Photocatalytic Dye Degradation and Biological Activities of Cu-Doped ZnSe Nanoparticles and Their Insights
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
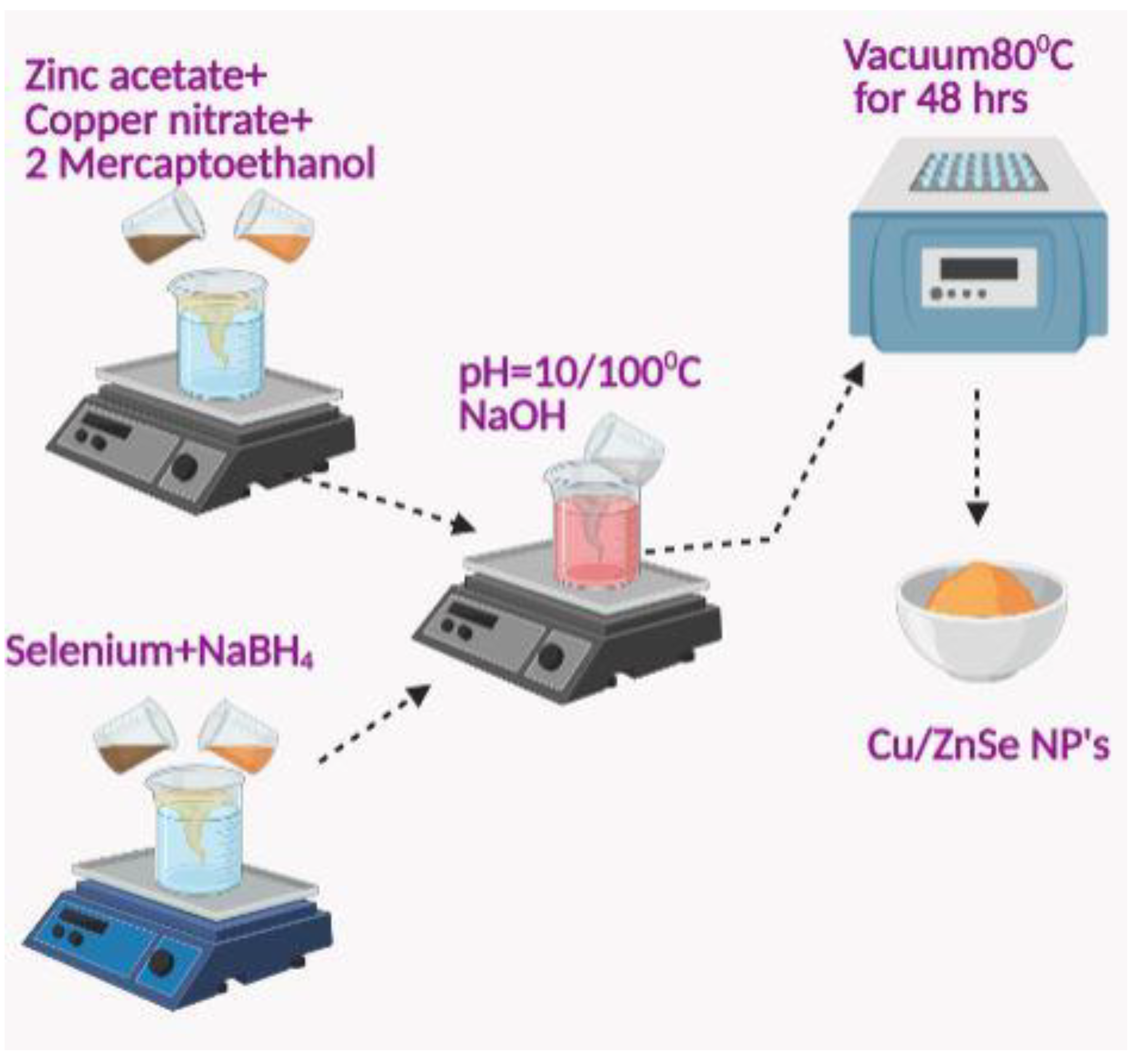
2.2. Synthesis Process
2.3. Characterization
2.4. Antibacterial Activity
2.5. Photocatalytic Dye Degradation
3. Results and Discussion
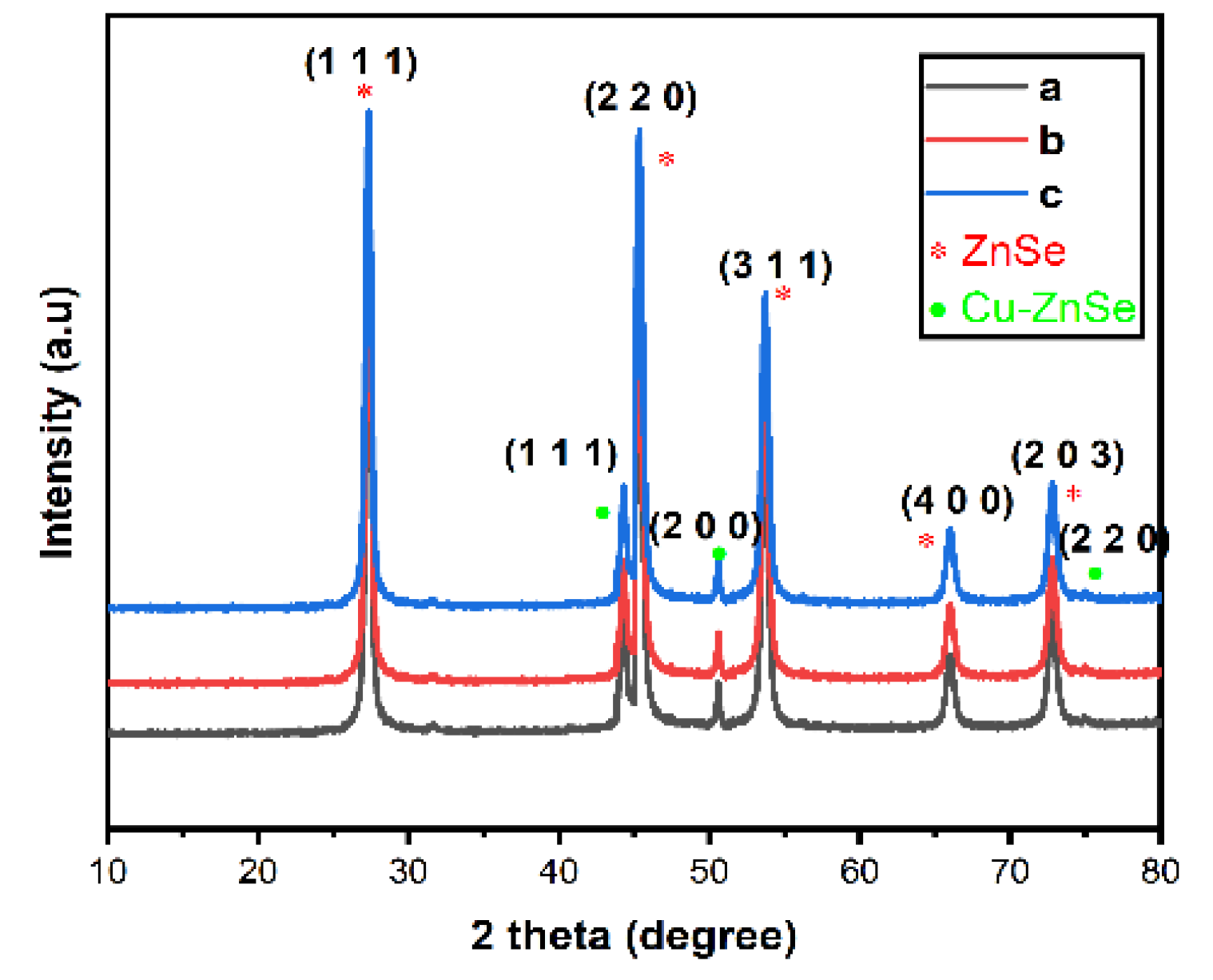
3.1. XRD Analysis
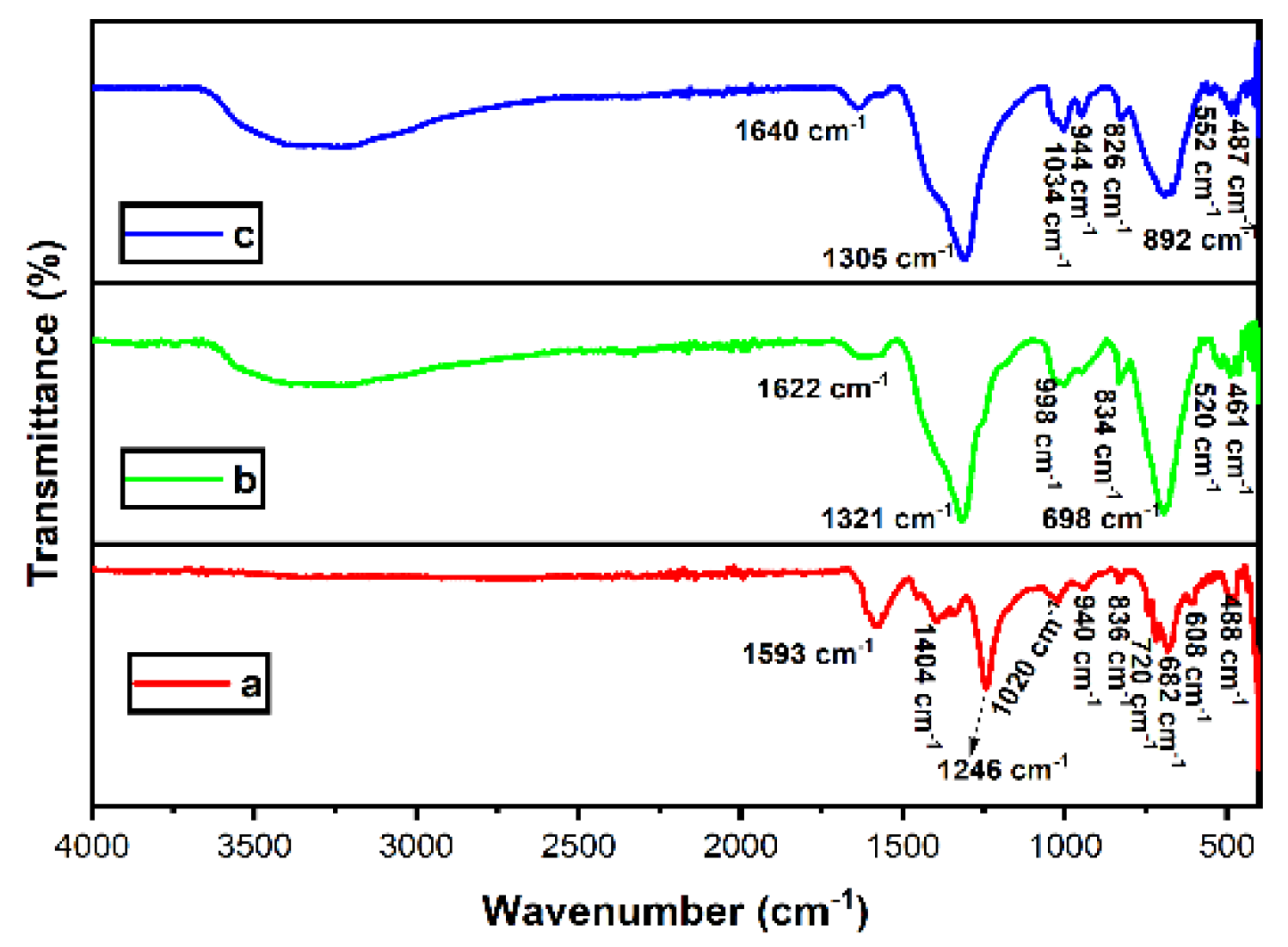
3.2. FTIR Analysis
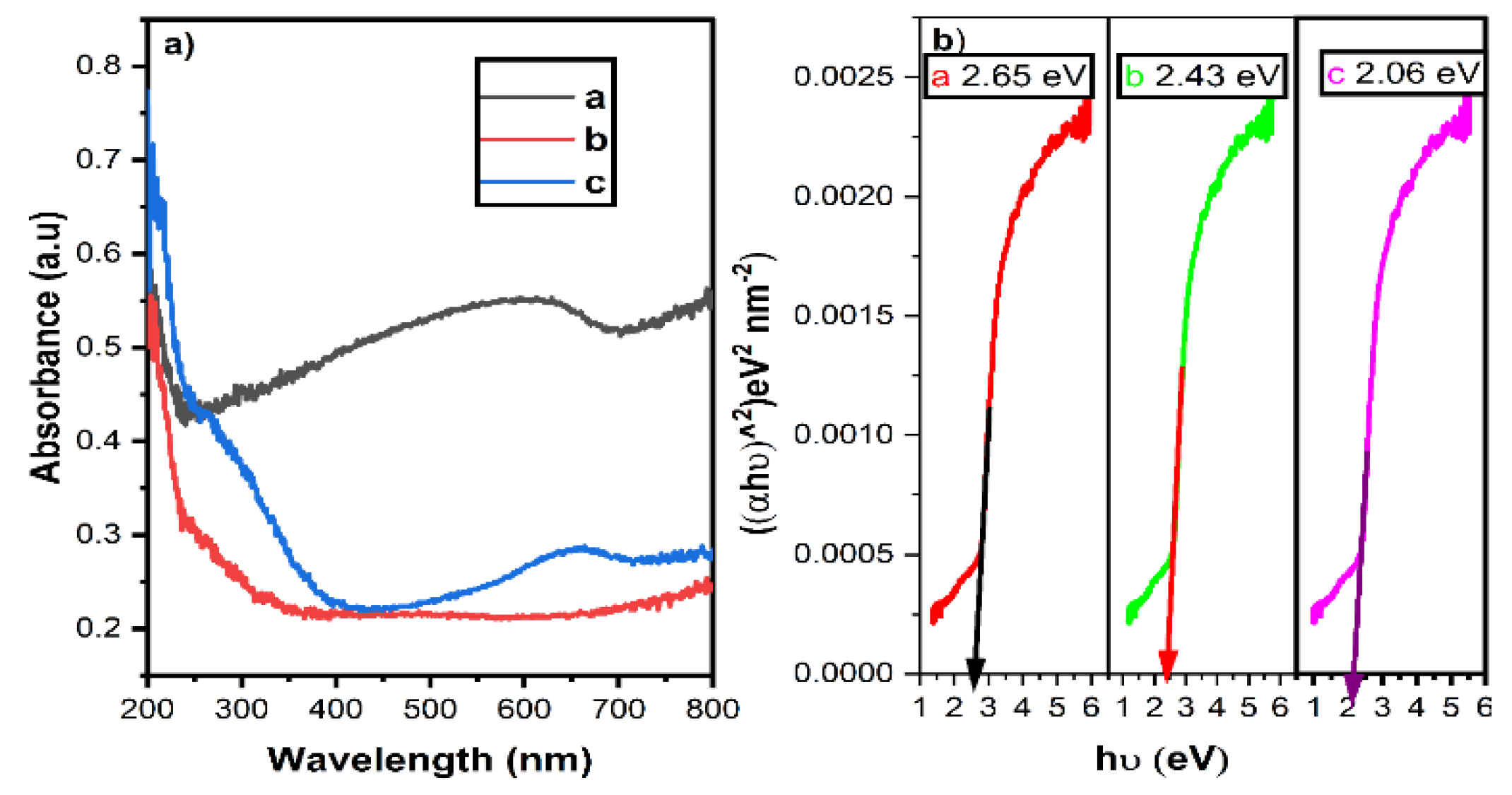
3.3. UV-DRS Analysis
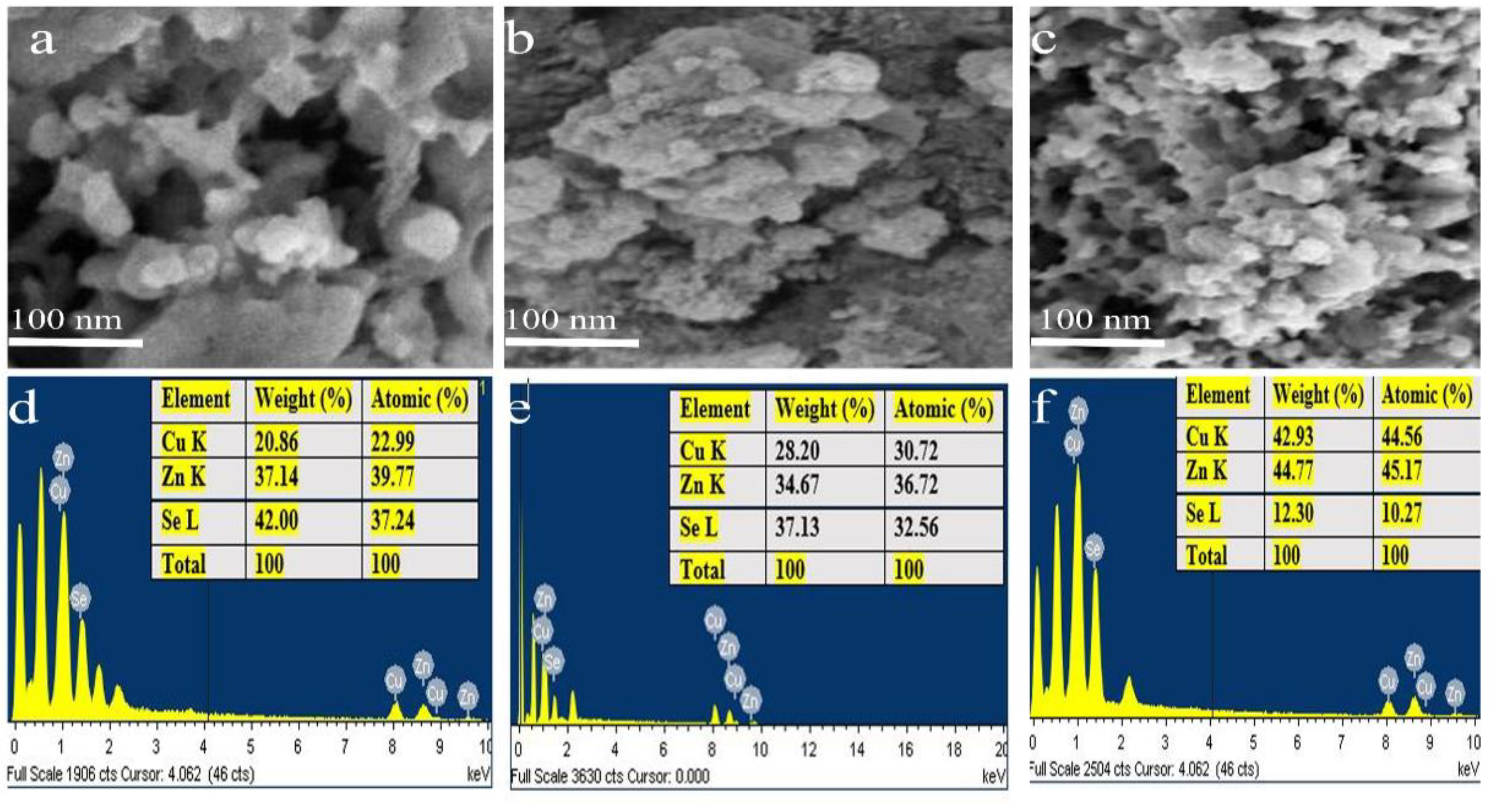
3.4. FE-SEM and EDX Analysis
3.5. HR-TEM Analysis
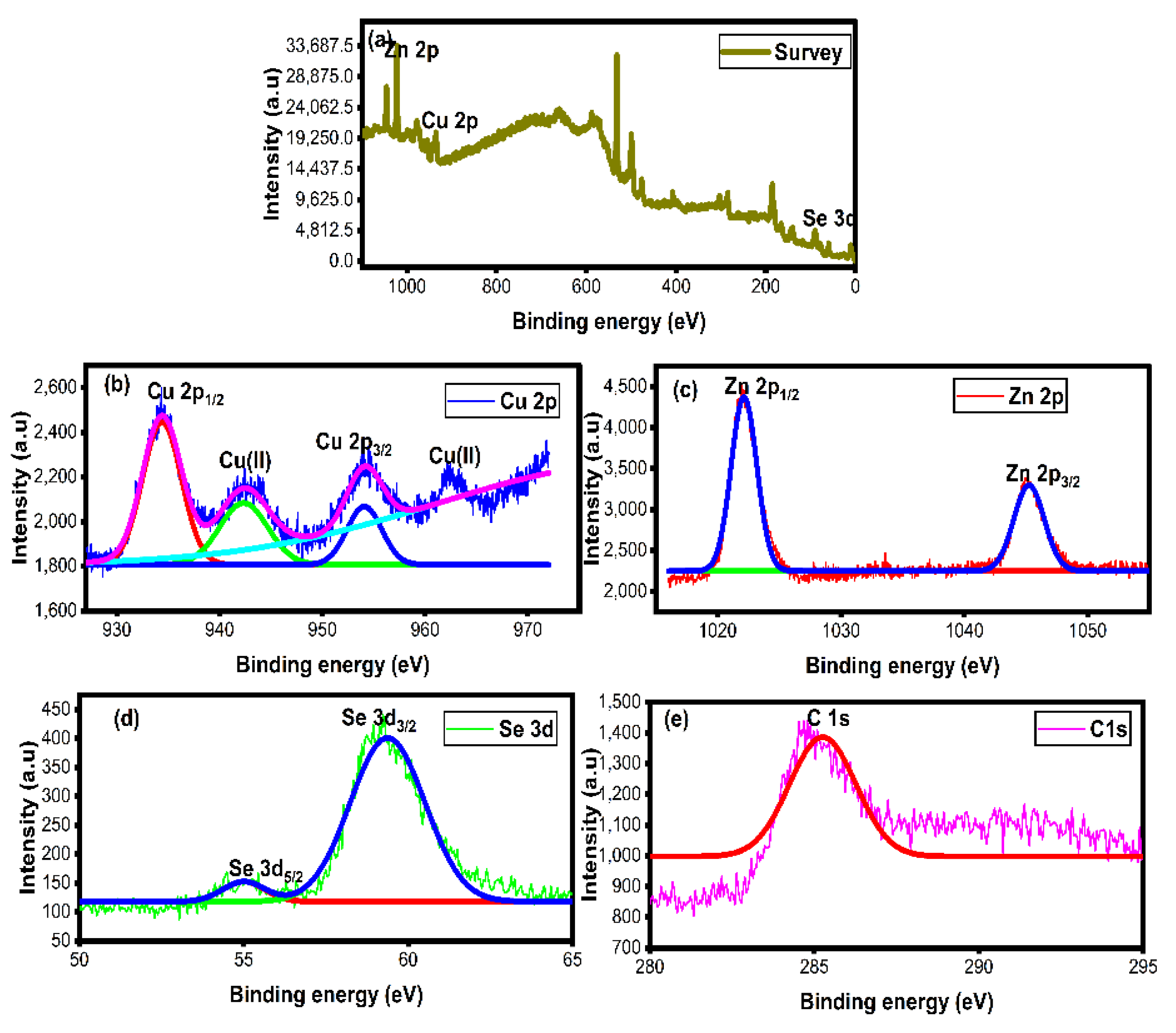
3.6. XPS Analysis
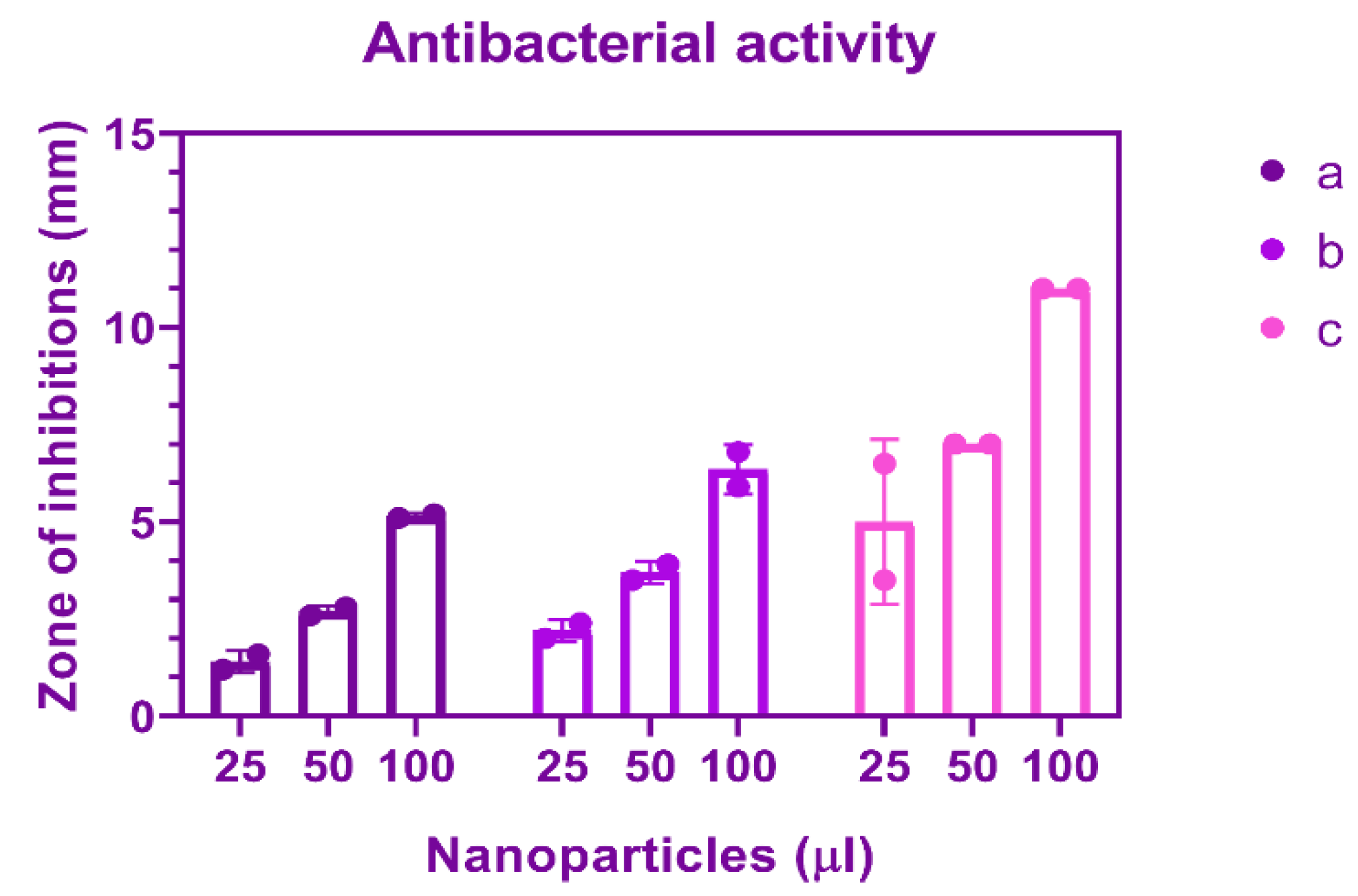
3.7. Antibacterial Activity
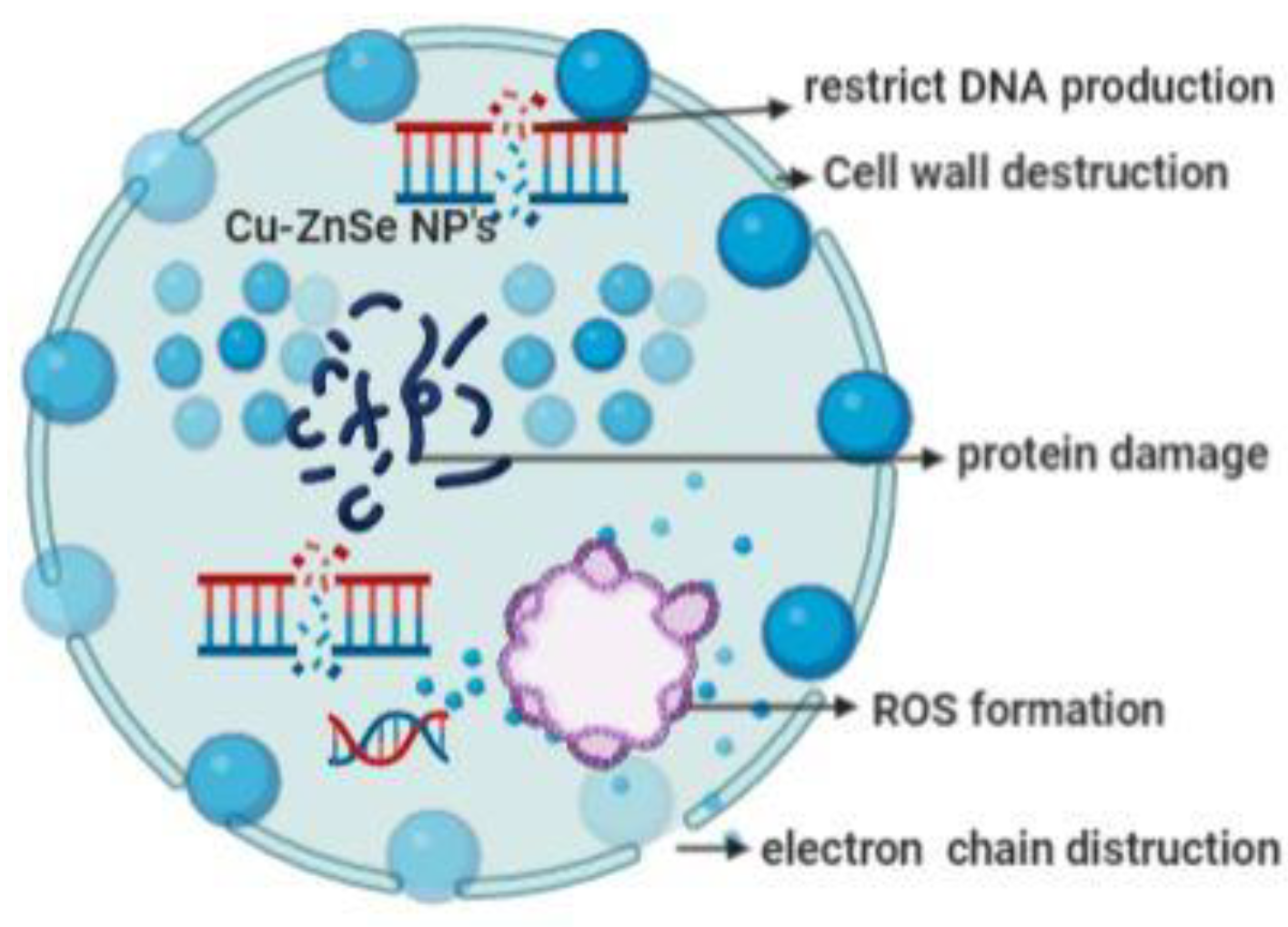
3.8. Mechanism of Antibacterial Activity of Cu-Doped ZnSe Nanoparticles
- Cell wall destruction;
- ROS formation (reactive oxygen species).
3.9. Photocatalytic Dye Degradation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Julkapli, N.M.; Bagheri, S.; Bee Abd Hamid, S. Recent Advances in Heterogeneous Photocatalytic Decolorization of Synthetic Dyes. Sci. World J. 2014, 2014, 692307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bibi, S.; Ahmad, A.; Anjum, M.A.R.; Haleem, A.; Siddiq, M.; Shah, S.S.; Al Kahtani, A. Photocatalytic degradation of malachite green and methylene blue over reduced graphene oxide (rGO) based metal oxides (rGO-Fe3O4/TiO2) nanocomposite under UV-visible light irradiation. J. Environ. Chem. Eng. 2021, 9, 105580. [Google Scholar] [CrossRef]
