Leaching and Transformation of Film Preservatives in Paints Induced by Combined Exposure to Ultraviolet Radiation and Water Contact under Controlled Laboratory Conditions
Abstract
:1. Introduction
2. Materials & Methods
2.1. Paint Formulations
2.2. Exposure of Test Specimens to UV Radiation in a Weathering Chamber
2.3. Exposure of Test Specimens to Water Contact
2.4. Exposure Schemes
2.5. Sampling
2.6. Analysis
2.7. Determination of Recovery Rates
2.8. Screening for Potential Transformation Products Using Mass Spectroscopy Methods
3. Results and Discussion
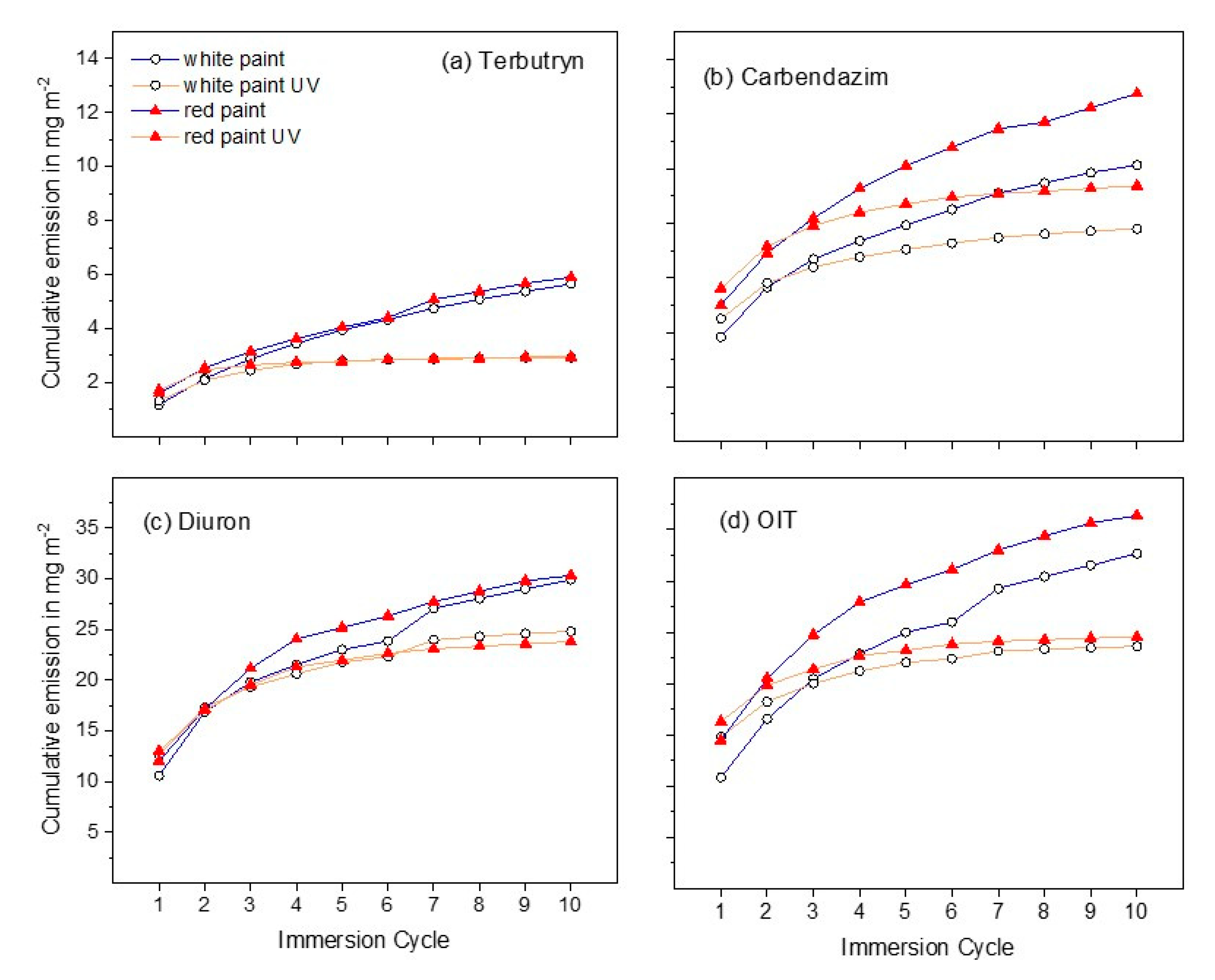
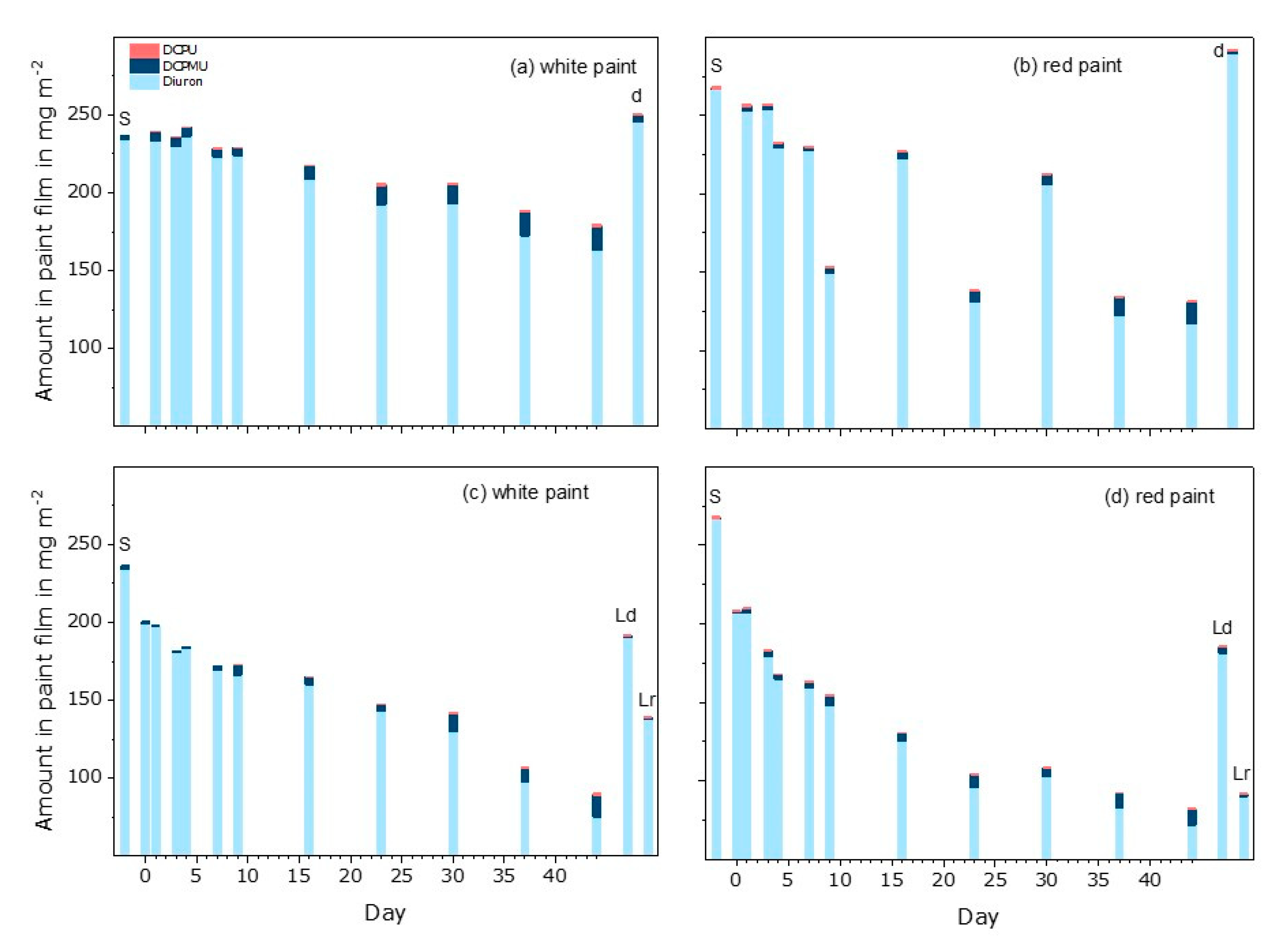
3.1. Leaching of Active Substances
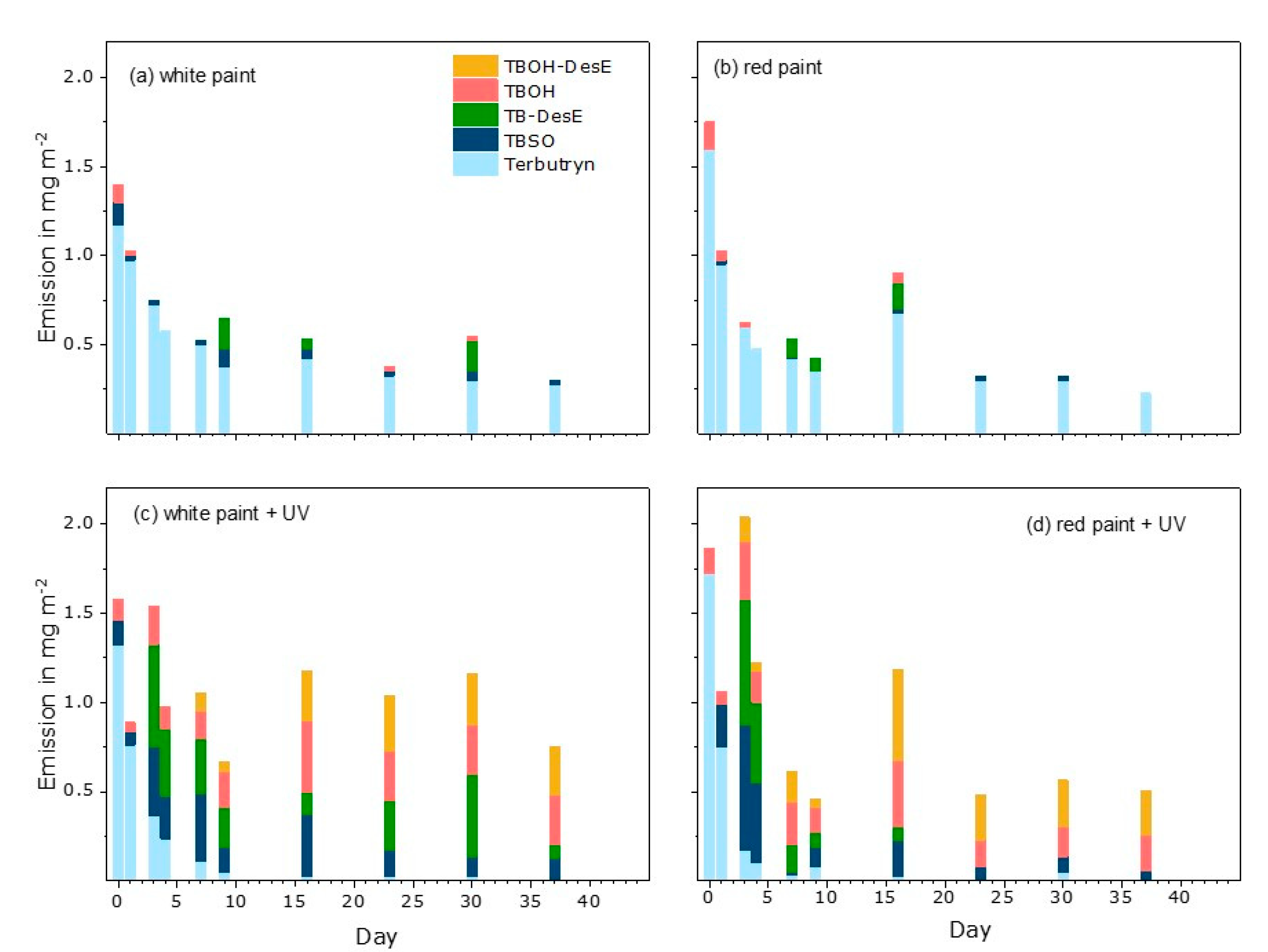
