Phosphorus Release from Sediments in a Raw Water Reservoir with Reduced Allochthonous Input
Abstract
:1. Introduction
1.1. Phosphorus Biogeochemistry and Indexes
1.2. Shifting from External to Internal P Sources
2. Materials and Methods
2.1. Study Area
2.2. Sampling and Analysis
2.3. Sorption Index and P Saturation
2.4. Sorption Experiments Fordetermination of EPC0
2.5. Phosphorus Release Experiment
2.6. Data Statistics
3. Results and Discussion
3.1. Sediment Core Characteristics and Variablity
3.2. Governing Factors for Adsorption and Desorption of P in the Sediments
3.3. Phosphorus Adsorption Isotherms
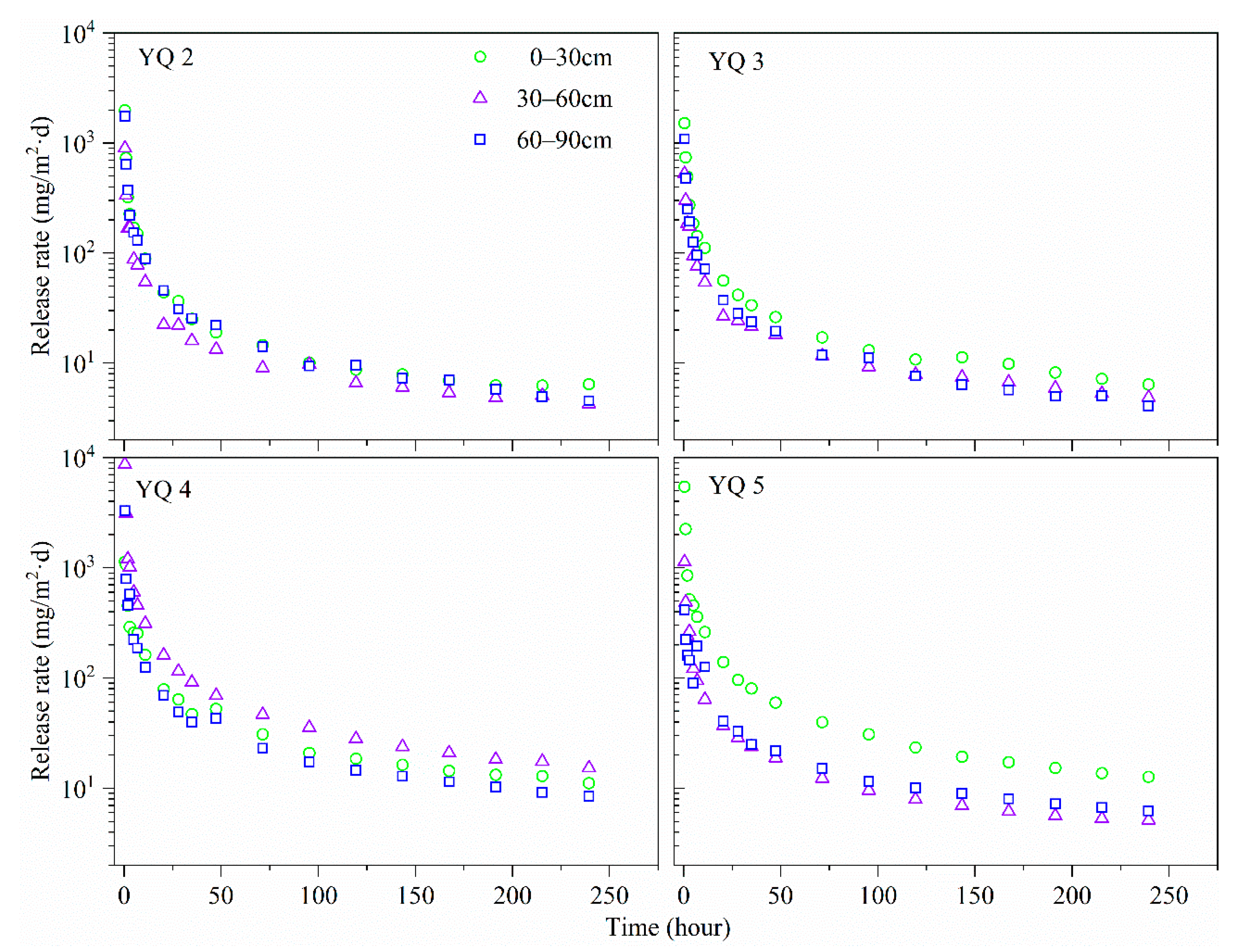
3.4. Phosphorus Release Rate from Sediment Cores
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, Q.; Chen, J.G.; Wang, J.F.; Guo, J.Y.; Jin, Z.X.; Yu, P.P.; Ma, Z.Z. In situ, high–resolution evidence of phosphorus release from sediments controlled by the reductive dissolution of iron–bound phosphorus in a deep reservoir, southwestern China. Sci. Total Environ. 2019, 666, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Nowlin, W.H.; Evarts, J.L.; Vanni, M.J. Release rates and potential fates of nitrogen and phosphorus from sediments in a eutrophic reservoir. Freshw. Biol. 2005, 50, 301–322. [Google Scholar] [CrossRef]
- Cerco, C.F.; Noel, M.R. Impact of Reservoir Sediment Scour on Water Quality in a Downstream Estuary. J. Environ. Qual. 2016, 45, 894–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maavara, T.; Parsons, C.T.; Ridenour, C.; Stojanovic, S.; Durr, H.H.; Powley, H.R.; Van Cappellen, P. Global phosphorus retention by river damming. Proc. Natl. Acad. Sci. USA 2015, 112, 15603–15608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sondergaard, M.; Jensen, J.P.; Jeppesen, E. Role of sediment and internal loading of phosphorus in shallow lakes. Hydrobiologia 2003, 506, 135–145. [Google Scholar] [CrossRef]
