The Impact of Bioaugmentation on the Performance and Microbial Community Dynamics of an Industrial-Scale Activated Sludge Sequencing Batch Reactor under Various Loading Shocks of Heavy Oil Refinery Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Screening and Evaluation of Mixed Microbial Strains
2.2. Preparation of SBA
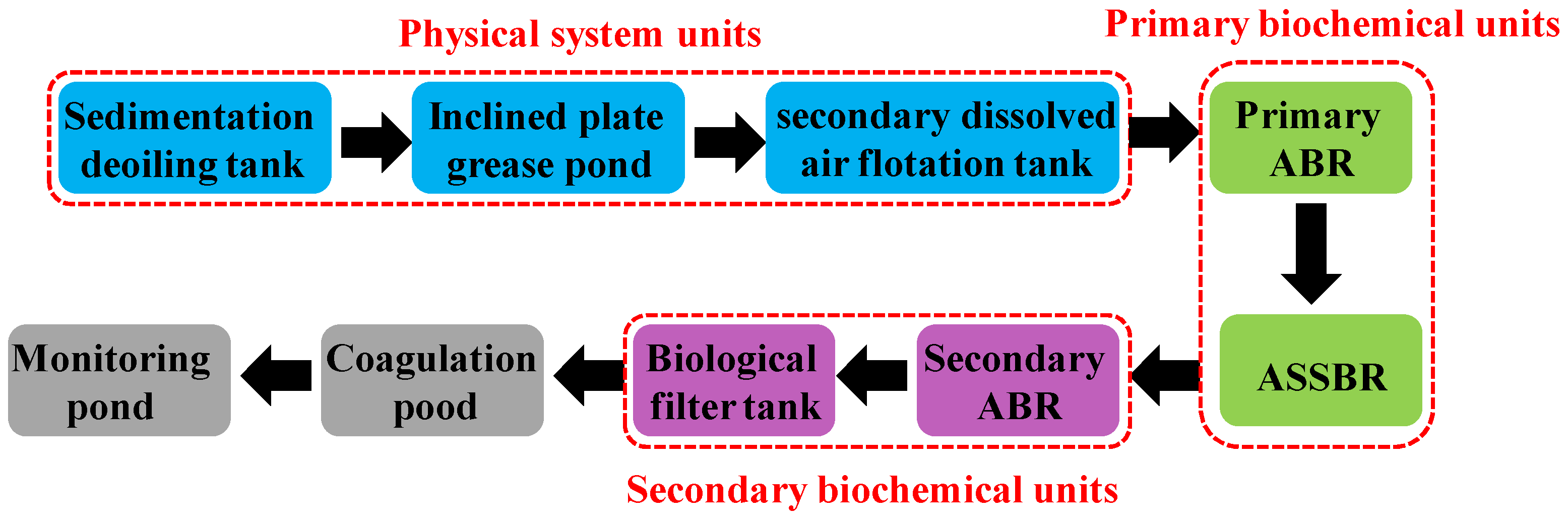
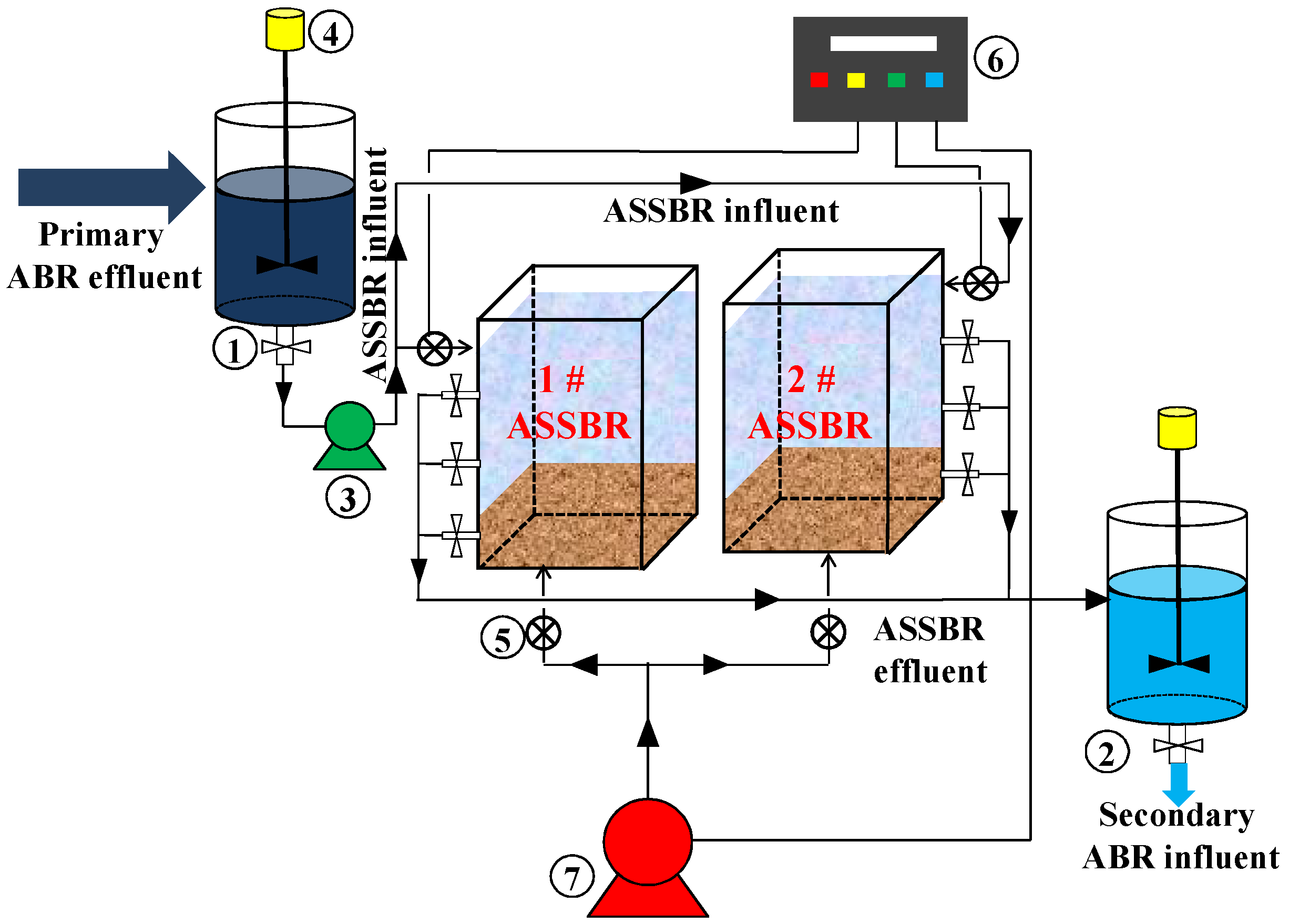
2.3. Industrial-Scale ASSBR Process
2.4. ASSBR Start-Up and Operation
2.5. Design of Loading Shock Experiments
2.6. Water Analysis
2.7. Collection of DNA Sequencing Samples
2.8. DNA Extraction and Microbial Diversity Analysis
3. Results and Discussion
3.1. Construction and Optimization of SBA
3.1.1. Optimization of Nutrient Concentration
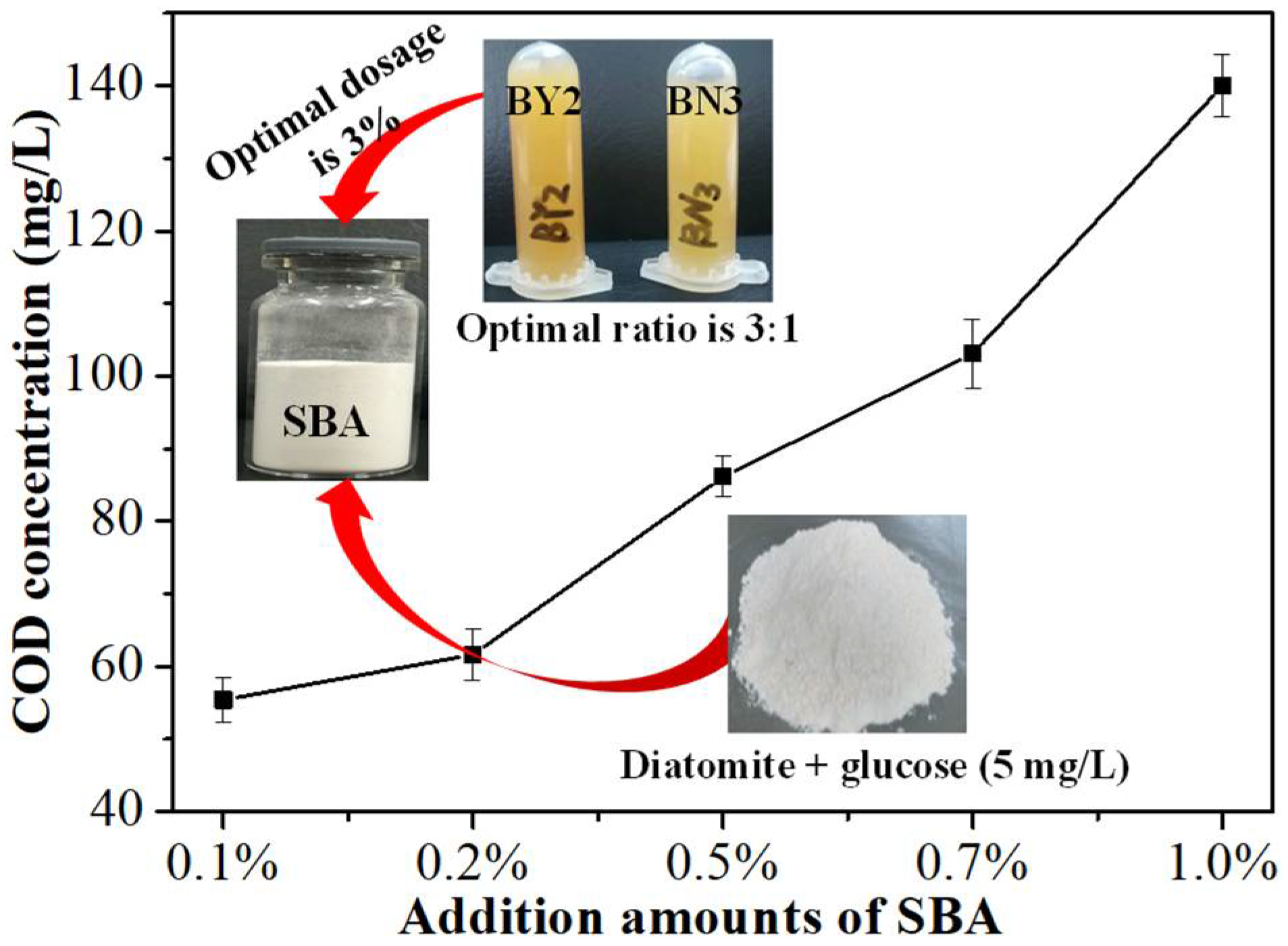
3.1.2. Optimization of Dosage of SBA
3.2. Effects of Bioaugmentation on the Dynamics of Microbial Communities
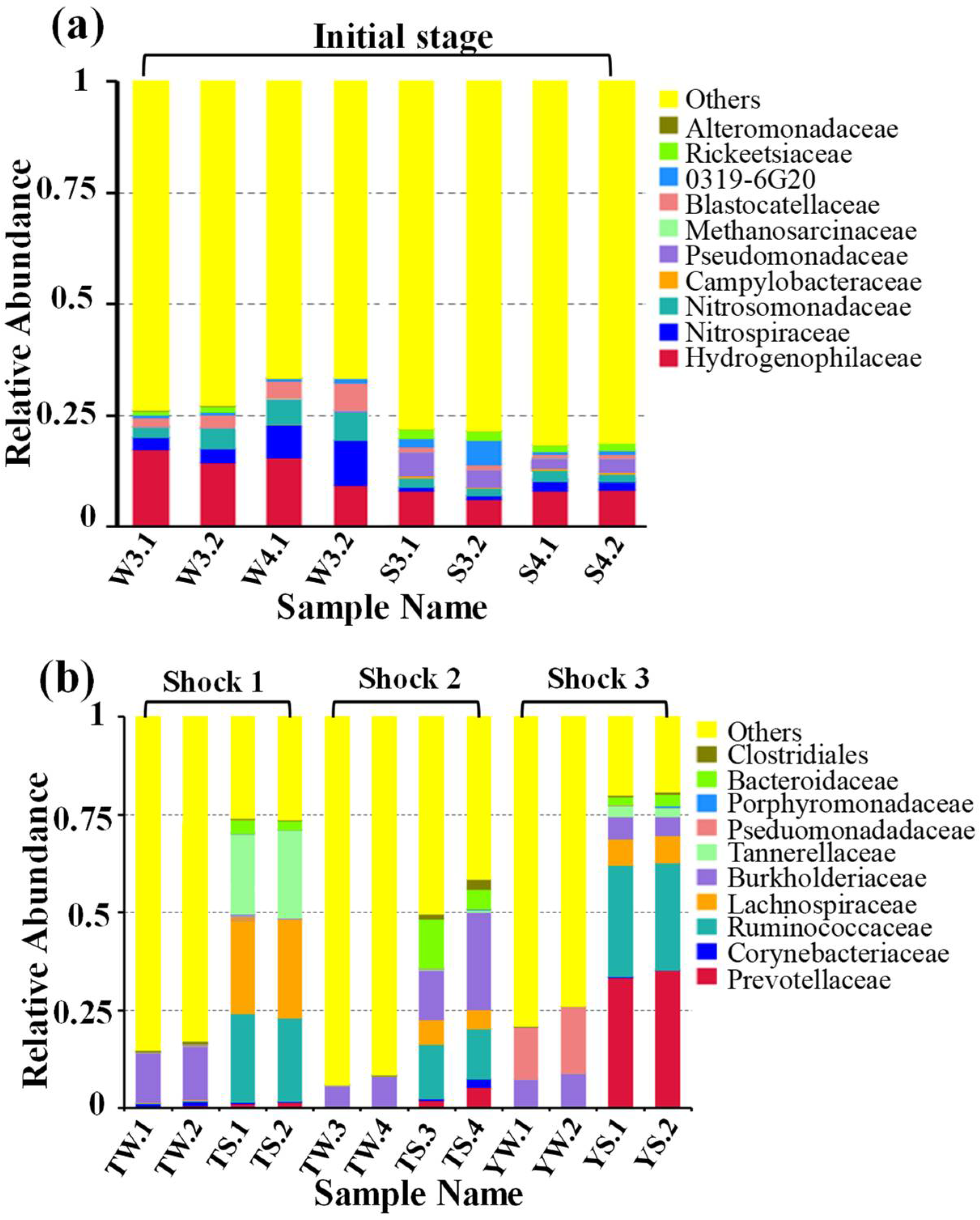
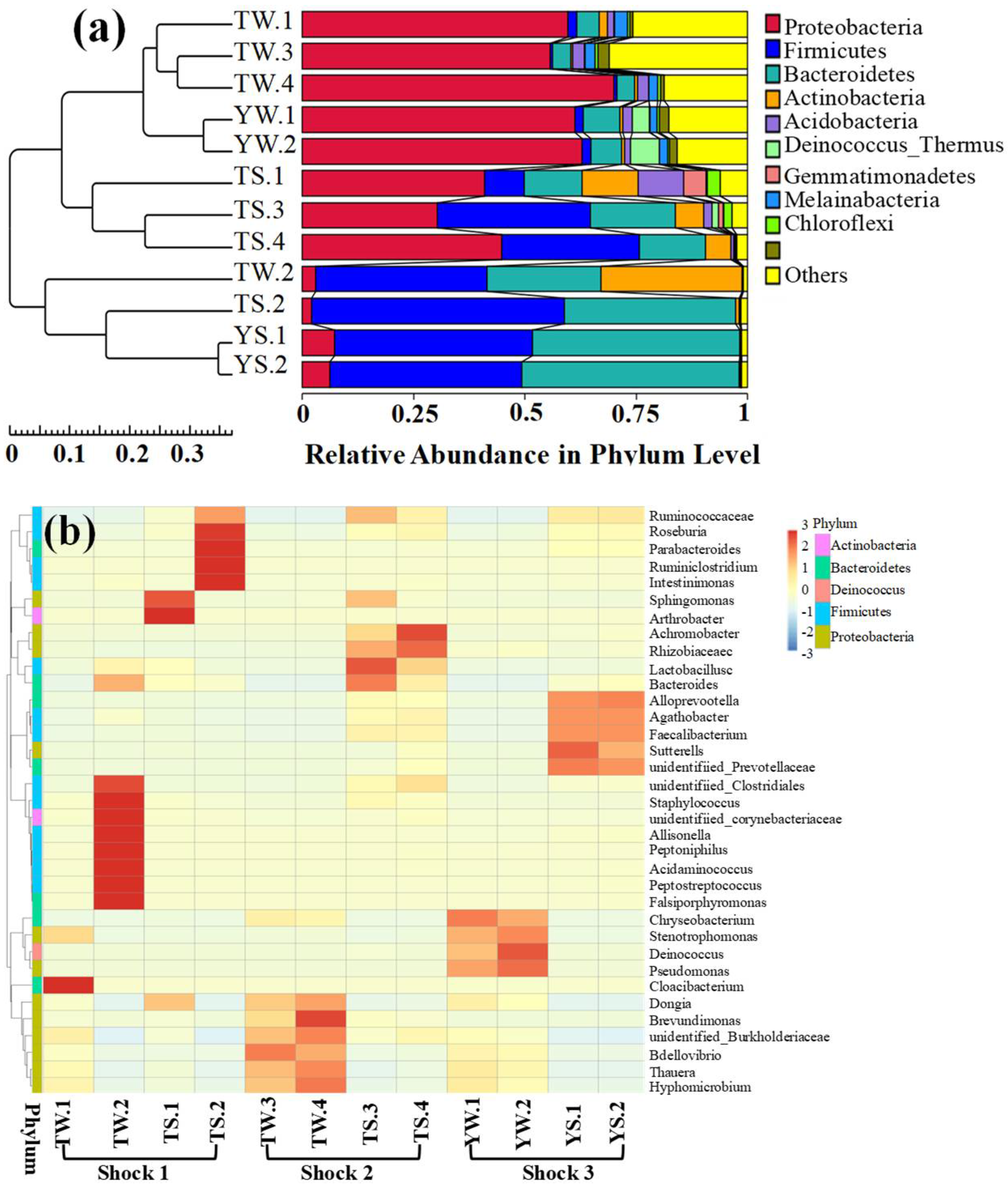
3.2.1. Evolutionary Characteristics of Microbial Communities
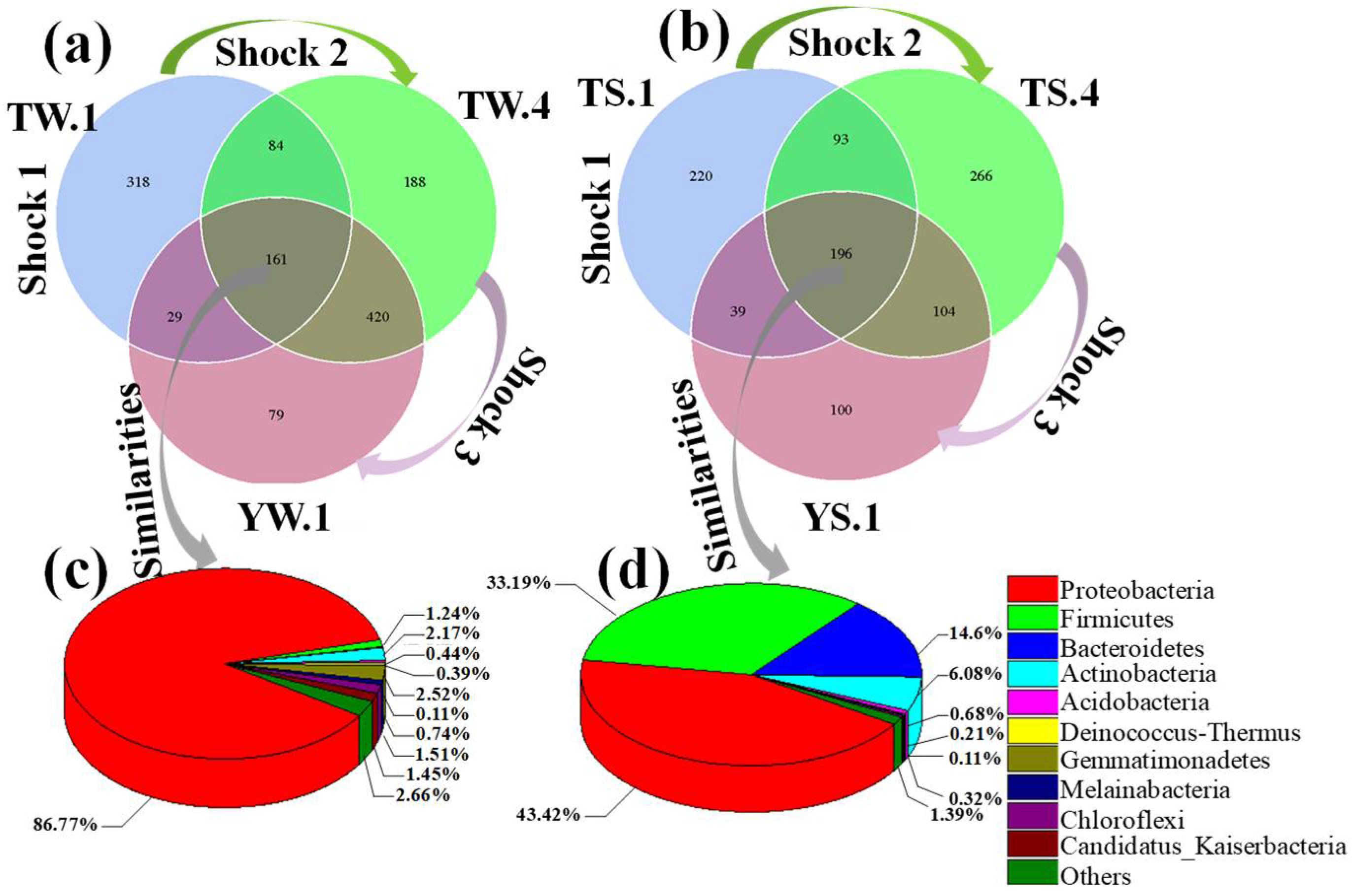
3.2.2. Similarities and Differences between Microbial Communities
3.3. Start-Up of the Two ASSBRs
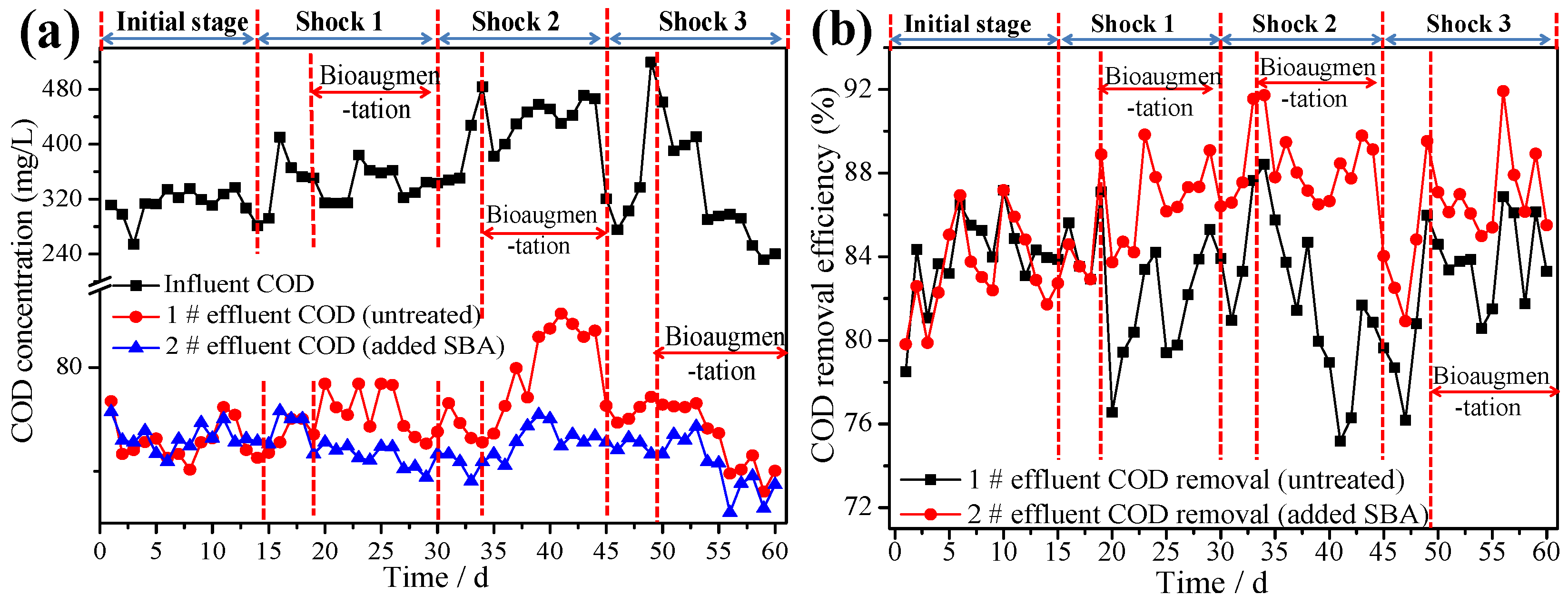
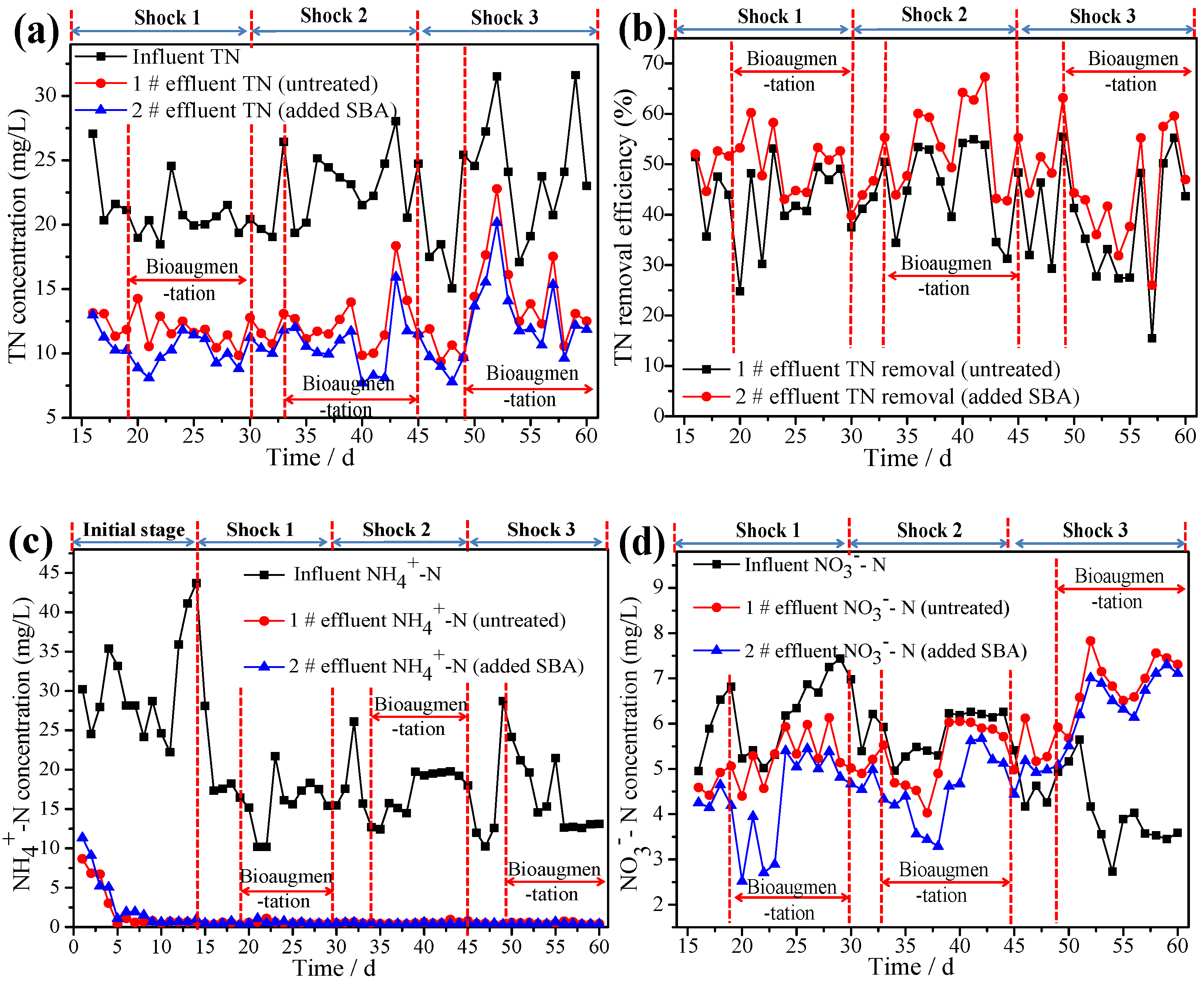
3.4. Reinforcing Effects of Bioaugmentation on ASSBRs Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dong, H.H.; Jiang, X.Y.; Sun, S.S.; Fang, L.; Wang, W.; Cui, K.; Zhang, Z.Z.; Fu, P.C. A cascade of a denitrification bioreactor and anaerobic biofilm reactor for heavy oil refinery wastewater treatment. RSC. Adv. 2019, 6, 7495. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, H.F.; Han, H.J.; Shan, S.D. Treatment of real coal gasification wastewater using a novel integrated system of anoxic hybrid two-stage aerobic processes: Performance and the role of pure oxygen microbubble. Environ. Sci. Pollut. Res. 2016, 23, 11916. [Google Scholar] [CrossRef]
