A Sustainable and Low-Cost Soil Filter Column for Removing Pathogens from Swine Wastewater: The Role of Endogenous Soil Protozoa
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Microcosm Experiment
2.2. Microorganisms and Culture
2.3. Protozoa Growth in Soil Columns
2.4. Soil Filtration and Protozoa Predation
2.5. Quantification of Microorganisms
3. Results
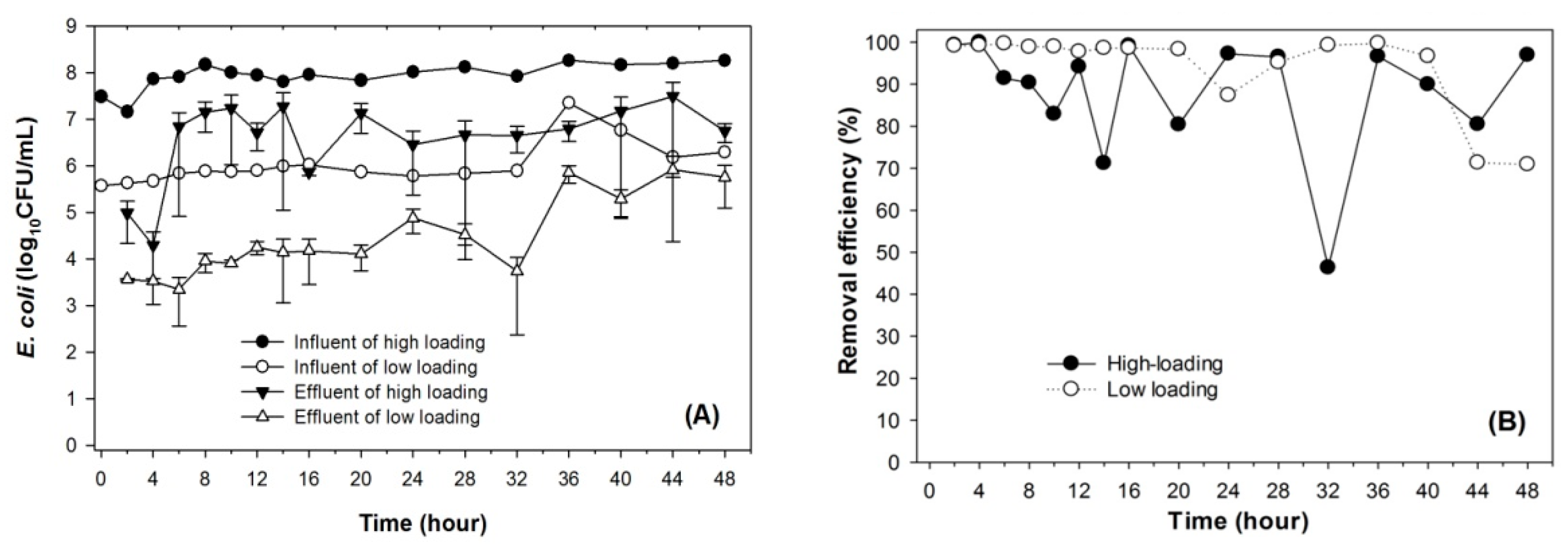
3.1. Sorption of Bacteria by Leilehua Soil
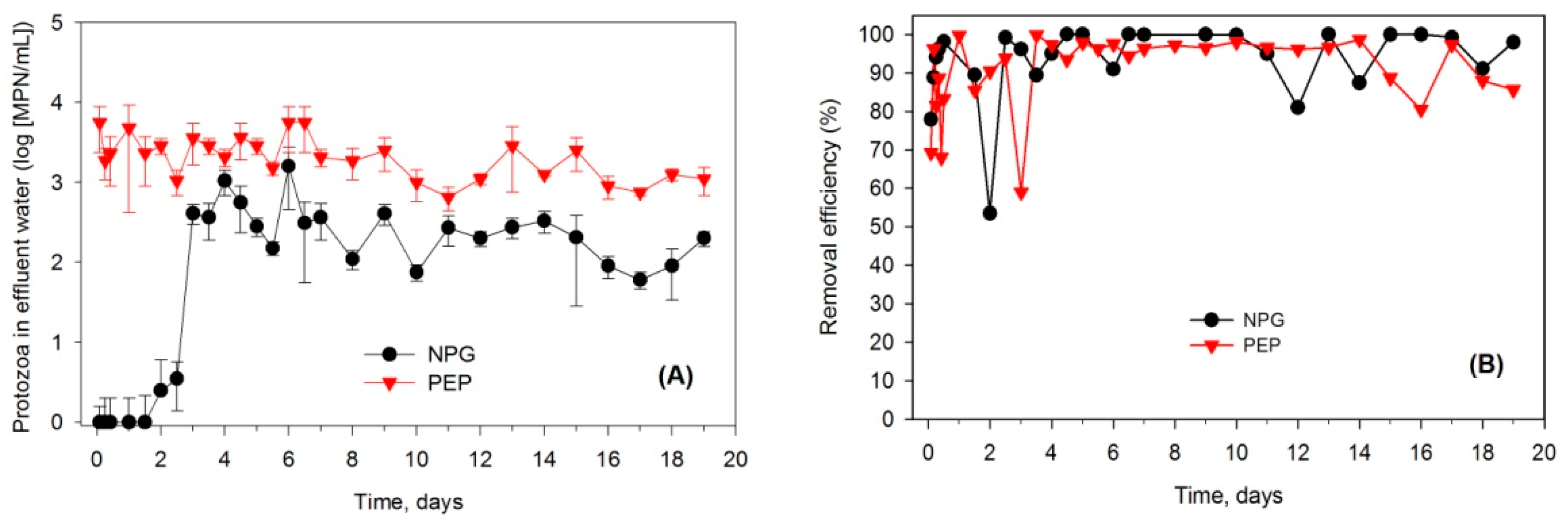
3.2. Protozoa Response to the Addition of E. coli
3.3. Effects of the Presence of Protozoa on Removal Efficiency of E. coli within Soil Columns
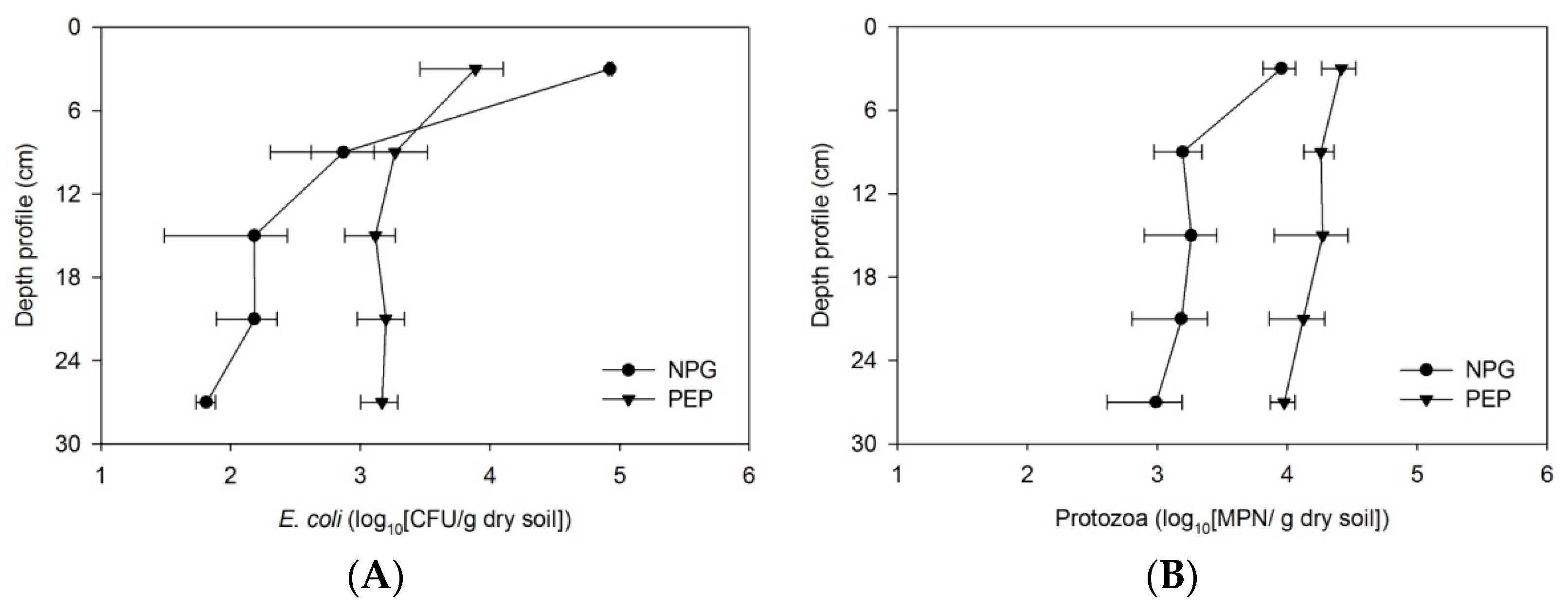
3.4. Enhancement of Bacterial Removal in Two-Stage Filtration
4. Discussion
4.1. Adsorption Capacity of Leilehua to E. coli
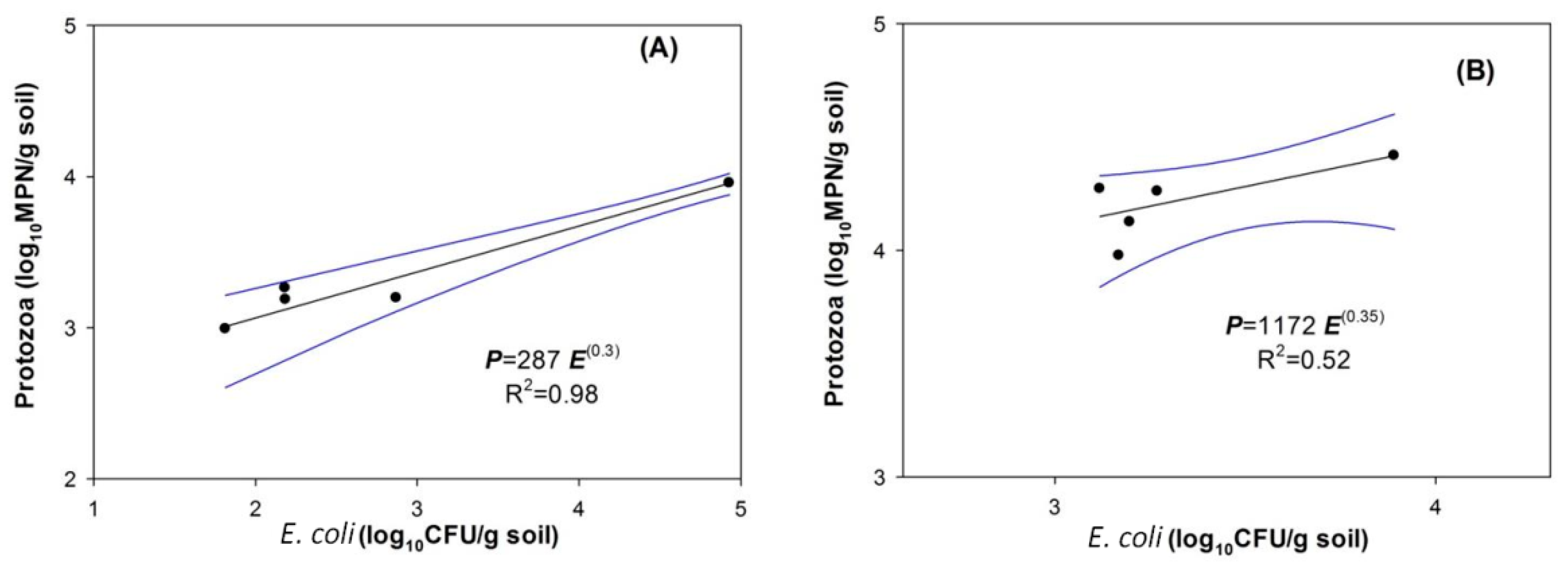
4.2. Indigenous Soil Protozoa and Bacterial Regulation by Protozoa
4.3. Effects of Protozoa on E. coli Removal Efficiencies in the Soil Columns
4.4. Sequential Design Filters for Better Removal
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Choi, E. Piggery Waste Management; IWA Publishing: London, UK, 2007; Volume 6. [Google Scholar]
- Owusu-Twum, M.Y.; Sharara, M.A. Sludge management in anaerobic swine lagoons: A review. J. Environ. Manag. 2020, 271, 110949. [Google Scholar] [CrossRef]
- McLaughlin, M.R.; Brooks, J.P.; Adeli, A. Temporal flux and spatial dynamics of nutrients, fecal indicators, and zoonotic pathogens in anaerobic swine manure lagoon water. Water Res. 2012, 46, 4949–4960. [Google Scholar] [CrossRef] [Green Version]
- Chee-Sanford, J.C.; Aminov, R.I.; Krapac, I.J.; Garrigues-Jeanjean, N.; Mackie, R.I. Occurrence and Diversity of Tetracycline Resistance Genes in Lagoons and Groundwater Underlying Two Swine Production Facilities. Appl. Environ. Microbiol. 2001, 67, 1494–1502. [Google Scholar] [CrossRef] [Green Version]
- Deng, S.; Yan, X.; Zhu, Q.; Liao, C. The utilization of reclaimed water: Possible risks arising from waterborne contaminants. Environ. Pollut. 2019, 254, 113020. