Investigation of Biofilm Growth within a Monodisperse Porous Medium under Fluctuating Water Level Assessed by Means of MRI
Abstract
:1. Introduction
2. Materials and Methods
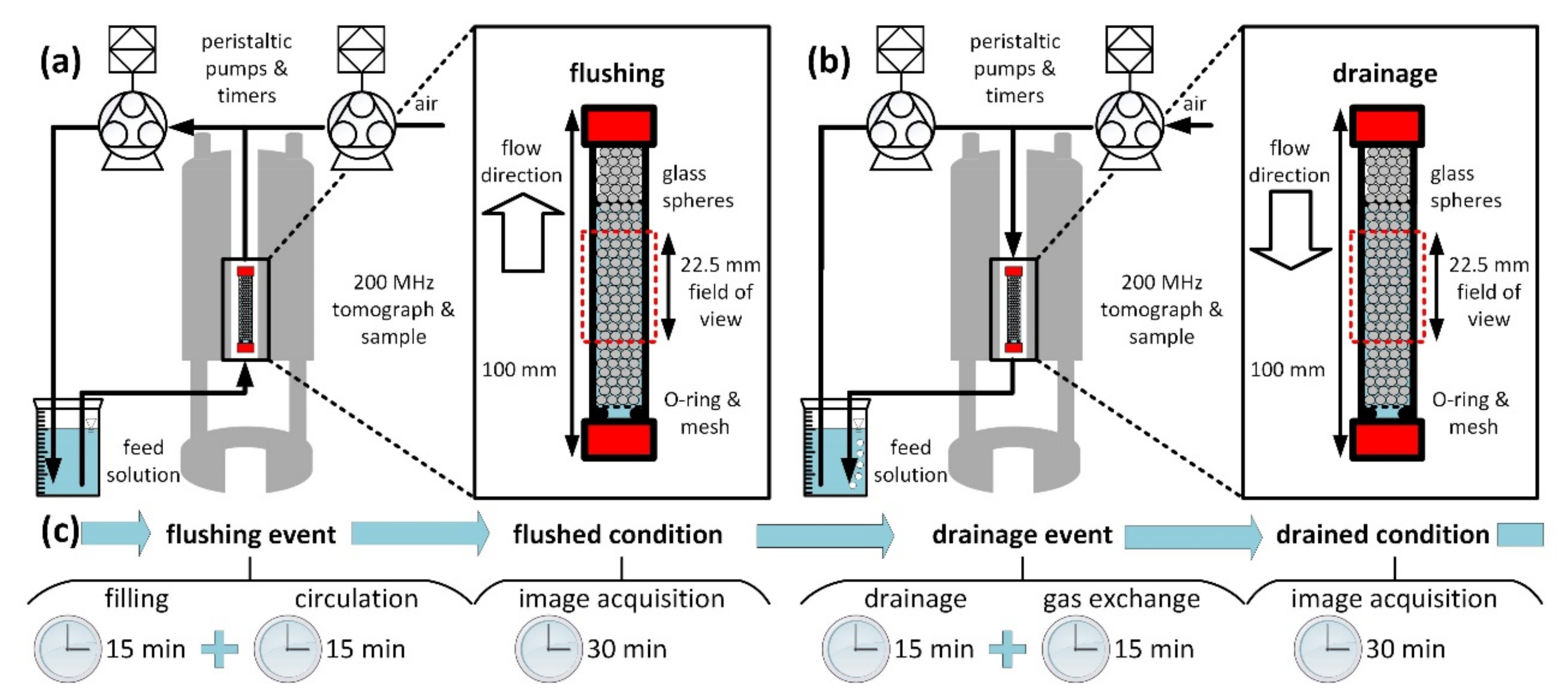
2.1. Experimental Setup and Procedure
2.2. Cultivation
2.3. Optical Coherence Tomography (OCT)
2.4. Magnetic Resonance Imaging (MRI)
2.5. Image Analysis
3. Results
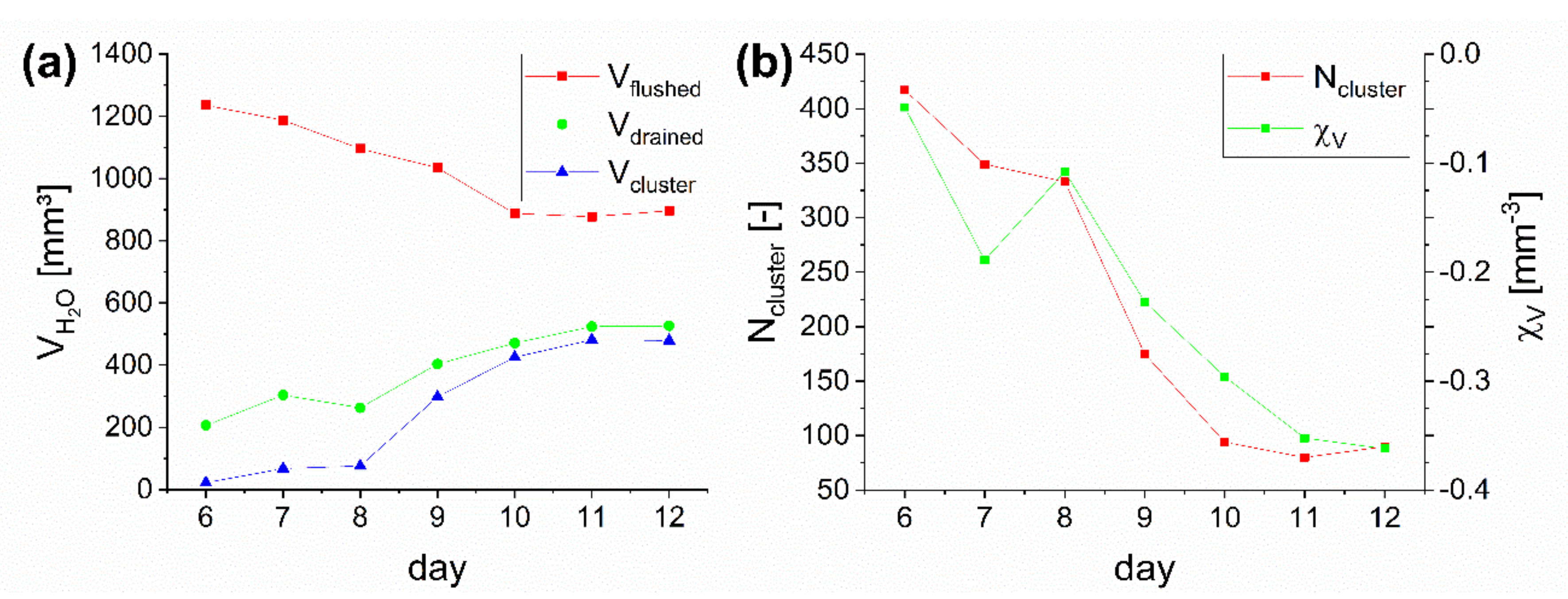
3.1. Dynamics of Water Content
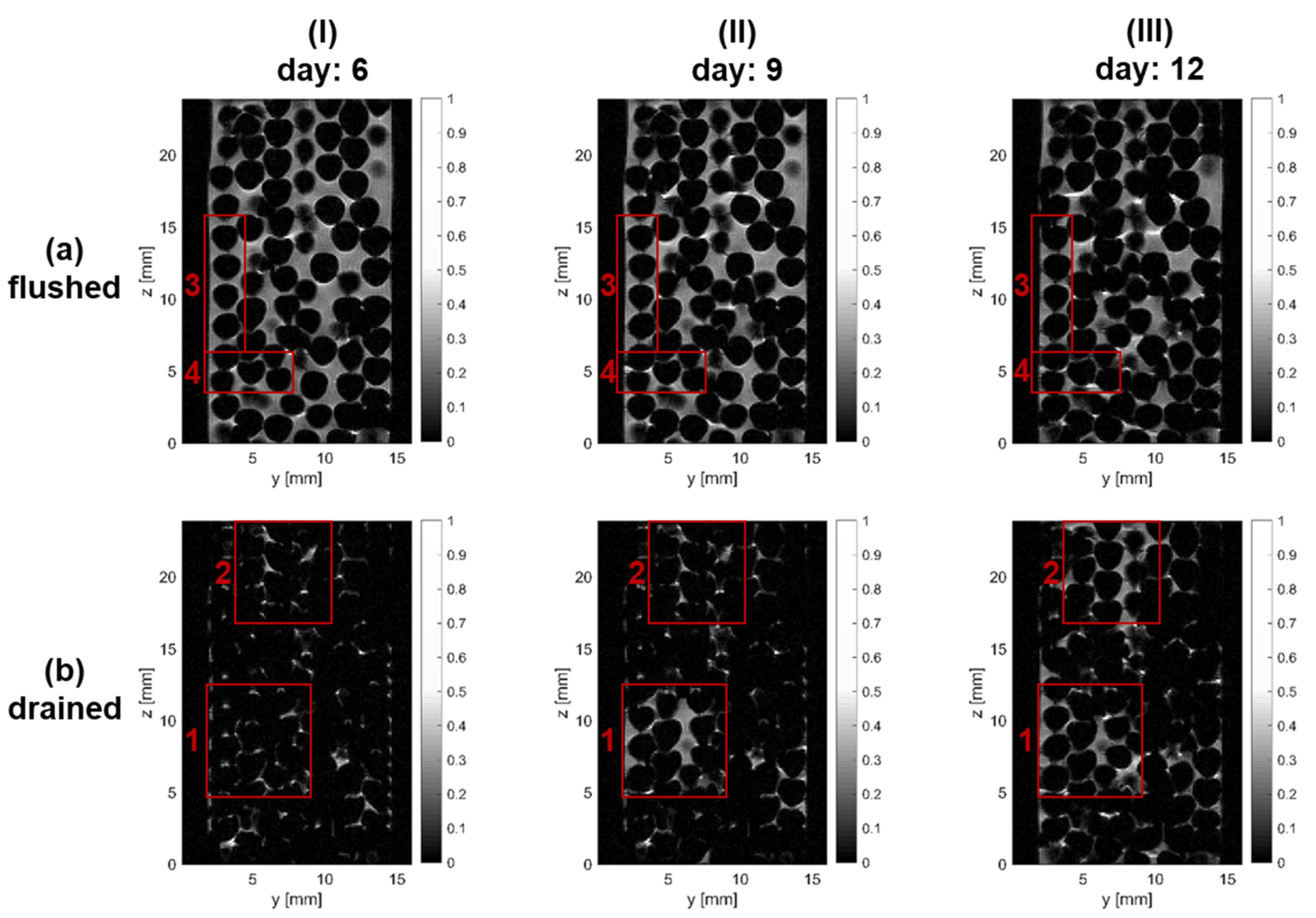
3.2. Biofilm Growth
3.3. Analysis of Liquid Phase
4. Discussion
4.1. Applicability of the MRI Approach
4.2. Biofilm Growth under Fluctuating Water Level
4.3. Impact of Biofilm on the Liquid Phase
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Cultivation under Saturated Conditions for OCT Measurements
| Substance | Concentration [mg/L] |
|---|---|
| Tryptophan | 5 |
| Phenylalanine | 5 |
| Thiamine hydrochloride | 0.67 |
| ZnCl2 | 0.136 |
| MnCl2 | 9.9 |
| FeCl2 | 0.5 |
| CaCl2 | 110 |
| MgCl2 | 410 |
| Glycerol | 500 |
Appendix B

References
- Tecon, R.; Or, D. Biophysical processes supporting the diversity of microbial life in soil. FEMS Microbiol. Rev. 2017, 41, 599–623. [Google Scholar] [CrossRef] [PubMed]
- Brussaard, L. Ecosystem services provided by the soil biota. In Soil Ecology and Ecosystem Services; Oxford University Press: Oxford, UK, 2012; pp. 45–58. [Google Scholar]
- Or, D.; Smets, B.F.; Wraith, J.M.; Dechesne, A.; Friedman, S.P. Physical constraints affecting bacterial habitats and activity in unsaturated porous media—A review. Adv. Water Resour. 2007, 30, 1505–1527. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Zeng, S.; Bais, H.; LaManna, J.M.; Hussey, D.S.; Jacobson, D.L.; Jin, Y. Plant Growth-Promoting Rhizobacteria (PGPR) Reduce Evaporation and Increase Soil Water Retention. Water Resour. Res. 2018, 54, 3673–3687. [Google Scholar] [CrossRef]