- Corzo, A.; Jiménez–Arias, J.L.; Torres, E.; García-Robledo, E.; Lara, M.; Papaspyrou, S. Biogeochemical changes at the sediment–water interface during redox transitions in an acidic reservoir: Exchange of protons, acidity and electron donors and acceptors. Biogeochemistry 2018, 139, 241–260. [Google Scholar] [CrossRef] [Green Version]
- Santschi, P.; Höhener, P.; Benoit, G.; Buchholtz-ten Brink, M. Chemical processes at the sediment-water interface. Mar. Chem. 1990, 30, 269–315. [Google Scholar] [CrossRef]
- Wen, S.L.; Wang, H.W.; Wu, T.; Yang, J.; Jiang, X.; Zhong, J.C. Vertical profiles of phosphorus fractions in the sediment in a chain of reservoirs in North China: Implications for pollution source, bioavailability, and eutrophication. Sci. Total Environ. 2020, 704, 135318. [Google Scholar] [CrossRef]
- Zeng, J.; Han, G. Preliminary copper isotope study on particulate matter in Zhujiang River, southwest China: Application for source identification. Ecotoxicol. Environ. Saf. 2020, 198, 110663. [Google Scholar] [CrossRef] [PubMed]
- Boström, B.; Andersen, J.M.; Fleischer, S.; Jansson, M. Exchange of phosphorus across the sediment-water interface. Hydrobiologia 1988, 170, 229–244. [Google Scholar] [CrossRef]
