Abstract
The multiple anthropogenic activities taking place in the marine environment increase and create a high demand for maritime space. The pressures generated thereof on coastal and marine resources require an integrated planning and management approach. The MSPD (2014/89/EU) forms the legislative framework to regulate maritime activities and ensure a sustainable use of the marine environment. Based on the MSFD (2008/56/EC), the present study provides an overview of the state of the marine environment in Greece and the existing pressures while examining the potential contribution of MSFD knowledge to the MSPD implementation, identifying possible knowledge gaps for the subsequent MSP process phases. It is supported that the MSFD constitutes the best available scientific knowledge about the ecosystem and its dynamics, and provides an open access database which should be optimally used in the MSP process. The MSFD data provided concerning Greece can support the implementation of the MSP process, as the MSFD qualitative descriptors of state and pressures feed MSP sectors with environmental data essential for the MSP application, and weigh the intensity of each descriptor contribution to this interrelationship. Considering MSPD requires the adoption of an ecosystem-based approach, it can only attain its goals based on MSFD input and aspirations.
1. Introduction
The various anthropogenic activities such as maritime shipping, fishing, aquaculture, tourism, renewable energy production, oil and gas exploration and exploitation, extraction of raw materials, ecosystem and biodiversity conservation, and underwater cultural heritage create a high and rapidly increasing demand for maritime space [1]. The unplanned use of the marine space taking place in the course of the last decades, in addition to the increasing competition among the human activities at sea, has caused irreversible damages to the marine ecosystem [2]. Thus, the multiple pressures on coastal resources require an integrated planning and management approach.
The Maritime Spatial Planning Directive (MSPD) 2014/89/EU is the legislative tool used to address these issues, aimed at the sustainable growth of maritime and coastal economies and the sustainable use of marine and coastal resources. It provides an operational framework for MSs to analyse and organize human activities in their marine areas in order to maintain the value of marine biodiversity and allow the sustainable use of the economic potential of their seas [3].
Action on MSP at the EU level must be developed in full coordination with existing and potential policies and initiatives within the field of maritime policy, including in particular the implementation of the Marine Strategy Framework Directive or MSFD (2008/56/EC) and ICZM [4]. The MSFD constitutes an overarching framework directive that sets out environmental objectives to be applied across all European marine regions, integrating all activities that affect marine ecosystems and ensuring sustainable use, although not actually regulating them [5]. Therefore, the directive, strictly following the ecosystem approach, is supposed to comprehensively integrate existing relevant sectorial policies [6] and as a result, alignment and coordination with other sectoral policies is essential to attain the directive objectives both at national and EU levels.
The MSPD as a legislative instrument should indirectly apply the ecosystem-based approach as referred to in Article 1(3) of the MSFD (2008/56/EC) with the aim to ensure that the collective pressure of all activities is kept within levels that are compatible with the achievement of GES [2]. The final objective is using the ecosystem approach for evaluating the cumulative impacts and informing and developing marine spatial planning that is able to guarantee the availability and durable use of goods and services [7], as assessing the status of marine ecosystems under an ecosystem approach is fundamental to informing management decisions [8].
MSPD and MSFD represent different perspectives on the marine environment. Jones et al. [9] compared the ‘soft sustainability’ of the MSPD, in which the needs of different maritime sectors are balanced, with the ‘hard’ sustainability of the MSFD, in which ecosystem conservation is the core of the ecosystem-based approach. Although both directives have specific objectives, Boyes and Elliott (2014) [10] have highlighted the importance of linking their efforts. In addition, Maza et al. (2020) [11] stressed the need to streamline the widely scattered marine policy landscape and consider all relevant European policies while implementing their obligations. At the end of the day, by resolving conflicts and regulating maritime activities that are drivers of state changes, MSP can make a significant contribution to achieving GES [12].
The first round of MSP reporting in accordance with the requirements of MSPD was due in March 2021 but only six countries managed to respond to the requirements [13]. To progress with the MSPD implementation, the MSFD can be a powerful instrument for integrated marine management in the European seas whose integration with the ongoing MSP processes can provide concrete data from the assessment and monitoring of the marine environment; can ensure a robust consideration of the marine environment in the planning process; and can promote the application of ecosystem-based management [14]. As MSP is based on the best available scientific knowledge about the ecosystem and its dynamics, and there is a need to clarify how national processes concerning the marine environment and ecosystem-based management can support the implementation of EBA in MSP, planners should make optimal use of information gathered and/or reported in relevant policies, especially those referred to the MSFD [15].
The responsibility for MSP lies at the national level and addresses maritime activities in a MS’s Exclusive Economic Zone. A transnational, subregional, and even a regional sea perspective is utilized when maritime activities and/or their effects cross national borders [16]. Both MSP and MSFD require a regional cooperation and coordination of activities between member states (MS), and, whenever possible, third countries sharing the same marine region or subregion. For example, to improve adequacy and coherence in MSFD implementation, MSs need to act in a coordinated manner by exchanging experiences and best practices, and aligning approaches, establishing common thresholds and baselines regionally/subregionally [17]. Such cooperation activities are strictly linked to the several priorities that each MS adopts in its marine waters, sharing the management of threats and risks of the region or subregion [18].
As Dassenakis et al. (2019) [19] mentioned, a major challenge in applying the ecosystem-based MSP is the gathering of knowledge on the structures and functions of ecosystems, as well as on the pressures and impacts induced on these ecosystems from various human activities. Given the necessity for undertaking an MSP process, the present analysis focused on the characteristics of the Greek marine environment and the pressures induced by anthropogenic activities, with main objectives to: (i) provide an overview of the state of the marine environment in Greece; (ii) examine/indicate the potential contribution of MSFD knowledge to the MSPD implementation; and (iii) state concerns, critical issues, and knowledge gaps in order to frame the subsequent phases in implementing the MSP process.
This study has been based on existing information gathered during the implementation of the Marine Strategy Framework Directive [20]. With a view to acquire a comprehensive snapshot of the state of the marine and coastal environment and existing pressures, we focused on the implementation of the second cycle of the MSFD in Greece initiated in 2018, which is considerably updated related to the initial assessment conducted in Greece in 2012, as it is based on recent monitoring data. More precisely, the overview was based on the implementation of Articles 8, 9, and 10 as reported to the European Commission by Greece in 2018 according to the requirements of the 2008/56/EC Directive, found in the associated Eionet Central Data Repository hosted by the European Environmental Agency [21]. In some cases, information was enriched with additional published data.
2. Area under Investigation
In Greece, about 85% of the population is concentrated along the coastline in a zone extending up to 50 km inland, whereas over 75% of the industry, 90% of the tourism, a large part of agriculture, and almost the whole of fisheries and fish farming takes place in this zone [22]. Although Greece has especially significant marine economic activities, experience in implementing MSP is rather limited. Spatial frameworks covering the productive sectors of aquaculture and renewable energy sources and industry have been established, whereas the existing marine parks in Zakynthos and Sporades for the protection of marine turtle and monk seal, respectively, could be considered as early maritime spatial plans adopted in the 1990’s [23].
Situated in the eastern Mediterranean Basin, including the Ionian Sea, Aegean (or the Archipelagos), and Levantine Sea, Greece is known for its highly insular and coastal nature. In fact, Greece has mainly insular border limits (Diapontia islands to the west, Castelorizo island to the east, and Gavdos island to the south) neighbouring several coastal countries: Turkey, Cyprus, Egypt, Libya, Malta, Italy, and Albania.
The jurisdictions of Greece over its surrounding seas are the following:
- Territorial waters: The Territorial Waters of Greece were very recently (January 2021) set to 12 nm in the Ionian Sea, whereas they are set to 6nm from the baseline in the Aegean and Levantine Sea.
- Contiguous Zone: Greece has no designated contiguous zone.
- Continental Shelf: The continental shelf was only designated in the common maritime borders with Italy in the Ionian Sea with an agreement in 1978 (Greek Law 786).
- Exclusive Economic Zone (EEZ): Greece proclaimed an EEZ towards the west in 2020 in agreement with Italy (Law 4716/2020, O.J. 163/A/28-8-2020) and towards the south with Egypt (Law 4717/2020, O.J. 164/A/28-8-2020) (Figure 1).
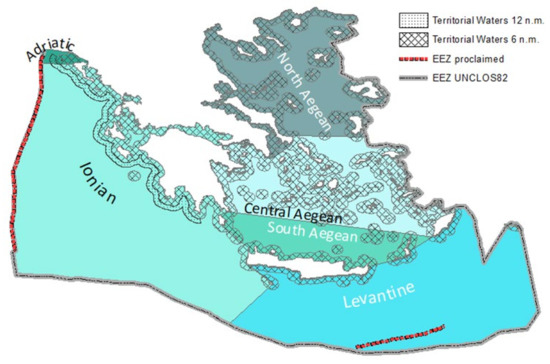 Figure 1. The territorial waters of Greece and marine reporting units as in MSFD.
Figure 1. The territorial waters of Greece and marine reporting units as in MSFD.
The total surface of Greece is 132,000 km2, having a coastline of more than 20,000 km. Greece is a highly insular country with 7500 km of its coastline belonging to more than 6000 islands and islets, 117 of which are inhabited. The underwater morphology of Greece is diverse with the greatest depth reaching 5127 km (Oinouses Well) in the offshore area southwest of Peloponnese. The continental shelf (defined by slopes < 2%) represents about 4% of the subaqueous relief in the Ionian Sea and 21% in the Aegean Sea [24]. The circulation in the Aegean Sea is generally cyclonic with warm, saline water entering from the Levantine Sea through the eastern Cretan Straits and continuing northwards to the Eastern Aegean Sea in which it subducts below the lighter water coming from the Black Sea. The Black Sea water mass moves along the east coast of Greece towards the south [25]. The unconsolidated offshore surficial sediments of the Aegean seabed are mixtures of mud (silt and clay) and sand, while only in some areas silty or clayey-sized sediment replaces the mud content, deposited under the influence of rivers in shelf areas, as well as in most of the deep basins of the Aegean Sea. The mud component found on the shelves is of a terrigenous origin [26].
The Mediterranean subregions, as in 2008/56/EC, in which Greece has territorial waters include: (i) the Aegean–Levantine Sea (≤6 nautical miles), (ii) the Ionian Sea and the Central Mediterranean Sea (≤12 nautical miles), and (iii) the Adriatic Sea (≤6 nautical miles).
In the aforementioned marine regions, in the first MSFD implementation cycle (2012), the demarcation of assessment areas was performed based on hydrological, geomorphological, and biological criteria. The Aegean Sea and Levantine Sea, which are considered a unified region, were divided into the North, Central, South Aegean, and Levantine Sea for some descriptors. The south-eastern part of the Adriatic Sea, in which Greece has territorial waters, were of the same characteristics with the Greek part of the Ionian Sea. In the second MSFD implementation cycle (2018), these areas are mentioned as marine reporting units (MRUs) used within the reporting obligations to link the implementation of the different articles to specific marine areas. Thus, MRUs in Greece include: the Ionian–Adriatic, North Aegean, Central Aegean, South Aegean, and Levantine Sea (Figure 1).
3. The state of the Marine Environment in Greece
The information in the analysis that follows is presented in accordance with MSFD Articles 8a (state of the marine environment) and 8b (pressures affecting the marine environment). In order to depict the state of the environment, the state descriptors of the MSFD are used, i.e., Descriptors 1, 4, and 6 (D6C4 and D6C5), whereas the presentation of the pressures component is based on pressure descriptors, i.e., Descriptors 2, 3, 5, 6 (D6C1–D6C3), 7, 8, 9, 10, and 11.
3.1. The State of the Marine Environment in Greece
3.1.1. Biological Diversity; Descriptor 1
The Greek biodiversity is considered to be one of the richest both in the European and Mediterranean seas and is characterised by a high degree of endemism despite the prevailing oligotrophic conditions [27]. According to the National Biodiversity Strategy and Action Plan [27], the monitoring and assessment of the conservation status aims to halt the loss of biodiversity and the degradation of ecosystem services in Greece by 2026 and restore them as far as feasible.
Until now, 3500 animal species have been reported in the marine environment. The species found in coastal and marine ecosystems [27] include: (a) macrophytes: more than 503 taxa of marine algae, including 313 taxa of red algae (Rhodophyta), 103 taxa of brown algae (Phaeophyceae), and 87 taxa of green algae (Chlorophyta), as well as 7 taxa of angiosperms in the sea and brackish water (e.g., Posidonia oceanica, Cymodocea nodosa, Zostera noltei, and Halophila stipulacea); (b) marine mammals: 12 taxa of cetaceans and the Mediterranean monk seal Monachus monachus); (c) marine reptiles: 3 taxa of sea turtles including the Caretta caretta that is reproduced on the Greek shores; (d) fish: approximately 480 marine fish species; and (e) zoobenthos: more than 2650 taxa of benthic macrofauna, not including the fauna on the hard substrate of the coast and bathyal zone. Among the 37 species of seabirds recorded in Greek waters, there are six that are considered purely seabird species in the Greek context [28].
