Evaluation of Calcium Alginate-Based Biopolymers as Potential Component of Membranes for Recovering Biosurfactants from Corn Steep Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Corn Steep Water
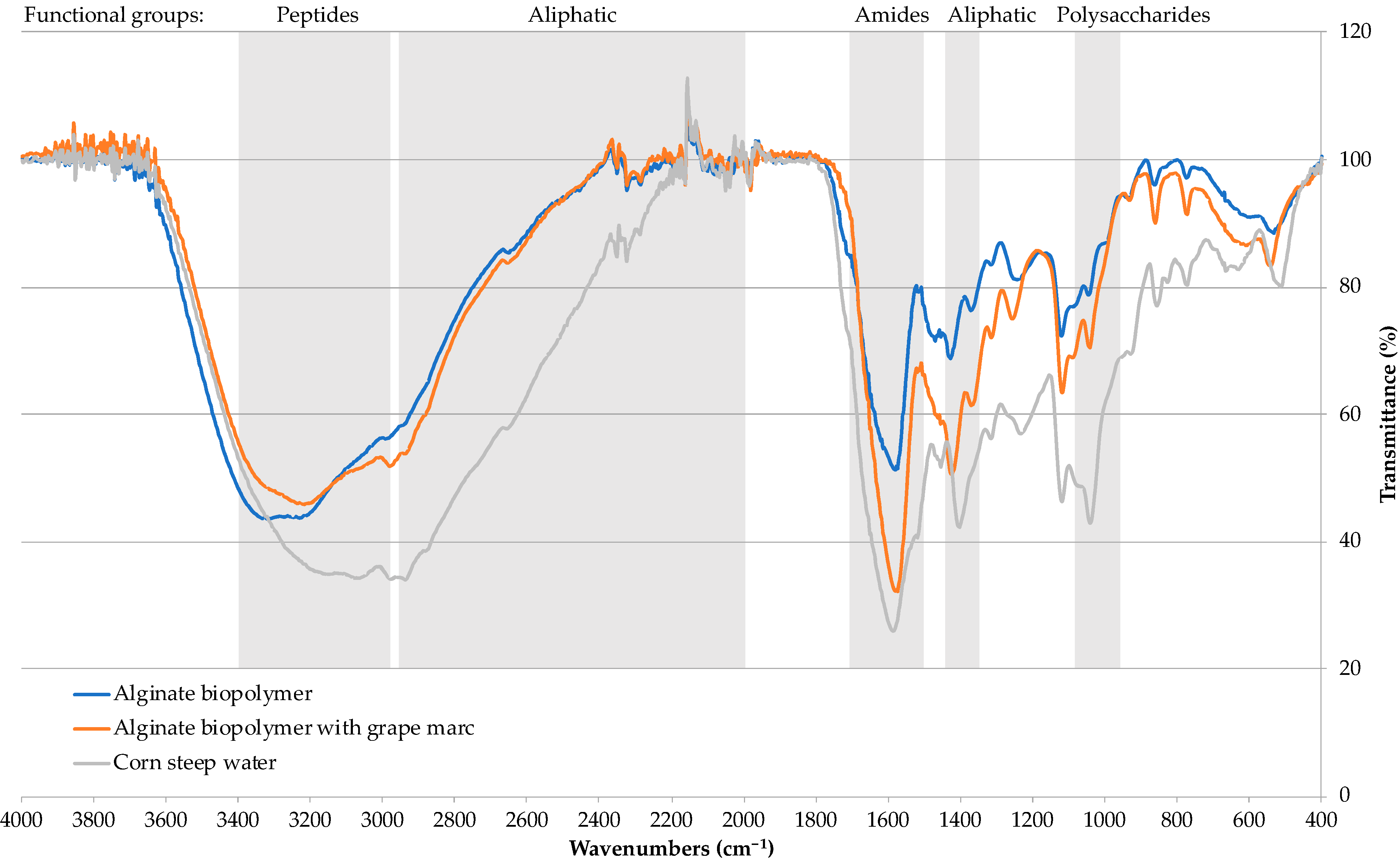
2.1.1. Fourier-Transform Infrared Spectroscopy (FTIR)
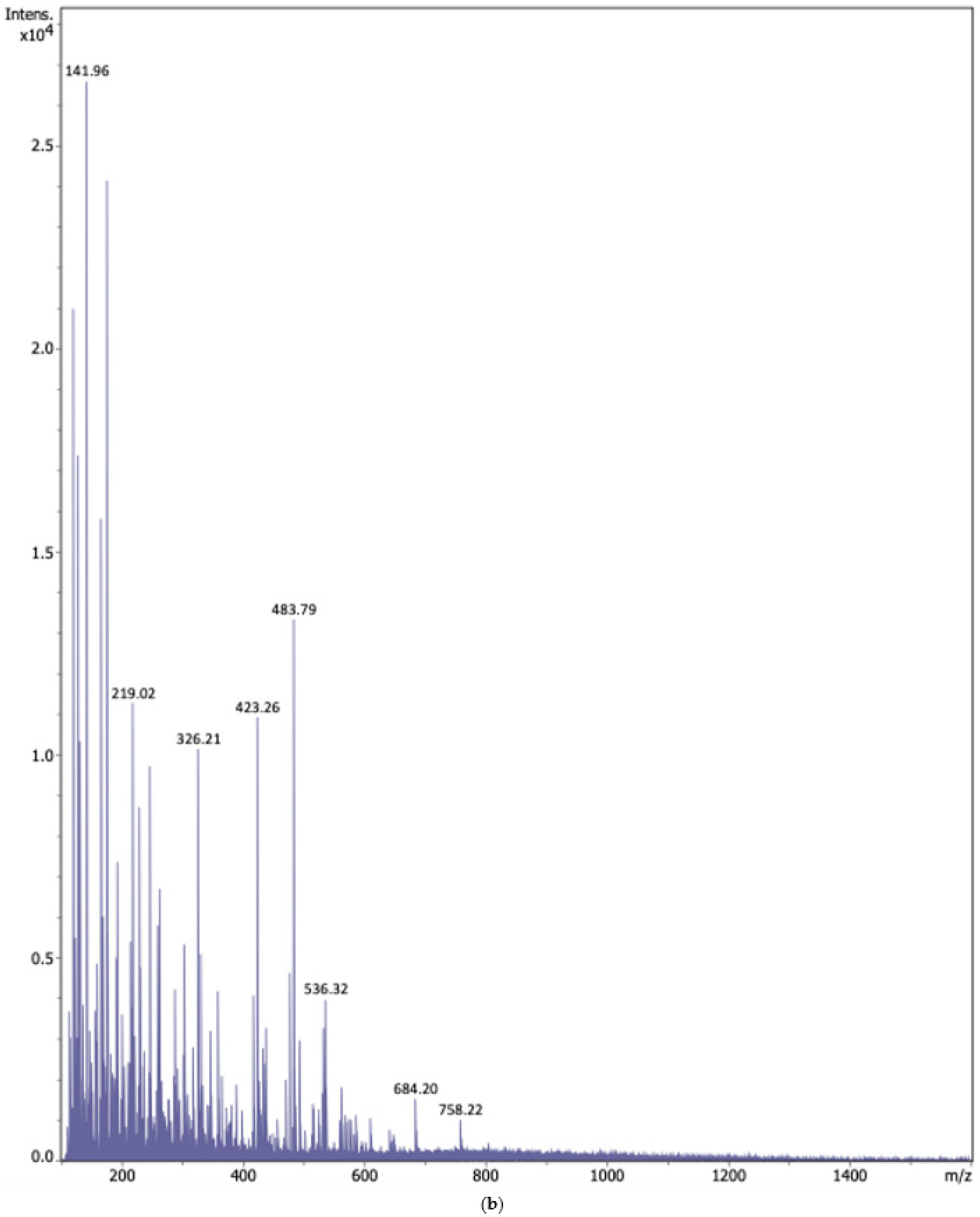
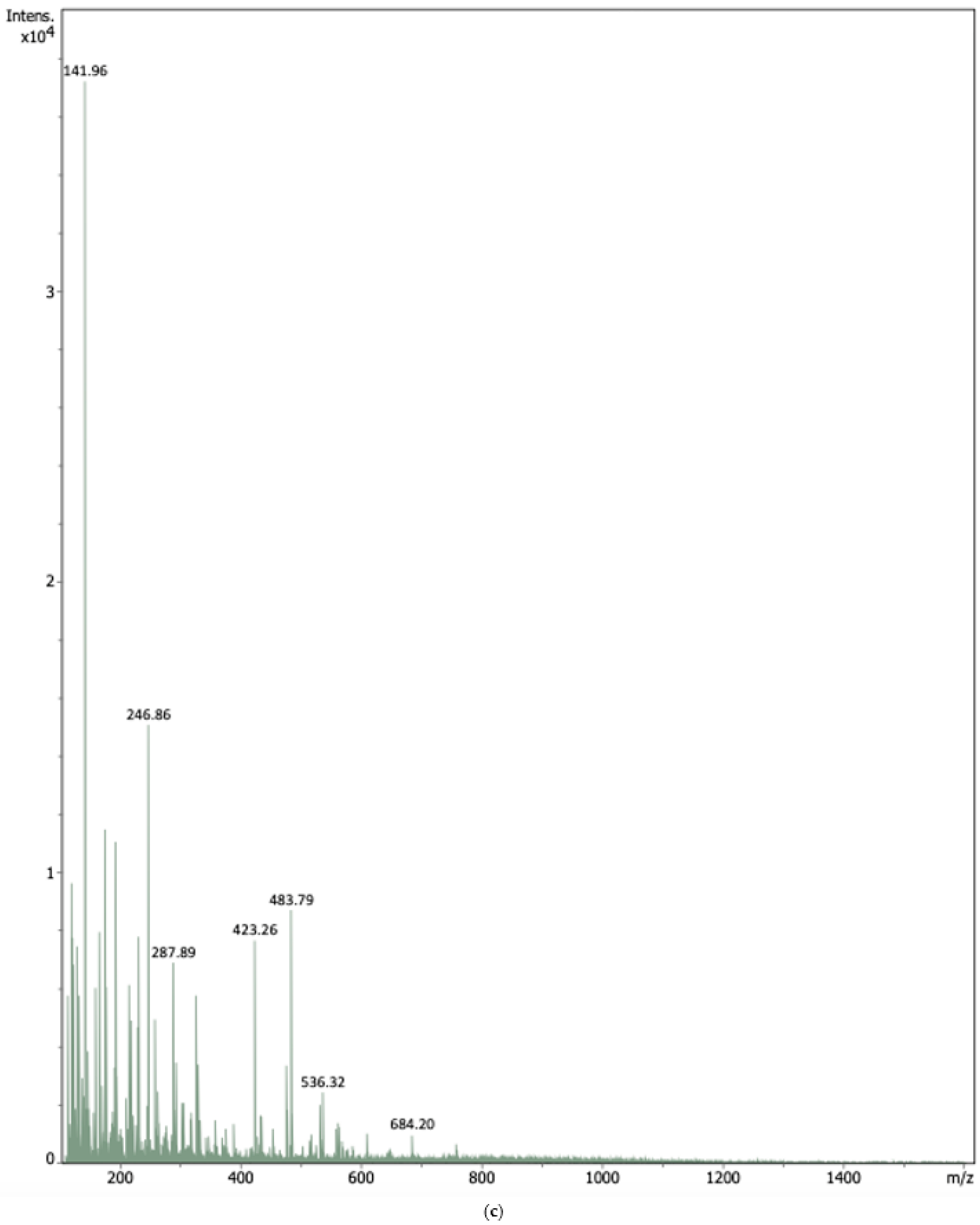
2.1.2. Electrospray Ionization Mass Spectrometry (ESI-MS)
2.2. Calcium Alginate-Based Biopolymers
Biopolymers Morphology Characterization
2.3. Adsorption Studies
Adsorption Removal and Capacity Determination
2.4. Extraction of Biosurfactants from Corn Steep Water
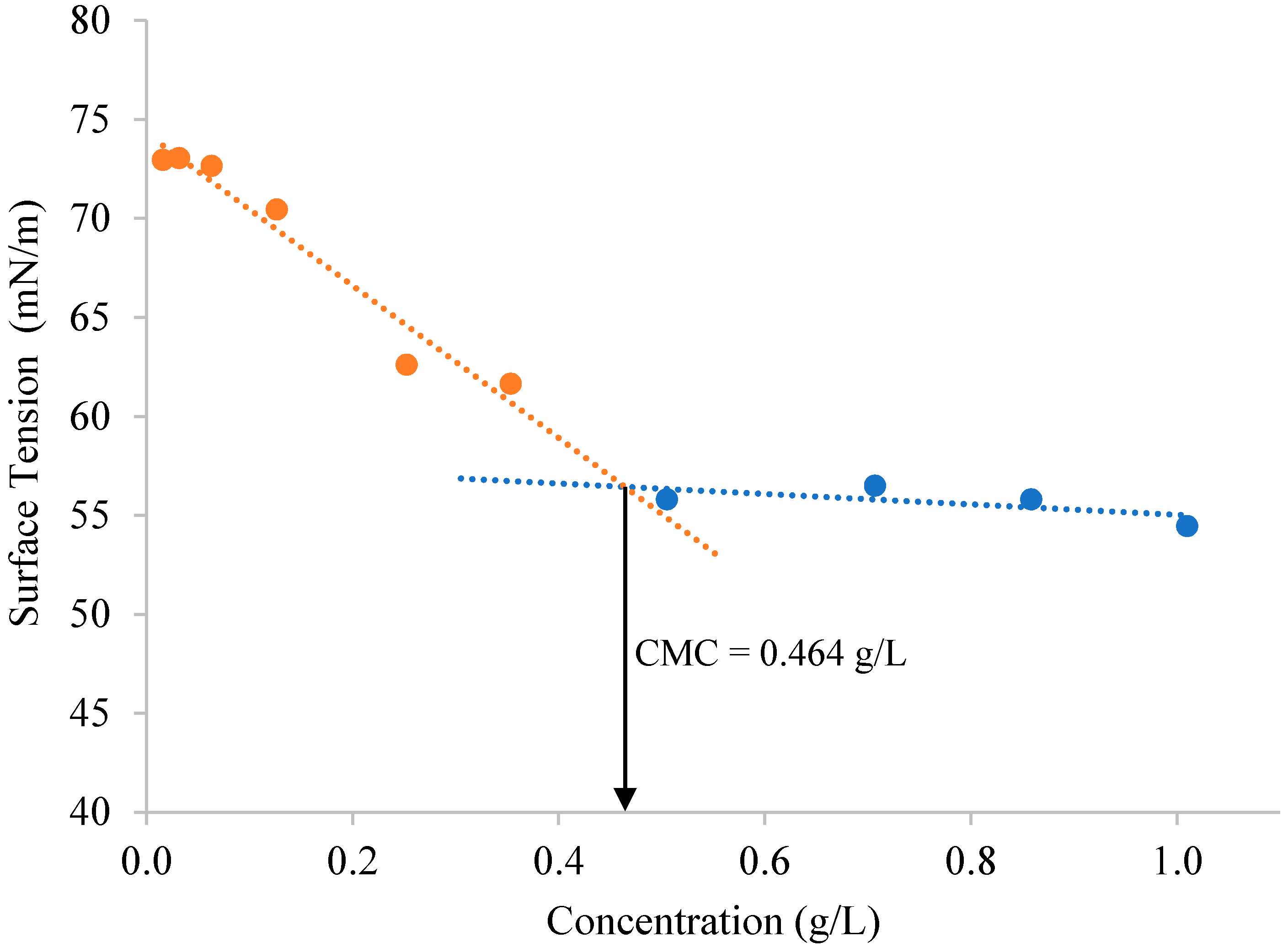
Surface Activity and Critical Micellar Concentration Determination
2.5. Measurement of Other Inorganic Solutes and Biomolecules from Corn Steep Water
2.5.1. TOC and TN
2.5.2. Anions, Organic Acids and Sugars
2.5.3. Cations and Metals
3. Results and Discussion
3.1. Corn Steep Water Composition
3.2. Calcium Alginate-Based Biopolymers Characterization
3.3. Calcium Alginate-Based Biopolymers Performance in Liquid-Solid Process to Recover Biosurfactants from Corn Steep Water
3.4. Characterization of Corn Steep Water before and after Adsorption Processes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vecino, X.; Barbosa-Pereira, L.; Devesa-Rey, R.; Cruz, J.M.; Moldes, A.B. Study of the Surfactant Properties of Aqueous Stream from the Corn Milling Industry. J. Agric. Food Chem. 2014, 62, 5451–5457. [Google Scholar] [CrossRef]
- Rivas, B.; Moldes, A.B.; Domínguez, J.M.; Parajó, J.C. Development of Culture Media Containing Spent Yeast Cells of Debaryomyces Hansenii and Corn Steep Liquor for Lactic Acid Production with Lactobacillus Rhamnosus. Int. J. Food Microbiol. 2004, 97, 93–98. [Google Scholar] [CrossRef]
- Gudiña, E.J.; Fernandes, E.C.; Rodrigues, A.I.; Teixeira, J.A.; Rodrigues, L.R. Biosurfactant Production by Bacillus Subtilis Using Corn Steep Liquor as Culture Medium. Front. Microbiol. 2015, 6, 1–7. [Google Scholar] [CrossRef]
