Environmental Drivers of Macroinvertebrate Assemblages within Peat Pool Habitat-Implication for Bioassessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Water Analysis
2.3. Macroinvertebrates Sampling
2.4. Statistical Analysis
3. Results
3.1. Environmental Variables
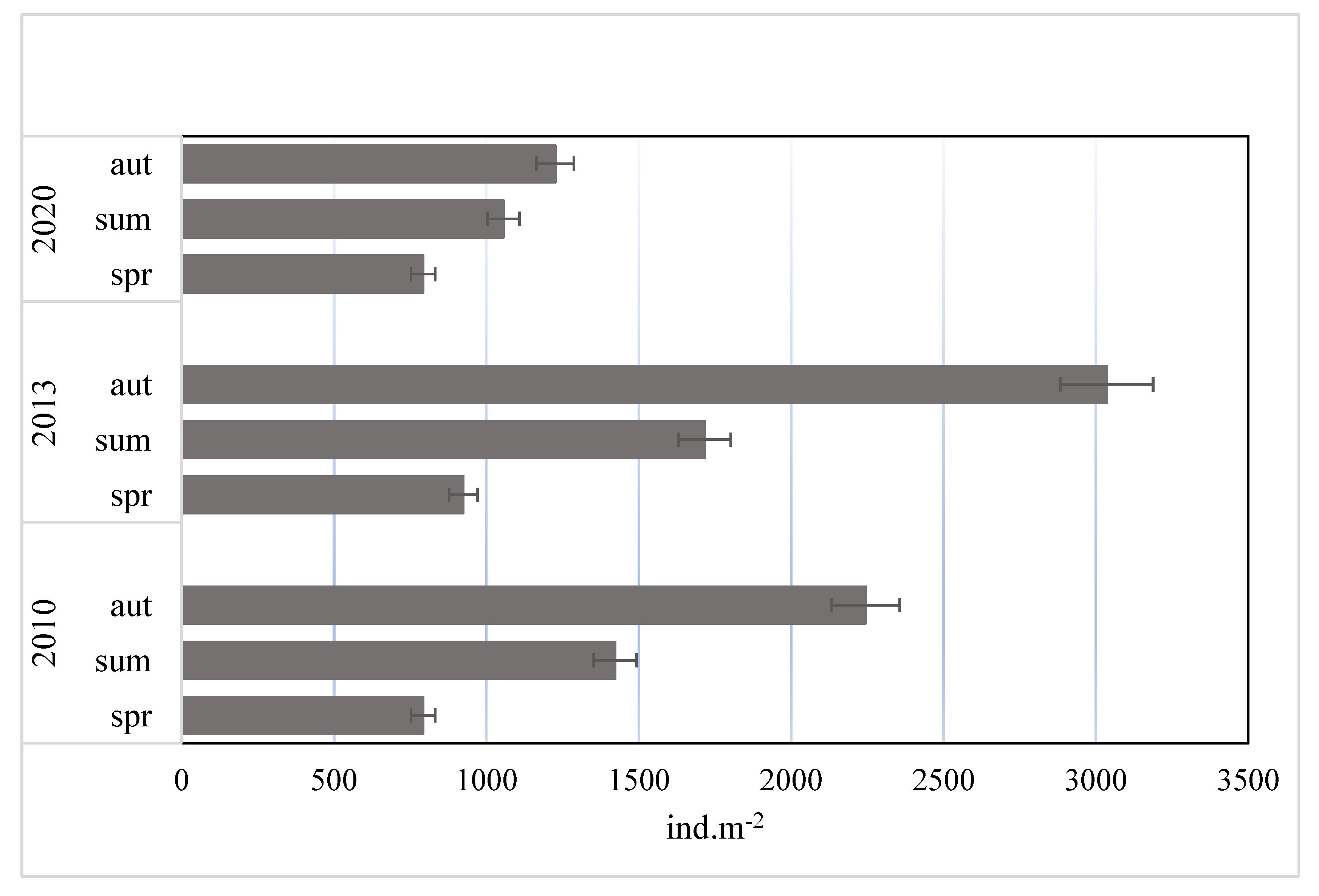
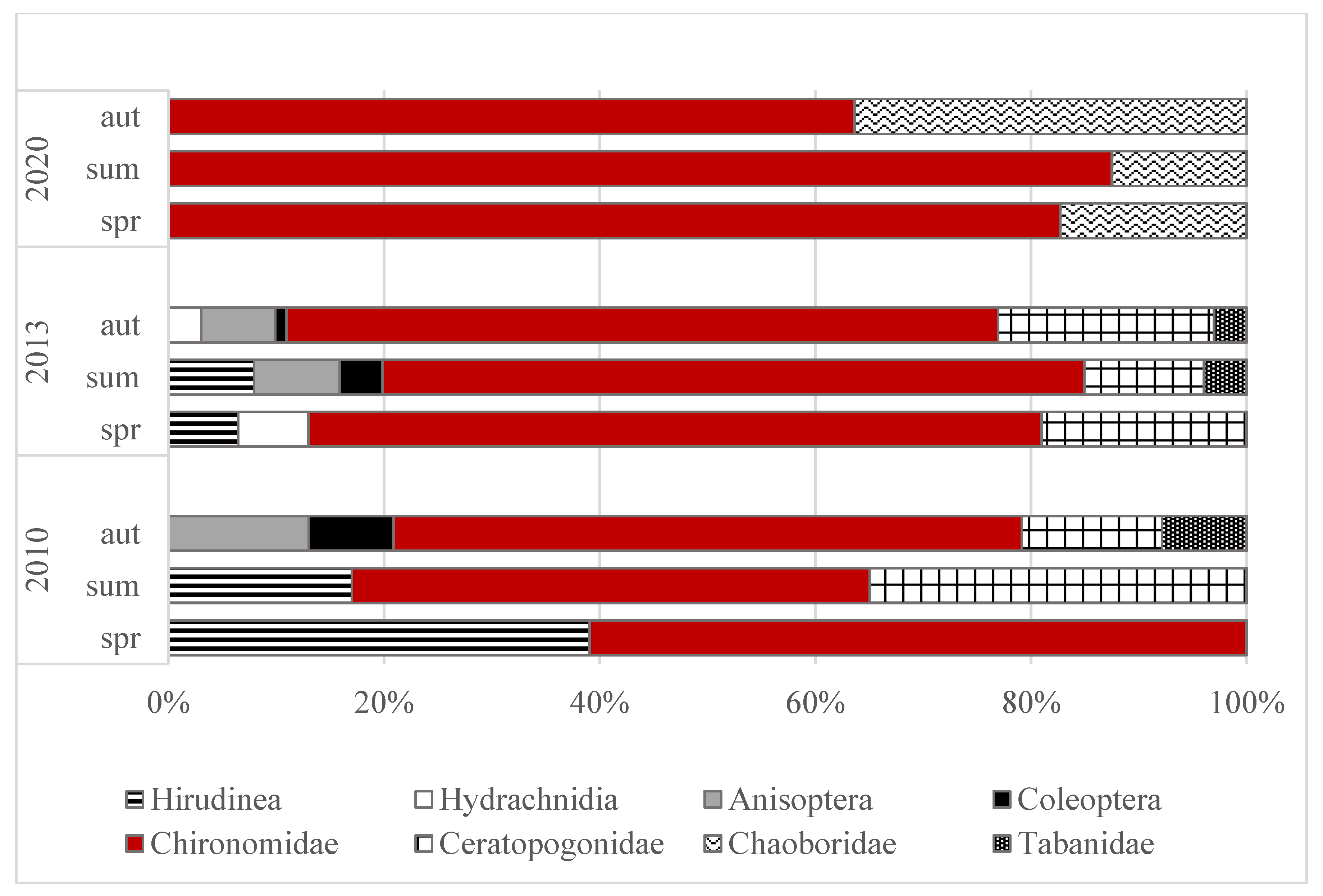
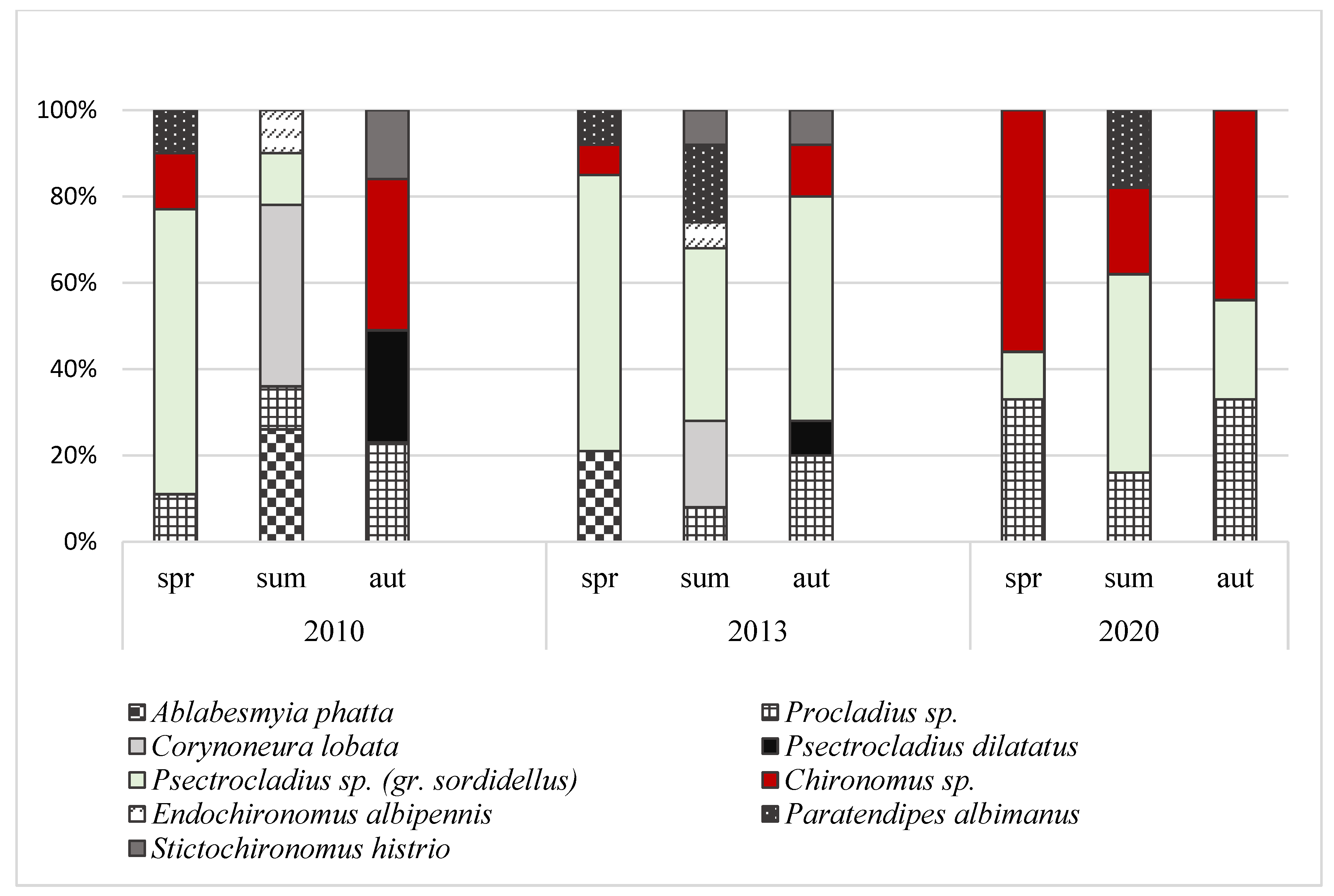
3.2. Abundance and Domination Structure of Macroinvertebrates
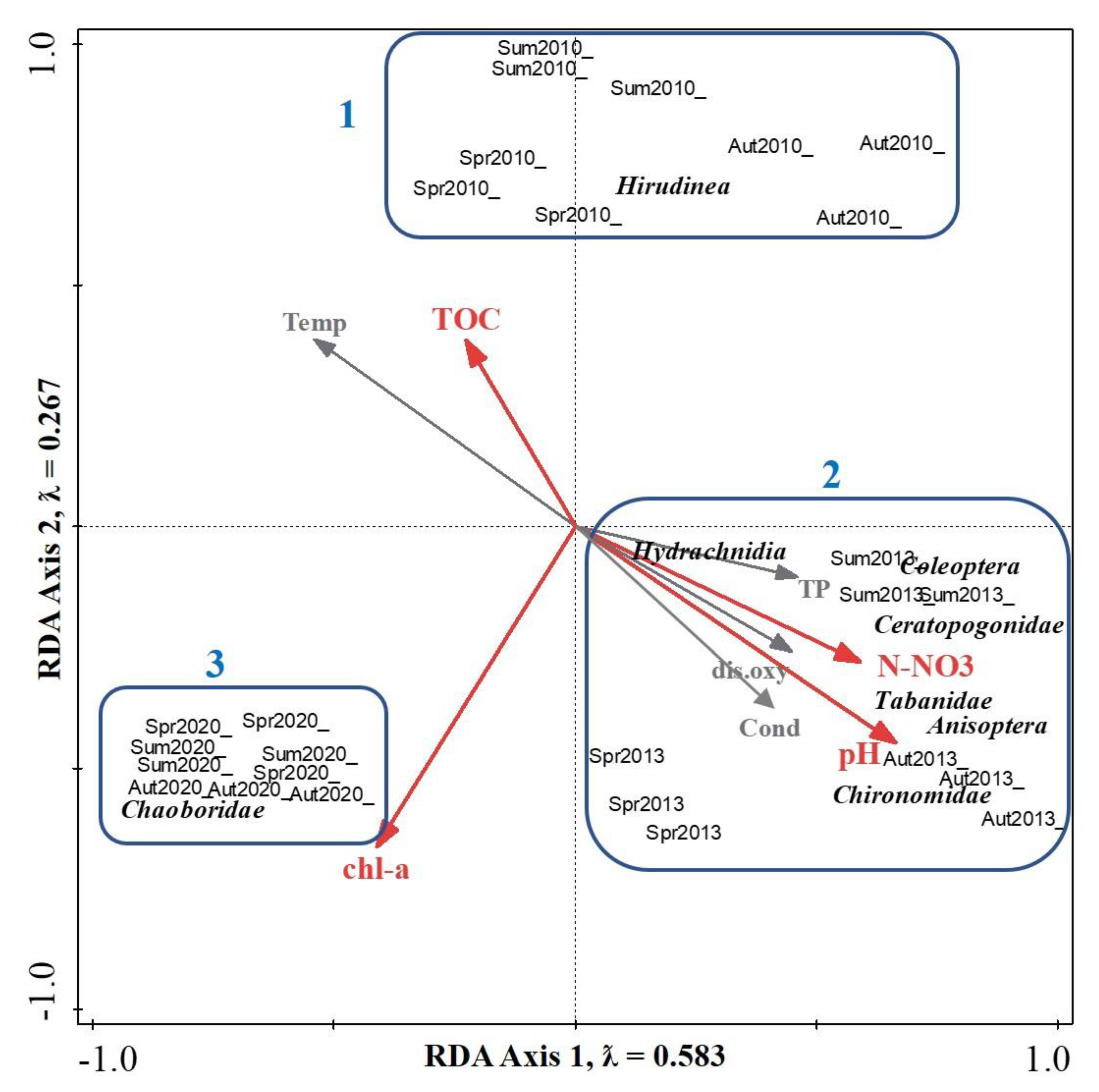
3.3. Ordination Analysis
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Beadle, J.M.; Brown, L.E.; Holden, J. Biodiversity and ecosystem functioning in natural bog pools and those created by rewetting schemes. WIREs Water 2015, 2, 65–84. [Google Scholar] [CrossRef]
- Maitland, P.S. New horizons—new species? The invertebrate fauna of unexplored aquatic habitats in Scotland. Aquat. Conserv. Mar. Freshw. Ecosyst. 1999, 9, 529–534. [Google Scholar] [CrossRef]
- Williams, P.; Whitfield, M.; Biggs, J.; Bray, S.; Fox, G.; Nicolet, P.; Sear, D.A. Comparative biodiversity of rivers, streams, ditches and ponds in an agricultural landscape in Southern England. Biol. Conserv. 2003, 115, 329–341. [Google Scholar] [CrossRef]
- Drinan, T.J.; Foster, G.N.; Nelson, B.H.; O’Halloran, J.; Harrison, S.S.C. Macroinvertebrate assemblages of peatland lakes: Assessment of conservation value with respect to anthropogenic land-cover change. Biol. Conserv. 2013, 158, 175–187. [Google Scholar] [CrossRef]