- Ahmad, I.; Jamal, M.A.; Iftikhar, M.; Ahmad, A.; Hussain, S.; Asghar, H.; Saeed, M.; Yousaf, A.B.; Karri, R.R.; Al-kadhi, N.S.; et al. Lanthanum-Zinc Binary Oxide Nanocomposite with Promising Heterogeneous Catalysis Performance for the Active Conversion of 4-Nitrophenol into 4-Aminophenol. Coatings 2021, 11, 537. [Google Scholar] [CrossRef]
- Kashif, M.; Jafaar, E.; Sahari, S.K.; Low, F.W.; Hoa, N.D.; Ahmad, A.; Abbas, A.; Ngaini, Z.; Shafa, M.; Qurashi, A. Organic sensitization of graphene oxide and reduced graphene oxide thin films for photovoltaic applications. Int. J. Energy Res. 2021, 45, 9657–9666. [Google Scholar] [CrossRef]
- Saleem, M.; Irfan, M.; Tabassum, S.; Albaqami, M.D.; Javed, M.S.; Hussain, S.; Pervaiz, M.; Ahmad, I.; Ahmad, A.; Zuber, M. Experimental and theoretical study of highly porous lignocellulose assisted metal oxide photoelectrodes for dye-sensitized solar cells. Arab. J. Chem. 2021, 14, 102937. [Google Scholar] [CrossRef]
- Aravind, M.; Ahmad, A.; Ahmad, I.; Amalanathan, M.; Naseem, K.; Mary, S.M.M.; Parvathiraja, C.; Hussain, S.; Algarni, T.S.; Zuber, M.P.M. Critical green routing synthesis of silver NPs using jasmine flower extract for biological activities and photocatalytical degradation of methylene blue. J. Environ. Chem. Eng. 2021, 9, 104877. [Google Scholar] [CrossRef]
- Hussain, S.; Khan, A.J.; Arshad, M.; Javed, M.S.; Ahmad, A.; Shah, S.S.A.; Khan, M.R.; Akram, S.; Zulfiqar; Ali, S.; et al. Charge storage in binder-free 2D-hexagonal CoMoO4 nanosheets as a redox active material for pseudocapacitors. Ceram. Int. 2021, 47, 8659–8667. [Google Scholar] [CrossRef]
