Predicted and Measured Concentration of Pharmaceuticals in Surface Water of Areas with Increasing Anthropic Pressure: A Case Study in the Coastal Area of Central Italy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Sample Collection
- (i)
- Rural area with low inhabitant density (SwRur);
- (ii)
- Periurban area with medium inhabitant density (SwPurb);
- (iii)
- Urban area with high inhabitant density (SwUrb).
2.2. Analytical Determinations
2.3. Predicted Environmental Concentration of the Selected PhCs and Comparison Method
2.4. Environmental Risk Assessment
2.5. Statistical Analyses of Results
3. Results
3.1. Measured Environmental Concentration of PhCs
3.2. Validation of the Predictive Method
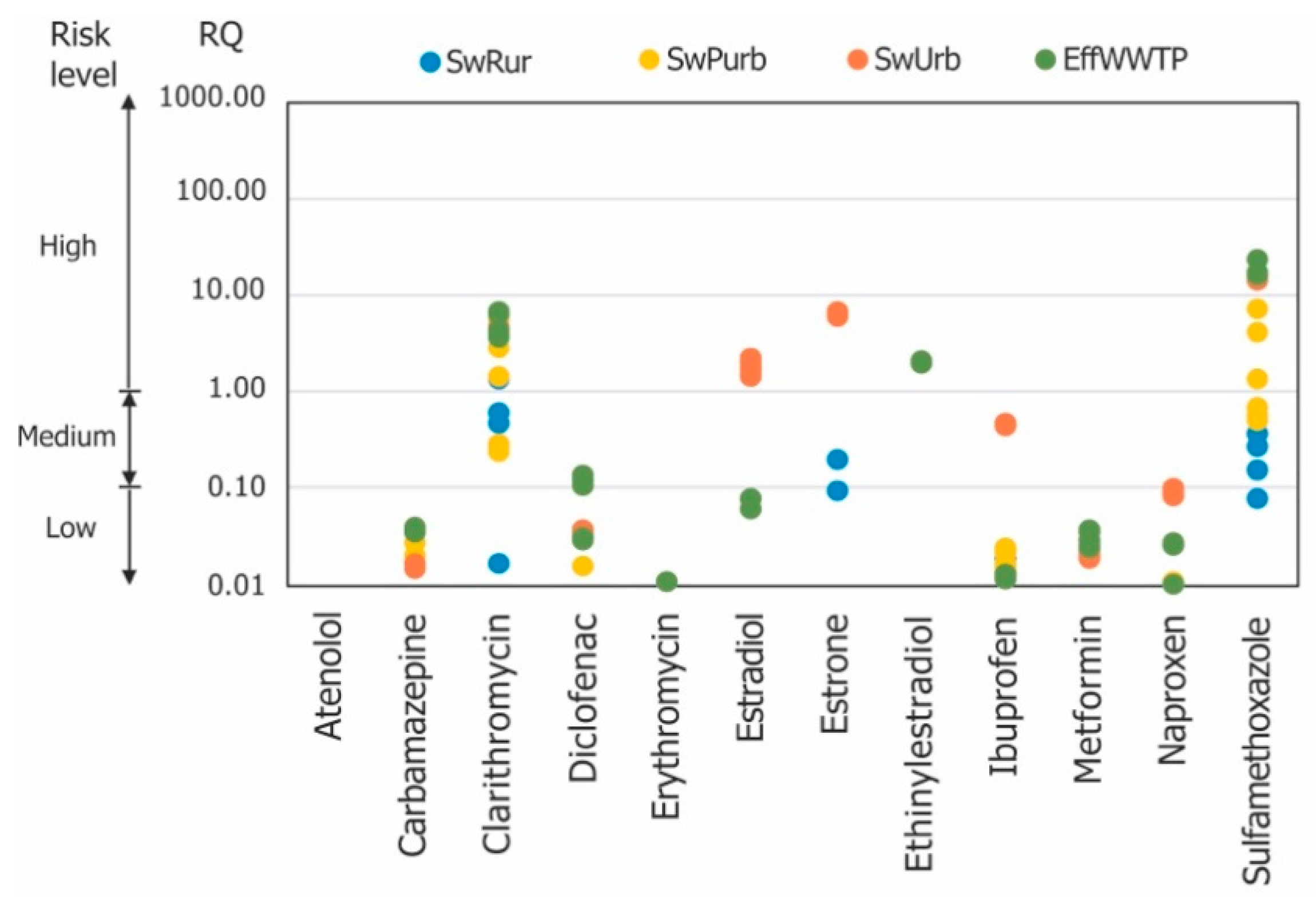
3.3. Environmental Risk Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hignite, C.; Azarnoff, D.L. Drugs and drug metabolites as environmental contaminants: Chlorophenoxyisobutyrate and salicylic acid in sewage water effluent. Life Sci. 1977, 20, 337–341. [Google Scholar] [CrossRef]
- Cortecci, G.; Dinelli, E.; Boschetti, T.; Arbizzani, P.; Pompilio, L.; Mussi, M. The Serchio River catchment, northern Tuscany: Geochemistry of stream waters and sediments, and isotopic composition of dissolved sulfate. Appl. Geochem. 2008, 23, 1513–1543. [Google Scholar] [CrossRef]
- Khatri, N.; Tyagi, S. Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Front. Life Sci. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Baran, N.; Stuart, M.E.; Ward, R.S. Emerging organic contaminants in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef] [Green Version]
- Figoli, A.; Fuoco, I.; Apollaro, C.; Chabane, M.; Mancuso, R.; Gabriele, B.; De Rosa, R.; Vespasiano, G.; Barca, D.; Criscuoli, A. Arsenic-contaminated groundwaters remediation by nanofiltration. Sep. Purif. Technol. 2020, 238, 116461. [Google Scholar] [CrossRef]
- Mateo-Sagasta, J.; Zadeh, S.M.; Turral, H. More People, More Food, Worse Water?: A Global Review of Water Pollution from Agriculture; Food and Agriculture Organization of the United Nations, FAO, and the International Water Management institute on Behalf of the Water, Land and Ecosystems Research Program of the CGIAR Colombo: Rome, Italy, 2018. [Google Scholar]
- De Filippis, G.; Ercoli, L.; Rossetto, R. A spatially distributed, physically-based modeling approach for estimating agricultural nitrate leaching to groundwater. Hydrology 2021, 8, 8. [Google Scholar] [CrossRef]
- Sall, M.L.; Diaw, A.K.D.; Gningue-Sall, D.; Efremova Aaron, S.; Aaron, J.J. Toxic heavy metals: Impact on the environment and human health, and treatment with conducting organic polymers, a review. Environ. Sci. Pollut. Res. 2020, 27, 29927–29942. [Google Scholar] [CrossRef] [PubMed]
- Meffe, R.; de Bustamante, I. Emerging organic contaminants in surface water and groundwater: A first overview of the situation in Italy. Sci. Total Environ. 2014, 481, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Petrie, B.; Barden, R.; Kasprzyk-Hordern, B. A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res. 2015, 72, 3–27. [Google Scholar] [CrossRef]
- Richardson, S.D.; Ternes, T.A. Water analysis: Emerging contaminants and current issues. Anal. Chem. 2018, 90, 398–428. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N.; Tomul, F.; Ha, N.T.H.; Nguyen, D.T.; Lima, E.C.; Le, G.T.; Chang, C.T.; Masindi, V.; Woo, S.H. Innovative spherical biochar for pharmaceutical removal from water: Insight into adsorption mechanism. J. Hazard. Mater. 2020, 394, 122255. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Hooda, P.S.; Swinden, J.; Barker, J.; Barton, S. Spatial (bio)accumulation of pharmaceuticals, illicit drugs, plasticisers, perfluorinated compounds and metabolites in river sediment, aquatic plants and benthic organisms. Environ. Pollut. 2018, 234, 864–875. [Google Scholar] [CrossRef] [Green Version]
- Carey, R.O.; Migliaccio, K.W. Contribution of wastewater treatment plant effluents to nutrient dynamics in aquatic systems: A review. Environ. Manag. 2009, 44, 205–217. [Google Scholar] [CrossRef]
