Estimating the Importance of Hydrologic Conditions on Nutrient Retention and Plant Richness in a Wetlaculture Mesocosm Experiment in a Former Lake Erie Basin Swamp
Abstract
:1. Introduction
2. Methods
2.1. Site Description
2.2. Wetland Mesocosm Experiments
2.2.1. Hydrologic Experiment
2.2.2. Sampling and Analysis of Water
2.2.3. Vegetation Survey
2.3. Calculation and Statistics
2.3.1. Water Quality Removal Rates and Fluxes
2.3.2. Statistical Analyses
3. Results
3.1. Hydrologic Regime and Budgets
3.2. Temporal Patterns of Nutrients in the Wetland Mesocosms
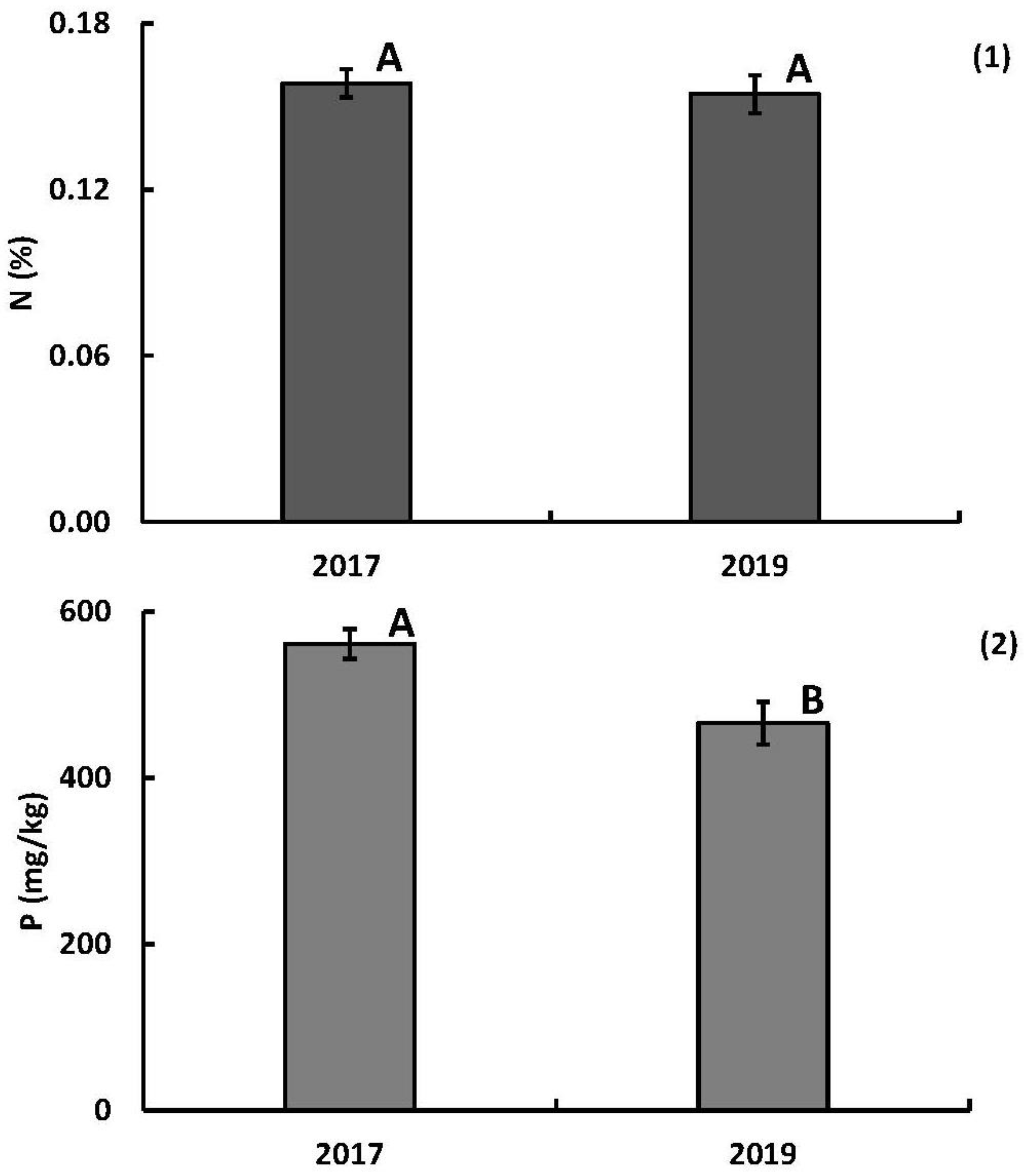
3.2.1. Annual Nutrient Concentrations
3.2.2. Yearly and Monthly Nutrient Removal Efficiency
3.3. Effects of Hydrologic Conditions on Nutrient Retention
3.3.1. Removal Efficiency
3.3.2. Mass Retention
3.4. Vegetation Cover and Species Richness
4. Discussion
4.1. Nutrient Retention by Treatment Wetlands
4.2. Role of Water Level in Treatment Wetlands
4.3. Role of Hydraulic Loading Rate in Treatment Wetlands
4.4. Response and Contribution of Wetland Plants in Treatment Wetlands
4.5. Nutrient Accumulation in Soils
4.6. Implication of Future Wetlaculture Research
- The below- and above-ground biomass of plants and nutrient concentrations in plants;
- A profile of nutrient concentrations in the new accreted sediment of the topsoil;
- The impact of draining the mesocosm wetlands in the winter;
- Annual decomposition rates of plant detritus.
