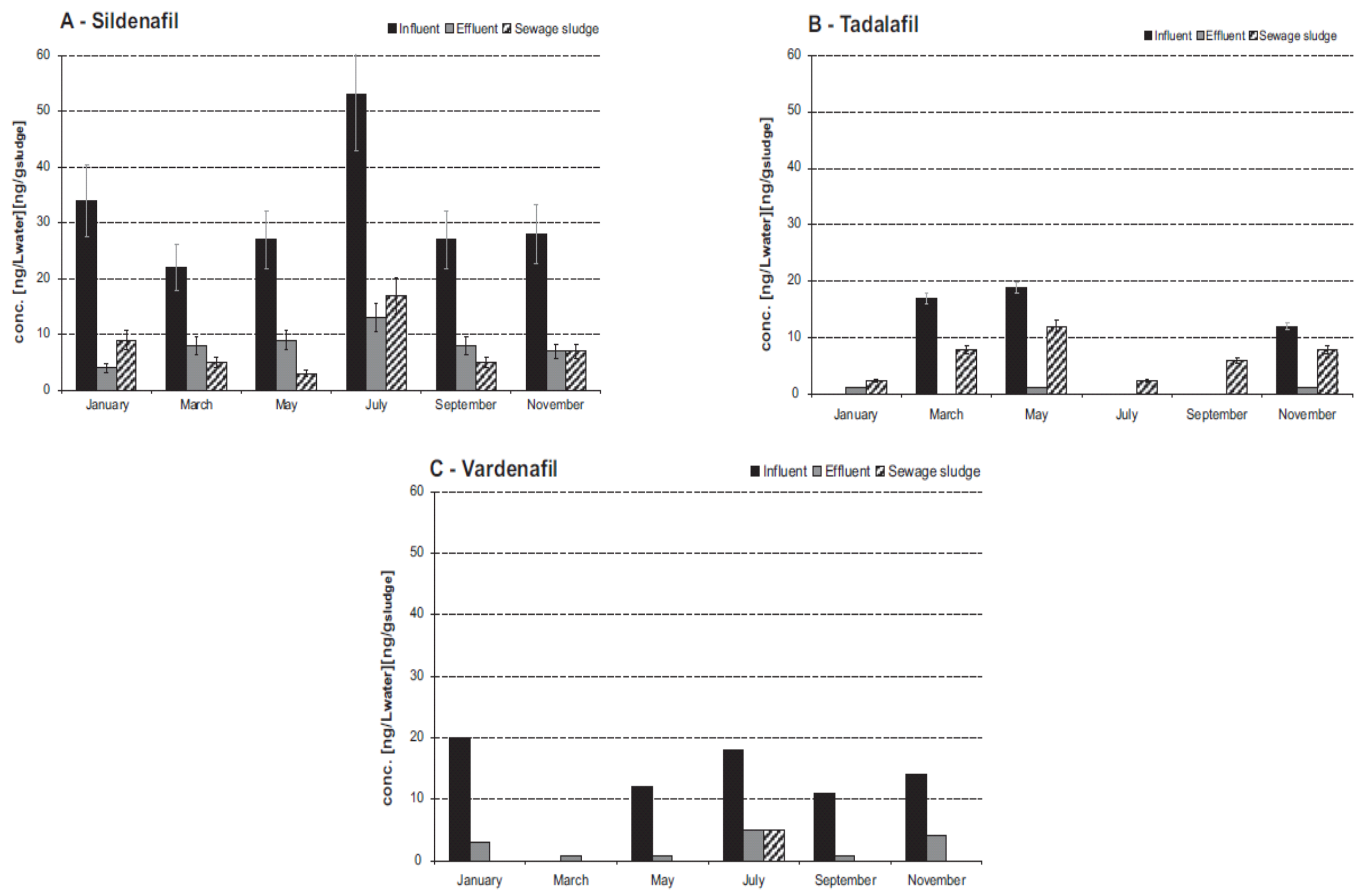
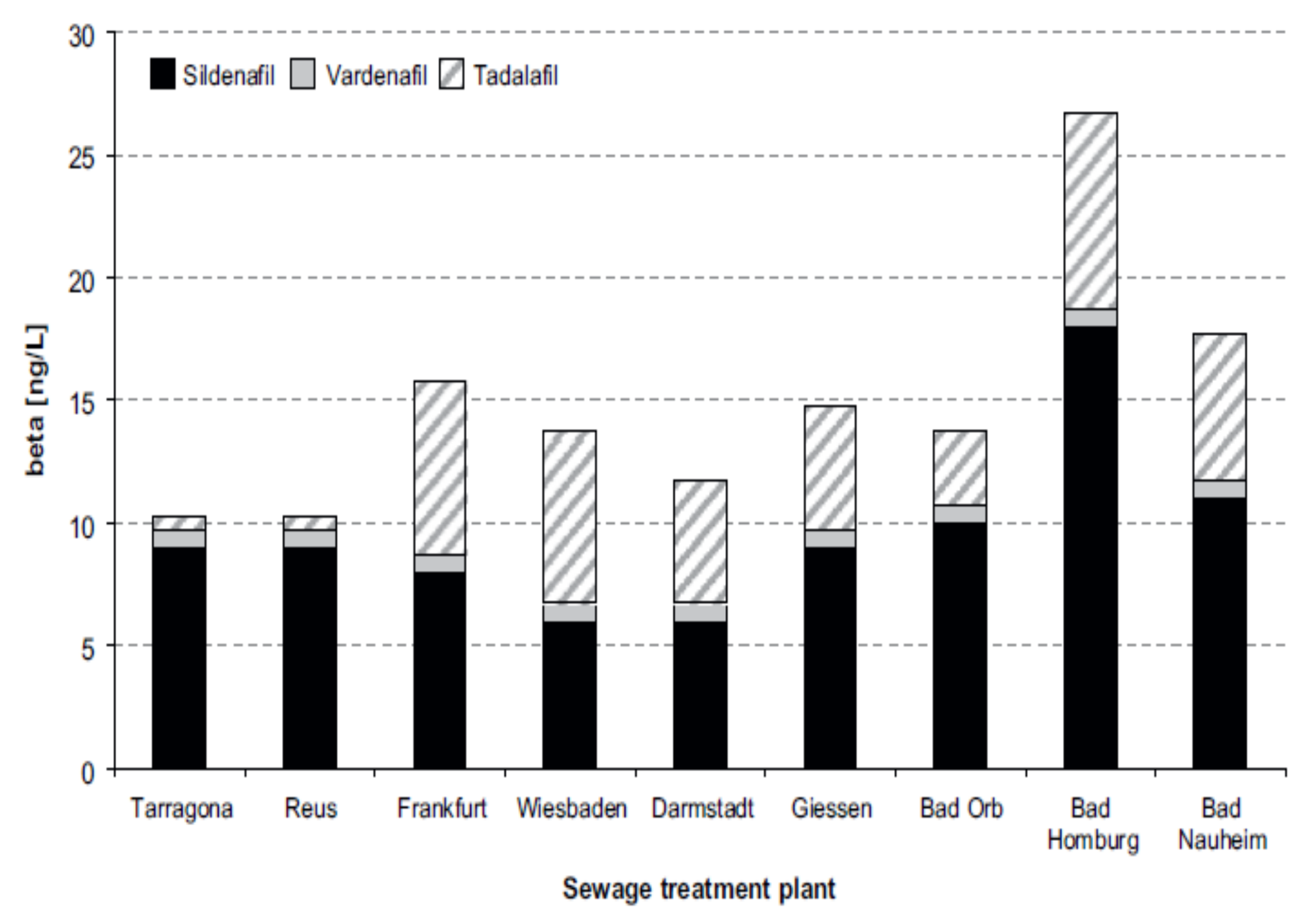
4. Content of PDE-5 Inhibitors in WWTPs and STPs
5. Removal Treatments
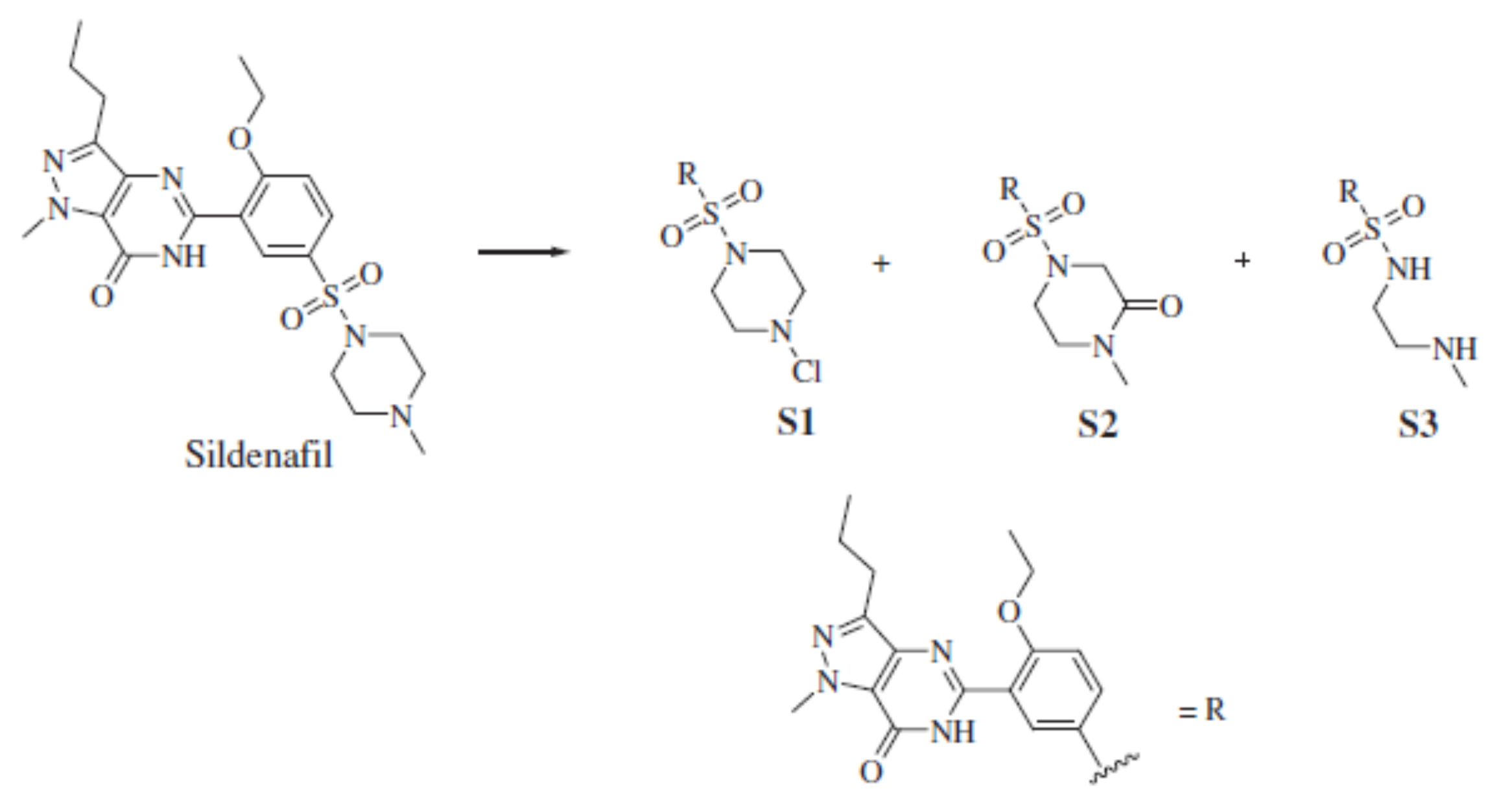
5.1. Advanced Oxidation Treatment
5.2. Biological Degradation
5.3. Adsorption on Activated Carbon
6. Toxicity Assessment
7. Impact on the Aquatic Organisms
8. Analytical Methods for PDE-5 Inhibitors Detection
8.1. Sample Preparation
8.2. Spectrophotometric Methods
8.3. Chromatographic Methods Coupled with UV/Vis Detectors and Mass Spectrometry (MS)
9. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Geissen, V.; Mol, H.; Klumpp, E.; Umlauf, G.; Nadal, M.; van der Ploeg, M.; van de Zee, S.E.A.T.M.; Ritsema, C.J. Emerging pollutants in the environment: A challenge for water resource management. Int. Soil Water Conserv. Res. 2015, 3, 57–65. [Google Scholar] [CrossRef]
- Tran, N.H.; Reinhard, M.; Yew-Hoong Gin, K. Occurrence and fate of emerging contaminants in municipal wastewater treatment plants from different geographical regions—A review. Water Res. 2017, 133, 182–207. [Google Scholar] [CrossRef]
- Zuccato, E.; Calamari, D.; Natangelo, M.; Fanelli, R. Presence of therapeutic drugs in the environment. Lancet 2000, 355, 1789–1790. [Google Scholar] [CrossRef]
- Susa, S.T.; Preuss, C.V. Drug Metabolism. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Halling-Sørensen, B.; Nors Nielsen, S.; Lanzky, P.F.; Ingerslev, F.; Holten Lützhøft, H.C.; Jørgensen, S.E. Occurrence, fate and effects of pharmaceutical substances in the environment—A review. Chemosphere 1998, 36, 357–393. [Google Scholar] [CrossRef]
- Zhou, J.; Broodbank, N. Sediment-water interactions of pharmaceutical residues in the river environment. Water Res. 2014, 48, 61–70. [Google Scholar] [CrossRef]
- Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. Removal of pharmaceuticals in sewage treatment plants in Italy. Environ. Sci. Technol. 2006, 40, 357–363. [Google Scholar] [CrossRef]
- Buser, H.R.; Müller, M.D.; Theobald, N. Occurrence of the pharmaceutical drug clofibric acid and the herbicide mecoprop in various Swiss Lakes and in the North Sea. Environ. Sci. Technol. 1998, 32, 188–192. [Google Scholar] [CrossRef]
- Calamari, D.; Zuccato, E.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Strategic survey of therapeutic drugs in the rivers Po and Lambro in Northern Italy. Environ. Sci. Technol. 2003, 37, 1241–1248. [Google Scholar] [CrossRef]
- Cedergreen, N. Quantifying synergy: A systematic review of mixture toxicity studies within environmental toxicology. PLoS ONE 2014, 9, e96580. [Google Scholar] [CrossRef]
- Hernández, A.F.; Gil, F.; Lacasaña, M. Toxicological interactions of pesticide mixtures: An update. Arch. Toxicol. 2017, 91, 3211–3223. [Google Scholar] [CrossRef]
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 1092–2172. [Google Scholar] [CrossRef] [PubMed]
- Directive 2008/105/EC of the European Parliament and of the Council of 16 December 2008 on Environmental Quality Standards in the Field of Water Policy. 2008. Available online: https://eur-lex.europa.eu/eli/dir/2008/105/oj (accessed on 4 May 2020).