- Richardson, M.L.; Bowron, J.M. The fate of pharmaceutical chemicals in the aquatic environment. J. Pharm. Pharmacol. 1985, 37, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Crane, M.; Watts, C.; Boucard, T. Chronic aquatic environmental risks from exposure to human pharmaceuticals. Sci. Total Environ. 2006, 367, 23–41. [Google Scholar] [CrossRef] [PubMed]
- Roig, B.; Greenwood, R.; Barceló, D. An international conference on “Pharmaceuticals in the Environment in a frame of EU Knappe project. Environ. Int. 2009, 35, 763–765. [Google Scholar] [CrossRef] [PubMed]
- Malchi, T.; Maor, Y.; Tadmor, G.; Shenker, M.; Chefetz, M. Irrigation of root vegetables with treated wastewater: Evaluating uptake of pharmaceuticals and the associated human health risks. Environ. Sci. Technol. 2014, 48, 9325–9333. [Google Scholar] [CrossRef]
- Richardson, S.D.; Ternes, T.A. Water analysis: Emerging contaminants and current issues. Anal. Chem. 2014, 86, 2813–2848. [Google Scholar] [CrossRef] [PubMed]
- Proia, L.; von Schiller, D.; Sànchez-Melsió, A.; Sabater, S.; Borrego, C.M.; Rodrìguez-Mozaz, S.; Balcázar, J.L. Occurrence and persistence of antibiotic resistance genes in river biofilms after wastewater inputs in small rivers. Environ. Pollut. 2016, 210, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. Removal of pharmaceuticals in sewage treatment plants in Italy. Environ. Sci. Technol. 2006, 40, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Petrović, M.; Ginebreda, A.; Barceló, D. Removal of pharmaceuticals during wastewater treatment and environmental risk assessment using hazard indexes. Environ. Int. 2010, 36, 15–26. [Google Scholar] [CrossRef]
- Hughes, S.R.; Kay, P.; Brown, L.E. Global synthesis and critical evaluation of pharmaceutical data sets collected from river systems. Environ. Sci. Technol. 2012, 47, 661–677. [Google Scholar] [CrossRef]
- Jjemba, P.K. Excretion and ecotoxicity of pharmaceutical and personal care products in the environment. Ecotoxicol. Environ. Saf. 2006, 63, 113–130. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Hooda, P.S.; Barker, J.; Barton, S.; Swinden, J. Ecotoxic pharmaceuticals, personal care products, and other emerging contaminants: A review of environmental, receptor-mediated, developmental, and epigenetic toxicity with discussion of proposed toxicity to humans. Crit. Rev. Environ. Sci. Technol. 2016, 46, 336–381. [Google Scholar] [CrossRef] [Green Version]
- Sabourin, D.L.; Beck, A.; Duenk, P.W.; Kleywegt, S.; Lapen, D.R.; Li, H.; Metcalfe, C.D.; Payne, M.; Topp, E. Runoff of pharmaceuticals and personal care products following application of dewatered municipal biosolids to an agricultural field. Sci. Total Environ. 2009, 407, 4596–4604. [Google Scholar] [CrossRef]
- Gottschall, N.; Topp, E.; Metcalfe, C.; Edwards, M.; Payne, M.; Kleywegt, S.; Russell, P.; Lapen, D.R. Pharmaceutical and personal care products in groundwater, subsurface drainage, soil, and wheat grain, following a high single application of municipal biosolids to a field. Chemosphere 2012, 87, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Verlicchi, P.; Al Aukidy, M.; Zambello, M. Occurrence of pharmaceutical compounds in urban wastewater: Removal, mass load and environmental risk after a secondary treatment-a review. Sci. Total Environ. 2012, 429, 123–155. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Grandclément, C.; Seyssiecq, I.; Piram, A.; Wong-Wah-Chung, P.; Vanot, G.; Tiliacos, T.; Roche, N.; Doumenq, P. From the conventional biological wastewater treatment to hybrid processes, the evaluation of organic micropollutant removal: A review. Water Res. 2017, 111, 297–317. [Google Scholar] [CrossRef] [Green Version]
- Joss, A.; Zabczynski, S.; Göbel, A.; Hoffmann, B.; Löffler, D.; McArdell, C.S.; Ternes, T.A.; Thomsen, A.; Siegrist, H. Biological degradation of pharmaceuticals in municipal wastewater treatment: Proposing a classification scheme. Water Res. 2006, 40, 1686–1696. [Google Scholar] [CrossRef] [PubMed]
- Statista. Available online: https://www.statista.com/statistics/978889/wastewater-treatment-plants-by-population-served-in-italy/ (accessed on 11 June 2021).