5. Conclusions
- These mesocosm wetlands created with hydric soils (Hoytville clay) left behind by the drained Black Great Black Swamp in northwestern Ohio and upstream of the western basin of Lake Erie became nutrient sinks almost immediately, with average removal efficiencies of TP and TN of 60% and 28% in 2018, and of 83% and 54% in 2019, respectively.
- The combination of a high loading rate and 10 cm of standing water achieved the best phosphorus and nitrogen removal efficiencies through the two-year hydroperiod, averaging 76% and 60%, respectively.
- During the two-year period, the average net mass retention of phosphorus from water sampling was estimated to be 1.0 g P m−2 in the wetland mesocosms with a higher hydraulic loading rate, while the highest estimated net nitrogen mass retention (average 22 g N m−2) was shown in the wetland mesocosms with standing water and higher hydraulic loading rates. The mass retention of P in the high loading rate treatment was almost three times the retention of the low loading rate treatment.
- A total of eleven new species were identified as volunteering in the wetland mesocosms to supplement the planted S. tabernaemontani. The coverage and species richness of wetland plants were both higher in the standing water treatments than in the no standing water treatment.
- This study has established that the wetlaculture mesocosms under a variety of loading rates and water depths effectively removed phosphorus and nitrogen from agricultural runoff. Future wetland research in these or other mesocosm experiments should consider intersystem transformations such as plant decomposition, plant root nutrient retention, sediment retention, and other nutrient transformations, in addition to considering water quality changes.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bellmore, R.A.; Compton, J.E.; Brooks, J.R.; Fox, E.W.; Hill, R.A.; Sobota, D.J.; Thornbrugh, D.J.; Weber, M.H. Nitrogen inputs drive nitrogen concentrations in US streams and rivers during summer low flow conditions. Sci. Total Environ. 2018, 639, 1349–1359. [Google Scholar] [CrossRef]
- Hamilton, H.A.; Ivanova, D.; Stadler, K.; Merciai, S.; Schmidt, J.; Van Zelm, R.; Moran, D.; Wood, R. Trade and the role of non-food commodities for global eutrophication. Nat. Sustain. 2018, 1, 314. [Google Scholar] [CrossRef]
- Harrison, S.; McAree, C.; Mulville, W.; Sullivan, T. The problem of agricultural ‘diffuse’pollution: Getting to the point. Sci. Total Environ. 2019, 677, 700–717. [Google Scholar] [CrossRef]
- Sharpley, A.; Wang, X. Managing agricultural phosphorus for water quality: Lessons from the USA and China. J. Environ. Sci. 2014, 26, 1770–1782. [Google Scholar] [CrossRef] [PubMed]
- Odum, H.T.; Ewel, K.C.; Mitsch, W.J.; Ordway, J.W. Recycling treated sewage through cypress wetlands. In Wastewater Renovation and Reuse; F.M. D’Itri ed.; Marcel Dekker Press: New York, NY, USA, 1977; pp. 35–67. [Google Scholar]
- Kadlec, R.H.; Reddy, K. Temperature effects in treatment wetlands. Water Environ. Res. 2001, 73, 543–557. [Google Scholar] [CrossRef]
- Nichols, D.S. Capacity of natural wetlands to remove nutrients from wastewater. J. Water Pollut. Control. Fed. 1983, 55, 495–505. [Google Scholar]
- Land, M.; Granéli, W.; Grimvall, A.; Hoffmann, C.C.; Mitsch, W.J.; Tonderski, K.S.; Verhoeven, J.T. How effective are created or restored freshwater wetlands for nitrogen and phosphorus removal? A systematic review. Environ. Evid. 2016, 5, 9. [Google Scholar] [CrossRef] [Green Version]
- Jarvie, H.P.; Johnson, L.T.; Sharpley, A.N.; Smith, D.R.; Baker, D.B.; Bruulsema, T.W.; Confesor, R. Increased soluble phosphorus loads to Lake Erie: Unintended Consequences of Conservation Practices? J. Environ. Qual. 2017, 46, 123–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kane, D.D.; Conroy, J.D.; Richards, R.P.; Baker, D.B.; Culver, D.A. Re-eutrophication of Lake Erie: Correlations between tributary nutrient loads and phytoplankton biomass. J. Great Lakes Res. 2014, 40, 496–501. [Google Scholar] [CrossRef]
- Michalak, A.M.; Anderson, E.J.; Beletsky, D.; Boland, S.; Bosch, N.S.; Bridgeman, T.B.; Chaffin, J.D.; Cho, K.; Confesor, R.; Daloğlu, I.; et al. Record-setting algal bloom in Lake Erie caused by agricultural and meteorological trends consistent with expected future conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 6448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartig, J.H.; Krantzberg, G.; Alsip, P. Thirty-five years of restoring Great Lakes Areas of Concern: Gradual progress, hopeful future. J. Great Lakes Res. 2020, 46, 429–442. [Google Scholar] [CrossRef]
- Ohio Environmental Protection Agency(EPA), Ohio Department of Natural Resources. Ohio Nutrient Reduction Strategy 2015 Addendum; U.S. EPA: Washington, DC, USA, 2016; p. 15.