- Wang, G.; Shi, B.; Zhang, P.; Zhao, T.; Yin, H.; Qiao, C. Effects of Corn Steep Liquor on β-Poly(l-Malic Acid) Production in Aureobasidium Melanogenum. AMB Express 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Azizi-Shotorkhoft, A.; Sharifi, A.; Mirmohammadi, D.; Baluch-Gharaei, H.; Rezaei, J. Effects of Feeding Different Levels of Corn Steep Liquor on the Performance of Fattening Lambs. J. Anim. Physiol. Anim. Nutr. 2016, 100, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ullah, Z.; Yousaf, M.; Shami, M.M.; Sharif, M.; Mahrose, K. Effect of Graded Levels of Dietary Corn Steep Liquor on Growth Performance, Nutrient Digestibility, Haematology and Histopathology of Broilers. J. Anim. Physiol. Anim. Nutr. 2018, 102, e395–e402. [Google Scholar] [CrossRef] [PubMed]
- Vecino, X.; Barbosa-Pereira, L.; Devesa-Rey, R.; Cruz, J.M.; Moldes, A.B. Optimization of Liquid–Liquid Extraction of Biosurfactants from Corn Steep Liquor. Bioprocess. Biosyst. Eng. 2015, 38, 1629–1637. [Google Scholar] [CrossRef] [PubMed]
- López-Prieto, A.; Martínez-Padrón, H.; Rodríguez-López, L.; Moldes, A.B.; Cruz, J.M. Isolation and Characterization of a Microorganism That Produces Biosurfactants in Corn Steep Water. CyTA-J. Food 2019, 17, 509–516. [Google Scholar] [CrossRef]
- López-Prieto, A.; Rodríguez-López, L.; Rincón-Fontán, M.; Cruz, J.M.; Moldes, A.B. Characterization of Extracellular and Cell Bound Biosurfactants Produced by Aneurinibacillus Aneurinilyticus Isolated from Commercial Corn Steep Liquor. Microbiol. Res. 2021, 242, 126614. [Google Scholar] [CrossRef]
- Marchant, R.; Banat, I.M. Biosurfactants: A Sustainable Replacement for Chemical Surfactants? Biotechnol. Lett. 2012, 34, 1597–1605. [Google Scholar] [CrossRef]
- Marchant, R.; Banat, I.M. Microbial Biosurfactants: Challenges and Opportunities for Future Exploitation. Trends Biotechnol. 2012, 30, 558–565. [Google Scholar] [CrossRef]
- Santos, D.K.F.; Rufino, R.D.; Luna, J.M.; Santos, V.A.; Sarubbo, L.A. Biosurfactants: Multifunctional Biomolecules of the 21st Century. Int. J. Mol. Sci. 2016, 17, 401. [Google Scholar] [CrossRef]