- European Union. Commission Implementing Decision (EU) 2018/840 of 5 June 2018 Establishing a Watch List of Substances for Union-Wide Monitoring in the Field of Water Policy Pursuant to Directive 2008/105/EC of the European Parliament and of the Council and Repealing Commission Implementing Decision (EU) 2015/495 (Notified under Document C(2018) 3362. 2018. Available online: https://eur-lex.europa.eu/eli/dec_impl/2018/840/oj (accessed on 4 May 2020).
- Nieto, A.; Peschka, M.; Borrull, F.; Pocurull, E.; Marcé, R.M.; Knepper, T.P. Phosphodiesterase type V inhibitors: Occurrence and fate in wastewater and sewage sludge. Water Res. 2010, 44, 1607–1615. [Google Scholar] [CrossRef] [PubMed]
- Mas, M. Molecular mechanisms of penile erection. Arch. Esp. Urol. 2010, 63, 589–598. [Google Scholar] [PubMed]
- White, A. The State of Men’s Health in Europe: Extended Report; European Union: Brussels, Belgium, 2011; ISBN 978-92-79-20167-7. [Google Scholar] [CrossRef]
- Goldstein, I.; Burnett, A.L.; Rosen, R.C.; Park, P.W.; Stecher, V.J. The Serendipitous Story of Sildenafil: An Unexpected Oral Therapy for Erectile Dysfunction. Sex. Med. Rev. 2019, 7, 115–128. [Google Scholar] [CrossRef]
- Maiorino, M.I.; Bellastella, G.; Esposito, K. Lifestyle modifications and erectile dysfunction: What can be expected? Asian J. Androl. 2015, 17, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Maiorino, M.I.; Bellastella, G.; Esposito, K. Diabetes and sexual dysfunction: Current perspectives. Diabetes Metab. Syndr. Obes. Targets Ther. 2014, 7, 95–105. [Google Scholar] [CrossRef]
- Yafi, F.A.; Jenkins, L.; Albersen, M.; Corona, G.; Isidori, A.M.; Goldfarb, S.; Maggi, M.; Nelson, C.J.; Parish, S.; Salonia, A.; et al. Erectile dysfunction. Nat. Rev. Dis. Prim. 2016, 2, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Taichman, D.B.; Ornelas, J.; Chung, L.; Klinger, J.R.; Lewis, S.; Mandel, J.; Palevsky, H.I.; Rich, S.; Sood, N.; Rosenzweig, E.B.; et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST guideline and expert panel report. Chest 2014, 146, 449–475. [Google Scholar] [CrossRef]
- Rashid, J.; Patel, B.; Nozik-Grayck, E.; McMurtry, I.F.; Stenmark, K.R.; Ahsan, F. Inhaled sildenafil as an alternative to oral sildenafil in the treatment of pulmonary arterial hypertension (PAH). J. Control. Release 2017, 250, 96–106. [Google Scholar] [CrossRef]
- Chiang, J.; Yafi, F.A.; Dorsey, P.J.; Hellstrom, W.J.G. The dangers of sexual enhancement supplements and counterfeit drugs to “treat” erectile dysfunction. Transl. Androl. Urol. 2017, 6, 12–19. [Google Scholar] [CrossRef]
- Venhuis, B.J.; De Voogt, P.; Emke, E.; Causanilles, A.; Keizers, P.H.J. Success of rogue online pharmacies: Sewage study of sildenafil in the Netherlands. Br. Med. J. 2014, 349, g4317. [Google Scholar] [CrossRef]
- Keizers, P.H.J.; Wiegard, A.; Venhuis, B.J. The quality of sildenafil active substance of illegal source. J. Pharm. Biomed. Anal. 2016, 131, 133–139. [Google Scholar] [CrossRef]
- Italian Medicine Agency. National Report on Medicines Use in Italy: Year 2018; Italian Medicine Agency: Rome, Italy, 2018. [Google Scholar]
- Dhaliwal, A.; Gupta, M. PDE5 Inhibitor. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2021; Available online: http://www.ncbi.nlm.nih.gov/books/nbk549843/ (accessed on 2 September 2021).
- Ghofrani, H.A.; Osterloh, I.H.; Grimminger, F. Sildenafil: From angina to erectile dysfunction to pulmonary hypertension and beyond. Nat. Rev. Drug Discov. 2006, 5, 689–702. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.A.; Lie, J.D. Phosphodiesterase-5 (PDE5) inhibitors in the management of erectile dysfunction. Pharm. Ther. 2013, 38, 407–419. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3776492/ (accessed on 2 September 2021).
- Nichols, D.J.; Muirhead, G.J.; Harness, J.A. Pharmacokinetics of sildenafil citrate after single oral doses in healthy male subjects: Absolute bioavailability, food effects and dose proportionality. Br. J. Clin. Pharmacol. Suppl. 2002, 53, 5S–12S. [Google Scholar] [CrossRef]
- Stark, S.; Sachse, R.; Liedl, T.; Hensen, J.; Rohde, G.; Wensing, G.; Horstmann, R.; Schrott, K.M. Vardenafil increases penile rigidity and tumescence in men with erectile dysfunction after a single oral dose. Eur. Urol. 2001, 40, 181–190. [Google Scholar] [CrossRef]
- Jung, J.; Choi, S.; Cho, S.H.; Ghim, J.L.; Hwang, A.; Kim, U.; Kim, B.S.; Koguchi, A.; Miyoshi, S.; Okabe, H.; et al. Tolerability and pharmacokinetics of avanafil, a phosphodiesterase type 5 inhibitor: A single- and multiple-dose, double-blind, randomised, placebo-controlled, dose-escalation study in healthy Korean male volunteers. Clin. Ther. 2010, 32, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Patterson, B.; Bedding, A.; Jewelll, H.; Payne, C.; Mitchell, M. The effect of intrinsic and extrinsic factors on the pharmacokinetic properties of tadalafil (IC351). Int. J. Impot. Res. 2001, 13, S62. [Google Scholar]
- Pfizer Labs. VIAGRA (Sildenafil Citrate); Pfizer: New York, NY, USA, 2010. [Google Scholar]
- Bayer HealthCare. LEVITRA (Vardenafil HCl); Bayer Pharmaceuticals Corporation: West Haven, CT, USA, 2014. [Google Scholar]
- Vivus. STENDRA (Avanafil); Vivus: Campbell, CA, USA, 2012. [Google Scholar]
- Shim, Y.S.; Pae, C.U.; Cho, K.J.; Kim, S.W.; Kim, J.C.; Koh, J.S. Effects of daily low-dose treatment with phosphodiesterase type 5 inhibitor on cognition, depression, somatisation and erectile function in patients with erectile dysfunction: A double-blind, placebo-controlled study. Int. J. Impot. Res. 2014, 26, 76–80. [Google Scholar] [CrossRef]
- Schnetzler, G.; Banks, I.; Kirby, M.; Zou, K.H.; Symonds, T. Characteristics, behaviors, and attitudes of men bypassing the healthcare system when obtaining phosphodiesterase type 5 inhibitors. J. Sex. Med. 2010, 7, 1237–1246. [Google Scholar] [CrossRef]
- Gaudiano, M.C.; Manna, L.; Rodomonte, A.L.; Bartolomei, M.; Bertocchi, P.; Gallinella, B.; Antoniella, E.; Muleri, N.; Civitelli, G.; Alimonti, S.; et al. A Survey on Illegal and Counterfeit Medicines for the Treatment of Erectile Dysfunctions in Italy. J. Sex. Med. 2012, 9, 2130–2137. [Google Scholar] [CrossRef]