According to Commission Decision 2017/848/EU [29], there are six criteria for Descriptor 1: D1C1/species mortality rate; D1C2/species population abundance; D1C3/species population demographic characteristics; D1C4/species distributional range and pattern; D1C5/habitat extent and condition; and D1C6/pelagic habitats’ condition and functions. However, only D1C2 and D1C4 can be addressed appropriately as there is not enough data to address all the criteria.
For species population abundance (D1C2), cetaceans, marine reptiles and marine mammals have been considered. With regard to cetaceans, there are no long-term monitoring data of all species and, moreover, the lack of data for some cetacean species provide rather uncertain estimates. During the recent years, scientific data on cetacean distribution and ecology are collected by different research groups more systematically [30,31,32,33,34,35]. The cetacean species occur mostly along the Hellenic Trench (from the western Ionian islands and the Peloponnese to the south of Crete and southeast of Rhodes Island), in Myrtoon Sea, and in areas in the Aegean with steep slopes and great depths. However, some species of Delphinidae such as Delphinus delphis, Stenella coeruleoalba, and Tursiops truncatus occur in coastal areas, straits, and gulfs of Greece. Cetaceans such as Delphinus delphis, Grampus griseus, Stenella coeruleoalba, Tursiops truncatus, Phocoena phocoena, and Physeter macrocephalus are highly threatened by fishing activities (e.g., mainly pelagic surface fishing gear and illegal fishing with dynamite), marine pollution (e.g., plastic bag-swallowing and noise pollution), and other maritime activities (collisions with ships, disturbance by navy exercises, and geological research) [30,31,33].
As for marine reptiles, Greece hosts 61% of loggerhead turtle nests (Caretta caretta) in the Mediterranean (2015), nesting mainly in the Laganas Bay (Zakynthos Island) and Kyparissiakos Gulf (W. Peloponnese). Green turtles (Chelonia mydas) do not nest in Greece but are regularly detected at sea, albeit at a much lower density than loggerhead turtles. The Lakonikos Gulf is a foraging area for juvenile individuals of Chelonia mydas. Finally, the leatherback sea turtle (Dermochelys coriacea) has a worldwide distribution and its presence in the Mediterranean is 60–200 times lower than in the Atlantic. Degradation of nesting sites mainly due to touristic activities commonly occurs, as well as the predation of eggs and young turtles by foxes, gulls, and other predators. Most importantly, plastic materials consumption, incidental capture to fishing gears, deliberate killing, and collisions with boats constitute major threats for marine reptiles [34,35,36,37].
Monachus monachus is an endangered species whose habitat deterioration and loss are attributed to human intervention to coasts, deliberate killing, and accidental catching in fishing gears, as well as attributed to marine pollution and diseases. Monachus monachus is mainly distributed in the coastal and insular of Greece but its important populations are located on the Northern Sporades, Kimolos, Gyaros, and Karpathos islands (Mom, 2005). According to Dendrinos et al. (2009) [38], the Ionian islands of Zakynthos and Kefalonia host lower populations. The Greek monk seal population has been estimated at 250 seals, which is equal to 40–51% of the species’ global population [39,40]. For the protection of the species, the Marine Park of Northern Sporades has been established.
The marine protection areas in Greece for the five most important species of seabirds extend to 9943 km2 and approximately represent 8.7% of the Greek territory [28].
For Monachus Monachus, cetaceans, marine reptiles, and seabirds, protection areas have been extended for better protection of the species (D1C2) so that GES will be achieved in the second MSFD implementation cycle.
To assess benthic ecosystems (D1C4), it was considered that benthic organisms react to various forms of anthropogenic pressure including organic pollution, disturbance of sediments by discharges, excavations, and son. Under pressure, resistant benthic organisms increase in number or density, while sensitive organisms recede. Thus, moderate to high dominance of opportunistic or resilient species indicates some degree of environmental pressure. Due to this inherent stability of the benthic ecosystem, zoobenthic and phytobenthic bio-communities are used to assess the state of the marine environment. The Bentix Index combined diversity indices [41] is used for Greek coastal ecosystems and the results also address D6C3.
Macroalgae are used as biomarkers of the ecological quality of coastal waters due to their wide distribution, important ecological role, and high sensitivity to anthropogenic stress. Chronic anthropogenic disturbance such as pollution and eutrophication degrades the ecosystem and opportunistic species rapidly reproduce and dominate. The EEI-c Index used in 94 water bodies in Greece showed that 40% were classified in the “high” class of ecological quality, 27% in the “good”, 18% in the “moderate”, 10% in the “incomplete”, and 5% in the “poor”. It is obvious that in closed and semi-closed bays, e.g., in Thermaikos, Saronikos, Amvrakikos Gulf, and the Gulf of Kavala, wastewater discharges and agricultural effluents and/or industrial waste have affected benthic vegetation. The water bodies of the inner Thermaikos and Amvrakikos Gulf were classified in the “poor” class of ecological quality. The water bodies of the Saronikos Gulf were classified in the “bad” class in Psyttalia (municipal waste treatment center) and “good” class in Sounio. Overall, North, Central, and South Aegean were classified in the “high” class of ecological quality, whereas the Ionian–Adriatic and Levantine Sea were in the “good” class [41,42].
Posidonia oceanica is used for the assessment of the ecological quality. The Posidonia oceanica Rapid Easy Index (PREI) is a quantitative expression of the condition of a seagrass meadow and the ecological status of the water. Where Posidonia oceanica is absent, the index CymosSkew is used based on Cymodocea nodosa (common in the North Aegean). Overall, in Greece, 60.71% of the sampling stations are in the “good” class of ecological quality and 17.86% in the “high” class. Therefore, 78.57% of the stations achieve GES. Most of the “moderate” class stations are located in the North Aegean (11 stations). In the Central Aegean Sea, only three stations were classified in the “moderate” class of ecological quality, located on the sea front of the metropolitan area of Athens [41,43].
3.1.2. Food Webs; Descriptor 4
Food webs are dynamic and change in response to various factors. In the Greek Seas, as well as worldwide, fishing affects population dynamics, environmental indicators related to size, relative biomass, and trophic indicators [44,45]. In addition, climate change favours thermophilic species including non-indigenous species (NIS), over the psychrophilic ones [46,47,48], whereas nutrient concentrations can affect populations of highly variable species such as small pelagic fish [48].
The decline of fishing and spawning sites has reduced the commercial stocks since the mid-1990s with respect to all major groups of species (e.g., marine pelagic, marine demersal, and cephalopods), although the annual catches of crustaceans are almost stable during the past three decades [49]. A variety of fish stocks across the Mediterranean Sea are being rapidly depleted due to overfishing. A steady deterioration of the biomass of 42 Mediterranean stocks in 1990–2010 is reported. Moreover, unreported fisheries and misreporting estimates in landings, illegal fisheries, and the lack of enforcement of laws and agreements lead to stock depletion [50]. Further contributing factors are the loss and degradation of natural habitats due to pollution and/or climate change impacts, as well as marine invasive species. Overall, the Greek seas are heavily impacted by human activities resulting from multiple drivers rather than one individual use or stressor [51]. Some viable populations of elasmobranchs still exist in the eastern Mediterranean (e.g., Turkish coasts) but the rapid increasing catches of elasmobranchs in non-EU waters in the last 20 years is a worrisome indication of an increased depletion risk for these “residual” populations [49].
Nowadays, dramatic spatial increases and temporal shifts in jellyfish distributions have been reported in Greece, associated with ecological ramifications such as food web and biogeochemical pathway alterations. Aurelia aurita was indicated to be one of the main occurring medusa in the northern Ionian Sea. Even though Aurelia aurita was not the main species responsible for the Mediterranean bloom, its biomass was reported to increase more than 30 times during the 1980s. Furthermore, the biomass of Cotylorhiza tuberculata was reported to increase in northern Crete, Cyclades, south and northeast Aegean, and the Gulfs of Thermaikos and Kavala [52].
According to Commission Decision 2017/848/EU, there are 4 criteria for Descriptor 4: D4C1/trophic guild diversity; D4C2/abundance balance; D4C3/individuals’ size distribution; and D4C4/productivity. Those addressed are D4C2–D4C3 as data is not adequate to safely estimate all of the criteria.
In North Aegean, mesozooplankton was designated as the most important group as it had the greatest impact on the food web [53]. In addition, squid, cilia, macro-zooplankton, anchovies, and medium-sized pelagic fish (bonito, small tuna, etc.) also had a significant effect on the food web [53]. Accordingly, in the Ionian Sea, seals, octopuses, benthic (non-commercial), micro and mesozooplankton, anchovies, small pelagic sharks, and sardines have the greatest impact on the food web [54].
In the Aegean, the biomass of small pelagic fish, i.e., anchovies and sardines, was estimated at relatively high levels (2016), whereas in the Ionian Sea, anchovy biomass exhibited a slight decrease [55]. For sardines, the decrease was higher. However, the time series available are not adequate to define GES.
Large pelagic fish (Atlantic bluefin tuna) stock assessments concern broader areas and not specifically the Greek seas as they are migratory species. Thus, the assessment is conducted at the eastern Atlantic and Mediterranean region level for Atlantic bluefin tuna and at the Mediterranean level for swordfish. Bluefin tuna biomass has improved and thus the stock is sustainable [56], whereas swordfish biomass is considered overfished [57].
3.1.3. Habitats; Descriptor 6
The most common habitats in Greece are seagrass meadows. Their estimated regression amounted to 34% in the last 50 years. The spatial distribution of Posidonia oceanica in Greece was mapped in detail for the needs of the DG Fisheries of the Ministry of Rural Development and Food [58]. Direct or indirect human induced disturbances include mechanical damage mainly caused by destructive fishing practices, pollution, sedimentation, diver frequentation, biological invasions, mass mortality outbreaks related to temperature anomalies, and the synergistic effects of these stressors. Bottom-trawling is also expressly forbidden on seagrass meadows (Ministerial Decision 2442/51879/2016 as amended in 2826/68784/26-6-2017).
In consideration of the importance of these habitats for biodiversity and their vulnerability to environmental threats and pressures, they are protected by both European directives and international treaties (e.g., Ramsar Convention and Habitats Directive). In addition, according to the European Regulation, habitats other than Posidonia oceanica seagrass beds are considered to be equally important for conservation, including mäerl beds, coralligenous habitats, and marine caves [27]. Coralligenous formations and marine caves are reported mostly in the Aegean Sea. There is limited data regarding the presence of coralligenous formations in the Ionian Sea and northern Levantine coasts [59]. Following Commission Decision 2017/848/EU, there are two criteria relating to state for Descriptor 6: D6C4/benthic habitat loss and D6C5/benthic habitats alteration.
It is estimated that benthic habitat loss (D6C4) due to anthropogenic activities does not exceed 5% of the total habitat extent, thus GES is achieved. For benthic habitats alteration (D6C5), it is estimated that less than 15% of the Greek assessment areas are impacted and it is suggested that the GES threshold is set at 15%. Thus, GES is also achieved.
3.2. Anthropogenic Pressures Affecting the Marine Environment
According to ANNEX III, Table 2 of the Directive 2017/845/EU amending Directive 2008/56/EC, the anthropogenic pressures affecting the marine environment include biological and physical pressures as well as the introduction of substances, litter, and energy. The pressures affecting the marine environment in Greece are presented in accordance with MSFD Articles 8b and 9 2018 reported data.
3.2.1. Biological Pressures
The biological pressures occurring in the Greek marine environment include the input and spread of NIS, the input of microbial pathogens, and extraction of species by commercial and recreational fishing.
(Ι) Non-Indigenous Species; Descriptor 2
NIS constitute a considerable biological pressure on the Greek marine environment as many species of Indo–Pacific origin enter the Mediterranean from the Red Sea and are dispersed according to the prevailing currents, considering Greece is located in this spreading corridor. In addition, indigenous flora and fauna are threatened by NIS entering in other ways apart from the introductions via the Suez Canal. The most important vectors of introduction are imports for aquaculture purposes and trade; fouling on merchant ships and/or yachts; in ballast water; and escapes/releases from aquariums [60,61].
A total of 242 marine NIS have been recorded in Greek waters as of 2020. The taxonomic group with the highest number of NIS are fishes, contributing with 51 species, followed by 50 molluscs, 43 crustaceans, and 33 annelids [61].
Most marine biota has spread mainly in the South Aegean Sea (n = 196) and Levantine Sea (n = 113), and less to the North Aegean Sea (n = 92) and Ionian Sea (n = 78). There are currently 7 NIS in the Adriatic Sea. Freshwater biota occurs mostly in northern Greece. For all source regions and taxonomic groups considered, the invasion rate has been increasing since the end of the last century; this trend is indirectly related to the “tropicalization” of the Mediterranean Sea [60,61].