- Horvath, E.K.; Christensen, J.R.; Mehaffey, M.H.; Neale, A.C. Building a potential wetland restoration indicator for the contiguous United States. Ecol. Indic. 2017, 83, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Mitsch, W.J. Solving Lake Erie’s harmful algal blooms by restoring the Great Black Swamp in Ohio. Ecol. Eng. 2017, 108, 406–413. [Google Scholar] [CrossRef]
- Mitsch, W.J. Solving Harmful Algal Blooms: Progress in 2016–2017 at a Wetlaculture Experiment at Buckeye Lake; Ohio Wetland Association Wetland Trumpeter Newsletter. Ohio Wetland Association: Amherst, OH, USA, 2017; pp. 8, 9, 11. [Google Scholar]
- Jiang, B.; Mitsch, W.J. Influence of hydrologic conditions on nutrient retention, and soil and plant development in a former central Ohio swamp: A wetlaculture mesocosm experiment. Ecol. Eng. 2020, 157, 105969. [Google Scholar] [CrossRef]
- Devito, K.J.; Dillon, P.J. The influence of hydrologic conditions and peat oxia on the phosphorus and nitrogen dynamics of a conifer swamp. Water Resour. Res. 1993, 29, 2675–2685. [Google Scholar] [CrossRef]
- Marton, J.M.; Creed, I.F.; Lewis, D.B.; Lane, C.R.; Basu, N.B.; Cohen, M.J.; Craft, C.B. Geographically isolated wetlands are important biogeochemical reactors on the landscape. Bioscience 2015, 65, 408–418. [Google Scholar] [CrossRef] [Green Version]
- Ahn, C.; Mitsch, W.J. Scaling considerations of mesocosm wetlands in simulating large created freshwater marshes. Ecol. Eng. 2002, 18, 327–342. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Zhang, L.; Marois, D.; Song, K. Protecting the Florida Everglades wetlands with wetlands: Can stormwater phosphorus be reduced to oligotrophic conditions? Ecol. Eng. 2015, 80, 8–19. [Google Scholar] [CrossRef]
- Marois, D.E.; Mitsch, W.J. Modeling phosphorus retention at low concentrations in Florida Everglades mesocosms. Ecol. Model. 2016, 319, 42–62. [Google Scholar] [CrossRef]
- Messer, T.L.; Burchell II, M.R.; Birgand, F.; Broome, S.W.; Chescheir, G. Nitrate removal potential of restored wetlands loaded with agricultural drainage water: A mesocosm scale experimental approach. Ecol. Eng. 2017, 106, 541–554. [Google Scholar] [CrossRef]
- Balster, L. Black Swamp Savior: How Bringing Back Conquered Wetlands Could Help Solve Harmful Algal Blooms. Environmental Monitor, Fondriest Environmental, Fairborn, OH. Available online: https://www.fondriest.com/news/black-swamp-savior-how-bringing-back-conquered-wetlands-could-help-solve-harmful-algal-blooms.htm (accessed on 20 May 2020).
- Scavia, D.; DePinto, J.V.; Bertani, I. A multi-model approach to evaluating target phosphorus loads for Lake Erie. J. Great Lakes Res. 2016, 42, 1139–1150. [Google Scholar] [CrossRef]
- Femeena, P.; Sudheer, K.; Cibin, R.; Chaubey, I. Spatial optimization of cropping pattern for sustainable food and biofuel production with minimal downstream pollution. J. Environ. Manag. 2018, 212, 198–209. [Google Scholar] [CrossRef]
- Kieta, K.A.; Owens, P.N.; Lobb, D.A.; Vanrobaeys, J.A.; Flaten, D.N. Phosphorus dynamics in vegetated buffer strips in cold climates: A review. Environ. Rev. 2001, 26, 255–272. [Google Scholar] [CrossRef]
- National Oceanic and Atmospheric Administration, Climate Data Oline. Available online: https://www.ncdc.noaa.gov/cdoweb/datasets/GSOM/stations/GHCND:USC00332098/detail (accessed on 4 May 2020).
- Mitsch, W.J.; Gosselink, J. Wetlands, 5th ed.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 111–160. [Google Scholar]
- American Public Health Association, APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 1998. [Google Scholar]
- United State Environmental Protection Agency, USEPA. Methods for the Determination of Inorganic Substances in Environmental Samples. Method 353.2: Determination of Nitrate-Nitrite by automated Colorimetry (Revision 2.0); USEPA: Cincinnati, OH, USA, 1993.
- United State Environmental Protection Agency, USEPA. Methods for the Determination of Inorganic Substances in Environmental Samples. Method 365.1: Determination of Phosphorus by automated Colorimetry (Revision 2.0); USEPA: Cincinnati, OH, USA, 1993.
- United State Environmental Protection Agency, USEPA. Methods for the Determination of Inorganic Substances in Environmental Samples. Method 351.2: Determination of Total Kjeldahl Nitrogen by Semi-automated Colorimetry (Revision 2.0); USEPA: Cincinnati, OH, USA, 1993.
