Shift towards Opportunistic Life-History of Sleeper in Response to Multi-Decadal Overfishing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fish Sampling and Processing
2.3. Statistical Analysis
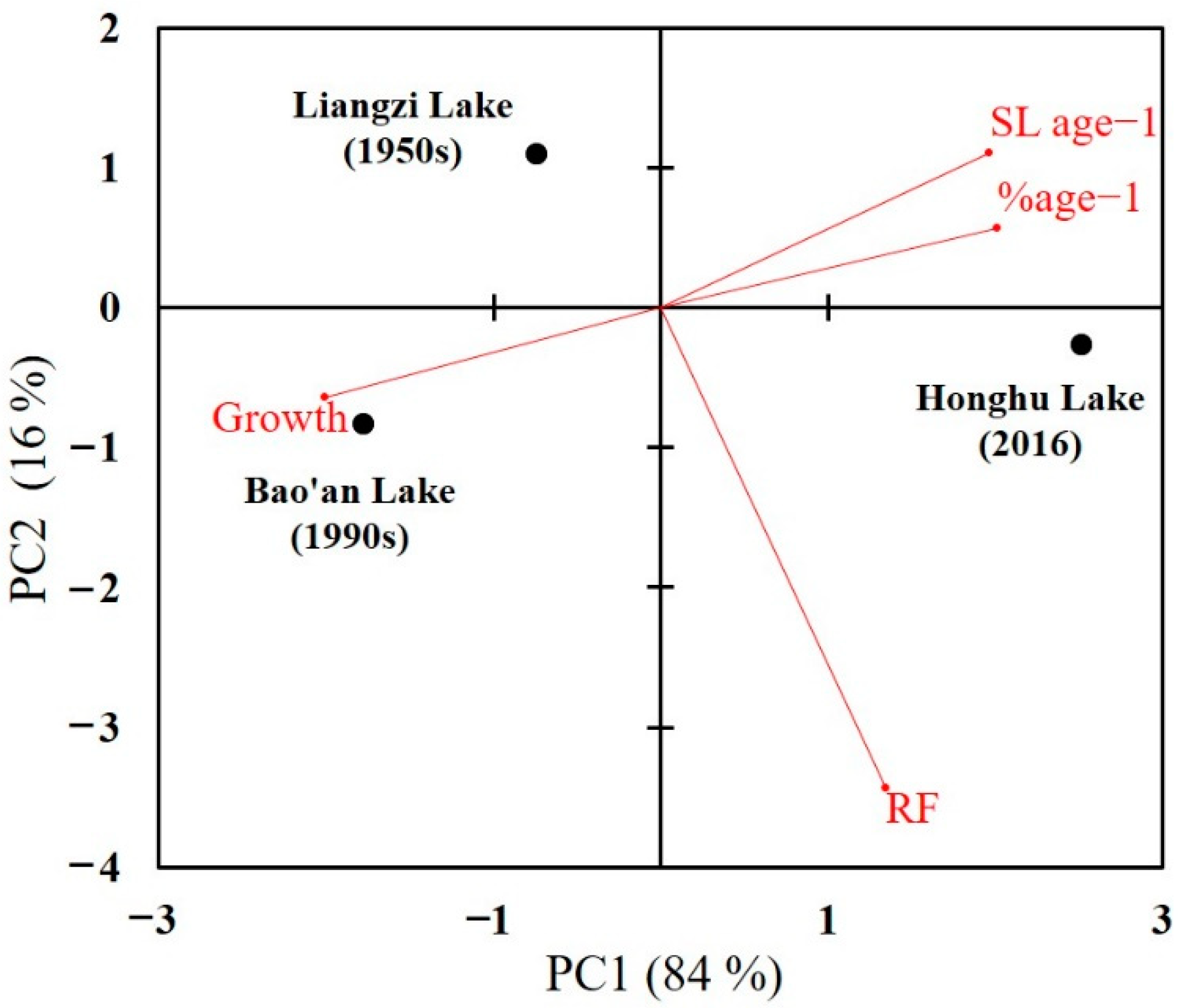
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pianka, E.R. On r- and K-selecion. Am. Nat. 1970, 104, 592–597. [Google Scholar] [CrossRef]
- Stearns, S.C. Evolution of Life Histories; Oxford University Press: Oxford, UK, 1992. [Google Scholar]
- Roff, D. Evoultion of Life Histories: Theory and Analysis; Chapman & Hall: New York, NY, USA, 1993. [Google Scholar]
- Rypel, A.L. Meta-analysis of growth rates for a circumpolar fish, the northern pike (Esox lucius), with emphasis on effects of continent, climate, and latitude. Ecol. Freshw. Fish 2012, 21, 521–532. [Google Scholar] [CrossRef]
- Rypel, A.L. The cold-water connection: Bergmann’s rule in north American freshwater fishes. Am. Nat. 2013, 183, 147–156. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, R.H.; Wilson, E.O. The Theory of Island Biogeography; Princeton University Press: Princeton, NJ, USA, 1967. [Google Scholar]
- Murphy, G.I. Pattern in life history and the environment. Am. Nat. 1968, 102, 391–403. [Google Scholar] [CrossRef]
- Stearns, S.C. Life-history tactics: A review of the ideas. Q. Rev. Biol. 1976, 51, 3–47. [Google Scholar] [CrossRef] [PubMed]
- Stearns, S.C. The evolution of life history traits: A critique of the theory and a review of the data. Annu. Rev. Eco. Syst. 1977, 8, 145–171. [Google Scholar] [CrossRef] [Green Version]
- Reznick, D.; Bryant, M.J.; Bashey, F. r- and K-selection revisited: The role of population regulation in life-history evolution. Ecology 2002, 83, 1509–1520. [Google Scholar] [CrossRef] [Green Version]
- Winemiller, K.O.; Rose, K.A. Patterns of life-history diversification in North American fishes: Implications for population regulation. Can. J. Fish. Aquat. Sci. 1992, 49, 2196–2218. [Google Scholar] [CrossRef]
- Juan-Jorda, M.J.; Mosqueira, I.; Freire, J.; Dulvy, N.K. Life in 3-D life history strategies in tunas, mackerels and bonitos. Rev. Fish Biol. Fish. 2013, 23, 135–155. [Google Scholar] [CrossRef]
- Olden, J.D.; Poff, N.L.; Bestgen, K.R. Life-history strategies predict fish invasions and extirpations in the Colorado River basin. Ecol. Monogr. 2006, 76, 25–40. [Google Scholar] [CrossRef] [Green Version]
- Riesch, R.; Martin, R.A.; Langerhans, R.B. Multiple traits and multifarious environments: Integrated divergence of morphology and life history. Oikos 2020, 129, 480–492. [Google Scholar] [CrossRef]