- Mustazza, C.; Gaudiano, M.C.; Borioni, A.; Valvo, L. The evolution of the illegal market of falsified medicines and the experience of the Italian OMCL: From control to research. Ann. Dell’istituto Super. Sanita 2018, 54, 267–269. [Google Scholar] [CrossRef]
- European Union. Directive 2011/62/EU of the European Parliament and of the Council of 8 June 2011 Amending Directive 2001/83/EC on the Community Code Relating to Medicinal Products for Human Use; European Union: Brussels, Belgium, 2011. [Google Scholar]
- Kee, C.L.; Ge, X.; Gilard, V.; Malet-Martino, M.; Low, M.Y. A review of synthetic phosphodiesterase type 5 inhibitors (PDE-5i) found as adulterants in dietary supplements. J. Pharm. Biomed. Anal. 2018, 147, 250–277. [Google Scholar] [CrossRef]
- Causanilles, A.; Rojas Cantillano, D.; Emke, E.; Bade, R.; Baz-Lomba, J.A.; Castiglioni, S.; Castrignanò, E.; Gracia-Lor, E.; Hernández, F.; Kasprzyk-Hordern, B.; et al. comparison of phosphodiesterase type V inhibitors use in eight European cities through analysis of urban wastewater. Environ. Int. 2018, 115, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.F.; Gebhardt, W.; Thevis, M. Anabolic, doping, and lifestyle drugs, and selected metabolites in wastewater-detection, quantification, and behaviour monitored by high-resolution MS and MS n before and after sewage treatment. Anal. Bioanal. Chem. 2010, 398, 1207–1229. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, M.; Kosma, C.; Lambropoulou, D. Seasonal occurrence, removal, mass loading and environmental risk assessment of 55 pharmaceuticals and personal care products in a municipal wastewater treatment plant in Central Greece. Sci. Total Environ. 2016, 543, 547–569. [Google Scholar] [CrossRef] [PubMed]
- Temussi, F.; DellaGreca, M.; Pistillo, P.; Previtera, L.; Zarrelli, A.; Criscuolo, E.; Lavorgna, M.; Russo, C.; Isidori, M. Sildenafil and tadalafil in simulated chlorination conditions: Ecotoxicity of drugs and their derivatives. Sci. Total Environ. 2013, 463, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Eichhorn, P.; Pérez, S.; Aceña, J.; Gardinali, P.; Abad, J.L.; Barceló, D. Identification of phototransformation products of sildenafil (Viagra) and its N-demethylated human metabolite under simulated sunlight. J. Mass Spectrom. 2012, 47, 701–711. [Google Scholar] [CrossRef]
- Arditsoglou, A.; Voutsa, D. Partitioning of endocrine disrupting compounds in inland waters and wastewaters discharged into the coastal area of Thessaloniki, Northern Greece. Environ. Sci. Pollut. Res. 2010, 17, 529–538. [Google Scholar] [CrossRef]
- Fent, K.; Weston, A.A.; Caminada, D. Ecotoxicology of human pharmaceuticals. Aquat. Toxicol. 2006, 76, 122–159. [Google Scholar] [CrossRef]
- Kolpin, D.W.; Furlong, E.T.; Meyer, M.T.; Thurman, E.M.; Zaugg, S.D.; Barber, L.B.; Buxton, H.T. Pharmaceuticals, hormones, and other organic wastewater contaminants in US streams, 1999–2000: A national reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef]
- Stasinakis, A.S.; Gatidou, G.; Mamais, D.; Thomaidis, N.S.; Lekkas, T.D. Occurrence and fate of endocrine disrupters in Greek sewage treatment plants. Water Res. 2008, 42, 1796–1804. [Google Scholar] [CrossRef]
- Deblonde, T.; Cossu-Leguille, C.; Hartemann, P. Emerging pollutants in wastewater: A review of the literature. Int. J. Hyg. Environ. Health 2011, 214, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Wang, B.; Yin, L.; Zhang, Y.; Deng, S.; Huang, J.; Wang, Y.; Yu, G. Characterization of pharmaceutically active compounds in Beijing, China: Occurrence pattern, spatiotemporal distribution and its environmental implication. J. Hazard. Mater. 2017, 323, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Ferro-García, M.Á.; Prados-Joya, G.; Ocampo-Pérez, R. Pharmaceuticals as emerging contaminants and their removal from water. A review. Chemosphere 2013, 93, 1268–1287. [Google Scholar] [CrossRef]
- Coimbra, R.N.; Calisto, V.; Ferreira, C.I.A.; Esteves, V.I.; Otero, M. Removal of pharmaceuticals from municipal wastewater by adsorption onto pyrolysed pulp mill sludge. Arab. J. Chem. 2019, 12, 3611–3620. [Google Scholar] [CrossRef]
- Jaria, G.; Calisto, V.; Silva, C.P.; Gil, M.V.; Otero, M.; Esteves, V.I. Obtaining granular activated carbon from paper mill sludge—A challenge for application in the removal of pharmaceuticals from wastewater. Sci. Total Environ. 2019, 653, 393–400. [Google Scholar] [CrossRef]
- Husein, D.Z.; Hassanien, R.; Al-Hakkani, M.F. Green-synthesized copper nano-adsorbent for the removal of pharmaceutical pollutants from real wastewater samples. Heliyon 2019, 5, e02339. [Google Scholar] [CrossRef]
- Delgado, N.; Capparelli, A.; Navarro, A.; Marino, D. Pharmaceutical emerging pollutants removal from water using powdered activated carbon: Study of kinetics and adsorption equilibrium. J. Environ. Manag. 2019, 236, 301–308. [Google Scholar] [CrossRef]
- Oulton, R.L.; Kohn, T.; Cwiertny, D.M. Pharmaceuticals and personal care products in effluent matrices: A survey of transformation and removal during wastewater treatment and implications for wastewater management. J. Environ. Monit. 2010, 12, 1956–1978. [Google Scholar] [CrossRef]
- Doll, T.E.; Frimmel, F.H. Fate of pharmaceuticals—Photodegradation by simulated solar UV-light. Chemosphere 2003, 52, 1757–1769. [Google Scholar] [CrossRef]
- Escher, B.I.; Fenner, K. Recent advances in environmental risk assessment of transformation products. Environ. Sci. Technol. 2011, 45, 3835–3847. [Google Scholar] [CrossRef]
- De Rosa, M.; Ledia Triana Alonso, J. Studies of the Mechanism of Chlorination of Indoles. Detection of IV-Chloroindole and 3-Chloro-3H-indole as Intermediates. J. Org. Chem. 1978, 43, 2639–2643. [Google Scholar] [CrossRef]
- De Felice, B.; Argenziano, C.; Guida, M.; Trifuoggi, M.; Russo, F.; Condorelli, V.; Inglese, M. Molecular characterisation of microbial population dynamics during sildenafil citrate degradation. Mol. Biotechnol. 2009, 41, 123–132. [Google Scholar] [CrossRef]
- Grossberger, A.; Hadar, Y.; Borch, T.; Chefetz, B. Biodegradability of pharmaceutical compounds in agricultural soils irrigated with treated wastewater. Environ. Pollut. 2014, 185, 168–177. [Google Scholar] [CrossRef]