- Jimoh, A.A.; Lin, J. Biosurfactant: A New Frontier for Greener Technology and Environmental Sustainability. Ecotoxicol. Environ. Saf. 2019, 184, 109607. [Google Scholar] [CrossRef]
- Desai, J.D.; Banat, I.M. Microbial Production of Surfactants and Their Commercial Potential. Microbiol. Mol. Biol. Rev. 1997, 61, 47–64. [Google Scholar] [CrossRef]
- Dubey, K.V.; Juwarkar, A.A.; Singh, S.K. Adsorption-Desorption Process Using Wood-Based Activated Carbon for Recovery of Biosurfactant from Fermented Distillery Wastewater. Biotechnol. Prog. 2005, 21, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Montastruc, L.; Gancel, F.; Zhao, L.; Nikov, I. Integrated Process for Production of Surfactin. Part 1: Adsorption Rate of Pure Surfactin onto Activated Carbon. Biochem. Eng. J. 2007, 35, 333–340. [Google Scholar] [CrossRef]
- Montastruc, L.; Liu, T.; Gancel, F.; Zhao, L.; Nikov, I. Integrated Process for Production of Surfactin. Part 2. Equilibrium and Kinetic Study of Surfactin Adsorption onto Activated Carbon. Biochem. Eng. J. 2008, 38, 349–354. [Google Scholar] [CrossRef]
- Carmen dos Santos Mendes de Oliveira, A.; da Silva Bezerra, M.; de Araujo Padilha, C.E.; Melchuna, A.M.; de Macedo, G.R.; dos Santos, E.S. Recovery of Rhamnolipids Produced by Pseudomonas Aeruginosa Using Acidic Precipitation, Extraction, and Adsorption on Activated Carbon. Sep. Sci. Technol. 2013, 48, 2852–2859. [Google Scholar] [CrossRef]
- Chen, H.L.; Lee, Y.S.; Wei, Y.H.; Juang, R.S. Purification of Surfactin in Pretreated Fermentation Broths by Adsorptive Removal of Impurities. Biochem. Eng. J. 2008, 40, 452–459. [Google Scholar] [CrossRef]
- Jauregi, P.; Kourmentza, K. Membrane filtration of biosurfactants. In Separation of Functional Molecules in Food by Membrane Technology; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 79–112. ISBN 9780128150566. [Google Scholar]
- Díaz De Rienzo, M.A.; Kamalanathan, I.D.; Martin, P.J. Comparative Study of the Production of Rhamnolipid Biosurfactants by B. Thailandensis E264 and P. Aeruginosa ATCC 9027 Using Foam Fractionation. Process. Biochem. 2016, 51, 820–827. [Google Scholar] [CrossRef]
