Copper and Zinc Removal Efficiency of Two Reactive Filter Media Treating Motorway Runoff—Model for Service Life Estimation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Characterization of Filter Materials and Stormwater Used in the Experiments
2.2. Batch Experiment
2.3. Packed Bed Reactor Experiment
2.4. Conceptual Model
2.5. Simulation and Statistical Analysis
3. Results and Discussion
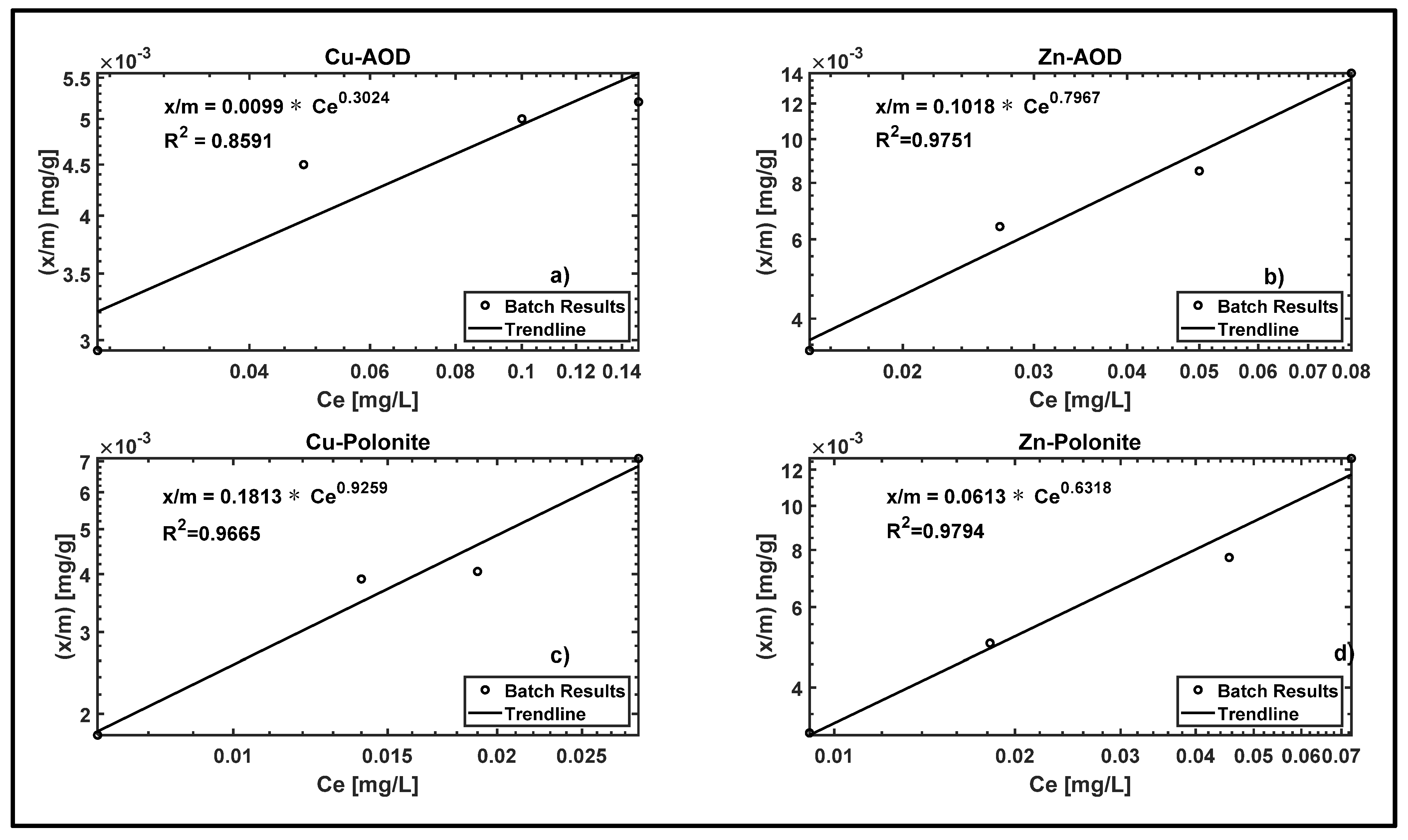
3.1. Batch Experiment
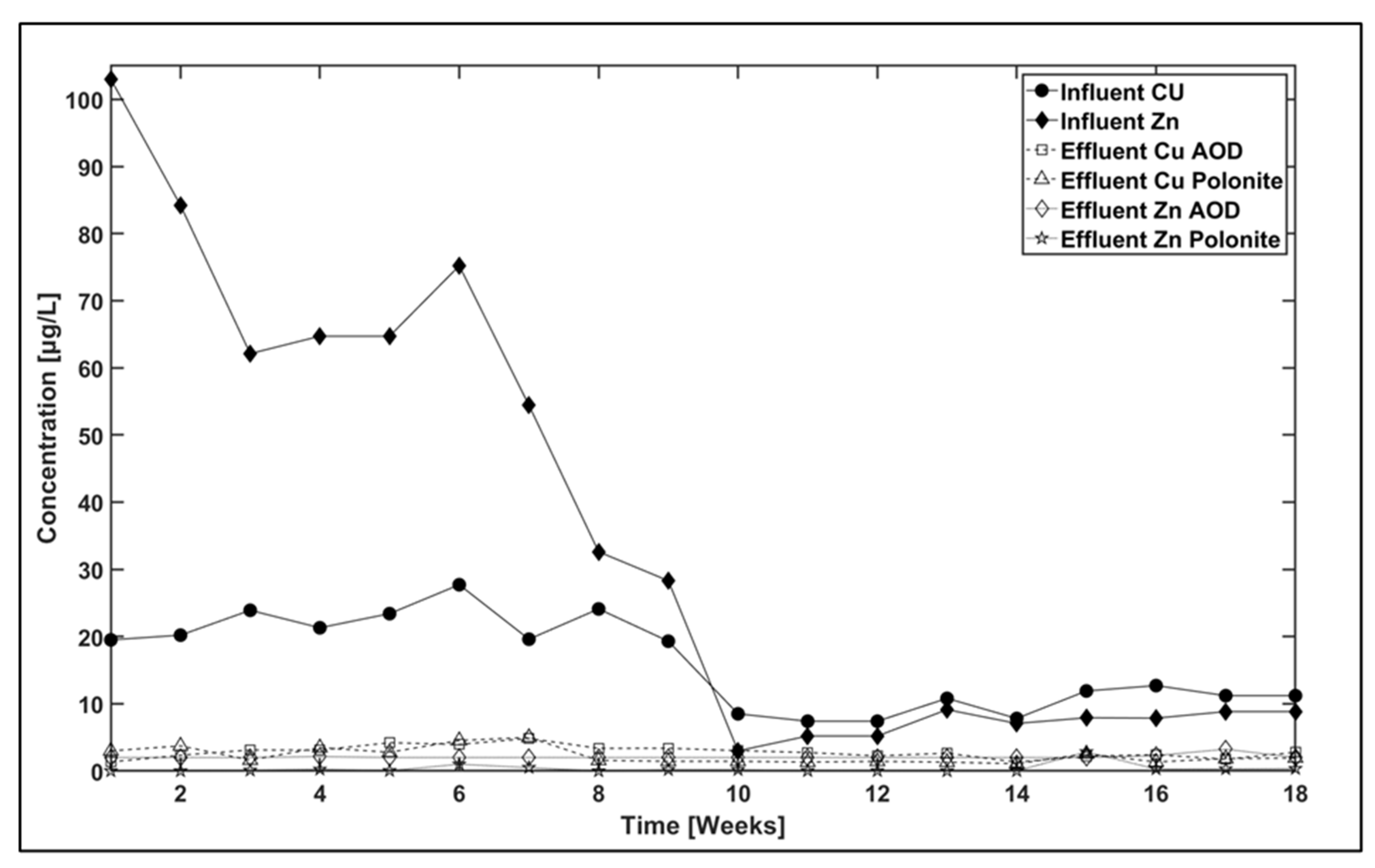
3.2. Packed Bed Reactor
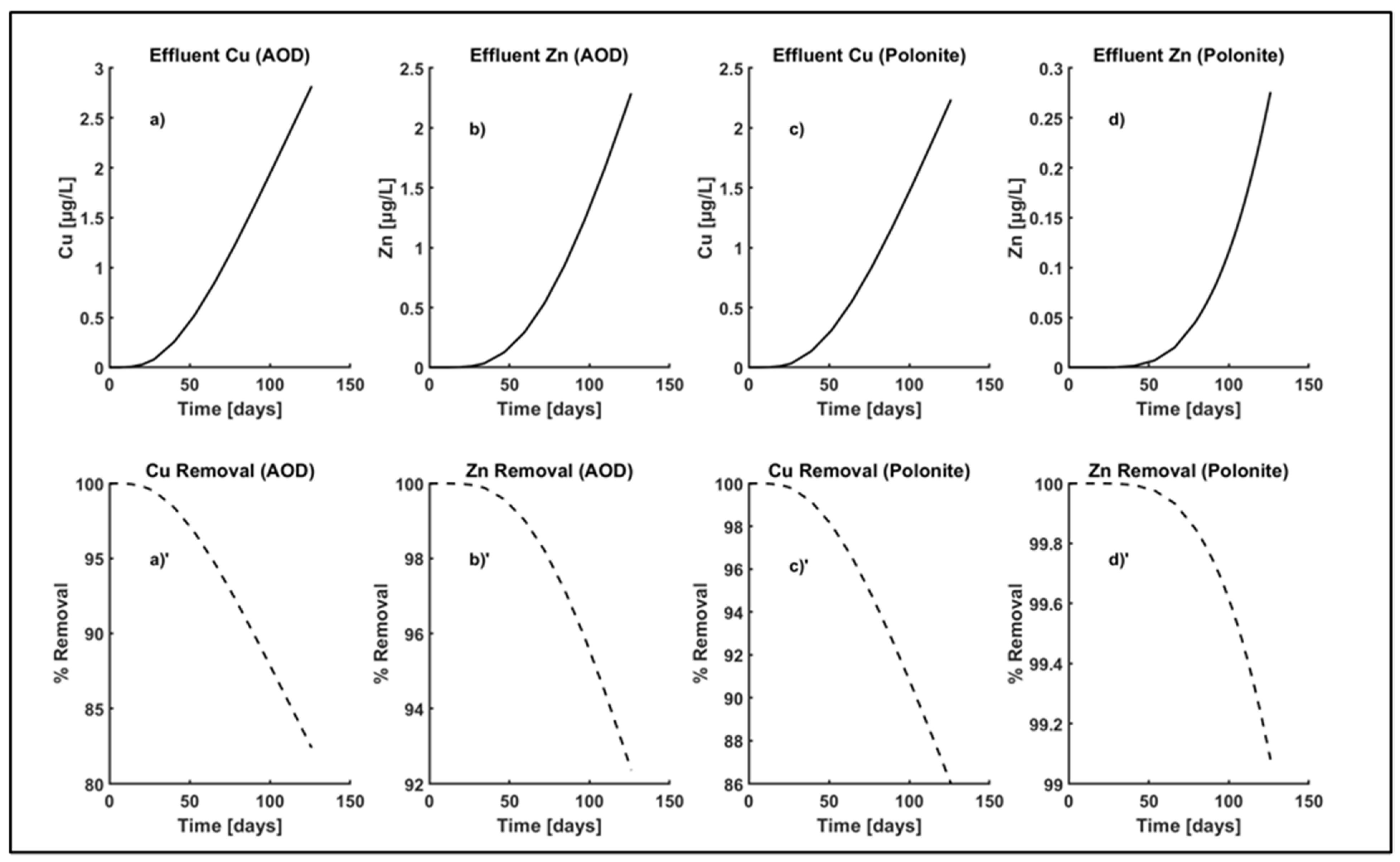
3.3. Simulation
3.4. Capability of the Model
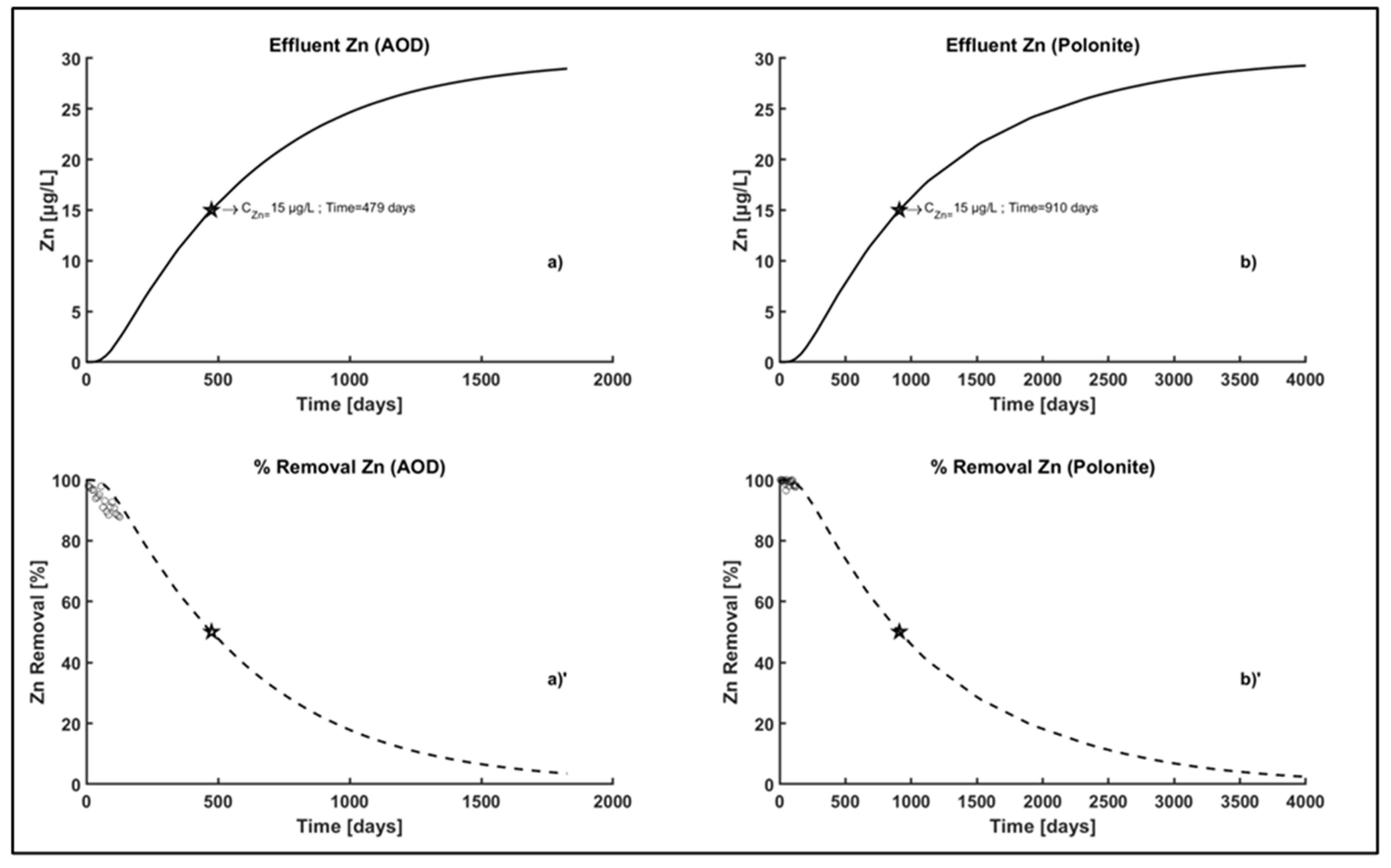
3.4.1. Profile of Zn Concentration and Percentage Removal
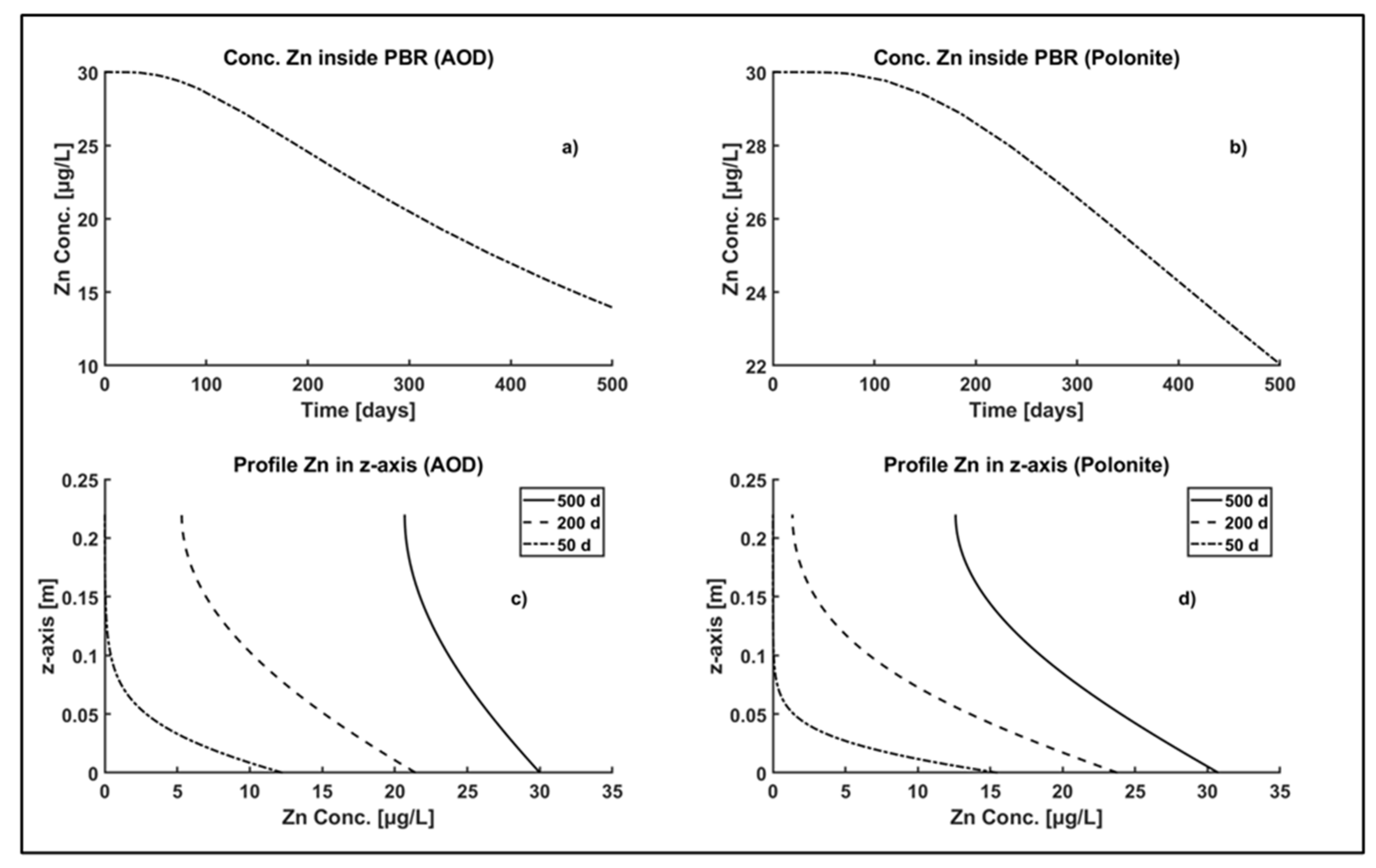
3.4.2. Profile of Zn Concentration
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balades, J.D.; Cathelain, M.; Marchandise, P.; Peybernard, J.; Pilloy, J.C. Chronic pollution of intercity motorway runoff waters. Water Sci. Technol. 1985, 17, 1165–1174. [Google Scholar] [CrossRef]
- Kayhanian, M.; Fruchtman, B.D.; Gulliver, J.S.; Montanaro, C.; Ranieri, E.; Wuertz, S. Review of highway runoff characteristics: Comparative analysis and universal implications. Water Res. 2012, 46, 6609–6624. [Google Scholar] [CrossRef]
- Huber, M.; Welker, A.; Helmreich, B. Critical review of heavy metal pollution of traffic area runoff: Occurrence, influencing factors, and partitioning. Sci. Total. Environ. 2016, 541, 895–919. [Google Scholar] [CrossRef]
- Spellerberg, I.A.N. Ecological effects of roads and traffic: A literature review. Glob. Ecol. Biogeogr. Lett. 1998, 7, 317–333. [Google Scholar] [CrossRef]
- Schuler, M.S.; Relyea, R.A. A review of the combined threats of road salts and heavy metals to freshwater systems. BioScience 2018, 68, 327–335. [Google Scholar] [CrossRef]