According to Commission Decision 2017/848/EU, there are three criteria for Descriptor 2: D2C1/NIS number; D2C2/NIS abundance and distribution; and D2C3/species and habitat extent NIS impacted. Despite the extensive research, there are still knowledge gaps regarding data availability, missing taxa, uncertainty in taxonomy, uncertainty of introductions via the Strait of Gibraltar, and the effect of climate. NIS numbers that entered the Greek marine environment due to anthropogenic activities in the period of 2012–2017 compared to 2006–2011 seem to have decreased to 36 from 42. However, it is considered premature to establish a reliable indicator for the assessment of GES and GES thresholds [62].
(II) Microbial pathogens
Bathing water quality is monitored and assessed based on the provisions of Directive 2006/7/EC (repealing 76/160/EEC). The quality of the coastal bathing waters shows compliance to 2006/7/EC Directive, exceeding 93% since 1995 and in 2018, the level of compliance reached 97%. The 2018 program included 1598 bathing waters points, of which 1595 are coastal waters and there are inland [63].
(III) Extraction of Species; Descriptor 3
Fisheries exert various pressures on the environment. Overfishing is causing the loss of the biodiversity of targeted and dependent species, disrupting food webs and the flows of biomass (and energy) across the ecosystem with major ecosystem, social, and economic consequences. In addition, fisheries by-catch may include birds, mammals, and turtles. Some fishing techniques cause habitat destruction of seagrass meadows, algal beds, and coral reefs, while illegal fishing techniques using dynamite or cyanides can cause irreversible damage to the marine environment. Pollution from fish processing plants, use of ozone-depleting refrigerants, and dumping at sea of plastic debris from fishing activity are some other processes that deteriorate marine habitats [62]. The decline of commercial stocks due to direct predation or competition for resources (food or space) is the presumed mechanism of negative impact in the cases of the decapods Homarus americanus and Paralithodes camtschaticus; the fish Fistularia commersonii, Neogobius melastomus, Saurida undosquamis, Liza haematocheila, Siganus luridus, and S. rivulatus; the bivalves Crassostrea gigas and Pictada imbricata radiata; and the gastropods Urosalpinx cinerea and Rapana venosa [64].
Recreational fisheries (RF) exhibit an upward trend in Greece. There is a great uncertainty about the estimation of catches considering they are not recorded, while the studies are sporadic and scarce [65,66]. In addition, few things are known regarding the impact of recreational fishing in the fish stocks [66].
Fishing is prohibited in areas with Posidonia οceanica meadows. Bottom trawl fishing is prohibited in Greek national waters from June to September. There are local closures either throughout the year or seasonally, in certain regions, mainly river mouths and gulfs. Fisheries Restricted Areas have also been established by FAO [67].
Sixteen (16) fishing subareas have been defined in the Greek seas of multi-species composition and multi-gear fisheries [68]. According to the National Fisheries Registry (17/6/ 2020), the Greek fishing fleet represents the largest fishing fleet among the EU Mediterranean countries. Three major categories depending on the fishing activity exist: (a) coastal-small length vessels operating mostly in a zone if 3–10 nm wide along the coasts; (b) pelagic-purse seine fishing that operates mostly in open seas rich in plankton and uplift currents; and (c) demersal-otter trawl fishing that exploits fishing grounds covering the continental shelf up to 800 m depth but mainly within 400 m. Fishing grounds are not uniformly located along the coastal zone of the country; the major fishing grounds are located within the range of the continental shelf in areas rich in brackish water, plankton, and local upwellings. The north-eastern Aegean Sea is the major fishing as well as spawning ground of Greek waters for most fish species. The largest artisanal fleets operate in the Aegean Sea (GSAs 20) where the majority of the bottom trawlers are exploiting the fishing grounds, but in terms of fishing pressure on the shelf, the area with the highest number of artisanal vessels per km2 is the eastern Ionian Sea (GSAs 20). The total number of fishing vessels has decreased considerably during the last years, following Regulation 1198/2006. The total fishing effort is rather homogeneously distributed among the months [69].
There are uncertainties in the evaluation of the Greek fisheries’ status because existing databases are not updated and there are no operational relevant portals; there is extremely limited stock assessments and insufficient implementation of EU regulations for illegal fishing. There is difficulty in estimating the balance between the fishing capacity, fishing effort, and making a reliable stock assessment due to the lack of relevant data [68]. The National Fisheries Data Collection Programme was not implemented in the period of 2009–2013 due to administrative and financial constraints. In fact, during 2009–2018, there were only two years in which data collection was fully implemented on a 12-month basis. According to Commission Decision 2017/848/EU, there are three criteria for Descriptor 3: D3C1/fishing mortality; D3C2/spawning stock biomass; and D3C3/age and size distribution.
With respect to fishing mortality (D3C1), amongst 24 major investigated commercial stocks, three appear to be overfished: Merluccius merluccius and Sepia officinalis in the Aegean Sea, and Xiphias gladius in the Mediterranean. Criteria D3C2 and D3C3 could not be assessed due to the lack of data. B/BMSY was assessed: of the 24 major commercial stocks investigated, four appear to be below safe biological thresholds: Merluccius merluccius, Sardina pilchardus, and Sepia officinalis in the Aegean Sea, and Xiphias gladius in the Mediterranean. Overall, among the investigated species in the Greek seas, stocks are sustainable except for Merluccius merluccius, Sardina pilchardus, Sepia officinalis, and Xiphias gladius which are overfished. Therefore, all the species are considered to be in GES except for Merluccius merluccius and Sepia officinalis in the Aegean Sea, and Xiphias gladius in the Mediterranean, while the status of Sardina pilchardus is not certain [62].
3.2.2. Physical Pressures
The physical pressures identified in the Greek Marine environment include physical disturbance to seabed, physical loss, and changes to hydrological conditions.
(I) Seafloor Integrity; Descriptor 6
The pressures that cause physical loss mainly include infrastructure to protect the coast from erosion, infrastructure for wind farms, and so on, while the pressures that are more related to disturbance concern the extraction of materials from the bottom or fishing with trawlers, and so on.
Although there are no existing offshore wind parks in Greece (D6C1), there are ongoing procedures foreseeing the legislative framework for future installation in licensing areas. Up to now, 12 indicative areas have been proposed to host wind parks [70] while currently many other areas are scheduled as potentially licensed areas.
There are three criteria concerning pressures according to Commission Decision 2017/848/EU for Descriptor 6: D6C1/physical loss; D6C2/physical disturbance; and D6C3/affected habitat extent. Coastal erosion (D6C1) has been induced mainly by human interference (road construction close to the shoreline, establishment of fishing shelters, and development of tourist marinas) but has been exacerbated by climatic variability (sea level rise). More than 70% of the coastal zones with existing coastal residential areas, mainly close to big urban agglomerations, are intensively exploited. Low land coastal landforms, that is, beaches, frequently (>75%) host some kind of artificial structures [71] to prevent from erosion. Nearly one-third (28.6%) of the Hellenic Aegean coastline has been characterized as extremely vulnerable [72]. The phenomenon is more intense in delta areas as the result of human presence in both catchment and deltaic areas (e.g., dam constructions and river regulation) [73,74,75]. In many cases, coastal structures favour seabed alterations, the spatial extent of which is not provided.
The current state of hydrocarbons exploitation in Greece (D6C2) is in its infancy as there is only one active extraction site in the Greek territory (Prinos complex, located offshore in the Gulf of Kavala, in the North Aegean). The total percentage of the Prinos production offers 0.6% of the national demand, producing a gross added value of 0.5% of national GDP [76]. Beyond the necessary infrastructure for storing and processing the intermediate products, six submarine pipelines are used for the transfer of hydrocarbons. The extraction of oil and gas follows the policy and National Law 4001/2011 concerning the prospection, exploration, and exploitation of hydrocarbons in the Greek legislation. Most recently, based on the geologic and tectonic regime of Greece, the government has designated 20 distinct marine areas for search, exploration, and exploitation of hydrocarbons, focusing on the western part of the mainland and the Ionian Sea, Libyan Sea under the Island of Crete, and the Thermaikos Gulf. However, there are knowledge gaps relative to the areas with promising geologic regimes that are to be explored in the near future, where the geophysical research, which will show the actual possibilities of those areas, has not been conducted yet.
Marine aggregate extraction (D6C2) has taken place in Greece since the 1960s to a limited extent, mainly from the inner continental shelf of the Greek islands (e.g., Andros and Mykonos), producing material for various construction uses [77]. Recently marine aggregate exploitation has been concentrated off the coasts of Trikeri (Northern Evoikos Gulf) and southern Evia [78], while the prospective of MA exploitation has been taking place sporadically since the 1980s in Kissamos Bay (NW Crete) [79]. Although there is no available information on annual extraction volumes, they are rather of the order of hundreds of thousands of m3 rather than millions. The relevant regulatory framework is multi-level and comprises a series of laws [80,81], which also define the constitution of committees that make decisions regarding the granting of MA extraction concessions [82]. Total seabed physical damage or biodiversity disturbance have not been identified.
Dumping operations (D6C2) with significant direct impacts and adverse effects on benthic organisms have been conducted numerous times in the past. The most characteristic examples are those of industrial waste disposal, e.g., in Antikyra–Korinthiakos Gulf, Larimna–Northern Evoikos Gulf [83], and dredged material disposal e.g., in Saronikos Gulf [78].
Despite temporal and spatial restrictions, trawling is mainly responsible for physical disturbance pressures on the seabed (D6C2). The north Aegean region is the most important fishing field in the country, accounting for 32% of the annual fishing effort of trawling. Overall, in the Aegean, fishing effort for trawling represents 88.3%, while in the Ionian it represents 11.7%. According to Eigaard et al. (2016) [84], the seafloor extent disturbed by trawling in the Aegean is extensive; 56.8% of seafloor extent is disturbed in the 0–200 m depth zone, and 13.3% of seafloor extent is disturbed in the 201–1000 m depth zone. The Seabed Integrity Index is 0.56% in the 0–200 m depth zone and 0.9% in the 201–1000 m depth zone, with the value 1 assigned to undisturbed sea bottom.
Regarding D6C3, the assessment of benthic communities of soft substrate, according to the BENTIX Index, water bodies were found as not achieving the “good” ecological status and thus not in GES. Benthic bio-communities include: the Amvrakikos Gulf, the Vistonikos Gulf, the Bay of Navarino, the Argolic Gulf, the inner Saronikos Gulf and Psyttalia, the western basin of the Saronikos Gulf, the Gulf of Thessaloniki and the inner and outer Thermaikos, the Bay of Avlida, V. Evoikos, the Elefsis Gulf, the Bay of Faneromeni, the Laconikos Gulf, the eastern shores of the Corfu Sea, the Gulf of Igoumenitsa, the Gulf of Ierissos, the Gulf of Kavala, the shores of Alexandroupolis, the Bay of Alexandroupolis, the Gulf of Ierapetra, and the Gulf of Gera [62].
Hard substrate habitats assessed with the EEI-c index showed that water bodies with a poor ecological condition of macroalgae include: the Amvrakikos Gulf, Navarinou Bay, Methoni Bay, Corinthian Gulf, Gulf of Argostoli, the inner Saronikos Gulf and Psyttalia, the western basin of the Saronikos Gulf, the Gulf of Thessaloniki and the inner Thermaikos, the North Evoikos, the Gulf of Elefsina, Mr. Faneromenis, the Laconikos, the eastern shores of the Corfu Sea, the shores of Alexandroupolis, the Gulf of Ierapetra, the shores of Chania, and the Gulf of Agios Nikolaos. The status of Posidonia οceanica meadows assessed with the PREI index showed that water bodies with poor ecological condition of the Posidonia oceanica meadows include: the Bay of Ierissos, the canal of Thassos, the Bay of Agios Nikolaos, the Bay of Gera, and the Bay of Heraklion [62].
According to a holistic assessment, using NEAT, the spatial extent of habitats of water bodies not in GES due to physical disturbance (D6C3) in the Ionian and Adriatic Sea is 6% of the total area, 7.53% in the North Aegean, 5.17% in Central Aegean, and 3.14% in South Aegean. In the Levantine Sea, 100% of water bodies are in GES [62].
(II) Alteration of Hydrographical Conditions; Descriptor 7
Following Commission Decision 2017/848/EU, there are two criteria for Descriptor 7: D7C1/hydrographical alterations extent and distribution, and D7C2/benthic habitat extent adversely affected by hydrographical alterations. Increasing sea temperature constitutes the most obvious and documented change in the characteristics of the Mediterranean and Greek marine waters (D7C1). The warming trends depend on the estimation period and are often hidden behind large multiannual variability. A rising tendency of the temperature has been recorded since mid-1990’s at a maximum rate of ~0.04 °C/per year in semi enclosed basins, i.e., the Saronikos Gulf. In the open sea areas of Greece (Aegean and Ionian) the increasing trend of temperature is expected to be less than ~0.04 °C/per year due to the greater depth and lateral mixing of seawater with other marine masses [62].