- Streever, W.J.; Portier, K.M.; Crisman, T.L. A comparison of dipterans from ten created and ten natural wetlands. Wetlands 1996, 16, 416–428. [Google Scholar] [CrossRef]
- Nelson, S.M.; Roline, R.A.; Thullen, J.S.; Sartoris, J.J.; Boutwell, J.E. Invertebrate assemblages and trace element bioaccumulation associated with constructed wetlands. Wetlands 2000, 20, 406–415. [Google Scholar] [CrossRef]
- King, T.S.; Wrubleski, D.A. Spatial and diel availability of flying insects as potential duckling food in prairie wetlands. Wetlands 1998, 18, 100–114. [Google Scholar] [CrossRef]
- Hájková, P.; Bojková, J.; Fránková, M.; Opravilová, V. Disentangling the effects of water chemistry and substratum structure on moss-dwelling unicellular and multicellular microorganisms in spring-fens. J. Limnol. 2011, 70, 54–64. [Google Scholar] [CrossRef][Green Version]
- Cao, Y.; Langdon, P.G.; Yan, Y.; Wang, S.; Zheng, Z.; Zhang, Z. Chironomid communities from subalpine peatlands in subtropical China as indicators of environmental change. J. Paleolimnol. 2019, 62, 165–179. [Google Scholar] [CrossRef]
- Galbrand, C.; Lemieux, I.G.; Ghaly, A.E.; Cote, A.; Verma, M. Assessment of wetland biological integrity using aquatic macroinvertebrates. J. Biol. Sci. 2007, 7, 52–65. [Google Scholar] [CrossRef][Green Version]
- Spieles, D.J.; Mitsch, W.J. Macroinvetrebrate community structure in high- and low- nutrient constructed wetlands. Wetlands 2000, 20, 716–729. [Google Scholar] [CrossRef]
