Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. MINLAKE Overview
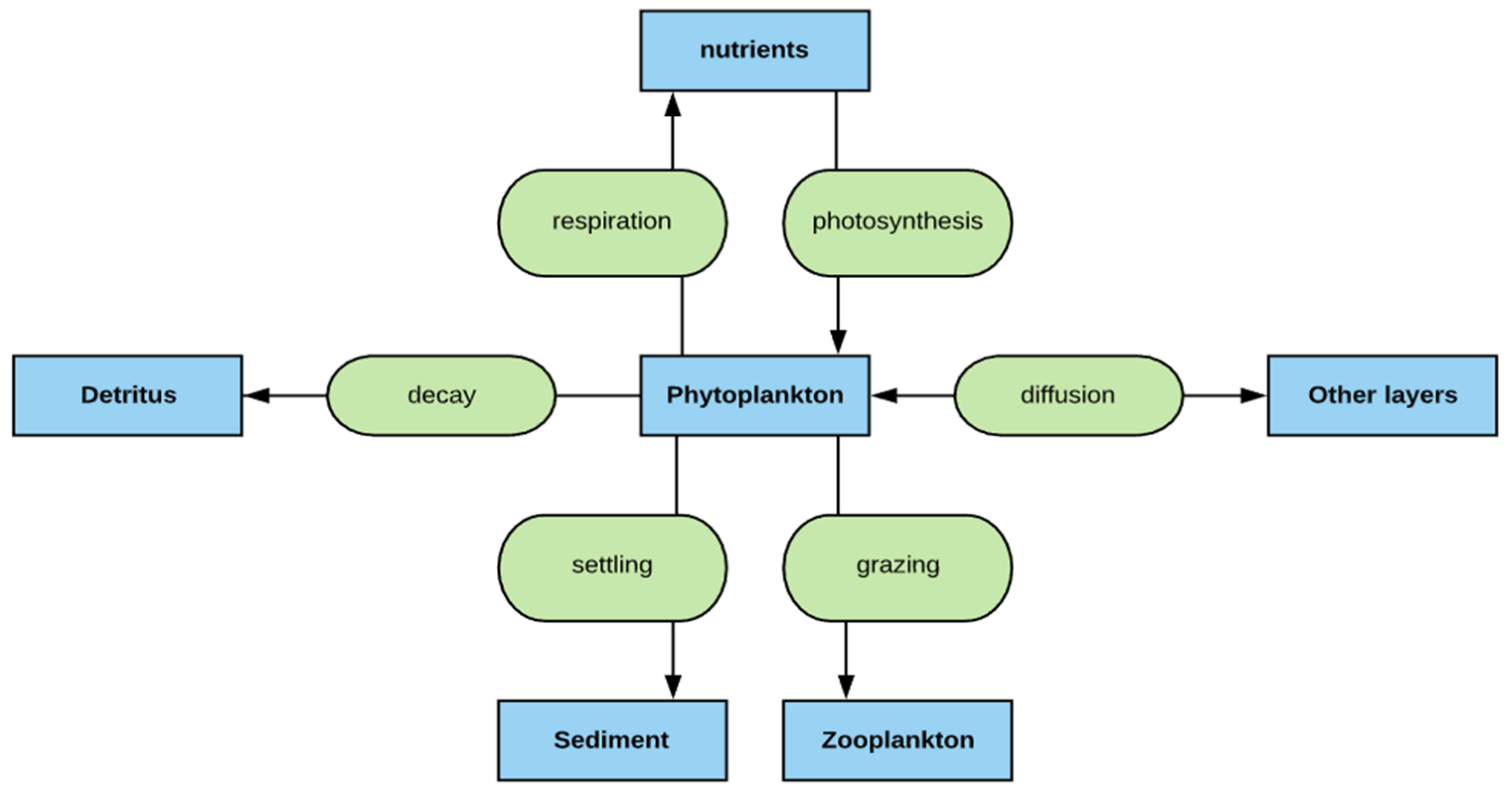
2.2. Phytoplankton Simulation

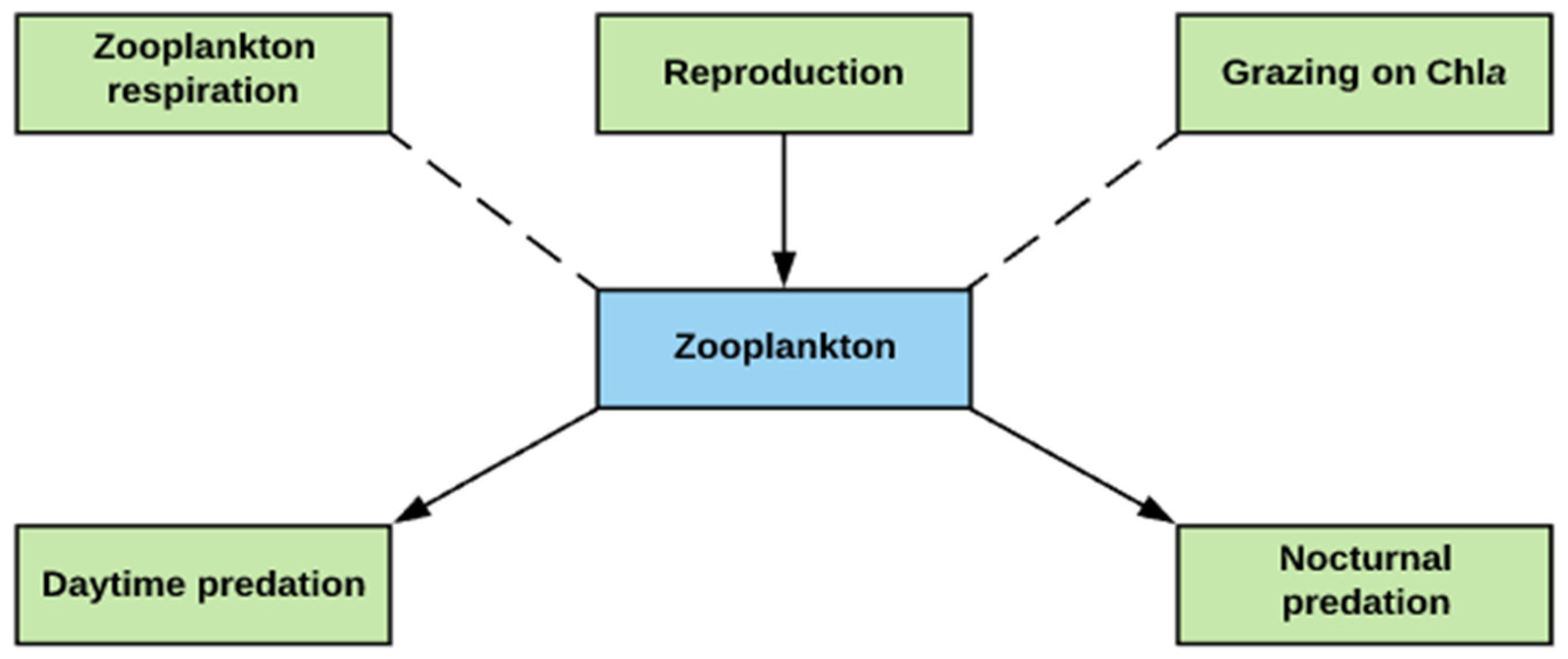
2.3. Zooplankton Simulation
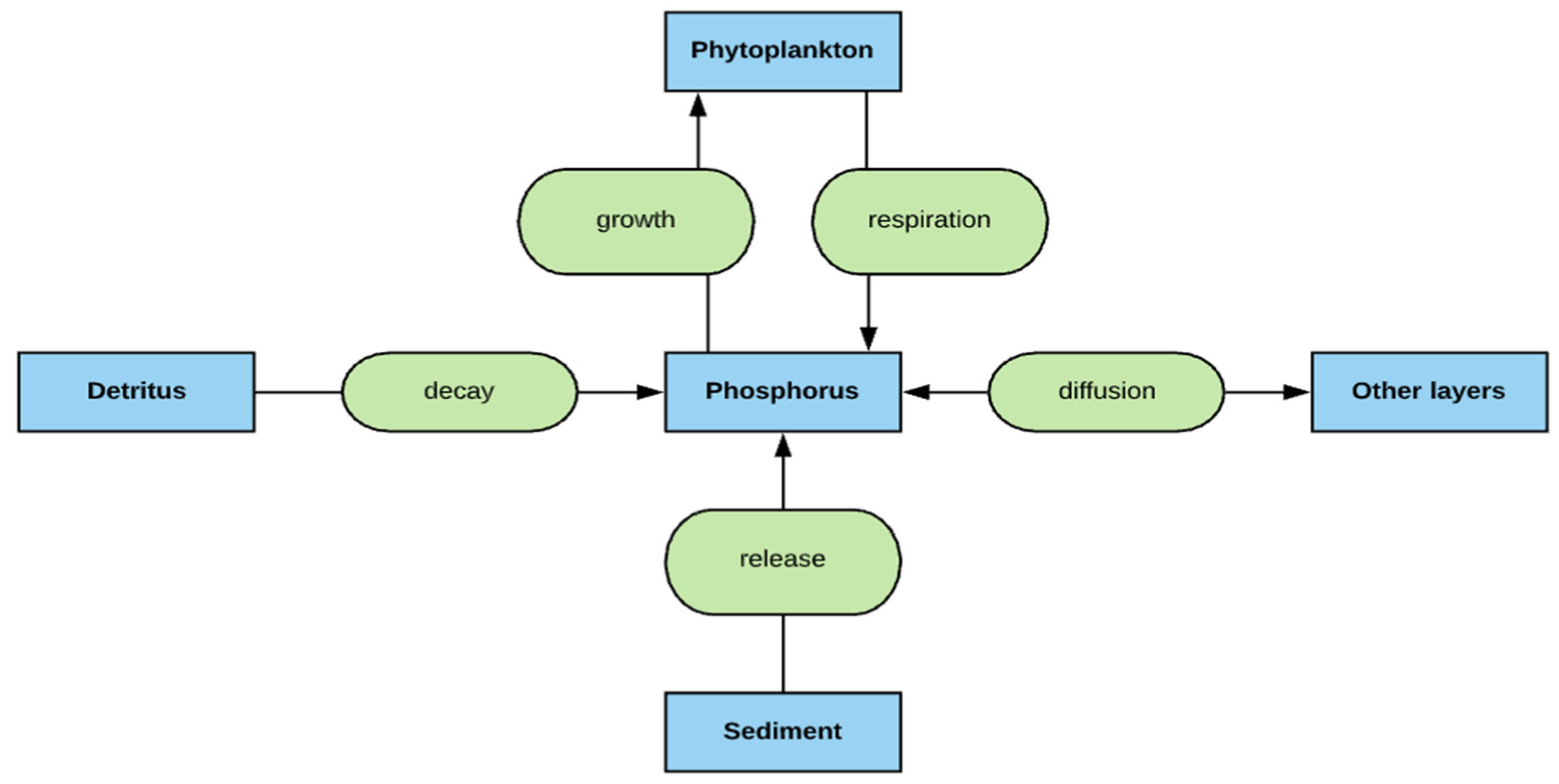
2.4. Phosphorus Simulation

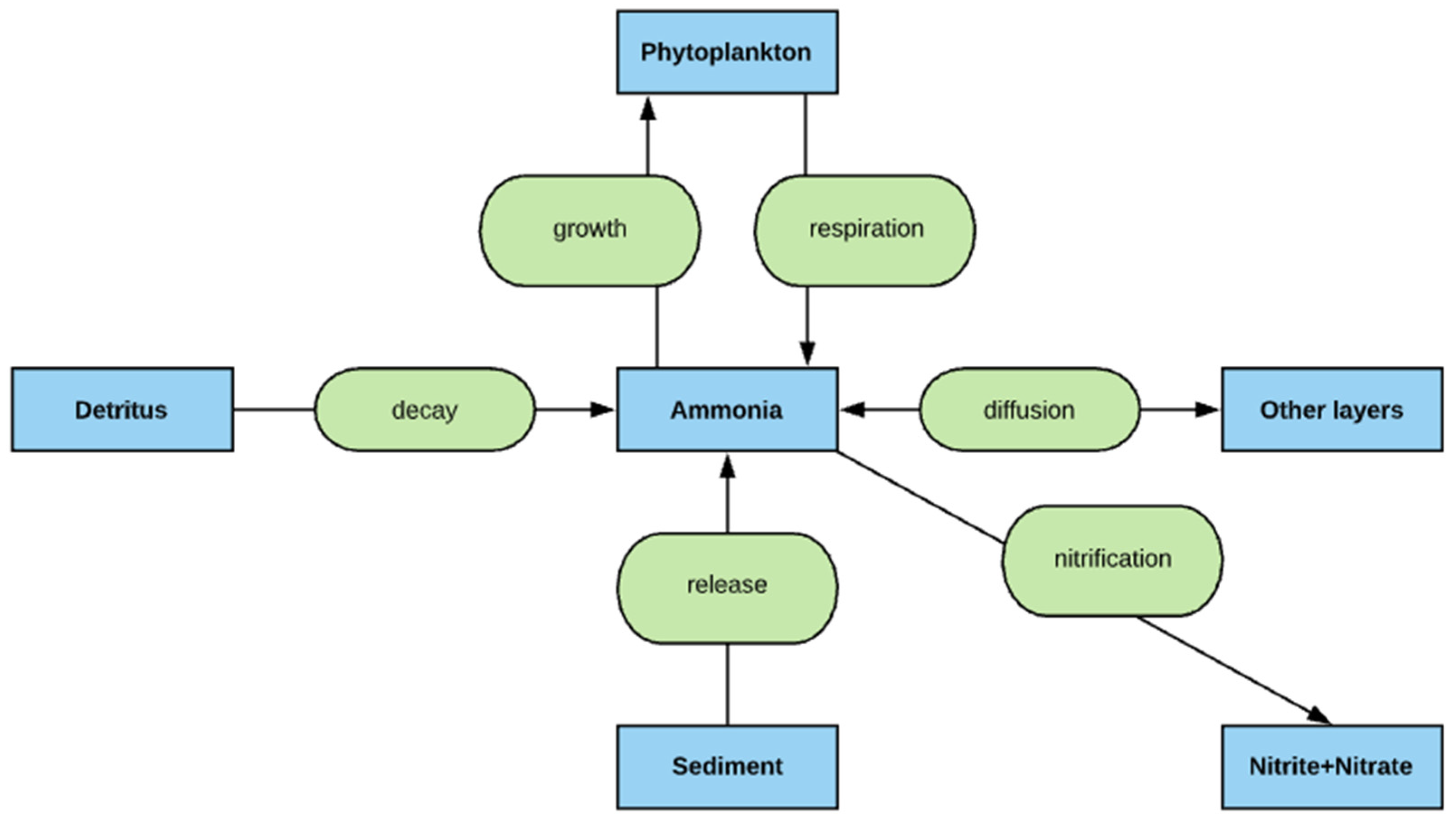
2.5. Nitrogen Simulation


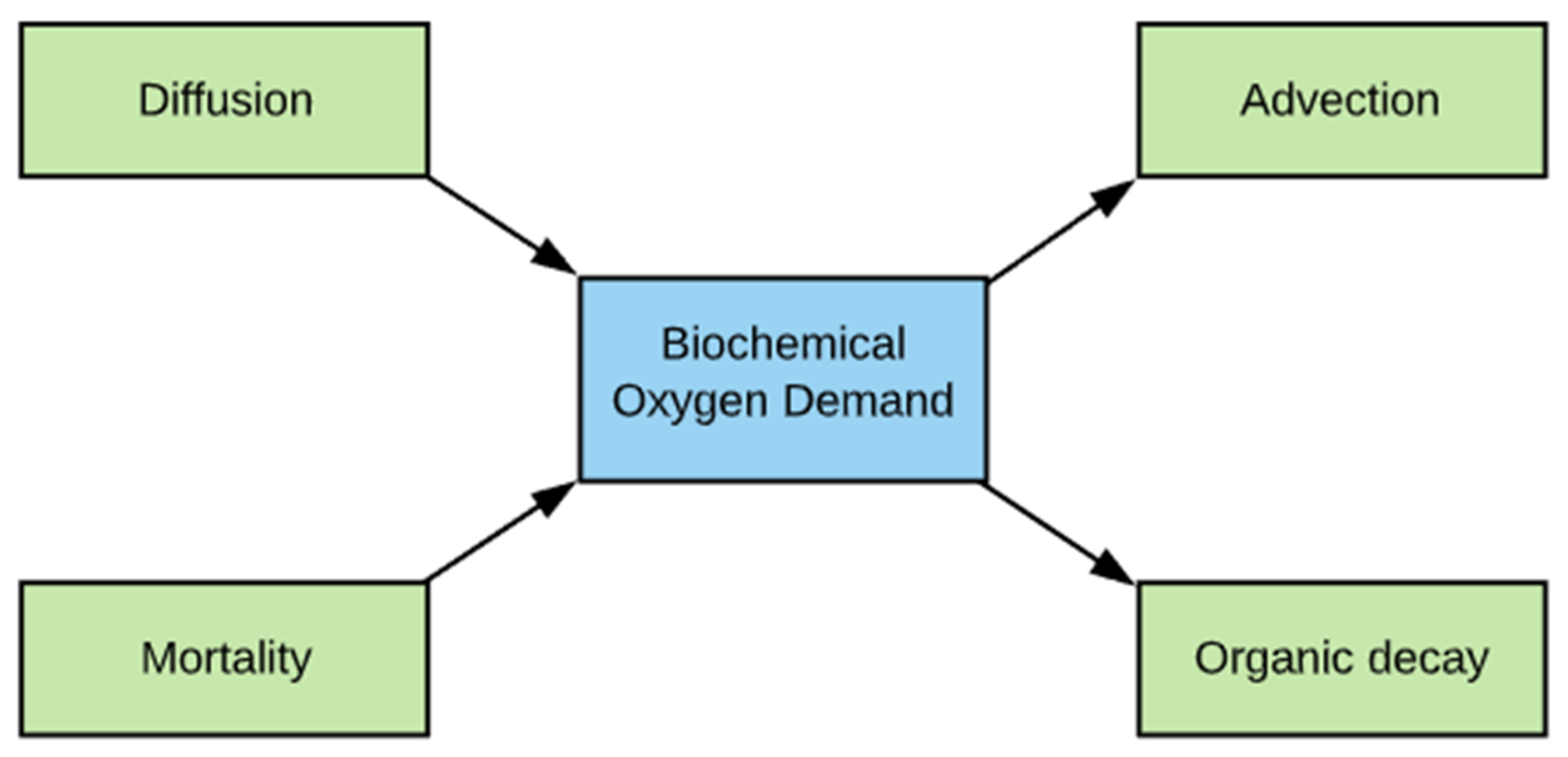
2.6. BOD Simulation
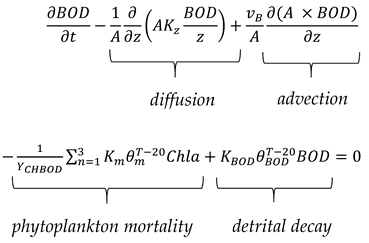
2.7. DO Simulation

2.8. Interaction and Connection among Modeling Variables