- Reed, P.B. National list of plant species that occur in wetlands: Northeast (Region 1); Report no. 88-26.1; Department of the Interior, Fish and Wildlife Service, Research and Development: Washington, DC, USA, 1988. [Google Scholar]
- Song, K.; Kang, H.; Zhang, L.; Mitsch, W.J. Seasonal and spatial variations of denitrification and denitrifying community structure in created wetlands. Ecol. Eng. 2012, 38, 130–134. [Google Scholar] [CrossRef]
- Song, K.; Hernandez, M.E.; Batson, J.A.; Mitsch, W.J. Long-term denitrification rates in created riverine wetlands and their relationship with environmental factors. Ecol. Eng. 2014, 72, 40–46. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Zhang, L.; Stefanik, K.C.; Nahlik, A.M.; Anderson, C.J.; Bernal, B.; Hernandez, M.; Song, K. Creating wetlands: Primary succession, water quality changes, and self-design over 15 years. Bioscience 2012, 62, 237–250. [Google Scholar] [CrossRef] [Green Version]
- Mitsch, W.J.; Zhang, L.; Waletzko, E.; Bernal, B. Validation of the ecosystem services of created wetlands: Two decades of plant succession, nutrient retention, and carbon sequestration in experimental riverine marshes. Ecol. Eng. 2014, 72, 11–24. [Google Scholar] [CrossRef]
- Schultz, R.; Andrews, S.; O’Reilly, L.; Bouchard, V.; Frey, S. Plant community composition more predictive than diversity of carbon cycling in freshwater wetlands. Wetlands 2011, 31, 965. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Bouchard, V.; Zhang, L.; Hunter, M. Biogeochemical and nutrient removal patterns of created riparian wetlands: Sixth-year results. In Olentangy River Wetland Research Park at The Ohio State University, Annual Report 1999; The Ohio State University: Columbus, OH, USA, 2000; pp. 83–90. [Google Scholar]
- Clément, J.-C.; Shrestha, J.; Ehrenfeld, J.G.; Jaffé, P.R. Ammonium oxidation coupled to dissimilatory reduction of iron under anaerobic conditions in wetland soils. Soil Biol. Biochem. 2005, 37, 2323–2328. [Google Scholar] [CrossRef]