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.P.; Adeli, A.; McLaughlin, M.R. Microbial ecology, bacterial pathogens, and antibiotic resistant genes in swine manure wastewater as influenced by three swine management systems. Water Res. 2014, 57, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Guan, T.Y.; Holley, R.A. Pathogen survival in swine manure environments and transmission of human enteric illness-a review. J. Environ. Qual. 2003, 32, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, L.; Liu, Y.; Shen, Y.; Liu, H.; Xiong, Y. Effect of limited artificial aeration on constructed wetland treatment of domestic wastewater. Desalination 2010, 250, 915–920. [Google Scholar] [CrossRef]
- Gerba, C.P.; Lance, J.C. Poliovirus removal from primary and secondary sewage effluent by soil filtration. Appl. Environ. Microbiol. 1978, 36, 247–251. [Google Scholar] [CrossRef] [Green Version]
- Gilbert, R.G.; Gerba, C.P.; Rice, R.C.; Bouwer, H.; Wallis, C.; Melnick, J.L. Virus and bacteria removal from wastewater by land treatment. Appl. Environ. Microbiol. 1976, 32, 333–338. [Google Scholar] [CrossRef] [Green Version]
- Pattnaik, R.; Yost, R.S.; Porter, G.; Masunaga, T.; Attanandana, T. Improving multi-soil-layer (MSL) system remediation of dairy effluent. Ecol. Eng. 2008, 32, 1–10. [Google Scholar] [CrossRef]
- Powelson, D.K.; Simpson, J.R.; Gerba, C.P. Effects of organic matter on virus transport in unsaturated flow. Appl. Environ. Microbiol. 1991, 57, 2192–2196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wakatsuki, T.; Esumi, H.; Omura, S. High performance and nitrogen and phosphorus-removable on-site domestic waste water treatment system by multi-soil-layering method. Water Sci. Technol. 1993, 27, 31–40. [Google Scholar] [CrossRef]
- Wang, D.S.; Gerba, C.P.; Lance, J.C. Effect of soil permeability on virus removal through soil columns. Appl. Environ. Microbiol. 1981, 42, 83–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyer, T.H.; Persaud, A.; Banerjee, P.; Palomino, P. Comparison of low-cost and engineered materials for phosphorus removal from organic-rich surface water. Water Res. 2011, 45, 4803–4814. [Google Scholar] [CrossRef]
- Latrach, L.; Ouazzani, N.; Masunaga, T.; Hejjaj, A.; Bouhoum, K.; Mahi, M.; Mandi, L. Domestic wastewater disinfection by combined treatment using multi-soil-layering system and sand filters (MSL–SF): A laboratory pilot study. Ecol. Eng. 2016, 91, 294–301. [Google Scholar] [CrossRef]