- Isidori, M.; Nardelli, A.; Pascarella, L.; Rubino, M.; Parrella, A. Toxic and genotoxic impact of fibrates and their photoproducts on non-target organisms. Environ. Int. 2007, 33, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Isidori, M.; Parrella, A.; Pistillo, P.; Temussi, F. Effects of ranitidine and its photoderivatives in the aquatic environment. Environ. Int. 2009, 35, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Vasquez, M.I.; Garcia-Käufer, M.; Hapeshi, E.; Menz, J.; Kostarelos, K.; Fatta-Kassinos, D.; Kümmerer, K. Chronic ecotoxic effects to Pseudomonas putida and Vibrio fischeri, and cytostatic and genotoxic effects to the hepatoma cell line (HepG2) of ofloxacin photo(cata)lytically treated solutions. Sci. Total Environ. 2013, 450, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Rocco, L.; Frenzilli, G.; Zito, G.; Archimandritis, A.; Peluso, C.; Stingo, V. Genotoxic effects in fish induced by pharmacological agents present in the sewage of some Italian water-treatment plants. Environ. Toxicol. 2012, 27, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Zanuri, N.B.M.; Bentley, M.G.; Caldwell, G.S. Assessing the impact of diclofenac, ibuprofen and sildenafil citrate (Viagra®) on the fertilisation biology of broadcast spawning marine invertebrates. Mar. Environ. Res. 2017, 127, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.H.; Vacquier, V.D. Cyclic GMP-specific phosphodiesterase-5 regulates motility of sea urchin spermatozoa. Mol. Biol. Cell 2006, 17, 114–121. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schnurstein, A.; Braunbeck, T. Tail moment versus tail length—Application of an in vitro version of the comet assay in biomonitoring for genotoxicity in native surface waters using primary hepatocytes and gill cells from zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2001, 49, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Diekmann, M.; Waldmann, P.; Schnurstein, A.; Grummt, T.; Braunbeck, T.; Nagel, R. On the relevance of genotoxicity for fish populations II: Genotoxic effects in zebrafish (Danio rerio) exposed to 4-nitroquinoline-1-oxide in a complete life-cycle test. Aquat. Toxicol. 2004, 68, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Kari, G.; Rodeck, U.; Dicker, A.P. Zebrafish: An emerging model system for human disease and drug discovery. Clin. Pharmacol. Ther. 2007, 82, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Hafez, E.M.; Shiekh, R.E.; Amin, A.S.; Gouda, A.A. Cloud point extraction of vardenafil HCl from pharmaceutical formulations prior to spectrophotometric determination. Int. J. Res. Pharm. Pharm. Sci. 2017, 2, 3–10. [Google Scholar]
- Reddy, T.V. Spectrophotometric quantification of vardenafil in bulk and tablet. Int. J. Biol. Chem. Sci. 2015, 3, 185–192. [Google Scholar]
- Sunil Kumar, A.V.V.N.K.; Reddy, T.V.; Sekaran, C.B. Spectrophotometric analysis of vardenafil in tablet dosage forms by using electrophilic coupling reagents. Anal. Bioanal. Chem. Res. 2016, 3, 29–39. [Google Scholar] [CrossRef]
- El Sheikh, R.; Gouda, A.A.; Abo, S.; Ezz, A.L. Utilitization of Charge Transfer Complexation Reaction for the Spectrophotometric Determination of Vardenafil HCl and Yohimbin HCl in Pharmaceutical Formulations. Chem. Sci. 2016, 5, 986–1000. [Google Scholar] [CrossRef][Green Version]
- Sakur, A.A.; Affas, S. Validated Spectrophotometric Method to Determine Vardenafil and Sildenafil in Pharmaceutical Forms Using Potassium Iodide and Potassium Iodate. Int. J. Pharm. Pharm. Sci. 2017, 9, 65–69. [Google Scholar] [CrossRef]
- Ahmed, N.R.; Ahmed, I.A.; Qasim, H.Y. Ultra-Sensitive Estimation of Vardenafil. Hcl in Pharmaceutical Preparations and Environmental Waste Water Samples. Eur. J. Biomed. Pharm. Sci. 2020, 7, 85–89. [Google Scholar]
- Idris, A.M.; Alnajjar, A.O. Multi-response optimisation of a capillary electrophoretic method for determination of vardenafil in the bulk drug and in a tablet formulation. Acta Chromatogr. 2007, 19, 97–109. [Google Scholar]
- Mohamed, S.K.; Shalaby, N.M. Microdetermination of sildenafil, tadalafil and vardenafil drugs employed in the erectile dysfunction therapy in pharmaceutical formulations and urine samples of diabetic patients type-II in taif area, saudia arabia using atomic emission and atomic absorp. Int. J. Pharma Bio Sci. 2013, 4, 1037–1046. [Google Scholar]
- Manisha, G.; Usha, P.; Vandana, P. Development and validation of RP-HPLC method for estimation of vardenafil in bulk and pharmaceutical formulation. Am. J. PharmTech Res. 2013, 3, 928–938. [Google Scholar]
- Di, Y.; Zhao, M.; Nie, Y.; Wang, F.; Lv, J. A High-Performance Liquid Chromatography: Chemiluminescence Method for Potential Determination of Vardenafil in Dietary Supplement. J. Autom. Methods Manag. Chem. 2011, 2011. [Google Scholar] [CrossRef][Green Version]
- Nickum, E.A.; Flurer, C.L. Determination of phosphodiesterase-5 inhibitors and analogs using high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. Sci. 2015, 53, 38–46. [Google Scholar] [CrossRef]
- Carlucci, G.; Palumbo, P.; Iuliani, P.; Palumbo, G. Development of a method for the determination of vardenafil in human plasma by high performance liquid chromatographywith UV detection. Biomed. Chromatogr. 2009, 23, 759–763. [Google Scholar] [CrossRef]
- Kumar, K.K.; Rao, C.K.; Rama, Y.; Reddy, K.; Mukkanti, K. A Validated Rapid Stability-Indicating Method for the Determination of Related Substances in Vardenafil Hydrochloride by Ultra-Performance Liquid Chromatography. Am. J. Anal. Chem. 2012, 3, 59–66. [Google Scholar] [CrossRef]
- Jeong, Y.; Suh, S.I.; Kim, J.Y.; In, M.K.; Paeng, K.J. Simultaneous Determination of Sildenafil, Tadalafil, and Vardenafil in Pharmaceutical Preparations by High-Temperature Gas Chromatography/Mass Spectrometry. Chromatographia 2016, 79, 1671–1678. [Google Scholar] [CrossRef]
- Mokhtar, S.U.; Chin, S.T.; Kee, C.L.; Low, M.Y.; Drummer, O.H.; Marriott, P.J. Rapid determination of sildenafil and its analogues in dietary supplements using gas chromatography-triple quadrupole mass spectrometry. J. Pharm. Biomed. Anal. 2016, 121, 188–196. [Google Scholar] [CrossRef]