- Stewart, T.W.; Downing, J.A. Macroinvertebrate communities and environmental conditions in recently constructed wetlands. Wetlands 2008, 28, 141–150. [Google Scholar] [CrossRef]
- Hermanowicz, W.; Dojlido, J.; Dożańska, W.; Kosiorowski, B.; Zerze, J. Fizycznochemiczne Badanie Wody i Ścieków; Arkady: Warszawa, Poland, 1999. [Google Scholar]
- Golterman, H.L. Methods for chemical analysis of freshwaters. In IBP Handbook No. 8; Blackwell Scientific Publications: Oxford, UK, 1969. [Google Scholar]
- Wiederholm, T. Chironomidae of the holartic region. Keys and diagnoses. Part 1. Larvae. Entomol. Scand. 1986, 19, 1–457. [Google Scholar]
- Kołodziejczyk, A.; Koperski, P. Bezkręgowce Słodkowodne Polski. Klucz do Oznaczania oraz Podstawy Biologii I Ekologii Makrofauny; Wydawnictwo Uniwersytetu Warszawskiego: Warszawa, Poland, 2000; pp. 1–250. [Google Scholar]
- Mataloni, G.G.; González-Garraza, G.; Vinocur, A. Landscape-driven environmental variability largely determines abiotic characteristics and phytoplankton patterns in peat bog pools (Tierra del Fuego, Argentina). Hydrobiologia 2015, 751, 105–125. [Google Scholar] [CrossRef]
- Küppers, G.C.; González-Garraza, G.C.; Quiroga, M.V.; Lombardo, R.; Marinone, M.C.; Vinocur, A.; Mataloni, G. Drivers of highly diverse planktonic ciliate assemblages in peat bog pools from Tierra del Fuego (Argentina). Hydrobiologia 2016, 773, 117–134. [Google Scholar] [CrossRef]
