Simultaneous Biosorption of Arsenic and Cadmium onto Chemically Modified Chlorella vulgaris and Spirulinaplatensis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Adsorbent Preparation and Modification
2.2. Characterizations and Analysis
2.3. Biosorption Experiments
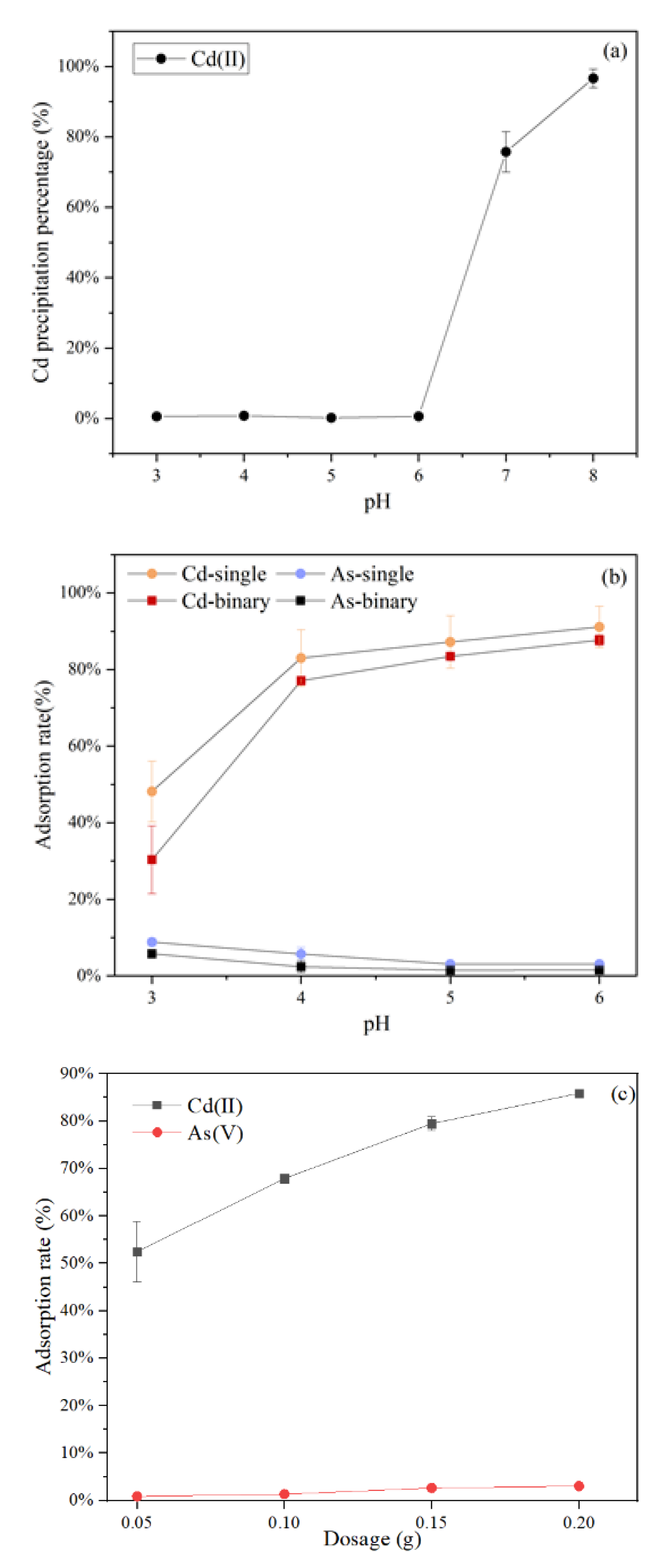
2.3.1. Effects of Initial pH and Adsorbents Dosage
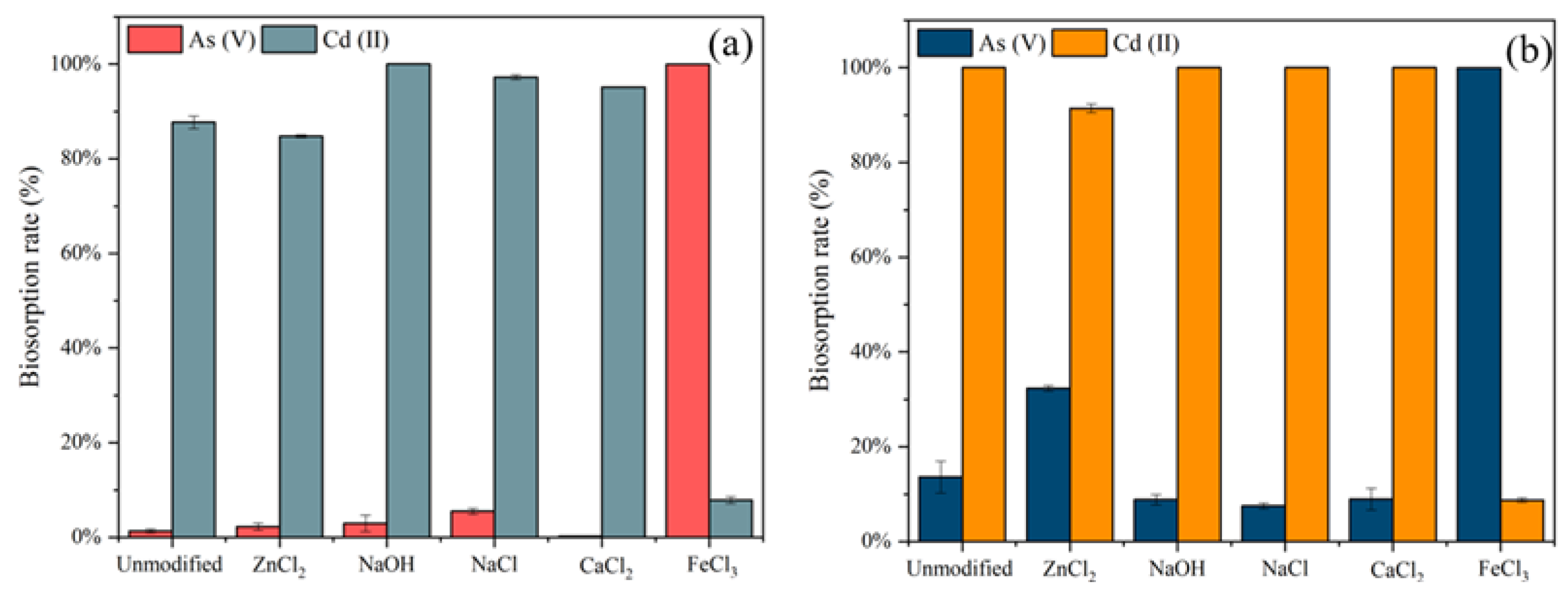
2.3.2. Effects of Different Modifications
2.3.3. Effects of Chemical Reagents Concentration
2.4. Biosorption Isotherm
2.5. Biosorption Kinetics
3. Results and Discussion
3.1. Effects of Initial pH and Adsorbents Dosage
3.2. Biosorption Efficiency of Different Modified Adsorbents
3.3. Effects of the Concentration of Modification Reagents
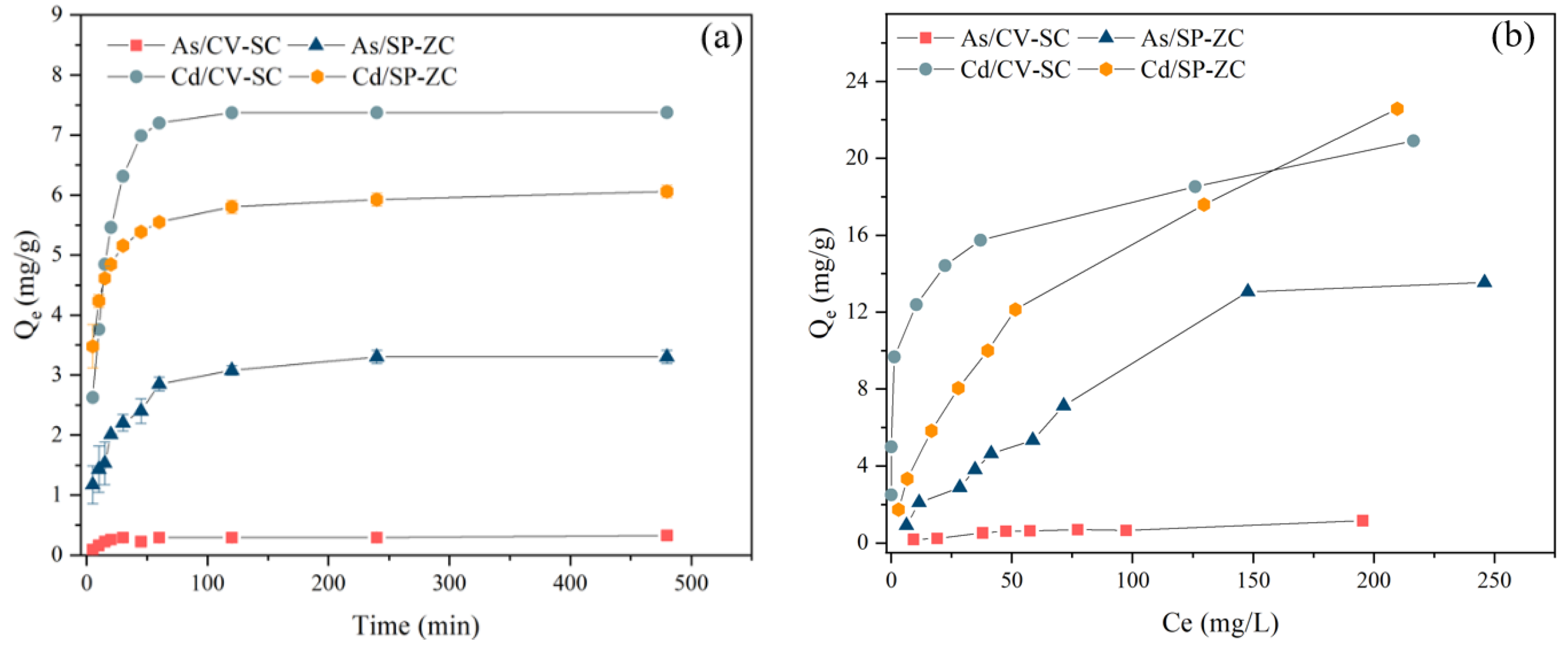
3.4. Biosorption Kinetic Analysis
3.5. Biosorption Isotherm
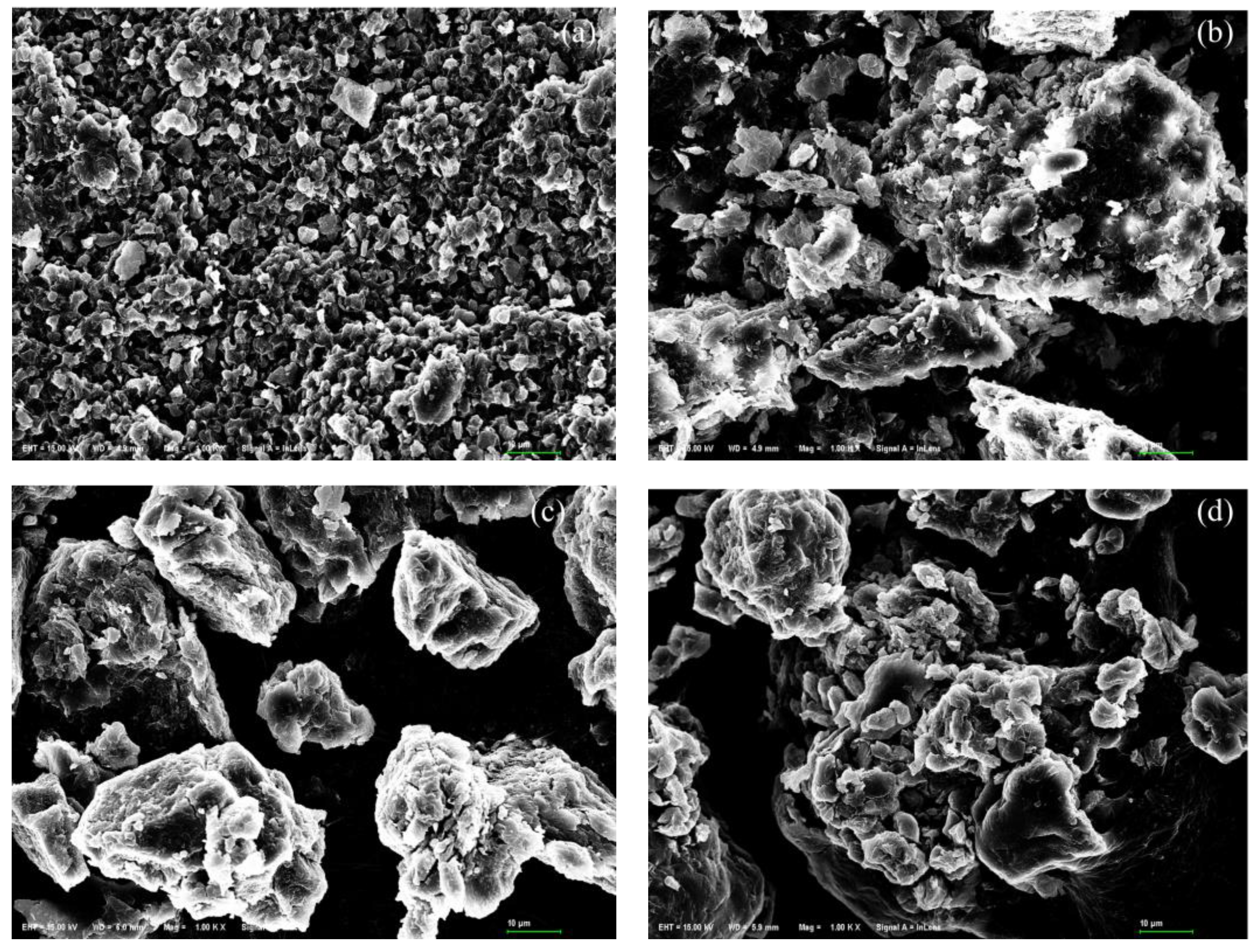
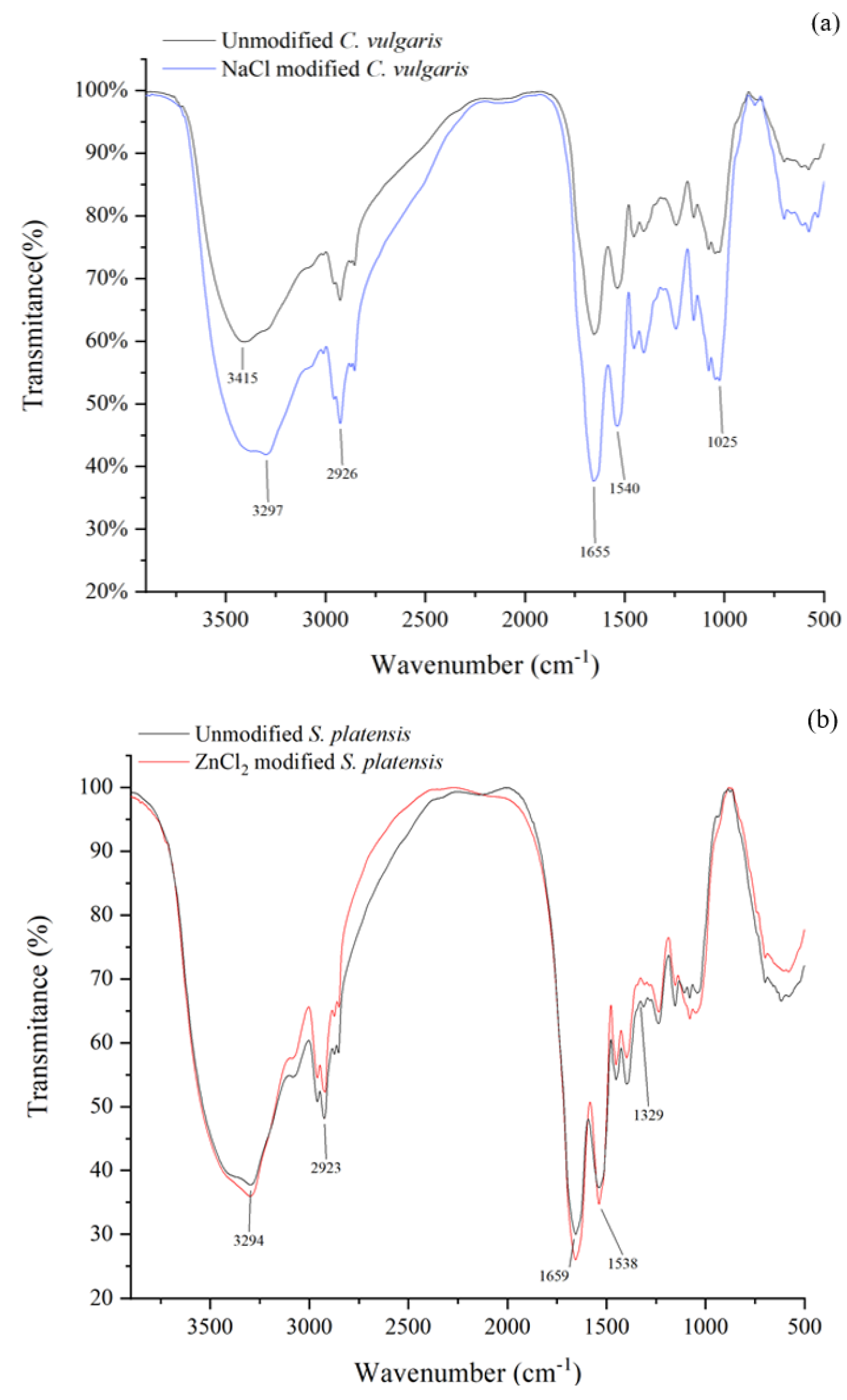
3.6. Adsorbents Characterization
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gogoi, A.; Biswas, S.; Bora, J.; Bhattacharya, S.S.; Kumar, M. Effect of vermicomposting on copper and zinc removal in activated sludge with special emphasis on temporal variation. Ecohydrol. Hydrobiol. 2015, 15, 101–107. [Google Scholar] [CrossRef]
- Gräfe, M.; Nachtegaal, M.; Sparks, D.L. Formation of Metal−Arsenate Precipitates at the Goethite−Water Interface. Environ. Sci. Technol. 2004, 38, 6561–6570. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Wang, W.; Li, X.; Duan, J.; Jing, C. Evaluating adsorption media for simultaneous removal of arsenate and cadmium from metallurgical wastewater. J. Environ. Chem. Eng. 2016, 4, 2795–2801. [Google Scholar] [CrossRef]