- Li, S.Y.; Fei, X.N.; Cao, L.Y.; Chi, Y.Z. Insights into the effects of carbon source on sequencing batch reactors: Performance, quorum sensing, and microbial community. Sci. Total Environ. 2019, 691, 799–809. [Google Scholar] [CrossRef]
- Jaramillo, F.; Orchard, M.; Muñoz, C.; Zamorano, M.; Antileo, C. Advanced strategies to improve nitrification process in sequencing batch reactors—A review. J. Environ. Manag. 2018, 218, 154–164. [Google Scholar] [CrossRef]
- Trelles, I.J.; Mahamud, M.M.; Lavín, A.G.; Díaz, M. Sludge settling prediction in sequencing batch reactor plants. J. Clean. Prod. 2017, 152, 115–124. [Google Scholar] [CrossRef]
- Ketheesan, B.; Stuckey, D.C. Effects of hydraulic/organic shock/transient loads in anaerobic wastewater treatment: A review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 2693–2727. [Google Scholar] [CrossRef]
- Steinberg, L.M.; Regan, J.M. Response of lab-scale methanogenic reactors inoculated from different sources to organic loading rate shocks. Bioresour. Technol. 2011, 102, 8790–8798. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Ji, F.; Yuan, S.J.; Hao, W.T.; Wang, W.; Hu, Z.H. The performance of activated sludge exposed to arsanilic acid amprolium hydrochloride in sequencing batch reactors. Int. Biodeter. Biodegr. 2017, 116, 260–265. [Google Scholar] [CrossRef] [Green Version]
- Ma, Q.; Qu, Y.; Zhang, X.; Liu, Z.; Li, H.; Zhang, Z.; Wang, J.; Shen, W.; Zhou, J. Systematic investigation and microbial community profile of indole degradation processes in two aerobic activated sludge systems. Sci. Rep. 2015, 5, 17674. [Google Scholar] [CrossRef]
- Sarkar, P.; Roy, A.; Pal, S.; Mohapatra, B.; Kazy, S.K.; Maiti, M.K.; Sar, P. Enrichment and characterization of hydrocarbon-degrading bacteria from petroleum refinery waste as potent bioaugmentation agent for in situ bioremediation. Bioresour. Technol. 2017, 242, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Christou, M.L.; Vasileiadis, S.; Angelidaki, I.; Kotsopoulos, T.A. Effects of organic loading rate and hydraulic retention time on bioaugmentation performance to tackle ammonia inhibition in anaerobic digestion. Bioresour. Technol. 2020, 334, 125246. [Google Scholar] [CrossRef]