- Volk, E.; Iden, S.C.; Furman, A.; Durner, W.; Rosenzweig, R. Biofilm effect on soil hydraulic properties: Experimental investigation using soil-grown real biofilm. Water Resour. Res. 2016, 52, 5813–5828. [Google Scholar] [CrossRef] [Green Version]
- Bozorg, A.; Gates, I.D.; Sen, A. Using bacterial bioluminescence to evaluate the impact of biofilm on porous media hydraulic properties. J. Microbiol. Methods 2015, 109, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Roberson, E.B.; Firestone, M.K. Relationship between desiccation and exopolysaccharide production in a soil Pseudomonas sp. Appl. Environ. Microbiol. 1992, 58, 1284–1291. [Google Scholar] [CrossRef] [Green Version]
- Or, D.; Phutane, S.; Dechesne, A. Extracellular polymeric substances affecting pore-scale hydrologic conditions for bacterial activity in unsaturated soils. Vadose Zone J. 2007, 6, 298–305. [Google Scholar] [CrossRef]
- Chenu, C.; Roberson, E. Diffusion of glucose in microbial extracellular polysaccharide as affected by water potential. Soil Biol. Biochem. 1996, 28, 877–884. [Google Scholar] [CrossRef]
- Carrel, M.; Morales, V.L.; Beltran, M.A.; Derlon, N.; Kaufmann, R.; Morgenroth, E.; Holzner, M. Biofilms in 3D porous media: Delineating the influence of the pore network geometry, flow and mass transfer on biofilm development. Water Res. 2018, 134, 280–291. [Google Scholar] [CrossRef] [Green Version]
- Peszynska, M.; Trykozko, A.; Iltis, G.; Schlueter, S.; Wildenschild, D. Biofilm growth in porous media: Experiments, computational modeling at the porescale, and upscaling. Adv. Water Resour. 2016, 95, 288–301. [Google Scholar] [CrossRef] [Green Version]
- Iltis, G.C.; Armstrong, R.T.; Jansik, D.P.; Wood, B.D.; Wildenschild, D. Imaging biofilm architecture within porous media using synchrotron-based X-ray computed microtomography. Water Resour. Res. 2011, 47. [Google Scholar] [CrossRef]