Such temperature increase, the so-called Mediterranean tropicalization, provides a suitable environment for the propagation of Lessepsian species entering through the Suez Canal. Therefore, in several cases, the environmental status in terms of biodiversity is affected. The degradation of plant communities of macroalgae due to herbivorous NIS, which have no local natural competitors and thus multiply excessively, is typical (D7C2) [62].
Similarly, a decreasing trend in salinity (D7C1) has been observed in semi enclosed basins, i.e., the Saronikos Gulf reaching maximum values of 0.01 psu/per year due to the vertical stratification that determines the ability of the water column to mix and ventilate in depth and thus maintain GES. Future projections predict with little confidence a deceleration of the vertical mixing in the Mediterranean, thus establishing a lower ventilation rate and hypoxic deep-water conditions, related to seabed biodiversity degradation (D7C2) [62].
Apart from the increasing trends described above which concern all Greek waters, there are changes in temperature and salinity on a smaller spatial scale in coastal areas in which there are desalination units and cooling infrastructures in industries, affecting coastal marine ecosystems by producing hot water and saline waste.
Further modifications that may affect the overall future clockwise circulation pattern of the Aegean Sea in water masses include the exchange through the Dardanelles straits, i.e., changes in the influx of Black Sea colder and less saline waters and/or changes (in volume and physical properties) of the Levantine waters entering the Aegean through its south-eastern section, related to changes in seawater properties in the eastern Mediterranean Sea. Finally, the current estimate of future etesian wind power in the Aegean Sea will be significantly increased by the end of the century, affecting its circulation and relative sea level coastal variations (meteorological tide) [62].
3.2.3. Introduction of Substances, Litter, and Energy
Coastal and marine environments are usually influenced by human-induced and natural pressures which may alter their functioning and contribute to ecosystem degradation and pollution problems [85]. The marine environment of Greece is impacted by the introduction of nutrients, organic matter, synthetic and non-synthetic substances, and radionuclides from point and/or diffuse sources, atmospheric deposition, or acute events. In addition, litter (macro and micro-sized) and anthropogenic sound affect the Greek marine environment.
(I) Nutrient Enrichment; Descriptor 5
Most areas of Greece are characterized as oligotrophic in terms of nutrients [86], while an increase in eutrophication is observed in coastal areas that receive more intense anthropogenic pressures. In Greece, nutrient inputs due to agricultural activity, fish farming, fisheries, river influxes, and municipal and industrial waste influx are associated with eutrophication phenomena [87]. Thermaikos, Elefsis, inner Saronikos, Patraikos (close to Patras), Pagasitikos, North Evoikos, Korinthiakos, and Amvrakikos are gulfs mostly affected by anthropogenic activity and, in fact, constitute hot spots.
Commission Decision 2017/848/EU criteria for Descriptor 5 include eight criteria: D5C1/nutrient concentrations; D5C2/Chlorophyll-a concentration; D5C3/harmful algal bloom events; D5C4/water column transparency; D5C5/dissolved oxygen concentration; D5C6/opportunistic macroalgae abundance; D5C7/macrophyte communities; and D5C8/ macrofaunal communities.
Nutrients (D5C1) and chlorophyll-α (D5C2) concentrations were assessed for the marine subregions of Greece. The WFD good ecological quality status (EQS) is considered as the GES threshold. Using the Eutrophication Index E.I. for the coastal zone of Greece on the 2012–2015 WFD monitoring program data, most areas (66%) are characterized by good trophic status except for areas with riverine and agricultural impacts, industrial and municipal waste, and fish farming where GES values are low [87].
In the open sea of the northern Aegean, nutrients are affected by Black Sea waters that enrich the surface layer of the water column mainly with organic nitrogen and phosphorus and less with inorganic nutrients. In the south of the Aegean, Levantine Sea, and Ionian Sea, nutrient concentrations are low.
According to (D5C2), in most cases, coastal marine waters in Greece are in good and high ecological condition (2018/229), therefore, they achieve GES.
Regarding (HABs) (D5C3), one of the main challenges concerns the low availability of high-frequency in situ data that adequately reflects the spatio–temporal variability of phytoplankton and coastal water blooms [88]. HABs are observed sporadically in various Greek coastal areas but the Thermaikos Gulf is the area with the most common HAB events with serious economic consequences for shellfish farming, in which 85% of the shellfish farming activity in Greece takes place [62]. The Maliakos Gulf is another coastal area characterized by the frequent presence of potentially harmful microalgae [88].
In a holistic approach using ΝΕΑΤ (nested environmental status assessment tool), it was estimated that in North Aegean, 75% of the areas are in good and high environmental condition, i.e., GES is achieved, whereas 11.6% of the water bodies are in poor environmental condition due to intense anthropogenic pressures (the Thermaikos, Strymonikos, Kavala, and Maliakos Gulfs, and the Thracian coast). In Central Aegean, 69% of the areas achieve GES, while 5.15% of the areas do not achieve GES (intense anthropogenic pressures and diffusion of treated wastewater with areas of reduced oxygen conditions near the bottom, e.g., Gulf of Elefsina and Saronikos Gulf). In the southern Aegean and Levantine Sea, 100% GES is achieved. The Levantine Sea is in a high environmental condition. In the Ionian Sea, 88% of the area achieves GES with only 2% not achieving GES (Navarino Bay and the Amvrakikos Gulf with relatively low water renewal in which conditions of hypoxia and even anoxia occur) [62].
(II) Contaminants; Descriptor 8
Heavy metals in the coastal marine environment derive largely from terrestrial inputs (natural or anthropogenic). In Greece, heavy metals’ concentrations in the coastal zone are 1.5 to 2 times higher than those of the high seas, whereas areas with intense anthropogenic and industrial activity (e.g., the Gulf of Elefsis and the Bay of Thessaloniki) are enriched in heavy metals 2 to 5 times in relation to the Aegean Sea. Fish farms also enrich the marine environment with certain minerals, used either in cages or food [62].
Sediment enrichment is observed in industrial areas including [89]: the Gulf of North Evoikos (mining plants), Gulf of Kavala (oil platforms and fertilizer plant), port of Stratoni (aluminium production plant), Gulf of Elefsina (industrial waste), the Saronikos Gulf (Psyttalia WWTP), the Thermaikos Gulf, and Northern Evoikos (waste from an iron–nickel smelter). In addition, sediments in or close to fish farms are enriched in heavy metals such as Zn, Cu, Cd, Pb, Ni, and Fe from food that is not consumed, secretions from organisms, and antifouling [90,91]. However, bioaccumulation of metals in organisms (mussels and red molluscs) collected from various Greek coastal areas are low and similar to that of unpolluted Mediterranean areas.
PAHs and organochlorine compounds enter the marine environment either through rivers and land outflows or through atmospheric deposition. PAHs are easily adsorbed, are transported on suspended particulate matter, and accumulate in marine sediments. High concentrations of PAHs in sediments (in some cases >10,000 μg kg−1) are observed in areas directly affected by coastal industrial activities (Antikyra Bay, Larymna Bay, Aliveri Bay, Elefsis Bay, and the Pagasitic Gulf) or urban waste (Psyttalia WWTP in the Saronikos Gulf). Organochlorine compound concentration values in marine organisms (mussels and two common species of fish) are much lower than those considered dangerous to consumers [62].
The inflow of Black Sea surface water through the Dardanelles Straits constitutes a continuous point source of radionuclides (137Cs) in the North Aegean Sea [92]. The concentrations of 226Ra in samples of marine sediments in the port of Stratoni (an area with mining activities) showed increased values, higher than the other industrial areas of Greece [89,93].
Overall, there are certain hot spots and coastal areas affected by high pressures from point and diffuse pollution sources including: the Saronikos Gulf–Psyttalia area, Gulf of Elefsis, Gulf of Thessaloniki, Gulf of Patras, Pagasitikos Gulf, Gulf of Larymna, Gulf of Antikyra, and Amvrakikos Gulf [62].
There are four criteria concerning contaminants according to Commission Decision 2017/848/EU for Descriptor 8: D8C1/contaminant concentrations; D8C2/species and habitats affected; D8C3/acute pollution events; and D8C4/acute pollution events effects. In coastal and territorial waters, most of the contaminants (D8C1) are below the detection limit at all stations, never exceeding the environmental quality standard (EQS) values. All concentrations of pollutants in sediments (D8C1) are considered very low, not enriched with toxic metals (North and South Aegean) or PAHs (polycyclic aromatic hydrocarbons) (Aegean and Levantine). They are comparable to values in non-polluted areas and can be used as a reference with the exception of some high heavy metals and PAHs values in the Saronikos Gulf (Elefsis Gulf and WWTP estuary) [62].
Species health (D8C2) cannot be correlated with pollutant concentrations due to limited data on marine organisms’ biomarkers.
In the cases of acute pollution events (D8C3/ D8C4), monitoring programs were conducted to assess the impact on the marine environment of the events that took place in 2012–2018. Complete recovery of the affected areas was observed in a period ranging from three months to one year after the accident, depending on the type and amount of oil released, oceanographic and weather conditions, and recovery efforts [94,95,96,97,98,99].
(III) Contaminants in Seafood; Descriptor 9
For Descriptor 9, there is one criterion according to Commission Decision 2017/848/EU: D9C1/contaminants’ levels in seafood. In Greece, the levels of contaminants (D9C1) in fish (Cd, Hg, and Pb in fish muscle or edible tissues) and other seafood are compliant with community legislation levels (regulation 1881/2006 and 1259/2011 amendment). PCBs and DDTs’ concentrations (not included in Regulation 1881/2006) do not exceed the maximum permissible levels for human health set by other authorities [100]. Consequently, the marine waters of Greece are in GES for D9.
(IV) Marine Litter; Descriptor 10
Marine litter (ML) enters the marine environment from land or sea-based human activities. However, it is attested that about 80% originate from land-based sources. Based on Jambeck at al. (2015) [101], it is estimated that in Greece, 2.5 to 15 KT of plastic waste per year enters the marine environment from land-based sources. Commission Decision 2017/848/EU criteria for Descriptor 10 include four criteria: D10C1/litter composition, amount, and spatial distribution; D10C2/micro litter composition, amount, and distribution; D10C3/litter ingested by marine animals; and D10C4/species individuals affected.
Regarding marine litter on the coasts (D10C1/ 10.1.1), in the North Ionian Sea, present items of ML on coasts are in most cases much lower than the values proposed by UNEP (2015) as baseline values are of 450–1400 items/100 m (DeFishGear, 2015–2016). Plastic items are by far the most common (93.23% according to DefishGear). There are no data available for the Aegean and Levantine coasts for the period 2012–2017 according to the MSFD protocol [102].
Regarding marine litter on the sea bottom (D10C1/10.1.2), all studied areas including the South Adriatic, North Ionian (around Corfu), and Central Ionian (Patraikos and Echinades gulfs) showed higher density values than those proposed by UNEP (2015) as baseline limit values for the Mediterranean (130–230/km2). The density values of ML recorded on the bottom of the Aegean Sea were lower than those recorded in the Ionian Sea, whereas in the Cretan Sea, the values found are higher than those proposed by UNEP [62].
As far as floating micro litter is concerned (D10C2/ 10.1.3), the South Adriatic and North Ionian Sea are enriched in floating microplastics as maximum concentrations (DeFishGear 2014–2015) exceed the proposed maximum value for the Mediterranean. In the southern Ionian waters, microplastics are close to or below the minimum reference value of 200,000–500,000/km2 [103]. South and Central Aegean are within the proposed range, with the exception of the Paros–Naxos Strait (654,000 pieces/km2) [104]. In addition, for the highly urbanized Saronikos Gulf, the maximum value (924,051 pieces/km2) exceeds the suggested range of base values.
For D10C3/10.2.1, the ingestion of microplastics by fish is generally comparable to the values reported in other parts of the Mediterranean for the same species [105]. The values found in the fish of the northern Ionian Sea were lower than those reported in the respective species from the Adriatic and Slovenian Croatian waters [106]. The values of microplastics ingested by Engraulis encrasicolus fish in North Evia (HCMR, 2017) are similar to those reported for the same species on the Spanish coast [107] and lower compared to the values reported in various Adriatic fish species in Slovenian and Croatian waters [106].
The average value of macro-litter ingested in the South Adriatic and North Ionian Seas was relatively small and similar to that of other studies on species in the Atlantic and Mediterranean [108]. Anastasopoulou et al. (2018) [106] found that the average ingestion values of macro-waste in fish of the North Ionian were much lower than those of the Adriatic Sea. There is no relevant data available for the Levantine Sea.