2.9. Model Coefficients and Parameters
3. Lakes Simulated
4. Result and Discussion
4.1. Model Calibration
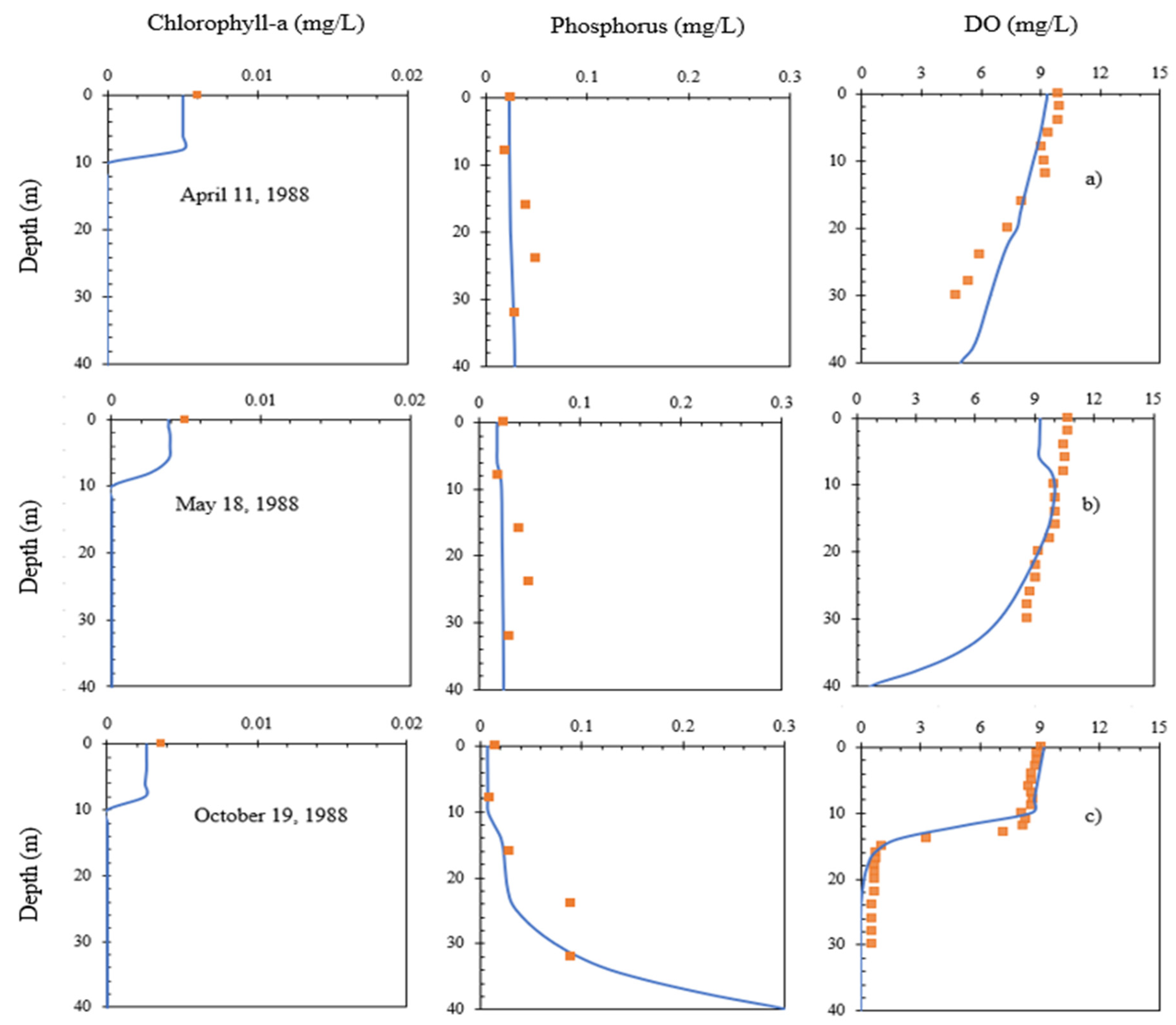
4.2. Chla and Phosphorus Profiles
4.3. Chlorophyll-a and Phosphorus Interaction
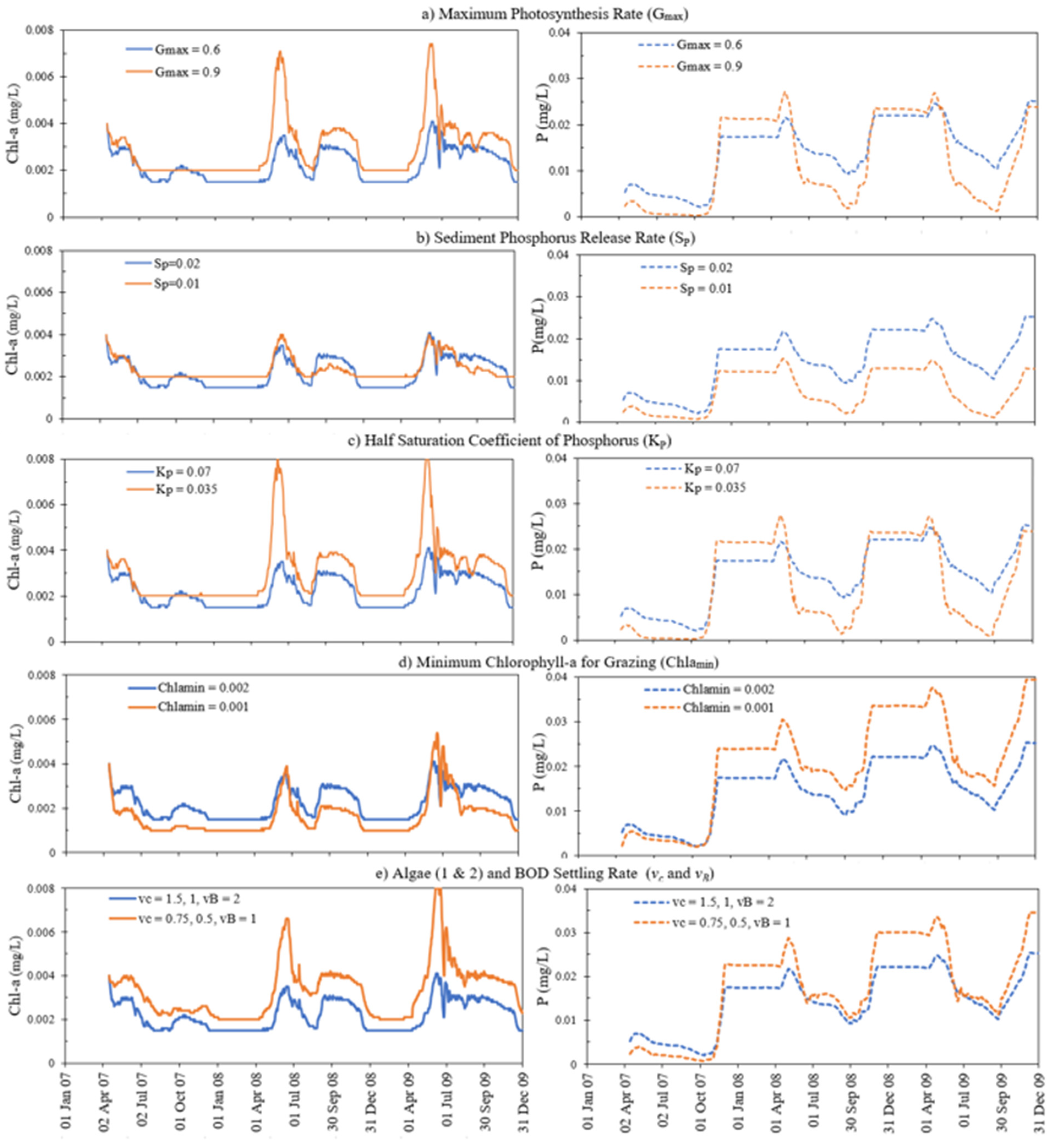
4.4. Sensitivity Analysis
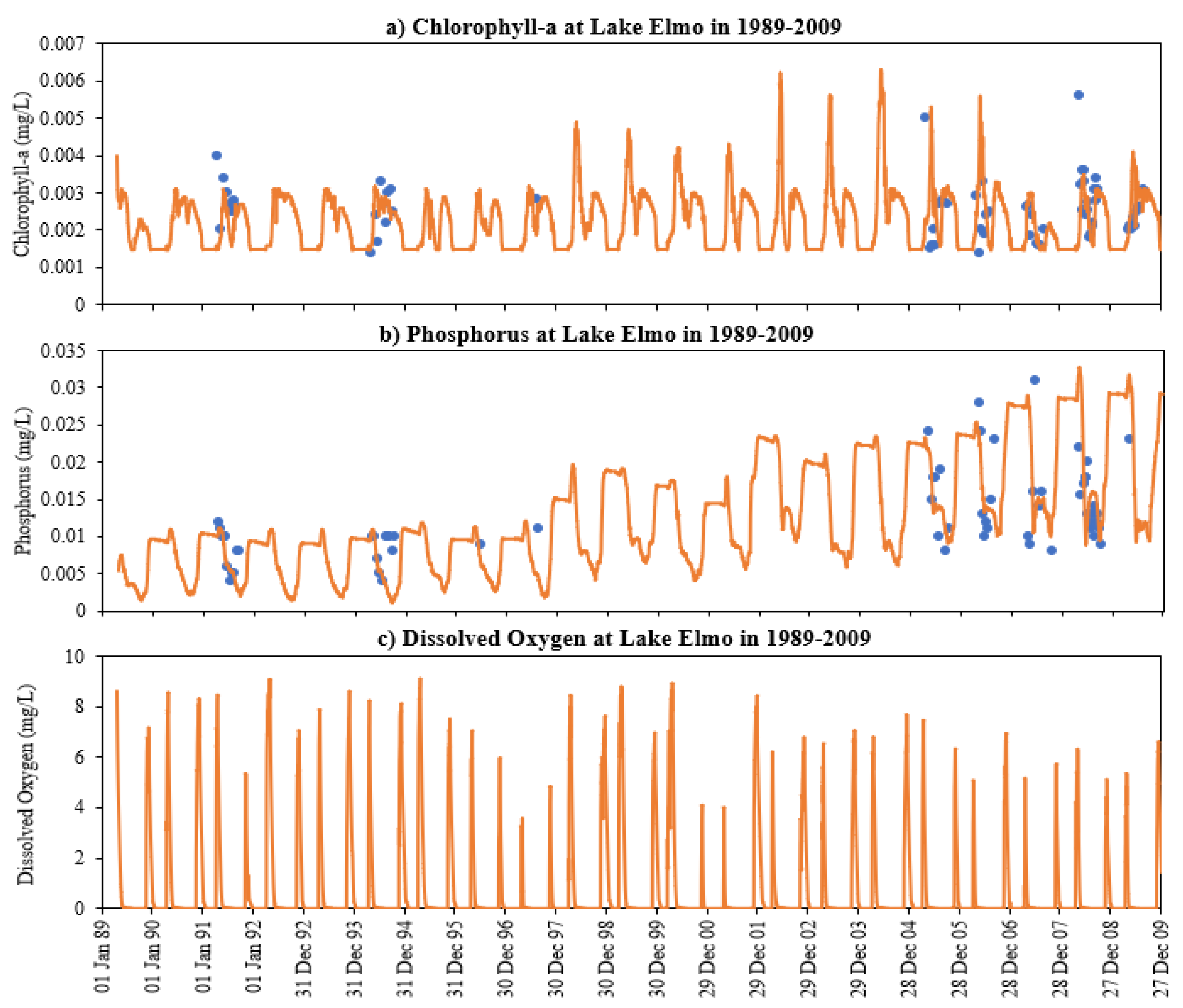
4.5. Long-Term Simulations Using MINLAKE2020
5. Conclusions
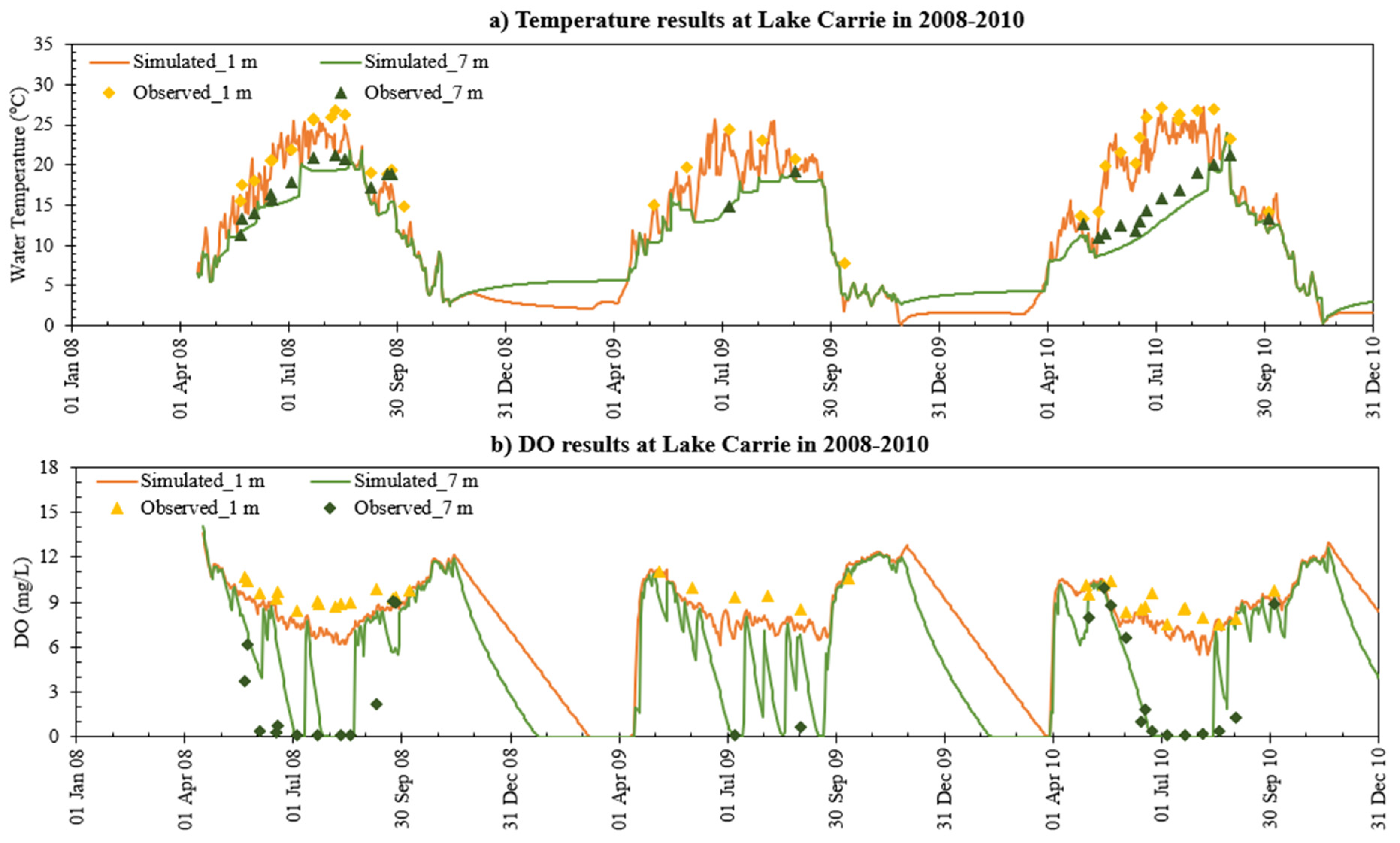
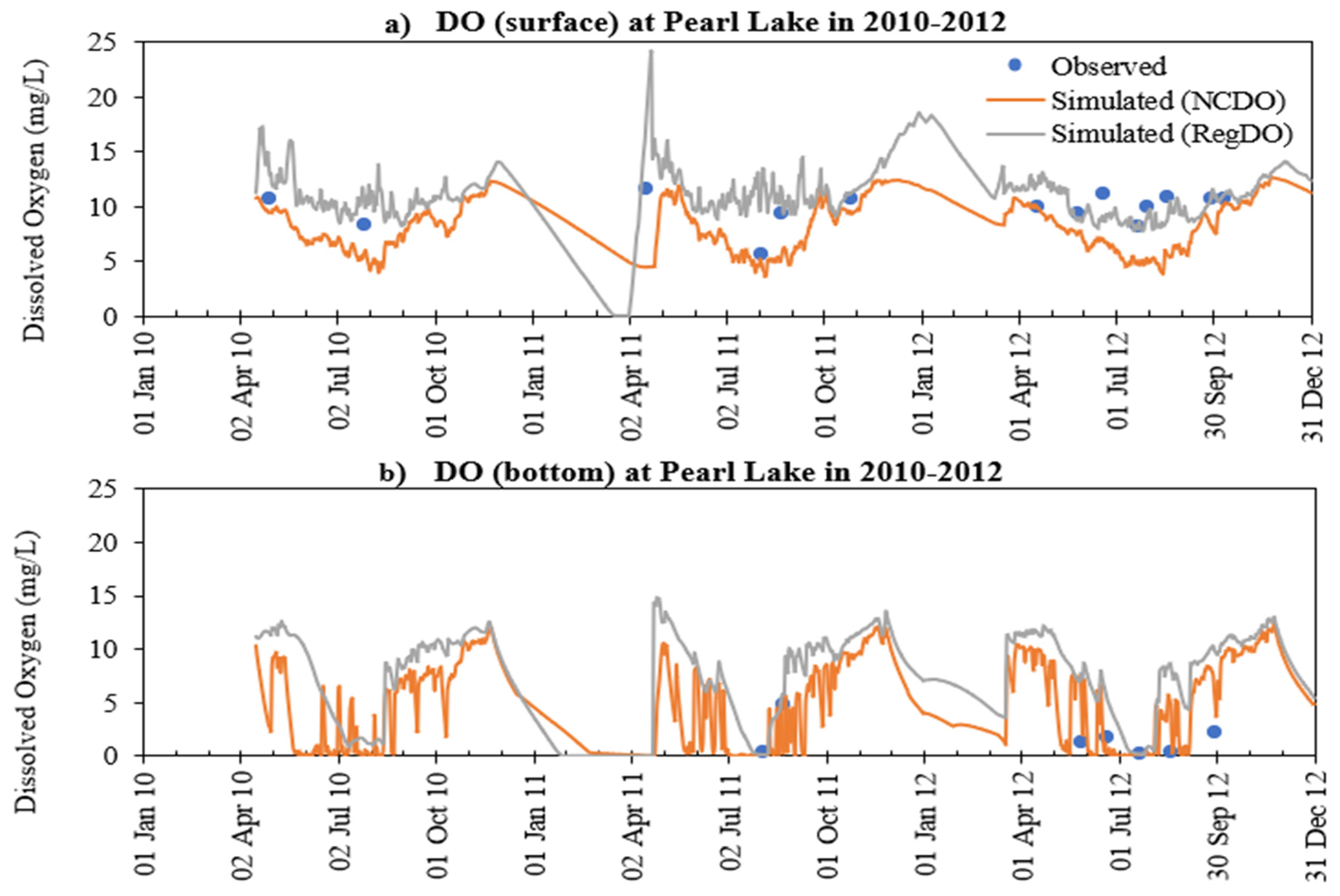