- Najmi, Z.; Ebrahimipour, G.; Franzetti, A.; Banat, I.M. In Situ Downstream Strategies for Cost-Effective Bio/Surfactant Recovery. Biotechnol. Appl. Biochem. 2018, 65, 523–532. [Google Scholar] [CrossRef]
- Juang, R.S.; Chen, H.L.; Chen, Y.S. Membrane Fouling and Resistance Analysis in Dead-End Ultrafiltration of Bacillus Subtilis Fermentation Broths. Sep. Purif. Technol. 2008, 63, 531–538. [Google Scholar] [CrossRef]
- Chen, H.L.; Chen, Y.S.; Juang, R.S. Flux Decline and Membrane Cleaning in Cross-Flow Ultrafiltration of Treated Fermentation Broths for Surfactin Recovery. Sep. Purif. Technol. 2008, 62, 47–55. [Google Scholar] [CrossRef]
- De Andrade, C.J.; de Andrade, L.M.; Rocco, S.A.; Sforça, M.L.; Pastore, G.M.; Jauregi, P. A Novel Approach for the Production and Purification of Mannosylerythritol Lipids (MEL) by Pseudozyma Tsukubaensis Using Cassava Wastewater as Substrate. Sep. Purif. Technol. 2017, 180, 157–167. [Google Scholar] [CrossRef]
- Vicente, R.; de Andrade, C.J.; de Oliveira, D.; Ambrosi, A. A Prospection on Membrane-Based Strategies for Downstream Processing of Surfactin. Chem. Eng. J. 2021, 415, 1–9. [Google Scholar] [CrossRef]
- Dalheim, M.; Omtvedt, L.A.; Bjørge, I.M.; Akbarzadeh, A.; Mano, J.F.; Aachmann, F.L.; Strand, B.L. Mechanical Properties of Ca-Saturated Hydrogels with Functionalized Alginate. Gels 2019, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Aburabie, J.H.; Puspasari, T.; Peinemann, K.V. Alginate-Based Membranes: Paving the Way for Green Organic Solvent Nanofiltration. J. Memb. Sci. 2020, 596, 117615. [Google Scholar] [CrossRef]
- Moldes, A.B.; Vázquez, M.; Domínguez, J.M.; Díaz-Fierros, F.; Barral, M.T. Evaluation of Mesophilic Biodegraded Grape Marc as Soil Fertilizer. Appl. Biochem. Biotechnol. 2007, 141, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ameneiro, M.; Vecino, X.; Barbosa-Pereira, L.; Cruz, J.M.; Moldes, A.B. Removal of Pigments from Aqueous Solution by a Calcium Alginate-Grape Marc Biopolymer: A Kinetic Study. Carbohydr. Polym. 2014, 101, 954–960. [Google Scholar] [CrossRef]