- Górniak, A.; Jekaterynczuk-Rudczyk, E.; Dobrzy, P. Hydrochemistry of three dystrophic lakes in Northeastern Poland (in Polish). Acta Hydrohichim. Hydrobiol. 1999, 27, 12–18. [Google Scholar] [CrossRef]
- Luoto, T.P. Dystrophy in determining midge community composition in boreal lakes. Écoscience 2013, 20, 391–398. [Google Scholar] [CrossRef]
- Foster, G.N.; Bilton, D.T. The aquatic coleoptera associated with patterned mires. In Patterned Mires and Mire Pools: Origin and Development: Flora and Fauna: Proceedings, University of Durham, 6–7 April 1998; Standen, V., Tallis, J.H., Meade, R., Eds.; British Ecological Society: London, UK, 1998; pp. 65–84. [Google Scholar]
- Fairchild, G.W.; Faulds, A.M.; Matta, J.F. Beetle assemblages in ponds: Effects of habitat and site age. Freshw. Biol. 2000, 44, 523–534. [Google Scholar] [CrossRef]
- Ribera, I.; Foster, G.N.; Holt, W.V. Functional types of diving beetle (Coleoptera: Hygrobiidae and Dytiscidae), as identified by comparative swimming behaviour. Biol. J. Linn. Soc. 1997, 61, 537–558. [Google Scholar] [CrossRef]
- Batzer, D.P.; Wissinger, S.A. Ecology of insect communities in nontidal wetlands. Annu. Rev. Entomol. 1996, 41, 75–100. [Google Scholar] [CrossRef]
- Richardson, W.B.; Ziegler, S.J.; Dewey, M.R. Bioenergetic relations in submerged aquatic vegetation: An experimental test of prey use by juvenile bluegills. Ecol. Freshw. Fish 1998, 7, 1–12. [Google Scholar] [CrossRef]