- MINLAKE2020 was calibrated against measured profiles in six Minnesota lakes (Table 4) for the short term (2–4 years) with an average standard error of 1.51 °C for temperature and 2.33 mg/L for DO. The average standard error for DO simulation of these lakes decreased by 24.2% from the original MINLAKE2012 model, which indicates better model performance. DO results reflect/integrate reasonably simulated phosphorus, Chla, and BOD results at different layers (see Figure 10, Figure 11, Figure 12, Figure 13 and Figure 14 and Figure 17).
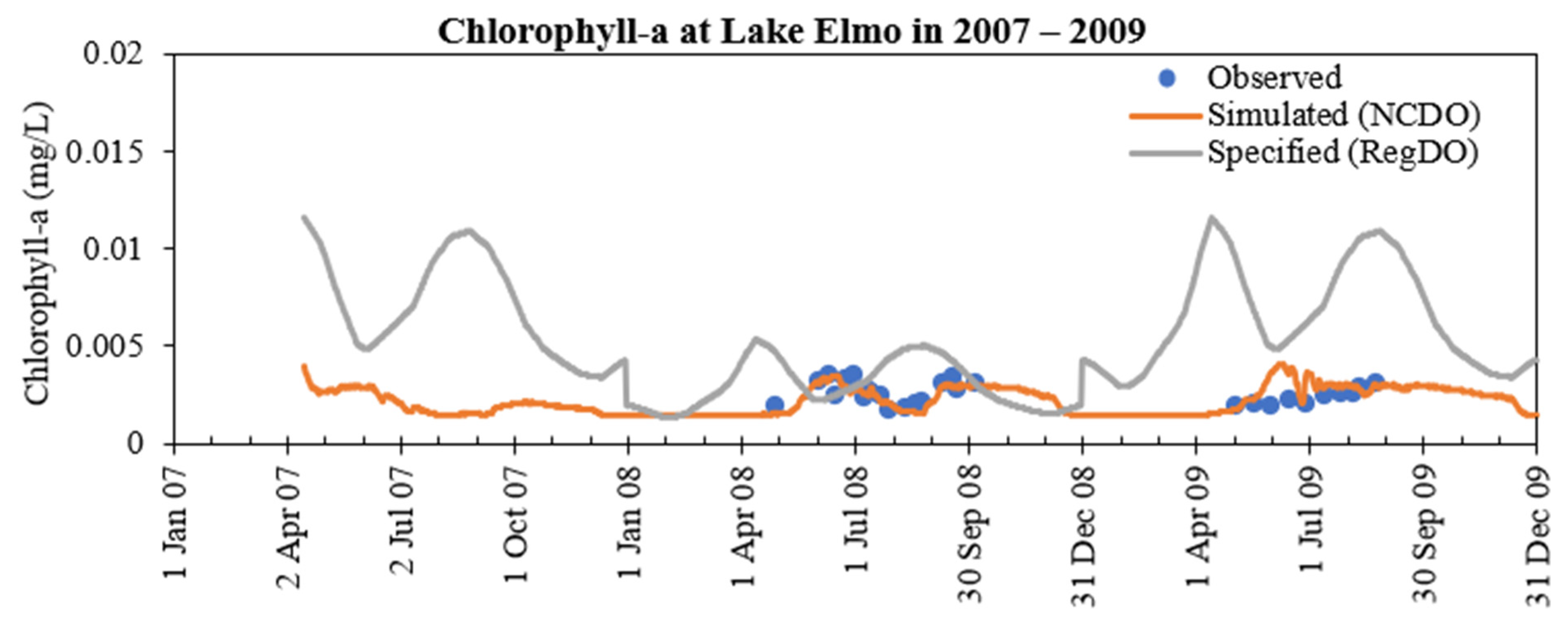
- The addition of phosphorus and Chla simulation in MINLAKE2020 improved model performance in comparison to MINLAKE2012 where Chla was specified input. It greatly affects the DO concentration in some lakes such as Pearl Lake (Figure 10). Thrush Lake and Carlos Lake also showed significant improvement in DO simulation with MINLAKE2020. The standard error decreased by 2.12 mg/L and 1.76 mg/L for Thrush and Carlos Lake, respectively.