- Seymour, J.D.; Gage, J.P.; Codd, S.L.; Gerlach, R. Anomalous fluid transport in porous media induced by biofilm growth. Phys. Rev. Lett. 2004, 93, 198103. [Google Scholar] [CrossRef] [PubMed]
- Graf von der Schulenburg, D.A.; Akpa, B.S.; Gladden, L.F.; Johns, M.L. Non-invasive mass transfer measurements in complex biofilm-coated structures. Biotechnol. Bioeng. 2008, 101, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Metzger, U.; Lankes, U.; Hardy, E.H.; Gordalla, B.C.; Frimmel, F.H. Monitoring the formation of an Aureobasidium pullulans biofilm in a bead-packed reactor via flow-weighted magnetic resonance imaging. Biotechnol. Lett. 2006, 28, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Vogt, S.J.; Sanderlin, A.B.; Seymour, J.D.; Codd, S.L. Permeability of a growing biofilm in a porous media fluid flow analyzed by magnetic resonance displacement-relaxation correlations. Biotechnol. Bioeng. 2013, 110, 1366–1375. [Google Scholar] [CrossRef] [Green Version]
- Valiei, A.; Kumar, A.; Mukherjee, P.P.; Liu, Y.; Thundat, T. A web of streamers: Biofilm formation in a porous microfluidic device. Lab Chip 2012, 12, 5133–5137. [Google Scholar] [CrossRef]
- Sharp, R.R.; Stoodley, P.; Adgie, M.; Gerlach, R.; Cunningham, A. Visualization and characterization of dynamic patterns of flow, growth and activity of biofilms growing in porous media. Water Sci. Technol. 2005, 52, 85–90. [Google Scholar] [CrossRef]
- Karimifard, S.; Li, X.; Elowsky, C.; Li, Y. Modeling the impact of evolving biofilms on flow in porous media inside a microfluidic channel. Water Res. 2021, 188, 116536. [Google Scholar] [CrossRef]
- Ranzinger, F.; Hille-Reichel, A.; Zehe, E.; Guthausen, G.; Horn, H. Quantification of evaporation and drainage processes in unsaturated porous media using Magnetic Resonance Imaging (MRI). Water Resour. Res. 2020. [Google Scholar] [CrossRef] [Green Version]