- Man, C.N.; Nor, N.M.; Lajis, R.; Harn, G.L. Identification of sildenafil, tadalafil and vardenafil by gas chromatography-mass spectrometry on short capillary column. J. Chromatogr. A 2009, 1216, 8426–8430. [Google Scholar] [CrossRef]
- Saisho, K.; Scott, K.S.; Morimoto, S.; Nakahara, Y. Hair analysis for pharmaceutical drugs. II. Effective extraction and determination of sildenafil (Vigra®) and its N-desmethyl metabolite in rat and human hair by GC-MS. Biol. Pharm. Bull. 2001, 24, 1384–1388. [Google Scholar] [CrossRef][Green Version]
- Acquavia, M.A.; Foti, L.; Pascale, R.; Nicolò, A.; Brancaleone, V.; Cataldi, T.R.I.; Martelli, G.; Scrano, L.; Bianco, G. Detection and quantification of COVID-19 antiviral drugs in biological fluids and tissues. Talanta 2021, 224, 121862. [Google Scholar] [CrossRef] [PubMed]
- Baokar, S.; Pawar, V.; Patil, R.; Jagatap, R.; Ekatpure, N. Validation of simple and rapid UV-spectrophotometric method with stress degradation study for sildenafil citrate. Res. J. Pharm. Technol. 2012, 5, 214–218. [Google Scholar]
- Sakur, A.; Affas, S. Simple and Sensitive Spectrophotometric Determination of Sildenafil Citrate in Pharmaceutical Formulations. Res. J. Pharm. Technol. 2017, 10, 4242–4246. [Google Scholar] [CrossRef]
- Salem, H.; Abdel Aziz, B.E. Utility of Gold Nanoparticles for Spectrofluorimetric and Spectrophotometric Determination of Sildenafil Citrate, Dapoxetine, Vardenafil and Tadalafil in their Dosage Forms and Biological Fluids. Anal. Chem. Lett. 2020, 10, 321–335. [Google Scholar] [CrossRef]
- Fidan, A.K.; Bakirdere, S. Simultaneous determination of sildenafil and tadalafil in legal drugs, illicit/counterfeit drugs, and wastewater samples by high-performance liquid chromatography. J. AOAC Int. 2016, 99, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Fejos, I.; Neumajer, G.; Béni, S.; Jankovics, P. Qualitative and quantitative analysis of PDE-5 inhibitors in counterfeit medicines and dietary supplements by HPLC-UV using sildenafil as a sole reference. J. Pharm. Biomed. Anal. 2014, 98, 327–333. [Google Scholar] [CrossRef]
- Patel, D.N.; Li, L.; Kee, C.L.; Ge, X.; Low, M.Y.; Koh, H.L. Screening of synthetic PDE-5 inhibitors and their analogues as adulterants: Analytical techniques and challenges. J. Pharm. Biomed. Anal. 2014, 87, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Liigand, P.; Kaupmees, K.; Haav, K.; Liigand, J.; Leito, I.; Girod, M.; Antoine, R.; Kruve, A. Think Negative: Finding the Best Electrospray Ionization/MS Mode for Your Analyte. Anal. Chem. 2017, 89, 5665–5668. [Google Scholar] [CrossRef]
- Öztürk Er, E.; Özbek, B.; Bakırdere, S. A sensitive and selective analytical method for the simultaneous determination of sildenafil and tadalafil in water, energy drinks and sewage sludge matrices by LC-QTOF-MS/MS. Meas. J. Int. Meas. Confed. 2018, 124, 64–71. [Google Scholar] [CrossRef]
- Abdel-Hamid, M. Determination of sildenafil, tadalafil, and vardenafil in tablets and adulterated herbal products by ESI-MS-MS. J. Liq. Chromatogr. Relat. Technol. 2006, 29, 591–603. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, Z.; Ding, L.; Yan, H.; Wang, M.; Zhu, S. Simultaneous determination of yohimbine, sildenafil, vardenafil and tadalafil in dietary supplements using high-performance liquid chromatography-tandem mass spectrometry. J. Sep. Sci. 2010, 33, 2109–2114. [Google Scholar] [CrossRef]
- De Orsi, D.; Pellegrini, M.; Marchei, E.; Nebuloni, P.; Gallinella, B.; Scaravelli, G.; Martufi, A.; Gagliardi, L.; Pichini, S. High performance liquid chromatography-diode array and electrospray-mass spectrometry analysis of vardenafil, sildenafil, tadalafil, testosterone and local anesthetics in cosmetic creams sold on the Internet web sites. J. Pharm. Biomed. Anal. 2009, 50, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Öztürk Er, E.; Özbek, B.; Bakırdere, S. Accurate and sensitive determination of sildenafil, tadalafil, vardenafil, and avanafil in illicit erectile dysfunction medications and human urine by LC with quadrupole-TOF-MS/MS and their behaviors in simulated gastric conditions. J. Sep. Sci. 2019, 42, 475–483. [Google Scholar] [CrossRef]
- Baker, D.R.; Kasprzyk-Hordern, B. Critical evaluation of methodology commonly used in sample collection, storage and preparation for the analysis of pharmaceuticals and illicit drugs in surface water and wastewater by solid phase extraction and liquid chromatography-mass spectrometry. J. Chromatogr. A 2011, 1218, 8036–8059. [Google Scholar] [CrossRef]
- 1Baker, D.R.; Barron, L.; Kasprzyk-Hordern, B. Illicit and pharmaceutical drug consumption estimated via wastewater analysis. Part A: Chemical analysis and drug use estimates. Sci. Total Environ. 2014, 487, 629–641. [Google Scholar] [CrossRef]
- Boleda, M.R.; Galceran, M.T.; Ventura, F. Validation and uncertainty estimation of a multiresidue method for pharmaceuticals in surface and treated waters by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2013, 1286, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Aceña, J.; Pérez, S.; Gardinali, P.; Abad, J.L.; Eichhorn, P.; Heuett, N.; Barceló, D. Structure elucidation of phototransformation products of unapproved analogs of the erectile dysfunction drug sildenafil in artificial freshwater with UPLC-Q Exactive-MS. J. Mass Spectrom. 2014, 49, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Herbert, L.P.; Becker-Krail, D.B.; Cory, W.C. Persistent phototransformation products of vardenafil (Levitra®) and sildenafil (Viagra®). Chemosphere 2015, 134, 557–562. [Google Scholar] [CrossRef]
- Causanilles, A.; Emke, E.; De Voogt, P. Determination of phosphodiesterase type V inhibitors in wastewater by direct injection followed by liquid chromatography coupled to tandem mass spectrometry. Sci. Total Environ. 2016, 565, 140–147. [Google Scholar] [CrossRef]
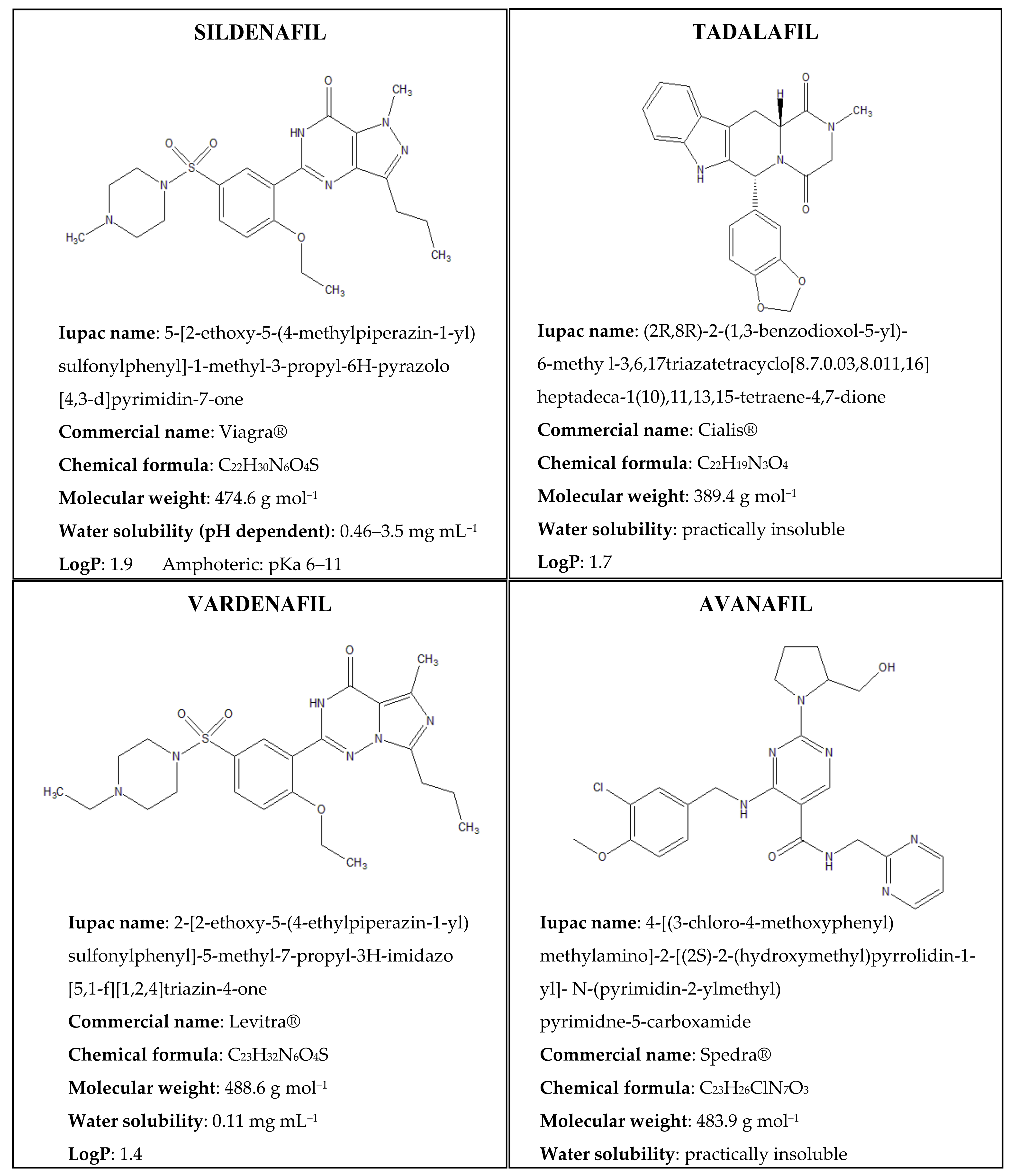
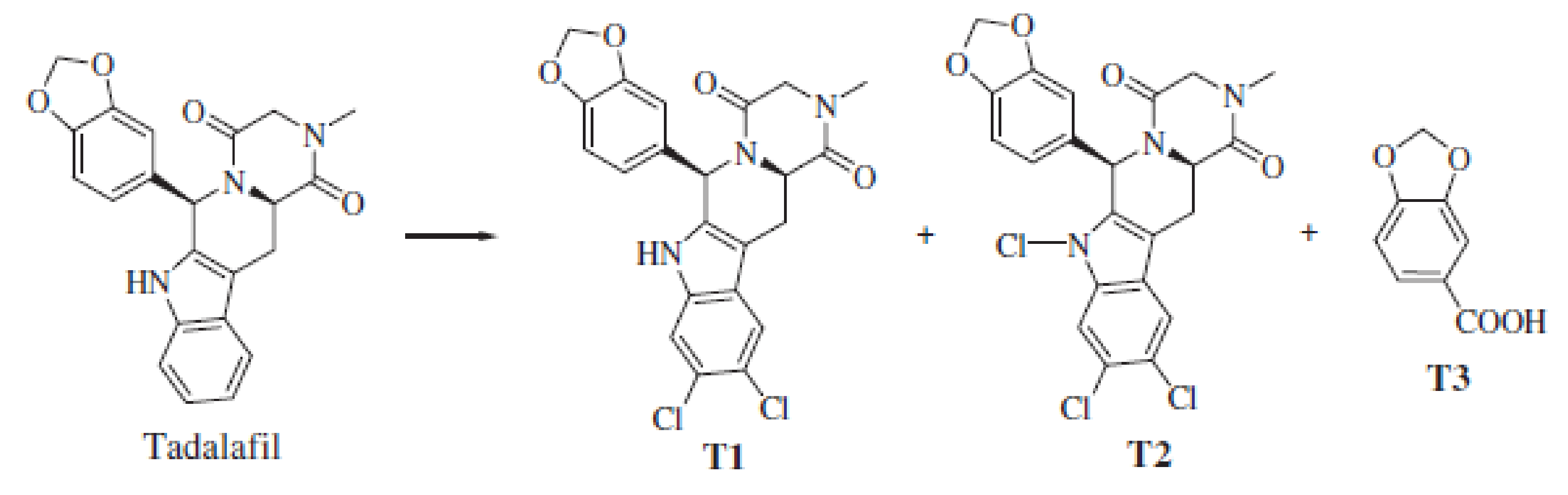
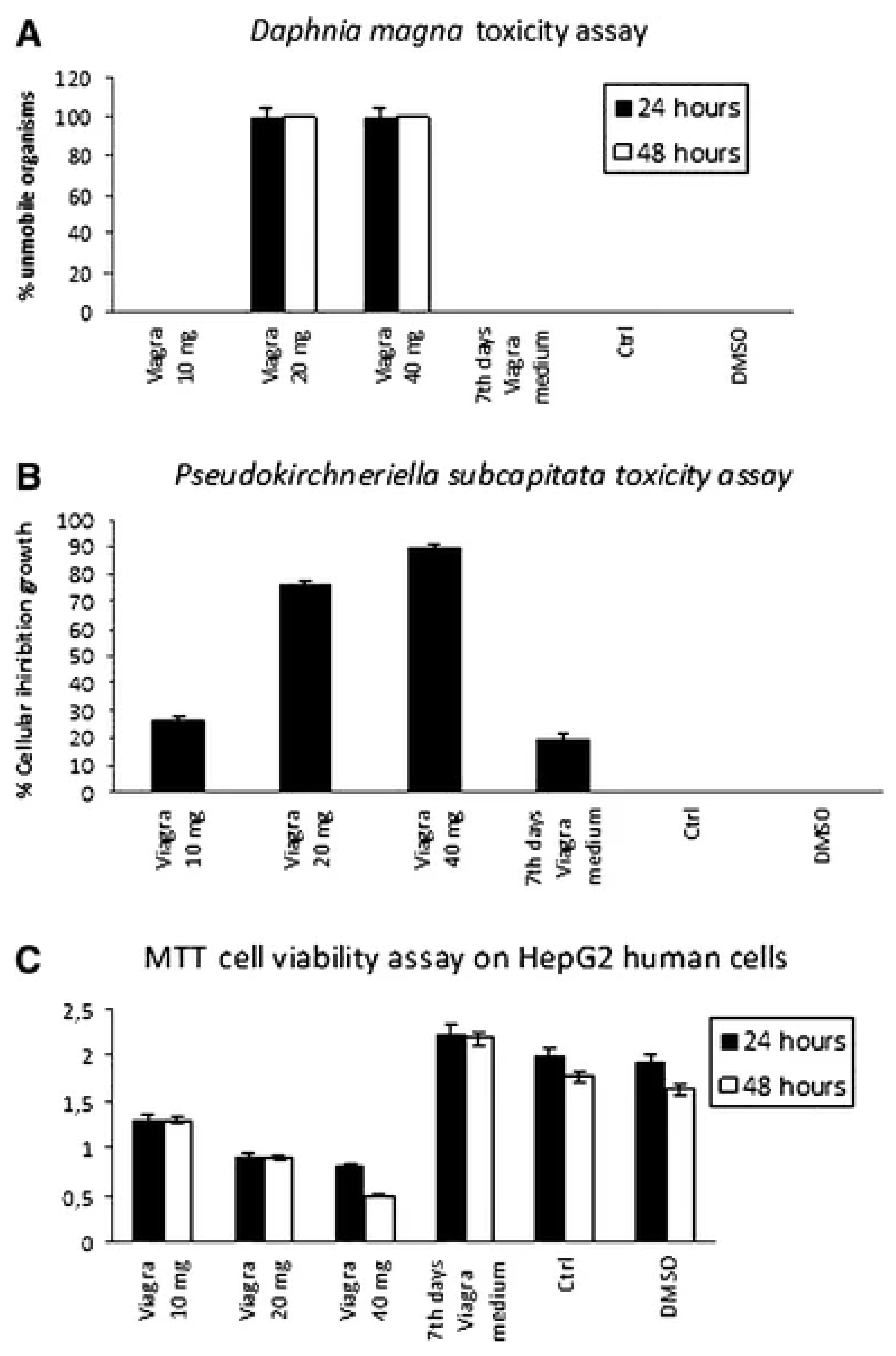
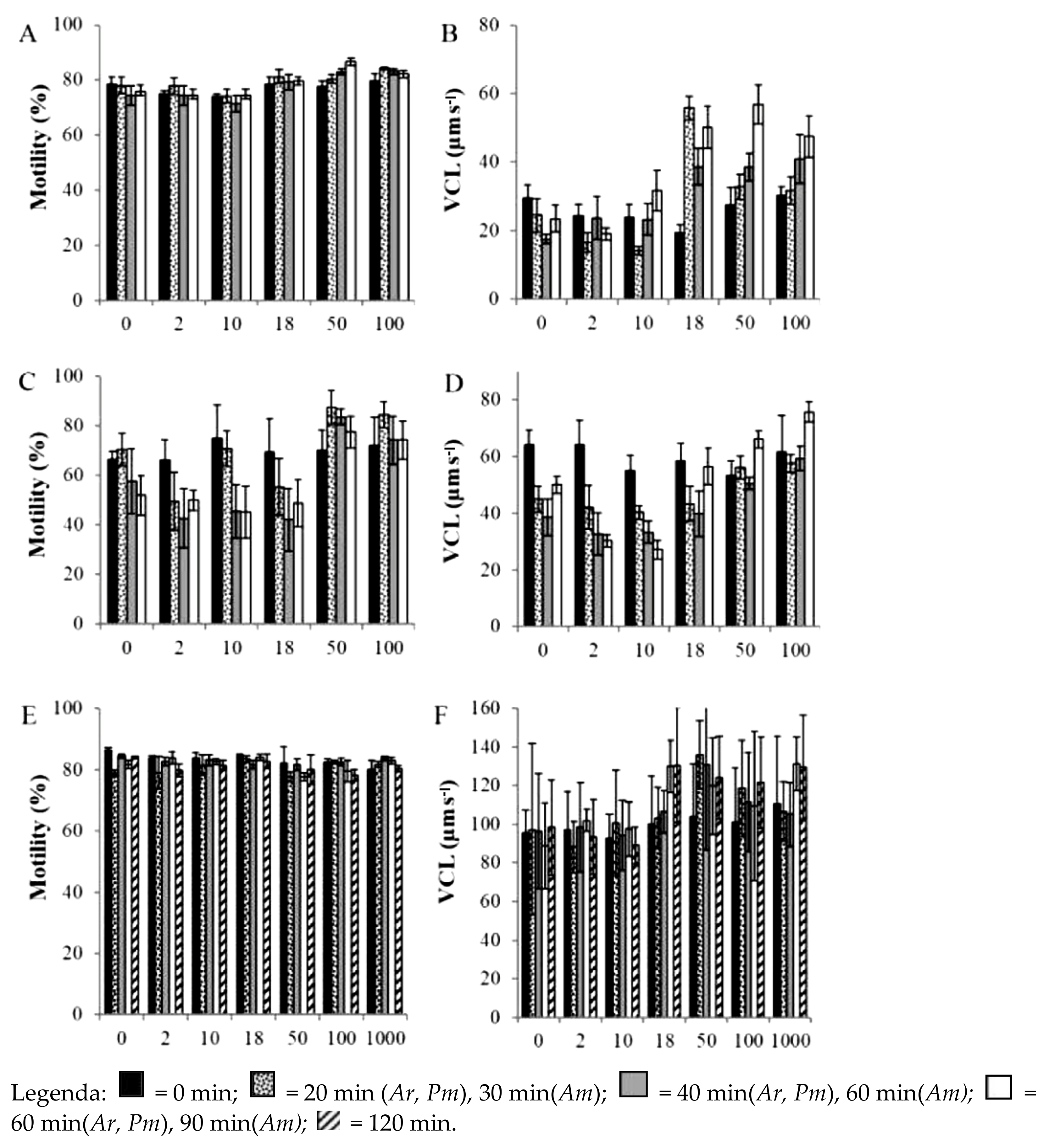
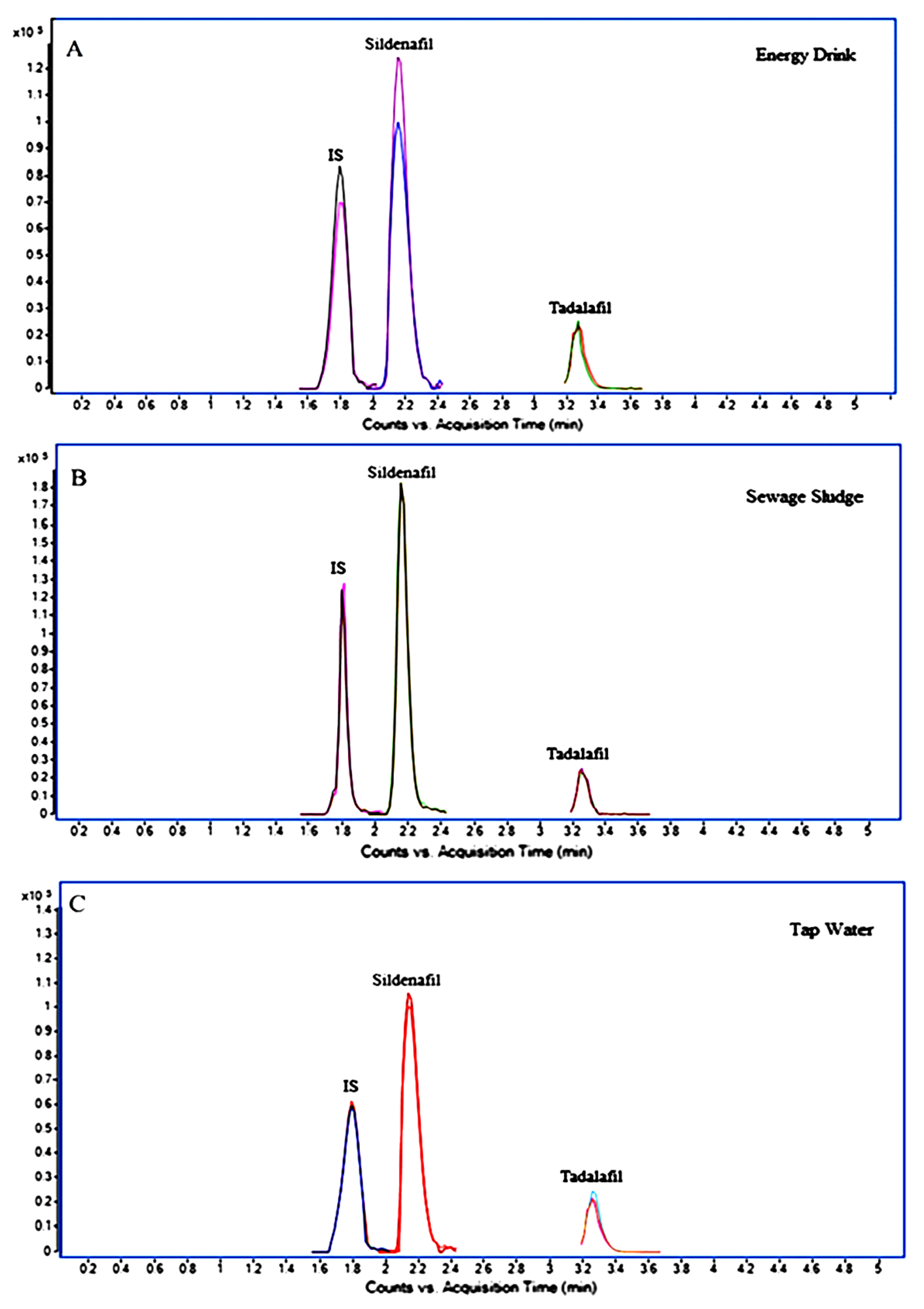
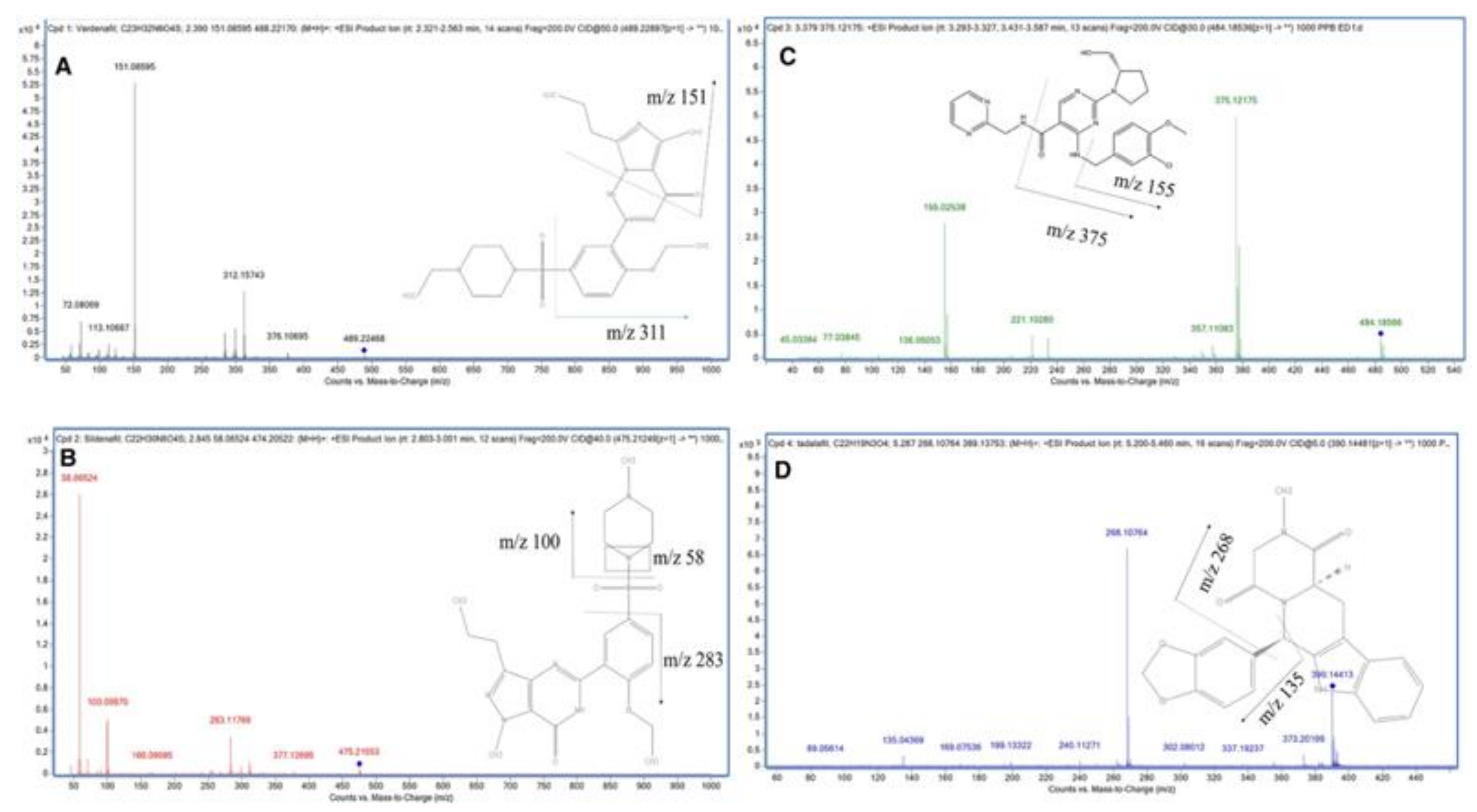