- Nzila, A. Mini review: Update on bioaugmentation in anaerobic processes for biogas production. Anaerobe 2017, 46, 3–12. [Google Scholar] [CrossRef]
- Germany, S.; Favier, L.; Djelal, H.; Taha, S.; Amrane, A. Bioaugmentation: Possible solution in the treatment of bio-Refractory organic compounds (Bio-ROCs). Biochem. Eng. J. 2012, 69, 75–86. [Google Scholar]
- Ma, F.; Guo, J.; Zhao, L.; Chang, C.; Cui, D. Application of bioaugmentation to improve the activated sludge system into the contact oxidation system treating petrochemical wastewater. Bioresour. Technol. 2009, 100, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Onyia, C.O.; Uyu, A.M.; Akunna, J.C.; Norulaini, N.A.; Omar, A. Increasing the fertilizer value of palm oil mill sludge: Bioaugmentation in nitrification. Water Sci. Technol. 2001, 44, 157–162. [Google Scholar] [PubMed]
- Raper, E.; Stephenson, T.; Anderson, D.R.; Fisher, R.; Soares, A. Industrial wastewater treatment through bioaugmentation. Process Saf. Environ. 2018, 118, 178–187. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Liu, Y.; Wang, C.; Xing, B.; Zhu, S.; Huang, J.; Xu, X.; Zhu, L. Biochar facilitates rapid restoration of methanogenesis by enhancing direct interspecies electron transfer after high organic loading shock. Bioresour. Technol. 2021, 320, 124360. [Google Scholar] [CrossRef]
- Cao, W.Z.; Wang, H.R.; Zeng, J.P.; Zhu, Y.; Lu, A.H.; Wang, C.Q. An experimental study on bioreduction of Fe3+ in montmorillonite by Cronobacter Sakazakii. Bull. Mineral. Petrol. Geochem. 2015, 34, 974–980. [Google Scholar]
- Huang, L.; Jiang, X.Y.; Xu, Q.S.; Zhang, Z.Z. Screening of efficient degradation bacteria for organic pollutants and their application in petroleum refinery wastewater treatment. Chem. Bioeng. 2018, 10, 44–48. [Google Scholar]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association (APHA): Washington, DC, USA, 2012; p. 1360. [Google Scholar]