- Beena, V.; Rayar, S.L.; Ajitha, S.; Ahmad, A.; Albaqami, M.D.; Alsabar, F.A.A.; Sillanpää, M. Synthesis and Characterization of Sr-Doped ZnSe Nanoparticles for Catalytic and Biological Activities. Water 2021, 13, 2189. [Google Scholar] [CrossRef]
- Ahmad, A.; Jini, D.; Aravind, M.; Parvathiraja, C.; Ali, R.; Kiyani, M.Z.; Alothman, A. A novel study on synthesis of egg shell based activated carbon for degradation of methylene blue via photocatalysis. Arab. J. Chem. 2020, 13, 8717–8722. [Google Scholar] [CrossRef]
- Vaez, Z.; Javanbakht, V. Synthesis, characterization and photocatalytic activity of ZSM-5/ZnO nanocomposite modified by Ag nanoparticles for methyl orange degradation. J. Photochem. Photobiol. A Chem. 2020, 388, 112064. [Google Scholar] [CrossRef]
- Deng, H.; Zhang, M.; Cao, Y.; Lin, Y. Decolorization of Reactive Black 5 by Mesoporous Al2O3 @TiO2 Nanocomposites. Environ. Prog. Sustain. Energy 2019, 38, S230–S242. [Google Scholar] [CrossRef]
- Rajendran, S.; Khan, M.M.; Gracia, F.; Qin, J.; Gupta, V.K.; Arumainathan, S. Ce3+-ion-induced visible-light photocatalytic degradation and electrochemical activity of ZnO/CeO2 nanocomposite. Sci. Rep. 2016, 6, 31641. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, A.F.; Arbuj, S.S.; Tamboli, M.S.; Naik, S.D.; Rane, S.B.; Kale, B.B. ZnSe/ZnO nano-heterostructures for enhanced solar light hydrogen generation. ChemistrySelect 2017, 2, 9174–9180. [Google Scholar] [CrossRef]