- Yoon, K.; Cho, D.-W.; Tsang, D.; Bolan, N.; Rinklebe, J.; Song, H. Fabrication of engineered biochar from paper mill sludge and its application into removal of arsenic and cadmium in acidic water. Bioresour. Technol. 2017, 246, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Lv, J.; Luo, L.; Yang, K.; Lin, Y.; Hu, F.; Zhang, J.; Zhang, S. Arsenate and cadmium co-adsorption and co-precipitation on goethite. J. Hazard. Mater. 2013, 262, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.G.; Debsarkar, A.; Dutta, A. Technology alternatives for decontamination of arsenic-rich groundwater—A critical review. Environ. Technol. Innov. 2018, 13, 277–303. [Google Scholar] [CrossRef]
- Nishijo, M.; Nakagawa, H.; Suwazono, Y.; Nogawa, K.; Kido, T. Causes of death in patients with Itai-itai disease suffering from severe chronic cadmium poisoning: A nested case–control analysis of a follow-up study in Japan. BMJ Open 2017, 7, e015694. [Google Scholar] [CrossRef] [Green Version]
- Sahmoune, M.N. The role of biosorbents in the removal of arsenic from water. Chem. Eng. Technol. 2016, 39, 1617–1628. [Google Scholar] [CrossRef]
- Ghosh, D.; Saha, R.; Ghosh, A.; Nandi, R.; Saha, B. A review on toxic cadmium biosorption from contaminated wastewater. Desalination Water Treat. 2013, 53, 413–420. [Google Scholar] [CrossRef]
- Du, L.; Yu, R.; Wang, H.-y.; LU Yun; Zheng, L. T. Pollution and toxicity of cadmium: A review of recent studies. J. Environ. Health 2013, 30, 167–174. [Google Scholar] [CrossRef]