- Woodcock, T.; Longcore, J.; McAuley, D.; Mingo, T.; Bennatti, C.R.; Stromborg, K. The role of pH in structuring communities of maine wetland macrophytes and chironomid larvae (Diptera). Wetlands 2005, 25, 306–316. [Google Scholar] [CrossRef]
- Rudolph, H.J.; Samland, J. Occurrence and metabolism of sphagnum acid in the cell walls of bryophytes. Phytochemistry 1985, 24, 745–749. [Google Scholar] [CrossRef]
- Jassey, V.E.; Chiapusio, G.; Binet, P.; Buttler, A.; Laggoun, F.; Delarue, F.; Bernard, N.; Mitchell, E.A.; Toussaint, M.; Francez, A.; et al. Above- and belowground linkages in Sphagnum peatland: Climate warming affects plant–microbial interactions. Glob. Change Biol. 2013, 19, 811–823. [Google Scholar] [CrossRef] [PubMed]
- Aerts, R.; Wallen, B.; Malmer, N. Growth-limiting nutrients in Sphagnum-dominated bogs subject to low a high atmospheric nitrogen supply. J. Ecol. 1992, 80, 131–140. [Google Scholar] [CrossRef]
- Limpens, J.; Berendse, F.; Klees, H. N deposition affects N availability in interstitial water, growth of Sphagnum and invasion of vascular plants in bog vegetation. New Phytol. 2003, 157, 339–347. [Google Scholar] [CrossRef]
- van Duinen, G.A.; Vermonden, K.; Brock, A.M.T.; Leuven, R.S.E.W.; Smolders, A.J.P.; van der Velde, G.; Verberk, W.C.E.P.; Esselink, H. Basal food sources for the invertebrate food web in nutrient poor and nutrient enriched raised bog pools. Proc. Neth. Entomol. Soc. Meet. 2006, 17, 37–44. [Google Scholar]
- Taylor, A.N.; Batzer, D.P. Spatial and temporal variation in invertebrate consumer diets in forested and herbaceous wetlands. Hydrobiologia 2010, 651, 145–159. [Google Scholar] [CrossRef]