- Perez-Ameneiro, M.; Vecino, X.; Cruz, J.M.; Moldes, A.B. Wastewater Treatment Enhancement by Applying a Lipopeptide Biosurfactant to a Lignocellulosic Biocomposite. Carbohydr. Polym. 2015, 131, 186–196. [Google Scholar] [CrossRef]
- Hull, S.R.; Montgomery, R. Myo-Inositol Phosphates in Corn Steep Water. J. Agric. Food Chem. 1995, 43, 1516–1523. [Google Scholar] [CrossRef]
- Hull, S.R.; Yang, B.Y.; Venzke, D.; Kulhavy, K.; Montgomery, R. Composition of Corn Steep Water during Steeping. J. Agric. Food Chem. 1996, 44, 1857–1863. [Google Scholar] [CrossRef]
- Moldes, A.B.; Cruz, J.M.; Devesa, R.; Vecino, X. Method for Separating the Surfactants Present in the Washing Liquors of Corn, and Uses. Patent WO2014044876A1, 27 March 2014. [Google Scholar]
- Perez-Ameneiro, M.; Vecino, X.; Cruz, J.M.; Moldes, A.B. Physicochemical Study of a Bio-Based Adsorbent Made from Grape Marc. Ecol. Eng. 2015, 84, 190–193. [Google Scholar] [CrossRef]
- Perez-Ameneiro, M.; Vecino, X.; Vega, L.; Devesa-Rey, R.; Cruz, J.M.; Moldes, A.B. Elimination of Micronutrients from Winery Wastewater Using Entrapped Grape Marc in Alginate Beads. CYTA-J. Food 2014, 12, 73–79. [Google Scholar] [CrossRef][Green Version]
- Bustos, G.; Calvar, S.; Vecino, X.; Cruz, J.M.; Moldes, A.B. Industrial Symbiosis Between the Winery and Environmental Industry Through the Utilization of Grape Marc for Water Desalination Containing Copper ( II ). Water Air Soil Pollut. 2018, 229, 1–11. [Google Scholar] [CrossRef]
- Ndiaye, B.; Bustos, G.; Calvar, S.; Vecino, X.; Cruz, J.M.; Moldes, A.B.; Pérez-Cid, B. Selective Adsorption Capacity of Grape Marc Hydrogel for Adsorption of Binary Mixtures of Dyes. Water Air Soil Pollut. 2020, 231, 1–14. [Google Scholar] [CrossRef]
- Pérez-Cid, B.; Calvar, S.; Moldes, A.B.; Manuel Cruz, J. Effective Removal of Cyanide and Heavy Metals from an Industrial Electroplating Stream Using Calcium Alginate Hydrogels. Molecules 2020, 25, 5183. [Google Scholar] [CrossRef]