| Parameters/Drugs | Sildenafil (Viagra) | Vardenafil (Levitra) | Tadalafil (Cialis) | Avanafil (Spedra) |
|---|---|---|---|---|
| Bioavailability | 41% (mean) 25–63% (range) | 15% (mean) | - | - |
| Tmax | 1 h (median) 0.5–2 h (range) | 1 h (median) 0.5–2 h (range) | 2 h (median) 0.5–6 h (range) | 0.5–0.75 h (range) |
| Protein binding | 96% | 95% | 94% | 99% |
| Metabolism | Major: CYP3A4 Minor: CYP2C9 | Major: CYP3A4 Minor: CYP3A5, CYP2C | CYP3A4 | Major: CYP3A4 Minor: CYP2C |
| Active metabolite(% effect) | Yes (20%) N-demethylation | Yes (7%) Demethylation | No | Yes (4%) Methylation, glucuronidation |
| Half-life(T1/2) | 4 h | 4–5 h | 17.5 h | 5 h |
| Elimination | 80% faeces 13% urine | 91–95% faeces 2–6% urine | 61% faeces 36% urine | 62% faeces 21% urine |
| Ingestion with high-fat meals | ↓ Cmax 29% ↑ Tmax by 1 h | ↓ Cmax 18–50% | Not affected | ↓ Cmax 24–39% ↑ Tmax by 1.12–1.25 h |
| Additional PDE inhibition | PDE1, PDE6 | PDE1, PDE6 | PDE11 | - |
| B. calyciflorus | C. dubia | |
|---|---|---|
| Sildenafil citrate | 42.74 (34.32–53.21) | 5.74 (3.08–10.71) |
| S1 | 19.82 (17.51–22.43) | 6.60 (5.73–7.60) |
| Tadalafil | NE up to 20 | NE up to 20 |
| T1 | NE up to 20 | NE up to 20 |
| Compounds | Ames Test | |||||
|---|---|---|---|---|---|---|
| TA98 | TA100 | |||||
| Concentration (μg mL−1) | Mean Revertants/Plate (±SD) | MR a | Concentration (μg mL−1) | Mean Revertants/Plate (±SD) | MR a | |
| Negative Control2-Nitrofluoren | - | 29.6 ± 2.7 | - | - - | 190.1 ± 47.2 - | - - |
| 2.5 | 61.0 ± 5.7 | 2.1 | ||||
| 5 | 83.2 ± 11.3 | 2.8 | ||||
| 10 | 178.0 ± 15.0 | 6 | ||||
| Sodium azide | - | - | - | 5 | 488.0 ± 96.9 | 2.6 |
| 10 | 794.7 ± 90.9 | 4.2 | ||||
| 20 | 1153.3 ± 239.8 | 6.1 | ||||
| Sildenafil citrate | 0.625 | 46.7 ± 13.1 | 1.6 | 0.625 | 161.0 ± 18.7 | 0.8 |
| 1.25 | 48.7 ± 5.1 | 1.6 | 1.25 | 164.6 ± 8.7 | 0.9 | |
| 2.5 | 72.0 ± 11.3 | 2.4 | 2.5 | 190.6 ± 12.6 | 1 | |
| 5 | 93.0 ± 4.4 | 3.1 | 5 | 198.3 ± 24.0 | 1 | |
| 10 | 170.7 ± 32.0 | 5.8 | 10 | 220.0 ± 46.8 | 1.2 | |
| S1 | 0.3125 | 27.8 ± 3.2 | 0.9 | 0.3125 | 241.1 ± 27.5 | 1.3 |
| 0.625 | 37.6 ± 4.7 | 1.3 | 0.625 | 291.1 ± 21.6 | 1.5 | |
| 1.25 | 37.3 ± 11.6 | 1.3 | 1.25 | 303.1 ± 44.7 | 1.6 | |
| 2.5 | 53.6 ± 9.0 | 1.8 | 2.5 | 376.6 ± 45.3 | 2 | |
| 5 | 64.7 ± 20.2 | 2.2 | 5 | 405.1 ± 82.5 | 2.1 | |
| 10 | 112.4 ± 41.5 | 3.8 | 10 | 444.2 ± 83.2 | 2.3 | |
| Tadalafil | 0.3125 | 49.5 ± 7.7 | 1.7 | 0.3125 | 298.5 ± 38.7 | 1.6 |
| 0.625 | 67.2 ± 12.2 | 2.3 | 0.625 | 318.6 ± 65.2 | 1.7 | |
| 1.25 | 149.3 ± 33.2 | 5 | 1.25 | 325.6 ± 94.4 | 1.7 | |
| 2.5 | 223.0 ± 54.8 | 7.5 | 2.5 | 425.3 ± 80.9 | 2.2 | |
| 5 | 337.3 ± 32.3 | 11.4 | 5 | 556.0 ± 41.7 | 2.9 | |
| 10 | 544.0 ± 92.1 | 18.4 | 10 | 880.0 ± 237.6 | 4.6 | |
| T1 | 0.625 | 43.6 ± 11.9 | 1.5 | 0.625 | 286.0 ± 14.1 | 1.5 |
| 1.25 | 43.5 ± 14.0 | 1.5 | 1.25 | 310.7 ± 43.9 | 1.6 | |
| 2.5 | 85.4 ± 19.2 | 2.9 | 2.5 | 322.7 ± 28.4 | 1.7 | |
| 5 | 221.8 ± 57.2 | 7.5 | 5 | 288.0 ± 40.6 | 1.5 | |
| 10 | 391.2 ± 69.4 | 13.2 | 10 | 410.7 ± 70.0 | 2.2 | |
| Species | Test | Pharmaceutical | NOEC or LOEC (µg L−1) | EC50 (µg L−1) | Classification |
|---|---|---|---|---|---|
| Asterias rubens | Sperm motility | Sildenafil citrate | NOEC = 0.18 | 60 min = 2.25 × 1012 | Non-toxic |
| Fertilisation: sperm pre-incubation | Sildenafil citrate | NOEC = 0.010 | 60 min = 7.15 × 1013 | Non-toxic | |
| Fertilisation: oocyte pre-incubation | Sildenafil citrate | NOEC = 0.10 | N/A | N/A | |
| Fertilisation: sperm and oocyte pre-incubation | Sildenafil citrate | NOEC = 0.01 | 60 min = 2.37 × 1012 | Non-toxic | |
| Psammechinus miliaris | Sperm motility | Diclofenac Ibuprofen Sildenafil citrate | NOEC = 0.01 NOEC = 0.1 NOEC = 0.018 | 60 min = 378.22 60 min = 845.98 60 min = 7.23 × 1010 | Very toxic Very toxic Non-toxic |
| Fertilisation: sperm pre-incubation | Sildenafil citrate | NOEC = 0.10 | 60 min = 6.241 × 1010 | Non-toxic | |
| Fertilisation: oocyte pre-incubation | Sildenafil citrate | NOEC = 0.01 | N/A | N/A | |
| Fertilisation: sperm and oocyte pre-incubation | Diclofenac Ibuprofen Sildenafil citrate | LOEC = 0.01 NOEC = 0.10 NOEC = 1.0 | 60 min = 247.31 60 min = 792.96 N/A | Very toxic Very toxic N/A | |
| Arenicola marina | Sperm motility | Sildenafil citrate | NOEC = 1.0 | N/A | N/A |
| Fertilisation: sperm pre-incubation | Diclofenac Ibuprofen Sildenafil citrate | NOEC = 1.00 NOEC = 0.10 NOEC = 1.0 | 120 min = 565.53 120 min = 3.24 × 109N/A | Very toxic Non toxic N/A | |
| Fertilisation: oocyte pre-incubation | Sildenafil citrate | NOEC = 1.00 | N/A | N/A | |
| Fertilisation: sperm and oocyte pre-incubation | Sildenafil citrate | NOEC = 1.0 | N/A | N/A |
| Pharmaceutical Compound | Mean Concentration ng L−1 | Number of Specimens Examined | Days of Treatment | Tail DNA Mean % ± SD | p-Value Tail DNA | Apoptotic Cells Mean % ± SD | p-Value Apoptotic Cells | ||
|---|---|---|---|---|---|---|---|---|---|
| NT | T | NT | T | ||||||
| Atorvastatin | 13 | 20 | 5 | 20.41 ± 4.44 | 50.47± 9.19 | 0.003 * | 1.0 ± 0.21 | 6.5 ± 1.85 | 0.003 * |
| 7 | 27.68 ± 13.49 | 40.81± 16.34 | 0.535 | 1.5 ± 0.31 | 5.7 ± 1.78 | 0.020 * | |||
| 14 | 19.96 ± 4.99 | 31.05 ± 13.03 | 0.426 | 1.2 ± 0.34 | 4.3 ± 1.86 | 0.101 | |||
| Gemfibrozil | 380 | 20 | 5 | 25.76 ± 12.94 | 55.12 ± 7.42 | 0.045 * | 6.9 ±0.22 | 8.6 ± 0.68 | 0.017 * |
| 7 | 27.24 ± 11.74 | 60.67 ± 11.78 | 0.044 * | 5.7 ± 0.84 | 9.6 ± 1.34 | 0.013 * | |||
| 14 | 12.45 ± 2.32 | 18.47 ± 2.78 | 0.096 | 4.5 ± 0.94 | 5.7 ± 0.75 | 0.298 | |||
| Sildenafil citrate | 26.25 | 20 | 5 | 20.41 ± 4.44 | 25.79 ± 10.13 | 0.627 | 1.0 ±0.28 | 1.8 ± 0.42 | 0.113 |
| 7 | 27.68 ± 13.49 | 34.00 ± 8.19 | 0.688 | 1.5 ± 0.25 | 2.5 ± 1.45 | 0.497 | |||
| 14 | 19.96 ± 4.99 | 29.93 ± 18.02 | 0.593 | 1.2 ± 0.21 | 2 ± 0.56 | 0.181 | |||
| 22 | 21 | 36.56 ± 13.33 | 39.29 ± 14.78 | 0.890 | 4.9 ± 0.25 | 5.3 ± 0.21 | 0.220 | ||
| 28 | 15.87 ± 5.08 | 12.99 ± 4.25 | 0.663 | 1.4 ± 0.25 | 1.1 ± 0.56 | 0.625 | |||
| 21 | 35 | 15.36 ± 4.86 | 49.15 ± 16.67 | 0.051 * | 1.3 ± 0.23 | 7.6 ±2.95 | 0.033 * | ||
| Compound/Photoproduct | Measured Ion Mass of [M + H]+ | Elemental Composition | Theoretical Ion Mass (m/z) | Relative Error (ppm) (DBE) |
|---|---|---|---|---|