- Yuan, Y.; Yu, Y.; Xi, H.B.; Zhou, Y.X.; He, X.W. Comparison of four test methods for toxicity evaluation of typical toxicants in petrochemical wastewater on activated sludge. Sci. Total Environ. 2019, 685, 273–279. [Google Scholar] [CrossRef]
- Gupta, P.; Sreekrishnan, T.R.; Ahammad, S. Role of sludge volume index in anaerobic sludge granulation in a hybrid anaerobic reactor. Chem. Eng. J. 2016, 283, 338–350. [Google Scholar] [CrossRef]
- He, S.; Chen, Y.; Qin, M.; Mao, Z.; Yuan, L.; Niu, Q.; Tan, X. Effects of temperature on anammox performance and community structure. Bioresour. Technol. 2018, 260, 186–195. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yanto, D.H.Y.; Hidayat, A.; Tachibana, A. Periodical biostimulation with nutrient addition and bioaugmentation using mixed fungal cultures to maintain enzymatic oxidation during extended bioremediation of oily soil microcosms. Int. Biodeter. Biodegr. 2017, 116, 112–123. [Google Scholar] [CrossRef]
- Zhou, J.; Li, H.S.; Chen, X.L.; Wan, D.J.; Mai, W.N.; Sun, C.Q. Cometabolic degradation of low-strength coking wastewater and the bacterial community revealed by high-throughput sequencing. Bioresour. Technol. 2017, 245, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Oberoi, A.S.; Huang, H.Q.; Khanal, S.K.; Sun, L.P.; Lu, H. Electron distribution in sulfur-driven autotrophic denitrification under different electron donor and acceptor feeding schemes. Chem. Eng. J. 2020, 404, 126486. [Google Scholar] [CrossRef]
- Hosseinlou, D. Determination of design loading rates for simultaneous anaerobic oxidation/partial nitrification-denitrification process and application in treating dairy industry effluent. J. Environ. Chem. Eng. 2021, 9, 105176. [Google Scholar] [CrossRef]
- Li, J.; Sun, S.; Yan, P.; Fang, L.; Yu, Y.; Xiang, Y.; Wang, D.; Gong, Y.; Gong, Y.; Zhang, Z. Microbial communities in the functional areas of a biofilm reactor with anaerobic–aerobic process for oily wastewater treatment. Bioresour. Technol. 