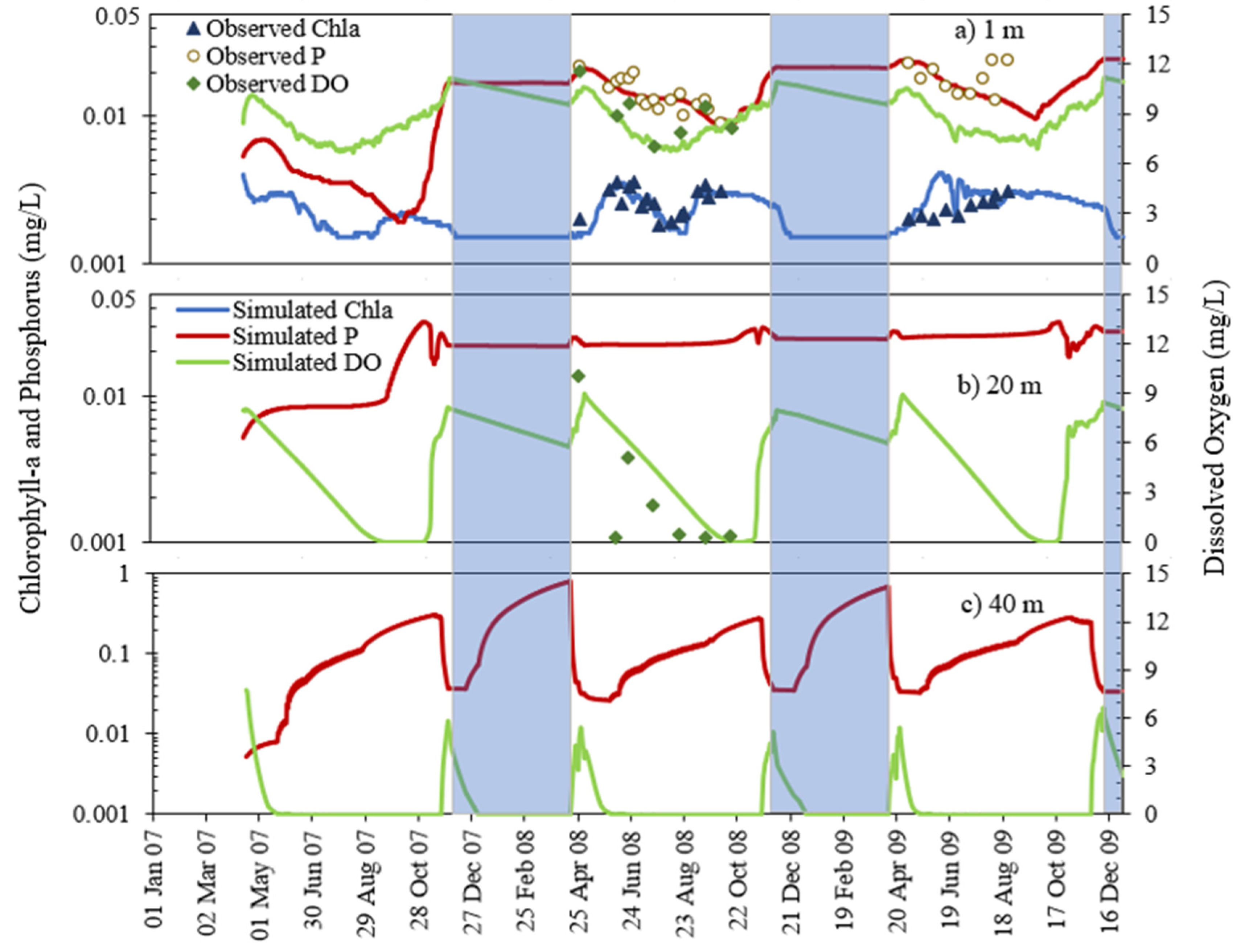
- The deep lakes exhibit a certain trend for phosphorus and Chla simulation year by year whereas the shallow lakes might show a significant change in phosphorus and Chla concentration year by year due to two overturn periods (complete mixing) and the complex interactions/connections among phosphorus, Chla, and DO (Figure 13 and Figure 14), which are evident through governing Equations (9) and (15)–(19) and processes simulated (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7).
- DO concentration is a primary control of internal loading via anoxic release of phosphorus from the lake sediment. MINLAKE2020 was applied to Lake Elmo for a 20-year (1989 to 2009) continuous simulation with a single set of calibration parameters with regression coefficients of 0.99 and 0.94 for temperature and DO profiles, respectively. An increasing trend of surface phosphorus was simulated from 1997 to 2009 which matches well with the observed condition and is directly related to sediment phosphorus release. The average yearly sediment phosphorus release increased from 151.8 kg during the period 1990–1996 to 244.1 kg during the period 1997–2009. This increase is caused by the average 25-day increase in the anoxic condition at the bottom depth (41 m) and the increase in the anoxic horizontal area (as a result of anoxia at lower depths) in later years.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Symbols, Corresponding Description and Units
| Symbols | Description and Units |
| A | Area (m2) |
| BOD | Biochemical oxygen demand (mg/L) |
| Chla | Chlorophyll concentration (mg/L) |
| Chlamin | Minimum chlorophyll concentration for grazing to occur (mg/L) |
| DO | Dissolved Oxygen concentration (mg/L) |
| DOsat | Saturated oxygen concentration (mg/L) |
| f(L) | Light limiting growth factor (between 0 and 1) |
| f(S) | Michalis-Menten growth limiting factor [-] |
| f(T) | Temperature function for growth [-] |
| Gmax | Maximum growth rate (d−1) |
| GRmax | Grazing maximum (mg Chla/ind. zooplankton/d) |
| Kgchla | Half-saturation constant for grazing (mg/L) |
| I(z) | Intensity of photosynthetically active radiation (µE/m2/h) |
| K1 | Light limitation coefficient (µE/m2/h) |
| K2 | Light inhibition coefficient (µE/m2/h) |
| KBOD | Organic decomposition rate (d−1) |
| Ke | Surface oxygen gas exchange (transfer) coefficient (m/d) |
| Km | Mortality rate (d−1) |
| KN | Half-saturation constant for nitrogen (mg/L) |
| KNI | Nitrification rate (d−1) |
| KNH(n) | Half-saturation constant for preferential uptake of ammonia over nitrate [-] |
| KTN(n) | Half-saturation constant of nitrogen for each algal class (mg/L) |
| KP | Half-saturation constant for phosphorus (mg/L) |
| Kr | Respiration rate (d−1) |
| Kzr | Zooplankton respiration rate (d−1) |
| Kz | Eddy diffusivity (cm2/s) |
| N | Nitrogen concentration |
| NH4 | Ammonia concentration (mg N/L) |
| NO3 | Nitrate concentration (mg N/L) |
| NO2 | Nitrite concentration (mg N/L) |
| NO2-3 | Nitrite + Nitrate concentration (mg N/L) |
| n | Phytoplankton species [-] |
| P | SRP or soluble reactive phosphorus concentration (mg/L) |
| θBOD | Temperature adjustment coefficient for BOD [-] |
| θSOD | Temperature adjustment coefficient for SOD [-] |
| θm | Temperature adjustment coefficient for mortality [-] |
| θr | Temperature adjustment coefficient for respiration [-] |
| θzr | Temperature adjustment coefficient for zooplankton grazing [-] |
| θNH | Temperature adjustment coefficient for ammonia [-] |
| θNO | Temperature adjustment coefficient for nitrate [-] |
| θNI | Temperature adjustment coefficient for nitrification [-] |
| RAD | Solar radiation (cal/cm2/d) |
| Sb | Sedimentary oxygen demand rate at 20°C (g O/m2/d) |
| SNH | Ammonia release rate from sediment (g N/m2/d) |
| SNO | Nitrite release rate from sediment (g N/m2/d) |
| SP | Phosphorus release rate from sediment (g P/m2/d) |
| t | Time (d) |
| T | Air temperature (°C) |
| TD | Length of day light (h) |
| Tmax | Maximum temperature at which phytoplankton growth is reduced 90 percent (°C) |
| Tmin | Minimum temperature at which phytoplankton growth is reduced 90 percent (°C) |
| Topt | Optimal temperature at which maximum phytoplankton growth occurs (°C) |
| TP | Total phosphorus (mg/L) |
| vc | Phytoplankton settling velocity (m/d) |
| vB | Detritus settling velocity (m/d) |
| Viz | Volume of day depth (m3) |
| Vr | Volume day depth/layer ratio [-] |
| YCHO2 | Mass yield ratio of chlorophyll to oxygen |
| YNChla | Mass yield ratio of Nitrogen to Chlorophyll-a |
| YNHBOD | Mass yield ratio of ammonia to BOD [-] |
| YPBOD | Mass yield ratio of ammonia to BOD [-] |
| YPCHLA | Mass yield ratio of ammonia to BOD [-] |
| z | Depth (m) |
| ZP | Zooplankton concentration (#/m3) |
| ZPmin | Minimum concentration of zooplankton for predation to occur (#/m3) |
| PDd | Daytime predation rate (d−1) |
| Pd | Daily predation rate (d−1) |
| XI | Light intensity at the day depth (µE/m2/s) |
| XImax | Light intensity at which no predation occurs (µE/m2/s) |
| XImin | Light intensity at which no predation occurs (µE/m2/s) |
| Pmax | Maximum predation rate (d−1) |
| DY | Julian day of last day of minimum predation rate [-] |
| DYmin | Julian day of last day of minimum predation rate [-] |
| DYmax | Julian day of beginning of maximum predation rate [-] |
| PD(t,I) | Nocturnal predation rate in layer I (#/m3) |
| PDn | Nocturnal predation rate (d−1) |
| ΔTg(I) | Time that zooplankton spent in a layer I during the night (d) |
References
- Le Moal, M.; Gascuel-Odoux, C.; Menesguen, A.; Souchon, Y.; Etrillard, C.; Levain, A.; Moatar, F.; Pannard, A.; Souchu, P.; Lefebvre, A.; et al. Eutrophication: A new wine in an old bottle? Sci. Total Environ. 2019, 651, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moss, B. Cogs in the endless machine: Lakes, climate change and nutrient cycles: A review. Sci. Total Environ. 2011, 434, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Takolander, A.; Cabeza, M.; Leskinen, E. Climate change can cause complex responses in Baltic Sea macroalgae: A systematic review. J. Sea Res. 2017, 123, 16–29. [Google Scholar] [CrossRef] [Green Version]
- Yao, X.; Zhang, Y.; Zhang, L.; Zhou, Y. A bibliometric review of nitrogen research in eutrophic lakes and reservoirs. J. Environ. Sci. 2017, 66, 274–285. [Google Scholar] [CrossRef]
- Liu, Y.; Villalba, G.; Ayres, R.U.; Schroder, H. Global phosphorus flows and environmental impacts from a consumption perspective. J. Ind. Ecol. 2008, 12, 229–247. [Google Scholar] [CrossRef]
- Turner, R.E. Linking landscape and water quality in the Mississippi river basin for 200 years. Bioscience 2003, 53, 563–572. [Google Scholar] [CrossRef]
- Wolfe, A.H.; Patz, J.A. Reactive nitrogen and human health: Acute and longterm implications. AMBIO J. Hum. Environ. 2002, 31, 120–125. [Google Scholar] [CrossRef]
- Townsend, A.R.; Howarth, R.W.; Bazzaz, F.A.; Booth, M.S.; Cleveland, C.C.; Collinge, S.K.; Dobson, A.P.; Epstein, P.R.; Holland, E.A.; Keeney, D.R.; et al. Human health effects of a changing global nitrogen cycle. Front. Ecol. Environ. 2003, 1, 240–246. [Google Scholar] [CrossRef]
- Hudnell, H.K.; Dortch, Q. Cyanobacterial Harmful Algal Blooms: Chapter 2: A Synopsis of Research Needs Identified at the Interagency, International Symposium on Cyanobacterial Harmful Algal Blooms (ISOC-HAB); US Environmental Protection Agency: Washington, DC, USA, 2008; Volume 38, pp. 17–43.