(V) Noise; Descriptor 11
The main sources of low and medium frequency impulsive sounds (D11C1) in Greek marine waters include the arrays of airguns used during seismic surveys for the exploration of hydrocarbons, the sonars used during naval exercises in military/defence activities for research purposes, and the impact of pile drivers during the expansion/reconstruction of jetties. Seismic surveys take place in the Ionian Sea, the North Aegean, and south of Crete. Sonar emissions due to military/defence activities are responsible for cetacean stranding and mortality [109]. Data is available only for seismic surveys and for the impact of pile drivers from the port operations and constructions. Following Commission Decision 2017/848/EU, there are two criteria for Descriptor 11: D11C1/levels of anthropogenic impulsive sound sources and D11C2/levels of anthropogenic continuous low-frequency sound.
The main source of anthropogenic continuous low-frequency sounds (D11C2) is shipping and marine traffic which is intense due to the high number of islands. In addition, there are corridors of marine transport connecting the Black Sea and ports in the Mediterranean. Moreover, the Greek fishing fleet is big and contributes to the sound production. The existing data concern the spatial distribution, time span, and levels of continuous low-frequency, man-made sound [62].
Thresholds follow the guidelines of the TG Noise [110] but conclusions cannot be provided for (D11C1) GES due to the fact that data are not accessible yet.
Regarding anthropogenic pulsed and continuous low-frequency sound (D11C2), GES cannot be determined as thresholds for the Greek waters are not available. In addition, the Greek seas are characterized by complex topography, by the presence of a large number of islands and islets, areas of great depth, steep slopes, and a difference in the profile of sound speed during the summer and winter months which make such an evaluation very difficult [62].
4. Aligning MSFD and MSPD
4.1. MSFD–MSPD Interrelation
MSPD with sustainability and an ecosystem approach as key principles can only attain its goals based on MSFD aspirations. MSFD provisions to achieve or maintain GES, as well as the information and data gathered under its umbrella encompassing crucial environmental directives and policies (i.e., WFD, CFP, HD and BD, and MPAs), ensure the application of the most up-to-date environmental knowledge and avoids duplication of efforts under the implementation of MSPD.
The data gathered under the 11 Descriptors of the MSFD concerning Greece can support the implementation of organizing the process through pre-planning (Step 3) and defining and analysing existing conditions (Step 5) of the proposed “Step-by-step approach for MSP” by Ehler and Douvere (2009) [3]. The MSP process is provided with all the necessary information on the state of the Greek marine environment collected under the MSFD state descriptors, the pressures on it, and the activities taking place in it or affecting it, collected under the MSFD pressure descriptors. Moreover, the MSFD Monitoring Programmes (Article 11) contribute significantly to monitoring and evaluating of the planning performance (Step 9), whereas the MSFD Programmes of Measures established (Article 13) facilitate the defining and analysing of future conditions (Step 6). Figure 2 summarizes the relations among MSFD descriptors, MSP processes, and MSP sectors under consideration.
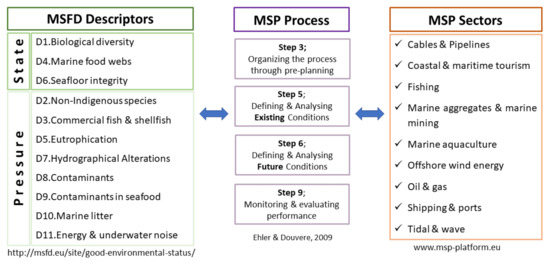
Figure 2.
Relations among MSFD descriptors, MSP processes, and MSP sectors.
Yet, the extent and means to which the MSFD information can support the MSP process in Greece needs to be addressed. Regarding extent, it can be assumed that it is strongly related to two factors: (i) the potential linkage among individual MSFD descriptors and MSP sectors which explains whether existing background information for one descriptor can constitute valuable input information for an activity and consequently a sector, and (ii) the extent to which one activity occurs in the marine environment of interest. The former has been discussed in detail above for individual descriptors, providing the available knowledge derived from the MSFD process. With respect to the analysis of the latter, one relies on existing information regarding the development of the maritime activities in Greece and the degree of their intensity in the Greek environment [111]. For instance, both fishing (trawling in particular) and marine mining activities are linked to sea floor integrity. However, in the case of Greece, fishing constitutes one of the best-developed sectors as opposed to marine mining which is at an early development stage and thus they do not equally make use of the MSFD knowledge. Therefore, based on the integration of these factors, a qualitative classification of the utility of the MSFD knowledge is reasonable and shows that the weight of each descriptor information is in a different range (low, medium, and high) (Figure 3). In fact, it seems that the highest the intensity of an MSP activity, the closest the relationship with MSFD knowledge.
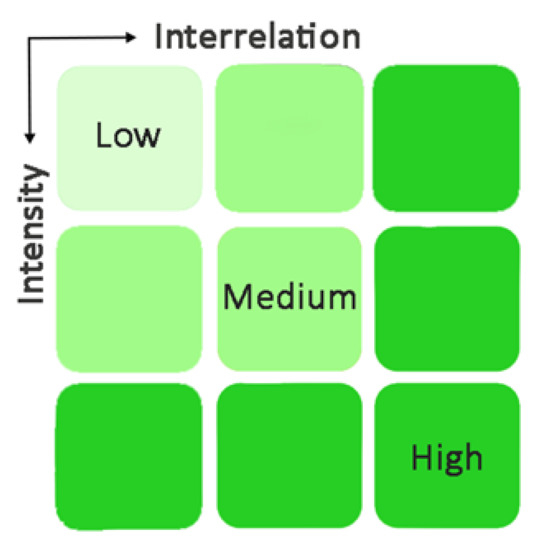
Figure 3.
Assessment of the interrelation extent of MSFD–MSPD information.
Accordingly, Figure 4 illustrates the means the information regarding MSFD descriptors and MSP sectors is interrelated in the Greek context based on the qualitative integration of the information presented in Section 3. The links developed between the MSFD descriptors and MSP Sectors depict that all MSFD descriptors feed MSP sectors more or less with environmental knowledge essential for the MSP application, while the intensity of each descriptor contribution to this interrelation is weighed. The width of the curves corresponds to the frequency of the linkage of MSFD descriptors and MSP sectors. Therefore, the descriptors contributing more intensely to MSP sectors are biological diversity and seafloor integrity (D1 and D6) and contaminants (D8 and D9). Commercial fish and shellfish (D3), eutrophication (D5), and marine litter (D10) follow with comparatively lower participation, whereas there is less contribution to the data feeding process by marine food webs (D4), non-indigenous species (D2), and energy and underwater noise (D11). Hydrological conditions (D7) are not included in this linkage as it is not assumed to directly supply information in the interrelation.
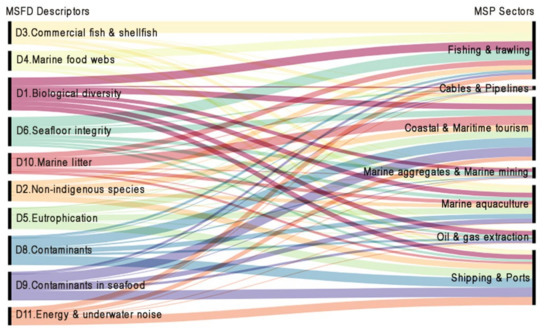
Figure 4.
The interrelations between MSFD descriptors and MSP sectors in the Greek context.
Correspondingly, the development of MSP sectors in Greek waters under the examined interrelation also supports the idea that the implementation of MSPD can be assisted to a great extent by the implementation of MSFD (Figure 4). As shown, most of the pressures in the Greek marine environment are induced by tourism and the fishing industry in addition to shipping activity. Marine aquaculture also constitutes an activity significantly affecting the coastal environment. Non-living resource exploitation, i.e., oil and gas extraction, marine aggregates, and mining, as well as cable and pipeline installation, is still not systematically developed in Greece; thus, they still have minor influence on the marine environment. Finally, tidal and wave energy and offshore wind energy are non-existing activities currently in Greece. The aforementioned, raised by this interrelation, reveal that MSP sectors’ intensity is in accordance with the existing knowledge regarding the development of current maritime activities in the Greek environment [110].
4.2. Spatial Coverage and Gaps
Adopting the ecosystem approach as required by MSPD indicates that MSP should no longer be sectorial but within ecosystem boundaries. This in turn entails that the spatial scale of management has to extend across different biological units and jurisdictions to encompass an entire ecosystem [112]. Therefore, MSFD can significantly assist the choice of scale for certain components. Overall, the different spatial scales depend on the peculiarities and characteristics of local areas, the type of planned or existing activities, and their impact on the environment.
According to Commission Decision 2017/848/EU [29], providing the assessment scale for each descriptor, the assessment of the marine environment and pressures (Article 8a/b), and the definition of GES (Article 9) are performed at different scales according to the component addressed and the specificities of each member state.
In 2018, the assessment for Greece was mainly conducted using the Marine Reporting Units (MRUs) scale for Descriptors 1, 2, 5, 6, 8, 9, and 10, i.e., for the North, Central, and South Aegean, and the Levantine Sea and Ionian Sea. In addition, the region (Mediterranean) and subregion (the Ionian/Central Mediterranean, Adriatic, and Aegean/Levantine Sea) scale was used for Descriptors 3 and 4, and the subregion scale for Descriptors 7 and 11. Specific parameters are measured by the MSFD monitoring network designated in the MSFD Monitoring Programmes [113], including 305 stations. Figure 5 presents the MSFD network indicating the stations’ monitoring parameters for state and/or pressure descriptors. A considerable extent of the Greek territorial waters is not monitored, while more extensive knowledge concerning the coastal waters cannot be extrapolated for the open sea waters. This is also due to the fact that Greece presents certain complexities; extensive coastlines both in the continental and insular part of the country; limited parts of the sea that the country has absolute jurisdiction over; and multiple isolated water territories formed due to the great dispersion of the Greek islands in the Aegean and Ionian Seas. However, currently the monitoring network is considered sufficient to support the monitoring of MSFD and thus meets the MSPD needs.
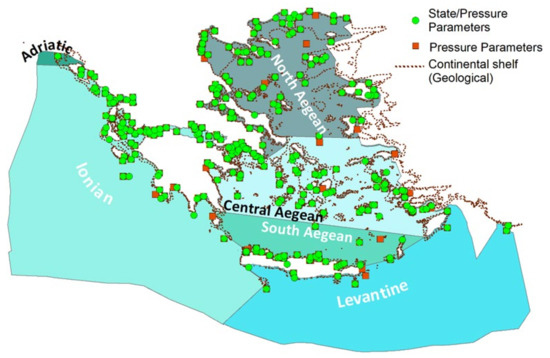
Figure 5.
Integrated Greek Monitoring Network depicting all monitoring stations (i.e., WFD, open seas MSFD, Natura 2000, Data Collection Framework, Mytimed, and Posidon stations) in which parameters are measured for state and/or pressure descriptors.
Identified hot spots and areas where GES is not achieved in the Greek marine environment, including mainly coastal areas and closed embayments or gulfs, require special attention in the course of MSP in order to regulate existing activities, progress towards GES, and identify potential new activities that could be introduced in the planning process. In addition, knowledge delivered based on the ecosystem approach enables spatial solutions for the management of specific maritime activities in Greece in a way that is compatible with the achievement of GES and capacity of the marine ecosystem to respond to human-induced changes [114], and ensures the good quality state of the sea in parallel with sustainable economic growth [115].
Despite the extensive assessment and monitoring conducted in the framework of MSFD implementation, there are still gaps at various levels of information. Nevertheless, the MSP process in Greece can make full use of the MSFD data.
Although further mapping is required to determine the full extent of highly variable habitats, MSFD Descriptors 1 and 6 provide data on species, habitats, and ecosystems that can in turn inform the MSP on the significance of the Greek marine environment in terms of biodiversity and thus allow MSP to decide upon the availability and use of marine space and regulate activities accordingly.
Descriptor 2 states that the introduction of NIS numbers appear slightly reduced. However, NIS still constitutes a considerable pressure on the marine environment in Greece as their introduction cannot be easily hindered and MSP has to regulate shipping and yachting activities in order to minimise and ultimately eliminate NIS entering the Greek waters due to anthropogenic activities.
Regarding fisheries (Descriptor 3), even though data gaps do not allow for a stock assessment of high certainty for all species, there is information on fishing mortality and spawning stock biomass, as well as stock assessments and determination of GES for certain commercial species. Based on this and by integrating the MSFD requirements [20], MSP has to regulate the fishing activity considering that populations of all commercially exploited fish and shellfish should be kept within safe biological limits, exhibiting characteristics of a healthy stock. In the future, an uninterrupted implementation of the Data Collection Framework will provide more detailed data on all stocks and marine food webs.