- Wagner, M.; Horn, H. Optical coherence tomography in biofilm research: A comprehensive review. Biotechnol. Bioeng. 2017, 114, 1386–1402. [Google Scholar] [CrossRef] [PubMed]
- Vogel, H.-J.; Roth, K. Quantitative morphology and network representation of soil pore structure. Adv. Water Resour. 2001, 24, 233–242. [Google Scholar] [CrossRef]
- Caizán-Juanarena, L.; Krug, J.R.; Vergeldt, F.J.; Kleijn, J.M.; Velders, A.H.; Van As, H.; Ter Heijne, A. 3D biofilm visualization and quantification on granular bioanodes with magnetic resonance imaging. Water Res. 2019, 167, 115059. [Google Scholar] [CrossRef] [PubMed]
- Ranzinger, F.; Herrling, M.P.; Lackner, S.; Grande, V.W.; Baniodeh, A.; Powell, A.K.; Horn, H.; Guthausen, G. Direct surface visualization of biofilms with high spin coordination clusters using magnetic resonance imaging. Acta Biomater. 2016, 31, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Nott, K.P.; Paterson-Beedle, M.; Macaskie, L.E.; Hall, L.D. Visualisation of metal deposition in biofilm reactors by three-dimensional magnetic resonance imaging (MRI). Biotechnol. Lett. 2001, 23, 1749–1757. [Google Scholar] [CrossRef]
- Peulen, T.-O.; Wilkinson, K.J. Diffusion of nanoparticles in a biofilm. Environ. Sci. Technol. 2011, 45, 3367–3373. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Kolter, R. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: A genetic analysis. Mol. Microbiol. 1998, 28, 449–461. [Google Scholar] [CrossRef]
- Hu, X.; Chen, K.; Lai, X.; Ji, S.; Kaiser, K. Effects of Fe (III) on biofilm and its extracellular polymeric substances (EPS) in fixed bed biofilm reactors. Water Sci. Technol. 2016, 73, 2060–2066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rittmann, B.E. The significance of biofilms in porous media. Water Resour. Res. 1993, 29, 2195–2202. [Google Scholar] [CrossRef]