- Press release European Commission. Available online: https://ec.europa.eu/commission/presscorner/detail/en/IP_19_1475 (accessed on 16 June 2021).
- EU Decision 495/2015, Commission Implementing Decision (EU) 2015/495 of 20 March 2015 Establishing a Watch List of Substances for Union-Wide Monitoring in the Field of Water Policy Pursuant to Directive 2008/105/EC of the European Parliament and of the Council, Off. J. Eur. Union L 78 (2015) 40–42. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv%3AOJ.L_.2015.078.01.0040.01.ENG (accessed on 11 June 2021).
- EU Decision 2018/840, Commission Implementing Decision (EU) 2018/840 of 5 June 2018 Establishing a Watch List of Substances for Union-Wide Monitoring in the Field of Water Policy Pursuant to Directive 2008/105/EC of the European Parliament and of the Council and Repealing Commission Implementing Decision (EU) 2015/495, Off. J. Eur. Union L 141/9 (7.6.2018). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32018D0840 (accessed on 11 June 2021).
- EU Decision 2020/1161, Commission Implementing Decision (EU) 2018/840 of 4 August 2020 Establishing a Watch List of Substances for Union-Wide Monitoring in the Field of Water Policy Pursuant to Directive 2008/105/EC of the European Parliament and of the Council, Off. J. Eur. Union L 257/32 (6.8.2020). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2020.257.01.0032.01.ENG&toc=OJ:L:2020:257:TOC (accessed on 11 June 2021).
- Calamari, D.; Zuccato, E.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Strategic survey of therapeutic drugs in the rivers Po and Lambro in northern Italy. Environ. Sci. Technol. 2003, 37, 1241–1248. [Google Scholar] [CrossRef]
- Castiglioni, S.; Fanelli, R.; Calamari, D.; Bagnati, R.; Zuccato, E. Methodological approaches for studying pharmaceuticals in the environment by comparing predicted and measured concentrations in River Po, Italy. Regul. Toxicol. Pharmacol. 2004, 39, 25–32. [Google Scholar] [CrossRef]
- Escher, B.I.; Baumgartner, R.; Koller, M.; Treyer, K.; Lienert, J.; McArdell, C.S. Environmental toxicology and risk assessment of pharmaceuticals from hospital wastewater. Water Res. 2011, 45, 75–92. [Google Scholar] [CrossRef] [Green Version]
- European Medicines Agency, EMA. CHMP Committee for Medicinal Products for Human Use; Guideline on the Environmental Risk Assessment of Medicinal Products for Human Use. Doc. Ref. EMEA/CHMP/SWP/4447/00 corr 1, June; European Medicine Agency, EMA: London, UK, 2006. [Google Scholar]
- Verlicchi, P.; Al Aukidy, M.; Jelic, A.; Petrović, M.; Barcelò, D. Comparison of measured and predicted concentrations of selected pharmaceuticals in wastewater and surface water: A case study of a catchment area in the Po Valley (Italy). Sci. Total Environ. 2014, 470, 844–854. [Google Scholar] [CrossRef]
- Riva, F.; Zuccato, E.; Castiglioni, S. Prioritization and analysis of pharmaceuticals for human use contaminating the aquatic ecosystem in Italy. J. Pharm. Biomed. Anal. 2015, 106, 71–78. [Google Scholar] [CrossRef]
- Ong, T.T.; Blanch, E.W.; Jones, O.A. Predicted environmental concentration and fate of the top 10 most dispensed Australian prescription pharmaceuticals. Environ. Sci. Pollut. Res. 2018, 25, 10966–10976. [Google Scholar] [CrossRef]
- He, K.; Borthwick, A.G.; Lin, Y.; Li, Y.; Fu, J.; Wong, Y.; Liu, W. Sale-based estimation of pharmaceutical concentrations and associated environmental risk in the Japanese wastewater system. Environ. Int. 2020, 139, 105690. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.C.; Mol, M.P.G. Theoretical environmental risk assessment of ten used pharmaceuticals in Belo Horizonte, Brazil. Environ. Monit. Assess. 2019, 191, 275. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Canela, C.; Pueyo, V.; Barata, C.; Lacorte, S.; Marcé, R.M. Development of predicted environmental concentrations to prioritize the occurrence of pharmaceuticals in rivers from Catalonia. Sci. Total Environ. 2019, 666, 57–67. [Google Scholar] [CrossRef]
- Keller, V.D.J.; Williams, R.J.; Lofthouse, C.; Johnson, A.C. Worldwide estimation of river concentrations of any chemical originating from sewage-treatment plants using dilution factors. Environ. Toxicol. Chem. 2014, 33, 447–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, W.; Reich, M.; Beier, S.; Behrendt, J.; Gulyas, H.; Otterpohl, R. Measured and predicted environmental concentrations of carbamazepine, diclofenac, and metoprolol in small and medium rivers in northern Germany. Environ. Monit. Assess. 2016, 188, 487. [Google Scholar] [CrossRef]
- Pellegrino, E.; Bosco, S.; Ciccolini, V.; Pistocchi, C.; Sabbatini, T.; Silvestri, N.; Bonari, E. Agricultural abandonment in Mediterranean reclaimed peaty soils: Long-term effects on soil chemical properties, arbuscular mycorrhizas and CO2 flux. Agric. Ecosyst. Environ. 2014, 199, 164–175. [Google Scholar] [CrossRef]
- Ciccolini, V.; Bonari, E.; Ercoli, L.; Pellegrino, E. Phylogenetic and multivariate analyses to determine the effect of agricultural land-use intensification and soil physico-chemical properties on N-cycling microbial communities in drained Mediterranean peaty soils. Biol. Fertil. Soils 2016, 52, 811–824. [Google Scholar] [CrossRef] [Green Version]
- Ciccolini, V.; Ercoli, E.; Davison, J.; Vasar, M.; Öpik, M.; Pellegrino, E. Land-use intensity and host plant simultaneously shape the composition of arbuscular mycorrhizal fungal communities in a Mediterranean drained peatland. FEMS Microbiol. Ecol. 2016, 92, fiw186. [Google Scholar] [CrossRef]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Köppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Zuccato, E.; Castiglioni, S.; Fanelli, R. Identification of the pharmaceuticals for human use contaminating the Italian aquatic environment. J. Hazard. Mater. 2005, 122, 205–209. [Google Scholar] [CrossRef]
- Local Health Agency of Tuscany 5 (ASL5). Available online: https://www.ars.toscana.it/2-articoli/4473-rapporto-farmaci-in-toscana.html (accessed on 11 June 2021).