- Mkhalid, I.A. Improved photocatalytic performance in Bi2S3-ZnSe nanocomposites for hydrogen production. Ceram. Int. 2018, 44, 22198–22204. [Google Scholar] [CrossRef]
- Ehsan, M.F.; Bashir, S.; Hamid, S.; Zia, A.; Abbas, Y.; Umbreen, K.; Ashiq, M.N.; Shah, A. One-pot facile synthesis of the ZnO/ZnSe heterostructures for efficient photocatalytic degradation of azo dye. Appl. Surf. Sci. 2018, 459, 194–200. [Google Scholar] [CrossRef]
- Zhang, J.; Tian, P.; Tang, T.; Huang, G.; Zeng, J.; Cui, B.; Shen, Z.; Wang, H.; Kong, Z.; Xi, J.; et al. Excellent photoelectrochemical hydrogen evolution performance of FeSe2 nanorod/ZnSe 0D/1D heterostructure as efficiency carriers migrate channel. Int. J. Hydrogen Energy 2020, 45, 8526–8539. [Google Scholar] [CrossRef]
- Sanchez-Martinez, A.; Ortiz-Beas, J.P.; Huerta-Flores, A.M.; López-Mena, E.R.; Pérez-Álvarez, J.; Ceballos-Sanchez, O. ZnSe nanoparticles prepared by coprecipitation method for photocatalytic applications. Mater. Lett. 2021, 282, 128702. [Google Scholar] [CrossRef]
- Hao, E.; Zhang, H.; Yang, B.; Ren, H.; Shen, J. Preparation of Luminescent Polyelectrolyte/Cu-Doped ZnSe Nanoparticle Multilayer Composite Films. J. Colloid Interface Sci. 2001, 238, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Ummartyotin, S.; Bunnak, N.; Juntaro, J.; Sain, M.; Manuspiya, H. Synthesis and luminescence properties of ZnS and metal (Mn, Cu)-doped-ZnS ceramic powder. Solid State Sci. 2014, 14, 299–304. [Google Scholar] [CrossRef]