- Cheng, S.Y.; Show, P.-L.; Lau, B.F.; Chang, J.-S.; Ling, T.C. New Prospects for Modified Algae in Heavy Metal Adsorption. Trends Biotechnol. 2019, 37, 1255–1268. [Google Scholar] [CrossRef]
- Liao, Q.; Tu, G.; Yang, Z.; Wang, H.; He, L.; Tang, J.; Yang, W. Simultaneous adsorption of As(III), Cd(II) and Pb(II) by hybrid bio-nanocomposites of nano hydroxy ferric phosphate and hydroxy ferric sulfate particles coating on Aspergillus niger. Chemosphere 2019, 223, 551–559. [Google Scholar] [CrossRef]
- He, J.; Chen, J.P. A comprehensive review on biosorption of heavy metals by algal biomass: Materials, performances, chemistry, and modeling simulation tools. Bioresour. Technol. 2014, 160, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Lebron, Y.; Moreira, V.; Santos, L. Studies on dye biosorption enhancement by chemically modified Fucus vesiculosus, Spirulina maxima and Chlorella pyrenoidosa algae. J. Clean. Prod. 2019, 240, 118197. [Google Scholar] [CrossRef]
- Pereira, L.; Alves, M. Dyes—Environmental Impact and Remediation. In Environmental Protection Strategies for Sustainable Development; Malik, A., Grohmann, E., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 111–162. [Google Scholar]
- Lebron, Y.; Moreira, V.; Santos, L.; Jacob, R. Remediation of methylene blue from aqueous solution by Chlorella pyrenoidosa and Spirulina maxima biosorption: Equilibrium, kinetics, thermodynamics and optimization studies. J. Environ. Chem. Eng. 2018, 6, 6680–6690. [Google Scholar] [CrossRef]
