Dolomitization of Paleozoic Successions, Huron Domain of Southern Ontario, Canada: Fluid Flow and Dolomite Evolution
Abstract
:1. Introduction
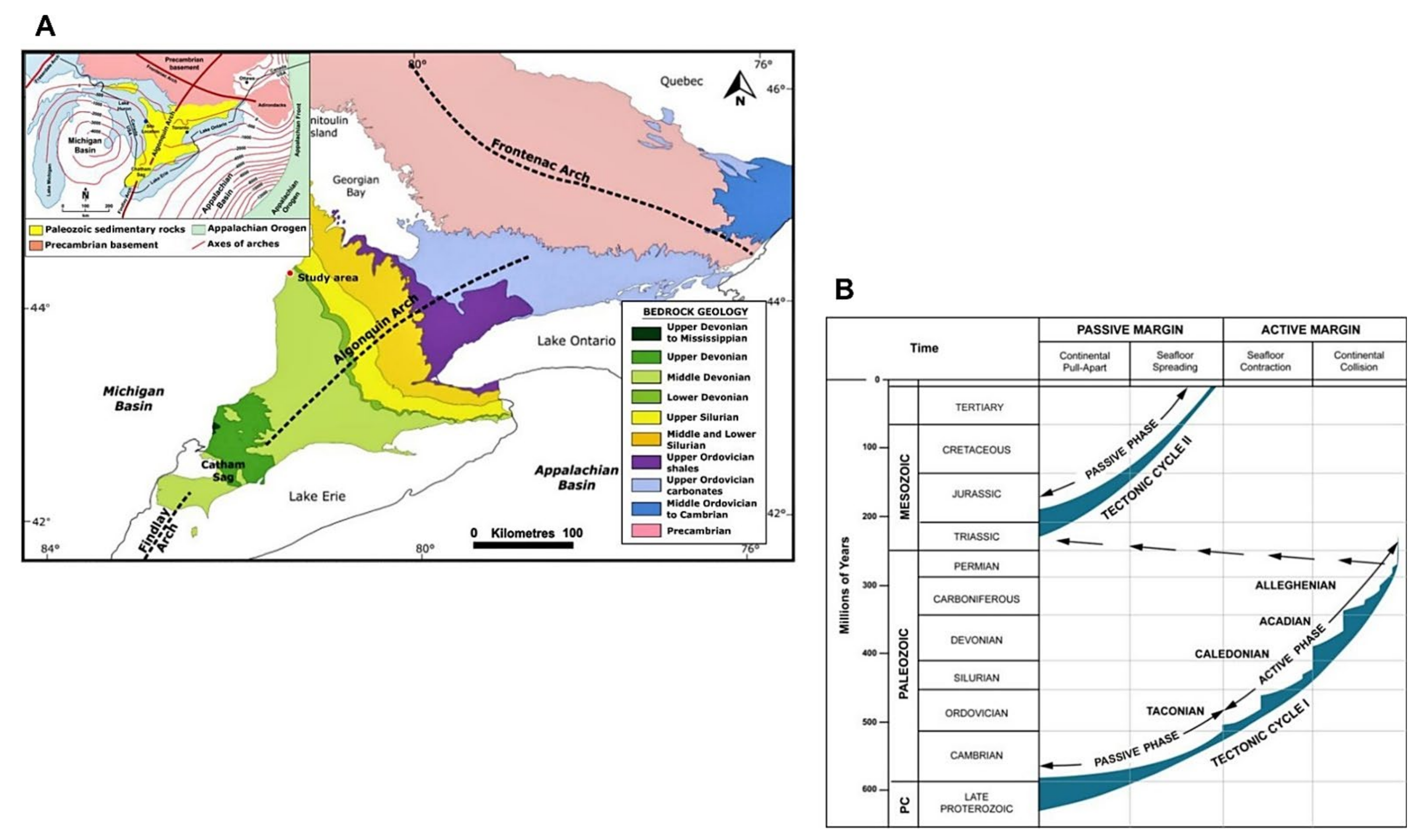
2. Regional Geologic, Tectonics, and Stratigraphic Framework
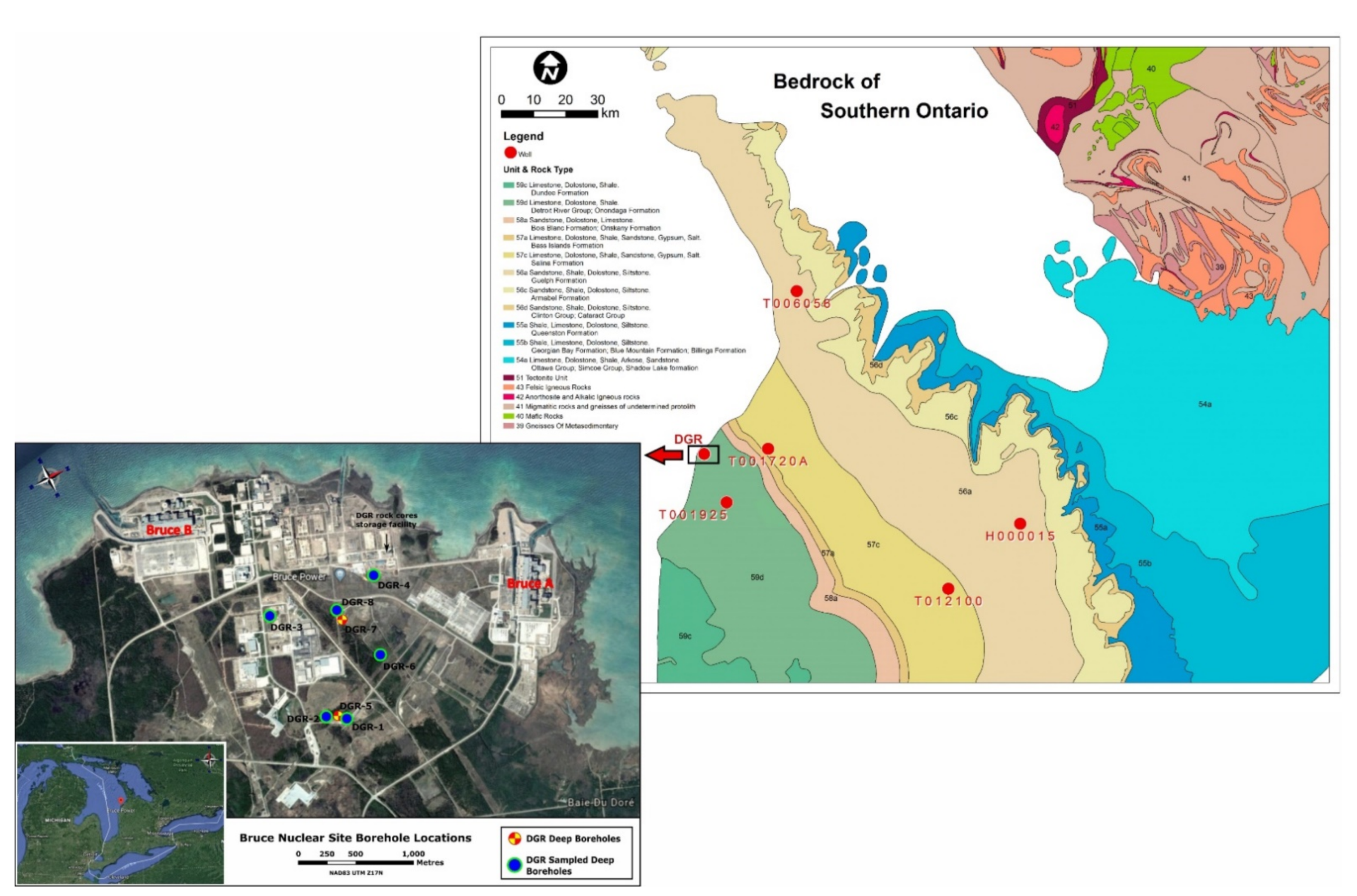
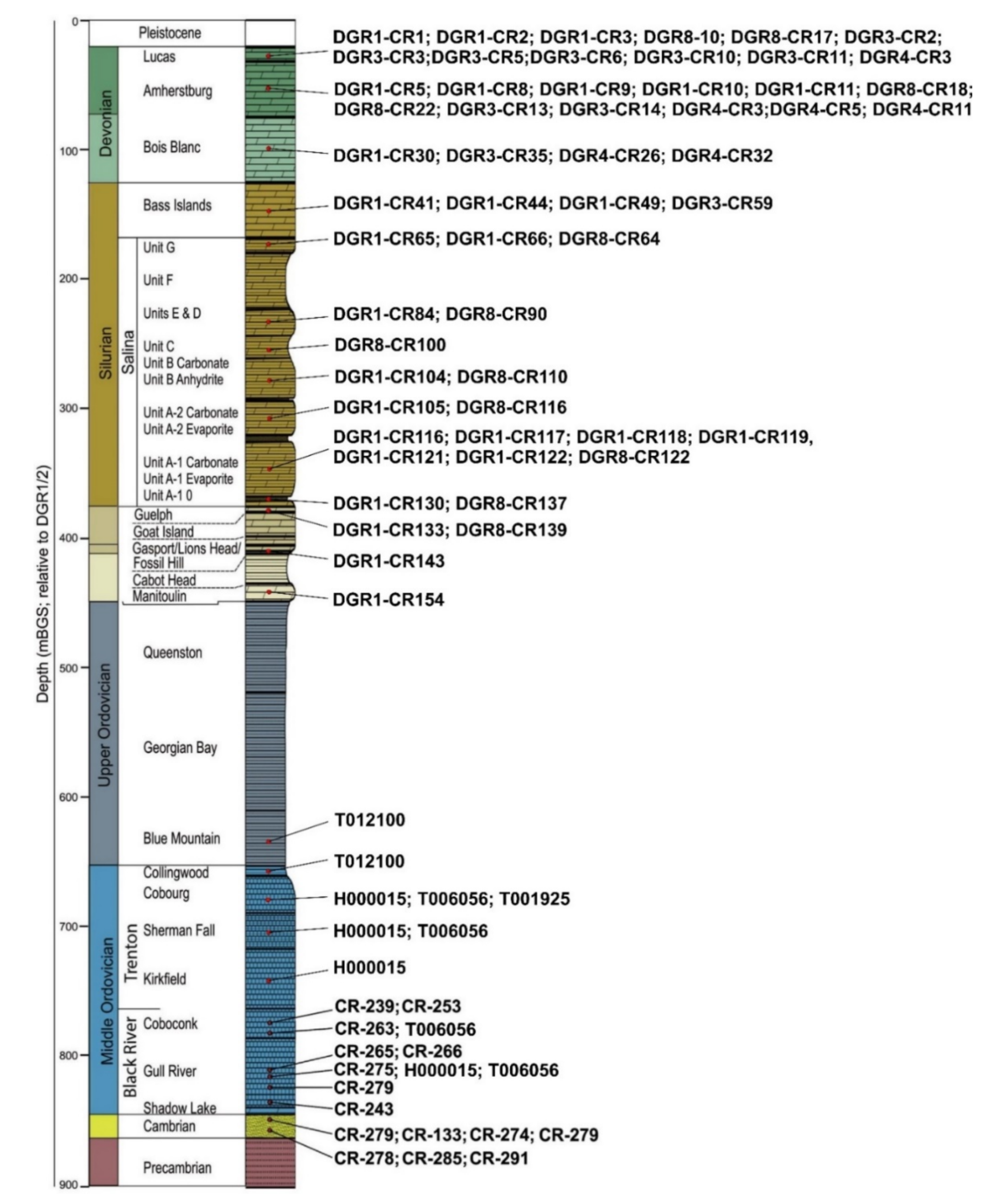
3. Sampling and Analytical Methods
4. Results
4.1. Petrographic Analysis
4.1.1. Dolomite Types
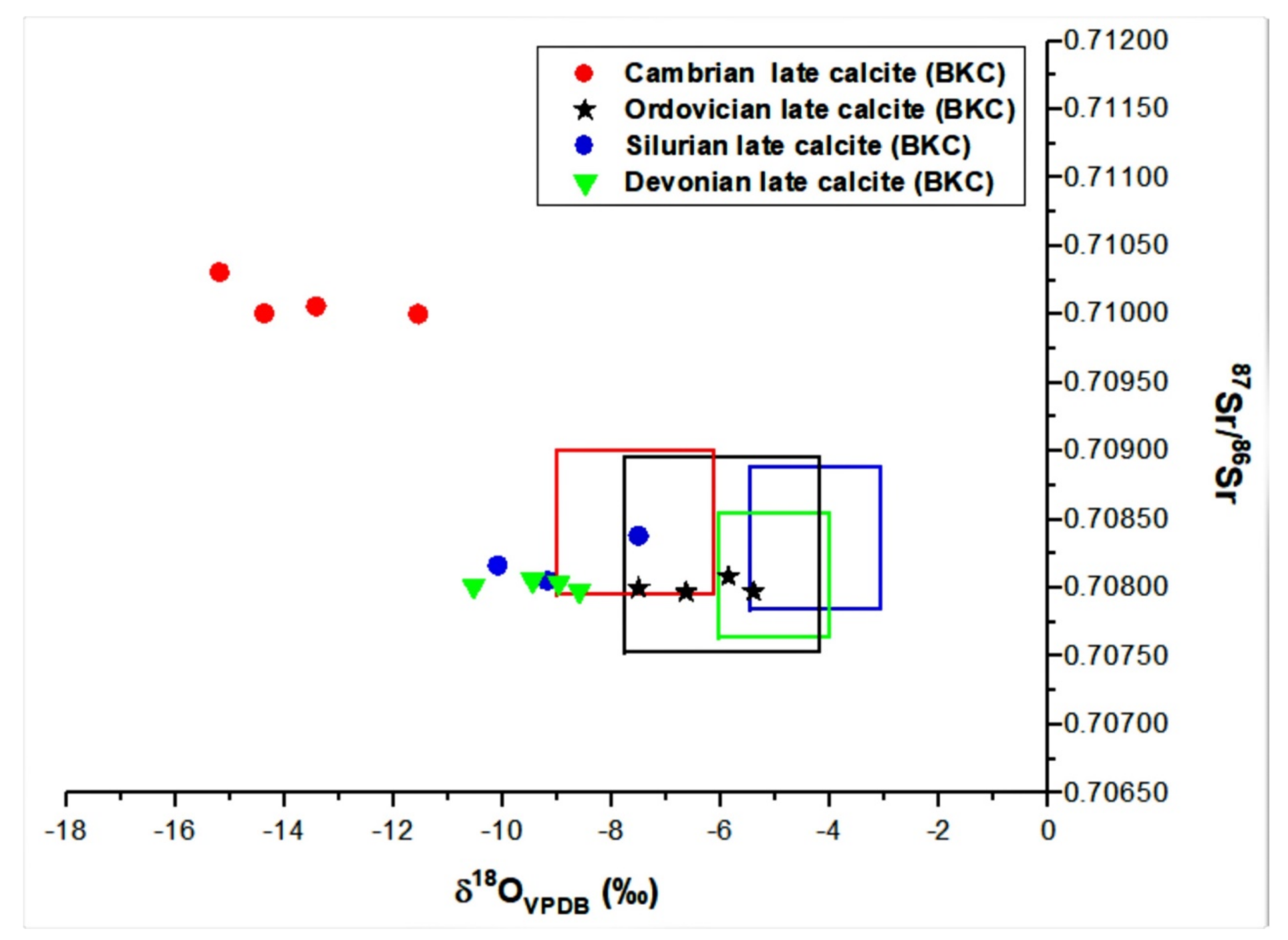
4.1.2. Calcite Cementation
4.1.3. Other Diagenetic Phases
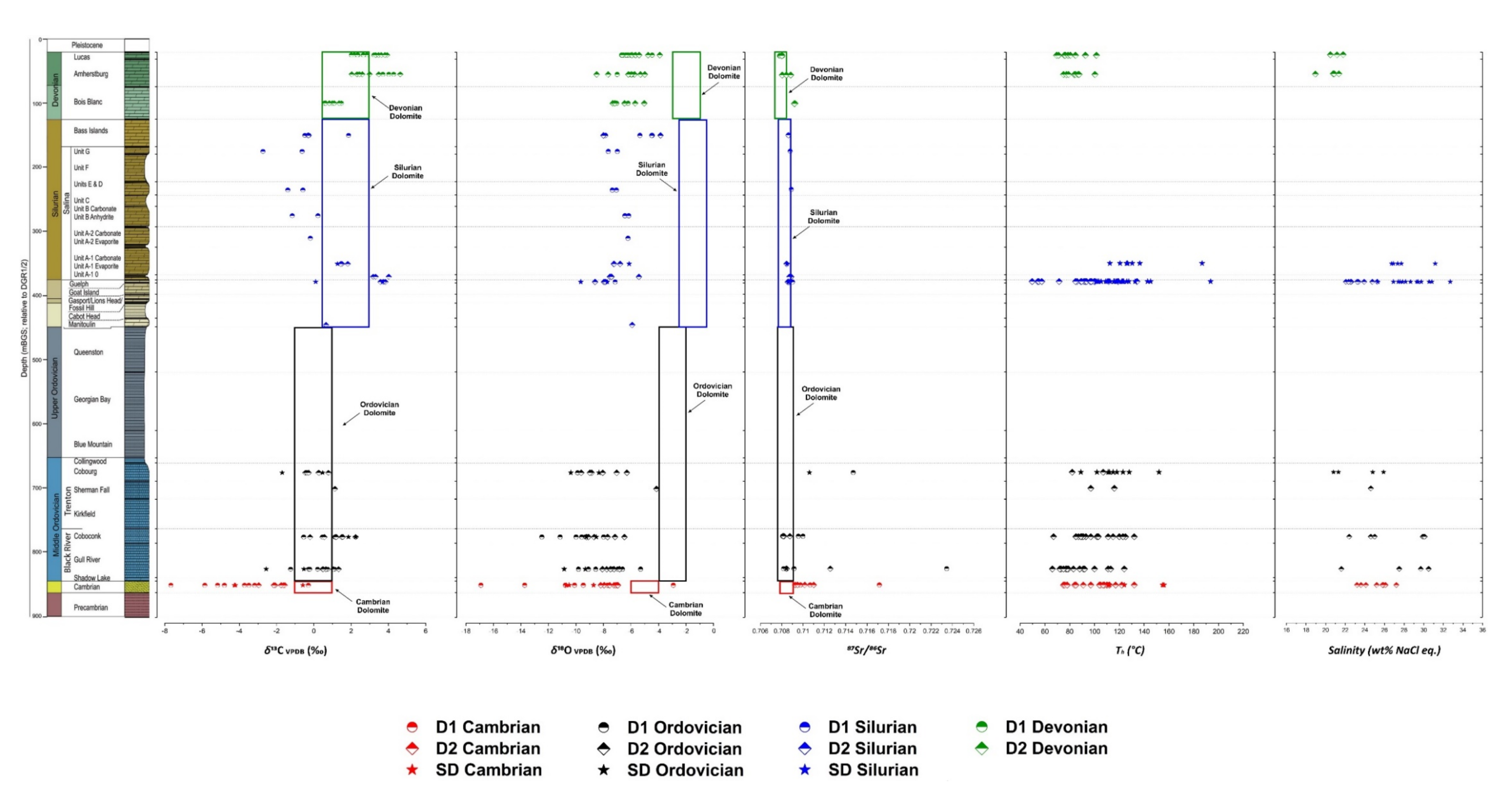
4.2. Geochemical and Fluid Inclusion Results
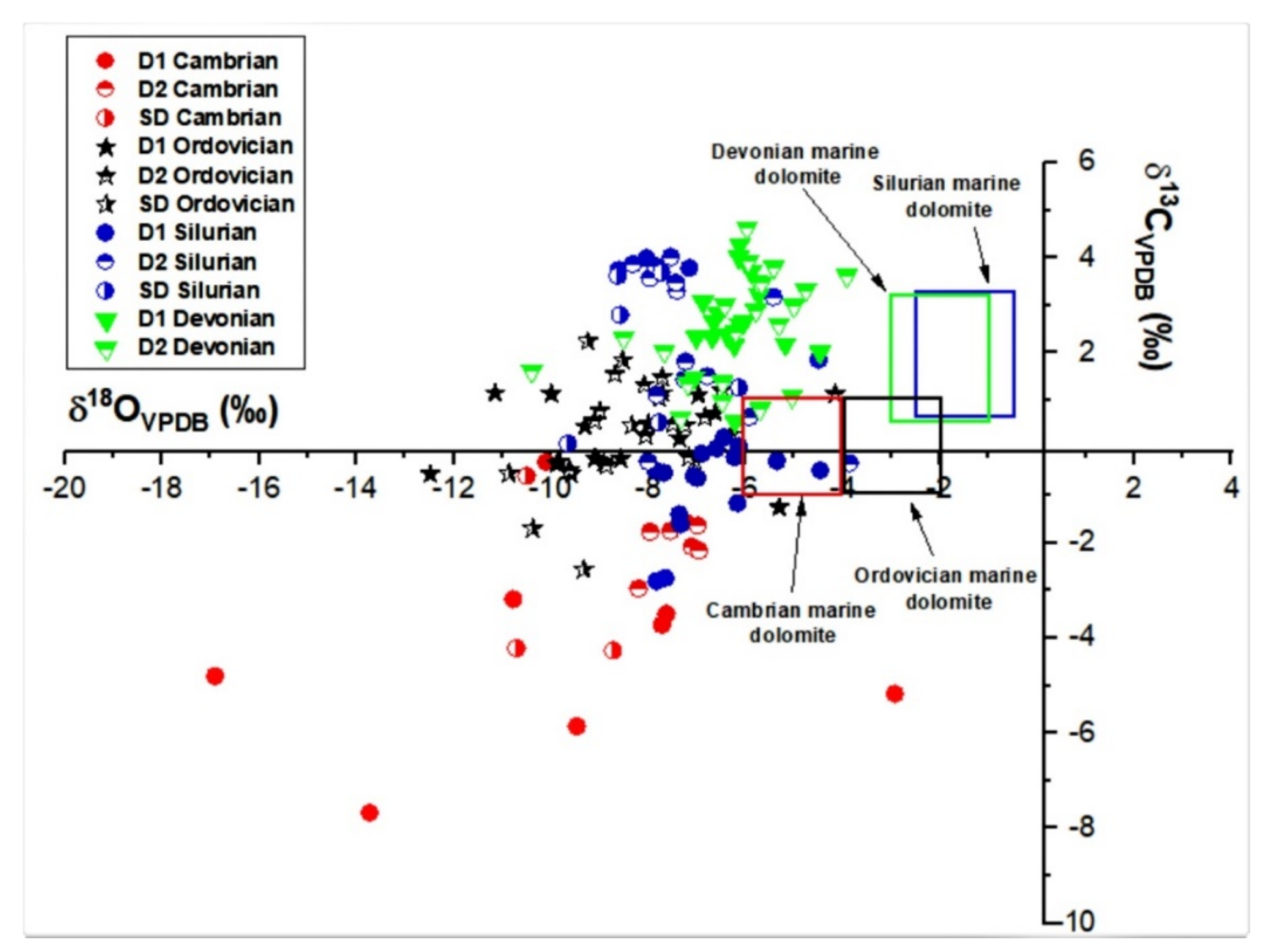
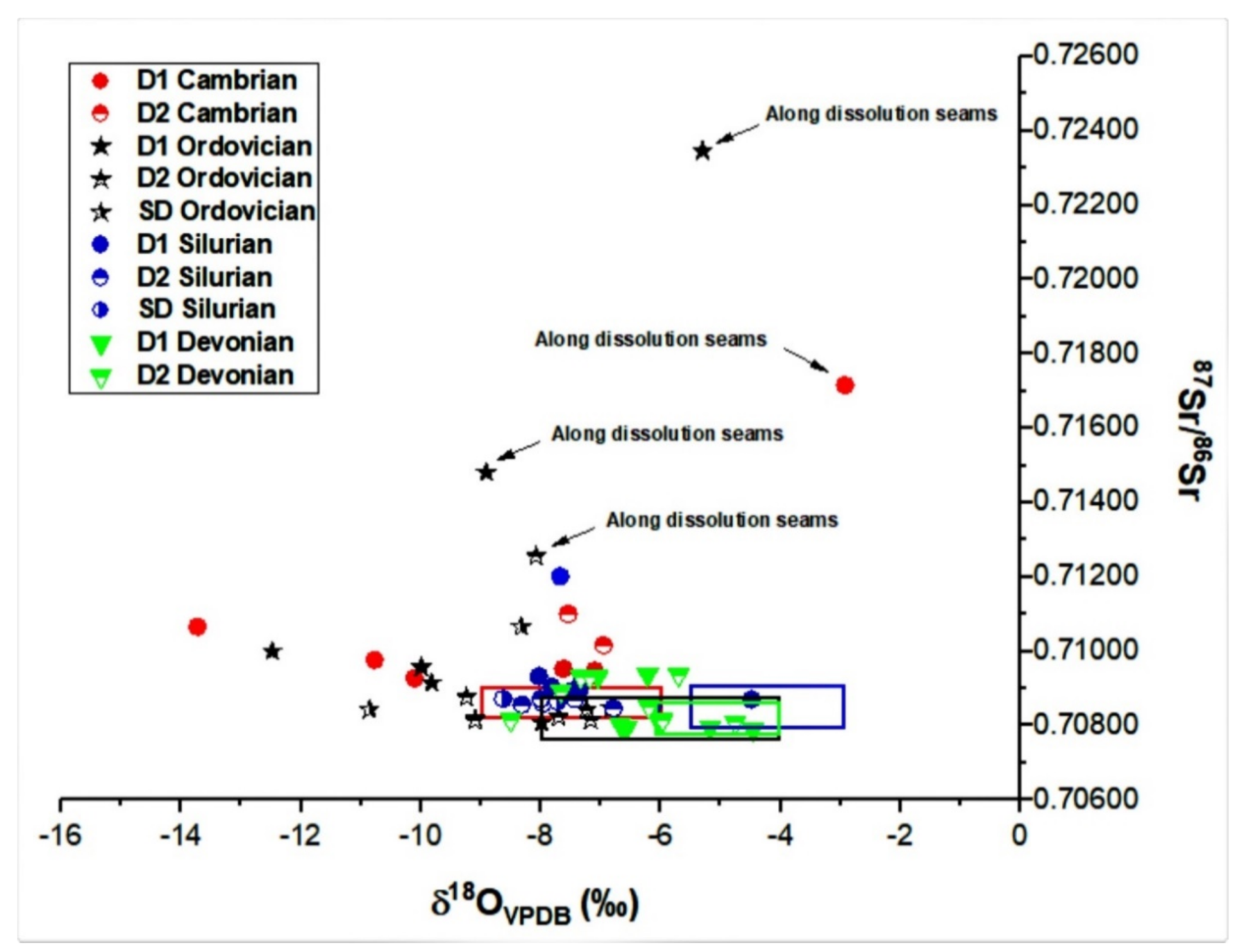
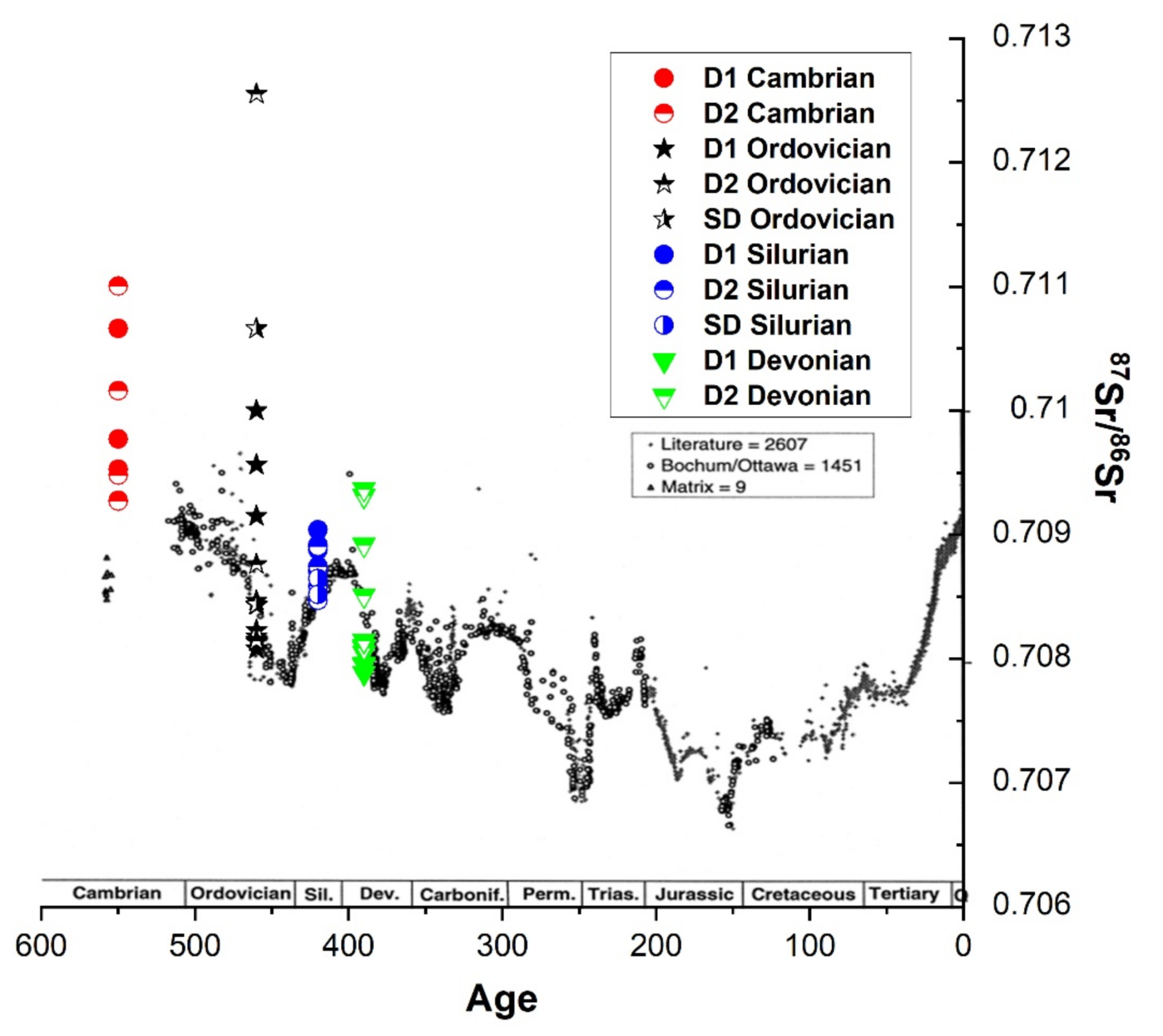
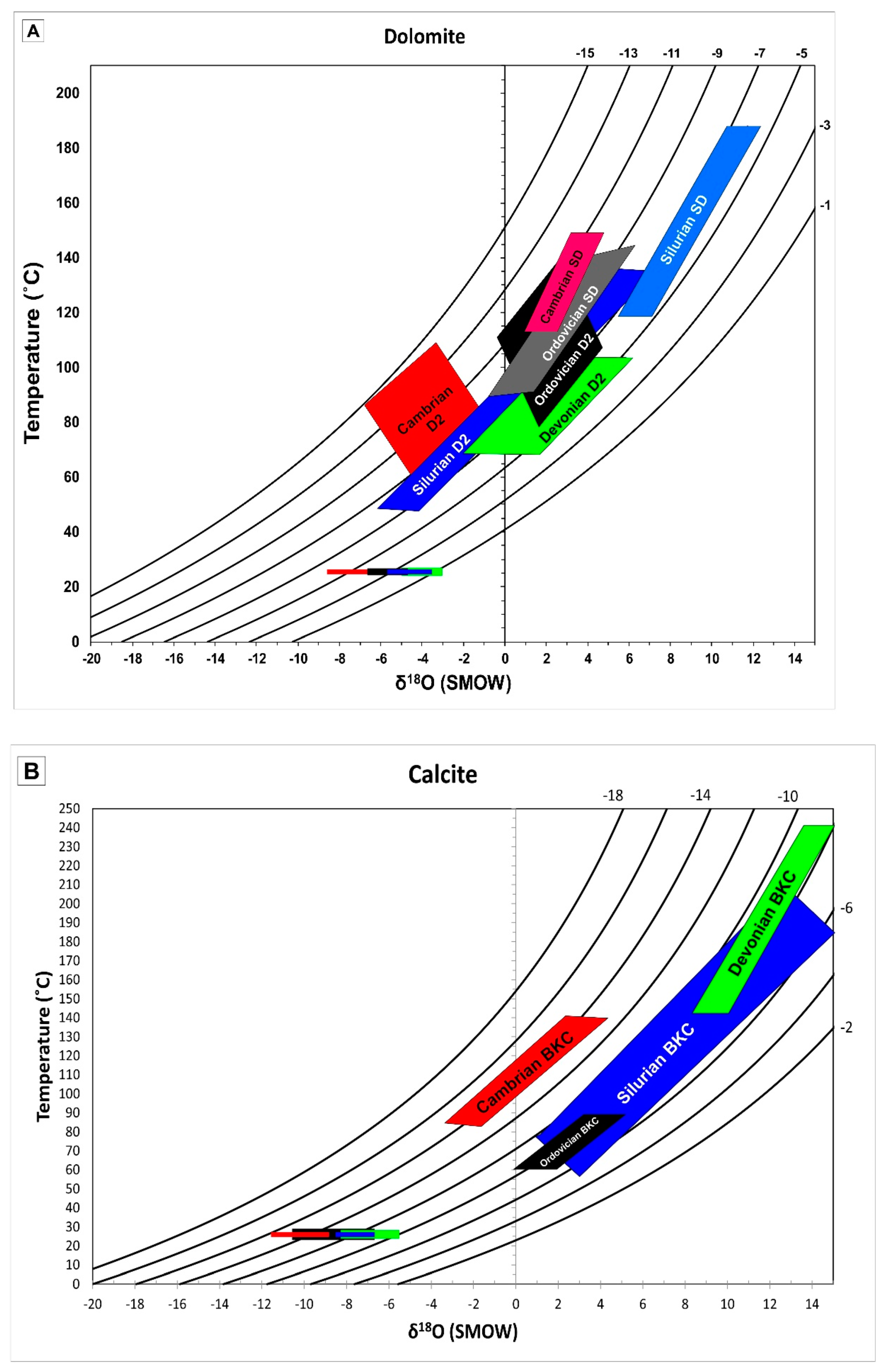