- Osservatorio Nazionale sull’Impiego dei Medicinali (OsMed). L’uso dei Farmaci in Italia; Rapporto Nazionale Anno 2014; Agrnzia Italiana del Farmaco (AIFA): Rome, Italy, 2015. [Google Scholar]
- ISTAT. Rapporto Annuale 2016. Available online: https://www.istat.it/it/files/2016/05/Ra2016.pdf (accessed on 11 June 2021).
- WHO Collaborating Centre for Drug Statistics Methodology. Available online: http://www.whocc.no (accessed on 11 June 2021).
- Demo-Geodemo, ISTAT. Available online: http://demo.istat.it/ricostruzione/index.php?lingua=ita (accessed on 11 June 2021).
- Ort, C.; Hollender, J.; Schaerer, M.; Siegrist, H. Model-based evaluation of reduction strategies for micropollutants from wastewater treatment plants in complex river networks. Environ. Sci. Technol. 2009, 43, 3214–3220. [Google Scholar] [CrossRef] [PubMed]
- Verlicchi, P.; Galletti, A.; Petrović, M.; Barcelò, D. Hospital effluents as a source of emerging pollutants: An overview of micropollutants and sustainable treatment options. J. Hydrol. 2010, 389, 416–428. [Google Scholar] [CrossRef]
- Zhang, Y.; Geißen, S.U.; Gal, C. Carbamazepine and diclofenac: Removal in wastewater treatment plants and occurrence in water bodies. Chemosphere 2008, 73, 1151–1161. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.C.; Williams, R.J. A model to estimate influent and effluent concentrations of estradiol, estrone, and ethinylestradiol at sewage treatment works. Environ. Sci. Technol. 2004, 38, 3649–3658. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, D.J.; Mastrocco, F.; Anderson, P.D.; Länge, R.; Sumpter, P.J. Predicted-no-effect concentrations for the steroid estrogens estrone, 17β-estradiol, estriol, and 17α-ethinylestradiol. Environ. Toxicol. Chem. 2012, 31, 1396–1406. [Google Scholar] [CrossRef] [PubMed]
- Oosterhuis, M.; Sacher, F.; Ter Laak, T.L. Prediction of concentration levels of metformin and other high consumption pharmaceuticals in wastewater and regional surface water based on sales data. Sci. Total Environ. 2013, 442, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Helwig, K.; Hunter, C.; McNaughtan, M.; Roberts, J.; Pahl, O. Ranking prescribed pharmaceuticals in terms of environmental risk: Inclusion of hospital data and the importance of regular review. Environ. Toxicol. Chem. 2016, 35, 1043–1050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, A.C.; Keller, V.; Dumont, E.; Sumpter, J.P. Assessing the concentrations and risks of toxicity from the antibiotics ciprofloxacin, sulfamethoxazole, trimethoprim and erythromycin in European rivers. Sci. Total Environ. 2015, 511, 747–755. [Google Scholar] [CrossRef] [Green Version]