- Latrach, L.; Ouazzani, N.; Hejjaj, A.; Mahi, M.; Masunaga, T.; Mandi, L. Two-stage vertical flow Multi-Soil-Layering (MSL) technology for efficient removal of coliforms and human pathogens from domestic wastewater in rural areas under arid climate. Int. J. Hyg. Environ. Health 2018, 221, 64–80. [Google Scholar] [CrossRef] [PubMed]
- Latrach, L.; Ouazzani, N.; Hejjaj, A.; Zouhir, F.; Mahi, M.; Masunaga, T.; Mandi, L. Optimization of hydraulic efficiency and wastewater treatment performances using a new design of vertical flow Multi-Soil-Layering (MSL) technology. Ecol. Eng. 2018, 117, 140–152. [Google Scholar] [CrossRef]
- Latrach, L.; Masunaga, T.; Ouazzani, N.; Hejjaj, A.; Mahi, M.; Mandi, L. Removal of bacterial indicators and pathogens from domestic wastewater by the multi-soil-layering (MSL) system. Soil Sci. Plant Nutr. 2015, 61, 337–346. [Google Scholar] [CrossRef] [Green Version]
- Camper, A.K.; Hayes, J.T.; Sturman, P.J.; Jones, W.L.; Cunningham, A.B. Effects of Motility and Adsorption Rate Coefficient on Transport of Bacteria through Saturated Porous Media. Appl. Environ. Microbiol. 1993, 59, 3455–3462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fontes, D.E.; Mills, A.L.; Hornberger, G.M.; Herman, J.S. Physical and chemical factors influencing transport of microorganisms through porous media. Appl. Environ. Microbiol. 1991, 57, 2473–2481. [Google Scholar] [CrossRef] [Green Version]
- Weber-Shirk, M.L.; Dick, R.I. Biological mechanisms in slow sand filters. J. Am. Water Works Assoc. 1997, 89, 72–83. [Google Scholar] [CrossRef]
- Hendry, M.J.; Lawrence, J.R.; Maloszewski, P. The role of sorption in the transport of Klebsiella oxytoca through saturated silica sand. Ground Water 1997, 35, 574–584. [Google Scholar] [CrossRef]
- Stumpp, C.; Lawrence, J.R.; Hendry, M.J.; Maloszewski, P. Transport and Bacterial Interactions of Three Bacterial Strains in Saturated Column Experiments. Environ. Sci. Technol. 2011, 45, 2116–2123. [Google Scholar] [CrossRef] [PubMed]
- Camesano, T.A.; Logan, B.E. Influence of Fluid Velocity and Cell Concentration on the Transport of Motile and Nonmotile Bacteria in Porous Media. Environ. Sci. Technol. 1998, 32, 1699–1708. [Google Scholar] [CrossRef]