- Kazaz, B.; Poddar, S.; Arabi, S.; Perez, M.A.; Sharma, A.; Whitman, J.B. Deep Learning-Based Object Detection for Unmanned Aerial Systems (UASs)-Based Inspections of Construction Stormwater Practices. Sensors 2021, 21, 2834. [Google Scholar] [CrossRef] [PubMed]
- Sörme, L.; Lagerkvist, R. Sources of Heavy Metals in Urban Wastewater in Stockholm. Sci. Total. Environ. 2002, 298, 131–145. [Google Scholar] [CrossRef]
- Hallberg, M.; Renman, G. Removal of heavy metals from road runoff by filtration in granular slag columns. In Proceedings of the 11th Conference on Urban Drainage, Edinburgh, Scotland, 31 August–5 September 2008; pp. 1–11. [Google Scholar]
- Rentz, R.; Öhlander, B. Urban impact on water bodies in the Luleå area, Northern Sweden, and the role of redox processes. Hydrol. Res. 2012, 43, 917–932. [Google Scholar] [CrossRef]
- Al-Rubaei, A.; Engström, M.; Viklander, M.; Blecken, G. Long-term hydraulic and treatment performance of a 19-year old constructed stormwater wetland—Finally maturated or in need of maintenance? Ecol. Eng. 2016, 95, 73–82. [Google Scholar] [CrossRef]
- Huber, M.; Welker, A.; Dierschke, M.; Drewes, J.E.; Helmreich, B. A novel test method to determine the filter material service life of decentralized systems treating runoff from traffic areas. J. Environ. Manag. 2016, 179, 66–75. [Google Scholar] [CrossRef] [PubMed]
- European Parliament, Council of the European Union. Directive 2000/60/ECc of the European parliament and of the council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Commun. 2000, L327/1–L327/72. [Google Scholar]
- Dierkes, C.; Lucke, T.; Helmreich, B. General Technical Approvals for Decentralised Sustainable Urban Drainage Systems (SUDS)—The Current Situation in Germany. Sustainability 2015, 7, 3031–3051. [Google Scholar] [CrossRef] [Green Version]