Regarding Descriptor 5 (eutrophication), there is extensive data on hotspot areas in terms of nutrients and organic matter loads entering the marine environment from point and diffused sources. MSP should regulate human activities contributing to eutrophication such as marine aquaculture and coastal tourism, with a view to minimising ecosystem degradation and losses in biodiversity.
The extensive data provided by Descriptors 8 and 9 (contaminants), indicating hot spot areas in terms of contaminants and highlighting the activities that generate pressures, should be considered by the MSP process to plan activities integrating the MSFD requirements [20], with the vision to maintain low or eliminate the levels of contaminants entering the Greek marine environment, degrading its quality or adversely impacting organisms. Therefore, shipping and ports, oil and gas, marine aquaculture, and marine mining should be regulated accordingly. In addition, although the data provided by Descriptor 10 (marine litter) is not spatially abundant, there is information on activities that introduce litter in the marine environment (e.g., coastal and maritime tourism, shipping and ports, fishing, and marine aquaculture) that should be considered in the planning process. Finally, based on the data produced under Descriptor 11 (noise), the main sources of anthropogenic continuous low-frequency sound that mainly disturb marine mammals are shipping, marine traffic, and the fishing fleet. Therefore, in order to maintain GES, MSP should consider where to allow maritime transport corridors and fishing.
5. Conclusions
The information gathered in the framework of the MSFD 2008/56/EC implementation procedure is assumed to constitute valuable background knowledge that can feed the MSP process in Greece. This extensive material operates as an open access information tank that can be used by those involved in the planning process as it is quite manageable and of a well-structured nature, providing a concrete presentation of the Greek marine environment.
The information was elaborated based on the assumption that it constitutes a major task in the application of a step-by-step approach for MSP (Step 5) [3]. This can be well defined by describing the state and pressures as determined by the 11 Qualitative Descriptors of MSFD. The arising interrelation between the MSFD descriptors and the MSP sectors showed that all MSFD descriptors provide valuable environmental information for MSP application, while the validity of the information ranges depending upon the intensity and the type of the activity.
Additionally, MSFD can significantly assist the choice of scale for certain components. Overall, the different spatial scales depend on the peculiarities and characteristics of local areas, the type of planned or existing activities, and their impact on the environment.
Most importantly, the identification of hot spots and areas where GES is not achieved constitutes valuable background information for the regulation of activities in the course of the MSP implementation so as to assist improvement and maintenance of GES. Therefore, although in some cases there may be spatial gaps in the monitoring network of MSFD, the MSP process can still benefit from using the MSFD data.
Author Contributions
Conceptualization, visualization, methodology, formal analysis, writing—original draft preparation, T.P.; conceptualization, visualization, methodology, formal analysis, writing—original draft preparation, A.K.; formal analysis, writing—review and editing, N.M.; formal analysis writing—review and editing, S.P. (Stelios Petrakis); writing—review and editing, P.M.; writing—review and editing, S.P. (Serafim Poulos); writing—review and editing, supervision, M.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by “HIMIOFOTS” (MIS 5002739), implemented under the action “Reinforcement of the Research & Innovation Infrastructure”, funded by the Operational Programme “Competitiveness, Entrepreneurship and Innovation” (NSRF 2014–2020) and co-financed by Greece and the European Union (ERDF).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data supporting reported results in this study are openly available at https://cdr.eionet.europa.eu/gr/eu/msfdart17/2018reporting/textreport/envxcouua/GR_PAPER_REPORT_31_12_2018.pdf accessed on 16 February 2021.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Abbreviations
| BD | Birds Directive |
| CFP | Common Fisheries Policy |
| EQS | Environmental Quality Standards |
| EU | European Union |
| FAO | Food and Agriculture Organisation |
| GES | Good Environmental Status |
| GDP | Gross Domestic Product |
| GSAs | Geographical subareas |
| HABs | Harmful Algal Blooms |
| HD | Habitats Directive |
| ICZM | Integrated Coastal Zone Management |
| MPA(s) | Marine Protected Area(s) |
| MRUs | Marine Reporting Units |
| MS(s) | Member State(s) |
| MSFD | Marine Strategy Framework Directive |
| MSP | Maritime Spatial Planning |
| MSPD | Maritime Spatial Planning Directive |
| NEAT | Nested Environmental Status Assessment Tool |
| NIS | Non-Indigenous Species |
| PAHs | Polycyclic Aromatic Hydrocarbons |
| UNEP | United Nations Environmental Programme |
| WFD | Water Framework Directive |
| WWTP | Waste Water Treatment Plant |
References
- European Commission. Directive 2014/89/EU of the European Parliament and of the Council of 23 July 2014 establishing a framework for maritime spatial planning. Off. J. Eur. Union 2014, L 257, 135. [Google Scholar]
- Douvere, F. The importance of marine spatial planning in advancing ecosystem-based sea use management. Mar. Policy 2008, 32, 762–771. [Google Scholar] [CrossRef]
- Ehler, C.; Douvere, F. Marine Spatial Planning: A Step-by-Step Approach toward Ecosystem-Based Management. Intergovernmental Oceanographic Commission and Man and the Biosphere Programme; IOC Manual and Guides No. 53; ICAM Dossier No. 6.; UNESCO: Paris, France, 2009. [Google Scholar]
- European Union. Maritime Spatial Planning in the EU—Achievements and Future Development; European Union: Brussels, Belgium, 2011; p. 12. [Google Scholar] [CrossRef]
- EC. Report from the Commission to the European Parliament and the Council on the Implementation of the Marine Strategy Framework Directive; (Directive 2008/56/EC) {COM(2020) 259 Final}—{SWD(2020) 60 Final}—{SWD(2020) 61 Final}; EC: Brussels, Belgium, 2020. [Google Scholar]
- Gorjanc, S.; Klančnik, K.; Murillas-Maza, A.; Uyarra, M.C.; Papadopoulou, N.K.; Paramana, T.; Smith, C.; Chalkiadaki, O.; Dassenakis, M.; Peterlin, M. Coordination of pollution-related MSFD measures in the Mediterranean—Where we stand now and insights for the future. Mar. Pollut. Bull. 2020, 159, 111476. [Google Scholar] [CrossRef]
- Kelly, C.; Gray, L.; Shucksmith, R.; Tweddle, J. Investigating options on how to address cumulative impacts in marine spatial planning. Ocean Coast Manag. 2014, 102, 139–148. [Google Scholar] [CrossRef]
- Eborja, A.; Elliott, M.; Andersen, J.H.; Eberg, T.; Ecarstensen, J.; Halpern, B.S.; Heiskanen, A.-S.; Ekorpinen, S.; Lowndes, J.S.S.; Emartin, G.; et al. Overview of Integrative Assessment of Marine Systems: The Ecosystem Approach in Practice. Front. Mar. Sci. 2016, 3. [Google Scholar] [CrossRef] [Green Version]
- Jones, P.J.S.; Qiu, W.; Lieberknecht, L.M. Typology of Conflicts in MESMA Case Studies; MESMA Work Package 6 (Governance); Deliverable 6.1; University College London: London, UK, 2013. [Google Scholar]
- Boyes, S.J.; Elliott, M. Marine legislation—The ultimate ‘horrendogram’: International law, European directives & national implementation. Mar. Pollut. Bull. 2014, 86, 39–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murillas-Maza, A.; Uyarra, M.C.; Papadopoulou, N.K.; Smith, C.; Gorjanc, S.; Klančnik, K.; Paramana, T.; Chalkiadaki, O.; Dassenakis, M.; Pavicic, M. Programmes of measures of the marine strategy framework directive: Are they contributing to achieving good environmental status in the Mediterranean? Mar. Pollut. Bull. 2020, 161, 111715. [Google Scholar] [CrossRef]
- Gilbert, A.J.; Alexander, K.; Sardá, R.; Brazinskaite, R.; Fischer, C.; Gee, K.; Jessopp, M.; Kershaw, P.; Los, H.J.; Morla, D.M.; et al. Marine spatial planning and Good Environmental Status: A perspective on spatial and temporal dimensions. Ecol. Soc. 2015, 20, 64. [Google Scholar] [CrossRef] [Green Version]
- WWF-European Policy Office. Guidance Paper, Ecosystem Based Maritime Spatial Planning in Europe and How to Assess It; WWF European Policy Office: Brussels, Belgium, 2021. [Google Scholar]
- Abramic, A.; Nogueira, N.; Sepulveda, P.; Cavallo, M.; Fernández-Palacios, Y.; Andrade, C.; Kaushik, S.; Haroun, R. Implementation of the Marine Strategy Framework Directive in Macaronesia and synergies with the Maritime Spatial Planning process. Mar. Policy 2020, 122, 104273. [Google Scholar] [CrossRef]
- Altvater, S. EBA in MSP—A SEA Inclusive Handbook; Pan Baltic Scope: Federal Maritime and Hydrographic Agency (BSH): Rostock, Germany, 2019; ISBN 978-3-86987-990-1. [Google Scholar]
- Gee, K.; Kannen, A.; Heinrichs, B. BaltSeaPlan Vision 2030—Towards the Sustainable Planning of Baltic Sea Space. 2011. Available online: http://www.baltseaplan.eu/index.php/BaltSeaPlan-Vision-2030;859/1 (accessed on 17 February 2021).
- Paramana, T.; Chalkiadaki, O.; Katsouras, G.; Dassenakis, M. Implementing the 1st MSFD cycle in the Mediterranean: Lessons learnt. In Proceedings of the 14th MEDCOAST Congress on Coastal and Marine Sciences, Engineering, Management and Conservation, MEDCOAST 2019, Marmaris, Turkey, 22–26 October 2019; Volume 1, pp. 33–43. [Google Scholar]
- Maccarrone, V.; Filiciotto, F.; de Vincenzi, G.; Mazzola, S.; Buscaino, G. An Italian proposal on the monitoring of underwater noise: Relationship between the EU Marine Strategy Framework Directive (MSFD) and marine spatial planning directive (MSP). Ocean Coast. Manag. 2015, 118, 215–224. [Google Scholar] [CrossRef]
- Dassenakis, M.; Poulos, S.; Megalofonou, P.; Paramana, T.; Karditsa, A.; Petrakis, S.; Milatou, N. Environmental status of Lakonikos Gulf in connection to MSP. In Proceedings of the 14th MEDCOAST Congress on Coastal and Marine Sciences, Engineering, Management and Conservation, MEDCOAST 2019, Marmaris, Turkey, 22–26 October 2019; Volume 1, pp. 361–372. [Google Scholar]
- European Union. MSFD 2008/56/EC of the European Parliament and of the Council of 17 June 2008 establishing a framework for Community action in the field of marine environmental policy. Off. J. Eur. Union 2008, L 164, 25. [Google Scholar]
- Eionet Central Data Repository. Available online: https://cdr.eionet.europa.eu/ (accessed on 16 February 2021).
- Poulos, S.E.; Karditsa, A. Applied & Environmental Oceanography. An introduction to Coastal Zone Management; DISIGMA: Thessaloniki, Greece, 2020; p. 512. ISBN 978–618-5242–93-0. (In Greek) [Google Scholar]
- Coccossis, H.; Stefani, F.; Lagiou, E.; Asprogerakas, E.; Lalou, E. Greece: Development of a Governance Scheme and Monitoring Mechanism; UTH: Thessaly, Greece; YPEN: Athens, Greece, 2020. [Google Scholar]
- Poulos, S.; Kotinas, V. Physio-geographical characteristics of the marine regions and their catchment areas of the Mediterranean Sea and Black Sea marine system. Phys. Geogr. 2020, 1–37. [Google Scholar] [CrossRef]
- SoHelME. State of the Hellenic Marine Environment; Papathanassiou, E., Zenetos, A., Eds.; HCMR Publications: Athens, Greece, 2005; p. 360. ISBN 960-86651-8. [Google Scholar]
- Poulos, S.E. Origin and distribution of the terrigenous component of the unconsolidated surface sediment of the Aegean floor: A synthesis. Cont. Shelf Res. 2009, 29, 2045–2060. [Google Scholar] [CrossRef]
- Ministry of Environment, Energy & Climate Change. National Biodiversity Strategy & Action Plan; Ministry of Environment, Energy & Climate Change: Athens, Greece, 2014.
- Birdlfe International. Available online: http://datazone.birdlife.org (accessed on 7 June 2021).
- European Commission (EC). Commission Decision (EU) 2017/848 of 17 May 2017 laying down criteria and methodological standards on good environmental status of marine waters and specifications and standardized methods for monitoring and assessment, and repealing Decision 2010/477/EU. Off. J. Eur. Union 2017, L 125, 43–74. [Google Scholar]
- Revees, R.; Notarbartolo di Sciara, G. The Status and Distribution of Cetaceans in the Black Sea and Mediterranean Sea; IUCN Centre for Mediterranean Cooperation: Malaga, Spain, 2006; p. 137. [Google Scholar]
- Frantzis, A. Fisheries interactions with Cetacean species in Hellas. In State of Hellenic Fisheries; Papaconstantinou, C., Zenetos, A., Vassilopoulou, V., Tsepres, G., Eds.; HCMR: Anavyssos, Greece, 2007; pp. 275–279. [Google Scholar]
- EIONET Directive 92/43/EEC, Article 17 Web Tool—Species Assessments. 2008. Available online: http://biodiversity.eionet.europa.eu/article17/speciesreport (accessed on 5 May 2021).