- Cronk, J.K.; Fennessy, M.S. Wetland plants: Biology and Ecology; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Dierberg, F.E.; DeBusk, T.A.; Jackson, S.D.; Chimney, M.J.; Pietro, K. Submerged aquatic vegetation-based treatment wetlands for removing phosphorus from agricultural runoff: Response to hydraulic and nutrient loading. Water Res. 2002, 36, 1409–1422. [Google Scholar] [CrossRef]
- Engelhardt, K.A.; Ritchie, M.E. Effects of macrophyte species richness on wetland ecosystem functioning and services. Nature 2001, 411, 687. [Google Scholar] [CrossRef] [PubMed]
- Fraser, L.H.; Carty, S.M.; Steer, D. A test of four plant species to reduce total nitrogen and total phosphorus from soil leachate in subsurface wetland microcosms. Bioresour. Technol. 2004, 94, 185–192. [Google Scholar] [CrossRef]
- Hopfensperger, K.N. A review of similarity between seed bank and standing vegetation across ecosystems. Oikos 2007, 116, 1438–1448. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Kadlec, J.A. Wetlands and water quality. In Wetlands Functions and Values: The State of Our Understanding; Greeson, P.E., Clark, J.R., Clark, J.E., Eds.; American Water Resources Association: Minneapolis, MN, USA, 1979. [Google Scholar]
- Mitsch, W.J. Experimental wetlaculture (wetlands + agriculture) mesocosm compound established in Naples, Florida, to restore wetlands, solve harmful algal blooms, and develop sustainable agriculture. Wetland Sci. Pract. 2018, 35, 33–34. [Google Scholar]
- Mitsch, W.J.; Horne, A.J.; Nairn, R.W. Nitrogen and phosphorus retention in wetlands-ecological approaches to solving excess nutrient problems. Ecol. Eng. 2000, 14, 1–7. [Google Scholar]