- Blanck, A.; Lamouroux, N. Large-scale intraspecific variation in life-history traits of European freshwater fish. J. Biogeogr. 2007, 34, 862–875. [Google Scholar] [CrossRef]
- Williams, E.H.; Shertzer, K.W. Effects of fishing on growth traits: A simulation analysis. Fish. Bull. 2005, 103, 392–403. [Google Scholar]
- Ziegler, P.E.; Lyle, J.M.; Haddon, M.; Ewing, G.P. Rapid changes in life-history characteristics of a long-live temperate reef fish. Mar. Freshwater Res. 2007, 58, 1096–1107. [Google Scholar] [CrossRef]
- Bishop, J.M.; Leslie, A.J.; Bourquin, S.L.; O’Ryan, C. Reduced effective population size in an overexploited population of the Nile crocodile (Crocodylus niloticus). Biol. Conserv. 2009, 142, 2335–2341. [Google Scholar] [CrossRef]
- He, W.P.; Li, Y.X.; Liu, M.; Radhakrishnan, K.V.; Li, Z.J.; Xie, S.G. Reproductive biology of Coilia mystus (Linnaeus) from the Yangtze Estuary, China: Responses to overexploitation. J. Appl. Ichthyol. 2011, 27, 1197–1202. [Google Scholar] [CrossRef]
- Ojuok, J.E.; Njiru, M.; Ntiba, M.J.; Mavuti, K.M. The effect of overfishing on the life-history strategies of Nile tilapia, Oreochromis niloticus (L.) in the Nyanza Gulf of Lake Victoria, Kenya. Aquat. Ecosyst. Health Manag. 2007, 10, 443–448. [Google Scholar] [CrossRef]
- Smith, V.H.; Schindler, D.W. Eutrophication science: Where do we go from here? Trends Ecol. Evol. 2009, 24, 201–207. [Google Scholar] [CrossRef]
- Crozier, L.G.; Hutchings, J.A. Plastic and evolutionary responses to climate change in fish. Evol. Appl. 2014, 7, 68–87. [Google Scholar] [CrossRef]
- Zhang, H.; Kang, M.; Li, S.; Wu, J.M.; Li, J.Y.; Du, H.; Wang, C.Y.; Yang, H.L.; Zhou, Q.; Liu, Z.G.; et al. Rapid changes in Yangtze fisheries and its implications for global freshwater ecosystem management. Fish Fish. 2020, 21, 601–620. [Google Scholar] [CrossRef]
- Chen, D.Q.; Duan, X.B.; Liu, S.P.; Shi, W.G.; Wang, B. On the dynamics of fisheries resources of the Yangtze River and its management. Acta Hydrobiol. Sin. 2002, 26, 685–690. [Google Scholar]
- He, F.Z.; Zarfl, C.; Bremerich, V.; David, J.N.W.; Hogan, Z.; Kalinkat, G.; Tockner, K.; Jaehnig, S.C. The global decline of freshwater megafauna. Glob. Chang. Biol. 2019, 25, 3883–3892. [Google Scholar] [CrossRef]
- Oke, K.B.; Cunningham, C.J.; Westley, P.A.H.; Baskett, M.L.; Carlson, S.M.; Clark, J.; Hendry, A.P.; Karatayev, V.A.; Kendall, N.W.; Kibele, J.; et al. Recent declines in salmon body size impact ecosystems and fisheries. Nat. Commun. 2020, 11, 4155. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.V.S.; Silva, W.M.M.L.; Novoa-Pabon, A.M.; Silva, H.M.; Pinho, M.R. Long-term changes in the diversity, abundance and size composition of deep sea demersal teleosts from the Azores assessed through surveys and commercial landings. Aquat. Living Resour. 