- Bolster, C.H.; Cook, K.L.; Marcus, I.M.; Haznedaroglu, B.Z.; Walker, S.L. Correlating transport behavior with cell properties for eight porcine Escherichia coli isolates. Environ. Sci. Technol. 2010, 44, 5008–5014. [Google Scholar] [CrossRef]
- Chen, G.; Walker, S.L. Fecal Indicator Bacteria Transport and Deposition in Saturated and Unsaturated Porous Media. Environ. Sci. Technol. 2012, 46, 8782–8790. [Google Scholar] [CrossRef]
- Wang, L.; Xu, S.; Li, J. Effects of Phosphate on the Transport of Escherichia coli O157:H7 in Saturated Quartz Sand. Environ. Sci. Technol. 2011, 45, 9566–9573. [Google Scholar] [CrossRef] [PubMed]
- Dhand, N.K.; Toribio, J.-A.L.M.L.; Whittington, R.J. Adsorption of Mycobacterium avium subsp. paratuberculosis to Soil Particles. Appl. Environ. Microbiol. 2009, 75, 5581–5585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powelson, D.K.; Mills, A.L. Bacterial enrichment at the gas-water interface of a laboratory apparatus. Appl. Environ. Microbiol. 1996, 62, 2593–2597. [Google Scholar] [CrossRef] [Green Version]
- Stevik, T.; Aa, K.; Ausland, G.; Hanssen, J. Retention and Removal of Pathogenic Bacteria in Wastewater Percolating through Porous Media: A Review. Water Res. 2004, 38, 1355–1367. [Google Scholar] [CrossRef]
- Zhang, L.; Seagren, E.A.; Davis, A.P.; Karns, J.S. The Capture and Destruction of Escherichia coli from Simulated Urban Runoff Using Conventional Bioretention Media and Iron Oxide-coated Sand. Water Environ. Res. 2010, 82, 701–714. [Google Scholar] [CrossRef]
- Mills, A.L.; Herman, J.S.; Hornberger, G.M.; DeJesús, T.H. Effect of Solution Ionic Strength and Iron Coatings on Mineral Grains on the Sorption of Bacterial Cells to Quartz Sand. Appl. Environ. Microbiol. 1994, 60, 3300–3306. [Google Scholar] [CrossRef] [Green Version]
- Casida, L.E. Protozoan Response to the Addition of Bacterial Predators and Other Bacteria to Soil. Appl. Environ. Microbiol. 1989, 55, 1857–1859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González, J.M.; Iriberri, J.; Egea, L.; Barcina, I. Differential Rates of Digestion of Bacteria by Freshwater and Marine Phagotrophic Protozoa. Appl. Environ. Microbiol. 1990, 56, 1851–1857. [Google Scholar] [CrossRef] [Green Version]