- Pezeshki, S.R.; DeLaune, R.D. Soil oxidation-reduction in wetlands and its impact on Plant Functioning. Biology 2012, 1, 196–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, J.R.; DeLaune, R.D.; Justic, D.; Day, J.W.; Pahl, J.; Lane, R.R.; Boynton, W.R.; Twilley, R.R. Consequences of Mississippi River diversions on nutrient dynamics of coastal wetland soils and estuarine sediments: A review. Estuar. Coast. Shelf Sci. 2019, 224, 209–216. [Google Scholar] [CrossRef]
- Neff, K.P.; Rusello, K.; Baldwin, A.H. Rapid seed bank development in restored tidal freshwater wetlands. Restor. Ecol. 2009, 17, 539–548. [Google Scholar] [CrossRef]
- Pavelic, P.; Dillon, P.; Mucha, M.; Nakai, T.; Barry, K.; Bestland, E. Laboratory assessment of factors affecting soil clogging of soil aquifer treatment systems. Water Res. 2011, 45, 3153–3163. [Google Scholar] [CrossRef]
- Ridolfi, L.; D’Odorico, P.; Laio, F. Effect of vegetation–water table feedbacks on the stability and resilience of plant ecosystems. Water Resour. Res. 2006, 42, W01201. [Google Scholar] [CrossRef] [Green Version]
- Alsadi, N. Treatment Wetland Vegetation Harvesting for Phosphorus Removal in Upper Midwest Agricultural Water-sheds. Ph.D. Thesis, University of Minnesota, Minneapolis, MN, USA, December 2019. [Google Scholar]
- Gordon, B.A.; Lenhart, C.; Peterson, H.; Gamble, J.; Nieber, J.; Current, D.; Brenke, A. Reduction of nutrient loads from agricultural subsurface drainage water in a small, edge-of-field constructed treatment wetland. Ecol. Eng. 2021, 160, 106128. [Google Scholar] [CrossRef]
- Baker, D.B.; Johnson, L.T.; Confesor, R.B.; Crumrine, J.P. Vertical stratification of soil phosphorus as a concern for dissolved phosphorus runoff in the Lake Erie ba-sin. J. Environ. Qual. 2017, 4, 1287–1295. [Google Scholar] [CrossRef] [Green Version]
- Shao, W.; Zhu, J.; Teng, Z.; Zhang, K.; Liu, S.; Li, M. Distribution of inorganic phosphorus and its response to the physicochemical characteristics of soil in Yeyahu Wetland, China. Process Saf. Environ. Protect. 2019, 125, 1–8. [Google Scholar] [CrossRef]