4.2.1. Oxygen, Carbon and Sr Isotopes
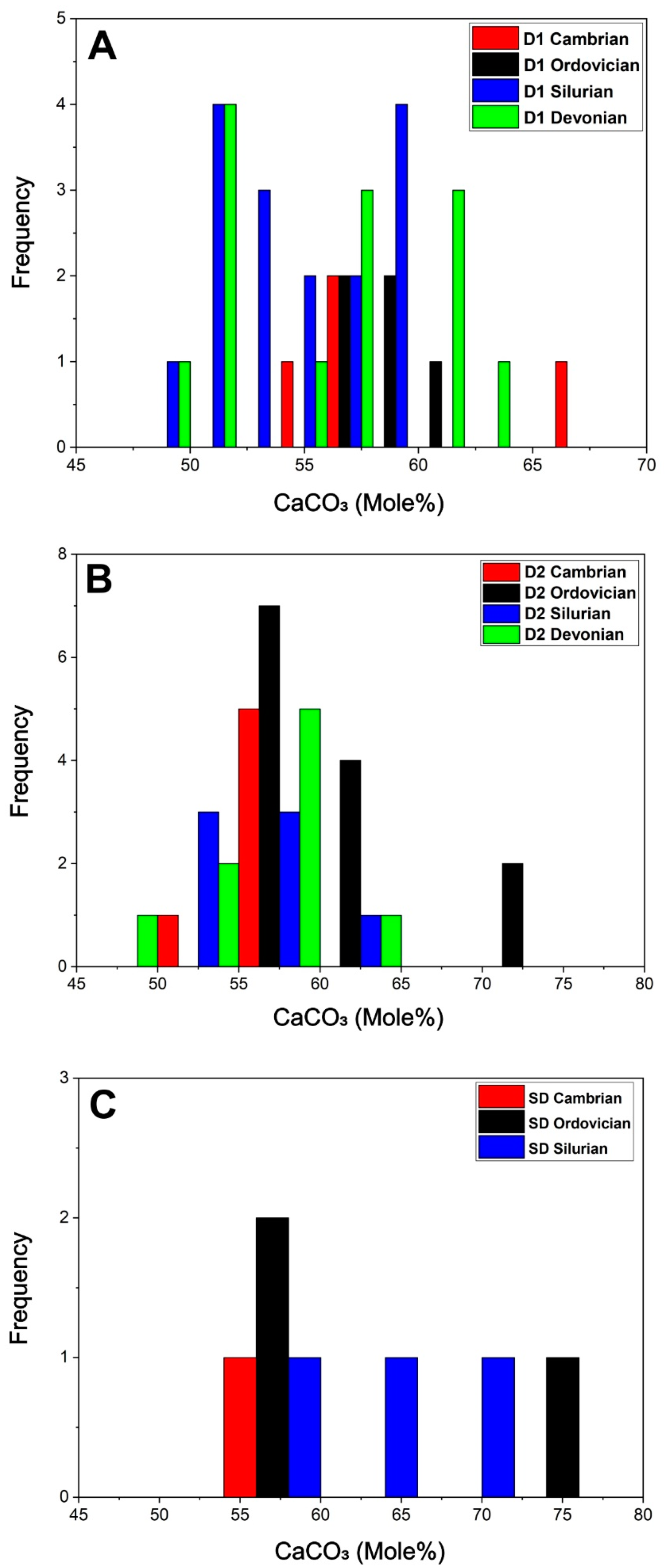
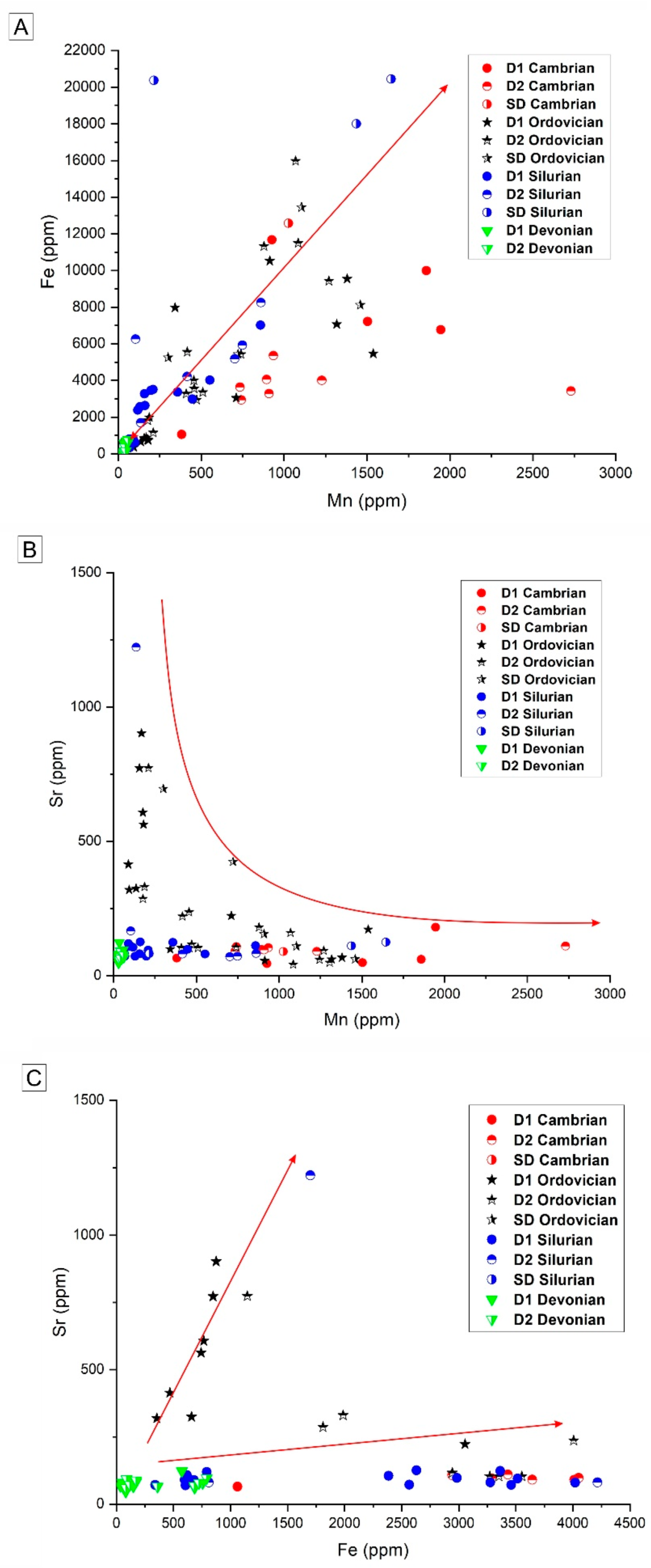
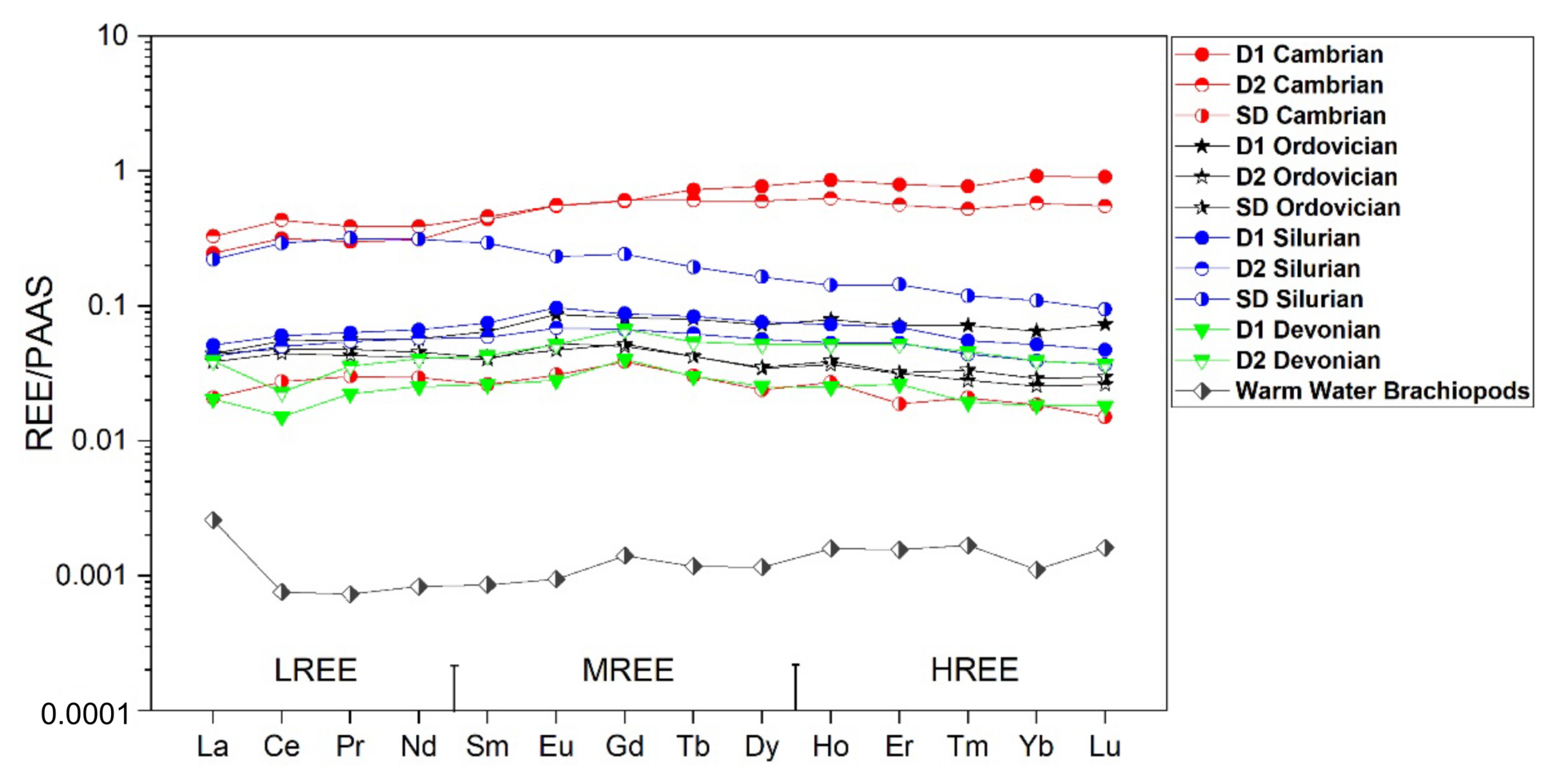
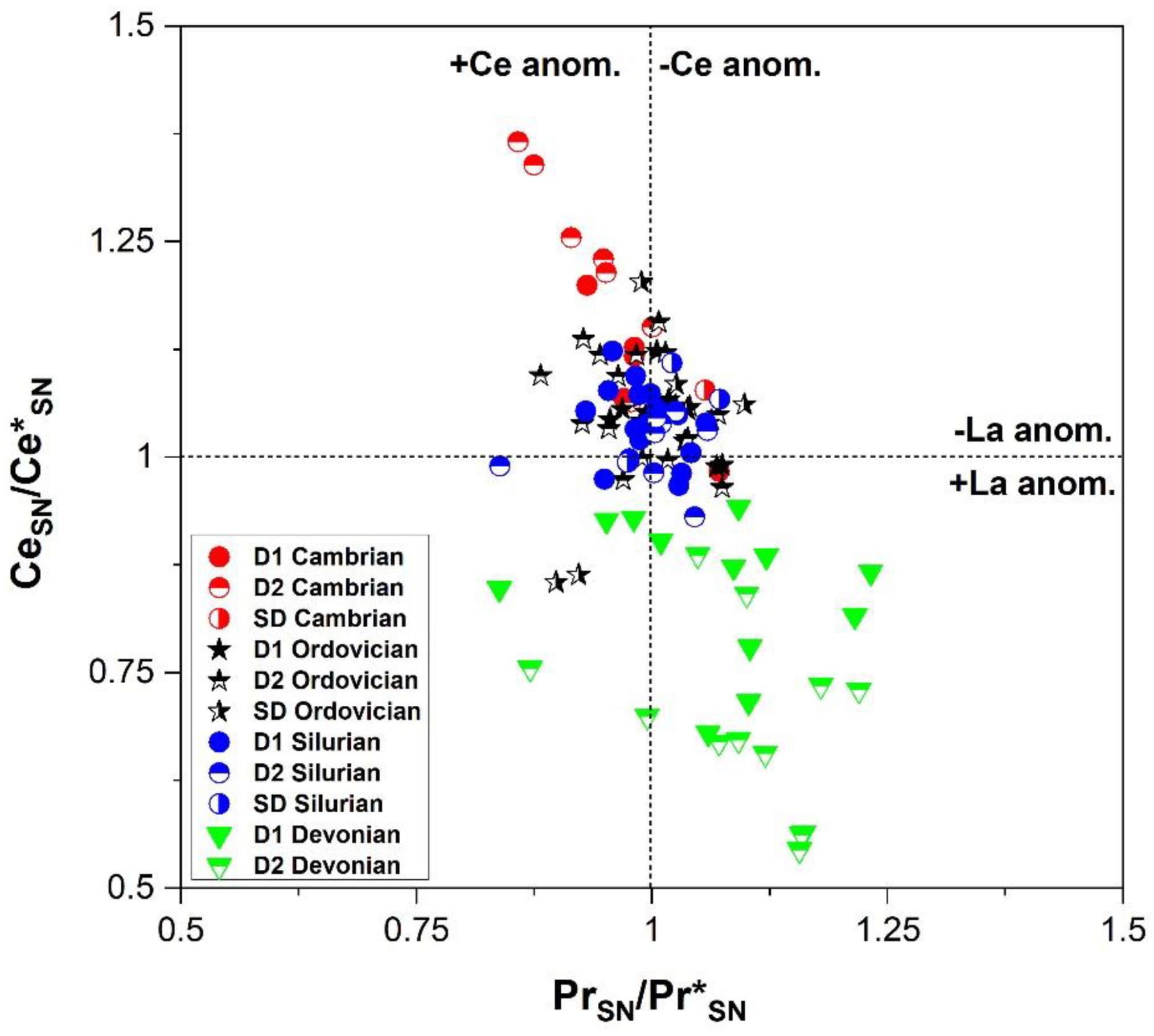
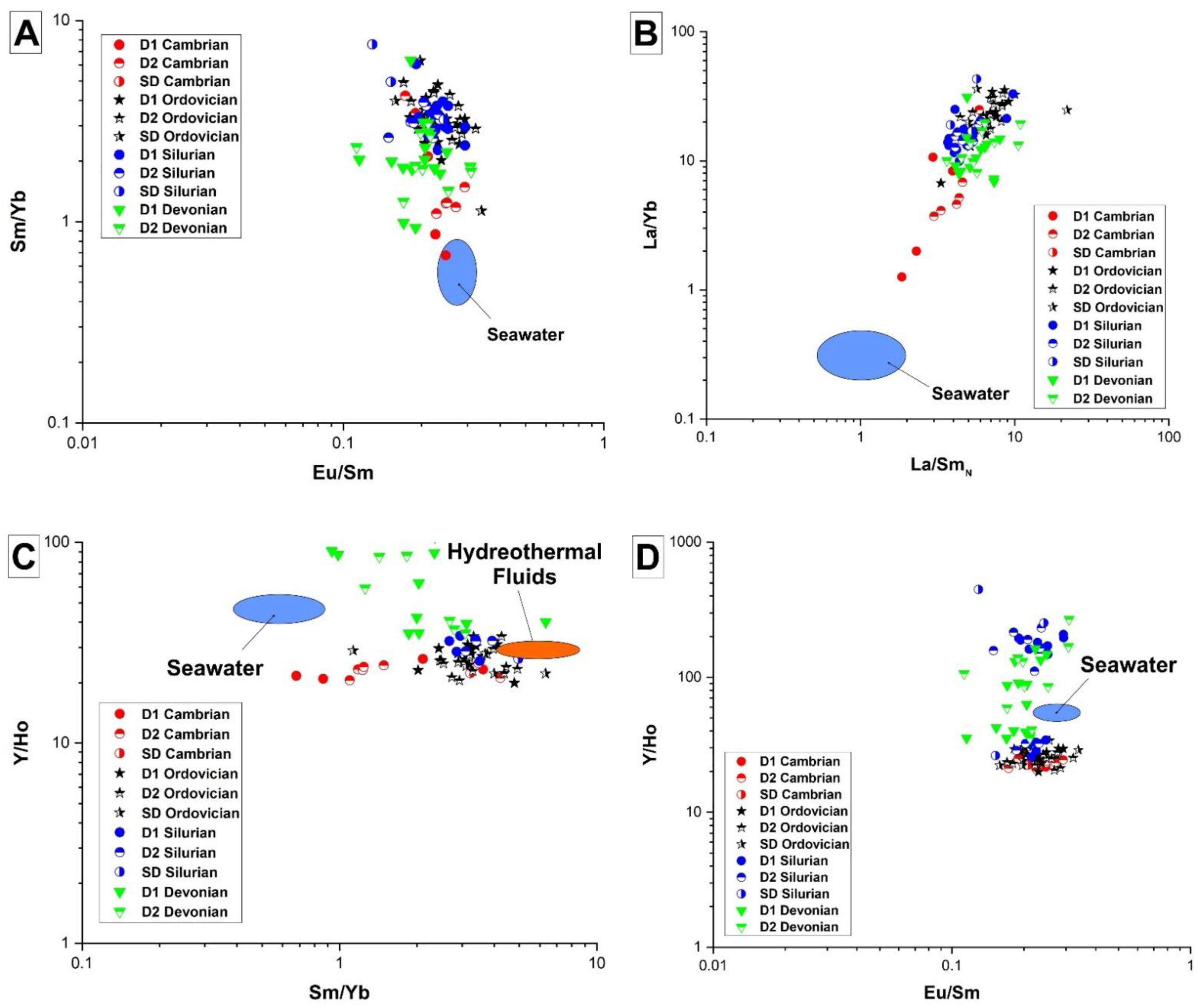
4.2.2. Major, Minor, Trace and REE Elements Content
4.2.3. Fluid Inclusions Microthermometry
5. Data Analysis and Discussion
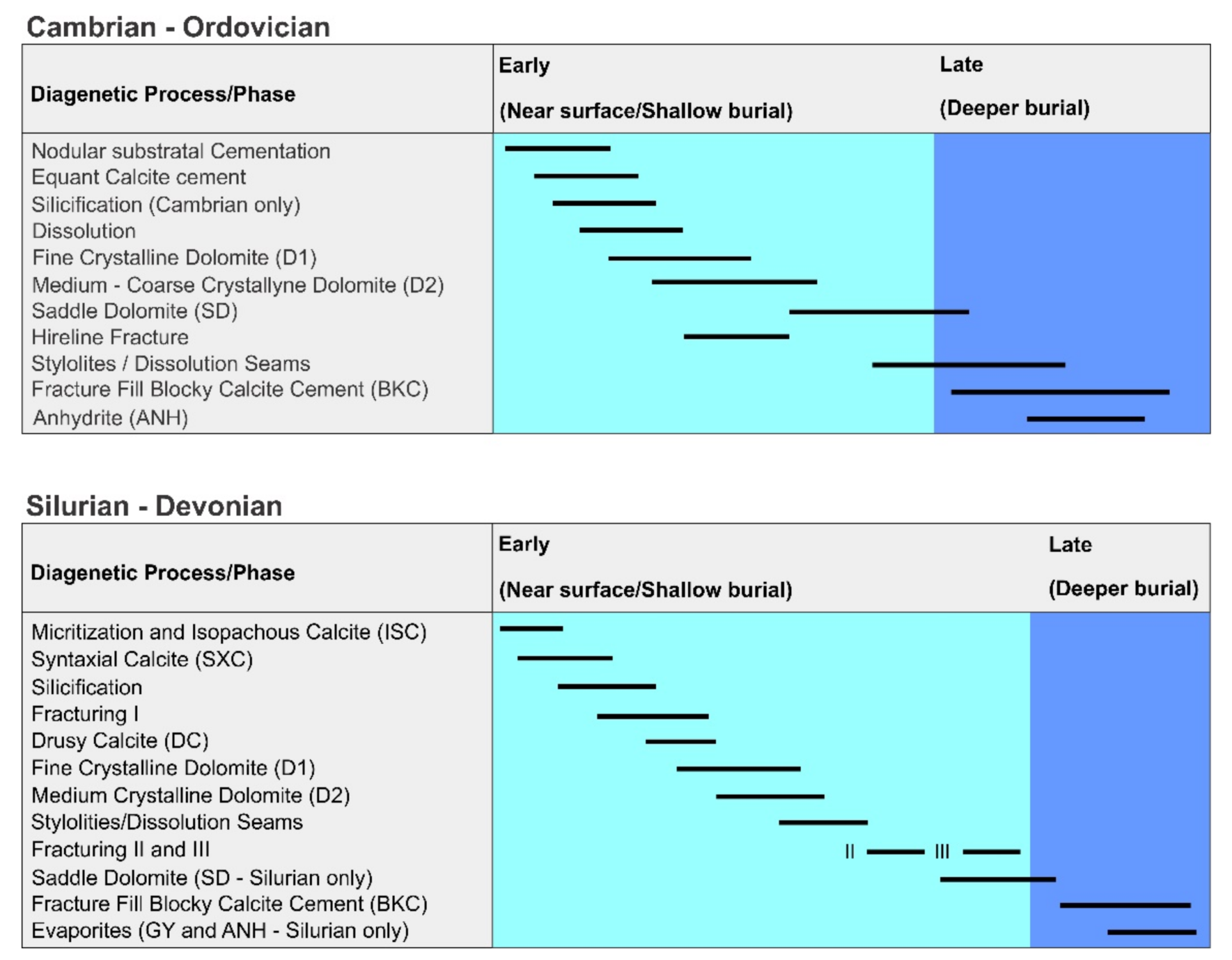
5.1. Constraints from Petrography-Paragenetic Sequence
5.2. Controls of Dolomitization-Geochemical Evidence
5.2.1. Stable and Radiogenic Isotopes
5.2.2. Major, Minor, and REE Elements
5.2.3. Fluid Inclusions
5.3. Dolomite Recrystallization
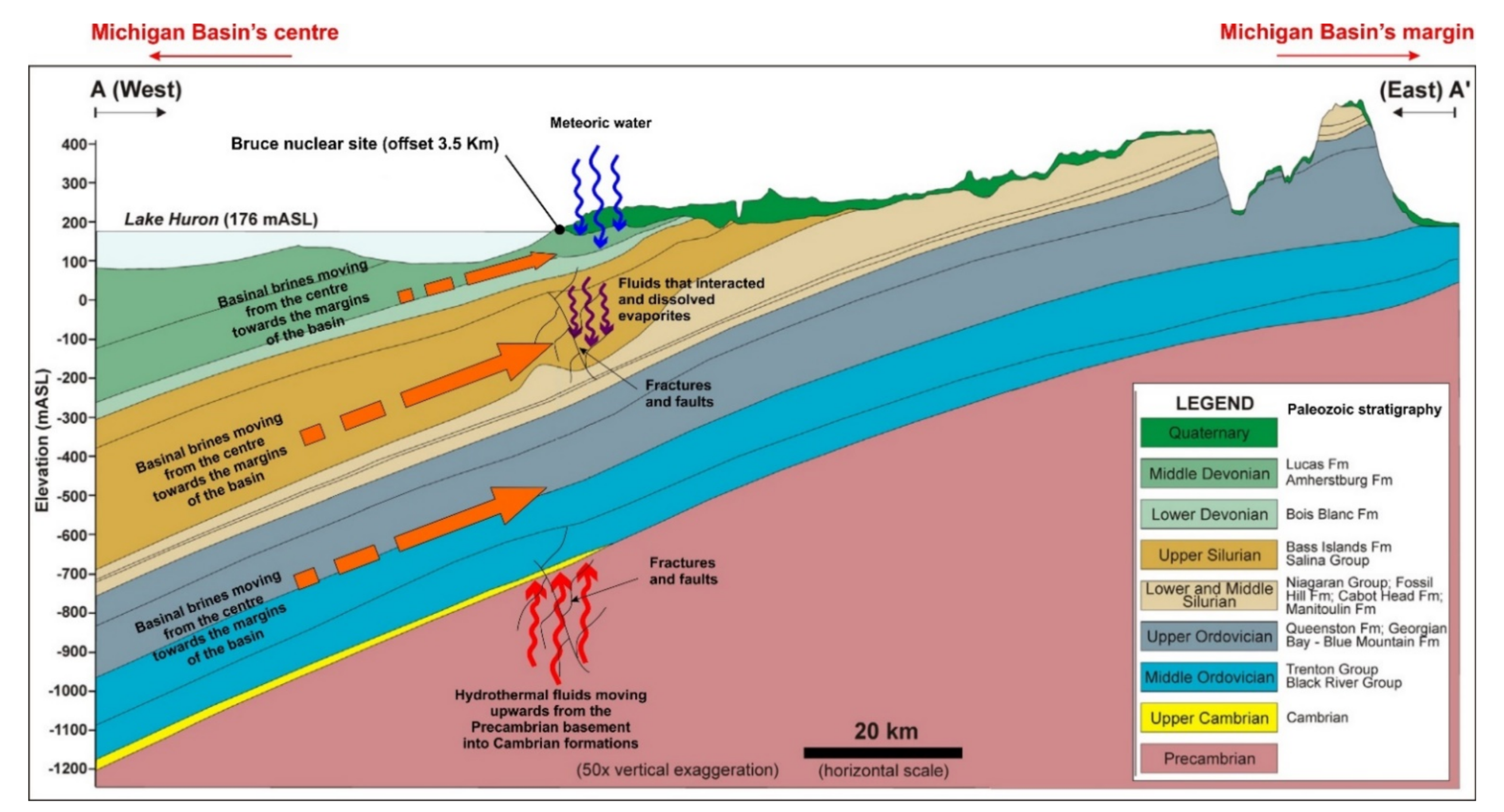
5.4. Evolution and Origin of the Diagenetic Fluids