- Herbert, R.; Björkvald, L.; Wällstedt, T.; Johansson, K. Background Levels of Metals in Swedish Inland and Coastal Waters; Report 2009:12; Department of Aquatic Sciences and Assessment, Swedish University of Agricultural Sciences: Uppsala, Sweden, 2009. (In Swedish) [Google Scholar]
- Winston, R.J.; Hunt, W.F.; Kennedy, S.G.; Merriman, L.S.; Chandler, J.; Brown, D. Evaluation of floating treatment wetlands as retrofits to existing stormwater retention ponds. Ecol. Eng. 2013, 54, 254–265. [Google Scholar] [CrossRef]
- Setälä, H.; Kuoppamäki, K.; Pflugmacher, L.; Scopetani, C. The ability of selected filter materials in removing nutrients, metals, and microplastics from stormwater in biofilter structures. J. Environ. Qual. 2021, 50, 465–475. [Google Scholar]
- Okaikue-Woodi, F.E.; Cherukumilli, K.; Ray, J.R. A critical review of contaminant removal by conventional and emerging media for urban stormwater treatment in the United States. Water Res. 2020, 187, 116434. [Google Scholar] [CrossRef] [PubMed]
- Behbahani, A.; Ryan, R.J.; McKenzie, E.R. Impacts of salinity on the dynamics of fine particles and their associated metals during stormwater management. Sci. Total. Environ. 2021, 493, 146135. [Google Scholar] [CrossRef]
- Björklund, K.; Li, L. Evaluation of low-cost materials for sorption of hydrophobic organic pollutants in stormwater. J. Environ. Manag. 2015, 159, 106–114. [Google Scholar] [CrossRef]
- Shafiquzzaman, M.; Haider, H.; Ghazaw, Y.M.; Alharbi, F.; S Al Saleem, S.; Almoshaogeh, M. Evaluation of a low-cost ceramic filter for sustainable reuse of urban stormwater in arid environments. Water 2020, 12, 460. [Google Scholar] [CrossRef] [Green Version]
- Deng, Y. Low-cost adsorbents for urban stormwater pollution control. Front. Environ. Sci. Eng. 2020, 14, 1–8. [Google Scholar] [CrossRef]
- Fronczyk, J.; Markowska-Lech, K. Treatment efficiency of synthetic urban runoff by low-cost mineral materials under various flow conditions and in the presence of salt: Possibilities and limitations. Sci. Total. Environ. 2021, 770, 145199. [Google Scholar] [CrossRef]