- Wu, Y.H.; Wen, Y.J.; Zhou, J.X.; Wu, Y.Y. Phosphorus release from lake sediments: Effects of pH, temperature and dissolved oxygen. KSCE J. Civ. Eng. 2014, 18, 323–329. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, W.N.; Huang, Y.X.; Gao, X.P. Analysing the correlations of long-term seasonal water quality parameters, suspended solids and total dissolved solids in a shallow reservoir with meteorological factors. Environ. Sci. Pollut. Res. 2017, 24, 6746–6756. [Google Scholar] [CrossRef] [PubMed]
- Gérard, F. Clay minerals, iron/aluminum oxides, and their contribution to phosphate sorption in soils—A myth revisited. Geoderma 2016, 262, 213–226. [Google Scholar] [CrossRef]
- Bao, L.L.; Li, X.Y.; Su, J.J. Alteration in the potential of sediment phosphorus release along series of rubber dams in a typical urban landscape river. Sci. Rep. 2020, 10, 2714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bache, B.W.; Williams, E.G. A PHOSPHATE SORPTION INDEX FOR SOILS. J. Soil Sci. 1971, 22, 289–301. [Google Scholar] [CrossRef]
- Cao, X.Y.; Chen, X.Y.; Song, C.L.; Zhou, Y.Y. Comparison of phosphorus sorption characteristics in the soils of riparian buffer strips with different land use patterns and distances from the shoreline around Lake Chaohu. J. Soils Sediments 2019, 19, 2322–2329. [Google Scholar] [CrossRef]
- Zhai, H.Y.; He, X.Z.; Zhang, Y.; Du, T.T.; Adeleye, A.S.; Li, Y. Disinfection byproduct formation in drinking water sources: A case study of Yuqiao reservoir. Chemosphere 2017, 181, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Huo, D.; Chen, Y.X.; Zheng, T.; Liu, X.; Zhang, X.Y.; Yu, G.L.; Qiao, Z.Y.; Li, R.H. Characterization of Microcystis (Cyanobacteria) Genotypes Based on the Internal Transcribed Spacer Region of rRNA by Next-Generation Sequencing. Front. Microbiol. 