Soil Hydrology Process and Rational Use of Soil Water in Desert Regions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Preparation
2.2. Canopy Interception Measurements
2.3. Runoff Measurement
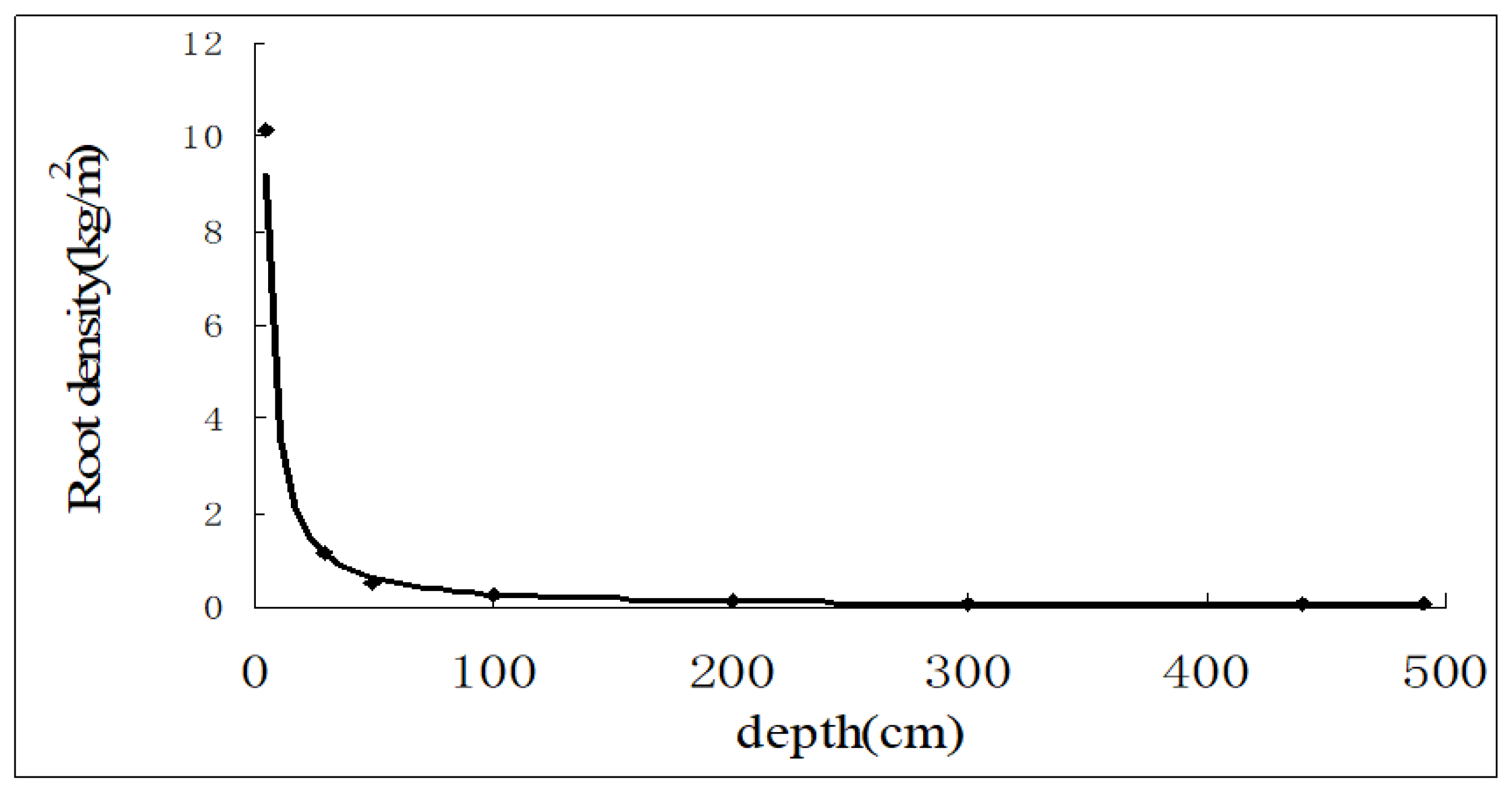
2.4. Root Distribution
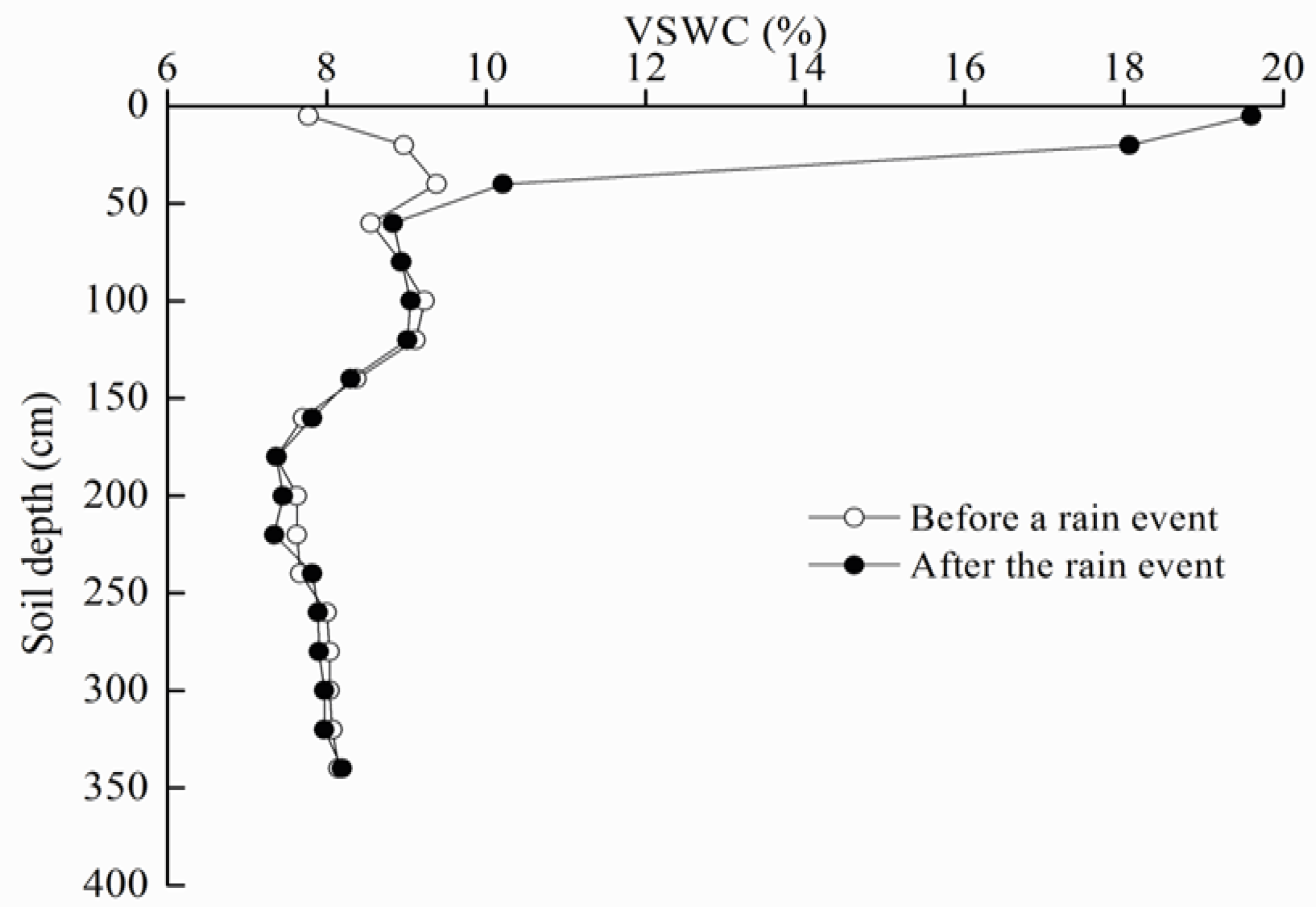
2.5. Soil Water Measurement
2.6. Moisture Content Measurement at Different Soil Suctions
2.7. Estimating Method for the Critical Period of Plant–Water Relationship Regulation
3. Results and Discussion
3.1. Soil Water Resources
3.2. Root Water Absorption
3.3. Use Limit of Soil Moisture Resource by Plants (ULSMRP)
3.4. Infiltration and Maximum Infiltration Depth
3.5. Carrying Capacity
3.6. Carrying Capacity of Soil Water for Vegetation
3.7. The Key Period of Regulating the Plant–Water Relationship
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lindh, M.; Zhang, L.; Falster, D.; Franklin, O.; Brännström, Å. Plant diversity and drought: The role of deep roots. Ecol. Model. 2014, 290, 85–93. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to whole plant. Funct. Plant. Biol. 2003, 30, 239–264. [Google Scholar]
- Jackson, R.B.; Canadell, J.; Ehleringer, J.R.; Mooney, H.A.; Sala, O.E.; Schulze, E.D. A global analysis of root distributions for terrestrial biomes (Review). Oecologia 1996, 108, 389–411. [Google Scholar] [CrossRef]
- Huang, L.M.; Shao, M.A. Advances and perspectives on soil water research on the Chin’s Loess plateau. Earth Sci. Rev. 2019, 199, 22. [Google Scholar] [CrossRef]
- Guo, Z.S.; Shao, M.A. Impact of afforestation density on soil and water conservation of the semiarid Loess Plateau. J. Soil Water Conserv. 2013, 68, 401–410. [Google Scholar] [CrossRef]