- Drizo, A.; Forget, C.; Chapuis, R.P.; Comeau, Y. Phosphorus removal by EAF steel slag—A parameter for the estimation of the longevity of constructed wetland systems. Environ. Sci. Technol. 2002, 36, 4642–4648. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Li, J.; Li, Y.; Chen, L. Field Performance of Bioretention Systems for Runoff Quantity Regulation and Pollutant Removal. Water Air Soil Pollut. 2017, 228, 1–13. [Google Scholar] [CrossRef]
- Meade, B.; Khorshidi, H.A.; Kandra, H.; Barton, A. Regression modelling for prediction of clogging in non-vegetated stormwater filters. In Proceedings of the 10th International Conference on Water Sensitive Urban Design: Creating Water Sensitive Communities (WSUD 2018 & Hydropolis 2018), Perth, Australia, 13 February 2018; Engineers Australia: Barton, Australia, 2018; p. 233. [Google Scholar]
- Chang, N.; Valencia, A.; Kilner, J.; Wanielista, M. Chemophysical Evaluation of Green Sorption Media for Copper Removal in Stormwater Runoff for Improving Ecosystem and Human Health. Water Air Soil Pollut. 2019, 230, 1–28. [Google Scholar]
- Jourak, A.; Frishfelds, V.; Lundström, T.S.; Herrmann, I.; Hedström, A. Modeling of phosphate removal by fitra P in fixed-bed columns. In Proceedings of the International Conference on Environmental Science and Technology, Dongguan, China, 26–28 February 2011; pp. 241–248. [Google Scholar]
- Behbahani, A.; Ryan, R.J.; McKenzie, E.R. Long-term simulation of potentially toxic elements (PTEs) accumulation and breakthrough in infiltration-based stormwater management practices (SMPs). J. Contam. Hydrol. 2020, 234, 103685. [Google Scholar] [CrossRef]
- Rodríguez-Gómez, R.; Renman, G. Phosphorus removal from UASB reactor effluent by reactive media filtration. Environ. Technol. 2017, 38, 2024–2031. [Google Scholar] [CrossRef]