2017, 238, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Quan, J.; Huang, K.; Zhao, J.; Xing, G.; Wu, P.; Chen, Y.; Ding, X.; Hu, Y. Effects of C/N ratio and dissolved oxygen on aerobic denitrification process: A mathematical modeling study. Chemosphere 2021, 272, 129521. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Hu, H.Y.; Shi, Z.J.; Yang, C.C.; Li, P.; Huang, M.; Ni, W.-M.; Shi, M.-L.; Jin, R.-C. Towards simultaneously removing nitrogen and sulfur by a novel process: Anammox and autotrophic desulfurization-denitrification (AADD). Chem. Eng. J. 2016, 297, 207–216. [Google Scholar] [CrossRef]
- Yi, X.H.; Wan, J.; Ma, Y.; Wang, Y. Characteristics and dominant microbial community structure of granular sludge under the simultaneous denitrification and methanogenesis process. Biochem. Eng. J. 2016, 107, 66–74. [Google Scholar] [CrossRef]
- Ma, F.; Sun, Y.; Li, A.; Zhang, X.; Yang, J. Activation of accumulated nitrite reduction by immobilized Pseudomonas stutzeri T13 during aerobic denitrification. Bioresour. Technol. 2015, 187, 30–36. [Google Scholar] [CrossRef]
- He, Y.; Tian, Z.; Luan, X.; Han, Z.; Zhang, Y.; Yang, M. Recovery of biological wastewater treatment system inhibited by oxytetracycline: Rebound of functional bacterial population and the impact of adsorbed oxytetracycline on antibiotic resistance. Chem. Eng. J. 2021, 418, 129364. [Google Scholar] [CrossRef]
- Lian, S.; Qu, Y.; Li, S.; Zhang, Z.; Zhang, H.; Dai, C.; Deng, Y. Interaction of graphene-family nanomaterials with microbial communities in sequential batch reactors revealed by high-throughput sequencing. Environ. Res. 2020, 184, 109392. [Google Scholar] [CrossRef]
- Dong, H.; Wang, W.; Song, Z.; Dong, H.; Wang, J.; Sun, S.; Zhang, Z.; Ke, M.; Zhang, Z.; Wu, W.M.; et al. A high-efficiency denitrification bioreactor for the treatment of acrylonitrile wastewater using waterborne polyurethane immobilized activated sludge. Bioresour. Technol. 2017, 239, 472–481. [Google Scholar] [CrossRef]