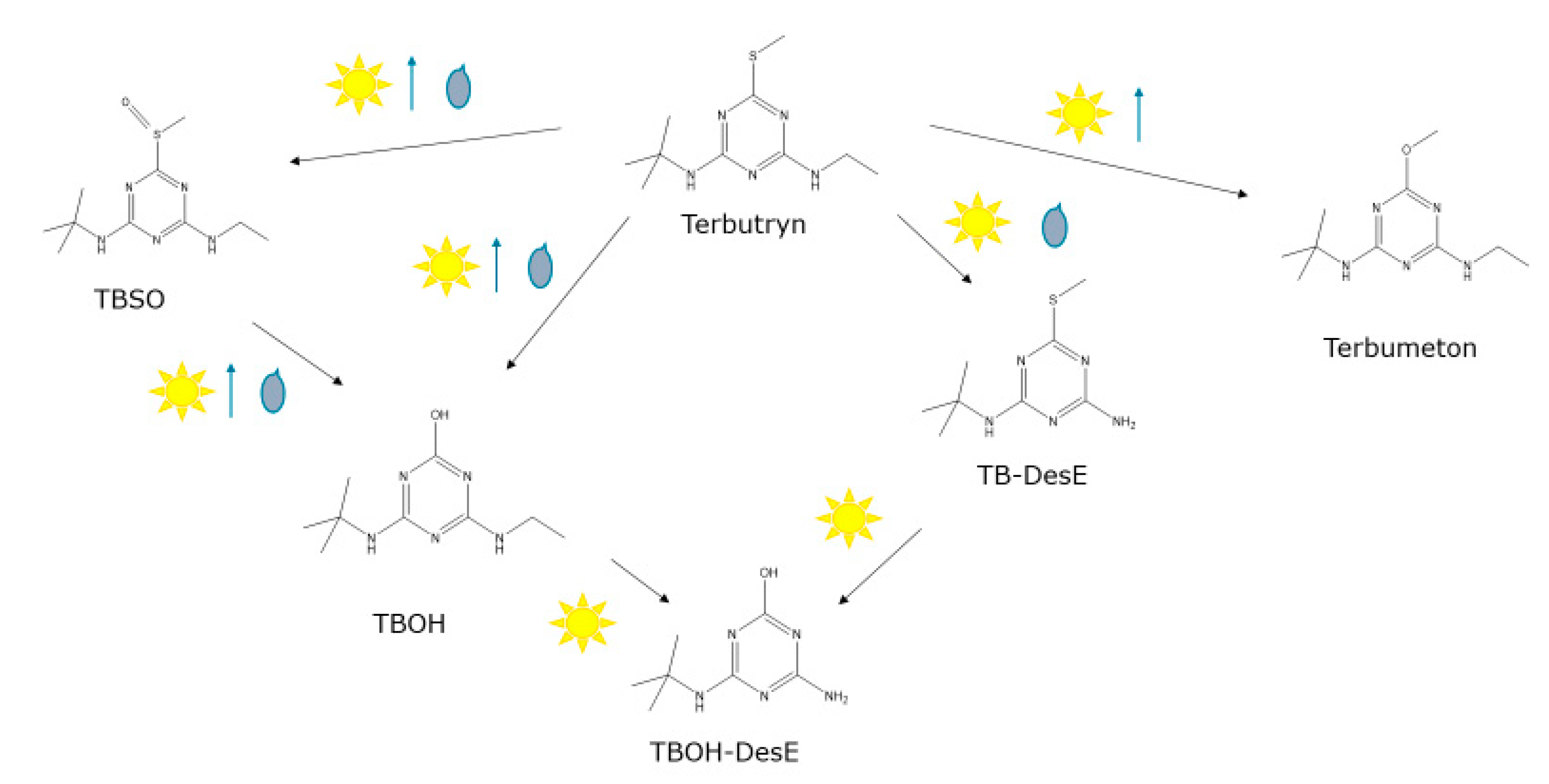
3.2. Terbutryn and Transformation Products in Eluates
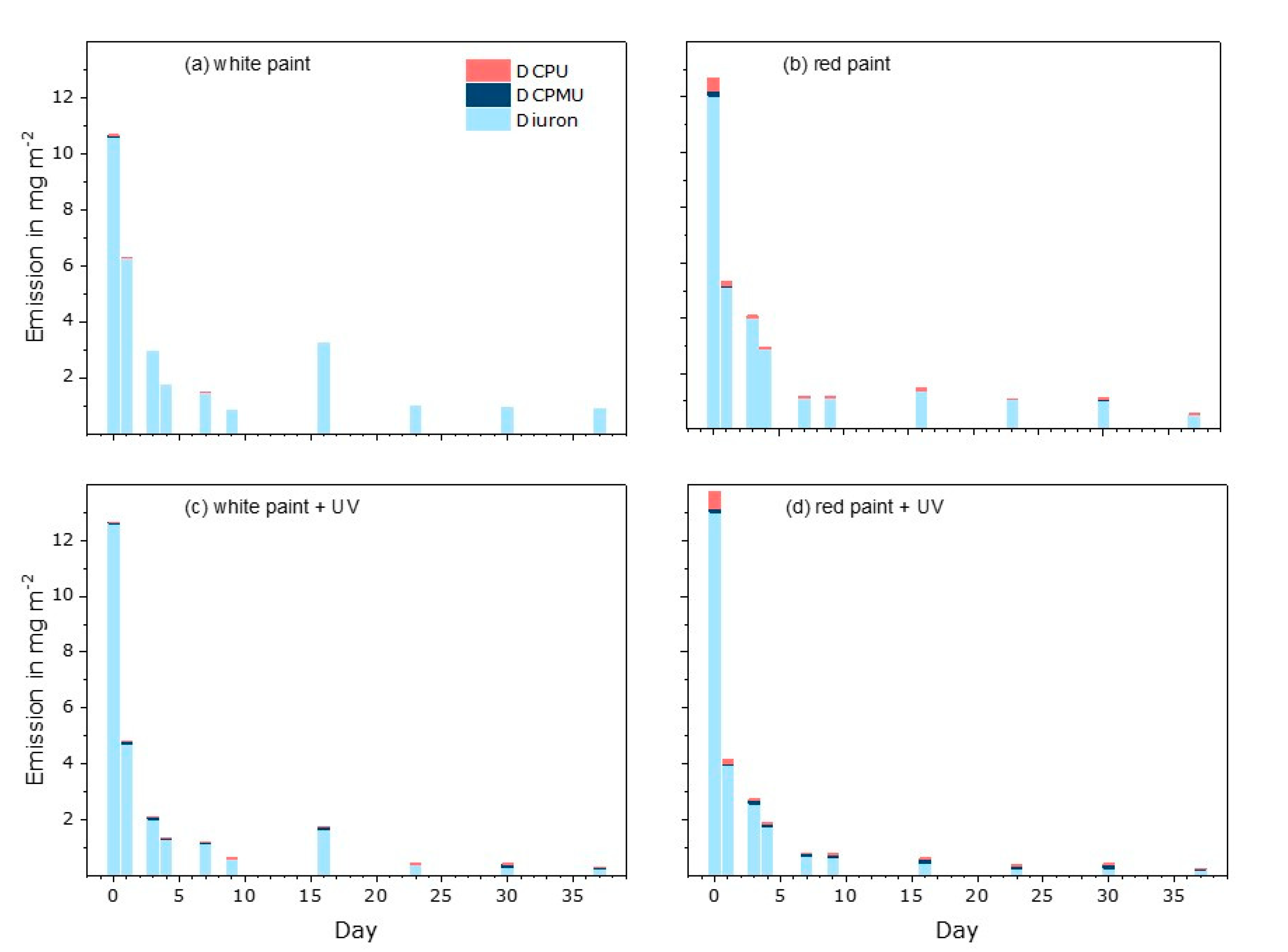
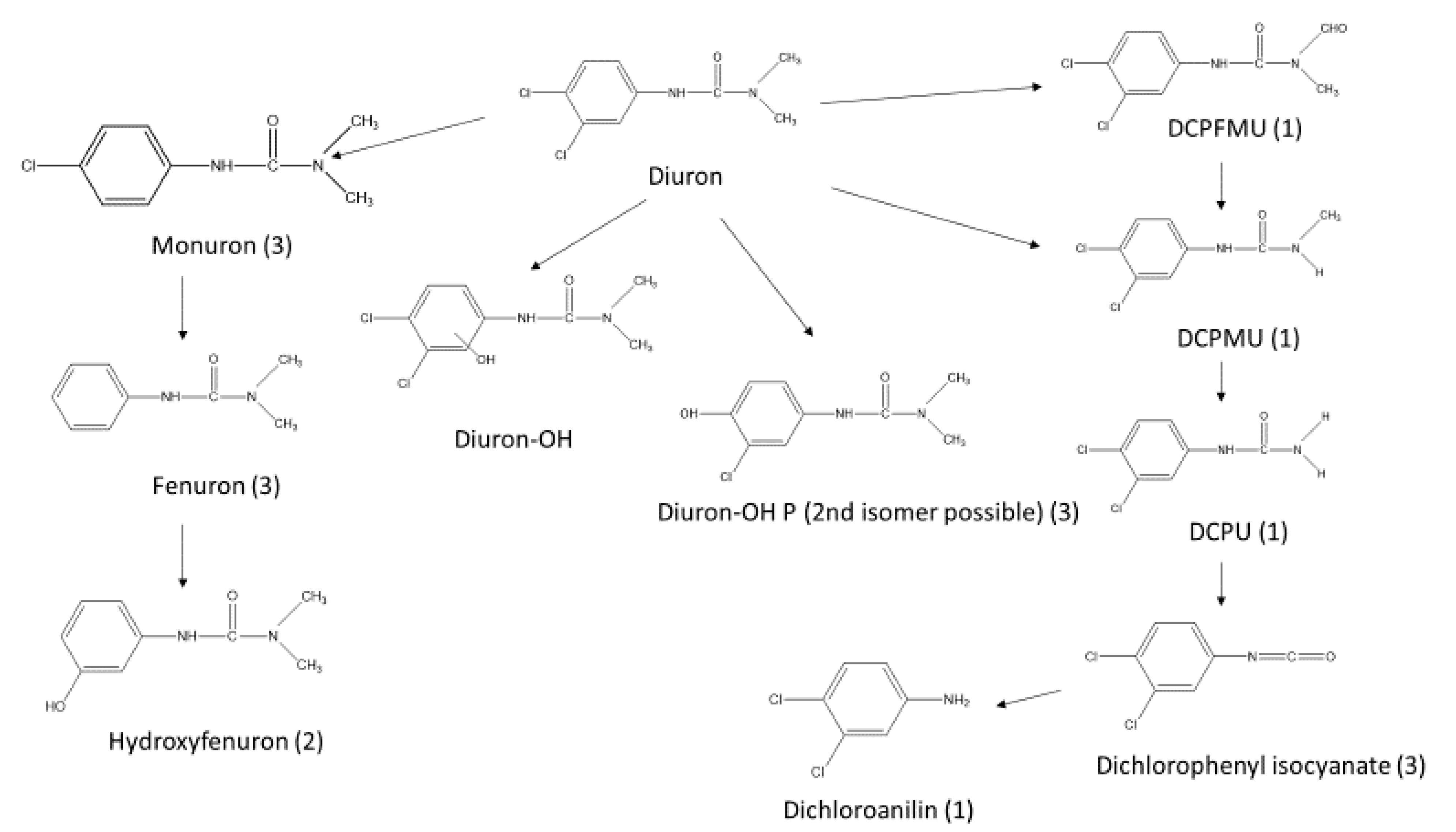
3.3. Diuron and Transformation Products in Eluates
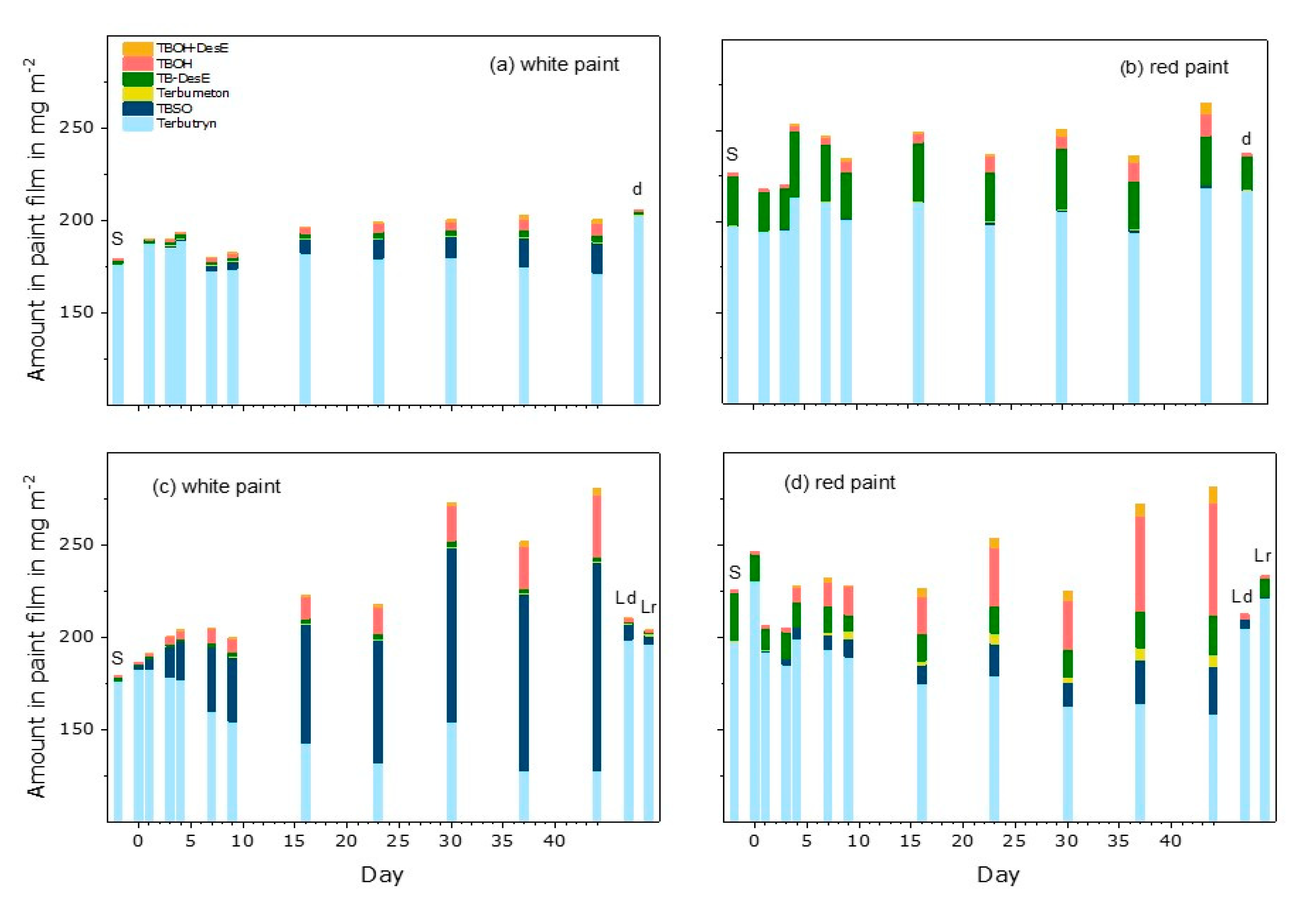
3.4. Terbutryn and Transformation Products in Paint Extracts
3.5. Diuron and Transformation Products in Paint Extracts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hensen, B.; Lange, J.; Jackisch, N.; Zieger, F.; Olsson, O.; Kümmerer, K. Entry of biocides and their transformation products into groundwater via urban stormwater infiltration systems. Water Res. 2018, 144, 413–423. [Google Scholar] [CrossRef]