- Thullen, J.S.; Nelson, S.M.; Cade, B.S.; Sartoris, J.J. Macrophyte decomposition in a surface-flow ammonia-dominated constructed wetland: Rates associated with environmental and biotic variables. Ecol. Eng. 2008, 32, 281–290. [Google Scholar] [CrossRef]
- Boutin, K.D.; Mitsch, W.J.; Everham, E.; Bakshi, B.R.; Zhang, L. An evaluation of corn production within a Wetlaculture™ system at Buckeye Lake, Ohio. Ecol. Eng. 2021, 171, 106366. [Google Scholar] [CrossRef]
- Lenhart, C.F.; Lenhart, P.C. Restoration of wetland and prairie on farmland in the former Great Black Swamp of Ohio, USA. Ecol. Restor. 2014, 32, 441–449. [Google Scholar] [CrossRef]

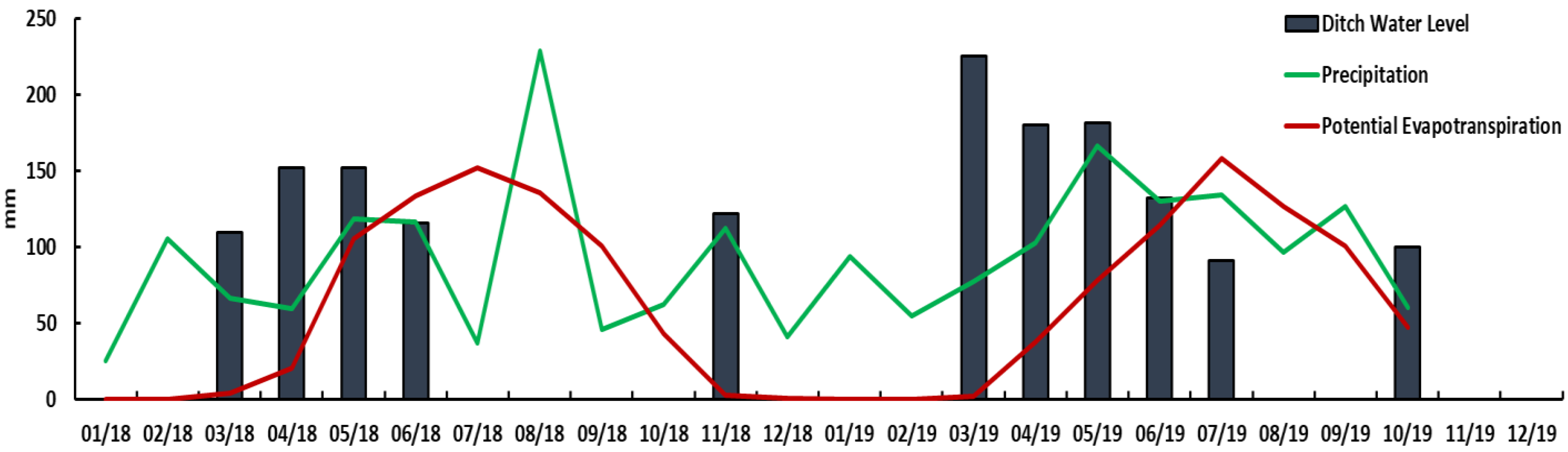
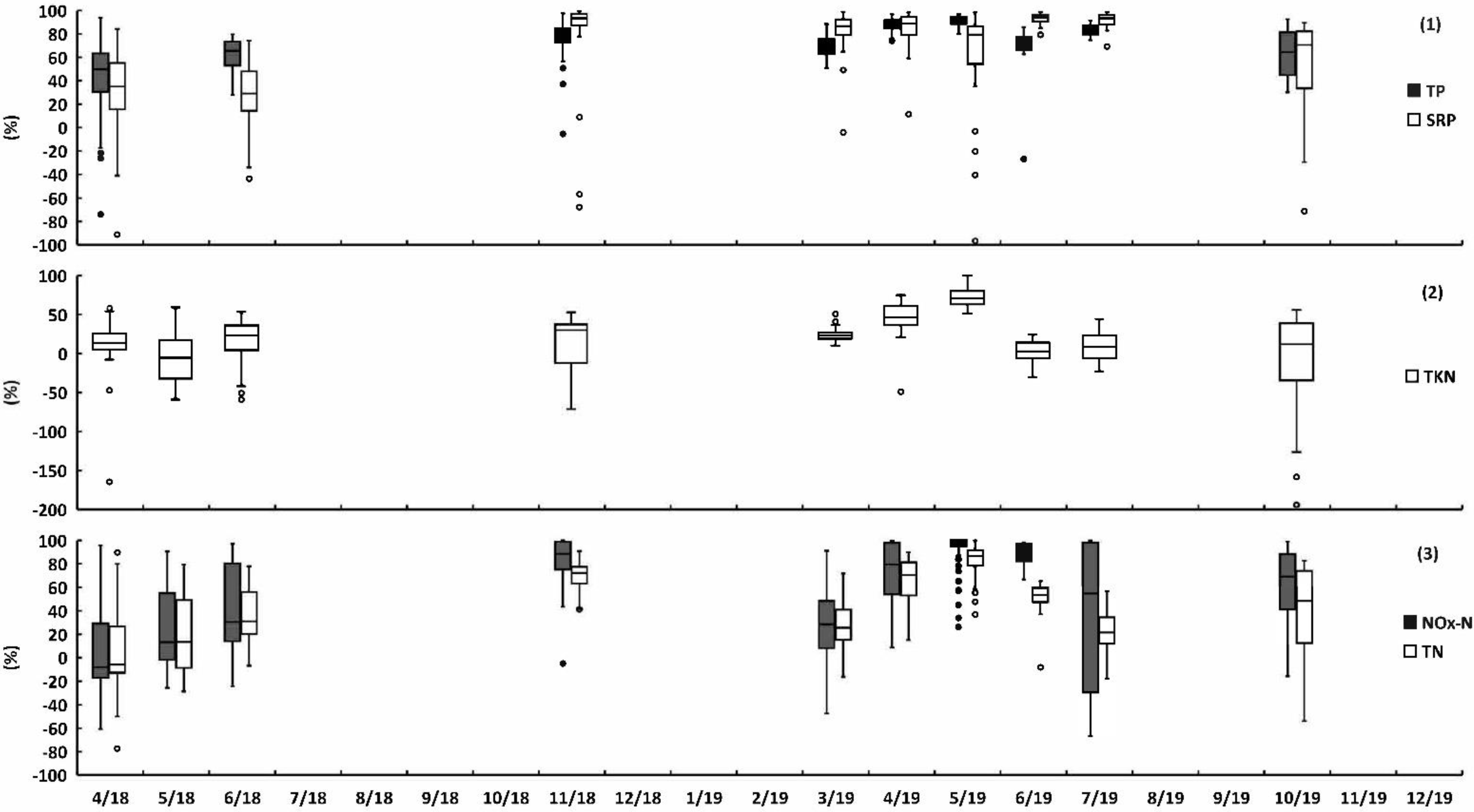
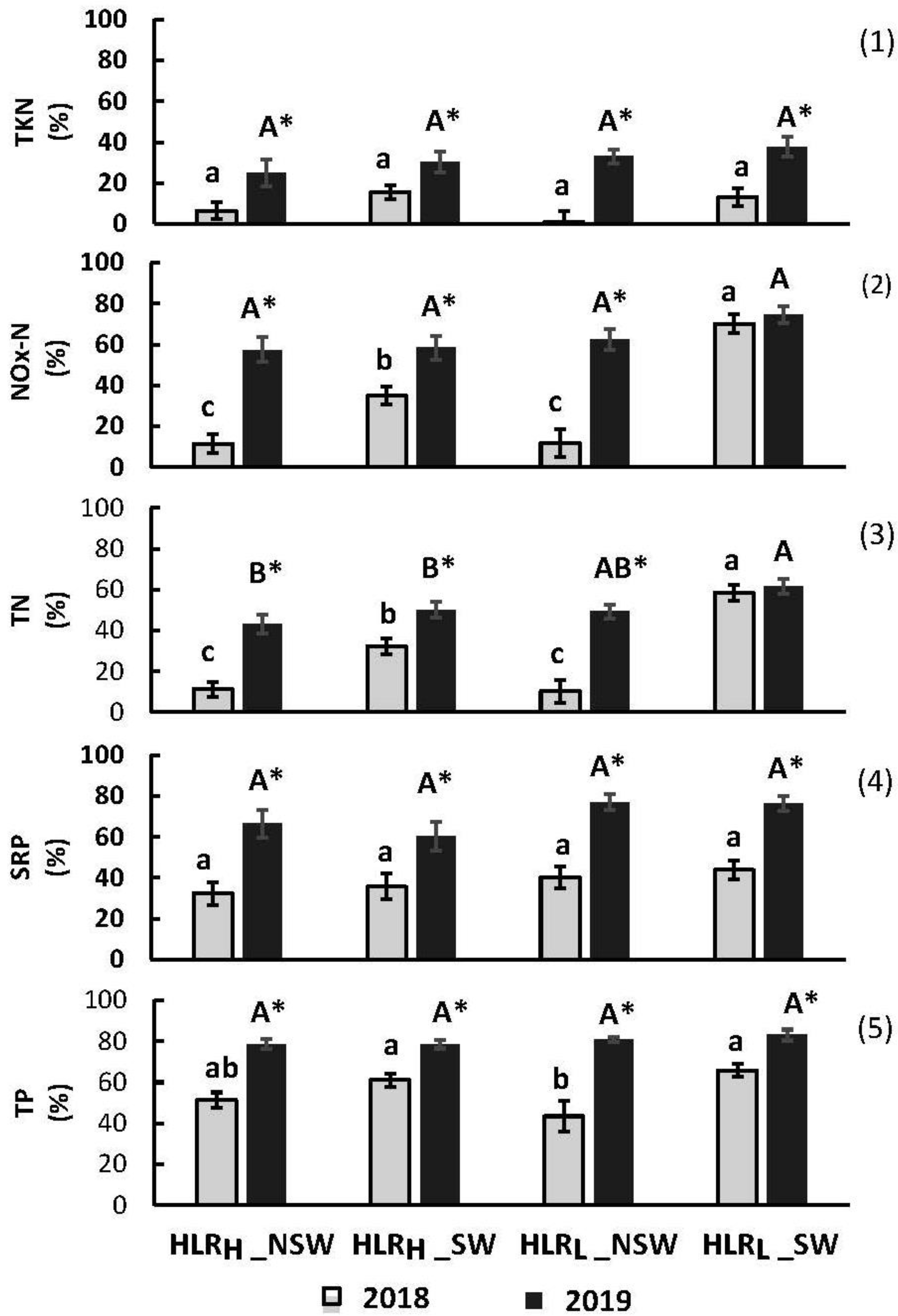
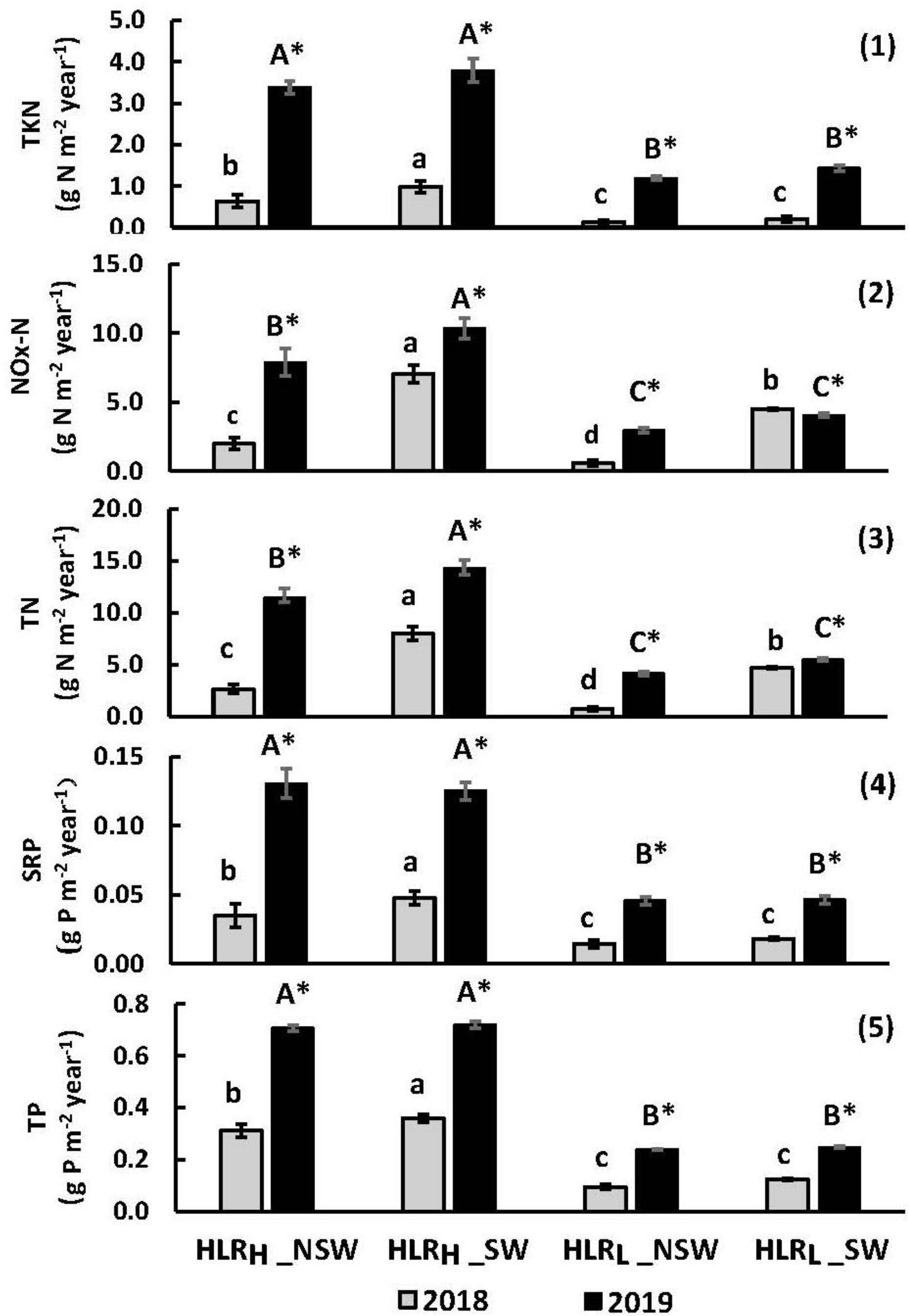
| 2018 | 2019 | ||
|---|---|---|---|
| TP | Inflow (μg·dm−3) | 142 ± 14 (5) | 162 ± 8 (8) |
| Outflow (μg·dm−3) | 57 ± 3 (139) | 28 ± 2 (206) * | |
| SRP | Inflow (μg·dm−3) | 23 ± 5 (6) | 35 ± 4 (8) |
| Outflow (μg·dm−3) | 11 ± 1 (168) | 7 ± 1 (231) * | |
| TN | Inflow (mg·dm−3) | 6.137 ± 0.841 (7) | 4.621 ± 0.494 (9) * |
| Outflow (mg·dm−3) | 4.403 ± 0.159 (195) | 2.144 ± 0.097 (231) * | |
| NOx-N | Inflow (mg·dm−3) | 4.913 ± 0.845 (7) | 2.757 ± 0.369 (9) * |
| Outflow (mg·dm−3) | 3.356 ± 0.16 (196) | 0.963 ± 0.073 (233) * | |
| TKN | Inflow(mg·dm−3) | 1.224 ± 0.141 (7) | 1.864 ± 0.184 (9) * |
| Outflow(mg·dm−3) | 1.055 ± 0.027 (195) | 1.175 ± 0.036 (233) * |
| 2018–2019 Removal Efficiency (%) | 2018–2019 Mass Retention (g m−2) | ||||
|---|---|---|---|---|---|
| HLRH | HLRL | HLRH | HLRL | ||
| TP | NSW | 67 ± 3 (80) b | 66 ± 4 (90) b | 1.008 ± 0.036 (6) A | 0.334 ± 0.009 (7) B |
| SW | 71 ± 2 (84) ab | 76 ± 2 (90) a | 1.076 ± 0.022 (6) A | 0.373 ± 0.007 (7) B | |
| TN | NSW | 28 ± 3 (101) c | 32 ± 4 (110) c | 14.454 ± 0.933 (6) B | 4.896 ± 0.313 (7) C |
| SW | 42 ± 3 (104) b | 60 ± 3 (111) a | 22.235 ± 1.135 (6) A | 10.177 ± 0.238 (7) B | |