- Srivastava, R.K.; Pandey, N.; Mishra, S.K. Effect of Cu concentration on the photoconductivity properties of ZnS nanoparticles synthesized by co-precipitation method. Mater. Sci. Semicond. Process. 2013, 16, 1659–1664. [Google Scholar] [CrossRef]
- Modwi, A.; Taha, K.K.; Khezami, L.; Boudina, M.; Khairy, M.; Al-Duaij, O.K.; Talab, S. Dependence of the electrical properties of Cu-doped ZnO nanoparticles decorated by Ag atoms. Z. Phys. Chem. 2021, 235, 745–767. [Google Scholar] [CrossRef]
- Mittal, M.; Sharma, M.; Pandey, O.P. UV–Visible light induced photocatalytic studies of Cu doped ZnO nanoparticles prepared by co-precipitation method. Sol. Energy 2014, 110, 386–397. [Google Scholar] [CrossRef]
- Chauhan, R.; Kumar, A.; Chaudhary, R.P. Photocatalytic degradation of methylene blue with Cu doped ZnS nanoparticles. J. Lumin. 2014, 145, 6–12. [Google Scholar] [CrossRef]
- Senda, S.; Arai, T.; Sato, Y.; Shinoda, K.; Jeyadevan, B.; Tohji, K. Influence of Cu on the photocatalytic activity of ZnS nanoparticles. AIP Conf. Proc. 2006, 833, 65. [Google Scholar] [CrossRef]
- Beena, V.; Ajitha, S.; Rayar, L.S.; Parvathiraja, C.; Kannan, K.; Palani, G. Enhanced Photocatalytic and Antibacterial Activities of ZnSe Nanoparticles. J. Inorg. Organomet. Polym. Mater. 2021, 6, 1–12. [Google Scholar]
- Bezverkhyy, I.; Skrzypski, J.; Safonova, O.; Bellat, J.P. Sulfidation mechanism of pure and Cu-doped ZnO nanoparticles at moderate temperature: TEM and in situ XRD studies. J. Phys. Chem. C 2012, 116, 14423–14430. [Google Scholar] [CrossRef]