- Hahn, M.W.; Hofle, M.G. Grazing of protozoa and its effect on populations of aquatic bacteria. FEMS Microbiol. Ecol. 2001, 35, 113–121. [Google Scholar] [CrossRef]
- Murase, J.; Noll, M.; Frenzel, P. Impact of Protists on the Activity and Structure of the Bacterial Community in a Rice Field Soil. Appl. Environ. Microbiol. 2006, 72, 5436–5444. [Google Scholar] [CrossRef] [Green Version]
- Rønn, R.; McCaig, A.E.; Griffiths, B.S.; Prosser, J.I. Impact of Protozoan Grazing on Bacterial Community Structure in Soil Microcosms. Appl. Environ. Microbiol. 2002, 68, 6094–6105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wright, D.A.; Killham, K.; Glover, L.A.; Prosser, J.I. Role of Pore Size Location in Determining Bacterial Activity during Predation by Protozoa in Soil. Appl. Environ. Microbiol. 1995, 61, 3537–3543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Decamp, O.; Warren, A.; Sanchez, R. The role of ciliated protozoa in subsurface flow wetlands and their potential as bioindicators. Water Sci. Technol. 1999, 40, 91–98. [Google Scholar] [CrossRef]
- Pinto, A.J.; Love, N.G. Bioreactor Function under Perturbation Scenarios Is Affected by Interactions between Bacteria and Protozoa. Environ. Sci. Technol. 2012, 46, 7558–7566. [Google Scholar] [CrossRef] [PubMed]
- Ravva, S.V.; Sarreal, C.Z.; Mandrell, R.E. Identification of protozoa in dairy lagoon wastewater that consume Escherichia coli O157:H7 preferentially. PLoS ONE 2010, 5, e15671. [Google Scholar] [CrossRef] [Green Version]
- Enzinger, R.M.; Cooper, R.C. Role of bacteria and protozoa in the removal of Escherichia coli from estuarine waters. Appl. Environ. Microbiol. 1976, 31, 758–763. [Google Scholar] [CrossRef] [Green Version]
- Holmes, D.E.; Nevin, K.P.; Snoeyenbos-West, O.L.; Woodard, T.L.; Strickland, J.N.; Lovley, D.R. Protozoan grazing reduces the current output of microbial fuel cells. Bioresour. Technol. 2015, 193, 8–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellegaard-Jensen, L.; Albers, C.N.; Aamand, J. Protozoa graze on the 2,6-dichlorobenzamide (BAM)-degrading bacterium Aminobacter sp. MSH1 introduced into waterworks sand filters. Appl. Microbiol. Biotechnol. 2016, 100, 8965–8973. [Google Scholar] [CrossRef] [PubMed]