- Gao, X.D.; Meng, T.T.; Zhao, X.N. Variations of Soil Organic Carbon Following Land Use Change on Deep-Loess Hillsopes in China. Land Degrad. Dev. 2018, 28, 1902–1912. [Google Scholar] [CrossRef]
- Guo, Z.S. Soil Water Carrying Capacity for Vegetation in a Semi-arid Region of Loess Hilly in the Loess Plateau. Ph.D. Thesis, Northwest Sci-Tech University of Agriculture and Forestry, Xianyang, China, 2004. [Google Scholar]
- Guo, Z.S. Soil Water Carrying Capacity for Vegetation. Land Degrad. Dev. 2021, 32, 3801–3811. [Google Scholar] [CrossRef]
- Guo, Z.S.; Shao, M.A. Soil Water infiltrating Process in Afforested land on Slopes of the Semiarid region of Loess Plateau. Chin. J. Pedologica Sinic. 2009, 46, 953–958. [Google Scholar]
- Feng, X.; Fu, B.; Piao, S.; Wang, S.; Ciais, P.; Zeng, Z.; Lü, Y.; Zeng, Y.; Li, Y.; Jiang, X.; et al. Revegetation in China’s Loess Plateau is approaching to sustainable water resources limits. Nat. Clim. Chang. 2016, 6, 1019–1022. [Google Scholar] [CrossRef]
- Li, L.J.; Song, X.Y.; Xia, L.; Fu, N.; Feng, D. Responnse of evaporation and transpiration of typical afforestation tree species to climate changechange in the gully region of loess plateau. Trans. Chin. Soc. Agric. Eng. 2018, 34, 148–159. [Google Scholar]
- Pierret, A.; Maeght, J.L.; Clément, C.; Montoroi, J.P.; Hartmann, C.; Gonkhamdee, S. Understanding deep roots and their functions in ecosystems: An advocacy for more unconventional research. Ann. Bot. 2016, 118, 621–635. [Google Scholar] [CrossRef] [Green Version]