2018, 9, 971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, B.; Vogt, R.D.; Xu, C.Y.; Lu, X.Q.; Xu, H.L.; Bishnu, J.P.; Zhu, L. Establishment and Validation of an Amended Phosphorus Index: Refined Phosphorus Loss Assessment of an Agriculture Watershed in Northern China. Water Air Soil Pollut. 2014, 225, 2103. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Xu, Y.; Zhao, G.; Shi, C.L.; Wang, Z.L.; Wang, Y.Q. Assessing threshold values for eutrophication management using Bayesian method in Yucliao Reservoir, North China. Environ. Monit. Assess. 2015, 187, 195. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Xu, Y.P.; Vogt, R.D.; Lu, X.Q.; Li, X.M.; Deng, X.W.; Yue, A.; Zhu, L. Effects of Land Use Change on Phosphorus Levels in Surface Waters-a Case Study of a Watershed Strongly Influenced by Agriculture. Water Air Soil Pollut. 2016, 227, 160. [Google Scholar] [CrossRef]
- Wen, S.L.; Wu, T.; Yang, J.; Jiang, X.; Zhong, J.C. Spatio-Temporal Variation in Nutrient Profiles and Exchange Fluxes at the Sediment-Water Interface in Yuqiao Reservoir, China. Int. J. Environ. Res. Public Health 2019, 16, 3071. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.Y.; Zhang, C.; Gao, X.P.; Wang, L.Y. Long-term variations of water quality in a reservoir in China. Water Sci. Techol. 2012, 65, 1454–1460. [Google Scholar] [CrossRef]
- Chang, C.; Sun, D.M.; Feng, P.; Zhang, M.; Ge, N. Impacts of Nonpoint Source Pollution on Water Quality in the Yuqiao Reservoir. Environ. Eng. Sci. 2017, 34, 418–432. [Google Scholar] [CrossRef]