- Tarkowska-Kukuryk, M. Periphytic algae as food source for grazing chironomids in a shallow phytoplankton dominated lake. Limnologica 2013, 43, 254–264. [Google Scholar] [CrossRef]
- Wissel, B.; Yan, N.D.; Ramcharan, C.W. Predation and refugia: Implications for Chaoborus abundance and species composition. Freshw. Biol. 2003, 48, 1421–1431. [Google Scholar] [CrossRef]
- Jäger, I.S.; Walz, N. Chaoborus flavicans (Diptera) is an oxy-regulator. Arch. Hydrobiol. 2002, 155, 401–411. [Google Scholar] [CrossRef]
- Walag, A.M.P.; Canencia, M.O.P. Physico-chemical parameters and macrobenthic invertebrates of the intertidal zone of Gusa, Cagayan de Oro city, Philippines. Adv. Environ. Sci. 2016, 8, 71–82. [Google Scholar]
- Tokeshi, M. Species abundance patterns and community structure. Adv. Ecol. Res. 1993, 24, 111–186. [Google Scholar]
- Toth, M.; Arva, D.; Sandor, A.N.; Specziar, A. Species diversity and abundance of plant-dwelling chironomids across hierarchical habitat and seasonal scales in the oxbow lakes of River Tisza, Hungary. Fundam. Appl. Limnol. 2013, 182, 309–321. [Google Scholar] [CrossRef]
- Wazbinski, K.E.; Quinlan, R. Midge (Chironomidae, Chaoboridae, Ceratopogonidae) assemblages and their relationship with biological and physicochemical variables in shallow, polymictic lakes. Freshw. Biol. 2013, 58, 2464–2480. [Google Scholar] [CrossRef]