- Hao, H.; Yao, X.; Wang, M. Preparation and optical characteristics of ZnSe nanocrystals doped glass by sol–gel in situ crystallization method. Opt. Mater. 2007, 29, 573–577. [Google Scholar] [CrossRef]
- Eren, E.; Afsin, B. An investigation of Cu (II) adsorption by raw and acid-activated bentonite: A combined potentiometric, thermodynamic, XRD, IR, DTA study. J. Hazard. Mater. 2008, 151, 682–691. [Google Scholar] [CrossRef]
- Chen, R.; Guo, C.; Chu, W.; Jiang, N.; Li, H. ATR-FTIR study of Bacillus sp. and Escherichia coli settlements on the bare and Al2O3 coated ZnSe internal reflection element. Chin. Chem. Lett. 2019, 30, 115–119. [Google Scholar] [CrossRef]
- Majumdar, S.S.; Das, S.K.; Saha, T.; Panda, G.C.; Bandyopadhyoy, T.; Guha, A.K. Adsorption behavior of copper ions on Mucor rouxii biomass through microscopic and FTIR analysis. Colloids Surf. B Biointerfaces 2008, 63, 138–145. [Google Scholar] [CrossRef]
- Hines, M.A.; Guyot-Sionnest, P. Bright UV-blue luminescent colloidal ZnSe nanocrystals. J. Phys. Chem. B. 1998, 102, 3655–3657. [Google Scholar] [CrossRef]
- Giordanino, F.; Vennestrøm, P.N.; Lundegaard, L.F.; Stappen, F.N.; Mossin, S.; Beato, P.; Bordiga, S.; Lamberti, C. Characterization of Cu-exchanged SSZ-13: A comparative FTIR, UV-Vis, and EPR study with Cu-ZSM-5 and Cu-β with similar Si/Al and Cu/Al ratios. Dalton Trans. 2013, 42, 12741–12761. [Google Scholar] [CrossRef]