| Scientific Name | HLR | Water Level | Indicator | Coverage (%) |
|---|---|---|---|---|
| Schoenoplectus tabernaemontani | H | SW | OBL | 98 ± 2 (6) |
| L | SW | OBL | 91 ± 3 (7) | |
| L | NSW | OBL | 69 ± 4 (7) | |
| H | NSW | OBL | 49 ± 8 (6) | |
| Alisma plantago-aquatica | H | SW | OBL | 10 ± 0 (1) |
| L | SW | OBL | 6 ± 1 (5) | |
| Carex vulpinoidea | H | NSW | OBL | 12 ± 1 (5) |
| L | NSW | OBL | 12 ± 1 (5) | |
| Typha spp. | L | SW | OBL | 9 ± 2 (4) |
| Polygonum pensylvanicum | H | NSW | FACW | 15 ± 7 (2) |
| L | NSW | FACW | 8 ± 3 (3) | |
| Cyperus esculentus | H | NSW | FACW | 8 ± 1 (4) |
| L | NSW | FACW | 9 ± 1 (5) | |
| Eupatorium perfoliatum | H | NSW | FACW | 5 ± 0 (1) |
| Rumex crispus | H | NSW | FACU | 20 ± 5 (3) |
| L | NSW | FACU | 13 ± 1 (3) | |
| Echinochloa crusgalli | H | NSW | FACU | 10 ± 2 (3) |
| L | NSW | FACU | 12 ± 1 (3) | |
| Ambrosia artemisiifolia | H | NSW | FACU | 15 ± 8 (5) |
| Setaria viridis | H | NSW | NL | 5 ± 0 (2) |
| L | NSW | NL | 10 ± 0 (1) | |
| Erigeron canadensis | H | NSW | NL | 5 ± 0 (1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, B.; Mitsch, W.J.; Lenhart, C. Estimating the Importance of Hydrologic Conditions on Nutrient Retention and Plant Richness in a Wetlaculture Mesocosm Experiment in a Former Lake Erie Basin Swamp. Water 2021, 13, 2509. https://doi.org/10.3390/w13182509
Jiang B, Mitsch WJ, Lenhart C. Estimating the Importance of Hydrologic Conditions on Nutrient Retention and Plant Richness in a Wetlaculture Mesocosm Experiment in a Former Lake Erie Basin Swamp. Water. 2021; 13(18):2509. https://doi.org/10.3390/w13182509
Chicago/Turabian StyleJiang, Bingbing, William J. Mitsch, and Chris Lenhart. 2021. "Estimating the Importance of Hydrologic Conditions on Nutrient Retention and Plant Richness in a Wetlaculture Mesocosm Experiment in a Former Lake Erie Basin Swamp" Water 13, no. 18: 2509. https://doi.org/10.3390/w13182509