- Huq, E.; Fahad, S.; Shao, Z.; Sarven, M.S.; Khan, I.A.; Alam, M.; Saeed, M.; Ullah, H.; Adnan, M.; Saud, S.; et al. Arsenic in a groundwater environment in Bangladesh: Occurrence and mobilization. J. Environ. Manag. 2020, 262, 110318. [Google Scholar] [CrossRef]
- Kousha, M.; Daneshvar, E.; Sohrabi, M.S.; Jokar, M.; Bhatnagar, A. Adsorption of acid orange II dye by raw and chemically modified brown macroalga Stoechospermum marginatum. Chem. Eng. J. 2012, 192, 67–76. [Google Scholar] [CrossRef]
- Daneshvar, E.; Vazirzadeh, A.; Niazi, A.; Sillanpää, M.; Bhatnagar, A. A comparative study of methylene blue biosorption using different modified brown, red and green macroalgae—Effect of pretreatment. Chem. Eng. J. 2017, 307, 435–446. [Google Scholar] [CrossRef]
- Feng, N.; Guo, X.; Liang, S.; Zhu, Y.; Liu, J. Biosorption of heavy metals from aqueous solutions by chemically modified orange peel. J. Hazard. Mater. 2011, 185, 49–54. [Google Scholar] [CrossRef] [PubMed]
- García-Rosales, G.; Olguín, M.T.; Colin-Cruz, A.; Romero-Guzmán, E.T. Effect of the pH and temperature on the biosorption of lead(II) and cadmium(II) by sodium-modified stalk sponge of Zea mays. Environ. Sci. Pollut. Res. 2011, 19, 177–185. [Google Scholar] [CrossRef]
- Wassie, A.B.; Srivastava, V.C. Chemical treatment of teff straw by sodium hydroxide, phosphoric acid and zinc chloride: Adsorptive removal of chromium. Int. J. Environ. Sci. Technol. 2016, 13, 2415–2426. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Yang, Z.; Dong, H.; Guan, X.; Ren, Q.; Lv, X.; Jin, X. Simple combination of oxidants with zero-valent-iron (ZVI) achieved very rapid and highly efficient removal of heavy metals from water. Water Res. 2015, 88, 671–680. [Google Scholar] [CrossRef]