- Huang, X.Q.; Liu, L.L.; Zhao, J.; Zhang, J.B.; Cai, Z.C. The families Ruminococcaceae, Lachnospiraceae, and Clostridiaceae are the dominant bacterial groups during reductive soil disinfestation with incorporated plant residues. Appl. Soil. Ecol. 2019, 135, 65–72. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, R.R.; Zhang, Y.M.; Li, R.D.; Wang, G.; Li, K.M. Impact of nutrient addition on diversity and fate of fecal bacteria. Sci. Total Environ. 2018, 636, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, Z.; Hu, Y.; Song, C.; Li, F.; He, W.; Wang, X.; Li, Z.; Lin, H. Immobilization of nitrifying bacteria in magnetic PVA–SA-diatomite carrier for efficient removal of NH4+-N from effluents. Environ. Technol. Innov. 2021, 22, 101407. [Google Scholar] [CrossRef]
- Wang, M.M.; Cao, G.; Feng, N.X.; Pan, Y.Z. Bioaugmentation of two-stage aerobic sequencing batch reactor with mixed strains for high nitrate-nitrogen wastewater treatment. Chin. J. Chem. Eng. 2020, 28, 3103–3109. [Google Scholar] [CrossRef]
- Cai, J.; Zheng, P.; Qaisar, M. Simultaneous sulfide and nitrate removal in anaerobic reactor under shock loading. Bioresour. Technol. 2009, 100, 3010–3014. [Google Scholar]
- Martínez-Pachón, D.; Echeverry-Gallego, R.A.; Serna-Galvis, E.A.; Villarreal, J.M.; Botero-Coy, A.M.; Hernández, F.; Torres-Palma, R.A.; Moncayo-Lasso, A. Treatment of wastewater effluents from Bogotá—Colombia by the photo-electro-Fenton process: Elimination of bacteria and pharmaceutical. Sci. Total Environ. 2021, 772, 144890. [Google Scholar] [CrossRef]
| Loading Shock Pattern | ASSBR Number | Influent Source | Influent Toxicity Level | Added SBA | Running Time (d) | Influent Characteristics |
|---|---|---|---|---|---|---|
| Initial stage | #1 | Primary ABR effluent | Low | Untreated | 15 | Normal COD (55 mg/L) |
| #2 | Primary ABR effluent | Low | 0.2% | 15 | Normal COD (55 mg/L) | |
| Shock 1 | #1 | SDAF effluent | Medium | Untreated | 15 | High COD (400 mg/L) |
| #2 | SDAF effluent | Medium | 0.2% | 15 | High COD (400 mg/L) | |
| Shock 2 | #1 | COP effluent | Heavy | Untreated | 15 | High COD +high toxicity (800 mg/L) |