- Tarkowska-Kukuryk, M.; Mieczan, T. Distribution and environmental determinants of chironomids(Diptera: Chironomidae) in Sphagnum microhabitats. Pol. J. Environ. Stud. 2014, 23, 483–490. [Google Scholar]
- Brodersen, K.P.; Quinlan, R. Midges as palaeoindicators of lake productivity eutrophication and hypolimnetic oxygen. Quat. Sci. Rev. 2006, 25, 1995–2012. [Google Scholar] [CrossRef]
- Weller, M.O.; Bossart, J.L. Insect community diversity tracks degradation and recovery of a wastewater assimilation marsh in southeast Louisiana. Wetlands 2017, 37, 1–13. [Google Scholar] [CrossRef]
- Jyväsjärvi, J.; Virtanen, R.; Ilmonen, J.; Paasivirta, L.; Muotka, T. Identifying taxonomic and functional surrogates for spring biodiversity conservation. Conserv. Biol. 2018, 32, 883–893. [Google Scholar] [CrossRef]
| 2010 | 2013 | 2020 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Spr | Sum | Aut | Spr | Sum | Aut | Spr | Sum | Aut | |
| Temp (°C) | 17.8 ± 3.4 | 24.8 ± 5.3 | 8.2 ± 0.8 | 20.2 ± 2.5 | 21.4 ± 0.1 | 14.8 ± 0.2 | 20.3 ± 0.9 | 26.7 ± 0.5 | 18.8 ± 0.5 |
| pH | 4.7 ± 0.1 | 3.8 ± 1.2 | 5.3 ± 0.9 | 4.2 ± 0.5 | 5.2 ± 0.3 | 4.9 ± 0.4 | 4.2 ± 0.1 | 4.5 ± 0.3 | 4.3 ± 0.2 |
| Cond (µS cm−1) | 43 ± 9 | 50 ± 5 | 62 ± 2 | 45 ± 3 | 49 ± 2 | 54 ± 9 | 35 ± 13 | 56 ± 3 | 44 ± 9 |
| Dis.oxy (mg dm−3) | 5.0 ± 2.5 | 5.4 ± 1.9 | 6.8 ± 1.1 | 6.2 ± 1.9 | 6.7 ± 1.8 | 5.4 ± 2.3 | 6.0 ± 1.6 | 5.2 ± 2.6 | 5.9 ± 2.5 |
| N-NO3 (mg dm−3) | 0.234 ± 0.09 | 0.734 ± 0.24 | 1.276 ± 0.21 | 0.540 ± 0.04 | 0.5904 ± 0.07 | 0.670 ± 0.09 | 0.019 ± 0.02 | 0.013 ± 0.01 | 0.024 ± 0.05 |