- European Agency for the Evaluation of Medicinal Products, EMEA. CPMP Safety Working Party. Note for Guidance on Environmental Risk Assessments of Non-Genetically Modified Organism (Non-GMO) Containing Medicinal Products for Human Use; CPMP/SWP/4447/00; EMEA: London, UK, 2003. [Google Scholar]
- Zuccato, E.; Castiglioni, S.; Fanelli, R.; Reitano, G.; Bagnati, R.; Chiabrando, C.; Pomati, F.; Rossetti, C.; Calamari, D. Pharmaceuticals in the environment in Italy: Causes, occurrence, effects and control. Environ. Sci. Pollut. Res. 2006, 13, 15–21. [Google Scholar] [CrossRef]
- Lindim, C.; Van Gils, J.; Georgieva, D.; Mekenyan, O.; Cousins, I.T. Evaluation of human pharmaceutical emissions and concentrations in Swedish river basins. Sci. Total Environ. 2016, 572, 508–519. [Google Scholar] [CrossRef]
- Zhang, R.; Du, J.; Dong, X.; Huang, Y.; Xie, H.; Chen, J.; Li, X.; Kadokami, K. Occurrence and ecological risks of 156 pharmaceuticals and 296 pesticides in seawater from mariculture areas of Northeast China. Sci. Total Environ. 2021, 792, 148375. [Google Scholar] [CrossRef]
- Pescod, M.B. Wastewater Treatment and Use in Agriculture; Food and Agriculture Organization: Rome, Italy, 1992. [Google Scholar]
- Sigmund, C. Teoria e Pratica Della Depurazione Delle Acque Reflue. Procedure di Smaltimento e Progettazione (Theory and Practice of Wastewater Treatment. Disposal Procedures and Design); S.F. Flaccovio Editore: Palermo, Italy, 2008. [Google Scholar]
- Randhawa, G.K.; Kullar, J.S. Bioremediation of pharmaceuticals, pesticides, and petrochemicals with gomeya/cow dung. Int. Sch. Res. Netw. ISRN Pharmacol. 2011, 2011, 362459. [Google Scholar] [CrossRef] [Green Version]
- Sridevi, V.; Chandana, M.V.V.; Lakshmi, M.V.V.C.; Manasa, M. An overview on Bioremediation. Asian J. Biochem. Pharm. Res. 2012, 3, 99–104. [Google Scholar]
- Matamoros, V.; Nguyen, L.X.; Arias, C.A.; Salvadó, V.; Brix, H. Evaluation of aquatic plants for removing polar microcontaminants: A microcosm experiment. Chemosphere 2012, 88, 1257–1264. [Google Scholar] [CrossRef]
- Caracciolo, A.B.; Topp, E.; Grenni, P. Pharmaceuticals in the environment: Biodegradation and effects on natural microbial communities. A review. J. Pharm. Biomed. Anal. 2015, 106, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Gersberg, R.M.; Ng, W.J.; Tan, S.K. Removal of pharmaceuticals and personal care products in aquatic plant-based systems: A review. Environ. Pollut. 2014, 184, 620–639. [Google Scholar] [CrossRef] [PubMed]