| #2 | COP effluent | Heavy | 0.2% | 15 | High COD +high toxicity (800 mg/L) | |
| Shock 3 | #1 | 1.5 times COP effluent | High | Untreated | 15 | High flow rate (22.5 L/h) |
| #2 | 1.5 times COP effluent | High | 0.2% | 15 | High flow rate (22.5 L/h) |
| Sample Number | COD (mg/L) (0 h) | BOD5 (mg/L) | B/C | COD (mg/L) (5 d) | COD Removal Efficiency (%) |
|---|---|---|---|---|---|
| Blank | 93.31 | 25.65 | 0.27 | 66.31 | 28.95 |
| BY2 + 0 mg/L glucose | 93.31 | 55.25 | 0.59 | 65.23 | 30.09 |
| BY2 + 5 mg/L glucose | 100.08 | 62.00 | 0.62 | 64.75 | 35.30 |
| BY2 + 10 mg/L glucose | 106.45 | 54.29 | 0.51 | 72.30 | 32.08 |
| BY2 + 15 mg/L glucose | 112.45 | 35.85 | 0.32 | 81.80 | 27.26 |
| BY2 + 20 mg/L glucose | 122.64 | 29.05 | 0.24 | 70.05 | 26.88 |
| Blank | 93.31 | 25.65 | 0.27 | 66.30 | 28.95 |
| BN3 + 0 mg/L glucose | 93.31 | 32.20 | 0.35 | 80.10 | 14.16 |
| BN3 + 5 mg/L glucose | 100.08 | 40.10 | 0.40 | 85.60 | 29.76 |
| BN3 + 10 mg/L glucose | 106.45 | 35.60 | 0.33 | 90.10 | 15.36 |
| BN3 + 15 mg/L glucose | 112.45 | 32.61 | 0.29 | 97.98 | 12.87 |
| BN3 + 20 mg/L glucose | 122.64 | 24.06 | 0.20 | 100.08 | 11.07 |
| Samples | Experimental Stage | OUTs | Shannon | Simpson | Chao1 | ACE | Good’s Coverage |
|---|---|---|---|---|---|---|---|
| W3.1 | Initial stage (ASSBR #1) | 780 | 6.938 | 0.976 | 850.678 | 862.989 | 0.995 |
| W3.2 | 794 | 6.556 | 0.956 | 1658.794 | 1045.369 | 0.991 | |
| W4.1 | 818 | 6.956 | 0.976 | 1074.079 | 1020.877 | 0.992 | |
| W4.2 | 786 | 7.148 | 0.981 | 865.813 | 873.86 | 0.995 | |
| S3.1 | Initial stage (ASSBR #2) | 938 | 7.439 | 0.974 | 997.692 | 980.363 | 0.997 |
| S3.2 | 1417 | 8.972 | 0.995 | 1478.546 | 1469.695 | 0.996 | |
| S4.1 | 849 | 7.028 | 0.974 | 983.384 | 981.789 | 0.994 | |
| S4.2 | 826 | 6.359 | 0.944 | 955.412 | 953.948 | 0.994 | |
| TW.1 | Shock 1 (ASSBR #2) | 899 | 7.223 | 0.974 | 1043.827 | 1031.856 | 0.994 |
| TW.2 | 865 | 7.551 | 0.986 | 944.138 | 949.443 | 0.995 | |
| TS.1 | 592 | 4.866 | 0.89 | 711.087 | 749.699 | 0.994 | |
| TS.2 | 659 | 6.503 | 0.947 | 738.222 | 712.084 | 0.997 | |