- Bollmann, U.E.; Tang, C.; Eriksson, E.; Jönsson, K.; Vollertsen, J.; Bester, K. Biocides in urban wastewater treatment plant influent at dry and wet weather: Concentrations, mass flows and possible sources. Water Res. 2014, 60, 64–74. [Google Scholar] [CrossRef]
- Bollmann, U.E.; Vollertsen, J.; Carmeliet, J.; Bester, K. Dynamics of biocide emissions from buildings in a suburban stormwater catchment–Concentrations, mass loads and emission processes. Water Res. 2014, 56, 66–76. [Google Scholar] [CrossRef]
- Lange, J.; Olson, O.; Jackisch, N.; Weber, T.; Hensen, B.; Zieger, F.; Schuetz, T.; Kümmerer, K. Urbane Regenwasserversick-erung als Eintragspfad für biozide Wirkstoffe in das Grundwasser. Korresp. Wasserwirtsch. 2017, 10, 198–202. [Google Scholar]
- Wicke, D.; Matzinger, A.; Rouault, P. Relevanz Organischer Spurenstoffe im Regenwasserabfluss Berlins Abschlussbericht. 2015. Available online: https://publications.kompetenz-wasser.de/pdf/Wicke-2015-801.pdf (accessed on 26 May 2021).
- Paijens, C.; Bressy, A.; Frère, B.; Tedoldi, D.; Mailler, R.; Rocher, V.; Neveu, P.; Moilleron, R. Urban pathways of biocides towards surface waters during dry and wet weathers: Assessment at the Paris conurbation scale. J. Hazard. Mater. 2021, 402, 123765. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, M.; Zuleeg, S.; Vonbank, R.; Bester, K.; Carmeliet, J.; Boller, M.; Wangler, T. Leaching of Biocides from Façades under Natural Weather Conditions. Environ. Sci. Technol. 