- Hudnell, H.K. The state of U.S. freshwater harmful algal blooms assessments, policy and legislation. Toxicon 2010, 55, 1024–1034. [Google Scholar] [CrossRef] [PubMed]
- Seewer, J. Ohio wants to put Lake Erie on a new, strict pollution diet. AP News, 15 February 2020. [Google Scholar]
- Moomaw, W.R.; Birch, M.B. Cascading costs: An economic nitrogen cycle. Sci. China Ser. C Life Sci. 2005, 48, 678–696. [Google Scholar]
- Pretty, J.N.; Mason, C.F.; Nedwell, D.B.; Hine, R.E.; Leaf, S.; Dils, R. Environmental Costs of Freshwater Eutrophication in England and Wales. Environ. Sci. Technol. Libr. 2003, 37, 201–208. [Google Scholar] [CrossRef] [Green Version]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of U.S. freshwaters: Analysis of potential economic damages. Environ. Sci. Technol. 2008, 43, 12–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartram, J.; Chorus, I. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; World Health Organization: London, UK, 1999; p. 400. [Google Scholar]
- Imboden, D.M. Phosphorus model of lake eutrophication. Limnol. Oceanogr. 1974, 19, 297–304. [Google Scholar] [CrossRef] [Green Version]
- Vollenweider, R.A.; Kerekes, J. Eutrophication of Waters. Monitoring, Assessment and Control; OECD Cooperative Programme on Monitoring of Inland Waters; Organization for Economic Co-Operation and Development (OECD): Paris, France, 1982; Volume 156. [Google Scholar]
- De Senerpont Domis, L.N.; Van de Waal, D.B.; Helmsing, N.R.; Donk, E.V.; Mooij, W.M. Community stoichiometry in a changing world: Combined effects of warming and eutrophication on phytoplankton dynamics. Ecology 2014, 95, 1485–1495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reynolds, C.S.; Irish, A.E.; Elliott, J.A. The ecological basis for simulating phytoplankton responses to environmental change (PROTECH). Ecol. Model. 2001, 140, 271–291. [Google Scholar] [CrossRef]
- Riley, M.J.; Stefan, H.G. MINLAKE: A dynamic lake water quality simulation model. Ecol. Model. 1988, 43, 155–182. [Google Scholar] [CrossRef]
- Gu, R.; Stefan, H.G. Year-round temperature simulation of cold climate lakes. Cold Reg. Sci. Technol. 1990, 18, 147–160. [Google Scholar] [CrossRef]
- Fang, X.; Stefan, H.G. Modeling of Dissolved Oxygen Stratification Dynamics in Minnesota Lakes under Different Climate Scenarios; St Anthony Falls Hydraulic Laboratory, University of Minnesota: Minneapolis, MN, USA, 1994; Volume 55414, p. 260. [Google Scholar]
- West, D.; Stefan, H.G. Simulation of Lake Water Quality Using a One-Dimensional Model with Watershed Input. In Model Description and Application to Lake Riley and Lake Elmo; Project Report No.430; St. Anthony Falls Laboratory, University of Minnesota: Minneapolis, MN, USA, 1998. [Google Scholar]
- Jamily, J.A. Developing an Hourly Water Quality Model to Simulate Diurnal Water Temperature and Dissolved Oxygen Variations in Shallow Lakes. Master’s Thesis, Auburn University, Auburn, AL, USA, 2018. [Google Scholar]
- Batick, B.M. Modeling Temperature and Dissolved Oxygen in the Cheatham Reservoir with CE-QUAL-W2; Vanderbilt University: Nashville, TN, USA, 2011. [Google Scholar]
- Chapra, S.C.; Martin, J.L. LAKE2K, a Modeling Framework for Simulating Lake Water Quality (Version 1.2): Documentation and users Manual; Civil and Environmental Engineering Department, Tufts University: Medford, MA, USA, 2004. [Google Scholar]
- Cole, T.M.; Buchak, E.M. CE-QUAL-W2: A Two-Dimensional, Laterally Averaged, Hydrodynamic and Water Quality Model, Version 2.0. User Manual; DTIC Document; DTIC: Fort Belvoir, VA, USA, 1995. [Google Scholar]
- Janssen, A.B.G.; Teurlincx, S.; Beusen, A.H.W.; Huijbregts, M.A.J.; Rost, J.; Schipper, A.M.; Seelen, L.M.S.; Mooij, W.M.; Janse, J.H. PCLake+: A process-based ecological model to assess the trophic state of stratified and non-stratified freshwater lakes worldwide. Ecol. Model. 2019, 396, 23–32. [Google Scholar] [CrossRef]
- Hipsey, M.R.; Romero, J.R.; Antenucci, J.P.; Hamilton, D. Computational Aquatic Ecosystem Dynamics Model: CAEDYM v2; Contract Research Group, Centre for Water Research, University of Western Australia: Crawley, WA, Australia, 2005; p. 102. [Google Scholar]
- Hannoun, I.; List, E.J.; Kavanagh, K.B.; Chiang, W.-L.; Ding, L.; Preston, A.; Karafa, D.; Rachkley, I. Use of ELCOM and CAEDYM for Water Quality Simulation in Boulder Basin; Water Environment Federation: Alexandria, VA, USA, 2006. [Google Scholar]
- Carraro, E.; Guyennon, N.; Hamilton, D.; Valsecchi, L.; Manfredi, E.C.; Viviano, G.; Salerno, F.; Tratari, G.; Copetti, D. Coupling high-resolution measurements to a three dimensional lake model to assess the spatial and temporal dynamics of the cyanobacterium Planktothrix rubescens in a medium-sized lake. Hydrobiologia 2012, 698, 77–95. [Google Scholar] [CrossRef]
- Pomati, F.; Matthews, B.; Seehausen, O.; Ibelings, B.W. Eutrophication and climate warming alter spatial (depth) co-occurrence patterns of lake phytoplankton assemblages. Hydrobiologia 2017, 787, 375–385. [Google Scholar] [CrossRef]
- Anderson, D.M.; Cembella, A.D.; Hallegraeff, G.M. Progress in Understanding Harmful Algal Blooms: Paradigm shifts and new technologies for research, monitoring, and management. Annu. Rev. Mar. Sci. 2012, 4, 143–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shimoda, Y.; Arhonditsis, G.B. Phytoplankton functional type modelling: Running before we can walk? A critical evaluation of the current state of knowledge. Ecol. Model. 2016, 320, 29–43. [Google Scholar] [CrossRef]
- Zhang, X.; Recknagel, F.; Chen, Q.; Cao, H.; Li, R. Spatially-explicit modelling and forecasting of cyanobacteria growth in Lake Taihu by evolutionary computation. Ecol. Model. 2015, 306, 216–225. [Google Scholar] [CrossRef]
- Elliott, A.H. Predicting the impact of changing nutrient load and temperature on the phytoplankton of England’s largest lake, Windermere. Freshw. Biol. 2011, 57, 400–413. [Google Scholar] [CrossRef] [Green Version]
- Marsden, M.W. Lake restoration by reducing external phosphorus loading: The influence of sediment phosphorus release. Freshw. Biol. 1989, 21, 139–162. [Google Scholar] [CrossRef]
- Sondergaard, M.; Jeppesen, E.; Jensen, J.P.; Amsinck, S.L. Water framework directive: Ecological classification of Danish lakes. J. Appl. Ecol. 2005, 42, 616–629. [Google Scholar] [CrossRef]
- Philips, G.; Kelly, A.; Pitt, J.A.; Sanderson, R.; Taylor, E. The recovery of a very shallow eutrophic lake, 20 years after the control of effluent derived phosphorus. Freshw. Biol. 2005, 50, 1628–1638. [Google Scholar] [CrossRef]
- Taguchi, V.J.; Olsen, T.A.; Janke, B.D.; Gulliver, J.S.; Finlay, J.C.; Stefan, H.G. Internal loading in stormwater ponds as a phosphorus source to downstream waters. Limnol. Oceanogr. Lett. 2020, 5, 322–330. [Google Scholar] [CrossRef]