- Mallem, S.P.R.; Koduru, M.; Chandrasekhar, K.; Prabhakar Vattikuti, S.V.; Manne, R.; Reddy, V.R.; Lee, J.H. Potato Chip-Like 0d Interconnected Znco2o4 Nanoparticles High-Perform. Supercapacitors. Crystals 2021, 11, 469. [Google Scholar] [CrossRef]
- Shakir, M.; Kushwaha, S.K.; Maurya, K.K.; Bhagavannarayana, G.; Wahab, M.A. Characterization of ZnSe nanoparticles synthesized by microwave heating process. Solid State Commun. 2009, 149, 2047–2049. [Google Scholar] [CrossRef]
- Liang, Q.; Bai, Y.; Han, L.; Deng, X.; Wu, X.; Wang, Z.; Liu, X.; Meng, J. Hydrothermal synthesis of ZnSe: Cu quantum dots and their luminescent mechanism study by first-principles. J. Lumin. 2013, 143, 185–192. [Google Scholar] [CrossRef]
- Ghodselahi, T.; Vesaghi, M.A.; Shafiekhani, A.; Baghizadeh, A.; Lameii, M. XPS study of the Cu@ Cu2O core-shell nanoparticles. Appl. Surf. Sci. 2008, 255, 2730–2734. [Google Scholar] [CrossRef]
- Feliu, S., Jr.; Barranco, V.X.P.S. XPS study of the surface chemistry of conventional hot-dip galvanised pure Zn, galvanneal and Zn–Al alloy coatings on steel. Acta Mater. 2003, 51, 5413–5424. [Google Scholar] [CrossRef]
- Agostinelli, E.; Battistoni, C.; Fiorani, D.; Mattogno, G.; Nogues, M. An XPS study of the electronic structure of the ZnxCd1− xCr2 (X = S, Se) spinel system. J. Phys. Chem. Solids 1989, 50, 269–272. [Google Scholar] [CrossRef]
- Nigussie, G.Y.; Tesfamariam, G.M.; Tegegne, B.M.; Weldemichel, Y.A.; Gebreab, T.W.; Gebrehiwot, D.G.; Gebremichel, G.E. Antibacterial activity of Ag-doped TiO2 and Ag-doped ZnO nanoparticles. Int. J. Photoenergy 2018, 2018, 5927485. [Google Scholar] [CrossRef] [Green Version]