- Kang, K.; Kim, J.; Kim, H.; Byun, I. Performance of carbon nanotube-coated steel slag filter media for the removal of phosphorus and heavy metal in stormwater. Desalination Water Treat. 2019, 149, 58–68. [Google Scholar] [CrossRef]
- Reddy, K.R.; Gopakumar, A.; Chetri, J.K. Critical review of applications of iron and steel slags for carbon sequestration and environmental remediation. Rev. Environ. Sci. Bio Technol. 2019, 18, 127–152. [Google Scholar] [CrossRef]
- Esfandiar, N.; Suri, R.; McKenzie, E.R. Simultaneous removal of multiple polycyclic aromatic hydrocarbons (PAHs) from urban stormwater using low-cost agricultural/industrial byproducts as sorbents. Chemosphere 2021, 274, 129812. [Google Scholar] [CrossRef]
- Hallberg, M.; Renman, G.; Lundbom, T. Seasonal variations of ten metals in highway runoff and their partition between dissolved and particulate matter. Water Air Soil Pollut. 2007, 181, 183–191. [Google Scholar] [CrossRef]
- Levec, J.; Nemeca, D. Flow through packed bed reactors: 1. Single-phase flow. Chem. Eng. Sci. 2005, 60, 6947–6957. [Google Scholar]
- Ryu, S.; Naidu, G.; Moon, H.; Vigneswaran, S. Continuous and selective copper recovery by multi-modified and granulated SBA-15. Chemosphere 2021, 271, 129820. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowski, A. Adsorption—From theory to practice. Adv. Colloid Interface Sci. 2001, 93, 135–224. [Google Scholar] [CrossRef]
- Muca, R.; Kołodziej, M.; Piątkowski, W.; Carta, G.; Antos, D. Effects of negative and positive cooperative adsorption of proteins on hydrophobic interaction chromatography media. J. Chromatogr. A 2020, 1625, 461309. [Google Scholar] [CrossRef]
- Koopal, L.K.; Van Riemsdijk, W.H.; De Wit, J.C.M.; Benedetti, M.F. Analytical isotherm equations for multicomponent adsorption to heterogeneous surfaces. J. Colloid Interface Sci. 1994, 166, 51–60. [Google Scholar] [CrossRef]