- Moldes, A.; Vecino, X.; Rodrıíguez-López, L.; Rincón-Fontán, M.; Cruz, J.M. Biosurfactants: The use of biomolecules in cosmetics and detergents. In New and Future Developments in Microbial Biotechnology and Bioengineering; Rodrigues, A.G., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 163–185. ISBN 9780444643018. [Google Scholar]
- Vecino, X.; Barbosa-Pereira, L.; Devesa-Rey, R.; Cruz, J.M.; Moldes, A.B. Optimization of Extraction Conditions and Fatty Acid Characterization of Lactobacillus Pentosus Cell-Bound Biosurfactant/Bioemulsifier. J. Sci. Food Agric. 2015, 95, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Andrade, R.F.S.; Silva, T.A.L.; Ribeaux, D.R.; Rodriguez, D.M.; Souza, A.F.; Lima, M.A.B.; Lima, R.A.; Alves Da Silva, C.A.; Campos-Takaki, G.M. Promising Biosurfactant Produced by Cunninghamella Echinulata UCP 1299 Using Renewable Resources and Its Application in Cotton Fabric Cleaning Process. Adv. Mater. Sci. Eng. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Rodríguez-López, L.; Rincón-Fontán, M.; Vecino, X.; Cruz, J.M.; Moldes, A.B. Extraction, Separation and Characterization of Lipopeptides and Phospholipids from Corn Steep Water. Sep. Purif. Technol. 2020, 248, 117076. [Google Scholar] [CrossRef]
- Li, J.; Deng, M.; Wang, Y.; Chen, W. Production and Characteristics of Biosurfactant Produced by Bacillus Pseudomycoides BS6 Utilizing Soybean Oil Waste. Int. Biodeterior. Biodegrad. 2016, 112, 72–79. [Google Scholar] [CrossRef]
- Ma, Y.; Kong, Q.; Qin, C.; Chen, Y.; Chen, Y.; Lv, R.; Zhou, G. Identification of Lipopeptides in Bacillus Megaterium by Two-Step Ultrafiltration and LC–ESI–MS/MS. AMB Express 2016, 6, 1–15. [Google Scholar] [CrossRef]
- LeClere, S.; Schmelz, E.A.; Chourey, P.S. Phenolic Compounds Accumulate Specifically in Maternally-Derived Tissues of Developing Maize Kernels. Cereal Chem. 2007, 84, 350–356. [Google Scholar] [CrossRef]
- Rodríguez-López, L.; Vecino, X.; Barbosa-Pereira, L.; Moldes, A.B.; Cruz, J.M. A Multifunctional Extract from Corn Steep Liquor: Antioxidant and Surfactant Activities. Food Funct. 2016, 7, 3724–3732. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, S.A.; Mou, H.; Ma, Y.; Li, M.; Hu, X. Characterization of Lipopeptide Biosurfactants Produced by Bacillus Licheniformis MB01 from Marine Sediments. Front. Microbiol. 