- Jeppesen, E.; Sondergaard, M.; Jensen, J.P.; Havens, K.E.; Anneville, O.; Carvalho, L.; Coveney, M.F.; Deneke, R.; Dokulil, M.T.; Foy, B.; et al. Lake responses to reduced nutrient loading: An analysis of contemporary long-term data from 35 case studies. Freshw. Biol. 2005, 50, 1747–1771. [Google Scholar] [CrossRef]
- Welch, E.B.; Cooke, G.D. Internal phosphorus loading in shallow lakes: Importance and control. Lake Reserv. Manag. 2009, 21, 209–217. [Google Scholar] [CrossRef] [Green Version]
- Riley, M.J.; Stefan, H.G. Dynamic Lake Water Quality Simulation Model “MINLAKE”; St. Anthony Falls Hydraulic Laboratory, University of Minnesota: Minneapolis, MN, USA, 1987; Volume 55414, p. 140. [Google Scholar]
- Hondzo, M.; Stefan, H.G. Lake water temperature simulation model. J. Hydraul. Eng. 1993, 119, 1251–1273. [Google Scholar] [CrossRef]
- Fang, X.; Stefan, H.G. Modeling of dissolved oxygen stratification dynamics in Minnesota lakes under different climate scenarios. Ecol. Model. 1994, 71, 37–68. [Google Scholar]
- Fang, X.; Stefan, H.G. Temperature and Dissolved Oxygen Simulations for a Lake with Ice Cover; Project Report 356; St. Anthony Falls Hydraulic Laboratory, University of Minnesota: Minneapolis, MN, USA, 1994. [Google Scholar]
- Fang, X.; Alam, S.R.; Jacobson, P.; Pereira, D.; Stefan, H.G. Simulations of Water Quality in Cisco Lakes in Minnesota; St. Anthony Falls Laboratory, University of Minnesota: Minneapolis, MN, USA, 2010. [Google Scholar]
- West, D.; Stefan, H.G. Simulation of Water Quality and Primary Productivity Control Strategies for Lake McCarrons; Project Report No.426; St. Anthony Falls Laboratory, University of Minnesota: Minneapolis, MN, USA, 2000. [Google Scholar]
- Tasnim, B.; Jamily, J.A.; Fang, X.; Zhou, Y.; Hayworth, J.S. Simulating diurnal variations of water temperature and dissolved oxygen in shallow Minnesota lakes. Water 2021, 13, 1980. [Google Scholar] [CrossRef]
- Monod, J. The growth of bacterial cultures. Annu. Rev. Microbiol. 1949, 3, 371–394. [Google Scholar] [CrossRef] [Green Version]
- Lehman, J.T.; Botkins, D.B.; Likens, G.E. The assumptions and rationales of a computer model of phytoplankton population dynamics. Limnol. Oceanogr. 1975, 20, 343–364. [Google Scholar] [CrossRef]
- Megard, R.O.; Tonkyn, D.W.; Senft, W.H. Kinetics of oxygenic photosynthesis in planktonic algae. J. Plankton Res. 1984, 6, 325–337. [Google Scholar] [CrossRef]
- Megard, R.O.; Combs, W.S.; Smith, P.D.; Knoll, A.S. Attenuation of light and daily integral rates of photosynthesis attained by planktonic algae1. Limnol. Oceanogr. 1979, 24, 1038–1050. [Google Scholar] [CrossRef]
- Shapiro, J.; Forsberg, B.; Lamarra, V.; Lindmark, G.; Lynch, M.; Smeltzer, E.; Zoto, G. Experiments and Experiences in Biomanipulation; Interim Report No. 19; Limnological Research Center, University of Minnesota: Minneapolis, MN, USA, 1982. [Google Scholar]
- Wright, D.; O’Brien, W.J.; Vinyard, G.L. Adaptive value of vertical migration: A simulation model argument for the predation hypothesis, Evolution and Ecology of Zooplankton Communities. In Evolution and Ecology of Zooplankton Communities; Kerfoot, C.W., Ed.; University Press of New England: Lebanon, NH, USA, 1980; pp. 138–147. [Google Scholar]
- Dillon, P.J.; Rigler, F.H. The phosphorus-chlorophyll relationship in lakes: Phosphorus-chlorophyll relationship. Limnol. Oceanogr. 1974, 19, 767–773. [Google Scholar] [CrossRef]
- Thomann, R.V.; Mueller, J.A. Principles of Surface Water Quality Modeling and Control; Harper Collins Publishers Inc: New York, NY, USA, 1987; p. 644. [Google Scholar]
- Goldman, C.R.; Horne, A.J. Limnology; McGraw-Hill: New York, NY, USA, 1983. [Google Scholar]
- Cole, T.M.; Wells, S.A. CE-QUAL-W2: A Two-Dimensional, Laterally Averaged, Hydrodynamic and Water Quality Model, Version 3.72 User Manual; Department of Civil and Environmental Engineering, Portland State University: Portland, OR, USA, 2015. [Google Scholar]
- Hamrick, M.J. The Environmental Fluid Dynamics Code Theory and Computation Volume 3: Water Quality Module; Tetra Tech, Inc.: Fairfax, VA, USA, 2007; p. 90. [Google Scholar]
- Nürnberg, G.K. The prediction of internal phosphorus load in lakes with anoxic hypolimnia. Limnol. Oceanogr. 1984, 29, 111–124. [Google Scholar] [CrossRef]
- Stigebrandt, A.; Rahm, L.; Viktorsson, L.; Ödalen, M.; Hall, P.O.J.; Liljebladh, B. A new phosphorus paradigm for the Baltic proper. Ambio 2014, 43, 634–643. [Google Scholar] [CrossRef] [Green Version]
- Stigebrandt, A.; Andersson, A. The eutrophication of the Baltic Sea has been boosted and perpetuated by a major internal phosphorus source. Front. Mar. Sci. 2020, 7, 572994. [Google Scholar] [CrossRef]
- Jiang, L.; Fang, X. Simulations and validation of cisco lethal conditions in Minnesota lakes under past and future climate scenarios using constant survival limits. Water 2016, 8, 279. [Google Scholar] [CrossRef] [Green Version]
- Edwards, R.; Owens, M. The oxygen balance of streams. Ecol. Ind. Soc. 1965, 6, 149–172. [Google Scholar]
- Henderson-Sellers, B. Engineering Limnology; Pitman Advanced Pub. Program: Boston, MA, USA, 1984; p. 356. [Google Scholar]
- Tasnim, B. Enhancement and Redevelopment of the Regional Lake Water Quality Model with Applications. Master’s Thesis, Auburn University, Auburn, AL, USA, 2020. [Google Scholar]
- Fang, X.; Stefan, H.G. Long-term lake water temperature and ice cover simulations/measurements. Cold Reg. Sci. Technol. 1996, 24, 289–304. [Google Scholar] [CrossRef]
- Brown, L.C.; Barnwell, T.O. The Enhanced Stream Water Quality Models QUAL2E and QUAL2W-UNCAS: Documentation and User Manual; U.S. Environmental Protection Agency: Athens, GA, USA, 1987.
- Zison, S.W.; Mills, W.B.; Diemer, D.; Chen, C.W. Rates, Constants and Kinetic Formulations in Surface Water Quality Modeling; Tetra Tech, Inc., for USEPA, ORD: Athens, GA, USA, 1978; p. 317. [Google Scholar]
- Stefan, H.G.; Hondzo, M.; Fang, X.; Eaton, J.G.; McCormick, J.H. Simulated long-term temperature and dissolved oxygen characteristics of lakes in the north-central United States and associated fish habitat limits. Limnol. Oceanogr. 1996, 41, 1124–1135. [Google Scholar] [CrossRef]
- Stefan, H.G.; Fang, X. Dissolved oxygen model for regional lake analysis. Ecol. Model. 1994, 71, 37–68. [Google Scholar] [CrossRef]
- NAS and NAE. Water Quality Criteria 1972—A Report of the Committee on Water Quality Criteria; Environmental Protection Agency: Washington, DC, USA, 1973.
- Barr Engineering. Pearl Lake and Mill Creek Bacterial Total Maximum Daily Load Report; Barr Engineering: Minneapolis, MN, USA, 2012. [Google Scholar]
- Engel, L.; Valley, R.; Beck, D.; Anderson, J. Sentinel Lake Assessment Report Lake Carlos (21-0057) Douglas County, Minnesota; Minnesota Pollution Control Agency & MN Minnesota Department of Natural Resources: St. Paul, MN, USA, 2010.
- Engel, L.; Heiskary, S.; Valley, R.; Tollefson, D. Sentinel Lake Assessment Report Carrie Lake (34-0032) Kandiyohi County, Minnesota; Minnesota Pollution Control Agency & Minnesota Department of Natural Resources: St. Paul, MN, USA, 2012.