- Li, H.J. Root Water Uptake Process in Deep Soil for Forest Growing on the Loess Plateau and Its Effect on Water Stress and Soil Carbon Input; Northwest A&F University: Xianyang, China, 2019; pp. 36–37. [Google Scholar]
- Nepstad, D.C.; De Carvalho, C.R.; Davidson, E.A.; Jipp, P.H.; Lefebvre, P.A.; Negreiros, G.H.; Da Silva, E.D.; Stone, T.A. The role of deep roots in the hydrological and carbon circles of amazonnian forests and pasture. Nature 1994, 372, 666–669. [Google Scholar] [CrossRef] [Green Version]
- Markewitz, D.; Devine, S.; Davidson, E.A.; Brando, P.; Nepstad, D.C. Soil moisture depletion under simulated drought in the Amazon: Impacts on deep root uptake. New Phytol. 2010, 187, 592–607. [Google Scholar] [CrossRef]
- Wang, Y.; Shao, M.A. A preliminary investigation of the dynamic characteristics of dried soil layers on the Loess Plateau of China. J. Hydrol. 2010, 381, 9–17. [Google Scholar] [CrossRef]
- Chen, H.S.; Shao, M.A.; Li, Y.Y. Soil desiccation in the Loess Plateau of China. Geoderma 2008, 143, 91–100. [Google Scholar] [CrossRef]
- Li, Y.S. Effects of forest on water circle on the Loess Plateau. Chinese. J. Nat. Resour. 2001, 16, 427–432. [Google Scholar]
- Guo, Z.S.; Shao, M.A.; Zhang, Y.P.; Wu, Q.X. A Layer-dividing method to soil water in forest land. In Proceedings of Soil Physics and Ecological Environment Construction; Shao, M.A., Ed.; Shaanxi Science and Technology Press: Xian, China, 2002; pp. 75–79. [Google Scholar]
- Guo, Z.S.; Shao, M.A. Carrying capacity of soil water for Vegetation in the Loess Plateau. In Water—Saving Agriculture and Sustainable Use of Water and Land Resources; Shaanxi Science and Technology Press: Xian, China, 2003; pp. 704–711. [Google Scholar]