- Chenxi, W.; Spongberg, A.L.; Witter, J.D. Determination of the persistence of pharmaceuticals in biosolids using liquid-chromatography tandem mass spectrometry. Chemosphere 2008, 73, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Guasch, H.; Ginebreda, A.; Geiszinger, A. (Eds.) Emerging and Priority Pollutants in Rivers: Bringing Science into River Management Plans; Springer Science & Business Media: Berlin, Germany, 2012; Volume 19. [Google Scholar]
- Mandaric, L.; Diamantini, E.; Stella, E.; Cano-Paoli, K.; Valle-Sistac, J.; Molins-Delgado, D.; Bellin, A.; Chiogna, G.; Majone, B.; Diaz-Cruz, M.S.; et al. Contamination sources and distribution patterns of pharmaceuticals and personal care products in Alpine rivers strongly affected by tourism. Sci. Total Environ. 2017, 590, 484–494. [Google Scholar] [CrossRef]
- Borja, A.; Galparsoro, I.; Solaun, O.; Muxika, I.; Tello, E.M.; Uriarte, A.; Valencia, V. The European Water Framework Directive and the DPSIR, a methodological approach to assess the risk of failing to achieve good ecological status. Estuar. Coast. Shelf Sci. 2006, 66, 84–96. [Google Scholar] [CrossRef]
- Zhang, Y.; Geißen, S.U. Elimination of carbamazepine in a non-sterile fungal bioreactor. Bioresource Technol. 2012, 112, 221–227. [Google Scholar] [CrossRef]
- Margot, J.; Bennati-Granier, C.; Maillard, J.; Blánquez, P.; Barry, D.A.; Holliger, C. Bacterial versus fungal laccase: Potential for micropollutant degradation. AMB Express 2013, 3, 63. [Google Scholar] [CrossRef] [Green Version]
- Domaradzka, D.; Guzik, U.; Wojcieszyńska, D. Biodegradation and biotransformation of polycyclic non-steroidal anti-inflammatory drugs. Rev. Environ. Sci. Biotechnol. 2015, 14, 229–239. [Google Scholar] [CrossRef] [Green Version]
- Rodarte-Morales, A.I.; Feijoo, G.; Moreira, M.T.; Lema, J.M. Degradation of selected pharmaceutical and personal care products (PPCPs) by white-rot fungi. World J. Microbiol. Biotechnol. 2011, 27, 1839–1846. [Google Scholar] [CrossRef]
- Mariotti, M.; Masoni, A.; Ercoli, L.; Arduini, I. Nitrogen leaching and residual effect of barley/field bean intercropping. Plant Soil Environ. 2015, 61, 60–65. [Google Scholar] [CrossRef] [Green Version]
- Afonso-Olivares, C.; Sosa-Ferrera, Z.; Santana-Rodríguez, J.J. Occurrence and environmental impact of pharmaceutical residues from conventional and natural wastewater treatment plants in Gran Canaria (Spain). Sci. Total Environ. 2017, 599, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Piazza, G.; Pellegrino, E.; Moscatelli, M.C.; Ercoli, L. Long-term conservation tillage and nitrogen fertilization effects on soil aggregate distribution, nutrient stocks and enzymatic activities in bulk soil and occluded microaggregates. Soil Tillage Res. 2020, 196, 104482. [Google Scholar] [CrossRef]