- Bulgariu, L.; Bulgariu, D. Enhancing Biosorption Characteristics of Marine Green Algae (Ulva lactuca) for Heavy Metals Removal by Alkaline Treatment. J. Bioprocess. Biotech. 2014, 4, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Yu, Q.; Matheickal, J.T.; Yin, P.; Kaewsarn, P. Heavy metal uptake capacities of common marine macro algal biomass. Water Res. 1999, 33, 1534–1537. [Google Scholar] [CrossRef]
- Teodoro, F.S.; Ramos, S.N.D.C.; Elias, M.M.C.; Mageste, A.B.; Ferreira, G.M.D.; Da Silva, L.H.M.; Gil, L.F.; Gurgel, L.V.A. Synthesis and application of a new carboxylated cellulose derivative. Part I: Removal of Co2+, Cu2+ and Ni2+ from monocomponent spiked aqueous solution. J. Colloid Interface Sci. 2016, 483, 185–200. [Google Scholar] [CrossRef]
- Moreira, V.; Lebron, Y.; Freire, S.; Santos, L.; Palladino, F.; Jacob, R. Biosorption of copper ions from aqueous solution using Chlorella pyrenoidosa: Optimization, equilibrium and kinetics studies. Microchem. J. 2018, 145, 119–129. [Google Scholar] [CrossRef]
- Asere, T.G.; Stevens, C.V.; Du Laing, G. Use of (modified) natural adsorbents for arsenic remediation: A review. Sci. Total. Environ. 2019, 676, 706–720. [Google Scholar] [CrossRef]
- Choi, H.-J.; Lee, S.-M. Heavy metal removal from acid mine drainage by calcined eggshell and microalgae hybrid system. Environ. Sci. Pollut. Res. 2015, 22, 13404–13411. [Google Scholar] [CrossRef] [PubMed]