2019, 32, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Cao, W.X.; Zhang, G.H.; Ma, J.; Yu, D.H. Preliminary studies on the phenomenon of size diminution of the fish resource in Lake Honghu. In Studies on Comprehensive Exploitation of Aquatic Biological Productivity and Improvement of Ecological Environment in Lake Honghu; China Ocean Press: Beijing, China, 1991. [Google Scholar]
- Chen, D.Q.; Xiong, F.; Wang, K.; Chang, Y.H. Status of research on Yangtze fish biology and fisheries. Environ. Biol. Fish. 2009, 85, 337–357. [Google Scholar] [CrossRef]
- Segurado, P.; Branco, P.; Jauch, E.; Neves, R.; Ferreira, M.T. Sensitivity of river fishes to climate change: The role of hydrological stressors on habitat range shifts. Sci. Total Environ. 2016, 562, 435–445. [Google Scholar] [CrossRef]
- Hutchings, J.A.; Kuparinen, A. Implications of fisheries-induced evolution for population recovery: Refocusing the science and refining its communication. Fish Fish. 2020, 21, 453–464. [Google Scholar] [CrossRef] [Green Version]
- Hao, T.H. An ecological study of a fresh-water goby Odontobutis obscura (Temm. et schl.) in Liangtze Lake of Hupei. Acta Hydrobiol. Sin. 1960, 2, 145–158. [Google Scholar]
- Zhu, B.K.; Xie, C.X.; Wang, M.X.; Jin, K. A study on feeding, reproduction, age and growth of dark sleeper Odontobutis obscura in Bao’an Lake. Acta Hydrobiol. Sin. 1999, 23, 316–323. [Google Scholar]
- Xie, S.G.; Cui, Y.B.; Zhang, T.L.; Fang, R.L.; Li, Z.J. The spatial pattern of the small fish community in the Biandantang Lake-a small shallow lake along the middle reach of the Yangtze River, China. Environ. Biol. Fish. 2000, 57, 170–190. [Google Scholar] [CrossRef]
- Institute of Hydrobiology. Fishes in the Yangtze River; Beijing Science Press: Beijing, China, 1976. [Google Scholar]
- Zhao, S.S.; Cheng, F.; Hou, G.; Hu, Z.Y.; Xie, S.G. Opportunistic-tended life history traits of Siniperca kneri in the Three Gorges Reservoir, China: Potential responses to impoundment. J. Oceanol. Limnol. 2018, 37, 694–705. [Google Scholar] [CrossRef]
- Wang, H.Z.; Liu, X.Q.; Wang, H.J. The Yangtze River Floodplain: Threats and rehabilitation. In Fishery Resources, Environment, and Conservation in the Mississippi and Yangtze; Chen, Y., Chapman, D.C., Jackson, J.R., Chen, D., Li, Z., Killgore, K.J., Phelps, Q., Eggleton, M.A., Eds.; American Fisheries Society: Bethesda, MD, USA, 2016; Volume 84, pp. 263–291. [Google Scholar]
- Guo, C.B.; Ye, S.W.; Lek, S.; Liu, J.S.; Zhang, T.L.; Yuan, J.; Li, Z.J. The need for improved fishery management in a shallow macrophytic lake in the Yangtze River Basin: Evidence from the food web structure and ecosystem analysis. Ecol. Model. 2013, 26, 137–147. [Google Scholar] [CrossRef]