- Besse, J.P.; Garric, J. Human pharmaceuticals in surface waters: Implementation of a prioritization methodology and application to the French situation. Toxicol. Lett. 2008, 176, 104–123. [Google Scholar] [CrossRef] [PubMed]
- Cooper, E.R.; Siewicki, T.C.; Phillips, K. Preliminary risk assessment database and risk ranking of pharmaceuticals in the environment. Sci. Total Environ. 2008, 398, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Bouissou-Schurtz, C.; Houeto, P.; Guerbet, M.; Bachelot, M.; Casellas, C.; Mauclaire, A.C.; Panetier, P.; Delval, C.; Masset, D. Ecological risk assessment of the presence of pharmaceutical residues in a French national water survey. Regul. Toxicol. Pharmacol. 2014, 69, 296–303. [Google Scholar] [CrossRef]
- Tahar, A.; Tiedeken, E.J.; Clifford, E.; Cummins, E.; Rowan, N. Development of a semi-quantitative risk assessment model for evaluating environmental threat posed by the three first EU watch-list pharmaceuticals to urban wastewater treatment plants: An Irish case study. Sci. Total Environ. 2017, 603, 627–638. [Google Scholar] [CrossRef]
- Grill, G.; Khan, U.; Lehner, B.; Nicell, J.; Ariwi, J. Risk assessment of down-the-drain chemicals at large spatial scales: Model development and application to contaminants originating from urban areas in the Saint Lawrence River Basin. Sci. Total Environ. 2016, 541, 825–838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fent, K.; Weston, A.A.; Caminada, D. Ecotoxicology of human pharmaceuticals. Aquat. Toxicol. 2006, 76, 122–159. [Google Scholar] [CrossRef]

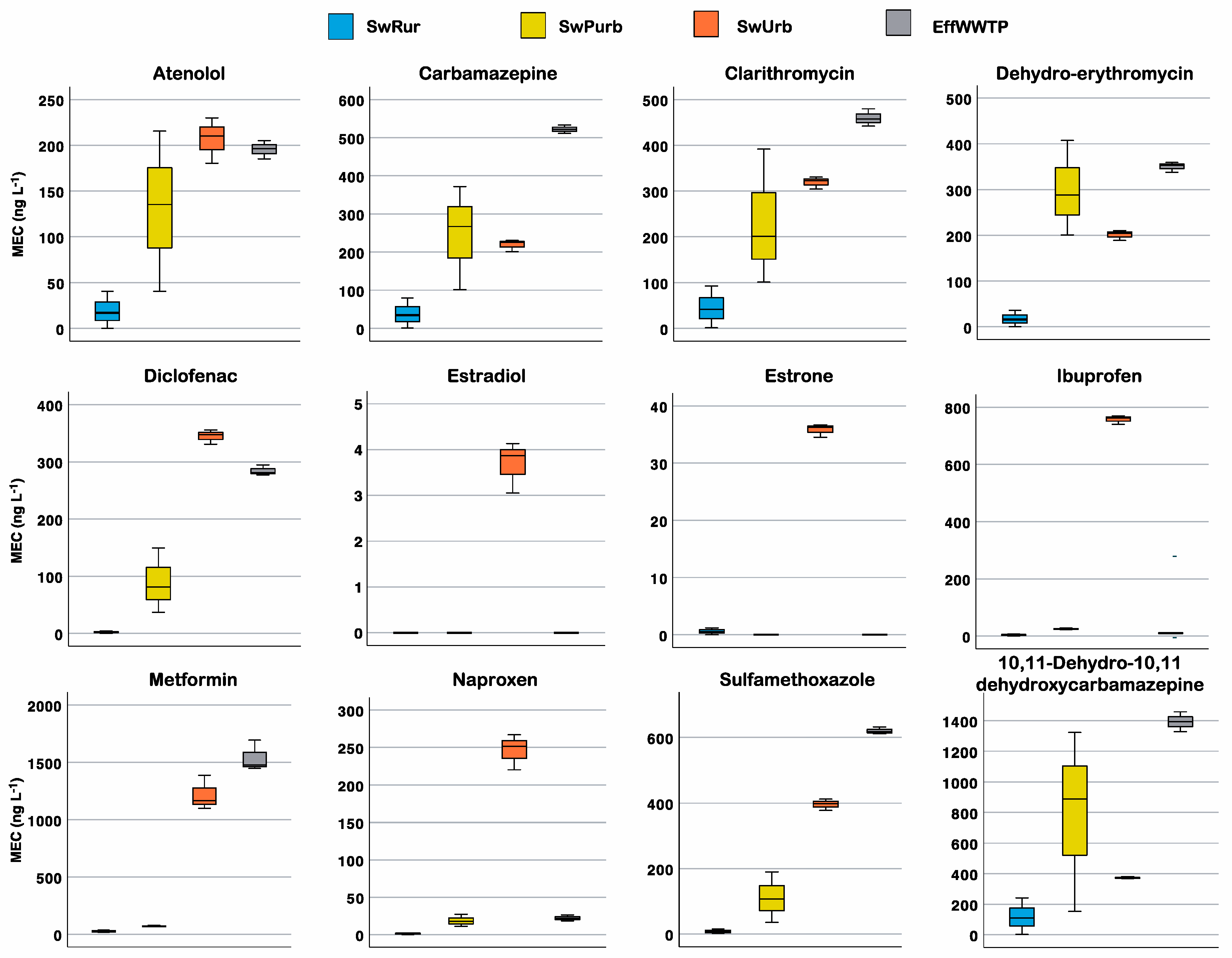
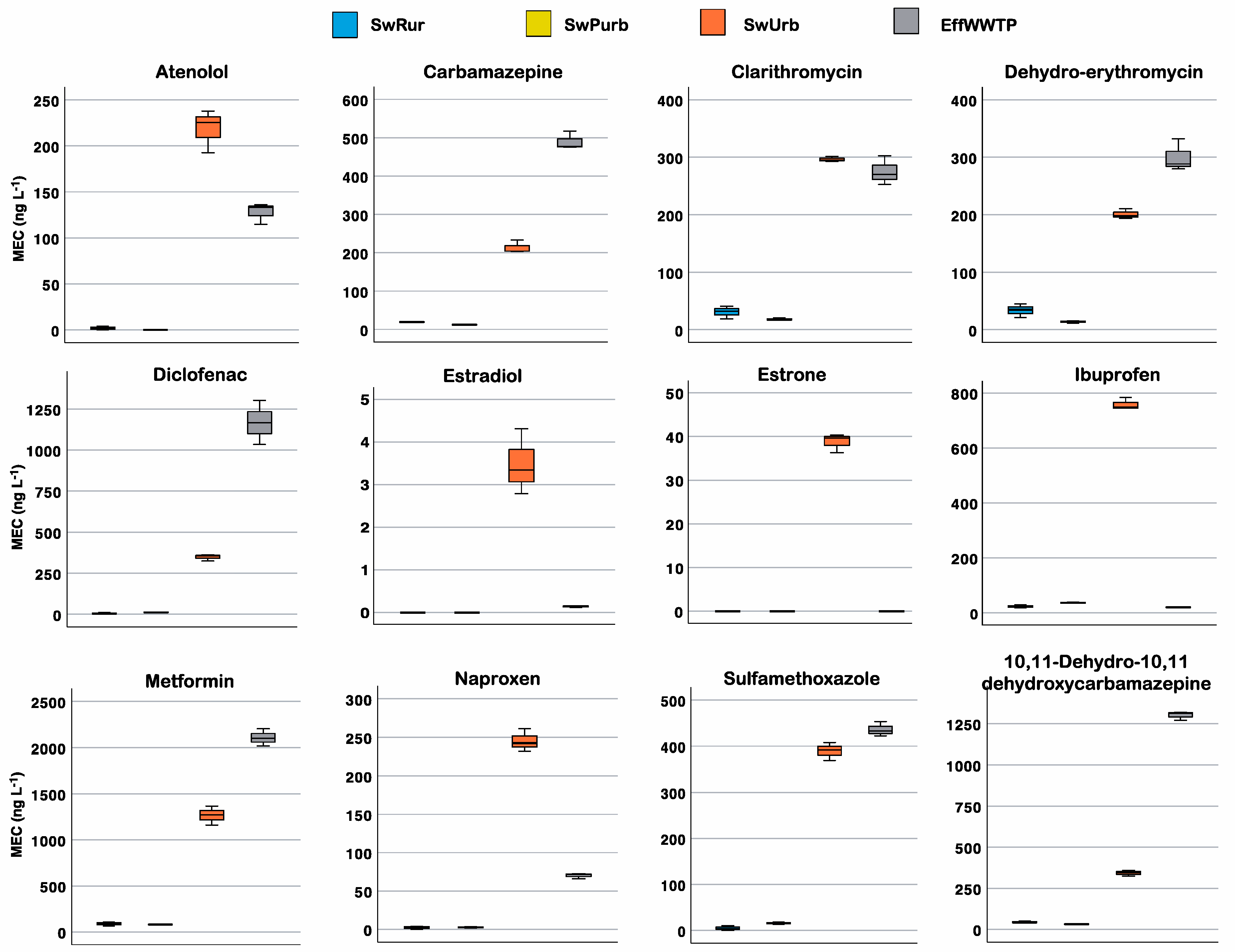
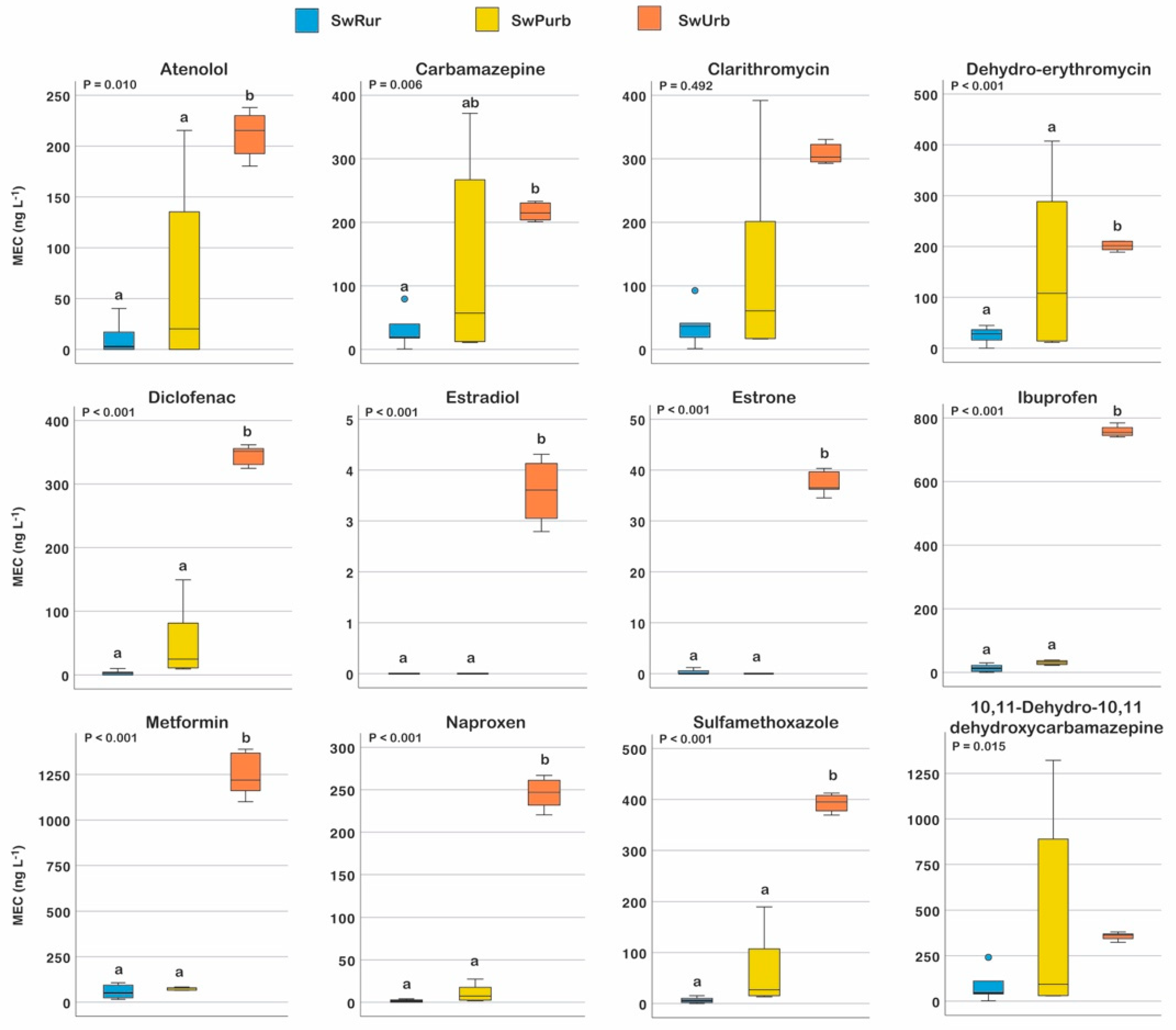
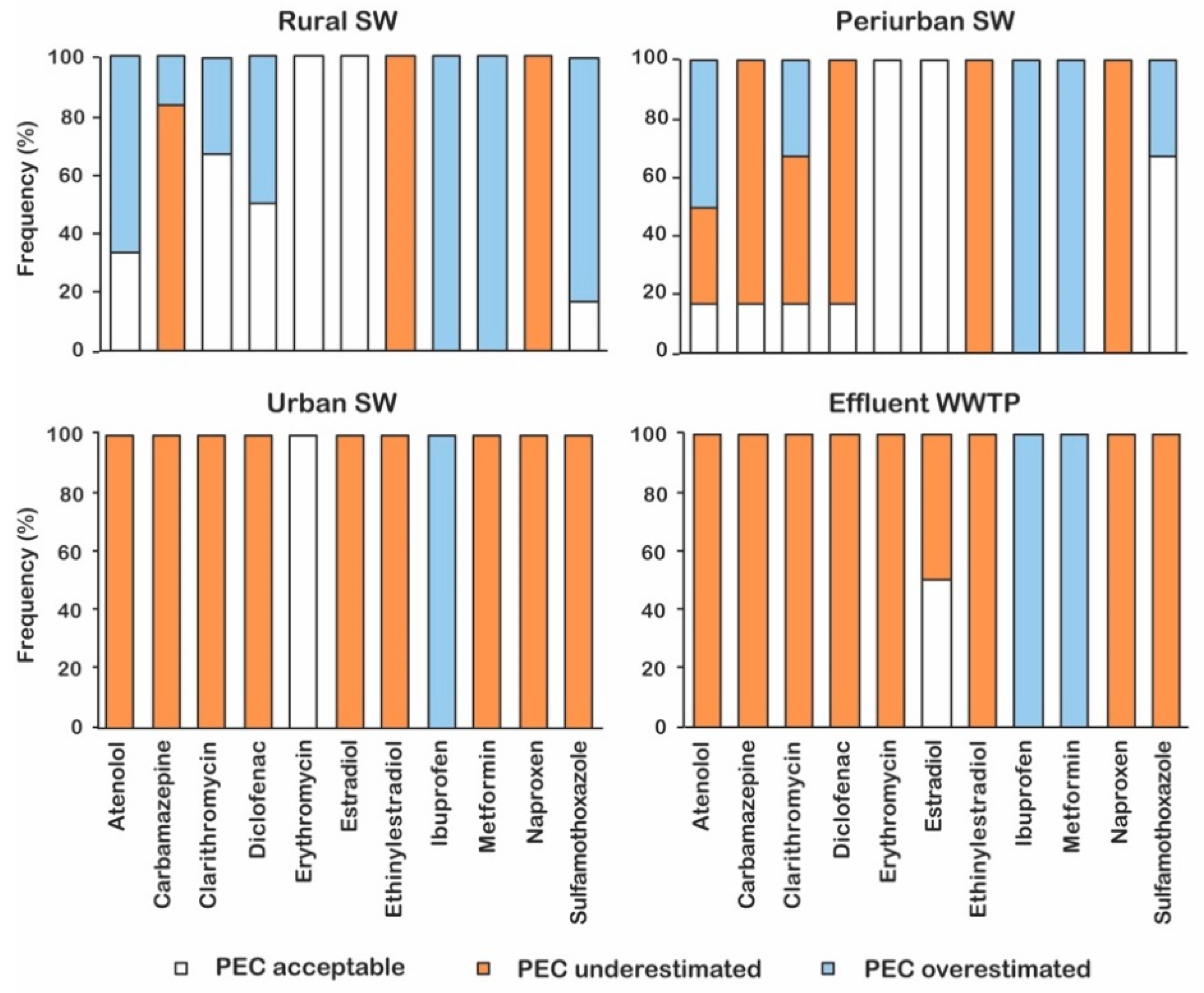
| Pharmaceutical | Therapeutic Class | DDDj a | OsMed b | ASL5 c | |||
|---|---|---|---|---|---|---|---|
| mg | DDDj,1000 | Qj | Total Sale (Aj) | Qj | Total Sale (Aj) | ||
| n 1000 inhab−1 day−1 | mg inh−1 year−1 | kg | mg inh−1 year−1 | kg | |||
| Atenolol | Cardiovascular drug | 75 | 10.0 | 273.8 | 94.62 | 281.8 | 97.41 |
| Carbamazepine | Antiepileptic | 1000 | - | 510.3 | 176.38 | ||
| Clarithromycin | Antibiotic | 500 | 2.7 | 492.8 | 170.32 | 511.4 | 176.76 |
| Diclofenac | Nonsteroidal anti-inflammatory drug | 100 | 12.7 | 463.6 | 160.23 | 193.4 | 66.85 |
| Erythromycin | Antibiotic | 1000 | - | 6.9 | 2.38 | ||
| Estradiol | Hormone | 2 | - | 0.7 | 0.24 | ||
| Estrone | Hormone | 1 | - | - | - | ||
| Ethinylestradiol | Synthetic hormone | 0.025 | - | 0.1 | 0.02 | ||
| Ibuprofen | Nonsteroidal anti-inflammatory drug | 1200 | 4.6 | 2014.8 | 696.42 | 1128.4 | 390.02 |
| Metformin | Antidiabetic | 2000 | 19.9 | 14,527.0 | 5122.20 | 15,778.1 | 5453.70 |
| Naproxen | Nonsteroidal anti-inflammatory drug | 500 | 0.8 | 146.0 | 50.47 | 121.1 | 41.87 |