- Zhou, B.; Vogt, R.D.; Lu, X.Q.; Yang, X.G.; Lu, C.W.; Mohr, C.W.; Zhu, L. Land use as an explanatory factor for potential phosphorus loss risk, assessed by P indices and their governing parameters. Environ. Sci. Process. Impacts 2015, 17, 1443–1454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharpley, A.N. Dependence of Runoff Phosphorus on Extractable Soil Phosphorus. J. Environ. Qual. 1995, 24, 920–926. [Google Scholar] [CrossRef] [Green Version]
- Asher, L.E. An Automated Method for the Determination of Orthophosphate in the Presence of Labile Polyphosphates. Soil Sci. Soc. Am. J. 1980, 44, 173–175. [Google Scholar] [CrossRef]
- Rydin, E. Potentially mobile phosphorus in Lake Erken sediment. Water Res. 2000, 34, 2037–2042. [Google Scholar] [CrossRef]
- Zhou, B.; Vogt, R.D.; Lu, X.Q.; Xu, C.Y.; Zhu, L.; Shao, X.L.; Liu, H.L.; Xing, M.N. Relative Importance Analysis of a Refined Multi-parameter Phosphorus Index Employed in a Strongly Agriculturally Influenced Watershed. Water Air Soil Pollut. 2015a, 226, 25. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhou, L.Y.; Wang, X.; Shen, B.Z. Investigation and evaluation on some elements in the substrate sludge of Yuqiao Reservoir. Acta Agric. Boreali-Sin. 1991, 6, 122–126. [Google Scholar]
- van Dael, T.; De Cooman, T.; Verbeeck, M.; Smolders, E. Sediment respiration contributes to phosphate release in lowland surface waters. Water Res. 2020, 168, 115168. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.L.; Wang, J.M.; Li, H.; Zhou, B.; Xing, M.N.; Liu, H.L. The Accumulation Characteristic of Sedimentary Phosphorus and Its Release Potential in Yuqiao Reservoir. Environ. Prot. Sci. 2020, 46, 56–61. [Google Scholar]
- Smith, E.A.; Mayfield, C.I.; Wong, P.T.S. Physical and chemical characterization of selected natural apatites in synthetic and natural aqueous solutions. Water Air Soil Pollut. 1977, 8, 401–415. [Google Scholar]