- Gary, R. Bleaching and Purifying Fats and Oils. In Theory and Practice, 2nd ed.; AOCS Press: Urbana, IL, USA, 2009; pp. 210–212. [Google Scholar]
- Zhou, M.; Li, Y. Phosphorus-sorption characteristics of calcareous soils and limestone from the southern Everglades and adjacent farmlands. Soil Sci. Soc. Am. J. 2001, 65, 1404–1412. [Google Scholar] [CrossRef] [Green Version]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2018, 148, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Febrianto, J.; Kosasih, A.N.; Sunarso, J.; Ju, Y.H.; Indraswati, N.; Ismadji, S. Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: A summary of recent studies. J. Hazard. Mater. 2009, 162, 616–645. [Google Scholar] [CrossRef]
- Renman, A.; Renman, G.; Gustafsson, J.P.; Hylander, L. Metal removal by bed filter materials used in domestic wastewater treatment. J. Hazard. Mater. 2009, 166, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Trenouth, W.R.; Gharabaghi, B. Soil amendments for heavy metals removal from stormwater runoff discharging to environmentally sensitive areas. J. Hydrol. 2015, 529, 1478–1487. [Google Scholar] [CrossRef]
- Renman, A.; Renman, G. Long-term phosphate removal by the calcium-silicate material Polonite in wastewater filtration systems. Chemosphere 2010, 79, 659–664. [Google Scholar] [CrossRef]
- Rostvall, A.; Zhang, W.; Dürig, W.; Renman, G.; Wiberg, K.; Ahrens, L.; Gago-Ferrero, P. Removal of pharmaceuticals, perfluoroalkyl substances and other micropollutants from wastewater using lignite, Xylit, sand, granular activated carbon (GAC) and GAC+ Polonite® in column tests–Role of physicochemical properties. Water Res. 2018, 137, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Kolosov, P.; Yargeau, V. Impact of catalyst load, chemical oxygen demand and nitrite on disinfection and removal of contaminants during catalytic ozonation of wastewater. Sci. Total. Environ. 2019, 651, 2139–2147. [Google Scholar] [CrossRef] [PubMed]