- Guo, C.B.; Lek, S.; Ye, S.W.; Li, W.; Liu, J.S.; Chen, Y.S.; Li, Z.J. Predicting fish species richness and assemblages with climatic, geographic and morphometric factors: A broad-scale study in Chinese lakes. Limnologica 2015, 54, 66–74. [Google Scholar] [CrossRef]
- Guo, Z.Q.; Cucherousset, J.; Lek, S.; Li, Z.J.; Zhu, F.Y.; Tang, J.F.; Liu, J.S. Comparative study of the reproductive biology of two congeneric and introduced goby species: Implications for management strategies. Hydrobiologia 2013, 709, 89–99. [Google Scholar] [CrossRef] [Green Version]
- Cao, J.F.; Yang, X.F.; Yang, R.B.; Wei, K.J. Length-weight relationships and biological data of Odontobutis sinensis (Wu, Chen & Chong, 2002) from Liangzi Lake, China. J. Appl. Ichthyol. 2014, 31, 798–799. [Google Scholar] [CrossRef]
- Thiese, M.S.; Ronna, B.; Ott, U. P value interpretations and considerations. J. Thorac. Dci. 2016, 8, E928–E931. [Google Scholar] [CrossRef] [Green Version]
- Olsvik, P.A.; Larsen, A.K.; Berntssen, M.H.G.; Goksoyr, A.; Karlsen, O.A.; Yadetie, F.; Sanden, M.; Kristen, T. Effects of agricultural pesticides in aquafeeds on wild fish feeding on leftover pellets near fish farms. Front. Genet. 2019, 10, 1–18. [Google Scholar] [CrossRef]
- Xie, S.G.; Cui, Y.B.; Li, Z.J. Small fish communities in two regions of the Liangzi Lake, China, with or without submersed macrophytes. J. Appl. Ichthyol. 2001, 17, 89–92. [Google Scholar] [CrossRef]
- Lu, S.; Hu, J.H.; Xiao, C.Y.; Hu, H.J. Fish species composition in Lake Honghu and estimation of influencing factors. Chin. J. Wildlife 2006, 27, 14–17. [Google Scholar]
- Rypel, A.L.; Lyons, J.; Griffin, J.D.T.; Simonson, T.D. Seventy-year retrospective on size-structure changes in the recreational fisheries of Wisconsin. Fisheries 2016, 41, 230–243. [Google Scholar] [CrossRef]
- Huang, L.X. Flood water level along the main channel of the lower reaches of Yangtze River in the past 40 years: Variations and cause. J. Lake Sci. 1999, 11, 99–104. [Google Scholar]
- Dong, X.H.; Xiang, T.; Ju, T.; Li, R.J.; Ye, S.W.; Lek, S.; Liu, J.S.; Grenouillet, G. Age, growth, mortality and recruitment of thin sharpbelly Toxabramis swinhonis Günther, 1873 in three shallow lakes along the middle and lower reaches of the Yangtze River basin, China. PeerJ 2019, 7, e6772. [Google Scholar] [CrossRef] [Green Version]
- Zhu, F.Y.; Rypel, A.L.; Murphy, B.R.; Li, Z.J.; Zhang, T.L.; Yuan, J.; Guo, Z.Q.; Tang, J.F.; Liu, J.S. Rapid life-history diversification of an introduced fish species across a localized thermal gradient. PLoS ONE 2014, 9, e88033. [Google Scholar] [CrossRef]