6. Conclusions
- Dolomitization of Cambrian to Devonian carbonates occurred through multiple events, with the formation of the earliest replacive dolomite phase (D1) occurring at shallow burial depths. This was followed by the precipitation of a later replacive dolomite phase (D2) and the formation of dolomite cement (SD) occluding vugs and fractures, which were formed at an intermediate burial depth. Evaporite and late blocky calcite cement followed the formation of these dolomite phases.
- These types of dolomite types display unique geochemical and microthermometric signatures suggesting the flow of diagenetic fluids that were driven by tectonic processes over the history of the Michigan Basin. The diagenetic fluid composition was dominated initially by seawater and later modified upon burial and interaction with basinal brines. For Cambrian and Ordovician carbonates two possibly isolated diagenetic fluid systems affected them. These are: (i) an earlier fluid system that is characterized by a pronounced negative shift in oxygen and carbon isotopic composition, more radiogenic ratios, warm and saline signature with higher average ∑REE, negative La anomaly and positive Ce anomaly; and (ii) a later Ordovician system, characterized by less negative shifts in oxygen and carbon isotopes, comparable homogenization temperature, hypersaline, a less radiogenic fluid system, less negative La anomaly and positive and negative Ce anomaly, but also higher average ∑REE compared to warm water marine brachiopods.