- Butkus, M.A.; Riegner, D.E. Lead stabilization by Polonite and phosphate amended Polonite: Modeling. J. Environ. Chem. Eng. 2016, 4, 3562–3569. [Google Scholar] [CrossRef]
- Froment, G.F.; Bischoff, K.B.; de Wilde, J. Chemical Reactor Design Analysis, 3rd ed.; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Bang, K.W.; Joo, J.C.; Kim, J.H.; Kang, E.; Choi, J.; Lee, J.M.; Kim, Y. Application of Bottom Ash as Filter Media for Construction Site Runoff Control. Water 2020, 12, 990. [Google Scholar] [CrossRef] [Green Version]
- Conley, G.; Beck, N.; Riihimaki, C.A.; Tanner, M. Quantifying clogging patterns of infiltration systems to improve urban stormwater pollution reduction estimates. Water Res. X 2020, 7, 100049. [Google Scholar] [CrossRef]
- Kandra, H.S.; McCarthy, D.; Fletcher, T.D.; Deletic, A. Assessment of clogging phenomena in granular filter media used for stormwater treatment. J. Hydrol. 2014, 512, 518–527. [Google Scholar] [CrossRef] [Green Version]
- Kandra, H.; McCarthy, D.T.; Deletic, A.; Zhang, K. Modelling the clogging of a field filtration system used for stormwater harvesting. Environ. Sci. Water Res. Technol. 2020, 6, 993–1003. [Google Scholar] [CrossRef]
- Wakida, F.T.; Martinez-Huato, S.; Garcia-Flores, E.; Piñon-Colin, T.D.J.; Espinoza-Gomez, H.; Ames-López, A. Pollutant association with suspended solids in stormwater in Tijuana, Mexico. Int. J. Environ. Sci. Technol. 2014, 11, 319–326. [Google Scholar] [CrossRef] [Green Version]
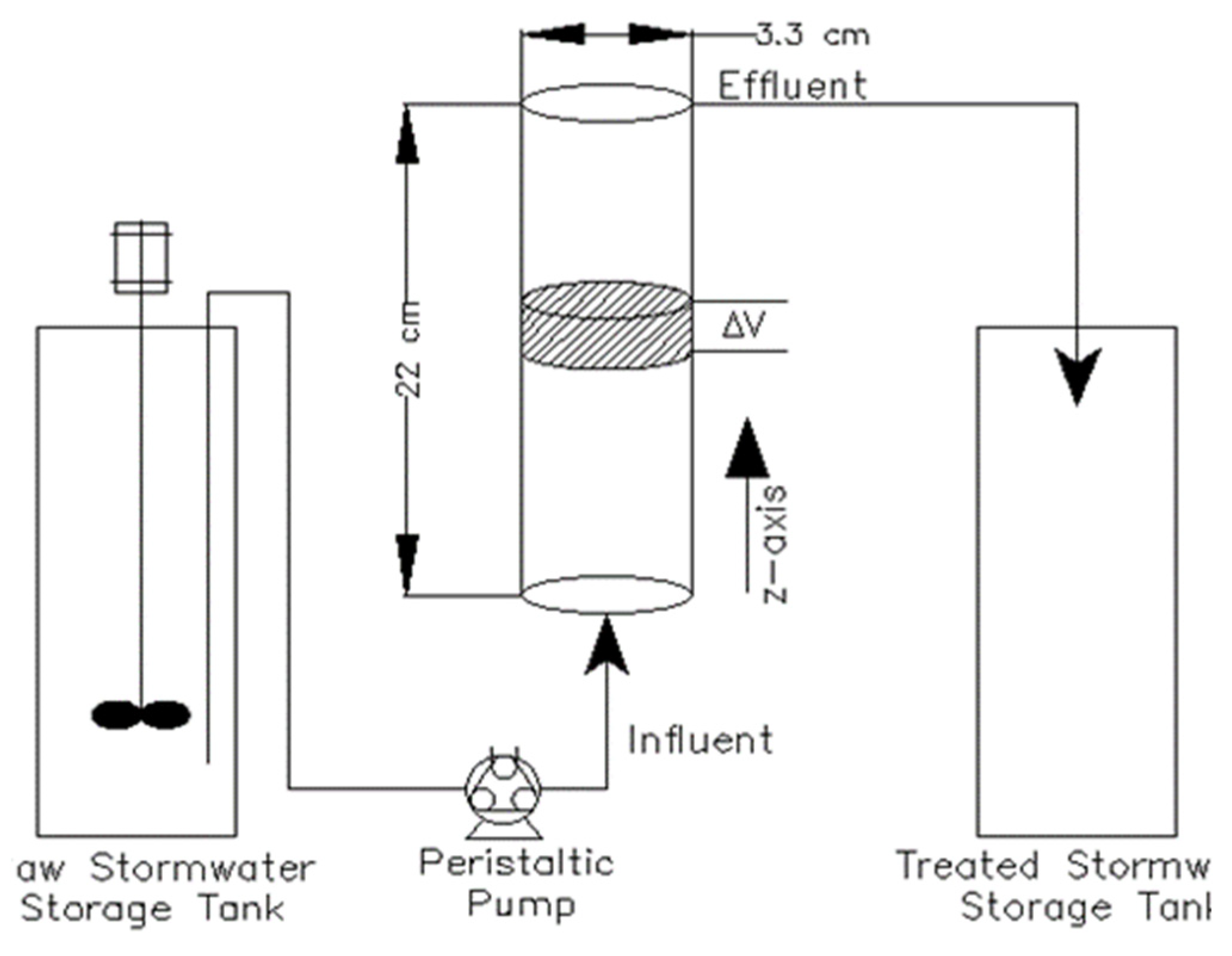
| Parameter | Symbol | Units | AOD | Polonite |
|---|---|---|---|---|
| Height of the PBR | m | 0.22 | 0.22 | |
| Diameter of the PBR | m | 3.3 × 10−2 | 3.3 × 10−2 | |