| P-PO4 (mg dm−3) | 0.031 ± 0.014 | 0.182 ± 0.01 | 0.017 ± 0.01 | 0.077 ± 0.04 | 0.074 ± 0.48 | 0.058 ± 0.48 | 0.017 ± 0.04 | 0.105 ± 0.001 | 0.014 ± 0.002 |
| TP (mg dm−3) | 0.129 ± 0.031 | 0.547 ± 0.026 | 0.497 ± 0.123 | 0.098 ± 0.154 | 0.103 ± 0.163 | 0.521 ± 0.162 | 0.046 ± 0.011 | 0.429 ± 0.041 | 0.039 ± 0.009 |
| TOC (mg dm−3) | 51.5 ± 9.1 | 63.3 ± 11.2 | 60.3 ± 6.4 | 48.5 ± 8.5 | 30.4 ± 2.3 | 10.6 ± 0.4 | 27.3 ± 1.5 | 55.7 ± 3.4 | 41.9 ± 2.2 |
| Chl-a (µg dm−3) | 4.8 ± 1.3 | 26.8 ± 1.9 | 69.7 ± 10.4 | 6.1 ± 71.8 | 6.6 ± 0.5 | 16.3 ± 1.3 | 37.8 ± 4.6 | 74.6 ± 7.1 | 101.8 ± 17.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tarkowska-Kukuryk, M. Environmental Drivers of Macroinvertebrate Assemblages within Peat Pool Habitat-Implication for Bioassessment. Water 2021, 13, 2369. https://doi.org/10.3390/w13172369
Tarkowska-Kukuryk M. Environmental Drivers of Macroinvertebrate Assemblages within Peat Pool Habitat-Implication for Bioassessment. Water. 2021; 13(17):2369. https://doi.org/10.3390/w13172369
Chicago/Turabian StyleTarkowska-Kukuryk, Monika. 2021. "Environmental Drivers of Macroinvertebrate Assemblages within Peat Pool Habitat-Implication for Bioassessment" Water 13, no. 17: 2369. https://doi.org/10.3390/w13172369
APA StyleTarkowska-Kukuryk, M. (2021). Environmental Drivers of Macroinvertebrate Assemblages within Peat Pool Habitat-Implication for Bioassessment. Water, 13(17), 2369. https://doi.org/10.3390/w13172369