- There is a significant overlap of δ13C and δ18O values and 87Sr/86Sr ratios in Silurian and Devonian carbonates reflecting both a modified marine source of fluids as well recrystallization of earlier formed dolomite. High homogenization temperatures and salinity in saddle dolomite (SD) in the Silurian sequences and its REE shale-normalized patterns suggest that in both age groups the diagenetic fluids were originally of coeval seawater composition and were subsequently modified via water-rock interaction. The different evolution of the diagenetic fluids is more prominent in REESN patterns from Silurian samples and it is possibly related to brines, which were modified by the dissolution of Silurian evaporites from the Salina series.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Aasm, I.S. Dolomitization: Cambrian and Ordovician Formations in the Huron Domain; Report NWMO-TR-2016-05; Nuclear Waste Management Organization: Toronto, ON, Canada, 2016. [Google Scholar]
- Al-Aasm, I.S.; Crowe, R. Fluid compartmentalization and dolomitization in the Cambrian and Ordovician successions of the Huron Domain, Michigan Basin. Mar. Pet. Geol. 2018, 92, 160–178. [Google Scholar] [CrossRef]
- Tortola, M.; Al-Aasm, I.S.; Crowe, R. Diagenetic Pore Fluid Evolution and Dolomitization of the Silurian and Devonian Carbonates, Huron Domain of Southwestern Ontario: Petrographic, Geochemical and Fluid Inclusion Evidence. Minerals 2020, 10, 140. [Google Scholar] [CrossRef] [Green Version]
- Muchez, P.; Slobodnik, M.; Sintubin, M.; Viaene, W.; Keppens, E. Origin and migration of palaeofluids in the Lower Carboniferous of southern and eastern Belgum. Zent. Geol. Palaeontol. 1997, 11 Pt 1, 1107–1112. [Google Scholar]
- Bethke, C.M.; Marshak, S. Brine migrations across North America—The plate tectonics of groundwater. Annu. Rev. Earth Planet. Sci. 1990, 18, 287–315. [Google Scholar] [CrossRef]
- Kharaka, Y.K.; Hanor, J.S. Deep fluids in the continents: I. sedimentary basins. In Treatise on Geochemistry; Drever, J.I., Holland, H.D., Turekian, K.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2004; Volume 5, pp. 499–540. [Google Scholar] [CrossRef]
- Morad, S.; Al-Ramadan, K.; Ketzer, J.M.; De Ros, L.F. The impact of diagenesis on the heterogeneity of sandstone reservoirs: A review of the role of depositional facies and sequence stratigraphy. AAPG Bull. 2010, 94, 1267–1309. [Google Scholar] [CrossRef]
- Durocher, S.; Al-Aasm, I.S. Dolomitization and neomorphism of Mississippian (Visean) upper Debolt Formation, Blueberry Field, NE British Columbia, Canada: Geologic, petrologic and chemical evidence. Am. Assoc. Pet. Geol. Bull. 1997, 81, 954–977. [Google Scholar]
- Lonnee, J.; Al-Aasm, I.S. Dolomitization and fluid evolution in the Middle Devonian Sulphur. Point Formation, Rainbow South Field, Alberta: Petrographic and geochemical evidence. Bull. Can. Soc. Pet. Geol. 2000, 90, 262–283. [Google Scholar] [CrossRef]
- Machel, H.G.; Lonnee, J. Hydrothermal dolomite—A product of poor definition and imagination. Sediment. Geol. 2002, 152, 163–171. [Google Scholar] [CrossRef]
- Cioppa, M.T.; Al-Aasm, I.S.; Symons, D.T.A.; Gillen, K.P. Dating penecontemporaneous dolomitization in carbonate reservoirs: Paleomagnetic, petrographic, and geochemical constraints. AAPG Bull. 2003, 87, 71–88. [Google Scholar]
- Wendte, J.; Chi, G.; Al-Aasm, I.; Sargent, D. Fault/fracture controlled hydrothermal dolomitization and associated diagenesis of the Upper Devonian Jean Marie Member (Redknife Formation) in the July Lake area of northeastern British Columbia. Bull. Can. Pet. Geol. 2009, 57, 275–322. [Google Scholar] [CrossRef]
- Al-Aasm, I.S.; Mrad, C.; Packard, J. Fluid compartmentalization of Devonian and Mississippian dolostones, Western Canada Sedimentary Basin: Petrologic and geochemical evidence from fracture mineralization. Can. J. Earth Sci. 2019, 56, 265–305. [Google Scholar] [CrossRef]
- Dong, S.; Chen, D.; Zhou, X.; Qian, Y.; Tian, M.; Qing, H. Tectonically driven dolomitization of Cambrian to Lower Ordovician carbonates of the Quruqtagh area, north-eastern flank of Tarim Basin, north-west China. Sedimentology 2016, 64, 1079–1106. [Google Scholar] [CrossRef]
- Land, L.S. The origin of massive dolomite. J. Geol. Educ. 1985, 33, 112–125. [Google Scholar] [CrossRef]
- Morrow, D.W. Dolomite—Part 2: Dolomitization Models and Ancient Dolostones. In Diagenesis; Mcllreath, I.A., Morrow, D.W., Eds.; Geoscience Canada Series 4; Geological Association of Canada: St John’s, NL, Canada, 1990; pp. 125–139. [Google Scholar]
- Packard, J.J.; Pellegrin, G.J.; Al-Aasm, I.S.; Samson, I.; Gagnon, J. Diagenesis and dolomitization associated with hydrothermal karst in Famennian upper Wabamun ramp sediments, northwestern Alberta. The development of porosity in carbonate reservoirs. In Canadian Society of Petroleum Geologists Short Course; The Development of Porosity in Carbonate Reservoirs: Calgary, AB, Canada, 1990; Volume 9-1. [Google Scholar]
- Sperber, C.M.; Wilkinson, B.H.; Peacor, D.R. Rock composition, dolomite stoichiometry, and rock/water reactions in dolomitic carbonate rocks. J. Geol. 1984, 92, 609–622. [Google Scholar] [CrossRef]
- Gregg, J.M.; Shelton, K.L. Dolomitization and dolomite Neomorphism in the back-reef facies of the Bonneterre and Davis formations (Cambrian), southwestern Missouri. J. Sediment. Res. 1990, 60, 549–562. [Google Scholar]
- Gao, G.; Land, L.S. Early Ordovician cool creel dolomite, middle Arbuckle group, silick Hills, SW Oklahoma, USA: Origin and modification. J. Sediment. Res. 1991, 61, 161–173. [Google Scholar]
- Mazzullo, S.J. Geochemical and neomorphic alteration of dolomite: A review. Carbonate Evaporites 1992, 7, 21–37. [Google Scholar] [CrossRef]
- Kupecz, J.A.; Land, L.S. Progressive recrystallization and stabilization of early-stage dolomite: Lower Ordovician Ellenburger Group, west Texas. In Dolomites. A Volume in Honor of Dolomieu; Purser, B., Tucker, M., Zenger, D., Eds.; International Association of Sedimentologists: Gent, Belgium, 1994; pp. 155–166. [Google Scholar]
- Al-Aasm, I.S. Chemical and isotopic constraints for recrystallization of sedimentary dolomites from the Western Canada Sedimentary basin. Aquat. Geochem. 2000, 6, 227–248. [Google Scholar] [CrossRef]
- Oliver, J. Fluids expelled tectonically from orogenic belts: Their role in hydrocarbon migration and other geologic phenomena. Geology 1986, 14, 99–102. [Google Scholar] [CrossRef]
- Nesbitt, B.E.; Muehlenbachs, K. Paleohydrogeology of the Canadian Rockies and origins of brines, Pb-Zn deposits and dolomitization in the Western Canada Sedimentary Basin. Geology 1994, 22, 243–246. [Google Scholar] [CrossRef]
- Al-Aasm, I.S. Origin and characterization of hydrothermal dolomite in the Western Canada Sedimentary Basin. J. Geochem. Explor. 2003, 78, 9–15. [Google Scholar] [CrossRef]
- Kareem, H.K.; Al-Aasm, I.S.; Mansurbeg, H. Stucturally-controlled hydrothermal fluid flow in an extensional tectonic regime: A case study of Cretaceous Qamchuqa Formation, Zagros Basin, Kurdistan Iraq. Sediment. Geol. 2019, 386, 52–78. [Google Scholar] [CrossRef]
- Tucker, M.E.; Wright, V.P. Carbonate Sedimentology; Blackwell Scientific Publications: Oxford, UK, 1990; 482p. [Google Scholar]
- Warren, J. Dolomite: Occurrence, evolution, and economically important associations. Earth Sci. Rev. 2000, 52, 1–81. [Google Scholar] [CrossRef]
- Machel, H.G. Concepts and models of dolomitization: A critical reappraisal. In The Geometry and Petrogenesis of Dolomite Hydrocarbon Reservoirs; Braithwaite, C.J.R., Rizzi, G., Drake, G., Eds.; Geological Society Special Publication: London, UK, 2004; Volume 235, pp. 7–63. [Google Scholar]
- Davies, G.R.; Smith, L.B., Jr. Structurally controlled hydrothermal dolomite reservoir facies: An overview. Am. Assoc. Pet. Geol. 2006, 90, 1641–1690. [Google Scholar] [CrossRef]
- Haeri-Ardakani, O.; Al-Aasm, I.S.; Coniglio, M. Fracture mineralization and fluid flow evolution: An example from Ordovician-Devonian carbonates, southwestern Ontario, Canada. Geofluids 2013, 13, 1–20. [Google Scholar] [CrossRef]
- Haeri-Ardakani, O.; Al-Aasm, I.S.; Coniglio, M. Petrologic and geochemical attributes of fracture-related dolomitization in Ordovician carbonates and their spatial distribution in southwestern Ontario, Canada. Mar. Pet. Geol. 2013, 43, 409–422. [Google Scholar] [CrossRef]
- Carter, T.R.; Fortner, L.D.; Russell, H.J.A.; Skuce, M.E.; Longstaffe, F.J.; Sun, S. A Hydrostratigrapic Framework for the Paleozoic Bedrock of Southern Ontario. Geosci. Can. 2021, 48, 23–58. [Google Scholar] [CrossRef]
- Ontario Geological Survey. Bedrock Geology of Ontario, Southern Sheet, Map 2544, Scale 1:1,000,000; Ontario Geological Survey: Sudbury, ON, Canada, 1991.
- Ontario Power Generation’s (OPG). Deep Geologic Repository for Low and Intermediate Level Waste: Preliminary Safety Report; Program Document n 00216-SR-01320-00001; OPG: Thunder Bay, ON, Canada, 2011. [Google Scholar]
- Sanford, B.V.; Thompson, F.J.; McFall, G.H. Plate tectonics—A possible controlling mechanism in the development of hydrocarbon traps in southwestern Ontario. Bull. Can. Pet. Geol. 1985, 33, 52–71. [Google Scholar]
- Cruden, A. Outcrop Fracture Mapping; NMWO DGR-TR-2011-43; Nuclear Waste Management Organization: Toronto, ON, Canada, 2011. [Google Scholar]
- Hamilton, D.G.; Coniglio, M. Reservoir Development in the Middle Devonian of southwestern Ontario. In Proceedings of the Ontario Petroleum Institute Thirty-First Annual Conference, Niagara Falls, ON, Canada, 28–30 October 1992; Technical Paper No. 9. Volume 32. 20p. [Google Scholar]
- Armstrong, D.K.; Carter, T.R. The Subsurface Paleozoic Stratigraphy of Southern Ontario; Ontario Geological Survey: Sudbury, ON, Canada, 2010; Special Volume 7, 301p.
- Klein, G.V.; Hsui, A.T. Origin of Cratonic Basins. Geology 1987, 15, 1094–1098. [Google Scholar] [CrossRef]
- Sleep, N.H. Thermal effects of the formation of Atlantic continental margins by continental break up. Geophys. J. R. Astron. Soc. 1971, 24, 325–350. [Google Scholar] [CrossRef]
- Sleep, N.H.; Snell, N.S. Thermal contraction and flexure of mid-continent and Atlantic marginal basins. Geophys. J. R. Astron. Soc. 1976, 45, 125–154. [Google Scholar] [CrossRef] [Green Version]
- Nunn, J.A.; Sleep, N.H.; Moore, W.E. Thermal subsidence and generation of hydrocarbon in Michigan Basin. AAPG Bull. 1984, 68, 296–315. [Google Scholar]
- Howell, P.D.; van der Pluijm, B.A. Early history of the Michigan basin: Subsidence and Appalachian tectonics. Geology 1990, 18, 1195–1198. [Google Scholar] [CrossRef]
- Howell, P.D.; van der Pluijm, B.A. Structural sequences and styles of subsidence in the Michigan basin. Geol. Soc. Am. Bull. 1999, 111, 974–991. [Google Scholar] [CrossRef]
- AECOM; Itasca Consulting Canada. Regional Geology—Southern Ontario; Report NWMO DGR-TR-2011-15 R000; Nuclear Waste Management Organization: Toronto, ON, Canada, 2011. [Google Scholar]
- Coakley, B.J.; Nadon, C.; Wang, H.F. Spatial variations in tectonic subsidence during Tippecanoe I in the Michigan Basin. Basin Res. 1994, 6, 131–140. [Google Scholar] [CrossRef]
- Coakley, B.; Gurnis, M. Far-field tilting of Laurentia during the Ordovician and constraints on the evolution of a slab under an ancient continent. J. Geophys. Res. Solid Earth 1995, 100, 6313–6327. [Google Scholar] [CrossRef] [Green Version]
- Johnson, M.D.; Armstrong, D.K.; Sanford, V.V.; Telford, P.G.; Rutka, M.A. Paleozoic and Mesozoic Geology of Ontario. In Geology of Ontario; Ontario Geologic Survey: Peterborough, ON, Canada, 1992; Special Volume 4, Part 2, pp. 907–1008. [Google Scholar]
- Armstrong, D.K.; Carter, T.R. An updated guide to the subsurface Paleozoic stratigraphy of Southern Ontario. Ont. Geol. Surv. 2006, 6191, 214. [Google Scholar]
- Beaumont, C.; Quinlan, G.; Hamilton, J. Orogeny and stratigraphy: Numerical models of the Paleozoic in the eastern interior of North America. Tectonics 1988, 7, 389–416. [Google Scholar] [CrossRef]
- Nuclear Waste Management Organization. Deep Geologic Repository, Geoscientific Site Characterization. 2011. Available online: http//www.nmwo.ca/dggeoscientifcsitecharaterization (accessed on 29 August 2021).
- Nuclear Waste Management Organization. Geosynthesis: Nuclear Waste Management Organization. Report NWMO DGR-TR-2011-11. Toronto, Canada. 2011. Available online: http//www.nmwo.ca/uploads.DGR%20PDF/Geo/Geosynthesis/pdf (accessed on 29 August 2021).
- Middleton, K.; Coniglio, M.; Sherlock, R.; Frape, S.K. Dolomitization of Middle Ordovician carbonate reservoirs, southwestern Ontario. Bull. Can. Pet. Geol. 1993, 41, 150–163. [Google Scholar]
- Intera Engineering. Descriptive Geosphere Site Model; Report NWMO DGR-TR-2011-24 R000; Nuclear Waste Management Organization: Toronto, ON, Canada, 2011. [Google Scholar]
- Van der Voo, R. Paleozoic paleogeography of North America, Gondwana, and intervening displaced terranes: Comparison of paleomagnetism with paleoclimatology and biogeographical patterns. Geol. Soc. Am. Bull. 1988, 100, 311–324. [Google Scholar] [CrossRef]
- Sonnenfeld, P.; Al-Aasm, I.S. The Salina evaporites in the Michigan Basin. In Early Sedimentary Evolution of the Michigan Basin; Special Paper, 256; Catacosinos, P.A., Daniels, P.A., Jr., Eds.; Geological Society of America: Boulder, CO, USA, 1991; pp. 139–153. [Google Scholar]
- Birchard, M.C.; Rutka, M.A.; Brunton, F.R. Lithofacies and Geochemistry of the Lucas Formation in the Subsurface of Southwestern Ontario: A High Purity Limestone and Potential High Purity Dolostone Resource; Open File Report 6137; Ontario Geological Survey: Sudbury, ON, Canada, 2004; 57p.