| Stormwater velocity | m/s | 1.35 × 10−3 | 1.35 × 10−3 | |
| Porosity | 0.595 | 0.45 | ||
| Density | Kg/m3 | 1180 | 730 | |
| Operation time | d | 126 | 126 | |
| Peclet number | 10 | 10 | ||
| Diffusion coefficient | m2/s |
| Filter Material [Metal] | Freundlich Parameter | Langmuir Parameter | ||||
|---|---|---|---|---|---|---|
| Kf [L/g] | n | R2 | a [mg/g] | b [L/mg] | R2 | |
| Polonite [Cu] | 0.1813 | 1.0801 | 0.9665 | 0.0549 | 4.8564 | 0.131 |
| AOD [Cu] | 0.0099 | 3.3069 | 0.8591 | 0.0043 | 40.485 | 0.4305 |
| Polonite [Zn] | 0.0613 | 1.5827 | 0.9794 | 0.0211 | 16.8472 | 0.8115 |
| AOD [Zn] | 0.1018 | 1.2552 | 0.9751 | 0.0393 | 6.4368 | 0.6924 |
| Filter Material [Metal] | Influent [μg/L] | Effluent [μg/L] | Removal [%] |
|---|---|---|---|
| AOD [Cu] | 16 | 2.80 | 82 |
| Polonite [Cu] | 16 | 2.30 | 86 |
| AOD [Zn] | 30 | 2.09 | 92 |
| Polonite [Zn] | 30 | 0.34 | 99 |
| Material | Cu Influent [μg/L] | Zn Influent [μg/L] | Cu Adsorption Rate [1/d] | Zn Adsorption Rate [1/d] |
|---|---|---|---|---|
| AOD | 16 | 30 | 0.18 | 0.35 |
| Polonite | 16 | 30 | 0.22 | 0.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Gómez, R.; Renman, A.; Mahmoudzadeh, B.; Renman, G. Copper and Zinc Removal Efficiency of Two Reactive Filter Media Treating Motorway Runoff—Model for Service Life Estimation. Water 2021, 13, 2592. https://doi.org/10.3390/w13182592
Rodríguez-Gómez R, Renman A, Mahmoudzadeh B, Renman G. Copper and Zinc Removal Efficiency of Two Reactive Filter Media Treating Motorway Runoff—Model for Service Life Estimation. Water. 2021; 13(18):2592. https://doi.org/10.3390/w13182592
Chicago/Turabian StyleRodríguez-Gómez, Raúl, Agnieszka Renman, Batoul Mahmoudzadeh, and Gunno Renman. 2021. "Copper and Zinc Removal Efficiency of Two Reactive Filter Media Treating Motorway Runoff—Model for Service Life Estimation" Water 13, no. 18: 2592. https://doi.org/10.3390/w13182592