2017, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Béchet, M.; Caradec, T.; Hussein, W.; Abderrahmani, A.; Chollet, M.; Leclére, V.; Dubois, T.; Lereclus, D.; Pupin, M.; Jacques, P. Structure, Biosynthesis, and Properties of Kurstakins, Nonribosomal Lipopeptides from Bacillus spp. Appl. Microbiol. Biotechnol. 2012, 95, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Hathout, Y.; Ho, Y.P.; Ryzhov, V.; Demirev, P.; Fenselau, C. Kurstakins: A New Class of Lipopeptides Isolated from Bacillus Thuringiensis. J. Nat. Prod. 2000, 63, 1492–1496. [Google Scholar] [CrossRef]


| Component | Concentration (mg/L) |
|---|---|
| Biosurfactant extract | 2007 ± 39 |
| Total organic carbon | 5613 ± 45 |
| Total nitrogen | 1655 ± 21 |
| Lactic acid | 6714 ± 394 |
| Acetic acid | 2822 ± 1 |
| Formic acid | 4.9 ± 0.7 |
| Phytic acid | 3141 ± 3 |
| Arabinose | 204 ± 5 |
| Glucose | 287 ± 10 |
| Sucrose | 41 ± 3 |
| Chloride | 163 ± 3 |
| Phosphate | 511 ± 16 |
| Sulphate | 242 ± 8 |
| Ammonium | 183 ± 4 |
| Calcium | 3.6 ± 0.2 |
| Aluminum | 0.1 ± 0.005 |
| Boron | 0.7 ± 0.03 |
| Chromium | 0.03 ± 0.001 |
| Copper | 0.07 ± 0.003 |
| Iron | 3.1 ± 0.1 |
| Cobalt | ND * |
| Potassium | 847 ± 45 |
| Magnesium | 301 ± 15 |
| Sodium | 183 ± 1 |
| Manganese | 1.5 ± 0.1 |
| Nickel | 0.09 ± 0.005 |
| Zinc | 4.9 ± 0.2 |
| Silicon | 8.0 ± 0.4 |
| Component | Alginate Biopolymer (%) | Alginate Biopolymer with Grape Marc (%) |
|---|---|---|
| Biosurfactant | 46.6 ± 1.5 | 54.6 ± 1.5 |
| Total organic carbon | 36.7 ± 0.5 | 48.8 ± 0.1 |
| Total nitrogen | 42.7 ± 2.4 | 57.5 ± 0.2 |
| Lactic acid | 50.0 ± 1.6 | 99.0 ± 0.1 |
| Acetic acid | 100 ± 0.1 | 83.8 ± 2.6 |
| Formic acid | 73.7 ± 4.3 | 69.1 ± 3.9 |
| Phytic acid | 72.7 ± 0.1 | 82.6 ± 1.7 |
| Arabinose | 88.5 ± 0.6 | 99.2 ± 0.1 |
| Glucose | 98.5 ± 0.6 | 99.4 ± 0.2 |
| Sucrose | 92.5 ± 1.8 | 99.8 ± 0.1 |
| Phosphate | 59.1 ± 1.1 | 75.1 ± 0.5 |
| Sulphate | 48.5 ± 2.2 | 42.0 ± 1.0 |
| Ammonium | 44.0 ± 1.4 | 27.1 ± 1.5 |
| Boron | 40.4 ± 0.8 | 8.8 ± 0.4 |
| Iron | 77.0 ± 0.1 | 98.5 ± 0.1 |