- Chaliha, C.; Nath, B.K.; Verma, P.K.; Kalita, E. Synthesis of functionalized Cu: ZnS nanosystems and its antibacterial potential. Arab. J. Chem. 2019, 12, 515–524. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beena, V.; Rayar, S.L.; Ajitha, S.; Ahmad, A.; Iftikhar, F.J.; Abualnaja, K.M.; Alomar, T.S.; Ouladsmne, M.; Ali, S. Photocatalytic Dye Degradation and Biological Activities of Cu-Doped ZnSe Nanoparticles and Their Insights. Water 2021, 13, 2561. https://doi.org/10.3390/w13182561
Beena V, Rayar SL, Ajitha S, Ahmad A, Iftikhar FJ, Abualnaja KM, Alomar TS, Ouladsmne M, Ali S. Photocatalytic Dye Degradation and Biological Activities of Cu-Doped ZnSe Nanoparticles and Their Insights. Water. 2021; 13(18):2561. https://doi.org/10.3390/w13182561
Chicago/Turabian StyleBeena, V., S. L. Rayar, S. Ajitha, Awais Ahmad, Faiza Jan Iftikhar, Khamael M. Abualnaja, Taghrid S. Alomar, Mohmed Ouladsmne, and Shafaqat Ali. 2021. "Photocatalytic Dye Degradation and Biological Activities of Cu-Doped ZnSe Nanoparticles and Their Insights" Water 13, no. 18: 2561. https://doi.org/10.3390/w13182561
APA StyleBeena, V., Rayar, S. L., Ajitha, S., Ahmad, A., Iftikhar, F. J., Abualnaja, K. M., Alomar, T. S., Ouladsmne, M., & Ali, S. (2021). Photocatalytic Dye Degradation and Biological Activities of Cu-Doped ZnSe Nanoparticles and Their Insights. Water, 13(18), 2561. https://doi.org/10.3390/w13182561