| SDF | 475.2128 | C22H31N6O4S | 475.2132 | +1.9 (10.5) |
| 377.1285 | C17H21N4O4S | 377.1279 | +1.6 (9.5) | |
| 311.1514 | C17H19N4O2 | 311.1503 | +3.5 (10.5) | |
| 299.1153 | C15H15N4O3 | 299.1139 | +4.7 (10.5) | |
| 283.1183 | C15H15N4O2 | 283.119 | −2.5 (10.5) | |
| 100.1033 | C5H12N2 | 100.0995 | +38 (1.0) | |
| 99.0938 | C5H11N2 | 99.0917 | +21.2 (1.5) | |
| TP490 | 491.2089 | C22H31N6O5S | 491.2072 | +3.5 (10.5) |
| 473.1948 | C22H29N6O4S | 473.1966 | −3.8 (11.5) | |
| 404.1382 | C18H22N5O4S | 404.1387 | −1.2 (10.5) | |
| 377.1285 | C17H21N4O4S | 377.1279 | +1.6 (9.5) | |
| 311.1518 | C17H19N4O2 | 311.1503 | +4.8 (10.5) | |
| 299.1152 | C15H15N4O3 | 299.1139 | +4.3 (10.5) | |
| 283.12.5 | C15H15N4O2 | 283.1190 | +4.6 (10.5) | |
| 100.1012 | C5H12N2 | 100.0995 | +17 (1.0) | |
| 99.0940 | C5H11N2 | 99.0917 | +28.3 (1.5) | |
| TP488-C | 489.1952 | C22H29N6O5S | 489.191 | +4.5 (11.5) |
| 461.1997 | C21H29N6O4S | 461.1966 | +6.7 (10.5) | |
| 377.1296 | C17H21N4O4S | 377.1279 | +4.5 (9.5) | |
| 311.1511 | C17H19N4O2 | 311.1503 | +2.6 (10.5) | |
| 299.1146 | C15H15N4O3 | 299.1139 | +2.3 (10.5) | |
| 283.1183 | C15H15N4O2 | 283.1190 | −2.5 (10.5) | |
| 85.0727 | C4H9N2 | 85.0760 | −38.8 (1.5) | |
| TP462 | 463.1777 | C20H27N6O5S | 463.1758 | +4.1 (10.5) |
| 435.1836 | C19H27N6O4S | 435.1809 | +4.1 (9.5) | |
| 418.1517 | C19H24N5O4S | 418.1544 | −6.5 (10.5) | |
| 311.153 | C17H19N4O2 | 311.1503 | +8.7 (10.5) | |
| 299.1155 | C15H15N4O3 | 299.1139 | +5.3 (10.5) | |
| 283.1178 | C15H15N4O2 | 283.1190 | −4.2 (10.5) | |
| TP460 | 461.1986 | C21H29N6O4S | 461.1966 | +4.3 (10.5) |
| 377.1285 | C17H21N4O4S | 377.1279 | +1.6 (9.5) | |
| 311.1511 | C17H19N4O2 | 311.1503 | +2.6 (10.5) | |
| 283.1185 | C15H18N4O2 | 283.119 | −1.8 (10.5) | |
| 85.0769 | C4H9N2 | 85.0760 | +9.4 (1.5) | |
| TP448-A | 449.2163 | C20H29N6O4S | 449.1966 | +5.1 (9.5) |
| 418.1529 | C19H24N5O4S | 418.1544 | −3.6 (10.5) | |
| 392.1363 | C19H24N5O4S | 392.1387 | −6.1 (9.5) | |
| 377.129 | C17H21N4O4S | 377.1279 | +2.9 (9.5) | |
| 311.1489 | C17H19N4O2 | 311.1503 | −4.5 (10.5) | |
| 299.1151 | C15H15N4O3 | 299.1139 | +4.0 (10.5) | |
| 283.1203 | C15H15N4O2 | 283.1190 | +4.6 (10.5) | |
| TP434 | 435.1834 | C19H27N6O4S | 435.1814 | +5.7 (9.5) |
| 418.1567 | C19H24N5O4S | 418.1544 | +5.5 (10.5) | |
| 392.1451 | C17H22N5O4S | 392.1387 | +5.6 (9.5) | |
| 377.1245 | C17H21N4O4S | 377.1279 | −9.0 (9.5) | |
| 311.1516 | C17H19N4O2 | 311.1503 | +4.2 (10.5) | |
| 299.1159 | C15H15N4O3 | 299.1139 | +6.7 (10.5) | |
| 283.1205 | C15H15N4O2 | 283.1190 | +5.3 (10.5) | |
| TP392 | 393.1234 | C17H21N4O5S | 393.1227 | +1.5 (9.5) |
| 365.0903 | C15H17N4O5S | 365.0914 | −3.0 (9.5) | |
| 336.0637 | C14H14N3O5S | 336.0649 | −3.6 (9.5) | |
| 284.1279 | C15H16N4O2 | 284.1268 | 3.9 (10.0) | |
| 256.1070 | C14H14N3O2 | 256.1081 | −4.3 (9.5) | |
| TP391 | 392.1365 | C17H22N5O4S | 392.1387 | −5.6 (9.5) |
| 364.1056 | C15H18N5O4S | 364.1079 | −6.3 (9.5) | |
| 299.1157 | C15H15N4O3 | 299.1139 | +6.0 (10.5) | |
| 283.1169 | C15H15N4O2 | 283.1190 | −7.4 (10.5) |
| Compound/Photoproduct | Measured Ion Mass of [M + H]+ | Elemental Composition | Theoretical Ion Mass (m/z) | Relative Error (ppm) |
|---|---|---|---|---|
| DM-SDF | 461.1978 | C21H29N6O4S C17H21N4O4S C17H19N4O2 | 461.1966 | +2.6 (10.5) |
| 377.1270 | C15H15N4O3 | 377.1279 | −2.4 (9.5) | |
| 311.1518 | C15H15N4O2 | 311.1503 | +4.8 (10.5) | |
| 299.1155 | C4H9N2 | 299.1139 | +5.3 (10.5) | |
| 283.1203 | 283.1190 | +4.6 (10.5) | ||
| 85.0803 | 85.0760 | −11.8 (1.5) | ||
| TP476 | 477.1959 | C21H29N6O5S | 477.1915 | +9.2 (10.5) |
| 404.1404 | C18H22N5O4S | 404.1387 | +7.9 (10.5) | |
| 377.1295 | C17H21N4O4S | 377.1278 | +4.5 (9.5) | |
| 331.0836 | C15H15N4O3S | 331.0865 | −5.7 (10.5) | |
| 312.1597 | C17H20N4O2 | 312.1581 | +5.1 (10.0) | |
| 283.1205 | C15H15N4O2 | 283.1190 | +4.6 (10.5) | |
| 83.0628 | C4H7N2 | 83.0604 | +28.9 (2.5) | |
| TP462 | 463.1777 | C20H27N6O5S | 463.1758 | +4.1 (10.5) |
| 435.1839 | C19H27N6O4S | 435.1809 | +6.9 (9.5) | |
| 418.1569 | C19H24N5O4S | 418.1544 | +6.0 (10.5) | |
| 311.1520 | C17H19N4O2 | 311.1503 | +5.5 (10.5) | |
| 299.1122 | C15H15N4O3 | 299.1139 | −5.7 (10.5) | |
| 283.1175 | C15H15N4O2 | 283.1190 | −5.3 (10.5) | |
| TP448-B | 449.162 | C19H25N6O5S | 449.1602 | +4.0 (10.5) |
| 431.1507 | C19H23N6O4S | 431.1496 | +2.6 (11.5) | |
| 404.1393 | C18H22N5O4S | 404.1387 | +0.2 (10.5) | |
| 311.1519 | C17H19N4O2 | 311.1503 | +5.1 (10.5) | |
| 299.1122 | C15H15N4O3 | 299.1139 | −5.7 (10.5) | |
| 283.1206 | C15H15N4O2 | 283.1190 | +5.7 (10.5) | |
| TP434 | 435.1801 | C19H25N6O5S | 435.181 | −2.1 (9.5) |
| 418.1539 | C19H22N5O4S | 418.1544 | −1.2 (10.5) | |
| 392.1353 | C17H22N5O4S | 392.1387 | −8.7 (9.5) | |
| 377.1281 | C17H21N4O4S | 377.1279 | +0.5 (9.5) | |
| 311.1514 | C17H19N4O2 | 311.1503 | +3.5 (10.5) | |
| 299.1126 | C15H15N4O3 | 299.1139 | −4.3 (10.5) | |
| 283.1197 | C15H15N4O2 | 283.1190 | +2.5 (10.5) | |
| TP392 | 393.1248 | C17H21N4O5S | 393.1227 | +5.3 (9.5) |
| 365.0922 | C15H17N4O5S | 365.0914 | +2.2 (9.5) | |
| 365.0922 | C15H17N4O5S | 365.0914 | +2.2 (9.5) | |
| 336.0627 | C14H14N3O5S | 336.0649 | −6.5 (9.5) | |
| 284.1290 | C15H16N4O2 | 284.1268 | +7.7 (10.0) | |
| 256.1060 | C14H14N3O2 | 256.1081 | −8.2 (9.5) | |
| TP391 | 392.1405 | C17H22N5O4S | 392.1387 | +3.1 (9.5) |
| 364.1105 | C15H18N5O4S | 364.1079 | −0.5 (9.5) | |
| 299.1158 | C15H15N4O3 | 299.1139 | +6.4 (10.5) | |
| 283.1172 | C15H15N4O2 | 283.1190 | −6.4 (10.5) |
| VRD Phototransformation (PT) Products | SLD Phototransformation (PT) Products | ||||||
|---|---|---|---|---|---|---|---|
| Rt(m) | m/z (Da) | Proposed Structure | Name | Rt(m) | m/z (Da) | Proposed Structure | Name |
| 2.8 | 393 | 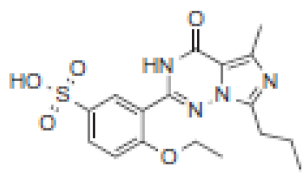 | VRD-392 | 3.8 | 393 | 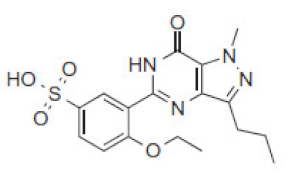 | SLD-392 |
| 5.3 | 392 | 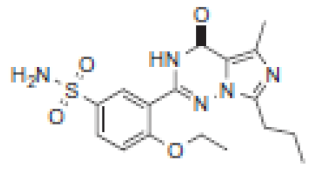 | VRD-391 | 9.6 | 392 | 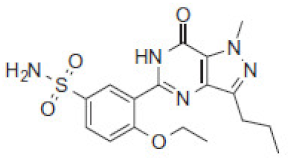 | SLD-391 |
| 5.6 | 463 | 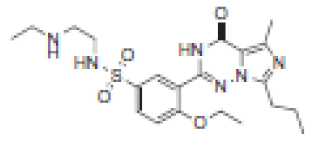 | VRD-462 | 6.9 | 449 | 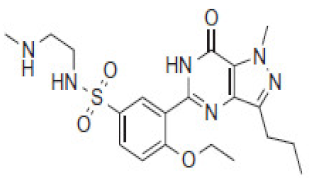 | SLD-448 |
| 435 | ND | 6.6 | 435 | 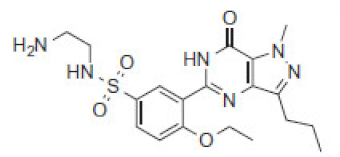 | SLD-434 | ||