- Heino, M.; Pauli, D.; Dieckmann, U. Fisheries-induced evolution. Annu. Rev. Ecol. Evol. Syst. 2015, 46, 461–480. [Google Scholar] [CrossRef] [Green Version]
- Dunlop, E.S.; Enberg, K.; Jorgensen, C.; Heino, M. Toward Darwinian fisheries management. Evol. Appl. 2009, 2, 246–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Embke, H.S.; Rypel, A.L.; Carpenter, S.R.; Greg, G.; Ogle, S.D.; Cichosz, T.; Hennessy, J.; Essington, T.E.; Vander Zanden, M.J. Production dynamics reveal hidden overharvest of inland recreational fisheries. Proc. Natl. Acad. Sci. USA 2019, 116, 24676–24681. [Google Scholar] [CrossRef] [PubMed]
- Rypel, A.L.; Goto, D.; Sass, G.G.; Vander Zanden, M.J. Production rates of walleye and their relationship to exploitation in Escanaba Lake, Wisconsin, 1965–2009. Can. J. Fish. Aquat. Sci. 2015, 72, 834–844. [Google Scholar] [CrossRef]
- Uusi-Heikkilä, S. Implications of size-selective fisheries on sexual selection. Evol. Appl. 2020, 13, 1487–1500. [Google Scholar] [CrossRef]
- Mims, M.C.; Olden, J.D. Life history theory predicts fish assemblage response to hydrologic regimes. Ecology 2012, 93, 35–45. [Google Scholar] [CrossRef]
- Tao, J.; He, D.K.; Kennard, M.J.; Ding, C.Z.; Bunn, S.E.; Liu, C.L.; Jia, Y.T.; Che, R.X.; Chen, Y.F. Strong evidence for changing fish reproductive phenology under climate warming on the Tibetan Plateau. Glob. Chang. Biol. 2018, 24, 2093–2104. [Google Scholar] [CrossRef] [Green Version]
- Li, X.Y.; Peng, S.S.; Deng, X.W.; Su, M.; Zeng, H. Attribution of lake warming in four shallow lakes in the middle and lower Yangtze River Basin. Environ. Sci. Technol. 2019, 53, 12548–12555. [Google Scholar] [CrossRef]
- Lin, M.; Chevalier, M.; Lek, S.; Zhang, L.; Gozlan, R.E.; Liu, J.; Zhang, T.; Ye, S.; Li, W.; Li, Z. Eutrophication as a driver of r-selection traits in a freshwater fish. J. Fish. Biol. 2014, 85, 343–354. [Google Scholar] [CrossRef]
- Jin, X.; Xu, Q.; Huang, C. Current status and future tendency of lake eutrophication in China. Sci. China Ser. C 2005, 48, 948–954. [Google Scholar]
- Rypel, A.L.; Goto, D.; Sass, G.G.; Vander Zanden, M.J. Eroding productivity of walleye populations in northern Wisconsin lakes. Can. J. Fish. Aquat. Sci. 2018, 75, 2291–2301. [Google Scholar] [CrossRef] [Green Version]
- Post, J.R.; Sullivan, M.; Cox, S.R.; Lester, N.P.; Walters, C.J.; Parkinson, E.A.; Paul, A.J.; Jackson, L.; Shuter, B.J. Canada’s recreational fisheries: The invisible collapse? Fisheries 2002, 27, 6–17. [Google Scholar] [CrossRef]