- Jaiswal, D.; Pandey, J. Hypoxia and associated feedbacks at sediment-water interface as an early warning signal of resilience shift in an anthropogenically impacted river. Environ. Res. 2019, 178, 108712. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Wang, J.; Chen, J.; Guo, J.; Yang, H.; Chen, Q. Successful control of phosphorus release from sediments using oxygen nano-bubble-modified minerals. Sci. Total Environ. 2019, 663, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Tammeorg, O.; Nürnberg, G.; Horppila, J.; Haldna, M.; Niemistö, J. Redox-related release of phosphorus from sediments in large and shallow Lake Peipsi: Evidence from sediment studies and long-term monitoring data. J. Great Lakes Res. 2020, 46, 1595–1603. [Google Scholar] [CrossRef]
- Yang, C.; Yang, P.; Geng, J.; Yin, H.; Chen, K. Sediment internal nutrient loading in the most polluted area of a shallow eutrophic lake (Lake Chaohu, China) and its contribution to lake eutrophication. Environ. Pollut. 2020, 262, 114292. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.M.; Li, K.F.; Liang, R.F.; Han, S.Q.; Li, Y. Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alam, M.S.; Barthod, B.; Li, J.Y.; Liu, H.; Zastepa, A.; Liu, X.C.; Maria, D. Geochemical controls on internal phosphorus loading in Lake of the Woods. Chem. Geol. 2020, 558, 119873. [Google Scholar] [CrossRef]
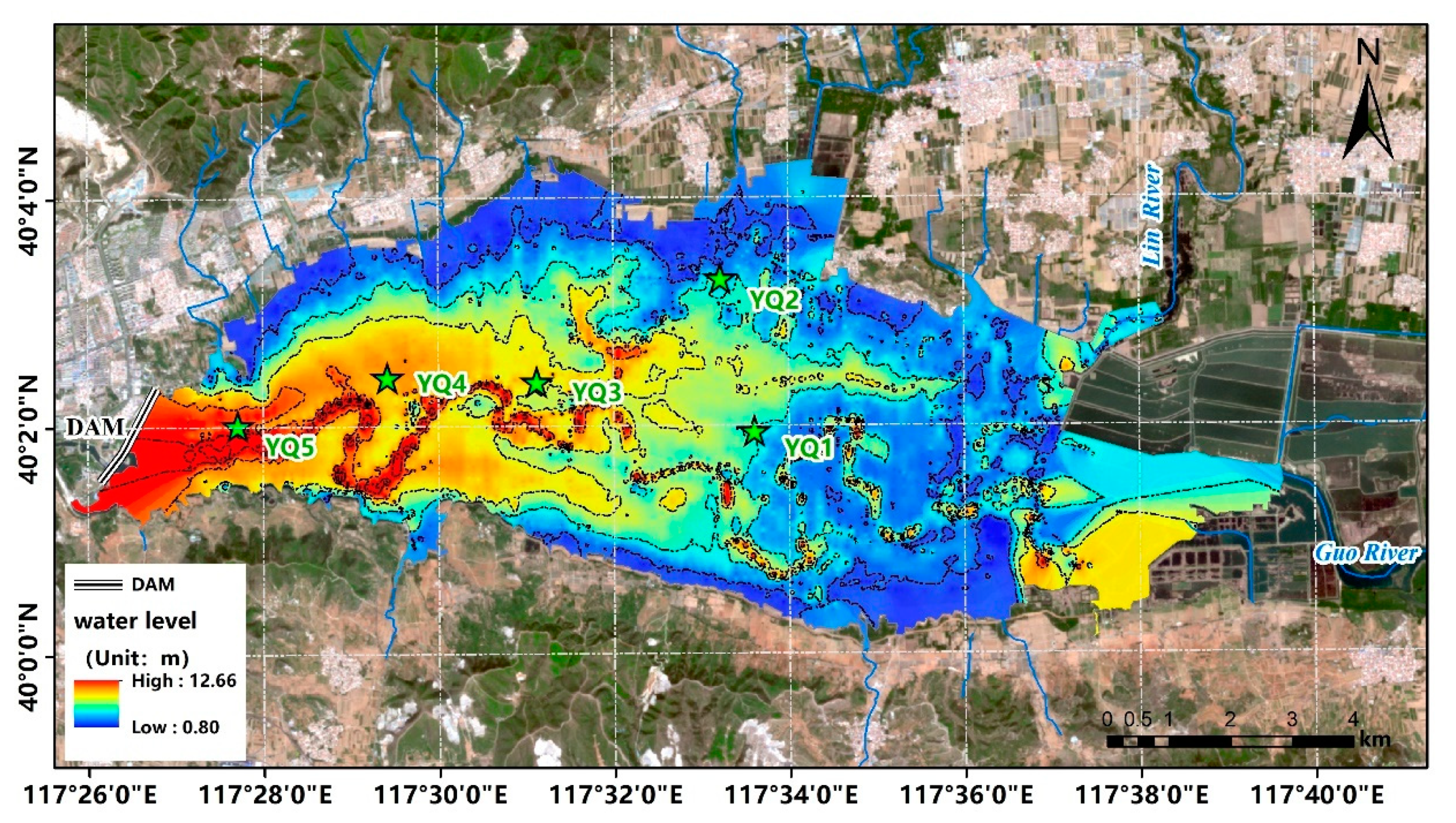
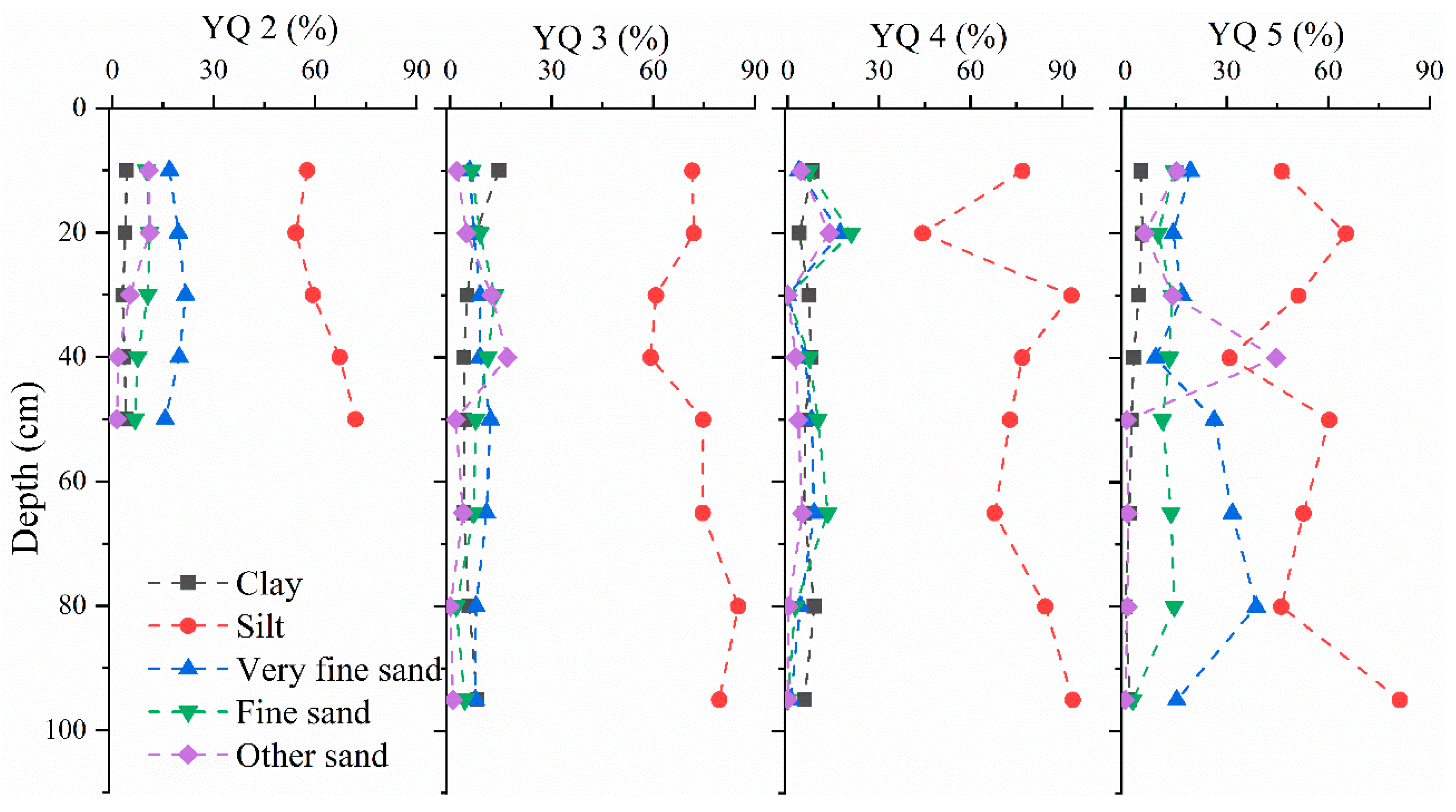
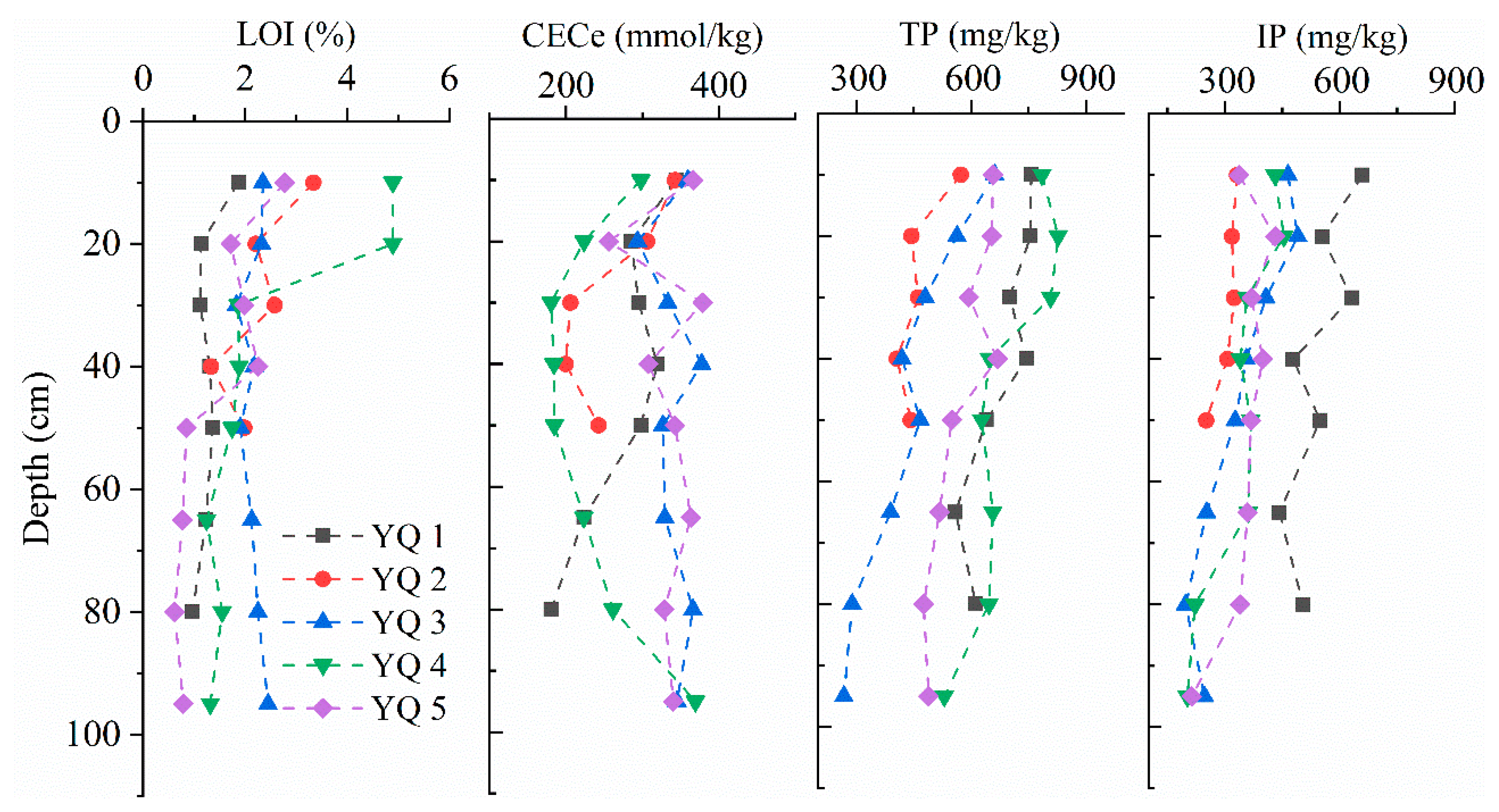



| Core Depth (cm) Subsections | YQ 1 80 7 | YQ 2 50 5 | YQ 3 95 8 | YQ 4 95 8 | YQ 5 95 8 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Avg | CV a | Avg | CV a | Avg | CV a | Avg | CV a | Avg | CV a | |