| 6.7 | 461 | 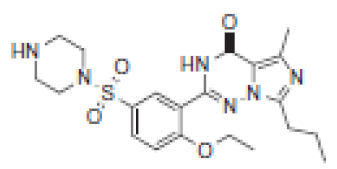 | VRD-460 | 461 |  | SLD-460 | |
| 6.8 | 489 | 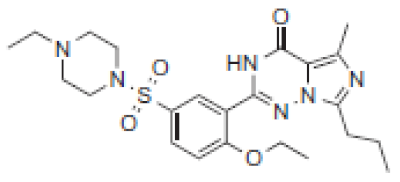 | VRD | 475 |  | SLD | |
| (463) | ND | 463 | 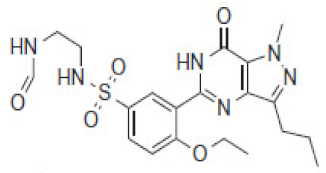 | SLD-462 | |||
| 7.1 | 505 | 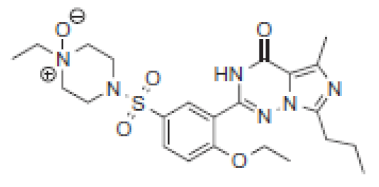 | VRD-504 | 491 | 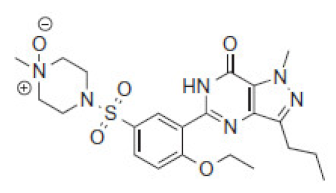 | SLD-490 | |
| 8.4 | 491 | 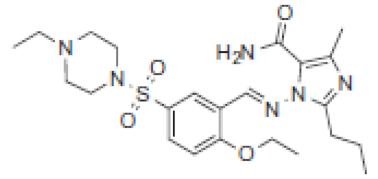 | VRD-490 | 477 | 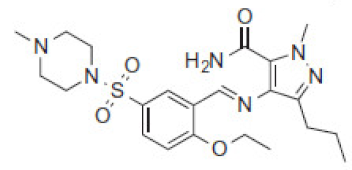 | SLD-476 | |
| Investigated Compound | Sample Matrix | Sample Preparation | Detection Method | LOD/LOQ | Reference |
|---|---|---|---|---|---|
| Sildenafil, Tadalafil, Vardenafil | Influent wastewater, effluent wastewater and sewage sludge samples | Wastewater samples Solid-phase extraction (SPE) Sewage sludge samples Pressurised liquid extraction (PLE) | HPLC-MS/MS | Sildenafil Influent wastewater LOD: 1 ng L−1 LOQ: 2.5 ng L−1 Effluent wastewater LOD: 1 ng L−1 LOQ: 1 ng L−1 Sewage sludge LOD: 1 ng g−1 LOQ: 3 ng g−1 Tadalafil Influent wastewater LOD: 5 ng L−1 LOQ: 6 ng L−1 Effluent wastewater LOD: 1.5 ng L−1 LOQ: 2.5 ng L−1 Sewage sludge LOD: 3 ng g−1 LOQ: 5 ng g−1 Vardenafil Influent wastewater LOD: 2 ng L−1 LOQ: 4 ng L−1 Effluent wastewater LOD: 1 ng L−1 LOQ: 1.5 ng L−1 Sewage sludge LOD: 2 ng g−1 LOQ: 5 ng g−1 | [15] |
| Sildenafil, Tadalafil, Vardenafil | Wastewater samples | Homogenisation, spiking with deuterated analogue and filtration with 0.2 µm syringe filter | UPLC-MS/MS | Sildenafil LOD: 2 ng L−1 LOQ: 6 ng L−1 Tadalafil LOD: 2 ng L−1 LOQ: 8 ng L−1 Vardenafil LOD: 7 ng L−1 LOQ: 24 ng L−1 | [44] |
| Sildenafil, Tadalafil, Vardenafil and their metabolites | Spiked urine and wastewater samples | Solid-phase extraction (SPE) | HPLC-(HR)-MS/MS | Sildenafil Spiked urine LLOD: <2 ng L−1 LLOQ: 2 ng L−1 Spiked wastewater LLOD: <1 ng L−1 LLOQ: 2 ng L−1 Tadalafil Spiked urine LLOD: <2 ng L−1 LLOQ: 2 ng L−1 Spiked wastewater LLOD: <1 ng L−1 LLOQ: 2 ng L−1 Vardenafil Spiked urine LLOD: <2 ng L−1 LLOQ: 2 ng L−1 Spiked wastewater LLOD: <1 ng L−1 LLOQ: 2 ng L−1 | [45] |
| Sildenafil, N-desmethyl sildenafil and their photo-transformation products | Ultrapure water, artificial freshwater, natural river water | Irradiation of tests solution in the 3 matrices, with an initial concentration of 10 mg L−1 | UPLC-ESI-QToF-MS | Not reported | [48] |
| Sildenafil and Tadalafil | Standard solutions, tap water, energy drinks, sewage sludge | Energy drink Direct injection Tap water and sewage sludge Dilution with 50/50 (v/v) ACN/water and filtration with a 0.45 µm syringe filter | LC-QTOF-MS/MS | Sildenafil LOD: 0.16 ng g−1 LOQ: 0.53 ng g−1 Tadalafil LOD: 0.28 ng g−1 LOQ: 0.93 ng g−1 | [100] |
| Sildenafil, Tadalafil, Vardenafil | Standard solutions, pharmaceutical dosage forms and herbal products | Tablet samples and herbal products Mixed with 50 mL of ACN: water 50:50 (v/v), sonication for 15 min and filtration through a 0.45 µm syringe filter | LC-ESI-MS/MS | Sildenafil, Tadalafil, Vardenafil LLOQ: 20 ng mL−1 | [101] |
| Sildenafil, Tadalafil, Vardenafil, Avanafil | Standard solutions, urine samples and illicit medications | Urine samples Dilution of urine samples in ultrapure water (1:10), filtration through a 0.22 µm syringe filter and spiking with standard solutions. Illicit medications Homogenisation of pills, dissolution in ultrapure water, sonication for 30 min and filtration with a 0.22 µm syringe filter. | LC-qTOF-MS/MS | Sildenafil LOD: 2.19 ng g−1 LOQ: 7.28 ng g−1 Tadalafil LOD: 9.81 ng g−1 LOQ: 32.71 ng g−1 Vardenafil LOD: 2.28 ng g−1 LOQ: 7.59 ng g−1 Avanafil LOD: 1.63 ng g−1 LOQ: 5.43 ng g−1 | [104] |
| Sildenafil | Wastewater samples | Solid-phase extraction (SPE) | SPE-LC-MS/MS | Not reported | [106] |
| Sildenafil & N-desmethyl sildenafil | Surface water and drinking water | Solid-phase extraction (SPE) | UHPLC-MS/MS | Sildenafil Surface water LOD: 1.0 pg L−1 LOQ: 0.9 ng L−1 Drinking water LOD: 1.0 pg L−1 LOQ: 0.8 ng L−1 N-desmethyl sildenafil Surface water LOD: 1.0 pg L−1 LOQ: 1.4 ng L−1 Drinking water LOD: 1.0 pg L−1 LOQ: 1.3 ng L−1 | [107] |
| Homosildenafil, Hydroxyhomo-sildenafil, Norneosildenafil, Thiosildenafil | Artificial freshwater & wastewater samples | Irradiation of tests solution in artificial freshwater with an initial concentration of 10 mg L−1 and 1 µg L−1 Wastewater samples Liquid–liquid extraction | UHPL-ESI-MS/MS | Not reported | [108] |
| Sildenafil and Vardenafil | Phosphate buffer (pH = 7.4) | Irradiation of tests solution in 5 mM phosphate buffer diluted with distilled water, with an initial concentration of 10 mg L−1 | LC-ESI-MS/MS | Not reported | [109] |
| Sildenafil, Tadalafil, Vardenafil and their metabolites | Spiked wastewater samples | Homogenisation and filtration through 0.2 µm cellulose filter | HPLC-(QqQ)-MS/MS | Sildenafil LOD: 1.8 ng L−1 LOQ: 6 ng L−1 Tadalafil LOD: 2.3 ng L−1 LOQ: 7.5 ng L−1 Vardenafil LOD: 7.2 ng L−1 LOQ: 24 ng L−1 | [110] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianco, G.; Foti, L.; Pascale, R.; Lelario, F.; Coviello, D.; Brienza, M.; Bufo, S.A.; Scrano, L. Phosphodiesterase-5 (PDE-5) Inhibitors as Emergent Environmental Contaminants: Advanced Remediation and Analytical Methods. Water 2021, 13, 2859. https://doi.org/10.3390/w13202859
Bianco G, Foti L, Pascale R, Lelario F, Coviello D, Brienza M, Bufo SA, Scrano L. Phosphodiesterase-5 (PDE-5) Inhibitors as Emergent Environmental Contaminants: Advanced Remediation and Analytical Methods. Water. 2021; 13(20):2859. https://doi.org/10.3390/w13202859
Chicago/Turabian StyleBianco, Giuliana, Luca Foti, Raffaella Pascale, Filomena Lelario, Donatella Coviello, Monica Brienza, Sabino Aurelio Bufo, and Laura Scrano. 2021. "Phosphodiesterase-5 (PDE-5) Inhibitors as Emergent Environmental Contaminants: Advanced Remediation and Analytical Methods" Water 13, no. 20: 2859. https://doi.org/10.3390/w13202859
APA StyleBianco, G., Foti, L., Pascale, R., Lelario, F., Coviello, D., Brienza, M., Bufo, S. A., & Scrano, L. (2021). Phosphodiesterase-5 (PDE-5) Inhibitors as Emergent Environmental Contaminants: Advanced Remediation and Analytical Methods. Water, 13(20), 2859. https://doi.org/10.3390/w13202859