- Legakis, A.; Maragou, P. The Red Data Book of Endangered Animals of Greece; Hellenic Zoological Society: Athens, Greece, 2009; p. 525. [Google Scholar]
- Paximadis, G.; Frantzis, A. Whales, porpoises and dolphins. In Red Data Book of Greek Animals; Legakis, A., Maragou, P., Eds.; Hellenic Zoological Society: Athens, Greece, 2009; pp. 366–369. [Google Scholar]
- Milani, C.B.; Vella, A.; Vidoris, P.; Christidis, A.; Koutrakis, E.; Frantzis, A.; Miliou, A.; Kallianiotis, A. Cetacean stranding and diet analyses in the North Aegean Sea (Greece). J. Mar. Biol. Assoc. UK 2017, 98, 1011–1028. [Google Scholar] [CrossRef]
- Margaritoulis, D.; Argano, R.; Baran, I.; Bantivenga, F.; Brandai, M.N.; Caminas, J.A.; Casale, P.; De Metrio, G.; Dimitropoulos, A.; Gerosa, G.; et al. Loggerhead turtles in the Mediterranean Sea: Present knowledge and conservation perspectives. In Loggerhead Sea Turtles; Bolten, A.B., Witherington, B.E., Eds.; Smithsonian Books: Washington, DC, USA, 2003; pp. 175–198. [Google Scholar]
- Margaritoulis, D.; Panagopoulou, A.; Rees, A. Loggerhead nesting in Rethymno, Island of Crete, Greece: Fifteen-year nesting data (1990–2004) indicate a declining population. In Proceedings of the Second Mediterranean Conference on Marine Turtles, Kemer, Turkey, 4–7 May 2005; pp. 116–119. [Google Scholar]
- Dendrinos, P.; Karamanlidis, A.; Paravas, V. Mediterranean monk seal. In Red Data Book of Greek Animals; Legakis, A., Maragou, P., Eds.; Hellenic Zoological Society: Athens, Greece, 2009; p. 525. [Google Scholar]
- Johnson, W.M.; Karamanlidis, A.A.; Dedrinos, P.; de Larrinoa, P.F.; Gazo, M.; Gonzalez, L.M.; Guecluesoy, H.; Pires, R.; Schnellmann, M. Monk Seal Fact Files. Biology, Behavior, Status and Conservation of the Mediterranean Monk Seal, Monachus Monachus. 2006. The Monachus Guardian. Available online: http://www.monachus-guardian.org/factfiles/medit01.htm (accessed on 5 May 2021).
- Aguilar, A.; Lowry, L. Monachus monachus. In IUCN Red List of Threatened Species; Version 2009; IUCN: Gland, Switzerland, 2008; Volume 1. [Google Scholar]
- Simboura, N.; Tsapakis, M.; Pavlidou, A.; Assimakopoulou, G.; Pagou, K.; Kontoyannis, H.; Zeri, C.; Krasakopoulou, E.; Rousselaki, E.; Katsiaras, N.; et al. Assessment of the environmental status in Hellenic coastal waters (Eastern Mediterranean): From the Water Framework Directive to the Marine Strategy Water Framework Directive. Mediterr. Mar. Sci. 2014, 16, 46–64. [Google Scholar] [CrossRef] [Green Version]
- Orfanidis, S.; Panayotidis, P.; Ugland, K.I. Ecological Evaluation Index continuous formula (EEI-c) application: A step forward for functional groups, the formula and reference condition values. Mediterr. Mar. Sci. 2011, 12, 199. [Google Scholar] [CrossRef] [Green Version]
- Orfanidis, S.; Papathanasiou, V.; Mittas, N.; Theodosiou, T.; Ramfos, A.; Tsioli, S.; Kosmidou, M.; Kafas, A.; Mystikou, A.; Papadimitriou, A. Further improvement, validation, and application of CymoSkew biotic index for the ecological status as-sessment of the Greek coastal and transitional waters. Ecol. Indic. 2020, 118, 106727. [Google Scholar] [CrossRef]
- Tsikliras, A.C.; Dinouli, A.; Tsalkou, E. Exploitation trends of the Mediterranean and Black Sea fisheries. Acta Adriat. 2013, 54, 273–282. [Google Scholar]
- Tsagarakis, K.; Palialexis, A.; Vassilopoulou, V. Mediterranean fishery discards: Review of the existing knowledge. ICES J. Mar. Sci. 2013, 71, 1219–1234. [Google Scholar] [CrossRef] [Green Version]
- Tzanatos, E.; Raitsos, D.E.; Triantafyllou, G.; Somarakis, S.; Tsonis, A.A. Indications of a climate effect on Mediterranean fisheries. Clim. Change 2013, 122, 41–54. [Google Scholar] [CrossRef]
- Tsikliras, A.; Dinouli, A.; Tsiros, V.-Z.; Tsalkou, E. The Mediterranean and Black Sea Fisheries at Risk from Overexploitation. PLoS ONE 2015, 10, e0121188. [Google Scholar] [CrossRef] [Green Version]
- Stergiou, K.I.; Somarakis, S.; Triantafyllou, G.; Tsiaras, K.P.; Giannoulaki, M.; Petihakis, G.; Machias, A.; Tsikliras, A.C. Trends in productivity and biomass yields in the Mediterranean Sea large marine ecosystem during climate change. Environ. Dev. 2016, 17, 57–74. [Google Scholar] [CrossRef]
- Colloca, F.; Scarcella, G.; Libralato, S. Recent Trends and Impacts of Fisheries Exploitation on Mediterranean Stocks and Ecosystems. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Food and Agriculture Organization of the United Nations. Reviving Ukraine’s Breadbasket; FAO: Kyiv, Ukraine, 2018. [Google Scholar]
- Micheli, F.; Halpern, B.S.; Walbridge, S.; Ciriaco, S.; Ferretti, F.; Fraschetti, S.; Lewison, R.; Nykjaer, L.; Rosenberg, A.A. Cumulative Human Impacts on Mediterranean and Black Sea Marine Ecosystems: Assessing Current Pressures and Opportunities. PLoS ONE 2013, 8, e79889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- CIESM. The Mediterranean Science Commission. Available online: http://www.ciesm.org/gis/JW/build/JellyBlooms.php (accessed on 7 June 2021).
- Tsagarakis, K.; Coll, M.; Giannoulaki, M.; Somarakis, S.; Papaconstantinou, C.; Machias, A. Food-WEB traits of the North Aegean Sea ecosystem (Eastern Mediterranean) and comparison with other Mediterranean ecosystems. Estuar. Coast. Shelf Sci. 2010, 88, 233–248. [Google Scholar] [CrossRef]
- Moutopoulos, D.K.; Libralato, S.; Solidoro, C.; Stergiou, K.I. Toward an ecosystem approach to fisheries in the Mediterranean Sea: Multi-gear/multi-species implications from an ecosystem model of the Greek Ionian Sea. J. Mar. Syst. 2013, 113–114, 13–28. [Google Scholar] [CrossRef]
- Pyrounaki, M.M.; Giannoulaki, M.; Tsagarakis, K.; Machias, A. ; Biomass estimation of anchovy (Engraulis encrasicolus) and sardine (Sardina pilchardus) in the eastern Ionian Sea using acoustic data. In Proceedings of the 16th Panhellenic Conference of Ichthyologists, Kavala, Greece, 6–9 October 2016; pp. 157–160. [Google Scholar]
- ICCAT. BFT Stock Assessment Meeting—Madrid 2017. In Proceedings of the 2017 ICCAT Bluefin Stock Assessment Meeting, Madrid, Spain, 20–28 July 2017; p. 106. Available online: https://www.iccat.int/Documents/Meetings/Docs/2017_BFT_ASS_REP_ENG.pdf (accessed on 12 June 2021).
- ICCAT. SWO MED Stock Assessment. In Proceedings of the 2016 Mediterranean Swordfish Stock Assessment Meeting, Casablanca, Morocco, 11–16 July 2016; p. 60. Available online: https://www.iccat.int/Documents/Meetings/Docs/2016_SWO_MED_SA_REPORT_ENG.pdf (accessed on 12 May 2021).
- Ministry of Rural Development and Food. Available online: http://www.alieia.minagric.gr/node/32 (accessed on 8 June 2021).
- Telesca, L.; Belluscio, A.; Criscoli, A.; Ardizzone, G.; Apostolaki, E.T.; Fraschetti, S.; Gristina, M.; Knittweis, L.; Martin, C.S.; Pergent, G. Seagrass meadows (Posidonia oceanica) distribution and trajectories of change. Sci. Rep. 2015, 5, srep12505. [Google Scholar] [CrossRef] [Green Version]
- Zenetos, A.; Pancucci-Papadopoulou, M.A.; Zogaris, S.; Papastergiadou, E.; Vardakas, L.; Aligizaki, K.; Economou, A.N. Aquatic alien species in Greece: Tracking sources, patterns and effects on the ecosystem. J. Biol. Res. 2009, 12, 135–172. [Google Scholar]
- Zenetos, A.; Karachle, P.K.; Corsini-Foka, M.; Gerovasileiou, V.; Simboura, N.; Xentidis, N.J.; Tsiamis, K. Is the trend in new introductions of marine non-indigenous species a reliable criterion for assessing good environmental status? Τhe case study of Greece. Mediterr. Mar. Sci. 2020, 21, 775–793. [Google Scholar] [CrossRef]
- Ministry of Environment & Energy, Special Secretariat for Waters. Marine Strategy Framework Directive (2008/56/ΕC) First Six Years Cycle of Implementation Articles 8 (A, B), 9 & 10 and Requirements of the New Directive (2017/845/ΕC); Technical Report; Ministry of Environment & Energy, Special Secretariat for Waters: Athens, Greece, 2018.
- Greek Bathing Water Profiles Registry. Available online: www.bathingwaterprofiles.gr (accessed on 22 April 2021).
- Katsanevakis, S.; Wallentinus, I.; Zenetos, A.; Leppäkoski, E.; Çinar, M.E.; Oztürk, B.; Grabowski, M.; Golani, D.; Cardoso, A.C. Impacts of invasive alien marine species on ecosystem services and biodiversity: A pan-European review. Aquat. Invasions 2014, 9, 391–423. [Google Scholar] [CrossRef]
- Kapiris, K.; Mytilineou, C.; Kavadas, S.; Capezzuto, F. Age and Growth of the Deep Water Rose Shrimp Parapenaeus longirostris in the Hellenic Ionian Sea; CIESM: Madrid, Spain, 2016. [Google Scholar]
- Giovos, I.; Keramidas, I. Recreational fishing in Greece through social media. In Proceedings of the 16th Panhellenic Conference of Ichthyologists, Kavala, Greece, 6–9 October 2016. [Google Scholar]
- Food and Agriculture Organisation of the United Nations. Available online: http://www.fao.org/gfcm/data/map-fisheries-restricted-areas/en/ (accessed on 11 June 2021).
- Moutopoulos, D.K.; Tsikliras, A.C.; Stergiou, K.I. Reconstruction of Greek Fishery Catches by Fishing Gear and Area (1950–2010); Working Paper Series 11; Fisheries Centre, The University of British Columbia: Vancouver, BC, Canada, 2015; p. 14. [Google Scholar]
- Ministry of Rural Development and Food Directorate. Greek Fishing Fleet 2019 Annual Report Pursuant to Article 22 of Regulation (EU) No 1380/2013 of the European Parliament and of the Council; Ministry of Rural Development and Food Directorate: Athens, Greece, 2020.