- Haig, S.J.; Schirmer, M.; D’Amore, R.; Gibbs, J.; Davies, R.L.; Collins, G.; Quince, C. Stable-isotope probing and metagenomics reveal predation by protozoa drives E. coli removal in slow sand filters. ISME J. 2015, 9, 797–808. [Google Scholar] [CrossRef] [Green Version]
- Siqueira-Castro, I.C.V.; Greinert-Goulart, J.A.; Bonatti, T.R.; Yamashiro, S.; Franco, R.M.B. First report of predation of Giardia sp. cysts by ciliated protozoa and confirmation of predation of Cryptosporidium spp. oocysts by ciliate species. Environ. Sci. Pollut. Res. Int. 2016, 23, 11357–11362. [Google Scholar] [CrossRef]
- Wang, M.; Zhu, J.; Mao, X. Removal of Pathogens in Onsite Wastewater Treatment Systems: A Review of Design Considerations and Influencing Factors. Water 2021, 13, 1190. [Google Scholar] [CrossRef]
- Bomo, A.-M.; Husby, A.; Stevik, T.K.; Hanssen, J.F. Removal of fish pathogenic bacteria in biological sand filters. Water Res. 2003, 37, 2618–2626. [Google Scholar] [CrossRef]
- Bomo, A.-M.; Stevik, T.K.; Hovi, I.; Hanssen, J.F. Bacterial Removal and Protozoan Grazing in Biological Sand Filters. J. Environ. Qual. 2004, 33, 1041–1047. [Google Scholar] [CrossRef]
- Pfannes, K.R.; Langenbach, K.M.W.; Pilloni, G.; Stührmann, T.; Euringer, K.; Lueders, T.; Neu, T.R.; Müller, J.A.; Kästner, M.; Meckenstock, R.U. Selective elimination of bacterial faecal indicators in the Schmutzdecke of slow sand filtration columns. Appl. Microbiol. Biotechnol. 2015, 99, 10323–10332. [Google Scholar] [CrossRef] [PubMed]
- Kadam, A.M.; Nemade, P.D.; Oza, G.H.; Shankar, H.S. Treatment of municipal wastewater using laterite-based constructed soil filter. Ecol. Eng. 2009, 35, 1051–1061. [Google Scholar] [CrossRef]
- Schlimme, W.; Marchiani, M.; Hanselmann, K.; Jenni, B. Gene transfer between bacteria within digestive vacuoles of protozoa. FEMS Microbiol. Ecol. 1997, 23, 239–247. [Google Scholar] [CrossRef]
- USEPA. Method 1603: Escherichia coli (E. coli) in Water by Membrane Filtration Using Modified Membrane-Thermotolerant Escherichia coli Agar (Modified mTEC); Office of Water: Washington, DC, USA, 2002. [Google Scholar]