- Al-Aasm, I.S.; Taylor, B.; South, B. Stable isotope analysis of multiple carbonate samples using selective acid extraction. Chem. Geol. 1990, 80, 119–125. [Google Scholar] [CrossRef]
- Chi, G.; Ni, P. Equations for calculation of NaCl/(NaCl+CaCl2) ratios and salinities from hydrohalite-melting and ice-melting temperatures in the H2O-NaCl-CaCl2 system. Acta Petrol. Sin. 2007, 23, 33–37. [Google Scholar]
- Steele-MacInnis, M.; Bondnar, R.J.; Naden, J. Numerical model to determine the composition of H2O-NaCl-CaCl2 fluid inclusions based on microthermometirc and microanalytical data. Geochem. Cosmochim. Acta 2011, 75, 21–41. [Google Scholar] [CrossRef] [Green Version]
- Lawler, J.P.; Crawford, M.L. Stretching of fluid inclusions resulting from a low temperature microthermometric technique. Econ. Geol. 1983, 78, 527–529. [Google Scholar] [CrossRef]
- Jenner, G.A.; Longerich, H.P.; Jackson, S.E.; Fryer, B.J. ICP-MS—A powerful tool for high-precision trace-element analysis in Earth sciences: Evidence from analysis of selected USGS refence samples. Chem. Geol. 1990, 83, 133–148. [Google Scholar] [CrossRef]
- Bau, M.; Dulski, P. Distribution of yttrium and rare-earth elements in the Penge and Kuruman iron-formations, Transvaal Supergroup, South Africa. Precambrian Res. 1996, 79, 37–55. [Google Scholar] [CrossRef]
- Kucera, J.; Cempirek, J.; Dolnicek, Z.; Muchez, P.; Prochaska, W. Rare earth elements and yttrium geochemistry of dolomite from post-Variscan vein-type mineralization of the Nizky Jesenik and Upper Silesian Basins, Czech Republic. J. Geochem. Explor. 2009, 103, 69–79. [Google Scholar] [CrossRef]
- Felitsyn, S.; Morad, S. REE patterns in latest Neoproterozoic–early Cambrian phosphate concretions and associated organic matter. Geochem. Geol. 2002, 187, 257–265. [Google Scholar] [CrossRef]
- Sibley, D.F.; Gregg, J.M. Classification of dolomite rock textures. J. Sediment. Petrol. 1987, 57, 967–975. [Google Scholar]
- Veizer, J.; Ala, D.; Azmy, K.; Bruckschen, P.; Buhl, D.; Bruhn, F.; Carden, G.A.F.; Diener, A.; Ebneth, S.; Godderis, Y.; et al. 87Sr ⁄ 86Sr and δ13C and δ18O evolution of Phanerozoic seawater. Chem. Geol. 1999, 161, 59–88. [Google Scholar] [CrossRef] [Green Version]
- Azmy, K.; Brand, U.; Sylvester, P.; Gleeson, S.A.; Logan, A.; Bitner, M.A. Biogenic and abiogenic low-Mg calcite (bLMC and aLMC): Evaluation of seawater-REE composition, water masses and carbonate diagenesis. Chem. Geol. 2011, 280, 180–190. [Google Scholar] [CrossRef] [Green Version]
- Goldstein, R.H.; Reynolds, T.J. Systematics of Fluid Inclusions in Diagenetic Minerals. Society of Economic Paleontologists and Mineralogists; Society for Sedimentary Geology: Tulsa, OK, USA, 1994; 199p. [Google Scholar]
- Morad, S.; Al-Aasm, I.S.; Nader, F.H.; Ceriani, A.; Gasparrani, M. Impact of diagenesis on the spatial and temporal of reservoir quality in C and D Members of the Arab Formation (Jurassic), offshore Abu Dhabi oilfield. GeoArabia 2012, 17, 17–56. [Google Scholar]
- Harper, D.A.; Longstaffe, F.J.; Wadleigh, M.A.; McNutt, R.H. Secondary K-feldspar at the Precambrian-Paleozoic unconformity, southwestern Ontario. Can. J. Earth Sci. 1995, 32, 1432–1450. [Google Scholar] [CrossRef]
- Ziegler, K.; Longstaffe, F.J. Clay mineral anthogenesis along a mid-continental scale fluid conduit in Palaeozoic sedimentary rocks from southern Ontario, Canada. Clay Miner. 2001, 35, 239–260. [Google Scholar] [CrossRef]
- Paganoni, M.; Al Harthi, A.; Morad, D.; Morad, S.; Ceriani, A.; Mansurbeg, H. Impact of stylolitization on diagenesis of a Lower Cretaceous carbonate reservoir from a giant oilfield, Abu Dhabi, United Arab Emirates. Sediment. Geol. 2016, 335, 70–92. [Google Scholar] [CrossRef]
- Sanford, B.V. Devonian of Ontario and Michigan. Int. Symp. Devonian Syst. 1968, 1, 973–999. [Google Scholar]
- Armstrong, D.K.; Goodman, W.R. Stratigraphy and Depositional Environments of Niagaran Carbonates, Bruce Peninsula, Ontario. In Proceedings of the Spring Eastern Section Meeting, University Park, PA, USA, 7–8 April 1990; Field Trip No. 4 Guidebook; American Association of Petroleum Geologists: London, ON, Canada; Ontario Petroleum Institute: Tulsa, OK, USA, 1990. [Google Scholar]
- James, N.P.; Jones, B. Origin of Carbonate Sedimentary Rocks; John Wiley & Sons: West Sussex, UK, 2016; p. 446. [Google Scholar]
- Cercone, K.R. Evaporative sea-level drawdown in the Silurian Michigan Basin. Geology 1988, 16, 109–130. [Google Scholar] [CrossRef]
- Zheng, Q. Carbonate Diagenesis and Porosity Evolution in the Guelph Formation, Southwestern Ontario. Ph.D. Thesis, University of Waterloo, Waterloo, ON, Canada, 1999. [Google Scholar]
- Swart, P.K. The geochemistry of carbonate diagenesis: The past, present and future. Sedimentology 2015, 62, 1233–1304. [Google Scholar] [CrossRef]
- Frank, T.D.; Lohmann, K.C. Chronostratigraphic significance of cathodoluminescence zoning in syntaxial cement: Mississippian Lake Valley Formation, New Mexico. Sediment. Geol. 1996, 105, 29–50. [Google Scholar] [CrossRef] [Green Version]
- McNutt, R.H.; Frape, S.K.; Dollar, P. A strontium, oxygen and hydrogen isotopic composition of brines, Michigan and Appalachian Basins, Ontario and Michigan. Appl. Geochem. 1987, 2, 495–505. [Google Scholar] [CrossRef]
- Baldermann, A.; Deditius, A.; Dietzel, M.; Fichtner, V.; Fischer, C.; Hippler, D.; Leis, A.; Baldermann, C.; Mavromatis, V.; Stickler, C.P.; et al. The role of bacterial sulfate reduction during dolomite precipitation: Implications from Upper Jurassic platform carbonates. Chem. Geol. 2015, 412, 1–14. [Google Scholar] [CrossRef]
- Azomani, E.; Azmy, K.; Blemey, N.; Brand, U.; Al-Aasm, I.S. Dolomitization of the Catoche Formation in Daniel’s Harbour, Northern Peninsula, western Newfoundland. Mar. Pet. Geol. 2013, 40, 99–114. [Google Scholar] [CrossRef] [Green Version]
- Al-Aasm, I.; Veizer, J. Diagenetic stabilization of aragonite and low-Mg calcite-1: Trace elements in rudists. J. Sed. Petrol. 1986, 56, 138–152. [Google Scholar] [CrossRef]
- Land, L.S. The isotopic and trace element geochemistry of dolomite: The state of the art. In Concepts and Models of Dolomitization; Zenger, D.H., Dunham, J.B., Ethington, R.L., Eds.; SEPM Special Publication: Tulsa, OK, USA, 1980; Volume 28, pp. 87–110. [Google Scholar]
- Nothdurft, L.; Webb, G.; Kamber, B. Rare earth element geochemistry of Late Devonian reefal carbonates, Canning Basin, Western Australia: Confirmation of a seawater REE proxy in ancient limestones. Geochim. Cosmochim. Acta 2004, 68, 263–283. [Google Scholar] [CrossRef]
- Webb, G.E.; Nothdurft, L.; Kamber, B.; Kloprogge, T.; Zhao, J.-X. Rare earth element geochemistry of scleractinian coral skeleton during meteoric diagenesis: A before and-after sequence through neomorphism of aragonite to calcite. Sedimentology 2009, 56, 1433–1463. [Google Scholar] [CrossRef]
- Özyurt, M.; Kirmachi, M.Z.; Al-Aasm, I.; Hollis, C.; Tasli, K.; Kandemir, R. REE Characteristics of Lower Cretaceous Limestone Succession in Gümüshane, NE Turkey: Implications for Ocean Paleoredox Conditions and Diagenetic Alteration. Minerals 2020, 10, 683. [Google Scholar] [CrossRef]
- Sholkovitz, E.; Shen, G.T. The incorporation of rare earth elements in modern coral. Geochim. Cosmochim. Acta 1995, 59, 2749–2756. [Google Scholar] [CrossRef]
- Allwood, A.C.; Kamber, B.S.; Walter, M.R.; Burch, I.W.; Kanik, I. Trace elements record depositional history of an Early Archean stromatolitic carbonate platform. Chem. Geol. 2010, 270, 148–163. [Google Scholar] [CrossRef]
- Shields, G.; Stille, P. Diagenetic constraints on the use of cerium anomalies as paleoseawater redox proxies: An isotopic and REE study of Cambrian phosphorites. Chem. Geol. 2001, 175, 29–48. [Google Scholar] [CrossRef]
- Sverjensky, D.A. Europium equilibria in aqueous solution. Earth Planet. Sci. Lett. 1984, 67, 70–78. [Google Scholar] [CrossRef]
- Zaky, A.H.; Brand, U.; Azmy, K. A new sample processing protocol for procuring seawater REE signatures in biogenic and abiogenic carbonates. Chem. Geol. 2015, 416, 36–50. [Google Scholar] [CrossRef]
- Caetano-Filho, S.; Paula-Santos, G.M.; Dias-Brito, D. Carbonate REE + Y signatures from the restricted early marine phase of South Atlantic Ocean (late Aptian—Albian): The influence of early anoxic diagenesis on shale-normalized REE + Y patterns of ancient carbonate rocks. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2008, 500, 69–83. [Google Scholar] [CrossRef] [Green Version]
- Hood, A.S.; Planavsky, N.J.; Wallace, M.W.; Wang, X. The effects of diagenesis on geochemical paleoredox proxies in sedimentary carbonates. Geochim. Cosmochim. Acta 2018, 232, 265. [Google Scholar] [CrossRef]
- Worthington, S.R.H. Karst Assessment; Report NWMO DGR-TR-2011-22; Nuclear Waste Management Organization: Toronto, ON, Canada, 2011. [Google Scholar]
- Banner, J.L.; Hanson, G.N. Calculation of simultaneous isotopic and trace element variations during water-rock interaction with applications to carbonate diagenesis. Geochim. Cosmochim. Acta 1990, 54, 3123–3137. [Google Scholar] [CrossRef]
- Özyurt, M.; Kirmachi, M.Z.; Al-Aasm, I.S. Geochemical characteristics of Late Jurassic-Early Cretaceous Berdiga carbonates in Hazine Mağara, Gümüşhane (NE Turkey): Implications for dolomitization and recrystallization. Can. J. Earth Sci. 2019, 56, 306–320. [Google Scholar]
- Luczaj, J.A. Evidence against the Dorag (mixing-zone) model for dolomitization along the Wisconsin arch-A case for hydrothermal diagenesis. Am. Assoc. Pet. Geol. Bull. 2006, 90, 1719–1738. [Google Scholar] [CrossRef] [Green Version]
- Barnes, D.A.; Parris, T.M.; Grammer, G.M. Hydrothermal Dolomitization of Fluid Reservoirs in the Michigan Basin, USA. Search and Discovery Article #50087. In Proceedings of the AAPG Annual Convention, San Antonio, TX, USA, 20–23 April 2008. [Google Scholar]
- Simo, J.A.; Johnson, C.M.; Vandrey, M.R.; Brown, P.E.; Castro Giovanni, E.; Drzewiecki, P.E.; Valley, J.W.; Boyer, J. Burial dolomitization of the Middle Ordovician Glenwood Formation by evaporitic brines, Michigan Basin. In Dolomites—A Volume in Honour of Dolomieu; Purser, B., Tucker, M., Zenger, D., Eds.; The International Association of Sedimentologists, Blacwell Scientific Publications: Hoboken, NJ, USA, 1994; pp. 169–186. [Google Scholar]
- Braithwaite, C.J.R.; Rizzi, G.; Darke, G. The geometry and petrogenesis of dolomite hydrocarbon reservoirs: Introduction. Geol. Soc. Lond. Spec. Publ. 2004, 235, 1–6. [Google Scholar] [CrossRef]
- Machel, H.G. Recrystallization versus neomorphism, and the concept of “significant recrystallization” in dolomite research. Sediment. Geol. 1997, 113, 161–168. [Google Scholar] [CrossRef]
- Kaczmarek, S.E.; Sibley, D.F. Direct physical evidence of dolomite recrystallization. Sedimentology 2014, 61, 1862–1882. [Google Scholar] [CrossRef]
- Adam, J.; Al-Aasm, I.S. Petrologic and geochemical attributes of calcite cementation, dolomitization and dolomite recrystallization: An example from the Mississippian Pekisko Formation, west-central Alberta. Bull. Can. Pet. Geol. 2017, 65, 235–261. [Google Scholar] [CrossRef]