- Dhar, R.; Zheng, Y.; Rubenstone, J.; van Geen, A. A rapid colorimetric method for measuring arsenic concentrations in groundwater. Anal. Chim. Acta 2004, 526, 203–209. [Google Scholar] [CrossRef]
- Kataria, N.; Garg, V. Green synthesis of Fe3O4 nanoparticles loaded sawdust carbon for cadmium (II) removal from water: Regeneration and mechanism. Chemosphere 2018, 208, 818–828. [Google Scholar] [CrossRef]
- Lee, J.S.; Nriagu, J.O. Stability constants for metal arsenates. Environ. Chem. 2007, 4, 123–133. [Google Scholar] [CrossRef]
- Sun, X.; Huang, H.; Zhu, Y.; Du, Y.; Yao, L.; Jiang, X.; Gao, P. Adsorption of Pb2+ and Cd2+ onto Spirulina platensis harvested by polyacrylamide in single and binary solution systems. Colloids Surf. A Physicochem. Eng. Asp. 2019, 583, 123926. [Google Scholar] [CrossRef]
- Anwar, J.; Shafique, U.; Zaman, W.U.; Salman, M.; Dar, A.; Anwar, S. Removal of Pb(II) and Cd(II) from water by adsorption on peels of banana. Bioresour. Technol. 2010, 101, 1752–1755. [Google Scholar] [CrossRef]
- Gupta, V.K.; Rastogi, A.; Nayak, A. Biosorption of nickel onto treated alga (Oedogonium hatei): Application of isotherm and kinetic models. J. Colloid Interface Sci. 2010, 342, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Vatutsina, O.; Soldatov, V.; Sokolova, V.; Johann, J.; Bissen, M.; Weissenbacher, A. A new hybrid (polymer/inorganic) fibrous sorbent for arsenic removal from drinking water. React. Funct. Polym. 2007, 67, 184–201. [Google Scholar] [CrossRef]