- Li, B.; Zhang, W.; Li, S.; Wang, J.; Liu, G.; Xu, M. Severe depletion of available deep soil water induced by revegetation on the arid and semiarid Loess Plateau. For. Ecol. Manag. 2021, 491. [Google Scholar] [CrossRef]
- Guo, Z.S. Estimating Method of Maximum Infiltration Depth and Soil Water Supply. Sci. Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.S.; Shao, M.A. Mathmatics Model for determining soil water Carrying capacity for Vegetation. Chin. J. of Hydraul. Eng. 2004, 10, 95–99. [Google Scholar]
- Guo, Z.S. Theory and Practice on Soil Water Carrying Capacity for Vegetation; Chinese Science Press: Beijing, China, 2014; Available online: http://www.geobooks.com.cn (accessed on 25 August 2021).
- Hauser, V.L. Neutron meter calibration and error control. Trans. Am. Soc. Agric. Eng. 1984, 27, 722–728. [Google Scholar] [CrossRef]
- Evett, S.R.; Schwartz, R.C.; Casanova, J.J.; Heng, L.K. Soil water sensing for water balance, ET and WUE. Agric. Water Manage. 2012, 104, 1–9. [Google Scholar] [CrossRef]
- Guo, Z.S.; Li, Y.L. Initiation stage to regulate the caragana growth and soil water in the semiarid area of Loess Hilly Region, China. Chin. J. Ecol. 2009, 29, 5721–5729. [Google Scholar]
- Guo, Z.S.; Shao, M.A. Use of the theory of Soil Water Carrying Capacity for Vegetation in Practice. Chin. J. Nat. Resour. 2009, 24, 2187–2193. [Google Scholar]
- Budagovski, A.I. Soil water resources and available water supply of the vegetation cover. Water Resour. 1986, 12, 317–325. [Google Scholar]
- Lvovich, M.I. Soil trend in hydrology. Hydrol. Sci. Bull. 1980, 25, 33–45. [Google Scholar] [CrossRef]