| pH | 6.5 | 6 | 7.6 | 1 | 6.9 | 3 | 6.9 | 3 | 7.2 | 3 |
| Clay b | – | – | 3.85 | 13 | 6.58 | 53 | 6.5 | 25 | 2.58 | 69 |
| LOI b | 1.28 | 22 | 2.29 | 33 | 2.17 | 10 | 2.42 | 64 | 1.47 | 56 |
| CECe c | 278 | 20 | 259 | 24 | 341 | 8 | 240 | 28 | 335 | 12 |
| TP d | 681 | 12 | 465 | 14 | 442 | 30 | 691 | 15 | 575 | 14 |
| IP d | 545 | 14 | 306 | 11 | 342 | 31 | 341 | 26 | 352 | 18 |
| OP d | 136 | 52 | 159 | 36 | 99 | 56 | 352 | 19 | 223 | 28 |
| STP d | – | – | 16.1 | 73 | 21.5 | 72 | 14.0 | 71 | 20.2 | 58 |
| Ex–P d | – | – | 3.9 | 30 | 7.3 | 31 | 4.6 | 30 | 9.5 | 39 |
| Fe–P d | – | – | 7.1 | 40 | 50.4 | 58 | 106 | 53 | 82.3 | 16 |
| Ca–P d | – | – | 164 | 12 | 187 | 31 | 194 | 29 | 247 | 17 |
| PSI d | – | – | 1135 | 14 | 1106 | 17 | 1424 | 19 | 1338 | 7 |
| PSC d | – | – | 1151 | 14 | 1128 | 17 | 1438 | 19 | 1358 | 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, B.; Fu, X.; Wu, B.; He, J.; Vogt, R.D.; Yu, D.; Yue, F.; Chai, M. Phosphorus Release from Sediments in a Raw Water Reservoir with Reduced Allochthonous Input. Water 2021, 13, 1983. https://doi.org/10.3390/w13141983
Zhou B, Fu X, Wu B, He J, Vogt RD, Yu D, Yue F, Chai M. Phosphorus Release from Sediments in a Raw Water Reservoir with Reduced Allochthonous Input. Water. 2021; 13(14):1983. https://doi.org/10.3390/w13141983
Chicago/Turabian StyleZhou, Bin, Xujin Fu, Ben Wu, Jia He, Rolf D. Vogt, Dan Yu, Fujun Yue, and Man Chai. 2021. "Phosphorus Release from Sediments in a Raw Water Reservoir with Reduced Allochthonous Input" Water 13, no. 14: 1983. https://doi.org/10.3390/w13141983