- Stevik, T.K.; Hanssen, J.F.; Jenssen, P.D. A comparison between DAPI direct count (DDC) and most probable number (MPN) to quantify protozoa in infiltration systems. J. Microbiol. Methods 1998, 33, 13–21. [Google Scholar] [CrossRef]
- Feng, F.; Goto, D.; Yan, T. Effects of autochthonous microbial community on the die-off of fecal indicators in tropical beach sand. FEMS Microbiol. Ecol. 2010, 74, 214–225. [Google Scholar] [CrossRef] [Green Version]
- Bradley, I.; Straub, A.; Maraccini, P.; Markazi, S.; Nguyen, T.H. Iron oxide amended biosand filters for virus removal. Water Res. 2011, 45, 4501–4510. [Google Scholar] [CrossRef] [Green Version]
- Lukasik, J.; Cheng, Y.-F.; Lu, F.; Tamplin, M.; Farrah, S.R. Removal of microorganisms from water by columns containing sand coated with ferric and aluminum hydroxides. Water Res. 1999, 33, 769–777. [Google Scholar] [CrossRef]
- Gülay, A.; Tatari, K.; Musovic, S.; Mateiu, R.V.; Albrechtsen, H.-J.; Smets, B.F. Internal Porosity of Mineral Coating Supports Microbial Activity in Rapid Sand Filters for Groundwater Treatment. Appl. Environ. Microbiol. 2014, 80, 7010–7020. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.-B.; Park, S.-J.; Lee, C.-G.; Kim, H.-C. Transport and retention of Escherichia coli in a mixture of quartz, Al-coated and Fe-coated sands. Hydrol. Process. 2008, 22, 3856–3863. [Google Scholar] [CrossRef]
- Cunningham, A.B.; Characklis, W.G.; Abedeen, F.; Crawford, D. Influence of biofilm accumulation on porous media hydrodynamics. Environ. Sci. Technol. 1991, 25, 1305–1311. [Google Scholar] [CrossRef]
- Surbeck, C.Q.; Jiang, S.C.; Grant, S.B. Ecological Control of Fecal Indicator Bacteria in an Urban Stream. Environ. Sci. Technol. 2010, 44, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Barcina, I.; Lebaron, P.; Vives-Rego, J. Survival of allochthonous bacteria in aquatic systems: A biological approach. FEMS Microbiol. Ecol. 1997, 23, 1–9. [Google Scholar] [CrossRef]
- Posch, T.; Pitsch, G.; Bruni, E.P. Protists: Ciliates. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Eisenmann, H.; Harms, H.; Meckenstock, R.; Meyer, E.I.; Zehnder, A.J.B. Grazing of a Tetrahymena sp. on Adhered Bacteria in Percolated Columns Monitored by In Situ Hybridization with Fluorescent Oligonucleotide Probes. Appl. Environ. Microbiol. 1998, 64, 1264–1269. [Google Scholar] [CrossRef] [Green Version]