- Eletaen. Institutional Framework for Marine Wind Parks: The International Experience and the Basics Planning Principles for Greece; Draft Report; Hellenic Republic Ministry of Development and Investments: Athens, Greece, 2021. [Google Scholar]
- Karditsa, A.; Poulos, S.; Velegrakis, A.; Adreadis, O.; Rigos, A.; Alexandrakis, A.; Petrakis, S.; Ghionis, G. Development of an Inventory of Hellenic Beaches. Rapport du Commission Internationale Mer Mediterranee CIESM. In Proceedings of the 41th CIESM Congress, Kiel, Germany, 12–16 September 2016. [Google Scholar]
- Alexandrakis, G.; Karditsa, A.; Poulos, S.; Ghionis, G.; Kampanis, N.A. Vulnerability assessment for the erosion of the coastal zone to a potential seal level rise: The case of the Aegean Hellenic Coast, in Environmental Systems. In Encyclopedia of Life Support Systems (EOLSS); Developed under the Auspices of the UNESCO; Eolss Publishers: Oxford, UK, 2010. [Google Scholar]
- Karditsa, A.; Tsapanou, A.; Poulos, S.E. The evolution of the transboundary Evros river delta (Northeast Aegean Sea) under human intervention: A seven-decade analysis. Phys. Geogr. 2020, 41, 291–314. [Google Scholar] [CrossRef]
- Petropoulos, G.; Kalivas, D.; Griffiths, H.M.; Dimou, P.P. Remote sensing and GIS analysis for mapping spatio-temporal changes of erosion and deposition of two Mediterranean river deltas: The case of the Axios and Aliakmonas rivers, Greece. Int. J. Appl. Earth Obs. Geoinf. 2015, 35, 217–228. [Google Scholar] [CrossRef]
- Ghionis, G.; Poulos, S.E.; Karditsa, A. Deltaic coastline retreat due to dam construction: The case of the River Alfios mouth area (Kyparissiakos Gulf, Ionian Sea). J. Sea Res. 2013, 65, 1. [Google Scholar] [CrossRef]
- WWF Hellas. Blue Growth in the Mediterranean Sea: The Challenge of Good Environmental Status; WWF Hellas: Athens, Greece, 2015; p. 318. [Google Scholar]
- Stamatakis, M.; Poulos, S.; Tsoutsia, A.; Anastasatou, M.; Karditsa, K.; Koukounari, I.; Papavlassopoulou, N.; Velegrakis, A.F.; Hasiotis, T.; Evaggelopoulos, A.; et al. Marine Aggregates Pro-specting and Exploitation (MARE project) in Greek waters: Μethods, Environmental Impact and Usage possibilities. In Proceedings of the 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene, Greece, 13–17 May 2015; pp. 1053–1056. [Google Scholar]
- Kapsimalis, V.; Rousakis, G.; Panagiotopoulos, I.; Hasiotis, T.; Tsoutsia, A.; Anastasatou, M.; Stamatakis, M.; Petrakis, S.; Karditsa, A.; Poulos, S. Searching for Marine Aggregate deposits on the continental shelf of SE Euboea, Greece. In Proceedings of the 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene, Greece, 13–17 May 2015; pp. 1065–1068. [Google Scholar]
- Anastasatou, M.; Karditsa, A.; Petrakis, S.; Tsoutsia, A.; Kapsimalis, V.; Foutrakis, P.; Hasiotis, T.; Rousakis, G.; Evangelopoulos, A.; Poulos, S.; et al. Marine Aggregates Deposits of Kissamos Bay, Crete, Greece. In Proceedings of the 14th International MEDCOAST Congress on Coastal and Marine Sciences, Engineering, Management & Conservation, Marmaris, Turkey, 22–26 October 2019. [Google Scholar]
- Velegrakis, A.; Karditsa, A.; Paramana, T.; Poulos, S.; Anastasatou, M.; Tsoutsia, A.; Papavlassopoulou, N.; Stamatakis, M.; Hasiotis, T. Marine Aggregates extraction regulations in Greece in accordance to European Practices. In Proceedings of the 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene, Greece, 13–17 May 2015; pp. 1073–1076. [Google Scholar]
- Hasiotis, T.; Gazis, I.-Z.; Anastasatou, M.; Manoutsoglou, E.; Velegrakis, A.F.; Kapsimalis, V.; Karditsa, A.; Stamatakis, M. Searching for potential marine sand resources to mitigate beach erosion in island settings. Mar. Georesour. Geotechnol. 2021, 39, 527–542. [Google Scholar] [CrossRef]
- Radzevičius, R.; Velegrakis, A.; Bonne, W.; Kortekaas, S.; Gare, E.; Blažauskas, N.; Asariotis, R. Marine Aggregate Extraction Regulation in EU Member States. J. Coast. Res. 2020, 51, 15–38. [Google Scholar]
- HCMR. Study of the Environmental Impact of Dumping Coarse Metalliferous Waste in N. Evvoikos Gulf; Technical Report; Tsangaris, C., Ed.; HCMR: Athens, Greece, 2013. [Google Scholar]
- Eigaard, O.R.; Bastardie, F.; Hintzen, N.; Buhl-Mortensen, L.; Mortensen, P.; Catarino, R.; Dinesen, G.E.; Egekvist, J.; Fock, H.O.; Geitner, K.; et al. The footprint of bottom trawling in European waters: Distribution, intensity, and seabed integrity. ICES J. Mar. Sci. 2017, 74, 847–865. [Google Scholar] [CrossRef] [Green Version]
- Borja, A.; Elliott, M.; Carstensenc, J.; Heiskanen, A.S.; Woutervan de Bunde. Marine management—Towards an integrated implementation of the European Marine Strategy Framework and the Water Framework Directives. Mar. Pollut. Bull. 2010, 60, 2175–2186. [Google Scholar] [CrossRef] [PubMed]
- Pavlidou, A.; Souvermezoglou, E. Nutrients in the Mediterranean Sea. Pollution in the Mediterranean Sea: An Overview from IASON SSA; HCMR: Athens, Greece, 2006; pp. 5–12. [Google Scholar]
- Pavlidou, A.; Simboura, N.; Rousselaki, E.; Tsapakis, M.; Pagou, K.; Drakopoulou, P.; Assimakopoulou, G.; Kontoyiannis, H.; Panayotidis, P. Methods of eutrophication assessment in the context of the water framework directive: Examples from the Eastern Mediterranean coastal areas. Cont. Shelf Res. 2015, 108, 156–168. [Google Scholar] [CrossRef]
- Varkitzi, I. Pelagic habitats in the Mediterranean Sea: A review of Good Environmental Status (GES) determination for plankton components and identification of gaps and priority needs to improve coherence for the MSFD implementation. Ecol. Indic. 2018, 95, 203–218. [Google Scholar] [CrossRef] [Green Version]
- Simboura, N.; Maragou, P.; Paximadis, G.; Kapiris, K.; Papadopoulos, V.P.; Sakellariou, D.; Pavlidou, A.; Hatzianestis, I.; Salomidi, M.; Arvanitidis, C.; et al. Greece. In World Seas. An Environmental Evaluation, 2nd ed.; Elsevier, Ltd.: Amsterdam, The Netherlands, 2019; pp. 227–260. [Google Scholar] [CrossRef]
- Basaran, A.K.; Aksu, M.; Egemen, Ö. Impacts of the fish farms on the water column nutrient concentrations and accumulation of heavy metals in the sediments in the eastern Aegean Sea (Turkey). Environ. Monit. Assess. 2009, 162, 439–451. [Google Scholar] [CrossRef]
- Belias, C.; Bikas, V.G.; Dassenakis, M.J.; Scoulos, M.J. Environmental Impacts of Coastal Aquaculture in Eastern Mediterranean Bays. The Case of Astakos Gulf, Greece. Environ. Sci. Pollut. Res. 2003, 10, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Tsabaris, C.; Zervakis, V.; Kaberi, H.; Delfanti, R.; Georgopoulos, D.; Lampropoulou, M.; Kalfas, C. 137Cs vertical distribution at the deep basins of the North and Central Aegean Sea, Greece. J. Environ. Radioact. 2014, 132, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Pappa, P. Radioactivity and metal concentrations in marine sediments associated with mining activities in Ierissos Gulf, North Aegean Sea, Greece. Appl. Radiat. Isot. 2016, 116, 22–33. [Google Scholar] [CrossRef] [PubMed]
- HCMR. Study of the Effects of the Pollution Caused by the Accident of the M/V “YUSUF CEPNIOGLOU” in the Marine Area of the North Mykonos. Results of the Measurements in June–August 2014; Technical Report; HCMR: Athens, Greece, 2014. [Google Scholar]
- HCMR. Study and Evaluation of Open Sea Ecological Status Around Crete Island; Final Report; HCMR: Athens, Greece, 2015. [Google Scholar]
- HCMR. PERSEUS Final Scientific Report Subtask 1.3.5; HCMR: Athens, Greece, 2015. [Google Scholar]
- HCMR. Study of the Effects of the Pollution Caused by the Accident of the M/V “YUSUF CEPNIOGLOU” in the Marine Area of the North Mykonos. Results of the Measurements in September 2015; Technical Report; HCMR: Athens, Greece, 2015. [Google Scholar]
- HCMR. Investigation of Possible Pollutant Substances Release from the M/V CABRERA Wreck in the Marine Environment of Northern Andros; Technical Report; HCMR: Athens, Greece, 2017. [Google Scholar]
- HCMR. Study of the Effects of the Pollution Caused by the AGHIA ZONI II Accident in Saronikos Gulf; Final Report; HCMR: Athens, Greece, 2018. [Google Scholar]
- Hatzianestis, A. Levels and temporal trends of organochlorine compounds in marine organisms from Greek waters. Rapp. Comm. Int. Mer. Médit. 2016, 41, 1. [Google Scholar]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Vlachogianni, T.; Anastasopoulou, A.; Fortibuoni, T.; Ronchi, F.; Zeri, C. Marine Litter Assessment in the Adriatic and Ionian Seas; IPA-Adriatic DeFishGear Project; HCMR: Anavyssos, Greece, 2017; p. 168. ISBN 978-960-6793-25-7. [Google Scholar]
- UNEP/MAP. UNEP/MAP Marine Litter Assessment in the Mediterranean; UNEP/MAP: Athens, Greece, 2015; ISBN 978-92-807-3564-2. [Google Scholar]
- Adamopoulou, A.; Zeri, C.; Kafetzis, A. Floating microplastics in the Greeks Seas. In Proceedings of the 12th Panhellenic Symposium of Oceanography & Fisheries, Corfu, Greece, 30 May–3 June 2018; Available online: https://www.symposia.gr/wp-content/uploads/2018/07/Book_of_Abstracts.pdf (accessed on 14 May 2021).
- Digka, N.; Tsangaris, C.; Torre, M.; Anastasopoulou, A.; Zeri, C. Microplastics in mussels and fish from the Northern Ionian Sea. Mar. Pollut. Bull. 2018, 135, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Anastasopoulou, A.; Viršek, M.C.; Varezić, D.B.; Digka, N.; Fortibuoni, T.; Koren, Š.; Mandić, M.; Mytilineou, C.; Pešić, A.; Ronchi, F.; et al. Assessment on marine litter ingested by fish in the Adriatic and NE Ionian Sea macro-region (Mediterranean). Mar. Pollut. Bull. 2018, 133, 841–851. [Google Scholar] [CrossRef]
- Compa, M.; Ventero, A.; Iglesias, M.; Deudero, S. Ingestion of microplastics and natural fibres in Sardina pilchardus (Walbaum, 1792) and Engraulis encrasicolus (Linnaeus, 1758) along the Spanish Mediterranean coast. Mar. Pollut. Bull. 2018, 128, 89–96. [Google Scholar] [CrossRef]
- Bellas, J.; Martínez-Armental, J.; Martínez-Cámara, A.; Besada, V.; Martínez-Gómez, C. Ingestion of microplastics by demersal fish from the Spanish Atlantic and Mediterranean coasts. Mar. Pollut. Bull. 2016, 109, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Foskolos, I.; Gkikopoulou, K.; Frantzis, A. Current State of Knowledge and Conservation Perspectives on the Cetaceans of the Aegean Sea in The Aegean Sea Environment: The Natural System; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Dekeling, R.P.A. Monitoring Guidance for Underwater Noise in European Seas, Parts I-II-III; JRC Scientific and Policy Report EUR 26557 EN; Publications Office of the European Union: Luxembourg, 2014. [Google Scholar]
- European MSP Platform. Available online: https://www.msp-platform.eu/sites/default/files/download/greece_may_2021.pdf (accessed on 14 June 2021).
- Wang, H. Ecosystem Management and Its Application to Large Marine Ecosystems: Science, Law, and Politics. Ocean Dev. Int. Law 2004, 35, 41–74. [Google Scholar] [CrossRef]
- Republic of Greece Ministry of Environment and Energy. Report on the Monitoring Programs of Greece for the Implementation of the Article 11 of the Marine Strategy Framework Directive (2008/56/EC); Republic of Greece Ministry of Environment and Energy: Athens, Greece, 2016. [Google Scholar]
- Ansong, J.; Gissi, E.; Calado, H. An approach to ecosystem-based management in maritime spatial planning process. Ocean Coast Manag. 2017, 141, 65–81. [Google Scholar] [CrossRef] [Green Version]
- McLeod, K.; Lubchenco, J.; Palumbi, S.; Rosenberg, A. Scientific Consensus Statement on Marine Ecosystem-Based Management; Signed by 221 Academic Scientists and Policy Experts with Relevant Expertise. 2005. Communication Partnership for Science and the Sea. Available online: http://compassonline.org/?q=EBM (accessed on 29 June 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).