| TW.3 | Shock 2 (ASSBR #2) | 804 | 6.735 | 0.962 | 963.725 | 932.642 | 0.994 |
| TW.4 | 853 | 7.197 | 0.98 | 1004.208 | 1021.559 | 0.993 | |
| TS.3 | 505 | 6.98 | 0.977 | 594.062 | 572.949 | 0.997 | |
| TS.4 | 548 | 5.232 | 0.924 | 640.158 | 607.693 | 0.996 | |
| YW.1 | Shock 3 (ASSBR #2) | 672 | 6.666 | 0.968 | 723.663 | 733.41 | 0.997 |
| YW.2 | 689 | 6.375 | 0.955 | 740.528 | 765.932 | 0.996 | |
| YS.1 | 407 | 5.345 | 0.949 | 530.051 | 543.07 | 0.996 | |
| YS.2 | 439 | 5.495 | 0.949 | 514.333 | 549.455 | 0.996 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, K.; Xu, Q.; Sheng, X.; Meng, Q.; Shang, G.; Ma, Y.; Zhang, Z.; Guo, K. The Impact of Bioaugmentation on the Performance and Microbial Community Dynamics of an Industrial-Scale Activated Sludge Sequencing Batch Reactor under Various Loading Shocks of Heavy Oil Refinery Wastewater. Water 2021, 13, 2822. https://doi.org/10.3390/w13202822
Cui K, Xu Q, Sheng X, Meng Q, Shang G, Ma Y, Zhang Z, Guo K. The Impact of Bioaugmentation on the Performance and Microbial Community Dynamics of an Industrial-Scale Activated Sludge Sequencing Batch Reactor under Various Loading Shocks of Heavy Oil Refinery Wastewater. Water. 2021; 13(20):2822. https://doi.org/10.3390/w13202822
Chicago/Turabian StyleCui, Kai, Quanshu Xu, Xiaoying Sheng, Qingfan Meng, Gaoyuan Shang, Yingqun Ma, Zhongzhi Zhang, and Kun Guo. 2021. "The Impact of Bioaugmentation on the Performance and Microbial Community Dynamics of an Industrial-Scale Activated Sludge Sequencing Batch Reactor under Various Loading Shocks of Heavy Oil Refinery Wastewater" Water 13, no. 20: 2822. https://doi.org/10.3390/w13202822
APA StyleCui, K., Xu, Q., Sheng, X., Meng, Q., Shang, G., Ma, Y., Zhang, Z., & Guo, K. (2021). The Impact of Bioaugmentation on the Performance and Microbial Community Dynamics of an Industrial-Scale Activated Sludge Sequencing Batch Reactor under Various Loading Shocks of Heavy Oil Refinery Wastewater. Water, 13(20), 2822. https://doi.org/10.3390/w13202822