2012, 46, 5497–5503. [Google Scholar] [CrossRef]
- Schoknecht, U.; Mathies, H.; Wegner, R. Biocide leaching during field experiments on treated articles. Environ. Sci. Eur. 2016, 28, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bollmann, U.E.; Minelgaite, G.; Schluesener, M.P.; Ternes, T.A.; Vollertsen, J.; Bester, K. Leaching of Terbutryn and Its Photodegradation Products from Artificial Walls under Natural Weather Conditions. Environ. Sci. Technol. 2016, 50, 4289–4295. [Google Scholar] [CrossRef] [PubMed]
- Bollmann, U.E.; Minelgaite, G.; Schlüsener, M.; Ternes, T.A.; Vollertsen, J.; Bester, K. Photodegradation of octylisothiazolinone and semi-field emissions from facade coatings. Sci. Rep. 2017, 7, srep41501. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and Council. Regulation (EU) No 528/2012 Concerning the Making Available on the Market and Use of Biocidal Products. Available online: https://echa.europa.eu/regulations/biocidal-products-regulation/legislation (accessed on 26 May 2021).
- Paijens, C.; Bressy, A.; Frère, B.; Moilleron, R. Biocide emissions from building materials during wet weather: Identification of substances, mechanism of release and transfer to the aquatic environment. Environ. Sci. Pollut. Res. 2019, 27, 3768–3791. [Google Scholar] [CrossRef]
- Luft, A.; Wagner, M.; Ternes, T.A. Transformation of Biocides Irgarol and Terbutryn in the Biological Wastewater Treatment. Environ. Sci. Technol. 2013, 48, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Hensen, B.; Olsson, O.; Kümmerer, K. A strategy for an initial assessment of the ecotoxicological effects of transformation products of pesticides in aquatic systems following a tiered approach. Environ. Int. 2020, 137, 105533. [Google Scholar] [CrossRef] [PubMed]
- EN 16105 2011-09: Paints and varnishes—Laboratory method for the determination of release of substances from coatings in intermittent contact with water. Available online: https://www.beuth.de/de/norm/din-en-16105/140443204 (accessed on 30 August 2021).