- Leidy, R.A.; Moyle, P.B. Conservation status of the world’s fish fauna: An overview. In Conservation Biology; Fiedler, P.L., Kareiva, P.M., Eds.; Springer: Boston, MA, USA, 1998; pp. 187–227. [Google Scholar] [CrossRef]
- Barausse, A.; Michieli, E.; Riginella, E.; Palmeri, L.; Mazzoldi, C. Long-term changes in community composition and life history traits in a highly exploited basin (northern Adriatic Sea): The role of environment and anthropogenic pressure. J. Fish Biol. 2011, 79, 1453–1486. [Google Scholar] [CrossRef] [PubMed]
- Sansom, B.J.; Bennett, S.J.; Atkinson, J.F.; Vaughn, C.C. Long-term persistence of freshwater mussel beds in labile river channels. Freshw. Biol. 2018, 63, 1469–1481. [Google Scholar] [CrossRef]
- Haimovici, M.; Cavole, L.M.; Cope, J.M.; Cardoso, L.G. Long-term changes in population dynamics and life history contribute to explain the resilience of a stock of Micropogonias furnieri (Sciaenidae, Teleostei) in the SW Atlantic. Fish. Res. 2021, 237, 105878. [Google Scholar] [CrossRef]
- Sass, G.G.; Rypel, A.L.; Stafford, J.D. Inland fisheries habitat management: Lessons learned from wildlife ecology and a proposal for change. Fisheries 2017, 42, 197–209. [Google Scholar] [CrossRef]
- Chen, Y.S.; Qu, X.; Xiong, F.Y.; Lu, Y.; Wang, L.Z.; Hughes, R.M. Challenges to saving China’s freshwater biodiversity: Fishery exploitation and landscape pressure. Ambio 2020, 49, 926–938. [Google Scholar] [CrossRef] [PubMed]



| Population | Year of Collection | Age Structure (Number Percentage, %) | Absolute Fecundity (Eggs) | Relative Fecundity (Eggs·g−1) | |||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | Range (Mean ± SD) | Range (Mean ± SD) | ||
| Liangzi Lake 1 | 1957 | 30.8 | 25.6 | 43.6 | 245–2505 | 21.8–43.3 | |
| Bao’an Lake | 1993–1994 | 14.2 | 43.7 | 30.9 | 11.3 | 540–1943 (1154 ± 317) | 33.3–63.3 (45.4 ± 7.5) |
| Honghu Lake | 2016 | 54.5 | 43.4 | 2.1 | 481–1869 (1053 ± 366) | 28.7–117.4 (58.0 ± 20.5) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiang, M.; Rypel, A.L.; Cheng, F.; Qin, J.; Zhang, L.; Chen, Y.; Xie, S. Shift towards Opportunistic Life-History of Sleeper in Response to Multi-Decadal Overfishing. Water 2021, 13, 2582. https://doi.org/10.3390/w13182582
Xiang M, Rypel AL, Cheng F, Qin J, Zhang L, Chen Y, Xie S. Shift towards Opportunistic Life-History of Sleeper in Response to Multi-Decadal Overfishing. Water. 2021; 13(18):2582. https://doi.org/10.3390/w13182582
Chicago/Turabian StyleXiang, Miao, Andrew L. Rypel, Fei Cheng, Jiao Qin, Lei Zhang, Yushun Chen, and Songguang Xie. 2021. "Shift towards Opportunistic Life-History of Sleeper in Response to Multi-Decadal Overfishing" Water 13, no. 18: 2582. https://doi.org/10.3390/w13182582