- Kadam, A.M.; Oza, G.H.; Nemade, P.D.; Shankar, H.S. Pathogen removal from municipal wastewater in Constructed Soil Filter. Ecol. Eng. 2008, 33, 37–44. [Google Scholar] [CrossRef]
- Vanotti, M.B.; Szogi, A.A.; Hunt, P.G.; Millner, P.D.; Humenik, F.J. Development of environmentally superior treatment system to replace anaerobic swine lagoons in the USA. Bioresour. Technol. 2007, 98, 3184–3194. [Google Scholar] [CrossRef] [PubMed]



| Treatment | Total Mass E. coli in Water, CFU | Total Mass Absorbed E. coli in Soil, CFU | Active Predators in Soil Column, MPN | Grazing Rate (E. coli/Protozoa.h) | ||||
|---|---|---|---|---|---|---|---|---|
| Influent | Effluent | Retained Cells | Live Cells | Death Cells | ||||
| NPG | Column 1 | 7.44 × 109 | 1.10 × 109 | 6.35 × 109 | 5.98 × 107 | 6.29 × 109 | 1.00 × 107 | 146.0 |
| Column 2 | 1.06 × 1010 | 3.15 × 109 | 7.45 × 109 | 5.82 × 107 | 7.40 × 109 | 1.17 × 107 | 146.5 | |
| PEP | Column 1 | 8.36 × 109 | 5.24 × 108 | 7.84 × 109 | 1.50 × 107 | 7.82 × 109 | 5.37 × 107 | 33.9 |
| Column 2 | 9.38 × 109 | 4.23 × 108 | 8.96 × 109 | 6.30 × 106 | 8.95 × 109 | 6.74 × 107 | 30.9 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, L.; Yan, T.; Yost, R.; Porter, G. A Sustainable and Low-Cost Soil Filter Column for Removing Pathogens from Swine Wastewater: The Role of Endogenous Soil Protozoa. Water 2021, 13, 2472. https://doi.org/10.3390/w13182472
Kim L, Yan T, Yost R, Porter G. A Sustainable and Low-Cost Soil Filter Column for Removing Pathogens from Swine Wastewater: The Role of Endogenous Soil Protozoa. Water. 2021; 13(18):2472. https://doi.org/10.3390/w13182472
Chicago/Turabian StyleKim, Lavane, Tao Yan, Russell Yost, and Guy Porter. 2021. "A Sustainable and Low-Cost Soil Filter Column for Removing Pathogens from Swine Wastewater: The Role of Endogenous Soil Protozoa" Water 13, no. 18: 2472. https://doi.org/10.3390/w13182472
APA StyleKim, L., Yan, T., Yost, R., & Porter, G. (2021). A Sustainable and Low-Cost Soil Filter Column for Removing Pathogens from Swine Wastewater: The Role of Endogenous Soil Protozoa. Water, 13(18), 2472. https://doi.org/10.3390/w13182472