| Potassium | 49.2 ± 0.5 | 54.9 ± 0.4 |
| Magnesium | 45.8 ± 0.2 | 66.0 ± 0.3 |
| Manganese | 62.2 ± 0.2 | 90.4 ± 0.7 |
| Zinc | 58.9 ± 1.4 | 96.8 ± 0.2 |
| Silicon | 39.2 ± 0.4 | 10.1 ± 0.5 |
| Component | Alginate Biopolymer (mg/g) | Alginate Biopolymer with Grape Marc (mg/g) |
|---|---|---|
| Biosurfactant | 46.8 ± 0.4 | 54.8 ± 0.6 |
| Total organic carbon | 102.9 ± 1.3 | 136.9 ± 0.1 |
| Total nitrogen | 35.3 ± 2.0 | 47.6 ± 0.1 |
| Lactic acid | 167.9 ± 5.4 | 332.4 ± 0.1 |
| Acetic acid | 141.1 ± 0.2 | 118.2 ± 3.7 |
| Phytic acid | 114.2 ± 0.2 | 129.7 ± 2.7 |
| Arabinose | 9.0 ± 0.1 | 10.1 ± 0.1 |
| Glucose | 14.1 ± 0.1 | 14.3 ± 0.1 |
| Sucrose | 1.9 ± 0.1 | 2.0 ± 0.1 |
| Phosphate | 15.1 ± 0.3 | 19.2 ± 0.1 |
| Sulphate | 5.9 ± 0.3 | 5.1 ± 0.1 |
| Ammonium | 4.0 ± 0.1 | 2.5 ± 0.1 |
| Potassium | 20.8 ± 0.2 | 23.3 ± 0.2 |
| Magnesium | 6.9 ± 0.1 | 9.9 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Arcos, A.; Reig, M.; Cruz, J.M.; Cortina, J.L.; Moldes, A.B.; Vecino, X. Evaluation of Calcium Alginate-Based Biopolymers as Potential Component of Membranes for Recovering Biosurfactants from Corn Steep Water. Water 2021, 13, 2396. https://doi.org/10.3390/w13172396
Martínez-Arcos A, Reig M, Cruz JM, Cortina JL, Moldes AB, Vecino X. Evaluation of Calcium Alginate-Based Biopolymers as Potential Component of Membranes for Recovering Biosurfactants from Corn Steep Water. Water. 2021; 13(17):2396. https://doi.org/10.3390/w13172396
Chicago/Turabian StyleMartínez-Arcos, Andrea, Mònica Reig, José Manuel Cruz, José Luis Cortina, Ana Belén Moldes, and Xanel Vecino. 2021. "Evaluation of Calcium Alginate-Based Biopolymers as Potential Component of Membranes for Recovering Biosurfactants from Corn Steep Water" Water 13, no. 17: 2396. https://doi.org/10.3390/w13172396
APA StyleMartínez-Arcos, A., Reig, M., Cruz, J. M., Cortina, J. L., Moldes, A. B., & Vecino, X. (2021). Evaluation of Calcium Alginate-Based Biopolymers as Potential Component of Membranes for Recovering Biosurfactants from Corn Steep Water. Water, 13(17), 2396. https://doi.org/10.3390/w13172396