- Guo, Z.S. A Review of Soil Water Carrying Capacity for Vegetation. Chin.J. Sci. Silvae Sinicae. 2011, 47, 140–144. [Google Scholar]
- Guo, Z.S. Soil water resource use limit in semi-arid loess hilly area. Chin. J. Appl. Ecol. 2010, 21, 3029–3035. [Google Scholar]
- Price, D. Carrying capacity reconsidered. Popul. Environ. 1999, 21, 5–26. [Google Scholar] [CrossRef]
- Young, C.C. Defining the range: The development of carrying capacity in management. J. Hist. Biol. 1998, 31, 61–83. [Google Scholar] [CrossRef]

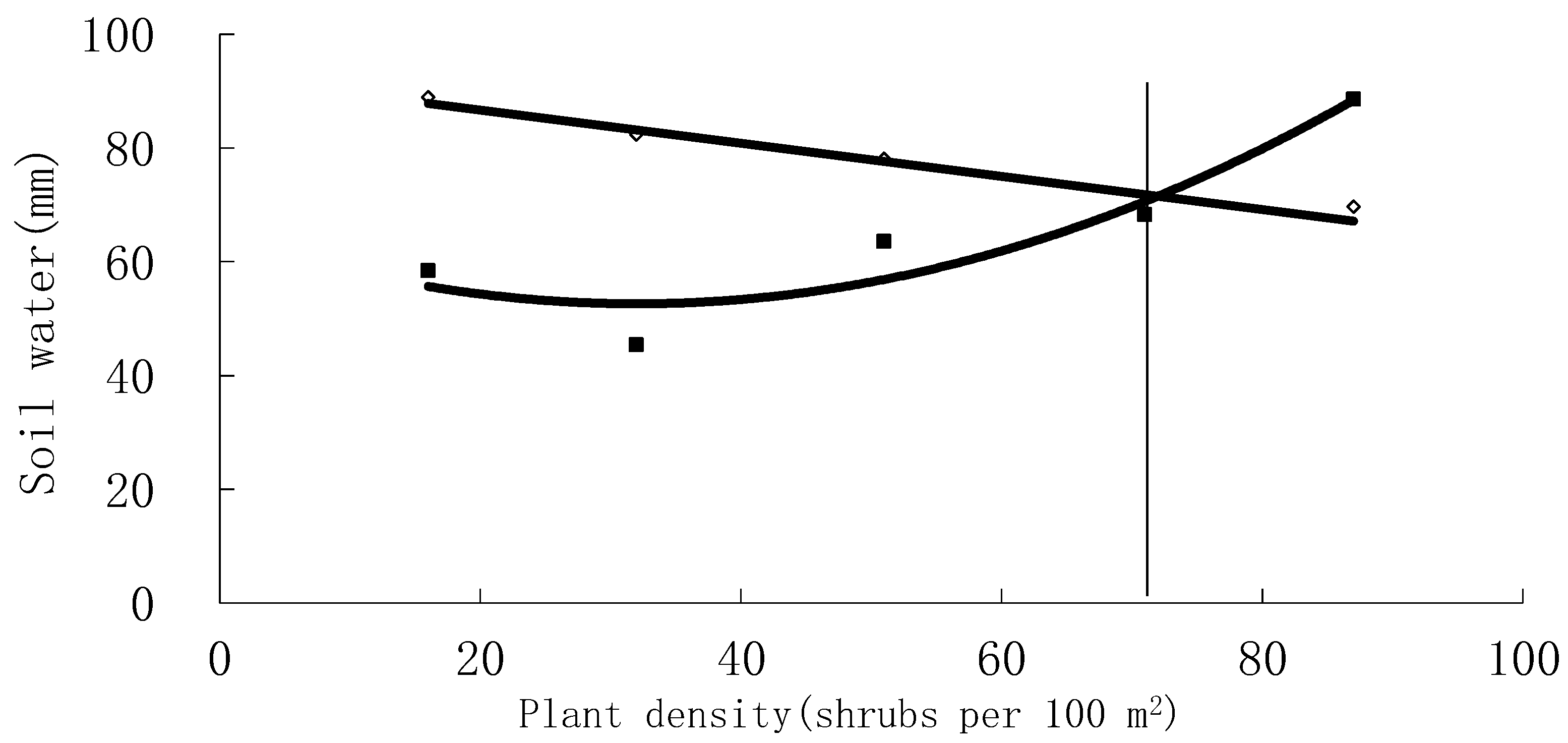

| Planting density | 87 | 71 | 51 | 32 | 16 |
| Slope gradient (°) | 7.9 | 7.8 | 8.5 | 7.7 | 7 |
| Projection area (m2) | 99.2 | 99.1 | 99.1 | 99.3 | 99.4 |
| Plant density (Brushes/100 m2) | 87 | 71 | 51 | 32 | 16 |
| Soil water supply (mm) | 69.64 | 68.53 | 78.11 | 82.35 | 88.97 |
| Soil water consumption (mm) | 88.57 | 68.28 | 63.58 | 45.42 | 58.4 |
| Seed yield (g/100 m2) | 39 | 76 | 130 | 63 | 45.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Z. Soil Hydrology Process and Rational Use of Soil Water in Desert Regions. Water 2021, 13, 2377. https://doi.org/10.3390/w13172377
Guo Z. Soil Hydrology Process and Rational Use of Soil Water in Desert Regions. Water. 2021; 13(17):2377. https://doi.org/10.3390/w13172377
Chicago/Turabian StyleGuo, Zhongsheng. 2021. "Soil Hydrology Process and Rational Use of Soil Water in Desert Regions" Water 13, no. 17: 2377. https://doi.org/10.3390/w13172377
APA StyleGuo, Z. (2021). Soil Hydrology Process and Rational Use of Soil Water in Desert Regions. Water, 13(17), 2377. https://doi.org/10.3390/w13172377