- Batzias, F.; Sidiras, D. Simulation of methylene blue adsorption by salts-treated beech sawdust in batch and fixed-bed systems. J. Hazard. Mater. 2007, 149, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Ozdes, D.; Duran, C.; Senturk, H.B. Adsorptive removal of Cd(II) and Pb(II) ions from aqueous solutions by using Turkish illitic clay. J. Environ. Manag. 2011, 92, 3082–3090. [Google Scholar] [CrossRef]
- Paulino, A.T.; Belfiore, L.A.; Kubota, L.T.; Muniz, E.; Almeida, V.C.; Tambourgi, E.B. Effect of magnetite on the adsorption behavior of Pb(II), Cd(II), and Cu(II) in chitosan-based hydrogels. Desalination 2011, 275, 187–196. [Google Scholar] [CrossRef]
- Segovia-Sandoval, S.J.; Ocampo-Pérez, R.; Berber-Mendoza, M.S.; Leyva-Ramos, R.; Jacobo-Azuara, A.; Medellín-Castillo, N.A. Walnut shell treated with citric acid and its application as biosorbent in the removal of Zn(II). J. Water Process. Eng. 2018, 25, 45–53. [Google Scholar] [CrossRef]
- Sheng, P.X.; Ting, Y.-P.; Chen, J.; Hong, L. Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: Characterization of biosorptive capacity and investigation of mechanisms. J. Colloid Interface Sci. 2004, 275, 131–141. [Google Scholar] [CrossRef]
- Gupta, V.; Rastogi, A. Equilibrium and kinetic modelling of cadmium(II) biosorption by nonliving algal biomass Oedogonium sp. from aqueous phase. J. Hazard. Mater. 2008, 153, 759–766. [Google Scholar] [CrossRef] [PubMed]

| Parameter | NaCl Modified C. vulgaris | ZnCl2 Modified S. platensis | ||
|---|---|---|---|---|
| As | Cd | As | Cd | |
| Pseudo-first-order | ||||
| K1 | 2.63 × 10−5 | 1.50 × 10−4 | 3.88 × 10−6 | 9.30 × 10−6 |
| qe | 0.99 | 3.34 | 5.64 | 2.96 |