- Rockhold, M.L.; Yarwood, R.R.; Niemet, M.R.; Bottomley, P.J.; Selker, J.S. Considerations for modeling bacterial-induced changes in hydraulic properties of variably saturated porous media. Adv. Water Resour. 2002, 25, 477–495. [Google Scholar] [CrossRef]
- Chenu, C. Clay—or sand—polysaccharide associations as models for the interface between micro-organisms and soil: Water related properties and microstructure. In Soil Structure/Soil Biota Interrelationships; Elsevier: Amsterdam, The Netherlands, 1993; pp. 143–156. [Google Scholar]


| MSME 2D | MSME 3D | |
|---|---|---|
| repetition time [sec] | 1.5 | 0.5 |
| echo time [sec] | 0.01 | 0.01 |
| field of view (x, y, z) [mm × mm × mm] | 16 × 24 (6 slices in y-direction) | 15 × 15 × 24 |
| matrix size (x, y, z) | 128 × 6 × 192 | 64 × 64 × 128 |
| voxel size (x, y, z) [µm × µm × µm] | 125 × 125 (200 µm slice thickness in y-direction) | 234 × 234 × 188 |
| number of averages [-] | 4 | 1 |
| measurement time [min:sec] | 12:48 | 34:08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ranzinger, F.; Schröter, K.; Horn, H.; Wagner, M. Investigation of Biofilm Growth within a Monodisperse Porous Medium under Fluctuating Water Level Assessed by Means of MRI. Water 2021, 13, 2456. https://doi.org/10.3390/w13182456
Ranzinger F, Schröter K, Horn H, Wagner M. Investigation of Biofilm Growth within a Monodisperse Porous Medium under Fluctuating Water Level Assessed by Means of MRI. Water. 2021; 13(18):2456. https://doi.org/10.3390/w13182456
Chicago/Turabian StyleRanzinger, Florian, Karin Schröter, Harald Horn, and Michael Wagner. 2021. "Investigation of Biofilm Growth within a Monodisperse Porous Medium under Fluctuating Water Level Assessed by Means of MRI" Water 13, no. 18: 2456. https://doi.org/10.3390/w13182456