- Urbanczyk, M.M.; Bester, K.; Borho, N.; Schoknecht, U.; Bollmann, U.E. Influence of pigments on phototransformation of biocides in paints. J. Hazard. Mater. 2018, 364, 125–133. [Google Scholar] [CrossRef]
- Sauer, F. Microbicides in Coatings; Hanover Vincentz Network: Hannover, Germany, 2017; ISBN 978-3-86630-897-8. [Google Scholar]
- COMMISSION IMPLEMENTING REGULATION (EU) 2021/348 of 25 February 2021. Approving carbendazim as an existing active substance for use in biocidal products of product-types 7 and 10. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32021R0348&from=EN (accessed on 26 May 2021).
- Schoknecht, U.; Sommerfeld, T.; Borho, N.; Bagda, E. Interlaboratory comparison for a laboratory leaching test procedure with façade coatings. Prog. Org. Coatings 2013, 76, 351–359. [Google Scholar] [CrossRef]
- Minelgaite, G.; Nielsen, A.H.; Pedersen, M.L.; Vollertsen, J. Photodegradation of three stormwater biocides. Urban Water J. 2015, 14, 53–60. [Google Scholar] [CrossRef]
- Kiss, A.; Rapi, S.; Csutorás, C. GC/MS studies on revealing products and reaction mechanism of photodegradation of pesticides. Microchem. J. 2007, 85, 13–20. [Google Scholar] [CrossRef]
- Johann, R.S.; Scherer, C.R. Verhalten von aus Baustoffen ausgelaugten Bioziden bei der Bodenpassage. Bauphysik 2018, 40, 369–378. [Google Scholar] [CrossRef]
- Lambropoulu, A.; Nollet, L.M.L. Transformation Products of Emerging Contaminants in the Environment; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Lányi, K.; Dinya, Z. Photodegradation study of some triazine-type herbicides. Microchem. J. 2003, 75, 1–13. [Google Scholar] [CrossRef]
- Hensen, B.; Olsson, O.; Kümmerer, K. The role of irradiation source setups and indirect phototransformation: Kinetic aspects and the formation of transformation products of weakly sunlight-absorbing pesticides. Sci. Total. Environ. 2019, 695, 133808. [Google Scholar] [CrossRef]
- Tanaka, F.S.; Hoffer, B.L.; Wien, R.G. Photolysis of 3-(3,4-dichlorophenyl)-1, 1-Dimethylurea (Diuron) in dilute aqueous solution. Toxicol. Environ. Chem. 1986, 11, 261–269. [Google Scholar] [CrossRef]
- Jirkovský, J.; Faure, V.; Boule, P. Photolysis of Diuron. Pestic. Sci. 1997, 50, 42–52. [Google Scholar] [CrossRef]
- Tixier, C.; Sancelme, M.; Bonnemoy, F.; Cuer, A.; Veschambre, H. Degradation products of a phenylurea herbicide, diuron: Synthesis, ecotoxicity, and biotransformation. Environ. Toxicol. Chem. 2001, 20, 1381–1389. [Google Scholar] [CrossRef]
- López, M.C.; Fernández-Pérez, M.-I.; Rodríguez, S.; Santaballa, J.A.; Steenken, S.; Vulliet, E. Mechanisms of Direct and TiO2-Photocatalysed UV Degradation of Phenylurea Herbicides. ChemPhysChem 2005, 6, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Khongthon, W.; Jovanovic, G.; Yokochi, A.; Sangvanich, P.; Pavarajarn, V. Degradation of diuron via an electrochemical advanced oxidation process in a microscale-based reactor. Chem. Eng. J. 2016, 292, 298–307. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schoknecht, U.; Mathies, H.; Lisec, J. Leaching and Transformation of Film Preservatives in Paints Induced by Combined Exposure to Ultraviolet Radiation and Water Contact under Controlled Laboratory Conditions. Water 2021, 13, 2390. https://doi.org/10.3390/w13172390
Schoknecht U, Mathies H, Lisec J. Leaching and Transformation of Film Preservatives in Paints Induced by Combined Exposure to Ultraviolet Radiation and Water Contact under Controlled Laboratory Conditions. Water. 2021; 13(17):2390. https://doi.org/10.3390/w13172390
Chicago/Turabian StyleSchoknecht, Ute, Helena Mathies, and Jan Lisec. 2021. "Leaching and Transformation of Film Preservatives in Paints Induced by Combined Exposure to Ultraviolet Radiation and Water Contact under Controlled Laboratory Conditions" Water 13, no. 17: 2390. https://doi.org/10.3390/w13172390
APA StyleSchoknecht, U., Mathies, H., & Lisec, J. (2021). Leaching and Transformation of Film Preservatives in Paints Induced by Combined Exposure to Ultraviolet Radiation and Water Contact under Controlled Laboratory Conditions. Water, 13(17), 2390. https://doi.org/10.3390/w13172390