- Hardie, L.A. Perspectives—dolomitization: A critical view of some current views. J. Sediment. Petrol. 1987, 57, 166–183. [Google Scholar] [CrossRef]
- Al-Aasm, I.S.; Clarke, J.D. The effect of hydrothermal fluid flow on early diagenetic dolomitization: An example from the Devonian Slave Point Formation, northwest Alberta, Canada. In Deformation, Fluid Flow, and Reservoir Appraisal in Foreland Fold and Thrust Belts: AAPG Hedberg Series; Swennen, R., Roure, F., Granath, J.W., Eds.; American Association of Petroleum Geologists: Tulsa, OK, USA, 2004; pp. 297–316. [Google Scholar]
- Malone, M.J.; Baker, P.A.; Burns, S.J. Recrystallization of dolomite: An experimental study from 50–200 °C. Geochim. Cosmochim. Acta 1996, 60, 2189–2207. [Google Scholar] [CrossRef]
- Coniglio, M.; William-Jones, A.E. Diagenesis of Ordovician carbonates from the northeast Michigan Basin, Manitoulin Island area, Ontario: Evidence from petrography, stable isotopes and fluid inclusions. Sedimentology 1992, 39, 813–836. [Google Scholar] [CrossRef]
- Coniglio, M.; Sherlock, R.; Williams-Jones, A.E.; Middleton, K.; Frape, S.K. Burial and hydrothermal diagenesis of Ordovician carbonates from the Michigan Basin, Ontario, Canada. Int. Assoc. Sedimentol. 1994, 21, 231–254. [Google Scholar]
- Yoo, C.M.; Gregg, J.M.; Shelton, K.L. Dolomitization and dolomite neomorphism: Trenton and Black River limestone (Middle Ordovician) Northern Indiana, U.S.A. J. Sediment. Res. 2000, 70, 265–274. [Google Scholar] [CrossRef]
- Hobbs, M.Y.; Frape, S.K.; Shouakar-Stash, O.; Kennell, L.R. Regional Hydrochemistry-Southern Ontario; Report NMWO-DGR-TR-2011-12; Nuclear Waste Management Organization: Toronto, ON, Canada, 2011. [Google Scholar]
- Petts, D.; Saso, J.; Diamond, L.; Aschwanden, L.; Al, T.; Jensen, M. The source and evolution of paleofluids responsible for secondary minerals in low-permeability Ordovician limestones of the Michigan Basin. Appl. Geochem. 2017, 86, 121–137. [Google Scholar] [CrossRef]
- Bouchard, L.; Veizer, J.; Kennell-Morrison, L.; Jensen, M.; Raven, K.G.; Clark, I.D. Origin and 87Rb–87Sr age of porewaters in low permeability Ordovician sediments on the eastern flank of the Michigan Basin, Tiverton, Ontario, Canada. Can. J. Earth Sci. 2019, 56, 201–208. [Google Scholar] [CrossRef]
- Nagia, N.G.; Hu, M.; Gao, D.; Hu, Z.; Sun, C.-Y. Application of stable strontium isotope geochemistry and fluid inclusion microthermometry to studies of dolomitization of the deeply buried Cambrian carbonate successions in west-central Tarim Basin, NW China. J. Earth Sci. 2019, 30, 176–193. [Google Scholar] [CrossRef]
- Land, L.S. The application of stable isotopes to studies of the origin of dolomite and to problems of diagenesis of clastic sediments. In Stable Isotopes in Sedimentary Geology; Arthur, M.A., Anderson, T.F., Kaplan, I.R., Veizer, J., Land, L.S., Eds.; SEPM Society for Sedimentary Geology: Tulsa, OK, USA, 1983. [Google Scholar]
- Friedman, I.; O’Neil, J.R. Compilation of stable isotope fractionation factors of geochemical interest. In Data of Geochemistry, 6th ed.; Chapter, K.K., Ed.; U.S. Government Printing Office: Washington, DC, USA, 1977. [Google Scholar]
- Wang, H.F.; Crowley, K.D.; Nadon, G.C. Thermal history of the Michigan Basin from Apatite fission-track analysis and vitrinite reflectance. Am. Assoc. Petrol. Geol. Mem. 1994, 61, 167–177. [Google Scholar]
- Ma, L.; Castro, M.C.; Hall, C.M. Atmospheric noble gas signatures in deep Michigan Basin brines as indicators of a past thermal event. Earth Planet Sci. Lett. 2009, 277, 137–147. [Google Scholar] [CrossRef]
- Slater, B.; Smith, L. Outcrop Analogue for Trenton-Black River Hydrothermal Reservoirs, Mohawk Valley, New York. AAPG Bull. 2012, 96, 1369. [Google Scholar] [CrossRef]
- Beauheim, R.L.; Roberts, R.M.; Avis, J.D. Hydraulic testing of low-permeability Silurian and Ordovician strata, Michigan Basin, southwestern Ontario. J. Hydrol. 2014, 509, 163–178. [Google Scholar] [CrossRef]
- Sutcliffe, C.N.; Thibodeau, A.M.; Davis, D.W.; Al-Aasm, I.S.; Parmenter, A.; Zajacz, Z.; Jensen, M. Hydrochronology of a proposed deep geological repository for Low and Intermediate nuclear waste in southern Ontario from U-Pb dating of secondary minerals: Response to Alleghanian events. Can. J. Earth Sci. 2019, 57, 494–505. [Google Scholar] [CrossRef]









| Age | Mineral | Description | Texture | Size | Luminescence |
|---|---|---|---|---|---|
| Cambrian | D1 | Non-ferroan replacive microcrystalline dolomite matrix | Planar-s | <50 μm | Dull red |
| Cambrian | D2 | Non-ferroan medium crystalline dolomite matrix | Planar-s | >50 μm up to 500 μm | Dull red/bright |
| Cambrian | SD | Minor coarse crystalline saddle dolomite cement filling vugs | Curved crystal faces and undulose extinction | >500 μm | Dull/non-luminescent |
| Cambrian | BKC | Non ferroan/ferroan calcite cement mainly filling fractures and voids | Blocky/equant | 200 μm to >500 μm | Red/bright orange |
| Ordovician | D1 | Non-ferroan replacive microcrystalline dolomite matrix | Non-planar to planar-s | 25–50 μm | Dull red |
| Ordovician | D2 | Non-ferroan medium crystalline, partly zoned dolomite matrix | Planar e/planar-s | >50 μm up to 500 μm | Dull red |
| Ordovician | SD | Minor coarse crystalline saddle dolomite cement filling vugs | Curved crystal faces and undulose extinction | >500 μm | Dull/non-luminescent |
| Ordovician | SXC | Ferroan syntaxial calcite overgrowth cement around echinoderm fragments | 100 μm to >500 μm | Bright red | |
| Ordovician | BKC | Non ferroan/ferroan calcite cement mainly filling fractures and voids | Blocky/equant | 200 μm to >500 μm | Red/bright Orange, zoned |
| Silurian | D1 | Non-ferroan pervasive replacive micro to fine crystalline dolomite matrix | Non-planar to planar-s | <50 μm | Dull red/bright |
| Silurian | D2 | Non-ferroan pervasive replacive medium to coarse crystalline dolomite matrix | Planar-e/planar-s | >50 μm up to 150 μm | Dull red/non-luminescent |
| Silurian | D2 (Dissolution seams) | Selective replacive medium crystalline dolomite matrix (commonly associated with dissolution seams) | Planar-e | >50 μm up to 150 μm | Dull red |
| Silurian | SD | Ferroan coarse crystalline saddle dolomite cement filling fractures and vugs | Curved crystal faces and undulose extinction | >500 μm | Dull/non-luminescent |
| Silurian | ISC | Non-ferroan isopachous calcite cement rimming coated grains | bladed | 50–100 μm | Dull red |
| Silurian | SXC | Ferroan syntaxial calcite overgrowth cement around echinoderm fragments | - | 100 μm to >500 μm | Bright red |
| Silurian | DC | Ferroan void-filling and pore-lining cement in intergranular and intraskeletal pores, molds and fractures | Equant to elongate, anhedral to subhedral | 75–250 μm Size increase towards the center | Dull red |
| Silurian | BKC | Non ferroan/ferroan calcite cement mainly filling fractures and voids | blocky | 200 μm to >500 μm | Red/bright orange zoned |
| Devonian | D1 | Non-ferroan pervasive replacive micro to fine crystalline matrix dolomite | Non-planar to planar-s | <50 μm | Dull/red |
| Devonian | D2 | Non-ferroan pervasive replacive medium crystalline matrix dolomite | Planar-e/planar-s | >50 μm up to 100 μm | Dull/non-luminescent |
| Devonian | D2 (Dissolution seams) | Non-feroan selective replacive medium crystalline dolomite matrix (commonly associated with dissolution seams) | Planar-e | >50 μm up to 100 μm | Red/bright |
| Devonian | SXC | Non-ferroan syntaxial calcite overgrowth cement | - | 100 μm to >500 μm | Non-luminescent |
| Devonian | DTC | Non-ferroan dogtooth calcite cement growing normal to the substrate (mainly skeletal grains) | Elongate scalenohedral or rhombohedral | 50 μm | Red/bright mostly zoned |
| Devonian | DC | Non-ferroan void-filling and pore-lining cement in intergranular and intraskeletal pores, molds and fractures | Equant to elongate, anhedral to subhedral | 75–250 μm Size increase towards the center | Dull red to bright, zoned in most cases |
| Devonian | BKC | Non-ferroan calcite cement mainly filling fractures and voids | blocky | 200 μm to >500 μm | Red/bright zoned |
| Age | Phase | δ13C VPDB (‰) | δ18O VPDB (‰) | 87Sr/86Sr |
|---|---|---|---|---|
| Cambrian | D1 | |||
| n. | 8 | 8 | 5 | |
| Avg. | −4.27 | −9.91 | 0.71128 | |
| Stdev. | 2.03 | 3.93 | 0.00298 | |
| Max. | −0.30 | −2.92 | 0.71716 | |
| Min. | −7.67 | −16.91 | 0.70928 | |
| Cambrian | D2 | |||
| n. | 7 | 7 | 4 | |
| Avg. | −1.98 | −7.42 | 0.70998 | |
| Stdev. | 0.45 | 0.46 | 0.00068 | |
| Max. | −1.56 | −6.95 | 0.71100 | |
| Min. | −2.96 | −8.20 | 0.70927 | |
| Cambrian | SD | |||
| n. | 3 | 3 | 0 | |
| Avg. | −3.02 | −9.98 | ||
| Stdev. | 1.72 | 0.89 | ||
| Max. | −0.59 | −8.72 | ||
| Min. | −4.26 | −10.71 | ||
| Cambrian | BKC | |||
| n. | 4 | 4 | 3 | |
| Avg. | −4.47 | −12.27 | 0.71011 | |
| Stdev. | 0.63 | 2.85 | 0.00014 | |
| Max. | −3.55 | −7.93 | 0.71031 | |
| Min. | −5.30 | −15.20 | 0.71000 | |
| Ordovician | D1 | |||
| n. | 15 | 15 | 7 | |
| Avg. | 0.11 | −8.87 | 0.71189 | |
| Stdev. | 0.70 | 1.77 | 0.00517 | |
| Max. | 1.16 | −5.30 | 0.72346 | |
| Min. | −1.25 | −12.48 | 0.70808 | |
| Ordovician | D2 | |||
| n. | 19 | 19 | 7 | |
| Avg. | 0.70 | −7.71 | 0.70896 | |
| Stdev. | 0.71 | 1.26 | 0.00148 | |
| Max. | 2.24 | −4.15 | 0.71255 | |
| Min. | −0.46 | −9.62 | 0.70814 | |
| Ordovician | SD | |||
| n. | 6 | 6 | 2 | |
| Avg. | −0.04 | −9.44 | 0.70955 | |
| Stdev. | 1.75 | 0.91 | 0.00111 | |
| Max. | 2.24 | −8.33 | 0.71066 | |
| Min. | −2.56 | −10.86 | 0.70844 | |
| Ordovician | Early Calcite | |||
| n. | 41 | 41 | 1 | |
| Avg. | 0.30 | −5.67 | ||
| Stdev. | 0.85 | 1.24 | ||
| Max. | 2.16 | −3.89 | 0.70809 | |
| Min. | −3.19 | −9.21 | 0.70809 | |
| Ordovician | BKC | |||
| n. | 30 | 30 | 4 | |
| Avg. | −0.15 | −6.78 | 0.70801 | |
| Stdev. | 0.70 | 1.20 | 0.00005 | |
| Max. | 1.46 | −3.94 | 0.70809 | |
| Min. | −1.84 | −9.88 | 0.70797 | |
| Silurian | D1 | |||
| n. | 21 | 21 | 8 | |
| Avg. | 0.21 | −6.83 | 0.70935 | |
| Stdev. | 1.99 | 1.03 | 0.00103 | |
| Max. | 3.99 | −4.46 | 0.71201 | |
| Min. | −2.80 | −8.03 | 0.70871 | |
| Silurian | D2 | |||
| n. | 14 | 14 | 7 | |
| Avg. | 2.23 | −7.06 | 0.70867 | |
| Stdev. | 1.51 | 1.18 | 0.00013 | |
| Max. | 4.02 | −3.85 | 0.70891 | |
| Min. | −0.32 | −8.31 | 0.70847 | |
| Silurian | SD | |||
| n. | 6 | 6 | 3 | |
| Avg. | 2.00 | −8.08 | 0.70863 | |
| Stdev. | 1.44 | 1.08 | 0.00008 | |
| Max. | 3.69 | −6.13 | 0.70872 | |
| Min. | 0.10 | −9.65 | 0.70852 | |
| Silurian | Early Calcite | |||
| n. | 12 | 12 | 1 | |
| Avg. | 0.96 | −6.65 | ||
| Stdev. | 1.21 | 1.38 | ||
| Max. | 3.53 | −4.73 | 0.70815 | |
| Min. | −0.88 | −8.85 | 0.70815 | |
| Silurian | BKC | |||
| n. | 10 | 10 | 3 | |
| Avg. | −0.26 | −7.92 | 0.70820 | |
| Stdev. | 1.98 | 1.66 | 0.00014 | |
| Max. | 3.17 | −4.54 | 0.70838 | |
| Min. | −3.75 | −10.19 | 0.70805 | |
| Devonian | D1 | |||
| n. | 16 | 16 | 8 | |
| Avg. | 2.65 | −6.16 | 0.70832 | |
| Stdev. | 0.90 | 0.66 | 0.00059 | |
| Max. | 4.28 | −4.46 | 0.70937 | |
| Min. | 0.55 | −7.04 | 0.70788 | |
| Devonian | D2 | |||
| n. | 22 | 22 | 8 | |