| Sulfamethoxazole | Antibiotic | 2000 | - | 200.6 | 69.33 | ||
| Pharmaceutical * | Excretion Factor | Reference | Removal Factor | Reference | PNEC ng L−1 | Reference |
|---|---|---|---|---|---|---|
| Atenolol | 0.80 | [42] | 0.57 | [42] | 30,000 | [28] |
| Carbamazepine | 0.03 | [60] | 0.07 | [61] | 13,800 | [28] |
| Clarithromycin | 0.25 | [41] | 0.08 | [41] | 70 | [28] |
| Diclofenac | 0.10 | [60] | 0.35 | [42] | 9700 | [28] |
| Erythromycin | 0.05 | [60] | 0.73 | [41] | 20 | [28] |
| Estradiol | - | - | 0.13 | [62] | 2 | [63] |
| Ethinylestradiol | 0.27 | [41] | 0.21 | [41] | 0.1 | [63] |
| Ibuprofen | 0.10 | [60] | 0.92 | [42] | 1650 | [28] |
| Metformin | 0.79 | [42] | 0.98 | [64] | 60,000 | [65] |
| Naproxen | 0.10 | [57] | 0.68 | [42] | 2620 | [28] |
| Sulfamethoxazole | 0.18 | [66] | 0.48 | [66] | 27 | [28] |
| Pharmaceutical * | PEC Rur | PEC Purb | PEC Urb | Unrefined PEC | PEC EffWWTP |
|---|---|---|---|---|---|
| Atenolol | 37.0 | 148.0 | 369.9 | 136.1 | 1361.2 |
| Carbamazepine | 5.3 | 21.2 | 53.0 | 19.5 | 195.0 |
| Clarithromycin | 43.8 | 175.1 | 437.8 | 161.1 | 1611.2 |
| Diclofenac | 4.7 | 18.7 | 46.8 | 41.3 | 172.2 |
| Erythromycin | 0.0 | 0.1 | 0.3 | 0.1 | 1.3 |
| Estradiol | 0.2 | 0.9 | 2.2 | 0.8 | 8.2 |
| Ethinylestradiol | 0.0 | 0.0 | 0.1 | 0.0 | 0.2 |
| Ibuprofen | 3.4 | 13.4 | 33.6 | 21.1 | 123.7 |
| Metformin | 92.8 | 371.2 | 928.0 | 341.5 | 3415.0 |
| Naproxen | 1.4 | 5.8 | 14.4 | 6.4 | 53.1 |
| Sulfamethoxazole | 7.0 | 28.0 | 69.9 | 25.7 | 257.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardini, A.; Pellegrino, E.; Ercoli, L. Predicted and Measured Concentration of Pharmaceuticals in Surface Water of Areas with Increasing Anthropic Pressure: A Case Study in the Coastal Area of Central Italy. Water 2021, 13, 2807. https://doi.org/10.3390/w13202807
Cardini A, Pellegrino E, Ercoli L. Predicted and Measured Concentration of Pharmaceuticals in Surface Water of Areas with Increasing Anthropic Pressure: A Case Study in the Coastal Area of Central Italy. Water. 2021; 13(20):2807. https://doi.org/10.3390/w13202807
Chicago/Turabian StyleCardini, Alessio, Elisa Pellegrino, and Laura Ercoli. 2021. "Predicted and Measured Concentration of Pharmaceuticals in Surface Water of Areas with Increasing Anthropic Pressure: A Case Study in the Coastal Area of Central Italy" Water 13, no. 20: 2807. https://doi.org/10.3390/w13202807
APA StyleCardini, A., Pellegrino, E., & Ercoli, L. (2021). Predicted and Measured Concentration of Pharmaceuticals in Surface Water of Areas with Increasing Anthropic Pressure: A Case Study in the Coastal Area of Central Italy. Water, 13(20), 2807. https://doi.org/10.3390/w13202807