| R2 | 0.31 | 0.86 | 0.46 | 0.37 |
| Pseudo-second-order | ||||
| K2 | 0.28 | 0.02 | 0.21 | 0.03 |
| qe | 0.33 | 7.51 | 3.4089 | 6.111 |
| R2 | 0.9970 | 0.9996 | 0.9994 | 0.9999 |
| Parameter | NaCl Modified C. vulgaris | ZnCl2 Modified S. platensis | ||
|---|---|---|---|---|
| As | Cd | As | Cd | |
| Langmuir | ||||
| qm | 1.61 | 17.60 | 24.88 | 29.37 |
| KL | 1.06 × 10−2 | 0.62 | 5.60 × 10−3 | 1.36 × 10−2 |
| R2 | 0.9211 | 0.7498 | 0.9634 | 0.9888 |
| Freundlich | ||||
| Kf | 6.40×10−2 | 8.93 | 0.42 | 1.44 |
| N | 1.83 | 6.45 | 1.54 | 1.93 |
| R2 | 0.9356 | 0.9904 | 0.9391 | 0.9918 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, W.; Xu, Y.; Liang, C.; Musah, B.I.; Peng, L. Simultaneous Biosorption of Arsenic and Cadmium onto Chemically Modified Chlorella vulgaris and Spirulinaplatensis. Water 2021, 13, 2498. https://doi.org/10.3390/w13182498
Lu W, Xu Y, Liang C, Musah BI, Peng L. Simultaneous Biosorption of Arsenic and Cadmium onto Chemically Modified Chlorella vulgaris and Spirulinaplatensis. Water. 2021; 13(18):2498. https://doi.org/10.3390/w13182498
Chicago/Turabian StyleLu, Wenlong, Yifeng Xu, Chuanzhou Liang, Baba Imoro Musah, and Lai Peng. 2021. "Simultaneous Biosorption of Arsenic and Cadmium onto Chemically Modified Chlorella vulgaris and Spirulinaplatensis" Water 13, no. 18: 2498. https://doi.org/10.3390/w13182498
APA StyleLu, W., Xu, Y., Liang, C., Musah, B. I., & Peng, L. (2021). Simultaneous Biosorption of Arsenic and Cadmium onto Chemically Modified Chlorella vulgaris and Spirulinaplatensis. Water, 13(18), 2498. https://doi.org/10.3390/w13182498