| Avg. | 2.50 | −6.22 | 0.70873 | |
| Stdev. | 1.11 | 1.35 | 0.00053 | |
| Max. | 4.63 | −3.91 | 0.70937 | |
| Min. | 0.64 | −10.39 | 0.70809 | |
| Devonian | Early Calcite | |||
| n. | 19 | 19 | 1 | |
| Avg. | 1.45 | −6.33 | ||
| Stdev. | 1.48 | 1.21 | ||
| Max. | 4.12 | −4.42 | 0.70805 | |
| Min. | −2.42 | −9.69 | 0.70805 | |
| Devonian | BKC | |||
| n. | 5 | 5 | 4 | |
| Avg. | −1.58 | −9.12 | 0.70803 | |
| Stdev. | 0.43 | 0.85 | 0.00003 | |
| Max. | −1.22 | −8.01 | 0.70806 | |
| Min. | −2.40 | −10.54 | 0.70798 |
| Age-Phase | Statistics | CaCO3 | Mn | Fe | Sr |
|---|---|---|---|---|---|
| Cambrian-D1 (n = 4) | Avg. | 59.3 | 1558.3 | 8912.8 | 83.6 |
| Stdev. | 4.2 | 400.4 | 2013.8 | 56.2 | |
| Max. | 66.5 | 1945 | 11,672.6 | 180.4 | |
| Min. | 56 | 926.9 | 6773.7 | 44.8 | |
| Cambrian-D2 (n = 6) | Avg. | 55.3 | 907.2 | 3883.6 | 97.8 |
| Stdev. | 0.3 | 163.7 | 767.4 | 6.3 | |
| Max. | 55.7 | 1227.7 | 5364.1 | 107.7 | |
| Min. | 734.3 | 2942.3 | 90.6 | 0 | |
| Cambrian-SD (n = 1) | Avg. | ||||
| Stdev. | |||||
| Max. | 57.7 | 1026.4 | 12,579.1 | 89.5 | |
| Min. | 57.7 | 1026.4 | 12,579.1 | 89.5 | |
| Ordovician-D1 (n = 5) | Avg. | 58.4 | 1097.9 | 8115.6 | 90.9 |
| Stdev. | 1.2 | 430.4 | 1787.9 | 43.2 | |
| Max. | 60.2 | 1537.4 | 10,521.4 | 172 | |
| Min. | 56.7 | 341.8 | 5462.3 | 55.5 | |
| Ordovician-D2 (n = 13) | Avg. | 61.8 | 793.3 | 10,128.9 | 120.9 |
| Stdev. | 5.7 | 347 | 8459 | 59.2 | |
| Max. | 75 | 1305.5 | 29,800 | 236.4 | |
| Min. | 56.5 | 409.5 | 2942.8 | 41.5 | |
| Ordovician-SD (n = 3) | Avg. | 63.3 | 1157.6 | 15,055.2 | 108.6 |
| Stdev. | 7.2 | 227.7 | 6420.9 | 38.1 | |
| Max. | 73.5 | 1459.1 | 23,597.3 | 154.9 | |
| Min. | 57.1 | 909 | 8116.3 | 61.4 | |
| Silurian-D1 (n = 16) | Avg. | 54.3 | 223.8 | 2428 | 94.6 |
| Stdev. | 3.4 | 217.9 | 1729.4 | 18.7 | |
| Max. | 59.4 | 857.8 | 7017.6 | 125.7 | |
| Min. | 48.3 | 34.2 | 339.3 | 70.2 | |
| Silurian-D2 (n = 7) | Avg. | 55.9 | 433.3 | 4621.3 | 253.7 |
| Stdev. | 3.1 | 313.2 | 2423.3 | 396.5 | |
| Max. | 62.1 | 860.9 | 8249.6 | 1222.1 | |
| Min. | 52.6 | 66.3 | 809.7 | 70.7 | |
| Silurian-SD (n = 3) | Avg. | 62.3 | 1098.9 | 19,606.4 | 105.9 |
| Stdev. | 4.4 | 631.1 | 1132.4 | 18 | |
| Max. | 67 | 1645.5 | 20,439.8 | 124.8 | |
| Min. | 56.3 | 214.6 | 18,005.3 | 81.8 | |
| Devonian-D1 (n = 13) | Avg. | 55.6 | 33.2 | 219.8 | 71.8 |
| Stdev. | 4.4 | 11.2 | 239.8 | 17.4 | |
| Max. | 62.2 | 54.2 | 690.5 | 123.5 | |
| Min. | 50 | 14.3 | 33.9 | 46.8 | |
| Devonian-D2 (n = 9) | Avg. | 56.8 | 48 | 419.9 | 78.2 |
| Stdev. | 4.1 | 10.7 | 296.6 | 12.2 | |
| Max. | 61.6 | 60.8 | 791.4 | 97.1 | |
| Min. | 47.9 | 32.9 | 83 | 60.3 |
| Age-Phase | Stats. | La | Ce | Pr | Nd | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | ΣREE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cambrian-D1 (n = 5) | Avg. | 10.96 | 27.92 | 3.04 | 11.58 | 3.01 | 0.67 | 3.62 | 0.65 | 4.11 | 0.90 | 2.45 | 0.35 | 2.77 | 0.40 | 72.43 |
| Stdev. | 11.60 | 27.91 | 2.69 | 9.58 | 2.13 | 0.48 | 2.60 | 0.53 | 3.68 | 0.82 | 2.26 | 0.34 | 2.80 | 0.40 | 59.82 | |
| Max. | 33.20 | 80.29 | 7.84 | 27.95 | 5.25 | 1.26 | 6.71 | 1.48 | 10.10 | 2.26 | 6.16 | 0.91 | 7.51 | 1.08 | 170.43 | |
| Min. | 0.72 | 1.86 | 0.27 | 1.08 | 0.24 | 0.05 | 0.24 | 0.03 | 0.15 | 0.03 | 0.07 | 0.01 | 0.07 | 0.01 | 4.83 | |
| Cambrian-D2 (n = 7) | Avg. | 14.68 | 38.36 | 3.95 | 14.53 | 3.16 | 0.68 | 3.67 | 0.54 | 3.19 | 0.66 | 1.73 | 0.24 | 1.74 | 0.24 | 87.38 |
| Stdev. | 15.90 | 41.71 | 4.24 | 15.06 | 2.79 | 0.52 | 2.91 | 0.43 | 2.56 | 0.53 | 1.37 | 0.18 | 1.33 | 0.19 | 84.89 | |
| Max. | 52.30 | 138.00 | 13.90 | 49.38 | 8.87 | 1.53 | 8.42 | 1.45 | 8.95 | 1.88 | 4.81 | 0.64 | 4.66 | 0.66 | 282.20 | |
| Min. | 3.48 | 8.93 | 0.90 | 3.18 | 0.76 | 0.22 | 1.11 | 0.16 | 1.01 | 0.20 | 0.54 | 0.07 | 0.44 | 0.06 | 21.20 | |
| Cambrian-SD (n = 1) | Avg. | |||||||||||||||
| Stdev. | ||||||||||||||||
| Max. | 0.932 | 2.428 | 0.306 | 1.103 | 0.180 | 0.038 | 0.235 | 0.027 | 0.127 | 0.029 | 0.058 | 0.009 | 0.056 | 0.007 | 5.533 | |
| Min. | 0.932 | 2.428 | 0.306 | 1.103 | 0.180 | 0.038 | 0.235 | 0.027 | 0.127 | 0.029 | 0.058 | 0.009 | 0.056 | 0.007 | 5.533 | |
| Ordovician-D1 (n = 13) | Avg. | 2.01 | 4.88 | 0.57 | 2.13 | 0.44 | 0.11 | 0.49 | 0.07 | 0.38 | 0.08 | 0.22 | 0.03 | 0.19 | 0.03 | 11.63 |
| Stdev. | 3.04 | 8.62 | 1.06 | 4.15 | 0.96 | 0.23 | 1.05 | 0.16 | 0.93 | 0.20 | 0.54 | 0.08 | 0.48 | 0.08 | 21.56 | |
| Max. | 12.41 | 34.49 | 4.24 | 16.42 | 3.74 | 0.89 | 4.12 | 0.64 | 3.60 | 0.78 | 2.07 | 0.28 | 1.85 | 0.28 | 85.79 | |
| Min. | 0.20 | 0.45 | 0.06 | 0.21 | 0.03 | 0.01 | 0.05 | 0.01 | 0.03 | 0.01 | 0.01 | 0.00 | 0.01 | 0.00 | 1.07 | |
| Ordovician D2 (n = 16) | Avg. | 1.68 | 3.85 | 0.43 | 1.52 | 0.27 | 0.06 | 0.30 | 0.04 | 0.18 | 0.04 | 0.09 | 0.01 | 0.07 | 0.01 | 8.54 |
| Stdev. | 1.31 | 3.37 | 0.39 | 1.36 | 0.25 | 0.05 | 0.28 | 0.03 | 0.16 | 0.03 | 0.08 | 0.01 | 0.06 | 0.01 | 7.36 | |
| Max. | 5.62 | 14.02 | 1.60 | 5.65 | 1.05 | 0.23 | 1.17 | 0.15 | 0.70 | 0.14 | 0.34 | 0.05 | 0.24 | 0.04 | 30.99 | |
| Min. | 0.44 | 0.83 | 0.09 | 0.36 | 0.06 | 0.02 | 0.08 | 0.01 | 0.03 | 0.01 | 0.02 | 0.00 | 0.02 | 0.00 | 1.98 | |
| Ordovician SD (n = 6) | Avg. | 1.97 | 4.26 | 0.49 | 1.69 | 0.29 | 0.06 | 0.32 | 0.04 | 0.19 | 0.04 | 0.10 | 0.02 | 0.09 | 0.01 | 9.55 |
| Stdev. | 0.57 | 2.05 | 0.28 | 0.97 | 0.18 | 0.02 | 0.16 | 0.02 | 0.11 | 0.02 | 0.05 | 0.01 | 0.04 | 0.01 | 4.36 | |
| Max. | 3.08 | 7.75 | 0.98 | 3.28 | 0.61 | 0.10 | 0.61 | 0.09 | 0.40 | 0.08 | 0.21 | 0.03 | 0.15 | 0.03 | 17.41 | |
| Min. | 1.17 | 2.24 | 0.19 | 0.59 | 0.08 | 0.03 | 0.15 | 0.02 | 0.09 | 0.02 | 0.06 | 0.01 | 0.05 | 0.01 | 5.43 | |
| Silurian D1 (n = 16) | Avg. | 2.30 | 5.32 | 0.64 | 2.49 | 0.52 | 0.12 | 0.53 | 0.07 | 0.40 | 0.08 | 0.21 | 0.03 | 0.16 | 0.02 | 12.89 |
| Stdev. | 1.07 | 2.84 | 0.35 | 1.38 | 0.31 | 0.07 | 0.30 | 0.04 | 0.24 | 0.04 | 0.12 | 0.01 | 0.09 | 0.01 | 6.84 | |
| Max. | 4.47 | 11.58 | 1.35 | 5.49 | 1.19 | 0.26 | 1.15 | 0.17 | 0.89 | 0.17 | 0.43 | 0.05 | 0.34 | 0.05 | 27.60 | |
| Min. | 0.78 | 1.42 | 0.15 | 0.52 | 0.10 | 0.02 | 0.10 | 0.01 | 0.06 | 0.01 | 0.04 | 0.00 | 0.03 | 0.00 | 3.25 | |
| Silurian D2 (n = 9) | Avg. | 1.89 | 4.43 | 0.55 | 2.15 | 0.41 | 0.08 | 0.41 | 0.06 | 0.30 | 0.06 | 0.16 | 0.02 | 0.12 | 0.02 | 10.65 |
| Stdev. | 1.32 | 3.34 | 0.43 | 1.67 | 0.31 | 0.06 | 0.31 | 0.04 | 0.23 | 0.04 | 0.12 | 0.02 | 0.08 | 0.01 | 7.95 | |
| Max. | 3.64 | 9.45 | 1.23 | 4.86 | 0.88 | 0.18 | 0.87 | 0.12 | 0.65 | 0.12 | 0.36 | 0.05 | 0.23 | 0.03 | 22.67 | |
| Min. | 0.28 | 0.50 | 0.05 | 0.23 | 0.05 | 0.01 | 0.05 | 0.01 | 0.04 | 0.01 | 0.02 | 0.00 | 0.02 | 0.00 | 1.30 | |
| Silurian SD (n = 4) | Avg. | 8.84 | 23.37 | 2.95 | 10.78 | 1.87 | 0.27 | 1.36 | 0.16 | 0.83 | 0.14 | 0.41 | 0.05 | 0.30 | 0.04 | 51.37 |
| Stdev. | 6.96 | 17.27 | 2.13 | 7.40 | 1.26 | 0.17 | 0.89 | 0.10 | 0.52 | 0.09 | 0.26 | 0.03 | 0.20 | 0.03 | 37.09 | |
| Max. | 19.39 | 47.98 | 5.92 | 20.35 | 3.42 | 0.44 | 2.30 | 0.27 | 1.34 | 0.23 | 0.69 | 0.08 | 0.52 | 0.06 | 102.93 | |
| Min. | 0.36 | 0.77 | 0.09 | 0.39 | 0.07 | 0.02 | 0.04 | 0.01 | 0.05 | 0.01 | 0.02 | 0.00 | 0.02 | 0.00 | 1.85 | |
| Devonian D1 (n = 13) | Avg. | 0.91 | 1.34 | 0.23 | 0.95 | 0.18 | 0.03 | 0.24 | 0.03 | 0.14 | 0.03 | 0.08 | 0.01 | 0.06 | 0.01 | 4.17 |
| Stdev. | 1.42 | 2.06 | 0.39 | 1.67 | 0.29 | 0.06 | 0.33 | 0.04 | 0.20 | 0.04 | 0.10 | 0.01 | 0.06 | 0.01 | 6.60 | |
| Max. | 4.80 | 7.60 | 1.35 | 5.77 | 0.98 | 0.18 | 0.97 | 0.11 | 0.64 | 0.12 | 0.33 | 0.04 | 0.23 | 0.03 | 22.88 | |
| Min. | 0.11 | 0.17 | 0.02 | 0.07 | 0.01 | 0.00 | 0.03 | 0.00 | 0.02 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.44 | |
| Devonian D2 (n = 14) | Avg. | 2.01 | 2.23 | 0.40 | 1.65 | 0.32 | 0.07 | 0.43 | 0.05 | 0.30 | 0.06 | 0.18 | 0.02 | 0.13 | 0.02 | 7.80 |
| Stdev. | 2.37 | 2.46 | 0.49 | 2.07 | 0.40 | 0.09 | 0.47 | 0.06 | 0.35 | 0.07 | 0.20 | 0.02 | 0.14 | 0.02 | 9.16 | |
| Max. | 7.20 | 7.62 | 1.48 | 6.15 | 1.18 | 0.25 | 1.32 | 0.18 | 1.03 | 0.20 | 0.57 | 0.06 | 0.42 | 0.05 | 27.72 | |
| Min. | 0.14 | 0.20 | 0.03 | 0.10 | 0.03 | 0.00 | 0.03 | 0.00 | 0.02 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.56 |
| Age | Host Mineral | Size (µm) | Th (°C) | Salinity |
|---|---|---|---|---|
| Cambrian | D2 | |||
| n. | 19 | 19 | 10 | |
| Avg. | 7.26 | 102.89 | 25.42 | |
| Stdev. | 2.88 | 17.05 | 1.32 | |
| Max. | 17.00 | 132.00 | 27.20 | |
| Min. | 4.00 | 75.00 | 23.20 | |
| Cambrian | SD | |||
| n. | 5 | 5 | 0 | |
| Avg. | 8.40 | 131.60 | ||
| Stdev. | 1.96 | 20.05 | ||
| Max. | 10.00 | 156.00 | ||
| Min. | 6.00 | 111.00 | ||
| Cambrian | BKC | |||
| n. | 11 | 11 | 2 | |
| Avg. | 11.00 | 108.00 | 22.85 | |
| Stdev. | 4.24 | 22.24 | 0.75 | |
| Max. | 20.00 | 141.00 | 23.60 | |
| Min. | 6.00 | 85.00 | 22.10 | |
| Ordovician | D2 | |||
| n. | 41 | 41 | 12 | |
| Avg. | 8.20 | 96.34 | 26.46 | |
| Stdev. | 2.37 | 17.08 | 3.39 | |
| Max. | 13.00 | 132.00 | 30.50 | |
| Min. | 4.00 | 66.00 | 21.60 | |
| Ordovician | SD | |||
| n. | 10 | 10 | 8 | |
| Avg. | 9.00 | 115.20 | 23.34 | |
| Stdev. | 2.05 | 16.28 | 2.32 | |
| Max. | 12.00 | 152.00 | 25.90 | |
| Min. | 6.00 | 89.00 | 20.80 | |
| Ordovician | BKC | |||
| n. | 27 | 27 | 7 | |
| Avg. | 11.77 | 109.67 | 27.19 | |
| Stdev. | 6.27 | 16.75 | 1.87 | |
| Max. | 22.00 | 153.00 | 30.30 | |
| Min. | 5.00 | 68.00 | 24.80 | |
| Silurian | D2 | |||
| n. | 42 | 42 | 15 | |
| Avg. | 3.52 | 93.45 | 23.10 | |
| Stdev. | 2.05 | 16.20 | 0.87 | |
| Max. | 10.00 | 134.10 | 25.21 | |
| Min. | 1.00 | 49.70 | 15.17 | |
| Silurian | SD | |||
| n. | 53 | 53 | 29 | |
| Avg. | 6.02 | 123.51 | 28.54 | |
| Stdev. | 2.83 | 16.44 | 1.74 | |
| Max. | 15.00 | 193.40 | 32.63 | |
| Min. | 2.00 | 101.20 | 25.21 | |
| Silurian | BKC | |||
| n. | 53 | 53 | 25 | |
| Avg. | 5.70 | 129.18 | 27.68 | |
| Stdev. | 3.31 | 31.92 | 2.55 | |
| Max. | 18.00 | 194.30 | 32.33 | |
| Min. | 2.00 | 70.20 | 21.40 | |
| Devonian | D2 | |||
| n. | 29 | 29 | 12 | |
| Avg. | 3.34 | 83.79 | 20.74 | |
| Stdev. | 1.67 | 8.26 | 0.90 | |
| Max. | 6.00 | 102.30 | 21.82 | |
| Min. | 1.00 | 69.90 | 18.96 | |
| Devonian | BKC | |||
| n. | 48 | 48 | 25 | |
| Avg. | 5.73 | 202.04 | 20.75 | |
| Stdev. | 2.33 | 27.83 | 2.17 | |
| Max. | 12.00 | 255.10 | 23.95 | |
| Min. | 2.00 | 146.20 | 15.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Aasm, I.S.; Crowe, R.; Tortola, M. Dolomitization of Paleozoic Successions, Huron Domain of Southern Ontario, Canada: Fluid Flow and Dolomite Evolution. Water 2021, 13, 2449. https://doi.org/10.3390/w13172449
Al-Aasm IS, Crowe R, Tortola M. Dolomitization of Paleozoic Successions, Huron Domain of Southern Ontario, Canada: Fluid Flow and Dolomite Evolution. Water. 2021; 13(17):2449. https://doi.org/10.3390/w13172449
Chicago/Turabian StyleAl-Aasm, Ihsan S., Richard Crowe, and Marco Tortola. 2021. "Dolomitization of Paleozoic Successions, Huron Domain of Southern Ontario, Canada: Fluid Flow and Dolomite Evolution" Water 13, no. 17: 2449. https://doi.org/10.3390/w13172449
APA StyleAl-Aasm, I. S., Crowe, R., & Tortola, M. (2021). Dolomitization of Paleozoic Successions, Huron Domain of Southern Ontario, Canada: Fluid Flow and Dolomite Evolution. Water, 13(17), 2449. https://doi.org/10.3390/w13172449