- Nash, J.E.; Sutcliffe, J.V. River flow forecasting through conceptual models part I—A discussion of principles. J. Hydrol. 1970, 10, 282–290. [Google Scholar] [CrossRef]




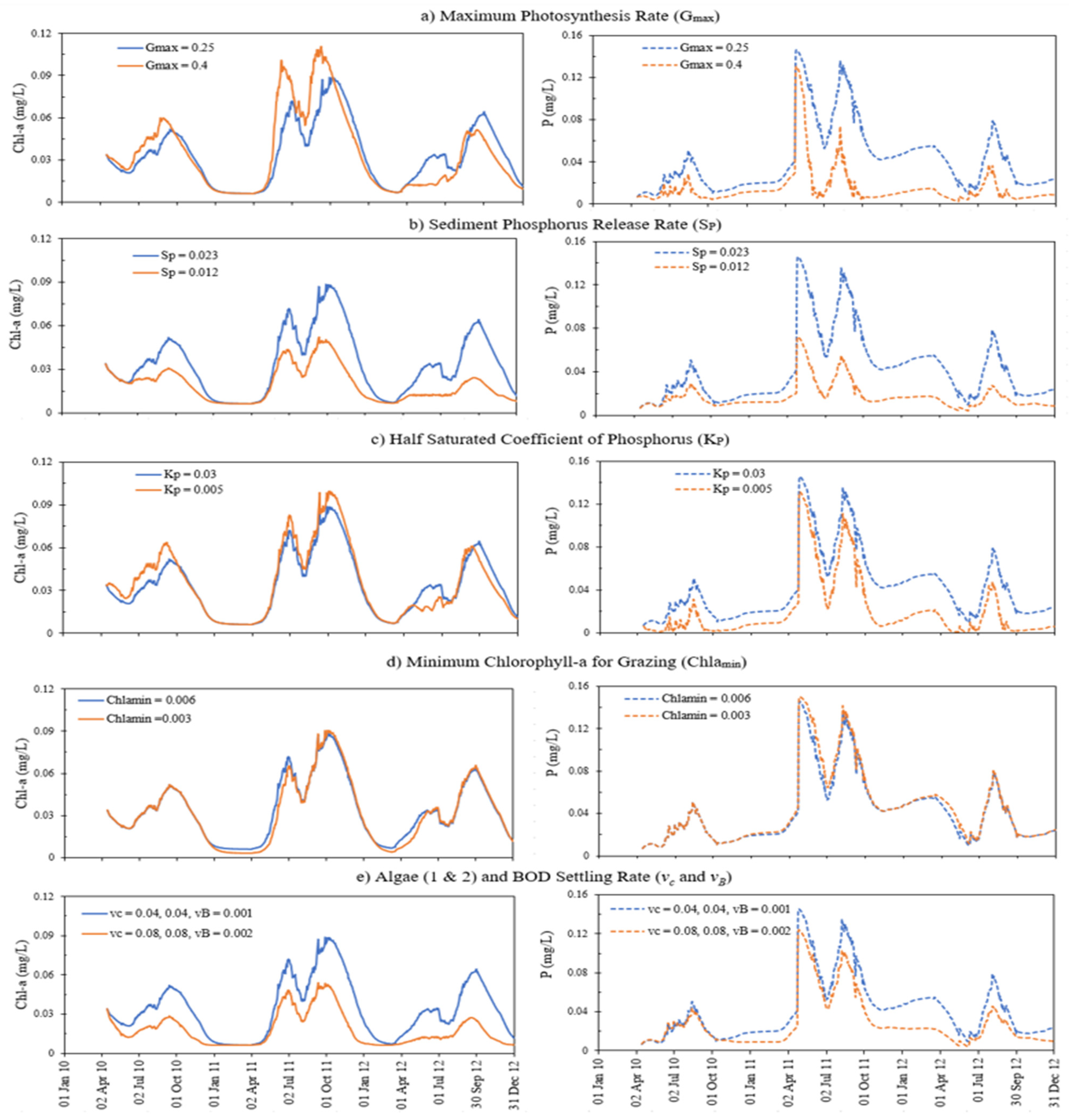
| Parameters | Unit | Effect on Model Results | Description of the Parameter |
|---|---|---|---|
| KBOD | d−1 | P, BOD, and DO profiles | Detrital decay rate |
| Sb | g/m2/d | DO profiles | SOD coefficient at 20 °C |
| EMCOE (2) | DO profiles | Multiplier for SOD below the euphotic zone | |
| Kr1 | d−1 | Chla, P, BOD, DO profiles | Respiration rate of algae |
| Gmax1 | d−1 | Chla, P, DO profiles | Maximum growth rate of algae |
| Km1 | d−1 | Chla and BOD profiles | Non-predatory mortality rate of algae |
| KP1 | m/d | Chla, P and DO profiles | Half saturation coefficient for phosphorus |
| SP | g/m2/d | P and DO profiles | Sediment phosphorus release rate |
| vc1 | m/d | Chla and DO profiles | Settling velocity of algae |
| vB | m/d | Chla and DO profiles | Settling velocity of detritus |
| Topt1 | °C | Chla, P and DO profiles | Optimum temperature for growth of algae |
| Tmax1 | °C | Chla, P and DO profiles | Maximum temperature for growth of algae |
| Tmin1 | °C | Chla, P and DO profiles | Minimum temperature for growth of algae |
| ZP | m−3 | Chla and DO profiles | Zooplankton population |
| ZPmin | m−3 | Chla and DO profiles | Minimum zooplankton population for predation |
| Chlamin1 | mg Chla | Chla and P profiles | Minimum chlorophyll-a for grazing |
| Lake | Surface Area, As, (km2) | Max. Depth Hmax, (m) | Geometry Ratio (m)0.5 | Mean Chla (μg/L) | Trophic Status | Simulation Years | Number of Profile Days (Data Points) |
|---|---|---|---|---|---|---|---|
| Pearl | 3.05 | 5.55 | 7.53 | 16.91 | Eutrophic | 2010–2012 | 15 (134) |
| Carrie | 0.37 | 7.90 | 3.12 | 6.71 | Mesotrophic | 2007–2010 | 36 (342) |
| Riley | 1.19 | 14.9 | 2.22 | 24.00 | Eutrophic | 1985–1987 | 16 (148) |
| Thrush | 0.048 | 14.63 | 1.01 | 1.71 | Oligotrophic | 2008–2015 | 18 (100) |
| Elmo | 1.039 | 42.63 | 0.75 | 4.45 | Mesotrophic | 1989–2009 | 70 (864) |
| Carlos | 10.54 | 50.00 | 1.15 | 3.84 | Oligotrophic | 2008–2015 | 54 (308) |
| Lake Name | NCDO Model (MINLAKE2020) | |||||
|---|---|---|---|---|---|---|
| Water Temperature | Dissolved Oxygen | |||||
| RMSE a (°C) | NSE b | R2 c | RMSE (mg/L) | NSE | R2 | |
| Elmo Lake | 0.98 | 0.98 | 0.99 | 2.02 | 0.70 | 0.92 |
| Carlos Lake | 1.66 | 0.83 | 0.97 | 2.39 | 0.61 | 0.90 |
| Riley Lake | 1.50 | 0.50 | 0.98 | 1.79 | 0.80 | 0.93 |
| Thrush Lake | 1.88 | 0.70 | 0.95 | 2.43 | 0.40 | 0.92 |
| Carrie Lake | 1.75 | 0.68 | 0.99 | 1.95 | 0.70 | 0.93 |
| Pearl Lake | 1.30 | 0.87 | 0.97 | 3.42 | −0.12 | 0.87 |
| Average ± STD d | 1.51 ± 0.33 | 0.76 ± 0.17 | 0.97 ± 0.01 | 2.33 ± 0.59 | 0.52 ± 0.34 | 0.91 ± 0.02 |
| Lake Name | RegDO Model (MINLAKE2012) | |||||
| Elmo Lake | 1.03 | 0.98 | 0.99 | 1.89 | 0.70 | 0.92 |
| Carlos Lake | 1.52 | 0.85 | 0.98 | 4.15 | −0.19 | 0.85 |
| Riley Lake | 1.55 | 0.5 | 0.98 | 2.61 | 0.55 | 0.91 |
| Thrush Lake | 2.01 | 0.69 | 0.95 | 2.91 | 0.1 | 0.92 |
| Carrie Lake | 1.76 | 0.64 | 0.99 | 1.98 | 0.69 | 0.94 |
| Pearl Lake | 1.04 | 0.97 | 0.98 | 3.30 | 0.01 | 0.89 |
| Average ± STD | 1.49 ± 0.39 | 0.77 ± 0.19 | 0.98 ± 0.02 | 3.08 ± 1.11 | 0.11 ± 0.69 | 0.90 ± 0.03 |
| Sensitive Parameters | Description of Parameter | Uncalibrated Value | Calibrated Value | Regression Coefficient |
|---|---|---|---|---|
| KBOD | Detrital decay rate (d−1) | 0.5 | 0.05 | 0.8955 |
| Sb | SOD coefficient at 20 °C (g O/m2/d) | 0.5 | 1.7 | 0.7449 |
| EMCOE (2) | Multiplier of SOD below euphotic zone [-] | 3 | 1 | 0.8931 |
| Kr | Algal respiration rate (d−1) | 0.03, 0.03 | 0.06, 0.06 | 0.8924 |
| Gmax | Maximum growth rate of algae (d−1) | 0.9, 0.9 | 0.6, 0.6 | 0.8966 |
| Km | Algal mortality rate (d−1) | 0.015, 0.015 | 0.03, 0.03 | 0.8944 |
| KP | Half saturation coefficient of phosphorus (mg/L) | 0.035, 0.035 | 0.07, 0.07 | 0.895 |
| SP | Benthic phosphorus release rate (g P/m2/d) | 0.01 | 0.02 | 0.8931 |
| vc | Settling velocity for algae (m/d) | 0.05, 0.05 | 0.15, 0.1 | 0.8931 |
| vB | Settling velocity for detritus (m/d) | 0.05 | 0.15 | 0.8931 |
| Topt | Optimum temperature for algal growth (°C) | 27,27 | 20, 20 | 0.8931 |
| Tmax | Maximum temperature for algal growth (°C) | 42, 42 | 25, 25 | 0.8951 |
| Tmin | Minimum temperature for algal growth (°C) | 0, 0 | 3, 10 | 0.8931 |
| ZP | Zooplankton population (m−3) | 200 | 100 | 0.8966 |
| ZPmin | Minimum zooplankton for grazing (m−3) | 50 | 10 | 0.8966 |
| Chlamin | Minimum chlorophyll-a for grazing (mg/L) | 0.001, 0.001 | 0.002, 0.002 | 0.8924 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tasnim, B.; Fang, X.; Hayworth, J.S.; Tian, D. Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications. Water 2021, 13, 2088. https://doi.org/10.3390/w13152088
Tasnim B, Fang X, Hayworth JS, Tian D. Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications. Water. 2021; 13(15):2088. https://doi.org/10.3390/w13152088
Chicago/Turabian StyleTasnim, Bushra, Xing Fang, Joel S. Hayworth, and Di Tian. 2021. "Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications" Water 13, no. 15: 2088. https://doi.org/10.3390/w13152088
APA StyleTasnim, B., Fang, X., Hayworth, J. S., & Tian, D. (2021). Simulating Nutrients and Phytoplankton Dynamics in Lakes: Model Development and Applications. Water, 13(15), 2088. https://doi.org/10.3390/w13152088







